Apr 3, 2026

At the cellular level, something remarkable happens when tirzepatide binds to its receptors. Two separate signaling cascades activate simultaneously. GIP receptors fire in the gut, the pancreas, and deep in the hypothalamus. GLP-1 receptors respond in parallel, amplifying insulin secretion, slowing gastric emptying, and sending satiety signals through the vagus nerve and into the brainstem. This is not a single-target drug. It is a coordinated biochemical intervention that works through two of the most powerful metabolic hormones the body produces naturally.
Compounded versions of this dual agonist, often labeled GLP-1 GIP B12, add a third element: vitamin B12. The combination is not random. It is deliberate. B12 addresses specific vulnerabilities that come with GLP-1 receptor agonist therapy, from nausea at initiation to nutrient absorption deficits that develop over months of use. Understanding why all three components appear together, and how they interact, is the foundation of using this compound well.
Most guides cover one piece. They explain tirzepatide in isolation, or they mention B12 as a footnote. This guide covers the full picture. You will understand the dual GIP and GLP-1 mechanism at a level most users never reach, the specific science behind B12 inclusion, every major compounded formulation available, the escalation schedule that produces results while minimizing side effects, storage requirements that protect potency, and the emerging microdosing protocols that go beyond weight loss entirely. If you are researching this compound, this is the reference you keep.
What GLP-1 GIP with B12 actually is
The name GLP-1 GIP B12 describes the pharmacological mechanism, not the drug itself. The active compound is tirzepatide, a synthetic peptide that acts as a dual agonist at both the glucose-dependent insulinotropic polypeptide receptor (GIPR) and the glucagon-like peptide-1 receptor (GLP-1R). Tirzepatide was engineered by modifying the native GIP peptide sequence with a fatty acid chain that extends its half-life to approximately five days, making once-weekly dosing possible.
B12 enters the picture through compounding pharmacies. When 503A or 503B compounding pharmacies prepare tirzepatide formulations for patients, they frequently add cyanocobalamin or methylcobalamin to the vial. The result is a single injectable solution containing both the peptide and the vitamin. Patients inject once weekly. The formulation is simple to administer. The rationale behind the combination is scientifically grounded.
This is not the same as brand-name Mounjaro or Zepbound. Those are FDA-approved tirzepatide products manufactured by Eli Lilly. Compounded versions are prepared by licensed pharmacies under physician supervision, using bulk pharmaceutical-grade active pharmaceutical ingredients. During periods when brand-name tirzepatide has been on the FDA shortage list, compounding has been legally permitted. Patients working with telehealth providers and licensed compounding pharmacies have accessed these formulations at different price points and in customized concentrations that the commercial product does not offer.
The compounded tirzepatide with B12 formulation is distinct from the tirzepatide glycine blend and the tirzepatide niacinamide formulation, though all three are compounded preparations. Each additive serves a different purpose. Understanding which formulation you have matters for both efficacy and side effect management.
People searching for GLP-1 GIP B12 are almost always looking for tirzepatide-based compounds. If you have seen a syringe with a pink or magenta liquid inside, that color is almost certainly from the B12. It is one of the most common visual identifiers of this formulation. Our complete guide to what color tirzepatide with B12 looks like covers this in detail, including when color variation indicates a problem and when it is completely normal.
How the dual GLP-1 and GIP mechanism works
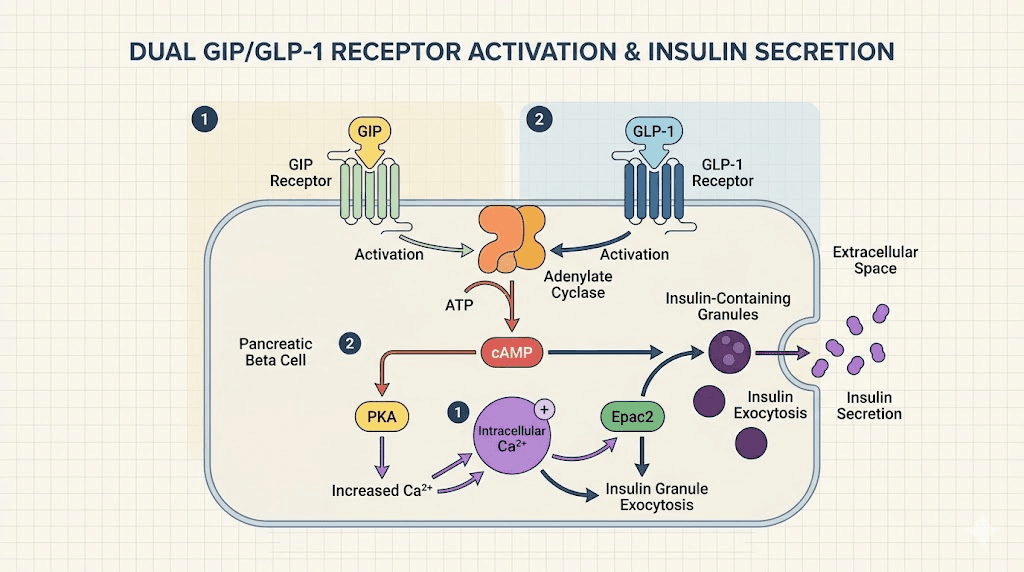
To understand why a dual agonist outperforms a single agonist, you need to understand what each receptor does independently and what happens when both are activated together.
The GLP-1 receptor pathway
Glucagon-like peptide-1 is secreted by L-cells in the distal small intestine and colon in response to food intake. It was first characterized as an incretin hormone, meaning it enhances glucose-stimulated insulin secretion from the pancreatic beta cells. This is the glucose-dependent part of the mechanism. GLP-1R agonists do not cause insulin secretion when blood glucose is low, which is why hypoglycemia risk is low compared to older diabetes medications.
But GLP-1 receptor activation does far more than regulate insulin. GLP-1R is expressed in the brainstem nucleus tractus solitarius, in the hypothalamic arcuate nucleus, in the vagus nerve, in the stomach, and in the heart. When activated, GLP-1R slows gastric emptying, reduces glucagon secretion, reduces appetite through central mechanisms, and reduces cardiovascular risk through pathways that are still being elucidated. The benefits of tirzepatide beyond weight loss stem in large part from these downstream GLP-1R effects.
Semaglutide, the drug in Ozempic and Wegovy, is a GLP-1R agonist only. It does not activate GIPR. Understanding this distinction helps explain why tirzepatide produces greater weight loss in head-to-head comparisons. If you are curious about the differences between semaglutide and tirzepatide in clinical terms, the receptor difference is the starting point for that entire conversation.
The GIP receptor pathway
Glucose-dependent insulinotropic polypeptide is secreted by K-cells in the proximal small intestine. It was identified before GLP-1 and was actually the first incretin hormone described in the literature. Despite being discovered first, it received less clinical attention for decades because early research suggested that GIP signaling was impaired in type 2 diabetes, making it seem like an unlikely therapeutic target.
That conclusion turned out to be incomplete. GIPR agonism in the context of a functioning GLP-1R signal produces synergistic effects that neither pathway achieves alone. GIPR is expressed in adipose tissue, bone, and the central nervous system, particularly in brain regions involved in reward processing and energy homeostasis. GIP receptor activation in the hypothalamus reduces food intake through mechanisms that are distinct from and complementary to GLP-1 pathways. This central GIPR activity is one reason tirzepatide reduces cravings and alters food preferences in ways patients frequently describe as different from their experience with GLP-1-only agents.
Research published in the context of the SURPASS trial program demonstrated that tirzepatide produced dose-dependent HbA1c reductions of 1.24 to 2.58 percent across the studied dose range, and body weight reductions of 5.4 to 11.7 kilograms in patients with type 2 diabetes.
The SURMOUNT trials in patients without diabetes showed even greater weight loss, with the 15 mg dose producing an average loss of 20.9 percent of body weight at 72 weeks compared to 14.9 percent for semaglutide in similar populations. The dual mechanism is not incremental. It is multiplicative.
Synergy between the two pathways
The additive effect happens at several levels. At the pancreas, simultaneous GIPR and GLP-1R activation produces greater insulin secretion than either pathway alone. At the brain, different neural circuits receive activation signals through both receptor types, creating broader coverage of appetite-regulating regions. At the level of adipose tissue, GIPR activation promotes fatty acid uptake during energy surplus and fatty acid release during energy deficit, effects that complement the appetite suppression driven by GLP-1R. The result is a compound that changes both how much you eat and how your body handles the energy you do consume.
For researchers interested in the comparison between tirzepatide and next-generation compounds like retatrutide, which adds a third receptor (GCG), our guide to retatrutide versus semaglutide provides useful context, as does our three-way comparison of semaglutide, tirzepatide, and retatrutide. The receptor pharmacology is where future obesity medicine is heading, and understanding tirzepatide dual mechanism positions you to interpret that research intelligently.
Why B12 is included in the compound
Vitamin B12 does not enhance tirzepatide potency directly. It is not added because it makes the drug work better at the receptor level. It is added because it addresses two specific and significant clinical problems that arise from GLP-1 receptor agonist therapy: nausea at treatment initiation and progressive B12 depletion over the course of long-term use. Both problems are real. Both have meaningful consequences if unaddressed.
B12 and nausea reduction
Nausea is the most common side effect of GLP-1 and GLP-1/GIP receptor agonists. In the SURMOUNT trials, nausea affected 12 to 18 percent of participants at lower doses and higher percentages at escalating doses. It typically peaks in the first few days after each injection, then subsides. For most patients, it resolves substantially after the first few weeks of a given dose. But for some, it remains disruptive enough to cause dose reductions or treatment discontinuation.
The mechanism of GLP-1-induced nausea involves central GLP-1R activation in the brainstem, specifically in the area postrema, which is the chemoreceptor trigger zone for vomiting. When GLP-1R agonists penetrate the blood-brain barrier and reach this region, they can trigger nausea signals. Research published in peer-reviewed journals has explored B12-conjugated GLP-1 agonists specifically to test whether B12 attachment reduces central nervous system penetration while preserving peripheral metabolic effects.
The findings are interesting. B12-conjugated GLP-1 analogs show reduced brain penetration due to the active transport mechanisms that move B12 through specific carrier proteins rather than through passive diffusion. This means the peptide reaches peripheral GLP-1R in the gut and pancreas efficiently but enters the brainstem region that triggers nausea at lower concentrations. The result, in preclinical and early clinical data, is maintained blood sugar regulation with reduced nausea and vomiting. Compounding pharmacies have applied this research pragmatically by including B12 in their tirzepatide formulations.
For patients who have struggled with nausea on GLP-1 therapy, our guides to tirzepatide fatigue and GLP-1 fatigue cover the overlapping symptom profile. Nausea, fatigue, and reduced appetite frequently occur together at initiation. Understanding which symptoms require medical attention and which are expected parts of the dose escalation process is important for staying on protocol.
B12 deficiency prevention during GLP-1 therapy
This is the more serious reason for B12 inclusion, and it is one that many patients are not informed about.
GLP-1 receptor agonists slow gastric emptying significantly. This is intentional and beneficial for glucose regulation and satiety. But the slowed gastric transit affects the absorption of multiple nutrients, and B12 is particularly vulnerable. Vitamin B12 absorption from food requires several steps: intrinsic factor secretion from gastric parietal cells, acid-dependent release of B12 from food proteins, and physical contact time in the ileum where transcobalamin receptors capture the B12-intrinsic factor complex. Slowed gastric emptying disrupts the timing and conditions required for optimal absorption.
Additionally, GLP-1 agonists significantly reduce food intake. Patients often eat 30 to 50 percent fewer calories than before treatment. If the foods they are eating less of included B12-rich sources like meat, fish, eggs, and dairy, their total dietary B12 intake falls sharply. The combination of reduced intake and impaired absorption creates a trajectory toward deficiency over months and years of treatment.
B12 deficiency is not a minor inconvenience. Mild deficiency causes fatigue, brain fog, and mood changes. Moderate deficiency causes peripheral neuropathy, characterized by numbness, tingling, and weakness in the hands and feet. Severe deficiency causes irreversible neurological damage and subacute combined degeneration of the spinal cord, a condition that does not fully reverse even with aggressive B12 repletion. This is why including B12 in the compound is medically justified, not merely cosmetic.
The form of B12 in the compound matters. Cyanocobalamin is the most common form used in pharmaceutical preparations. It is stable, inexpensive, and well-absorbed. However, it requires conversion in the liver to methylcobalamin and adenosylcobalamin before it can be used in enzymatic reactions. Methylcobalamin is the bioactive form. It does not require hepatic conversion. For patients with impaired liver function or genetic variants affecting B12 metabolism, methylcobalamin may be preferable. Our guide to tirzepatide with methylcobalamin covers this distinction in depth, and our cyanocobalamin tirzepatide guide covers the more common formulation. The tirzepatide hydroxocobalamin variant represents a third option with its own absorption characteristics.
B12 and energy during caloric deficit
There is a third, more practical role for B12. Patients on tirzepatide enter significant caloric deficits. The drug is effective partly because it makes eating less feel effortless rather than effortful. But caloric restriction, combined with the reduced nutrient density of a smaller total food volume, creates conditions where B vitamins including B12 become limiting factors for mitochondrial energy production. B12 is a cofactor for succinyl-CoA synthesis and for the conversion of homocysteine to methionine. Both reactions are fundamental to cellular energy metabolism.
Patients who supplement B12 during tirzepatide therapy commonly report better sustained energy levels compared to those who do not. This is consistent with B12 role in energy metabolism and with the depletion pattern described above. Our guide to whether tirzepatide gives you energy addresses this question specifically, including the mechanisms by which both the drug and its adjunct nutrients affect energy levels. You can also review our comprehensive list of supplements to take with tirzepatide for a broader framework of nutritional support during treatment.
Benefits beyond weight loss
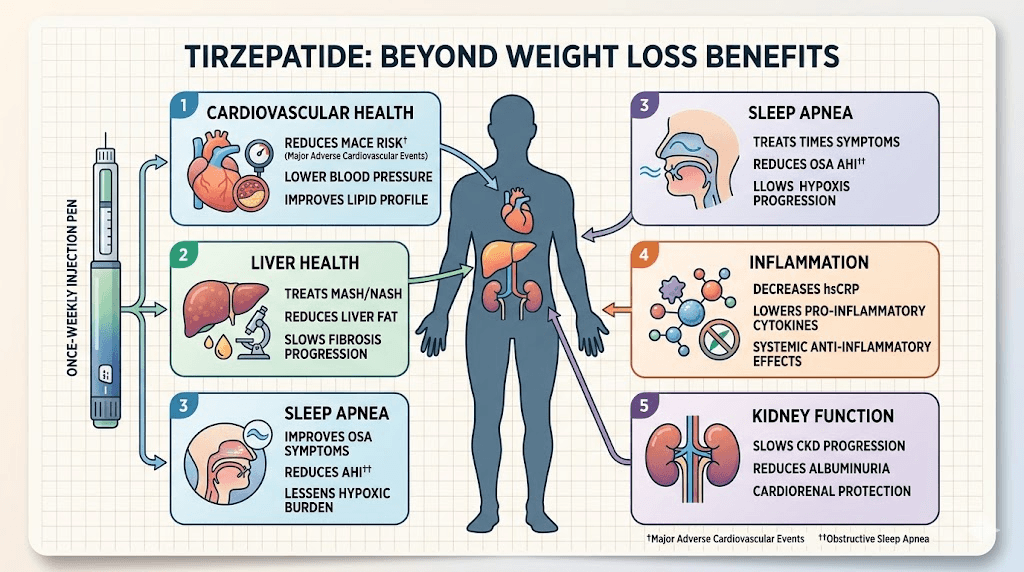
Weight loss is the headline. It is not the whole story. The clinical research and emerging data on tirzepatide-based compounds, including GLP-1 GIP B12 formulations, point to a range of metabolic, cardiovascular, and anti-inflammatory benefits that extend well beyond the number on the scale.
Cardiovascular outcomes
GLP-1 receptor agonists have demonstrated cardiovascular risk reduction in large outcome trials. The LEADER trial for liraglutide and SUSTAIN-6 for semaglutide established cardiovascular benefit for that class. Tirzepatide cardiovascular outcome data from the SURPASS-CVOT trial extends these findings to the dual agonist. Patients with established cardiovascular disease showed reductions in major adverse cardiovascular events. The mechanisms include blood pressure reduction, improvement in lipid profiles, anti-inflammatory effects on the arterial wall, and possible direct cardioprotective effects through GLP-1R expressed on cardiomyocytes.
The B12 component adds a separate cardiovascular benefit. B12 deficiency elevates homocysteine, a sulfur-containing amino acid that damages arterial endothelium and increases atherosclerosis risk. Maintaining adequate B12 levels keeps homocysteine in the normal range, supporting the cardiovascular benefit tirzepatide already provides through its primary mechanism.
Glycemic control and insulin sensitivity
For patients with type 2 diabetes or prediabetes, the HbA1c reduction from tirzepatide is substantial. The 2.58 percent HbA1c reduction seen at higher doses represents a clinical effect comparable to two or three additional antidiabetic medications. Many patients in the SURPASS trials achieved HbA1c below 5.7 percent, which is the threshold for normal glycemia. For patients with prediabetes, tirzepatide can produce what clinical researchers describe as remission of the prediabetic state.
Insulin sensitivity improves alongside insulin secretion enhancement. As body weight decreases, hepatic fat content falls, reducing hepatic insulin resistance. Skeletal muscle insulin sensitivity improves as visceral adipose tissue volume decreases. The drug creates a positive feedback loop where weight loss improves the metabolic environment, which makes further metabolic normalization easier.
Non-alcoholic fatty liver disease
Hepatic steatosis, or fatty liver, is closely linked to insulin resistance and visceral adiposity. Both improve dramatically with tirzepatide. The SURPASS-3 MRI substudy demonstrated significant reductions in liver fat content at 52 weeks. For patients with non-alcoholic fatty liver disease or non-alcoholic steatohepatitis, tirzepatide represents one of the most effective interventions currently available. The combination of weight loss, improved insulin sensitivity, and direct GLP-1R effects on hepatic lipid metabolism drives these improvements.
Sleep apnea
The SURMOUNT-OSA trial demonstrated that tirzepatide reduced the severity of obstructive sleep apnea significantly, with some patients achieving complete resolution. The mechanisms include weight loss reducing upper airway fat deposition, improved respiratory muscle tone with reduced adiposity, and possible direct effects on central respiratory control through GLP-1R in the brainstem. For patients whose sleep apnea has been attributed to obesity, the weight loss driven by GLP-1 GIP therapy often produces dramatic improvements in sleep quality.
Inflammation and immune modulation
Tirzepatide reduces circulating inflammatory markers including interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-alpha). These cytokines are produced by adipose tissue macrophages in proportion to the amount of visceral fat. As fat mass decreases, their production falls. But there appear to be direct anti-inflammatory effects as well, independent of weight loss alone, through GLP-1R expressed on immune cells. This anti-inflammatory profile has generated significant interest in conditions beyond metabolic disease. Our guide to tirzepatide and autoimmune conditions covers this emerging area, as does our guide to GLP-1 for Hashimoto thyroiditis. Researchers exploring microdosing GLP-1 for autoimmune conditions are targeting specifically this anti-inflammatory mechanism.
For patients with conditions like lupus, endometriosis, or lipedema, the anti-inflammatory effects combined with weight loss represent a potentially significant quality of life intervention.
Neurological and cognitive benefits
GLP-1R is expressed throughout the brain. In preclinical models of neurodegeneration, GLP-1R agonists reduce amyloid plaque formation, improve neuronal survival, and reduce neuroinflammation. Human trials of GLP-1R agonists in Parkinson disease have shown promising signals. Cognitive function in obese individuals improves substantially with weight loss alone, but the direct central GLP-1R effects may contribute additional benefit beyond what weight reduction alone would predict. This is an area of active research and one of the primary motivations behind longevity-focused microdosing protocols explored later in this guide.
Common GLP-1 GIP B12 formulations compared
Compounding pharmacies prepare tirzepatide in several distinct formulations. Each adds a different companion ingredient to the tirzepatide base. Understanding what is in your vial, and why, allows you to make informed decisions and to recognize if a formulation switch is warranted.
Tirzepatide with B12 only
The simplest compounded version. Tirzepatide plus either cyanocobalamin or methylcobalamin, in bacteriostatic water, at a concentration typically ranging from 2 mg/mL to 10 mg/mL. The complete guide to tirzepatide with B12 covers this base formulation in full. This is the formulation most likely to appear bright pink or magenta in the vial, since B12 has a characteristic intense red pigment. The color intensity varies with B12 concentration and formulation specifics.
Tirzepatide, glycine, and B12
The most widely prescribed compounded formulation during periods of shortage availability. Glycine is a simple amino acid that serves as a stabilizer and pH buffer in the solution. It helps maintain the peptide in its properly folded conformation and reduces degradation during storage. The tirzepatide glycine B12 compound guide covers this three-component formulation, and our guide specifically on tirzepatide with glycine explains the stabilizer role in more detail. The glycine addition generally does not change the patient experience but may extend shelf life and maintain potency across the storage period.
Tirzepatide with niacinamide and B12
Niacinamide is a form of vitamin B3 that has been added to some compounded tirzepatide preparations.
Like B12, it supports energy metabolism during caloric restriction. Niacinamide has anti-inflammatory properties and supports NAD+ biosynthesis, which is relevant for metabolic function and cellular repair. The tirzepatide niacinamide complete guide covers this formulation, and you can compare it to the semaglutide niacinamide formulation if you are considering a switch between base peptides.
Tirzepatide with L-carnitine
A newer formulation that adds L-carnitine to support fatty acid transport into mitochondria. During weight loss, the body is mobilizing stored fat for energy. Carnitine is the carrier molecule that moves long-chain fatty acids across the mitochondrial membrane for beta-oxidation. Deficiency of carnitine can limit fat oxidation efficiency. The tirzepatide levocarnitine complete guide covers this formulation in detail. Some preparations combine carnitine with B12 for dual metabolic support, and our guide to the tirzepatide levocarnitine blend addresses that combination specifically.
Semaglutide with B12
For completeness: compounded semaglutide is also available with B12. This is the GLP-1-only version of a similar concept. The rationale for B12 inclusion is identical, though semaglutide lacks the GIP receptor component. Our semaglutide with B12 complete guide and compounded semaglutide with B12 dosage chart cover that parallel formulation. If you are considering switching from tirzepatide to semaglutide or vice versa, the formulation comparison matters for continuity of your B12 support. Our guide to the semaglutide glycine B12 blend covers the three-component semaglutide version.
Our comprehensive tirzepatide color guide explains what each visual appearance tells you about the compound in your vial. The why is my semaglutide pink guide covers the same question for those using the semaglutide analog.
Dosage protocols and escalation schedules
Dosing compounded tirzepatide B12 correctly is where outcomes are made or lost. The escalation schedule exists for a reason. Rushing it produces more side effects without meaningfully accelerating results. Moving too slowly leaves efficacy on the table. Understanding the logic behind the schedule helps you follow it intelligently.
Standard escalation protocol
The most widely used escalation schedule for compounded tirzepatide B12 formulations follows this structure. These doses assume a 2 mg/mL concentration vial using an insulin syringe marked in units. Confirm your concentration with your pharmacy before calculating your volume.
Weeks 1 to 4: 20 units (0.2 mL) once weekly
Weeks 5 to 8: 40 units (0.4 mL) once weekly
Weeks 9 to 12: 70 units (0.7 mL) once weekly
Weeks 13 to 16: 90 units (0.9 mL) once weekly
These are unit measurements based on insulin syringe markings. The actual milligram dose depends entirely on your formulation concentration. A 2 mg/mL vial and a 5 mg/mL vial with the same number of units represent very different milligram doses. This is one of the most important details to confirm with your prescribing provider and pharmacy before starting. Our comprehensive guide to tirzepatide dosage in units explains this conversion in detail, and you can use the peptide calculator to determine your exact dose in milligrams based on your specific formulation concentration.
The compound tirzepatide dosage chart and compound tirzepatide dosing chart provide visual references for different concentration formulations. For Empower Pharmacy specifically, the Empower tirzepatide dosage chart and Empower tirzepatide dosing chart are formatted for their specific concentrations. The tirzepatide dose chart and tirzepatide dosing conversion chart for weight loss provide additional reference formats.
Extended escalation for sensitive patients
Some patients metabolize tirzepatide more slowly or have greater sensitivity to GI side effects. These patients benefit from a slower escalation. Rather than moving to the next dose tier at 4 weeks, they hold each dose for 6 to 8 weeks before escalating. This extends the total time to reach the maintenance dose but substantially reduces the likelihood of side effects that cause discontinuation.
The compounded tirzepatide starting dose guide covers how to assess whether you are a candidate for modified escalation and what markers suggest you are tolerating your current dose well enough to advance. The compounded tirzepatide dosage calculator allows you to model different escalation timelines against your weight loss goals. The semaglutide dosage calculator serves the same function for patients on the semaglutide analog.
Maintenance dosing and long-term management
Once you reach a dose that produces adequate weight loss and acceptable side effects, that becomes your maintenance dose. Most patients find their optimal maintenance dose somewhere between 5 mg and 15 mg weekly. Some achieve their goals at lower doses and find that higher doses produce diminishing returns with more side effects. Others require maximum doses to overcome metabolic adaptation.
The GLP-1 maintenance dose guide addresses the critical question of how long to stay on treatment and what happens when you stop. This is important for long-term planning. If you are considering weaning off tirzepatide after reaching your goal, the protocol for doing so safely is different from simply stopping the injections. Our guide to getting off GLP-1 covers the full discontinuation framework.
Converting between formulations and products
If you switch pharmacies, switch from compounded to brand-name tirzepatide, or switch from semaglutide to tirzepatide, the dose conversion requires attention. The semaglutide to tirzepatide conversion chart provides conversion references. The key principle is that tirzepatide is more potent on a milligram basis than semaglutide, so direct milligram equivalents do not apply. Our guide on tirzepatide syringe dosage covers how to read your syringe accurately after a concentration change.
Injection timing and day selection
Once-weekly dosing gives you flexibility in choosing your injection day. Most patients choose a day that positions the peak side effect window away from important professional or social commitments. Since nausea and fatigue tend to peak 12 to 48 hours after injection, choosing a Friday injection means the worst of any side effects falls on the weekend for most people. Our guide to the best time to take your tirzepatide shot and the best time to take a GLP-1 shot cover the timing considerations in full.
How to reconstitute and store GLP-1 GIP B12
Many compounded tirzepatide B12 formulations arrive as lyophilized powder rather than pre-mixed solution. Reconstitution is a simple process, but doing it incorrectly degrades the peptide and reduces efficacy. Storage after reconstitution requires consistent attention. These details determine whether you get the potency you paid for.
Reconstitution procedure
You will need bacteriostatic water (BAC water) for reconstitution. Do not use sterile water, saline, or tap water. Bacteriostatic water contains 0.9% benzyl alcohol, which prevents microbial growth in the multi-use vial over the storage period. Using plain sterile water requires single-use handling and significantly shortens stability.
The most important principle in reconstitution is gentle handling. Tirzepatide is a peptide with a molecular structure that can be damaged by mechanical agitation. Do not shake the vial. Do not draw the reconstitution water in rapidly. Inject the BAC water slowly down the side of the vial, allowing it to run down the glass rather than hitting the lyophilized cake directly. Then swirl the vial gently until fully dissolved. This takes 30 to 60 seconds. A clear solution indicates complete dissolution.
Our step-by-step guide on how to reconstitute tirzepatide covers this procedure with detailed instructions. If you have a 10 mg vial and need to determine the right BAC water volume, our guide on how much BAC water to mix with 10 mg tirzepatide provides the calculation. For 30 mg vials, the 30 mg tirzepatide BAC water guide applies. Use the peptide reconstitution calculator to determine the exact volume for any combination of vial size and target concentration. For comparison, our guide on how to reconstitute semaglutide covers the parallel process for the GLP-1-only compound.
Storage requirements
Temperature is the most critical storage factor. Lyophilized tirzepatide is relatively stable at room temperature for short periods. Reconstituted solution must be refrigerated. The storage parameters are straightforward and non-negotiable.
Unreconstituted powder: Refrigerate at 36 to 46 degrees F (2 to 8 degrees C). Some formulations tolerate room temperature for up to 30 days, but refrigeration is strongly preferred throughout.
Reconstituted solution: Must be refrigerated. Stable for approximately 28 days after reconstitution when stored properly and prepared with bacteriostatic water.
Never freeze: Freezing destroys the peptide structure and renders it inactive. This is absolute. Our guide to whether you can freeze tirzepatide explains the degradation mechanism.
Protect from light: B12 in particular is photosensitive. Store in the original amber vial or a dark location. Extended light exposure degrades B12 and can affect overall formulation stability.
Our guides to whether tirzepatide needs refrigeration, how long tirzepatide lasts in the fridge, and tirzepatide shelf life cover the specific scenarios in depth. If you have accidentally left your vial out, our guide on how long compounded tirzepatide can be out of the fridge helps you assess viability, and what happens if tirzepatide gets warm explains the degradation mechanism. For semaglutide users, the same principles apply and are covered in our compounded semaglutide refrigeration guide and semaglutide out-of-fridge time guide.
Traveling with GLP-1 GIP B12
Travel introduces real storage challenges. Airlines allow insulin syringes and medically necessary injectables, but keeping your vial at refrigerator temperature during a long trip requires planning. Insulated medication travel cases with gel ice packs maintain 2 to 8 degrees C for 24 to 48 hours, which is sufficient for most travel scenarios. Our guide on how to travel with tirzepatide covers airline rules, cold pack options, and temperature excursion risk management in detail. The GLP-1 travel case guide identifies specific product options. The parallel guide on traveling with semaglutide covers the same logistics for the semaglutide compound.
Visual inspection before each injection
Before drawing each dose, inspect the vial. The solution should be clear to pink, free of particles, and without unusual cloudiness or dramatic color changes beyond the expected B12 pink. Our compounded tirzepatide expiration date guide covers how to interpret the date on your vial. Do not inject a vial that appears cloudy, shows visible particles, or has changed color unexpectedly. When in doubt, contact your compounding pharmacy before proceeding.
Injection technique for GLP-1 GIP B12
Correct injection technique is straightforward, but the details matter for both efficacy and minimizing injection site reactions. Tirzepatide is administered subcutaneously, meaning into the fat layer just beneath the skin, not into muscle.
Choosing an injection site
The abdomen, outer thighs, and upper arms are the three standard subcutaneous injection sites for tirzepatide. The abdomen is most commonly used. Rotate sites with each injection to prevent lipohypertrophy, which is the buildup of fibrous tissue at repeatedly injected locations. Rotating by at least one inch from the previous injection site within the same region is a minimum standard. Rotating between regions, for example abdomen one week and thigh the next, is preferable for long-term use.
Our detailed guides to how to inject GLP-1, where to inject GLP-1, and how to inject tirzepatide in the stomach cover the mechanics step by step. The guide to tirzepatide injection in the thigh covers the specific positioning and needle angle for that alternative site. Our best injection site guide and best GLP-1 injection site for weight loss compare absorption rates and side effect profiles across sites. The guide on where you can inject tirzepatide addresses common questions about alternative injection locations.
Syringe selection and technique
Use an insulin syringe with a fine-gauge needle (28 to 31 gauge) for subcutaneous injection. The needle length of 4 to 8 mm is appropriate for most adults. For thicker subcutaneous tissue, slightly longer needles ensure reliable subcutaneous rather than intradermal placement. Our GLP-1 syringe complete guide covers needle selection in detail. The guide to how to inject tirzepatide with a syringe covers the injection mechanics from prep to post-injection. The guide to how to give a semaglutide injection with a syringe covers the parallel procedure for the semaglutide compound.
Managing injection site reactions
Redness, swelling, or itching at the injection site affects a subset of patients. These reactions are more common when injecting cold solution directly from the refrigerator. Allowing the vial to reach room temperature for 15 to 20 minutes before drawing your dose reduces this substantially. Injecting slowly, using a new needle for every injection, and not rubbing the site after injection all reduce local reactions. Our guide to injection site reactions with tirzepatide and treatment options for injection site reactions cover management strategies. The GLP-1 injection site reaction guide addresses the class-wide pattern and distinguishes normal reactions from those requiring medical attention.
Side effects and how to manage them
The side effect profile of tirzepatide B12 compounds is shaped primarily by the tirzepatide mechanism. B12 modulates some of these effects but does not eliminate them entirely. Managing side effects well is the difference between successful treatment and discontinuation.
Gastrointestinal side effects
Nausea, vomiting, diarrhea, and constipation are the most common side effects. They are most intense during the first few weeks at each dose level and typically improve as the body adapts. The B12 component reduces nausea through the mechanism described earlier, but does not eliminate it at higher doses.
Nausea management strategies include eating smaller meals, avoiding high-fat foods around injection time, staying well-hydrated, and taking the injection at a time that positions peak nausea during sleep hours. Constipation is managed with adequate fiber, hydration, and if needed, gentle fiber supplements. Our guides to tirzepatide constipation treatment, GLP-1 constipation relief, tirzepatide diarrhea duration, tirzepatide bloating, and sulfur burps on GLP-1 address each GI manifestation specifically. The best fiber supplement for GLP-1 and best probiotic for semaglutide guides offer practical adjunct support strategies.
Heartburn and acid reflux are common because gastric emptying slows and food sits longer in the stomach. Our guides to heartburn on tirzepatide and semaglutide acid reflux cover management options including dietary adjustments and positioning strategies. Dry mouth is a less discussed but frequent complaint. Our guides to tirzepatide dry mouth and GLP-1 dry mouth address this symptom.
Fatigue and energy fluctuations
Fatigue is a frequently reported side effect, particularly in the first few weeks of treatment and after dose escalations. Multiple mechanisms contribute. Reduced caloric intake means less fuel for daily activity. The hormonal shifts accompanying rapid weight loss affect energy regulation. GLP-1R effects on the vagus nerve can produce a general sense of tiredness. B12 inclusion addresses the nutritional component of fatigue but does not fully prevent the physiological adaptation period.
Our complete guide to GLP-1 fatigue covers this topic thoroughly, including the distinction between expected adaptation fatigue and fatigue that warrants medical attention. The guide to whether tirzepatide makes you tired addresses the specific mechanisms. The related guide on semaglutide fatigue covers the GLP-1-only version of these same symptoms. Electrolyte support is frequently helpful during this period. Our best electrolytes for GLP-1 guide identifies the specific formulations that support energy and prevent dehydration during treatment.
Headaches and dizziness
Headaches are reported by a significant minority of patients, particularly early in treatment. Dehydration is the most common cause, since nausea reduces fluid intake while GI effects increase fluid losses. Electrolyte imbalance can also trigger headaches. Our guides to whether tirzepatide causes headaches, GLP-1 headaches complete guide, semaglutide dizziness, and semaglutide lightheadedness cover assessment and management in detail.
Hair loss
Telogen effluvium, or temporary hair loss related to metabolic stress, is reported by a meaningful subset of patients on GLP-1 therapy. It is not caused by the drug directly. It is caused by the caloric restriction and rapid weight loss, which trigger a shift of hair follicles from the growth phase to the resting and shedding phase. B12 deficiency, when present, can worsen hair loss. This is another reason adequate B12 support during treatment matters. Our guides to GLP-1 hair loss and whether tirzepatide makes your hair fall out explain the mechanism, timeline, and recovery pattern. Ensuring adequate protein intake significantly reduces this side effect, which connects to our nutrition section below.
Muscle pain and body aches
Muscle pain and general body aches occur in some patients, particularly during early treatment or after dose escalation. These symptoms typically resolve within a few days. If they are severe or persistent, they warrant contact with your prescribing provider. Our guides to tirzepatide muscle pain and tirzepatide body aches cover the mechanisms and management approaches.
Sleep, mood, and cognition
Some patients report insomnia, particularly in the initial weeks. The causes are multiple: metabolic changes, sleep disruption from GI symptoms, and changes in energy metabolism that affect sleep architecture. Our guides to tirzepatide insomnia, semaglutide insomnia, tirzepatide and anxiety, and tirzepatide brain fog cover these neurological and psychological side effects. Most resolve with dose stabilization and time. B12 adequacy is directly relevant here since B12 deficiency independently causes cognitive symptoms and mood disturbance, which is another reason the B12-containing formulation is preferred for long-term use.
SeekPeptides members access comprehensive side effect management guides, protocol optimization resources, and a community of experienced researchers who have navigated these exact situations. If you are managing multiple side effects simultaneously, the ability to ask questions of people who have been through the same process is genuinely valuable.
Microdosing GLP-1 GIP B12 for longevity
A separate and distinct use case has emerged for tirzepatide and GLP-1 class agents: microdosing for metabolic health, anti-inflammatory effects, and longevity, independent of weight loss goals. This is not FDA-approved. It is driven by the emerging research on GLP-1R biology and by the clinical experience of practitioners working in longevity medicine.
What microdosing means in this context
Microdosing for GLP-1 agents typically refers to doses significantly below the weight loss threshold. Where standard weight loss protocols use doses of 2.5 mg to 15 mg weekly, microdosing protocols use doses of 0.5 mg to 1.5 mg weekly. These doses are too low to produce significant appetite suppression or meaningful weight loss in most people. They are chosen specifically for their anti-inflammatory and cellular signaling effects.
Our dedicated guide to microdosing tirzepatide chart and microdose tirzepatide schedule provide the specific dose structures used in these protocols. The guide to how to microdose tirzepatide covers the practical aspects of drawing and administering these very small volumes accurately.
Anti-inflammatory mechanism at low doses
The anti-inflammatory effects of GLP-1R agonism are not dose-dependent in the same way that weight loss is. Research suggests that even sub-therapeutic doses from a weight loss perspective produce measurable reductions in circulating IL-6 and TNF-alpha. These cytokines are markers of chronic low-grade inflammation, the kind associated with accelerated biological aging, cardiovascular disease, neurodegeneration, and metabolic syndrome. Reducing them through a mechanism with a favorable safety profile is the core rationale for longevity microdosing.
Our guide to microdosing tirzepatide for inflammation covers the specific inflammatory pathways targeted and the emerging clinical experience with this approach. For patients with autoimmune conditions, the microdosing GLP-1 for autoimmune conditions guide addresses this specific application. The broader guide on tirzepatide and autoimmune disease provides additional context.
Metabolic benefits at low doses
Even at microdose levels, GLP-1R agonists improve glucose variability, reducing post-meal blood glucose spikes. Continuous glucose monitor data from patients on microdosing protocols shows improved time-in-range metrics even without significant weight loss. This reduction in glycemic variability is associated with reduced advanced glycation end-product formation, one of the cellular mechanisms linked to accelerated aging.
GIP receptor activation at low doses supports mitochondrial function in adipose tissue and may contribute to improved fatty acid metabolism independent of the dramatic appetite suppression seen at full doses. The synergy between the two receptor pathways is present even at low doses, which is an argument for tirzepatide-based microdosing over semaglutide-based microdosing from a metabolic standpoint. The comparison between tirzepatide and next-generation triple agonists like retatrutide is relevant for researchers tracking the evolution of this field. Our guide to the difference between tirzepatide and retatrutide covers how the addition of glucagon receptor agonism changes the metabolic and longevity profile.
Combining tirzepatide microdosing with other longevity protocols
Longevity-focused researchers frequently combine tirzepatide microdosing with other interventions. NAD+ precursors, in particular, are a common pairing. Our guide on NAD and tirzepatide together covers the rationale and protocol considerations for this combination. The niacinamide-containing tirzepatide formulations discussed earlier are partly motivated by this logic, since niacinamide is a NAD+ precursor. The combination of adequate B12 with microdose tirzepatide supports the neurological aspects of longevity protocols specifically. B12 adequacy is associated with better cognitive aging outcomes, and the combination of neural anti-inflammatory effects from GLP-1R activation and neuroprotective effects from B12 creates a complementary pair even at doses where weight loss is not the goal.
What to eat while using GLP-1 GIP B12
Nutrition during tirzepatide therapy requires more attention than most patients expect. The drug significantly reduces appetite, which is the point. But appetite reduction alone does not guarantee optimal nutrition. The reduced food volume makes every bite count more, not less. Poor food choices on tirzepatide produce worse outcomes than on any other weight loss approach because the absolute food quantity is so much lower.
Protein is the priority
Protein intake preservation is the single most important dietary focus during tirzepatide treatment. The target for most adults is 0.7 to 1.0 grams of protein per pound of target body weight daily. This is substantially higher than average dietary protein intake. Achieving this on a significantly reduced calorie intake requires deliberate planning and typically means protein-first eating at every meal.
Adequate protein serves two critical functions: it preserves lean muscle mass during rapid weight loss, and it supports satiety independent of the drug mechanism. Patients who prioritize protein report better body composition outcomes at the same body weight. Our guides to how much protein to eat on semaglutide, what to eat on tirzepatide, and tirzepatide diet plan provide specific meal frameworks and examples. The guide to how many calories to eat on tirzepatide provides specific calculation methods and caloric targets by starting weight and activity level.
Foods to avoid
High-fat foods, particularly fried foods and fatty animal products, significantly worsen GI side effects on tirzepatide. Gastric emptying is already slowed by the drug. High-fat meals slow it further, increasing the time food sits in the stomach and amplifying nausea, bloating, and reflux. This is not a permanent dietary restriction. Many patients find that as they adapt to their maintenance dose, they can tolerate occasional higher-fat meals without significant symptoms. But during escalation, avoiding these foods reduces the burden substantially.
Alcohol has complex interactions with tirzepatide. Rapid weight loss reduces alcohol tolerance. Some patients report unusual sensitivity to alcohol during treatment. Our guides to tirzepatide foods to avoid, foods to avoid on semaglutide, whether you can drink on tirzepatide, and whether you can drink on semaglutide address these considerations.
Best foods to support treatment
Lean proteins (chicken breast, fish, egg whites, low-fat Greek yogurt, cottage cheese) should form the foundation of most meals. Non-starchy vegetables provide fiber and micronutrients at very low calorie density, meaning they contribute nutritional value without crowding out protein capacity. Gentle complex carbohydrates in small portions (oatmeal, sweet potato, legumes) provide energy for exercise and support gut motility.
Our guides to best foods to eat on semaglutide, foods to eat while on semaglutide, and GLP-1 friendly meals provide specific meal ideas. The semaglutide diet plan provides a structured framework applicable to tirzepatide users as well.
Supplements to support treatment
B12 is covered within the compound itself, but additional nutritional gaps commonly develop during tirzepatide therapy. Vitamin D, magnesium, and iron deficiencies are frequently documented in patients on long-term GLP-1 therapy. Our comprehensive guide to supplements to take with GLP-1 and GLP-1 vitamin complete guide cover the full supplementation framework. The electrolyte guide and fiber supplement guide address two of the most practically important nutritional supports during treatment. The probiotic guide covers gut health support that helps manage the GI side effects common to this class of medications.
SeekPeptides provides detailed nutrition protocols specifically calibrated for GLP-1 therapy, including macro targets, supplement stacks, and meal planning frameworks developed from the collective experience of thousands of researchers. These resources go substantially beyond what generic dietary guidelines offer for this specific context.
When results are not what you expected
Not everyone sees the same results on tirzepatide B12 compounds. Weight loss varies significantly across individuals. Understanding why results vary, and what to do about it, saves patients from unnecessary frustration and premature discontinuation.
Understanding the timeline
Weight loss on tirzepatide is not linear. The first month often produces rapid initial loss driven by reduced sodium retention and water weight. Then a slower, more consistent fat loss phase follows. Most patients do not see the full weight loss benefit of a given dose until they have been on it for 8 to 12 weeks. This means that impatience during the escalation phase leads to premature dose increases that drive more side effects without proportional benefit.
Our guides to how fast tirzepatide works, how long tirzepatide takes to work, and the tirzepatide weight loss timeline set realistic expectations. The tirzepatide first month guide covers what to expect in the critical initial period. For those tracking results against specific benchmarks, our tirzepatide before and after results guide and the guide to men tirzepatide before and after provide comparative reference points. The guide to GLP-1 before and after results covers the broader class perspective.
When the drug stops working
Metabolic adaptation occurs on tirzepatide just as it does on any weight loss intervention. As body weight decreases, total daily energy expenditure decreases, and the deficit that produced rapid early weight loss narrows. Some patients experience what they describe as a plateau. Our guides to why you might not be losing weight on tirzepatide, tirzepatide not working anymore, and why you might be gaining weight on tirzepatide address the specific scenarios and solutions. Understanding whether tirzepatide burns fat or just suppresses appetite and whether tirzepatide increases metabolism helps calibrate realistic expectations about the mechanism. The parallel semaglutide guides for not losing weight on semaglutide and four weeks on semaglutide with no weight loss cover similar scenarios for that compound.
Special populations and health conditions
Tirzepatide B12 compounds are used across a diverse population. Several specific groups warrant additional consideration before starting or continuing treatment.
Thyroid conditions
Patients taking levothyroxine for hypothyroidism frequently use GLP-1 therapy concurrently. The interaction requires attention: tirzepatide slows gastric emptying, which can affect levothyroxine absorption timing and completeness. Levothyroxine is typically recommended to be taken on an empty stomach, 30 to 60 minutes before food. The changed gastric transit dynamics on tirzepatide may require dose adjustments monitored by thyroid labs. Our guide to tirzepatide and levothyroxine covers this specific interaction. For Hashimoto thyroiditis, the anti-inflammatory effects of GLP-1 therapy are particularly relevant. Our guide to GLP-1 for Hashimoto disease covers the clinical rationale and practical considerations.
Hormonal therapy interactions
Patients combining tirzepatide with hormone replacement therapy represent a significant and growing population. The metabolic effects of tirzepatide interact with the hormonal environment created by estrogen, progesterone, and testosterone replacement. Weight loss itself affects hormone levels, sex hormone binding globulin, and the distribution of steroid hormones between free and bound fractions. Our guide to HRT and GLP-1 together covers the specific considerations for patients on combined protocols.
Autoimmune and inflammatory conditions
Patients with autoimmune conditions including lupus, rheumatoid arthritis, and Hashimoto thyroiditis have increasing access to GLP-1 therapy, and the anti-inflammatory mechanism provides specific rationale for this use. Our guides to tirzepatide and lupus, GLP-1 and endometriosis, and GLP-1 and lipedema address these specific conditions. The question of whether GLP-1 therapy qualifies as direct immunomodulation or whether the benefits are primarily mediated through weight loss and reduced adipose inflammation is still under active investigation.
Other specific considerations
Patients with migraines should be aware that dehydration and electrolyte shifts early in treatment can trigger or worsen migraines. Our guide to tirzepatide and migraines covers this relationship. GLP-1 therapy is generally not recommended during breastfeeding due to theoretical concerns about infant exposure and caloric restriction during lactation. Our guide to whether you can take GLP-1 while breastfeeding covers the current evidence. For patients researching compounding pharmacy options, our guides to Empower tirzepatide, Strive tirzepatide, 503B compounding pharmacy tirzepatide, and Orderly Meds tirzepatide provide pharmacy-specific information.
Frequently asked questions
What does GLP-1 GIP B12 mean?
It refers to a compounded tirzepatide formulation that combines the dual GLP-1 and GIP receptor agonist (tirzepatide) with vitamin B12. The B12 is added to reduce nausea, prevent B12 deficiency during long-term GLP-1 therapy, and support energy metabolism. See our compounded tirzepatide with B12 guide for a full breakdown.
Why does my GLP-1 GIP B12 compound look pink?
The pink color comes from vitamin B12. Cyanocobalamin and hydroxocobalamin both have intense red pigmentation that turns the solution pink or magenta. The intensity of the color reflects the B12 concentration. Our guide to what color tirzepatide with B12 should be covers normal color variation and when discoloration indicates a problem.
Is tirzepatide B12 the same as Mounjaro?
No. Mounjaro is brand-name tirzepatide manufactured by Eli Lilly. Compounded tirzepatide B12 is prepared by licensed compounding pharmacies using pharmaceutical-grade tirzepatide API with added B12 and other stabilizers. The active receptor targets are the same, but the formulation, source, and regulatory pathway differ. Our guide on whether GLP-1 is the same as Ozempic addresses the broader question of brand versus compound distinctions.
What is the starting dose for compounded tirzepatide B12?
The standard starting dose is 20 units on an insulin syringe (0.2 mL). The actual milligram dose depends on your formulation concentration. At 2 mg/mL, this equals 0.4 mg. Our compounded tirzepatide starting dose guide covers the full protocol. Use the peptide calculator to confirm the milligram dose for your specific vial concentration.
How long does compounded tirzepatide B12 last after reconstitution?
Approximately 28 days when stored at 36 to 46 degrees F (2 to 8 degrees C) in a refrigerator and prepared with bacteriostatic water. Do not freeze. Protect from light. Our tirzepatide fridge life guide covers the specific storage conditions and stability factors. The tirzepatide shelf life guide addresses the broader question of how long the compound remains viable under various conditions.
Does tirzepatide cause B12 deficiency?
Not directly, but the mechanisms of GLP-1 therapy, including slowed gastric emptying and significantly reduced food intake, create conditions that increase B12 deficiency risk over time. This is why B12 is included in compounded formulations. Patients on brand-name tirzepatide without supplemental B12 should have B12 levels monitored periodically, especially after 6 to 12 months of treatment.
Which B12 form is better in compounded tirzepatide, cyanocobalamin or methylcobalamin?
Methylcobalamin is bioactive and does not require hepatic conversion. It is better absorbed and retained by most tissues. Cyanocobalamin is more stable in solution and less expensive. For most patients, cyanocobalamin provides adequate B12 support. For patients with genetic variants affecting B12 metabolism or impaired liver function, methylcobalamin is preferable. See our tirzepatide methylcobalamin guide and cyanocobalamin tirzepatide guide for the full comparison.
What is the difference between tirzepatide glycine B12 and tirzepatide B12 only?
Glycine is an amino acid stabilizer that helps maintain the tirzepatide peptide structure and pH during storage. It does not change the active compound or the patient experience but may improve vial-to-vial consistency and extend stability. Most patients experience identical outcomes on either formulation. See our tirzepatide glycine B12 guide for a detailed comparison, and our tirzepatide glycine guide for the glycine-only formulation context.
Can I take additional oral B12 alongside my compound?
Yes, and it is often advisable for long-term users. The B12 in the compounded vial is injected alongside tirzepatide and is absorbed well. But the dose is not always optimized for B12 repletion specifically. Many practitioners recommend supplementing with oral methylcobalamin or sublingual B12 in addition to the compound, particularly for patients who have been on therapy for more than six months or who have documented low B12 levels. Our GLP-1 vitamin complete guide covers the full supplementation framework for long-term users.
External resources
For researchers serious about optimizing their GLP-1 GIP B12 protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides covering every aspect of compounded tirzepatide therapy, proven escalation protocols, side effect management frameworks, and a community of thousands who have navigated these exact questions with real-world experience.
In case I do not see you, good afternoon, good evening, and good night. May your B12 stay adequate, your dose escalation stay smooth, and your results stay worth it.