Mar 18, 2026

Most people believe tirzepatide works through one simple mechanism: it kills your appetite, you eat less, and you lose weight. End of story. That explanation is not wrong, exactly, but it is dangerously incomplete. And understanding why matters more than you might realize.
The reality is far more interesting.
Tirzepatide does suppress appetite. Powerfully. But research from the SURMOUNT clinical trials, combined with emerging data on adipose tissue signaling and brown fat activation, reveals something that separates this medication from every other weight loss drug that came before it. Tirzepatide appears to engage fat tissue directly, influencing how the body stores, mobilizes, and burns fat through mechanisms that operate independently of how much food you put on your plate. A 28-week study published in Diabetes Care found that tirzepatide reduced fat mass by 9.6 kg more than placebo, and here is the part that surprised researchers, the difference in calorie reduction between tirzepatide and semaglutide was not statistically significant, yet tirzepatide produced 3.8 kg more fat loss. Something beyond appetite suppression is happening.
This guide breaks down every mechanism researchers have identified so far, from the well-established appetite pathways to the cutting-edge discoveries about GIP receptor signaling in fat cells. Whether you are already on a tirzepatide protocol or evaluating your options, understanding how this medication actually works will help you optimize your results and set realistic expectations. SeekPeptides has analyzed the clinical evidence to give you the complete picture.
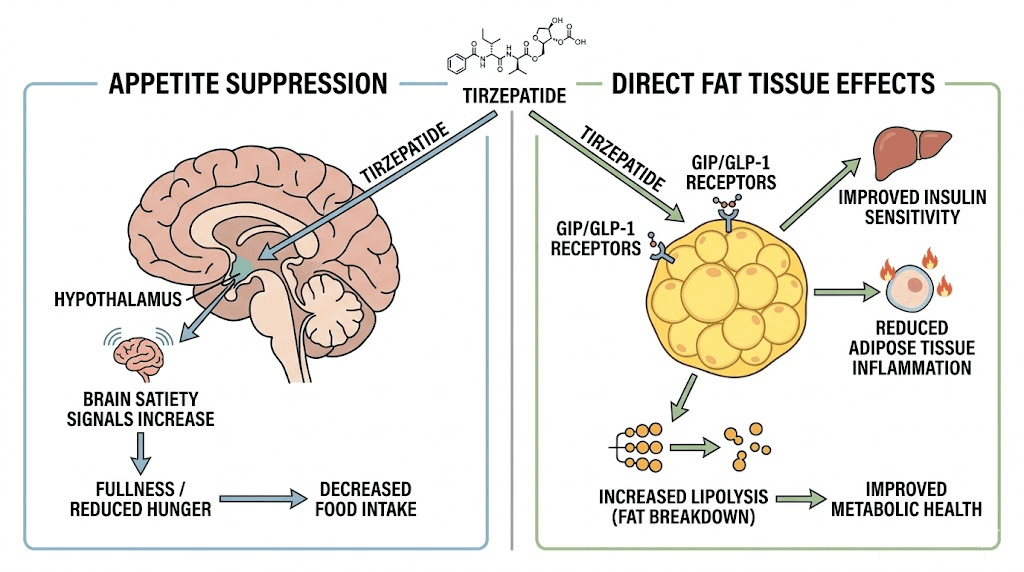
The dual mechanism that makes tirzepatide different
To understand why tirzepatide does more than suppress appetite, you need to understand what makes it structurally unique. Every other GLP-1 medication on the market, including semaglutide, liraglutide, and their branded counterparts, targets a single receptor. Tirzepatide targets two.
It is a dual GIP and GLP-1 receptor agonist.
That distinction matters enormously. The GLP-1 receptor is primarily responsible for the appetite suppression and blood sugar regulation that most people associate with these medications. The GIP receptor, on the other hand, is found directly on fat cells, and its activation triggers a cascade of metabolic effects that researchers are only beginning to fully understand. This dual targeting is why tirzepatide consistently outperforms single-receptor medications in head-to-head clinical comparisons, producing greater weight loss even when appetite reduction between the drugs appears similar.
GLP-1: the appetite suppression pathway
The GLP-1 component of tirzepatide works through well-established mechanisms. When tirzepatide activates GLP-1 receptors in the brain, it amplifies satiety signals, making you feel full faster and stay satisfied longer after meals. It also slows gastric emptying, meaning food moves through your stomach more slowly, which extends that feeling of fullness and reduces the urge to eat again quickly.
These effects are significant. In clinical trials, tirzepatide reduced energy intake by approximately 310 calories per meal compared to placebo during ad libitum feeding tests. For context, that is roughly equivalent to skipping a full meal every day without feeling deprived. Over weeks and months, this calorie deficit compounds dramatically. A 310-calorie daily reduction translates to roughly 2,170 fewer calories per week, or about 0.6 pounds of fat loss per week from appetite suppression alone. The appetite suppression is so effective that many users report needing reminders to eat rather than fighting cravings. Planning meals becomes more about ensuring adequate nutrition and meal variety than fighting hunger.
But here is the critical finding that changed how researchers think about this medication.
When scientists compared the appetite suppression between tirzepatide and semaglutide in a 28-week study, they found something unexpected. The appetite score reductions were nearly identical between the two drugs. The difference in calorie intake during test meals was just 64 calories, which was not statistically significant. Yet tirzepatide produced substantially more fat loss. The researchers stated explicitly that differences in energy intake were not sufficient to explain the different weight outcomes. Something else was contributing to the additional fat loss, and the metabolic effects of GIP receptor activation became the prime suspect.
GIP: the direct fat tissue pathway
This is where tirzepatide separates itself from every other weight loss medication available. The GIP receptor is expressed directly on human adipocytes, which are fat cells. GLP-1 receptors are not found on adipocytes. This means tirzepatide has a direct line of communication with your fat tissue that semaglutide simply does not have.
Imaging studies have confirmed this. When researchers used fluorescent tracers to track where tirzepatide goes in the body, they found it associates with all adipocytes in both human and mouse tissue, as well as along the vasculature surrounding fat deposits. GLP-1 receptor tracers showed no such association with fat cells. Tirzepatide is literally binding to your fat tissue and changing how it functions.
What happens when tirzepatide activates GIP receptors on fat cells? The research reveals a sophisticated, context-dependent response that adapts to your metabolic state. In the fed state, when insulin levels are high, GIP receptor activation enhances insulin signaling in fat tissue, augments glucose uptake into fat cells, and increases the conversion of glucose to glycerol. This improves how efficiently your body handles the nutrients from meals. In the fasted state, when insulin levels drop, GIP receptor activation increases lipolysis, which is the breakdown of stored fat into fatty acids that can be burned for energy. This dual response means tirzepatide helps your body store nutrients efficiently after meals while also promoting fat breakdown between meals.
A 2024 study published in Cell Metabolism confirmed these findings at the molecular level, describing how long-acting GIP receptor agonism modulates both fasted and fed adipose tissue function. The researchers concluded that tirzepatide cooperates with insulin to manage fat storage in the fed state while enhancing fat release when insulin levels are low. This is not appetite suppression. This is direct metabolic regulation of fat tissue.
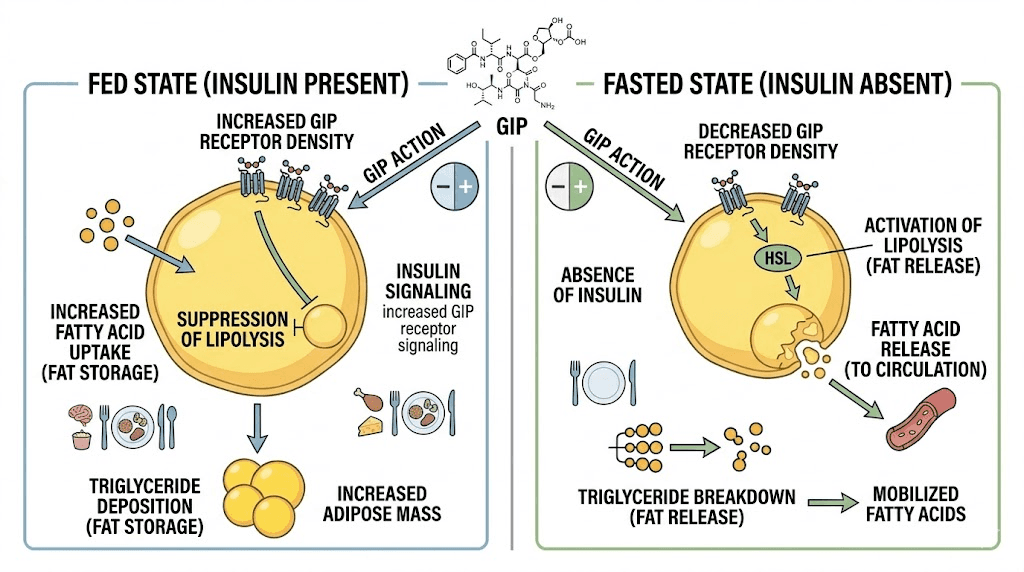
Brown fat activation: the metabolic furnace
One of the most exciting discoveries about tirzepatide came from a study published in early 2026. Researchers at the University of Barcelona found that tirzepatide directly activates brown adipose tissue, a specialized type of fat that burns calories to generate heat rather than storing energy. This is a fundamentally different mechanism from appetite suppression, and it could explain a significant portion of the additional weight loss that tirzepatide produces compared to GLP-1-only medications.
Brown fat is not like the white fat most people think of when they hear the word fat. White fat stores excess calories. Brown fat burns them. It is packed with mitochondria, the cellular powerhouses that convert nutrients into energy, and its primary function is thermogenesis, the production of heat. Newborns have large amounts of brown fat to maintain body temperature, and for years scientists believed adults had little to none. We now know that adults retain metabolically active brown fat, particularly in the neck, collarbone, and spinal regions, and its activity correlates strongly with metabolic health and leanness.
The Barcelona study used obese mice on high-fat diets and compared drug-treated groups with calorie-matched controls. This design was crucial because it isolated the metabolic effects of the drug from the effects of eating less. When the tirzepatide-treated mice ate the exact same number of calories as the control group, they still showed enhanced brown fat activation and greater fat loss. This is the clearest possible demonstration that tirzepatide does something beyond appetite suppression. Same calories in, but more fat burned. Active brown adipose tissue was burning glucose and fat within the body, contributing to weight loss through direct energy expenditure rather than reduced intake. The implications for human users are profound, even if the exact magnitude of this effect in humans still needs to be quantified.
The study also identified that this brown fat activation triggered the production of batokines, signaling molecules released by brown fat that improve metabolism throughout the body. These batokines produce downstream benefits including reduced blood glucose levels, improved fat metabolism, and enhanced overall metabolic function. It is as though activating brown fat sends a ripple effect through the entire metabolic system, improving metabolic health through pathways that have nothing to do with eating less food.
Researchers do stress an important caveat.
These findings come from mouse studies, and the magnitude of brown fat activation in humans may differ. As the lead researcher noted, there may be significant differences between species in metabolism regulation, adipose tissue distribution, and response to drugs. However, the discovery provides a plausible mechanism for the unexplained gap between appetite reduction and fat loss that clinical trials have consistently documented with tirzepatide.
Fat oxidation: burning fat for fuel
Separate from brown fat activation, tirzepatide also appears to increase fat oxidation in white adipose tissue. Fat oxidation is the process by which stored fat is broken down and used as fuel, and it can be measured through changes in the respiratory exchange ratio, a metric that indicates whether the body is primarily burning carbohydrates or fats for energy.
In animal studies, tirzepatide-treated subjects showed decreased respiratory exchange ratios, indicating a shift toward burning fat rather than carbohydrates as a primary fuel source. This finding was confirmed in a clinical study published in Cell Metabolism in early 2025, which found that tirzepatide led to increased fat oxidation in human participants. Importantly, the study also found that tirzepatide did not impact metabolic adaptation, the frustrating phenomenon where the body reduces its metabolic rate during weight loss to conserve energy. This is significant because metabolic adaptation is one of the biggest obstacles to sustained weight loss, and most diets and medications fail to prevent it.
The combination of increased fat oxidation without metabolic adaptation means that tirzepatide users may continue burning fat efficiently even as they lose significant amounts of weight. This could explain why tirzepatide continues working effectively for extended treatment periods, as demonstrated in the SURMOUNT clinical trial program that followed participants for up to 72 weeks.
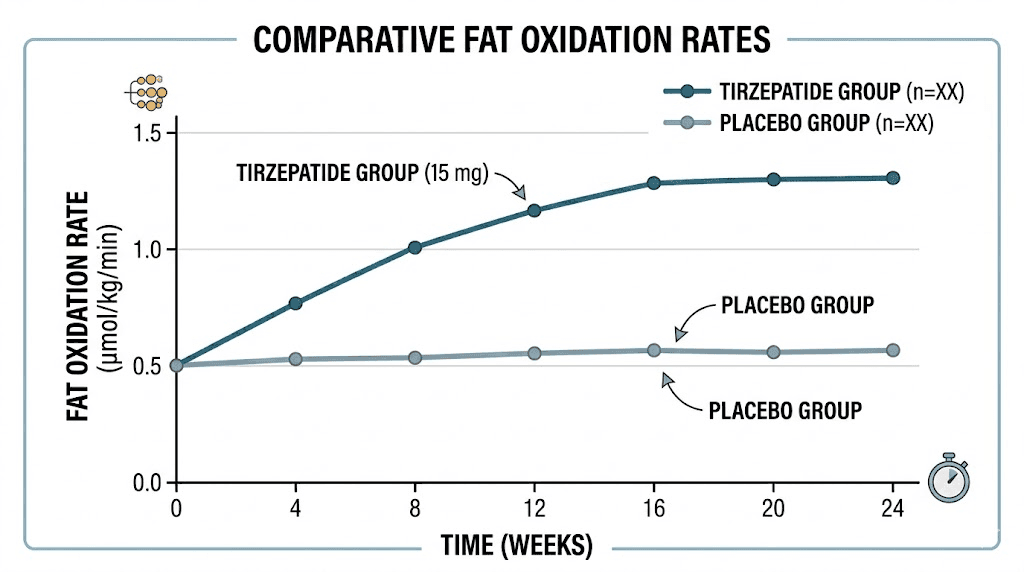
What the SURMOUNT trials reveal about body composition
Clinical data always matters more than mechanistic theories, and the SURMOUNT-1 trial provides some of the most detailed body composition data available for any weight loss medication. A substudy of 160 participants used dual-energy X-ray absorptiometry, the gold standard for measuring body composition, to track exactly what type of tissue people were losing on tirzepatide.
The numbers tell a compelling story.
Over 72 weeks, participants on tirzepatide lost an average of 21.3% of their body weight. That alone is remarkable, but the breakdown is what matters most. Of that total weight loss, 74% came from fat mass and 26% from lean mass. The placebo group, which lost just 5.3% of body weight, showed a nearly identical ratio of 75% fat and 25% lean mass. This means tirzepatide does not preferentially destroy muscle. It produces the same fat-to-lean loss ratio as natural weight loss, just dramatically more of it.
The fat mass numbers are staggering. Tirzepatide reduced fat mass by 33.9% compared to just 8.2% with placebo. In absolute terms, that is a massive reduction in body fat that far exceeds what most people achieve through diet and exercise alone.
Lean mass preservation: the muscle question
One of the biggest concerns with rapid weight loss is muscle loss, and this is a legitimate worry. Lean mass decreased by 10.9% with tirzepatide compared to 2.6% with placebo. However, when you look at the proportional breakdown, the 74/26 fat-to-lean ratio is actually favorable compared to many aggressive diet interventions, where lean mass losses of 30-40% of total weight loss are common.
A separate analysis from the SURPASS-3 trial, which used MRI to assess muscle composition specifically, found that indicators of muscle composition remained stable or showed signs of improvement during tirzepatide treatment. This suggests that while absolute lean mass decreases during significant weight loss, the quality and function of remaining muscle tissue may actually improve. For researchers concerned about maintaining muscle mass during a tirzepatide protocol, combining treatment with resistance training and adequate protein intake can further shift the ratio toward greater fat loss and better lean mass preservation.
These body composition findings have important implications for the original question. If tirzepatide only suppressed appetite, you would expect the fat-to-lean loss ratio to mirror that of calorie restriction alone. The fact that tirzepatide produces 33.9% fat mass reduction while maintaining proportional lean mass suggests that its direct effects on fat tissue, through GIP receptor activation, brown fat stimulation, and enhanced fat oxidation, are contributing to preferential fat loss.
Results across different doses
The SURMOUNT-1 trial tested three doses of tirzepatide: 5mg, 10mg, and 15mg weekly. All three doses produced significant fat loss, but the higher doses showed progressively greater effects. The 15mg dose produced the most dramatic results, with some participants losing over 25% of their starting body weight. Importantly, the fat-to-lean mass ratio remained consistent across all dose groups, meaning higher doses produce more fat loss without disproportionately increasing muscle loss.
This dose-response relationship supports the idea that the fat-burning mechanisms of tirzepatide scale with dose intensity. If the medication only suppressed appetite, you would expect diminishing returns at higher doses as calorie intake reaches a floor. Instead, the progressive improvement suggests that the direct metabolic effects, the GIP-mediated fat tissue changes, intensify with higher doses.
For those considering dose adjustments, SeekPeptides offers dosage calculators and starting dose guides to help researchers navigate the titration process safely. Understanding your exact dose in units is essential for accurate administration and consistent results.

Tirzepatide vs semaglutide: why more fat loss with similar appetite suppression?
The comparison between tirzepatide and semaglutide provides perhaps the strongest evidence that tirzepatide does more than suppress appetite. Both medications are powerful appetite suppressors. Both produce significant weight loss. But tirzepatide consistently produces more fat loss, and the difference cannot be fully explained by differences in how much people eat.
In the 28-week comparative study, both drugs reduced appetite scores by similar amounts. The 5.3-point difference between tirzepatide and semaglutide on appetite measures was not statistically significant. Energy intake during test meals was reduced by both drugs, with tirzepatide showing only a 64-calorie greater reduction than semaglutide, again not statistically significant. Yet tirzepatide produced 4.3 kg more total weight loss and 3.8 kg more fat mass loss than semaglutide.
Where did that extra fat loss come from?
The researchers themselves pointed to tirzepatide unique GIP receptor activity. Because GIP receptors are found on fat cells but GLP-1 receptors are not, tirzepatide has a direct pathway to influence fat metabolism that semaglutide simply lacks. The additional fat loss likely comes from enhanced lipolysis during fasting periods, improved fat oxidation, and potentially brown fat activation, all mechanisms that operate independently of how much food someone consumes.
This comparison is particularly informative because it controls for the appetite suppression variable. If both drugs suppress appetite equally but one produces significantly more fat loss, the additional fat loss must come from non-appetite mechanisms. For anyone deciding between semaglutide and tirzepatide, this distinction matters. Semaglutide is an excellent medication for appetite suppression and weight loss. Tirzepatide does everything semaglutide does, and adds direct fat tissue engagement on top.
The GIP advantage explained
Understanding why GIP receptor activation matters requires a closer look at what happens at the cellular level. When tirzepatide binds to GIP receptors on adipocytes, it triggers a signaling cascade that fundamentally changes how those fat cells operate. The research describes a model where long-acting GIP receptor activation modulates both storage and release functions in fat tissue, acting as a metabolic switch that responds to the body nutritional state.
In practical terms, this means tirzepatide helps your body become better at using fat as fuel. During the hours between meals, when insulin levels are naturally low, the GIP-mediated increase in lipolysis means your fat cells are releasing more stored fatty acids into the bloodstream, where they can be burned for energy. During meals, when insulin rises, the enhanced insulin signaling means nutrients are handled more efficiently, reducing the likelihood that excess glucose gets converted to new fat stores.
This bidirectional effect, improving both fat burning and nutrient handling, is unique to tirzepatide and other GIP-targeting medications. It is why the comparison data consistently favors tirzepatide for total fat mass reduction, even when appetite effects are similar. And it is why some researchers who have plateaued on semaglutide find renewed results when switching to tirzepatide, which provides that additional metabolic pathway for fat loss.
For those currently on semaglutide who are considering a switch, the conversion chart provides guidance on appropriate dose equivalencies.
The CNS-adipose tissue axis: a newly discovered pathway
Research published in early 2025 uncovered another mechanism by which tirzepatide influences fat metabolism. Scientists discovered a central nervous system to adipose tissue axis, a direct neural connection between the brain and fat tissue that tirzepatide activates to promote fat utilization.
The study, published in PubMed, found that tirzepatide effects on fat tissue were mediated by increased lipolytic capacity in white adipose tissue and enhanced thermogenesis in brown and beige adipose tissues. But the truly novel finding was how these effects were transmitted. Rather than acting directly on fat cells alone, tirzepatide also activates neural pathways that connect the brain to fat deposits through the sympathetic nervous system.
Specifically, the researchers found that these metabolic effects were primarily mediated by sympathetic nervous system innervation of adipose tissues. Changes in neuronal activity were detected in two key brain regions: the dorsomedial hypothalamus and the nucleus of the solitary tract within the hindbrain. These brain areas are known to regulate energy balance, body temperature, and metabolic rate, and tirzepatide appears to modulate their activity in ways that increase fat utilization throughout the body.
This finding adds another layer to the fat-burning question. Tirzepatide does not just suppress appetite through brain signaling and engage fat cells through GIP receptors. It also activates a neural network that tells your body to increase fat burning at the systemic level. The brain, in essence, sends instructions to fat tissue to release and burn stored energy, creating a coordinated metabolic response that goes far beyond simply eating less. This three-pronged approach, brain-level appetite control, direct fat cell engagement, and neural-adipose communication, is unlike anything seen in previous weight loss medications. It represents a fundamentally new paradigm in how pharmaceutical interventions can address excess body fat.
This CNS-adipose tissue connection may also help explain why tirzepatide users often report feeling increased energy even while eating significantly fewer calories. If the medication is actively directing the body to mobilize fat for fuel, the energy from those fat stores could compensate for reduced calorie intake, preventing the fatigue and low energy that typically accompany aggressive dieting.
Does tirzepatide increase your metabolism?
This is a question that researchers frequently ask, and the answer requires some nuance. Tirzepatide does not appear to increase basal metabolic rate in the traditional sense. A clinical study found that tirzepatide did not impact metabolic adaptation, meaning it neither increased nor decreased the expected metabolic rate for a given body weight. But that finding is actually good news.
Why? Because during significant weight loss, the body normally reduces its metabolic rate by more than what would be expected from the loss of tissue alone. This is metabolic adaptation, and it is the primary reason why most diets fail long term. Your body fights back against weight loss by becoming more energy efficient, burning fewer calories at rest and during activity, making it progressively harder to continue losing weight.
Tirzepatide prevents this. By maintaining expected metabolic rates during weight loss, tirzepatide allows fat burning to continue at normal efficiency even as body weight drops significantly. Combined with increased fat oxidation, this means the body continues to use fat as fuel at expected rates rather than downregulating energy expenditure to conserve fat stores.
Think of it this way. Most weight loss interventions are like driving a car while someone gradually reduces the engine power. Tirzepatide keeps the engine running at full capacity while simultaneously improving fuel efficiency, directing the body to burn fat rather than other fuel sources. The result is sustained, progressive fat loss that does not hit the typical metabolic wall.
The thermogenic contribution
While tirzepatide may not increase overall metabolic rate, the brown fat activation discussed earlier does represent a form of increased energy expenditure. Active brown fat generates heat through a process called non-shivering thermogenesis, which burns calories without requiring physical activity. This localized increase in energy expenditure may not show up as a dramatic change in overall metabolic rate measurements, but it does contribute to total daily energy expenditure and, consequently, to fat loss.
Some tirzepatide users report feeling cold during treatment, which may seem counterintuitive if brown fat is generating extra heat. However, the sensation of feeling cold can result from reduced calorie intake and lower body fat insulation, even if brown fat thermogenesis is simultaneously increasing. These are separate mechanisms, and the cold sensation is a common side effect that does not indicate a problem with the medication fat-burning mechanisms.
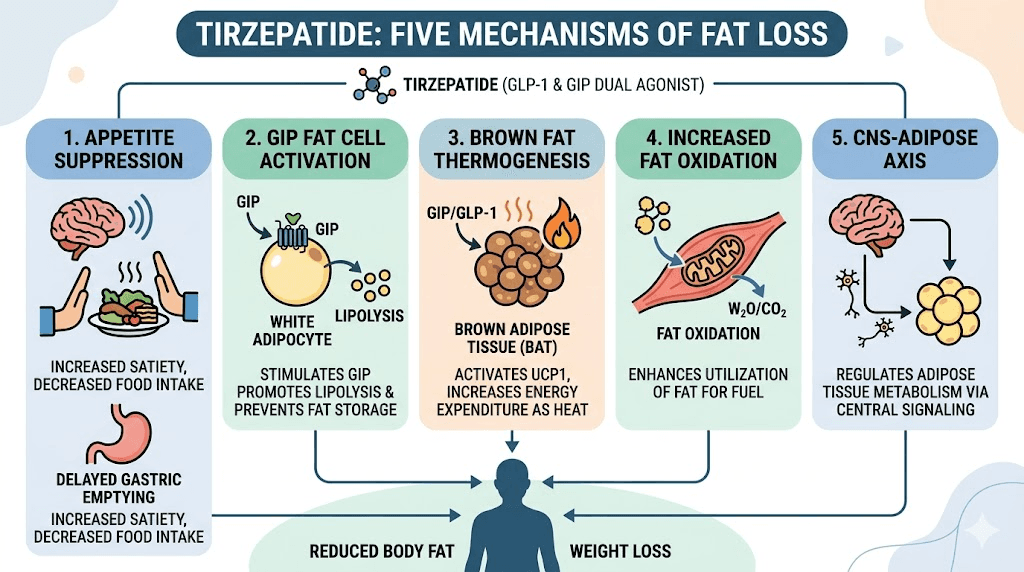
How to maximize fat burning on tirzepatide
Understanding that tirzepatide works through multiple fat-loss mechanisms has practical implications for how you approach your protocol. While the medication does significant metabolic work on its own, certain strategies can amplify each of its fat-burning pathways.
Optimize your dosing schedule
The GIP-mediated fat-burning effects of tirzepatide are influenced by your metabolic state, particularly the balance between fed and fasted conditions. Because GIP receptor activation increases lipolysis during fasting (when insulin is low) and improves nutrient handling during feeding (when insulin is high), maintaining clear metabolic transitions between fed and fasted states may enhance these effects.
This does not mean extreme fasting protocols. It means avoiding constant snacking that keeps insulin elevated throughout the day. Eating structured meals with clear breaks between them allows your body to cycle between the fed-state benefits (improved nutrient handling) and fasted-state benefits (enhanced fat release) that tirzepatide GIP component provides.
Consistency in injection timing also matters. Taking your injection at the same time each week, ideally on the same day, ensures steady drug levels that support consistent metabolic effects. Some users find that injecting in the evening helps manage side effects while allowing the appetite suppression to align with overnight fasting.
Prioritize protein to preserve muscle
The SURMOUNT-1 body composition data showed that 26% of weight loss comes from lean mass. While this ratio is acceptable and consistent with natural weight loss, you can shift it further toward fat loss by prioritizing protein intake. Adequate protein stimulates muscle protein synthesis, signals your body to preserve lean tissue during calorie restriction, and has a higher thermic effect than carbohydrates or fats, meaning more calories are burned simply digesting it.
Aim for 1.0-1.2 grams of protein per pound of goal body weight daily. This is higher than standard recommendations, but the research supports higher protein intake during active weight loss to preserve muscle mass. Protein shakes can help bridge the gap when appetite suppression makes eating large meals difficult. Focus on high-quality sources like lean meats, fish, eggs, and dairy, and spread intake across multiple meals rather than concentrating it in one sitting.
For specific meal planning guidance, the tirzepatide meal plan provides structured templates that balance protein needs with the reduced appetite most users experience.
Add resistance training
If there is one intervention that dramatically improves body composition outcomes during tirzepatide treatment, it is resistance training. Lifting weights sends a powerful signal to your body that muscle tissue is needed and should be preserved, even during calorie deficit. The combination of tirzepatide fat-burning mechanisms with resistance training muscle-preserving stimulus creates a synergistic effect that maximizes the fat-to-lean loss ratio.
You do not need an extreme program. Three to four sessions per week focusing on compound movements like squats, deadlifts, presses, and rows is sufficient. The key is progressive overload, gradually increasing the weight or volume over time to maintain the muscle-preservation signal. Even moderate resistance training can shift the body composition ratio from the 74/26 fat-to-lean split observed in the trials toward 80/20 or better. Research on peptides for muscle growth shows that certain peptides can further support this process, and combining resistance training with tirzepatide metabolic effects creates a synergy that maximizes body recomposition.
The timing of workouts relative to meals matters too. Training in a slightly fasted state, when the GIP-mediated lipolysis is most active, may theoretically enhance fat utilization during exercise. However, having adequate fuel for performance is equally important, so a small protein-rich meal 1-2 hours before training often strikes the right balance. Post-workout protein intake within 1-2 hours helps maximize the muscle-preservation signal while the metabolic effects of both exercise and tirzepatide are elevated.
Support fat oxidation with strategic supplements
Several supplements can support the fat oxidation pathways that tirzepatide activates. L-carnitine, which shuttles fatty acids into mitochondria for burning, may enhance the increased fat oxidation that tirzepatide promotes. Some compounded formulations already include levocarnitine alongside tirzepatide for this reason.
B vitamins play essential roles in energy metabolism, and B12 in particular supports the metabolic processes involved in fat utilization. Glycine supports multiple metabolic pathways and is commonly combined with tirzepatide in compounded formulations. Niacinamide (vitamin B3) supports cellular energy production and has been shown to influence fat metabolism.
Fiber supplements support gut health and can help manage some of the digestive side effects while also promoting healthy blood sugar regulation that complements tirzepatide metabolic effects. The complete GLP-1 supplement guide covers additional options worth considering.
Manage side effects to maintain consistency
The fat-burning benefits of tirzepatide require consistent use, and side effects that lead to missed doses or early discontinuation will undermine results. The most common side effects, including constipation, bloating, and headaches, are typically manageable and often improve over time as the body adjusts.
If you experience significant muscle pain, body aches, or persistent fatigue, these may indicate a need for dose adjustment rather than discontinuation. Microdosing protocols allow for a more gradual introduction that minimizes side effects while still providing metabolic benefits. The key is finding a dose that you can maintain consistently, because the fat-burning mechanisms of tirzepatide work best with steady, uninterrupted treatment.
For those experiencing GLP-1 related fatigue, proper nutrition and hydration often resolve the issue. Hair thinning, another concern for some users, can be managed with appropriate nutritional support including adequate protein and specific micronutrients.
What happens when you stop: does the fat come back?
Understanding the fat-burning mechanisms of tirzepatide raises an important question about what happens when treatment ends. If the medication is actively promoting fat oxidation, brown fat activation, and enhanced lipolysis through GIP receptor signaling, what happens when those signals stop?
The clinical data provides a sobering answer. In the SURMOUNT-4 trial, participants who were switched from tirzepatide to placebo after achieving significant weight loss regained a substantial portion of their lost weight over the following months. This is consistent with the understanding that tirzepatide metabolic effects require ongoing treatment to maintain.
However, the regain is not instantaneous, and it does not mean the medication only suppresses appetite. The weight regain follows a gradual trajectory that suggests the metabolic changes, including improved insulin sensitivity and fat utilization patterns, take time to fully reverse. This provides a window during which lifestyle modifications can be established to partially sustain the metabolic improvements achieved during treatment.
For those approaching the end of a treatment course, tapering protocols rather than abrupt cessation may help maintain more of the metabolic benefits while the body adjusts. The goal is to establish eating patterns and exercise habits during treatment that can sustain lower body weight even without the medication active metabolic support.
Common misconceptions about tirzepatide and fat burning
Several persistent myths circulate about how tirzepatide works, and understanding the science helps dispel them.
Myth: tirzepatide is just a fancy appetite suppressant
This is the most common misconception, and the research clearly contradicts it. While appetite suppression is a major component of tirzepatide weight loss effects, the GIP-mediated direct engagement with fat tissue, brown fat activation, increased fat oxidation, and CNS-adipose tissue axis activation all represent mechanisms that operate independently of appetite. The clinical evidence showing greater fat loss with similar appetite suppression compared to semaglutide is perhaps the strongest proof that tirzepatide does more than reduce food intake. If appetite suppression were the only mechanism, tirzepatide and semaglutide would produce identical fat loss, which they clearly do not.
Myth: all the weight loss is muscle
The SURMOUNT-1 body composition data directly refutes this. The 74% fat and 26% lean mass composition of weight loss is proportionally identical to natural weight loss patterns. Tirzepatide does not preferentially destroy muscle tissue. With proper nutrition and resistance training, the lean mass preservation can be even better than what the clinical trials demonstrated.
Myth: tirzepatide works the same way as semaglutide
While both medications activate GLP-1 receptors, tirzepatide additional GIP receptor activity makes it a fundamentally different medication. The direct effects on fat tissue, the dual metabolic regulation, and the greater fat mass reduction documented in head-to-head comparisons all demonstrate that tirzepatide works through pathways that semaglutide does not access. This is why people who have switched between the two medications often report different experiences with each.
Myth: higher doses just suppress appetite more
The dose-response data suggests otherwise. Higher doses of tirzepatide produce greater fat loss without proportionally greater appetite suppression, indicating that the additional fat loss at higher doses comes from intensified metabolic effects rather than simply eating even less. This is consistent with the GIP receptor activation model, where higher drug concentrations produce greater direct effects on fat tissue metabolism.
Myth: you cannot influence the results beyond taking the medication
While tirzepatide is powerful on its own, lifestyle factors significantly influence outcomes. Protein intake, resistance training, food choices, consistent dosing, adequate sleep, and stress management all modulate the metabolic environment in which tirzepatide operates. Users who combine the medication with intentional lifestyle changes consistently achieve better body composition outcomes than those who rely on the drug alone.
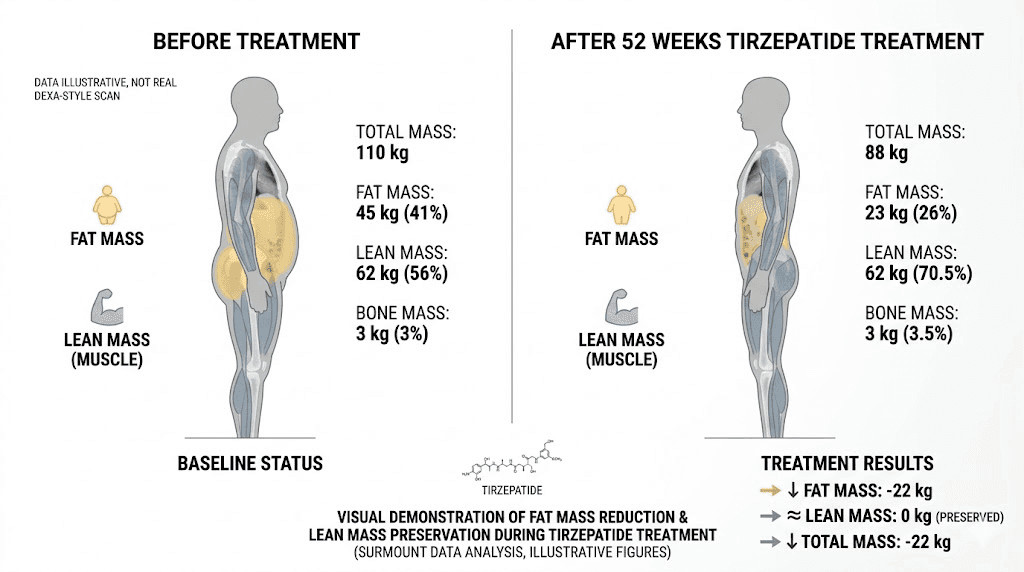
How tirzepatide compares to other weight loss approaches for fat burning
Putting tirzepatide fat-burning mechanisms in context helps illustrate just how different this medication is from traditional weight loss approaches.
Context matters when evaluating each approach. No single weight loss strategy works in isolation, and tirzepatide greatest strength may be how well it combines with other interventions. The medication handles the metabolic and appetite components while traditional approaches like exercise and nutrition handle the structural and behavioral components. Together, they produce outcomes that neither could achieve independently.
Tirzepatide vs calorie restriction alone
Pure calorie restriction works entirely through creating an energy deficit. Your body responds by burning stored energy, but it also triggers metabolic adaptation, reducing metabolic rate by 15-25% beyond what would be expected from tissue loss alone. Additionally, calorie restriction alone typically results in lean mass losses of 30-40% of total weight lost, worse than the 26% seen with tirzepatide. Tirzepatide adds direct fat tissue engagement and prevents metabolic adaptation, resulting in more fat loss and better lean mass preservation.
Tirzepatide vs exercise for fat burning
Exercise, particularly resistance training combined with cardiovascular work, is excellent for fat oxidation and muscle preservation. However, most people cannot exercise enough to overcome a poor diet, and the total fat loss achievable through exercise alone is modest compared to pharmaceutical intervention. The ideal approach combines tirzepatide metabolic effects with regular exercise, using each to amplify the other. Exercise enhances the fat oxidation pathways that tirzepatide activates, while tirzepatide creates the calorie deficit that makes exercise-induced fat burning more impactful.
Tirzepatide vs other GLP-1 medications
As discussed, semaglutide provides powerful appetite suppression and significant weight loss but lacks the GIP-mediated direct fat tissue effects. Liraglutide is a shorter-acting GLP-1 agonist with less potent effects overall. Neither can match tirzepatide combination of appetite suppression and direct metabolic fat-burning mechanisms.
Emerging medications like retatrutide, which targets three receptors (GLP-1, GIP, and glucagon), may add yet another fat-burning pathway through glucagon-mediated increases in energy expenditure. Survodutide and mazdutide represent other next-generation approaches, while cagrisema combines semaglutide with cagrilintide for a different dual-mechanism approach. The field is rapidly evolving, but tirzepatide remains the best-studied dual agonist with the most robust evidence for direct fat tissue engagement.
Tirzepatide vs phentermine and other appetite suppressants
Phentermine works purely as an appetite suppressant by stimulating the release of norepinephrine in the brain. It creates a calorie deficit through reduced food intake, nothing more. There is no direct fat tissue engagement, no improved insulin sensitivity, no brown fat activation. While phentermine can produce meaningful short-term weight loss of 5-10% over 3-6 months, the quality of that weight loss differs significantly from tirzepatide. Phentermine users typically lose a higher proportion of lean mass because the medication does not provide any of the metabolic support that preserves muscle or preferentially targets fat stores. Additionally, phentermine is only approved for short-term use due to cardiovascular concerns, whereas tirzepatide has demonstrated safety and efficacy over treatment periods exceeding 72 weeks.
Some researchers combine phentermine with tirzepatide for enhanced appetite suppression during specific phases of their protocol, but this is an off-label combination that requires careful medical supervision.
Tirzepatide vs traditional fat burners
Traditional over-the-counter fat burners, including caffeine, green tea extract, and thermogenic supplements, work primarily by modestly increasing metabolic rate or enhancing fat oxidation. Their effects are typically small, often 50-100 extra calories burned per day, and they do not address appetite or fat tissue signaling. Tirzepatide operates at a fundamentally different scale and through fundamentally different mechanisms, producing fat loss that no supplement can match.
That said, some supplements may complement tirzepatide effects. Berberine, for instance, has its own effects on glucose metabolism and may work synergistically with tirzepatide. Metformin is another medication that some researchers combine with tirzepatide for enhanced metabolic effects. And phentermine, while primarily an appetite suppressant, may provide additive effects when combined with tirzepatide under medical supervision.
The timeline of fat burning: what to expect and when
Understanding the timeline of tirzepatide fat-burning effects helps set realistic expectations and identify whether your protocol is working optimally. Too many people judge the medication effectiveness in the first two weeks, before the most powerful metabolic mechanisms have even had time to engage. Patience during the titration phase is not just a suggestion. It is essential for giving the full spectrum of fat-burning pathways time to activate.
Week 1-4: appetite effects dominate
During the initial titration period, appetite suppression is the primary driver of weight loss. Most users start at 2.5mg weekly and notice reduced hunger within the first few days. The metabolic effects of GIP receptor activation are beginning, but the dose is typically too low for these effects to be prominent. Weight loss during this phase, usually 2-5 pounds, comes primarily from reduced calorie intake.
The first dose experience varies significantly between individuals, but most people notice appetite changes before any other effects.
Week 4-12: metabolic effects ramp up
As doses increase through the titration schedule, the GIP-mediated fat tissue effects become more pronounced. This is when many users notice changes beyond just eating less, including shifts in body shape that suggest preferential fat loss, improved energy levels, and sometimes increased warmth or temperature sensitivity as brown fat activation contributes to thermogenesis.
The weight loss timeline typically shows acceleration during this phase, with many users losing 1-2 pounds per week. Fat loss becomes more noticeable as the direct metabolic effects layer on top of the appetite suppression.
Week 12-36: sustained fat burning
By this point, most users are on their maintenance dose and experiencing the full spectrum of tirzepatide fat-burning mechanisms. The combination of appetite suppression, GIP-mediated fat tissue engagement, brown fat activation, increased fat oxidation, and CNS-adipose axis signaling all contribute to continued, progressive fat loss.
This is the phase where before and after results become most dramatic. Body composition shifts are visible, and many users report that their body shape changes more than the scale suggests, indicating preferential fat loss from metabolically active visceral fat stores.
Week 36+: optimization and maintenance
After significant weight loss, the rate of fat burning naturally slows as there is less excess fat to lose. This is not a plateau in the traditional sense. It is the body approaching a new equilibrium. The metabolic effects of tirzepatide continue, but the calorie deficit narrows as the body requires fewer calories to maintain a lower weight.
Some users at this stage explore microdosing or adjusted schedules to maintain their results with a lower dose. Others continue at their full dose to maximize the metabolic benefits. The appropriate approach depends on individual goals, side effect tolerance, and how much additional fat loss is desired.
At this stage, the importance of body composition tracking becomes critical. The scale may show minimal movement while the body is still actively recomposing, replacing remaining excess fat with lean tissue, particularly if resistance training is part of the protocol. Waist measurements, progress photos, and how clothing fits often tell a more accurate story than body weight alone during this optimization phase. The compound dosage chart can help guide dose adjustments during this period.
The role of insulin sensitivity in fat burning
One mechanism that often gets overlooked in discussions of tirzepatide fat-burning effects is its impact on insulin sensitivity. Insulin resistance is both a cause and consequence of excess body fat, creating a vicious cycle where the body becomes less efficient at using glucose for energy and increasingly likely to store calories as fat.
Tirzepatide breaks this cycle through multiple pathways. The GIP component directly improves insulin signaling in adipose tissue, enhancing glucose disposal in fat cells. Research has shown that even in the absence of GLP-1 receptor-induced weight loss, the GIP agonist component of tirzepatide independently improved insulin sensitivity. This weight-independent insulin sensitization means that even before significant fat loss occurs, tirzepatide is improving the metabolic environment in ways that favor fat burning over fat storage.
As insulin sensitivity improves, the body becomes more metabolically flexible, better able to switch between burning carbohydrates and fats depending on availability. This metabolic flexibility supports the increased fat oxidation observed in clinical studies and may contribute to the sustained fat loss that tirzepatide users experience over extended treatment periods.
For users with type 2 diabetes or significant insulin resistance, these effects are particularly meaningful. The improvement in insulin sensitivity addresses a root cause of fat accumulation rather than just treating the symptom of excess appetite. This is another reason why tirzepatide does more than suppress appetite. It fundamentally changes the metabolic environment in ways that favor fat loss.
Visceral fat vs subcutaneous fat: which does tirzepatide target?
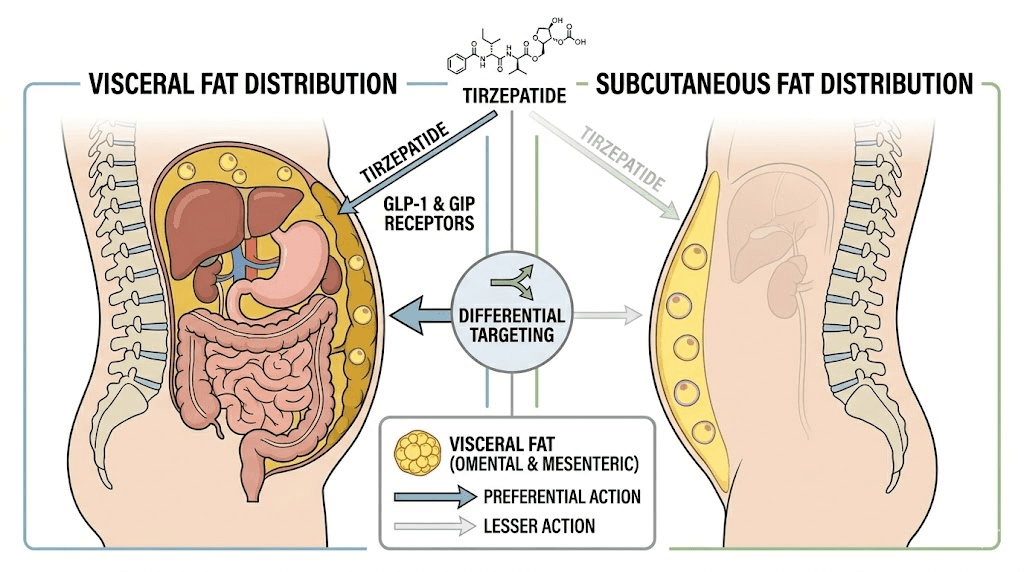
Not all fat is created equal, and understanding where tirzepatide has its greatest impact matters for both health outcomes and visible results. The human body stores fat in two primary locations: subcutaneous fat, which sits just beneath the skin and is what you can pinch, and visceral fat, which surrounds internal organs deep within the abdominal cavity. These two types of fat behave very differently, respond differently to metabolic signals, and carry dramatically different health risks.
Visceral fat is the dangerous one. It is metabolically active in harmful ways, secreting inflammatory cytokines, disrupting insulin signaling, and contributing to cardiovascular disease, type 2 diabetes, and metabolic syndrome. People with high visceral fat levels, even if their overall body weight appears normal, face elevated health risks. Subcutaneous fat, while cosmetically unwanted, is relatively benign from a health perspective and actually serves important functions including energy storage, insulation, and hormone regulation.
The clinical evidence suggests that tirzepatide preferentially reduces visceral fat. In the SURMOUNT trials, the improvements in metabolic markers like insulin sensitivity, blood glucose, triglycerides, and inflammatory markers exceeded what would be expected from total weight loss alone. This pattern is consistent with preferential visceral fat reduction, because visceral fat drives metabolic dysfunction more aggressively than subcutaneous fat. When visceral fat decreases, metabolic improvements are disproportionately large compared to the absolute amount of fat lost.
The GIP receptor signaling pathway may explain this preferential targeting. Visceral fat has higher metabolic activity and different receptor expression patterns than subcutaneous fat. The GIP-mediated increases in lipolysis during fasting states may be particularly effective at mobilizing visceral fat stores, which are more metabolically responsive than subcutaneous deposits. This could explain why many tirzepatide users report that their waist measurement decreases faster than other body dimensions, as visceral fat loss reduces abdominal girth more dramatically than subcutaneous fat loss elsewhere.
For researchers tracking their results, waist circumference is one of the best proxy measurements for visceral fat loss. A decrease of 2 inches or more in waist measurement typically indicates significant visceral fat reduction, with corresponding improvements in metabolic health. The dramatic transformations many users report, where they look substantially different despite modest scale weight changes, often reflect this preferential visceral fat targeting.
The metabolic cascade of visceral fat reduction
When tirzepatide reduces visceral fat, a cascade of metabolic improvements follows. Insulin sensitivity improves as the inflammatory burden from visceral fat decreases. Blood pressure often drops as vascular inflammation subsides. Triglyceride levels normalize as the liver receives fewer inflammatory signals from surrounding visceral fat. These improvements happen independently of total weight loss, meaning someone who loses 20 pounds primarily from visceral fat will see dramatically better health outcomes than someone who loses 20 pounds primarily from subcutaneous fat.
This is part of what makes the question of whether tirzepatide just suppresses appetite so important. If it only reduced food intake, the pattern of fat loss would be non-preferential, pulling from both visceral and subcutaneous stores proportionally. The clinical evidence of disproportionate metabolic improvement suggests that the GIP-mediated mechanisms are doing something more targeted, directing the body to mobilize the most metabolically harmful fat deposits first.
Who benefits most from tirzepatide fat-burning effects?
While tirzepatide is effective across a broad range of body types and conditions, certain individuals may benefit more from its dual fat-burning mechanisms.
People with high visceral fat
Individuals carrying significant visceral fat, often identified by a waist circumference exceeding 40 inches for men or 35 inches for women, may see outsized benefits from tirzepatide GIP-mediated fat tissue effects. The direct engagement with adipose tissue metabolism, combined with preferential visceral fat mobilization, addresses the most health-threatening fat deposits first. These individuals often see metabolic improvements that precede visible changes on the scale, including better blood glucose control, improved energy levels, and reduced inflammation.
People who have plateaued on semaglutide
Because tirzepatide offers fat-burning mechanisms that semaglutide lacks, individuals who have reached a plateau on semaglutide may experience renewed fat loss when switching to tirzepatide. The addition of GIP receptor-mediated fat tissue engagement provides a second pathway for fat loss that was completely inactive on semaglutide. The semaglutide-to-tirzepatide transition has become increasingly common for this reason.
People with insulin resistance
The GIP component of tirzepatide directly improves insulin signaling in adipose tissue, making it particularly beneficial for people with insulin resistance or type 2 diabetes. These individuals often have impaired ability to switch between burning carbohydrates and fats, a condition called metabolic inflexibility. Tirzepatide GIP-mediated effects improve this metabolic flexibility, allowing the body to more efficiently burn fat during fasting periods. The result is enhanced fat loss that goes beyond what appetite suppression alone would achieve in insulin-resistant individuals.
People with significant weight to lose
The SURMOUNT-1 data showed that the fat-to-lean mass ratio remained consistent across weight loss tertiles, but the absolute amount of fat lost was naturally greater in individuals with more excess weight. People with higher BMIs have more fat tissue with more GIP receptors, potentially amplifying the direct fat-burning effects. The clinical trials enrolled participants with BMIs of 30 or above (or 27 with comorbidities), and the greatest absolute fat mass reductions occurred in participants with the highest starting weights.
Active individuals who want body recomposition
Athletes and active individuals who want to reduce body fat while preserving or building muscle may benefit particularly from combining tirzepatide with structured exercise. The medication fat-burning mechanisms create an environment where fat is being actively mobilized and oxidized, while resistance training provides the signal to preserve and build lean tissue. This combination, supported by adequate protein intake and potentially testosterone optimization in appropriate candidates, can produce dramatic body recomposition that neither exercise nor medication alone could achieve.
Understanding your specific profile helps set appropriate expectations and optimize your protocol. SeekPeptides members access detailed protocol recommendations tailored to individual goals, body composition, and metabolic status.
Emerging research: what we still do not know
Despite the growing body of evidence, several important questions about tirzepatide fat-burning mechanisms remain unanswered.
First, the brown fat activation studies were conducted in mice, and the magnitude of this effect in humans has not been directly quantified. While the mechanism is plausible and consistent with clinical observations, we need human studies with brown fat imaging to confirm and measure this effect.
Second, the relative contribution of each fat-burning mechanism, appetite suppression, GIP receptor activation, brown fat thermogenesis, enhanced fat oxidation, and CNS-adipose axis signaling, has not been precisely quantified. We know all these pathways are active, but we do not know what percentage of total fat loss each one contributes. Future research with selective pathway inhibitors could help answer this question.
Third, individual variation in GIP receptor expression and brown fat activity means that some people may respond more to the metabolic fat-burning effects than others. This could explain why some tirzepatide users experience dramatic body composition changes while others with similar appetite suppression see more modest fat loss. Genetic and physiological factors that predict metabolic responsiveness are an active area of investigation.
Fourth, long-term effects beyond 72 weeks are still being studied. The SURMOUNT-MAINTAIN trial is examining whether the metabolic benefits of tirzepatide persist with continued treatment or whether the body develops tolerance to the fat-burning mechanisms over time. Early indications suggest that the metabolic effects remain active with continued treatment, but long-term data over years rather than months is still needed to confirm this.
Fifth, the interaction between tirzepatide fat-burning mechanisms and other medications remains an active area of investigation. How does metformin affect the GIP-mediated fat tissue changes? Does NAD supplementation enhance the brown fat activation? These are questions that clinicians and researchers are actively working to answer, and the results will help optimize treatment protocols for maximum fat loss.
SeekPeptides continues to follow this research closely, and members receive updates as new findings emerge. The science of GIP receptor signaling in fat tissue is still young, and each new study reveals additional complexity in how tirzepatide interacts with the body metabolic systems.
Practical implications: what this means for your protocol
If you are currently using tirzepatide or considering starting, understanding the dual mechanism has several practical implications:
Do not judge effectiveness by appetite alone. Some users worry that tirzepatide is not working because their appetite returns partially at certain doses. But the fat-burning mechanisms operate independently of appetite. If you are losing fat even when you feel hungry, the metabolic effects are working. Track body measurements and body fat percentage, not just subjective appetite changes.
Give higher doses time to work their metabolic magic. The GIP-mediated effects scale with dose. If you are tolerating your current dose well but feel the fat loss has slowed, a dose increase may activate stronger metabolic effects beyond just further appetite suppression. Discuss dose titration with your provider and refer to the dose chart for guidance.
Support the fat-burning pathways with your lifestyle. Structured meals with adequate protein support GIP cycling between fed and fasted states. Resistance training preserves lean mass and may enhance fat oxidation. Adequate sleep supports hormonal profiles that favor fat burning. Cold exposure, while speculative, could theoretically enhance brown fat activation.
Track the right metrics. Scale weight alone can be misleading because it does not distinguish between fat and lean mass loss. Body measurements (waist, hips, chest, arms, thighs) provide better feedback on fat loss specifically. If available, DEXA scans every 3-6 months give the most accurate body composition data. The peptide calculator tools on SeekPeptides can help with dosing precision.
Be patient with the process. The multiple fat-burning mechanisms of tirzepatide work on different timescales. Appetite suppression is immediate. GIP-mediated fat tissue changes take weeks to fully establish. Body composition changes become visible over months. Trust the process, maintain consistency, and allow the full spectrum of mechanisms to contribute to your results.
Frequently asked questions
Does tirzepatide directly burn fat?
Yes, through multiple mechanisms beyond appetite suppression. Tirzepatide activates GIP receptors directly on fat cells, promotes fat oxidation, stimulates brown adipose tissue thermogenesis, and engages a CNS-adipose tissue axis that increases fat utilization. These mechanisms work independently of reduced food intake, as demonstrated by clinical studies showing greater fat loss with tirzepatide compared to semaglutide despite similar appetite suppression.
How much of tirzepatide weight loss is from fat vs muscle?
According to the SURMOUNT-1 body composition substudy, 74% of weight loss on tirzepatide comes from fat mass and 26% from lean mass. This ratio is proportionally identical to natural weight loss and can be improved with adequate protein intake and resistance training.
Why does tirzepatide cause more fat loss than semaglutide?
Tirzepatide is a dual GIP and GLP-1 receptor agonist, while semaglutide targets only GLP-1 receptors. GIP receptors are found directly on fat cells, giving tirzepatide a direct pathway to influence fat metabolism that semaglutide lacks. In head-to-head studies, tirzepatide produced 3.8 kg more fat mass loss than semaglutide despite similar appetite suppression, confirming that the additional fat loss comes from metabolic mechanisms beyond reduced eating.
Does tirzepatide increase metabolism?
Tirzepatide does not increase basal metabolic rate, but it prevents metabolic adaptation, the natural reduction in metabolic rate that normally occurs during weight loss. It also increases fat oxidation, shifting the body toward burning fat as fuel, and activates brown adipose tissue, which generates heat through calorie burning. These combined effects create a metabolic environment that favors sustained fat loss.
Can I do anything to enhance tirzepatide fat-burning effects?
Yes. Resistance training preserves lean mass and may enhance fat oxidation. Adequate protein intake (1.0-1.2g per pound of goal body weight) supports muscle preservation. Structured meals with clear fasting periods allow the GIP-mediated metabolic cycling between fat storage and fat release. Certain supplements like L-carnitine, B12, and glycine may support the fat-burning pathways tirzepatide activates.
Does the fat come back when you stop tirzepatide?
Clinical data from the SURMOUNT-4 trial shows that weight regain occurs gradually after discontinuation, as the metabolic effects of tirzepatide require ongoing treatment. However, the regain is not immediate, and lifestyle modifications established during treatment, including exercise habits and eating patterns, can help maintain results. Maintenance strategies and tapering protocols can help preserve more of the metabolic benefits.
Is tirzepatide better than exercise for fat loss?
Tirzepatide produces substantially more fat loss than exercise alone, with clinical trials showing 20%+ body weight reduction versus the 3-5% typically achievable through exercise programs. However, the ideal approach combines both. Exercise enhances the fat oxidation pathways tirzepatide activates, preserves lean mass, and provides cardiovascular and mental health benefits that medication alone cannot. The combination produces better body composition outcomes than either approach independently.
How long until tirzepatide fat-burning effects kick in?
Appetite suppression begins within the first week, but the full spectrum of metabolic fat-burning effects develops over 4-12 weeks as doses increase during titration. The GIP-mediated fat tissue effects, brown fat activation, and enhanced fat oxidation become more pronounced at higher doses, which is why the weight loss timeline typically shows acceleration after the initial titration period.
External resources
Body Composition Changes During Weight Reduction with Tirzepatide in SURMOUNT-1 - PMC
Tirzepatide Modulates Adipocyte Nutrient Metabolism Through GIP Receptor - Cell Metabolism
Tirzepatide Once Weekly for the Treatment of Obesity - New England Journal of Medicine
Tirzepatide Reduces Body Weight by Increasing Fat Utilization via CNS-Adipose Tissue Axis - PubMed
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, personalized dosing recommendations, and a community of thousands who have navigated these exact questions. Whether you are just starting your research or looking to optimize an existing protocol, the membership provides the depth of information and support that makes the difference between good results and exceptional ones.
In case I do not see you, good afternoon, good evening, and good night. May your fat cells stay responsive, your metabolism stay efficient, and your body composition stay exactly where you want it.