Mar 24, 2026

What if the mental fog creeping into your afternoons has nothing to do with tirzepatide itself? What if the real culprit is something far simpler, something fixable in a matter of days?
Thousands of people report brain fog after starting tirzepatide. They describe it as a haze that rolls in after their weekly injection, dulling concentration, slowing recall, making familiar tasks feel oddly effortful. Some say it peaks on days two and three, then lifts. Others feel it constantly, a low-grade cloudiness that makes them question whether the medication is worth the cognitive cost.
But here is the thing most discussions about tirzepatide and brain fog miss entirely. The FDA prescribing information for both Mounjaro and Zepbound does not list brain fog as a side effect. The SURPASS and SURMOUNT clinical trials, which enrolled tens of thousands of participants, did not identify cognitive impairment as an adverse event. And emerging research suggests that GLP-1 receptor agonists may actually protect the brain, with over 100 preclinical studies demonstrating neuroprotective effects and retrospective analyses showing a 40 to 70 percent reduction in Alzheimer diagnosis risk among treated populations.
So what is happening? Why do real people experience real cognitive symptoms while taking a medication that research suggests should help the brain, not hurt it? The answer lies at the intersection of nutrition, hydration, blood sugar regulation, and the dramatic appetite suppression that makes tirzepatide so effective for weight loss. This guide breaks down every mechanism, every contributing factor, and every evidence-based strategy for clearing the fog without stopping your medication. SeekPeptides members have navigated these exact challenges, and the patterns that emerge from their experiences paint a remarkably consistent picture.
What brain fog actually means and why it matters on tirzepatide
Brain fog is not a medical diagnosis. It is a colloquial term describing a cluster of cognitive symptoms that include difficulty concentrating, mental fatigue, forgetfulness, slow processing speed, and a general sense of cognitive dullness. People use the phrase because it captures the subjective experience perfectly. Your brain feels wrapped in cotton. Thoughts that normally flow freely get stuck.
On tirzepatide, brain fog reports follow specific patterns. Some users notice it within the first week of treatment, particularly during the initial dose adjustment period. Others do not experience it until they reach higher doses, when appetite suppression becomes more pronounced and caloric intake drops dramatically. A smaller group reports cognitive improvement instead, describing newfound mental clarity they attribute to better blood sugar control, reduced sleep disruption, or freedom from constant food-related thoughts.
The variability matters. If tirzepatide directly caused brain fog through a pharmacological mechanism, we would expect consistent reports across users. Instead, we see a spectrum. That spectrum points toward indirect causes rather than a direct drug effect.
The difference between pharmacological and metabolic cognitive effects
Pharmacological cognitive effects happen when a drug crosses the blood-brain barrier and directly alters neurotransmitter activity in ways that impair cognition. Benzodiazepines do this. Anticholinergic medications do this. The cognitive impairment is predictable, dose-dependent, and consistent across users.
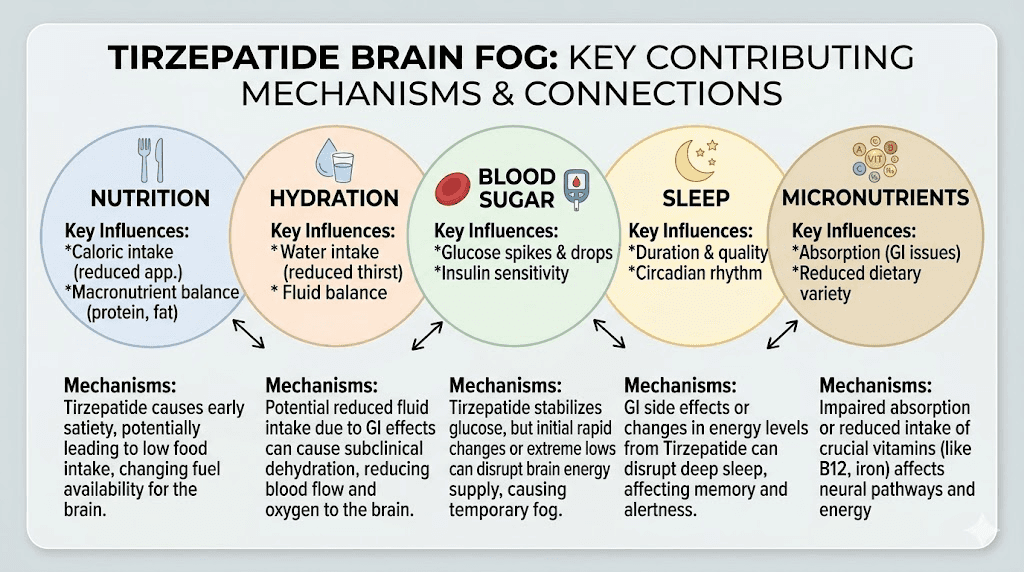
Metabolic cognitive effects happen when a drug changes the body metabolic environment in ways that secondarily affect brain function. This is what appears to happen with tirzepatide and dietary changes. The medication does not fog your brain directly. Instead, the dramatic appetite suppression it produces creates conditions, specifically nutritional deficiency, dehydration, and blood sugar fluctuation, that the brain responds to with reduced cognitive performance.
Understanding this distinction is critical because it determines your response. You do not fight a metabolic cognitive effect by reducing the dose or switching medications. You fight it by fixing the metabolic environment. And that, for most people, is entirely within their control. The same principle applies whether you are on a starting dose or have been titrating up through the full dosage chart.
The five mechanisms behind tirzepatide brain fog
Research and clinical observation point to five primary mechanisms that explain why some tirzepatide users experience cognitive symptoms. Most people dealing with brain fog have at least two of these factors operating simultaneously, which is why addressing just one often produces incomplete relief.
Mechanism 1: caloric restriction and glucose availability
Your brain consumes approximately 20 percent of your total daily energy despite representing only about 2 percent of your body weight. It runs primarily on glucose, and it is exquisitely sensitive to fuel availability. When caloric intake drops significantly, as it often does on tirzepatide, the brain may not receive adequate glucose to maintain optimal function.
Tirzepatide users frequently report eating 500 to 1,200 calories per day without realizing how dramatic the reduction is. The medication suppresses appetite so effectively that many people simply forget to eat, skip meals without hunger cues, or eat tiny portions that feel satisfying but fall far below their metabolic needs. People who track their intake on a structured GLP-1 diet plan are far less likely to experience this problem because they have caloric targets to hit regardless of appetite. Research on caloric restriction consistently shows that cognitive performance declines when intake drops below approximately 1,200 calories per day for women and 1,500 calories per day for men, with effects appearing within days rather than weeks.
The timing of brain fog often correlates with this mechanism. If your fog is worst in the late morning or mid-afternoon, when blood glucose naturally dips between meals, inadequate caloric intake is likely a primary contributor. Users who track their food intake and discover they have been eating under 1,000 calories frequently report immediate cognitive improvement when they increase intake to appropriate levels while following a structured tirzepatide eating plan.
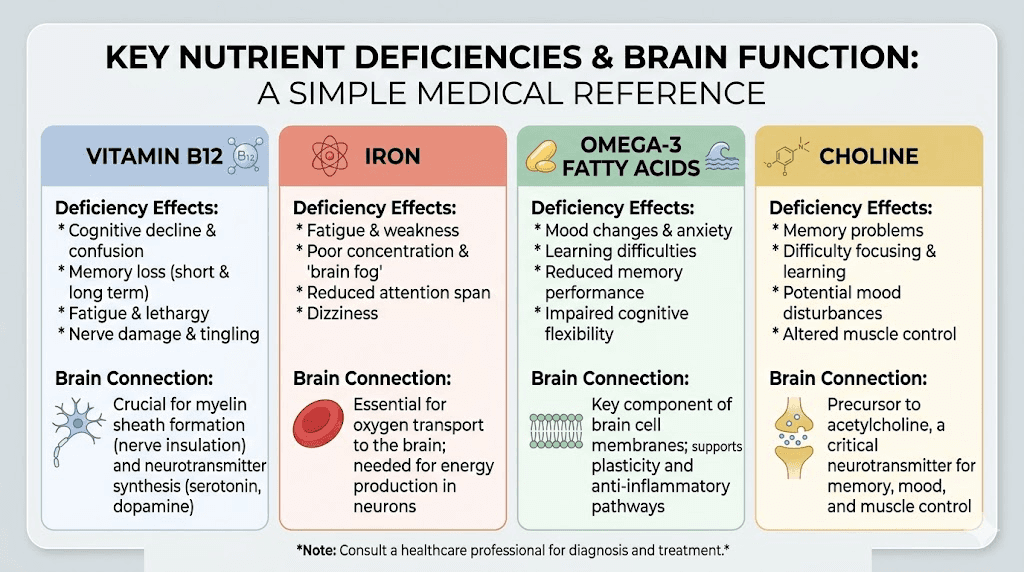
Mechanism 2: micronutrient depletion
Even when total caloric intake remains adequate, the types of foods people eat on tirzepatide often shift. Appetite suppression tends to reduce interest in nutrient-dense foods like meats, vegetables, and complex carbohydrates while sometimes preserving tolerance for simpler, easier-to-eat items. This dietary shift creates specific micronutrient deficiencies that directly affect brain function.
Vitamin B12 deficiency is the most significant concern. B12 is essential for myelin synthesis, the fatty insulation that allows nerve signals to travel quickly and accurately through the brain. Low B12 produces symptoms that look exactly like brain fog: poor concentration, memory problems, mental fatigue, and slowed thinking. Research has found that combining tirzepatide with B12 addresses this common deficiency pathway. Many compounded formulations now include methylcobalamin specifically to prevent these neurological effects.
Iron deserves attention too. Reduced meat intake while on tirzepatide can lower iron stores, and iron deficiency impairs cognitive function even before anemia develops. Iron-deficient individuals show measurable deficits in attention, memory, and processing speed that resolve with supplementation. Women are at particular risk, especially those whose menstrual patterns change on tirzepatide, since heavier or more frequent periods increase iron losses at the same time that dietary iron intake decreases.
Omega-3 fatty acids play a role as well. These essential fats make up a substantial portion of brain cell membranes and are critical for neurotransmitter function. When appetite suppression reduces fish consumption and overall fat intake, omega-3 levels may decline over weeks and months, contributing to gradually worsening cognitive symptoms. The right supplement strategy can prevent these deficiencies entirely.
Choline is another nutrient that rarely gets discussed but matters enormously for cognition. It serves as a precursor to acetylcholine, the primary neurotransmitter involved in memory and attention. Eggs, liver, and cruciferous vegetables are the primary dietary sources, and all three are foods that tirzepatide users commonly reduce or eliminate.
Mechanism 3: dehydration and electrolyte imbalance
This mechanism is the most underappreciated and often the easiest to fix. Tirzepatide causes gastrointestinal effects including nausea, reduced fluid intake from appetite suppression, and occasional vomiting or diarrhea that can all contribute to dehydration. The brain is approximately 75 percent water, and even mild dehydration, a 1 to 2 percent reduction in body water, produces measurable cognitive impairment.
Studies on dehydration and cognition show that fluid loss equivalent to just 1.5 percent of body weight impairs working memory, increases anxiety, and slows reaction time. At 2 percent dehydration, these effects become noticeable to the individual. Many tirzepatide users who report brain fog are simply not drinking enough water because their thirst cues, like their hunger cues, have been suppressed.
Electrolyte imbalance compounds the problem. Sodium, potassium, and magnesium all play essential roles in nerve signal transmission. When fluid intake drops and gastrointestinal losses increase, these electrolytes can fall below optimal levels. Low magnesium is particularly relevant because it affects over 300 enzymatic reactions in the body, many of which involve neurotransmitter synthesis and neural signaling. Common signs of magnesium depletion overlap significantly with brain fog symptoms: poor concentration, irritability, mental fatigue, and sleep disruption.
Mechanism 4: blood sugar volatility
Tirzepatide works by activating both GLP-1 and GIP receptors, which together regulate insulin secretion, glucagon suppression, and gastric emptying. For most people, this produces more stable blood sugar levels over time. But during the adjustment period, and especially at higher doses, some users experience periods of relative hypoglycemia that the brain perceives as a fuel crisis.
This is different from clinical hypoglycemia, which involves blood glucose dropping below 70 mg/dL. What tirzepatide users often experience is a rapid drop from their usual elevated baseline to a normal level. If someone has been running blood glucose at 140 to 180 mg/dL and tirzepatide brings it down to 90 mg/dL within days, the brain may interpret that 90 as "low" because it has adapted to higher levels. The result feels like hypoglycemia, with brain fog, shakiness, irritability, and difficulty concentrating, even though the absolute number is perfectly normal. This same relative blood sugar adjustment explains why feeling cold on tirzepatide often accompanies brain fog during the early weeks of treatment.
This mechanism explains why brain fog is more common in people with insulin resistance or prediabetes who start tirzepatide. Their brains have calibrated to higher glucose levels, and the medication rapid normalization feels like deprivation. The good news is that this type of brain fog is almost always temporary. The brain recalibrates to normal glucose levels within two to four weeks, and cognitive symptoms resolve on their own. Understanding the full timeline of tirzepatide effects helps set realistic expectations for when symptoms should improve. Users who wonder why their weight loss has stalled sometimes discover that blood sugar stabilization is still occurring, which temporarily obscures scale changes while the body adapts metabolically.
Mechanism 5: sleep disruption and fatigue cascading into cognition
Sleep quality affects cognitive function more powerfully than almost any other single variable. Even one night of poor sleep reduces attention, working memory, and executive function by measurable amounts. Several nights of disrupted sleep produce cumulative cognitive impairment that looks and feels exactly like brain fog.
Tirzepatide can disrupt sleep through multiple pathways. Nausea that worsens at night, acid reflux from delayed gastric emptying, and general discomfort can all fragment sleep without the person fully waking. The result is mornings that feel unrested and days that feel foggy, with the connection between poor sleep and poor cognition often going unrecognized. Users who experience tirzepatide-related fatigue are particularly vulnerable to this cascading effect.
The relationship between GLP-1 medications and fatigue is well documented. When fatigue leads to poor sleep, which leads to brain fog, which leads to reduced physical activity, which leads to deeper fatigue, the cycle becomes self-reinforcing. Breaking the cycle at any point, improving sleep hygiene, managing nausea, or addressing the nutritional factors described above, often resolves the cognitive symptoms as well.
What the research actually says about GLP-1 medications and the brain
The research on GLP-1 receptor agonists and cognitive function tells a story that might surprise people experiencing brain fog. The overwhelming direction of the evidence suggests these medications are neuroprotective, not neurotoxic.
Preclinical neuroprotection evidence
Over 100 preclinical studies have demonstrated neuroprotective effects of GLP-1 receptor agonists in animal models of neurodegeneration, stroke, and traumatic brain injury. The mechanisms include reduced neuroinflammation, decreased oxidative stress, improved mitochondrial function in neurons, and enhanced synaptic plasticity. These are not subtle effects. In many animal models, GLP-1 agonists significantly preserved cognitive function that would otherwise have declined.
Tirzepatide specifically has shown neuroprotective effects through brain glucose metabolism regulation. A study using APP/PS1 mice, an Alzheimer disease model, found that tirzepatide improved spatial learning and memory through modulation of insulin resistance and inflammation response. The dual action on both GLP-1 and GIP receptors may provide additional neuroprotective benefits, since GIP receptors are expressed throughout the brain and GIP signaling plays a role in neuronal survival and synaptic function.
However, the picture is not entirely clear. A separate study found that semaglutide and tirzepatide, while preserving metabolic benefits, did not detectably improve structural or functional parameters of neurodegeneration in two different mouse models of Alzheimer disease. This does not mean the drugs harm the brain. It means the neuroprotective effects may depend on the specific condition, dosing, and duration of treatment.
Human epidemiological data
Retrospective analyses of large patient populations have produced striking findings. One analysis found that semaglutide use was associated with a 40 to 70 percent lower risk of first-time Alzheimer diagnosis in patients with type 2 diabetes compared to other diabetes medications. A meta-analysis of 26 randomized trials involving over 160,000 patients showed that GLP-1 therapies were associated with a statistically significant reduction in dementia risk and cognitive impairment.
These are not small effects. A 40 to 70 percent risk reduction is the kind of signal that has prompted multiple clinical trials specifically investigating GLP-1 agonists as potential treatments for Alzheimer disease and other neurodegenerative conditions. If these medications were causing brain fog through direct pharmacological action, we would expect to see the opposite pattern in epidemiological data.
The distinction matters for understanding individual experiences. When someone reports brain fog on tirzepatide, the most likely explanation is not that the drug is damaging their brain. The most likely explanation is that the metabolic changes, specifically reduced caloric intake, dehydration, and micronutrient depletion, are creating conditions that impair cognition. Fix those conditions, and the fog lifts. Understanding how different GLP-1 medications compare in their cognitive effects can also help inform treatment decisions.
Timeline: when brain fog starts, peaks, and resolves
One of the most useful things about tirzepatide brain fog is that it follows a predictable timeline for most people. Knowing where you are on that timeline provides both reassurance and actionable guidance.
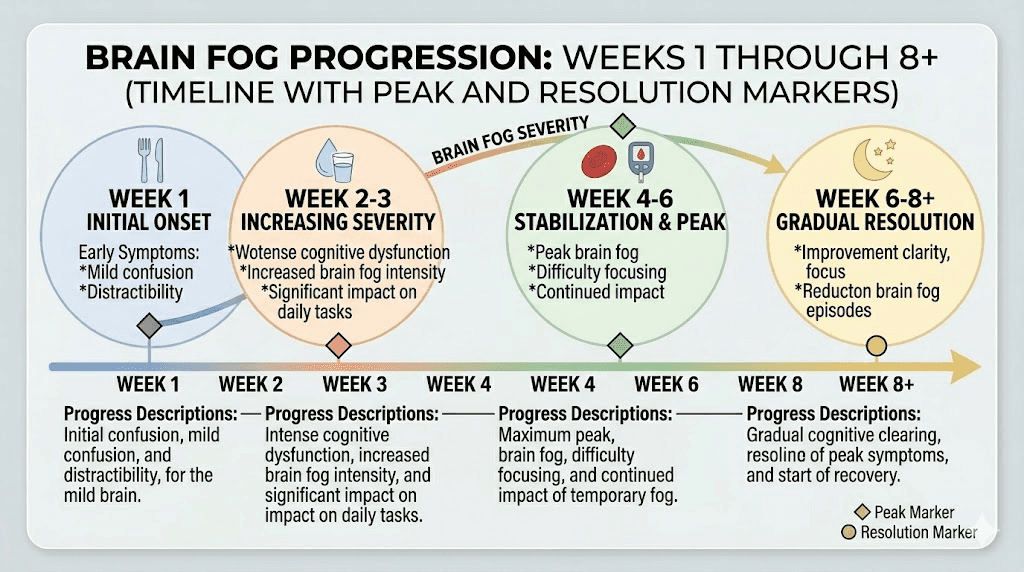
Week 1 to 2: initial adjustment
Brain fog is most commonly reported during the first two weeks of treatment or after a dose increase. During this period, the body is adapting to the medication effects on appetite, gastric emptying, and glucose regulation. Many users report that cognitive symptoms parallel their gastrointestinal adjustment, peaking on days two through four after injection and gradually improving as the week progresses.
This pattern aligns with the pharmacokinetics of tirzepatide, which reaches peak blood levels approximately 24 to 72 hours after subcutaneous injection. If brain fog consistently peaks in this window, the mechanism is likely related to peak medication effects on appetite suppression and glucose regulation rather than a persistent drug effect.
Week 3 to 6: the critical window
Most tirzepatide-related brain fog resolves within four to eight weeks as the body adapts to the new metabolic baseline. During this period, several things happen simultaneously. The brain recalibrates to lower glucose levels. Appetite suppression becomes more predictable, allowing better meal planning. Users develop strategies for maintaining adequate hydration and nutrition despite reduced hunger cues.
If brain fog has not improved by week six, it almost certainly indicates one of the ongoing metabolic factors, nutritional deficiency, chronic dehydration, or persistent caloric restriction, rather than medication adaptation. This is the point where active intervention becomes essential rather than optional.
Beyond week 8: persistent brain fog signals
Brain fog that persists beyond eight weeks on a stable dose warrants investigation. Blood work checking B12 levels, iron studies, thyroid function, fasting glucose, and a comprehensive metabolic panel can identify the specific deficiency or imbalance driving symptoms. Many people are surprised to discover that their B12 has dropped substantially, or their ferritin has fallen below optimal ranges, since these changes happen gradually and the symptoms overlap with what they attributed to the medication itself.
Persistent brain fog is not a reason to stop tirzepatide. It is a reason to identify and correct the specific metabolic factor causing it. The distinction between correlation and causation matters here: the fog started when tirzepatide started, but the fog is almost always caused by the behavioral changes the medication produces, not by the medication itself.
Who is most at risk for tirzepatide brain fog
Not everyone on tirzepatide experiences cognitive symptoms. Understanding the risk factors helps identify who should proactively implement prevention strategies rather than waiting for symptoms to appear.
People with insulin resistance or prediabetes
As discussed in the blood sugar volatility section, individuals whose brains have adapted to elevated glucose levels are more likely to experience cognitive symptoms when tirzepatide normalizes those levels. This group often reports the most dramatic initial brain fog, but also the most complete resolution once the brain recalibrates, typically within two to four weeks.
Aggressive caloric restrictors
Some people view tirzepatide appetite suppression as an opportunity to dramatically cut calories, eating 600 to 800 calories per day in pursuit of faster weight loss. This approach almost guarantees brain fog and potentially creates more serious nutritional deficiencies over time. Understanding appropriate caloric targets on GLP-1 medications is essential for maintaining cognitive function while still losing weight effectively.
Rapid dose escalators
People who increase their dose quickly, moving through the titration schedule faster than recommended or jumping straight to higher doses, give their body less time to adapt to each level of appetite suppression. Each dose increase restarts the adaptation clock, and stacking multiple adjustments can produce persistent cognitive symptoms that would not occur with a more gradual approach. Microdosing protocols have gained popularity partly because they allow gentler adaptation with fewer cognitive side effects.
People with pre-existing nutritional deficiencies
If you started tirzepatide with borderline B12, low iron stores, or suboptimal vitamin D levels, the medication-induced reduction in food intake can push you from "borderline" to "deficient" quickly. Women, vegetarians, people over 50, and those with digestive conditions are at highest risk for pre-existing deficiencies that tirzepatide exacerbates.
Those not drinking enough water
This sounds too simple to matter, but dehydration is one of the most common and most easily correctable causes of brain fog on any medication. People who relied on hunger-driven eating patterns to consume a significant portion of their daily fluids, through soups, fruits, and beverages consumed with meals, may inadvertently reduce fluid intake by 30 to 50 percent when appetite suppression reduces meal frequency and portion size.
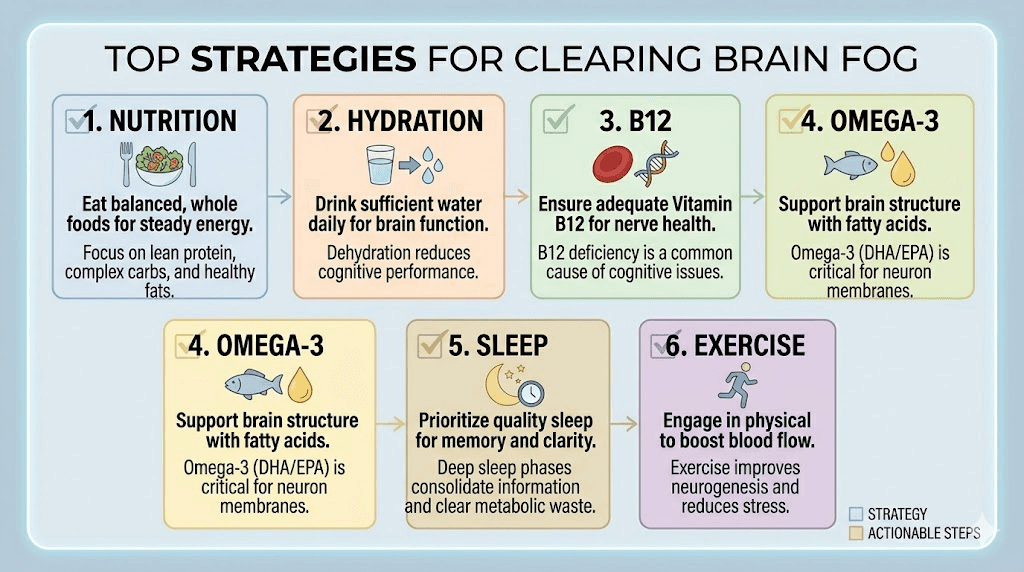
Twelve evidence-based strategies to clear tirzepatide brain fog
These strategies are ordered from most impactful to least impactful based on clinical evidence and user reports. Most people find that implementing the first three or four strategies produces significant improvement within days.
Strategy 1: eat enough, even when you are not hungry
This is the single most important intervention. Your brain needs fuel regardless of whether your appetite says so. Set a minimum caloric floor, typically 1,200 calories for women and 1,500 calories for men, and hit it every day regardless of hunger levels.
Structure three meals and one to two snacks at consistent times. Do not wait for hunger to eat because tirzepatide may have eliminated that cue entirely. Focus on nutrient-dense options from a comprehensive GLP-1 friendly food list that prioritizes protein, healthy fats, and complex carbohydrates. Each meal should include at least 20 to 30 grams of protein, which supports both muscle preservation and neurotransmitter production.
If solid food feels overwhelming due to nausea or early satiety, liquid nutrition counts. Smoothies made with protein powder, frozen fruit, spinach, and nut butter can deliver 400 to 600 calories in a form that most people tolerate well even when appetite is minimal. Protein shakes designed for GLP-1 users can bridge the gap between what your appetite allows and what your brain needs. See our GLP-1 recipe collection for more meal ideas that pack maximum nutrition into smaller portions.
Strategy 2: hydrate aggressively and consistently
Aim for a minimum of 64 ounces of water daily, with many tirzepatide users needing closer to 80 to 100 ounces, especially during the first few months. Set hourly reminders if necessary. Do not rely on thirst to guide intake because tirzepatide can blunt thirst perception just as it blunts appetite.
Add electrolytes to at least two glasses of water per day. A simple electrolyte mix containing sodium, potassium, and magnesium supports neural signaling and prevents the specific mineral depletions that contribute to brain fog. Avoid electrolyte products with large amounts of added sugar, which can cause blood sugar spikes and crashes that worsen cognitive symptoms.
Strategy 3: supplement B12 immediately
Begin taking methylcobalamin (the active form of B12) at 1,000 to 2,000 mcg daily as soon as you start tirzepatide. Do not wait for symptoms or blood work to confirm deficiency because B12 depletion happens gradually, and neurological symptoms appear before blood levels reach clinically "deficient" ranges. Many compounded tirzepatide formulations include B12 specifically to address this concern, and the tirzepatide-B12 combination has become one of the most popular compounding approaches.
Sublingual B12 tablets dissolve under the tongue and bypass the digestive system, making them effective even when gastrointestinal symptoms reduce oral supplement absorption. This matters because the same GI effects that cause nausea and bloating on tirzepatide can also reduce absorption of oral supplements taken in standard pill form.
Strategy 4: prioritize omega-3 fatty acids
Take a high-quality fish oil supplement providing at least 1,000 mg of combined EPA and DHA daily. These omega-3 fatty acids are structural components of brain cell membranes and play direct roles in neurotransmitter function, inflammation regulation, and synaptic plasticity. Low omega-3 status has been consistently linked to cognitive impairment in research.
If fish oil causes GI distress, which can happen when combined with tirzepatide gastrointestinal effects, try algae-based omega-3 supplements or take fish oil with your largest meal of the day when absorption is typically better and GI tolerance is higher.
Strategy 5: time your meals around cognitive demands
If your brain fog follows a predictable daily pattern, strategic meal timing can provide fuel when your brain needs it most. Eating a protein-rich breakfast within an hour of waking prevents the late-morning fog that comes from overnight fasting extended by appetite suppression. A mid-afternoon snack containing protein and complex carbohydrates addresses the 2 to 4 PM cognitive dip that many users report.
The timing of your injection also matters. If brain fog peaks two to three days after injection, schedule your dose so that peak effects occur during lower-demand days when cognitive performance is less critical. Some users find that injecting on Thursday evening allows the adjustment period to fall over the weekend, with cognitive function returning to baseline by Monday.
Strategy 6: add magnesium glycinate
Magnesium glycinate at 200 to 400 mg daily before bed serves double duty. It supports the 300+ enzymatic reactions involved in neurotransmitter synthesis and energy production, and the glycinate form has calming properties that may improve sleep quality. Better sleep means better cognitive function the next day, addressing two brain fog mechanisms simultaneously.
Magnesium oxide, the most common form in supplements, has poor absorption and can worsen the GI symptoms that tirzepatide already causes. Glycinate, citrate, or threonate forms are better absorbed and better tolerated. Magnesium L-threonate specifically crosses the blood-brain barrier and has shown cognitive benefits in clinical research, making it the theoretically optimal form for brain fog specifically.
Strategy 7: maintain consistent sleep habits
Set a fixed wake time and stick to it every day, including weekends. Keep the bedroom cool, dark, and quiet. Avoid screens for 30 minutes before sleep. These basic sleep hygiene practices become especially important on tirzepatide because the medication can disrupt sleep through gastrointestinal discomfort, nausea, or acid reflux.
If nausea disrupts sleep, try elevating the head of your bed by 4 to 6 inches and avoiding eating within 3 hours of bedtime. If headaches or anxiety are affecting sleep quality, address those symptoms directly rather than assuming they will resolve on their own. Users who experience dry mouth on tirzepatide often find that nighttime dehydration contributes to both poor sleep and morning brain fog.
Strategy 8: move your body daily, especially in the morning
Exercise increases cerebral blood flow, stimulates neurotrophin release, and improves glucose utilization by the brain. Even a 20-minute morning walk can noticeably improve cognitive function for the rest of the day. The effect is acute, meaning you do not need weeks of consistent exercise to see cognitive benefits. A single session helps.
Resistance training is particularly valuable because it improves insulin sensitivity, which stabilizes blood sugar and reduces the glucose volatility that contributes to brain fog. It also preserves muscle mass during weight loss, which is important for long-term metabolic health and consistent energy levels. The cognitive benefits of exercise add to the non-weight-loss benefits of tirzepatide that many users come to value.
Strategy 9: consider the B12-glycine-niacinamide compound
Compounded tirzepatide formulations that include B12, glycine, and niacinamide address multiple brain fog mechanisms simultaneously. The tirzepatide glycine B12 compound provides direct nutritional support, while niacinamide (vitamin B3) supports NAD+ production, which is essential for cellular energy metabolism throughout the brain.
Glycine itself functions as an inhibitory neurotransmitter that promotes relaxation and sleep quality, and research suggests it may improve cognitive function by enhancing sleep architecture. The combination of glycine with tirzepatide has become increasingly popular precisely because users report fewer cognitive and energy-related side effects compared to plain tirzepatide formulations.
Strategy 10: track your symptoms and identify patterns
Keep a simple daily log noting three things: what you ate, how much water you drank, and your brain fog severity on a scale of 1 to 10. After two weeks, patterns almost always emerge. You might discover that fog is worse on days when you skip breakfast, or that it correlates with days when water intake drops below a certain threshold, or that it consistently peaks two days after injection.
These patterns are your personal diagnostic data. They tell you which mechanisms are driving your symptoms and which interventions to prioritize. Without tracking, you are guessing. With tracking, you are problem-solving. SeekPeptides members use structured tracking tools that make pattern identification faster and more reliable than manual journaling alone.
Strategy 11: optimize injection timing relative to your schedule
If brain fog follows a predictable post-injection pattern, adjust when you take your dose. People who inject on Monday morning and experience fog on Tuesday and Wednesday might benefit from switching to a Friday evening injection, moving the fog window to the weekend. Injection timing is one of the most underutilized tools for managing side effects, and it requires no additional supplements or dietary changes.
Strategy 12: talk to your provider about dose adjustment
If brain fog persists despite implementing all the strategies above, a temporary dose reduction may be warranted. Stepping back one dose level for four to six weeks while optimizing nutrition and hydration gives the body more time to adapt before progressing again. This is not failure. It is strategic pacing. The microdosing approach to tirzepatide has gained traction partly because it reduces the incidence and severity of cognitive side effects while still delivering meaningful weight loss results.
The nutrition blueprint for cognitive clarity on tirzepatide
What you eat matters as much as how much you eat when it comes to preventing brain fog. This section provides specific food recommendations organized by the brain-supporting nutrient they provide.
Protein-first eating for neurotransmitter production
Neurotransmitters are made from amino acids. Dopamine comes from tyrosine. Serotonin comes from tryptophan. Acetylcholine comes from choline. Without adequate protein intake providing these amino acid precursors, neurotransmitter production slows and cognitive function suffers.
Aim for 1.2 to 1.6 grams of protein per kilogram of body weight daily, distributed across meals. For a 175-pound person, that means approximately 95 to 127 grams per day. Good sources include eggs (which also provide choline), Greek yogurt, chicken, fish (which also provides omega-3s), and legumes. If whole food protein intake feels impossible given appetite suppression, supplement with a high-quality whey or plant protein powder.
The tirzepatide diet plan should prioritize protein at every meal, but this becomes even more important when brain fog is present. Protein also slows gastric emptying and stabilizes blood sugar, reducing the glucose volatility that contributes to cognitive symptoms. Our GLP-1 dinner ideas and foods to avoid guides provide practical meal frameworks that keep protein high and brain-supporting nutrients flowing.
Brain-boosting fats
Your brain is approximately 60 percent fat by dry weight. It needs dietary fat to maintain cell membrane integrity, produce myelin, and support fat-soluble vitamin absorption. The specific fats that matter most for cognitive function include:
DHA (docosahexaenoic acid): Found in fatty fish like salmon, sardines, and mackerel. DHA makes up 25 percent of total brain fat and 90 percent of the omega-3 fat in the brain. Two to three servings of fatty fish per week provides adequate DHA for most people.
Monounsaturated fats: Found in olive oil, avocados, and nuts. These fats improve blood flow to the brain and reduce inflammation. Adding avocado to smoothies is an effective way to increase brain-healthy fat intake when appetite is low.
Medium-chain triglycerides (MCTs): Found in coconut oil and MCT oil supplements. MCTs are converted to ketones by the liver, providing an alternative fuel source for the brain that does not depend on glucose. Some users report that adding 1 to 2 tablespoons of MCT oil to their morning coffee or smoothie produces noticeable cognitive improvement, particularly on days when food intake is lower.
Complex carbohydrates for steady glucose delivery
The brain runs on glucose, and complex carbohydrates provide that glucose in a steady, sustained manner that avoids the spikes and crashes of simple sugars. Oatmeal, sweet potatoes, quinoa, brown rice, and whole grain bread are all excellent choices. Pair complex carbohydrates with protein and fat at every meal to further slow glucose absorption and maintain stable blood sugar levels.
Many tirzepatide users make the mistake of eliminating carbohydrates entirely, combining the medication with a ketogenic or very low-carb diet. While this combination can produce rapid weight loss, it also removes the brain primary fuel source during a period when the body is already adapting to reduced caloric intake. Unless you are already well-adapted to ketosis, adding moderate complex carbohydrates to your GLP-1 diet may be the fastest way to resolve brain fog.
Hydrating foods that count toward fluid intake
Cucumbers, watermelon, celery, strawberries, and broth-based soups all contribute to hydration while providing nutrients. On days when drinking large amounts of plain water feels difficult, incorporating these high-water-content foods into meals and snacks can meaningfully boost hydration status without requiring additional fluid consumption.
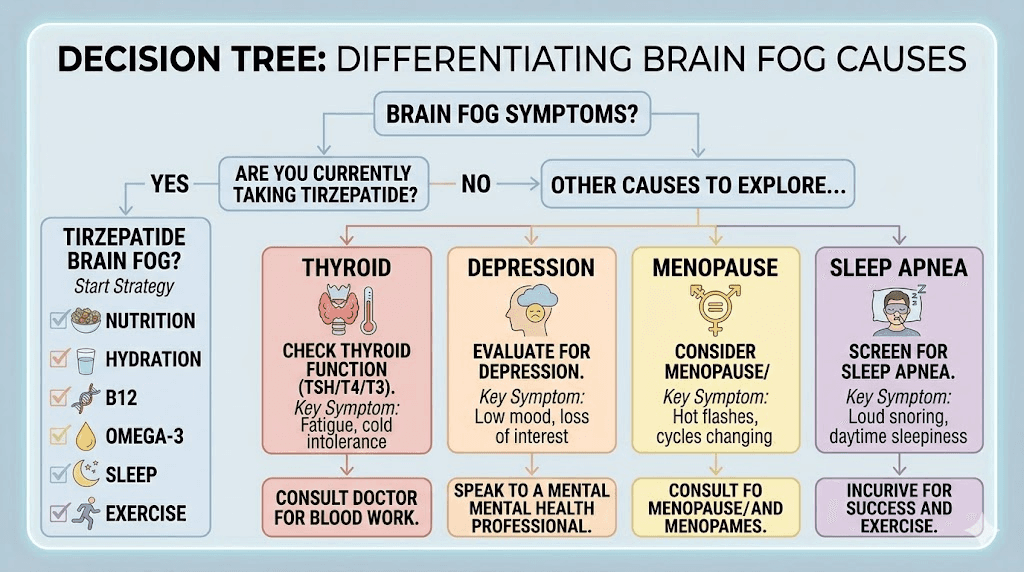
Tirzepatide brain fog versus other cognitive conditions
Not all brain fog while taking tirzepatide comes from tirzepatide-related mechanisms. It is important to recognize when cognitive symptoms might indicate a separate condition that requires its own evaluation and treatment.
Thyroid dysfunction
Hypothyroidism produces brain fog, fatigue, and cognitive slowing that can look identical to tirzepatide-related cognitive symptoms. Because weight changes and metabolic shifts can affect thyroid function, checking thyroid levels (TSH, free T3, and free T4) is reasonable for anyone experiencing persistent brain fog on tirzepatide. Users taking tirzepatide with levothyroxine should be aware that the medication may affect thyroid hormone absorption and should work with their provider to monitor levels.
Depression and anxiety
Cognitive symptoms of depression, including difficulty concentrating, mental fatigue, and reduced processing speed, overlap substantially with brain fog. The weight loss journey itself can trigger or worsen mood disorders in some people, and anxiety on tirzepatide has been reported by some users. If brain fog accompanies persistent low mood, loss of interest in activities, or feelings of hopelessness, a mental health evaluation is warranted.
Perimenopause and menopause
Women in their 40s and 50s who start tirzepatide may attribute brain fog to the medication when hormonal changes are the primary driver. Estrogen plays a significant role in cognitive function, and the cognitive effects of perimenopause can emerge or intensify during periods of significant weight change. Understanding how hormone therapy interacts with GLP-1 medications can help distinguish between medication-related and hormone-related cognitive symptoms. Tirzepatide effects on menstrual cycles further complicate this picture for premenopausal women.
Sleep apnea
Untreated or undertreated sleep apnea causes brain fog through chronic sleep disruption and intermittent hypoxia. Interestingly, weight loss from tirzepatide often improves sleep apnea significantly, so brain fog may actually get better over time for this group. However, during the weight loss phase, fluctuations in airway anatomy can temporarily change CPAP requirements or unmask previously subclinical sleep-disordered breathing.
Autoimmune conditions
For people managing autoimmune conditions while taking tirzepatide, brain fog may relate to underlying inflammation rather than medication effects. Conditions like lupus, Hashimoto thyroiditis, and rheumatoid arthritis all include cognitive dysfunction as a potential symptom, and distinguishing medication-related from disease-related brain fog requires careful clinical evaluation.
The paradox: how tirzepatide might actually improve long-term brain health
While short-term brain fog gets all the attention, the long-term cognitive story of tirzepatide may be overwhelmingly positive. Understanding this paradox helps put temporary cognitive symptoms in perspective.
Weight loss reduces chronic neuroinflammation
Excess body fat, particularly visceral fat, produces inflammatory cytokines that cross the blood-brain barrier and contribute to chronic neuroinflammation. This inflammation impairs synaptic function, reduces neuroplasticity, and accelerates cognitive decline over years and decades. By producing significant, sustained weight loss, tirzepatide may reduce this inflammatory burden and protect long-term cognitive function.
The irony is that the short-term brain fog some people experience while losing weight gives way to improved cognitive function once weight stabilizes at a lower level. Multiple studies have shown that sustained weight loss of 10 percent or more improves cognitive performance on standardized tests, with benefits in attention, memory, and executive function. Users who document their progress through before-and-after tracking frequently report cognitive improvements alongside physical changes once the initial adjustment period resolves.
Improved insulin sensitivity protects neurons
Insulin resistance does not just affect blood sugar regulation. The brain itself becomes insulin resistant, which impairs glucose utilization by neurons, reduces insulin-mediated neurotransmitter activity, and contributes to the accumulation of beta-amyloid plaques associated with Alzheimer disease. By improving insulin sensitivity throughout the body and brain, tirzepatide may provide neuroprotection that extends far beyond weight management.
GLP-1 receptor activation in the brain
GLP-1 receptors are expressed in brain regions critical for learning and memory, including the hippocampus and cortex. When tirzepatide activates these receptors, it stimulates pathways involved in neuronal survival, synaptic plasticity, and neurogenesis. The dual GIP/GLP-1 receptor activation that makes tirzepatide unique for weight loss may also make it uniquely beneficial for brain health, since GIP receptors in the brain are involved in neuroprotective signaling distinct from GLP-1 pathways.
This does not mean brain fog on tirzepatide is imaginary or unimportant. It means that while you address the short-term metabolic factors causing cognitive symptoms, the medication may simultaneously be providing long-term neuroprotective benefits that you will never subjectively notice but that could meaningfully reduce your risk of cognitive decline later in life.
Specific supplements and doses for brain fog prevention
Beyond the core strategies outlined above, specific supplement protocols have emerged from clinical practice and user experience that target cognitive function during GLP-1 therapy. SeekPeptides members consistently report that a targeted approach to supplementation outperforms generic multivitamins for preventing and resolving brain fog.
The cognitive support stack
This protocol addresses every major nutritional pathway involved in brain fog on tirzepatide. Start all supplements within the first week of treatment rather than waiting for symptoms to appear.
Morning (with breakfast):
Methylcobalamin (B12): 1,000 to 2,000 mcg sublingual
Fish oil: 1,000 to 2,000 mg combined EPA/DHA
Vitamin D3: 2,000 to 5,000 IU (with fat for absorption)
Iron bisglycinate: 18 to 27 mg (menstruating women only, with vitamin C for absorption)
Afternoon (with lunch or snack):
B-complex vitamin: covers B6, folate, and additional B12
Alpha-GPC or CDP-choline: 300 to 600 mg (direct acetylcholine precursor for memory and focus)
Evening (before bed):
Magnesium glycinate or L-threonate: 200 to 400 mg
Zinc: 15 to 30 mg (supports neurotransmitter metabolism)
This stack addresses the specific deficiencies that drive brain fog while supporting sleep quality and neurotransmitter production. Users taking semaglutide with B12 or semaglutide with methylcobalamin have reported similar cognitive benefits, confirming that B12 supplementation is one of the most impactful single interventions regardless of which GLP-1 medication you use.
The role of creatine in GLP-1 brain fog
Creatine is not just for muscles. The brain uses creatine to regenerate ATP, the cellular energy currency, and research shows that creatine supplementation improves cognitive performance under conditions of sleep deprivation, stress, and caloric restriction, all of which are relevant to tirzepatide users. A dose of 3 to 5 grams daily of creatine monohydrate provides cognitive support while also helping preserve lean mass during weight loss. The relationship between creatine and tirzepatide extends beyond muscle preservation into genuine cognitive territory, and users who add creatine frequently report noticeable improvements in mental energy and clarity.
L-carnitine for brain energy metabolism
Acetyl-L-carnitine (ALCAR) crosses the blood-brain barrier and supports mitochondrial energy production in neurons. At doses of 500 to 1,500 mg daily, ALCAR has shown cognitive benefits in clinical trials, particularly for fatigue-related cognitive impairment. L-carnitine combined with GLP-1 medications has gained attention for both metabolic and cognitive support, and the tirzepatide levocarnitine blend addresses this through a compounded formulation.
Exercise protocols that specifically target brain fog
Not all exercise is equal when it comes to cognitive benefits. Specific protocols have stronger evidence for improving brain function during periods of caloric restriction and metabolic change.
Zone 2 cardio for cerebral blood flow
Low-intensity aerobic exercise at 60 to 70 percent of maximum heart rate (Zone 2) maximizes cerebral blood flow without the fatigue that higher intensities can produce. Thirty to forty minutes of walking, cycling, or swimming at this intensity, performed most days of the week, provides a sustained cognitive boost that lasts for hours after the session. This is the single most effective exercise modality for brain fog during weight loss on any medication.
Resistance training for insulin sensitivity
Two to three resistance training sessions per week improve insulin sensitivity, stabilize blood glucose, and preserve the lean mass that contributes to metabolic rate. All of these effects reduce brain fog risk. Compound movements like squats, deadlifts, rows, and presses provide the greatest metabolic impact per minute of exercise. If you are new to resistance training, starting with bodyweight exercises or machines is perfectly effective. The exercise debate for GLP-1 users misses a crucial point: exercise on these medications is not just about weight loss, but about maintaining cognitive function and preserving the metabolic health that makes weight loss sustainable.
Morning sunlight exposure
This is technically not exercise, but 10 to 15 minutes of morning sunlight within the first hour of waking sets circadian rhythms, supports vitamin D synthesis, and improves sleep quality that night. All three effects reduce brain fog. Combining morning sunlight with a walk creates a powerful cognitive reset that costs nothing and requires no equipment.
Comparing cognitive effects: tirzepatide versus semaglutide versus retatrutide
People considering or switching between GLP-1 medications often want to know whether cognitive side effects differ across options. The side effect profiles of semaglutide and tirzepatide overlap considerably, but some differences are worth noting.
Semaglutide (Ozempic/Wegovy)
Brain fog reports on semaglutide follow similar patterns to tirzepatide, with the same underlying mechanisms of caloric restriction, dehydration, and nutritional depletion driving most cases. Semaglutide fatigue and cognitive symptoms have been reported at comparable rates to tirzepatide. Users who have experienced semaglutide dizziness alongside brain fog often find that both symptoms share the same root cause of dehydration and inadequate nutrition. Because semaglutide typically produces slightly less appetite suppression and less total weight loss than tirzepatide, the severity of nutrition-related brain fog may be somewhat lower, though this varies considerably between individuals. The overall experience on semaglutide includes similar cognitive fluctuations during the adjustment period.
Tirzepatide (Mounjaro/Zepbound)
The dual GIP/GLP-1 mechanism of tirzepatide produces more potent appetite suppression than semaglutide alone, which translates to greater potential for caloric restriction-related brain fog. However, the GIP receptor activation may provide additional neuroprotective effects not available with GLP-1 agonists alone. Understanding how quickly tirzepatide takes effect helps predict when cognitive adjustment symptoms are most likely. The net cognitive impact appears roughly neutral compared to semaglutide when nutritional factors are properly managed. Users tracking week-by-week results on either medication frequently note that cognitive symptoms improve in parallel with gastrointestinal adaptation.
Retatrutide
As a triple agonist targeting GLP-1, GIP, and glucagon receptors, retatrutide produces the most dramatic appetite suppression and weight loss of the three. This means the potential for nutrition-related brain fog is highest with retatrutide, and proactive nutritional strategies become even more important. Users considering switching from tirzepatide to retatrutide should be aware of this and have a solid nutritional plan in place before transitioning.
Factor | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
Appetite suppression intensity | Moderate | Strong | Very strong |
Brain fog risk (nutrition-related) | Moderate | Moderate-high | High |
Neuroprotective potential | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + glucagon |
B12 compound available | Yes | Yes | Limited |
Typical fog resolution | 3 to 6 weeks | 4 to 8 weeks | 4 to 8 weeks |
When brain fog is actually a sign of something more serious
While most tirzepatide brain fog is benign and correctable, certain patterns warrant immediate medical attention. Recognizing these red flags prevents serious conditions from being dismissed as routine side effects. SeekPeptides always recommends working with a healthcare provider when cognitive symptoms are severe or progressive.
Sudden onset of severe confusion: Brain fog is gradual and mild to moderate. If you experience sudden, severe disorientation, difficulty speaking, or inability to perform familiar tasks, seek immediate medical attention. This could indicate hypoglycemia below 50 mg/dL, stroke, or other acute conditions unrelated to tirzepatide.
Progressive worsening despite interventions: If brain fog steadily worsens over weeks despite adequate nutrition, hydration, and supplementation, this suggests a cause beyond metabolic factors. Thyroid dysfunction, B12 deficiency severe enough to cause neurological damage, or other conditions may need investigation.
Accompanying neurological symptoms: Numbness or tingling in extremities alongside brain fog could indicate B12 deficiency severe enough to cause peripheral neuropathy. Vision changes, weakness on one side, or difficulty with balance require immediate evaluation.
Brain fog with significant mood changes: If cognitive symptoms accompany new or worsening depression, anxiety, or personality changes, a comprehensive psychiatric and neurological evaluation is appropriate. While tirzepatide itself has not been conclusively linked to mood disorders, the metabolic and life changes associated with significant weight loss can unmask or exacerbate underlying conditions. People who notice changes in libido or sexual function alongside cognitive symptoms should mention both to their provider, as hormonal shifts from weight loss can affect multiple systems simultaneously.
The gut-brain connection on tirzepatide
Emerging research reveals a fascinating connection between tirzepatide effects on gut bacteria and cognitive function that adds nuance to the brain fog conversation.
Tirzepatide alters the gut microbiome through changes in gastric emptying, bile acid metabolism, and the dietary shifts it produces. Recent animal research found that tirzepatide treatment significantly reduced Lachnospiraceae, a family of bacteria that produces short-chain fatty acids essential for gut barrier integrity and neuroactive metabolite production. These bacteria produce butyrate, which directly modulates brain inflammation and supports the integrity of the blood-brain barrier.
The gut-brain axis, the bidirectional communication pathway between intestinal bacteria and the central nervous system, means that changes in gut bacteria composition can directly affect mood, cognition, and mental energy. If tirzepatide reduces populations of bacteria that produce brain-supporting metabolites, this could represent an additional mechanism for cognitive symptoms beyond the nutritional factors discussed above.
Practical implications include considering probiotic supplementation, particularly strains from the Lachnospiraceae family, and consuming prebiotic fiber from foods like onions, garlic, leeks, and asparagus that support beneficial bacterial populations. The gut response on GLP-1 medications is an area of active research, and supporting gut health may prove to be an important strategy for preventing brain fog that extends beyond simple nutrition. Users dealing with semaglutide bloating or sulfur burps on GLP-1 medications may find that gut microbiome support addresses both digestive and cognitive symptoms simultaneously, since the same bacterial disruption can drive both.
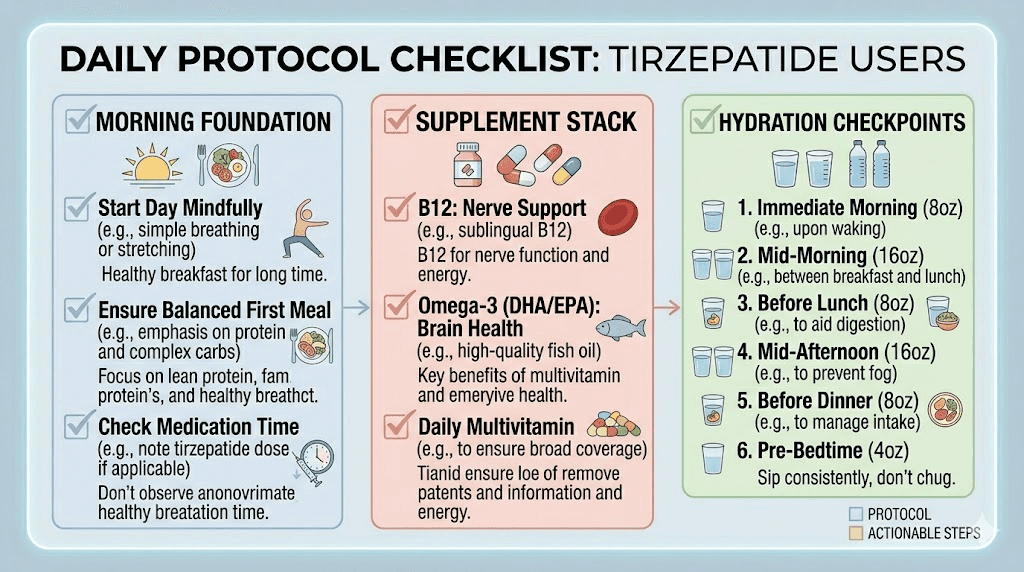
Real-world protocols that work: what experienced users do differently
Experienced tirzepatide users who have successfully managed brain fog consistently report three non-negotiable daily habits. These are not complicated protocols. They are simple, sustainable practices that address the most common causes.
The morning foundation protocol
Within 30 minutes of waking: 16 ounces of water with electrolytes, followed by a breakfast containing at least 30 grams of protein and 15 grams of healthy fat. Common choices include three eggs with avocado, a protein smoothie with almond butter and spinach, or Greek yogurt with nuts and berries. This single habit addresses caloric restriction, dehydration, and blood sugar volatility before they have a chance to create cognitive symptoms.
The supplement stack
With breakfast: methylcobalamin (1,000 to 2,000 mcg), fish oil (1,000 to 2,000 mg EPA/DHA), and a multivitamin with iron (for menstruating women). Before bed: magnesium glycinate (200 to 400 mg). This stack covers the most common micronutrient deficiencies that contribute to brain fog and costs less than a dollar per day. The full GLP-1 supplement guide provides additional options for specific symptoms, but this core stack addresses the cognitive essentials.
The hydration checkpoint
Four ounces of fluid every waking hour, tracked with a simple tally or phone reminder. By noon, you should have consumed at least 32 ounces. By dinner, 56 ounces. By bedtime, 72 to 80 ounces minimum. This consistent approach prevents the dehydration that builds gradually and produces afternoon brain fog that people attribute to the medication rather than to fluid deficit.
Blood work to request if brain fog persists
If brain fog continues beyond six to eight weeks despite implementing the strategies above, request these specific lab tests from your provider. They identify the most common correctable causes of persistent cognitive symptoms on tirzepatide.
Complete blood count (CBC): Checks for anemia, which reduces oxygen delivery to the brain and impairs cognition. Pay particular attention to hemoglobin, hematocrit, and MCV (mean corpuscular volume), which can indicate iron or B12 deficiency even before frank anemia develops.
Vitamin B12 and methylmalonic acid: Serum B12 alone can miss functional deficiency. Methylmalonic acid is a more sensitive marker that rises before B12 levels drop below the standard reference range. Request both.
Ferritin: Iron stores below 30 ng/mL can impair cognition even when hemoglobin is normal. Many providers consider ferritin "normal" at 12 ng/mL or above, but cognitive symptoms from iron depletion can appear at levels well above the lab low range. Optimal ferritin for cognitive function is typically above 50 ng/mL.
Thyroid panel (TSH, free T3, free T4): Thyroid dysfunction causes brain fog that closely mimics medication-related symptoms. Hashimoto disease in particular can cause cognitive symptoms that worsen during periods of metabolic change.
Comprehensive metabolic panel: Checks sodium, potassium, calcium, and magnesium levels. Electrolyte imbalances from reduced food and fluid intake are common on tirzepatide and directly affect neural function. Understanding how tirzepatide affects blood work helps interpret results accurately.
Fasting glucose and HbA1c: Establishes whether blood sugar regulation has changed significantly since starting tirzepatide, which could explain glucose-related cognitive symptoms.
Vitamin D: Low vitamin D levels are associated with cognitive impairment and are common in overweight and obese populations. Levels below 30 ng/mL warrant supplementation.
Frequently asked questions
Does tirzepatide directly cause brain fog?
No. Brain fog is not listed as a side effect in the FDA prescribing information for Mounjaro or Zepbound, and clinical trials did not identify cognitive impairment as an adverse event. Brain fog on tirzepatide is almost always caused by the metabolic changes the medication produces, specifically reduced caloric intake, dehydration, electrolyte depletion, and micronutrient deficiencies, rather than a direct pharmacological effect on the brain.
How long does tirzepatide brain fog last?
For most people, brain fog related to blood sugar recalibration resolves within two to four weeks. Brain fog related to nutritional and hydration factors resolves within days to two weeks once those factors are corrected. Without intervention, persistent brain fog from ongoing nutrient depletion can continue indefinitely, which is why proactive supplementation and adequate eating are essential from day one.
Can I take anything to help with brain fog on tirzepatide?
Yes. Methylcobalamin (B12) at 1,000 to 2,000 mcg daily, fish oil providing 1,000+ mg EPA/DHA, magnesium glycinate at 200 to 400 mg before bed, and consistent electrolyte supplementation address the most common causes. Many users also benefit from compounded tirzepatide with B12, which provides nutritional support built directly into the medication.
Is brain fog worse on higher doses of tirzepatide?
It can be, primarily because higher doses produce more appetite suppression, which leads to greater caloric restriction and more pronounced nutritional gaps. If brain fog worsens with each dose increase, it almost always indicates that your nutrition is not keeping pace with your appetite reduction. Review your dosing schedule and ensure caloric intake stays above minimum thresholds.
Should I stop tirzepatide if I get brain fog?
No. Stopping the medication is rarely necessary for brain fog. The cognitive symptoms are nearly always correctable through nutritional optimization, hydration, and supplementation. Stopping tirzepatide addresses the symptom by removing the underlying appetite suppression but does not address the actual nutritional and metabolic factors causing the fog. Instead, maintain the medication and fix the fuel supply. If brain fog is truly severe and unresponsive to all interventions, discuss dose adjustment with your provider before considering discontinuation.
Does brain fog mean tirzepatide is not working for me?
Not at all. Brain fog and effective weight loss can coexist. In fact, the same appetite suppression that causes weight loss is typically what creates the conditions for brain fog. If you are losing weight but experiencing cognitive symptoms, the medication is working exactly as intended. Your job is to maintain adequate nutrition within the new, lower caloric framework that tirzepatide creates.
Will switching to semaglutide eliminate brain fog?
Unlikely, if the brain fog is nutrition-related. Switching from tirzepatide to semaglutide will change the degree of appetite suppression but will not address the underlying nutritional deficiencies or dehydration driving cognitive symptoms. If anything, the adjustment period of switching medications may temporarily worsen brain fog before it improves.
Can keto diet make tirzepatide brain fog worse?
It can during the adaptation period. Combining ketogenic dieting with GLP-1 medications removes the brain primary fuel source (glucose) while simultaneously reducing total caloric intake. Until the brain adapts to using ketones, which typically takes one to three weeks, cognitive symptoms may intensify. If you choose a keto approach, the adaptation period for brain fog will be longer and potentially more pronounced. Following the tirzepatide foods to avoid guidance while maintaining adequate complex carbohydrates is generally a safer approach for preserving cognitive function.
Does tirzepatide brain fog affect everyone?
No. Many users report no cognitive changes at all, and some report improved mental clarity. The users most likely to experience brain fog are those who eat significantly less than they need, do not hydrate adequately, skip supplementation, or have pre-existing nutritional deficiencies. Users who follow a structured tirzepatide meal plan from day one are much less likely to develop cognitive symptoms.
Can I take nootropics with tirzepatide for brain fog?
Common nootropics like caffeine, L-theanine, and alpha-GPC are generally safe to use alongside tirzepatide, though you should discuss any supplements with your provider. However, nootropics address symptoms rather than root causes. If your brain fog stems from dehydration or B12 deficiency, no amount of cognitive enhancement supplements will provide lasting relief. Fix the fundamentals first, then consider nootropics as a complement if needed. The GLP-1 vitamin guide covers which supplements have evidence behind them and which are marketing hype.
External resources
Tirzepatide ameliorates spatial learning and memory impairment in diabetic rats (PubMed Central)
Neurodegeneration and stroke after semaglutide and tirzepatide (JAMA Network Open)
GLP-1 medications and Alzheimer disease (Alzheimer Association)
Exercise prevents bone and cognitive decline on weight loss drugs (Healthline)
For researchers serious about optimizing their cognitive performance while using tirzepatide, SeekPeptides provides the most comprehensive resource available, with evidence-based protocol guides, supplement recommendations, and a community of thousands who have navigated these exact challenges and found solutions that work.
In case I do not see you, good afternoon, good evening, and good night. May your thoughts stay sharp, your nutrition stay complete, and your fog stay lifted.