Feb 23, 2026

On paper, these three medications look like variations of the same thing. A weekly injection. Appetite suppression. Weight loss. In practice, the differences between semaglutide, tirzepatide, and retatrutide could mean 10 extra pounds lost, fewer side effects, or access to a medication that does not even exist on pharmacy shelves yet. That gap matters more than most people realize.
Semaglutide was the first to break through. It showed the world that a GLP-1 receptor agonist could produce meaningful, sustained weight loss in people without diabetes. Then tirzepatide arrived and raised the bar, adding a second receptor target and delivering results that made semaglutide look modest by comparison. Now retatrutide sits in late-stage clinical trials, targeting three receptors simultaneously and producing weight loss numbers that researchers once thought impossible with medication alone.
But weight loss percentages only tell part of the story. The real questions are harder. Which one causes fewer side effects? Which works best for someone with fatty liver disease? What about someone who has already tried semaglutide and hit a plateau? And what do you do when the most effective option is not yet available at your local pharmacy? This guide breaks down every meaningful difference between these three medications, with specific numbers from clinical trials, practical dosing information, and honest guidance about who should consider each option. Whether you are comparing semaglutide against tirzepatide or trying to understand where retatrutide fits into the picture, the data tells a clear story once you know how to read it.
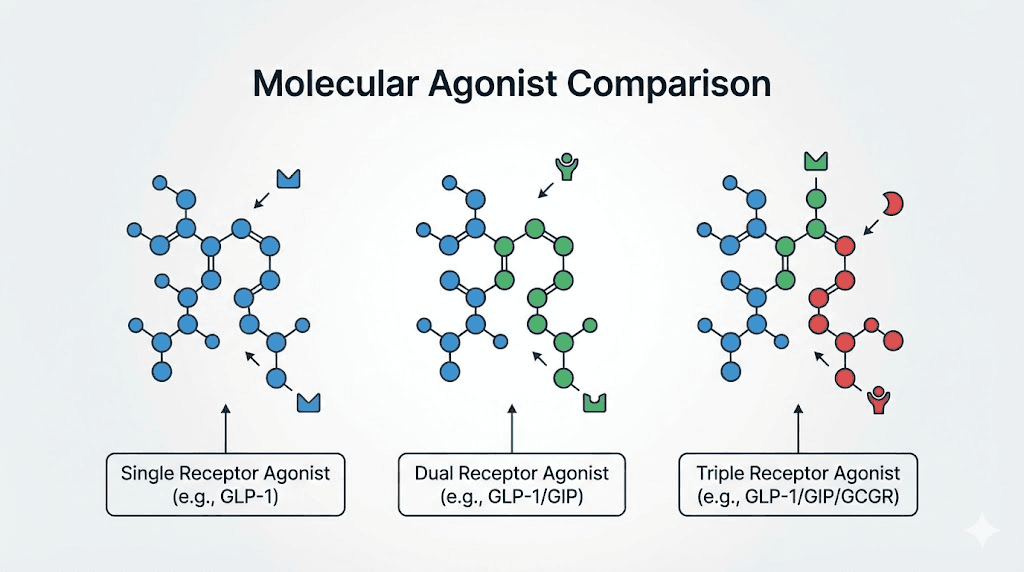
The fundamental difference: one receptor, two, or three
Every comparison between these three medications starts with the same basic question. How many hormone receptors does each one activate? The answer explains nearly everything else, from how much weight people lose to which side effects appear to what metabolic benefits extend beyond the scale.
Think of it this way. Your body has a complex signaling system that regulates hunger, blood sugar, fat storage, and energy expenditure. Semaglutide taps into one channel of that system. Tirzepatide taps into two. Retatrutide taps into three. More channels means more pathways for the medication to influence your metabolism, but it also means more complexity and, potentially, more side effects.
How semaglutide works: the GLP-1 single agonist
Semaglutide activates one receptor: the glucagon-like peptide-1 (GLP-1) receptor. That single target does a remarkable amount of work. When semaglutide binds to GLP-1 receptors in the brain, it reduces appetite and increases feelings of fullness. In the pancreas, it stimulates insulin release and suppresses glucagon, which helps regulate blood sugar. In the stomach, it slows gastric emptying, meaning food stays in your digestive system longer and you feel satisfied with smaller meals.
The science behind this is well established. GLP-1 is a natural hormone your body produces after eating. Semaglutide mimics that hormone but lasts much longer in the bloodstream, roughly seven days compared to the minutes a natural GLP-1 molecule survives. That extended duration is why a once-weekly injection works. If you are curious about how quickly semaglutide starts working, most people notice appetite changes within the first week or two, though meaningful weight loss takes longer to appear on the scale.
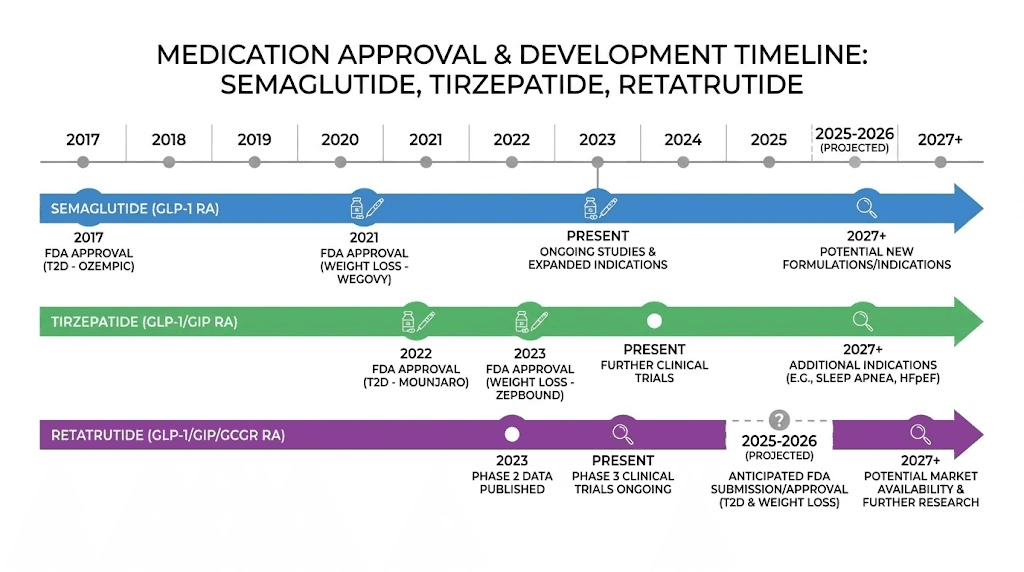
Available as both Wegovy (for weight management) and Ozempic (for type 2 diabetes), semaglutide has the longest track record of the three. It also comes in oral formulations, which neither tirzepatide nor retatrutide currently offer in approved form, though oral tirzepatide is in development.
How tirzepatide works: the GLP-1 and GIP dual agonist
Tirzepatide changed the game by adding a second receptor target: glucose-dependent insulinotropic polypeptide (GIP). This dual mechanism is not just semaglutide with an extra ingredient. The GIP receptor activation creates effects that complement and amplify what GLP-1 alone can do.
GIP is interesting. For years, researchers debated whether activating GIP receptors would even help with weight loss. Some early data suggested it might actually promote fat storage. But tirzepatide proved that when GIP and GLP-1 receptors are activated together, the combination produces substantially more weight loss than either pathway alone. The GIP component appears to enhance insulin sensitivity in fat tissue, improve lipid metabolism, and potentially increase energy expenditure in ways that pure GLP-1 agonists cannot match.
Sold as Mounjaro (for diabetes) and Zepbound (for weight management), tirzepatide has quickly become the most prescribed alternative for people who want more weight loss than semaglutide delivers. The tirzepatide dosing schedule follows a similar weekly injection pattern, and many providers now offer compounded tirzepatide as well.
How retatrutide works: the GLP-1, GIP, and glucagon triple agonist
Retatrutide takes the concept one step further. It activates all three receptors: GLP-1, GIP, and the glucagon receptor. That third receptor is the key differentiator, and it is also the most controversial.
Glucagon is traditionally thought of as the opposite of insulin. When blood sugar drops, glucagon tells the liver to release stored glucose. So why would you want a weight loss medication that activates glucagon receptors? Because glucagon does far more than regulate blood sugar. It increases energy expenditure, meaning your body burns more calories at rest. It promotes fat oxidation, meaning your body preferentially burns fat for fuel. And it has powerful effects on the liver, reducing fat accumulation in ways that neither GLP-1 nor GIP activation can match.
The retatrutide dosing protocol reflects this triple mechanism. The medication must be titrated carefully because three receptor pathways mean three sets of biological effects to manage. Currently in Phase 3 clinical trials under Eli Lilly, retatrutide is not FDA-approved and cannot be prescribed through standard pharmacies. However, understanding its data matters because it represents where the field is heading. You can track the latest updates on retatrutide availability as trials progress.
Why receptor count matters for your results
More receptors does not automatically mean better. It means different.
Single receptor activation (semaglutide) offers a well-understood, predictable response with years of safety data. Dual activation (tirzepatide) adds metabolic benefits that translate to roughly 30-50% more weight loss. Triple activation (retatrutide) pushes weight loss even further while adding unique benefits for liver health and energy expenditure, but with less long-term safety data and some novel side effects.
The clinical trial numbers reflect this progression clearly. And those numbers are what we need to examine next.
Weight loss results from clinical trials
Numbers do not lie, but they do need context. The weight loss figures from clinical trials for these three medications tell a compelling story, one where each generation of treatment meaningfully outperforms the last. But raw percentages can be misleading without understanding the trial designs, patient populations, and timelines involved.
Semaglutide STEP trials: the benchmark
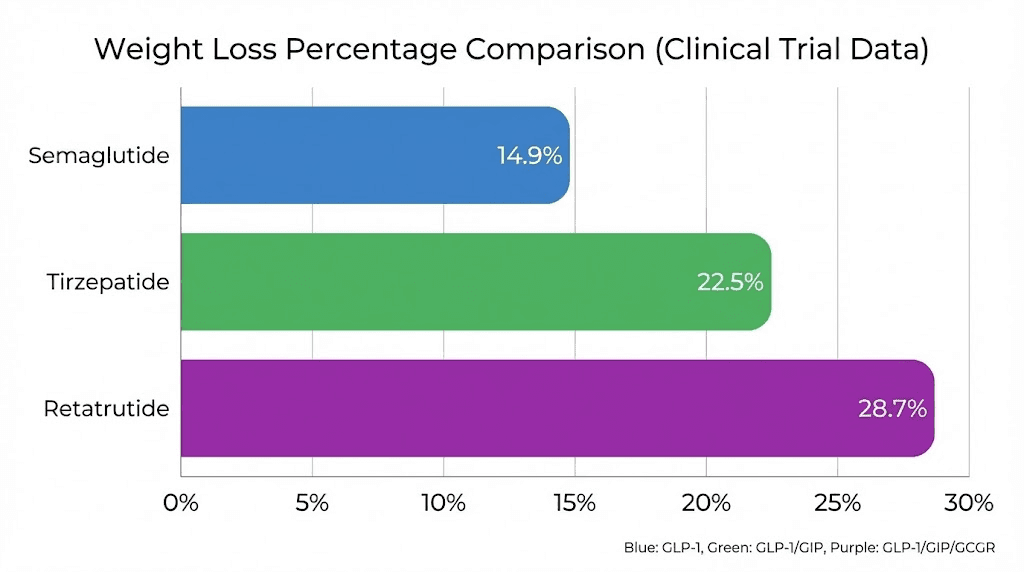
The STEP (Semaglutide Treatment Effect in People with Obesity) trial program established semaglutide as a legitimate weight loss medication. In STEP 1, participants without diabetes who received 2.4mg of semaglutide weekly lost an average of 14.9% of their body weight over 68 weeks. The placebo group lost 2.4%.
That 14.9% figure became the number to beat. For a 220-pound person, it means losing roughly 33 pounds. Significant. Life-changing for many. But not everyone responded equally. About one-third of participants achieved 20% or greater weight loss, while some achieved less than 10%. Individual variation matters enormously with any of these medications, and understanding why some people do not lose weight on semaglutide helps set realistic expectations.
The STEP trials also showed that weight loss continued throughout most of the treatment period, with most participants still losing weight at 68 weeks. The first-month results on semaglutide typically show 3-5 pounds of loss, with the pace accelerating as doses increase. For a visual sense of what these results look like in real people, semaglutide before-and-after results provide useful reference points.
Tirzepatide SURMOUNT trials: raising the bar
Then came the SURMOUNT trials. SURMOUNT-1 tested tirzepatide in people with obesity but without diabetes, and the results stunned the medical community. At the highest dose of 15mg, participants lost an average of 22.5% of their body weight over 72 weeks. Even the middle dose of 10mg produced 19.5% weight loss.
Those numbers represented a massive leap. For the same 220-pound person, 22.5% means losing roughly 50 pounds, nearly 17 more pounds than semaglutide would produce on average.
But the real proof came from the head-to-head trial. SURMOUNT-5, published in the New England Journal of Medicine, directly compared the maximum tolerated doses of tirzepatide and semaglutide in the same patient population. The results were unambiguous. Tirzepatide produced 20.2% weight loss compared to 13.7% for semaglutide at 72 weeks. Nearly one-third of tirzepatide participants achieved at least 25% total body weight loss, compared to 16% on semaglutide. The tirzepatide weight loss timeline shows that most people begin seeing noticeable changes within the first four to six weeks.
A meta-analysis across multiple studies confirmed the pattern: tirzepatide produces roughly 4.23 kg (about 9.3 pounds) more weight loss than semaglutide on average. That difference is clinically meaningful. Detailed tirzepatide before-and-after results show the visual impact clearly, and the before-and-after weight loss data remains consistent across different populations.
Retatrutide Phase 2 and Phase 3: breaking records
Retatrutide entered the picture with Phase 2 trial data that immediately captured attention. At the 12mg dose, participants lost an average of 24.2% of their body weight in just 48 weeks. That is more weight lost in less time than either semaglutide or tirzepatide achieved in their respective trials. And the weight loss curve had not yet plateaued, meaning participants were likely to lose even more with continued treatment.
The Phase 3 TRIUMPH-4 trial confirmed and exceeded those expectations. Participants with obesity who received the 12mg dose lost an average of 28.7% of their body weight at 68 weeks, translating to an average of 71.2 pounds from a baseline weight of approximately 248 pounds. Nearly half of all participants achieved at least 25% weight loss, and a meaningful portion exceeded 30% or even 35%.
These numbers are unprecedented for any medication. The retatrutide dosage chart shows the titration schedule that produced these results, and understanding the complete dosing schedule is essential for anyone tracking this medication through clinical development.
What these percentages mean for real people
Let us make this concrete. For someone starting at 250 pounds:
Semaglutide 2.4mg: approximately 37 pounds lost (to roughly 213 lbs)
Tirzepatide 15mg: approximately 56 pounds lost (to roughly 194 lbs)
Retatrutide 12mg: approximately 72 pounds lost (to roughly 178 lbs)
The difference between the lowest and highest performer is 35 pounds. That is not trivial. It is the difference between dropping a few clothing sizes and transforming your entire body composition. For someone starting at 300 pounds, the gap widens even further.
However, these are averages. Some people on semaglutide lose more than some people on retatrutide. Genetics, diet, exercise, starting weight, metabolic health, and adherence all influence individual outcomes. The medication creates the conditions for weight loss. Your body and your behaviors determine the actual result. Resources like the semaglutide dosage calculator and guides on what to eat while on semaglutide can help optimize individual outcomes regardless of which medication you choose.
The complete comparison table
Seeing all the data side by side makes the differences clearer. This table summarizes every major clinical trial metric across all three medications at their highest studied doses:
Factor | Semaglutide (2.4mg) | Tirzepatide (15mg) | Retatrutide (12mg) |
|---|---|---|---|
Receptor targets | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
Avg weight loss (%) | 14.9% | 22.5% | 28.7% |
Avg weight loss (lbs, 250lb person) | ~37 lbs | ~56 lbs | ~72 lbs |
Trial duration | 68 weeks (STEP 1) | 72 weeks (SURMOUNT-1) | 68 weeks (TRIUMPH-4) |
Achieved 20%+ loss | ~32% | ~57% | ~75% |
Nausea rate | ~18% | 17-22% | 16-25% |
Discontinuation rate | ~7% | 4-7% | 6-10% |
A1c reduction | 1.5-1.8% | 2.0-2.3% | 1.5-2.0% |
Liver fat reduction | Moderate | Significant | Up to 82% |
FDA status | Approved (Wegovy/Ozempic) | Approved (Mounjaro/Zepbound) | Phase 3 trials |
Brand cost (monthly) | $1,300-$1,900 | $1,400-$1,600 | Not available |
Compounded cost (monthly) | $100-$400 | $150-$500 | Research only |
Oral option available | Yes (Rybelsus) | In development | No |
CV outcome data | Yes (SELECT trial) | Ongoing trials | None yet |
Unique side effects | None beyond GI | None beyond GI | Dysesthesia (8-21%) |
The table reveals a clear progression. Each generation adds receptors, weight loss, and metabolic benefits, but also adds complexity and reduces the length of available safety data. That trade-off is the fundamental decision every person faces when choosing between these three options. No single medication is best for everyone. The right choice depends on your health profile, your weight loss goals, your budget, and your tolerance for newer versus more established treatments.
Response rates tell a deeper story
Average weight loss is one thing. Response rates tell you something different. They answer the question: what are my odds of hitting a specific target?
In STEP 1, roughly 86% of semaglutide participants lost at least 5% body weight, 69% lost at least 10%, and 50% lost at least 15%. Those are strong numbers for a single-agonist medication. But tirzepatide pushed every threshold higher. In SURMOUNT-1, 91% lost at least 5%, 79% lost at least 15%, and 36% lost at least 25%. Retatrutide pushed further still. Phase 2 data showed 100% of participants on the 8mg and 12mg doses achieved at least 5% weight loss. Ninety-three percent hit 10%. Eighty-three percent crossed the 15% mark.
What does that mean for you? If you need a 15% weight loss to reach a health-relevant threshold, semaglutide gives you roughly a coin flip. Tirzepatide gives you roughly an 80% chance. Retatrutide, based on available data, pushes that above 80% as well. These probabilities matter when you are deciding which medication to try first, because time spent on a less effective option is time without the metabolic benefits a more effective option might provide.
Side effects and tolerability compared
Weight loss numbers grab headlines. Side effects determine whether people actually stay on the medication long enough to see those numbers become reality. All three of these medications share a common side effect profile rooted in their GLP-1 activity, but each also has unique characteristics worth understanding.
Gastrointestinal side effects: the common thread
Nausea, diarrhea, vomiting, and constipation appear with all three medications. This is expected. GLP-1 receptor activation slows gastric emptying and alters gut signaling. Your digestive system needs time to adjust.
The rates are remarkably similar across all three, despite their different mechanisms:
Semaglutide: Nausea approximately 18%, diarrhea approximately 12%, vomiting approximately 8%
Tirzepatide: Nausea 17-22% (dose dependent), diarrhea 13-16%, vomiting 6-10%
Retatrutide: Nausea 16-25% (dose dependent), diarrhea 15-22%, vomiting 8-13%
The pattern holds consistently. Gastrointestinal side effects are most common during the titration phase, when doses are increasing. Most people find that symptoms peak around weeks two to four of each new dose level and then gradually improve. By the time someone reaches their maintenance dose, many of the early GI issues have resolved or become much more manageable.
For practical strategies on managing these effects, treating constipation on semaglutide and managing constipation on tirzepatide offer evidence-based approaches. Understanding how long tirzepatide diarrhea typically lasts can also help set expectations during the adjustment period. Some people also experience excessive burping on semaglutide or headaches on tirzepatide, both of which tend to be temporary.
Unique to retatrutide: dysesthesia
Here is where retatrutide diverges from the other two. The TRIUMPH-4 trial revealed a notable incidence of dysesthesia, an abnormal sensation affecting touch perception that can cause numbness, tingling, or unusual skin sensitivity. At the 9mg dose, 8.8% of participants experienced dysesthesia. At the 12mg dose, that figure jumped to 20.9%. In the placebo group, only 0.7% reported it.
One in five. That is a substantial rate for a side effect that neither semaglutide nor tirzepatide produces at any meaningful frequency.
The good news is that these dysesthesia events were generally mild. Eli Lilly reported that the sensation rarely caused participants to discontinue treatment. But the side effect is novel enough that researchers and analysts are watching closely for additional data from upcoming TRIUMPH trials. The glucagon receptor activation unique to retatrutide likely drives this effect, though the exact mechanism remains under investigation.
For context, semaglutide and tirzepatide do not produce dysesthesia at clinically significant rates, though a recent paper in PMC examined allodynia and dysesthesia reports across GLP-1 medications more broadly. The side effect profiles for semaglutide and tirzepatide are better characterized because both medications have been on the market longer.
Fatigue and energy levels
Fatigue is a common complaint across all three medications, particularly during the early titration weeks. Reduced caloric intake naturally leads to some decrease in energy levels, especially when the body is adjusting to a new metabolic state. Understanding semaglutide-related fatigue and whether tirzepatide causes tiredness helps people plan their titration around work and life demands.
Interestingly, some users report that semaglutide eventually increases their energy as weight decreases, and similar reports exist for tirzepatide and energy levels. Retatrutide, with its glucagon component that increases energy expenditure, may theoretically influence energy levels differently, but clinical trial data on subjective energy has not yet been published in detail.
Other side effects to be aware of include insomnia on tirzepatide, sleep disruption on semaglutide, dizziness on semaglutide, and body aches on tirzepatide. Anxiety on tirzepatide and muscle pain with tirzepatide have also been reported, though these are less common.
Discontinuation rates: who actually stays on treatment?
The most telling safety metric is how many people stop taking the medication because of side effects. In clinical trials:
Semaglutide: Approximately 7% discontinued due to adverse events
Tirzepatide: Approximately 4-7% discontinued due to adverse events (dose dependent)
Retatrutide: Approximately 6-10% discontinued due to adverse events (dose dependent)
These rates are relatively close, suggesting that despite the differences in mechanism and side effect profiles, all three medications are tolerable for the vast majority of people who start them. Proper titration, starting low and increasing gradually, is the single most important factor in tolerability across all three options.
Which is easiest to tolerate?
Based on the available data, tirzepatide has the lowest discontinuation rates and similar or slightly lower GI side effect rates compared to semaglutide. Retatrutide has the highest overall adverse event burden, primarily because of the dysesthesia issue. But tolerability is deeply individual. Some people breeze through semaglutide titration with no issues while struggling with tirzepatide, and vice versa.
The complete guide to GLP-1 fatigue and the GLP-1 headache guide offer broader perspectives on managing common side effects that apply to all three medications.
Dosing schedules and titration protocols
All three medications follow the same basic principle: start low, increase gradually, inject once per week. The specifics differ, and those details matter for both effectiveness and tolerability.
Semaglutide titration: 0.25mg to 2.4mg
The semaglutide titration schedule moves through five dose levels over approximately 16-20 weeks:
Weeks 1-4: 0.25mg weekly
Weeks 5-8: 0.5mg weekly
Weeks 9-12: 1.0mg weekly
Weeks 13-16: 1.7mg weekly
Week 17 onward: 2.4mg weekly (maintenance dose)
Each increase happens monthly. Some providers extend the time at lower doses if a patient experiences significant GI side effects, and that is perfectly reasonable. The semaglutide 5mg/ml dosage chart is particularly useful for people using compounded versions, while the semaglutide dosage in units guide helps convert between milligrams and injection units.
Timing matters too. Research suggests that the best time of day to take semaglutide varies by individual, but consistency matters more than the specific hour. The same applies to choosing an injection site and proper injection technique.
Tirzepatide titration: 2.5mg to 15mg
Tirzepatide uses a six-step titration over approximately 20-24 weeks:
Weeks 1-4: 2.5mg weekly
Weeks 5-8: 5mg weekly
Weeks 9-12: 7.5mg weekly
Weeks 13-16: 10mg weekly
Weeks 17-20: 12.5mg weekly
Week 21 onward: 15mg weekly (maximum dose)
Not everyone reaches 15mg. Many people achieve satisfactory results at 10mg or 12.5mg, and the clinical trials showed meaningful weight loss at every dose level above 5mg. The tirzepatide compound dosage chart and the dosing in units guide help people using compounded formulations calculate their exact amounts.
Understanding unit conversions like 2.5mg in units becomes important when working with compounded tirzepatide. The starting dose for compounded tirzepatide follows the same principles as the brand-name version, and resources on syringe dosage calculations ensure accuracy.
For those interested in a gentler approach, microdosing tirzepatide has gained attention as a way to start at even lower doses than the standard protocol suggests.
Retatrutide titration: 1mg to 12mg
The retatrutide titration protocol tested in Phase 3 trials follows a five-step escalation:
Weeks 1-4: 1mg weekly
Weeks 5-8: 2mg weekly
Weeks 9-12: 4mg weekly
Weeks 13-16: 8mg weekly
Week 17 onward: 12mg weekly (maximum dose)
The dose doubles at each step (with the exception of the 1mg to 2mg increase), which is a faster escalation rate than either semaglutide or tirzepatide. This aggressive titration schedule may contribute to the higher side effect rates seen in early treatment weeks. The complete retatrutide dosage chart details each step, and the dosing schedule guide provides context for each escalation.
TRIUMPH-4 also tested a 9mg dose, and additional trials are evaluating a 4mg maintenance dose. This suggests that the final approved dosing schedule (if and when retatrutide receives approval) may offer more flexibility than the Phase 2 protocol. Resources like the retatrutide dosing guide and the 20mg dosing guide cover various scenarios in detail.
Key differences in titration approach
All three medications use monthly dose increases. All three are once-weekly injections. But the pace differs. Semaglutide reaches its maximum dose in about 16-17 weeks. Tirzepatide takes 20-21 weeks. Retatrutide reaches 12mg in about 16 weeks but uses larger relative jumps at each step.
For people who are sensitive to GI side effects, slower titration generally produces better tolerability. Some providers extend the time at each dose level to two months instead of one, especially when transitioning to higher doses. This applies to all three medications. The best time to take your GLP-1 shot can also influence how well you tolerate each dose increase.
Metabolic benefits beyond the scale
Weight loss is the headline. But these medications do far more than help people shed pounds. The metabolic improvements they produce may ultimately matter more for long-term health than the number on the scale.
Blood sugar and A1c improvements
All three medications were originally developed for (or in the pipeline for) type 2 diabetes. Their blood sugar effects are substantial:
Semaglutide: Reduces A1c by 1.5-1.8% from baseline (Ozempic trials)
Tirzepatide: Reduces A1c by 2.0-2.3% from baseline (SURPASS trials). In the SURPASS-2 head-to-head against semaglutide for diabetes, tirzepatide produced significantly greater A1c reductions.
Retatrutide: Phase 2 data showed A1c reductions of 1.5-2.0%, with the glucagon component adding complexity to blood sugar management.
For people with type 2 diabetes, all three medications offer meaningful glycemic control. Tirzepatide currently leads in head-to-head comparisons against semaglutide for diabetes management. Retatrutide data in diabetes populations is still emerging from the TRIUMPH trial program.
Liver fat: where retatrutide separates itself
This is perhaps the most striking difference between the three medications.
Metabolic dysfunction-associated steatotic liver disease (MASLD, formerly known as NAFLD) affects millions of people worldwide. It is strongly associated with obesity, insulin resistance, and type 2 diabetes. Reducing liver fat is critical for preventing progression to more serious conditions like cirrhosis.
All three medications reduce liver fat to some degree. But retatrutide does something remarkable. In a Phase 2a substudy published in Nature Medicine, retatrutide at the 12mg dose reduced liver fat by an average of 82.4% from baseline at 24 weeks. At the 8mg dose, the reduction was 81.4%. By comparison, the placebo group showed essentially no change (0.3% increase).
Even more impressive, over 85% of participants receiving the two highest retatrutide doses achieved complete resolution of steatosis, defined as liver fat content dropping below 5%. At 48 weeks, 93% of patients on the 12mg dose had less than 5% liver fat.
Why does retatrutide outperform here? The glucagon receptor. Glucagon directly activates fat oxidation in the liver, promoting the breakdown and clearance of stored fat in hepatocytes. This is a metabolic pathway that neither GLP-1 nor GIP activation can access with the same potency. For anyone tracking the broader category of medications that target liver fat, understanding how mazdutide compares to retatrutide provides additional context on the competitive landscape.
Semaglutide and tirzepatide also reduce liver fat, with tirzepatide generally showing greater reductions than semaglutide, but neither approaches the 80%+ reductions seen with retatrutide. The survodutide comparison is worth noting here too, as survodutide is another glucagon-containing dual agonist that shows strong liver fat effects.
Cardiovascular benefits
Semaglutide has the strongest cardiovascular evidence. The SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events (heart attack, stroke, cardiovascular death) in people with obesity and established cardiovascular disease. This benefit appeared independent of weight loss, suggesting direct cardiovascular protective effects from GLP-1 receptor activation.
Tirzepatide cardiovascular outcome trials are ongoing. Real-world evidence suggests favorable cardiovascular effects, with some retrospective analyses showing tirzepatide may reduce major cardiovascular events by approximately 14% compared to semaglutide. However, this data comes from observational studies, not randomized controlled trials, so it should be interpreted cautiously.
Retatrutide cardiovascular data is limited to early-phase trials. The glucagon receptor raises theoretical concerns about heart rate increases (glucagon is known to increase heart rate), but clinical trial data has not shown significant safety issues in this regard so far.
Sleep apnea improvements
Weight loss from any of these medications can improve obstructive sleep apnea. But tirzepatide has specific clinical trial data showing impressive results. In a dedicated sleep apnea trial, tirzepatide reduced the apnea-hypopnea index (AHI) by 25.3 events per hour at 52 weeks compared to 5.3 with placebo. That is a dramatic improvement that can eliminate the need for CPAP in many patients.
Semaglutide also improves sleep apnea markers, though with somewhat less dramatic reductions in AHI compared to tirzepatide. The greater weight loss produced by tirzepatide likely contributes to its superior performance in this area. Retatrutide has not yet been studied in a dedicated sleep apnea trial, but given its greater weight loss, the expectation is that sleep apnea improvements would be at least as significant as those seen with tirzepatide, possibly more so. The mechanism is straightforward: less fat around the neck and upper airway means less airway collapse during sleep. For people whose sleep apnea is severe enough to affect daily functioning, the greater weight loss from tirzepatide or potentially retatrutide could be a deciding factor in medication choice.
Insulin sensitivity and metabolic markers
All three medications improve insulin sensitivity, but the magnitude varies. Retatrutide Phase 2 data showed insulin resistance reductions of over 50% from baseline at higher doses, with fasting insulin levels dropping by up to 71%. Adiponectin, a hormone associated with metabolic health, increased by 29.8% to 99.3% depending on dose and duration.
Tirzepatide also produces significant insulin sensitivity improvements, outperforming semaglutide in head-to-head diabetes trials. The GIP receptor activation in both tirzepatide and retatrutide appears to contribute substantially to insulin sensitization in fat tissue, which may explain why both outperform semaglutide on this metric.
Triglycerides, a blood fat marker linked to cardiovascular disease, dropped by over 40% in retatrutide participants at the higher doses. All three medications reduce triglycerides, but retatrutide produces the largest reductions, likely because of the glucagon-driven increase in fat oxidation.
Availability, approval status, and cost considerations
Having the best clinical trial data means nothing if you cannot actually get the medication. This is where the practical reality diverges sharply from the clinical promise.
Semaglutide: widely available, multiple options
Semaglutide has the broadest availability of the three. It is FDA-approved under two brand names: Wegovy for chronic weight management and Ozempic for type 2 diabetes. Both contain the same active ingredient at different dose ranges.
Brand-name semaglutide carries a retail price of approximately $1,300 to $1,900 per month without insurance. With insurance coverage (which has expanded significantly), out-of-pocket costs can range from $0 to $500 depending on the plan.
Compounded semaglutide offers a more affordable alternative, typically costing $100 to $400 per month. Compounded versions contain the same active ingredient but are prepared by compounding pharmacies rather than the brand-name manufacturer. Understanding how to reconstitute semaglutide is essential for anyone using compounded lyophilized (freeze-dried) formulations.
Semaglutide is also available in oral form (Rybelsus for diabetes, and a newer oral Wegovy formulation), making it the only one of the three with a non-injectable option currently on the market. The oral semaglutide guide covers this option in detail, and some compounding pharmacies now offer semaglutide with methylcobalamin or semaglutide with B12 as combination formulations.
Tirzepatide: available and gaining ground
Tirzepatide is FDA-approved as Mounjaro (for type 2 diabetes) and Zepbound (for chronic weight management). Both are manufactured by Eli Lilly.
Retail pricing sits between $1,400 and $1,600 per month without insurance. Insurance coverage is expanding but remains more variable than semaglutide coverage, partly because tirzepatide has been on the market for less time.
Compounded tirzepatide is available and often costs $150 to $500 per month, depending on the pharmacy, dose, and formulation. Guides on how to reconstitute tirzepatide help ensure proper preparation, and resources on tirzepatide storage and refrigeration are critical for maintaining potency. Understanding refrigeration requirements and expiration timelines prevents waste.
Some compounding pharmacies offer tirzepatide in combination formulations such as tirzepatide with B12, tirzepatide with niacinamide, tirzepatide with glycine, and tirzepatide with glycine and B12. Finding affordable compounded tirzepatide has become a priority for many people as brand-name costs remain high. Empower Pharmacy tirzepatide is one of the more well-known compounding options.
Tirzepatide is also being developed in oral form, with tablet versions and even sublingual drops in various stages of development and compounding availability.
Retatrutide: not yet available through standard channels
Retatrutide is not FDA-approved. It cannot be prescribed by physicians through normal channels. It is not covered by any insurance plan. It does not have an established retail price.
Eli Lilly has seven additional Phase 3 TRIUMPH trials expected to complete during the course of the coming months and into the following year. If those trials are successful, Lilly would likely submit a New Drug Application (NDA) to the FDA, which typically takes 10 months to review. The most optimistic approval timeline points to late the following year, with commercial availability potentially in the year after that.
Some research chemical suppliers and international compounding pharmacies offer retatrutide, though these sources operate outside normal pharmaceutical channels. The complete guide to buying retatrutide and the online purchasing guide cover what options currently exist. Resources on retatrutide pricing and bulk purchasing provide cost context for those exploring research-grade sources. Information on Peptide Sciences retatrutide and bulk sourcing from China represents the current landscape for those in the research community.
The peptide cost calculator at SeekPeptides can help compare the true cost per dose across different formulations and suppliers.
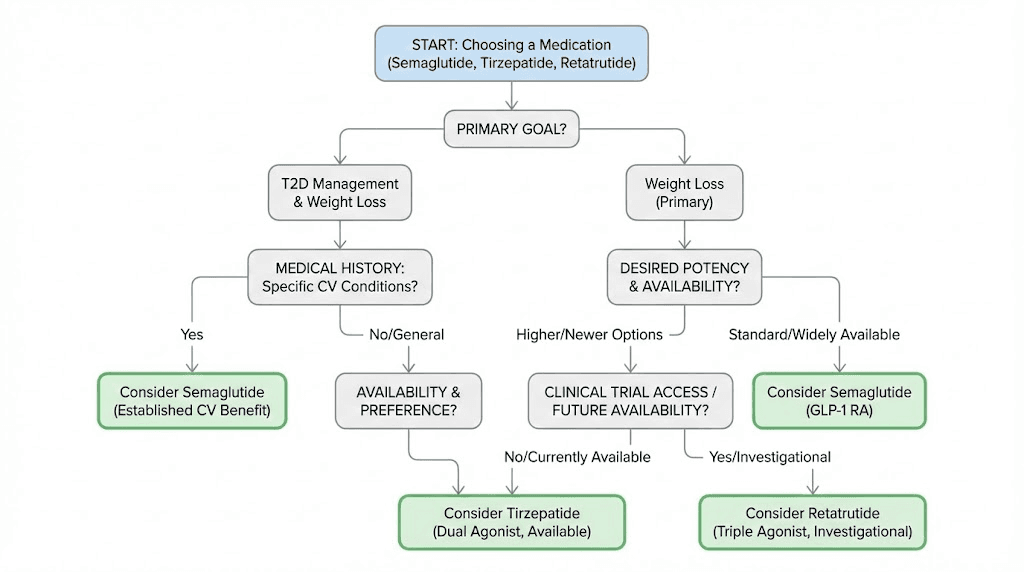
Choosing the right medication for your goals
The best medication is not necessarily the one with the highest weight loss numbers. It is the one that fits your specific situation, goals, medical history, and practical constraints.
For moderate, steady weight loss with the most safety data
Choose semaglutide. It has the longest track record, the most extensive post-market safety data, cardiovascular outcome evidence, and broad availability. If you need to lose 10-15% of your body weight and want the most established option, semaglutide is the clear choice. It is also the only option with a proven oral formulation, which matters if you prefer not to inject.
People who do well on semaglutide typically see significant appetite suppression within the first few weeks. If you are wondering how long semaglutide takes to work or whether appetite suppression is immediate, most people notice changes within days of the first injection, though weight loss takes weeks to become visible.
For maximum available weight loss right now
Choose tirzepatide. It is FDA-approved, widely available, and produces roughly 30-50% more weight loss than semaglutide in clinical trials. The head-to-head SURMOUNT-5 data leaves no room for debate: tirzepatide outperforms semaglutide for weight loss in the same patient population.
If you are currently on semaglutide and have hit a weight loss plateau, switching to tirzepatide is a well-supported option. The semaglutide-to-tirzepatide conversion chart provides dosing equivalencies for a smooth transition. Understanding how quickly tirzepatide works and the expected timeline for results helps set realistic expectations during the switch.
For potential future maximum weight loss
Watch retatrutide. If clinical trials continue to show the current results, retatrutide will likely become the most effective single medication for weight loss once approved. The 28.7% average weight loss at 68 weeks surpasses anything else in the pipeline.
But it is not available through standard medical channels yet. If you want to understand the timeline, the retatrutide availability guide tracks regulatory progress.
For type 2 diabetes management
Both semaglutide and tirzepatide are FDA-approved for type 2 diabetes. Tirzepatide produced greater A1c reductions in the SURPASS-2 head-to-head trial, making it the stronger option for glucose control. However, semaglutide has cardiovascular outcome data (the SELECT trial) that tirzepatide does not yet have from a completed dedicated outcomes trial.
Your endocrinologist can help weigh the blood sugar benefits against the cardiovascular evidence when choosing between them. The comparison between tirzepatide and semaglutide dosing provides practical guidance for understanding how the two stack up.
For liver fat reduction specifically
If MASLD or fatty liver disease is your primary concern, retatrutide shows the most promise by a wide margin. The 82% average liver fat reduction and 85%+ steatosis resolution rates are unmatched. Since retatrutide is not yet available, tirzepatide is the next best option among approved medications, as it produces greater liver fat reductions than semaglutide.
For those concerned about side effects
Tirzepatide has the best overall tolerability profile in clinical trials, with the lowest discontinuation rates. Semaglutide is a close second with a longer safety track record. Retatrutide has the unique dysesthesia concern that affects up to 20% of people at the highest dose.
If side effects have been a barrier in the past, supplements that support tirzepatide tolerance and dietary adjustments like following a tirzepatide-friendly diet plan or a semaglutide-compatible food list can make a meaningful difference. Knowing which foods to avoid on tirzepatide and foods to avoid on semaglutide reduces GI side effects significantly.
Switching between these three medications
Switching medications is increasingly common. Maybe semaglutide worked at first but the results have stalled. Maybe you want the additional weight loss tirzepatide offers. Maybe retatrutide becomes available and you want to try the triple agonist. Whatever the reason, switching requires careful planning.
Semaglutide to tirzepatide: the most common switch
This is the most well-documented transition. The conversion chart between semaglutide and tirzepatide provides specific dosing guidance. General principles include:
There is no need for a washout period. You can start tirzepatide the week after your last semaglutide injection.
Starting dose depends on your semaglutide dose. If you were on 2.4mg semaglutide, most providers start at 5mg or 7.5mg tirzepatide rather than the standard 2.5mg starting dose.
GI side effects may temporarily increase during the transition because tirzepatide adds GIP receptor activation.
Most people see additional weight loss resume within 4-8 weeks of switching.
The reverse switch from tirzepatide to semaglutide is less common but occasionally necessary, usually due to insurance coverage or cost considerations.
Why people switch
The most common reasons include weight loss plateaus, inadequate response at maximum dose, side effect intolerance, cost or insurance changes, and desire for greater efficacy. Understanding what to do when tirzepatide stops working or why you might not lose weight in the first four weeks on semaglutide can help determine whether a switch is truly needed or whether adjustments to the current medication might solve the problem.
Sometimes the issue is not the medication itself but lifestyle factors. The whether exercise is necessary with semaglutide discussion and understanding how tirzepatide affects metabolism provide important context.
Switching to retatrutide from tirzepatide
This switch will become relevant as retatrutide moves toward potential approval. For those in the research community already tracking this transition, how long retatrutide takes to work after tirzepatide and the dosing schedule after high-dose tirzepatide provide practical guidance for this specific scenario.
The key consideration is that retatrutide adds glucagon receptor activation that the body has never experienced with either semaglutide or tirzepatide. New side effects, particularly dysesthesia, may appear even if you tolerated the other medications well. Starting at a conservative dose and titrating slowly makes sense for anyone transitioning from a dual agonist to a triple agonist.
Timeline considerations for switching
Give each medication a fair trial before deciding to switch. That means at least 12-16 weeks at or near the maximum tolerated dose. Short trials at low doses do not provide meaningful data about how you will respond at therapeutic levels. Knowing how long tirzepatide takes to suppress appetite and how long semaglutide takes to suppress appetite helps establish reasonable evaluation periods.
Also consider that withdrawal symptoms from semaglutide are a real phenomenon. Stopping any of these medications abruptly can lead to rebound hunger, blood sugar fluctuations, and weight regain. A planned transition between medications is always preferable to an abrupt stop.
Reconstitution and preparation for compounded versions
Brand-name medications come in pre-filled pens ready to inject. Compounded versions typically come as lyophilized (freeze-dried) powder that requires reconstitution with bacteriostatic water before use. This is where proper technique directly impacts effectiveness.
How to reconstitute each medication
The reconstitution process is essentially the same for all three: inject bacteriostatic water into the vial containing the powder, swirl gently (never shake), and allow the solution to dissolve completely. But the specific amounts of water and resulting concentrations differ based on the formulation.
For semaglutide, the complete reconstitution guide walks through each step with exact calculations. The peptide reconstitution calculator at SeekPeptides automates the math, ensuring you get the correct concentration for your specific vial size and desired dose.
For tirzepatide, the reconstitution process follows similar principles. Tirzepatide is generally more stable in solution than semaglutide, but proper technique still matters.
For retatrutide, the bacteriostatic water calculations for 10mg vials are a common starting point. Since retatrutide is not available as a pharmaceutical product, all current formulations are research-grade and require reconstitution.
Storage requirements
All three medications should be stored refrigerated (36-46 degrees Fahrenheit / 2-8 degrees Celsius) after reconstitution. Unreconstituted lyophilized powder can typically be stored at room temperature in a cool, dark place, though refrigeration extends stability.
Understanding how long tirzepatide lasts in the fridge and whether semaglutide expires after 28 days prevents waste and ensures potency. Proper storage practices are not optional with peptide medications. Temperature excursions can degrade the active molecule and reduce effectiveness.
Injection technique
All three medications are injected subcutaneously, typically in the abdomen, thigh, or upper arm. The technique is straightforward once you learn the basics. Resources on how to inject GLP-1 medications and where to inject cover the fundamentals that apply to all three. For retatrutide specifically, injection site guidance is available, and stomach injection technique for tirzepatide provides location-specific instructions.
Rotating injection sites is important for all three medications. Using the same spot repeatedly can cause lipohypertrophy (hardened areas under the skin) that impair absorption. Rotate between the left and right sides of your abdomen, thighs, and upper arms on a consistent schedule.
The comparative landscape: other medications in the pipeline
Semaglutide, tirzepatide, and retatrutide are not the only contenders in the weight loss medication space. Understanding how they compare to emerging alternatives provides valuable context for anyone making decisions about treatment.
Several other comparison articles may be relevant depending on your specific interests. The cagrisema comparison with tirzepatide examines a novel semaglutide-cagrilintide combination. The orforglipron analysis looks at an oral GLP-1 agonist in development. And the mazdutide comparison examines another dual agonist being developed in China.
For those who have considered older medications like phentermine alongside semaglutide, the GLP-1 class has fundamentally changed expectations for what medication-assisted weight loss can achieve.
The Ozempic versus retatrutide comparison is another frequently searched topic, essentially pitting the established standard against the most promising newcomer. Understanding whether GLP-1 and Ozempic are the same thing helps clarify the broader category for people new to these medications.
Practical optimization: diet, supplements, and lifestyle
No medication works in isolation. What you eat, how you move, and what supplements you take all influence outcomes. The strategies differ slightly depending on which medication you use.
Protein intake becomes critical on any of these medications because rapid weight loss can include muscle loss. Aim for 0.7 to 1.0 grams of protein per pound of body weight daily. The best protein shakes for GLP-1 users offer convenient options when appetite suppression makes large meals difficult.
A structured meal plan for tirzepatide users can help ensure adequate nutrition despite reduced appetite. Similarly, knowing the best foods to eat on semaglutide supports both weight loss and nutritional adequacy. Understanding alcohol considerations with semaglutide and alcohol interactions with tirzepatide is important for social situations.
The supplements that complement tirzepatide article covers B vitamins, electrolytes, and other nutrients that may become depleted during rapid weight loss. Formulations that combine semaglutide with glycine or tirzepatide with glycine attempt to address some of these nutritional considerations directly.
Hair loss is a concern some people raise with rapid weight loss from any of these medications. The GLP-1 hair loss guide covers this comprehensively, and whether retatrutide causes hair loss specifically addresses the triple agonist.
The peptide calculator and peptide stack calculator at SeekPeptides are free tools that help optimize dosing across multiple peptides for those exploring broader protocols. Understanding whether these medications interact with other peptides for fat loss, peptides for muscle growth, or peptides for anti-aging helps build comprehensive protocols.
Special considerations for different populations
Not everyone responds to these medications the same way. Age, sex, starting weight, and existing medical conditions all influence which medication works best and which side effects are most likely to appear.
Women and reproductive considerations
Women make up the majority of GLP-1 medication users, and a few considerations are unique to this population. All three medications can affect menstrual cycles. Rapid weight loss changes estrogen levels, and the hormonal shifts that accompany significant fat loss can temporarily alter cycle timing, flow, and symptoms. Understanding how semaglutide affects your period and how tirzepatide affects your period helps set expectations for these changes.
Pregnancy is a critical concern. None of these medications should be used during pregnancy or breastfeeding. Semaglutide and tirzepatide carry warnings to discontinue at least two months before planned conception. GLP-1 medications can increase fertility by restoring ovulation in women with obesity-related anovulation, which means unplanned pregnancies can happen. The discussion around getting pregnant on tirzepatide and the guidance on GLP-1 use during breastfeeding address these important safety topics. Similar concerns apply to tirzepatide during breastfeeding specifically.
People with significant weight to lose
For individuals starting at a higher body weight, say 300 pounds or more, the absolute pounds lost with each medication become dramatic. At 300 pounds, the difference between semaglutide (approximately 45 pounds) and retatrutide (approximately 86 pounds) is 41 pounds. That gap is essentially the difference between remaining in the obese BMI category and potentially reaching the overweight category.
Higher starting weights also tend to produce better percentage-based outcomes. Clinical trial subgroup analyses consistently show that participants with a BMI of 35 or higher lose a larger percentage of body weight than those with lower BMIs. The SURMOUNT-1 trial specifically noted that participants with a BMI of 35 or higher who received the 15mg tirzepatide dose lost an average of 26.5% of their body weight, substantially higher than the overall trial average.
People with existing medication combinations
Many people considering these medications are already taking other drugs for blood pressure, cholesterol, diabetes, or mental health conditions. Drug interactions with GLP-1 agonists are relatively uncommon, but the slowed gastric emptying can affect absorption rates of oral medications taken at the same time.
The question of combining GLP-1 medications with other weight management approaches comes up frequently. Whether phentermine and semaglutide can be combined or phentermine and tirzepatide taken together are among the most searched questions in this space. Some people also explore combining GLP-1 medications with other peptides like AOD-9604 alongside tirzepatide for enhanced fat loss, though data on such combinations is limited.
Surgical considerations
GLP-1 medications require careful management around any surgical procedure. The slowed gastric emptying they cause increases aspiration risk during anesthesia. Most surgical guidelines now recommend stopping these medications at least one to two weeks before elective surgery. Understanding when to resume semaglutide after surgery helps plan the post-operative timeline. The same principles apply to tirzepatide and retatrutide, though specific guidelines for retatrutide will not exist until it reaches the market.
Understanding the BMI and eligibility question
Not everyone qualifies for these medications through traditional prescribing channels. FDA-approved weight loss medications (semaglutide and tirzepatide) require either a BMI of 30 or higher, or a BMI of 27 or higher with at least one weight-related comorbidity such as type 2 diabetes, hypertension, or dyslipidemia.
The BMI requirements for GLP-1 medications article breaks down the specific thresholds and explains how comorbidities affect eligibility. Understanding how long you stay on semaglutide also matters because these are increasingly understood as long-term or lifelong medications, not short-term treatments.
For those exploring whether microdosing GLP-1 medications for autoimmune conditions has merit, some emerging research suggests anti-inflammatory benefits at lower doses, though this remains investigational.
Long-term considerations and staying on treatment
What happens when you stop taking these medications? The data is consistent across all three: weight regain occurs in most people who discontinue treatment. The STEP 1 extension study showed that participants who stopped semaglutide regained approximately two-thirds of their lost weight within a year. Similar patterns are expected with tirzepatide and retatrutide, though long-term discontinuation data for retatrutide is not yet available.
This reality shapes how you should think about these medications. They are not temporary interventions. They are ongoing treatments for a chronic condition. Choosing the right one involves thinking not just about initial weight loss but about long-term tolerability, cost sustainability, and availability.
Cost becomes the dominant factor in long-term planning. A medication that costs $400 per month is manageable for many people indefinitely. One that costs $1,600 per month without insurance coverage is not. This financial reality is why compounded options have gained so much traction. If you plan to stay on treatment for years, and the data increasingly suggests you should, then the monthly expense matters as much as the clinical trial data.
The other long-term consideration is dose stability. Some people find they can reduce their dose over time while maintaining their weight loss, which lowers both cost and side effect burden. Others need their full maintenance dose indefinitely. The variability is considerable, and there is currently no reliable way to predict which category you will fall into before starting treatment. Starting with a microdose approach and finding the lowest effective dose is one strategy for long-term sustainability.
Weight regain after discontinuation follows a predictable pattern. Hunger signals return within one to two weeks. Weight begins creeping back within the first month. By six months, most people have regained a measurable portion of their lost weight, and by one year, the regain is typically substantial. This pattern holds regardless of whether the person exercises or follows a strict diet after stopping the medication, suggesting that these medications alter biological set points rather than simply reducing appetite.
The implications are straightforward. Choose a medication you can afford, tolerate, and access for the long term. Semaglutide wins on track record and availability. Tirzepatide wins on efficacy among currently available options. Retatrutide, when it arrives, may win on raw weight loss numbers. But the best medication is the one you actually take consistently, month after month, without financial strain or intolerable side effects derailing your progress.
Resources on how quickly GLP-1 medications start working and guides on managing expectations during the first weeks, such as one-month semaglutide results and men-specific tirzepatide results, help set a realistic long-term perspective.
For researchers serious about optimizing their weight management protocols, SeekPeptides offers comprehensive guides, dosing calculators, and a community of experienced members who have navigated these exact decisions. Whether you need a detailed compounded dosage calculator or want to explore the broader landscape of injectable versus oral peptide options, the tools and resources make complex decisions simpler.
Frequently asked questions
Is retatrutide better than tirzepatide for weight loss?
Based on clinical trial data, retatrutide produces greater average weight loss (28.7% at 68 weeks with 12mg) compared to tirzepatide (22.5% at 72 weeks with 15mg). However, retatrutide is not yet FDA-approved and is only available through clinical trials or research channels. Tirzepatide remains the most effective FDA-approved option. See the full retatrutide comparison guide for detailed data.
Can I switch from semaglutide to tirzepatide mid-treatment?
Yes. No washout period is required. Most providers recommend starting tirzepatide the week after your last semaglutide injection at a dose that accounts for your current semaglutide level. The conversion chart provides specific dosing guidance for this transition.
Which medication has the fewest side effects?
Tirzepatide shows the lowest discontinuation rates due to adverse events in clinical trials (4-7%), followed closely by semaglutide (approximately 7%). Retatrutide has a higher overall adverse event burden (6-10% discontinuation), partly because of the unique dysesthesia side effect at higher doses. For a complete side-by-side analysis, review the semaglutide versus tirzepatide side effects comparison.
When will retatrutide be FDA-approved?
The most likely timeline points to a potential FDA approval in late the next year, with commercial availability following shortly after. Eli Lilly has multiple Phase 3 TRIUMPH trials expected to complete throughout the current year. However, regulatory timelines frequently shift due to additional data requests, safety reviews, or manufacturing considerations. Track the latest updates in the retatrutide availability guide.
Do I need a prescription for any of these medications?
Semaglutide (Wegovy, Ozempic) and tirzepatide (Mounjaro, Zepbound) require a prescription. Compounded versions of both are available through telehealth providers and compounding pharmacies with a prescription. Retatrutide is not FDA-approved and cannot be obtained through standard prescriptions. The compounded semaglutide guide explains the pharmacy options in detail.
Which is better for someone with type 2 diabetes?
Tirzepatide produced greater A1c reductions than semaglutide in the SURPASS-2 head-to-head trial, making it the stronger option for glycemic control. However, semaglutide has dedicated cardiovascular outcome data from the SELECT trial showing a 20% reduction in major adverse cardiac events. The choice depends on whether glucose control or cardiovascular risk reduction is the higher priority for your specific situation.
How do the costs compare?
Brand-name semaglutide costs approximately $1,300-$1,900 per month. Brand-name tirzepatide costs approximately $1,400-$1,600 per month. Compounded versions of both range from $100-$500 per month. Retatrutide has no established retail price since it is not yet approved. Use the peptide cost calculator to compare specific formulations and sources.
Can I combine any of these medications?
No. Semaglutide, tirzepatide, and retatrutide should not be combined with each other. They all activate GLP-1 receptors, and using two GLP-1 agonists simultaneously would increase side effect risk without proportional benefit. If one medication is not producing adequate results, switching to another is preferable to stacking them.
Will I regain weight if I stop taking these medications?
The evidence is consistent across all three: most people regain a significant portion of lost weight after discontinuation. The STEP 1 extension study showed approximately two-thirds of weight was regained within one year of stopping semaglutide. This reflects the chronic nature of obesity as a disease, not a failure of the medication. Most experts now recommend viewing these medications as long-term treatments rather than temporary interventions. Maintaining results requires either continued medication use or significant, sustained lifestyle changes.
Does retatrutide cause more nausea than semaglutide or tirzepatide?
At the highest doses, retatrutide GI side effect rates are modestly higher than the other two medications. Nausea affects approximately 16-25% of retatrutide participants depending on dose, compared to roughly 18% for semaglutide and 17-22% for tirzepatide. The difference is not dramatic for GI symptoms specifically. What sets retatrutide apart is the dysesthesia side effect, which does not meaningfully occur with the other two medications.
Which one works fastest?
All three medications begin suppressing appetite within the first one to two weeks. However, the rate of weight loss differs. Retatrutide appears to produce the fastest weight loss trajectory, with participants in Phase 2 trials losing 24.2% of body weight in just 48 weeks, a pace that outstrips semaglutide and tirzepatide over comparable timeframes. For early timeline expectations, the semaglutide speed guide and the tirzepatide speed guide provide week-by-week benchmarks.
Can I use these medications if I do not have diabetes?
Yes. Both semaglutide (as Wegovy) and tirzepatide (as Zepbound) are specifically FDA-approved for weight management in people without diabetes who meet BMI criteria. In fact, the largest weight loss trials for all three medications were conducted in people without diabetes. Having diabetes is not a prerequisite.
External resources
New England Journal of Medicine: Retatrutide Phase 2 trial results
PMC meta-analysis: Comparative efficacy of GLP-1 receptor agonists
Nature Medicine: Retatrutide for MASLD randomized Phase 2a trial
Eli Lilly: TRIUMPH-4 Phase 3 retatrutide results press release
New England Journal of Medicine: Tirzepatide versus semaglutide head-to-head trial
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your titrations stay smooth, and your results stay consistent.