Feb 21, 2026

Your joints ache before you even get out of bed. The stiffness lasts an hour, sometimes two. Medications keep things manageable, but manageable is not the same as good, and you know the difference. Then someone mentions a weight loss drug, of all things, that made their joint pain disappear. Not a little better. Gone. You are skeptical. You should be. But the reports keep coming, from rheumatologists, from immunologists, from people who have tried everything and finally found something that shifted the needle on inflammation they could not control for years.
This is the story behind microdosing GLP-1 receptor agonists for autoimmune conditions. Not the full weight loss dose. Not the diabetes protocol. A fraction of that, sometimes one-tenth, targeted at the inflammatory machinery that drives autoimmune disease from the inside out. The idea is not new, not exactly. GLP-1 receptors exist on immune cells, not just in the pancreas or the gut. Researchers have known this for over a decade. What is new is the growing pile of evidence suggesting that activating those receptors, even at very low doses, can quiet the overactive immune response that defines conditions like rheumatoid arthritis, psoriasis, Hashimoto thyroiditis, and inflammatory bowel disease.
This guide breaks down every piece of current evidence, from the molecular mechanisms to the clinical observations, from the specific doses practitioners are using to the conditions showing the most promise.
Whether you are a researcher exploring new territory, a patient frustrated by partial relief, or a clinician curious about what your colleagues are seeing, every section that follows is built on what the science actually says right now, not what we hope it will say later. SeekPeptides has assembled the most comprehensive breakdown available on this emerging intersection of metabolic and immune health.
What microdosing GLP-1 actually means
The term microdosing gets thrown around loosely. In clinical pharmacology, it has a specific meaning. For GLP-1 receptor agonists, microdosing means using doses significantly below the standard therapeutic range, typically one-fifth to one-tenth of what is prescribed for weight loss or diabetes management.
Standard semaglutide dosing for weight loss starts at 0.25 mg per week and escalates to 2.4 mg. Standard tirzepatide starts at 2.5 mg and climbs to 15 mg. Microdosing protocols, by contrast, operate in a completely different range.
For semaglutide, microdoses typically fall between 0.05 mg and 0.1 mg per week. Some practitioners start as low as 0.025 mg. For tirzepatide, the equivalent range sits around 0.5 mg to 1.25 mg weekly. These are not weight loss doses. The appetite suppression effect is minimal or absent at these levels. The goal is different entirely.
At these concentrations, the GLP-1 receptor activation is sufficient to engage anti-inflammatory pathways without triggering the gastrointestinal side effects that make full-dose therapy difficult for many patients. No nausea. No constipation. No burping. Just targeted receptor activation where it matters most for immune modulation.
There is an important distinction here. This is not a dilution strategy. It is not about taking less of the same thing and hoping for the same result. The anti-inflammatory effects of GLP-1 receptor agonists appear to operate through pathways that are activated at lower receptor occupancy levels than those required for metabolic effects. In simpler terms, the immune system responds to less drug than the metabolic system needs.
How microdosing differs from standard GLP-1 therapy
Standard semaglutide dosing for obesity targets sustained appetite suppression and metabolic rate changes. These effects require consistent receptor activation above a threshold that typically corresponds to 1.0 mg or higher weekly doses. The timeline for weight loss effects reflects this, with meaningful appetite changes emerging at 4 to 8 weeks of escalating doses.
Microdosing skips this escalation entirely. The dose stays low. Stays there on purpose.
The anti-inflammatory benefit appears to operate independently of weight loss. This matters enormously for autoimmune patients, many of whom are not overweight and would not qualify for standard GLP-1 prescriptions. It also matters because the side effect profile at microdoses is dramatically different. Research from the STEP trials showed that GLP-1 side effects increase in a dose-dependent manner. At one-tenth the therapeutic dose, most patients report zero gastrointestinal symptoms.
The dosing landscape right now
No standardized microdosing protocol exists yet. This is important to acknowledge. What does exist is a growing number of practitioners, primarily in functional medicine and integrative rheumatology, who are documenting their approaches and tracking outcomes.
The most common protocols involve:
Semaglutide microdose protocol: Starting at 0.05 mg weekly, holding for 4 weeks, then reassessing inflammatory markers before considering any adjustment. Maximum microdose ceiling typically set at 0.25 mg, which is the standard starting dose for weight loss protocols.
Tirzepatide microdose protocol: Starting at 0.5 mg weekly, holding for 4 to 6 weeks, with similar marker-based reassessment. The compounded tirzepatide starting dose for this application often requires specialized compounding pharmacies that can prepare lower concentrations.
These protocols are based on clinical observation, not randomized controlled trials. That distinction matters. But one active clinical trial, registered as NCT07092605 on ClinicalTrials.gov, is now specifically studying the effectiveness of microdosed GLP-1 receptor agonists for improving health and quality of life in patients who do not meet traditional prescribing criteria. Results will provide the first controlled data on this approach.
The science behind GLP-1 and immune function
GLP-1 receptors are everywhere. Not just the pancreas. Not just the gut.
They sit on the surface of macrophages, those immune cells responsible for detecting threats and coordinating inflammatory responses. They are found on T cells, the adaptive immune system warriors that drive autoimmune tissue destruction when they malfunction. They appear on dendritic cells, the scouts of the immune system. Even brain microglia, the immune cells of the central nervous system, express GLP-1 receptors.
This distribution tells a story. GLP-1 signaling is not just about blood sugar. It is a broad regulatory signal that touches virtually every branch of the immune system.
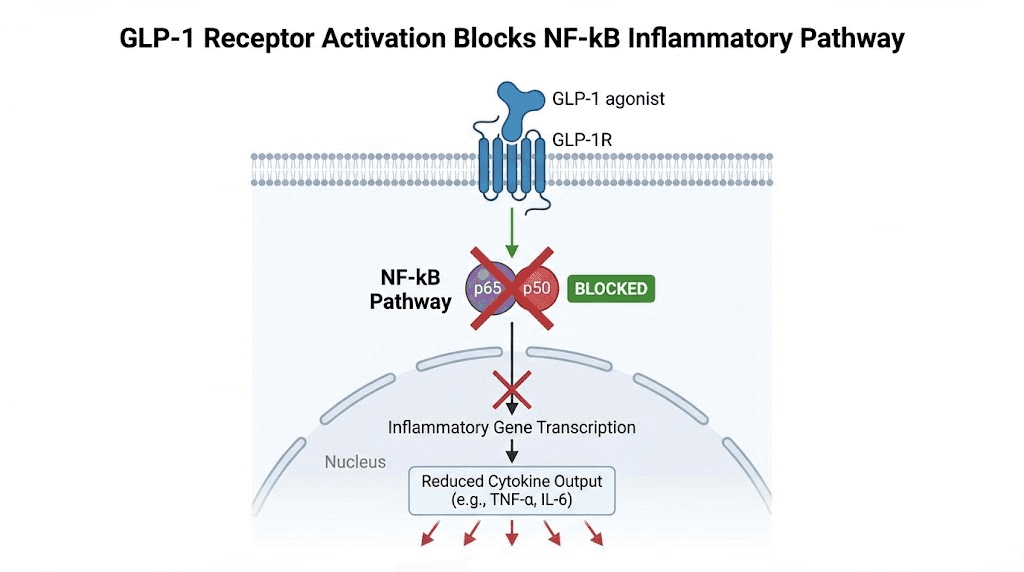
The NF-kB pathway: ground zero for autoimmune inflammation
Nuclear factor kappa B, or NF-kB, is the master switch for inflammatory gene expression. When activated, it drives production of TNF-alpha, IL-6, IL-1 beta, and dozens of other inflammatory mediators. In autoimmune disease, NF-kB is chronically overactivated. The switch gets stuck in the on position.
GLP-1 receptor agonists inhibit NF-kB activation. This has been demonstrated in human peripheral blood mononuclear cells, where exenatide blocked NF-kB binding to DNA and downregulated both TNF and IL-1 beta expression. In mouse microglia, a GLP-1 receptor agonist called NLY01 blocked nuclear translocation of NF-kB entirely when cells were stimulated with bacterial endotoxin.
This is not a subtle effect. It is a direct brake on the central inflammatory cascade that drives autoimmune damage.
Cytokine suppression: the numbers
The specific cytokines reduced by GLP-1 receptor activation read like a hit list of autoimmune mediators:
TNF-alpha drives joint destruction in rheumatoid arthritis, skin plaque formation in psoriasis, and intestinal inflammation in Crohn disease. GLP-1 receptor agonists reduce TNF-alpha production in multiple tissue types, including immune cells, cardiac tissue, and brain tissue.
IL-6 orchestrates the acute phase response and drives chronic inflammation across nearly every autoimmune condition. In a study of 40 men with type 2 diabetes, 1 mg of semaglutide per week for 6 months significantly reduced circulating IL-6 levels.
IL-1 beta amplifies inflammatory cascades and contributes to tissue destruction. GLP-1 receptor activation suppresses IL-1 beta through multiple pathways, including direct NF-kB inhibition and reduced inflammasome activation.
CRP (C-reactive protein) serves as the most commonly measured marker of systemic inflammation. Data from the STEP clinical trials showed semaglutide 2.4 mg reduced CRP by 44% in STEP 1, 39% in STEP 2, and 48% in STEP 3 compared to placebo. The SELECT trial demonstrated a 38% CRP reduction regardless of baseline BMI, statin use, or cholesterol levels. Even at lower doses, semaglutide 1.0 mg achieved a 42% CRP reduction in STEP 2.
Critically, researchers found the CRP reduction was only partially mediated by weight loss. There appears to be a direct anti-inflammatory effect independent of metabolic changes. This supports the rationale for microdosing, where weight loss does not occur but anti-inflammatory effects may still be present.
Regulatory T cells: restoring balance
Autoimmune disease fundamentally involves a loss of immune tolerance. The regulatory T cells, or Tregs, that normally keep the immune system from attacking the body's own tissues either decline in number or lose function. The attack proceeds unchecked.
GLP-1 receptor agonists enhance regulatory T cell function. This has been documented in multiple research models, where GLP-1 signaling promoted Treg differentiation and suppressive activity. At the same time, GLP-1 receptor activation reduces the activity of pathogenic Th1 and Th17 cells, the effector T cells most directly responsible for autoimmune tissue damage.
Think of it this way. The immune system has an accelerator and a brake. In autoimmune disease, the accelerator is stuck and the brake is weak. GLP-1 receptor activation appears to release the accelerator (reducing inflammatory effector cells) while strengthening the brake (enhancing regulatory cells). Both effects simultaneously.
Macrophage polarization
Macrophages exist on a spectrum. M1 macrophages are pro-inflammatory, releasing cytokines that drive tissue destruction. M2 macrophages are anti-inflammatory, promoting tissue repair and resolution of inflammation. In autoimmune disease, the balance tilts heavily toward M1.
GLP-1 receptor agonists shift macrophage polarization toward the M2 phenotype. This reduces the inflammatory burden in affected tissues while simultaneously promoting repair. In atherosclerotic plaque models, semaglutide reduced macrophage infiltration and suppressed the M1 inflammatory program. Similar effects have been observed in liver tissue, kidney tissue, and brain tissue.
The macrophage shift is one reason why GLP-1 receptor agonists show benefit across such diverse autoimmune conditions. Macrophages are involved in virtually every autoimmune process, regardless of the specific tissue being attacked.
Which autoimmune conditions respond to GLP-1 therapy
Not every autoimmune condition has equal evidence. Some have preliminary clinical data. Others have case reports. A few have only mechanistic rationale. Here is what the evidence currently supports, condition by condition, with an honest assessment of where the data stands.
Rheumatoid arthritis
This is where the strongest clinical evidence exists right now.
Research presented at the ACR Convergence 2025 conference showed that RA patients with elevated BMI who took GLP-1 receptor agonists experienced superior improvements in disease activity scores, pain levels, and weight loss compared to controls. More significantly, RA patients already on DMARDs (disease-modifying antirheumatic drugs) who added a GLP-1 agonist had fewer disease flares than those on DMARDs alone.
That second finding is crucial. It suggests an additive anti-inflammatory effect beyond what standard RA medications achieve.
A separate analysis found that semaglutide users had significantly lower 30-day, 3-month, and 1-year risk of joint-related outcomes, including stiffness, pain, swelling, and synovitis, compared to nonusers. This was observed even after controlling for weight loss, suggesting a direct joint-protective effect.
Large-scale observational data adds another layer. GLP-1 receptor agonist use is associated with a reduced risk of developing rheumatoid arthritis in the first place. This prevention signal, if confirmed in prospective studies, would have enormous implications for high-risk populations.
For microdosing specifically, clinical practitioners report that patients with RA who cannot tolerate or do not qualify for full-dose GLP-1 therapy are seeing meaningful reductions in morning stiffness, flare frequency, and body aches at microdose levels. These are not published studies. They are clinical observations being documented and shared among practitioners. But the consistency of these reports deserves attention.
Psoriasis and psoriatic arthritis
The evidence here comes from multiple directions.
Clinical studies show that GLP-1 receptor agonists improve psoriasis severity scores regardless of whether the patient has diabetes. This is important because it separates the skin benefit from the metabolic benefit. One mechanism involves regulation of invariant natural killer T cells (iNKT cells) in psoriatic plaques, plus AMPK activation that reduces keratinocyte proliferation.
For psoriatic arthritis specifically, patients show improvements in both skin involvement and joint inflammation on GLP-1 therapy. The muscle and joint pain that characterizes psoriatic arthritis appears responsive to the anti-inflammatory effects.
Research from ACR Convergence 2025 highlighted GLP-1 receptor agonists as showing particular promise in psoriatic arthritis management, where the dual burden of skin and joint disease creates challenges for conventional therapies.
Hashimoto thyroiditis
Hashimoto thyroiditis, the most common autoimmune condition globally, involves immune-mediated destruction of thyroid tissue. The evidence for GLP-1 therapy here is earlier stage but intriguing.
An important study published in the Annals of Clinical Endocrinology and Metabolism found that patients with Hashimoto thyroiditis have altered GLP-1 and GLP-2 serum levels compared to healthy controls. This suggests a potential role for GLP-1 signaling in thyroid autoimmunity.
Clinical observations from practitioners using microdosed GLP-1 in Hashimoto patients report improvements in thyroid antibody levels (TPO and thyroglobulin antibodies) and reductions in thyroid-related fatigue. These are not published trials. The findings have not been replicated in robust controlled studies. But the mechanistic rationale is sound, since the NF-kB and cytokine pathways that GLP-1 modulates are directly involved in Hashimoto pathogenesis.
One concern exists. GLP-1 receptor agonists carry a boxed warning about thyroid C-cell tumors based on animal studies with liraglutide. Patients with thyroid conditions should discuss this thoroughly with their healthcare provider before considering any GLP-1 therapy, including microdosing.
Inflammatory bowel disease
Crohn disease and ulcerative colitis involve immune-mediated destruction of the intestinal lining. GLP-1 receptors are densely expressed in gut tissue, making this a logical target.
The anti-inflammatory mechanisms, including NF-kB suppression, TNF-alpha reduction, and macrophage polarization, are directly relevant to IBD pathology. Animal models show intestinal protection from GLP-1 receptor agonists.
However, this is also where caution is warranted. One real-world evidence study found that patients receiving GLP-1 receptor agonists had a higher risk of ulcerative colitis compared to those on DPP-4 inhibitors (a different diabetes drug class). The causality is unclear, and confounding factors are significant, but it raises a flag that needs further investigation.
For patients with established IBD, the gastrointestinal side effects of full-dose GLP-1 therapy can be problematic. This is actually one argument in favor of microdosing, where gastrointestinal effects are minimized while anti-inflammatory receptor activation may still occur. But this remains theoretical until clinical data emerges.
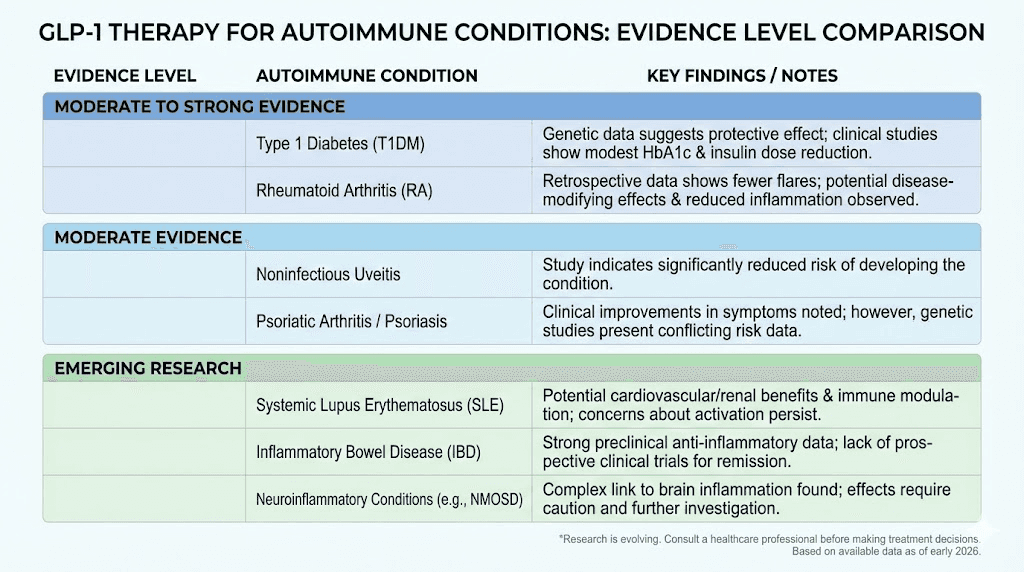
Multiple sclerosis
GLP-1 receptor agonists show neuroprotective properties that extend to autoimmune conditions of the central nervous system. In multiple sclerosis models, GLP-1 receptor activation reduces pathogenic Th1 and Th17 cell activity, inhibits microglial activation (the brain equivalent of macrophage inflammation), and may enhance remyelination processes.
A T-cell-dependent autoimmune model demonstrated that liraglutide, an earlier GLP-1 receptor agonist, inhibited T-cell proliferation and reduced disease severity. This suggests a direct effect on the adaptive immune response driving MS.
The evidence here is largely preclinical. No published clinical trials have tested GLP-1 microdosing specifically in MS patients. But the mechanistic data is compelling enough that several research groups are exploring this avenue.
Systemic lupus erythematosus
Lupus is one of the more complex autoimmune conditions, involving multiple organ systems and antibody-mediated damage. The evidence for GLP-1 therapy in lupus is limited.
Observational studies did not find a significant difference in lupus risk between GLP-1 receptor agonist users and non-users. This neither supports nor refutes potential benefit. Mechanistically, the NF-kB and cytokine effects should theoretically help, but lupus pathogenesis involves pathways (particularly type I interferon and complement activation) that may not be as responsive to GLP-1 signaling.
This is an honest knowledge gap. We do not have enough data to make strong claims either way.
Other conditions showing signals
Emerging research suggests potential benefit in gout (reduced risk of development in GLP-1 users), osteoarthritis (improved joint outcomes in semaglutide users), and endometriosis (anti-inflammatory mechanisms relevant to endometrial inflammation). Each of these has limited but suggestive data.
Microdosing protocols: what practitioners are actually using
No FDA-approved indication exists for GLP-1 microdosing in autoimmune disease. No professional medical society endorses it. No standardized dosing guideline has been published.
With those caveats stated clearly, here is what practitioners who are using this approach are documenting.
Protocol 1: semaglutide microdose for autoimmune inflammation
Goal: Reduce systemic inflammation and autoimmune flare frequency
Starting dose: 0.05 mg semaglutide subcutaneously, once weekly
Week 1 to 4: Hold at 0.05 mg. Monitor for any GI symptoms (rare at this dose). Track subjective symptoms, morning stiffness duration, pain levels, and energy.
Week 5 reassessment: Check inflammatory markers (CRP, ESR, relevant condition-specific markers). If symptoms improved and markers trended down, continue at current dose. If no change, consider increasing to 0.1 mg weekly.
Maximum microdose ceiling: 0.25 mg weekly (which is the standard lowest therapeutic dose for weight loss, so anything at or below this is considered a microdose)
Duration: Minimum 12 weeks before assessing overall efficacy. Some practitioners report that anti-inflammatory effects become more pronounced at weeks 8 to 12 compared to weeks 1 to 4.
Injection guidance: Standard subcutaneous technique applies. The best injection site for semaglutide remains the abdomen, though thigh and upper arm work too. Rotate sites per usual GLP-1 injection protocols.
One challenge is access to appropriate concentrations. Standard semaglutide pens deliver a minimum of 0.25 mg per dose. Achieving 0.05 mg requires either compounded semaglutide at a lower concentration or careful insulin syringe dosing from multi-dose vials. The syringe injection technique allows for more precise low-dose administration than pen devices.
Protocol 2: tirzepatide microdose for autoimmune inflammation
Goal: Leverage dual GIP/GLP-1 receptor activation for broader anti-inflammatory coverage
Starting dose: 0.5 mg tirzepatide subcutaneously, once weekly
Week 1 to 6: Hold at 0.5 mg. Tirzepatide has a longer pharmacokinetic profile, so a longer initial assessment window is appropriate.
Week 7 reassessment: Same marker and symptom assessment as semaglutide protocol.
Maximum microdose ceiling: 1.25 mg weekly
Tirzepatide brings an interesting additional dimension. Research shows that tirzepatide does not rely exclusively on neuronal GLP-1 receptors for its anti-inflammatory effects. In mouse models where semaglutide lost its acute anti-inflammatory effects when neuronal GLP-1 receptors were knocked out, tirzepatide still effectively prevented TNF-alpha induction. This suggests tirzepatide may engage anti-inflammatory pathways through its GIP receptor component as well, potentially providing broader immune modulation. The tirzepatide dosage calculator can help determine appropriate volumes for non-standard doses.

Monitoring markers
Practitioners using these protocols typically track several categories of markers to assess response.
General inflammatory markers:
High-sensitivity CRP (hs-CRP), the most responsive general marker
Erythrocyte sedimentation rate (ESR)
Fibrinogen
Condition-specific markers:
Rheumatoid arthritis: RF, anti-CCP, DAS28 score
Hashimoto thyroiditis: TPO antibodies, thyroglobulin antibodies, TSH
Psoriasis: PASI score, BSA involvement
IBD: Fecal calprotectin, SES-CD or Mayo score
Lupus: ANA, anti-dsDNA, complement C3/C4
Cytokine panels (when available):
TNF-alpha
IL-6
IL-1 beta
IL-17 (particularly relevant for psoriasis and spondyloarthritis)
The recommendation from most practitioners is baseline testing before starting, a 4 to 6 week interim check, and a full reassessment at 12 weeks. Changes in hs-CRP tend to be the most reliable early indicator of response.
The case report that started conversations
One case report deserves specific attention because it illustrates the potential.
A 37-year-old patient with primary Sjogren syndrome, comorbid Hashimoto thyroiditis, and seronegative rheumatoid arthritis was started on semaglutide. Before treatment, she experienced multi-week flares that significantly impacted quality of life. After starting semaglutide, her flare episodes decreased from lasting multiple weeks to approximately a single day.
Read that again. Multi-week flares reduced to single-day episodes.
This is one patient. One case report. It does not prove anything in isolation. But it was published in a peer-reviewed journal specifically because the dramatic clinical improvement demanded documentation. Multiple autoimmune conditions responding simultaneously to a single intervention is unusual in rheumatology, where treatment typically requires different medications targeting different pathways.
The case raises a question that researchers are now actively investigating. If GLP-1 receptor activation can produce this level of improvement in a patient with three overlapping autoimmune conditions, what happens when we study this systematically?
Semaglutide vs tirzepatide for autoimmune applications
Both molecules activate GLP-1 receptors. But they are not identical, and the differences matter for autoimmune applications.
Semaglutide is a pure GLP-1 receptor agonist. Its anti-inflammatory mechanisms operate entirely through GLP-1R activation. It has the most published data on CRP reduction and inflammatory marker improvement. The STEP and SUSTAIN trial data provide robust evidence of systemic anti-inflammatory effects. Semaglutide onset of action for metabolic effects is well documented, though anti-inflammatory timelines may differ.
Tirzepatide activates both GLP-1 and GIP receptors. This dual mechanism is significant because, as mentioned earlier, tirzepatide maintained anti-inflammatory effects even when neuronal GLP-1 receptors were knocked out in animal models. This means tirzepatide has anti-inflammatory pathways that semaglutide does not, operating through GIP receptor signaling. Tirzepatide onset may also differ from semaglutide in immune-relevant contexts.
Factor | Semaglutide | Tirzepatide |
|---|---|---|
Receptor targets | GLP-1 only | GLP-1 + GIP |
Anti-inflammatory pathways | NF-kB, cytokine suppression via GLP-1R | NF-kB, cytokine suppression via GLP-1R and GIP-R |
Published CRP data | Extensive (STEP, SUSTAIN, SELECT, PIONEER) | Growing (SURMOUNT trials) |
Neuronal GLP-1R dependence | Yes (loses effect without neuronal GLP-1R) | No (retains effect without neuronal GLP-1R) |
Microdose range | 0.05 to 0.25 mg weekly | 0.5 to 1.25 mg weekly |
Compounded availability | Widely available | Widely available |
GI side effects at microdose | Minimal | Minimal |
The choice between them for autoimmune applications is not settled. Semaglutide has more published anti-inflammatory data. Tirzepatide may have broader anti-inflammatory mechanisms. Some practitioners are using them sequentially, starting with semaglutide and switching to tirzepatide if response is suboptimal, similar to how switching between GLP-1 agents works in metabolic contexts.
How GLP-1 microdosing fits with existing autoimmune treatments
This is not a replacement therapy. It is important to be direct about that.
GLP-1 microdosing, based on current evidence, functions as a complementary approach, not a substitute for standard immunotherapies. DMARDs, biologics, corticosteroids, and other established treatments remain the backbone of autoimmune disease management. GLP-1 microdosing occupies the space of adjunctive therapy, something added alongside standard treatment to improve outcomes.
Compatibility with common autoimmune medications
The ACR Convergence 2025 data specifically showed benefit when GLP-1 agonists were added to existing DMARD therapy in RA patients. This suggests additive rather than competing mechanisms. The anti-inflammatory pathways modulated by GLP-1 receptor activation (NF-kB suppression, macrophage polarization, Treg enhancement) are largely distinct from the pathways targeted by:
Methotrexate: Folate metabolism disruption, different anti-inflammatory pathway
TNF inhibitors (adalimumab, etanercept): Direct TNF blockade, whereas GLP-1 reduces TNF production upstream
IL-6 inhibitors (tocilizumab): Receptor-level blockade, while GLP-1 reduces IL-6 production
JAK inhibitors (tofacitinib, baricitinib): Different signaling pathway entirely
Corticosteroids: Broad immunosuppression, while GLP-1 is immunomodulatory
This pathway complementarity is why additive benefit is biologically plausible. You are not doubling up on the same mechanism. You are addressing inflammation from a completely different angle.
One theoretical advantage of the GLP-1 approach is that it modulates rather than suppresses the immune system. Standard immunosuppressants reduce overall immune function, increasing infection risk. GLP-1 receptor agonists appear to shift immune activity from inflammatory to regulatory without compromising protective immunity. Research specifically notes that semaglutide does not appear to compromise immune defense against infections.
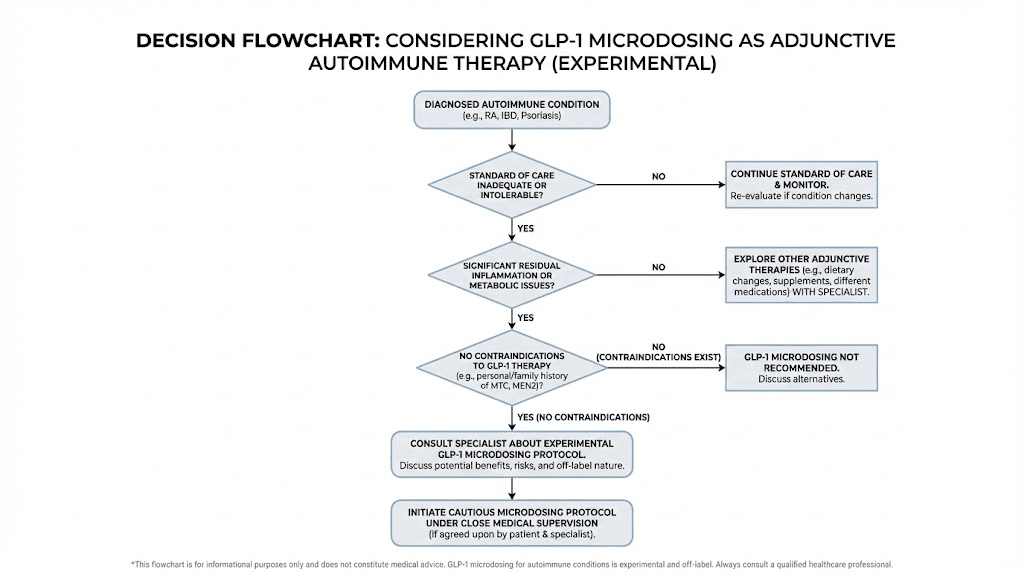
When to consider GLP-1 microdosing
Based on current clinical practice patterns, practitioners are most commonly recommending GLP-1 microdosing in these scenarios:
Partial responders: Patients on standard therapy who have improved but still experience breakthrough flares, residual inflammation, or persistent symptoms. Adding microdosed GLP-1 aims to address the remaining inflammatory burden through complementary pathways.
Side effect-limited patients: Those who cannot tolerate higher doses of standard immunosuppressants and need additional inflammatory control without additional immunosuppression.
Patients with metabolic comorbidities: Autoimmune patients with concurrent insulin resistance, metabolic syndrome, or cardiovascular risk factors benefit doubly, since the GLP-1 addresses both the autoimmune inflammation and the metabolic burden.
Prevention candidates: Given the observational data suggesting reduced autoimmune disease risk in GLP-1 users, some practitioners are considering microdosing for patients with strong family histories or early biomarker evidence of autoimmune activation before clinical disease manifests.
Side effects and safety at microdose levels
The side effect profile at microdose levels is fundamentally different from full-dose therapy. This is one of the primary arguments for the microdosing approach in autoimmune patients.
What you will probably not experience
At doses of 0.05 to 0.25 mg semaglutide weekly, the gastrointestinal effects that dominate full-dose therapy are largely absent. Constipation, nausea, burping, and appetite suppression are dose-dependent effects that require higher receptor occupancy than microdoses provide.
Most practitioners report that their microdosing patients experience zero gastrointestinal side effects. Some report mild, transient nausea in the first 1 to 2 weeks that resolves spontaneously. Digestive discomfort is rarely a concern at these levels.
Energy changes at microdose levels tend to be neutral or mildly positive, likely related to reduced systemic inflammation rather than direct metabolic effects. GLP-1 fatigue is primarily a higher-dose phenomenon.
What you should monitor
Thyroid considerations: GLP-1 receptor agonists carry a boxed warning regarding thyroid C-cell tumors based on rodent studies with liraglutide. While this has not been observed in humans at standard doses, and microdoses are far below standard doses, patients with existing thyroid conditions (including Hashimoto) should have regular thyroid monitoring. TSH, free T4, and thyroid ultrasound at baseline and at 6-month intervals is a reasonable monitoring schedule.
Pancreatic considerations: GLP-1 agonists increase pancreatic enzyme output. While pancreatitis risk at microdose levels is theoretical rather than documented, monitoring lipase levels at baseline and periodically is prudent, especially in patients with any history of pancreatic issues.
Gallbladder considerations: Full-dose GLP-1 therapy is associated with increased gallbladder events. At microdose levels, this risk is likely minimal, but patients with existing gallbladder disease should be monitored.
Injection site reactions: Mild redness or swelling at the injection site can occur regardless of dose. Standard site rotation protocols apply. Proper injection technique minimizes this risk.
Interactions with autoimmune medications
No significant drug interactions have been identified between GLP-1 receptor agonists and common autoimmune medications, including methotrexate, hydroxychloroquine, sulfasalazine, leflunomide, or biologic DMARDs.
The one consideration is timing. GLP-1 receptor agonists slow gastric emptying (though less so at microdoses). Medications that require specific absorption timing, particularly oral medications with narrow therapeutic windows, may have altered absorption kinetics. Timing the GLP-1 injection appropriately relative to oral medications is a practical consideration.
The inflammation-autoimmune connection most people miss
Here is something that changes how you think about autoimmune disease and GLP-1 therapy.
Most autoimmune patients have subclinical metabolic inflammation that exists independently of their autoimmune condition but amplifies it. Insulin resistance, visceral adiposity, gut permeability, and metabolic endotoxemia create a baseline inflammatory state that lowers the threshold for autoimmune flares.
GLP-1 receptor agonists address this metabolic inflammation directly. Even at microdoses, the improvement in insulin sensitivity, reduction in visceral fat inflammatory signaling, and modulation of gut barrier function create a less inflammatory internal environment. This does not cure the autoimmune condition. But it may reduce the inflammatory load enough that the immune system operates closer to its regulation threshold rather than chronically above it.
Think of it as noise reduction. The autoimmune signal still exists. But by reducing the metabolic inflammatory noise, the signal becomes more manageable. Flares trigger less easily. Resolution happens faster. The overall disease burden decreases even though the fundamental autoimmune process persists.
This framework explains why some patients see dramatic improvements while others see modest changes. Those with higher baseline metabolic inflammation have more noise to reduce. Those with primarily immune-driven (rather than inflammation-amplified) disease may see less benefit.
Gut health and the autoimmune-GLP-1 axis
The gut connection deserves special attention. Many autoimmune conditions have gut involvement, whether through intestinal permeability (leaky gut), microbiome disruption, or direct intestinal immune activation. The gut is the largest immune organ in the body, and its state directly influences systemic immune function.
GLP-1 is naturally produced in the gut. Its role in gut health extends beyond glucose regulation to include mucosal barrier maintenance and intestinal immune modulation. Peptides for gut health have been a growing area of interest, and GLP-1 receptor agonists fit into this framework through their effects on intestinal inflammation and barrier integrity.
For autoimmune patients whose conditions have a gut component, which includes most of them to varying degrees, the local gut effects of GLP-1 receptor activation may contribute to systemic immune improvement. This is distinct from the direct immune cell effects discussed earlier. It represents a second, complementary pathway through which microdosed GLP-1 could influence autoimmune activity.
What the skeptics say and where they have a point
Honest assessment requires addressing the criticism. And some of it is valid.
The evidence gap
No randomized controlled trial has specifically tested GLP-1 microdosing for autoimmune disease. The clinical trial NCT07092605 is underway but results are not yet available. Most of the evidence comes from standard-dose studies in metabolic populations, case reports, observational data, and clinical observations from practitioners.
This is a significant limitation. Observational data is prone to confounding. Case reports cannot establish causation. Clinical observations, while valuable, lack the controls necessary for confident conclusions.
The counter-argument is that mechanistic evidence is strong, multiple lines of clinical evidence converge, and the risk profile at microdose levels is favorable. But this does not substitute for controlled trials, and patients should understand they are, in effect, early adopters of an approach that is not yet evidence-based in the traditional sense.
The conflicting safety signal
One real-world evidence study found that patients receiving GLP-1 receptor agonists had higher risks of certain autoimmune conditions, including ulcerative colitis, rheumatoid arthritis, autoimmune thyroiditis, ankylosing spondylitis, and psoriasis, compared to those on DPP-4 inhibitors.
This finding directly contradicts other studies showing protective effects. How do we reconcile this?
Several possibilities exist. Detection bias (GLP-1 users are monitored more closely, leading to more diagnoses). Indication bias (patients prescribed GLP-1 agonists may have underlying risk factors for autoimmune disease). Confounding by obesity (which itself increases autoimmune risk). Or, the possibility that GLP-1 receptor agonists genuinely increase certain autoimmune risks in certain populations through immune activation effects that differ from the anti-inflammatory effects observed in other studies.
This unresolved tension means we cannot currently say with certainty that GLP-1 therapy is universally beneficial for autoimmune conditions. It may help some conditions while being neutral or potentially problematic for others. This is exactly why controlled trials are needed and why microdosing should be approached with appropriate medical supervision, not self-experimented with based on internet articles alone.
The dosing problem
Microdosing assumes that anti-inflammatory effects occur at lower doses than metabolic effects. While there is mechanistic support for this (different receptor occupancy thresholds), it has not been definitively demonstrated in human studies. It is possible that the anti-inflammatory CRP reductions observed in the STEP and SELECT trials require the full doses used in those studies. The 42% to 48% CRP reductions were achieved at 1.0 to 2.4 mg semaglutide, not at 0.05 mg.
Whether a proportional or threshold anti-inflammatory effect exists at microdose levels remains an open question. This is perhaps the most important unknown in the entire field.
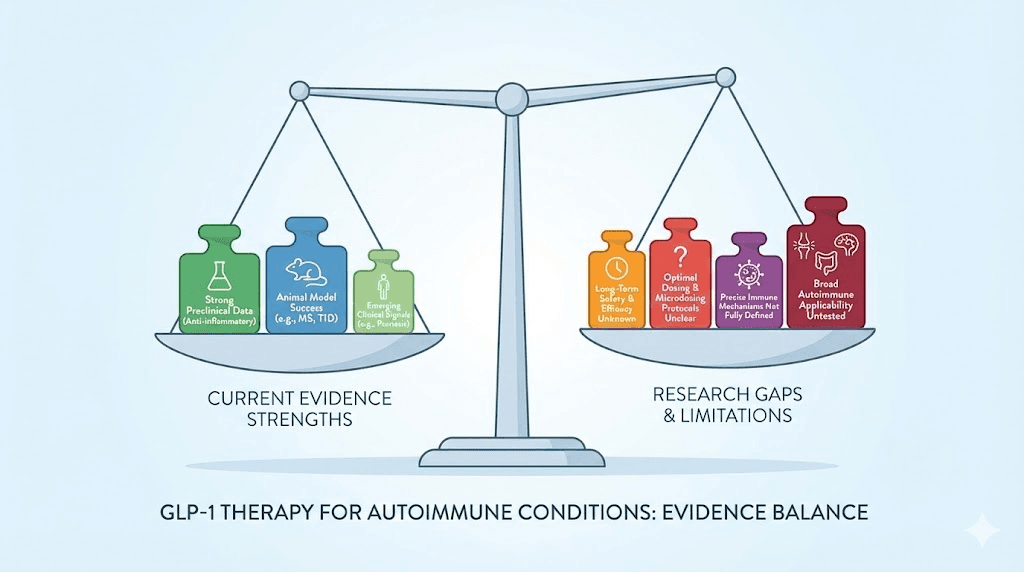
Practical considerations for getting started
If you and your healthcare provider decide that GLP-1 microdosing is worth exploring for your autoimmune condition, here are the practical details that matter.
Accessing microdose concentrations
Standard pharmaceutical GLP-1 products are not designed for microdosing. The lowest available pen dose for semaglutide (brand name Ozempic for diabetes, Wegovy for obesity) is 0.25 mg. Getting to 0.05 mg requires either:
Option 1: Compounded semaglutide. Compounded formulations can be prepared at custom concentrations that allow precise microdosing with standard insulin syringes. A typical compounded concentration for microdosing might be 1 mg/mL, allowing 0.05 mL draws for 0.05 mg doses. Reconstitution protocols must be followed precisely for accurate dosing.
Option 2: Insulin syringe from multi-dose vials. Some practitioners use standard semaglutide vials with insulin syringes to draw precise small volumes. This requires understanding semaglutide unit-to-mg conversions and having appropriate syringe measurement skills. The semaglutide dosage calculator can help with these conversions.
For tirzepatide, similar options exist through compounding pharmacies that can prepare custom concentrations. The compounded tirzepatide calculator helps determine correct volumes.
Storage at microdose volumes
When using compounded or multi-dose vial semaglutide for microdosing, storage becomes important because the vial will be used over a longer period (drawing smaller amounts means more doses per vial).
Semaglutide shelf life after reconstitution or first use depends on the formulation. Multi-dose vials typically have a 56-day in-use period when refrigerated. Compounded semaglutide storage varies by pharmacy, so check the specific beyond-use date on your vial. Refrigeration is essential for maintaining potency throughout the extended use period.
For tirzepatide, fridge storage guidelines apply similarly. Avoid temperature excursions, since warm temperatures degrade peptides regardless of dose.
Working with your healthcare team
This approach requires a prescriber. GLP-1 receptor agonists are prescription medications regardless of dose. Self-sourcing and self-dosing without medical supervision is strongly discouraged, particularly for autoimmune patients who are often on complex medication regimens.
When discussing this with your healthcare provider, bring the evidence. The ACR Convergence 2025 data, the Sjogren case report, the CRP reduction data from STEP and SELECT trials. Many physicians are aware of the emerging anti-inflammatory potential but may not have reviewed the autoimmune-specific data.
If your current provider is unfamiliar with or opposed to this approach, functional medicine practitioners and integrative rheumatologists are more likely to have experience with GLP-1 microdosing for autoimmune applications. SeekPeptides members connect with practitioners experienced in peptide-based approaches to inflammatory conditions.
The timeline: what to expect and when
Based on practitioner reports and the available clinical data, here is what the general timeline looks like for GLP-1 microdosing in autoimmune conditions.
Weeks 1 to 2: Most patients feel nothing different. The dose is too low for metabolic effects, and the anti-inflammatory effects are building but not yet clinically apparent. Some patients report slightly improved energy levels, possibly related to early inflammatory marker reduction.
Weeks 3 to 4: The first signs of benefit may appear. Reduced morning stiffness is the most commonly reported early change in RA patients. Psoriasis patients may notice slightly less plaque activity. Changes are subtle.
Weeks 4 to 8: If the approach is going to work, most patients begin seeing meaningful changes in this window. Flare frequency decreases. Pain levels drop. CRP measurements, if checked, may show the beginnings of a downward trend.
Weeks 8 to 12: This is the full assessment window. Inflammatory markers have had time to respond. Clinical symptoms have stabilized at whatever the new baseline will be. This is when practitioners typically decide whether to continue, adjust dose, or discontinue.
Month 3 and beyond: Patients who respond tend to continue responding. Some practitioners report progressive improvement through months 3 to 6, with the anti-inflammatory effect deepening over time. Whether this is due to cumulative immune modulation or other factors is unclear.
Not everyone responds. The honest estimate from practitioners using this approach is that roughly 60 to 70% of patients see some degree of improvement, while 30 to 40% see no meaningful change. Among responders, the degree of improvement varies from subtle symptom reduction to dramatic flare elimination, as illustrated by the Sjogren case report.
Combining GLP-1 microdosing with other anti-inflammatory strategies
GLP-1 microdosing does not operate in a vacuum. For autoimmune patients, optimizing the overall inflammatory environment amplifies whatever benefit the GLP-1 provides.
Nutritional support
Diet plays a significant role in modulating inflammation. For autoimmune patients on GLP-1 microdosing, an anti-inflammatory dietary pattern provides the foundation. Nutrition on GLP-1 therapy should emphasize omega-3 fatty acids, polyphenol-rich foods, and adequate protein to support immune function and tissue repair.
Specific supplements that practitioners commonly recommend alongside GLP-1 microdosing for autoimmune conditions include vitamin D (immune modulator), omega-3 fish oil (anti-inflammatory), and curcumin (NF-kB inhibitor through a complementary pathway). Protein intake is important for maintaining muscle mass and supporting immune function.
Glycine and B12 formulations
Several compounded GLP-1 formulations include glycine, B12, or both as stabilizers and potential synergistic agents. Semaglutide with glycine is a common compounded combination, as glycine itself has anti-inflammatory properties. Tirzepatide glycine B12 compounds combine three ingredients with potential anti-inflammatory effects.
For autoimmune patients, these combination formulations may provide modest additional anti-inflammatory benefit beyond the GLP-1 alone. Methylcobalamin (active B12) is particularly relevant for autoimmune patients, many of whom have compromised B12 absorption due to gut inflammation or medications like methotrexate.
Stress and sleep optimization
Chronic stress and poor sleep are both potent inflammatory stimuli that lower the flare threshold for autoimmune patients. Sleep quality can be affected by both the autoimmune condition and, at higher doses, by GLP-1 therapy itself. At microdose levels, sleep effects are typically neutral.
Addressing these foundational factors alongside GLP-1 microdosing creates a synergistic anti-inflammatory effect that exceeds what any single intervention provides alone.
Frequently asked questions
Does microdosing GLP-1 for autoimmune conditions cause weight loss?
At true microdose levels (0.05 to 0.1 mg semaglutide weekly), significant weight loss is uncommon. Appetite suppression requires higher receptor occupancy than these doses provide. Some patients report modest weight changes of 1 to 3 pounds, likely related to reduced inflammation-driven fluid retention rather than fat loss. If your goal is weight loss on semaglutide, standard therapeutic doses are needed.
Can I microdose GLP-1 without a prescription?
GLP-1 receptor agonists are prescription medications. They require a healthcare provider evaluation, prescription, and monitoring. This is especially important for autoimmune patients who may be on complex medication regimens. Self-sourcing is strongly discouraged due to quality control concerns and the need for proper medical supervision. Work with a qualified provider familiar with this approach.
How long before I know if GLP-1 microdosing is working for my autoimmune condition?
Most practitioners recommend a minimum 12-week trial before making efficacy judgments. Some patients notice improvements in morning stiffness and pain levels as early as weeks 3 to 4. Inflammatory marker changes (CRP, ESR) typically require 4 to 8 weeks to become measurable. Full assessment of how quickly semaglutide produces results varies by condition and individual response.
Is it safe to microdose GLP-1 with methotrexate or biologics?
No significant drug interactions have been identified between GLP-1 receptor agonists and standard autoimmune medications including methotrexate, biologics (TNF inhibitors, IL-6 inhibitors), JAK inhibitors, or hydroxychloroquine. The ACR Convergence 2025 data specifically showed additive benefit when GLP-1 agonists were combined with DMARDs. However, individual circumstances vary, so discuss combinations with your prescriber.
Which GLP-1 medication is better for autoimmune conditions, semaglutide or tirzepatide?
Both show anti-inflammatory potential. Semaglutide has more published data on inflammatory marker reduction. Tirzepatide may have broader anti-inflammatory pathways through its additional GIP receptor activation. The comparison between the two for autoimmune applications specifically is not settled. Some practitioners start with semaglutide (more data) and switch to tirzepatide (broader mechanism) if response is insufficient.
Will my insurance cover GLP-1 microdosing for autoimmune disease?
Almost certainly not. GLP-1 receptor agonists are FDA-approved for type 2 diabetes and obesity, not autoimmune conditions. Insurance coverage for off-label use is uncommon. Most patients access microdose GLP-1 through compounded formulations, which tend to be more affordable than brand-name products but are not typically covered by insurance. Costs vary significantly by pharmacy and formulation.
Can GLP-1 microdosing replace my current autoimmune medication?
No. Based on current evidence, GLP-1 microdosing is a complementary approach, not a replacement for standard immunotherapy. The integration of GLP-1 receptor agonists into autoimmune treatment has yielded partial improvements in disease control, not complete remission. Never discontinue prescribed autoimmune medications without direct guidance from your treating physician.
Are there autoimmune conditions where GLP-1 microdosing should be avoided?
The conflicting data regarding ulcerative colitis risk warrants caution for IBD patients until more data is available. Patients with a history of medullary thyroid carcinoma or MEN 2 syndrome should not use any GLP-1 receptor agonist. Patients with a history of pancreatitis should use extreme caution. For other autoimmune conditions, no specific contraindications have been identified beyond the general GLP-1 precautions, but individual risk-benefit assessment with a healthcare provider is essential.
External resources
American College of Rheumatology: GLP-1 therapies in rheumatic disease
Frontiers in Immunology: GLP-1 as modulator of innate immunity
Cell and Bioscience: GLP-1 receptor agonists in autoimmune diseases
For researchers serious about understanding how GLP-1 receptor agonists intersect with autoimmune management, SeekPeptides provides the most comprehensive resource available, with evidence-based protocols, detailed mechanism guides, and a community of thousands navigating these exact questions about peptide-based approaches to inflammation and immune health.
In case I do not see you, good afternoon, good evening, and good night. May your inflammation stay quiet, your immune system stay balanced, and your protocols stay evidence-based.