Feb 10, 2026
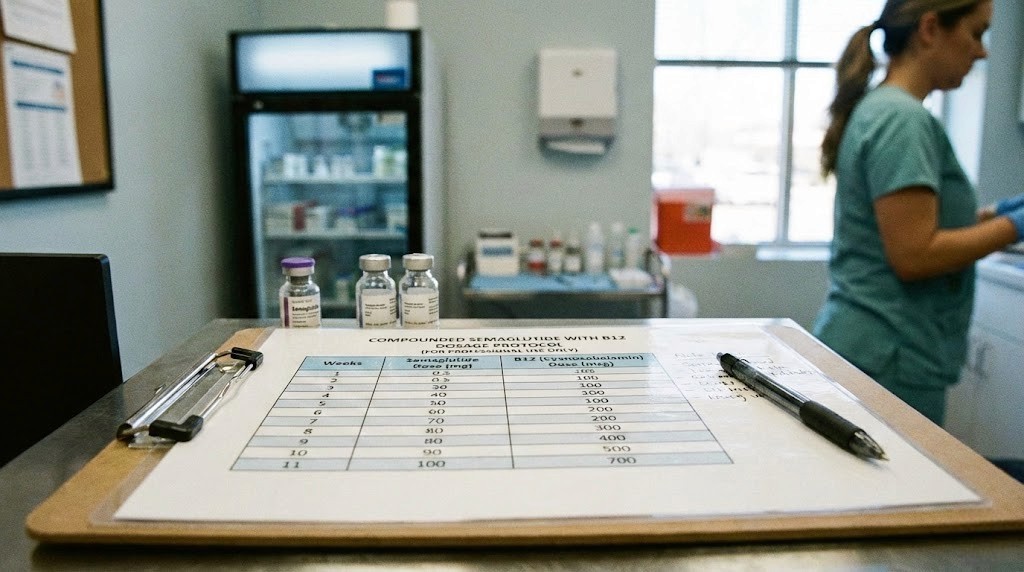
Your compounded semaglutide with B12 vial just arrived. You pull it out of the packaging, read the label, and immediately realize something. The concentration printed on that vial means nothing to you without a clear dosage chart that tells you exactly how many units to draw for each weekly dose.
You are not alone in this confusion.
Compounded semaglutide formulations come in wildly different concentrations depending on the pharmacy. One vial might contain 1 mg/mL. Another could hold 5 mg/mL. And the B12 component adds yet another layer of complexity that brand-name products like Ozempic and Wegovy simply do not have. Without a reliable dosage reference, you are left guessing, and guessing with a GLP-1 receptor agonist is a fast track to nausea, vomiting, and wasted medication. This guide breaks down every concentration you are likely to encounter, maps out the exact units to draw at each dose tier, explains why vitamin B12 is included in these formulations in the first place, and walks you through the complete titration schedule from your very first 0.25 mg injection to the maximum maintenance dose. Whether your provider prescribed a 1 mg/mL vial or a 5 mg/mL formulation, the charts below will show you precisely what to do. You will also find guidance on higher-concentration vials, syringe conversion math, and how to transition between different GLP-1 medications if your provider recommends a change.
What compounded semaglutide with B12 actually is
Compounded semaglutide with B12 is a custom-mixed injectable medication that combines two active ingredients into a single vial. The first is semaglutide, a GLP-1 receptor agonist that mimics the incretin hormone your gut naturally produces after eating. The second is cyanocobalamin, the synthetic form of vitamin B12 that your body converts into its active coenzyme forms, methylcobalamin and adenosylcobalamin.
This is not Ozempic. It is not Wegovy. Those are FDA-approved brand-name medications manufactured by Novo Nordisk under strict pharmaceutical controls. Compounded semaglutide with B12 is prepared by compounding pharmacies, either 503A state-licensed pharmacies that fill individual prescriptions or 503B outsourcing facilities that produce larger batches under federal oversight. The distinction matters because compounded medications are not FDA-approved, meaning no regulatory body has reviewed these specific formulations for safety, efficacy, or quality. If you are wondering about the differences between semaglutide and tirzepatide, that comparison involves an entirely different class of dual-agonist medications.
That said, the individual ingredients are well-studied. Semaglutide has robust clinical data behind it. The STEP trials demonstrated an average body weight reduction of approximately 15% over 68 weeks in participants receiving 2.4 mg weekly, making it one of the most effective peptides for fat loss currently available. And cyanocobalamin has decades of established safety data as a vitamin B12 supplement. The peptide calculator on SeekPeptides can help you understand dosing math across various peptide formulations.
Compounding pharmacies combine them for a practical reason. Semaglutide slows gastric emptying and reduces stomach acid production, both of which can impair the absorption of dietary vitamin B12. By delivering B12 directly via subcutaneous injection, the formulation bypasses the digestive system entirely, ensuring the vitamin reaches your bloodstream regardless of what semaglutide does to your gut.
Why B12 is included in compounded semaglutide formulations
There are three reasons compounding pharmacies add cyanocobalamin to semaglutide formulations, and only one of them has strong scientific support.
Reason 1: GLP-1 medications may impair B12 absorption
This is the most evidence-backed rationale. GLP-1 receptor agonists like semaglutide slow gastric emptying and reduce gastric acid secretion. Vitamin B12 absorption depends on a multi-step process that starts in the stomach. Stomach acid separates B12 from dietary proteins, then a protein called intrinsic factor binds to the freed B12 and escorts it to absorption sites in the ileum. When semaglutide reduces gastric acid, it disrupts the very first step in that chain. Less acid means less B12 gets freed from food proteins. Less freed B12 means less binding to intrinsic factor. And less binding means less absorption in the small intestine.
Research confirms this is not theoretical. A study examining once-weekly semaglutide treatment found that weight reduction was accompanied by significant decreases in serum albumin, vitamin B12, and zinc levels. Patients on long-term GLP-1 therapy, especially those also taking metformin or proton pump inhibitors, face compounded risk for B12 depletion.
The injectable B12 in compounded formulations sidesteps this problem completely. Subcutaneous cyanocobalamin goes directly into the bloodstream. No stomach acid needed. No intrinsic factor required. No absorption variability.
Reason 2: B12 may help counteract fatigue during caloric restriction
Semaglutide suppresses appetite significantly. Most people eat substantially less while on it, which is the entire point for weight loss. But reduced food intake means reduced nutrient intake across the board, and B12 plays a critical role in energy metabolism. It helps convert food into glucose, supports red blood cell production, and maintains myelin sheaths around nerve fibers.
When B12 levels drop, fatigue is one of the earliest symptoms. Adding it to the semaglutide formulation provides a consistent weekly dose that may help maintain energy levels even as caloric intake decreases.
Important caveat: if your B12 levels are already adequate, supplementation will not boost your energy beyond baseline. The fatigue-fighting benefit only applies to people who are deficient or trending toward deficiency.
Reason 3: glycine for muscle preservation
Many compounded semaglutide formulations also include glycine alongside B12. Glycine is a simple amino acid that serves as both a stabilizer for the semaglutide molecule and a potential support for muscle preservation during rapid weight loss. Some pharmacies market the semaglutide/glycine/B12 combination as a more comprehensive weight management formulation.
The evidence for glycine preventing muscle loss during GLP-1 therapy is limited, but the amino acid is safe at the doses included in these formulations. Researchers interested in peptides for athletic performance often look at glycine in the context of collagen synthesis and connective tissue support, though those applications involve much higher doses than what compounding pharmacies include in semaglutide formulations.
Compounded semaglutide with B12 dosage chart by concentration
Here is where most people get confused. Compounded semaglutide comes in different concentrations depending on the pharmacy, and the number of units you draw changes dramatically based on what concentration you have. Drawing the wrong amount from a higher-concentration vial can mean receiving two to five times your intended dose.
The two most common concentrations from compounding pharmacies are 1 mg/mL (with 0.5 mg/mL cyanocobalamin) and 5 mg/mL (with 0.5 mg/mL cyanocobalamin). Some pharmacies also produce 2.5 mg/mL formulations.
Dosage chart: 1 mg/mL concentration
This is the lower-concentration formulation. You will draw larger volumes for each dose, which can make precise dosing easier for beginners since small measurement errors have less impact.
Phase | Weeks | Semaglutide dose | Volume to draw | Insulin syringe units | B12 delivered |
|---|---|---|---|---|---|
Initiation | 1-4 | 0.25 mg | 0.25 mL | 25 units | 0.125 mg |
Titration 1 | 5-8 | 0.5 mg | 0.50 mL | 50 units | 0.25 mg |
Titration 2 | 9-12 | 1.0 mg | 1.00 mL | 100 units (full syringe) | 0.5 mg |
Titration 3 | 13-16 | 1.7 mg | 1.70 mL | Use 3 mL syringe | 0.85 mg |
Maintenance | 17+ | 2.4 mg | 2.40 mL | Use 3 mL syringe | 1.2 mg |
Notice that once you move past 1.0 mg per week with the 1 mg/mL concentration, you exceed the capacity of a standard 1 mL insulin syringe. At that point, you will need to switch to a 3 mL syringe or split the injection into two separate sites. Many providers transition patients to the 5 mg/mL concentration at this stage instead. Understanding how syringe units convert to milligrams is essential for avoiding dosing errors, especially when switching between concentrations. For patients using a different concentration, the 40-unit to milligram conversion guide provides additional reference points.
Dosage chart: 2.5 mg/mL concentration
This mid-range concentration offers a balance between the precision of lower concentrations and the convenience of higher ones. It works well across the entire dosage range without requiring syringe changes.
Phase | Weeks | Semaglutide dose | Volume to draw | Insulin syringe units | B12 delivered |
|---|---|---|---|---|---|
Initiation | 1-4 | 0.25 mg | 0.10 mL | 10 units | 0.05 mg |
Titration 1 | 5-8 | 0.5 mg | 0.20 mL | 20 units | 0.10 mg |
Titration 2 | 9-12 | 1.0 mg | 0.40 mL | 40 units | 0.20 mg |
Titration 3 | 13-16 | 1.7 mg | 0.68 mL | 68 units | 0.34 mg |
Maintenance | 17+ | 2.4 mg | 0.96 mL | 96 units | 0.48 mg |
Dosage chart: 5 mg/mL concentration
The higher-concentration formulation is what most compounding pharmacies dispense for patients who have already titrated up or who want fewer vials over the course of treatment. The volumes are much smaller, which means precision matters more. A mistake of just 5 units on an insulin syringe translates to a 0.25 mg dosing error.
Phase | Weeks | Semaglutide dose | Volume to draw | Insulin syringe units | B12 delivered |
|---|---|---|---|---|---|
Initiation | 1-4 | 0.25 mg | 0.05 mL | 5 units | 0.025 mg |
Titration 1 | 5-8 | 0.5 mg | 0.10 mL | 10 units | 0.05 mg |
Titration 2 | 9-12 | 1.0 mg | 0.20 mL | 20 units | 0.10 mg |
Titration 3 | 13-16 | 1.7 mg | 0.34 mL | 34 units | 0.17 mg |
Maintenance | 17+ | 2.4 mg | 0.48 mL | 48 units | 0.24 mg |
With the 5 mg/mL concentration, every dose fits comfortably within a standard 1 mL insulin syringe. This is the primary advantage of higher concentrations, you never need to switch syringes or split injections. For detailed reconstitution math with the 5 mg vial specifically, the semaglutide 5mg/mL dosage chart provides additional breakdowns. And if your pharmacy dispenses 10 mg vials, the 10 mg semaglutide mixing chart covers those calculations. Patients who need help determining the right reconstitution ratios should consult with their pharmacist, as compounded vials may arrive in lyophilized form requiring bacteriostatic water mixing.
The standard titration schedule explained week by week
Titration is not optional. It is the single most important factor in tolerating semaglutide without debilitating side effects. The medication works by activating GLP-1 receptors throughout your body, in your pancreas, your brain, your stomach, and your gut. Jumping straight to a therapeutic dose overwhelms these receptors and triggers severe gastrointestinal distress in most people.
Here is what to expect at each stage.
Weeks 1 through 4: the initiation phase (0.25 mg)
This is not a therapeutic dose for weight loss. Think of it as a training period for your GLP-1 receptors. At 0.25 mg weekly, you are introducing semaglutide to your system at the lowest level that produces measurable receptor activation without overwhelming your digestive tract.
What to expect during these four weeks:
Mild appetite reduction, some people notice it immediately while others feel nothing
Possible mild nausea in the 24 to 48 hours after injection, affecting roughly 20 to 30% of people at this dose
Minimal to no weight loss, perhaps 1 to 3 pounds, mostly from reduced food intake
Slight changes in food preferences, many people report decreased interest in sugary or fatty foods
If you tolerate 0.25 mg with zero issues, resist the temptation to increase early. Your body needs all four weeks to upregulate receptor sensitivity and adjust gastrointestinal motility. This patience applies equally to people transitioning from compounded tirzepatide or any other GLP-1 medication.
Weeks 5 through 8: the first therapeutic dose (0.5 mg)
Now things start happening. At 0.5 mg, most people experience noticeable appetite suppression. Meals feel satisfying sooner. Cravings diminish. The voice in your head that used to demand a snack at 3 PM goes quiet.
Side effects often peak during the first week or two at this dose. Nausea affects approximately 30 to 40% of people, though it typically resolves by the end of week 6 or 7. Constipation becomes more common as gastric motility slows further. And some people experience what researchers call "food noise reduction," a dramatic decrease in the mental chatter about food that many people with obesity experience constantly.
Weight loss at this stage typically ranges from 3 to 7 pounds total since starting treatment. Your provider will assess your response at the end of week 8 to determine whether to increase. If results are underwhelming, review the complete troubleshooting guide before assuming the medication is not working. For patients tracking their dosing in units rather than milligrams, the conversion charts above become especially important at this stage.
Weeks 9 through 12: building momentum (1.0 mg)
The 1.0 mg dose is where the clinical weight loss data starts getting compelling. This is the dose used in many of the pivotal semaglutide studies, and it produces meaningful metabolic changes beyond simple appetite suppression.
At this level, semaglutide meaningfully improves insulin sensitivity, reduces fasting glucose levels, and begins to shift body composition. Many people report not just eating less but genuinely feeling different about food. The compulsive relationship with eating loosens its grip.
Common experiences at 1.0 mg:
Consistent weekly weight loss of 0.5 to 1.5 pounds
Significant reduction in hunger between meals
Possible intermittent nausea, usually manageable and less intense than at 0.5 mg
Increased fullness after smaller portions
Some people develop mild acid reflux at this dose
If the B12 component of your compounded formulation is going to show its value, this is when it starts mattering. Caloric intake is significantly reduced by week 9, and the risk of nutritional gaps begins to increase.
Weeks 13 through 16: approaching maximum efficacy (1.7 mg)
Not everyone needs to reach 1.7 mg. If you are losing weight consistently at 1.0 mg with tolerable side effects, your provider may keep you there. The decision to increase depends on your rate of weight loss, your tolerance profile, and how far you are from your goal.
At 1.7 mg, the GLP-1 receptor activation is near its peak. Side effects that resolved at lower doses sometimes return temporarily as your body adjusts to the higher level. Gastrointestinal symptoms affect roughly 15 to 25% of people at this dose, though most are milder than what they experienced during initial titration.
Total weight loss by week 16 typically ranges from 8 to 15% of starting body weight for people who have been consistent with their injections and made reasonable dietary adjustments. The before and after weight loss results page shows real-world outcomes from GLP-1 users at similar timepoints. For patients who started on tirzepatide and are considering semaglutide, or vice versa, the switching dosage chart provides the transition framework.
Week 17 and beyond: maintenance dosing (2.0 to 2.4 mg)
The maximum recommended dose of semaglutide for weight management is 2.4 mg weekly, the same dose used in the Wegovy clinical program. The STEP 1 trial showed that participants receiving 2.4 mg weekly for 68 weeks lost an average of 14.9% of their body weight compared to 2.4% in the placebo group.
Not all compounded formulations support doses above 2.0 mg per week. Check your vial concentration and remaining volume before attempting to draw a 2.4 mg dose. With a 5 mg/mL concentration, 2.4 mg requires drawing 0.48 mL (48 units), which is well within a standard insulin syringe. With a 1 mg/mL concentration, 2.4 mg requires 2.4 mL, which needs a 3 mL syringe.
Maintenance phase strategy varies. Some providers keep patients at 2.4 mg indefinitely. Others cycle between 1.7 mg and 2.4 mg based on weight trajectory. And some begin tapering once the patient reaches their goal weight, reducing by one dose tier every four weeks to determine the minimum effective dose for weight maintenance. Understanding what happens when you stop semaglutide helps set realistic expectations if discontinuation ever becomes necessary. People curious about whether alcohol affects semaglutide should pay particular attention during the maintenance phase when social eating patterns resume.
How to read your vial label and calculate the correct dose
This trips up nearly everyone who receives their first compounded semaglutide vial. The label format varies between pharmacies, but most follow a pattern like this:
Semaglutide/Cyanocobalamin 5/0.5 mg/mL
That notation means each milliliter of liquid contains 5 mg of semaglutide and 0.5 mg of cyanocobalamin (vitamin B12). The first number is always semaglutide. The second is always B12.
The math for any concentration
To calculate how much liquid to draw for any dose at any concentration, use this formula:
Volume (mL) = Desired dose (mg) / Concentration (mg/mL)
Then convert to insulin syringe units:
Units = Volume (mL) x 100
Example: You want 0.5 mg from a 5 mg/mL vial.
Volume = 0.5 / 5 = 0.10 mL
Units = 0.10 x 100 = 10 units
Example: You want 1.7 mg from a 2.5 mg/mL vial.
Volume = 1.7 / 2.5 = 0.68 mL
Units = 0.68 x 100 = 68 units
For a faster approach, use the SeekPeptides semaglutide dosage calculator to plug in your vial concentration and desired dose. It handles the math instantly and shows you exactly where to draw on your syringe.
Common vial sizes and how many doses they contain
Vial size | Concentration | Total semaglutide | Doses at 0.25 mg | Doses at 0.5 mg | Doses at 1.0 mg | Doses at 2.4 mg |
|---|---|---|---|---|---|---|
1 mL | 5 mg/mL | 5 mg | 20 | 10 | 5 | 2 |
2 mL | 5 mg/mL | 10 mg | 40 | 20 | 10 | 4 |
2.5 mL | 5 mg/mL | 12.5 mg | 50 | 25 | 12 | 5 |
1 mL | 1 mg/mL | 1 mg | 4 | 2 | 1 | 0 |
2.5 mL | 1 mg/mL | 2.5 mg | 10 | 5 | 2 | 1 |
Knowing how many doses your vial contains helps you plan refills. Nothing is worse than running out mid-titration because you miscounted doses. The peptide cost calculator can help you estimate your total cost per month at each dose level, factoring in vial size and concentration.
Compounded vs. brand-name semaglutide: what the B12 addition changes
The differences between compounded semaglutide with B12 and brand-name products go deeper than just an added vitamin. Understanding these differences helps you make informed decisions about your treatment.
Regulatory status
Ozempic (for type 2 diabetes) and Wegovy (for weight management) are FDA-approved. They went through years of clinical trials involving thousands of participants. The FDA reviewed their manufacturing processes, purity standards, efficacy data, and safety profiles before granting approval.
Compounded semaglutide with B12 has no FDA approval. The FDA does not review compounded medications for safety or efficacy. This does not automatically mean they are unsafe, but it does mean the quality assurance burden shifts from a federal agency to the compounding pharmacy and your prescribing provider.
The semaglutide shortage context
Compounded semaglutide became widely available during the semaglutide drug shortage that lasted from early 2022 through February 2025. Federal law allows compounding pharmacies to produce copies of FDA-approved medications when those medications are on the official drug shortage list. When the FDA resolved the shortage in February 2025, it created regulatory uncertainty for compounding pharmacies.
The FDA established enforcement discretion periods, giving 503A pharmacies until April 2025 and 503B outsourcing facilities until May 2025 to wind down compounded semaglutide production. Legal challenges from compounding industry groups have complicated these timelines, and the regulatory landscape continues to evolve. Check with your provider about the current availability of compounded formulations in your area.
Formulation differences
Feature | Ozempic/Wegovy | Compounded semaglutide with B12 |
|---|---|---|
Active ingredient | Semaglutide only | Semaglutide + cyanocobalamin (B12) |
Delivery device | Pre-filled pen | Multi-dose vial + syringe |
Concentrations | Fixed per pen strength | Varies (1, 2.5, or 5 mg/mL) |
FDA approved | Yes | No |
B12 included | No | Yes (0.5 mg/mL typically) |
Dose flexibility | Fixed dose clicks | Any dose within vial range |
Storage after opening | 56 days at room temp (Ozempic) | 28 days refrigerated typically |
The dose flexibility of compounded formulations is actually an advantage for some patients. Brand-name pens only deliver fixed doses (0.25, 0.5, 1.0, 1.7, or 2.4 mg). Compounded vials let you draw any amount, which gives providers the ability to prescribe intermediate doses like 0.375 mg or 1.25 mg for patients who need a gentler microdosing approach. This flexibility also matters for patients using peptide injection pens rather than standard syringes, though most compounded semaglutide vials are designed for syringe withdrawal. The injectable versus oral peptide comparison explains why injectable delivery remains the gold standard for GLP-1 medications.
Injection technique for compounded semaglutide with B12
Drawing from a multi-dose vial is different from clicking a pre-filled pen. The process requires more steps but gives you full control over your dose accuracy.
Step-by-step injection protocol
Step 1: Gather your supplies. You need your compounded semaglutide with B12 vial, an appropriately sized syringe (insulin syringe for doses under 1 mL, 3 mL syringe for larger volumes), alcohol swabs, and a sharps container.
Step 2: Check the vial. Examine the liquid inside. Compounded semaglutide with B12 typically has a light pink to reddish hue from the cyanocobalamin. The solution should be clear with no visible particles, cloudiness, or crystallization. If anything looks off, do not use it.
Step 3: Clean the vial top. Wipe the rubber stopper with an alcohol swab and let it dry for 10 to 15 seconds. Do this every time, even if you just cleaned it an hour ago.
Step 4: Draw air into the syringe. Pull the plunger back to the volume you plan to withdraw. This creates positive pressure in the vial that makes drawing medication easier.
Step 5: Inject air and draw medication. Insert the needle through the rubber stopper, push the air in, then invert the vial and slowly pull the plunger back to your target dose. Tap the syringe gently to move any air bubbles to the top, then push the plunger slightly to expel them.
Step 6: Choose your injection site. The three approved subcutaneous injection sites are the abdomen (at least 2 inches from the navel), the front of the thigh (middle third), and the back of the upper arm. Rotate between these sites each week to prevent lipodystrophy, the formation of hard lumps under the skin from repeated injections in the same spot.
Step 7: Clean and inject. Swab the injection site with alcohol. Let it dry. Pinch a fold of skin, insert the needle at a 45 to 90 degree angle (depending on your body composition), push the plunger slowly and steadily, wait 5 to 10 seconds, then withdraw.
Step 8: Dispose properly. Place the used syringe immediately into a sharps container. Never recap needles. Never throw syringes in regular trash. The same injection technique applies to other reconstituted peptide medications and tirzepatide preparations.
Injection site rotation schedule
A simple rotation pattern prevents tissue damage and ensures consistent absorption. Try this four-week cycle:
Week 1: Right side of abdomen
Week 2: Left thigh
Week 3: Left side of abdomen
Week 4: Right thigh
Then repeat. If you have a partner who can help, the back of the upper arm is also an excellent injection site with consistent subcutaneous absorption.
Storage requirements for compounded semaglutide with B12
Improper storage destroys peptides. Semaglutide is a 31-amino-acid peptide that degrades when exposed to heat, light, or freezing temperatures. The B12 component is more stable, but the combined formulation should be treated with the same care as the semaglutide alone.
Temperature requirements
Store your vial between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). This means the refrigerator, not the door shelf where temperatures fluctuate every time you open it, but the middle or back of a shelf where the temperature stays consistent.
Never freeze compounded semaglutide. Freezing causes irreversible damage to the peptide structure. If your vial accidentally freezes, even briefly, discard it. The semaglutide molecule cannot recover from ice crystal formation.
Room temperature exposure is acceptable for short periods. Most compounding pharmacies rate their semaglutide formulations for up to 72 hours at room temperature (below 77 degrees Fahrenheit). Some formulations may tolerate longer, but check the specific guidance from your pharmacy. Storage guidelines vary between compounding facilities.
Light protection
The cyanocobalamin (B12) component is light-sensitive. Exposure to direct sunlight or strong artificial light can degrade it over time. Keep your vial in its original box or wrapping when not actively drawing a dose. If your pharmacy dispenses vials without light-protective packaging, store them in a dark area of your refrigerator.
After first puncture
Once you insert a needle through the rubber stopper for the first time, the clock starts ticking. Most compounding pharmacies assign a 28-day beyond-use date from the first puncture. Write the date of first use on the vial with a permanent marker so you never lose track.
After 28 days, discard the vial regardless of how much medication remains. The risk of bacterial contamination increases with each needle puncture, and the preservatives in the formulation have a limited window of effectiveness. Check how long compounded semaglutide lasts in the fridge for detailed stability data. The same storage principles apply to compounded tirzepatide and tirzepatide room temperature exposure. For general guidance on preserving peptide potency, the post-reconstitution storage guide covers the science behind degradation curves. Understanding lyophilized versus liquid peptide stability also helps contextualize why storage matters so much for these medications.
Side effects and how to manage them
Semaglutide side effects follow a predictable pattern. They are worst during the first two weeks at any new dose, then gradually improve as your body adapts. Understanding this pattern prevents unnecessary panic and premature dose reductions.
Gastrointestinal effects (most common)
Nausea affects 30 to 40% of people at some point during treatment. It typically peaks in the first 24 to 72 hours after an injection and fades by day 4 or 5. Eating smaller meals, avoiding fatty or greasy foods, and staying hydrated all help. Some people find that injecting in the evening rather than the morning reduces daytime nausea.
Constipation affects 10 to 15% of people, and it can be persistent. Semaglutide slows the entire digestive tract, which means stool spends more time in the colon and loses more water. Increasing fiber intake, drinking more water, and adding a gentle osmotic laxative (like polyethylene glycol) if needed are standard management strategies.
Diarrhea affects a similar percentage and sometimes alternates with constipation. It usually resolves within the first month at each dose level.
Abdominal pain and bloating affect approximately 10% of people. Eating slowly, chewing thoroughly, and avoiding carbonated beverages can help.
Vomiting affects 15 to 20% of people, primarily during dose increases. If vomiting occurs within 30 minutes of your weekly injection, contact your provider to ask whether a replacement dose is needed.
Non-gastrointestinal effects
Headache is common in the first few weeks and usually related to changes in blood sugar regulation or reduced caloric intake. Staying hydrated and maintaining consistent meal timing helps.
Fatigue affects some people, particularly in the first month. This is where the B12 component of your compounded formulation may prove beneficial. If fatigue persists beyond the first few weeks, ask your provider to check your B12 and iron levels.
Injection site reactions occur in 5 to 10% of people. Redness, mild swelling, or itching at the injection site is normal and typically resolves within a few hours.
Increased heart rate is documented in clinical data, typically an increase of 2 to 3 beats per minute at rest. This is generally not clinically significant but should be discussed with your provider if you have pre-existing cardiac conditions.
Serious side effects (rare but important)
Stop your medication and contact your healthcare provider immediately if you experience:
Severe, persistent abdominal pain that radiates to your back (possible pancreatitis, occurring in less than 1% of patients)
Signs of gallbladder problems including intense upper-right abdominal pain, fever, and jaundice (approximately 2 to 3% incidence)
Symptoms of allergic reaction including facial swelling, difficulty breathing, or widespread rash (less than 1%)
Signs of kidney problems including changes in urine output, swelling in legs or ankles, or unusual fatigue
Any thoughts of self-harm (reported in post-marketing surveillance)
The B12 advantage for side effect management
One theoretical advantage of the compounded B12 formulation is the potential to reduce fatigue and maintain neurological function during aggressive caloric restriction. B12 deficiency symptoms, including fatigue, weakness, numbness in extremities, cognitive fog, and mood changes, can mimic or worsen common semaglutide side effects. By proactively supplementing B12 through the injection itself, compounded formulations may help prevent this overlap.
However, it is important to note that no clinical trials have directly compared compounded semaglutide with B12 against standard semaglutide for side effect severity. The theoretical benefit is logical but unproven in controlled studies. For patients exploring other peptides with gut health benefits, some researchers combine GLP-1 therapy with supportive peptides, though this should always be done under medical supervision. The peptide stacking calculator can help assess potential interactions between different compounds.
Drug interactions to watch for
Semaglutide interacts with several common medications, and the B12 component introduces a few additional considerations. Bring your complete medication list to every appointment.
Medications that affect semaglutide efficacy
Oral medications in general: Because semaglutide slows gastric emptying, it can alter the absorption rate and peak concentration of any oral medication you take. This does not mean you cannot take other medications, but timing and monitoring may need adjustment.
Oral contraceptives: Delayed gastric emptying can reduce the absorption of hormonal birth control pills. Your provider may recommend switching to non-oral contraceptive methods (patch, ring, IUD, or injection) while on semaglutide.
Insulin and sulfonylureas: Combining semaglutide with these diabetes medications increases the risk of hypoglycemia. Providers typically reduce insulin or sulfonylurea doses when initiating semaglutide. Never adjust diabetes medications on your own.
Warfarin: Semaglutide can alter warfarin absorption, leading to INR fluctuations. More frequent monitoring is recommended, especially during dose changes.
Levothyroxine: Thyroid medication absorption can be affected. Thyroid function tests should be monitored more frequently during semaglutide titration.
Medications that affect B12 status
Metformin: This diabetes medication is independently associated with reduced B12 absorption. If you take both metformin and compounded semaglutide, you face a double hit on B12 levels. The injectable B12 in your compounded formulation helps offset this, but regular monitoring is still wise.
Proton pump inhibitors (PPIs): Medications like omeprazole and pantoprazole further reduce stomach acid, compounding the B12 absorption problem. Again, injectable B12 bypasses this concern, but your provider should know about all acid-reducing medications you take.
H2 receptor blockers: Similar to PPIs, medications like famotidine and ranitidine reduce gastric acid and can impair dietary B12 absorption.
Absolute contraindications
Do not use compounded semaglutide with B12 if you have:
A personal or family history of medullary thyroid carcinoma (MTC)
Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
Known hypersensitivity to semaglutide or cyanocobalamin
Leber hereditary optic neuropathy (a contraindication specific to the cyanocobalamin component)
Active pregnancy (semaglutide showed embryofetal toxicity in animal studies)
Understanding drug interactions is especially important for people using peptide therapy broadly, as multiple compounds can produce unpredictable interactions. The research versus pharmaceutical peptide comparison provides context on quality and safety standards across different peptide sources. Patients considering adding other compounds alongside their semaglutide protocol should review the prescribing landscape and discuss options with their healthcare provider.
Monitoring your progress and adjusting your protocol
Weight loss is the obvious metric, but smart monitoring goes much deeper than the scale. Tracking the right data points helps your provider make better dosing decisions and catches potential problems early.
What to track weekly
Body weight: Weigh yourself at the same time each week, same day, same conditions. Morning after using the bathroom, before eating or drinking, is the most consistent approach. Do not weigh daily. Water fluctuations will drive you insane.
Injection details: Record the date, time, dose, injection site, and lot number from your vial. This creates a complete treatment record.
Side effects: Note any symptoms, their severity (mild/moderate/severe), duration, and what helped. This data is invaluable when your provider is deciding whether to increase your dose.
Food intake: Not detailed calorie counting, but a general awareness of how much and what you are eating. Semaglutide can reduce intake so dramatically that some people under-eat, leading to nutritional deficiencies, muscle loss, and fatigue.
What to track monthly
Measurements: Waist circumference, hip circumference, and ideally a few other sites. The scale lies sometimes. You can lose inches while the scale barely moves if you are recomposing body tissue.
Energy levels: Rate your average daily energy on a 1 to 10 scale. A steady decline might indicate B12 depletion despite the supplementation in your formulation, caloric restriction that is too aggressive, or other nutritional gaps.
Mood and cognition: Note any changes in mental clarity, motivation, or emotional stability. Both semaglutide and B12 deficiency can affect neurological function.
Lab work schedule
Your provider should order baseline labs before starting treatment and then monitor periodically. Key markers include:
Lab test | Why it matters | When to check |
|---|---|---|
Complete metabolic panel | Kidney/liver function, electrolytes | Baseline, 3 months, 6 months |
Vitamin B12 level | Ensures supplementation is adequate | Baseline, 3 months, then annually |
Fasting glucose/HbA1c | Blood sugar control | Baseline, 3 months, 6 months |
Lipid panel | Cholesterol response to weight loss | Baseline, 6 months |
Thyroid function (TSH) | Especially if on levothyroxine | Baseline, 3 months if on thyroid meds |
Complete blood count | Detects megaloblastic anemia from B12 deficiency | Baseline, 6 months |
Iron/ferritin | Reduced food intake can cause iron depletion | Baseline, 6 months |
When to adjust your dose
Consider staying at current dose if: You are losing 1 to 2 pounds per week consistently, side effects are tolerable, and you feel good. There is no clinical benefit to increasing just because you can.
Consider increasing if: Weight loss has stalled for 3 or more weeks despite consistent injections and reasonable dietary habits, and you are below the maximum dose. Plateaus lasting less than 3 weeks are normal and do not require dose changes.
Consider decreasing if: Side effects are intolerable despite management strategies, weight loss is faster than 3 pounds per week consistently (which increases gallstone and muscle loss risk), or you have reached your goal weight and want to find the minimum effective maintenance dose.
Consider pausing if: You experience any serious side effects listed above, you are planning pregnancy (discontinue at least 2 months before conception), or you have a scheduled surgery (your surgeon may request discontinuation due to gastroparesis concerns during anesthesia). For patients who have been on semaglutide long-term and are considering discontinuation, the semaglutide withdrawal symptoms guide explains what to expect. Understanding how quickly GLP-1 medications take effect also provides context for how quickly their effects fade when stopped.
Weight loss timeline and realistic expectations
Setting realistic expectations prevents disappointment and premature discontinuation. Here is what the clinical data and real-world experience show for compounded semaglutide with B12, assuming consistent weekly injections and basic dietary awareness.
Month 1 (0.25 mg phase)
Expected weight loss: 2 to 5 pounds. Some people lose nothing. A few lose more. This is the adaptation phase, not the results phase. If you are measuring success at month one, you are measuring too early.
Month 2 (0.5 mg phase)
Expected cumulative weight loss: 5 to 10 pounds. Appetite suppression becomes noticeable and consistent. Many people describe this as the "turning point" where they realize the medication is actually working, even if the scale has not moved dramatically.
Months 3 and 4 (1.0 mg phase)
Expected cumulative weight loss: 8 to 15% of starting body weight by the end of month 4 for strong responders. Average responders see 5 to 10%. This is the period where weight loss accelerates and becomes visually noticeable to others.
Months 5 through 8 (1.7 to 2.4 mg phase)
Total weight loss approaches the 12 to 15% range documented in clinical trials. The rate of loss typically slows compared to months 3 and 4, but the trajectory continues downward. This is normal, not a plateau. Your body is approaching a new equilibrium point.
Beyond month 8
Weight loss continues but at a slower rate, typically reaching its maximum around month 15 to 17 in clinical trial data. The STEP 1 trial showed continued weight reduction through week 68 (approximately 17 months), with a final average loss of 14.9% of starting body weight at 2.4 mg weekly. For comparison, tirzepatide before and after results in men show similar or greater reductions over comparable timeframes, as the dual-agonist mechanism targets both GLP-1 and GIP receptors simultaneously.
After reaching your maximum weight loss, the goal shifts to maintenance. This is where long-term strategy matters, including finding the right maintenance dose, establishing sustainable eating habits, and incorporating physical activity to preserve lean muscle mass. Some patients explore complementary approaches including peptides specifically targeting abdominal fat or fat-burning peptides to support their GLP-1 protocol, though evidence for these combinations is still emerging. Patients interested in weight loss during menopause face unique hormonal challenges that may affect their semaglutide response timeline.
Special considerations for different populations
Not everyone follows the standard dosing chart. Several populations require modified approaches.
Patients also taking metformin
If you take metformin for type 2 diabetes or insulin resistance, the B12 component of your compounded semaglutide is especially important. Metformin independently reduces B12 absorption by approximately 5 to 10% over long-term use. Combined with semaglutide gastric acid reduction, you face a significant risk of B12 depletion.
Ask your provider to check B12 levels at baseline and every 3 months for the first year. The injectable B12 in your compounded formulation provides direct supplementation, but the dose may not be sufficient for everyone, particularly those who were already B12-deficient before starting treatment. The appetite suppression timeline for GLP-1 medications gives context for when reduced food intake typically begins affecting nutrient status.
Older adults (65 and over)
Older adults naturally produce less stomach acid and intrinsic factor, making B12 absorption already compromised before adding any medications. The compounded semaglutide with B12 formulation is particularly well-suited for this population because the injectable B12 bypasses age-related absorption issues entirely. The anti-aging peptide guide discusses additional compounds that older adults research for longevity support.
However, older adults may need slower titration. Starting at 0.25 mg for 6 weeks instead of 4, and increasing more gradually, helps prevent excessive nausea and dehydration, which carry higher risks in older populations. The risk of muscle loss during rapid weight loss is also greater in this group, making protein intake and resistance exercise especially important.
People with a history of eating disorders
GLP-1 medications like semaglutide dramatically alter the relationship with food. For people with a history of restrictive eating disorders, this can be a double-edged sword. The appetite suppression can trigger old patterns of under-eating. Monitoring caloric intake, working with a mental health professional, and ensuring adequate nutrition are all critical. The B12 component does not address these psychological considerations.
People transitioning from tirzepatide
If you are switching from tirzepatide to semaglutide, the dosing is not equivalent. Tirzepatide is a dual GIP/GLP-1 receptor agonist with different potency at each receptor. There is no direct milligram-to-milligram conversion. Most providers restart semaglutide at 0.5 mg or 1.0 mg depending on what tirzepatide dose you were taking, then titrate based on response. The semaglutide to tirzepatide dose conversion chart provides guidance for translating between these medications.
Recognizing and addressing B12 deficiency while on semaglutide
Even with injectable B12 in your compounded formulation, deficiency can still develop if your baseline levels were low, if your dose provides insufficient supplementation, or if other medications are depleting your stores faster than the weekly injection can replenish them.
Early warning signs of B12 deficiency
Fatigue that does not improve with rest. That is usually the first sign. Not the "I did not sleep well" kind of tired, but a deep, bone-level exhaustion that makes ordinary tasks feel monumental.
Then come the neurological symptoms. Numbness or tingling in the hands and feet, sometimes described as a "pins and needles" sensation. Difficulty concentrating. Memory that feels foggy. Mood changes, particularly increased irritability or unexplained sadness.
More advanced deficiency produces balance problems, weakness in the legs, a sore and inflamed tongue (glossitis), and in severe cases, megaloblastic anemia where red blood cells become abnormally large and inefficient at carrying oxygen.
Why injectable B12 might not be enough
The typical B12 concentration in compounded semaglutide is 0.5 mg/mL. At lower semaglutide doses, you draw smaller volumes, which means you receive less B12. For example:
At 0.25 mg semaglutide from a 5 mg/mL vial, you only draw 0.05 mL, delivering just 0.025 mg (25 mcg) of B12
At 2.4 mg from the same vial, you draw 0.48 mL, delivering 0.24 mg (240 mcg) of B12
The recommended daily intake of B12 for adults is 2.4 mcg. At the lowest dose, your weekly injection provides roughly 25 mcg, or about 3.6 mcg per day averaged across the week. That exceeds the RDI but may not be enough to correct an existing deficiency or counter the combined effects of semaglutide plus metformin plus age-related absorption decline.
If your provider identifies B12 deficiency despite your compounded formulation, they may prescribe additional B12 supplementation, either through separate injections, high-dose oral supplements (1000 mcg daily, which can overcome absorption limitations through passive diffusion), or sublingual formulations. The sublingual delivery route bypasses the GI tract entirely, similar to how oral tirzepatide attempts to solve the absorption challenge for GLP-1 medications. Patients concerned about broader nutritional deficiencies during aggressive fat loss protocols should request comprehensive nutrient panels, not just B12.
B12 level reference ranges
B12 level (pg/mL) | Status | Action needed |
|---|---|---|
Below 200 | Deficient | Immediate supplementation required |
200 to 300 | Borderline low | Supplementation recommended |
300 to 900 | Normal | Continue current protocol |
Above 900 | Elevated | Likely fine, discuss with provider |
Troubleshooting common problems
Things do not always go according to the chart. Here are the most common issues people encounter with compounded semaglutide with B12 and how to handle them.
Problem: nausea that does not improve after 2 weeks at a new dose
Standard advice says nausea resolves within 1 to 2 weeks of each dose increase. But for some people, it persists. Before asking your provider to reduce the dose, try these interventions:
Eat 5 to 6 small meals instead of 3 larger ones
Avoid lying down for at least 30 minutes after eating
Eliminate fried, fatty, and heavily seasoned foods temporarily
Ginger tea or ginger supplements (250 mg four times daily) have anti-nausea properties
Switch your injection day so the worst nausea falls on a weekend
Ask your provider about ondansetron (Zofran) for breakthrough nausea
If none of these work after 3 weeks, your provider may drop you back to the previous dose for another 4 weeks before attempting the increase again. Some people need 6 to 8 weeks at each dose instead of the standard 4.
Problem: weight loss stalled for more than 4 weeks
Plateaus are normal. But a stall lasting more than a month deserves investigation.
First, verify your dose is correct. Recalculate the units you are drawing against your vial concentration. Drawing errors are more common than people admit, especially with the 5 mg/mL concentration where small unit differences translate to meaningful dose variations.
Second, examine your eating habits. As semaglutide suppresses appetite, some people unconsciously compensate by choosing calorie-dense foods for the smaller amounts they do eat. A handful of nuts here, a piece of cheese there, and suddenly your caloric deficit has disappeared despite eating "less."
Third, check your activity level. Semaglutide reduces metabolic rate along with body weight. Adding or increasing physical activity can reignite a stalled scale.
Fourth, consider a dose increase if you are below the maximum. Your provider can help determine whether moving up is appropriate based on your specific situation and response history.
Problem: the liquid in your vial changed color
Compounded semaglutide with B12 has a characteristic light pink to reddish color from the cyanocobalamin. If the color darkens significantly, becomes cloudy, or develops visible particles, the formulation has likely degraded. Discard it immediately and contact your pharmacy for a replacement.
Minor color variation between batches is normal. But changes within a single vial during use are not.
Problem: missed a dose
If you remember within 5 days of your scheduled dose, take it immediately and resume your regular schedule. If more than 5 days have passed, skip the missed dose entirely and take the next one on your regular schedule. Do not double up. Doubling a semaglutide dose dramatically increases the risk of severe nausea and vomiting.
Problem: you ran out of medication before your refill arrives
Missing one weekly dose is unlikely to cause significant issues. Missing two or more consecutive doses may trigger a return of appetite and could require a brief re-titration period at a lower dose when you resume. If you anticipate gaps in your supply, talk to your provider about medication continuity strategies. Some patients explore alternative pharmacy sources as a backup, though switching pharmacies mid-treatment requires careful vial transition management. Other GLP-1 providers like Thrive, Evolv, and MMIT offer compounded formulations that may help bridge supply gaps.
Problem: anxiety or mood changes after starting semaglutide
Some patients report increased anxiety, mood changes, or irritability during treatment. While semaglutide does not directly cause anxiety in most people, the combination of rapid dietary changes, blood sugar fluctuations, and reduced caloric intake can trigger these symptoms. The relationship between GLP-1 medications and anxiety is still being studied. B12 deficiency itself can also cause mood disturbances, so if your compounded formulation delivers less B12 at lower doses, this could be a contributing factor. Report any persistent mood changes to your provider promptly.
Problem: hair thinning during weight loss
Rapid weight loss from any cause, including GLP-1 therapy, can trigger telogen effluvium, a temporary condition where hair shifts into its resting phase and falls out more than usual. This typically occurs 3 to 6 months after significant weight loss begins and resolves on its own within 6 to 12 months. The GLP-1 hair loss guide covers prevention strategies and treatment options. Adequate protein intake, iron levels, and B12 status all influence hair health during weight loss. Some researchers explore hair growth peptides as a supportive measure during this transition.
Problem: fatigue that does not improve despite B12 in formulation
If the B12 in your compounded semaglutide is not resolving your fatigue, the issue might not be B12 at all. Iron deficiency, inadequate caloric intake, poor sleep quality, dehydration, and thyroid dysfunction can all cause fatigue that mimics B12 deficiency. Ask your provider to run a comprehensive panel including ferritin, thyroid function, and a complete blood count. Some patients explore additional peptides for energy and mood support, though these should complement, not replace, proper medical evaluation. The cognitive support peptide guide discusses compounds that researchers study for mental clarity and focus.
Compounded semaglutide with B12 versus other formulations
The compounding pharmacy market offers several semaglutide formulations beyond the standard B12 combination. Understanding how they compare helps you discuss options with your provider.
Semaglutide with B12 versus semaglutide with niacinamide
Some pharmacies compound semaglutide with niacinamide (vitamin B3) instead of or in addition to B12. Niacinamide supports skin health, energy metabolism, and may help with hair thinning that some patients experience during rapid weight loss. For reference, the tirzepatide with niacinamide guide covers the niacinamide angle in detail for the related GLP-1 medication. The choice between B12 and niacinamide depends on your individual risk factors and nutritional status.
Semaglutide with B12 versus tirzepatide with B12
Tirzepatide with B12 is the other major compounded GLP-1 formulation on the market. Tirzepatide activates both GLP-1 and GIP receptors, which may produce greater weight loss in some patients. The tirzepatide versus semaglutide dosage chart shows that the two medications have completely different dose ranges and titration schedules. They are not interchangeable without provider guidance.
Semaglutide injection versus oral semaglutide
Brand-name oral semaglutide (Rybelsus) exists but requires specific administration protocols, taking it on an empty stomach with minimal water and waiting 30 minutes before eating. The bioavailability of oral semaglutide is only about 1%, meaning you need much higher oral doses to achieve the same blood levels as injections. There is no compounded oral semaglutide with B12 formulation currently available. The injectable versus oral peptide comparison explains why injection remains the preferred delivery method for most peptide therapies.
Semaglutide versus newer GLP-1 compounds
The GLP-1 landscape is evolving rapidly. Retatrutide, a triple-agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, has shown weight loss exceeding 24% in clinical trials. The retatrutide versus semaglutide comparison highlights how triple-agonist therapy may eventually surpass dual and single agonists. Even mazdutide, another dual-agonist, is entering the competitive landscape. And innovative delivery systems like the Onmorlo GLP-1 patches could change how these medications are administered entirely. For now, compounded semaglutide with B12 remains one of the most accessible and well-studied options available. The retatrutide dosage calculator provides early guidance for patients whose providers recommend transitioning to newer compounds.
Cost considerations and maximizing your investment
Compounded semaglutide with B12 typically costs significantly less than brand-name alternatives. Prices vary widely between pharmacies, but most patients pay between $100 and $500 per month depending on the dose, concentration, and vial size.
How to get the most from each vial
Draw accurately. Overfilling your syringe by even a few units adds up over multiple doses. If you consistently draw 12 units instead of 10 from a 5 mg/mL vial, you are receiving 0.6 mg instead of 0.5 mg per week, which means your vial runs out 20% faster.
Minimize waste. Some medication always remains in the vial after the last full dose. This "dead space" is unavoidable with standard syringes but can be reduced by using low-dead-space syringes designed to minimize trapped volume.
Store correctly. A vial that degrades due to improper temperature or light exposure is wasted money. Follow the storage guidelines precisely.
Plan your refills. Calculate exactly when your current vial will run out based on your dose and vial size. Order refills 1 to 2 weeks before your projected empty date. Shipping delays happen. Pharmacy compounding timelines vary. Running out mid-treatment is disruptive and expensive if you need to visit a clinic for a bridge prescription.
Insurance coverage
Most insurance plans do not cover compounded medications. Some flexible spending accounts (FSAs) and health savings accounts (HSAs) will reimburse compounded semaglutide with a valid prescription. Check with your benefits administrator. A letter of medical necessity from your provider can sometimes make the difference. The retatrutide cost guide and peptide therapy cost overview provide context for how compounded semaglutide pricing compares to other peptide therapies on the market.
Nutrition strategies while using compounded semaglutide with B12
Semaglutide reduces how much you eat, but it does not change what you eat. The quality of your reduced caloric intake matters enormously for preserving muscle mass, maintaining energy, and achieving sustainable results.
Protein first: the non-negotiable priority
When appetite is suppressed, every calorie needs to count. Protein should be your top priority at every meal. Aim for 0.7 to 1.0 grams per pound of body weight daily, or as close to that as your reduced appetite allows. This protects against the muscle loss that accompanies all forms of weight loss.
Practical protein sources that work well with reduced appetite include Greek yogurt, eggs, cottage cheese, lean poultry, fish, and protein shakes. Many people on semaglutide find that solid protein sources are easier to tolerate than carbohydrate-heavy meals.
Hydration becomes critical
Semaglutide slows gastric emptying, which can reduce the urge to drink as well as the urge to eat. Dehydration exacerbates constipation, headaches, and fatigue. Aim for at least 64 ounces of water daily, more if you are active.
Foods to emphasize
High-protein, moderate-volume foods that maximize nutrition per bite
Non-starchy vegetables for fiber and micronutrients
Healthy fats in moderate amounts (avocado, olive oil, nuts)
Foods rich in B12 to supplement the injectable dose: meat, fish, eggs, dairy
Iron-rich foods to prevent deficiency during caloric restriction
Collagen-rich bone broth for connective tissue and joint support
Foods to limit
Fried and greasy foods (worsen nausea dramatically)
Carbonated beverages (increase bloating and discomfort)
Extremely high-fiber meals (can cause severe bloating when gastric motility is already slowed)
Alcohol (increases hypoglycemia risk, adds empty calories, worsens GI symptoms)
Large volumes of food at a single sitting (the stomach simply cannot handle it)
Long-term protocol planning
Most people think about compounded semaglutide with B12 as a weight loss medication. It is. But the long-term question is what happens after you reach your goal weight. The answer determines whether you keep the weight off or join the majority who regain.
The maintenance dose strategy
Clinical data from the STEP 4 trial demonstrated that patients who discontinued semaglutide after reaching their weight loss goals regained approximately two-thirds of the lost weight within one year. This finding changed the medical consensus. Semaglutide is now widely viewed as a long-term medication, similar to blood pressure or cholesterol medications, rather than a short-term intervention.
For patients who want to reduce their dose after reaching goal weight, a gradual tapering approach works best. Drop one dose tier every 4 weeks (for example, from 2.4 mg to 1.7 mg, then to 1.0 mg) while monitoring weight. If weight starts creeping up at a lower dose, that tells you the previous dose was your minimum effective maintenance level.
Transitioning between compounded and brand-name
If regulatory changes make compounded semaglutide unavailable, you may need to transition to brand-name Ozempic or Wegovy. The active ingredient is the same, so the transition is straightforward from a pharmacological standpoint. The main adjustments involve:
Switching from vial-and-syringe to a pre-filled pen device
Losing the B12 component (you may need separate B12 supplementation)
Adjusting to fixed dose increments instead of flexible dosing
Potential insurance/cost changes
Discuss transition planning with your provider proactively rather than waiting until your compounding pharmacy can no longer fill your prescription. The latest peptide regulation updates track the evolving legal landscape around compounded GLP-1 medications.
Exercise integration
Semaglutide alone causes weight loss, but a portion of that loss comes from muscle mass. Adding resistance training 2 to 3 times per week significantly shifts the ratio toward fat loss and muscle preservation. This matters for long-term metabolic health, functional capacity, and the visual results most people are seeking.
Start conservatively if you are new to exercise. Reduced caloric intake means reduced recovery capacity. A simple program of compound movements (squats, deadlifts, presses, rows) performed twice weekly is sufficient for most people to maintain muscle mass during active weight loss. For patients dealing with joint issues that limit exercise, some researchers investigate peptides for joint and shoulder pain or injury recovery peptides alongside their weight management protocol. The autoimmune considerations page addresses whether GLP-1 medications interact with immune-modulating peptides.
For researchers serious about optimizing their peptide protocols, SeekPeptides provides the most comprehensive resource available. Members access evidence-based guides including online peptide therapy guidance, detailed dosing calculators for semaglutide and reconstitution math, proven protocols, and a community of thousands who have navigated these exact questions. Whether you are figuring out your first compounded semaglutide dose, comparing affordable compounded GLP-1 options, or optimizing a long-term maintenance strategy, SeekPeptides offers the tools and guidance that generic dosage charts simply cannot provide. Members also access comprehensive resources on peptide regulation updates, legal status, and the true cost of peptide therapy.
Frequently asked questions
Is compounded semaglutide with B12 the same as Ozempic?
No. Ozempic is an FDA-approved brand-name product manufactured by Novo Nordisk. Compounded semaglutide with B12 is a custom formulation prepared by compounding pharmacies. Both contain semaglutide as the active ingredient, but compounded versions also include cyanocobalamin (vitamin B12) and are not FDA-approved. The complete guide to semaglutide with B12 explains the differences in detail.
Can I switch from compounded semaglutide with B12 to brand-name Ozempic or Wegovy?
Yes. Since the active ingredient is the same, transitioning is straightforward. Your provider will match your current semaglutide dose to the appropriate pen strength. The main difference is losing the B12 component, so you may need separate B12 supplementation after switching. Discuss the transition with your prescriber to ensure continuity. The same principle applies when switching between semaglutide and tirzepatide, though that requires dose recalculation since the medications are not milligram-equivalent.
How much B12 am I actually getting with each injection?
The amount varies based on your semaglutide dose and vial concentration. At the standard B12 concentration of 0.5 mg/mL, a patient drawing 0.25 mg semaglutide from a 5 mg/mL vial receives only 0.025 mg (25 mcg) of B12 per week. At 2.4 mg from the same concentration, you receive 0.24 mg (240 mcg). Check the semaglutide dosage calculator to see your specific B12 delivery at each dose tier.
Do I still need to take B12 supplements if my compounded semaglutide already contains B12?
Possibly. The B12 delivered through your compounded formulation may be sufficient for maintaining normal levels, but it depends on your baseline B12 status, other medications you take (especially metformin), your age, and your diet. Ask your provider to check B12 levels at baseline and at 3-month intervals to determine if additional supplementation is needed.
What happens if I accidentally draw too much from my vial?
If you realize the error before injecting, simply squirt the excess back into the vial and redraw the correct amount. If you have already injected an overdose, do not panic, but do contact your provider. A single slightly higher dose is unlikely to cause serious harm, though you may experience more intense nausea and gastrointestinal symptoms. The biggest risk of consistent overdosing is running out of medication before your next refill.
Can I use compounded semaglutide with B12 if I am not overweight?
Semaglutide for weight management is approved for adults with a BMI of 30 or greater (obesity), or 27 or greater (overweight) with at least one weight-related comorbidity. Compounding pharmacies follow similar prescribing criteria. Your provider determines whether you meet the clinical criteria for treatment based on your individual health profile. For people without significant weight to lose who are interested in body composition optimization, other peptides for athletic performance and anti-aging peptides may be more appropriate.
Why does my compounded semaglutide with B12 look pink or red?
The color comes from cyanocobalamin (vitamin B12), which is naturally reddish-pink. This is completely normal. A clear or colorless solution would actually indicate that the B12 is missing or has degraded. Expect a consistent light pink to reddish hue throughout the life of the vial.
How long can compounded semaglutide with B12 stay out of the fridge?
Most formulations tolerate room temperature (below 77 degrees Fahrenheit or 25 degrees Celsius) for limited periods, typically 24 to 72 hours. Extended room temperature exposure accelerates peptide degradation. Always return the vial to the refrigerator immediately after drawing your dose. For detailed storage guidelines, check with your specific compounding pharmacy. The tirzepatide refrigeration guide follows similar principles if you use both medications.
Does compounded semaglutide with B12 cause fatigue?
Some people experience fatigue, particularly during the first few weeks at a new dose. This can result from reduced caloric intake, blood sugar adjustments, or the body adapting to GLP-1 receptor activation. The B12 component is specifically included to help counteract this effect, though the amount delivered at lower doses may be insufficient for people with pre-existing deficiency. If fatigue persists beyond 2 to 3 weeks at a stable dose, ask your provider to check B12, iron, and thyroid levels. The question of whether GLP-1 medications cause tiredness applies across the class, not just to semaglutide.
What is the difference between the semaglutide B12 dosage chart and a compounded semaglutide with B12 dosage chart?
Functionally, they describe the same thing. The semaglutide B12 dosage chart is a general reference for any semaglutide-B12 combination. The term "compounded" specifically refers to the pharmacy-prepared version as opposed to brand-name products. Since brand-name Ozempic and Wegovy do not contain B12, any semaglutide-with-B12 product is, by definition, compounded. The dosing schedules are identical.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your doses stay accurate, your B12 levels stay sufficient, and your progress stay consistent.