Mar 24, 2026

Your immune system is attacking your own body. And now someone suggested tirzepatide might help.
That is a bold claim. The kind of claim that deserves scrutiny, context, and real evidence before anyone makes a decision based on it. Because autoimmune diseases are not simple metabolic problems with tidy pharmaceutical solutions. They are complex, unpredictable, and deeply personal conditions that affect every aspect of daily life.
Yet something interesting is happening in the research world. GLP-1 receptor agonists, medications originally designed for blood sugar management and weight loss, are showing up in immunology journals. Rheumatologists are asking questions about them. And patients with conditions like rheumatoid arthritis, lupus, and inflammatory bowel disease are reporting unexpected changes after starting tirzepatide for weight management. Coincidence? Placebo? Or something worth investigating?
This guide examines everything researchers currently know about tirzepatide and autoimmune conditions. The mechanisms. The evidence. The gaps. The risks. We will look at what the science actually shows, not what social media claims, not what wellness influencers promise, and not what headlines imply. Because when your immune system is the problem, the last thing you need is bad information making it worse. SeekPeptides believes every researcher deserves access to accurate, evidence-based information, especially on topics this nuanced.
What tirzepatide actually is and why autoimmune researchers care
Tirzepatide is a dual agonist. It activates two receptor systems simultaneously: the GLP-1 receptor and the GIP receptor. This dual mechanism separates it from single-target medications like semaglutide and retatrutide, which activate different receptor combinations. The distinction matters because GIP receptors are found on immune cells, and that opens biological doors that single-receptor drugs cannot.
Originally approved for type 2 diabetes management in May 2022 and later for chronic weight management in November 2023, tirzepatide was never designed with autoimmune conditions in mind. Its standard dosing protocol starts at 2.5 mg weekly, escalating to a target range of 5 mg to 15 mg weekly through subcutaneous injection.
So why are autoimmune researchers paying attention?
Three reasons. First, GLP-1 receptors exist on immune cells, including macrophages, lymphocytes, and monocytes. Second, preclinical studies show GLP-1 receptor activation can reduce inflammatory cytokine production. Third, real-world data from large patient populations suggests GLP-1 receptor agonist users may have lower rates of certain inflammatory conditions. Each of these findings, on its own, is merely interesting. Together, they form a pattern that serious researchers cannot ignore.
The inflammation connection between obesity, autoimmunity, and GLP-1
Fat tissue is not just storage. It is an active endocrine organ.
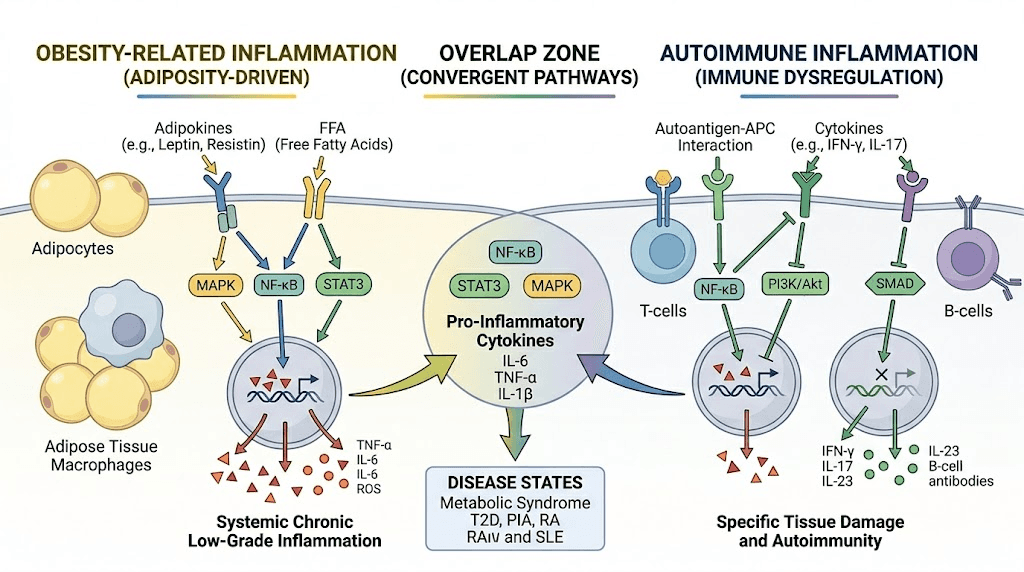
Visceral adipose tissue produces inflammatory cytokines, tumor necrosis factor alpha (TNF-alpha), interleukin-6 (IL-6), and C-reactive protein (CRP) among them. These are the same inflammatory mediators that drive disease activity in rheumatoid arthritis, lupus, psoriasis, and inflammatory bowel disease. When someone carries excess weight AND has an autoimmune condition, they face a compounding problem: obesity-driven inflammation amplifies autoimmune-driven inflammation, creating a feedback loop that standard immunosuppressive therapies struggle to fully address.
Tirzepatide produces significant weight reduction, up to 21% of body weight in clinical trials with patients who have obesity but not diabetes. That level of fat loss alone reduces circulating inflammatory markers substantially. A recent systematic review and meta-analysis confirmed that tirzepatide reduces both hsCRP and IL-6 compared to placebo. These are not trivial findings for someone whose disease activity correlates with systemic inflammation levels.
But here is the critical question. Are tirzepatide anti-inflammatory effects simply a downstream consequence of weight loss? Or does the drug exert direct immunomodulatory effects independent of metabolic changes? The answer appears to be both, though the relative contributions remain unclear.
Direct receptor-mediated immune effects
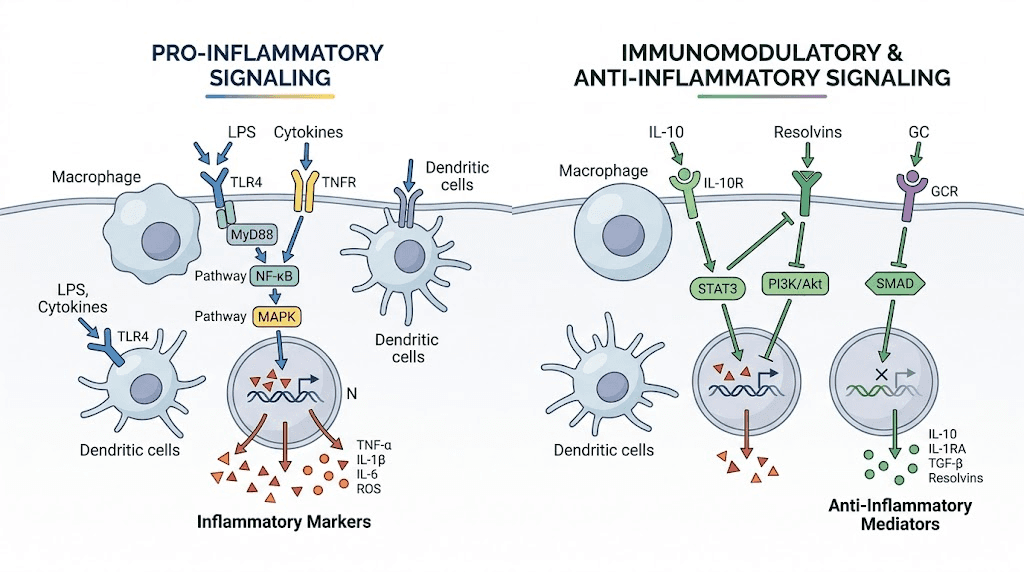
GLP-1 receptor agonists activate the AMP-activated protein kinase (AMPK) pathway and inhibit the nuclear factor-kappa B (NF-kB) signaling pathway. Both pathways are crucial regulators of inflammatory responses. NF-kB is particularly important in autoimmune disease because it controls the expression of genes involved in immune cell activation, cytokine production, and inflammatory cell survival.
Laboratory studies demonstrate that GLP-1 receptor agonists suppress macrophage secretion of multiple inflammatory cytokines including IFN-gamma, IL-17, IL-2, TNF-beta, IL-6, and IL-1beta. Perhaps more significantly, these drugs appear to shift macrophage phenotype toward an anti-inflammatory profile by increasing levels of IL-10, an immunosuppressive cytokine that helps resolve inflammation rather than perpetuate it.
These mechanisms have been documented in cell cultures and animal models. Human clinical data specifically examining tirzepatide direct immune effects in autoimmune disease patients remains limited. That distinction matters enormously.
Indirect metabolic benefits for autoimmune patients
The metabolic improvements that tirzepatide produces extend well beyond simple weight reduction. Improvements in insulin sensitivity, reductions in visceral fat specifically, improvements in lipid profiles, and better glycemic control all contribute to a less inflammatory metabolic environment. For autoimmune patients who also have metabolic syndrome, these changes address a significant contributor to their overall inflammatory burden.
Consider what this means practically. A patient with rheumatoid arthritis who also carries 40 excess pounds of visceral fat is fighting inflammation on two fronts. Reducing that fat burden does not cure the autoimmune condition, but it may lower the baseline inflammatory level enough that existing treatments work more effectively. Some rheumatologists describe this as "debulking the inflammatory load."
What the research actually shows for specific autoimmune conditions
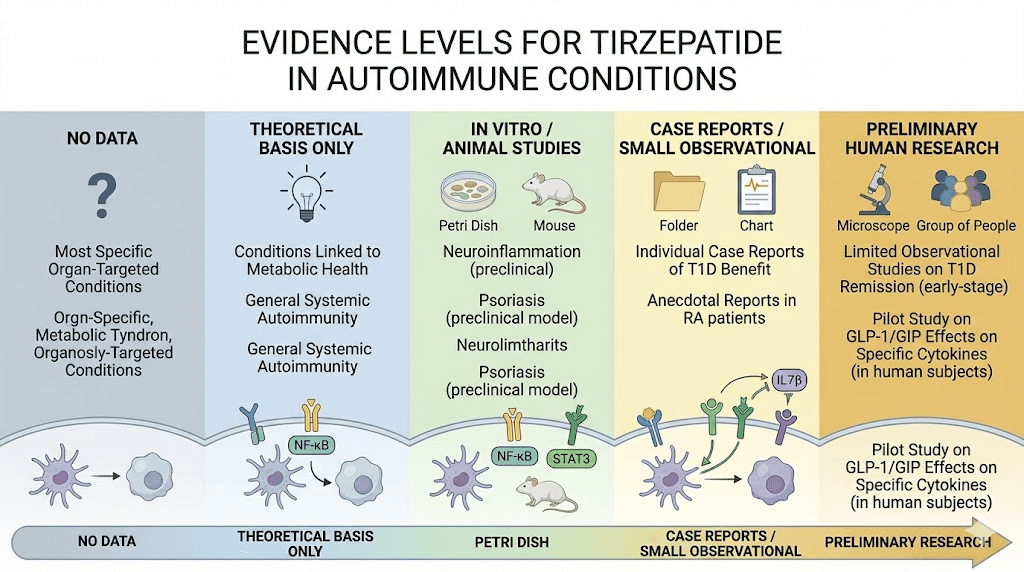
No randomized controlled trials have evaluated tirzepatide specifically for any autoimmune condition. That statement needs to be clear before we discuss what IS known. Everything that follows represents preliminary data, observational findings, preclinical evidence, or extrapolation from related GLP-1 receptor agonist research. Treat it accordingly.
Rheumatoid arthritis
A large multi-center study using the TriNetX database found that GLP-1 receptor agonist use was associated with lower risk of developing immune-mediated inflammatory diseases in patients with type 2 diabetes or obesity. For rheumatoid arthritis specifically, researchers observed that patients on GLP-1 therapy had fewer disease flares compared to matched controls not receiving these medications.
The American College of Rheumatology (ACR) Convergence 2025 conference highlighted emerging research on GLP-1 therapies in rheumatic disease management, noting potential benefits from the anti-inflammatory properties of this drug class. However, rheumatologists uniformly emphasize that these medications should not replace disease-modifying antirheumatic drugs (DMARDs), biologics, or standard treatment protocols.
The mechanism proposed for rheumatoid arthritis benefit is primarily indirect. Weight loss reduces mechanical joint stress. Reduced visceral fat lowers systemic inflammatory cytokines. Improved metabolic health may enhance the efficacy of existing RA medications. Direct GLP-1-mediated suppression of joint inflammation has been suggested by preclinical models but not demonstrated in human RA patients taking tirzepatide.
Inflammatory bowel disease
The relationship between GLP-1 receptor agonists and inflammatory bowel disease is complicated by the gastrointestinal side effects these medications commonly cause. Nausea, diarrhea, constipation, and abdominal discomfort affect 15 to 30 percent of tirzepatide users, especially during dose escalation. For someone with Crohn's disease or ulcerative colitis, distinguishing between medication side effects and disease flares becomes genuinely difficult.
Preclinical data suggests GLP-1 receptor activation may protect intestinal mucosal integrity and reduce intestinal inflammation. Some animal models show improvements in colitis markers with GLP-1 receptor agonist treatment. But translating these findings to human IBD patients taking tirzepatide requires caution. The GI side effect profile of the drug itself could mask, mimic, or potentially worsen IBD symptoms, particularly during the dose escalation period when gastrointestinal complaints peak.
Psoriasis and psoriatic arthritis
Obesity is a well-established risk factor for psoriasis severity. Weight loss through any method, surgical, dietary, or pharmacological, consistently improves psoriasis outcomes. This makes it difficult to determine whether tirzepatide offers any psoriasis benefit beyond what weight loss alone provides.
That said, the NF-kB pathway inhibition demonstrated by GLP-1 receptor agonists is directly relevant to psoriasis pathogenesis. NF-kB drives keratinocyte hyperproliferation and T-cell-mediated skin inflammation. Whether tirzepatide achieves sufficient NF-kB suppression in skin tissue specifically to produce clinically meaningful psoriasis improvement, independent of weight loss effects, remains unknown.
Lupus (systemic lupus erythematosus)
Research on GLP-1 receptor agonists in lupus is extremely limited. Lupus involves autoantibody production, complement activation, and immune complex deposition, pathways that are not directly targeted by GLP-1 or GIP receptor activation. The anti-inflammatory properties of tirzepatide might theoretically reduce some aspects of lupus-related inflammation, but there is no clinical evidence supporting this hypothesis.
Lupus patients considering tirzepatide for weight management should discuss potential interactions with their existing medications, particularly immunosuppressants that affect metabolism or renal function. The weight loss itself may be beneficial for lupus patients with metabolic comorbidities, but attributing any autoimmune benefit specifically to tirzepatide would be premature.
Multiple sclerosis
GLP-1 receptors are expressed in the central nervous system, and some preclinical research suggests neuroprotective effects from GLP-1 receptor agonism. This has generated interest in whether drugs like tirzepatide might influence neuroinflammatory conditions like multiple sclerosis. Animal models of experimental autoimmune encephalomyelitis (EAE), a model for MS, have shown some benefit from GLP-1 receptor agonist treatment.
However, a case report published in the Journal of the American Pharmacists Association documented a patient who developed anti-NMDA receptor autoimmune encephalitis after five weeks of tirzepatide treatment. An 18-year-old patient without prior risk factors presented with seizures, psychiatric symptoms, and speech impairment. Anti-NMDA receptor antibodies were detected in cerebrospinal fluid. The patient required intravenous methylprednisolone and immunoglobulin treatment. This case does not establish causation, but it does warrant attention and further investigation.
Hashimoto's thyroiditis
Hashimoto's thyroiditis is one of the more commonly discussed autoimmune conditions in the context of GLP-1 medications, partly because thyroid function monitoring is already recommended for tirzepatide users due to the medication boxed warning about medullary thyroid carcinoma risk. Researchers have explored whether GLP-1 receptor agonists might influence thyroid autoimmunity, but findings remain preliminary and inconsistent.
Patients with Hashimoto's who are overweight may see improvements in thyroid function simply from weight loss, as excess adiposity can impair thyroid hormone metabolism. Whether tirzepatide provides additional benefit beyond the weight loss effect for thyroid autoimmunity specifically has not been established.
The difference between tirzepatide and microdosing GLP-1 for autoimmune conditions
Some researchers have explored microdosing GLP-1 receptor agonists specifically for anti-inflammatory and immunomodulatory effects, using doses well below the standard weight management range. This approach differs fundamentally from standard tirzepatide use in both rationale and protocol.
Standard tirzepatide dosing (2.5 to 15 mg weekly) primarily targets metabolic endpoints: weight loss, blood sugar control, appetite suppression. Any anti-inflammatory effects are considered secondary. Microdosing protocols use substantially lower doses, sometimes as little as 0.5 to 1 mg weekly, specifically targeting inflammation modulation while minimizing metabolic effects and side effects.
The theoretical basis for microdosing involves the concept that GLP-1 receptor activation at lower levels may preferentially engage anti-inflammatory signaling pathways without strongly activating the appetite suppression and gastric motility changes that cause side effects. This remains a hypothesis. No randomized controlled trials have compared microdosing to standard dosing for autoimmune outcomes.
For autoimmune patients, the microdosing question is practically important. Someone with Crohn's disease might not tolerate standard tirzepatide doses due to GI side effects, but might tolerate microdoses that still provide some anti-inflammatory benefit. This is speculative, however, and should only be explored under medical supervision.
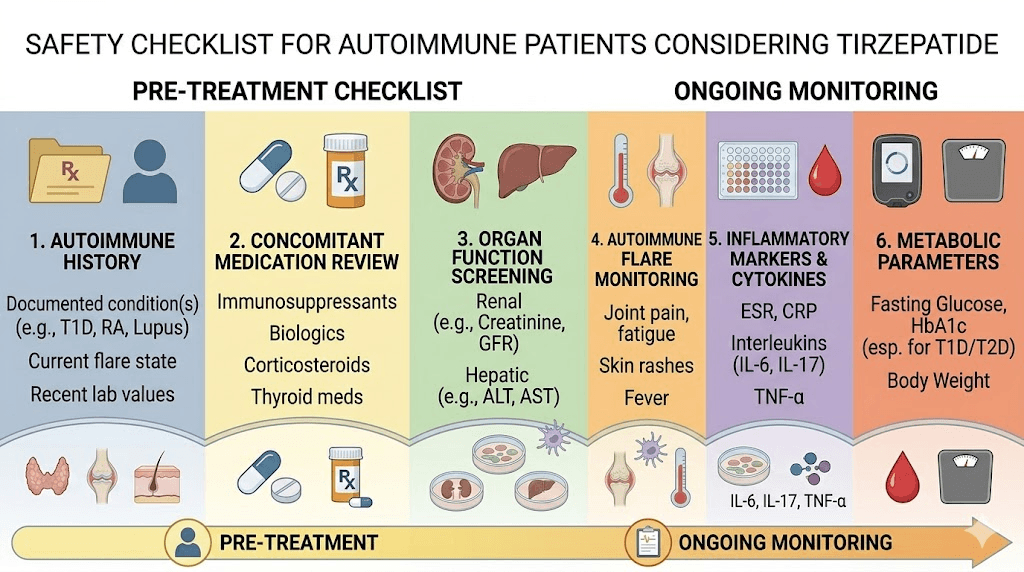
Safety considerations for autoimmune patients on tirzepatide
Taking tirzepatide when you have an autoimmune condition introduces specific safety considerations that healthy individuals do not face. These deserve detailed attention.
Drug interactions with immunosuppressants
Many autoimmune conditions are managed with immunosuppressive medications: methotrexate, azathioprine, mycophenolate, cyclophosphamide, or biologic agents like TNF inhibitors and IL-6 inhibitors. Tirzepatide slows gastric emptying, which can alter the absorption of oral medications. This is not a trivial concern. If methotrexate absorption changes unpredictably, the patient faces either inadequate immunosuppression or toxicity.
No formal drug interaction studies have examined tirzepatide with most immunosuppressive agents. The current recommendation from clinicians is to monitor drug levels more frequently when initiating tirzepatide in patients on immunosuppressants, and to consider separating oral medication timing from tirzepatide injection days.
For patients taking oral medications alongside tirzepatide, the delayed gastric emptying effect may be most pronounced during the first few weeks of treatment and during dose escalations. Planning medication timing around these periods is essential.
Infection risk considerations
Autoimmune patients on immunosuppressive therapy already have elevated infection risk. Whether tirzepatide immune-modulating properties compound this risk or, conversely, provide some protection through anti-inflammatory effects is unknown. The case report of autoimmune encephalitis after tirzepatide use, while isolated, raises the question of whether GLP-1/GIP receptor agonism can occasionally trigger aberrant immune responses rather than suppress them.
Monitoring for unusual infections or immune-related adverse events should be part of any autoimmune patient care plan when starting tirzepatide. Regular blood work to track inflammatory markers, immune cell counts, and autoimmune disease activity markers becomes even more important in this context.
Gastrointestinal autoimmune conditions
Patients with celiac disease, Crohn's disease, ulcerative colitis, or autoimmune hepatitis face particular challenges with tirzepatide. The medication gastrointestinal effects, nausea, bloating, altered bowel habits, and reduced appetite, directly overlap with symptoms of these conditions. This makes disease monitoring more difficult and can lead to delayed recognition of flares.
Additionally, the reduced caloric intake that tirzepatide promotes can be problematic for patients with malabsorptive autoimmune conditions. Someone with active Crohn's who is already struggling with nutritional absorption may not tolerate further appetite suppression and caloric restriction. Careful nutritional monitoring and supplementation planning are essential.
Thyroid autoimmunity concerns
Tirzepatide carries a boxed warning about the risk of thyroid C-cell tumors based on animal studies. For patients with Hashimoto's thyroiditis or Graves' disease, this warning takes on added significance. Thyroid function should be monitored more frequently in autoimmune thyroid patients taking tirzepatide, and any new thyroid nodules or changes in thyroid function should be investigated promptly.
The medication is contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients with multiple endocrine neoplasia syndrome type 2. These contraindications apply regardless of autoimmune status.
Real-world evidence: what population studies reveal
Two large-scale observational studies have examined the relationship between GLP-1 receptor agonist use and autoimmune disease outcomes, and their findings partially conflict.
A multi-center study using the TriNetX database found that GLP-1 receptor agonist use was associated with LOWER risk of developing immune-mediated inflammatory diseases among patients with type 2 diabetes or obesity. This protective association held across multiple autoimmune conditions, including a notably lower risk of noninfectious uveitis, an inflammatory eye condition that affects many autoimmune patients.
However, a separate analysis comparing GLP-1 receptor agonists to DPP-4 inhibitors found that GLP-1 receptor agonist use was linked to INCREASED risks of certain autoimmune conditions. The contradictory findings may reflect differences in comparison groups (comparing to no treatment versus comparing to another diabetes medication), patient populations, or unmeasured confounding variables.
What can we reasonably conclude from this conflicting data? Probably this: GLP-1 receptor agonists, tirzepatide included, are not strongly pro-inflammatory or strongly anti-inflammatory in the context of autoimmune disease. Their effects on autoimmune risk and disease activity are likely modest, variable across conditions, and heavily influenced by individual patient factors including weight, metabolic status, and specific autoimmune diagnosis.
Tirzepatide and autoimmune-related symptoms
Beyond disease activity itself, autoimmune patients often deal with a constellation of symptoms that significantly impact quality of life. Understanding how tirzepatide might affect these symptoms matters for practical decision-making.
Fatigue
Autoimmune-related fatigue is among the most debilitating and difficult-to-treat symptoms patients face. Tirzepatide itself can cause fatigue, particularly during dose escalation and in the early weeks of treatment. For autoimmune patients, this creates a challenging overlap where it becomes difficult to determine whether fatigue is disease-related, medication-related, or both.
On the other hand, some patients report improved energy levels after the initial adjustment period, likely related to weight loss, improved metabolic function, and better sleep quality. The energy effects of tirzepatide appear to be highly individual and may take 8 to 12 weeks to stabilize.
Joint pain
Weight loss reliably reduces mechanical joint stress, which benefits autoimmune arthritis patients. However, some tirzepatide users report joint pain as a side effect, possibly related to rapid body composition changes or dehydration. For someone with rheumatoid arthritis or psoriatic arthritis, distinguishing between disease flare and medication side effect requires careful clinical assessment.
The body aches reported by some tirzepatide users may overlap with the myalgias and arthralgias common in many autoimmune conditions. Keeping detailed symptom logs, including timing relative to injection days, can help identify whether new pain patterns are medication-related.
Brain fog and cognitive symptoms
Cognitive dysfunction, commonly called brain fog, affects patients across multiple autoimmune conditions. GLP-1 receptors are expressed in the brain, and some preclinical evidence suggests GLP-1 receptor agonism may have neuroprotective effects. Whether this translates to improved cognition in autoimmune patients is unknown, but some users report subjective cognitive improvements after stabilizing on tirzepatide, potentially related to improved metabolic health and reduced systemic inflammation.
Skin symptoms
Autoimmune skin conditions like psoriasis, dermatomyositis, and lupus rashes may respond to the systemic anti-inflammatory effects of weight loss and reduced metabolic inflammation. Injection site reactions are reported by some tirzepatide users and should not be confused with autoimmune skin flares. The injection site typically shows localized redness and itching that resolves within 24 to 48 hours, distinct from systemic autoimmune rash patterns.
Who should and should not consider tirzepatide with autoimmune disease
This section requires nuance. Tirzepatide is not approved for autoimmune disease treatment, and it should never be used as a substitute for established autoimmune therapies.
Potentially appropriate candidates
Autoimmune patients who may benefit from tirzepatide are those who independently meet criteria for the medication approved indications. This means patients who have type 2 diabetes requiring better glycemic control, or patients with obesity (BMI 30 or above, or BMI 27 or above with weight-related comorbidities) who need weight management assistance.
For these patients, the potential secondary anti-inflammatory benefits represent a bonus, not the primary rationale. The decision framework should be:
Does this patient need tirzepatide for metabolic reasons? If yes, the possible anti-inflammatory effects make the benefit-risk profile somewhat more favorable for someone who also happens to have an autoimmune condition. If no, prescribing tirzepatide solely for hoped-for autoimmune benefits is not supported by current evidence.
Patients who should exercise particular caution
Several autoimmune patient groups warrant extra caution. Patients with active inflammatory bowel disease, especially those with ongoing flares, may find tirzepatide GI effects intolerable or confusing from a symptom monitoring perspective. Patients on complex immunosuppressive regimens involving multiple oral medications should have potential drug absorption changes carefully monitored. And anyone with a personal or family history of medullary thyroid carcinoma should not use tirzepatide regardless of autoimmune status.
Patients with autoimmune conditions affecting the pancreas, such as type 1 diabetes or autoimmune pancreatitis, require specialized consideration. While tirzepatide has been investigated in type 1 diabetes contexts, its GLP-1 and GIP receptor agonism affects pancreatic function in ways that may interact unpredictably with pancreatic autoimmune processes.
The importance of coordinated care
Autoimmune patients considering tirzepatide should ideally have their prescribing provider coordinate with their rheumatologist, immunologist, or relevant specialist. The endocrinologist or weight management specialist prescribing tirzepatide may not be aware of autoimmune disease nuances, while the rheumatologist may not be familiar with GLP-1 receptor agonist pharmacology. Both perspectives are needed for safe, informed decision-making.
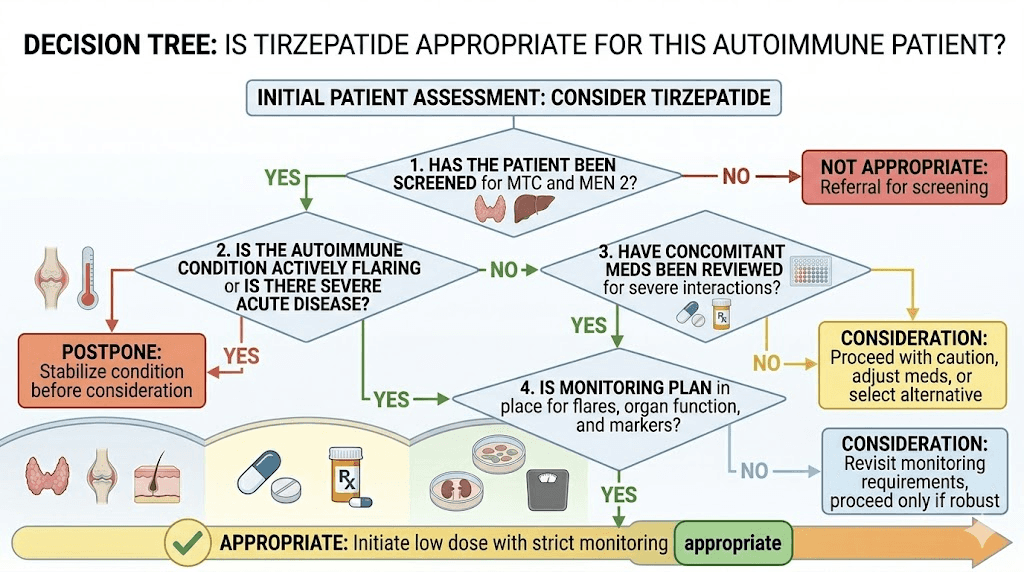
Practical considerations for autoimmune patients using tirzepatide
Starting protocol modifications
The standard tirzepatide starting dose is 2.5 mg weekly for four weeks before escalating. Autoimmune patients, particularly those with GI-affecting conditions or those on immunosuppressants, may benefit from a slower escalation schedule. Some clinicians recommend remaining at each dose level for six to eight weeks rather than the standard four before escalating, allowing more time to assess both metabolic response and any autoimmune disease activity changes.
Starting during a period of stable disease, rather than during a flare, reduces the risk of confounding symptom changes. This timing consideration applies to both the autoimmune condition and any ongoing medication adjustments.
Monitoring recommendations
Beyond the standard tirzepatide monitoring parameters, autoimmune patients should track additional markers. Baseline and periodic checks should include CRP or hsCRP, ESR (erythrocyte sedimentation rate), complete blood count with differential, disease-specific markers (RF and anti-CCP for RA, ANA for lupus, etc.), and any condition-specific monitoring that their specialist recommends.
A reasonable monitoring schedule might be: baseline labs before starting, repeat at 4 weeks, repeat at 12 weeks, then every 3 months thereafter. More frequent monitoring may be warranted during dose escalation periods or if symptoms change.
Nutritional considerations
Autoimmune patients often have specific nutritional needs or restrictions. The appetite suppression caused by tirzepatide can make it challenging to meet protein and micronutrient requirements, which is especially problematic for conditions where malnutrition worsens disease outcomes. Targeted supplementation becomes important.
Key nutritional priorities for autoimmune patients on tirzepatide include adequate protein intake (at least 1.2 to 1.5 g per kg of body weight daily to prevent muscle loss), vitamin D and calcium (particularly important for lupus and RA patients at risk for osteoporosis), B vitamins including B12 supplementation, iron and folate (especially for patients on methotrexate), and omega-3 fatty acids for their independent anti-inflammatory effects.
Knowing which foods to avoid while on tirzepatide helps manage GI side effects, but autoimmune patients should ensure that any dietary restrictions do not compromise their already-elevated nutritional needs.
Exercise and physical activity
Exercise is one of the most consistently beneficial interventions for autoimmune disease management. It reduces inflammation, improves mood, maintains muscle mass, and supports joint health. Tirzepatide's weight loss effects can occur without exercise, but for autoimmune patients specifically, maintaining physical activity during treatment is even more important than for the general population.
The risk of lean mass loss during rapid weight reduction on tirzepatide is a particular concern for autoimmune patients who may already have muscle wasting from disease activity, corticosteroid use, or reduced physical activity during flares. Resistance training and adequate protein intake are the primary strategies for preserving muscle during tirzepatide treatment.
Understanding autoimmune disease mechanisms and where tirzepatide fits
To evaluate tirzepatide potential role in autoimmune contexts, understanding the basic immune dysfunction at play is essential. Autoimmune diseases arise when the immune system loses its ability to distinguish between self and non-self. T cells, B cells, and various innate immune components begin targeting the body own tissues, producing chronic inflammation and progressive tissue damage.
The specific mechanisms vary by condition. Rheumatoid arthritis involves CD4+ T cells and inflammatory macrophages attacking synovial tissue. Lupus features autoantibody production against nuclear components. Multiple sclerosis involves T cell-mediated demyelination of central nervous system neurons. Psoriasis results from IL-17-driven keratinocyte hyperproliferation. Each condition has distinct pathophysiology, which means any medication relevance must be evaluated condition by condition, not as a blanket statement about "autoimmune disease."
Where does tirzepatide fit in this landscape? Its GLP-1 and GIP receptor activation primarily modulates innate immune responses, specifically macrophage polarization and cytokine production. It does not directly address T cell autoreactivity, autoantibody production, or complement-mediated tissue damage. This is why researchers describe tirzepatide potential autoimmune effects as "immunomodulatory" rather than "immunosuppressive," an important distinction that shapes realistic expectations.
The cytokine environment and tirzepatide
Autoimmune diseases are characterized by an imbalanced cytokine environment. Pro-inflammatory cytokines like TNF-alpha, IL-1beta, IL-6, IL-17, and interferon-gamma dominate, while anti-inflammatory cytokines like IL-10, IL-4, and TGF-beta are insufficient to restore immune homeostasis. Many successful autoimmune treatments, including biologics like adalimumab (anti-TNF) and tocilizumab (anti-IL-6), work by directly neutralizing specific pro-inflammatory cytokines.
Tirzepatide's documented effects on this cytokine environment are modest by comparison. Laboratory studies show it reduces macrophage production of several pro-inflammatory cytokines and increases IL-10 production. These effects, if they translate to clinically meaningful changes in human autoimmune patients, would complement rather than replace targeted biologic therapy. Think of it as potentially lowering the background noise of inflammation rather than silencing the specific alarm signals that drive autoimmune attacks.
This analogy helps set appropriate expectations. If your autoimmune condition produces a metaphorical "volume 10" of inflammatory signaling, and tirzepatide turns that down to "volume 8" through metabolic improvement and modest immunomodulation, you still need your disease-specific medications to address the remaining signal. But that lower baseline may mean your existing medications work more effectively, your flares are less severe, and your overall quality of life improves.
The gut-immune axis and GLP-1
An increasingly important concept in autoimmune disease is the gut-immune axis, the relationship between intestinal health, the gut microbiome, and systemic immune function. Approximately 70% of the immune system resides in the gut-associated lymphoid tissue (GALT). Disruptions to intestinal permeability, commonly called "leaky gut," have been implicated in the development of multiple autoimmune conditions including rheumatoid arthritis, type 1 diabetes, multiple sclerosis, and inflammatory bowel disease.
GLP-1 receptors are heavily expressed in the gastrointestinal tract, and GLP-1 receptor agonism appears to influence intestinal barrier function, gut motility, and potentially the gut microbiome composition. Preclinical research suggests that GLP-1 receptor activation may strengthen tight junctions between intestinal epithelial cells, reducing the translocation of bacterial products (like lipopolysaccharide) that trigger systemic immune activation.
For autoimmune patients, this gut-immune connection is particularly relevant. If tirzepatide improves intestinal barrier function while reducing systemic inflammation through weight loss and metabolic improvement, it could theoretically address one of the upstream drivers of autoimmune disease activity. This remains theoretical, but it represents one of the more scientifically plausible mechanisms by which tirzepatide might influence autoimmune outcomes beyond simple weight reduction.
The practical implication is that autoimmune patients starting tirzepatide should pay attention to digestive symptoms not just as side effects to manage, but as potential indicators of gut-immune axis changes. Improvements in bloating, constipation patterns, and overall digestive comfort may correlate with broader immune function changes, though this connection has not been formally studied in clinical trials.
Tirzepatide dosing strategies for autoimmune patients
The standard tirzepatide dosing protocol was designed for metabolic outcomes, not immune modulation. This raises important questions about optimal dosing for autoimmune patients who may prioritize anti-inflammatory effects alongside, or even over, maximum weight loss.
Standard escalation versus conservative approach
The FDA-approved tirzepatide escalation starts at 2.5 mg weekly for four weeks, then increases to 5 mg weekly. From there, doses can be escalated by 2.5 mg increments every four weeks up to a maximum of 15 mg weekly. This schedule prioritizes achieving therapeutic metabolic doses quickly.
For autoimmune patients, several arguments favor a more conservative approach. First, rapid dose escalation increases the likelihood and severity of GI side effects, which are particularly problematic for patients with IBD, celiac disease, or other GI-affecting autoimmune conditions. Second, the immune-modulating effects of tirzepatide likely begin at lower doses than the full metabolic effects, meaning autoimmune patients might achieve meaningful anti-inflammatory benefit at 5 mg or 7.5 mg weekly without needing higher doses. Third, lower doses reduce the magnitude of medication absorption changes that could affect immunosuppressant levels.
A conservative escalation for autoimmune patients might look like this: 2.5 mg weekly for six to eight weeks (instead of four), followed by 5 mg weekly for eight to twelve weeks with careful monitoring of both metabolic and autoimmune markers, then cautious escalation to 7.5 mg only if tolerated and if additional metabolic benefit is desired.
Some researchers exploring microdosing schedules suggest that doses as low as 1 to 2 mg weekly may provide measurable anti-inflammatory effects with minimal metabolic impact and side effects. This approach is entirely off-label and unproven, but it reflects the hypothesis that immune modulation and metabolic effects may have different dose-response curves.
Injection timing and technique for autoimmune patients
The timing of tirzepatide injection may matter more for autoimmune patients than for the general population. Patients who experience significant nausea or appetite suppression after injection may want to time their dose to minimize impact on medication absorption. Taking tirzepatide on a different day than oral immunosuppressants allows medication levels to stabilize before gastric emptying changes peak.
Injection technique also requires attention. Autoimmune patients on corticosteroids may have thinner, more fragile skin that bruises easily. Those with dermatomyositis, scleroderma, or lupus may have skin changes at potential injection sites that require careful site selection. Rotating between abdomen, thigh, and upper arm is standard practice, but autoimmune patients should avoid injecting into areas with active skin disease, fibrotic tissue, or compromised circulation.
Injection site reactions deserve special attention in autoimmune patients. While localized redness and itching at the injection site is common in all tirzepatide users, autoimmune patients may experience more pronounced or persistent reactions due to their altered immune response. Any injection site reaction that persists beyond 48 hours, spreads significantly beyond the injection site, or is accompanied by systemic symptoms should be reported to the healthcare provider.
The role of compounded tirzepatide for autoimmune patients
Some autoimmune patients explore compounded tirzepatide formulations that offer more flexible dosing options. Compounded preparations may allow for increments smaller than the standard 2.5 mg steps, which can be useful for autoimmune patients who need very gradual dose escalation or who want to maintain a specific dose between standard increments.
Compounded formulations sometimes include additional ingredients like vitamin B12, glycine, or niacinamide. For autoimmune patients, these additions deserve scrutiny. B12 supplementation is often beneficial, especially for those on methotrexate. Niacinamide has its own anti-inflammatory properties that may complement tirzepatide effects. However, any additional ingredient introduces another variable in an already complex medication regimen, and autoimmune patients should discuss these additions with their specialist before starting.
Specific autoimmune conditions: deeper analysis
Type 1 diabetes
Type 1 diabetes represents a unique intersection of autoimmune disease and metabolic dysfunction. The autoimmune destruction of pancreatic beta cells eliminates endogenous insulin production, making exogenous insulin essential. Tirzepatide was designed for type 2 diabetes, where insulin resistance rather than autoimmune beta cell destruction is the primary problem.
Research published in the Journal of Clinical Medicine examined the therapeutic potential of tirzepatide in type 1 diabetes and latent autoimmune diabetes in adults (LADA). Understanding the benefits of tirzepatide beyond weight loss is essential for evaluating its role in these complex metabolic-autoimmune conditions. The GLP-1 receptor activation component of tirzepatide may provide some beta cell protective effects, potentially slowing the autoimmune destruction of remaining beta cells in patients with residual beta cell function. However, this effect is far from established, and type 1 diabetes patients require specialized endocrine management that accounts for the fundamentally different pathophysiology of their condition.
For type 1 diabetes patients considering tirzepatide for weight management (which has become more common as insulin therapy-related weight gain is a recognized problem), the primary concern is hypoglycemia risk. Using a dose chart designed for type 2 diabetes does not account for the fundamentally different insulin dynamics in type 1. Tirzepatide reduces appetite and caloric intake while potentially improving insulin sensitivity, which can dramatically alter insulin requirements. Frequent blood glucose monitoring and proactive insulin dose adjustment are essential.
Ankylosing spondylitis
Ankylosing spondylitis (AS) is an autoimmune condition primarily affecting the spine and sacroiliac joints, driven by IL-17 and TNF-alpha-mediated inflammation. Obesity worsens AS outcomes by increasing mechanical spinal load and contributing to systemic inflammation. Tirzepatide's weight loss effects could theoretically benefit AS patients through both mechanisms.
The NF-kB pathway inhibition demonstrated by GLP-1 receptor agonists is directly relevant to AS pathogenesis. NF-kB activation drives the chronic spinal inflammation and new bone formation (syndesmophyte development) that characterizes AS progression. Whether tirzepatide achieves sufficient NF-kB suppression to clinically impact AS activity remains speculative, but the mechanistic plausibility exists.
AS patients considering tirzepatide should be aware that spinal and joint pain changes during treatment may reflect either disease activity changes or the normal musculoskeletal adaptations to significant weight loss. Documenting pain patterns relative to injection timing and weight loss milestones helps distinguish between these possibilities.
Sjogren's syndrome
Sjogren's syndrome, characterized by autoimmune destruction of moisture-producing glands causing dry eyes and dry mouth, has a complex relationship with metabolic health. Some Sjogren's patients report that tirzepatide worsens dry mouth, a symptom they already struggle with. The appetite-suppressing and GI-motility-altering effects of GLP-1 receptor agonists can reduce salivary flow in some patients, potentially exacerbating Sjogren's oral symptoms.
On the other hand, the anti-inflammatory properties of tirzepatide might theoretically reduce the glandular inflammation that drives Sjogren's symptoms. This creates a complex clinical picture where the medication might simultaneously worsen symptoms through one mechanism while potentially improving the underlying disease process through another. Careful symptom tracking and adequate hydration are essential for Sjogren's patients on tirzepatide.
Autoimmune eye conditions
A particularly interesting finding from population studies is that GLP-1 receptor agonist users have a significantly lower risk of developing noninfectious uveitis compared to non-users. This protective effect was observed across subgroups of patients with and without diabetes and across different subtypes of noninfectious uveitis. Uveitis is a common manifestation of several autoimmune conditions including ankylosing spondylitis, Behcet's disease, sarcoidosis, and juvenile idiopathic arthritis.
If this protective association proves causal rather than correlational, it would represent one of the clearest examples of GLP-1 receptor agonism providing direct autoimmune benefit. The eye is an immunologically privileged site with its own regulatory mechanisms, and GLP-1 receptors are expressed in ocular tissues, providing a plausible mechanism for local anti-inflammatory effects.
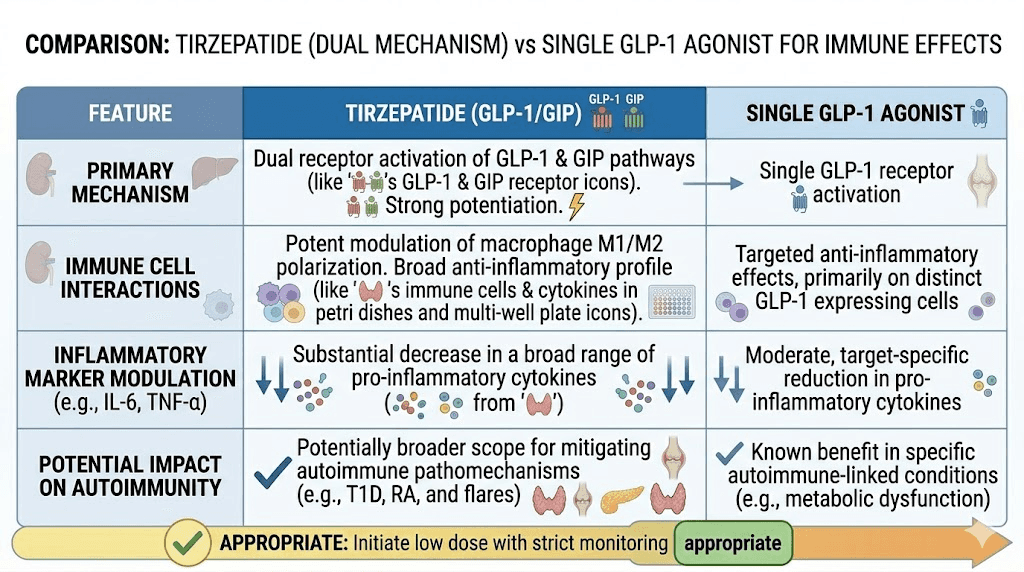
The GIP receptor advantage in autoimmune contexts
One aspect of tirzepatide that distinguishes it from semaglutide and other GLP-1-only agonists is its GIP receptor agonism. GIP receptors are expressed on bone cells, adipocytes, and certain immune cells. The relevance for autoimmune patients includes potential effects on bone density (important for patients on corticosteroids or with conditions like lupus that increase osteoporosis risk) and additional anti-inflammatory pathways that GLP-1-only drugs do not activate.
Research on tirzepatide and bone density is still emerging, but the GIP receptor role in bone metabolism suggests tirzepatide might have a more favorable bone density profile compared to GLP-1-only agonists during weight loss. For autoimmune patients already at risk for bone loss, this could be a clinically meaningful distinction when choosing between available GLP-1/GIP medications.
The dual receptor mechanism also appears to produce somewhat different gastrointestinal tolerability compared to single-receptor drugs. While both tirzepatide and semaglutide cause GI side effects, the specific profile differs, and some patients who do not tolerate one may tolerate the other. For autoimmune patients with GI-affecting conditions, this optionality matters.
Current clinical trials and future research directions
As of early 2026, no active clinical trials are specifically investigating tirzepatide for autoimmune disease treatment. The ClinicalTrials.gov registry shows multiple ongoing tirzepatide studies, but all focus on metabolic endpoints (weight loss, diabetes management, cardiovascular outcomes, obstructive sleep apnea) rather than autoimmune disease activity.
However, several research directions are likely to produce relevant data in coming years. The systematic review and meta-analysis of GLP-1 receptor agonist effects on inflammatory markers continues to grow, with new publications appearing regularly. A 2025 meta-analysis confirmed tirzepatide ability to reduce hsCRP and IL-6, establishing a foundation for future autoimmune-specific investigations.
Large database studies examining autoimmune disease incidence and outcomes in GLP-1 receptor agonist users will continue to emerge, providing increasingly refined estimates of any protective or risk-elevating associations. These studies are valuable because they capture real-world outcomes across diverse patient populations, even though they cannot establish causation.
The most clinically useful research will come from prospective studies that specifically enroll autoimmune patients starting tirzepatide for metabolic indications and track autoimmune disease activity alongside metabolic outcomes. Such studies would provide the first controlled human data on whether tirzepatide meaningfully affects autoimmune disease course in clinical settings.
Comparing tirzepatide to established autoimmune treatments
It bears repeating: tirzepatide is not an autoimmune treatment. Comparing it to methotrexate, biologics, or JAK inhibitors would be misleading. These medications directly target immune pathways involved in autoimmune disease pathogenesis. Tirzepatide does not.
The appropriate comparison is between tirzepatide and other weight management interventions for autoimmune patients who need metabolic support. In this context, tirzepatide offers several advantages over alternatives.
Compared to bariatric surgery, tirzepatide is reversible, does not require anesthesia (important for immunosuppressed patients), and does not permanently alter GI anatomy. Compared to phentermine, tirzepatide does not raise blood pressure or heart rate, both of which are relevant concerns for autoimmune patients with cardiovascular involvement. Compared to orlistat, tirzepatide does not cause fat malabsorption, which could worsen nutritional deficiencies in autoimmune patients.
The potential anti-inflammatory effects of tirzepatide represent an advantage over these alternatives, even if those effects are modest and not fully characterized. For an autoimmune patient who needs meaningful weight reduction, a medication that might also reduce inflammatory burden is preferable to one that does not, all else being equal.
Supplement and lifestyle strategies for autoimmune patients on tirzepatide
Managing an autoimmune condition while taking tirzepatide requires attention to several lifestyle factors that influence both metabolic and immune outcomes. These strategies do not replace medical treatment, but they can optimize results and minimize complications.
Protein optimization to prevent muscle loss
Autoimmune patients face a higher risk of sarcopenia (muscle wasting) than the general population, driven by chronic inflammation, corticosteroid use, and reduced physical activity during flares. Adding tirzepatide caloric restriction effects to this existing vulnerability makes protein optimization absolutely critical.
Research suggests autoimmune patients on tirzepatide should aim for 1.2 to 1.6 grams of protein per kilogram of body weight daily, distributed across at least three meals. This is higher than the general recommendation for tirzepatide users (1.0 to 1.2 g/kg) because of the additive muscle loss risk from autoimmune disease. For a 180-pound (82 kg) patient, this means 98 to 131 grams of protein daily, which can be challenging given reduced appetite.
Practical strategies include prioritizing protein at every eating occasion, using protein shakes or supplements to meet targets when whole food intake is limited, and timing protein intake around resistance training sessions. Quality supplements can bridge the gap between reduced appetite and increased nutritional needs.
Anti-inflammatory dietary patterns
The Mediterranean diet pattern consistently shows benefits for autoimmune disease management and aligns well with tirzepatide dietary recommendations. This eating pattern emphasizes omega-3 fatty acids from fish, monounsaturated fats from olive oil, abundant vegetables and fruits, whole grains, and limited processed foods. These components independently reduce inflammatory markers and complement tirzepatide anti-inflammatory properties.
Certain foods may be particularly beneficial for autoimmune patients on tirzepatide. Fatty fish (salmon, sardines, mackerel) provides omega-3 fatty acids that suppress inflammatory cytokine production through a different pathway than tirzepatide, creating an additive anti-inflammatory effect. Fermented foods (yogurt, kimchi, sauerkraut) support gut microbiome diversity, which may enhance the gut-immune axis benefits of GLP-1 receptor agonism. Turmeric, ginger, and other culinary anti-inflammatories provide complementary mechanisms of inflammatory cytokine suppression.
Conversely, foods to avoid while on tirzepatide, particularly those high in refined sugar, processed oils, and alcohol, are also foods that exacerbate autoimmune inflammation. The dietary overlap between optimal tirzepatide outcomes and optimal autoimmune management is substantial, making nutritional compliance doubly rewarding for this patient population.
Sleep and stress management
Both sleep disruption and chronic psychological stress are potent drivers of autoimmune flares and inflammatory cytokine production. Tirzepatide can affect sleep quality, particularly during dose changes, which creates a vulnerability window for autoimmune patients.
Prioritizing sleep hygiene during tirzepatide treatment is not optional for autoimmune patients. It is therapeutic. Seven to nine hours of quality sleep supports immune regulation, cortisol cycling, and inflammatory marker reduction. For patients experiencing tirzepatide-related sleep disruption, adjusting injection timing (morning versus evening), ensuring adequate hydration, and addressing any medication-related anxiety can help restore normal sleep patterns.
Stress management deserves equal attention. The hypothalamic-pituitary-adrenal (HPA) axis connects psychological stress directly to immune function. Chronic stress elevates cortisol, which initially suppresses immune function but eventually leads to cortisol resistance and rebound inflammation. Mindfulness, moderate exercise, social connection, and adequate rest form the foundation of stress management for autoimmune patients.
Resistance training protocols
Resistance training serves triple duty for autoimmune patients on tirzepatide: it preserves lean mass during weight loss, independently reduces inflammatory markers, and improves joint stability and function. The American College of Sports Medicine recommends two to three resistance training sessions per week, targeting all major muscle groups, for patients with autoimmune conditions.
Autoimmune patients should modify resistance training based on their specific condition. Patients with active joint inflammation should use lighter weights and higher repetitions to avoid exacerbating inflammation. Those with lupus should avoid exercising during peak sun exposure and should monitor for exercise-induced flares. Patients with muscle pain from either their autoimmune condition or tirzepatide should start with bodyweight exercises and progress gradually.
Long-term considerations for autoimmune patients on tirzepatide
Duration of treatment
Metabolic research suggests that tirzepatide weight loss benefits reverse after discontinuation, with most patients regaining significant weight within 12 to 18 months of stopping. For autoimmune patients, this raises a practical question: if tirzepatide anti-inflammatory benefits are primarily driven by weight loss and metabolic improvement, will autoimmune symptoms return when the medication is stopped?
The answer likely depends on whether lifestyle changes, improved diet, regular exercise, and stress management, are maintained after discontinuation. These changes have independent anti-inflammatory effects that persist regardless of medication status. Patients who use the tirzepatide treatment period to establish sustainable lifestyle habits may retain some autoimmune benefit even after stopping the drug, though metabolic-related improvements may diminish.
Decisions about discontinuing or weaning off tirzepatide should be made in consultation with both the prescribing provider and the autoimmune specialist. Abrupt discontinuation is generally not recommended, and autoimmune patients should have a plan for monitoring disease activity during and after the weaning process.
Pregnancy planning
Autoimmune patients of reproductive age face particular challenges when considering tirzepatide. Many autoimmune conditions affect fertility, and many autoimmune medications are teratogenic or require washout periods before conception. Tirzepatide itself should be discontinued at least two months before planned pregnancy due to insufficient safety data.
For autoimmune patients who benefit metabolically from tirzepatide, planning pregnancy requires coordinated timing between optimizing body weight and metabolic health, ensuring autoimmune disease is in remission or well-controlled, washing out tirzepatide with adequate lead time, and transitioning to pregnancy-safe autoimmune medications. The considerations around breastfeeding are equally complex and require specialist guidance.
Monitoring for new autoimmune manifestations
Although rare, the possibility that GLP-1/GIP receptor agonism could trigger new autoimmune manifestations in susceptible individuals has been raised by case reports. Autoimmune patients already have an elevated baseline risk for developing additional autoimmune conditions (a phenomenon called polyautoimmunity), which means they should be particularly vigilant about new symptoms while on tirzepatide.
New symptoms warranting prompt medical evaluation include unexplained rashes, new joint pain patterns, unexpected changes in thyroid function, neurological symptoms (numbness, tingling, vision changes), unexplained cytopenias on blood work, and any symptom suggesting an autoimmune condition they have not previously been diagnosed with. While these are unlikely to be caused by tirzepatide, the theoretical concern warrants awareness.
What autoimmune patients report anecdotally
Online communities and patient forums contain numerous reports from autoimmune patients using tirzepatide, primarily for weight management. These anecdotal reports should be interpreted cautiously, but patterns within them are worth noting.
Common positive reports include reduced joint stiffness and morning pain (likely primarily from weight loss), improved energy levels after initial adjustment, better mood and reduced brain fog, improved sleep quality, and feeling that their autoimmune medications "work better" than before starting tirzepatide. Some patients with psoriasis report improved skin clearance beyond what they would expect from weight loss alone.
Common negative reports include difficulty distinguishing GI side effects from autoimmune flares, insomnia and anxiety during dose adjustments, muscle and body aches that overlap with autoimmune symptoms, and concerns about drug interactions and medication absorption changes.
A minority of patients report what they perceive as autoimmune flares after starting tirzepatide, though it is impossible to determine from anecdotal reports whether these represent genuine medication-triggered flares or coincidental disease fluctuations.
The most consistent pattern in patient reports is that benefits appear gradually, typically emerging after 8 to 16 weeks of treatment as significant weight loss accumulates. This timeline aligns with the hypothesis that most autoimmune improvements are mediated through metabolic changes and reduced adipose-driven inflammation rather than direct immunomodulation, which would theoretically produce faster effects.
Several patients in online communities describe a phenomenon worth noting: their rheumatologist or specialist independently observed improved inflammatory markers on lab work without knowing the patient had started tirzepatide. While this does not constitute clinical evidence, it suggests measurable inflammatory changes that extend beyond subjective symptom reporting.
Common misconceptions about tirzepatide and autoimmune disease
The growing interest in tirzepatide for autoimmune conditions has unfortunately generated misconceptions that deserve direct correction.
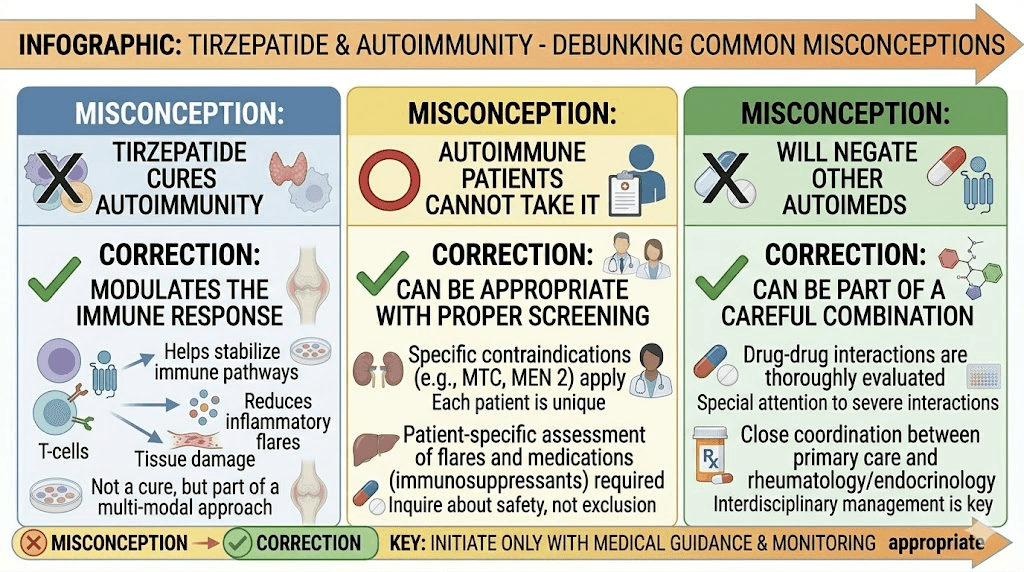
Misconception: tirzepatide is a natural alternative to immunosuppressants
It is not. Tirzepatide does not provide the level of immune suppression needed to control active autoimmune disease. Patients who discontinue their DMARDs, biologics, or other immunosuppressive medications in favor of tirzepatide risk serious disease flares and permanent organ damage. Any anti-inflammatory benefit tirzepatide provides supplements, not replaces, established autoimmune therapy.
Misconception: higher doses provide more autoimmune benefit
There is no evidence for a dose-response relationship between tirzepatide and autoimmune disease activity. Higher doses produce more weight loss and greater metabolic effects, but whether this translates to proportionally greater immune modulation is unknown. In fact, higher doses may be counterproductive for some autoimmune patients due to increased side effect burden and greater disruption of medication absorption.
Misconception: tirzepatide works for all autoimmune conditions equally
Autoimmune diseases are not a single entity. They involve different immune pathways, different target tissues, and different disease mechanisms. A medication relevance to rheumatoid arthritis says nothing definitive about its relevance to type 1 diabetes, multiple sclerosis, or myasthenia gravis. Each condition must be evaluated individually based on its specific pathophysiology and how it might interact with GLP-1/GIP receptor agonism.
Misconception: if tirzepatide reduces CRP, it is treating the autoimmune disease
CRP reduction reflects decreased systemic inflammation, which can result from weight loss, improved metabolic health, and reduced adipose tissue, none of which specifically address autoimmune pathology. Many interventions reduce CRP without affecting autoimmune disease activity. Using CRP reduction as evidence of autoimmune treatment efficacy is a logical error that conflates general inflammation with disease-specific immune dysregulation.
The bottom line for autoimmune patients considering tirzepatide
After examining hundreds of studies, case reports, meta-analyses, and real-world observations, a clear picture emerges. It is nuanced. It requires honesty about what we know and what we do not know. And it demands that we resist the temptation to overstate preliminary findings.
The evidence does not support using tirzepatide as a treatment for autoimmune disease. Full stop.
The evidence does suggest that tirzepatide produces anti-inflammatory effects, both directly through receptor-mediated immune modulation and indirectly through metabolic improvements and weight loss. These effects may provide secondary benefits for autoimmune patients who use tirzepatide for its approved metabolic indications.
For autoimmune patients who independently qualify for tirzepatide treatment based on metabolic criteria, the potential anti-inflammatory properties represent a reason to consider tirzepatide favorably among available GLP-1/GIP medication options. But these properties are not sufficient reason to prescribe tirzepatide to someone who would not otherwise be a candidate.
The critical takeaway: autoimmune patients should not stop, reduce, or modify their immunosuppressive medications based on the hope that tirzepatide will provide autoimmune benefit. The risk of disease flare from inadequate immunosuppression far outweighs any speculative benefit from tirzepatide anti-inflammatory properties. This is not a cautious hedge. It is a clinical reality that patient safety demands we state clearly.
What is genuinely exciting, and worth watching closely, is the growing body of evidence that metabolic health optimization has measurable impacts on autoimmune disease burden. Tirzepatide is one of the most powerful metabolic optimization tools available. For patients who need it for metabolic reasons and who also live with autoimmune conditions, the convergence of benefits represents real clinical opportunity. The key is maintaining perspective: treat the metabolism with tirzepatide, treat the autoimmunity with established therapies, and monitor both rigorously.
For researchers and patients following this space, SeekPeptides provides comprehensive, evidence-based guides covering microdosing GLP-1 for autoimmune conditions, tirzepatide microdosing for inflammation, and the latest research on tirzepatide protocols. As this field evolves, staying informed with reliable sources matters more than following headlines.
Frequently asked questions
Does tirzepatide suppress the immune system?
Tirzepatide is not classified as an immunosuppressant. It modulates certain inflammatory pathways through GLP-1 and GIP receptor activation, reducing production of specific inflammatory cytokines like TNF-alpha and IL-6. However, this is different from the broad immunosuppression caused by drugs like methotrexate or prednisone. The side effect profile does not include increased infection susceptibility at the rates seen with true immunosuppressants.
Can I take tirzepatide with my autoimmune medications?
No formal contraindications exist between tirzepatide and most autoimmune medications. However, tirzepatide slows gastric emptying, which can affect absorption of oral medications. Discuss timing and potential interactions with your healthcare provider. Monitoring drug levels of immunosuppressants may be warranted, especially during tirzepatide dose escalation.
Will tirzepatide cure my autoimmune condition?
No. Tirzepatide has no FDA approval for any autoimmune condition and has not been demonstrated to reverse or cure autoimmune disease in any clinical study. It may provide indirect anti-inflammatory benefits through weight loss and metabolic improvement, but it does not target the fundamental immune dysregulation that drives autoimmune disease.
Is tirzepatide better than semaglutide for autoimmune patients?
There is insufficient evidence to declare one superior for autoimmune patients. Tirzepatide's dual GIP/GLP-1 receptor activation theoretically provides broader anti-inflammatory coverage and may have advantages for bone density preservation. Semaglutide has a longer track record and more published safety data. The choice should be based on metabolic needs, tolerability, and individual clinical factors.
Should I tell my rheumatologist I am considering tirzepatide?
Absolutely. Coordinated care between your metabolic provider and your rheumatologist or immunologist ensures proper monitoring, appropriate medication timing, and informed assessment of any symptom changes. Your specialist needs to know about all medications you are taking to manage your autoimmune condition safely.
Can tirzepatide trigger an autoimmune flare?
Isolated case reports describe immune-related adverse events in tirzepatide users, including one case of autoimmune encephalitis and one case of immune thrombocytopenia. These are extremely rare. However, the possibility that tirzepatide could trigger aberrant immune responses in susceptible individuals cannot be completely excluded. Close monitoring during treatment initiation is advisable for autoimmune patients.
What autoimmune conditions might benefit most from tirzepatide anti-inflammatory effects?
Conditions where obesity-driven inflammation significantly contributes to disease burden, such as rheumatoid arthritis, psoriatic arthritis, and psoriasis, are most likely to show indirect benefit from tirzepatide-induced weight loss. Conditions primarily driven by autoantibody-mediated mechanisms, like lupus or myasthenia gravis, are less likely to see meaningful benefit from tirzepatide anti-inflammatory properties.
How long before I might notice changes in autoimmune symptoms after starting tirzepatide?
Any autoimmune symptom improvement would likely follow the timeline of significant weight loss and metabolic improvement, typically 12 to 24 weeks of treatment. Changes noticed in the first few weeks are more likely related to appetite changes, medication adjustment effects, or coincidental disease fluctuation rather than meaningful anti-inflammatory benefit.
Does tirzepatide interact with methotrexate?
No formal drug interaction study has been conducted between tirzepatide and methotrexate. However, tirzepatide slows gastric emptying, which could theoretically alter oral methotrexate absorption. Patients taking both should discuss monitoring methotrexate levels more frequently, especially during tirzepatide dose escalation. Subcutaneous methotrexate would bypass the gastric emptying concern entirely.
Can tirzepatide help with autoimmune-related weight gain from steroids?
Many autoimmune patients gain significant weight from corticosteroid therapy (prednisone, prednisolone). Tirzepatide can be effective for weight management in steroid-treated patients, though the metabolic environment created by corticosteroids, including insulin resistance, increased appetite, and altered fat distribution, may affect the magnitude of response. Some patients report that tirzepatide partially counteracts steroid-induced appetite increases, though this is anecdotal rather than studied.
Is there a difference between brand-name and compounded tirzepatide for autoimmune patients?
Brand-name tirzepatide (Mounjaro, Zepbound) and compounded tirzepatide contain the same active ingredient. Compounded formulations may offer dosing flexibility advantages for autoimmune patients who need non-standard dose increments. However, brand-name products have undergone rigorous quality testing and clinical trials. The choice should factor in insurance coverage, dosing needs, and individual provider recommendations.
Should I get extra blood tests while on tirzepatide for my autoimmune condition?
Yes. Beyond standard tirzepatide monitoring (A1c, kidney function, lipids), autoimmune patients should track disease-specific inflammatory markers at baseline, 4 weeks, 12 weeks, and quarterly thereafter. This includes CRP or hsCRP, ESR, disease-specific autoantibodies, and complete blood count with differential. Document these alongside weight and metabolic changes to identify correlations between metabolic improvement and autoimmune disease activity.
Can stopping tirzepatide trigger an autoimmune flare?
This has not been studied, but it is theoretically possible. If tirzepatide provides meaningful anti-inflammatory benefit, discontinuation could lead to a rebound in inflammatory markers as weight regain occurs and metabolic health deteriorates. Autoimmune patients planning to stop tirzepatide should taper gradually rather than stopping abruptly, and should have their autoimmune specialist monitor disease activity closely during and after the transition.
External resources
Anti-inflammatory role of GLP-1 receptor agonists and clinical implications (PMC)
Frontiers in Immunology: GLP-1 receptor agonist as modulator of innate immunity
Anti-inflammatory effects of tirzepatide systematic review and meta-analysis (PubMed)
For researchers serious about understanding the intersection of metabolic health and autoimmune disease management, SeekPeptides provides the most comprehensive evidence-based guides available, with detailed protocols, safety information, and a community of thousands who navigate complex health decisions with accurate data rather than speculation.
In case I do not see you, good afternoon, good evening, and good night. May your inflammation stay managed, your protocols stay evidence-based, and your immune system stay balanced.