Feb 24, 2026

Before you use that vial sitting on your counter, stop. Read this first.
Tirzepatide is not a supplement you can toss in a bag and forget about. It is a precision-engineered peptide, a delicate chain of amino acids folded into a specific three-dimensional shape that allows it to bind GLP-1 and GIP receptors in your body. Heat disrupts that shape. Sometimes gradually. Sometimes permanently. And the worst part? You cannot always see the damage.
Thousands of people using tirzepatide for weight loss have found themselves staring at a vial left on the kitchen counter, forgotten in a hot car, or delivered without proper cold packs, asking the same question. Is it still good? Can I still use it? Will it still work?
The answer depends on exactly how warm it got, for how long, and what form of tirzepatide you are using. Pharmaceutical-grade products from Eli Lilly follow strict FDA stability testing. Compounded tirzepatide formulations operate under different rules entirely. Both degrade when exposed to heat, but at different rates and with different consequences.
This guide covers every scenario you might encounter, from a single night on the counter to hours baking in a parked car on a summer afternoon. You will learn the exact temperature thresholds, the biochemistry behind peptide degradation, how to inspect your medication visually, what reduced potency actually means for your results, and when you absolutely must discard a vial versus when it is likely still safe. Whether you are using brand-name Mounjaro, Zepbound, or a compounded tirzepatide formulation, the storage principles matter more than most people realize.
What actually happens to tirzepatide at warm temperatures
Tirzepatide is a 39-amino-acid peptide. Each of those amino acids connects to the next through peptide bonds, and the entire chain folds into a precise configuration that determines how the molecule interacts with receptors in your body. Heat disrupts this configuration through several well-documented chemical pathways. Understanding these pathways helps explain why proper peptide storage is not optional.
It is not like a vitamin pill losing a small percentage of potency. The degradation process involves fundamental changes to the molecular structure.
The science behind peptide degradation
At the molecular level, heat accelerates three primary degradation pathways that affect how peptides work in the body. Each pathway damages the molecule differently, and they often occur simultaneously, compounding the overall loss of function.
Think of tirzepatide like a precisely folded origami crane. The crane only works when every fold is in place. Heat is like crumpling that crane slightly. Maybe the first crumple does not ruin it completely. But each additional crumple makes it less recognizable, less functional, less capable of doing what it was designed to do.
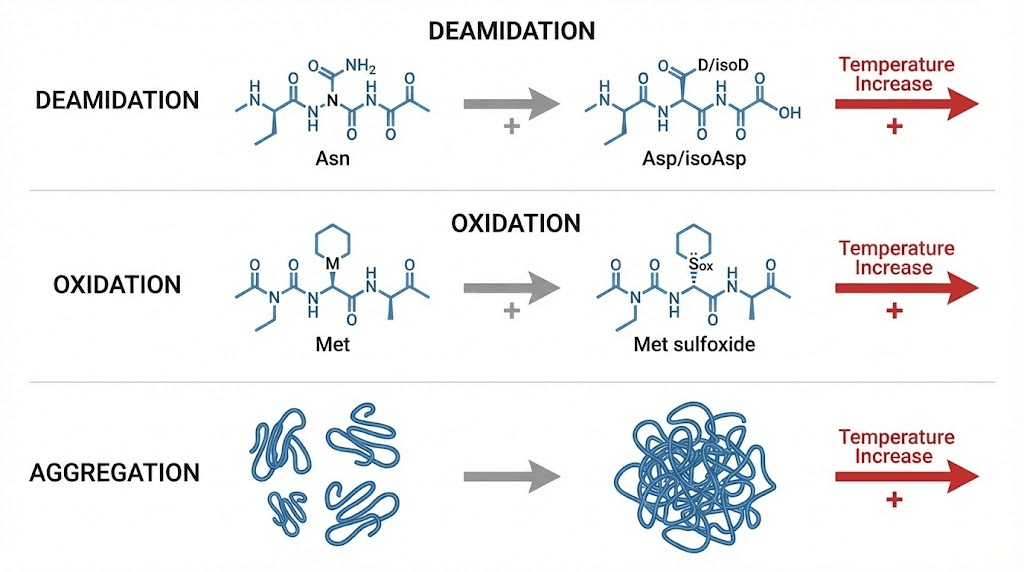
The three primary degradation mechanisms are deamidation, oxidation, and aggregation. Research published through the American Chemical Society and documented extensively in pharmaceutical stability studies shows these reactions accelerate exponentially with temperature increases. A 10-degree Celsius rise in temperature can roughly double the rate of most chemical degradation reactions in peptide formulations.
Deamidation, oxidation, and aggregation explained
Deamidation is the most common degradation pathway for peptide therapeutics. During deamidation, asparagine residues within the peptide chain convert into a mixture of aspartate and isoaspartate. This conversion changes the charge and shape of the molecule, altering how tirzepatide binds to its target receptors. The process is strongly temperature-dependent, accelerating significantly above 30 degrees Celsius. Every hour spent above that threshold chips away at the molecular integrity of the peptide.
Oxidation targets methionine, cysteine, and histidine residues. Heat increases the rate at which these amino acids react with oxygen in the solution, creating oxidized variants that bind receptors poorly or not at all. You cannot reverse this process. Once oxidized, those amino acid residues cannot return to their original state regardless of how carefully you refrigerate the vial afterward.
Then there is aggregation. This is the most visible form of degradation, though it often occurs long after invisible damage has already accumulated. Heat causes individual tirzepatide molecules to unfold slightly, exposing hydrophobic regions that are normally buried inside the folded structure. These exposed regions stick to each other, forming clumps. Small aggregates remain invisible to the naked eye. Larger ones appear as cloudiness or visible particles floating in the solution, a clear sign the medication should not be used.
These three mechanisms do not operate in isolation. Deamidation can trigger aggregation. Oxidation can accelerate deamidation. Heat amplifies all of them simultaneously, creating a cascade of molecular damage that worsens the longer the temperature remains elevated.
Why temperature matters more than you think
Most people underestimate how sensitive peptide medications are to temperature. Small molecules like aspirin or ibuprofen can tolerate significant temperature swings with minimal impact on potency. Peptides are fundamentally different. Their function depends entirely on their three-dimensional structure, and heat disrupts that structure in ways that cannot be undone.
Consider the numbers. At 4 degrees Celsius (standard refrigerator temperature), tirzepatide degradation proceeds so slowly that the medication remains potent for months. At 25 degrees Celsius (room temperature), degradation accelerates measurably. At 30 degrees Celsius (the FDA maximum for temporary storage), the rate increases further. At 40 degrees Celsius (a hot car on a mild day), the reaction rate roughly quadruples compared to refrigerated conditions.
This is why the shelf life of tirzepatide in the fridge extends for months, while the room temperature allowance is only 21 days. The math of chemical degradation does not lie. Higher temperatures mean faster breakdown. Always.
FDA storage guidelines for tirzepatide
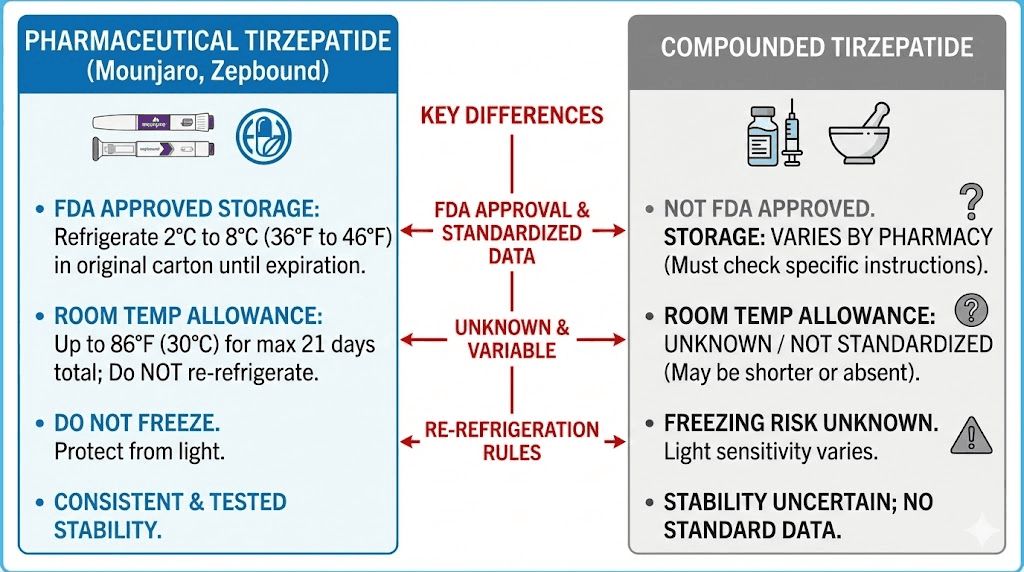
The FDA approved tirzepatide under two brand names, Mounjaro for type 2 diabetes and Zepbound for weight management, both manufactured by Eli Lilly. The tirzepatide refrigeration requirements come directly from stability data submitted during the approval process. These are not arbitrary numbers. They represent the tested boundaries within which the manufacturer guarantees the medication will maintain its stated potency.
Refrigerated storage requirements
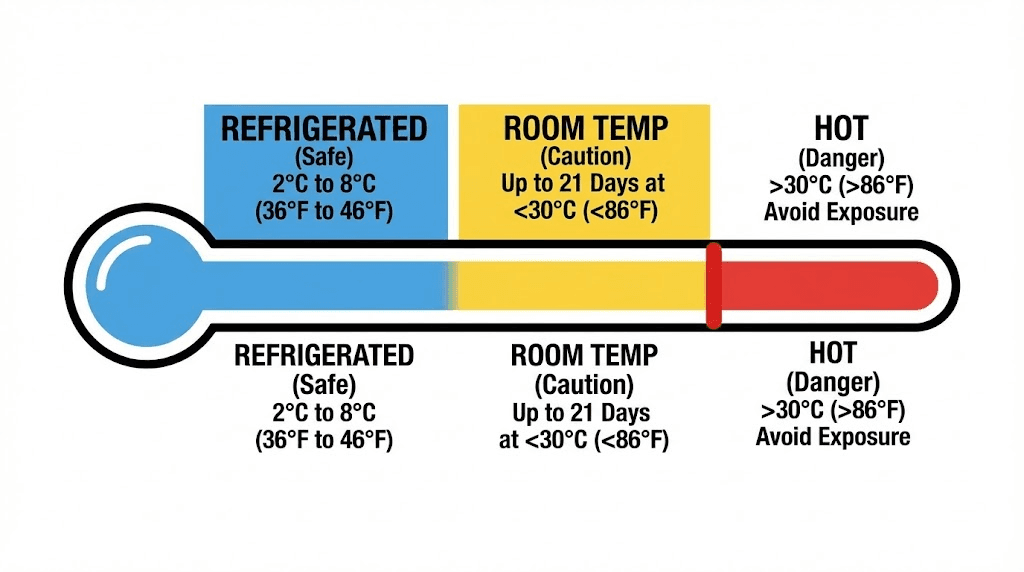
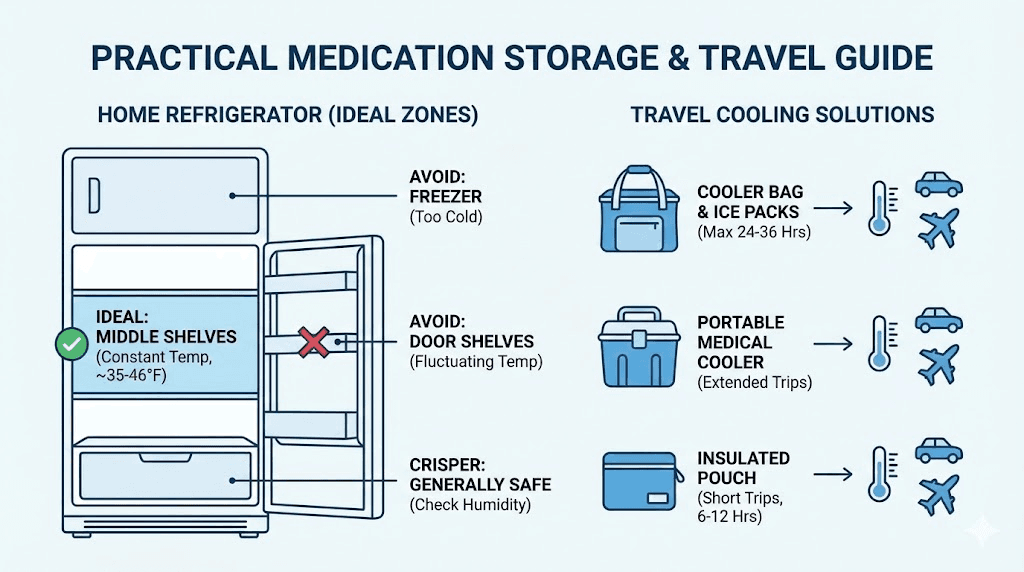
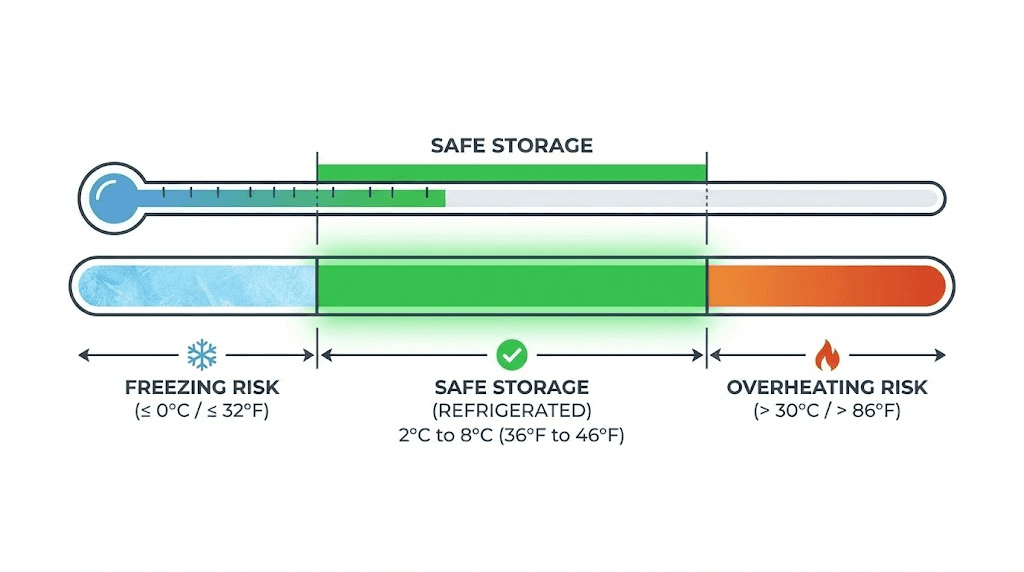
The primary storage recommendation is straightforward. Keep tirzepatide refrigerated between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). This temperature range slows all three degradation pathways to negligible rates, preserving the peptide structure and potency until the printed expiration date.
Store the pens or vials in their original packaging to protect from light exposure, which can trigger photo-oxidation and accelerate breakdown. Keep them away from the freezer compartment. The back of the refrigerator, where temperatures remain most stable, is the ideal location. Door shelves experience the most temperature fluctuation every time you open the fridge, making them a poor choice for medication storage.
At proper refrigerator temperatures, unopened pharmaceutical tirzepatide maintains potency through the manufacturer expiration date, which is typically 24 months from production. This long shelf life reflects the remarkable stability peptides can achieve when stored correctly. Every degree above the recommended range shortens that window.
Room temperature allowances and the 21-day rule
Life does not always cooperate with ideal storage conditions. Eli Lilly tested tirzepatide at controlled room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius) and established a 21-day window during which the medication maintains acceptable potency.
Twenty-one days. Not three weeks and a couple of extra days. Not roughly a month. Exactly 21 days.
Once tirzepatide leaves refrigeration, the 21-day clock starts ticking. The medication must be used within that window or discarded. This timeline applies whether the vial is opened or unopened. And critically, once tirzepatide has been stored at room temperature, do not return it to the refrigerator. Temperature cycling, moving repeatedly between cold and warm environments, can cause condensation and additional stress on the peptide structure.
If you are tracking tirzepatide dosage in units and using a multi-dose vial, mark the date it first left the fridge. Some researchers use a small piece of tape on the vial with the date written clearly. This simple habit prevents guesswork and protects against accidentally using degraded medication.
The 86-degree threshold
Eighty-six degrees Fahrenheit (30 degrees Celsius). That is the absolute ceiling for the 21-day room temperature allowance. Above this threshold, the FDA has not validated stability, and the manufacturer cannot guarantee potency for any specific duration.
To put that in perspective, 86 degrees Fahrenheit is a comfortable spring day in most climates. It is well below the temperature inside a parked car on a summer afternoon (which can reach 130 to 170 degrees Fahrenheit). It is below the temperature of a delivery truck without climate control. It is below the temperature of a kitchen counter near a stove or next to a sunny window.
This threshold matters because many people assume room temperature storage means any indoor environment. It does not. A kitchen that reaches 90 degrees during summer cooking exceeds the limit. A bathroom after a hot shower can exceed it. A bedroom in a home without air conditioning during a heat wave absolutely exceeds it.
What happens above 86 degrees
Above 86 degrees Fahrenheit, tirzepatide enters uncharted territory. The manufacturer has not tested long-term stability at these temperatures, which means there is no validated timeline for acceptable use.
Brief excursions slightly above 86 degrees (say, 88-90 degrees for an hour) likely cause minimal degradation. The chemistry of peptide breakdown is continuous, not binary. There is no magic temperature at which the medication instantly becomes useless. But without stability data, there is also no way to quantify exactly how much potency has been lost.
Extended exposure to temperatures well above 86 degrees, such as sitting in a hot car for several hours, represents a more serious concern. At 100+ degrees Fahrenheit, degradation rates accelerate dramatically. Protein aggregation can begin within hours. Deamidation rates increase four to eight times compared to refrigerated conditions. The practical effect is a medication that may look perfectly normal but delivers significantly less active compound per dose.
If your tirzepatide has been exposed to temperatures above 86 degrees, the safest approach is to discard it and obtain a replacement. The cost of a replacement vial is always less than the cost of weeks of reduced effectiveness from a degraded medication. For those using affordable tirzepatide options, this can feel frustrating, but using compromised medication is worse than the financial loss.
Common scenarios and what to do
Theory is helpful. But what you really need are answers for specific situations. Here are the most common temperature excursion scenarios, ranked from least to most concerning, with clear guidance on what to do in each case.
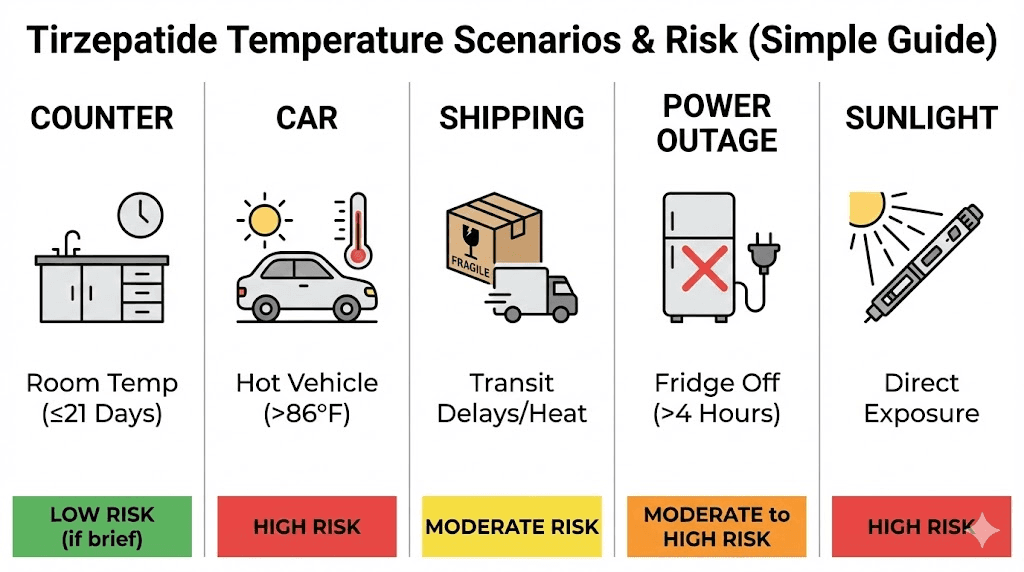
Left out on the counter overnight
This is the most common scenario by far. You pull the vial out of the fridge for your weekly injection, set it on the counter to warm slightly (cold injections can sting more), and then forget to put it back.
If your home is air-conditioned and the indoor temperature stayed below 86 degrees Fahrenheit overnight, the medication is almost certainly fine. One night at typical room temperature (65 to 75 degrees) causes minimal degradation in pharmaceutical tirzepatide. The peptide can handle this without significant potency loss.
Here is what to do. Check the solution visually. It should be clear and colorless to slightly yellow with no particles, cloudiness, or discoloration. If it passes visual inspection, the medication is safe to use. Start counting the 21-day room temperature clock from this point if you plan to keep it out. Otherwise, return it to the fridge, understanding that this single warm-to-cold cycle is acceptable, though you should avoid making it a habit.
The situation changes if your home was not air-conditioned during a hot night. If indoor temperatures exceeded 86 degrees for several hours, you have less certainty about potency. A single hot night likely causes modest degradation, perhaps a few percentage points of potency loss. For most people on standard tirzepatide dosing protocols, this is not enough to notice clinically. But repeated incidents accumulate.
Left in a hot car
This is the scenario that causes the most concern, and rightfully so.
The interior of a parked car in direct sunlight can reach 130 to 170 degrees Fahrenheit within 30 to 60 minutes, even when the outside temperature is only 80 to 90 degrees. These temperatures are catastrophically high for peptide stability. At 130+ degrees, significant degradation can occur within minutes, not hours.
If your tirzepatide spent more than a few minutes in a hot car during summer conditions, discard it. Do not use it even if it looks normal. At these temperatures, the invisible degradation (deamidation, oxidation) begins long before visible signs (cloudiness, particles) appear. You may inject what appears to be a normal dose and receive only a fraction of the active compound.
A cooler day changes the equation. If outside temperatures were moderate (60-75 degrees) and the car was parked in shade, car interior temperatures may have stayed within the 86-degree threshold. In this case, treat it like the counter overnight scenario, inspect visually and use with caution.
For regular travelers, consider how people manage traveling with GLP-1 medications. The same principles apply to tirzepatide. A medical cooling case is a worthwhile investment if you frequently transport your medication.
Shipped without cold packs or inadequate packaging
Online pharmacies and compounding pharmacies ship tirzepatide with cold packs to maintain temperature during transit. But cold packs have limited capacity. In summer heat, they may lose effectiveness within 12 to 24 hours, especially if the package sits on a hot doorstep or in a delivery vehicle without climate control.
If your package arrived warm to the touch with depleted cold packs, assess the situation carefully. How long was it in transit? What were ambient temperatures during shipping? Did the pharmacy use insulated packaging? If transit was overnight in moderate weather, the medication is likely fine. If the package spent two or three days in summer heat, the risk of significant degradation increases substantially.
When in doubt, contact the pharmacy. Reputable pharmacies will replace shipments that arrived outside temperature specifications. Document the warm package with a photo if possible, noting the condition of cold packs on arrival. This is especially relevant for compounded tirzepatide formulations that may have different stability profiles than pharmaceutical products.
Power outage and refrigerator failure
During a power outage, a closed refrigerator maintains safe temperatures for approximately four hours. A closed freezer maintains temperature for 24 to 48 hours depending on how full it is. If the outage lasts less than four hours and you did not repeatedly open the fridge, your tirzepatide should be fine.
Longer outages present a challenge. The refrigerator will gradually warm to room temperature, which triggers the 21-day countdown. If the power returns within a day and your home temperature stayed below 86 degrees, the medication has experienced a manageable excursion. If the outage lasted multiple days during hot weather, the same principles apply as extended room temperature exposure in warm conditions.
Consider keeping a small thermometer in your medication fridge. Some digital models record minimum and maximum temperatures, which provides exact data about how warm the fridge got during an outage. This eliminates guesswork when deciding whether to keep or discard the medication.
Exposed to direct sunlight
Sunlight compounds the temperature problem with an additional concern, photo-degradation. Ultraviolet radiation can directly break peptide bonds and trigger oxidation reactions independent of temperature. A vial sitting on a sunny windowsill faces both heat and UV damage simultaneously.
Even brief exposure to direct sunlight should prompt a careful visual inspection. The normal color of tirzepatide is clear and colorless to slightly yellow. Any shift toward cloudiness, darkening, or visible particles after sun exposure is cause for concern. This is why manufacturer packaging includes opaque cartons, not just for marketing but for light protection.
How to tell if your tirzepatide has gone bad
Visual inspection is your first line of defense, but it has significant limitations. Understanding both what you can see and what you cannot is essential for making informed decisions about medication safety.
Visual inspection checklist
Before every injection, check your tirzepatide against these criteria. Normal tirzepatide solution should be clear. Not hazy. Not cloudy. Completely transparent, like water. It should be colorless or have a very faint yellow tint. This slight yellow color is normal for some formulations and does not indicate degradation.
Look for particles. Hold the vial up to a light source and look through the solution. Tilt it gently. Any floating specks, fibers, or solid material means the medication should not be used. These particles can represent aggregated protein, bacterial contamination, or particulate from a compromised vial.
Check for cloudiness or haziness. Even slight turbidity that was not present when you first received the medication suggests protein aggregation has occurred. This is a definitive sign of degradation. Discard the vial.
Note any color changes. If the solution has become noticeably more yellow, brown, or any color other than clear-to-faintly-yellow, degradation has progressed significantly. This color shift indicates oxidation products forming in the solution.
Inspect the rubber stopper and any visible seals. Compromised seals can allow bacterial contamination in addition to accelerated chemical degradation.
When degradation is invisible
Here is the difficult truth. Most peptide degradation is invisible.
Deamidation does not change the appearance of the solution. A vial can lose 20 to 30 percent of its active tirzepatide through deamidation and look absolutely identical to a fresh vial. Oxidation in its early stages produces no visible changes. Even early-stage aggregation, where molecules form small clusters too tiny to see without analytical instruments, remains invisible to the naked eye.
This means visual inspection, while necessary, is not sufficient. A vial that passes visual inspection may still have experienced significant potency loss. This is why following temperature guidelines matters so much, not because you can detect every problem by looking, but because proper storage prevents the invisible problems that looking cannot catch.
Laboratory testing using high-performance liquid chromatography (HPLC) or mass spectrometry can detect these invisible changes with precision. But these tests are not available to most people using tirzepatide at home. The practical alternative is strict adherence to storage guidelines combined with vigilant visual inspection.
The potency question nobody answers
Everyone wants to know exactly how much potency their warm tirzepatide has lost. The honest answer is that nobody can tell you without laboratory analysis. But we can establish some general principles based on the biochemistry of peptide degradation.
Brief, mild exposure (one night at room temperature in an air-conditioned home) likely results in less than 1 to 2 percent potency loss. This is clinically insignificant. You will not notice any difference in appetite suppression, weight loss timeline, or blood sugar management.
Extended room temperature storage within guidelines (up to 21 days at or below 86 degrees) may result in 5 to 10 percent cumulative degradation, depending on actual temperature conditions. Eli Lilly has validated this range as acceptable for clinical use, meaning the medication still delivers enough active compound to produce therapeutic effects.
Extreme heat exposure (hours in a hot car, extended time above 100 degrees) can cause 20 to 50+ percent potency loss. At this level, the medication may not produce meaningful clinical effects. Someone expecting the appetite suppression of a 5mg dose might effectively be receiving 2.5mg or less, which could explain a sudden plateau in tirzepatide results.
If you suspect your tirzepatide has lost potency due to heat exposure, watch for these practical signs over your next few injections. Reduced appetite suppression compared to previous doses. Less pronounced fullness after meals. A return of cravings that had been absent. Slower or stalled weight loss despite no changes to diet or activity. Any of these could indicate you are receiving a lower effective dose than expected, whether from degradation or from other factors affecting tirzepatide effectiveness.
Compounded tirzepatide versus pharmaceutical tirzepatide and temperature
The storage conversation changes significantly when comparing pharmaceutical (brand-name) products to compounded formulations. Both contain tirzepatide as the active ingredient, but the surrounding formulation, testing standards, and stability profiles differ in important ways.
Why compounded formulations may be more sensitive
Pharmaceutical tirzepatide (Mounjaro and Zepbound) goes through extensive stability testing as part of the FDA approval process. Eli Lilly tests the product at multiple temperatures for months or years, generating precise data about degradation rates under various conditions. This data directly informs the storage guidelines printed on the label.
Compounded formulations do not undergo the same level of testing. Compounding pharmacies assign a Beyond-Use Date (BUD) based on USP guidelines and any available stability data, but this testing is typically less extensive than what pharmaceutical companies perform. The result is that compounded tirzepatide may have shorter validated stability windows and may be more sensitive to temperature excursions.
The excipients (inactive ingredients) in compounded formulations also differ from pharmaceutical products. These excipients can affect peptide stability. Some formulations include stabilizers like niacinamide or glycine that may help maintain peptide structure during mild temperature excursions. Others use simpler formulations that may offer less protection. If you use tirzepatide with niacinamide or tirzepatide with glycine, the added stabilizers may provide a small buffer against temperature stress, though they do not eliminate the need for proper storage.
Beyond-use dates versus expiration dates
The terminology matters. Pharmaceutical products carry an expiration date, which represents the date through which the manufacturer guarantees full potency under specified storage conditions. This date is backed by extensive stability testing.
Compounded products carry a Beyond-Use Date (BUD), which represents the date beyond which the preparation should not be used. BUDs are typically more conservative (shorter) than pharmaceutical expiration dates because the supporting stability data is less comprehensive.
For temperature-exposed compounded tirzepatide, this means even less certainty about remaining potency. If a pharmaceutical product gives you some margin for error within its validated 21-day room temperature window, a compounded product may give you less margin. Some compounding pharmacies specify shorter room temperature allowances for their formulations. Always check the specific storage instructions that came with your compounded medication, as they may differ from the general pharmaceutical guidelines.
Storage considerations for different compounded formulations
Compounded tirzepatide comes in several forms. Subcutaneous injection vials are the most common and follow similar storage principles to pharmaceutical products, with the caveats about shorter validated windows mentioned above.
Tirzepatide oral drops and orally disintegrating tablets may have different stability characteristics than injectable formulations. The delivery format, excipients, and concentration all affect how the peptide responds to temperature stress. Sublingual and oral formulations may include additional preservatives or stabilizers, but the underlying peptide remains equally temperature-sensitive.
Tirzepatide compounded with B12 is another common formulation. The addition of methylcobalamin (B12) can add its own stability considerations, as B12 is light-sensitive and can degrade independently of the tirzepatide component. This makes proper storage even more critical for combination formulations.
Lyophilized (freeze-dried) tirzepatide powder is generally more temperature-stable than reconstituted liquid formulations. If you reconstitute tirzepatide yourself, keep the unreconstituted powder in the fridge but know that it can tolerate brief temperature excursions better than the liquid form. Once reconstituted with bacteriostatic water, the solution becomes significantly more temperature-sensitive and should be refrigerated promptly.
Traveling with tirzepatide in hot weather
Travel is where temperature management gets complicated. You leave the controlled environment of your home refrigerator and enter a world of variable temperatures, limited resources, and situations you did not plan for. Proper preparation prevents most problems.
Cooling cases and travel solutions
A medical-grade cooling case is the single best investment for anyone who travels with tirzepatide. These cases use various technologies to maintain temperature, from simple insulated pouches with gel packs to battery-powered mini-coolers that actively regulate temperature.
For short trips (a few hours to a day), an insulated medication pouch with a partially thawed gel pack works well. The key word is partially thawed. A fully frozen gel pack placed directly against a vial can freeze the medication, which is equally destructive as overheating. Wrap the gel pack in a thin cloth or paper towel and place it in the case with the vial, ensuring no direct contact between ice and medication.
For longer trips, consider a Frio cooling wallet. These wallets use evaporative cooling technology, requiring only water activation to maintain cool temperatures for up to 48 hours. They are lightweight, reusable, and do not require ice or electricity. Many people who manage regular tirzepatide injections during travel rely on these wallets as their primary solution.
For extended travel to hot climates, a battery-powered medication cooler provides the most reliable temperature control. These devices maintain precise temperatures regardless of ambient conditions and can run for 8 to 24 hours on a single charge. They represent a higher upfront investment but offer the most protection for expensive medication.
Flying with tirzepatide
Air travel introduces unique considerations. Always carry tirzepatide in your carry-on luggage. Never check it. The cargo hold of an aircraft can reach temperatures ranging from extreme cold (below freezing at altitude) to significant heat (sitting on a hot tarmac), neither of which is acceptable for peptide storage.
TSA allows injectable medications through security. You do not need to remove them from your bag, though declaring them proactively can speed the screening process. Carry a copy of your prescription or pharmacy label to avoid questions.
During the flight, overhead bins and underseat storage maintain cabin temperature, which stays well within the acceptable range. The risk periods are before and after the flight, moving through hot airports, waiting on a sun-baked tarmac, or leaving medication in a car at the destination airport. Plan for these transitions with your cooling case ready.
International travelers should research destination-specific considerations. Pharmacy availability, hotel refrigerator reliability, and ambient temperatures all factor into planning. Some travelers carry an extra vial as backup in case one is compromised during transit.
Road trips and outdoor events
Road trips present continuous temperature management challenges. A car interior in summer becomes dangerously hot within minutes of parking. Never leave tirzepatide in a parked vehicle, even for a quick stop. Bring your medication with you or keep it in a powered cooler plugged into the vehicle cigarette lighter while driving.
For outdoor events like camping, festivals, or long hikes, plan your medication schedule around access to cooling. If you are due for your weekly injection during a camping trip, time it for when you have access to a cooler. Choosing the best injection timing around travel logistics is a practical skill worth developing.
Beach days require extra caution. Sand reflects heat, shade is limited, and ambient temperatures at the beach often exceed what the thermometer reads in town. A well-insulated cooler with gel packs keeps medication safe, but only if the cooler stays in shade and is not opened repeatedly for drinks and snacks (which warms the interior rapidly).
International travel considerations
Crossing time zones with tirzepatide requires coordinating both injection timing and storage logistics. If you are on a weekly injection schedule, minor adjustments to timing (a day early or late) are generally acceptable. The storage challenge is more critical than the timing question.
Hotels typically provide mini-fridges, which are adequate for medication storage. However, some hotel fridges run warmer than home refrigerators or cycle on and off, creating temperature fluctuations. If possible, set the fridge to its coldest setting and place the medication toward the back. Avoid the door compartment.
In tropical destinations, ambient temperatures may consistently exceed 86 degrees Fahrenheit. This means any time your medication is outside a cooled environment, the degradation clock runs at an accelerated rate. Minimize time outside refrigeration and use a cooling case for all transport.
Temperature tracking and prevention strategies
Prevention is always better than trying to assess damage after the fact. A few simple strategies eliminate most temperature excursion risks and give you confidence that your tirzepatide is maintaining full potency.
Digital thermometers and monitoring tools
A dedicated refrigerator thermometer is a small investment with outsized returns. Place it on the same shelf as your medication to get accurate readings of the actual temperature your tirzepatide experiences, not just what the fridge thermostat claims.
Min/max recording thermometers are even better. These devices continuously track temperature and display the lowest and highest readings since the last reset. After a power outage or if you suspect your fridge malfunctioned, you can check exactly how warm the interior got. This eliminates guesswork.
For travel, adhesive temperature indicators provide a simple yes-or-no answer. These small strips change color irreversibly when exposed to temperatures above a set threshold (typically 86 or 100 degrees Fahrenheit). Place one in your medication bag. If it changes color, your medication was exposed to excess heat regardless of whether you noticed.
Smart thermometers with phone alerts represent the most advanced option. These Bluetooth-connected devices send notifications to your phone if the temperature exceeds a set range, allowing real-time monitoring of your medication storage whether you are home or away.
Setting up a dedicated medication spot in the fridge
Location within the fridge matters more than most people realize. The ideal spot for tirzepatide is on a middle shelf toward the back of the refrigerator. This location offers the most stable temperature because it is farthest from the door (which experiences temperature swings every time you open it) and away from both the cooling element (which can create localized freezing) and the bottom (where condensation collects).
Do not store tirzepatide near raw food, especially on the same shelf. While contamination through sealed packaging is unlikely, separating medications from food is a basic safety practice. Some people dedicate a small container or bin within the fridge specifically for their medication, which also makes it easier to grab quickly and reduces the time the fridge door stays open.
Keep the medication in its original packaging until ready to use. The carton provides light protection and an extra layer of insulation during brief temperature fluctuations. If you use multiple supplements alongside tirzepatide, store each separately and clearly labeled to prevent mix-ups.
Emergency backup plans
What happens when your fridge dies at 2 AM on a Saturday? Having a plan prevents panic decisions.
Know where your nearest 24-hour pharmacy is. Many pharmacies can store medication temporarily in an emergency. Some even offer loaner coolers or gel packs for patients whose refrigeration has failed.
Keep a cooler and gel packs in your freezer at all times. If the fridge fails, immediately transfer your tirzepatide to the cooler with frozen gel packs. This buys you hours of safe storage while you arrange a permanent solution.
If you live in an area prone to extended power outages (hurricane zones, remote areas), consider a small battery-powered cooler dedicated to medication storage. The cost is modest compared to replacing degraded medication after a multi-day outage.
What reduced potency actually means for your results
Understanding the practical implications of degraded tirzepatide helps you make informed decisions about whether to use a vial that may have been compromised. The effects of reduced potency vary depending on how much degradation has occurred and what you are using tirzepatide for.
How degraded tirzepatide affects weight loss
Tirzepatide produces weight loss through several mechanisms, appetite suppression via GLP-1 receptor activation, delayed gastric emptying, and enhanced insulin sensitivity through GIP receptor activation. Each mechanism depends on adequate receptor binding, which requires intact, properly folded tirzepatide molecules.
Mildly degraded tirzepatide (less than 10 percent potency loss) will likely produce weight loss results indistinguishable from fresh medication. The therapeutic window for weight loss is relatively broad, meaning small reductions in effective dose do not translate into noticeably different outcomes. Most people on standard tirzepatide dosing protocols will not notice a difference at this level of degradation.
Moderately degraded tirzepatide (10 to 30 percent potency loss) may produce subtle differences. You might notice slightly less appetite suppression than usual, meals may feel less satisfying sooner, or cravings that had been well-controlled might edge back into consciousness. The speed of tirzepatide results could slow compared to previous doses. These effects are subtle enough that many people attribute them to diet changes, stress, or other factors rather than medication degradation.
Significantly degraded tirzepatide (30+ percent potency loss) produces noticeable effects. Appetite suppression may feel weak or absent. Nausea, if you normally experience it as a side effect, may decrease or disappear (because you are receiving a lower effective dose). Weight loss may stall or even reverse. At this point, you are effectively on a lower dose than prescribed, and the clinical effects reflect that reduction.
Blood sugar implications
For people using tirzepatide for type 2 diabetes management (Mounjaro), potency loss has more immediate clinical significance. Blood sugar control depends on consistent, reliable medication delivery. Even moderate degradation can result in blood glucose readings that creep upward from their established baseline.
Monitor blood glucose more frequently if you suspect your tirzepatide has been compromised by heat exposure. Watch for fasting glucose readings trending higher, post-meal spikes that are larger than usual, or HbA1c values that increase at your next check. These patterns may indicate reduced medication potency rather than disease progression or diet changes.
Contact your healthcare provider if you observe persistent blood glucose elevations after using tirzepatide that may have been heat-exposed. They may recommend replacing the medication, adjusting your dose, or increasing monitoring frequency.
Signs your medication may have lost potency
Because visual inspection cannot detect most degradation, tracking your clinical response provides a valuable secondary assessment. Keep a mental note (or a written log) of how your medication typically affects you, then watch for deviations.
Appetite changes. Tirzepatide normally suppresses appetite significantly, especially at higher doses like 5mg or above. If you notice your appetite returning to pre-medication levels, particularly right after using a vial that may have been heat-exposed, potency loss is a reasonable suspect.
Side effect changes. Paradoxically, a reduction in common tirzepatide side effects like nausea, constipation, or digestive changes after a suspected heat event could indicate reduced potency. Less active drug means fewer side effects.
Weight loss plateau. A sudden stall in weight loss coinciding with suspected medication degradation warrants investigation. While plateaus happen for many reasons, the timing coincidence is worth considering. Check our guide on why tirzepatide stops working for a comprehensive look at all possible causes.
Energy and fatigue. Some users report energy changes with tirzepatide. If your typical energy response changes after suspected heat exposure, degraded medication could be a factor.
The freezing problem and the other temperature extreme
While this guide focuses on heat exposure, freezing deserves attention as the opposite but equally destructive temperature extreme. Many people focus so much on keeping tirzepatide cool that they inadvertently freeze it.
Why frozen tirzepatide cannot be used
Freezing damages peptide formulations through a different mechanism than heat. When the solution freezes, ice crystals form within the liquid. These crystals can physically shear peptide molecules, breaking them apart mechanically rather than chemically. Freeze-thaw cycles are particularly destructive because repeated crystal formation amplifies the damage with each cycle.
The pharmaceutical packaging is clear on this point. Do not freeze tirzepatide. Do not use tirzepatide that has been frozen, even if it has thawed and appears normal. The mechanical damage from ice crystal formation cannot be reversed and may not be visible.
Common freezing scenarios include placing the vial too close to the back wall of the fridge (where temperatures can dip below freezing near the cooling element), packing medication with fully frozen gel packs during travel (where direct contact freezes the solution), and storing medication in a car during winter (where overnight temperatures drop below freezing).
Accidental freezing scenarios and what to do
If you suspect your tirzepatide has frozen, check for telltale signs. A partially frozen vial may have ice crystals visible in the solution, a slushy consistency, or a uniform cloudiness that clears as the solution warms. A fully frozen and thawed vial may appear normal but will have suffered significant molecular damage.
If you are certain the medication froze (ice crystals were visible or the solution was solid), discard it. If you are uncertain (the fridge was very cold but you did not observe ice), the conservative approach is still to discard, but you can also perform a visual inspection after the vial reaches room temperature. If the solution is clear with no particles or cloudiness, and you are not certain it actually froze, it may still be usable. When in doubt, err on the side of caution.
To prevent accidental freezing, avoid the coldest zones in your fridge (usually the back wall and areas directly below the freezer compartment). Use a fridge thermometer to identify these zones. And during winter travel, keep medication close to your body or in an insulated case rather than in a bag exposed to frigid outdoor temperatures.
Understanding the relationship between storage and injection technique
Proper storage is only one part of maintaining tirzepatide effectiveness. How you handle the medication during injection preparation also affects the compound you ultimately receive.
Warming tirzepatide before injection
Many people find that injecting cold tirzepatide causes more discomfort than injecting room temperature medication. The common practice of removing the vial from the fridge 15 to 30 minutes before injection to let it warm slightly is perfectly acceptable. This brief room temperature exposure causes negligible degradation.
Do not attempt to warm tirzepatide quickly using hot water, a microwave, or any heat source. Rapid temperature changes cause more peptide stress than gradual warming. The 15 to 30 minute counter method provides gentle warming without risk. If you prefer not to wait, injecting cold medication is equally effective, just slightly less comfortable at the injection site.
For those learning how to inject GLP-1 medications properly, temperature is just one factor. Injection site selection, technique, and rotation patterns all affect absorption and comfort. Our guide on tirzepatide injection site reactions covers these topics in detail.
Reconstitution and temperature sensitivity
If you use lyophilized (powder) tirzepatide that requires reconstitution, the temperature of your mixing water matters. Use room temperature bacteriostatic water for reconstitution. Cold water slows dissolution and may cause incomplete mixing. Hot water can denature the peptide on contact.
The correct amount of bacteriostatic water affects concentration and, indirectly, stability. More dilute solutions may degrade slightly faster than concentrated ones in some formulations, though this effect is minor compared to temperature. Follow your pharmacy instructions for reconstitution volumes.
After reconstitution, refrigerate the solution promptly. Reconstituted peptide solutions are inherently less stable than lyophilized powder because water enables the chemical reactions (hydrolysis, deamidation) that drive degradation. The clock starts ticking the moment water meets powder, and refrigeration is the primary tool for slowing that clock. Our tirzepatide reconstitution chart provides detailed guidance on proper mixing ratios and storage timelines.
Comparing tirzepatide temperature sensitivity to other GLP-1 medications
If you have used other GLP-1 receptor agonists before switching to tirzepatide, you may wonder how temperature sensitivity compares across medications. The principles are similar, but the specifics differ.
Tirzepatide versus semaglutide storage
Semaglutide (Ozempic, Wegovy) follows similar storage guidelines to tirzepatide, with refrigeration recommended and room temperature storage allowed for limited periods. The question of what happens when semaglutide gets warm has the same fundamental answer, peptide degradation through the same chemical pathways.
However, the specific stability profiles differ because semaglutide and tirzepatide have different molecular structures, different formulation excipients, and different concentrations. Semaglutide pens (Ozempic) allow up to 56 days at room temperature, significantly longer than tirzepatide at 21 days. This reflects differences in the specific formulation and stabilizers used rather than any inherent molecular advantage.
For people switching between tirzepatide and semaglutide, understanding these different storage windows prevents accidentally applying one medication rules to another. Always check the specific storage requirements for whichever medication you are currently using.
Why dual agonists face unique challenges
Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it has structural features for binding two different receptor types. This dual functionality makes the molecule slightly more complex than single-agonist peptides like semaglutide. The additional structural complexity means there are more molecular bonds that can be disrupted by heat, potentially making tirzepatide somewhat more sensitive to temperature excursions than simpler peptides.
This is one reason why tirzepatide has a shorter room temperature allowance than semaglutide. The manufacturer testing revealed faster degradation rates at elevated temperatures, which translated into a more conservative storage guideline. It does not mean tirzepatide is fragile, but it does mean the temperature rules deserve strict adherence.
For researchers interested in comparing tirzepatide with other emerging GLP-1 medications, our guides on survodutide vs tirzepatide, cagrisema vs tirzepatide, and orforglipron vs tirzepatide cover the broader landscape of next-generation weight loss peptides.
Long-term storage optimization for regular tirzepatide users
If you use tirzepatide weekly as part of an ongoing protocol, optimizing your storage routine pays dividends over months and years. Small improvements in handling accumulate into significantly better medication potency over time.
Building a consistent storage routine
Consistency eliminates most temperature excursion risks. Store your tirzepatide in the same fridge location every time. Retrieve it at the same point in your injection preparation routine. Return it to the same spot immediately after drawing your dose. This routine becomes automatic, reducing the chance of accidentally leaving a vial out.
Some people pair their tirzepatide storage with their injection day routine. Every injection day follows the same sequence: retrieve from fridge, let warm 15 minutes, draw dose, inject, return to fridge. This structured approach prevents the most common scenario, pulling out the vial and forgetting to return it because you got distracted.
If your tirzepatide protocol includes dietary planning, some people coordinate medication timing with meal preparation, creating a linked routine that is harder to break or forget. The key is consistency in any system you choose.
Multi-vial management
If you purchase multiple vials at once (common for compounded tirzepatide to save on shipping costs or take advantage of volume pricing), proper storage of backup vials matters. Keep all unopened vials in the fridge in their original packaging. Use them in order, first purchased first used, to ensure the oldest vial does not sit indefinitely.
Label each vial with the purchase date and, for compounded products, the Beyond-Use Date. This prevents accidentally using a vial past its recommended timeline. Some people also note the date each vial was first removed from the fridge (if applicable) to track the room temperature exposure window accurately.
For those managing costs, our guide on affordable compounded tirzepatide covers sourcing strategies that balance cost with quality. Proper storage protects your financial investment by ensuring every vial delivers its full therapeutic value.
Seasonal considerations
Storage challenges change with the seasons. Summer brings heat risk, winter brings freezing risk, and the transitions between seasons bring the less obvious risk of assuming your home temperature is stable when it may not be.
In summer, be aware that home temperatures fluctuate more than you think, especially without air conditioning. A second-floor bedroom can reach 85+ degrees on a hot day even when the main floor feels comfortable. If your fridge is in a garage (common in some homes), summer temperatures in the garage can stress the refrigerator, causing internal temperatures to rise above optimal levels.
In winter, the opposite risk applies. Medication shipped during cold weather can freeze during transit. A vial stored near a poorly insulated wall or in an unheated room can freeze overnight. Even some refrigerators, particularly older models or those set too cold, can create localized freezing conditions.
Spring and fall bring variable conditions that require attention rather than complacency. A warm spring day followed by a cool evening creates exactly the kind of temperature cycling that stresses peptide stability. Maintain vigilance year-round rather than only during extreme seasons.
The real cost of heat-damaged tirzepatide
Beyond the direct cost of replacing a compromised vial, heat-damaged tirzepatide carries hidden costs that people rarely consider.
Weeks of suboptimal results from degraded medication represent wasted time. If you are on a tirzepatide weight loss timeline, two or three weeks of reduced potency can delay your progress meaningfully. That delay cannot be recaptured simply by resuming fresh medication, it is time lost from your overall journey.
The psychological cost also matters. Unexplained plateaus or reduced effectiveness can be demoralizing, especially if you do not realize degraded medication is the cause. People blame their diet, their exercise routine, or their metabolism when the real culprit is a vial that sat too long in a warm room. This misdirected frustration can lead to unnecessary protocol changes, dose adjustments, or even discontinuation of treatment that was actually working, just with a damaged supply.
For those on microdosing tirzepatide protocols, the impact of potency loss is amplified. When your intended dose is already small, losing 20 to 30 percent of that dose to degradation can push you below the therapeutic threshold entirely.
SeekPeptides members access comprehensive storage guides, stability databases, and handling protocols designed to protect their investment in peptide research. Getting the storage right is just as important as getting the dose right, and for serious researchers, the two go hand in hand.
When to absolutely discard tirzepatide
Not every temperature excursion requires discarding your medication. But some situations demand it. Here is a clear framework for making that decision.
Always discard if:
The solution is cloudy, discolored, or contains visible particles
The medication was exposed to temperatures above 100 degrees Fahrenheit for more than 30 minutes
The medication was left in a hot car during summer conditions for any extended period
The medication has been frozen (ice crystals observed or vial was solid)
The total room temperature exposure exceeds 21 days (pharmaceutical) or the pharmacy-specified BUD (compounded)
The medication has passed its expiration date or Beyond-Use Date regardless of storage conditions
You have observed a significant reduction in clinical effects after using doses from the vial
Likely safe to use if:
Left on the counter overnight in an air-conditioned home (below 86 degrees) and visual inspection is normal
Brief exposure (under 30 minutes) to moderate warmth (86-95 degrees) with normal visual inspection
Shipped with depleted cold packs but transit was overnight in moderate weather
Fridge temperature rose slightly during a brief power outage (under 4 hours) but remained below room temperature
Use with caution and monitor closely if:
Extended room temperature storage approaching the 21-day limit
Multiple brief temperature excursions that individually were acceptable but may have accumulated
Storage in a room that may have briefly exceeded 86 degrees but returned to acceptable temperatures quickly
Compounded formulation without clear stability data for the specific temperature exposure
When the decision is borderline, ask yourself one question. Is the potential risk of reduced potency worth the cost of replacement? For most people, the answer is no. A fresh vial eliminates uncertainty entirely.
If you are ever unsure whether your tirzepatide has expired or degraded, err on the side of replacing it. The consequences of using compromised medication, wasted weeks and stalled progress, outweigh the cost of a new vial every time.
Prevention protocols that experienced researchers follow
People who have used tirzepatide long-term develop habits that minimize temperature risks. These practices come from experience, often from learning the hard way what happens when storage is neglected.
The fridge alarm. A small, battery-powered thermometer with an alarm function that sounds if the temperature rises above 46 degrees Fahrenheit. This catches fridge malfunctions, accidental door-ajar situations, and power outages before they compromise medication.
The travel kit. A pre-packed bag containing an insulated cooling case, gel packs (kept in the freezer), and a temperature indicator strip. When travel comes up, the kit is ready. No scrambling to find cooling solutions at the last minute.
The injection day checklist. A simple mental or written checklist: remove from fridge, warm for 15 minutes, inspect visually, draw dose, inject, return to fridge immediately. The checklist prevents the most common human error, getting distracted between injection and refrigerator return.
The backup vial. Keeping one extra vial in the fridge at all times ensures that if a primary vial is compromised, treatment continuity is maintained without gaps. This is especially important for people managing diabetes who cannot afford missed or underdosed injections.
The seasonal review. Twice a year (entering summer and entering winter), experienced users review their storage setup. Is the fridge performing correctly? Are travel cooling supplies in good condition? Are gel packs still sealed and functional? Has anything changed in the home environment that might affect medication storage?
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Can I put tirzepatide back in the fridge after it has been at room temperature?
For brief exposure (a few hours), returning the vial to the fridge is acceptable. For extended room temperature storage (multiple days), the manufacturer recommends against returning it to the fridge due to temperature cycling concerns. If the vial has been at room temperature for more than a day or two, use it within the 21-day room temperature window rather than refrigerating it again. The complete guide to tirzepatide out of the fridge covers this in more detail.
Is tirzepatide that was warm for a few hours still safe to inject?
If the temperature stayed below 86 degrees Fahrenheit and the solution passes visual inspection (clear, colorless to slightly yellow, no particles), it is almost certainly safe. A few hours of moderate warmth causes negligible degradation. The concern increases with higher temperatures and longer durations.
My tirzepatide was delivered warm but the solution looks fine. Should I use it?
Visual appearance alone does not guarantee potency. If the cold packs were completely depleted and the package was warm to the touch, contact the pharmacy. Most reputable pharmacies will replace warm-delivered medications. If you decide to use it, monitor your clinical response carefully over the next few doses.
Does compounded tirzepatide degrade faster than Mounjaro or Zepbound?
It depends on the specific formulation. Compounded products with added stabilizers like glycine and B12 may have comparable stability to pharmaceutical products. Simpler formulations without stabilizers may degrade somewhat faster. Always follow the specific storage instructions from your compounding pharmacy, as they may differ from brand-name guidelines.
What temperature does tirzepatide start to degrade?
Degradation occurs at all temperatures, even in the fridge, just very slowly. The rate increases meaningfully above 25 degrees Celsius (77 degrees Fahrenheit) and becomes a significant concern above 30 degrees Celsius (86 degrees Fahrenheit). The relationship is exponential, meaning each additional degree causes proportionally more damage than the last. Peptide safety considerations always include proper temperature management.
I left my tirzepatide in a hot car for two hours. Is it ruined?
Likely yes, especially during summer. Car interiors can reach 130 to 170 degrees Fahrenheit on hot days, far exceeding the safe threshold. At these temperatures, significant degradation occurs rapidly. Even if the solution looks normal, the invisible damage (deamidation, oxidation) is substantial. Discard and replace.
Can heat-damaged tirzepatide be harmful to inject?
Heat-degraded tirzepatide is generally not toxic, as the degradation products are typically inactive fragments rather than harmful compounds. The primary risk is reduced efficacy rather than adverse effects. However, aggregated protein (visible as particles or cloudiness) should never be injected, as it can cause injection site reactions or immune responses. Always discard visibly compromised medication.
How do I know if my fridge is cold enough for tirzepatide?
Use a refrigerator thermometer placed directly next to your medication. The reading should consistently show 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Check it weekly and after any suspected temperature events (power outages, door left open). Most home refrigerators run within this range when properly maintained, but older units or those with failing seals may run warmer than expected. For proper peptide storage after reconstitution, consistent fridge temperature is essential.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your storage habits stay consistent.