Feb 8, 2026

Tired of conflicting information about switching from tirzepatide to semaglutide? You are not alone. Thousands of people face this transition every month, and the advice they receive ranges from confusing to contradictory. Some providers say wait two weeks. Others suggest starting immediately. One forum tells you to match doses. Another warns you will regain weight. Here is what actually works, backed by pharmacology and real-world experience.
The truth is that switching between these two GLP-1 receptor agonists is more common than most people realize. Insurance changes, supply shortages, side effect management, and cost considerations drive many to make this transition. And while tirzepatide and semaglutide share similarities, they are not identical medications. Understanding the differences between their mechanisms, half-lives, and dosing protocols will determine whether your switch is smooth or rocky.
This guide walks you through the entire switching process. You will learn when to make the transition, how to dose semaglutide based on your current tirzepatide dose, what side effects to expect, and how to maintain your weight loss results during the changeover.
We cover the pharmacokinetics that determine timing, the titration schedules that minimize side effects, and the practical strategies that keep you on track. SeekPeptides members have navigated this transition thousands of times, and this guide distills the strategies that actually work into a clear, actionable protocol.
Why people switch from tirzepatide to semaglutide
The decision to switch from tirzepatide to semaglutide typically stems from practical rather than medical reasons. Understanding these motivations helps contextualize the transition strategy. Most people who switch are not doing so because tirzepatide failed them. They switch because external factors force their hand or because specific side effects become intolerable.
Insurance coverage represents the most common driver. Many insurance plans covered compounded tirzepatide during the FDA shortage period but discontinued coverage once the shortage resolved in December 2024. When brand-name Mounjaro costs exceed one thousand dollars monthly and peptide therapy costs for compounded versions become less favorable, switching to a more affordable option becomes necessary. Some people find that their insurance will cover semaglutide injections or even the oral form of semaglutide while excluding tirzepatide entirely.
Supply and availability issues create another switching scenario. The FDA compounding enforcement actions have made accessing tirzepatide more complicated for some patients. When your provider can no longer source reliable compounded tirzepatide, switching to semaglutide may be your only option for continuing GLP-1 therapy. This situation has become more common as regulatory oversight tightens around compounded weight loss medications.
Side effect management drives a smaller but significant group of switchers. While tirzepatide generally produces superior weight loss results compared to semaglutide, its dual mechanism as both a GLP-1 and GIP receptor agonist can amplify gastrointestinal side effects for some individuals. Persistent nausea, vomiting, or severe constipation that does not resolve after several months may prompt a switch to the single-mechanism semaglutide. Some people find that tirzepatide side effects like fatigue become problematic enough to warrant trying an alternative.
Provider preference occasionally influences the decision. Some healthcare providers have more experience prescribing semaglutide due to its longer market presence. Others may prefer semaglutide because of its availability in oral form for patients who develop needle anxiety or injection site reactions. The option to use oral versus injectable peptides can be appealing for certain patients, though injectable forms typically demonstrate better efficacy for weight loss.
Weight loss plateau is rarely a valid reason to switch from tirzepatide to semaglutide, since tirzepatide typically produces greater weight reduction. However, some people switch in the opposite direction after starting with semaglutide. If you are considering switching due to plateau, exploring why you are not losing weight on your current medication may reveal other factors at play.
Cost considerations extend beyond insurance coverage. Even with insurance, copays for brand-name tirzepatide often exceed those for semaglutide. Compounded versions of both medications vary significantly in price depending on your source and pharmacy. Using a peptide cost calculator can help you compare actual monthly expenses for each option. Many people discover that switching to semaglutide reduces their monthly expenditure by two hundred to five hundred dollars.
Understanding the key differences between tirzepatide and semaglutide
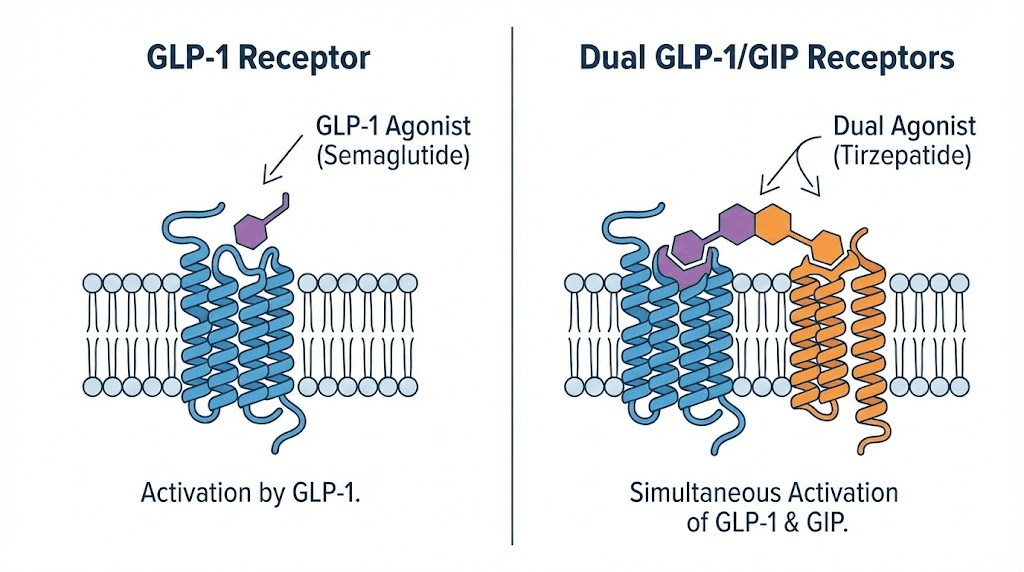
The fundamental distinction between these medications lies in their receptor targets. Semaglutide acts solely as a GLP-1 receptor agonist. It mimics the naturally occurring GLP-1 hormone that your intestines produce after eating. This hormone slows gastric emptying, increases insulin secretion, and suppresses appetite through both peripheral and central mechanisms.
Tirzepatide operates differently. It is a dual agonist that stimulates both GLP-1 and GIP receptors. The glucose-dependent insulinotropic polypeptide receptor activation adds another dimension to its metabolic effects. This dual mechanism typically translates to greater weight loss but also potentially more intense side effects. Understanding this distinction is crucial because it affects how your body will respond when you transition between medications.
The pharmacokinetic profiles differ in meaningful ways. Tirzepatide has a half-life of approximately five days, meaning that half of the medication clears from your system every five days. Complete clearance takes roughly twenty-five days, or five half-lives. Semaglutide has a slightly longer half-life of approximately seven days, reaching steady-state concentrations in your body after four to five weeks of consistent dosing. These differences influence when you should start your new medication after stopping the old one.
Peak plasma concentration timing also varies. Tirzepatide reaches peak levels between eight and seventy-two hours after injection, with most people experiencing peak effects around twenty-four to forty-eight hours post-injection. Semaglutide typically peaks between one and three days after injection. This timing affects when you might experience maximum appetite suppression and when side effects are most likely to occur. Understanding these patterns helps you plan your injection schedule and anticipate your body response.
Efficacy data from clinical trials shows that tirzepatide generally produces superior weight loss outcomes. The SURMOUNT-1 trial demonstrated that participants on the highest tirzepatide dose lost approximately twenty-two percent of their body weight over seventy-two weeks. The STEP trials for semaglutide showed weight loss of approximately fifteen percent at the highest dose. This difference matters when you switch, as you may experience slightly reduced appetite suppression initially, though proper titration typically maintains most of your weight loss progress.
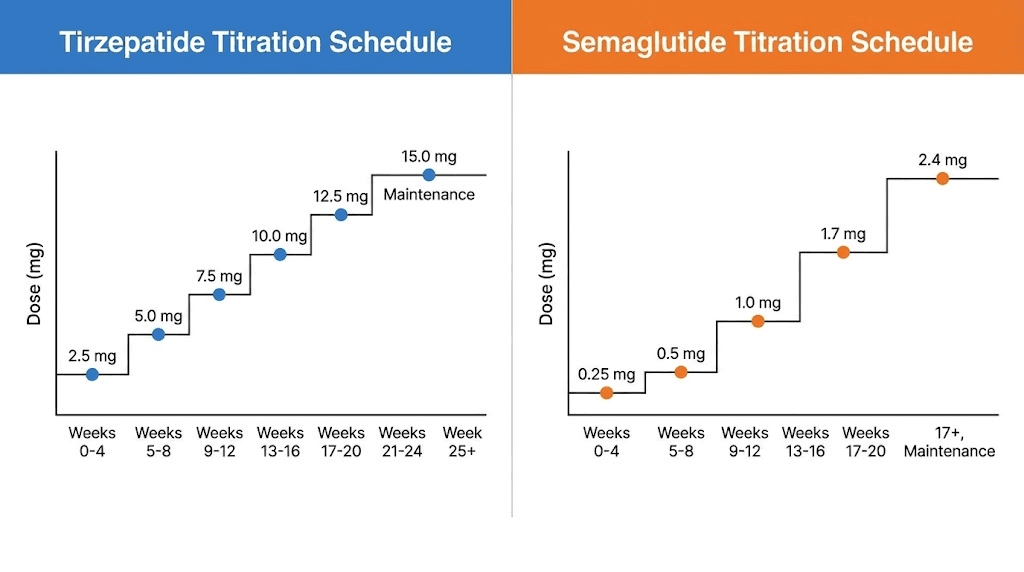
Dosing schedules follow different paths. The tirzepatide dosing protocol starts at 2.5 milligrams weekly and can escalate to fifteen milligrams over five months. Semaglutide begins at 0.25 milligrams and reaches a maximum of 2.4 milligrams for weight loss over approximately four months. These different escalation curves mean you cannot simply match your current dose when switching. Your provider will need to establish an appropriate starting point based on multiple factors.
Side effect profiles overlap considerably but differ in intensity and frequency. Both medications commonly cause nausea, vomiting, diarrhea, and constipation. Tirzepatide users report higher rates of gastrointestinal disturbance, likely due to the dual receptor mechanism. Semaglutide withdrawal symptoms when stopping the medication are similar to those experienced with tirzepatide, including appetite return and potential weight regain if proper strategies are not implemented.
Form factors provide another point of differentiation. Semaglutide is available as both an injectable and an oral tablet. The oral tirzepatide formulation is not yet widely available, though some compounding pharmacies offer it. This availability difference may influence your decision if needle anxiety or injection site issues have become problematic.
Brand name availability and marketing also differ. Semaglutide is marketed as Wegovy for weight loss and Ozempic for diabetes. Tirzepatide is sold as Mounjaro for diabetes and Zepbound for weight loss. Insurance coverage often depends on which indication your provider uses when prescribing. Some plans cover the diabetes indication but not the weight loss indication, even though the medication remains identical.
Reconstitution and storage requirements matter for those using compounded versions. Both peptides require proper handling, but protocols may vary depending on your source. Understanding how to reconstitute peptides correctly and how long compounded semaglutide lasts in the fridge ensures you maintain medication potency throughout your treatment.
Pharmacokinetics and timing your switch
The science of medication clearance determines when you should begin semaglutide after your last tirzepatide dose. This is not guesswork. It is pharmacology. Understanding the elimination kinetics prevents both medication overlap that could intensify side effects and gaps that might allow appetite return and weight regain.
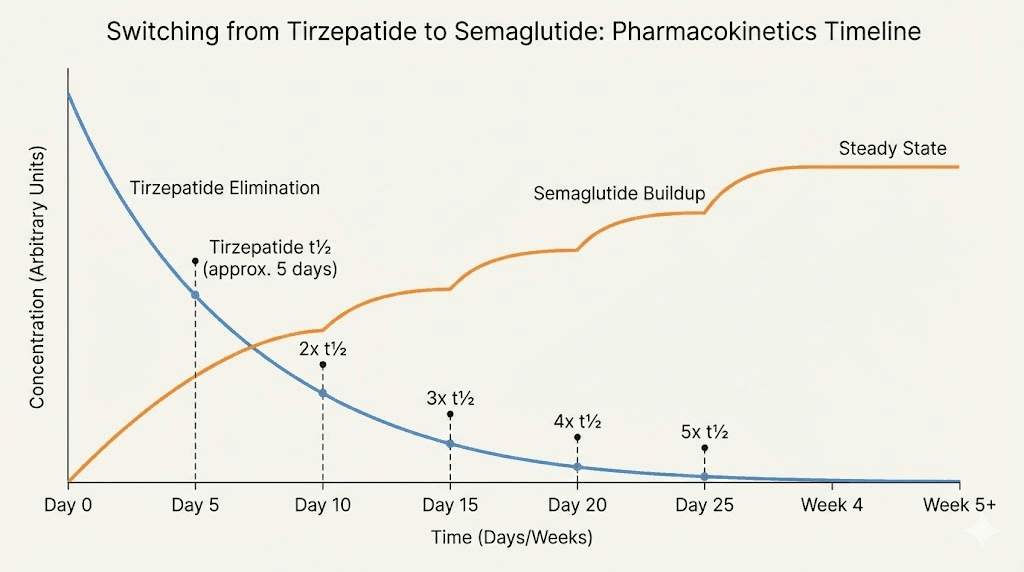
Tirzepatide elimination follows predictable mathematics. With a half-life of approximately five days, you can calculate clearance using the standard pharmacokinetic model. After one half-life, fifty percent remains. After two half-lives, twenty-five percent remains. After three, twelve point five percent remains. After four, six point two five percent. After five half-lives, less than three percent remains in your system, which pharmacologists consider complete elimination.
This means tirzepatide takes roughly twenty-five days to clear completely from your body. However, you do not need to wait for complete clearance before starting semaglutide. Most providers recommend beginning semaglutide between one and two weeks after your last tirzepatide injection. This timing allows tirzepatide levels to drop significantly while maintaining some GLP-1 receptor stimulation during the transition.
Starting semaglutide at your next scheduled injection day represents the most common approach. If you inject tirzepatide weekly on Sundays, you would simply inject your first semaglutide dose the following Sunday instead of taking another tirzepatide dose. This same-day switch approach works well for most people and minimizes the gap in appetite suppression. Your body will have approximately seventy-five percent of your tirzepatide dose still circulating when you inject your first semaglutide dose, providing overlapping coverage.
Some providers prefer a one-week gap between medications. This approach suits individuals who experienced significant side effects on tirzepatide and want to ensure medication levels drop before introducing semaglutide.
The one-week gap allows tirzepatide to decrease to roughly sixty-five percent of peak levels before semaglutide enters the picture. This reduction may help those who struggled with nausea or vomiting to tolerate the new medication more easily.
A two-week washout period is rarely necessary unless you experienced severe side effects requiring medication discontinuation. Two weeks after your last tirzepatide dose, approximately forty percent of peak tirzepatide levels remain. This longer gap increases the risk of appetite return and weight regain, so most providers avoid it unless specifically indicated for side effect management.
Semaglutide builds to steady-state concentrations more slowly than tirzepatide due to its longer half-life. You will not achieve full semaglutide effect until after four to five weeks of consistent dosing. This delayed peak explains why some people experience a temporary dip in appetite suppression during the first month after switching. Understanding how long it takes for semaglutide to work helps set realistic expectations during this transition period.
The overlapping receptor stimulation during week one provides a buffer against appetite return. While tirzepatide levels decline, semaglutide levels increase. This crossover typically prevents the complete loss of appetite control that might occur if you simply stopped tirzepatide without replacement. However, the transition is not always seamless. Some people notice increased hunger during weeks two through four as tirzepatide continues clearing while semaglutide has not yet reached therapeutic steady state.
Injectable versus oral semaglutide timing differs slightly. If switching to oral semaglutide, you begin daily tablets on the same schedule as you would start injectable semaglutide. However, oral absorption is less efficient and more variable, so the buildup to therapeutic levels may take longer. Most providers prefer injectable semaglutide for patients switching from injectable tirzepatide to maintain consistency in delivery method and bioavailability.
Peak effect timing matters for injection scheduling. If you experienced your worst nausea twelve to twenty-four hours after tirzepatide injections, you might choose to inject semaglutide on a different day of the week to avoid peak effects coinciding with work obligations or social events. Semaglutide appetite suppression timing follows a similar pattern to tirzepatide, with maximum effect occurring one to three days post-injection.
Storage considerations during the transition also matter. If you have remaining tirzepatide after switching, understanding how long tirzepatide lasts in the fridge helps you determine whether to keep it as backup or dispose of it properly. Some people maintain their tirzepatide supply temporarily in case the switch to semaglutide does not go smoothly and they need to revert.
Dose conversion strategies that actually work
No official conversion ratio exists between tirzepatide and semaglutide doses. This lack of standardization frustrates patients and providers alike. The medications work through different mechanisms, produce different magnitudes of effect, and follow different dose escalation curves. What works as a conversion for one person may fail for another. Despite this variability, clinical experience has established some general guidelines that providers use when transitioning patients.
The most important principle is this: always start semaglutide at the lowest dose regardless of your tirzepatide dose. This approach minimizes side effects and allows your body to adjust to the new medication gradually. Even if you were taking fifteen milligrams of tirzepatide weekly, you begin semaglutide at 0.25 milligrams. This conservative strategy has become standard practice among experienced providers.
Some approximate equivalencies have emerged from clinical observation, though these should guide rather than dictate dosing decisions. A starting tirzepatide dose of 2.5 milligrams roughly corresponds to the starting semaglutide dose of 0.25 milligrams in terms of tolerability and initial effect. Both represent the minimum therapeutic dose designed to introduce your body to the medication with minimal side effects. Using a peptide calculator can help you track your doses accurately during the transition.
Mid-range tirzepatide doses of five to seven point five milligrams may correspond loosely to semaglutide doses of 0.5 to one milligram. However, this comparison is imperfect because tirzepatide at these doses typically produces more weight loss than semaglutide at corresponding doses. You may find that semaglutide one milligram provides similar appetite suppression to tirzepatide five milligrams, but weight loss velocity may slow slightly during the transition period.
Higher tirzepatide doses of ten to fifteen milligrams do not have clear semaglutide equivalents. The maximum FDA-approved semaglutide dose for weight loss is 2.4 milligrams, which generally produces less weight loss than tirzepatide fifteen milligrams.
Some providers use off-label semaglutide dosing up to three milligrams for patients who need maximum effect, but this exceeds standard protocols. If you were thriving on high-dose tirzepatide, switching to semaglutide may require accepting slightly reduced efficacy or exploring peptide stacking strategies to maintain results.
The standard semaglutide titration schedule begins with 0.25 milligrams weekly for four weeks. This loading dose primes your body for the medication while producing minimal side effects. Most people experience modest appetite suppression at this dose but not the dramatic effect they experienced with therapeutic tirzepatide doses. The semaglutide dosage calculator can help you plan your entire escalation schedule from start to maintenance dose.
After four weeks at 0.25 milligrams, you increase to 0.5 milligrams for another four weeks. This represents the first therapeutic dose where most people experience significant appetite suppression and weight loss. Some providers allow patients who previously tolerated high-dose tirzepatide to advance through these early semaglutide doses more quickly, moving from 0.25 to 0.5 milligrams after two weeks instead of four. This accelerated titration must be done carefully and only under provider supervision.
The next escalation brings you to one milligram weekly, typically after eight weeks on semaglutide total. This dose produces robust effects for many people and represents a common maintenance level. Some people find that semaglutide one milligram provides comparable appetite control to their previous tirzepatide dose of five to seven point five milligrams, though individual responses vary widely.
Further increases to 1.7 milligrams and eventually 2.4 milligrams follow at four-week intervals. Not everyone needs to reach the maximum dose. Your provider will determine your optimal maintenance dose based on weight loss progress, side effect tolerance, and appetite control. Some people stabilize at one milligram while others require the full 2.4 milligrams to maintain the effects they experienced on tirzepatide.
Microdosing represents an alternative approach that some providers use during transitions. Instead of following the standard FDA titration schedule, they start with even smaller semaglutide doses and increase more gradually. This strategy may help people who are particularly sensitive to GLP-1 medications or who experienced severe side effects on tirzepatide. While microdosing tirzepatide protocols are well-established, microdosing semaglutide during transitions is less standardized but gaining interest.
Compounded semaglutide dosing may differ from brand-name protocols. Compounding pharmacies often provide semaglutide in different concentrations, requiring conversion calculations. Understanding how many milligrams are in your syringe units prevents dosing errors. Similarly, knowing conversion between units and milligrams ensures accuracy when switching between different formulations or suppliers.
Individual titration factors override any general conversion guidelines. Your switching protocol should account for how well you tolerated tirzepatide, your current weight loss progress, your previous experience with GLP-1 medications, and your side effect history.
Someone who breezed through tirzepatide escalation with minimal side effects might tolerate faster semaglutide titration.
Someone who struggled with nausea throughout their tirzepatide journey needs the slowest possible semaglutide introduction.
Weight loss goals also influence dosing strategy. If you have reached your target weight on tirzepatide and are switching primarily for cost or access reasons, you might aim for a lower maintenance semaglutide dose that preserves results without pushing for additional loss. If you still have significant weight to lose, you may need to escalate to maximum semaglutide doses to maintain momentum. Discussing your specific goals with your provider ensures your dosing protocol aligns with your objectives.
Step-by-step switching protocol
The actual mechanics of switching require careful planning and execution. A structured protocol minimizes complications and maintains your weight loss progress. The following step-by-step approach represents best practices synthesized from provider experience and patient outcomes. Adapt it to your specific situation in consultation with your healthcare provider.
Step one involves assessing your current status on tirzepatide. Document your current dose, how long you have been at that dose, your total weight loss to date, and any ongoing side effects. Record your appetite control level on a scale of one to ten, with ten being complete appetite suppression. Note your energy levels, gastrointestinal function, and any other medication effects. This baseline documentation will help you track changes during the transition and identify whether the switch is working or needs adjustment.
Step two requires obtaining your semaglutide supply before discontinuing tirzepatide. Do not stop your current medication until you have your new medication in hand. Insurance authorization delays, pharmacy stock issues, and compounding lead times can create gaps that trigger appetite return and weight regain. Order your semaglutide at least two weeks before your planned switch date to ensure timely arrival. Verify proper peptide storage requirements and gather necessary supplies like syringes, alcohol wipes, and sharps containers if using compounded versions.
Step three establishes your switching timeline. Decide with your provider whether you will do a same-week switch, a one-week gap, or a two-week washout. For most people, the same-week switch works best. Schedule your last tirzepatide injection and mark your calendar for your first semaglutide injection. If you typically inject on Sunday mornings, your last tirzepatide would be Sunday and your first semaglutide would be the following Sunday. This one-week interval provides optimal overlap without excessive medication accumulation.
Step four begins your semaglutide dosing at 0.25 milligrams weekly. Even if you were on maximum-dose tirzepatide, start here. Inject at the same time of day and day of week that you used for tirzepatide to maintain consistency. Use proper injection technique, rotating injection sites between abdomen, thigh, and upper arm to prevent lipohypertrophy. If using compounded semaglutide, verify your dose calculation using a peptide reconstitution calculator to ensure accuracy.
Step five involves close monitoring during your first four weeks. Track your appetite daily, noting any increases in hunger or cravings compared to your experience on tirzepatide. Weigh yourself weekly at the same time under the same conditions. Monitor for side effects, particularly nausea, vomiting, diarrhea, and constipation. Keep a simple journal noting dose, side effects, appetite level, and weight. This data will inform your titration decisions and help your provider optimize your protocol.
Step six addresses the expected appetite fluctuation during weeks two through four. As tirzepatide clears and semaglutide builds toward steady state, you will likely experience reduced appetite suppression compared to your tirzepatide peak. This is normal and temporary. Implement behavioral strategies to maintain control during this vulnerable period. Increase protein intake, stay hydrated, maintain your eating schedule, and avoid trigger foods. The appetite suppression will strengthen as semaglutide reaches therapeutic levels.
Step seven increases your dose to 0.5 milligrams after four weeks at 0.25 milligrams. Schedule this increase for your fifth semaglutide injection. Most people tolerate this escalation well, experiencing minimal increase in side effects while gaining noticeably stronger appetite suppression. Continue your monitoring practices, documenting any changes in effects or side effects. This dose represents the first truly therapeutic level where significant weight loss typically resumes if it had slowed during the initial transition period.
Step eight continues escalation every four weeks according to the standard titration schedule: one milligram at week nine, 1.7 milligrams at week thirteen, and potentially 2.4 milligrams at week seventeen. Not everyone will need to reach maximum dose. Work with your provider to determine your optimal maintenance dose based on weight loss progress, side effect tolerance, and appetite control. Some people achieve excellent results at one milligram and never need higher doses. Others require 2.4 milligrams to match their previous tirzepatide effects.
Step nine establishes your maintenance strategy once you reach your optimal semaglutide dose. Monitor for weight loss plateau, which may indicate the need for dose adjustment or the addition of complementary approaches. Consider whether peptide stacking for weight loss might enhance your results if you have not achieved the same outcomes as you experienced on tirzepatide. Some people add AOD-9604 or other complementary peptides to optimize fat loss.
Step ten involves ongoing assessment and adjustment. Schedule regular check-ins with your provider every four to eight weeks during the transition period and every three months once stabilized. Monitor labs including comprehensive metabolic panel, lipid panel, and hemoglobin A1C if applicable. Track body composition changes, not just scale weight, as you may lose fat while maintaining or gaining muscle. Adjust your protocol based on results, always prioritizing sustainable progress over rapid change.
Managing side effects during the transition
Side effect management can make or break your switching experience. The transition between tirzepatide and semaglutide creates a unique physiological situation where your body adapts to a new medication while clearing the old one. Understanding potential side effects and having management strategies ready prevents minor discomfort from becoming major obstacles to your success.
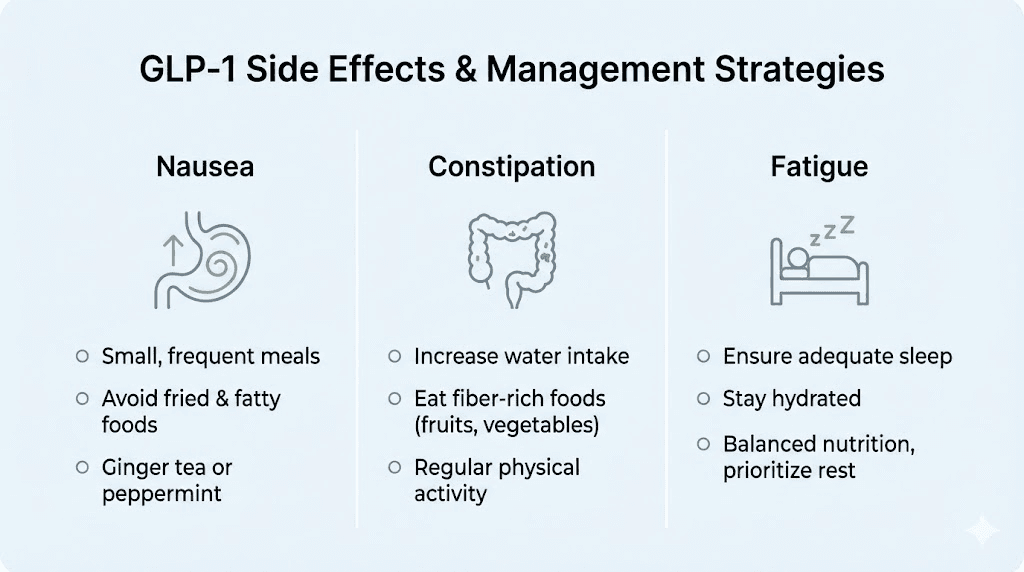
Nausea represents the most common side effect during the transition. The mechanisms differ slightly between medications, but both slow gastric emptying and alter gut hormone signaling in ways that can trigger nausea. During your first two weeks on semaglutide, you may experience less nausea than you had on tirzepatide, particularly if GI side effects were your reason for switching. However, as semaglutide doses increase, nausea may return or intensify.
Managing nausea starts with eating strategy. Small, frequent meals work better than large meals when your gastric emptying is pharmacologically slowed. Avoid high-fat foods that sit in your stomach longer and trigger more nausea. Cold foods often cause less nausea than hot foods. Ginger, whether as tea, candied ginger, or supplements, provides natural nausea relief for many people. Vitamin B6 supplementation at fifty to one hundred milligrams daily may reduce nausea severity. Prescription anti-nausea medications like ondansetron can be used short-term for severe cases.
Constipation affects a significant portion of people on GLP-1 medications. The slowed gut motility that helps with weight loss also slows bowel movements. During the transition from tirzepatide to semaglutide, your constipation pattern may change. Some people find that constipation improves when switching from tirzepatide, while others experience worsening as semaglutide doses increase.
Preventing and treating constipation requires a multi-pronged approach. Increase your water intake substantially, targeting at least eighty to one hundred ounces daily. Add fiber gradually, working up to twenty-five to thirty-five grams daily through food or supplements. Magnesium supplements, particularly magnesium citrate or magnesium oxide, provide gentle osmotic laxative effects at doses of three hundred to five hundred milligrams nightly. Regular movement and exercise stimulate gut motility. If constipation persists despite these measures, discuss additional options with your provider, including osmotic laxatives like polyethylene glycol or stimulant laxatives for occasional use.
Diarrhea can occur instead of or alternating with constipation. The gut adaptation to GLP-1 medications sometimes manifests as loose stools or increased bowel frequency. This side effect typically resolves within two to four weeks as your body adjusts to semaglutide. Probiotics may help stabilize gut function during the transition. Avoiding artificial sweeteners, particularly sugar alcohols and sucralose, prevents additional GI upset. Understanding peptides that support gut health may provide additional strategies for managing digestive challenges during your switch.
Fatigue sometimes increases during the medication transition. Multiple factors contribute to this effect. The caloric deficit from reduced appetite can leave you under-fueled. Dehydration from inadequate fluid intake compounds fatigue. Blood sugar fluctuations as your metabolism adjusts to the new medication may cause energy dips. Understanding whether your previous medication caused fatigue helps you anticipate whether switching will improve or worsen this symptom.
Combating transition-related fatigue requires attention to nutrition and hydration. Ensure you are consuming adequate protein, targeting at least 0.7 grams per pound of goal body weight daily. Do not let your calorie intake drop too low too fast. A moderate deficit of three hundred to five hundred calories below maintenance supports sustainable weight loss without excessive fatigue. Consider timing your injections for evenings if you notice fatigue peaks in the day or two following injection. Some people find that combining semaglutide with B12 helps address energy levels, though individual responses vary.
Appetite fluctuation during the transition period deserves special attention. You will likely notice reduced appetite suppression compared to your peak tirzepatide effect, particularly during weeks two through four after switching. This is not medication failure. It reflects the pharmacokinetic reality of clearing one medication while building another to steady state.
Expect this change and prepare behavioral strategies to maintain control during this window.
Structured eating helps tremendously during appetite fluctuation periods. Pre-plan your meals and snacks so you are not making food decisions when hungry. Keep high-protein, low-calorie options readily available. Maintain your eating schedule even if appetite returns, as chaotic eating patterns often trigger overeating. Track your food intake during this transition period to maintain awareness and prevent gradual increases that could stall weight loss. Many people find that their appetite suppression strengthens significantly after reaching semaglutide 0.5 or one milligram, making this temporary challenge worth enduring.
Injection site reactions may change when switching medications. Brand-name and compounded formulations have different excipients that can trigger different skin reactions. Some people who experienced injection site burning or redness with tirzepatide find that semaglutide causes less irritation, while others experience the opposite. Proper injection technique matters regardless of which medication you use. Understanding proper peptide injection technique minimizes site reactions and ensures optimal medication absorption.
Headaches occasionally increase during medication transitions. Dehydration, blood sugar fluctuations, and the physiological stress of metabolic adaptation can all trigger headaches. Address hydration first, ensuring you consume adequate water and electrolytes. Consider your caffeine intake, as changes in eating patterns during the transition may inadvertently reduce your usual caffeine consumption, triggering withdrawal headaches. Over-the-counter pain relievers can manage occasional headaches, but persistent or severe headaches warrant provider consultation to rule out other causes.
Hair loss concerns arise frequently in GLP-1 medication discussions. While GLP-1-related hair loss can occur, it typically results from rapid weight loss and nutritional deficiencies rather than direct medication effects. The transition between medications does not necessarily increase hair loss risk, but maintaining adequate protein, biotin, and other nutrients during your switch helps protect hair health. If you experienced hair thinning on tirzepatide, switching to semaglutide will not necessarily resolve it unless you also address the underlying nutritional factors.
Maintaining weight loss during the switch
Weight maintenance during the switching period requires deliberate strategy. The pharmacokinetic gap between peak tirzepatide effect and peak semaglutide effect creates a vulnerable window where appetite suppression decreases and weight regain risk increases. Understanding this challenge and implementing protective measures prevents the frustrating experience of losing hard-won progress during your transition.
The first protective factor is realistic expectation setting. You will very likely experience increased hunger during weeks two through four after switching. Your appetite will not disappear completely. Food will become more appealing. Cravings may return. This is temporary and normal. Accepting this reality psychologically prepares you to implement behavioral strategies rather than panicking when appetite increases. Remember that this phase is time-limited and appetite suppression will strengthen as semaglutide reaches therapeutic levels.
Protein intake becomes even more critical during the transition. Target at least one hundred grams daily if you are a woman and one hundred fifty grams if you are a man, adjusting based on your body size and activity level. Protein provides satiety, preserves lean muscle mass during continued caloric deficit, and supports metabolic rate. Structure each meal around a protein source. Supplement with protein shakes if needed to hit your targets. This single nutritional intervention provides more protection against weight regain than any other dietary factor during your switch.
Meal timing and structure help compensate for reduced appetite suppression. Eat at consistent times each day, even if you do not feel particularly hungry. This structure prevents the pattern of skipping meals during low-appetite periods and then overeating when appetite returns. Use smaller plates to manage portion sizes visually. Start meals with protein and vegetables before moving to starches or other foods. These behavioral approaches work synergistically with whatever appetite suppression your medication provides.
Hydration provides appetite control benefits beyond its other health effects. Drinking sixteen to twenty ounces of water before meals increases fullness and reduces calorie intake. Many perceived hunger signals are actually thirst signals. Target at least eighty ounces of water daily, more if you exercise significantly or live in a hot climate. Add electrolytes if needed to make increased water intake more palatable and to prevent electrolyte dilution.
Exercise consistency supports weight maintenance through multiple mechanisms. Physical activity burns calories, preserves muscle mass, improves insulin sensitivity, and provides psychological benefits that reduce stress eating. You do not need to increase exercise intensity during your transition, but maintaining your current activity level matters.
Resistance training two to three times weekly helps preserve muscle mass during continued caloric deficit. Daily walking provides low-impact calorie expenditure and appetite regulation benefits.
Sleep quality influences appetite regulation and weight management powerfully. Inadequate sleep increases hunger hormones, decreases satiety hormones, and impairs decision-making around food choices. Aim for seven to nine hours of quality sleep nightly. Maintain consistent sleep and wake times. The appetite regulation benefits of adequate sleep become particularly important when medication-driven appetite suppression is reduced during your transition period.
Stress management prevents stress-eating that can undermine your weight maintenance efforts. The transition between medications may create anxiety about regaining weight or losing progress. This psychological stress can trigger emotional eating patterns. Implement stress reduction practices like meditation, deep breathing, journaling, or therapy. Identify your stress-eating triggers and develop alternative coping mechanisms. Addressing the emotional components of eating behavior provides protection during vulnerable transition periods.
Monitoring frequency should increase during your transition period. Weigh yourself weekly rather than monthly to catch any upward trends early. Track your food intake using an app or journal to maintain awareness. Measure body composition if possible, as the scale does not tell the complete story. Take progress photos monthly.
These monitoring practices provide early warning if your maintenance strategies need adjustment and help you stay accountable during the challenging transition phase.
Contingency planning prepares you for worst-case scenarios. Decide in advance what you will do if you regain five pounds during the transition. At what point will you contact your provider to discuss accelerating your semaglutide titration or implementing additional interventions? What weight gain threshold triggers a return to tirzepatide if semaglutide proves inadequate? Having these decision points established in advance prevents panic-driven poor choices and provides a clear action plan if challenges arise. Reviewing common peptide mistakes helps you avoid errors that could compound transition challenges.
Understanding that temporary weight stabilization or even small gains do not represent failure helps maintain perspective. Many people experience a two to four week period during the medication transition where weight loss stalls or they regain two to five pounds. This does not mean the switch has failed. It reflects the pharmacokinetic reality of the transition. As semaglutide reaches therapeutic levels and you continue working through the titration schedule, weight loss typically resumes. Judge your transition by three-month outcomes, not week-to-week fluctuations.
When to consider switching back or trying alternatives
Not every switch from tirzepatide to semaglutide succeeds. Some people find that semaglutide does not provide adequate appetite suppression or weight loss compared to their tirzepatide experience. Others struggle with side effects that were not problematic on tirzepatide. Knowing when to persist with semaglutide titration versus when to consider alternative approaches helps you avoid prolonged frustration and suboptimal results.
Inadequate appetite suppression represents the most common reason people consider switching back or trying alternatives. If you have reached semaglutide one milligram or higher and still experience significantly more hunger than you had on tirzepatide, the difference may reflect the dual versus single receptor mechanism. Some people simply respond better to tirzepatide dual agonism than to semaglutide single agonism. If cost or access was your only reason for switching and those barriers have resolved, returning to tirzepatide may make sense.
Weight loss stalling or regain despite medication adherence signals the need for protocol evaluation. First, ensure you are truly at an adequate semaglutide dose. Many people achieve excellent results only after reaching 1.7 or 2.4 milligrams. If you have plateaued at lower doses, completing your titration should be the first intervention. If you have reached maximum dose and still cannot maintain your tirzepatide results, discuss with your provider whether switching back or trying alternative medications makes sense.
Intolerable side effects that worsen rather than improve over four to eight weeks warrant reconsideration. Most GLP-1 side effects diminish with continued use as your body adapts. However, if nausea, vomiting, or other effects remain severe after two months at a given dose, you may not tolerate semaglutide well. This is surprising if you tolerated tirzepatide without issues, as semaglutide typically causes fewer side effects. However, individual responses vary, and occasionally someone tolerates the dual agonist better than the single agonist.
Alternative medications beyond switching back to tirzepatide deserve consideration. Retatrutide versus semaglutide comparisons show that this triple agonist may provide even better results than tirzepatide for some people, though it remains in clinical development. Understanding retatrutide dosing protocols helps you evaluate whether pursuing this medication through clinical trials or other avenues makes sense for your situation.
Combination approaches may succeed where single medications partially fail. Adding cagrilintide to semaglutide enhances weight loss through an additional amylin receptor mechanism. This combination, marketed as CagriSema, represents a pipeline medication. Understanding CagriSema dosing protocols prepares you to discuss this option with your provider once it gains broader approval. The cagrilintide weight loss data and cagrilintide dosing information show promising results for enhancing GLP-1 effects.
Other peptide additions may enhance semaglutide effects without requiring a complete medication switch. BPC-157 provides gut healing properties that may reduce GI side effects and improve overall tolerance. 5-Amino-1MQ may enhance fat loss through different metabolic pathways. Ipamorelin benefits include preserving muscle mass during weight loss phases. Exploring how many peptides you can safely combine opens additional optimization options beyond single-medication approaches. Some people also explore lipotropic peptide formulations to support fat metabolism during transitions.
Timing considerations affect when you might switch back to tirzepatide if semaglutide proves inadequate. Ideally, complete your full semaglutide titration to maximum tolerated dose before making this decision. Give semaglutide at least twelve weeks at your highest dose to assess its true effects. Switching back prematurely means you will never know whether higher semaglutide doses would have worked. However, if you experience severe side effects or rapid weight regain despite adherence, waiting twelve weeks may not be appropriate.
The switching-back protocol mirrors your original switch. If returning to tirzepatide from semaglutide, start at the lowest tirzepatide dose regardless of your semaglutide dose. Follow the standard tirzepatide titration schedule. Semaglutide longer half-life means it will take five to six weeks to clear completely, providing some appetite suppression overlap as you rebuild tirzepatide levels. The transition back typically causes fewer challenges than the original switch because tirzepatide more potent effects build more quickly.
Provider communication throughout the switching and potential switching-back process is essential. Your provider needs to know if the transition is not going well. Many issues have solutions short of abandoning semaglutide entirely. Dose adjustments, side effect management strategies, adding complementary medications, or modifying your titration schedule may resolve problems without requiring a complete reversal. Open communication ensures you exhaust appropriate options before making major protocol changes. Exploring online peptide therapy options may give you access to providers with more specialized transition experience.
Real-world switching experiences and outcomes
Understanding how real people experience the transition from tirzepatide to semaglutide provides valuable context beyond pharmacological theory. While individual experiences vary widely, certain patterns emerge from the thousands of people who have made this switch. These real-world outcomes help set realistic expectations and identify common challenges you may encounter.
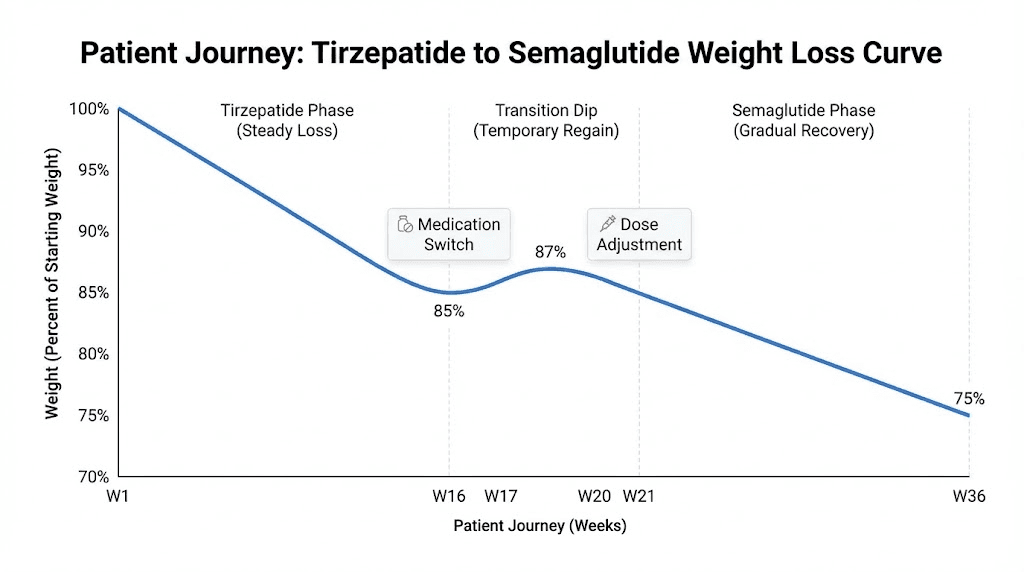
The most frequently reported experience involves a temporary increase in hunger during weeks two through four after switching. This pattern appears so consistently that providers who regularly manage these transitions now warn patients in advance. People describe feeling like their medication stopped working, even though they understand intellectually that semaglutide is still building to therapeutic levels. This hunger typically peaks around week three and then gradually improves as semaglutide doses increase through the titration schedule.
Weight loss velocity typically slows during and immediately after the transition. People who were losing two to three pounds weekly on tirzepatide often see this drop to one pound weekly or even weight maintenance during their first month on semaglutide. This slowing frustrates many people but does not necessarily indicate medication failure. As semaglutide doses escalate to one milligram and higher, weight loss typically resumes at rates of one to two pounds weekly, which represents excellent sustainable progress even if slower than peak tirzepatide results. Viewing tirzepatide before and after results alongside expected semaglutide outcomes helps calibrate expectations realistically.
Side effect experiences during the switch vary more than expected. The most common pattern involves reduced side effects compared to tirzepatide, particularly for people who struggled with nausea or GI distress on the dual agonist. These individuals often report that semaglutide feels gentler and more tolerable while still providing meaningful appetite suppression. However, a smaller subset reports increased side effects on semaglutide despite tolerating tirzepatide well.
Long-term success rates for maintaining weight loss after switching appear comparable between people who stay on tirzepatide and those who switch to semaglutide, provided the switch is done properly with full titration. Data from patient registries and clinical practices suggest that most people who complete their semaglutide titration to maximum tolerated dose maintain at least ninety percent of their tirzepatide weight loss results over six to twelve months. The critical factor is completing the titration rather than stopping at low doses due to impatience during the transition period.
Cost savings represent the most cited benefit among people who switched from brand-name or expensive compounded tirzepatide to more affordable semaglutide options. Many report saving three hundred to seven hundred dollars monthly, which makes the temporary increase in hunger and potential slight reduction in weight loss velocity acceptable trade-offs. Understanding overall peptide therapy costs helps you evaluate whether your specific transition makes financial sense in the context of your broader treatment plan.
Medication access concerns drive many transitions, and people report mixed experiences depending on their sources. Some who switched due to tirzepatide compounding pharmacy issues found reliable semaglutide sources and achieved stable long-term treatment. Others encountered similar supply chain problems with compounded semaglutide, discovering that regulatory pressures affect multiple GLP-1 medications. Having contingency plans and multiple supplier options helps people navigate these access challenges. Researching reliable peptide vendor options in advance prevents scrambling when supply disruptions occur.
The option to use oral semaglutide appeals to some people who make the switch, though real-world experience with oral forms is more mixed than many expect. While avoiding injections seems attractive, oral semaglutide requires daily dosing rather than weekly, must be taken on an empty stomach with specific timing restrictions, and demonstrates lower bioavailability than injectable forms. Most people who try oral semaglutide after injectable tirzepatide report preferring to return to weekly injections due to the superior convenience and effectiveness of the injectable route.
Psychological aspects of switching receive less attention than physical effects but significantly impact experiences. Some people report feeling discouraged or defeated by needing to switch, particularly if cost or access barriers forced the change rather than it being their choice. This emotional response can undermine adherence and motivation during the challenging transition period. Conversely, people who view the switch as an active choice to optimize their protocol often maintain better psychological adjustment and persistence through temporary difficulties.
Community and peer support help many people navigate switching challenges. Online communities dedicated to GLP-1 medications provide spaces where people share experiences, strategies, and encouragement. However, these communities also contain misinformation and anxiety-provoking anecdotes. Learning to evaluate information critically and focus on evidence-based guidance helps people benefit from community support without being derailed by outlier experiences.
The peptide forum landscape offers various communities where you can connect with others going through similar transitions.
Long-term perspective separates people who succeed with the switch from those who abandon treatment. Those who view the transition as a temporary adjustment phase within a long-term weight management journey typically persist through difficulties and achieve good outcomes. Those who expect seamless transition with no temporary setbacks often become discouraged during the normal adaptation period and sometimes discontinue treatment entirely. Maintaining focus on multi-year success rather than week-to-week perfection provides the psychological resilience needed to navigate medication transitions successfully.
Insurance, cost, and access considerations
The financial landscape surrounding tirzepatide and semaglutide drives many switching decisions and deserves detailed examination. Understanding insurance coverage patterns, cost comparisons, and access strategies helps you make informed decisions and avoid expensive surprises. The economics of these medications have shifted dramatically as shortages have resolved and FDA enforcement on compounding has intensified.
Brand-name medication costs create significant barriers for many people. Mounjaro and Zepbound, the brand names for tirzepatide, typically cost between one thousand and thirteen hundred dollars monthly without insurance coverage. Wegovy and Ozempic, the brand names for semaglutide, cost similarly, ranging from nine hundred to twelve hundred dollars monthly. These prices place brand-name GLP-1 medications out of reach for most people without insurance coverage or manufacturer copay assistance programs.
Insurance coverage patterns vary dramatically between plans and change frequently. Many insurance plans cover these medications for diabetes treatment but exclude or severely restrict coverage for weight loss indications. Some plans cover semaglutide but not tirzepatide, or vice versa. Prior authorization requirements, step therapy protocols requiring failure of other medications first, and body mass index thresholds create additional barriers. Understanding your specific plan coverage before switching prevents discovering mid-transition that your new medication is not covered.
Compounded medication costs represent a middle ground between prohibitive brand-name prices and manufacturer assistance programs. Compounded tirzepatide typically costs between one hundred fifty and four hundred dollars monthly depending on dose and source. Compounded semaglutide generally runs slightly less, averaging one hundred to three hundred dollars monthly. These compounded options have provided affordable access for millions of people but face ongoing regulatory uncertainty. The legal landscape around peptides continues to evolve in ways that affect compounded GLP-1 medication access.
The FDA stance on compounding shortages directly impacts medication access. When a medication is on the FDA shortage list, compounding pharmacies may legally produce it under specific conditions. Both tirzepatide and semaglutide have appeared on and been removed from shortage lists multiple times. Tirzepatide most recently came off the shortage list in December 2024, while semaglutide was removed in February 2025. Once removed from shortage status, compounding becomes more legally precarious, potentially reducing availability and increasing costs.
Quality concerns with compounded medications require careful evaluation. Not all compounding pharmacies maintain equivalent standards. Some use pharmaceutical-grade active ingredients sourced from FDA-registered facilities and maintain rigorous testing protocols. Others may use lower-quality chemicals or skip critical purity and potency testing. Understanding how to identify high-quality peptide vendors helps you avoid inferior or potentially dangerous products. Asking about third-party testing, certificate of analysis availability, and compounding pharmacy accreditation provides important quality indicators.
Telemedicine prescribing has expanded access to GLP-1 medications but varies in quality and cost. Several companies offer compounded semaglutide through telemedicine consultations, with all-inclusive pricing ranging from two hundred to four hundred dollars monthly. These services provide convenience and avoid insurance hassles but typically cost more than working with local compounding pharmacies directly. Exploring local peptide therapy options alongside telemedicine may reveal more cost-effective approaches for your situation.
Prior authorization processes create delays and frustration when switching medications. Even if your insurance covers your new medication, obtaining approval may require your provider to submit extensive documentation justifying medical necessity. This process can take days to weeks, creating supply gaps that trigger weight regain if not managed proactively. Starting the prior authorization process at least three weeks before you need your new medication helps prevent these gaps.
Alternative approaches may reduce costs when brand-name and compounded options prove too expensive. Ozempic alternatives include other GLP-1 medications, non-GLP-1 weight loss options, and complementary peptide approaches that may provide benefits at lower price points. Some people find that combining a lower-dose semaglutide with fat-burning peptides achieves comparable results to maximum-dose single-medication therapy at reduced cost.
Special populations and considerations
Certain groups face unique considerations when switching from tirzepatide to semaglutide. Individual circumstances affect optimal switching protocols, potential risks, and expected outcomes. Understanding how your specific situation influences the transition helps you and your provider develop the most appropriate strategy for your needs.
People with diabetes who use GLP-1 medications primarily for glucose control face different switching considerations than those using these medications solely for weight loss. Blood sugar management must remain stable throughout the transition. More frequent glucose monitoring during the switch helps detect any destabilization early. Some people find that semaglutide provides less robust glucose control than tirzepatide due to the single versus dual receptor mechanism. If your hemoglobin A1C begins rising after switching, discuss with your provider whether adjusting other diabetes medications or returning to tirzepatide makes sense.
Women navigating perimenopause or menopause face additional metabolic considerations during medication transitions. Hormonal changes already affect weight management, insulin sensitivity, and appetite regulation. The transition between GLP-1 medications adds another variable to an already complex metabolic picture. Understanding how peptides interact with menopause-related weight changes helps women anticipate and manage the additional challenges of switching medications during this life phase. The best peptide approaches for perimenopause may complement your GLP-1 transition strategy.
Individuals with gastroparesis or other gastrointestinal motility disorders require careful evaluation before switching. Both medications slow gastric emptying, but tirzepatide typically causes more pronounced GI effects. Some people with underlying motility issues tolerate semaglutide better than tirzepatide, making the switch potentially beneficial. However, others may find that even semaglutide exacerbates their baseline condition. Starting at the lowest possible dose and escalating more slowly than standard protocols helps identify tolerance issues before reaching problematic dose levels.
People with a history of pancreatitis face relative contraindications to GLP-1 medications generally. While both tirzepatide and semaglutide carry theoretical pancreatitis risk, the incidence appears low in clinical practice. If you tolerated tirzepatide without pancreatic issues, semaglutide will likely be similarly safe. However, any new or worsening abdominal pain during the switch warrants immediate medical evaluation. Understanding peptide safety considerations helps you recognize warning signs that require urgent attention.
Older adults over age sixty-five may experience different side effect profiles and weight loss patterns than younger users. Dehydration risk increases with age, and GLP-1 medications may compound this through reduced fluid intake from appetite suppression. Muscle loss concerns are more significant in older adults, making protein intake even more critical during switching transitions. Peptide strategies for women over forty address some of these age-related concerns, though both men and women benefit from age-appropriate protocol modifications.
Athletes and highly active individuals may notice performance changes when switching medications. Some athletes report that tirzepatide affects training capacity and recovery more than semaglutide, while others experience the opposite. Energy availability for training, muscle preservation during caloric deficit, and recovery capacity all deserve attention during the switching period. Understanding how to balance weight loss and muscle preservation with peptides helps active individuals optimize their protocols during the transition.
People considering compounded versions should understand the reconstitution requirements for compounded semaglutide before switching. Compounded formulations often require mixing with bacteriostatic water, and proper handling ensures medication efficacy and safety. Understanding semaglutide expiration and stability prevents using degraded medication that may produce suboptimal results.
For researchers serious about optimizing their medication transitions, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. The SeekPeptides platform provides personalized dosing calculators, expert-reviewed transition protocols, and member forums where you can connect with others going through similar switches for real-time support and guidance.
Frequently asked questions
How long should I wait between stopping tirzepatide and starting semaglutide?
Most providers recommend starting semaglutide one week after your last tirzepatide injection. This timing allows tirzepatide levels to decrease while maintaining some appetite suppression during the transition. You can start semaglutide on your next scheduled injection day, replacing tirzepatide with semaglutide. Some people benefit from a two-week gap if they experienced severe side effects on tirzepatide, but longer waiting periods increase weight regain risk. Understanding tirzepatide pharmacokinetics helps you appreciate why same-week switching typically works well.
What semaglutide dose equals my tirzepatide dose?
No official conversion ratio exists between tirzepatide and semaglutide doses. Always start semaglutide at 0.25 milligrams regardless of your tirzepatide dose. This conservative approach minimizes side effects while allowing gradual titration to your optimal therapeutic dose. Rough approximations suggest tirzepatide five milligrams may correspond to semaglutide 0.5 to one milligram, but individual variation is substantial. Follow the standard semaglutide dosing protocol rather than attempting direct dose conversion.
Will I regain weight when switching from tirzepatide to semaglutide?
Some temporary weight stabilization or small regain of two to five pounds commonly occurs during the switching period, particularly in weeks two through four. This reflects reduced appetite suppression as tirzepatide clears and semaglutide builds to therapeutic levels. Most people resume weight loss once they reach semaglutide doses of 0.5 to one milligram. Implementing strong behavioral strategies during the transition minimizes regain risk. Understanding how peptides support weight loss helps you maintain realistic expectations about the transition period.
Can I switch directly without any gap between medications?
Yes, same-day switching is possible and commonly practiced. On your next scheduled tirzepatide injection day, simply inject semaglutide instead. Both medications will overlap in your system for several weeks due to tirzepatide extended half-life, providing continuous GLP-1 receptor stimulation. This approach minimizes appetite return and weight regain risk during the transition. Most people tolerate this overlap well without excessive side effects.
What side effects should I expect when switching?
The most common side effects during switching include temporary nausea, changes in bowel habits, and increased hunger compared to your peak tirzepatide effect. Most people switching from tirzepatide to semaglutide report fewer side effects with semaglutide, particularly reduced GI distress. Side effects typically improve within two to four weeks as your body adjusts. If side effects worsen after one month, consult your provider about dose adjustments. Learning about peptide safety helps you distinguish normal adjustment effects from concerning symptoms.
How long does it take to feel semaglutide working after switching?
You will likely notice some appetite suppression from your first semaglutide injection due to overlapping tirzepatide effect. However, semaglutide reaches steady-state concentrations after four to five weeks of consistent dosing. Most people report meaningful appetite suppression once they reach 0.5 milligram doses, typically at week five. Understanding semaglutide onset timing helps set appropriate expectations during the early transition phase.
Should I stay at 0.25 milligrams semaglutide for four weeks even though I was on high-dose tirzepatide?
Standard practice recommends starting at 0.25 milligrams for four weeks regardless of previous tirzepatide dose. However, some providers allow accelerated titration for patients who tolerated high-dose tirzepatide well, advancing to 0.5 milligrams after two weeks instead of four. The conservative four-week approach minimizes side effect risk, while faster titration reduces the duration of suboptimal appetite suppression. Either approach works when implemented with proper monitoring and careful dose management.
Can I switch to oral semaglutide instead of injectable?
Oral semaglutide is an option, though injectable forms demonstrate superior efficacy and convenience for most people. Oral semaglutide requires daily dosing on an empty stomach with strict timing requirements, whereas injectable semaglutide continues your familiar weekly injection schedule. Bioavailability is lower with oral forms, potentially requiring higher milligram doses to achieve equivalent effects. Understanding injectable versus oral peptide differences helps inform this decision.
In case I do not see you, good afternoon, good evening, and good night. May your transitions stay smooth, your appetite stay controlled, and your progress stay sustainable.