Feb 13, 2026

Three weeks into your GLP-1 protocol and you can barely keep your eyes open by 2 PM. The scale is moving. Your appetite is under control. But the exhaustion is something nobody warned you about, and it is starting to interfere with everything else in your life.
You are not imagining it.
In clinical trials for semaglutide (Wegovy), approximately 11% of adult patients reported fatigue compared to just 5% in the placebo group. For tirzepatide, the numbers follow a dose-dependent pattern: 5% at the 5 mg dose, 6% at 10 mg, and 7% at 15 mg versus 3% on placebo. These are the reported numbers. The real-world incidence is almost certainly higher, because fatigue is one of those side effects people tend to dismiss or attribute to other causes rather than reporting it to their prescriber.
The frustrating part is that GLP-1 fatigue is not a simple problem with a simple fix. It involves at least six different mechanisms working simultaneously, from caloric deficit and blood sugar fluctuations to nutrient malabsorption, electrolyte imbalances, hormonal shifts, and disrupted sleep architecture. Understanding which mechanisms are driving your specific fatigue pattern determines whether you bounce back in two weeks or drag through months of brain fog and exhaustion.
This guide breaks down every mechanism behind GLP-1 related fatigue, covers the clinical data across all major GLP-1 medications, and provides specific protocols for restoring your energy without compromising your weight loss results. Whether you are on semaglutide or tirzepatide, just starting or months into treatment, the information here will help you understand exactly what is happening in your body and what to do about it.
What GLP-1 fatigue actually is (and what it is not)
GLP-1 fatigue is not laziness. It is not poor motivation. And it is not just the normal tiredness that comes with eating less food. It is a specific, identifiable pattern of exhaustion that stems from the way these peptide medications interact with your metabolism, your hormones, your digestive system, and your brain.
The distinction matters.
Normal tiredness from caloric restriction feels different from GLP-1 fatigue. When you simply eat less food, you feel hungry and somewhat lower energy. With GLP-1 fatigue, you can feel profound exhaustion even when you have eaten enough, because the medication is affecting energy regulation through multiple pathways simultaneously. Some researchers describe it as a metabolic recalibration period, where your body is adjusting to fundamentally different signals about food intake, appetite suppression, and energy utilization.
GLP-1 receptor agonists work by mimicking your natural incretin hormones. They bind to GLP-1 receptors throughout your body, not just in your pancreas, but in your brain, your gut, your cardiovascular system, and your nervous system. When these receptors activate, they trigger a cascade of effects: insulin secretion increases, glucagon decreases, gastric emptying slows dramatically, and appetite signaling in the hypothalamus shifts. Every single one of these changes has the potential to affect your energy levels.
Think of it this way. Your body has been running on one operating system for decades. GLP-1 medications install an update that changes how you process fuel, when you feel hungry, how fast food moves through your system, and how your brain interprets energy status signals. The fatigue is, in many cases, the buffering screen while that update installs.
The clinical data on GLP-1 fatigue across medications
Not all GLP-1 medications produce the same fatigue profile. The data from major clinical trials reveals important differences that rarely get discussed in standard prescribing information.
Semaglutide (Ozempic, Wegovy): The STEP clinical trial program showed fatigue rates of approximately 11% in adult patients on the 2.4 mg weekly dose. This was more than double the placebo rate of 5%. Fatigue was more commonly reported during the initial dose escalation phase and often coincided with gastrointestinal side effects like nausea and reduced appetite.
Tirzepatide (Mounjaro, Zepbound): As a dual GIP/GLP-1 receptor agonist, tirzepatide shows a dose-dependent fatigue pattern. The SURMOUNT trials documented fatigue in 5% at 5 mg, 6% at 10 mg, and 7% at the 15 mg dose versus 3% on placebo. Interestingly, some researchers theorize that the GIP component may partially offset some of the energy-depleting effects of GLP-1 activation, which could explain why tirzepatide fatigue rates are slightly lower despite producing greater weight loss. People often wonder whether tirzepatide makes them tired, and the answer is nuanced.
Liraglutide (Saxenda, Victoza): Earlier generation GLP-1 medications like liraglutide also produce fatigue, though the shorter half-life means the pattern is somewhat different. Daily injections create a more fluctuating energy pattern compared to the weekly injections of semaglutide and tirzepatide.
What the clinical trial data does not capture is the subjective quality of the fatigue. Forum discussions and patient reports consistently describe GLP-1 fatigue as qualitatively different from normal tiredness. Words like "bone-deep," "brain fog," and "hitting a wall" come up repeatedly. This suggests that the fatigue involves central nervous system pathways beyond simple caloric deficit.
Who gets hit hardest by GLP-1 fatigue
Certain populations experience more severe fatigue on GLP-1 medications. If you fall into any of these categories, you will want to be especially proactive with the management strategies covered later in this guide.
People with lower baseline caloric intake. If you were already eating relatively little before starting a GLP-1, the additional appetite suppression can push you into a caloric deficit that your body struggles to sustain. Women eating under 1,200 calories daily and men eating under 1,800 calories face significantly higher risks of nutrient deficiency and fatigue.
Those with pre-existing nutrient deficiencies also get hit harder. If your B12, iron, vitamin D, or magnesium levels were already borderline before treatment, GLP-1 medications can push them into clinical deficiency territory. Research shows that nutritional deficiencies were diagnosed in 12.7% of patients within 6 months after GLP-1 initiation and in 22.4% within 12 months.
Patients on higher doses report more fatigue. This is consistent across all GLP-1 medications. The dose-response relationship is clear: higher doses produce more appetite suppression, greater caloric deficit, more gastrointestinal side effects, and consequently more fatigue. Understanding proper dosing protocols and dosage charts becomes critical for managing this.
People who exercise heavily without adjusting their nutrition face a compounded energy deficit. The combination of reduced caloric intake from the medication plus increased energy expenditure from exercise can create a recovery gap that manifests as persistent fatigue, poor workout performance, and slow recovery times.

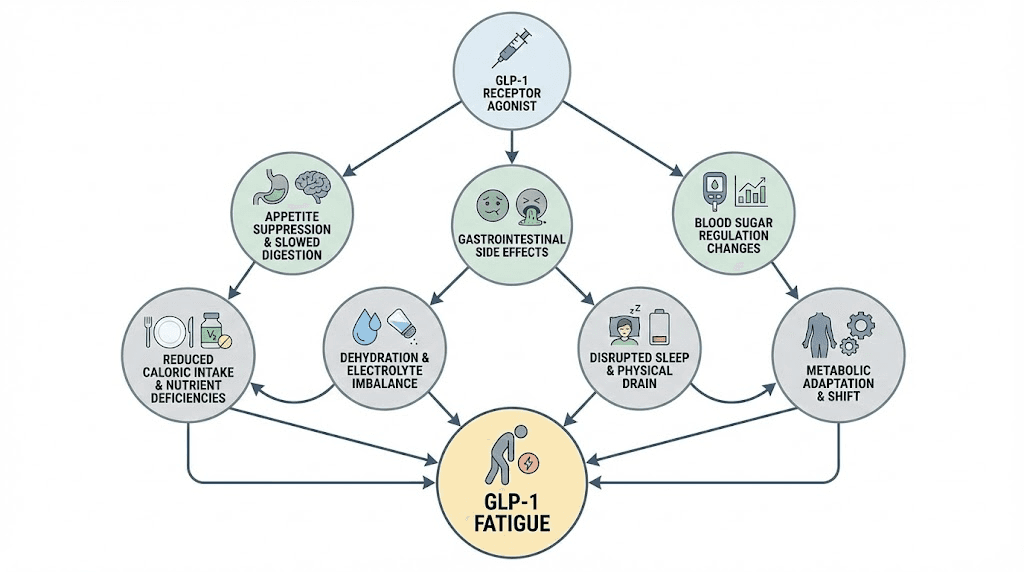
The six mechanisms behind GLP-1 fatigue
Understanding why GLP-1 medications cause fatigue requires examining six distinct but interconnected mechanisms. Most articles mention one or two. The reality is that all six contribute, and identifying which ones dominate in your case is the key to effective management.
Mechanism 1: the caloric deficit cascade
This is the most obvious mechanism, and it is the one most people assume is the entire explanation. It is not. But it is significant.
GLP-1 medications reduce caloric intake by 16-39% on average. That is an enormous reduction. If someone was eating 2,500 calories daily, a 30% reduction drops them to 1,750 calories, well below their maintenance needs and potentially below the threshold where their body can maintain normal energy production without adaptation.
The caloric deficit triggers several secondary effects that compound the fatigue. Your basal metabolic rate begins to slow as your body attempts to conserve energy. Thyroid hormone conversion from T4 to T3 can decrease. Growth hormone pulsatility may shift. Cortisol patterns can change. Your body interprets the sustained caloric deficit as a survival threat and responds accordingly, redirecting energy away from non-essential functions toward basic survival processes.
For people on semaglutide or tirzepatide, following a structured diet plan becomes essential to prevent the caloric deficit from becoming too severe. The goal is a moderate, sustainable deficit that produces weight loss without crashing your energy systems.
The caloric deficit also creates a nitrogen balance problem. Without adequate protein intake, your body begins breaking down muscle tissue for amino acids. Muscle loss has its own fatigue-producing effects: reduced metabolic rate, decreased insulin sensitivity, lower mitochondrial density, and reduced physical capacity. This is why what you eat on GLP-1 medications matters as much as how much you eat.
Mechanism 2: blood sugar dysregulation
GLP-1 medications were originally developed for type 2 diabetes, and their primary mechanism involves blood sugar regulation. Semaglutide mimics your natural GLP-1, telling your body to release more insulin and less glucagon. This lowers blood sugar. In people without diabetes, this glucose-lowering effect can sometimes overshoot, producing episodes of relative hypoglycemia.
You do not need to hit clinical hypoglycemia (below 70 mg/dL) to feel the effects. A rapid drop from 120 to 85 can produce fatigue, brain fog, shakiness, and difficulty concentrating. These reactive hypoglycemic episodes often occur 2-4 hours after eating, which is why many GLP-1 users report a specific mid-afternoon energy crash.
The blood sugar mechanism also explains why some people feel worse on certain days. Carbohydrate-heavy meals can trigger a larger insulin response from the medication, leading to a more pronounced blood sugar drop afterward. Conversely, protein and fat-focused meals produce more stable blood sugar curves and less post-meal fatigue. Knowing which foods to avoid while on GLP-1 medications can significantly reduce these glucose-related energy crashes.
This mechanism interacts with the caloric deficit mechanism in a particularly problematic way. When you are eating less overall AND each meal produces blood sugar fluctuations, you get a double hit to your energy levels. Your baseline energy is lower from the deficit, and then you get periodic drops below that already lowered baseline from glucose fluctuations.
Mechanism 3: nutrient malabsorption and deficiency
This mechanism gets criminally underreported. GLP-1 medications slow gastric emptying by 30-50%. Food sits in your stomach and upper digestive tract significantly longer. While this helps with appetite suppression, it also changes how nutrients get absorbed.
Vitamin B12 absorption is particularly affected. B12 requires intrinsic factor produced in the stomach and a specific pH environment for proper absorption. Altered gastric motility and reduced food intake can compromise both of these requirements. B12 is essential for red blood cell production, neurological function, and cellular energy metabolism. When levels drop, fatigue is one of the first and most prominent symptoms.
The research is compelling. A retrospective observational study found that nutritional deficiencies were diagnosed in 12.7% of patients within 6 months of starting GLP-1 therapy, climbing to 22.4% within 12 months. That is nearly one in four patients developing measurable nutritional deficiencies within their first year. The combination of semaglutide with B12 supplementation has become a standard recommendation for exactly this reason.
Beyond B12, iron absorption can be affected. Iron deficiency anemia produces fatigue, weakness, and exercise intolerance. Vitamin D levels may drop due to reduced dietary intake. Magnesium, zinc, and folate can all become insufficient. Each of these deficiencies has its own fatigue-producing pathway, and when multiple deficiencies co-occur, the fatigue effect is multiplicative, not additive.
SeekPeptides members frequently discuss the importance of comprehensive bloodwork panels before and during GLP-1 therapy. Knowing your baseline nutrient levels before starting treatment gives you a reference point for identifying deficiency-driven fatigue later.
Mechanism 4: electrolyte imbalance
Electrolyte imbalances are the silent fatigue driver that most people never check for. The gastrointestinal side effects of GLP-1 medications, including nausea, vomiting, and diarrhea, cause direct losses of sodium, potassium, and magnesium. At the same time, reduced food intake means less electrolyte replenishment from dietary sources.
Hypokalemia (low potassium), hyponatremia (low sodium), and hypomagnesemia (low magnesium) are the most common electrolyte disturbances associated with GLP-1 therapy. Even modest imbalances produce symptoms ranging from mild fatigue to muscle weakness, cramping, heart palpitations, and cognitive difficulty.
The FDA labels for GLP-1 medications include warnings about dehydration risk and acute kidney injury, particularly during episodes of significant gastrointestinal illness. But subclinical dehydration, the kind that does not trigger a warning label but absolutely tanks your energy, is far more common.
Here is the mechanism in detail. GLP-1 receptor agonists can affect kidney function by increasing sodium excretion. Combined with reduced fluid intake (because you are not eating as much and thirst signals may be dampened), you get a slow, steady depletion of electrolyte reserves. Potassium is critical for muscle contraction and nerve signaling. Magnesium participates in over 300 enzymatic reactions including energy production. Sodium maintains fluid balance and nerve function. When any of these drop, fatigue follows.
The tricky part is that GLP-1 medications can suppress thirst signals alongside appetite. You may not feel thirsty even when your body needs more fluids and electrolytes. This is why proactive hydration with electrolyte supplementation, rather than drinking only when thirsty, becomes important during treatment.
Mechanism 5: hormonal disruption
GLP-1 medications do not just affect insulin and glucagon. They influence a broader hormonal network that impacts energy, mood, and recovery.
Cortisol is one piece of this puzzle. Research shows that GLP-1 receptor activation potently activates the hypothalamic-pituitary-adrenal (HPA) axis in both humans and rodents, resulting in increased ACTH and cortisol concentrations. During the initial adaptation period, cortisol patterns can shift, with higher nighttime cortisol disrupting sleep quality and morning cortisol patterns affecting daytime energy.
The rapid weight loss induced by GLP-1 medications also triggers hormonal adaptations. Leptin levels drop as fat mass decreases. Ghrelin signaling changes. Thyroid function can be subtly affected, with some researchers recommending thyroid function tests as part of routine screening for patients on GLP-1 therapy. Testosterone may decrease in men experiencing rapid weight loss, and estrogen metabolism shifts in women.
These hormonal changes create a cascading fatigue effect. Lower leptin tells your brain that energy stores are depleting, triggering energy conservation. Altered thyroid function slows metabolism. Changed cortisol patterns disrupt sleep. Each hormonal shift independently produces some fatigue, and together they create the compound exhaustion that many GLP-1 users describe.
For people already exploring peptides for hormone balance, understanding how GLP-1 medications interact with the broader endocrine system provides important context for managing multiple protocols simultaneously.
Mechanism 6: sleep architecture disruption
Poor sleep on GLP-1 medications is more common than most prescribers acknowledge. Several factors contribute to disrupted sleep that then feeds back into daytime fatigue.
Delayed gastric emptying means food is still being processed when you go to bed. Feelings of fullness, bloating, or mild nausea can interfere with falling asleep and reduce sleep quality. Acid reflux, more common with slowed gastric emptying, can cause nighttime awakenings that fragment sleep architecture.
The hormonal changes discussed above also play a role. Elevated nighttime cortisol from HPA axis activation makes it harder to fall asleep and reduces time spent in deep, restorative sleep stages. Altered blood sugar regulation can cause nocturnal hypoglycemia that triggers cortisol and adrenaline release, causing you to wake up feeling alert at 3 AM and then exhausted by morning. Researchers who study peptides and sleep regulation have documented how these disruptions compound over time.
Reduced caloric intake can also affect sleep neurotransmitter production. Tryptophan, the amino acid precursor to serotonin and melatonin, comes primarily from dietary protein. If protein intake drops significantly, your body may produce less melatonin, directly affecting sleep onset and quality.
The sleep-fatigue connection creates a vicious cycle. Poor sleep causes daytime fatigue. Daytime fatigue reduces activity levels. Reduced activity affects sleep quality the next night. Breaking this cycle requires addressing the root causes rather than just the symptoms.
The GLP-1 fatigue timeline: what to expect and when
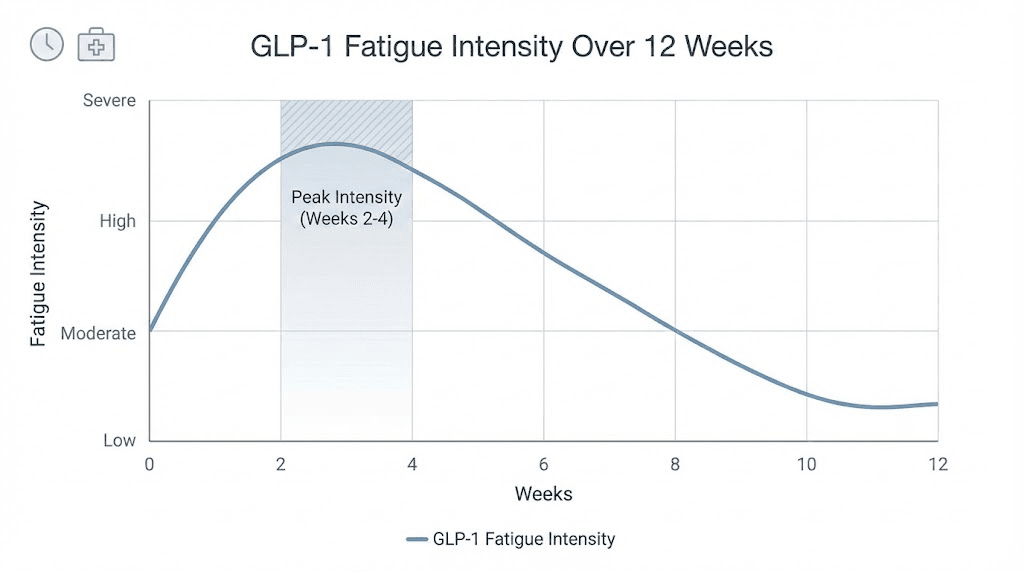
Understanding when fatigue peaks and when it resolves helps you plan your management strategy and set realistic expectations. The timeline is not identical for everyone, but the general pattern is remarkably consistent across the research literature and patient reports.
Weeks 1-2: the initial adjustment
Fatigue often appears within the first few days of starting a GLP-1 medication or escalating to a new dose. During this phase, your body is encountering the medication for the first time, and multiple adjustment processes happen simultaneously.
Appetite drops suddenly, often more dramatically than you expect. Caloric intake plummets before you have had time to adjust your eating habits to focus on nutrient density. Blood sugar patterns shift as the medication takes effect. Gastric emptying slows. Your brain is receiving signals it has never processed before.
The fatigue during this phase is typically moderate. Most people describe it as feeling "sluggish" or "a step behind." You can function, but everything requires more effort. This is the phase where many people make the mistake of pushing through with coffee and stimulants rather than addressing the underlying causes.
Weeks 2-4: the potential peak
For many people, fatigue intensifies during weeks 2-4, especially if they are escalating doses during this period. This is when the cumulative effects of reduced caloric intake, emerging nutrient depletions, and ongoing metabolic adjustment converge.
If you are on a microdosing protocol, the fatigue peak may be milder because the dose escalation is more gradual. If you are on standard escalation schedules, this period can feel significantly worse than the first two weeks.
The 2-4 week window is critical because it is when most people either develop effective management strategies or begin to struggle. The key insight is that this peak is temporary for the vast majority of users. Knowing that helps psychologically, but it does not replace the need for active management.
Weeks 4-8: the adaptation phase
Most people begin to notice improvement around weeks 4-6. Your body has adapted to the new metabolic environment. Blood sugar regulation stabilizes. You have hopefully adjusted your eating patterns to focus on nutrient density. The initial gastrointestinal side effects are typically improving, which means better nutrient absorption and fewer electrolyte losses.
This does not mean fatigue disappears completely at week 4. It means the trajectory shifts from worsening to improving. You might go from feeling exhausted all day to feeling tired only in the afternoons. From needing a nap daily to managing without one. The improvement is gradual but real.
Week 8 and beyond: the new normal
By week 8, most patients report significant improvement in fatigue. The body has largely adapted to the metabolic changes. Nutrient intake has been optimized. Electrolyte management is established. Sleep patterns have adjusted.
However, some people experience persistent fatigue beyond this timeline. If fatigue is not improving by week 8-10, it is time for a deeper investigation. Comprehensive blood work should check B12, iron, ferritin, vitamin D, thyroid function, electrolytes, and metabolic markers. The fatigue may be driven by a specific deficiency that requires targeted intervention rather than general adaptation.
Some patients require dose adjustment. Moving down one dose level, or switching between medications, can sometimes resolve persistent fatigue while maintaining adequate weight loss. People who have explored switching from tirzepatide to semaglutide or vice versa sometimes find that one medication produces less fatigue for their individual physiology.
Dose escalation fatigue: the recurring pattern
Every dose increase essentially resets the fatigue clock. When you escalate from 0.25 mg to 0.5 mg of semaglutide, or from 2.5 mg to 5 mg of tirzepatide, expect a mini-version of the initial fatigue pattern. It typically peaks within 3-7 days of the dose increase and resolves faster than the initial onset, usually within 1-2 weeks.
Understanding tirzepatide dosing in units and semaglutide dosage charts helps you anticipate these escalation periods and prepare your management strategies in advance. Many experienced users time their dose escalations strategically, avoiding weeks when they have important commitments that require peak energy.
GLP-1 fatigue versus other causes of tiredness
One of the biggest mistakes people make is attributing ALL their fatigue to the GLP-1 medication when other treatable causes are contributing. Before assuming your tiredness is purely medication-related, rule out these common confounders.
Thyroid dysfunction
Thyroid issues deserve special attention in the context of GLP-1 therapy. Weight loss itself can affect thyroid function, and the rapid metabolic changes induced by these medications create additional thyroid stress. If you had subclinical hypothyroidism before starting treatment, GLP-1 medications may unmask or worsen it.
The research on GLP-1 receptor agonists and thyroid function is still evolving, with somewhat conflicting results. Some studies suggest potential impacts on thyroid volume and TSH levels. Routine thyroid screening is advisable for anyone experiencing persistent fatigue on GLP-1 therapy, particularly if fatigue is not improving on the expected timeline.
Iron deficiency anemia
Reduced food intake means reduced iron intake. Women of reproductive age are especially vulnerable. Iron deficiency can develop gradually, with fatigue appearing long before a full-blown anemia shows up on standard blood work. Checking ferritin levels (not just hemoglobin) provides earlier detection of iron depletion.
Sleep apnea changes
This is a counterintuitive one. Weight loss from GLP-1 medications can actually improve sleep apnea, but the transition period can temporarily worsen sleep quality. If you use a CPAP machine, your pressure settings may need adjustment as you lose weight. Additionally, changes in neck circumference and airway anatomy during weight loss can create a transitional period where sleep quality fluctuates.
Depression and mood changes
Rapid weight loss, hormonal changes, and altered eating patterns can all affect mood. Some GLP-1 users report depressive symptoms that contribute to fatigue. If your fatigue is accompanied by persistent low mood, loss of interest in activities, or difficulty concentrating beyond what you would expect from tiredness alone, a mental health evaluation is warranted. Research into peptides for depression and anxiety provides additional context for understanding these mood-energy connections.
Medication interactions
GLP-1 medications can interact with other medications, particularly those that affect blood sugar or liver function. If you are on multiple medications, discuss potential fatigue-producing interactions with your prescriber. Some people taking phentermine and semaglutide together or tirzepatide with B12 find that combination approaches help address both weight loss and energy simultaneously.
The complete GLP-1 fatigue management protocol
This section is the actionable core of this guide. These are specific, evidence-based strategies organized by the mechanism they target. Implement them systematically rather than trying everything at once.
Nutrition strategies: the foundation
Nutrition is the single most impactful intervention for GLP-1 fatigue. The challenge is that the very medication making you tired is also making you not want to eat. You need to eat strategically, prioritizing nutrient density in smaller volumes.
Protein first, always. Aim for 1.6 to 2.2 grams of protein per kilogram of ideal body weight per day. At minimum, every meal should contain 25-30 grams of complete protein. Protein provides sustained energy, preserves muscle mass, supports neurotransmitter production, and has the highest thermic effect of any macronutrient. Good sources include chicken, fish, eggs, Greek yogurt, tofu, and lean beef.
Spread protein intake across all meals. Your body can only utilize approximately 25-40 grams of protein for muscle protein synthesis per meal. Eating 80 grams at dinner and skipping protein at breakfast is significantly less effective than distributing it across three or four meals. This even distribution also provides more stable energy throughout the day.
Complex carbohydrates for sustained energy. Despite what some advice suggests, you should not eliminate carbohydrates on GLP-1 therapy. Complex carbohydrates provide glucose for brain function, support serotonin production (which affects both mood and sleep), and prevent the blood sugar crashes that compound fatigue. Focus on whole grains, sweet potatoes, legumes, and vegetables. Avoid simple sugars and refined carbohydrates that cause rapid blood sugar spikes followed by crashes.
Healthy fats for satiety and nutrient absorption. Fat-soluble vitamins (A, D, E, K) require dietary fat for absorption. Including healthy fats at each meal supports nutrient status and provides slow-burning energy. Avocado, olive oil, nuts, seeds, and fatty fish are excellent choices.
Meal timing matters. Eat your last meal at least 3-4 hours before bedtime to allow adequate gastric emptying. With GLP-1 medications slowing digestion, going to bed with a full stomach is a recipe for poor sleep and next-day fatigue. Morning and midday meals should be your largest, providing fuel when you need energy most.
A structured semaglutide diet plan or tirzepatide diet plan can help ensure you are hitting these nutritional targets despite reduced appetite. Planning meals in advance prevents the common trap of simply eating whatever is convenient (which is usually nutrient-poor) when your appetite finally appears.
Hydration and electrolyte management
Aim for a minimum of 64 ounces (about 2 liters) of water daily. If you are exercising, in a hot environment, or experiencing gastrointestinal side effects, increase to 80-100 ounces. Do not wait until you feel thirsty. GLP-1 medications can suppress thirst signals alongside appetite.
Electrolyte supplementation protocol:
Sodium: 1,500-2,300 mg daily from food and supplementation. If you are experiencing nausea or vomiting, you may need more.
Potassium: 2,600-3,400 mg daily, primarily from food sources (bananas, potatoes, spinach, avocado). Supplement only under medical guidance.
Magnesium: 310-420 mg daily. Magnesium glycinate or citrate forms are well-absorbed and gentle on the stomach.
Commercial electrolyte supplements designed for daily use (not just sports drinks loaded with sugar) can help. Look for products that provide sodium, potassium, and magnesium without excessive sugar or artificial ingredients. Add electrolytes to your morning water before anything else. This single habit can noticeably improve energy levels within days.
Monitor for signs of dehydration: dark urine, dry mouth, headaches, dizziness when standing. These are warning signs that your fluid and electrolyte intake needs immediate attention. The FDA includes warnings about dehydration risk with GLP-1 medications for good reason. People who are also researching semaglutide with glycine or tirzepatide glycine supplementation should factor these hydration needs into their overall protocol.
Targeted supplementation
Based on the mechanisms discussed above, certain supplements directly address the causes of GLP-1 fatigue. This is not about throwing supplements at the problem. It is about targeted correction of specific deficiencies that the medication creates or worsens.
Vitamin B12: essential. Supplement with 1,000-2,000 mcg of methylcobalamin (the active form) daily. B12 deficiency is one of the most common and most impactful nutrient depletions on GLP-1 therapy. Sublingual forms bypass potential absorption issues in the gut. For those considering compounded semaglutide with B12 or tirzepatide with B12, these formulations address the deficiency concern directly within the medication itself.
Iron (if deficient): Only supplement iron based on blood work. Excess iron is harmful. If your ferritin is below 30 ng/mL, supplementation is typically warranted. Take iron with vitamin C for better absorption and on an empty stomach if tolerated.
Vitamin D: 2,000-5,000 IU daily, depending on baseline levels. Vitamin D is involved in immune function, mood regulation, and energy metabolism. Most people on GLP-1 therapy benefit from supplementation, especially during winter months or if predominantly indoors.
Magnesium: 200-400 mg of magnesium glycinate before bed. This serves double duty. It addresses potential magnesium depletion AND improves sleep quality. Magnesium glycinate is preferred because it does not cause the digestive issues that other forms can, which is important when your GI system is already stressed by the medication.
CoQ10: 100-200 mg daily. CoQ10 is essential for mitochondrial energy production. Some researchers suggest that metabolic stress from rapid weight loss can increase CoQ10 demand. It is particularly relevant for people also taking statins, which deplete CoQ10.
Get comprehensive blood work done before starting GLP-1 therapy and repeat it at 3-month and 6-month intervals. The panel should include CBC, comprehensive metabolic panel, B12, folate, iron studies (including ferritin), vitamin D, thyroid function (TSH, free T3, free T4), and magnesium. This data takes the guesswork out of supplementation.
Exercise and movement optimization
Exercise on GLP-1 medications requires a fundamentally different approach than exercise without them. Your recovery capacity is reduced. Your caloric buffer is smaller. And your body is already under metabolic stress from the medication and weight loss.
Start gentle, especially during weeks 1-4. Walking 15-30 minutes daily is the ideal starting point. Walking improves insulin sensitivity, aids digestion, boosts mood, and increases energy without creating significant recovery demand. Post-meal walks are especially beneficial because they help stabilize blood sugar and reduce the afternoon energy crash.
Add resistance training, but scale appropriately. Resistance training is critical for preserving muscle mass during GLP-1 mediated weight loss. Studies show that even modest resistance training twice weekly significantly benefits lean mass retention. However, reduce volume and intensity by 30-40% from your pre-medication levels during the first 4-6 weeks. Increase gradually as your energy and recovery improve.
The protein intake recommendations above become even more important if you are exercising. Training creates additional protein demand for muscle repair and growth. If you are exercising and eating less due to appetite suppression, the protein gap widens. Time protein intake around your workouts, with 25-30 grams within 1-2 hours post-training.
Avoid high-intensity training during the adaptation phase. HIIT, heavy compound lifts, and endurance training all create significant caloric and recovery demands. During the first 4-8 weeks of GLP-1 therapy, your body does not have the metabolic headroom for these demands. Pushing through with intense exercise while fatigued is a path to overtraining syndrome, injury, and worsened fatigue.
Listen to your body more carefully than you normally would. If a planned workout feels like an insurmountable obstacle, scale it back. A 20-minute walk is infinitely better than a skipped gym session because you were too exhausted to go. Consistency at lower intensity beats sporadic high-intensity efforts during this phase. Resources on peptides for athletic performance and muscle growth provide additional strategies for maintaining physical performance during this adjustment period.
Sleep optimization protocol
Fixing sleep is one of the highest-leverage interventions for GLP-1 fatigue, because poor sleep amplifies every other mechanism. Address these factors systematically.
Meal timing for sleep quality. Stop eating 3-4 hours before bed. With slowed gastric emptying, this is even more important than for the general population. If you must eat closer to bedtime, choose small, protein-focused snacks rather than full meals. Avoid fatty foods close to bedtime since they take longest to digest.
Manage acid reflux. Elevate the head of your bed 4-6 inches. Avoid trigger foods (citrus, tomatoes, chocolate, caffeine, alcohol) in the evening. Consider a proton pump inhibitor or H2 blocker if reflux is significantly disrupting sleep.
Blood sugar stability overnight. A small protein and fat snack (like a handful of almonds or a cheese stick) 1-2 hours before bed can help prevent nocturnal hypoglycemia without creating the gastric loading problems of a full meal. This is particularly important during the first few weeks when blood sugar regulation is still adjusting.
Sleep hygiene fundamentals. Cool bedroom (65-68 degrees F), complete darkness, consistent wake time (even on weekends), no screens 30-60 minutes before bed, and a wind-down routine. These basics become even more important when your sleep architecture is already being challenged by the medication.
Magnesium before bed. As mentioned in supplementation, 200-400 mg of magnesium glycinate 30-60 minutes before bed improves sleep onset and quality. This is one of the few supplements with solid evidence for sleep improvement that also addresses an actual deficiency risk on GLP-1 therapy.
If sleep remains problematic despite these interventions, consider a sleep study. Weight loss can unmask or change sleep apnea severity, and CPAP settings may need adjustment. People researching DSIP peptide benefits and pineal peptides for sleep often find that foundational sleep hygiene must be in place before any supplementation strategy works effectively.
Dose management strategies
Sometimes the most effective fatigue management strategy is adjusting the medication itself. This should always be done in consultation with your prescriber, but understanding the options empowers you to have an informed conversation.
Slower dose escalation. Standard escalation schedules move relatively quickly. For semaglutide, the typical schedule is 0.25 mg for 4 weeks, then 0.5 mg for 4 weeks, escalating every 4 weeks. For people who experience significant fatigue, extending each dose level to 6-8 weeks allows more time for adaptation before adding the next metabolic challenge. Many prescribers are comfortable with this approach when patients communicate their symptoms clearly.
Microdosing. Some prescribers now use microdosing protocols that use smaller, more frequent dose increases. Instead of jumping from 2.5 mg to 5 mg of tirzepatide, for example, an intermediate step at 3.75 mg gives the body more time to adjust. The compounded tirzepatide dosage calculator can help determine intermediate doses for these protocols.
Dose reduction. If fatigue is severe and persistent, stepping back one dose level while maintaining the management strategies above often produces the best overall outcome. Slightly slower weight loss with sustainable energy is better than faster weight loss while barely functioning.
Medication switching. Some people tolerate semaglutide better than tirzepatide or vice versa. The side effect profiles of semaglutide versus tirzepatide differ enough that switching can sometimes resolve fatigue while maintaining efficacy. The conversion charts between medications help ensure a smooth transition.
Injection timing. Some people find that the day of injection and the 1-2 days following produce the most fatigue. Timing your weekly injection for Thursday or Friday evening allows the peak fatigue period to fall on the weekend when you can rest more. Knowing the best time of day to take semaglutide and planning around your schedule can make a measurable difference in how manageable the fatigue feels.
Specific fatigue patterns and what they mean
Not all GLP-1 fatigue presents the same way. The specific pattern of your fatigue provides clues about which mechanisms are dominant, which helps you target your interventions more precisely.
Morning fatigue: waking up exhausted
If you wake up feeling like you did not sleep, even after 7-8 hours in bed, the primary suspects are sleep quality disruption and overnight blood sugar instability.
Check your sleep quality first. Are you waking during the night? Do you notice acid reflux symptoms? Is your partner reporting new or changed snoring? Any of these point to sleep architecture problems that need targeted intervention.
Overnight blood sugar drops can trigger cortisol and adrenaline release that disrupts deep sleep without fully waking you. You wake up feeling exhausted because your body spent part of the night in a stress response rather than restorative sleep. A small protein and fat snack before bed, as discussed above, often resolves this pattern.
Afternoon crash: the 2-3 PM wall
This is the most commonly reported GLP-1 fatigue pattern. The afternoon crash typically indicates blood sugar dysregulation combined with inadequate midday nutrition.
The mechanism usually works like this. Breakfast (if eaten) and lunch trigger an insulin response amplified by the GLP-1 medication. Blood sugar drops more aggressively than it would without the medication. By mid-afternoon, you hit a glucose nadir that produces fatigue, brain fog, and difficulty concentrating.
The fix involves eating a balanced lunch with adequate protein and complex carbohydrates, followed by a small afternoon snack combining protein and healthy fat around 2 PM. This pre-emptive approach prevents the crash rather than trying to recover from it with coffee or sugar, both of which create their own rebound crash later.
Post-injection fatigue: 24-72 hour window
Some people experience their worst fatigue in the 1-3 days following their weekly injection. This pattern is related to peak drug levels, when the medication concentration in your blood is highest and its effects on appetite, blood sugar, and gastric emptying are most pronounced.
Managing this pattern involves strategic injection timing (as discussed above) and being especially proactive about nutrition and hydration on injection days. Some people find that eating a protein-rich meal before their injection and having easy-to-consume nutrient-dense foods ready for the following day helps maintain energy through the peak-level period.
Exercise-related fatigue: the recovery gap
If your fatigue is primarily triggered or worsened by exercise, you are likely in a recovery deficit. Your caloric intake is not supporting the energy demands of both the medication-induced weight loss AND your exercise program.
The solution is not to stop exercising. It is to right-size your exercise to match your current recovery capacity. Reduce volume by 30-50%. Focus on compound movements with moderate intensity. Ensure protein intake is adequate. And give yourself permission to have lighter training days when energy is low. Understanding how to balance weight loss and muscle preservation is essential for navigating this phase.
Persistent, unrelenting fatigue
If your fatigue is constant, not improving on the expected timeline, and not responsive to the management strategies above, it is time for a thorough medical evaluation. Persistent fatigue may indicate a specific nutrient deficiency (B12, iron, vitamin D), thyroid dysfunction, adrenal insufficiency, or an unrelated medical condition that happened to emerge around the same time you started GLP-1 therapy.
Do not assume that all fatigue on a GLP-1 medication is caused by the medication. Use it as a prompt for comprehensive evaluation rather than a reason to simply tolerate feeling terrible.
GLP-1 fatigue and weight loss: finding the balance
One of the most common concerns about managing GLP-1 fatigue is whether the interventions will slow weight loss. This fear keeps many people suffering unnecessarily through fatigue rather than implementing changes that would help.
Here is the truth. Managing fatigue properly SUPPORTS weight loss rather than undermining it. When you are exhausted, you move less, exercise less, make worse food choices, sleep poorly, and produce more cortisol, all of which slow weight loss. Adequate nutrition, proper supplementation, and managed exercise actually optimize the weight loss process.
The research is clear. Combining high protein diet and consistent exercise with GLP-1 treatment has the greatest benefit in preserving bone and muscle mass compared to diet alone or high protein diet alone. Muscle preservation maintains metabolic rate. Higher metabolic rate means more efficient fat loss. Better energy means more activity, which means more calories burned. Managing fatigue creates a virtuous cycle rather than the vicious cycle of exhaustion and metabolic slowdown.
People who monitor their tirzepatide before and after results or track their semaglutide progress timeline often find that the phases where they managed fatigue best corresponded with the most consistent weight loss. The phases where they pushed through exhaustion while under-eating often produced plateaus.
The specific numbers support this. People who are not losing weight on semaglutide or not losing weight on tirzepatide often discover that metabolic adaptation from excessive caloric restriction is the culprit. Eating too little while fatigued creates a metabolic environment that resists weight loss. The solution, eating more (but still at a deficit), exercising moderately, and sleeping well, simultaneously resolves the fatigue AND restarts weight loss.
When to talk to your prescriber
Fatigue management for GLP-1 medications should be a collaborative process with your healthcare provider. Here are the specific situations that warrant a conversation.
Fatigue that prevents normal daily functioning. If you cannot work, care for your family, or drive safely due to fatigue, your provider needs to know. Dose adjustment or temporary discontinuation may be necessary.
Fatigue that has not improved after 8 weeks. Persistent fatigue beyond the expected adaptation period suggests an underlying cause that needs investigation.
Fatigue accompanied by other concerning symptoms. Chest pain, severe headaches, confusion, significant mood changes, muscle weakness, numbness or tingling, or severe gastrointestinal symptoms all warrant prompt medical evaluation.
Fatigue following dose increase that is significantly worse than previous levels. While some fatigue with dose escalation is expected, a dramatically worse response may indicate that a particular dose is not right for you.
New-onset fatigue after months of treatment. If you have been on a stable dose for months and suddenly develop new fatigue, this is not typical adaptation. It may indicate a developing nutrient deficiency, thyroid change, or other medical issue. Understanding semaglutide withdrawal symptoms is also important context, as some people experience fatigue after dose reduction or discontinuation.
When you talk to your prescriber, be specific. Track your fatigue patterns for 1-2 weeks before the appointment. Note when fatigue is worst (time of day, relation to injection, relation to meals), what makes it better, what makes it worse, and how it affects your daily functioning. This information helps your provider make informed decisions about management.
GLP-1 fatigue and alcohol
Alcohol and GLP-1 medications have a complicated relationship when it comes to fatigue. Many people notice significantly reduced alcohol tolerance on these medications, and even moderate drinking can produce disproportionate fatigue.
The mechanisms are straightforward. GLP-1 medications alter liver metabolism. Alcohol processing may be slower. Blood sugar effects of alcohol (initial spike followed by drop) are amplified by the medication. Dehydration from alcohol compounds the dehydration risk from the medication. Sleep quality, already vulnerable on GLP-1 therapy, is further degraded by alcohol.
If you choose to drink on semaglutide or tirzepatide, reduce your intake significantly from pre-medication levels. One drink on a GLP-1 medication may feel like two or three. Ensure extra hydration with electrolytes before, during, and after drinking. And expect more fatigue the following day than you would normally experience from the same amount of alcohol.
Specific management by medication
While the general principles above apply to all GLP-1 medications, there are nuances worth noting for specific drugs.
Semaglutide-specific considerations
Semaglutide has a half-life of approximately one week, meaning drug levels remain relatively stable between weekly injections. This produces a more consistent fatigue pattern than shorter-acting GLP-1 medications. The trade-off is that you cannot easily time your fatigue around specific days.
The appetite suppression from semaglutide tends to be steady rather than peaking and troughing. This means caloric intake reduction is relatively consistent, which makes nutritional planning more predictable. Use this stability to your advantage by establishing consistent meal times and nutrient targets.
For people using compounded semaglutide, the formulation may affect absorption kinetics and therefore fatigue patterns. Compounded versions with added B12 or glycine may help address some fatigue mechanisms directly within the medication. Understanding proper reconstitution is important for maintaining medication efficacy and consistent dosing.
Tirzepatide-specific considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist. The GIP component may provide some protective effect against fatigue, as GIP signaling is involved in nutrient absorption and energy metabolism. This could partially explain why tirzepatide fatigue rates are slightly lower than semaglutide despite producing greater weight loss.
The dual mechanism also means that tirzepatide takes a somewhat different course in terms of side effect patterns. Some people report that fatigue is more pronounced immediately after injection and fades more quickly compared to semaglutide. If this matches your experience, strategic injection timing becomes especially valuable.
Tirzepatide users should pay particular attention to the foods to avoid recommendations, as the dual receptor activation may produce stronger gastrointestinal responses to certain food triggers. Tirzepatide niacinamide combinations and tirzepatide glycine formulations are increasingly popular approaches to addressing both weight loss and side effect management.
Retatrutide and next-generation GLP-1 agents
As newer GLP-1 medications enter the market, fatigue profiles continue to evolve. Retatrutide, a triple receptor agonist (GIP/GLP-1/glucagon), produces particularly aggressive weight loss. Early data suggests that the fatigue profile may be different from existing medications, with the glucagon component potentially affecting energy regulation in unique ways.
The general management principles apply regardless of which GLP-1 medication you use. Nutrition, hydration, electrolytes, supplementation, sleep, and appropriate exercise are universally important. The specifics of timing and intensity may vary, and working with your prescriber to adjust your approach based on your individual medication and response is always advisable.
For comprehensive retatrutide dosing schedules and dosage charts, these resources provide the detailed protocol information that helps with planning around fatigue patterns.
GLP-1 fatigue in special populations
Women
Women experience GLP-1 fatigue differently than men, and the menstrual cycle adds a complicating variable. Estrogen and progesterone fluctuations throughout the month interact with the metabolic effects of GLP-1 medications, creating cyclical fatigue patterns on top of the medication-related fatigue.
The week before menstruation (luteal phase) often produces the worst fatigue for women on GLP-1 therapy. Progesterone is naturally higher, metabolic rate increases slightly, and fluid retention changes electrolyte dynamics. Proactively increasing caloric intake, electrolytes, and rest during this phase can prevent the compound fatigue that some women describe as debilitating.
Iron needs are also higher for menstruating women. The combination of menstrual blood loss and reduced dietary iron intake from GLP-1 appetite suppression creates a perfect storm for iron deficiency. Regular ferritin monitoring (every 3 months) is especially important. Resources covering peptides for women over 40 and peptides for menopause provide additional context for managing GLP-1 therapy across different life stages.
Older adults
Adults over 60 face additional fatigue risks on GLP-1 medications. Baseline muscle mass is lower. Nutrient absorption efficiency is already declining with age. Kidney function may be reduced, affecting electrolyte balance. And the risk of sarcopenia (age-related muscle loss) is amplified by the weight loss medication.
For older adults, the protein recommendations become even more critical. Some researchers recommend the higher end of the protein range (2.0-2.2 g/kg ideal body weight) for adults over 60 on GLP-1 therapy. Resistance training is not optional in this population. It is essential for preventing the muscle loss that drives fatigue and reduces functional independence.
People with pre-existing conditions
If you have hypothyroidism, diabetes, adrenal insufficiency, or other conditions that affect energy, GLP-1 fatigue may be more pronounced and require more aggressive management. Your existing conditions should be optimally managed before adding the metabolic stress of GLP-1 therapy, and medication dosages for existing conditions may need adjustment as your weight changes.
Diabetic patients need particular attention to blood sugar monitoring during GLP-1 therapy. The combination of diabetes medications with GLP-1 agonists increases hypoglycemia risk, and hypoglycemia is a direct cause of fatigue. If you are using semaglutide or tirzepatide for diabetes management alongside weight loss, close monitoring and frequent communication with your endocrinologist are essential.
The complete GLP-1 fatigue management checklist
Use this checklist as your daily and weekly framework for managing GLP-1 fatigue. It consolidates all the strategies discussed above into an actionable reference.
Daily essentials:
Consume 1.6-2.2 g protein per kg ideal body weight across 3-4 meals
Drink 64-100 oz water with electrolytes (start your morning with electrolyte water)
Take supplements: B12, vitamin D, magnesium (before bed)
Walk 15-30 minutes (post-meal walks ideal)
Stop eating 3-4 hours before bed
Maintain consistent sleep and wake times
Weekly checkpoints:
Resistance train 2-3 times (moderate intensity during adaptation)
Track fatigue patterns (time of day, severity, triggers)
Assess hydration (urine color check)
Review food intake quality (adequate protein at every meal?)
Note any new or worsening symptoms
Monthly reviews:
Evaluate overall fatigue trajectory (improving, stable, worsening?)
Adjust supplementation based on symptoms
Discuss progress with prescriber if fatigue is not improving
Consider blood work if persistent issues
Quarterly blood work:
CBC, metabolic panel, B12, folate, iron studies, ferritin, vitamin D, thyroid function, magnesium
Adjust supplementation and management based on results
For researchers serious about optimizing their GLP-1 protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed dosing guides, calculators, and expert-reviewed management strategies that account for the individual factors most resources ignore.
Frequently asked questions
How long does GLP-1 fatigue last?
For most people, GLP-1 fatigue peaks during weeks 2-4 of treatment and improves significantly by weeks 4-8. Each dose escalation may produce a temporary fatigue increase that typically resolves within 1-2 weeks. If fatigue persists beyond 8-10 weeks without improvement, consult your prescriber for blood work and potential dose adjustment. Our guides on how long semaglutide takes to work and how long tirzepatide takes to work cover the full treatment timeline.
Does tirzepatide cause less fatigue than semaglutide?
Clinical trial data suggests slightly lower fatigue rates with tirzepatide (5-7% depending on dose) compared to semaglutide (11%). The GIP receptor activation in tirzepatide may provide some protective effect against energy depletion. However, individual responses vary significantly, and some people experience more fatigue on tirzepatide. The complete side effect comparison can help you understand the differences.
Can B12 supplementation help with GLP-1 fatigue?
Yes. B12 deficiency is one of the most common and impactful nutrient depletions associated with GLP-1 therapy. Supplementing with 1,000-2,000 mcg of methylcobalamin daily addresses this specific mechanism. Some patients use compounded semaglutide with B12 or tirzepatide with B12 formulations for convenience.
Should I exercise if I am fatigued on a GLP-1 medication?
Yes, but adjust intensity. Walking 15-30 minutes daily and moderate resistance training 2-3 times weekly supports energy levels, preserves muscle mass, and improves sleep quality. Avoid high-intensity exercise during the adaptation phase. Scale effort to match your current energy level rather than pushing through exhaustion. Our athletic performance guide covers exercise optimization in detail.
Is GLP-1 fatigue a sign that the medication is not right for me?
Not necessarily. Some fatigue during the adaptation period is normal and expected. It becomes a concern if it is severe enough to prevent normal daily functioning, does not improve on the expected timeline, or is not responsive to the management strategies outlined in this guide. Persistent, severe fatigue warrants a conversation with your prescriber about dose adjustment or alternative medications.
Can I take caffeine for GLP-1 fatigue?
Moderate caffeine intake (200-400 mg daily, equivalent to 2-4 cups of coffee) is generally safe and can help with energy. However, caffeine is not addressing the root cause and can worsen dehydration and sleep quality if overused. Limit caffeine to morning and early afternoon only, and ensure you are increasing water intake proportionally.
Does the fatigue mean I am losing weight too fast?
Sometimes. If you are losing more than 1-2% of body weight per week, your caloric deficit may be too aggressive. Rapid weight loss increases the risk of muscle loss, nutritional deficiency, and metabolic adaptation, all of which produce fatigue. People experiencing weight loss plateaus after initial rapid loss often discover that excessive restriction was the cause of both their fatigue and their stalled progress.
What blood tests should I get for GLP-1 fatigue?
Request a comprehensive panel including CBC, basic metabolic panel, B12, folate, iron and ferritin, vitamin D, TSH and free T3/T4, and magnesium. This covers the most common treatable causes of fatigue on GLP-1 therapy. Get baseline levels before starting treatment and repeat every 3-6 months.
External resources
Nutritional Priorities to Support GLP-1 Therapy for Obesity - Joint Advisory (PMC)
Nutritional Deficiencies and Muscle Loss in Adults Using GLP-1 Receptor Agonists (PMC)
In case I do not see you, good afternoon, good evening, and good night. May your energy stay steady, your nutrients stay balanced, and your GLP-1 results stay on track.