Feb 28, 2026

Everyone talks about the weight loss. The dramatic before-and-after photos. The dropping numbers on the scale. And yes, tirzepatide delivers remarkable weight loss results. But researchers who stop there are missing the bigger picture, and it is a much bigger picture than most people realize.
Tirzepatide does not simply suppress appetite and call it a day. This dual GIP/GLP-1 receptor agonist triggers a cascade of biological changes that extend far beyond body composition. We are talking about cardiovascular protection confirmed in landmark clinical trials. Liver fat reduction so significant that researchers achieved MASH resolution in over half of participants. Anti-inflammatory effects that reach nearly every organ system in the body. Sleep apnea improvement so profound that some participants no longer met the diagnostic criteria for the condition.
The science here is not speculative. These are published, peer-reviewed findings from trials like SURPASS-CVOT, SURMOUNT-OSA, SYNERGY-NASH, and SUMMIT. Tens of thousands of participants. Years of follow-up data. Hard clinical endpoints that matter.
This guide covers every proven and emerging benefit of tirzepatide that has nothing to do with stepping on a scale. From your heart to your kidneys, from your liver to your brain, from your joints to your hormones, the evidence keeps expanding. SeekPeptides has analyzed the latest research to bring you the most comprehensive breakdown available. Whether you are already using tirzepatide for weight management or considering it for other reasons entirely, understanding these additional benefits changes the conversation about what this peptide can do.
How tirzepatide works beyond simple appetite suppression
Most conversations about tirzepatide begin and end with appetite. It makes you less hungry. You eat less. You lose weight. That explanation is accurate but wildly incomplete.
Tirzepatide is a dual agonist. It activates two distinct receptor systems simultaneously: the GLP-1 receptor and the GIP receptor. This dual mechanism separates it from semaglutide and other single-receptor GLP-1 agonists. And those two receptor systems do not just exist in your gut and brain. They appear throughout your body, in your heart, your liver, your kidneys, your blood vessels, your adipose tissue, and even your immune cells.
When tirzepatide binds to GLP-1 receptors outside the brain, it triggers effects that have nothing to do with hunger. Blood vessels relax. Inflammatory signaling decreases. Insulin sensitivity improves in tissues that were previously resistant. The GIP receptor activation adds another layer entirely, influencing lipid metabolism, bone health, and potentially even cognitive function through pathways researchers are still mapping.
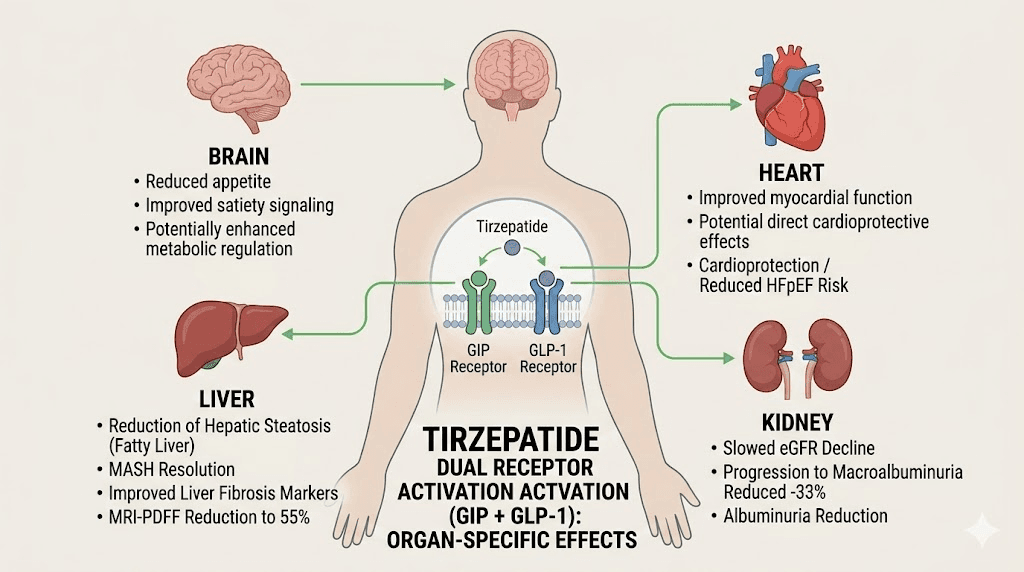
Think of it this way. A medication that only reduced appetite would produce weight loss and nothing else. But tirzepatide consistently improves biomarkers that weight loss alone cannot fully explain. The speed at which some benefits appear suggests direct pharmacological effects rather than secondary consequences of losing body mass. Blood pressure improvements emerge before significant weight is lost. Inflammatory markers drop faster than body composition changes. Liver fat decreases out of proportion to total weight reduction.
This matters for anyone evaluating tirzepatide purely through the lens of body weight. The scale captures maybe 30% of what this molecule does. The other 70% happens quietly, in organ systems you cannot see, measured by lab values most people never check.
Cardiovascular protection: the benefit that saves lives
Heart disease kills more people than any other condition on the planet. And tirzepatide, originally developed for diabetes and weight management, turns out to be a powerful cardiovascular protector.
SURPASS-CVOT: head-to-head cardiovascular data
The SURPASS-CVOT trial compared tirzepatide directly against dulaglutide in patients with type 2 diabetes and established cardiovascular disease. This was not a placebo comparison. Both drugs are active treatments. Over a median follow-up of 4 years, the results told a clear story.
The primary endpoint, a composite of cardiovascular death, heart attack, and stroke, occurred in 12.2% of tirzepatide patients versus 13.1% of dulaglutide patients. That met noninferiority. But the expanded MACE endpoint, which added coronary revascularization, showed a statistically significant 12% reduction with tirzepatide (HR 0.88). All-cause mortality was also lower in the tirzepatide group, driven by a reduction in non-cardiovascular death.
These are not trivial numbers. In a trial comparing two active cardiovascular medications, tirzepatide still demonstrated additional benefit. For researchers tracking their tirzepatide dosing protocols, knowing that every injection potentially protects the cardiovascular system adds significant context to the decision.
Heart failure with preserved ejection fraction
The SUMMIT trial examined tirzepatide in patients with heart failure with preserved ejection fraction (HFpEF) and obesity. This is a condition with very few effective treatments. The results were striking.
At one year, patients taking tirzepatide had a 38% lower rate of cardiovascular death or worsening heart failure compared to placebo. That is not a modest improvement. That is a paradigm shift for a condition that cardiologists have struggled to treat for decades.
How does it work? By producing sustained weight reduction and favorable changes in lipids, blood pressure, systemic inflammation, and endothelial biology, tirzepatide targets the central pathophysiological drivers of obesity-related HFpEF. It does not just mask symptoms. It addresses root causes. For anyone managing fatigue related to GLP-1 therapy, understanding that the medication actively protects heart function provides important perspective on the benefit-to-side-effect ratio.
Blood pressure reduction without additional medication
Blood pressure improvements with tirzepatide are dose-dependent and clinically meaningful. A meta-analysis of randomized controlled trials found systolic blood pressure reductions of 4.20 mmHg at 5 mg, 5.34 mmHg at 10 mg, and 5.77 mmHg at 15 mg. These numbers matter because every 5 mmHg reduction in systolic blood pressure correlates with approximately a 10% reduction in major cardiovascular events.
For some patients, this blood pressure reduction eliminates the need for an additional antihypertensive medication. For others, it brings readings from borderline-high into the normal range. Either way, it represents a cardiovascular benefit that occurs alongside weight management, not instead of it.
The supplements you take alongside tirzepatide can further support cardiovascular health, but the peptide itself does significant heavy lifting in this department without any additional interventions.
Liver protection and MASH resolution: reversing fatty liver disease
Metabolic dysfunction-associated steatohepatitis (MASH), formerly known as NASH, affects an estimated 5-6% of adults globally. It is the leading cause of liver transplant waitlist additions. And until recently, treatment options were extremely limited. Tirzepatide is changing that equation dramatically.
SYNERGY-NASH trial: the numbers that stunned hepatologists
The SYNERGY-NASH trial tested tirzepatide against placebo in patients with MASH. The results exceeded expectations across the board.
MASH resolution without worsening fibrosis occurred in 44-62% of tirzepatide-treated patients depending on dose. In the placebo group? Just 10%. That is a four-to-six-fold improvement. More than half of patients (51-55%) achieved at least one stage of fibrosis improvement compared to 30% on placebo. Fibrosis improvement matters enormously because fibrosis progression is what ultimately leads to cirrhosis, liver failure, and transplant.
These findings position tirzepatide as potentially one of the most effective MASH therapies ever studied. And unlike dedicated liver drugs, it simultaneously addresses the metabolic dysfunction driving the liver disease in the first place.
How tirzepatide reduces liver fat
The mechanism involves multiple pathways working simultaneously. Tirzepatide reduces liver fat accumulation through improved insulin sensitivity, decreased de novo lipogenesis (the liver making new fat), and enhanced fatty acid oxidation. Recent mechanistic research published in 2026 shows that tirzepatide improves hepatic steatosis, inflammation, oxidative stress, and fibrosis through the AMPK/NF-kB signaling pathway.
It also modulates lipid metabolism in extrahepatic tissues, reducing the overflow of fatty acids that typically end up stored in the liver. Think of it as both reducing the supply of fat reaching the liver and enhancing the liver ability to process and export fat it already contains.
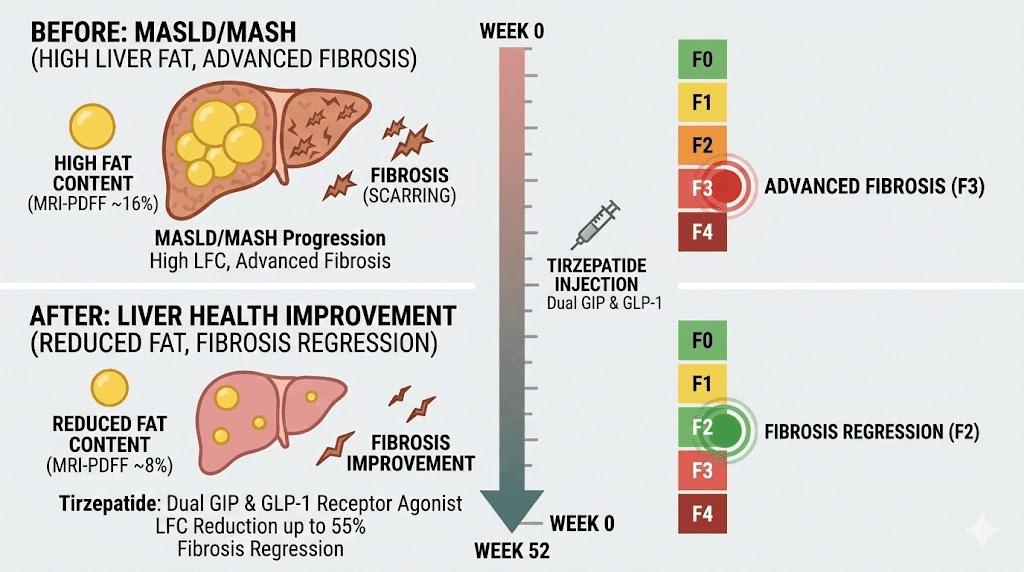
For researchers using tirzepatide combined with B12 or other compounds, the liver protection benefit adds another compelling reason to explore this peptide. And for anyone with elevated liver enzymes, fatty liver on imaging, or a diagnosis of MASH, the data from SYNERGY-NASH represents genuine hope.
Sleep apnea improvement: breathing better, sleeping deeper
Obstructive sleep apnea affects roughly 1 billion people worldwide. It causes fragmented sleep, drops blood oxygen levels, increases cardiovascular risk, and destroys quality of life. The standard treatment, continuous positive airway pressure (CPAP), works but adherence rates remain frustratingly low. Many people simply cannot tolerate sleeping with a mask strapped to their face every night.
Tirzepatide offers a fundamentally different approach. And the data from the SURMOUNT-OSA trial is remarkable.
SURMOUNT-OSA results: 63% reduction in breathing events
The trial enrolled participants with moderate-to-severe obstructive sleep apnea and obesity. It consisted of two studies, one with participants not using CPAP therapy and one with participants already on CPAP.
The numbers speak for themselves. In Study 1 (no CPAP), tirzepatide reduced the apnea-hypopnea index by 25.3 events per hour compared to just 5.3 for placebo. That is a treatment difference of 20 fewer breathing interruptions per hour of sleep. Study 2 (with CPAP) showed even larger improvements, with a treatment difference of 23.8 events per hour.
Overall, tirzepatide demonstrated a mean AHI reduction of up to 62.8%. That translates to approximately 30 fewer sleep interruptions per hour. For someone experiencing 45 events per hour (severe sleep apnea), dropping to 15 events per hour reclassifies their condition from severe to mild.
Disease resolution: some participants no longer had sleep apnea
Perhaps the most striking finding was the disease resolution rate. In Study 1, 43% of tirzepatide-treated participants met criteria for disease resolution. In Study 2, that number climbed to 51.5%. Disease resolution meant achieving an AHI below 5 events per hour (essentially normal) or an AHI of 5-14 with minimal daytime sleepiness.
Half of the participants in one study no longer qualified as having sleep apnea. That is not improvement. That is resolution.
Beyond the breathing events themselves, tirzepatide reduced hypoxic burden (time spent with low oxygen), lowered hsCRP (an inflammatory marker), decreased systolic blood pressure, and improved patient-reported sleep quality outcomes. Participants reported sleeping better, feeling more rested, and experiencing less daytime drowsiness.
For researchers tracking tirzepatide fatigue, it is worth noting that some initial tiredness during titration may be temporary, while long-term sleep quality often improves substantially once sleep apnea resolves. The relationship between tirzepatide and sleep is more complex than simple sedation or stimulation.
Cholesterol and lipid improvements: rewriting your lipid panel
High cholesterol and dyslipidemia contribute massively to cardiovascular disease risk. Statins have been the primary treatment for decades, but they do not address all lipid abnormalities equally. Tirzepatide fills important gaps.
Comprehensive lipid panel improvements
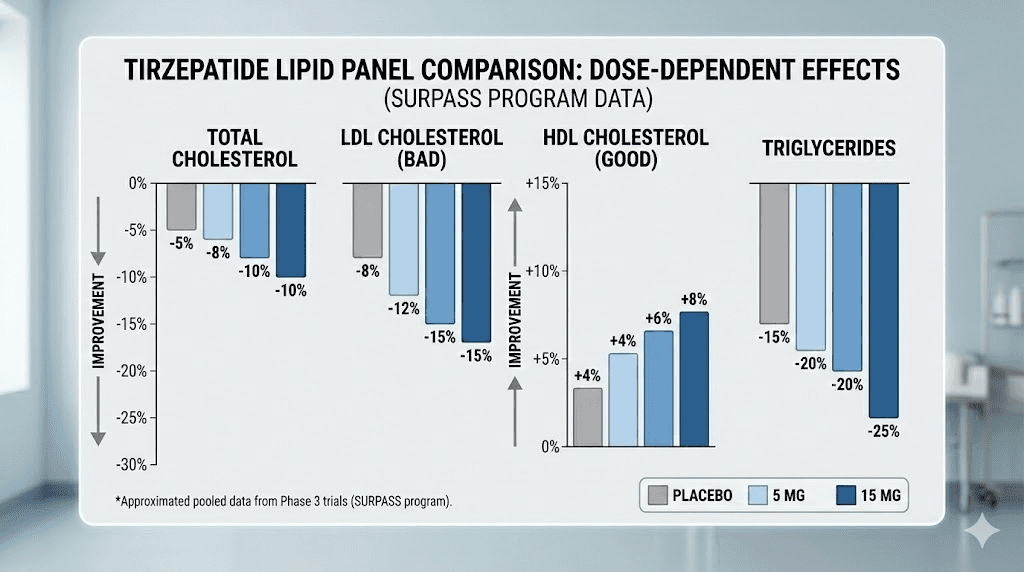
A systematic review and meta-analysis of randomized controlled trials found that tirzepatide improves every major dimension of the lipid profile in a dose-dependent manner.
Total cholesterol decreased by 3.76% at 5 mg, 4.63% at 10 mg, and 5.93% at 15 mg. But the really impressive numbers came from the SURPASS-5 trial, where tirzepatide reduced total cholesterol by 12.9%, LDL cholesterol by 15.5%, and triglycerides by a substantial 24.9%, while increasing HDL cholesterol by 0.9%.
A 24.9% triglyceride reduction is particularly noteworthy. Elevated triglycerides are notoriously difficult to treat with lifestyle changes alone, and they represent an independent risk factor for cardiovascular events. Many patients with metabolic syndrome have normal LDL but severely elevated triglycerides. Tirzepatide addresses this directly.
What this means practically
For someone with borderline-high LDL who does not want to start a statin, a 15% LDL reduction from tirzepatide might bring levels into the acceptable range. For someone already on a statin with persistently elevated triglycerides, tirzepatide provides additional improvement that the statin cannot deliver.
The lipid improvements also explain part of the cardiovascular protection observed in the SURPASS-CVOT trial. Better lipid profiles mean less plaque formation, less arterial inflammation, and lower risk of the cardiovascular events that kill millions each year. Anyone comparing semaglutide versus tirzepatide should note that these lipid improvements are consistently observed across tirzepatide trials at a magnitude that clinical practice cannot ignore.
Kidney protection: preserving renal function
Chronic kidney disease affects approximately 850 million people worldwide. It progresses silently, often detected only when significant damage has already occurred. And the kidneys, once damaged, do not regenerate easily. Prevention and slowing progression are everything.
Tirzepatide shows remarkable kidney-protective effects across multiple patient populations.
Slowing kidney decline in the SURPASS-CVOT trial
In patients with high or very high risk chronic kidney disease enrolled in SURPASS-CVOT, tirzepatide dramatically slowed the rate of kidney function decline compared to dulaglutide. Over 36 months, estimated glomerular filtration rate (eGFR) declined by just 3.0 mL/min per 1.73m2 with tirzepatide versus 7.2 mL/min per 1.73m2 with dulaglutide. That is a difference of 4.1 mL/min per 1.73m2, which translates to years of preserved kidney function over a patient lifetime.
The composite kidney outcome (a broad measure of major kidney events) was 33% lower with tirzepatide compared to dulaglutide: 16.7% versus 23.0% (HR 0.67). One-third fewer major kidney events. That is not a subtle finding.
Albuminuria reduction: the early warning system
Albuminuria, the presence of protein in urine, serves as both a marker and a driver of kidney disease progression. Pooled analysis from the SURPASS-1-5 trials showed that tirzepatide reduced urinary albumin-to-creatinine ratio (UACR) by 19-26% depending on dose, compared to all pooled comparators.
Mediation analysis suggested that approximately half of this albuminuria reduction is related to weight loss, but the other half appears to be a direct pharmacological effect. Tirzepatide likely reduces intraglomerular pressure through hemodynamic effects, which decreases the filtration of albumin into the urine.
For researchers managing their tirzepatide protocols long-term, requesting periodic kidney function tests (eGFR and UACR) makes sense. These are inexpensive blood and urine tests that can capture kidney protection benefits that would otherwise go unnoticed. SeekPeptides members access comprehensive monitoring guides that include kidney biomarkers alongside the standard weight and metabolic tracking most people focus on.
Joint pain and mobility improvements
Excess body weight places enormous mechanical stress on joints. Every pound of body weight translates to roughly four pounds of pressure on the knees during walking. Lose 30 pounds and your knees experience 120 fewer pounds of force with every step. The math alone explains why weight loss helps joint pain.
But tirzepatide appears to offer joint benefits beyond what simple weight reduction would predict.
Reduced joint pain and medication use
Large-scale analysis has found that patients using tirzepatide are significantly less likely to develop osteoarthritis than those on other weight-loss medications. Tirzepatide is associated with lower risk of joint pain across multiple sites, including low back pain, hip pain, and knee pain, with particularly strong benefits for knee pain (HR 0.84).
Perhaps even more telling, tirzepatide users had lower rates of NSAID and opioid prescriptions compared to users of other weight loss medications. When people need fewer pain medications, that signals genuine functional improvement rather than just numerical change on a pain scale.
For anyone who has experienced joint pain while on tirzepatide, it is worth distinguishing between transient side effects during titration and the long-term joint benefits that accumulate over months of treatment. Initial body aches sometimes occur but tend to resolve, while the reduction in inflammatory markers and mechanical joint stress builds progressively.
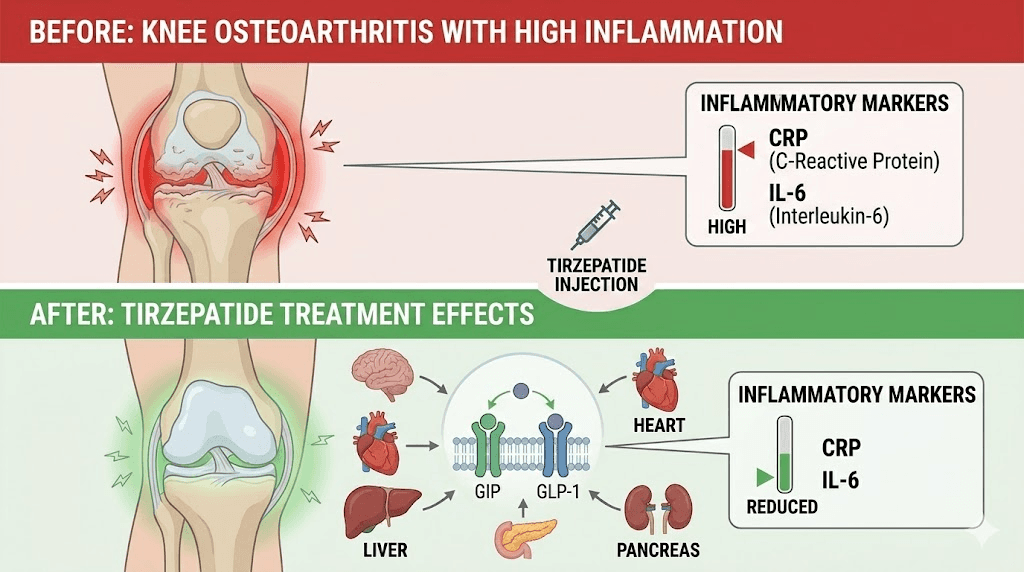
Anti-inflammatory mechanisms that protect cartilage
A systematic review and meta-analysis confirmed that tirzepatide significantly reduces inflammatory markers regardless of population studied or treatment regimen. Specifically, tirzepatide reduces high-sensitivity C-reactive protein (hsCRP) and interleukin-6 (IL-6), both of which play central roles in cartilage degradation and osteoarthritis progression.
GLP-1 based therapies may improve osteoarthritis symptoms both indirectly through weight loss and directly through anti-inflammatory mechanisms. By improving insulin sensitivity and reducing systemic inflammation, tirzepatide creates a healthier environment for tissue repair and movement. This dual mechanism, mechanical unloading plus biochemical inflammation reduction, explains why the joint benefits appear larger than weight loss alone would predict.
Recent cost-effectiveness analysis from Mass General Brigham found tirzepatide more cost-effective than semaglutide and other weight loss interventions specifically for patients with knee osteoarthritis and obesity. The researchers concluded that tirzepatide provided greater health benefits at lower costs, yielding an incremental cost-effectiveness ratio of $57,400 per quality-adjusted life year versus diet and exercise alone.
PCOS and reproductive health benefits
Polycystic ovary syndrome (PCOS) affects 8-13% of women of reproductive age. It involves insulin resistance, hormonal imbalances, irregular periods, ovarian cysts, and often significant difficulty with weight management. The metabolic dysfunction at the core of PCOS overlaps substantially with what tirzepatide treats. That overlap has produced encouraging results.
Insulin resistance improvement
Insulin resistance is both a driver and a consequence of PCOS. It fuels excess androgen production, disrupts ovulation, and makes weight loss exceptionally difficult. In clinical studies of women with PCOS on tirzepatide, insulin resistance improved from 80.4% prevalence to 50%. That is a 30-percentage-point reduction in insulin resistance prevalence.
Glycemic control improved significantly alongside. Fasting blood sugar dropped from 6.89 mg/dL to 5.57 mg/dL. HbA1c decreased from 5.7% to 4.9%. These improvements in glucose metabolism directly reduce the hyperinsulinemia that drives many PCOS symptoms.
Menstrual regularity and ovarian changes
The hormonal cascade triggered by improved insulin sensitivity produces tangible results for PCOS symptoms. In published data, irregular menstrual cycles decreased from 85.7% to 32.1% among women taking tirzepatide. Ovarian cyst prevalence dropped from 89.3% to 41%. These are not subtle improvements. They represent a fundamental shift in disease activity.
Weight loss plays a role here, as even a 5-10% reduction in body weight can restore ovulatory cycles in many women with PCOS. But the insulin sensitization effects of tirzepatide likely contribute independently, since improvements in hormonal markers sometimes precede significant weight change.
For women considering tirzepatide specifically for PCOS, the effects of tirzepatide on menstrual cycles deserve careful attention. Periods may become more regular, which for women with PCOS is actually a positive sign indicating restored ovulatory function. The growing adoption reflects this benefit, as prescribing of semaglutide or tirzepatide among women with PCOS increased more than seven-fold from 2.4% in 2021 to 17.6% in 2025.
Fertility considerations
Here is where caution becomes critical. Studies suggest GLP-1 agonists can enhance ovulation rates and fertility outcomes in women with PCOS by regulating hormonal imbalances. But this improved fertility means unintended pregnancy becomes possible for women who previously could not conceive due to anovulation.
Experts recommend a washout period of two to three months after stopping tirzepatide before actively trying to conceive. This is not because tirzepatide is proven harmful to pregnancy, but because sufficient safety data in pregnant women does not yet exist. The experiences of women who became pregnant while on tirzepatide underscore the importance of adequate contraception during treatment if pregnancy is not desired, and proper planning if it is.
Women who are breastfeeding while considering tirzepatide or GLP-1 medications in general should consult their healthcare provider, as current data on safety during lactation remains limited.
Mental health and mood regulation
The relationship between obesity, metabolic dysfunction, and mental health runs deeper than most people appreciate. Chronic inflammation, which tirzepatide reduces, is increasingly linked to depression and anxiety. Insulin resistance affects neurotransmitter function. Poor sleep from sleep apnea impairs mood regulation. Tirzepatide potentially addresses all three of these pathways simultaneously.
Depression and anxiety outcomes
Recent research suggests that people who started GLP-1 medications like tirzepatide were less likely to develop depression compared to those not on a GLP-1 medication. Post-hoc analysis of the SURMOUNT trial data found that tirzepatide relative to placebo was not associated with increased risk of depressive symptoms. In fact, there were no excess risks for any neurological or neuropsychiatric adverse effects.
The GIP receptor pathway may offer additional cognitive and emotional benefits that pure GLP-1 agonists do not provide. GIP signaling has been linked to improved cognitive processes in animal models, suggesting tirzepatide dual mechanism could provide broader neuropsychiatric benefits than semaglutide alone.
For anyone experiencing anxiety on tirzepatide, it is important to distinguish between initial adjustment effects and longer-term mood changes. Some individuals report transient anxiety during dose titration that resolves as the body adapts. The broader trend in research points toward mental health improvement, not deterioration.
The inflammation-mood connection
Chronic inflammation has been increasingly linked to mental health conditions including depression. hsCRP, a marker of systemic inflammation that tirzepatide consistently reduces, correlates with depressive symptom severity in multiple studies. IL-6, another inflammatory marker reduced by tirzepatide, crosses the blood-brain barrier and directly affects neurotransmitter production.
Researchers hypothesize that GLP-1 medications decrease inflammation throughout the body at a cellular level, and this anti-inflammatory effect in the brain contributes to mood stabilization. This is not merely a consequence of feeling better about weight loss. It appears to be a direct pharmacological effect on neuroinflammation. Resources on peptides for depression and anxiety and peptides for brain repair provide additional context on how various peptides interact with neurological function.
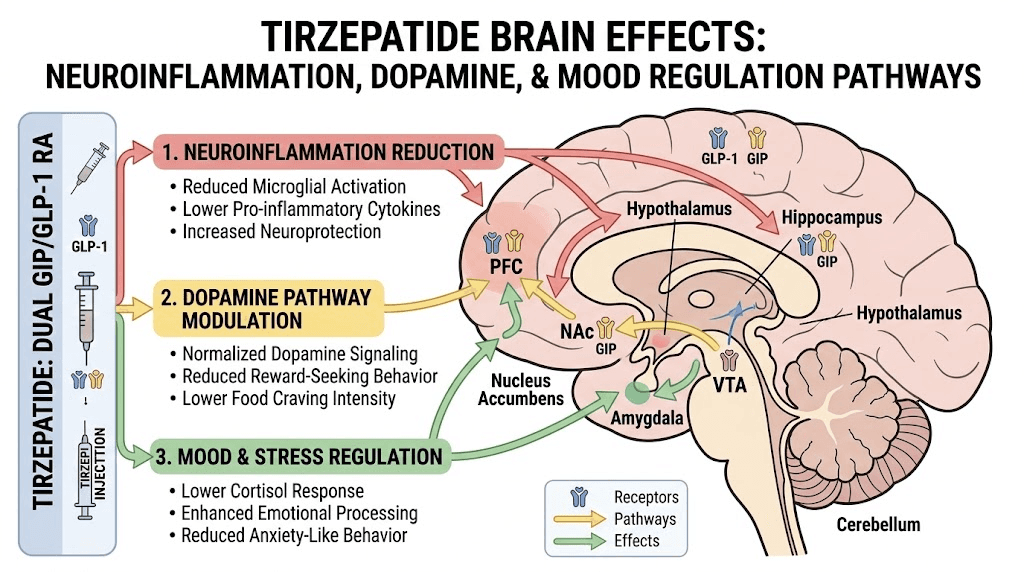
Addiction and compulsive behavior modulation
This might be the most unexpected benefit of tirzepatide. And the most fascinating.
Emerging research shows that tirzepatide affects the dopamine reward pathway in ways that could reduce addictive behaviors. This is not peripheral speculation. It involves direct modulation of the mesolimbic reward system, the same neural circuit that drives addiction to alcohol, nicotine, opioids, and other substances.
Alcohol consumption reduction in preclinical studies
Rodent studies have demonstrated significant and consistent effects. Tirzepatide effectively attenuated the rewarding properties of alcohol as measured through locomotor stimulation, conditioned place preference, and accumbal dopamine release. It dose-dependently reduced voluntary alcohol consumption, prevented binge drinking and relapse-like behavior, and maintained efficacy with repeated administration.
The mechanism is direct and measurable. When researchers gave mice tirzepatide before alcohol exposure, the typical dopamine surge was mostly blocked. Without the chemical reward, the drive to consume alcohol diminished. The brain simply found alcohol less rewarding.
Broader implications for compulsive behaviors
Tirzepatide binds to receptors in the ventral tegmental area, nucleus accumbens, and prefrontal cortex, all brain regions central to reward processing and impulse control. By blunting dopamine release and reducing reward signaling in these areas, tirzepatide could theoretically reduce cravings for various substances and even non-substance compulsive behaviors.
The research suggests applications beyond alcohol. GLP-1 receptor agonists affect multiple regions of the reward pathway that mediate addiction to foods and various other substances including alcohol, opioids, nicotine, and cocaine. Some researchers speculate about potential benefits for compulsive shopping, gambling, and other behavioral addictions, though human data for these applications remains very limited.
It is essential to note that tirzepatide has no FDA-approved indication for treating substance use disorders. The National Institute on Alcohol Abuse and Alcoholism (NIAAA) is currently funding studies to systematically evaluate whether incretin-based medications can effectively reduce substance use behaviors in humans. But the preclinical evidence is striking enough that major research institutions are investing significant resources to investigate further.
Many users of tirzepatide who drink alcohol have anecdotally reported decreased interest in drinking. Whether this reflects the reward pathway modulation seen in animal studies or simply reduced overall appetite (including appetite for alcohol) remains an active area of investigation.
Blood sugar control that extends beyond diabetes management
Tirzepatide received its initial approval for type 2 diabetes management, and its glucose-lowering effects remain among its most potent actions. But even people without diabetes benefit from improved glycemic control, and the degree of improvement tirzepatide provides deserves specific attention.
HbA1c reductions that rival insulin
Across the SURPASS trial program, tirzepatide reduced HbA1c by up to 2.24%, bringing many participants to levels below the diabetic threshold. For context, most oral diabetes medications reduce HbA1c by 0.5-1.5%. Insulin itself typically achieves 1.0-2.0% reduction. Tirzepatide matches or exceeds insulin effectiveness without the injection burden, hypoglycemia risk, and weight gain associated with insulin therapy.
For people with prediabetes or insulin resistance who do not yet meet diagnostic criteria for type 2 diabetes, tirzepatide can normalize glucose metabolism before the disease progresses. This preventive effect may ultimately prove more valuable than the therapeutic one, since preventing diabetes avoids decades of associated complications.
Insulin sensitivity improvement across tissues
Tirzepatide improves insulin sensitivity in muscle, liver, and adipose tissue simultaneously. This is unusual. Most diabetes medications primarily target one tissue type. Metformin mainly reduces hepatic glucose production. Thiazolidinediones mainly improve adipose tissue insulin sensitivity. Tirzepatide dual receptor mechanism allows it to affect all three simultaneously.
The practical result is more stable blood sugar throughout the day, fewer post-meal glucose spikes, reduced fasting insulin levels, and lower overall metabolic stress on the pancreas. For researchers using the compounded tirzepatide dosage calculator, these metabolic benefits begin at the lowest effective dose and generally increase with appropriate dose titration.
Anyone interested in the proper starting dose for compounded tirzepatide should understand that blood sugar improvements often appear among the earliest benefits, sometimes within the first week of treatment, well before significant weight loss occurs. This early metabolic improvement helps explain why the first week on GLP-1 medications can feel different even before the scale moves.
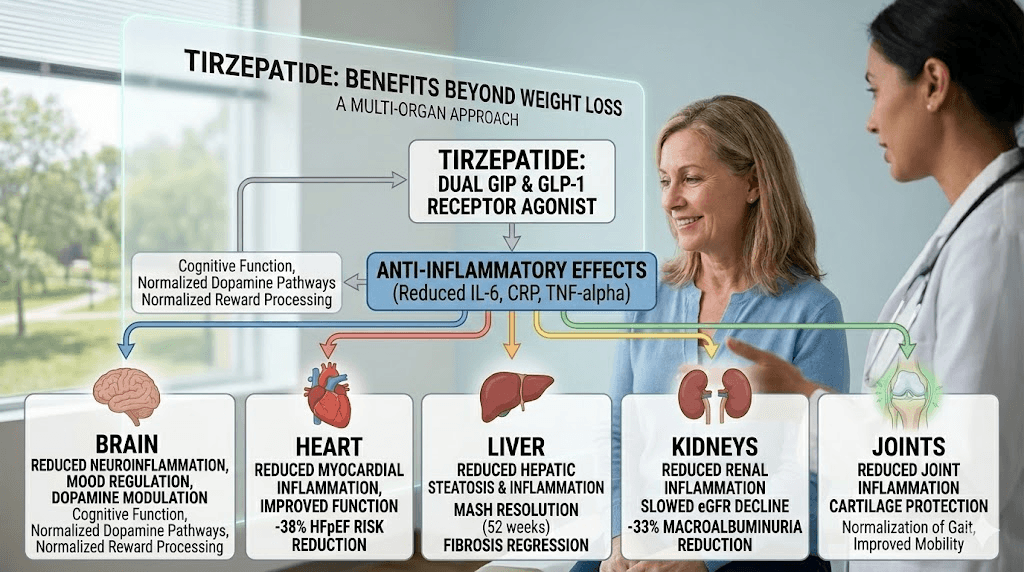
Systemic inflammation reduction: the benefit that amplifies everything else
If there is one thread connecting all of tirzepatide non-weight-loss benefits, it is inflammation reduction. Chronic low-grade inflammation underlies cardiovascular disease, fatty liver disease, insulin resistance, joint degeneration, cognitive decline, and cancer risk. Tirzepatide addresses this inflammatory burden directly and profoundly.
What the inflammatory markers show
A systematic review and meta-analysis confirmed that tirzepatide use is associated with significant reductions in inflammatory markers regardless of population studied or treatment regimen. High-sensitivity C-reactive protein (hsCRP) and interleukin-6 (IL-6) both decreased significantly compared to placebo.
hsCRP is a general marker of systemic inflammation. IL-6 is a pro-inflammatory cytokine that drives tissue damage in multiple organ systems. Reducing both simultaneously suggests that tirzepatide addresses inflammation at a fundamental level rather than simply masking one downstream marker.
The complete guide to inflammation peptides provides broader context on how various compounds modulate inflammatory pathways. Tirzepatide stands out because its anti-inflammatory effects are not its primary mechanism, they are a beneficial secondary effect that amplifies its primary actions across every organ system it touches.
How reduced inflammation amplifies other benefits
Consider how inflammation reduction connects to every other benefit discussed in this guide.
Cardiovascular protection: arterial inflammation drives plaque formation and rupture. Reduce inflammation, reduce heart attack risk. Liver health: hepatic inflammation drives fibrosis progression. Reduce inflammation, slow fibrosis. Joint health: synovial inflammation degrades cartilage. Reduce inflammation, preserve joint function. Kidney protection: renal inflammation accelerates nephron loss. Reduce inflammation, preserve kidney function. Mental health: neuroinflammation disrupts neurotransmitter signaling. Reduce inflammation, stabilize mood.
This interconnection means that the inflammatory benefit is not one benefit among many. It is the foundational benefit that makes many of the others possible. Researchers exploring microdosing tirzepatide specifically for inflammation or investigating GLP-1 microdosing for autoimmune conditions are tapping into this very principle.
How to maximize the non-weight-loss benefits of tirzepatide
Knowing that tirzepatide offers a dozen benefits beyond weight loss is valuable. Knowing how to maximize those benefits is actionable. Here is what the evidence suggests for getting the most out of every injection.
Dosing strategy matters
Many of the non-weight-loss benefits show dose-dependent improvements. Blood pressure reduction increases from 4.2 mmHg at 5 mg to 5.8 mmHg at 15 mg. Lipid improvements scale with dose. Anti-inflammatory effects strengthen at higher doses. This does not mean everyone should rush to the highest dose, because side effect burden also increases. But it does mean that finding the optimal therapeutic dose, not just the minimum weight-loss dose, matters for maximizing total benefit.
Working with a proper tirzepatide dosage protocol and following established titration schedules helps balance efficacy against tolerability. Some researchers find that splitting the tirzepatide dose improves tolerability while maintaining benefits.
Nutrition that supports multi-organ benefits
Diet choices can either amplify or undermine tirzepatide non-weight-loss benefits. A thoughtful tirzepatide diet plan should include:
Omega-3 rich foods to further reduce inflammation and support cardiovascular health
Fiber to support gut health and blood sugar stability
Adequate protein to preserve lean mass and support tissue repair (consider protein shakes designed for GLP-1 users)
Potassium-rich foods to support blood pressure benefits
Liver-friendly nutrients like cruciferous vegetables and adequate choline
Knowing which foods to avoid on tirzepatide is equally important. High-sodium processed foods can counteract blood pressure benefits. Excessive alcohol undermines liver protection. Refined carbohydrates blunt insulin sensitivity improvements.
A comprehensive tirzepatide meal plan should account for these multi-organ benefits rather than focusing exclusively on calorie restriction. Detailed guidance on what to eat on tirzepatide and the best foods for GLP-1 users can help optimize outcomes across all the benefit categories discussed in this guide.
Supplement support for enhanced benefits
Certain supplements can complement tirzepatide multi-system effects. Key supplements to take with tirzepatide include B vitamins for methylation support and energy, magnesium for cardiovascular and metabolic health, and vitamin D for immune modulation and bone health.
Compounded formulations like tirzepatide with B12, tirzepatide with glycine, and tirzepatide with glycine and B12 address specific nutritional needs that arise during GLP-1 therapy. Tirzepatide with niacinamide and tirzepatide with levocarnitine offer additional metabolic support through different mechanisms.
Monitoring beyond body weight
If you only track your weight, you are missing most of the story. To capture the full range of tirzepatide benefits, consider monitoring:
Blood pressure at home (weekly minimum)
Lipid panel every 3-6 months
Liver enzymes (ALT, AST) every 3-6 months
HbA1c and fasting glucose every 3 months
Kidney function (eGFR, UACR) annually or as directed
hsCRP as an inflammatory marker every 3-6 months
Sleep quality subjectively and, if applicable, with a sleep study
Joint pain and mobility using standardized assessments
SeekPeptides members get access to comprehensive monitoring protocols, tracking tools, and guidance on interpreting lab results in the context of peptide therapy. Understanding what your bloodwork means, and how it changes over time, transforms tirzepatide from a weight loss injection into a comprehensive health optimization tool.
Long-term consistency wins
Many of tirzepatide non-weight-loss benefits are cumulative. Cardiovascular protection builds over years. Kidney preservation compounds month after month. Liver improvement requires sustained treatment. The question of how long to stay on GLP-1 therapy takes on new dimensions when you factor in organ protection beyond weight maintenance.
Understanding how to maintain benefits after stopping tirzepatide and how to wean off properly matters because some benefits may persist after discontinuation (improved metabolic health, resolved sleep apnea) while others may diminish (direct anti-inflammatory effects, lipid improvements). Planning accordingly helps preserve the gains you have worked to achieve.
How tirzepatide compares to other GLP-1 medications for non-weight-loss benefits
Not all GLP-1 receptor agonists are created equal when it comes to benefits beyond the scale. Tirzepatide dual GIP/GLP-1 mechanism gives it advantages in several areas.
A post hoc analysis of the SURMOUNT-5 study found that tirzepatide was associated with a greater predicted 10-year cardiovascular disease risk reduction compared to semaglutide. Real-world cardiovascular outcome data published in Nature Medicine showed that treatment with tirzepatide lowered the risk of stroke, heart attack, and death by 13% compared to dulaglutide. The side effect profiles differ between semaglutide and tirzepatide, and these differences extend to the benefit profiles as well.
The GIP receptor component appears to contribute meaningfully to lipid metabolism improvements, insulin sensitization in adipose tissue, and potentially cognitive and bone health effects that pure GLP-1 agonists do not provide. For a detailed comparison across multiple dimensions, the semaglutide versus tirzepatide versus retatrutide comparison offers comprehensive analysis. Those considering switching from tirzepatide to semaglutide should weigh these non-weight-loss differences alongside cost and availability factors.
Newer compounds in development, like survodutide, cagrisema, and orforglipron, target similar or additional receptor pathways. Retatrutide, a triple agonist, adds glucagon receptor activation to the GLP-1 and GIP activation. Whether these next-generation compounds will offer even broader non-weight-loss benefits remains an active area of investigation.
Practical protocols for specific non-weight-loss goals
Different benefits may require different approaches. Here is how to think about optimizing for specific non-weight-loss outcomes.
For cardiovascular protection
Cardiovascular benefits appear most robustly at moderate to higher doses maintained consistently over time. Combine tirzepatide with a heart-healthy diet rich in omega-3 fatty acids, regular moderate exercise, and monitoring of blood pressure and lipids. The cardiovascular protection data comes primarily from studies lasting 1-4 years, suggesting this is a long-term strategy rather than a short-term intervention.
For liver health
Liver fat reduction begins relatively early in treatment, but MASH resolution and fibrosis improvement require sustained therapy. In the SYNERGY-NASH trial, the assessment point was 52 weeks. Avoid alcohol consumption during treatment to maximize liver benefits. Request liver enzyme testing and, if available, fibroscan imaging to track improvement objectively. For those managing gastrointestinal side effects like constipation, addressing these issues helps maintain consistent dosing, which in turn supports sustained liver improvement.
For sleep apnea
The SURMOUNT-OSA trial showed benefits at 52 weeks. Continue CPAP therapy during tirzepatide treatment until a repeat sleep study confirms sufficient improvement. Weight loss of even 10-15% can dramatically reduce sleep apnea severity. Some participants may eventually discontinue CPAP, but this decision should always be guided by objective sleep testing rather than subjective improvement alone.
For PCOS and reproductive health
Start at the lowest effective dose and titrate slowly, as hormonal changes can be significant. Monitor menstrual regularity as an indicator of ovulatory function restoration. Use reliable contraception if pregnancy is not desired, because restored fertility can occur before weight loss goals are met. If pregnancy is planned, discuss a washout timeline with your healthcare provider. The effects on menstrual cycles can begin within the first few months of treatment.
For inflammation reduction
Some researchers specifically explore microdosing tirzepatide for anti-inflammatory benefits at doses lower than those used for weight loss. A structured microdose schedule may provide meaningful inflammation reduction with minimal side effects. Track hsCRP and other inflammatory markers to objectively measure response. Dedicated microdosing protocols for inflammation are an emerging area of interest, particularly for individuals with autoimmune conditions or chronic inflammatory diseases.
What the future holds: ongoing and upcoming research
The non-weight-loss benefits of tirzepatide are still being actively investigated. The SURMOUNT-MMO trial is examining whether tirzepatide reduces overall morbidity and mortality in adults with obesity but without diabetes. This event-driven trial uses a five-component composite endpoint of heart attack, stroke, coronary revascularization, heart failure events, or death from any cause. If positive, it would establish tirzepatide as a life-extending medication, not merely a weight management tool.
Additional research is exploring tirzepatide potential benefits for Alzheimer disease, where insulin resistance and neuroinflammation play pathogenic roles. Trials examining kidney outcomes in dedicated renal populations are underway. And the addiction research, while still preclinical, has attracted funding from the National Institute on Alcohol Abuse and Alcoholism for human studies.
The trajectory is clear. With each new study, the list of tirzepatide benefits beyond weight loss grows longer. What started as a diabetes medication has revealed itself as a multi-organ therapeutic that addresses the interconnected metabolic dysfunction underlying dozens of modern chronic diseases.
For researchers serious about optimizing their peptide protocols and understanding the full spectrum of benefits, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, detailed monitoring tools, and a community of thousands who have navigated these exact questions. The peptide calculator and compounded tirzepatide dosage calculator provide precise dosing guidance, while the compound dosage charts and dosing charts in units offer quick visual references for daily use.
Frequently asked questions
What are the main benefits of tirzepatide besides weight loss?
Tirzepatide offers cardiovascular protection (38% reduction in heart failure events), liver fat reduction and MASH resolution (44-62% of patients), sleep apnea improvement (up to 63% AHI reduction), blood pressure reduction (up to 5.8 mmHg), cholesterol improvement (24.9% triglyceride reduction), kidney protection (33% fewer major kidney events), joint pain reduction, PCOS symptom improvement, and systemic inflammation reduction. These benefits stem from tirzepatide dual GIP/GLP-1 mechanism affecting receptors throughout the body.
Does tirzepatide protect the heart even without significant weight loss?
Some cardiovascular benefits appear to occur independently of weight loss. Blood pressure improvements and reductions in inflammatory markers like hsCRP often emerge before significant weight is lost. The SURPASS-CVOT trial showed cardiovascular benefit compared to another active GLP-1 treatment, suggesting direct pharmacological effects beyond what weight reduction alone would produce. However, sustained weight loss amplifies and maintains these cardiovascular benefits over time.
Can tirzepatide help with fatty liver disease?
Yes. The SYNERGY-NASH trial demonstrated that tirzepatide achieved MASH resolution in 44-62% of patients compared to 10% with placebo. More than half of patients achieved fibrosis improvement. Tirzepatide reduces liver fat through improved insulin sensitivity, decreased lipogenesis, and enhanced fatty acid oxidation. These effects work through the AMPK/NF-kB pathway and are partially but not entirely explained by weight loss.
How much does tirzepatide improve sleep apnea?
In the SURMOUNT-OSA trial, tirzepatide reduced the apnea-hypopnea index by up to 62.8%, translating to approximately 30 fewer breathing interruptions per hour of sleep. Between 43% and 51.5% of participants met criteria for disease resolution, meaning they essentially no longer had clinically significant sleep apnea. Benefits also included reduced hypoxic burden, lower blood pressure, and improved sleep quality.
Does tirzepatide help with PCOS?
Emerging evidence suggests significant benefits for PCOS. Studies show insulin resistance prevalence decreased from 80.4% to 50%, irregular menstrual cycles decreased from 85.7% to 32.1%, and ovarian cyst prevalence dropped from 89.3% to 41%. Weight loss and insulin sensitization both contribute to these improvements. However, tirzepatide is not FDA-approved for PCOS, and women should be aware that restored fertility is a possibility requiring appropriate contraception planning.
Can tirzepatide reduce alcohol cravings?
Preclinical research shows tirzepatide attenuates dopamine reward signaling and dose-dependently reduces voluntary alcohol consumption in rodent models. It blocks the typical dopamine surge associated with alcohol, reducing the rewarding properties of drinking. However, no human clinical trials have been completed yet, and tirzepatide has no FDA approval for substance use disorders. The NIAAA is currently funding studies to evaluate this potential application in humans.
How long does it take to see non-weight-loss benefits from tirzepatide?
Different benefits appear on different timelines. Blood sugar improvements often appear within the first one to two weeks. Blood pressure reductions emerge within weeks to months. Inflammatory marker improvements appear within months. Liver fat reduction requires 6-12 months for maximal effect. Cardiovascular protection builds over years of consistent use. Sleep apnea improvement depends on both direct effects and the degree of weight loss achieved.
Should I monitor anything beyond weight while on tirzepatide?
Absolutely. Consider regular monitoring of blood pressure, lipid panels (every 3-6 months), liver enzymes, HbA1c and fasting glucose, kidney function tests (eGFR and UACR), and hsCRP as an inflammatory marker. Sleep quality tracking is valuable for anyone with known or suspected sleep apnea. SeekPeptides provides comprehensive monitoring guides that help members track the full range of benefits beyond body weight.
External resources
SURPASS-CVOT: Cardiovascular Outcomes with Tirzepatide (New England Journal of Medicine)
SYNERGY-NASH Trial Results (American College of Gastroenterology)
SURMOUNT-OSA: Tirzepatide for Sleep Apnea and Obesity (PubMed Central)
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your inflammation stay low, and your benefits extend far beyond the scale.