Feb 8, 2026

You just picked up your tirzepatide. Maybe it is Mounjaro. Maybe Zepbound. Maybe a compounded vial from your pharmacy. And now you are staring at it wondering one thing.
Does this need to go in the fridge?
The short answer is yes. But the full answer involves temperature ranges, timing windows, formulation differences, and a handful of mistakes that quietly destroy potency before you ever notice. Tirzepatide is a peptide, which means it is a chain of amino acids folded into a precise three-dimensional structure. That structure is what makes it work. Heat unfolds it. Freezing shatters it. Even small temperature swings, repeated over days, chip away at its effectiveness in ways you cannot see, smell, or feel until your results start sliding backward. The medication looks exactly the same in the vial whether it is fully potent or halfway degraded, and that invisibility is what makes storage so critically important. This guide covers every angle of tirzepatide refrigeration, from FDA-approved brand products to compounded formulations, from daily home storage to traveling across time zones with your medication in tow. You will learn the exact temperatures, the precise time limits, and the specific mistakes that cost people real money and real results. SeekPeptides has compiled every storage detail into one resource so you never have to guess.
The short answer: yes, tirzepatide needs refrigeration
Tirzepatide must be stored in a refrigerator at 36 to 46 degrees Fahrenheit, which is 2 to 8 degrees Celsius. This applies to every form of the medication. Mounjaro pens. Zepbound pens. Multi-dose vials. Compounded tirzepatide from a pharmacy. The temperature range is the same across the board because the molecule itself demands it.
This is not a suggestion. It is a requirement straight from Eli Lilly, the manufacturer, and reinforced by the FDA prescribing information for both Mounjaro and Zepbound. The reason comes down to chemistry.
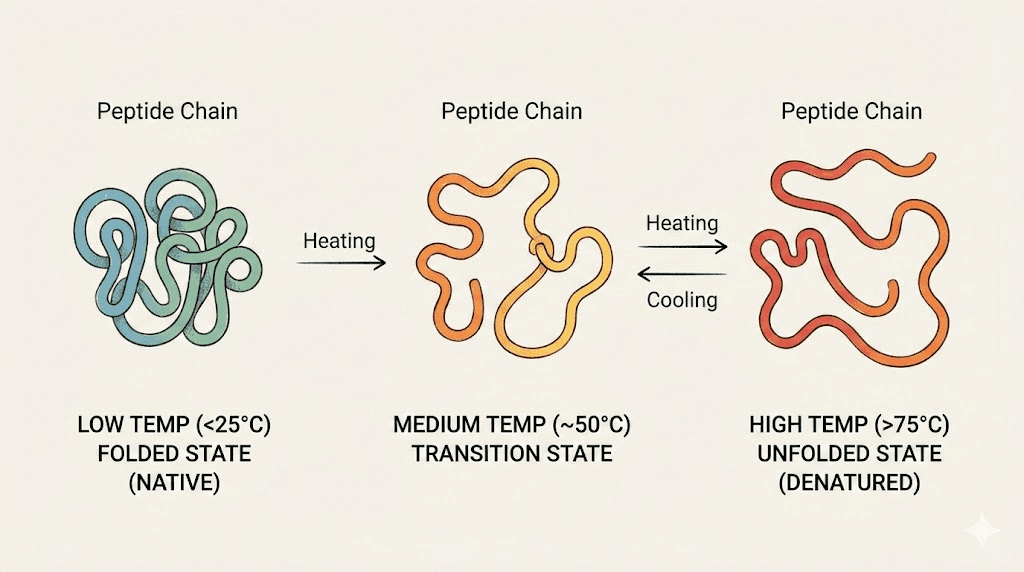
Tirzepatide is a 39-amino-acid peptide that acts as a dual GIP and GLP-1 receptor agonist. Its biological activity depends entirely on its three-dimensional folding pattern. When temperatures rise above 46 degrees Fahrenheit, the kinetic energy of the molecules increases. Bonds that hold the peptide in its active shape start to loosen. Chemical reactions that break the peptide chain, particularly hydrolysis, accelerate. The result is a medication that progressively loses its ability to bind receptors and produce the effects you need for weight management and metabolic health.
None of this is visible. The liquid stays clear. The color stays the same. You draw up the dose and inject it and everything feels normal. But the molecule has changed.
Why tirzepatide is temperature sensitive
Understanding why tirzepatide needs refrigeration helps you make better storage decisions. Not every medication requires cold storage. Tablets usually sit in a cabinet just fine. But peptide medications are fundamentally different from pills, and tirzepatide is no exception.
Peptide structure and folding
Every peptide has a specific amino acid sequence that determines how it folds. Tirzepatide has 39 amino acids connected in a precise order. The chain folds into a shape that fits into two different receptor types: the GIP receptor and the GLP-1 receptor. This dual action is what makes tirzepatide different from medications like semaglutide, which only activates the GLP-1 receptor.
Heat disrupts folding. Think of it like a key that warps in hot temperatures. A warped key might still fit the lock, partially. Or it might not fit at all. Either way, the mechanism does not work properly.
Hydrolysis: water breaks the chain
Tirzepatide sits in an aqueous solution. Water surrounds every molecule. At proper refrigeration temperatures, the water molecules move slowly enough that they rarely have enough energy to break peptide bonds. But as temperature increases, molecular motion increases. Water molecules collide with the peptide chain with greater force. Some collisions break bonds. Each broken bond creates a smaller, inactive fragment. Over time, you end up with a vial of fragments that used to be functional injectable peptide.
Research on GLP-1 peptide stability shows that recoveries were unaffected by freezing cycles or storage on ice for up to three hours, but were significantly impaired when samples stood at room temperature for more than one hour in plasma studies. While those studies measured endogenous GLP-1 in blood samples rather than pharmaceutical preparations, the principle holds: peptides and temperature are not friends.
Aggregation and fibrillation
GLP-1 class peptides have a known tendency to self-assemble into structures called amyloid fibrils. This is essentially the peptide molecules sticking together in long chains, like tangled yarn. Once aggregation begins, the individual peptide molecules are no longer available to bind receptors. Research published in Biophysical Journal demonstrated that GLP-1 aggregates into low-molecular-weight oligomers that can progress to full fibrillation. Temperature accelerates this process. Proper peptide storage keeps aggregation rates minimal.
Oxidation
Tirzepatide contains amino acids that are vulnerable to oxidation, including methionine residues. Oxygen dissolved in the solution reacts with these residues faster at higher temperatures. The oxidized peptide loses binding affinity. This is one reason why manufacturers recommend keeping the medication in its original packaging.
The packaging reduces light exposure, and light catalyzes oxidation reactions. Understanding the safety considerations behind peptide storage helps you protect your investment.
FDA storage guidelines for Mounjaro and Zepbound
The FDA prescribing information for both Mounjaro and Zepbound provides clear storage instructions. These are not recommendations. They are the conditions under which the manufacturer guarantees the medication will remain safe and effective until its expiration date.
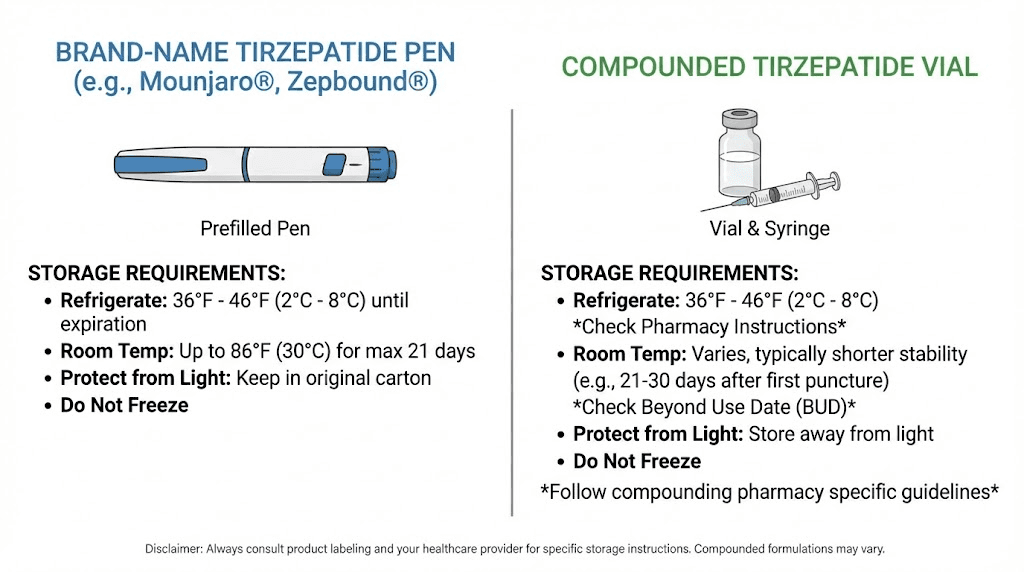
Refrigerated storage (primary)
Store unopened pens and vials at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Keep them in the original carton to protect from light. That is the baseline requirement and the condition under which the labeled expiration date applies. When stored this way, your tirzepatide remains potent until the date printed on the packaging.
Room temperature storage (temporary)
Each single-dose pen or single-dose vial can be stored at room temperature, up to 86 degrees Fahrenheit (30 degrees Celsius), for a maximum of 21 days. This 21-day window is cumulative, not resettable. Once a pen leaves the fridge, the clock starts. If you put it back in the fridge and take it out again, the counter does not reset.
This is critical. Many people assume that returning the pen to the fridge buys them more time. It does not.
After 21 days at room temperature, discard the pen or vial even if it appears normal. Even if there are doses remaining. The manufacturer cannot guarantee potency beyond that window outside refrigeration. For tirzepatide dosing to work properly, the medication must retain its full potency.
Multi-dose vials
The newer multi-dose vials for Zepbound and Mounjaro follow slightly different rules. Store them refrigerated at 36 to 46 degrees Fahrenheit. Do not freeze. Once opened (first punctured with a needle), the vial can be used for up to 28 days. Keep it refrigerated during those 28 days. This is a separate consideration from the 21-day room temperature allowance for single-dose products.
Never freeze
Do not freeze tirzepatide in any form. Never use a pen or vial that has been frozen. Freezing causes water in the solution to form ice crystals, and those crystals physically tear apart peptide molecules. The damage is irreversible. A thawed pen might look fine. The solution might be clear. But the peptide chains have been mechanically disrupted at the molecular level. This is the same reason you should never put reconstituted peptides in the freezer unless the specific formulation is designed for it.
Compounded tirzepatide storage requirements
Compounded tirzepatide deserves its own section because the rules are different. Not slightly different. Meaningfully different. And if you are using compounded tirzepatide, which many people are due to the cost of brand-name GLP-1 medications, you need to understand these distinctions.
Why compounded is different
Brand-name Mounjaro and Zepbound go through extensive stability testing by Eli Lilly. The manufacturer stores batches at various temperatures for months and years, measuring degradation at every step. This testing determines the expiration date and the 21-day room temperature window.
Compounding pharmacies do not perform the same scale of stability testing. Instead, they assign a Beyond-Use Date (BUD) based on United States Pharmacopeia (USP) guidelines and any stability data they have generated. This BUD is typically shorter than a manufacturer expiration date.
Typical beyond-use dates
Most compounded tirzepatide carries a BUD of 28 to 90 days from the date of preparation. Some pharmacies, particularly those with in-house stability testing, may assign longer dates. But the industry standard range falls in that window. When stored properly in the refrigerator at 2 to 8 degrees Celsius, compounded tirzepatide can remain stable for the duration of its BUD.
The important difference: compounded tirzepatide does not carry the same 21-day room temperature allowance as brand-name products. Unless your specific pharmacy provides written documentation that their formulation is stable at room temperature for a defined period, assume it needs refrigeration at all times.
Lyophilized versus liquid compounded tirzepatide
Some compounding pharmacies provide tirzepatide in lyophilized (freeze-dried) form. Others provide it as a pre-mixed liquid solution. The storage requirements differ significantly between these two forms.
Lyophilized (freeze-dried) tirzepatide: This is the most stable form because removing water eliminates hydrolysis as a degradation pathway. Lyophilized peptides can often be stored at refrigerator temperatures for longer periods than liquid formulations. Some lyophilized peptides remain stable at room temperature for extended periods, though you should always follow your pharmacy specific instructions. For more detail on this topic, see our guide on lyophilized versus liquid peptides.
Liquid (reconstituted) tirzepatide: Once tirzepatide is in solution, all the degradation pathways discussed earlier become active. Hydrolysis. Oxidation. Aggregation. The clock is ticking. Liquid formulations almost always require constant refrigeration and have shorter beyond-use dates. Understanding how long peptides last in the fridge once reconstituted helps you plan your usage schedule.
Preservatives and stabilizers matter
Compounded formulations may contain different preservatives and stabilizers than the brand-name product. Common additions include:
Benzyl alcohol as a preservative in multi-dose vials
Buffer solutions to maintain optimal pH (typically around 4.5 to 5.0 for tirzepatide)
Mannitol or trehalose as stabilizers in lyophilized formulations
These additives affect stability profiles. A compounded vial with good preservatives and proper buffering may tolerate brief temperature excursions better than a bare peptide in water. But do not test this assumption. Follow your pharmacy storage instructions exactly. If those instructions are unclear, call and ask specifically about temperature requirements and room temperature tolerance. The resources at SeekPeptides can help you understand what questions to ask your provider about peptide protocols and storage requirements.
What happens when tirzepatide is not refrigerated properly
Storing tirzepatide outside its required temperature range triggers a cascade of molecular changes. None of them are good. All of them reduce the effectiveness of your medication. Some of them create byproducts you definitely do not want to inject.
Progressive potency loss
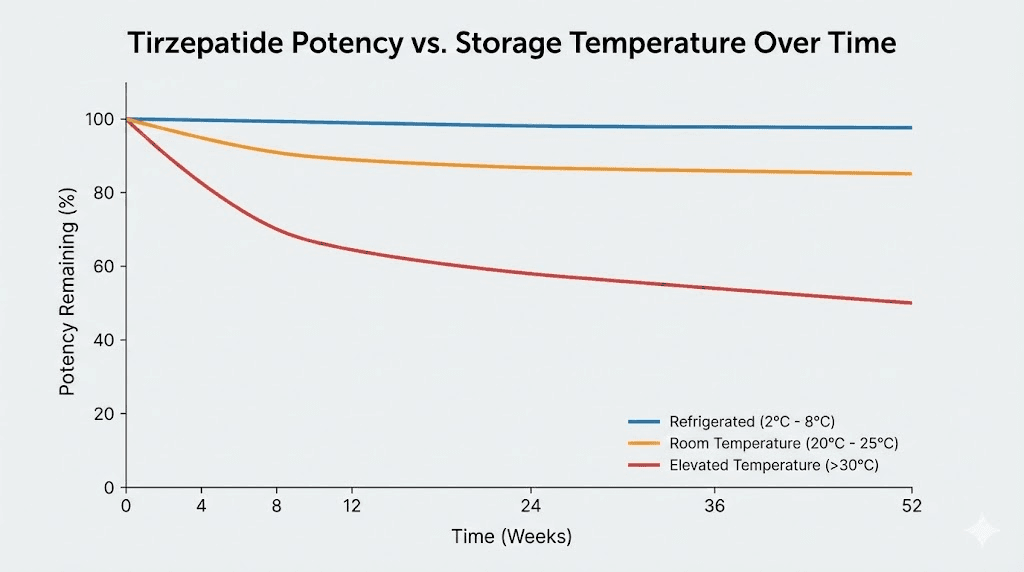
The first thing that happens is a gradual loss of active peptide. Not all at once. Not overnight. But steadily, like a slow leak in a tire. Each day at elevated temperature, more peptide molecules degrade. The rate depends on the temperature. At 77 degrees Fahrenheit (25 degrees Celsius), degradation is relatively slow. At 95 degrees Fahrenheit (35 degrees Celsius), it accelerates dramatically.
This matters for dosing accuracy. If your vial has lost 20% of its potency, you are effectively under-dosing by 20%. If you are on a specific dose chart for weight management, that under-dosing means slower results. If you are on tirzepatide for blood sugar management, it means less glycemic control.
Degradation products
When tirzepatide breaks down, it does not just disappear. It creates degradation products. Truncated peptide fragments. Oxidized variants. Deamidated forms. These fragments are not biologically neutral. Some may trigger immune responses at the injection site. Others may compete with intact tirzepatide for receptor binding without activating the receptor, effectively blocking the medication from working.
Research on tirzepatide synthesis identified diketopiperazine (DKP) formation as a significant degradation pathway. This cyclization reaction occurs more readily at higher temperatures and creates peptide fragments that have no therapeutic value. The pharmaceutical science journal ACS Omega published detailed kinetic modeling of this degradation, confirming that temperature control is the primary factor in preventing DKP-related impurity formation.
Visual signs of severe degradation
Most degradation is invisible. But severe degradation can produce visible changes:
Cloudiness or particles in the solution indicate protein aggregation
Color changes from clear to yellow or brown suggest oxidation
Unusual viscosity (thicker or thinner than normal) indicates structural changes
If you notice any of these signs, do not use the medication. Discard it. But remember, the absence of visible changes does not guarantee potency. Significant degradation can occur while the solution looks perfectly normal. When you are comparing before and after results, inconsistent storage could explain why some people see better outcomes than others on the same dose.
The compounding factor: time plus temperature
Degradation is not just about temperature. It is about the combination of temperature and time. One hour at 90 degrees might cause negligible degradation. Ten hours at 90 degrees is a different story. Three weeks at 80 degrees is worse than three days at 90 degrees, even though the peak temperature was lower. This time-temperature relationship is why the FDA gives a specific number of days (21) at a specific maximum temperature (86 degrees Fahrenheit) rather than just a temperature limit.
The 21-day room temperature rule explained
This rule causes more confusion than any other aspect of tirzepatide storage. Let me break it down clearly.
How the 21-day window works
When an unopened Mounjaro or Zepbound pen or single-dose vial is removed from the refrigerator, you have 21 calendar days to use it. During those 21 days, the temperature must stay below 86 degrees Fahrenheit (30 degrees Celsius) at all times. After 21 days, discard the pen regardless of remaining medication. The pen does not need to be at room temperature continuously. It can sit in a cool room, a temperature-controlled travel case, or anywhere that stays under 86 degrees. The critical factor is the cumulative time outside the fridge.
The no-return policy
Once a pen has been stored at room temperature, do not return it to the refrigerator. This is the official guidance from Eli Lilly, and it catches many people off guard.
Why? Temperature cycling, moving between warm and cold repeatedly, creates additional stress on the peptide molecule. Each cycle causes slight condensation inside the pen mechanism. This moisture can affect the injection mechanism and introduce contaminants. The peptide itself also experiences thermal stress during each transition. While a single warm-to-cold transition is unlikely to cause dramatic degradation, the manufacturer has not tested repeated cycling, so they cannot guarantee stability under those conditions.
Practical translation: if you take your pen out for a trip, use it within 21 days. Do not put it back in the fridge when you get home and try to extend its life. The same principle applies to other peptide injections unless the manufacturer specifically states otherwise.
What 86 degrees actually means in practice
Most people do not walk around with thermometers. So what does 86 degrees Fahrenheit feel like? It is a warm day. Not blistering. Not desert heat. Just a warm summer afternoon. Most air-conditioned homes stay well below 86 degrees. Most offices are well below it. But a car in summer? A car parked in sunlight in most of the United States will exceed 86 degrees within minutes. A glove compartment can reach 130 degrees or higher.
Never leave tirzepatide in a parked car. Not for five minutes. Not while you run into a store. The temperature inside a car can spike faster than most people realize.
Accidentally left tirzepatide out: what to do
It happens. You forget. You fall asleep. You leave the pen on the counter overnight. Maybe it sat out for a few hours, maybe a full day. Panic is understandable but rarely helpful. Here is a decision framework instead.
Step 1: check the temperature
What was the ambient temperature where the pen was sitting? If your home thermostat was set to 72 degrees and the pen sat on a kitchen counter, you are probably fine for a single overnight episode. That scenario falls well within the 21-day, sub-86-degree window. If the pen was in a hot car, on a sunny windowsill, or in a room without air conditioning during summer, the situation is more concerning.
Step 2: check the duration
How long was it out? A few hours at moderate room temperature is almost certainly fine and falls within the manufacturer allowance. A full day? Still within the 21-day window if the temperature stayed under 86 degrees. Several days in a warm environment? Call your pharmacy or healthcare provider for guidance.
Step 3: inspect the medication
Look at the solution. Is it clear and colorless? Are there any particles, cloudiness, or color changes? If the solution looks normal, and the temperature stayed within acceptable range, and the total unrefrigerated time is under 21 days, the medication is likely still usable according to the manufacturer guidelines.
Step 4: for compounded tirzepatide, call your pharmacy
If you are using compounded tirzepatide and it was left out, contact your compounding pharmacy directly. Provide the approximate duration and temperature. They can advise based on their specific formulation stability data. Compounded products do not have the same room temperature allowance as brand-name pens, so professional guidance is important here. Understanding how long peptides last at room temperature gives you useful context for these situations.
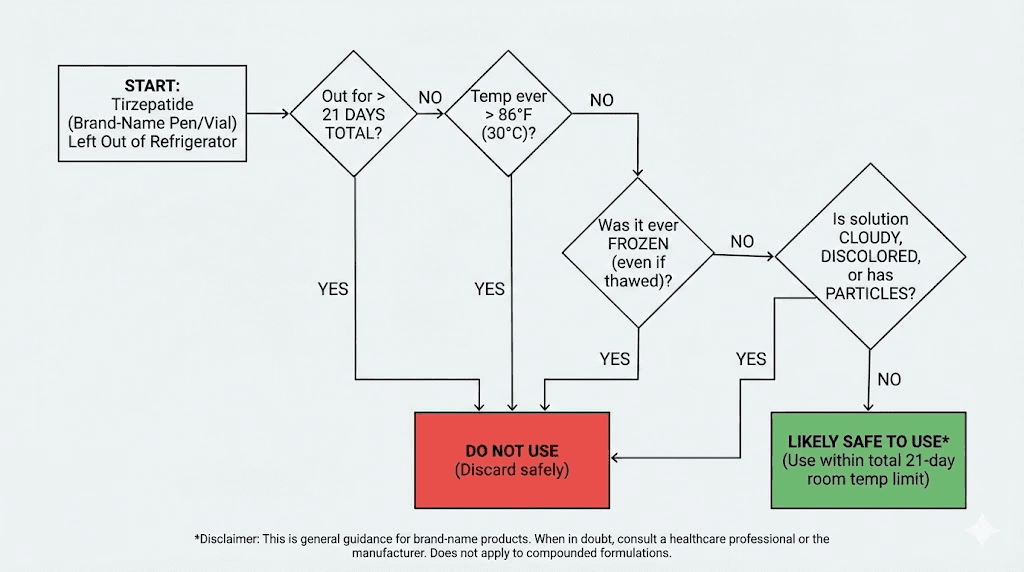
When to discard without question
Throw it away if:
The solution is cloudy, discolored, or contains particles
The medication was exposed to temperatures above 86 degrees Fahrenheit
The medication was frozen at any point
The total time at room temperature exceeds 21 days (for brand-name products)
You are uncertain about the conditions and cannot verify them
The cost of a replacement pen is always less than the cost of injecting degraded medication. Reduced potency means reduced results. Degradation products mean potential side effects. Neither outcome is worth the gamble.
How to store tirzepatide at home
Proper home storage is not complicated, but it requires attention to a few details that most people overlook. The fridge is not a uniform-temperature box. Some spots are colder than others. Some spots are warmer. Where you put your tirzepatide matters.
Best location in the refrigerator
Place tirzepatide in the main body of the refrigerator, toward the back. Not on the door. The door experiences the most temperature fluctuation because it swings open and closed throughout the day. Each time the door opens, warm air rushes in and directly hits items stored on door shelves. The back of the main compartment stays the most consistently cold.
Avoid the back wall in older refrigerators where the cooling element sits. Items placed directly against the cooling element can partially freeze, and freezing tirzepatide is permanent damage. A middle shelf, toward the back, is the sweet spot.
Keep it in the original carton
The carton is not just packaging. It serves two protective functions. First, it blocks light. Light, especially fluorescent light from the refrigerator bulb, catalyzes oxidation reactions in the peptide. Second, the cardboard provides a small amount of thermal insulation, buffering the pen against momentary temperature spikes when the fridge door opens. For the same reason, peptides in powder form also benefit from light-protected storage.
Temperature monitoring
Consider placing a small digital thermometer in your refrigerator. They cost a few dollars and give you peace of mind. The target range is 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Most home refrigerators run between 35 and 40 degrees, which is perfect. If your fridge runs warmer than 46 degrees, adjust the temperature setting. If it regularly dips below 32 degrees in certain spots, those spots are freezing territory, and you need to keep medication away from them.
Away from food with strong odors
This might sound unusual, but some volatile compounds from strongly flavored foods can permeate packaging over time. While this does not directly affect the peptide chemistry, it is good practice to store medications in a clean, dedicated area of the refrigerator. A small plastic container or bin works well for keeping medication organized and separated.
Multi-dose vial handling
If you are using a multi-dose vial, additional precautions apply. After each use, wipe the rubber stopper with an alcohol swab before returning it to the fridge. This prevents bacterial contamination that could grow between injections. Keep the vial upright. Mark the date you first punctured it so you know when the 28-day use window expires. Do not draw up doses in advance and store them in syringes, as the peptide can adsorb to syringe surfaces over time, reducing the effective dose. The proper handling of peptide vials extends naturally to tirzepatide products.
Traveling with tirzepatide
Travel is where tirzepatide storage gets complicated. You cannot always guarantee refrigeration. Temperatures fluctuate. TSA has opinions about needles and medication. And if your trip lasts longer than 21 days, you need a plan for keeping your medication cold the entire time.
Short trips (under 21 days)
For trips shorter than three weeks, you can take advantage of the 21-day room temperature window (brand-name products only). Remove the pen from the fridge on your departure day. Keep it below 86 degrees Fahrenheit throughout your trip. Use it within 21 days. This is the simplest approach and requires no special equipment.
However, even on short trips, a basic insulated pouch is a smart investment. Hotel rooms can get warm. Luggage in airport holds or car trunks can exceed 86 degrees easily. A simple insulated pouch with a cool pack provides a buffer against temperature spikes without the complexity of active cooling. Many people who are managing their tirzepatide dosing schedule find that trip planning is an important part of staying on protocol.
Long trips (over 21 days)
Trips longer than 21 days require active refrigeration for brand-name products and for most compounded formulations. Options include:
Medical-grade travel coolers: These are insulated bags designed specifically for medication transport. Some use evaporative cooling technology that maintains temperatures below 80 degrees Fahrenheit for 45+ hours without electricity. Others use USB-powered cooling elements that keep medication at refrigerator temperatures (36-46 degrees Fahrenheit) indefinitely when plugged in.
Hotel refrigerators: Most hotel rooms have a mini-fridge. The temperature inside hotel mini-fridges varies widely, from barely cool to nearly freezing. If you rely on a hotel fridge, place a thermometer inside and verify the temperature before storing medication. Some mini-fridges run warmer than 46 degrees, especially when set to a low cooling level.
Portable USB refrigerators: These compact units can maintain 2-8 degree Celsius temperatures using a USB power source. They are designed for medications and can hold two to four pens. They are invaluable for travelers who need guaranteed cold chain maintenance.
Flying with tirzepatide
Keep tirzepatide in your carry-on bag. Never put it in checked luggage. Cargo holds are not temperature-controlled, and temperatures in the cargo area of an aircraft can drop below freezing at cruising altitude. A pen that freezes at 30,000 feet is destroyed, even if it thaws normally by the time you land.
TSA allows injectable medications through security checkpoints. You do not need a prescription label on the medication itself, though having one (or carrying a copy of your prescription) can smooth the process. Needles are permitted when accompanied by injectable medication. Declare your medication to the TSA agent if you want to keep it out of the X-ray machine, though X-rays do not affect peptide medications.
Gel packs and ice packs for medication cooling are also permitted through TSA, even if they are partially frozen. They fall under the medical exemption for liquids. However, fully frozen gel packs may receive additional screening. When in doubt, use packs that are cool but not solid ice to avoid delays. These travel considerations apply equally to semaglutide and other GLP-1 injectable medications you might be carrying.
International travel considerations
If you are crossing borders, carry a copy of your prescription and a letter from your healthcare provider. Some countries have strict regulations about bringing injectable medications across borders. Having documentation prevents delays and confiscation. Keep medication in its original packaging when possible, as this provides the clearest identification.
Time zone changes can affect your dosing schedule. If you are on weekly tirzepatide injections and crossing multiple time zones, plan your injection day so it falls at a consistent interval. A day or two of schedule shift is generally not problematic, but discuss with your provider if your travel involves significant time changes.
Tirzepatide storage comparison: brand vs compounded vs lyophilized
Storage factor | Brand-name (Mounjaro/Zepbound) | Compounded liquid | Compounded lyophilized |
|---|---|---|---|
Refrigerated temperature | 36-46F (2-8C) | 36-46F (2-8C) | 36-46F (2-8C) preferred |
Room temp allowance | 21 days below 86F | Check with pharmacy | Often more tolerant |
Shelf life refrigerated | Until expiration date | 28-90 days (BUD) | Varies, often longer |
Freeze tolerance | None, discard if frozen | None, discard if frozen | More tolerant before reconstitution |
Light sensitivity | Keep in carton | Keep in carton or foil | Store in original container |
After opening (multi-dose) | 28 days refrigerated | 28 days typical | Follow pharmacy BUD post-reconstitution |
Stability testing | Extensive (manufacturer) | Limited (USP guidelines) | Limited (USP guidelines) |
This table illustrates why brand-name products offer more predictable storage parameters. The extensive stability testing gives you confidence in the 21-day room temperature window. Compounded products, while often more affordable, require stricter adherence to refrigeration because fewer data points support temperature excursions. For help choosing between formulations, our semaglutide versus tirzepatide comparison covers the broader landscape of GLP-1 options.
Common storage mistakes that destroy tirzepatide potency
After analyzing hundreds of user reports and forum discussions, certain mistakes come up repeatedly. Each one seems minor in isolation. Together, they can slash the effectiveness of your medication before you realize what happened.
Mistake 1: storing on the fridge door
The door is the warmest part of the refrigerator and the most temperature-variable. Every time you open the fridge, door shelves get hit with room temperature air. Over the course of a day, items on the door shelf can experience temperature swings of 10 to 15 degrees. For milk, this is negligible. For a peptide medication, these repeated thermal cycles add up. Move your tirzepatide to the main body of the fridge.
Mistake 2: freezing by accident
It happens more often than you think. A pen gets pushed to the back of the fridge where it contacts the cooling element. The fridge thermostat drifts lower than intended. Someone adjusts the temperature dial without realizing medication is stored inside. Once frozen, the damage is done. Ice crystals form within the solution and mechanically disrupt peptide structures. Even partial freezing at the edges of a vial can cause localized damage. The question of whether peptides expire becomes irrelevant if they are frozen first.
Mistake 3: leaving in a hot car
A car interior can reach 120 to 170 degrees Fahrenheit on a sunny day. Even on a mild 75-degree day, a car in direct sunlight can exceed 100 degrees within 20 minutes. Five minutes in a hot car during summer is enough to push tirzepatide past its temperature tolerance. Never leave medication in a vehicle, even briefly.
Mistake 4: assuming the 21-day clock resets
It does not. If your pen spent three days out of the fridge last week and you are taking it out again for a trip, you have 18 days remaining, not 21. The cumulative time at room temperature is what matters. Some people try to track this informally and lose count. If you are going to use the room temperature allowance, write the date you first removed the pen on a piece of tape attached to it.
Mistake 5: using expired medication
The expiration date assumes continuous refrigeration. If your tirzepatide spent any time outside the fridge, the effective expiration date is shorter than printed. If you are nearing the expiration date and the pen has also spent time at room temperature, the compound risk of degradation increases. Err on the side of discarding medication that is both near expiration and has been temperature-stressed. Understanding whether expired GLP-1 medications are still usable applies the same logic to tirzepatide.
Mistake 6: exposing to direct sunlight
UV light accelerates oxidation of methionine and tryptophan residues in peptides. Even indirect sunlight through a window provides enough UV to matter over hours or days. Keep tirzepatide in its carton or in a drawer, away from windows, when it is not in the fridge. This applies to daily storage and to travel situations where the pen might be sitting on a hotel nightstand near a window.
Mistake 7: drawing up doses in advance
Some people pre-fill syringes from multi-dose vials to save time. This creates multiple problems. The peptide can adsorb to syringe surfaces, reducing the dose. Air exposure increases oxidation. The sterility of a pre-filled syringe degrades over time. Draw your dose immediately before injection, not hours or days in advance. If you need help with preparation, our guide to mixing peptides with bacteriostatic water covers proper technique.
Power outages and emergency storage situations
Power outages present a real storage challenge, especially during summer storms when they can last days. Here is what to do when your fridge stops working.
The first few hours
A closed refrigerator maintains temperature for about four hours without power. Do not open the fridge door during this time. Every time the door opens, cold air escapes and warm air enters. If you know the power will be out for a while, resist the urge to check on your medication. The fridge is doing its job as an insulated box. Leave it alone.
After four hours
If power has not returned after four hours, your fridge is warming. At this point, you have options depending on the situation:
Ice coolers: Pack your tirzepatide in an insulated cooler with ice packs. Monitor the temperature with a thermometer. The goal is keeping it below 46 degrees Fahrenheit. With adequate ice, a quality cooler can maintain medication-safe temperatures for 24 to 48 hours.
Neighbor or friend: If someone nearby has power, ask to store your medication in their fridge temporarily.
Room temperature window: For brand-name products, remember the 21-day room temperature allowance. If your home stays below 86 degrees, the medication is safe for up to 21 days without refrigeration. This provides significant breathing room during most power outages.
Extended outages
If you are facing a multi-day outage during extreme heat, prioritize getting the medication to a refrigerated location. Pharmacies, hospitals, and some community cooling centers may be willing to store medication temporarily if you explain the situation. The cost of replacing a tirzepatide pen is significant, and most healthcare providers understand the urgency.
How storage affects tirzepatide effectiveness and your results
Storage is not just a theoretical concern. It directly impacts how well your tirzepatide protocol works. People who follow optimal storage practices consistently report better and more predictable outcomes than those who are casual about temperature management.
Dose consistency depends on potency consistency
Your tirzepatide dose in units is calibrated to a specific amount of active peptide. If degradation reduces the active peptide by 15%, your 5mg dose is effectively a 4.25mg dose. That difference matters, especially during the critical early weeks when your body is adjusting and you are trying to find the right dose for microdosing protocols or standard escalation.
Inconsistent storage creates inconsistent dosing, which creates inconsistent results, which makes it nearly impossible to optimize your protocol because you cannot tell whether the problem is the dose or the storage.
Why some people plateau on tirzepatide
Not all plateaus are biological. Some are pharmaceutical. If you are not losing weight despite being on what should be an effective dose, and your diet and activity are consistent, storage issues are worth investigating. Ask yourself:
Has the medication been properly refrigerated at all times?
Has it spent time at room temperature beyond the allowed window?
Is the compounded product past its beyond-use date?
Has it been exposed to heat, light, or freezing at any point?
If the answer to any of these is yes or uncertain, degraded medication may be contributing to your plateau. A fresh vial with confirmed proper storage can help you determine if dosing needs to change or if the problem was storage all along. Understanding how tirzepatide suppresses appetite requires recognizing that appetite suppression depends on full-potency medication reaching the right receptors.
The financial angle
Tirzepatide is not cheap. Brand-name Mounjaro and Zepbound carry significant price tags. Even compounded versions represent a meaningful investment. Poor storage is literally money down the drain. One pen ruined by a hot car or improper freezing can cost hundreds of dollars. Over the course of a year, casual storage habits can waste thousands of dollars in degraded medication that delivers sub-therapeutic doses. The side effects of tirzepatide are worth managing, but only if the medication is potent enough to deliver benefits alongside those effects.
Storage for specific tirzepatide brands and formulations
Different tirzepatide products have slightly different packaging and storage nuances. Here is a brand-by-brand breakdown.
Mounjaro (tirzepatide) pens
Mounjaro pens are single-dose prefilled injectors. Each pen contains one dose, and each dose is used in its entirety. Storage: refrigerate at 36-46 degrees Fahrenheit. Room temperature allowance: 21 days below 86 degrees. Do not freeze. Keep in original carton. Discard used pens even if they appear to have medication remaining.
Zepbound (tirzepatide) pens
Zepbound pens follow identical storage requirements as Mounjaro because they contain the same molecule. The primary difference between Mounjaro and Zepbound is the indication (Mounjaro for type 2 diabetes, Zepbound for weight management) and the dosing options available. Storage-wise, they are interchangeable in their requirements. For details on how Zepbound compares to other options, see our GLP-1 comparison guides.
Zepbound and Mounjaro multi-dose vials
Multi-dose vials are a newer format. Store refrigerated. Once the vial is first punctured, use within 28 days while keeping it refrigerated. These vials require careful handling with proper sterile technique for each draw to prevent contamination. The multi-dose format is often more cost-effective but demands more attention to storage and handling than single-use pens.
Compounded tirzepatide vials
Compounded tirzepatide comes in a standard pharmaceutical vial. Storage requirements depend entirely on the compounding pharmacy formulation. General guidelines: refrigerate at all times unless your pharmacy specifically states a room temperature allowance. Follow the beyond-use date on the label exactly. Once reconstituted (if provided in lyophilized form), follow your pharmacy instructions for post-reconstitution storage. Our guide on reconstituted peptide shelf life provides additional context for compounded formulations.
Reconstituting tirzepatide: storage before and after
If you are working with lyophilized (freeze-dried) compounded tirzepatide, the reconstitution process introduces a critical storage transition. The rules change the moment you add water to the powder.
Before reconstitution (lyophilized powder)
Lyophilized tirzepatide is significantly more stable than the liquid form. Without water, hydrolysis cannot occur. The powder form is also less susceptible to aggregation and oxidation, though it is not immune to either. Store lyophilized tirzepatide refrigerated for maximum shelf life. Many lyophilized peptides can tolerate short periods at room temperature better than their reconstituted counterparts, but there is no reason to test this when a fridge is available.
After reconstitution
The moment you add bacteriostatic water or sterile water to the lyophilized powder, every degradation pathway activates. Hydrolysis begins. Oxidation begins. Aggregation potential increases. The reconstituted solution must be refrigerated immediately and used within the timeframe specified by your pharmacy.
Typical post-reconstitution storage life for compounded peptides is 28 to 30 days refrigerated. Some formulations with preservatives like benzyl alcohol may last longer. Check your pharmacy documentation for the specific beyond-use date. Our peptide reconstitution calculator helps with preparation, and the interactive tool on our site makes the math simple.
Reconstitution best practices for stability
How you reconstitute affects how long the solution stays stable. Follow these practices:
Use the right diluent: Bacteriostatic water (containing 0.9% benzyl alcohol) provides antimicrobial protection that extends vial life. Sterile water for injection does not contain preservatives and should be used in single-dose applications only.
Add water gently: Direct the stream of water along the vial wall, not directly onto the peptide cake. Direct impact can denature peptide at the surface.
Swirl, do not shake: Shaking creates foam, and foam means air exposure at a massive surface area. Air exposure means oxidation. Gently swirl the vial until the powder dissolves.
Refrigerate immediately after reconstitution: Do not leave the freshly reconstituted vial at room temperature while you prepare your injection supplies. Reconstitute, draw your dose, and refrigerate the remainder promptly.
For a complete walkthrough of the reconstitution process, see our detailed peptide reconstitution guide.
Tirzepatide storage vs other GLP-1 medications
If you are transitioning between GLP-1 medications or considering a switch, understanding how tirzepatide storage compares to alternatives helps you plan. Different GLP-1 receptor agonists have different stability profiles.
Medication | Refrigeration required | Room temp window | Max room temp | Freeze safe |
|---|---|---|---|---|
Tirzepatide (Mounjaro/Zepbound) | Yes (36-46F) | 21 days | 86F (30C) | No |
Semaglutide (Ozempic) | Yes (36-46F) | 56 days | 86F (30C) | No |
Semaglutide oral (Rybelsus) | No (room temp) | N/A | 86F (30C) | N/A |
Liraglutide (Saxenda) | Yes (36-46F) | 30 days | 86F (30C) | No |
Compounded tirzepatide | Yes (36-46F) | Pharmacy-specific | Pharmacy-specific | No |
Notice that semaglutide (Ozempic) has a significantly longer room temperature window at 56 days compared to tirzepatide at 21 days. This is relevant for travelers and for people who have difficulty maintaining consistent refrigeration. If storage flexibility is a priority, this is one factor to discuss with your provider when comparing semaglutide versus tirzepatide. For people investigating the newer triple-agonist retatrutide, storage requirements follow similar cold-chain principles.
The oral formulation of semaglutide (Rybelsus) is the only GLP-1 medication that does not require refrigeration at all. It stores at room temperature like a regular tablet. However, oral semaglutide has different bioavailability and absorption characteristics, which is why most people on injectable GLP-1 therapy are working with products that need the fridge. Our guide on oral tirzepatide covers the emerging alternatives.
Storage and the tirzepatide supply chain: what happens before it reaches you
Your tirzepatide goes through a cold chain before it ever reaches your refrigerator. Understanding this chain helps you evaluate whether your medication arrived in optimal condition.
Manufacturing and distribution
Eli Lilly manufactures Mounjaro and Zepbound in temperature-controlled facilities. The medication is stored at 2 to 8 degrees Celsius from production through warehouse storage. Shipping uses insulated packaging with temperature monitoring. Major distributors maintain cold-chain logistics with refrigerated trucks and temperature-logged shipping containers.
Pharmacy storage
Pharmacies store tirzepatide in pharmaceutical-grade refrigerators that maintain temperatures between 2 and 8 degrees Celsius. These refrigerators have alarm systems that alert staff if temperatures drift outside acceptable ranges. Chain pharmacies and hospital pharmacies undergo regular inspection of their cold storage equipment.
The last mile risk
The greatest risk in the supply chain is the "last mile," the trip from the pharmacy to your home. If you pick up tirzepatide and then run errands for two hours with it in your car, you have introduced a temperature risk. In summer, car interiors can exceed safe temperatures within minutes. In winter, the medication could freeze in a cold car trunk.
When you pick up tirzepatide from the pharmacy, go home and refrigerate it. If you need to make stops, bring an insulated bag. Some pharmacies offer insulated bags for medication transport, ask if yours does. If you receive tirzepatide by mail order, check the condition of the cold pack upon delivery. If the cold pack is warm and the medication feels warm, contact the pharmacy about a replacement. These best practices apply to all peptide storage situations, not just tirzepatide.
The science of peptide stability: why cold matters at the molecular level
For those who want to understand the deeper science behind why tirzepatide needs refrigeration, this section explains the molecular mechanisms. Understanding these mechanisms makes all the practical advice in this guide make more sense.
Arrhenius equation and degradation kinetics
The Arrhenius equation describes how chemical reaction rates change with temperature. For most chemical reactions, including peptide degradation, the rate approximately doubles for every 10-degree Celsius increase in temperature. This means tirzepatide at 25 degrees Celsius (77 degrees Fahrenheit) degrades roughly four times faster than at 5 degrees Celsius (41 degrees Fahrenheit, the middle of the recommended range). At 35 degrees Celsius (95 degrees Fahrenheit), it degrades roughly eight times faster than at proper refrigeration temperature.
These are not small differences. An eightfold increase in degradation rate means that one day at 95 degrees is equivalent to eight days at refrigerator temperature in terms of damage to the peptide. This mathematical relationship is why the manufacturer sets both a time limit and a temperature limit for room temperature storage.
Deamidation
Asparagine and glutamine residues in peptides can undergo deamidation, a reaction where the amide group is replaced by a carboxyl group. This changes the charge and shape of the peptide at that position. Deamidation rates increase with temperature and also depend on pH and the sequence of neighboring amino acids. Deamidated tirzepatide has reduced receptor binding affinity, which directly translates to reduced therapeutic effect.
Methionine oxidation
Methionine residues are particularly vulnerable to oxidation by dissolved oxygen and reactive oxygen species. The rate of oxidation increases with temperature, light exposure, and oxygen concentration. Oxidized methionine residues can significantly alter the local structure of the peptide, potentially disrupting receptor binding. This is one reason the manufacturer specifies storage in the original carton, to reduce light-catalyzed oxidation. Similar considerations apply to copper peptide storage and other peptide shelf life questions.
Disulfide bond scrambling
While tirzepatide does not contain traditional disulfide bonds, it does contain a C20 fatty diacid chain attached via a linker to a lysine residue. This fatty acid modification is what gives tirzepatide its extended half-life by promoting albumin binding. Temperature-induced changes in the conformation around this modification site could affect how well the fatty acid chain interacts with albumin, potentially altering the medication pharmacokinetics, how long it stays active in your body.
Seasonal storage considerations
Your storage needs change with the seasons. What works in January may not work in July.
Summer challenges
Summer heat is the primary enemy of tirzepatide storage. Power outages from storms are more common. Air conditioning failures happen. Cars become ovens. Even brief lapses in cold chain can matter when ambient temperatures are high.
During summer, be especially vigilant about:
Getting medication from pharmacy to home quickly
Monitoring fridge temperature (fridges work harder in hot kitchens)
Having backup cooling for power outages
Never leaving medication in a vehicle, even for minutes
Keeping medication away from sunny windows
Winter challenges
Cold weather brings the opposite risk: freezing. If your medication is shipped during winter, the cold pack in the shipping box and the ambient temperature could push the medication below freezing. Mail-order prescriptions left on a doorstep in sub-zero temperatures can freeze before you bring them inside.
During winter:
Request signature-required delivery so packages are not left outside
Track deliveries and bring them inside promptly
Do not store tirzepatide in a garage or unheated room
Check the temperature of your car before placing medication inside during transport
Whether you are managing tirzepatide through summer or winter, maintaining the proper storage conditions keeps your medication effective. SeekPeptides members access detailed storage protocols and seasonal management guides for every peptide in their regimen.
Signs your tirzepatide may have been compromised
Sometimes you suspect a storage breach but are not sure. Maybe the fridge felt warm. Maybe a family member reorganized the fridge. Maybe you cannot remember if you left it out. Here are the signs to watch for.
Visual indicators
Cloudiness: The solution should be clear and colorless. Any cloudiness suggests protein aggregation.
Particles: Floating specks or sediment indicate peptide breakdown and aggregation.
Color change: Any shift from clear/colorless to yellow, amber, or brown suggests oxidation.
Changed consistency: If the solution seems thicker or thinner than expected, structural changes have occurred.
Functional indicators
Reduced appetite suppression: If the same dose that previously suppressed your appetite is suddenly less effective, potency loss is possible.
Increased injection site reactions: Degradation products can trigger local immune responses that the intact peptide does not.
Plateau or weight regain: Unexpected changes in your progress pattern could signal compromised medication.
None of these indicators are definitive on their own. Diet changes, stress, hormonal shifts, and other factors can also cause these patterns. But if you suspect a storage issue AND notice reduced effectiveness, the most prudent action is to start with a fresh, properly stored supply and see if your results improve. Many factors can contribute to feeling like tirzepatide is making you tired or less effective than expected, and storage is one variable you can control completely.
Frequently asked questions
Can I put tirzepatide back in the fridge after it has been at room temperature?
For brand-name Mounjaro and Zepbound, the manufacturer recommends against returning the medication to the refrigerator once it has been stored at room temperature. The 21-day clock continues regardless. For compounded tirzepatide, consult your compounding pharmacy for their specific guidance, as formulations vary. Learn more in our complete tirzepatide fridge storage guide.
Does compounded tirzepatide need to be refrigerated at all times?
Yes. Unless your compounding pharmacy provides specific documentation that their formulation tolerates room temperature for a defined period, assume compounded tirzepatide requires constant refrigeration. Compounded products have not undergone the same stability testing as brand-name products, so the conservative approach is the safe approach.
What happens if tirzepatide freezes?
Freezing irreversibly damages tirzepatide. Ice crystals form within the solution and mechanically disrupt the peptide molecular structure. Discard any tirzepatide that has been frozen, even if it appears normal after thawing. This is consistent with storage guidelines for all injectable peptides.
How can I tell if my tirzepatide has gone bad?
Visible signs include cloudiness, particles, color changes, or unusual consistency. However, significant degradation can occur without any visible changes. If you suspect a storage breach, the safest approach is to obtain a fresh supply from your pharmacy rather than risk injecting compromised medication.
Is it safe to use tirzepatide that was left out overnight?
If the ambient temperature stayed below 86 degrees Fahrenheit (30 degrees Celsius) and the solution appears clear and colorless, one overnight at room temperature falls well within the 21-day allowance for brand-name products. For compounded tirzepatide, contact your pharmacy. Understanding room temperature peptide stability helps you assess these situations confidently.
Do I need a special cooler for traveling with tirzepatide?
For trips under 21 days with brand-name products, a basic insulated pouch with a cool pack is sufficient as long as temperatures stay below 86 degrees Fahrenheit. For longer trips or compounded products requiring constant refrigeration, invest in a medical-grade travel cooler with active cooling, either evaporative or USB-powered. Check our injection pen guide for additional travel preparation advice.
Can I ship tirzepatide to myself at a travel destination?
Yes, many pharmacies offer shipping to alternate addresses. Ensure the shipping includes cold chain packaging (insulated box with cold packs) and arrange for the package to be refrigerated upon arrival. Do not ship to a location where the package might sit unrefrigerated on a doorstep in heat.
How long does tirzepatide last in the fridge once opened?
Multi-dose vials can be used for up to 28 days after first puncture, as long as they remain refrigerated. Single-dose pens are designed for one-time use and should not be stored after use. For a deeper look at this topic, read our complete tirzepatide fridge storage guide.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based storage guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. Join here.