Feb 18, 2026

You have been told tirzepatide speeds up your metabolism. The internet says it. Clinic ads repeat it. Even some healthcare providers imply it when describing how tirzepatide works for weight loss. But the clinical data tells a different story, one that is more nuanced, more interesting, and honestly more useful than a simple yes-or-no answer.
Here is what the research actually shows. Tirzepatide does not raise your basal metabolic rate the way a stimulant would. It does not make your body burn more calories at rest in the traditional sense. But it does something arguably more powerful. It shifts how your body burns fuel, increasing fat oxidation, activating brown adipose tissue, improving insulin sensitivity through mechanisms that operate independently of weight loss, and fundamentally reshaping the metabolic environment at the cellular level.
That distinction matters. It matters for anyone trying to understand why tirzepatide produces weight loss results that outperform nearly every other pharmaceutical intervention in history. It matters for researchers studying the tirzepatide dosing protocols that produce the best outcomes. And it matters for the growing number of people who want to know whether this dual GIP/GLP-1 receptor agonist is doing something fundamentally different to their metabolic machinery.
This guide breaks down every metabolic mechanism tirzepatide engages, from the cellular pathways in brown adipose tissue to the clinical trial data on energy expenditure, fat oxidation, insulin sensitivity, and adiponectin levels. No vague claims. No oversimplified answers. Just what the science says, what it means for you, and how to use that knowledge to optimize results.
The short answer: tirzepatide changes metabolism without raising metabolic rate
Before diving into mechanisms and studies, the headline finding needs to be clear. A phase 1 clinical trial published in Cell Metabolism examined whether tirzepatide affects metabolic adaptation during weight loss. Researchers placed 55 participants with obesity into metabolic chambers and measured sleeping metabolic rate, 24-hour energy expenditure, and respiratory quotient at baseline and after 18 weeks of treatment.
The result? Tirzepatide did not prevent the normal decrease in metabolic rate that comes with weight loss. When you lose weight, your body burns fewer calories at rest. That is metabolic adaptation, and tirzepatide did not change it.
But here is where it gets interesting.
Tirzepatide significantly decreased the respiratory quotient, a measurement that indicates what type of fuel your body is burning. A lower respiratory quotient means greater fat oxidation. Your body shifts from burning a mix of carbohydrates and fats to preferentially burning fat. The participants on tirzepatide were not burning more total calories, but they were burning a dramatically higher proportion of those calories from stored fat.
That is a fundamentally different kind of metabolic change. And when you combine it with the appetite suppression, the appetite-reducing effects that cut caloric intake by over 500 calories per day in some studies, and the improvements in how the body handles glucose and lipids, the picture of tirzepatide as a metabolic agent becomes far more complete than a simple "does it speed up metabolism" question can capture.
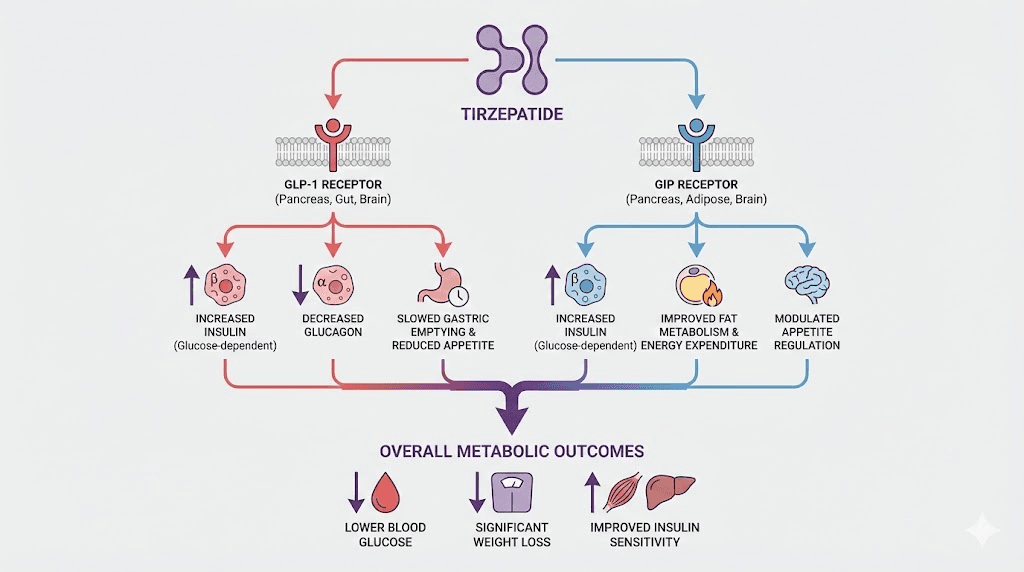
How tirzepatide works at the receptor level
Understanding tirzepatide requires understanding its dual mechanism. Unlike semaglutide, which only activates GLP-1 receptors, tirzepatide activates both GIP (glucose-dependent insulinotropic polypeptide) receptors and GLP-1 (glucagon-like peptide-1) receptors. This dual agonism is not just a marketing distinction. It produces measurably different metabolic effects.
The GLP-1 receptor pathway
GLP-1 receptor activation is responsible for many of the effects people associate with tirzepatide appetite suppression. When GLP-1 receptors in the brain are stimulated, appetite decreases. Food intake drops. Gastric emptying slows, meaning food stays in the stomach longer. These are the same pathways that make GLP-1 medications like Ozempic effective for weight loss.
But the metabolic effects of GLP-1 activation go beyond appetite. GLP-1 receptor stimulation enhances glucose-dependent insulin secretion from pancreatic beta cells. It suppresses glucagon release, which helps prevent blood sugar spikes. And it improves the overall insulin signaling environment that determines how efficiently your body processes nutrients.
The GIP receptor pathway: the metabolic differentiator
This is where tirzepatide separates itself from pure GLP-1 agonists. GIP receptor activation in adipose tissue produces metabolic effects that do not occur with GLP-1 stimulation alone.
Research published in Cell Metabolism demonstrated that tirzepatide modulates adipocyte nutrient metabolism through long-acting activation of the GIP receptor. Specifically, GIP receptor stimulation in fat cells triggers the adenylyl cyclase-cAMP-protein kinase A (PKA) pathway. This cascade directly stimulates glucose transport into adipocytes, lipid metabolism, and amino acid processing.
In practical terms, GIP receptor activation tells fat cells to process nutrients differently. It enhances glucose uptake. It increases lipid oxidation. And critically, it boosts the production of adiponectin, a hormone that improves insulin sensitivity throughout the entire body. These are not small, incremental effects. In SURPASS-2 clinical trial data, tirzepatide increased adiponectin levels by 12-26% depending on dose, a change that cascades through multiple metabolic systems.
The dual activation of both receptors creates synergistic metabolic effects. GLP-1 handles appetite and glucose regulation from the brain and pancreas. GIP handles fat tissue metabolism and insulin sensitization from the periphery. Together, they produce metabolic changes that neither pathway achieves alone.
Fat oxidation: the metabolic shift that matters most
When people ask "does tirzepatide increase metabolism," what they usually want to know is whether the drug helps them burn more fat. The answer is yes, but not through the mechanism most people assume.
What fat oxidation actually means
Your body constantly switches between burning carbohydrates and burning fat for fuel. The ratio depends on multiple factors, including insulin levels, food intake, exercise status, and hormonal signaling. Fat oxidation is the process of breaking down stored triglycerides into fatty acids and glycerol, then processing those fatty acids through beta-oxidation and the TCA cycle to produce ATP, the energy currency your cells run on.
Higher fat oxidation means your body is preferentially using fat stores for energy rather than relying on glucose. This is metabolically significant because it directly reduces adipose tissue, the stored fat that drives obesity-related health complications.
The clinical evidence for increased fat oxidation
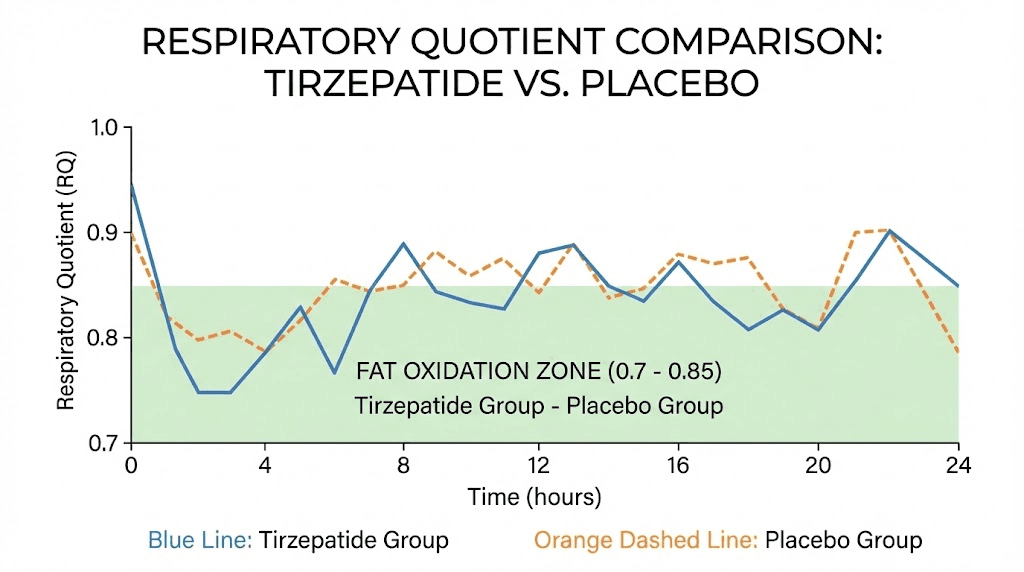
The Cell Metabolism study measured respiratory quotient (RQ) in participants using metabolic chambers. RQ values range from 0.7 (pure fat burning) to 1.0 (pure carbohydrate burning). Tirzepatide significantly lowered RQ compared to placebo, confirming that participants were oxidizing substantially more fat.
This happened even though total energy expenditure was not different between groups. Think about what that means. Two groups of people are burning roughly the same number of calories, but the tirzepatide group is burning a much larger percentage of those calories from fat. Over weeks and months, that preferential fat burning compounds into significant differences in body composition.
The tirzepatide group in this study lost 16.7 kg on average, compared to 8.3 kg in the placebo group (both groups were on caloric restriction). The difference, 8.4 kg more weight loss, came not from burning more total energy but from burning more fat specifically. Understanding how tirzepatide affects body composition over time requires grasping this distinction.
How tirzepatide increases fat oxidation
Multiple mechanisms contribute to the increased fat oxidation observed with tirzepatide.
First, GIP receptor activation in adipose tissue directly stimulates lipolysis, the breakdown of stored fat into free fatty acids. Research shows that tirzepatide enhances cAMP-dependent modulation of lipid metabolism in both mouse and human adipocytes. More fat gets released from storage and made available for burning.
Second, tirzepatide improves insulin sensitivity, which paradoxically enhances fat oxidation. When insulin resistance is high, fat storage is favored over fat burning. By improving insulin signaling, tirzepatide creates a metabolic environment where the body can more easily access and burn stored fat. The side effect profile of tirzepatide reflects these metabolic changes, with many users reporting changes in appetite, energy, and body composition that suggest enhanced fat metabolism.
Third, the appetite suppression itself contributes indirectly. When caloric intake drops, the body must rely more heavily on stored fuel. Combined with the direct fat-mobilizing effects of GIP receptor activation, this creates a powerful fat-burning environment that accelerates fat loss beyond what caloric restriction alone would achieve.
Brown adipose tissue activation: tirzepatide thermogenic effects
One of the most exciting areas of tirzepatide metabolism research involves brown adipose tissue (BAT). Unlike white fat, which stores energy, brown fat burns energy to produce heat. It is the body closest thing to a natural metabolic accelerator, and tirzepatide appears to significantly enhance its activity.
What brown adipose tissue does
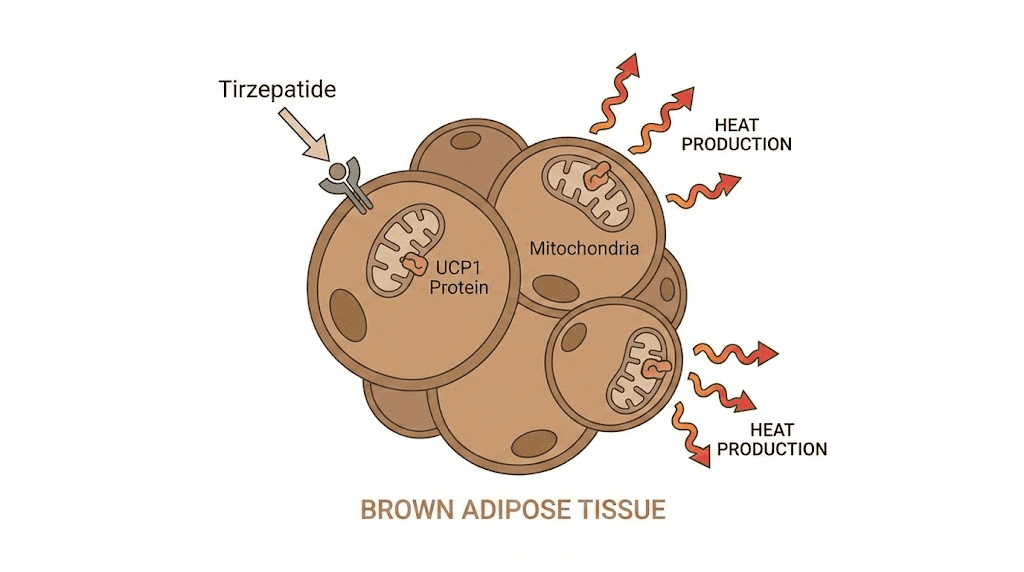
Brown fat is packed with mitochondria, the cellular powerhouses that produce energy. These mitochondria contain a special protein called UCP1 (uncoupling protein 1) that allows them to convert energy directly into heat rather than ATP. When brown fat is active, it literally burns calories to maintain body temperature. This is called non-shivering thermogenesis, and it represents genuine metabolic energy expenditure beyond what shows up as basal metabolic rate in most measurements.
Adults have relatively small amounts of brown fat compared to infants, but the amount they do have can be metabolically significant. Active brown fat burns between 50-100 extra calories per day in some estimates, and it preferentially burns fatty acids as fuel. For anyone interested in fat-burning peptides and compounds, brown fat activation represents one of the most promising mechanisms available.
How tirzepatide activates brown fat
A landmark study published in Molecular Metabolism demonstrated that chronic tirzepatide treatment induces a thermogenic-like amino acid signature in brown adipose tissue. Here is what researchers found.
Tirzepatide treatment significantly upregulated UCP1, the key thermogenic protein in brown fat. It also increased Dio2 and Ppargc1a, two well-established regulators of thermogenesis. Additionally, tirzepatide boosted levels of respiratory complex subunits SDHB (Complex II) and COXI (Complex IV), indicating enhanced mitochondrial oxidative capacity.
In simpler terms, tirzepatide makes brown fat cells better equipped to burn calories. More UCP1 means more heat production. More mitochondrial complex proteins means more capacity for energy metabolism. The brown fat is not just present. It is more active, more capable, and burning more fuel.
The BCAA connection
One of the most specific findings involves branched-chain amino acids (BCAAs). Elevated blood levels of BCAAs, specifically leucine, isoleucine, and valine, are strongly associated with insulin resistance and metabolic disease. Tirzepatide treatment was shown to increase BCAA catabolic gene expression specifically in brown adipose tissue.
This means brown fat under tirzepatide treatment actively pulls BCAAs from the bloodstream and breaks them down. The downstream metabolites, including glutamate, alanine, and 3-hydroxyisobutyric acid, were all elevated in BAT after tirzepatide treatment. This BCAA clearance is metabolically significant because reducing circulating BCAAs improves systemic insulin sensitivity.
The study also found that tirzepatide increased TCA cycle intermediates in brown fat, suggesting enhanced production of both oxidative and anaplerotic substrates. The brown fat is not just making heat. It is actively metabolizing amino acids, lipids, and glucose at an accelerated rate, functioning as a metabolic sink that helps clear problematic metabolites from the bloodstream. For researchers tracking precise tirzepatide dosing, understanding these dose-dependent BAT effects adds another dimension to protocol optimization.
The TABFAT trial: ongoing research
The scientific community considers brown fat activation so important that a dedicated clinical trial, the Tirzepatide Brown and Beige Adipose Tissue Activation (TABFAT) trial, has been designed to specifically measure these effects in humans. This randomized, placebo-controlled study will provide definitive human data on how tirzepatide changes brown and beige fat activity, filling a critical gap between the robust animal data and clinical outcomes.
Insulin sensitivity: the weight-independent metabolic improvement
Perhaps the most clinically significant metabolic effect of tirzepatide is its improvement of insulin sensitivity, and specifically, the portion of that improvement that occurs independently of weight loss.
Why insulin sensitivity matters for metabolism
Insulin is the master metabolic hormone. When cells respond properly to insulin (high sensitivity), glucose gets efficiently transported into cells for energy, fat storage and release are properly regulated, and metabolic processes run smoothly. When insulin sensitivity declines (insulin resistance), the body compensates by producing more insulin, fat storage increases, glucose metabolism deteriorates, and a cascade of metabolic problems follows.
Insulin resistance is not just a diabetes concern. It fundamentally changes how the body handles every calorie consumed. In an insulin-resistant state, the body favors fat storage over fat burning, struggles to efficiently use glucose, and creates an inflammatory environment that further worsens metabolic function. Improving insulin sensitivity reverses all of these problems simultaneously.
Tirzepatide insulin sensitivity data
Clinical trial data from the SURPASS program demonstrated that tirzepatide improves insulin sensitivity beyond what weight loss alone explains. A study published in the Journal of Clinical Endocrinology and Metabolism found that tirzepatide, as a dual GIP/GLP-1 receptor agonist, improved both beta-cell function markers and insulin sensitivity compared to the selective GLP-1 receptor agonist dulaglutide.
Even more striking, in the SURPASS-2 trial, tirzepatide produced significantly greater reductions in HOMA2-IR (a measure of insulin resistance), fasting insulin, and C-peptide concentrations compared to semaglutide at week 40. The weight-loss effect of tirzepatide accounted for only approximately 70% of the improvement in insulin sensitivity. The remaining 30% came from mechanisms independent of weight loss entirely.
This is a critical finding. It means tirzepatide is not just helping people lose weight, which secondarily improves metabolism. It is directly improving metabolic function through pathways that operate whether or not weight is lost. For anyone comparing tirzepatide versus semaglutide, this weight-independent insulin sensitization represents a meaningful pharmacological difference.
The GIP-mediated insulin sensitization mechanism
Researchers have identified GIP receptor activation in adipose tissue as the primary driver of weight-independent insulin sensitization. When tirzepatide stimulates GIP receptors on fat cells, several things happen.
Adiponectin production increases. Adiponectin is an adipokine (a signaling molecule produced by fat tissue) that directly improves insulin sensitivity. It enhances Glut4-mediated glucose uptake in white adipocytes, reduces visceral fat deposits, and decreases hepatic lipid accumulation. The 12-26% increase in adiponectin seen with tirzepatide treatment cascades through the entire metabolic system.
Lipid partitioning improves. GIP receptor activation changes how fat cells store and release lipids, promoting healthier fat distribution and reducing the harmful visceral fat that drives metabolic disease. Circulating triglycerides decrease. HDL cholesterol increases. The lipid profile shifts toward a pattern associated with metabolic health.
BCAA clearance accelerates. As discussed in the brown fat section, tirzepatide promotes breakdown of circulating BCAAs, which are independently associated with insulin resistance. Reducing BCAA levels removes one of the metabolic signals that perpetuates insulin resistance.
These effects combine to create a metabolic environment that is fundamentally more efficient at processing nutrients, storing energy appropriately, and maintaining glucose homeostasis. The metabolism has not "sped up" in the traditional sense. It has become better organized, more efficient, and less wasteful. People tracking their tirzepatide dosage progression often notice improvements in blood glucose markers before significant weight loss occurs, reflecting these weight-independent metabolic improvements.
Energy expenditure: what the metabolic chamber studies show
For those wanting hard numbers on whether tirzepatide increases total calorie burning, the metabolic chamber data provides the clearest answer.
How metabolic chamber studies work
Metabolic chambers are sealed rooms where researchers can precisely measure every calorie a person burns over 24 hours. Sensors track oxygen consumption and carbon dioxide production, which allows calculation of both total energy expenditure and the ratio of fat to carbohydrate being burned. These are considered the gold standard for measuring metabolic rate because they capture everything, from sleeping metabolism to the thermic effect of food to fidgeting.
The key findings
The Pennington Biomedical Research Center study placed participants in metabolic chambers at baseline and again after 18 weeks of tirzepatide treatment (with matched caloric restriction in the placebo group). Here is what the data showed.
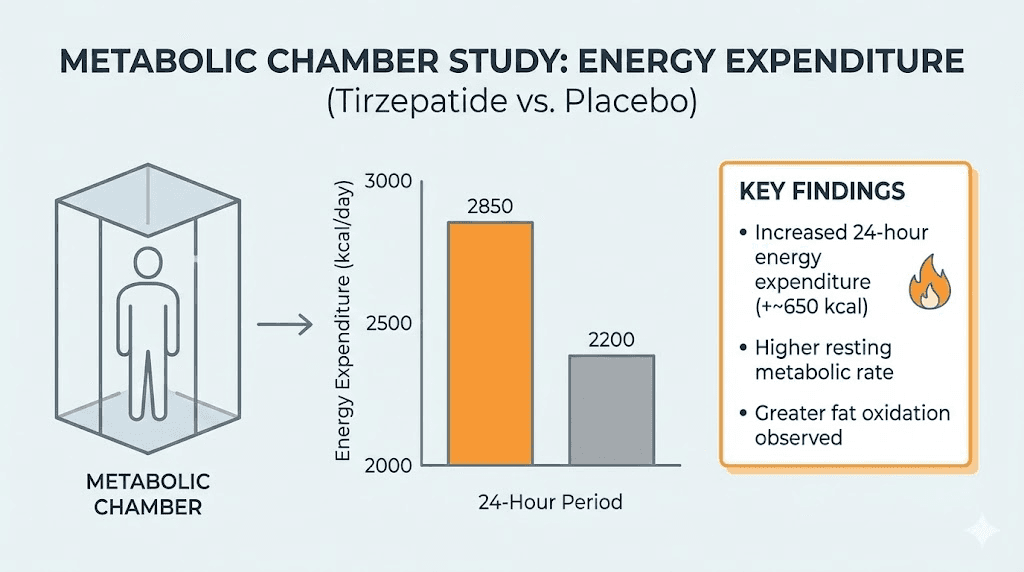
Sleeping metabolic rate: Decreased in both groups proportionally to weight lost. No significant difference between tirzepatide and placebo after adjusting for changes in body weight and body composition. Tirzepatide did not prevent the normal metabolic slowdown that accompanies weight loss.
24-hour energy expenditure: Also decreased in both groups proportionally to weight lost. Again, no significant difference between groups after appropriate adjustments. The total number of calories burned per day was not meaningfully different.
Respiratory quotient: Significantly lower in the tirzepatide group. This is the fat oxidation finding discussed earlier. The body burns the same total energy but draws a larger proportion from fat stores.
Food intake: During an ad libitum test meal (eat as much as you want), the tirzepatide group consumed significantly fewer calories. Appetite was measurably suppressed.
The animal vs. human discrepancy
Interestingly, animal studies told a different story. In calorie-restricted obese mice, chronic tirzepatide treatment reduced the drop in energy expenditure that occurred in control mice. In other words, tirzepatide did attenuate metabolic adaptation in mice. It prevented the metabolic slowdown.
Why the difference? Several possibilities exist. Mouse brown fat is proportionally much larger and more metabolically active than human brown fat. The doses used in animal studies may produce different receptor activation patterns. And the duration and degree of caloric restriction differed between the animal and human protocols.
The TABFAT trial currently underway may help resolve this discrepancy by directly measuring brown and beige fat activation in humans at doses and durations more comparable to the animal studies. For now, the honest answer is that tirzepatide does not appear to increase total energy expenditure in humans based on the best available data, but it meaningfully alters the composition of that expenditure toward fat burning.
Why metabolic adaptation still matters
One concern with any weight loss approach is metabolic adaptation, the body reduction in calorie burning as weight decreases. This is what makes maintaining weight loss so difficult. Your body literally becomes more efficient, burning fewer calories to perform the same functions.
While tirzepatide does not prevent metabolic adaptation, the clinical weight loss results suggest this adaptation is not a significant barrier to its effectiveness. SURMOUNT trial participants achieved average weight reductions of 19.5% (10 mg dose) and 20.9% (15 mg dose) over 72 weeks. These are extraordinary results that suggest tirzepatide weight loss mechanisms, primarily appetite suppression and enhanced fat oxidation, are powerful enough to overcome the metabolic adaptation that normally limits weight loss. People wondering why they are not losing weight on tirzepatide should understand that metabolic adaptation is a normal physiological process, not a sign that the medication has stopped working.
Tirzepatide versus semaglutide: metabolic differences
Since many people choose between tirzepatide and semaglutide for weight management, understanding the metabolic differences matters. Both medications reduce appetite and promote weight loss. But the metabolic mechanisms differ in ways that may be clinically meaningful.
The GIP receptor advantage
Semaglutide activates only GLP-1 receptors. Tirzepatide activates both GLP-1 and GIP receptors. This difference is not trivial. The GIP receptor component adds metabolic effects that semaglutide simply cannot produce.
GIP receptor activation in adipose tissue drives the insulin-sensitizing effects discussed above, the adiponectin increase, the BCAA clearance, the lipid partitioning improvements. It also drives the brown fat activation and thermogenic amino acid signature changes. These are metabolic effects unique to compounds that activate the GIP receptor.
In head-to-head data from SURPASS-2, tirzepatide produced greater improvements in fasting triglycerides, HDL cholesterol, and body mass index compared to semaglutide. The dosage comparison between tirzepatide and semaglutide shows different escalation patterns, but the metabolic outcomes consistently favor tirzepatide at equipotent doses.
Weight loss magnitude
The metabolic differences translate into weight loss differences. In clinical trials, tirzepatide 15 mg produced approximately 20-22% body weight reduction, compared to approximately 15-17% with semaglutide 2.4 mg. That additional 5-7% weight loss represents the contribution of the GIP-mediated metabolic pathways on top of the shared GLP-1 effects.
For people evaluating the comparative side effect profiles, the metabolic differences may help explain why some individuals respond better to one medication than the other. If someone has significant insulin resistance, the additional insulin-sensitizing effects of tirzepatide GIP pathway may provide more benefit. If appetite suppression is the primary need, the differences may be smaller.
Metabolic markers comparison
The SURPASS-2 trial directly compared tirzepatide and semaglutide on metabolic markers. Here is how they stacked up.
HbA1c reduction: Tirzepatide 15 mg reduced HbA1c by 2.46%, compared to 1.86% with semaglutide 1 mg. Greater glucose metabolism improvement.
Fasting insulin: Tirzepatide produced greater reductions in fasting insulin at all doses, indicating more robust insulin sensitivity improvement.
HOMA2-IR: Tirzepatide showed significantly greater reductions in this insulin resistance marker.
Triglycerides and lipids: Greater numerical improvements in fasting triglycerides and HDL cholesterol with tirzepatide.
These metabolic advantages persist even after accounting for the greater weight loss. The dual receptor mechanism produces metabolic changes beyond what weight reduction alone explains. Understanding these differences helps people make informed decisions when considering switching between medications or when working with their healthcare provider to choose the right GLP-1 medication.
Adiponectin: the hidden metabolic hormone boost
Most discussions about tirzepatide and metabolism overlook adiponectin entirely. That is a mistake.
What adiponectin does
Adiponectin is a protein hormone produced by fat cells that acts as a metabolic regulator throughout the body. Despite being produced by fat tissue, adiponectin levels actually decrease as body fat increases, particularly visceral fat. This creates a paradox where the people who most need adiponectin metabolic benefits have the lowest levels.
Adiponectin functions include enhancing glucose uptake in muscle cells, increasing fatty acid oxidation in both muscle and liver, reducing hepatic glucose production, decreasing inflammation in blood vessels, and improving overall insulin sensitivity. It is essentially a metabolic optimization hormone, and higher levels are consistently associated with better metabolic health, lower diabetes risk, and reduced cardiovascular disease.
Tirzepatide adiponectin data
In the SURPASS clinical trials, tirzepatide treatment increased circulating adiponectin levels by 12-26% depending on the dose. This is a substantial increase. The 5 mg dose produced approximately 12% elevation. The 10 mg dose pushed it higher. And the 15 mg dose achieved the full 26% increase.
This adiponectin increase has cascading metabolic effects. More adiponectin means better glucose handling, more fat oxidation, less inflammation, and improved insulin sensitivity. It is one of the mechanisms through which tirzepatide achieves metabolic improvements that exceed what weight loss alone would predict.
The adiponectin boost also helps explain why some metabolic markers improve early in tirzepatide treatment, before significant weight loss has occurred. Fat cells respond to GIP receptor activation quickly, increasing adiponectin production within the first weeks of treatment. This early metabolic improvement is one reason people sometimes notice changes in energy levels, blood sugar stability, and overall well-being before the scale shows dramatic weight loss. For those interested in maximizing metabolic outcomes, supplementation strategies alongside tirzepatide may further support these beneficial hormonal changes.
Glucose metabolism improvements
The metabolic effects of tirzepatide on glucose processing deserve dedicated attention, especially because glucose metabolism and energy metabolism are deeply intertwined.
How tirzepatide changes glucose handling
Tirzepatide affects glucose metabolism through multiple simultaneous pathways. At the pancreatic level, both GLP-1 and GIP receptor activation stimulate glucose-dependent insulin secretion. This means the pancreas releases more insulin when blood sugar is elevated but does not over-produce insulin when blood sugar is normal, reducing hypoglycemia risk.
At the cellular level, GIP receptor activation triggers the adenylyl cyclase-cAMP-PKA pathway in adipocytes, directly stimulating glucose transport into fat cells. Acute treatment with insulin, GIP, and tirzepatide independently increased glucose transport into adipocytes in dose-response studies. This means tirzepatide helps clear glucose from the bloodstream by making fat cells more willing to absorb it.
At the liver level, GLP-1 receptor activation suppresses glucagon release. Glucagon normally signals the liver to produce and release glucose. By suppressing this signal, tirzepatide reduces hepatic glucose output, helping keep blood sugar stable between meals.
The metabolic significance of better glucose control
Better glucose control is not just a diabetes concern. It directly affects energy levels, fat storage, hunger, and metabolic efficiency. When blood sugar spikes and crashes repeatedly (as happens with insulin resistance), the body responds by storing excess glucose as fat, triggering hunger signals, and creating an inflammatory environment that worsens metabolic function.
Stable blood sugar, facilitated by tirzepatide improved glucose metabolism, means more consistent energy throughout the day, reduced cravings for high-carbohydrate foods, less fat storage from glucose overflow, and a more stable hormonal environment for metabolic processes. People who monitor how quickly tirzepatide begins working often notice blood sugar improvements within the first week, well before meaningful weight loss begins.
Clinical outcomes: HbA1c and beyond
The SURPASS clinical trial program demonstrated HbA1c reductions of 1.24-2.58% with tirzepatide, numbers unprecedented for a single glucose-lowering agent. In SURPASS-2, up to 92% of participants achieved HbA1c below 5.7% (the normal range) with the highest tirzepatide dose.
But the metabolic improvements extend beyond HbA1c. Fasting glucose, postprandial glucose, insulin resistance indices, and lipid profiles all improved. These are not isolated glucose effects. They represent a comprehensive metabolic recalibration. The body becomes more efficient at processing every macronutrient, not just glucose.
Lipid metabolism: how tirzepatide changes fat processing
Beyond burning stored fat, tirzepatide fundamentally changes how the body processes dietary and circulating lipids.
Triglyceride reduction
Elevated triglycerides are a hallmark of metabolic dysfunction. They indicate the body is struggling to process dietary fats efficiently. In clinical trials, tirzepatide produced significant reductions in fasting triglycerides, with greater improvements than semaglutide at comparable effectiveness levels.
Lower triglycerides mean less circulating fat available for storage, reduced cardiovascular risk, and improved metabolic efficiency. When the body processes dietary fat more effectively, less ends up in problematic storage locations like visceral fat depots and liver tissue.
HDL cholesterol improvement
Tirzepatide consistently increased HDL ("good") cholesterol in clinical trials. HDL particles help transport excess cholesterol away from tissues for processing and elimination. Higher HDL levels are associated with better cardiovascular health and improved metabolic function. The HDL increase seen with tirzepatide likely reflects improved reverse cholesterol transport, a metabolic process that is suppressed when insulin resistance is high.
Adipose tissue lipid partitioning
Research shows that tirzepatide changes how fat tissue stores and releases lipids. Through GIP receptor activation, tirzepatide promotes healthier lipid partitioning. This means fat is distributed more favorably (less visceral, more subcutaneous), fat cells respond better to metabolic signals, and the inflammatory profile of adipose tissue improves.
Visceral fat, the type that accumulates around organs, is particularly metabolically harmful. It produces inflammatory cytokines, contributes to insulin resistance, and disrupts liver function. Tirzepatide preferential reduction of visceral fat represents a metabolic improvement that goes far beyond simple weight loss. Two people could lose the same number of pounds, but the one losing more visceral fat achieves substantially better metabolic outcomes. Understanding visceral fat reduction strategies adds context to why tirzepatide metabolic effects matter clinically.
Practical implications: optimizing metabolism while on tirzepatide
Understanding the science is valuable, but applying it practically is what produces results. Here is how the metabolic research translates into actionable strategies for people using tirzepatide.
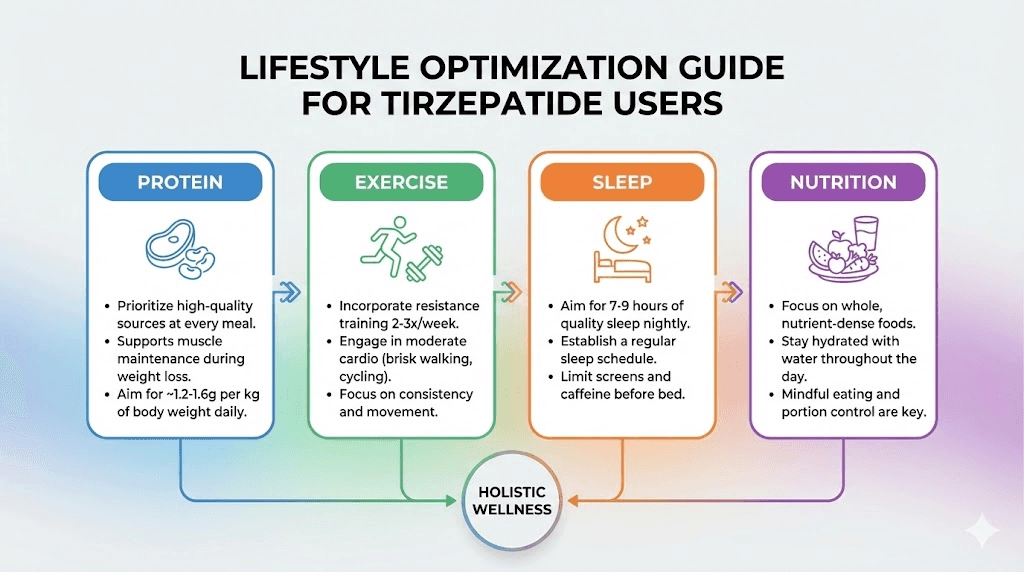
Protein intake and muscle preservation
One of the biggest concerns with significant weight loss is muscle loss. Muscle tissue is metabolically active, burning more calories at rest than fat tissue. Losing muscle during weight loss reduces basal metabolic rate, creating a vicious cycle where the body burns fewer calories and regains weight more easily.
Research supports consuming 1.2-1.6 grams of protein per kilogram of body weight daily during tirzepatide treatment to preserve lean mass. Higher protein intake also increases the thermic effect of food (the calories burned processing nutrients), partially offsetting the metabolic adaptation that comes with weight loss. People following a tirzepatide diet plan should prioritize protein at every meal, even when appetite is significantly reduced.
Resistance training for metabolic support
Exercise does not just burn calories during the activity. Resistance training builds and maintains muscle mass, which keeps basal metabolic rate higher throughout weight loss. For tirzepatide users specifically, resistance training may enhance the fat oxidation effects already provided by the medication while preserving the metabolically valuable muscle tissue.
The combination is synergistic. Tirzepatide shifts fuel utilization toward fat burning. Resistance training provides a demand signal that protects muscle. Together, they promote body recomposition, losing fat while maintaining or even building muscle, which is the optimal metabolic outcome. Finding the optimal timing for GLP-1 medications may also help coordinate exercise and medication effects.
Nutrition strategies that support metabolic health
The metabolic environment tirzepatide creates responds well to specific nutritional strategies.
Adequate fiber intake supports the gut microbiome changes associated with GLP-1 receptor agonism and helps maintain stable blood sugar. People following advice on what to eat on tirzepatide should include diverse fiber sources.
Healthy fats support the lipid metabolism improvements tirzepatide provides. Omega-3 fatty acids, monounsaturated fats from olive oil and avocado, and adequate essential fatty acid intake all complement the medication metabolic effects.
Micronutrient sufficiency becomes critical when food intake decreases. Tirzepatide appetite suppression can lead to insufficient intake of vitamins and minerals that support metabolic function, including B vitamins, magnesium, zinc, and iron. Targeted supplementation may be appropriate during active weight loss phases.
Avoiding processed foods matters more on tirzepatide because the body improved insulin sensitivity means nutrients are processed more efficiently. High-quality whole foods provide the substrates needed for the enhanced metabolic pathways tirzepatide activates. The foods to avoid on tirzepatide are the same ones that would undermine any metabolic improvement: highly processed, high-sugar, and nutrient-poor options.
Sleep and stress management
Tirzepatide metabolic benefits can be undermined by poor sleep and chronic stress. Both sleep deprivation and elevated cortisol increase insulin resistance, promote visceral fat storage, and reduce the adiponectin levels that tirzepatide works to increase. Prioritizing 7-9 hours of quality sleep and managing stress through evidence-based methods (regular exercise, adequate social connection, stress reduction techniques) supports the metabolic improvements the medication provides.
Some users report that tirzepatide affects their energy levels and fatigue patterns, particularly during dose escalation. Understanding these effects and adjusting sleep patterns accordingly helps maintain the metabolic benefits throughout treatment.
Metabolic adaptation during weight loss: what to expect
Metabolic adaptation is the body natural response to weight loss. Understanding it prevents unrealistic expectations and helps people stay on track when results inevitably slow.
What metabolic adaptation looks like in practice
As you lose weight on tirzepatide, your body will burn fewer calories at rest. This is not the medication failing. It is basic physics and biology. A smaller body requires less energy to maintain. Additionally, your body may become more metabolically efficient, extracting more energy from the same amount of food.
In practical terms, the rapid weight loss many people experience in the first 12-16 weeks of tirzepatide treatment will naturally slow over time. Weekly weight loss of 2-3 pounds early on may decrease to 0.5-1 pound per week later. This is normal. It does not mean tirzepatide stopped working or that your metabolism is "broken."
How tirzepatide mitigates some adaptation effects
While tirzepatide does not prevent metabolic adaptation entirely, several of its mechanisms partially offset the problem.
The sustained appetite suppression means caloric intake remains low even as metabolic rate decreases. In many weight loss scenarios, hunger increases as the body adapts, driving people to eat more and regain weight. Tirzepatide appetite suppression fights this compensatory hunger, maintaining the caloric deficit needed for continued weight loss.
The enhanced fat oxidation means the body preferentially burns stored fat rather than shifting to other energy sources. During non-medicated weight loss, the body often increases muscle catabolism (breaking down muscle for fuel) as adaptation progresses. Tirzepatide fat oxidation preference may partially protect against this metabolically destructive response.
The insulin sensitization effects maintain efficient nutrient processing even as total metabolism slows. Better insulin sensitivity means the body does not need to produce as much insulin, which reduces the fat-storage signaling that can counteract weight loss efforts.
Strategies for managing weight loss plateaus
Understanding metabolic adaptation helps people respond appropriately to weight loss plateaus on tirzepatide.
Do not panic. A plateau lasting 2-4 weeks is normal and expected. Body weight fluctuates daily based on water retention, digestive contents, hormonal cycles, and other factors that have nothing to do with fat loss. The scale may stall while fat loss continues.
Maintain protein intake. As discussed, protein preserves muscle and keeps metabolic rate higher. During a plateau, increasing protein slightly (to the upper end of the 1.2-1.6 g/kg range) may help.
Adjust activity, not calories. Adding moderate activity, even walking 30 minutes daily, can break through plateaus more sustainably than further restricting food intake. For people exploring the relationship between activity and tirzepatide energy effects, increased movement often improves both metabolic rate and subjective energy levels.
Consider dose optimization. If weight loss has stalled significantly and lifestyle factors are optimized, discussing dose adjustment with a healthcare provider is appropriate. The tirzepatide dose escalation chart provides structured guidelines for this conversation. Some people respond to higher doses with renewed metabolic response, though side effects also increase.
Track body composition, not just weight. If you are exercising and maintaining protein intake, you may be gaining muscle while losing fat. The scale may not change, but your metabolic health is improving. Waist circumference, clothing fit, and body composition measurements tell a more complete story than weight alone.
Common misconceptions about tirzepatide and metabolism
Several popular beliefs about tirzepatide and metabolism are either oversimplified or outright wrong. Correcting these misconceptions helps people make better decisions about their treatment.
Misconception 1: "Tirzepatide speeds up your metabolism"
As covered extensively above, tirzepatide does not increase basal metabolic rate or total energy expenditure in humans based on the best available evidence. What it does is shift fuel utilization toward fat burning, improve metabolic efficiency, and enhance specific metabolic pathways in brown fat and adipose tissue. Calling this "speeding up metabolism" is technically inaccurate, even though the practical effect, burning more fat, is what most people actually want.
Misconception 2: "Tirzepatide only works through appetite suppression"
Appetite suppression is a major mechanism, but it is far from the only one. The GIP-mediated effects on insulin sensitivity, adiponectin production, BCAA clearance, brown fat activation, and lipid metabolism all contribute to outcomes that exceed what appetite suppression alone would achieve. The approximately 30% of insulin sensitivity improvement that is weight-independent proves that tirzepatide has direct metabolic effects beyond simply reducing food intake.
Misconception 3: "Once you stop tirzepatide, your metabolism is ruined"
The SURMOUNT-4 trial examined what happens when tirzepatide is discontinued. Participants who switched from tirzepatide to placebo regained approximately two-thirds of their lost weight over 52 weeks. This is not because tirzepatide "damaged" their metabolism. It is because the physiological drivers of weight regain, increased appetite, loss of enhanced fat oxidation, and return of insulin resistance, reassert themselves when the medication is removed.
Metabolic adaptation during weight loss is a normal physiological response that occurs with any weight loss method, from dieting to surgery. Tirzepatide does not make this worse. The weight regain seen after discontinuation is consistent with what happens when any effective weight loss intervention is stopped. The take-home message is not that tirzepatide damages metabolism but that obesity is a chronic condition that may require ongoing treatment, similar to hypertension or diabetes.
Misconception 4: "Higher doses mean more metabolic benefit"
While higher tirzepatide doses generally produce more weight loss, the relationship between dose and metabolic benefit is not purely linear. The adiponectin increase, for example, scales with dose (12% at 5 mg, up to 26% at 15 mg). But side effects also scale with dose, and gastrointestinal issues at higher doses can impair nutrition absorption and reduce the dietary quality needed to support metabolic health.
The optimal dose is the one that produces meaningful metabolic improvement while remaining tolerable enough to maintain good nutrition and quality of life. For many people, that is not the highest available dose. Working with a healthcare provider to find the right balance based on individual response is critical. The tirzepatide dosage calculator can help model different dose scenarios, but clinical judgment should guide the final decision.
Misconception 5: "Tirzepatide metabolic effects are the same as semaglutide"
As discussed in the comparison section, this is demonstrably false. The GIP receptor component of tirzepatide produces metabolic effects that pure GLP-1 agonists like semaglutide do not achieve. The weight-independent insulin sensitization, enhanced adiponectin production, and brown fat activation patterns differ between the two medications. People considering a conversion from semaglutide to tirzepatide should understand that they are not just switching brands but accessing a different metabolic mechanism.
How tirzepatide compares to other metabolic interventions
Putting tirzepatide metabolic effects in context helps people understand where it fits among other approaches to improving metabolism.
Tirzepatide vs. exercise for metabolic improvement
Exercise and tirzepatide affect metabolism through different but complementary mechanisms. Exercise increases energy expenditure during and after activity, builds metabolically active muscle tissue, improves insulin sensitivity through skeletal muscle adaptations, and enhances mitochondrial function. Tirzepatide enhances fat oxidation, improves insulin sensitivity through adipose tissue pathways, boosts adiponectin, and activates brown fat.
Neither replaces the other. The combination of exercise and tirzepatide produces metabolic improvements greater than either alone. Exercise preserves muscle mass that tirzepatide weight loss might otherwise reduce. Tirzepatide improves the metabolic environment in ways that make exercise more effective. For anyone looking to optimize peptide and weight loss strategies, exercise remains an essential complement.
Tirzepatide vs. caloric restriction alone
Pure caloric restriction reduces metabolic rate proportionally to weight lost and often leads to muscle loss, increased hunger, and eventual weight regain. Tirzepatide combined with caloric restriction produces greater weight loss, better body composition (more fat loss relative to muscle loss), improved metabolic markers beyond what the weight loss itself would predict, and more sustainable appetite control.
The study that compared tirzepatide to placebo with matched caloric restriction found an additional 8.4 kg of weight loss in the tirzepatide group. Same caloric deficit, dramatically different results. That difference represents the value of tirzepatide enhanced fat oxidation, improved insulin sensitivity, and metabolic optimization on top of simple energy restriction.
Tirzepatide vs. other GLP-1 medications
Among injectable GLP-1 medications, tirzepatide produces the greatest metabolic improvements and weight loss. Semaglutide comes next, followed by liraglutide and other older GLP-1 agonists. The dual receptor mechanism of tirzepatide provides metabolic advantages that single-receptor agonists cannot match.
For people interested in the next generation of metabolic medications, triple agonists like retatrutide (GIP/GLP-1/glucagon receptor agonist) are in clinical trials showing even greater metabolic effects. The addition of glucagon receptor agonism to the GIP/GLP-1 combination may produce genuine increases in energy expenditure that tirzepatide alone does not achieve, potentially representing the first true "metabolism booster" in this drug class.
Long-term metabolic health on tirzepatide
The metabolic benefits of tirzepatide extend beyond weight loss into long-term health improvements that affect multiple organ systems.
Cardiovascular metabolic benefits
The improvements in triglycerides, HDL cholesterol, insulin sensitivity, and visceral fat all contribute to reduced cardiovascular risk. These are not just laboratory numbers. They translate into lower rates of heart attack, stroke, and cardiovascular death in populations with metabolic disease. The comprehensive metabolic improvement tirzepatide provides, addressing glucose, lipid, and inflammatory pathways simultaneously, makes it one of the most broadly effective metabolic interventions available.
Liver health
Non-alcoholic fatty liver disease (NAFLD) affects a significant proportion of people with obesity and insulin resistance. Tirzepatide metabolic effects, particularly the reduction in hepatic lipid accumulation driven by improved insulin sensitivity and adiponectin increase, may significantly benefit liver health. Early clinical data suggests improvements in liver fat content with tirzepatide treatment, though dedicated liver outcome trials are still underway.
Inflammatory markers
Chronic low-grade inflammation is both a cause and consequence of metabolic dysfunction. Tirzepatide reduces inflammatory markers through multiple mechanisms: weight loss itself reduces inflammation, improved insulin sensitivity decreases inflammatory signaling, increased adiponectin has direct anti-inflammatory effects, and reduced visceral fat removes a major source of inflammatory cytokines.
This anti-inflammatory effect is metabolically significant because inflammation directly impairs insulin signaling, promotes fat storage, and disrupts the metabolic pathways tirzepatide is trying to improve. By reducing inflammation, tirzepatide creates a positive feedback loop where metabolic improvements beget further metabolic improvements.
Who benefits most from tirzepatide metabolic effects?
Not everyone responds equally to tirzepatide. Understanding who benefits most from the metabolic effects helps set appropriate expectations.
People with significant insulin resistance
The greater the baseline insulin resistance, the more room tirzepatide has to improve metabolic function. People with type 2 diabetes, prediabetes, or metabolic syndrome see the most dramatic metabolic improvements because tirzepatide is addressing active metabolic dysfunction, not just optimizing already-healthy systems.
People with high visceral fat
Visceral fat is metabolically active in harmful ways, producing inflammatory signals and contributing to insulin resistance. Tirzepatide preferential effect on visceral fat means people carrying significant abdominal fat may see disproportionate metabolic benefits. Those interested in targeted belly fat reduction should understand that tirzepatide effects on visceral fat are among the most well-documented.
People who have plateaued on other interventions
If diet and exercise alone have not produced adequate metabolic improvement, tirzepatide adds mechanisms that cannot be achieved through lifestyle changes. The GIP-mediated adipose tissue effects, brown fat activation, and adiponectin increase are pharmacological effects that no amount of exercise or dietary optimization can replicate.
People with poor metabolic markers despite normal weight
"Metabolically obese, normal weight" individuals, people who carry excess visceral fat and have poor metabolic markers despite not appearing overweight, may benefit from tirzepatide metabolic effects even at lower doses. The insulin sensitization and adiponectin improvements address the underlying metabolic dysfunction regardless of overall body weight.
Frequently asked questions
Does tirzepatide increase resting metabolic rate?
No. Clinical trial data from metabolic chamber studies shows that tirzepatide does not increase sleeping metabolic rate or 24-hour energy expenditure compared to placebo after adjusting for weight loss. However, it significantly increases fat oxidation, meaning your body burns more fat without burning more total calories. This shift in fuel utilization drives the substantial fat loss seen in clinical trials. Learn more about how quickly tirzepatide effects begin.
How does tirzepatide affect metabolism differently than semaglutide?
Tirzepatide activates both GIP and GLP-1 receptors, while semaglutide only activates GLP-1 receptors. The additional GIP receptor activation produces weight-independent insulin sensitization, increased adiponectin levels (12-26%), enhanced brown adipose tissue activity, and improved lipid metabolism. These effects contribute to approximately 5-7% greater total weight loss and superior metabolic marker improvements compared to semaglutide. See the full semaglutide versus tirzepatide comparison.
Will tirzepatide permanently change my metabolism?
The metabolic improvements from tirzepatide are largely dependent on continued use. The SURMOUNT-4 trial showed that participants who discontinued tirzepatide regained approximately two-thirds of lost weight over 52 weeks. The metabolic benefits, including improved insulin sensitivity, adiponectin levels, and lipid profiles, also partially reversed. This suggests tirzepatide metabolic effects require ongoing treatment to maintain, similar to medications for blood pressure or cholesterol. Explore how long people typically stay on GLP-1 medications.
Can I boost my metabolism further while on tirzepatide?
Yes. Resistance training preserves muscle mass and maintains a higher metabolic rate during weight loss. Adequate protein intake (1.2-1.6 g/kg/day) supports muscle preservation. Quality sleep and stress management prevent cortisol-driven metabolic impairment. And certain supplements taken alongside tirzepatide may support the enhanced metabolic pathways the medication activates.
Does tirzepatide cause metabolic adaptation?
Tirzepatide does not prevent metabolic adaptation (the natural decrease in metabolic rate that accompanies weight loss), but its mechanisms partially offset the consequences. Sustained appetite suppression prevents the compensatory hunger that normally accompanies adaptation. Enhanced fat oxidation ensures the body preferentially burns stored fat. And improved insulin sensitivity maintains efficient nutrient processing even as total calorie needs decrease. If weight loss has stalled, check our guide on what to do when tirzepatide stops working.
How long does it take for tirzepatide metabolic effects to appear?
Different metabolic effects appear on different timelines. Appetite suppression and reduced food intake begin within the first week. Blood glucose improvements often appear within 2-4 weeks. Insulin sensitivity improvements develop over 4-8 weeks. Significant fat oxidation changes are measurable by 12-18 weeks. And the full metabolic profile, including adiponectin increases, lipid improvements, and brown fat activation, develops over 6-12 months of treatment. Learn more about the complete tirzepatide timeline.
Is the metabolic benefit dose-dependent?
Generally, yes. Higher tirzepatide doses produce greater adiponectin increases (12% at 5 mg vs. 26% at 15 mg), more weight loss, and larger improvements in metabolic markers. However, side effects also increase with dose, and the optimal dose balances metabolic benefit with tolerability. Use our dosage calculator to explore different dose scenarios, but always work with a healthcare provider for final dose decisions.
Does tirzepatide help with metabolic syndrome?
Tirzepatide addresses multiple components of metabolic syndrome simultaneously: it reduces waist circumference (visceral fat), improves fasting glucose and HbA1c, increases HDL cholesterol, decreases triglycerides, and may lower blood pressure through weight loss. It is one of the most comprehensive single-agent treatments for metabolic syndrome available, addressing the underlying insulin resistance that drives the condition rather than treating each component separately.
External resources
Cell Metabolism: Tirzepatide metabolic adaptation and fat oxidation study
PMC: Tirzepatide thermogenic amino acid signature in brown adipose tissue
JCEM: Tirzepatide dual GIP/GLP-1 agonist improves beta-cell function and insulin sensitivity
NEJM: Tirzepatide once weekly for treatment of obesity (SURMOUNT-1)
Cell Metabolism: Tirzepatide modulates adipocyte nutrient metabolism through GIP receptor
For researchers serious about understanding and optimizing their metabolic health during peptide-based protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, detailed protocol databases, and a community of thousands who have navigated these exact questions about metabolism, dosing, and long-term outcomes.
In case I do not see you, good afternoon, good evening, and good night. May your fat oxidation stay elevated, your insulin sensitivity stay sharp, and your metabolic health stay on an upward trajectory.