Feb 25, 2026
That vial of compounded tirzepatide sitting in your refrigerator has a ticking clock. Most people do not realize it. They order a month supply, maybe two, and assume the medication will stay effective until the last drop is gone. Then they notice something strange. The weight loss stalls. The appetite suppression fades. The results that seemed so promising in week one quietly disappear by week six.
The problem is not the medication itself. It is the expiration date.
Compounded tirzepatide operates on a fundamentally different timeline than the brand-name versions sitting on pharmacy shelves. And understanding that timeline, truly understanding it, is the difference between getting full value from every milligram and quietly injecting degraded peptide that does nothing for your body. A single vial of compounded tirzepatide is not cheap. Wasting even 30% of it because you misjudged the expiration date means throwing money away while wondering why your weight loss timeline has stalled.
This guide covers everything you need to know about compounded tirzepatide expiration dates. You will learn what a beyond-use date actually means, how long your vial truly lasts under different conditions, how to spot signs of degradation before you inject, and exactly how to store your medication for maximum potency. Whether you are new to compounded tirzepatide or months into your protocol, the information here will help you protect your investment and your results. SeekPeptides has compiled the most current research, pharmacy guidelines, and practical storage strategies into one comprehensive resource so you never have to guess about your medication stability again.
What is a beyond-use date and why does it matter for compounded tirzepatide
If you have ever looked at the label on your compounded tirzepatide vial, you have probably noticed a date. But that date is not an expiration date in the traditional sense. It is called a beyond-use date, or BUD, and understanding the distinction could save you from using degraded medication or throwing away perfectly good tirzepatide prematurely.
An expiration date appears on FDA-approved medications like Mounjaro or Zepbound. Manufacturers determine these dates through extensive stability testing, often spanning years, that proves the medication maintains at least 90% potency through the labeled date. A beyond-use date works differently. It is assigned by the compounding pharmacy based on USP Chapter 797 guidelines and represents the last date the pharmacy can guarantee the sterility and potency of the preparation.
Think of it this way. A manufacturer has spent millions testing their product under controlled conditions for 24 to 36 months. A compounding pharmacy, working with smaller batches and different equipment, assigns a more conservative date because they have less long-term data to support extended shelf life claims.
The BUD on your compounded tirzepatide is typically calculated from the date the medication was prepared, not the date you received it and not the date you first opened or punctured the vial. This distinction matters enormously. If your pharmacy compounded the medication on January 1st and assigned a 60-day BUD, that medication expires on March 2nd regardless of when it arrived at your door or when you drew your first dose.
For researchers comparing tirzepatide expiration timelines, the BUD system creates a unique challenge. Unlike picking up a branded pen at your local pharmacy, compounded medications require you to actively track dates and plan your dosing around them. The peptide reconstitution calculator can help you determine exact volumes so you do not waste any medication before the BUD arrives.
USP 797, the chapter governing sterile compounding, was significantly updated in 2023. The revised guidelines shifted from categorizing preparations by contamination risk level to a system based on the compounding environment itself. Category 1 preparations, made in segregated compounding areas, receive shorter BUDs. Category 2 preparations, made in ISO-classified cleanrooms with stricter environmental controls, can receive longer BUDs of up to 60 days refrigerated or even longer with stability testing data.
What does this mean for your vial? The pharmacy environment directly influences how long your compounded tirzepatide is considered safe and effective. A state-of-the-art 503B outsourcing facility with full environmental monitoring may assign a 90-day BUD on the same formulation that a smaller 503A pharmacy labels with a 28-day BUD. Same active ingredient. Same concentration. Different expiration dates, all because of the compounding environment and available stability data.
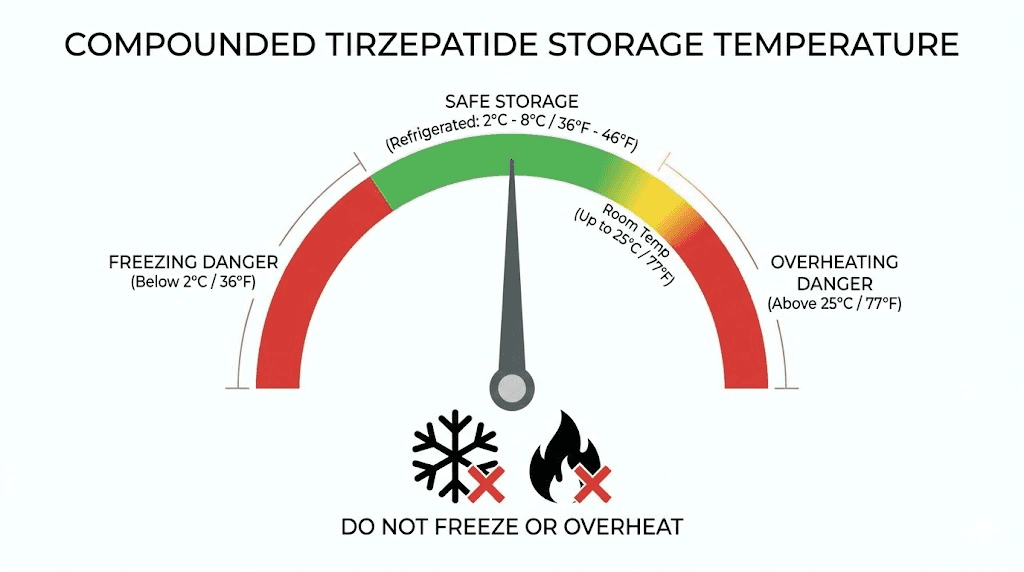
This is why checking tirzepatide refrigeration requirements is not optional. The BUD assumes you are storing the medication exactly as directed. Break the cold chain, and that date becomes meaningless.

How long does compounded tirzepatide last before it expires
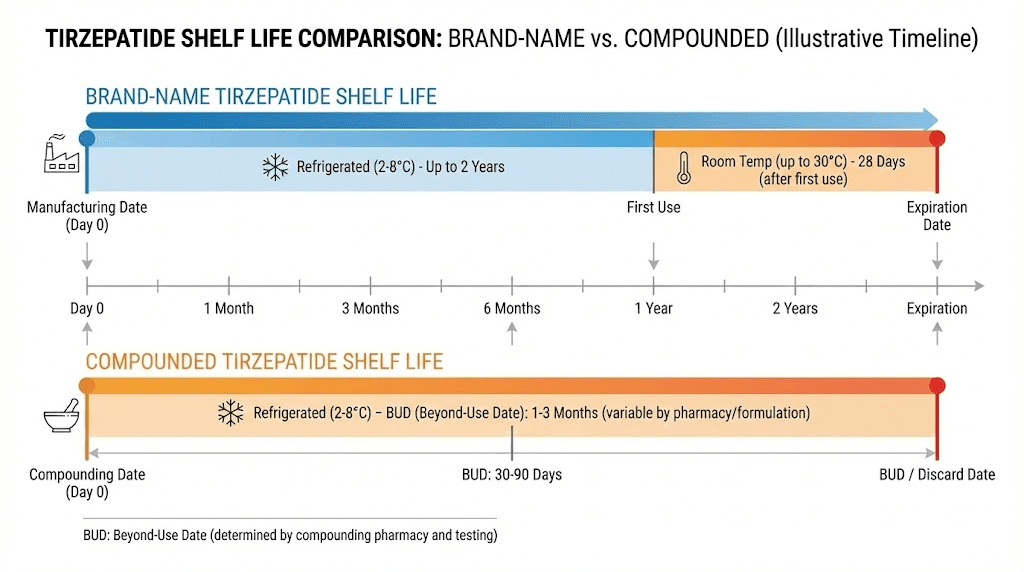
The short answer is 28 to 90 days from the date of compounding. The longer answer involves several variables that can push your specific vial toward either end of that range.
Most compounding pharmacies assign BUDs based on three factors: the sterility of their compounding environment, the results of any stability testing they have conducted, and the default guidelines from USP 797. Without proprietary stability data, the default BUD for a refrigerated sterile preparation is typically 28 days. Pharmacies that invest in third-party stability testing, and many reputable ones do, can extend this to 60 or even 90 days when data supports it.
Unopened refrigerated vials
An unopened vial of compounded tirzepatide stored consistently at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit) will remain stable through its assigned BUD. Some stability studies have found that properly formulated compounded tirzepatide retains over 95% potency for 120 days under consistent refrigeration. However, your pharmacy BUD is the only date you should rely on, because you cannot verify potency without laboratory testing.
The type of formulation also matters. Reconstituted tirzepatide from lyophilized powder typically has a shorter post-reconstitution shelf life than pre-mixed liquid formulations. If you are mixing your own vials using bacteriostatic water, the clock starts ticking faster. Most guidelines suggest using reconstituted peptide solutions within 28 to 30 days, even if the original powder had a longer shelf life.
After first puncture
Once you insert a needle into your compounded tirzepatide vial, you introduce a potential contamination pathway. Even with proper technique, sterile needles, and alcohol-swabbed stoppers, each puncture slightly increases contamination risk. Most pharmacies recommend using multi-dose vials within 28 days of first puncture, and this 28-day window may be shorter than the original BUD on the label.
Here is the critical detail that catches people off guard. If your vial has a 90-day BUD from the compounding date but you first puncture it 70 days after compounding, you do not get another 28 days. The BUD still applies. Your medication expires at the 90-day BUD mark, giving you only 20 days of use after opening, not 28.
This is where understanding your compounded tirzepatide starting dose and planning your ordering schedule becomes essential. You need to calculate how much medication you will use before the BUD arrives and order accordingly. The tirzepatide dosage in units guide can help you map out exactly how many doses each vial contains.
At room temperature
Compounded tirzepatide can survive at room temperature for limited periods. Most pharmacies allow up to 21 days at temperatures not exceeding 30 degrees Celsius (86 degrees Fahrenheit). Some formulations may have shorter room temperature windows depending on their preservative system and base formulation.
But here is what many people miss. Room temperature time is cumulative. Every minute your vial spends outside the refrigerator counts toward that 21-day limit. Drawing your morning dose takes 5 minutes. Your evening dose takes another 5 minutes. After four weeks of twice-daily dosing, you have accumulated roughly 4 to 5 hours of room temperature exposure, which is still well within limits. But leaving the vial on the counter for 3 hours because you forgot to put it back? That adds up quickly.
If your tirzepatide has been out of the fridge for extended periods, the expiration timeline may have shortened significantly. And once the total room temperature exposure exceeds the pharmacy guidelines, the medication should be discarded regardless of the original BUD.
Frozen tirzepatide
Never freeze compounded tirzepatide. Freezing irreversibly damages the peptide structure, breaking the molecular bonds that allow tirzepatide to bind to GIP and GLP-1 receptors. A frozen vial may look identical to an unfrozen one after thawing. The solution might appear clear. The color might seem normal. But the peptide inside is structurally compromised and will not produce the expected therapeutic effects.
If your refrigerator tends to run cold or you store the vial near the back wall where temperatures dip below 2 degrees Celsius, you risk partial freezing. A small thermometer placed near your medication can prevent this expensive mistake.
Compounded tirzepatide vs brand-name tirzepatide expiration dates
The gap between compounded and brand-name tirzepatide expiration dates is enormous. Understanding why helps you plan better and worry less.
Brand-name tirzepatide, sold as Mounjaro for type 2 diabetes and Zepbound for weight management, comes with manufacturer-assigned expiration dates typically 18 to 24 months from the production date. Eli Lilly conducts extensive stability testing under accelerated and real-time conditions to support these dates. The medication arrives in a pre-filled pen with a controlled formulation, tested preservative system, and a manufacturing process that has been validated thousands of times.
Compounded tirzepatide exists in a different world entirely.
Your compounding pharmacy may produce dozens or hundreds of vials per batch rather than millions. The equipment, while regulated, differs from pharmaceutical manufacturing lines. The stability testing, if conducted at all, covers a fraction of the timeframe that Eli Lilly studies. And the formulation itself varies from pharmacy to pharmacy, with different concentrations, preservatives, and added compounds like B12, glycine, or niacinamide.
Here is a direct comparison:
Factor | Brand-name (Mounjaro/Zepbound) | Compounded tirzepatide |
|---|---|---|
Shelf life (unopened, refrigerated) | 18-24 months | 28-90 days (BUD) |
Room temperature window | 21 days (up to 30C/86F) | Up to 21 days (varies by pharmacy) |
After first use | 21 days at room temp, longer refrigerated | 28 days or remaining BUD (whichever is shorter) |
Stability testing | Years of FDA-required data | Varies by pharmacy (28 days default) |
Formulation consistency | Identical across all units | Varies by pharmacy and batch |
Preservative system | Proprietary, extensively tested | Varies (typically bacteriostatic water) |
Delivery format | Pre-filled pen | Multi-dose vial (usually) |
The practical takeaway? If you are using compounded tirzepatide, you need to be significantly more attentive to storage and timing than someone using a branded pen. The margin for error is smaller. The consequences of poor storage are faster. And the cost of wasting a vial, per milligram, can be just as significant even though compounded tirzepatide costs less than brand-name options.
For those weighing their options between different tirzepatide and semaglutide formulations, expiration date differences should factor into your decision. A lower per-unit cost means nothing if you consistently lose 20 to 30% of each vial to expiration. The peptide cost calculator can help you model the true cost per dose when accounting for potential waste from BUD constraints.
Some people also explore alternative delivery methods like oral tirzepatide, tirzepatide drops, or orally disintegrating tablets partly because of storage and expiration concerns. Each format has its own stability profile, and understanding these differences is part of making an informed choice about your protocol.
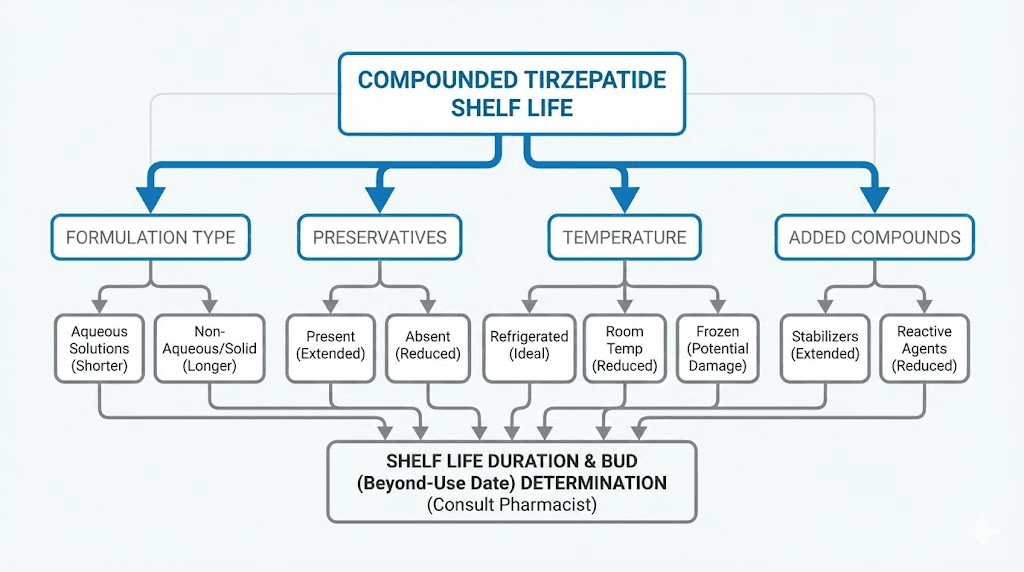
Factors that affect compounded tirzepatide shelf life
Not all compounded tirzepatide vials are created equal. Several variables influence how long your specific medication remains potent, and some of these are within your control while others depend entirely on your pharmacy.
Formulation type
Compounded tirzepatide comes in two primary forms: liquid (pre-mixed) and lyophilized (freeze-dried powder). Pre-mixed liquid formulations are ready to inject straight from the vial. Lyophilized formulations require reconstitution with bacteriostatic water before use.
Lyophilized tirzepatide generally has a longer shelf life before reconstitution because the dry powder is inherently more stable than the peptide in solution. Once you add bacteriostatic water to your tirzepatide, the stability clock accelerates significantly. Most reconstituted peptide solutions should be used within 28 to 30 days when refrigerated properly.
Pre-mixed liquid formulations from compounding pharmacies often contain stabilizers and preservatives designed to extend the shelf life. These may last 60 to 90 days depending on the specific formulation. The trade-off is convenience versus shelf life flexibility.
Added compounds and their impact on stability
Many compounding pharmacies add additional active ingredients to their tirzepatide formulations. Tirzepatide with B12 is one of the most common combinations, designed to help counteract the fatigue and energy dips some people experience on GLP-1 medications. Tirzepatide with glycine and B12 is another popular formulation, where glycine serves as a stabilizer and may also reduce gastrointestinal side effects.
Each added compound changes the stability profile. Vitamin B12 (cyanocobalamin or methylcobalamin) is light-sensitive and can degrade when exposed to UV or fluorescent light. If your compounded tirzepatide contains B12, light protection becomes even more critical for maintaining potency through the BUD. You might notice color changes in your tirzepatide with B12 over time, which can indicate degradation.
Glycine, when used as a stabilizer, can actually help extend shelf life by buffering the pH of the solution and protecting the peptide from degradation. Tirzepatide with glycine formulations may maintain potency slightly longer than formulations without it, though the difference is modest under proper storage conditions.
Preservative system
The preservative in your compounded tirzepatide vial serves a dual purpose: preventing microbial growth and maintaining a stable environment for the peptide. Bacteriostatic water, which contains 0.9% benzyl alcohol, is the most common preservative vehicle. Some pharmacies use alternative preservative systems or additional antimicrobial agents.
The quality and concentration of the preservative directly influence how long the multi-dose vial remains safe to use after first puncture. Insufficient preservative means faster microbial risk. Too much preservative can interact with the peptide and accelerate degradation. Reputable pharmacies conduct preservative effectiveness testing to ensure their formulations maintain antimicrobial protection throughout the assigned BUD.
Storage temperature consistency
Temperature is the single most impactful factor under your control. A vial stored at a rock-steady 4 degrees Celsius will outlast the same formulation subjected to temperature cycling between 2 and 8 degrees multiple times per day. Every time the temperature fluctuates, the peptide structure experiences thermal stress. Over days and weeks, these micro-stresses accumulate and can reduce potency faster than the BUD suggests.
This is why understanding what happens when tirzepatide gets warm matters even for brief exposures. A car interior on a hot day easily reaches 50 to 60 degrees Celsius. Even 2 to 3 hours at these temperatures can cause 10 to 20% potency reduction, and the damage is permanent and cumulative.
Container type and integrity
Most compounded tirzepatide arrives in glass vials with rubber stoppers sealed under aluminum crimps. Glass provides excellent chemical stability and does not interact with the peptide. The rubber stopper, however, can be a weak point. Repeated needle punctures create micro-channels through the stopper that can allow air ingress and potential contamination.
If you are using a vial that requires more than 20 to 25 needle punctures to empty, consider whether the stopper integrity is compromised. Some pharmacies offer smaller vial sizes specifically to minimize puncture-related risks. The tirzepatide syringe dosage guide can help you determine exactly how many draws you need per vial based on your concentration and dose.
How to read your compounded tirzepatide expiration label
Your compounded tirzepatide vial contains critical information on its label. Knowing how to read it properly prevents confusion and ensures you never accidentally use expired medication.
Every compounded medication label must include several key pieces of information, though the exact format varies by pharmacy. Look for these elements:
Beyond-use date (BUD): This is the most important date on the label. It may appear as "BUD," "Exp," "Use By," or "Do Not Use After." Some pharmacies print the date in MM/DD/YYYY format while others use DD/MMM/YYYY. Make sure you understand which format your pharmacy uses. Confusing month and day could mean using medication that expired weeks ago.
Date of compounding: Some labels include the date the medication was prepared. This helps you calculate how much shelf life remains and plan your dosing schedule accordingly. If your label shows a compounding date of February 1st and a BUD of April 1st, you know the pharmacy assigned a 59-day BUD.
Concentration: Typically expressed as milligrams per milliliter (mg/mL). Common concentrations include 5 mg/mL, 10 mg/mL, and 20 mg/mL. This number determines how many units you need to draw for your prescribed dose. The tirzepatide dosage chart in units can translate concentration and dose into exact syringe markings.
Lot number: A tracking number that identifies the specific batch. If you experience any issues with the medication, this number helps the pharmacy trace the problem to its source. Write it down somewhere accessible in case you need it later.
Storage instructions: Usually "Store refrigerated at 2-8C" or similar language. Some labels include additional instructions about light protection or shaking restrictions.
Total volume: The amount of liquid in the vial. Combined with the concentration, this tells you the total milligrams of tirzepatide available. A 5 mL vial at 10 mg/mL contains 50 mg total. Knowing this helps you calculate how many doses your vial contains and whether you can finish it before the BUD arrives.
One common source of confusion: some people receive compounded tirzepatide with the BUD expressed as a number of days rather than a specific calendar date. For example, "Use within 60 days of compounding" or "30-day BUD from first puncture." If your label uses this format, mark the calendar date yourself the day you receive or first use the medication. Do not rely on memory. A simple note on the vial with a permanent marker, or a reminder in your phone, prevents any guesswork about when your medication expires.
Proper storage to maximize compounded tirzepatide shelf life
Storage is the one factor entirely within your control, and it has the biggest impact on whether your compounded tirzepatide reaches its BUD with full potency or degrades silently weeks before.
Refrigeration fundamentals
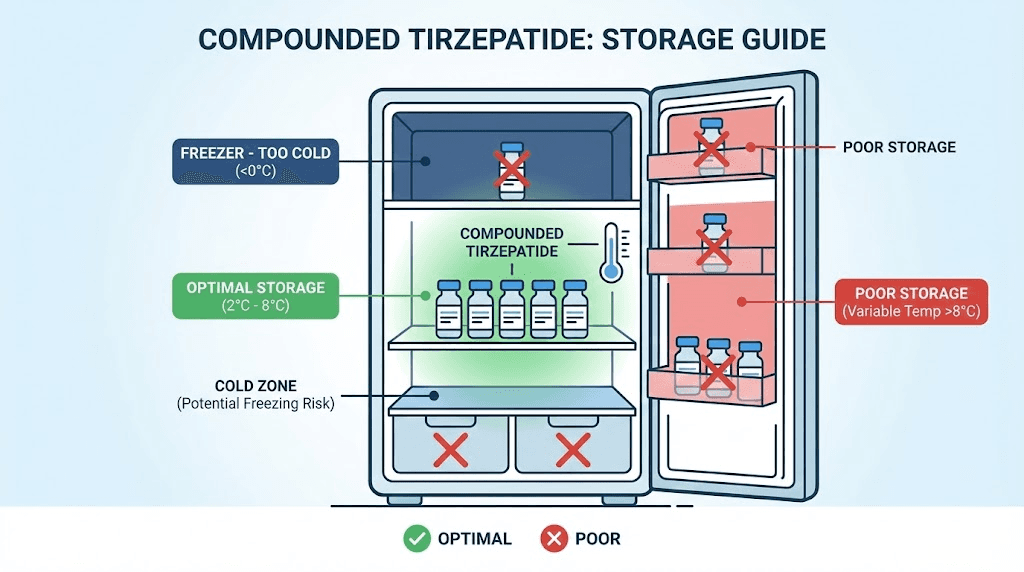
The target temperature range is 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit). Most home refrigerators operate within this range, but not all parts of the refrigerator maintain consistent temperatures. The door shelf is the worst location. It experiences the most temperature fluctuation every time you open the fridge. The crisper drawer is often too cold and risks partial freezing. The middle shelf, toward the back, typically provides the most stable temperature.
Invest in a small refrigerator thermometer. They cost a few dollars and provide peace of mind that your medication is stored within range. Place it next to your tirzepatide vial and check it periodically, especially during summer months when ambient temperatures rise and your refrigerator works harder to maintain its setpoint.
For detailed guidance on refrigeration specifics, the complete guide to tirzepatide fridge storage covers every scenario from power outages to temporary warming events.
Room temperature protocols
Sometimes refrigeration is not immediately possible. You might be traveling, preparing an injection, or simply forgot to return the vial to the fridge. Compounded tirzepatide can tolerate room temperature (up to 30 degrees Celsius / 86 degrees Fahrenheit) for limited periods, typically up to 21 cumulative days.
The key word is cumulative. Every minute outside the refrigerator counts toward that total, whether it is five minutes for an injection or five hours because you left it on the counter. If you are concerned about tracking this precisely, a simple log noting when the vial comes out and goes back in can prevent uncertainty. Most people who use compounded tirzepatide responsibly accumulate only a few hours of total room temperature exposure over the life of a vial, well within safe limits.
If your vial has been out of the fridge for an extended period and you are unsure whether it exceeds the 21-day cumulative limit, err on the side of caution. Potency loss from temperature exposure is invisible. The medication may look, smell, and feel normal while delivering significantly reduced efficacy.
Light protection
Tirzepatide itself is not highly light-sensitive, but formulations containing vitamin B12 are. Cyanocobalamin and methylcobalamin degrade under fluorescent and UV light exposure. If your compounded tirzepatide contains B12, store it in the original packaging or box if possible. If the vial is exposed, keeping it in a dark area of the refrigerator is a simple precaution.
Even for formulations without B12, avoiding direct light exposure is good practice. Glass vials do not fully block UV light, and prolonged exposure to bright light can contribute to peptide degradation over weeks.
Positioning and handling
Store vials upright with the rubber stopper facing up. This minimizes the contact area between the solution and the stopper, reducing the potential for leachables and extractables from the rubber to enter the solution over time. It also prevents the solution from sitting against the stopper seal, which can degrade faster than the glass walls of the vial.
Never vigorously shake your compounded tirzepatide. Peptides are fragile molecules, and aggressive agitation can cause denaturation and aggregation, essentially damaging the protein structure. If you need to mix the contents, gently roll the vial between your palms. The same principle applies to compounded semaglutide storage and other peptide medications.
Signs your compounded tirzepatide has expired or degraded
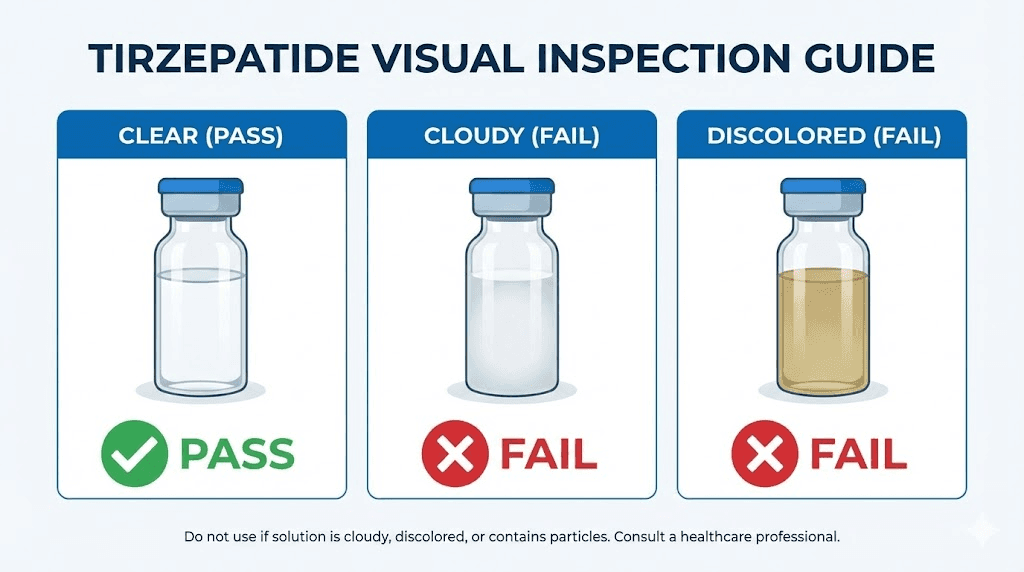
Visual inspection is your first line of defense against using degraded medication. While you cannot test potency at home, several observable changes indicate that your compounded tirzepatide may no longer be safe or effective.
Color changes
Fresh compounded tirzepatide without additives should be a clear, colorless solution. Formulations with B12 typically range from light yellow to light pink. Any deviation from the expected color is a warning sign. If your normally clear tirzepatide develops a yellow tint, or your B12 formulation shifts toward brown or orange, degradation is likely occurring.
Red or dark pink coloration is a particularly concerning sign. While some color variation is normal in B12 formulations, dramatic color shifts suggest breakdown of the cyanocobalamin component. Similarly, red or discolored semaglutide indicates degradation, and the same principle applies to tirzepatide.
Cloudiness and particles
Compounded tirzepatide should be crystal clear. Hold the vial up to a light source and look carefully for any haze, cloudiness, or floating particles. Even small specks or fibers indicate potential contamination or peptide aggregation. Aggregated peptide particles form when denatured tirzepatide molecules clump together, a process that is irreversible and renders the affected portion of the solution inactive.
Do not confuse tiny air bubbles (which are normal after drawing from the vial) with particles. Air bubbles rise to the top and disappear within seconds. Particles remain suspended or settle to the bottom. If you see anything that does not resolve after gently rolling the vial and letting it sit for a minute, do not use the medication.
Odor changes
Normal compounded tirzepatide in bacteriostatic water has a very faint chemical smell from the benzyl alcohol preservative, or no detectable odor at all. Any strong, unusual, or unpleasant smell is a clear indicator of contamination or degradation. If something smells off, trust your senses and discard the vial.
Reduced effectiveness
This is the subtlest sign and the hardest to attribute definitively to expiration. If you have been on a consistent dose and suddenly notice that your tirzepatide is not working anymore, several factors could be responsible. But if the timing coincides with a vial approaching its BUD, or if you suspect storage conditions were compromised, potency loss should be high on your list of possibilities.
Weight loss plateaus, return of appetite, increased blood sugar readings, and decreased metabolic effects can all signal that your medication has lost potency. Before assuming the drug itself is the problem, check whether other factors might explain the stall, and inspect your vial carefully for any of the physical signs described above.
Texture or viscosity changes
The solution should flow freely, like water. If it seems thicker, more viscous, or if you notice any gel-like consistency when drawing with a syringe, the formulation has likely degraded. Changes in viscosity suggest breakdown of the solution matrix or bacterial contamination producing biofilm. Either way, discard the vial immediately.
What happens if you use expired compounded tirzepatide
Using compounded tirzepatide past its beyond-use date is not the same as eating yogurt a day after the sell-by date. The risks are real, even if the consequences are not always immediate or dramatic.
Reduced potency and effectiveness
The most common outcome of using expired compounded tirzepatide is simply that it does not work as well. The peptide degrades over time, breaking down into inactive fragments that cannot bind to GIP and GLP-1 receptors effectively. You might inject your full dose and receive only 60 or 70% of the intended therapeutic effect.
This is insidious because the reduction happens gradually. You do not go from full effectiveness to zero overnight. Instead, you might notice slightly less appetite suppression, marginally slower weight loss progress, or a modest increase in blood glucose. These small changes are easy to attribute to other factors like diet, stress, or tolerance, when the real culprit is sitting in your refrigerator with a past-due label.
If you have experienced these symptoms, the complete guide to expired tirzepatide effects covers the full range of what to expect and how to respond.
Contamination risks
As the preservative system degrades over time, its ability to suppress microbial growth diminishes. A vial that was sterile when compounded may harbor bacteria, fungi, or other microorganisms weeks or months after the BUD, especially if it has been punctured multiple times. Injecting contaminated solution introduces these organisms directly into subcutaneous tissue, potentially causing:
Localized injection site reactions (redness, swelling, warmth)
Abscess formation requiring medical drainage
Cellulitis (spreading skin infection)
In rare cases, systemic infection
The risk is highest for vials that are significantly past their BUD, have been stored improperly, or have been punctured many times.
Degradation byproducts
When tirzepatide degrades, it does not simply become inactive. The peptide breaks down into various fragments and deamidation products. While most of these degradation byproducts are biologically inactive and relatively harmless, some can potentially trigger immune responses. Your body might recognize these degraded fragments as foreign proteins, leading to increased injection site reactions or antibody development that could theoretically reduce the effectiveness of future tirzepatide doses.
When to absolutely discard expired medication
Certain situations demand immediate disposal regardless of how close to or past the BUD:
Any visible cloudiness, particles, or color change
Vial was frozen at any point
Extended exposure to temperatures above 30C (86F)
More than 21 cumulative days at room temperature
Unusual smell from the vial
More than 7 days past the assigned BUD
For comparison, the same principles apply to using expired semaglutide and other compounded GLP-1 medications. The risks and degradation patterns are similar across the class.
How to avoid wasting compounded tirzepatide before it expires
Compounded tirzepatide is too expensive to waste. With BUDs as short as 28 days, poor planning means lost medication and lost money. Here are practical strategies to ensure you use every milligram before expiration.
Match your order size to your dosing schedule
Before ordering, calculate exactly how much tirzepatide you will use before the expected BUD. If you are on a starting dose of 2.5 mg per week and your pharmacy sends 10 mg vials with a 30-day BUD, you will use 10 mg in four weeks, perfectly matching the vial size. But if you are titrating up and your dose increases mid-vial, the math changes.
Use the compounded tirzepatide dosage calculator to map out your exact usage. Input your current dose, injection frequency, vial concentration, and BUD timeline. The calculator will tell you whether your vial will run out before the BUD or whether you will have leftover medication that risks expiring.
Coordinate ordering with your BUD
Many people order their next vial immediately after finishing the current one. This creates a problem if shipping takes 3 to 5 days, leaving a gap in your protocol. But ordering too early means overlapping vials with competing BUDs.
The ideal approach: order your next vial when you have approximately 7 to 10 days of medication remaining. This gives enough buffer for shipping delays while ensuring you start the new vial as close to its compounding date as possible, maximizing usable shelf life.
Consider dose timing strategies
If you find yourself consistently wasting the last 10 to 20% of each vial, explore whether splitting your tirzepatide dose could help. Some protocols divide the weekly dose into two smaller injections, which can smooth out side effects while also giving you more flexibility in timing doses around your BUD.
Microdosing tirzepatide is another approach where smaller, more frequent doses are used. While the clinical evidence for microdosing GLP-1 medications is still emerging, some users report that it helps them manage side effects while using medication more efficiently. The microdose tirzepatide schedule guide outlines common approaches.
Know your pharmacy BUD policies
Not all pharmacies assign the same BUD. If your current pharmacy consistently assigns 28-day BUDs and you regularly waste medication, explore whether other compounding pharmacies offer longer BUDs based on proprietary stability testing. A 60 or 90-day BUD gives you dramatically more flexibility in your dosing schedule and ordering cadence.
Ask specific questions when evaluating pharmacies: What BUD do you assign to your tirzepatide formulation? Is this based on default USP 797 guidelines or proprietary stability data? Can you provide documentation of your stability testing? Pharmacies with robust quality programs are usually happy to share this information.
SeekPeptides members access detailed pharmacy comparison guides and reviews that include BUD information, helping you choose a source that aligns with your dosing schedule and minimizes waste.
Traveling with compounded tirzepatide without compromising expiration
Travel introduces the biggest threat to your compounded tirzepatide shelf life: uncontrolled temperatures. Whether you are driving across town or flying across the country, maintaining the cold chain is essential to reaching your destination with potent medication.
Car travel
Never leave compounded tirzepatide in a parked car, even for a few minutes in warm weather. Vehicle interiors can reach 60 degrees Celsius (140 degrees Fahrenheit) within an hour on a sunny day, even when outside temperatures seem moderate. This extreme heat causes rapid and irreversible peptide degradation.
For short car trips, a small insulated bag with a cold pack works well. Place a barrier (cloth, paper towel) between the cold pack and the vial to prevent direct contact and accidental freezing. For longer road trips, a travel cooler with a digital thermometer lets you verify the temperature stays within the 2 to 8 degree range.
Air travel
Compounded tirzepatide can be carried through airport security. Keep it in your carry-on luggage, never in checked bags where cargo hold temperatures can fluctuate dramatically. Bring a small insulated case with a cold pack, along with your prescription label or pharmacy documentation showing it is a compounded medication.
TSA allows injectable medications and associated supplies (syringes, alcohol swabs) through security. If questioned, having the original pharmacy label visible on the vial speeds the process. The travel guide for GLP-1 medications covers additional tips for navigating airports with injectable medications, and the same principles apply to compounded tirzepatide.
Hotel storage
Most hotel rooms have a mini-fridge or minibar. These are typically set to around 4 to 7 degrees Celsius, which falls within the acceptable storage range. Place your vial toward the back of the fridge where temperatures are most stable, and check that the fridge is actually running.
If no refrigerator is available, request one from the front desk. Most hotels accommodate medical requests without charge. In an absolute pinch, your insulated travel case with a refreshed cold pack can maintain safe temperatures for 12 to 24 hours, giving you time to find proper refrigeration.
International travel considerations
When traveling internationally, consider the total time your medication will be outside controlled storage. Long flights, layovers, customs delays, and transfers to your accommodation can add up to 12 hours or more of unrefrigerated time. While this falls well within the 21-day room temperature allowance, combined with warm ambient temperatures in tropical destinations, the thermal stress is real.
Proper injection technique matters during travel too. Maintaining sterility with correct injection procedures in less-than-ideal environments reduces contamination risk that could accelerate vial degradation. Choosing the right injection site and using fresh supplies for each dose helps maintain both your safety and the medication sterility.
Common mistakes that shorten compounded tirzepatide shelf life
Even careful users make mistakes that quietly reduce their medication potency. These are the most frequent errors and how to fix them.
Storing in the refrigerator door
The refrigerator door is the warmest, most unstable location in your fridge. Every time you open the door, the contents experience a temperature swing of 3 to 5 degrees. Over days and weeks, these fluctuations stress the peptide structure and can reduce potency faster than the BUD anticipates. Move your vial to the middle shelf, toward the back.
Drawing from the vial with a used needle
Every needle puncture through the rubber stopper should use a fresh, sterile needle. Reusing needles introduces bacteria, dead skin cells, and other contaminants into the vial. Even if you only used it once, that needle is no longer sterile. The preservative in bacteriostatic water can handle minor contamination from sterile punctures, but contaminated needles overwhelm the preservative system and dramatically shorten vial life.
Vigorous shaking instead of gentle mixing
Shaking introduces air bubbles and generates mechanical stress on the peptide molecules. Tirzepatide, like all proteins, can denature when subjected to aggressive agitation. Once denatured, the peptide cannot refold correctly and loses its biological activity. If your vial has settled or you see slight concentration gradients, gently roll it between your palms for 10 to 15 seconds.
Using non-bacteriostatic water for reconstitution
If you are reconstituting lyophilized tirzepatide, the water you use matters enormously. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, allowing the reconstituted solution to remain safe for multiple draws over days to weeks. Sterile water for injection does NOT contain a preservative, which means a vial reconstituted with sterile water should ideally be used in a single session or discarded within 24 hours.
Using the wrong type of water is one of the most common reconstitution errors. The tirzepatide reconstitution chart specifies exactly how much bacteriostatic water to add for your target concentration, and following these guidelines precisely protects both the medication stability and your dosing accuracy.
Ignoring temperature excursions
Your vial was left out overnight. Or the power went out for 6 hours. Or you received a shipment that felt warm to the touch. Each of these situations represents a temperature excursion that may or may not have compromised your medication.
A better approach: note the approximate duration and temperature of the excursion. If the vial was at room temperature for less than a few hours, it is almost certainly fine. If it sat in a warm car for several hours or was exposed to temperatures above 30C for any significant period, the medication safety and potency are compromised. The same principle applies to other GLP-1 medications. Learning what happens when semaglutide gets warm or what to do if semaglutide is left out overnight provides useful context for understanding temperature sensitivity across the entire medication class.
How compounded tirzepatide expiration compares to other GLP-1 medications
If you use or have considered other GLP-1 receptor agonists alongside or instead of tirzepatide, understanding how expiration timelines compare helps you make informed decisions about your protocol.
Compounded semaglutide
Compounded semaglutide follows similar BUD guidelines to compounded tirzepatide. Most pharmacies assign 28 to 90 day BUDs depending on their facility classification and stability testing data. The storage requirements are nearly identical: refrigeration at 2 to 8 degrees Celsius, room temperature tolerance up to 21 days, and the same prohibition against freezing.
One notable difference is that semaglutide has been available in compounded form for longer, which means some pharmacies have accumulated more stability data and may offer slightly longer BUDs for their semaglutide formulations. The semaglutide fridge storage guide covers the specifics, and many of the principles apply directly to tirzepatide.
For detailed stability information on specific semaglutide formulations, how long semaglutide remains effective and whether semaglutide expires after 28 days provide granular details that parallel the tirzepatide information in this guide.
Switching between medications
If you are switching from tirzepatide to semaglutide or vice versa, the transition period creates a unique expiration management challenge. You may have partial vials of both medications in your refrigerator simultaneously. Track each vial BUD independently and prioritize using the one expiring soonest.
The semaglutide to tirzepatide conversion chart helps you determine equivalent doses when switching, which in turn helps you calculate how much of each vial you will need during the transition period.
Understanding the broader GLP-1 landscape
The expiration and storage challenges with compounded tirzepatide are not unique. They apply across the entire class of peptide-based GLP-1 medications. Whether you are researching retatrutide, cagrisema, survodutide, or mazdutide, the same storage fundamentals apply: refrigerate consistently, avoid freezing, minimize room temperature exposure, and always respect the assigned BUD or expiration date.
The side effect profiles of these medications also connect to the expiration discussion. Degraded medication can cause unexpected muscle pain, headaches, fatigue, or increased gastrointestinal issues that might be attributed to the medication itself rather than to degradation. If you experience unusual side effects from a vial that is approaching or past its BUD, consider that the medication condition, not the medication itself, might be the cause.
Tracking your expected timeline for results against your actual experience can also help you identify potential potency issues.
Building your protocol around compounded tirzepatide BUD constraints
The beyond-use date should influence your entire approach to tirzepatide, from the dose you choose to the pharmacy you select. Here is how to build a protocol that works with BUD constraints rather than against them.
Dose titration planning
Standard tirzepatide protocols start at 2.5 mg per week and increase in 2.5 mg increments every four weeks. This titration schedule was designed around brand-name pre-filled pens, not compounded multi-dose vials. When using compounded tirzepatide, you need to think ahead about how dose changes affect your vial usage.
For example, if you are at 5 mg per week and your pharmacy sends a 30 mg vial (10 mg/mL, 3 mL), you have six weeks of medication. But if the BUD is only 30 days, you can only use four doses before expiration, wasting roughly a third of the vial. If you plan to increase to 7.5 mg at week five, the math gets even more complicated.
The dosage calculator helps you model these scenarios. Understanding how many units correspond to each dose is essential for this planning. When you know exactly how many insulin units equal your target milligram dose, you can calculate the total number of doses per vial with precision.
Timing your injections strategically
The day of the week you inject matters more than most people realize when BUDs are tight. If your vial arrives on a Monday and the BUD falls on a Friday 30 days later, injecting on Mondays gives you exactly four doses plus a few days of buffer. Injecting on Fridays gives you the same four doses but your last injection happens the day before expiration with zero margin.
The best time to take your GLP-1 shot guide discusses timing from an effectiveness perspective, but timing also matters from a BUD management standpoint. If you take your tirzepatide a day early to use remaining medication before expiration, that is generally safe and preferable to wasting the dose.
Pharmacy selection based on BUD policies
BUD length varies significantly between pharmacies, and it should be a factor in your pharmacy selection. Consider these questions when evaluating options:
What BUD does the pharmacy assign to their tirzepatide formulation?
Is the BUD based on default USP guidelines (28 days) or proprietary stability testing (potentially 60-90 days)?
What is the typical compounding-to-shipping timeline?
Does the pharmacy offer different vial sizes to accommodate various dosing needs?
What cold-chain shipping practices does the pharmacy use?
Reviews from Empower Pharmacy, Strive Pharmacy, ProRx, and Orderly Meds on SeekPeptides include information about BUD policies, shipping practices, and formulation details that directly affect how long your medication lasts after delivery.
Optimizing your tirzepatide results while managing expiration concerns
Expiration management should support your results, not distract from them. The ultimate goal is consistent, effective dosing with minimal waste.
Consistency matters more than perfection
A consistent weekly injection of properly stored tirzepatide will always outperform sporadic dosing from a vial with questionable storage history. If you are focused on your tirzepatide results, the single most important thing is maintaining your injection schedule with medication you are confident is fully potent.
This means choosing a pharmacy, vial size, and ordering cadence that removes uncertainty. You should never be in a position where you are debating whether to inject from an expired or potentially compromised vial because your replacement has not arrived.
Supporting your protocol with proper nutrition
While medication potency is critical, your overall results depend on more than just the tirzepatide itself. Understanding foods to avoid while on tirzepatide and following a tirzepatide diet plan helps you maximize results from each fully potent dose. Many users find that supplements taken alongside tirzepatide support their overall wellbeing during treatment.
The tirzepatide meal plan provides structured nutrition guidance that complements your medication protocol. Proper nutrition becomes especially important if you suspect any potency loss from your current vial, as good dietary habits help maintain momentum even during brief periods of potentially reduced medication effectiveness.
Monitoring your progress
Track your results carefully enough to detect subtle changes that might indicate medication degradation. Weekly weigh-ins, appetite ratings, and progress photos create a data trail that can reveal potency issues before they become obvious. If your progress stalls unexpectedly mid-vial, cross-reference the timing with your vial BUD and storage conditions.
For those early in their journey, understanding how long it takes for tirzepatide to suppress appetite helps set realistic expectations separate from any expiration concerns.
Managing side effects in context
Some side effects that people attribute to tirzepatide itself may actually result from using degraded medication. If you experience unusual body aches, unexpected insomnia, or anxiety that differs from your typical side effect profile, consider whether the medication condition could be a factor.
This does not mean that every side effect is related to expiration. Tirzepatide commonly causes fatigue, joint discomfort, and gastrointestinal effects like constipation as normal therapeutic side effects, especially during dose titration. But ruling out medication degradation as a contributing factor gives you clearer data about your actual response to the drug.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Frequently asked questions
Can I use compounded tirzepatide after the beyond-use date?
Using compounded tirzepatide after its BUD is not recommended. The BUD represents the last date the pharmacy guarantees sterility and potency. While medication one day past the BUD may still be effective, you have no way to verify this without laboratory testing. The risk of reduced potency or contamination increases each day past the BUD, and the potential consequences, including injection site infections and lost therapeutic effect, outweigh the cost savings of finishing a nearly empty vial.
Does the BUD start from when the pharmacy compounds the medication or when I receive it?
The BUD is calculated from the date of compounding, not the date of delivery or first use. If your vial was compounded on February 1st with a 60-day BUD, it expires April 2nd regardless of when it arrived at your door. This is why shipping speed matters. A pharmacy that ships within 24 hours of compounding maximizes your usable window.
Is compounded tirzepatide expiration the same as the expiration on Mounjaro or Zepbound?
No. Brand-name tirzepatide carries manufacturer-assigned expiration dates based on years of stability testing, typically 18 to 24 months from production. Compounded tirzepatide carries a beyond-use date of 28 to 90 days. The difference reflects the gap in stability testing data between pharmaceutical manufacturers and compounding pharmacies. For a detailed comparison, see our guide on tirzepatide expiration.
How should I dispose of expired compounded tirzepatide?
Do not throw expired tirzepatide vials in the regular trash where children or pets might access them. The FDA recommends mixing the medication with an undesirable substance (used coffee grounds, dirt, or cat litter) in a sealed bag before disposing in household trash. Some pharmacies and medical offices accept expired medication for proper disposal.
Can I freeze compounded tirzepatide to extend the expiration date?
Never freeze compounded tirzepatide. Freezing irreversibly damages the peptide molecular structure, even if the solution appears normal after thawing. The ice crystal formation during freezing physically tears apart the protein chains that give tirzepatide its biological activity. A frozen and thawed vial should be discarded entirely.
My compounded tirzepatide looks cloudy but is not expired yet. Is it still safe?
Cloudiness in a compounded tirzepatide vial is a warning sign regardless of the BUD. Clear solution is normal. Cloudiness indicates peptide aggregation, contamination, or chemical instability. Do not inject cloudy medication. Contact your pharmacy to report the issue and request a replacement. Changes in medication color or clarity always warrant investigation.
Does adding B12 or glycine to compounded tirzepatide change the expiration date?
Added compounds can influence the overall stability profile of the formulation. Glycine may provide some stabilizing effect, while B12 adds light sensitivity as a consideration. However, the BUD assigned by the pharmacy already accounts for the complete formulation, including any additives. You do not need to adjust the BUD yourself based on what is in the vial.
What if my pharmacy did not put a BUD on the label?
A compounded medication without a clearly labeled BUD is a regulatory red flag. USP Chapter 797 and state pharmacy regulations require BUDs on all compounded sterile preparations. If your vial does not have a visible BUD, contact the pharmacy immediately. In the meantime, a conservative approach is to assume a 28-day BUD from the date you received the medication.
Can I tell how potent my compounded tirzepatide is at home?
No. There is no reliable home test for peptide potency. Visual inspection can reveal gross degradation (cloudiness, particles, color changes), but a vial can lose 20 to 30% of its potency while still appearing perfectly clear and colorless. Only laboratory HPLC testing can accurately measure remaining peptide concentration. This is why respecting the assigned BUD and proper storage practices are so important.
If I am titrating doses, should I order different vial sizes?
Absolutely. Ordering vial sizes matched to your current dosing phase minimizes waste. During the initial 2.5 mg phase, a smaller vial with a 30-day BUD gives you four to six doses with minimal leftover. As you increase to 5 mg, 7.5 mg, or 15 mg per week, larger vials become more practical since you consume them faster.
Does the concentration of compounded tirzepatide affect expiration?
Concentration can influence stability. Higher concentration formulations (20 mg/mL) contain less solvent relative to peptide, which can create a different degradation profile than lower concentrations (5 mg/mL). However, any reputable pharmacy will assign BUDs based on testing or validated guidelines for their specific concentration. The dosage chart for your specific concentration helps you calculate precise doses.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your storage stay consistent, and your results stay on track.