Feb 18, 2026

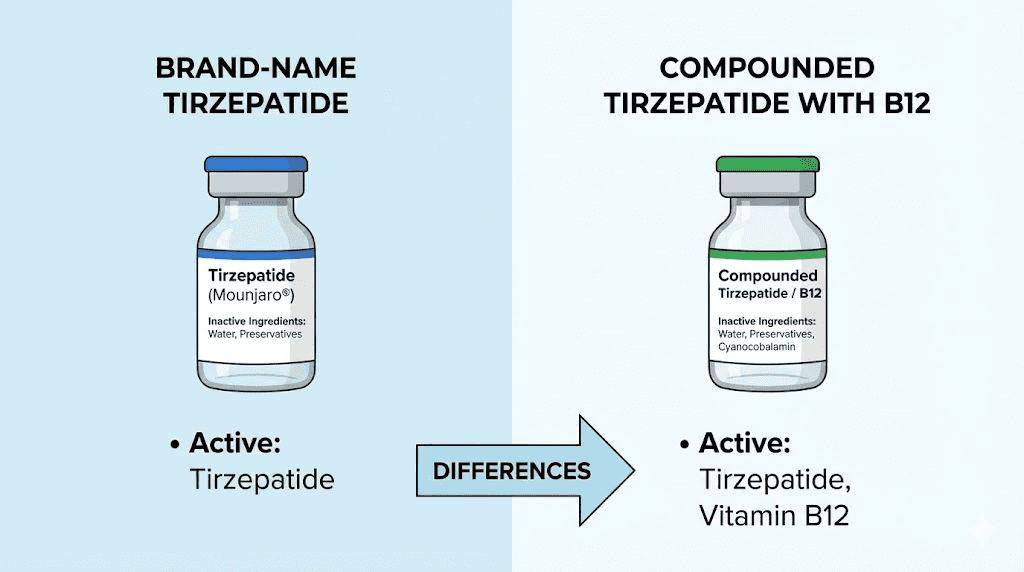
Compounded tirzepatide with B12 is a custom-formulated injectable medication that pairs the GLP-1/GIP receptor agonist tirzepatide with vitamin B12, typically in the form of cyanocobalamin. Compounding pharmacies create these formulations to address specific patient needs that the brand-name versions, Mounjaro and Zepbound, do not cover on their own. The B12 component serves multiple purposes in the formulation, from supporting energy levels during weight loss to potentially reducing common side effects.
But here is the part most guides skip entirely.
The addition of B12 to compounded tirzepatide is not purely a health decision. It also serves a regulatory function. By adding cyanocobalamin (or sometimes methylcobalamin) to the formulation, compounding pharmacies create what the FDA considers a "non-copy" of the commercially available product. This distinction matters enormously in the regulatory landscape surrounding compounded tirzepatide, because pharmacies that produce exact copies of FDA-approved medications face significantly more restrictions than those producing genuinely different formulations. The B12 addition transforms the product from a simple generic alternative into a unique compounded preparation with its own therapeutic rationale.
Understanding this dual purpose changes how you should evaluate these products.
Many compounding pharmacies also include glycine in their tirzepatide/B12 formulations. Glycine is an amino acid that serves as a stabilizer for the peptide and may offer its own benefits for muscle preservation during weight loss. The typical compounded formulation contains tirzepatide as the active weight loss ingredient, cyanocobalamin at doses ranging from 0.25mg to 1mg per injection, and glycine as both a stabilizer and supplementary amino acid. This three-ingredient combination has become the standard offering from most compounding pharmacies that still produce tirzepatide/glycine/B12 formulations.
How tirzepatide and B12 work together in the body
Tirzepatide operates through a dual-receptor mechanism that targets both the GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual action is what separates tirzepatide from single-receptor agonists like semaglutide. When tirzepatide binds to these receptors, it triggers a cascade of metabolic effects. Appetite decreases. Gastric emptying slows. Insulin sensitivity improves. Blood sugar regulation tightens.
The GLP-1 receptor activation handles appetite suppression and blood sugar management. The GIP receptor activation adds a layer that pure GLP-1 agonists cannot match, enhancing fat metabolism and potentially improving how the body handles lipids. Together, these mechanisms produced average weight loss of approximately 21% of body weight over 72 weeks in the SURMOUNT clinical trials, making tirzepatide one of the most effective peptides for fat loss ever studied.
Vitamin B12, meanwhile, fills a completely different but complementary role.
Your body needs B12 for three critical functions that become especially important during significant weight loss. First, B12 supports red blood cell production. Without adequate B12, your body produces abnormally large red blood cells that cannot carry oxygen efficiently, leading to a specific type of anemia called megaloblastic anemia. Second, B12 is essential for nerve function and the production of myelin, the protective coating around your nerves. Third, B12 plays a direct role in energy metabolism by helping convert carbohydrates into glucose and supporting the metabolism of fats and proteins.
Here is where the combination becomes genuinely interesting from a physiological standpoint. GLP-1 receptor agonists slow gastric emptying. That is one of their primary mechanisms for reducing appetite and improving blood sugar control. But slowed gastric emptying also affects nutrient absorption, and B12 is one of the nutrients most sensitive to changes in digestive function. The absorption of B12 requires intrinsic factor produced in the stomach, adequate stomach acid to release B12 from food proteins, and a functioning terminal ileum where absorption occurs. When tirzepatide slows gastric emptying, each of these steps can be affected.
Research shows that GLP-1 medications are associated with reductions in vitamin B12 levels over time. The mechanism is straightforward. Delayed gastric emptying can reduce stomach acid production and alter the environment needed for B12 liberation from food. This means that the longer someone uses tirzepatide, the greater their risk of developing B12 insufficiency or outright deficiency. Symptoms include fatigue, brain fog, mood changes, weakness, and tingling in the hands or feet. Many of these symptoms overlap with common tirzepatide side effects, making it difficult to determine whether the medication itself or a B12 deficiency is responsible for how a patient feels.
By including B12 directly in the injection, compounded formulations bypass the digestive system entirely. The B12 enters the bloodstream through intramuscular or subcutaneous delivery, avoiding the very absorption issues that tirzepatide creates. This is the strongest pharmacological argument for the combination.
Compounded versus brand-name tirzepatide: the real differences
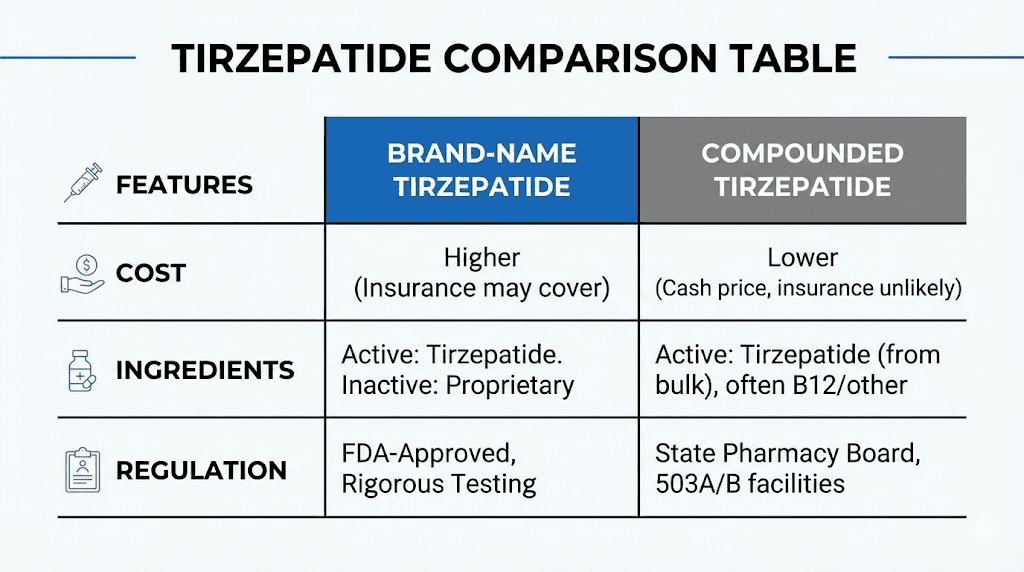
The differences between compounded tirzepatide with B12 and brand-name tirzepatide go deeper than ingredients. They span manufacturing standards, testing requirements, cost structures, and regulatory oversight. Understanding these differences is essential for anyone considering either option.
Manufacturing and quality control
Brand-name tirzepatide (Mounjaro for type 2 diabetes, Zepbound for weight management) is manufactured by Eli Lilly under current Good Manufacturing Practice (cGMP) regulations enforced by the FDA. Every batch undergoes rigorous testing for potency, purity, sterility, and stability. The manufacturing process is validated, meaning it has been proven to consistently produce a product that meets predetermined specifications. Clinical trials involving thousands of participants have established the safety and efficacy profile.
Compounded tirzepatide with B12 operates under a different regulatory framework. Compounding pharmacies that hold a 503A license prepare medications for individual patients based on prescriptions from licensed providers. Pharmacies with 503B outsourcing facility designation can produce larger batches without patient-specific prescriptions but must still comply with cGMP standards. The quality control requirements are real but less extensive than those applied to commercially manufactured pharmaceuticals. There are no clinical trials proving the safety and efficacy of the combined formulation. The individual ingredients have been studied separately, but not together in the specific ratios and concentrations used by compounding pharmacies.
This is not a minor distinction.
Formulation differences
Brand-name tirzepatide comes in single-dose autoinjector pens at fixed concentrations: 2.5mg, 5mg, 7.5mg, 10mg, 12.5mg, and 15mg. Each pen delivers exactly one dose with a pre-set needle and injection mechanism. The inactive ingredients include sodium phosphate dibasic heptahydrate, sodium chloride, and hydrochloric acid for pH adjustment.
Compounded tirzepatide with B12 typically comes in multi-dose vials at various concentrations. Common configurations include tirzepatide at concentrations of 5mg/mL, 10mg/mL, or even 20mg/mL, combined with cyanocobalamin at 0.25mg to 1mg per milliliter and often glycine as a stabilizer. Patients draw their own doses using insulin syringes based on their prescribed amount. This requires understanding tirzepatide dosage in units and proper injection technique.
The multi-dose vial format introduces variables that autoinjector pens eliminate. You need proper technique for drawing doses. You need to maintain sterility when accessing the vial multiple times. You need to understand how to reconstitute tirzepatide if the product arrives in lyophilized (freeze-dried) form rather than as a pre-mixed solution.
Cost comparison
The cost difference has historically been the primary driver of demand for compounded versions. Brand-name tirzepatide (Zepbound) carries a list price of approximately $1,000 or more per month without insurance. Many insurance plans cover Mounjaro for type 2 diabetes but provide limited or no coverage for Zepbound for weight management alone. Out-of-pocket costs for the brand-name product can be prohibitive for many patients.
Compounded tirzepatide with B12 has traditionally been available at significantly lower price points, often ranging from $150 to $500 per month depending on the pharmacy, dosage, and provider. This accessibility drove enormous demand when tirzepatide was in shortage and compounding pharmacies could legally produce essentially identical products. The affordable tirzepatide landscape has shifted dramatically with regulatory changes, which we will cover in detail below.
The B12 forms used in compounded tirzepatide: cyanocobalamin versus methylcobalamin
Not all B12 is created equal, and the form used in your compounded tirzepatide formulation matters more than most providers explain. The two most common forms found in compounded tirzepatide with B12 preparations are cyanocobalamin and methylcobalamin. Each has distinct characteristics that affect stability, absorption, and therapeutic value.
Cyanocobalamin
Cyanocobalamin is the most commonly used form of B12 in compounded tirzepatide formulations. It is a synthetic form that the body must convert to its active forms (methylcobalamin and adenosylcobalamin) before it can be used. The conversion process occurs in the liver and requires adequate folate, functional enzymes, and a healthy methylation pathway.
The advantages of cyanocobalamin in compounding are primarily practical. It is highly stable, which matters enormously in a multi-dose vial that may be stored for weeks. It is less sensitive to light and temperature variations. It has a longer shelf life than methylcobalamin. And it is significantly less expensive to source at pharmaceutical grade. For compounding pharmacies concerned with product stability and consistency, cyanocobalamin is the safer choice from a formulation standpoint.
The typical dose in compounded tirzepatide formulations ranges from 0.25mg (250mcg) to 1mg (1000mcg) of cyanocobalamin per injection. For reference, the daily recommended intake of B12 for adults is just 2.4mcg, so even the lowest compounded dose delivers roughly 100 times the daily requirement. This is not dangerous because B12 is water-soluble, and excess amounts are excreted through the kidneys. But it does raise the question of whether such doses provide meaningful additional benefit beyond correcting a deficiency.
Methylcobalamin
Some compounding pharmacies offer tirzepatide with methylcobalamin instead. Methylcobalamin is the naturally occurring, biologically active form of B12. It does not require conversion in the liver before the body can use it, which makes it theoretically more efficient, especially for individuals who have impaired methylation pathways (including those with certain MTHFR gene variants).
The drawback is stability. Methylcobalamin degrades more rapidly when exposed to light, heat, and certain pH conditions. In a compounded multi-dose vial that gets stored in a refrigerator, accessed multiple times, and occasionally exposed to light during dose preparation, methylcobalamin is more likely to lose potency over the life of the vial. This is why most compounding pharmacies default to cyanocobalamin despite the theoretical advantages of methylcobalamin.
If your provider specifically prescribes methylcobalamin in your compounded tirzepatide, pay extra attention to storage. Keep the vial protected from light. Maintain consistent refrigeration. Use the product within the timeframe specified by the pharmacy. These precautions help preserve the B12 potency throughout the life of the vial, similar to how you would handle any temperature-sensitive peptide. Check our guide on how long tirzepatide lasts in the fridge for general storage principles.
Why compounding pharmacies started adding B12 to tirzepatide
The story of B12 in compounded tirzepatide is equal parts pharmacology and regulatory strategy. Understanding both sides gives you a more complete picture of what you are getting and why.
The pharmacological rationale
The pharmacological argument for adding B12 to tirzepatide rests on several legitimate observations. GLP-1 receptor agonists slow gastric emptying, which can impair B12 absorption over time. Patients on significant caloric restriction (a natural consequence of tirzepatide use) may not consume enough B12-rich foods. Weight loss itself increases metabolic demands that B12 supports. And energy levels during weight loss can suffer without adequate B12.
These arguments are reasonable. They are not, however, supported by clinical trials specifically studying the tirzepatide/B12 combination. The evidence comes from what we know about each ingredient separately and from general research on GLP-1 agonists and nutrient absorption. No large-scale, placebo-controlled trial has specifically evaluated whether adding B12 to tirzepatide injections produces better outcomes than tirzepatide alone with oral B12 supplementation or than tirzepatide alone without any B12 supplementation at all.
The regulatory rationale
This is the part that most marketing materials from compounding pharmacies will not explain directly.
Under the Federal Food, Drug, and Cosmetic Act, compounding pharmacies face different restrictions depending on whether their product is an "essentially a copy" of a commercially available product or a genuinely different formulation. When tirzepatide was in shortage (which the FDA declared in late 2022 and maintained until late 2024), pharmacies could compound essentially identical versions under a shortage exception. But as shortage status changed, pharmacies needed a different legal basis for continued compounding.
Adding B12 (and often glycine or niacinamide) to the formulation creates a product that is not identical to Mounjaro or Zepbound. This "non-copy" distinction potentially allows pharmacies to continue compounding under certain conditions even after the shortage is resolved. The FDA has challenged this interpretation in some cases, and the legal landscape remains complex. But the regulatory incentive to add B12 is undeniable.
Does this mean the B12 is useless? No. The pharmacological rationale has merit. But it is important to recognize that the combination exists for regulatory reasons as much as therapeutic ones. This should not make you distrust the product, but it should inform your expectations about what the B12 component realistically adds to your treatment.
Dosing protocols for compounded tirzepatide with B12
The dosing protocol for compounded tirzepatide generally follows the same titration schedule as brand-name tirzepatide, with the B12 component delivered passively alongside each injection. Understanding both the tirzepatide and B12 dosing is important for managing expectations and outcomes.
Standard tirzepatide titration
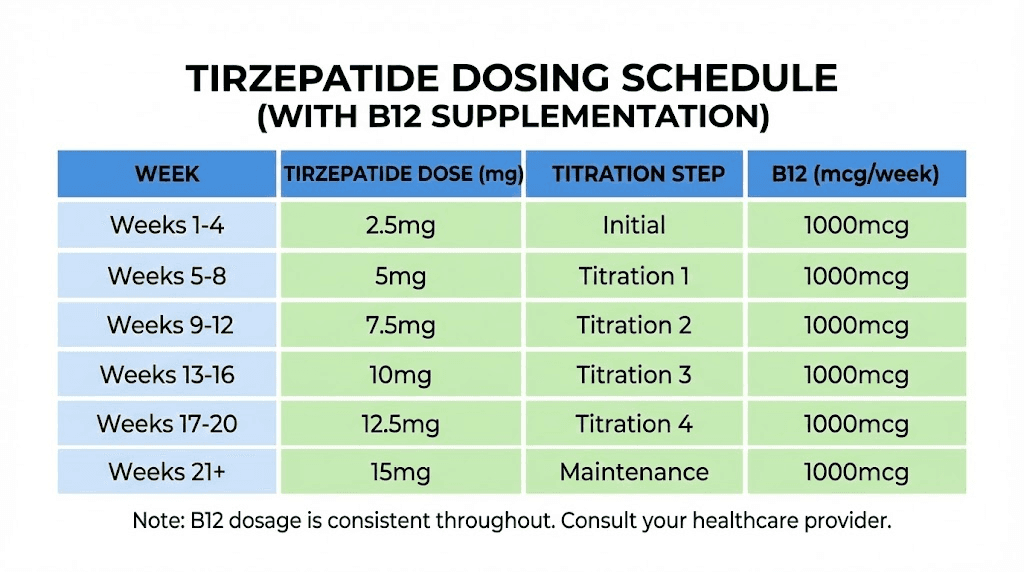
The FDA-approved titration schedule for tirzepatide starts at 2.5mg weekly for the first four weeks. This initial period allows the body to adjust to the GLP-1/GIP receptor activation and minimizes the severity of gastrointestinal side effects. After four weeks, the dose increases to 5mg weekly. From there, further increases of 2.5mg can occur every four weeks at the prescribing provider discretion, up to a maximum of 15mg weekly.
Most compounding pharmacies follow this same schedule with their tirzepatide/B12 formulations. A typical progression looks like this:
Weeks 1 through 4: 2.5mg tirzepatide with B12, once weekly. The focus during this phase is tolerance. Side effects are common but usually mild. Appetite suppression begins within the first week for many patients, though the full timeline for tirzepatide to work varies considerably.
Weeks 5 through 8: 5mg tirzepatide with B12, once weekly. This is where most patients begin seeing meaningful weight loss. The appetite suppression effect intensifies, and blood sugar improvements become more noticeable for those with insulin resistance or type 2 diabetes.
Weeks 9 through 12: 7.5mg tirzepatide with B12, once weekly. Weight loss typically accelerates at this dose. Side effects that appeared during dose increases usually diminish within one to two weeks.
Weeks 13 through 16: 10mg tirzepatide with B12, once weekly. Many patients find their maintenance dose somewhere between 10mg and 15mg. The decision to increase further depends on weight loss trajectory, side effect tolerance, and individual goals.
Weeks 17 and beyond: 12.5mg to 15mg tirzepatide with B12, once weekly, if needed. Not everyone requires the maximum dose. Some patients achieve their goals at 7.5mg or 10mg and maintain at that level indefinitely.
B12 dosing within the formulation
The B12 dose in compounded formulations is generally fixed per milliliter of solution. As the tirzepatide dose increases and the injection volume changes, the amount of B12 per injection may also change, depending on the concentration of the specific product. For example, a formulation containing 0.5mg B12 per milliliter will deliver different amounts of B12 depending on whether you inject 0.25mL (for a lower tirzepatide dose) or 0.75mL (for a higher dose).
In practice, this dosing variation is clinically insignificant. B12 is water-soluble and non-toxic at the doses used in compounded formulations. Whether you receive 125mcg or 750mcg of cyanocobalamin per injection, your body will use what it needs and excrete the rest. The more important factor is that you are receiving B12 through a parenteral route (injection) that bypasses the digestive absorption issues caused by tirzepatide.
How to calculate your dose
Calculating the correct injection volume from a multi-dose vial requires knowing the concentration of your specific product. If your vial contains tirzepatide at 10mg/mL and your prescribed dose is 5mg, you would draw 0.5mL (or 50 units on an insulin syringe). Use the dosage calculator or refer to the tirzepatide dosage chart in units for reference.
Common concentration and volume combinations:
At 5mg/mL concentration: 2.5mg = 0.5mL (50 units), 5mg = 1.0mL (100 units), 7.5mg = 1.5mL (150 units)
At 10mg/mL concentration: 2.5mg = 0.25mL (25 units), 5mg = 0.5mL (50 units), 7.5mg = 0.75mL (75 units), 10mg = 1.0mL (100 units)
At 20mg/mL concentration: 2.5mg = 0.125mL (12.5 units), 5mg = 0.25mL (25 units), 10mg = 0.5mL (50 units), 15mg = 0.75mL (75 units)
Higher concentrations mean smaller injection volumes, which generally means less discomfort at the injection site and less B12 per dose. Lower concentrations mean larger volumes but more B12 per injection. Our tirzepatide 20mg/mL dosing chart provides detailed calculations for the most common high-concentration formulations.
Benefits of the B12 addition: what the evidence actually supports
The marketing for compounded tirzepatide with B12 tends to overstate the benefits of the combination. The reality is more nuanced. Some claimed benefits have genuine support. Others are theoretical at best.
Supported by evidence
Prevention of B12 deficiency during GLP-1 treatment. This is the strongest argument for the combination. GLP-1 receptor agonists are associated with reduced B12 absorption over time. Parenteral B12 delivery bypasses the digestive system entirely. For patients on long-term tirzepatide therapy, maintaining adequate B12 levels is a legitimate clinical concern, and injectable B12 addresses it directly.
Correction of pre-existing B12 deficiency. Many patients starting tirzepatide for weight loss already have suboptimal B12 levels. Obesity is associated with lower B12 status, and dietary patterns common among people with metabolic dysfunction often lack adequate B12 sources. For these patients, the B12 in their compounded tirzepatide provides genuine therapeutic benefit.
Energy support during caloric restriction. B12 plays a direct role in energy metabolism. Patients on tirzepatide eat significantly less, sometimes dramatically so. When caloric intake drops substantially, ensuring adequate B12 through injection helps maintain the metabolic pathways that convert food into usable energy. This does not mean B12 "gives you energy" the way caffeine does. It means B12 prevents the fatigue and lethargy that accompany B12 deficiency. For context on managing fatigue during GLP-1 treatment, see our guide on GLP-1 fatigue.
Theoretically reasonable but unproven
Reduced nausea. Some compounding pharmacies claim that B12 helps reduce the nausea associated with tirzepatide use. The evidence for this is thin. While B12 deficiency can cause nausea, and correcting a deficiency would resolve that symptom, there is no clinical evidence that B12 supplementation reduces GLP-1-related nausea in patients who are not B12 deficient. The nausea from tirzepatide results from receptor activation in the GI tract and brain, not from B12 status.
Improved cognitive function. B12 is essential for neurological function, and some marketing materials suggest the B12 in compounded tirzepatide improves mental clarity and reduces brain fog. Again, this is true only if you have a B12 deficiency. For patients with adequate B12 levels, additional supplementation does not enhance cognitive performance.
Enhanced weight loss. Some sources imply that B12 helps "boost metabolism" and accelerate weight loss. The metabolic role of B12 is real but modest. B12 helps convert macronutrients into energy but does not independently accelerate fat burning. There is no evidence that adding B12 to tirzepatide increases weight loss beyond what tirzepatide alone produces.
Not supported by evidence
Muscle preservation. While glycine (often included alongside B12) has some data supporting muscle preservation, B12 itself does not directly prevent muscle loss during weight loss. Adequate protein intake, resistance training, and appropriate supplementation with tirzepatide are far more important factors for maintaining lean mass.
Detoxification support. Some marketing claims suggest B12 supports detoxification during weight loss as fat cells release stored toxins. While B12 does participate in methylation reactions that are part of the body detoxification processes, the clinical significance of this in the context of compounded tirzepatide is negligible.
Side effects and safety considerations
The side effects of compounded tirzepatide with B12 are predominantly the side effects of tirzepatide itself. B12 at the doses used in compounded formulations rarely causes adverse reactions. However, understanding the full side effect profile helps you manage your treatment effectively and know when to contact your provider.
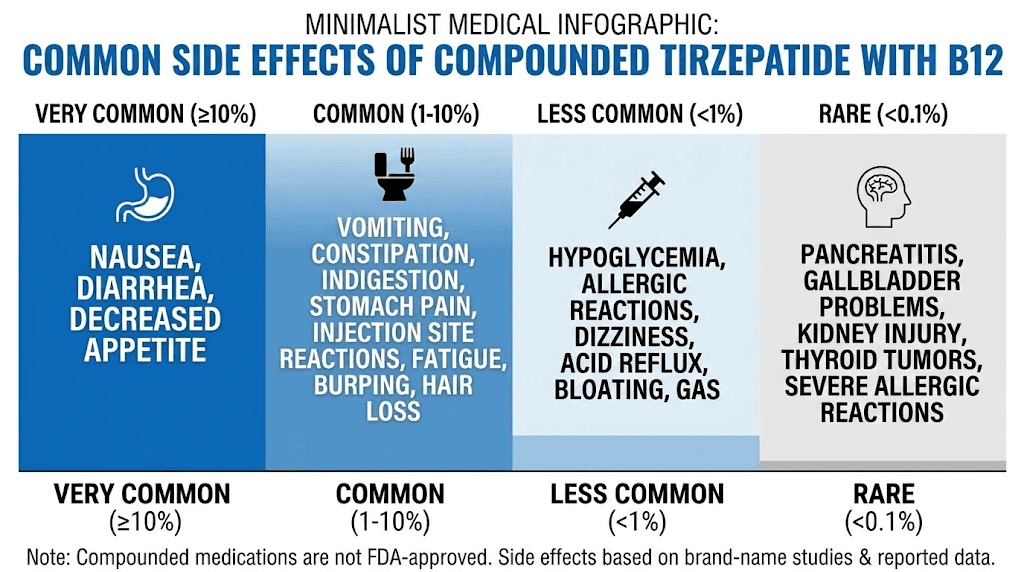
Common tirzepatide side effects
Nausea is the most frequently reported side effect, affecting a substantial percentage of patients, especially during dose initiation and increases. It tends to peak in the first few days after a dose and diminish over the following week. Strategies for managing nausea include injecting in the evening before bedtime (so you sleep through the worst of it), eating smaller meals, avoiding fatty or greasy foods, and staying hydrated. Our guide on foods to avoid on tirzepatide covers dietary adjustments in detail.
Other common GI side effects include constipation, diarrhea, reduced appetite (which is technically the therapeutic effect but can feel extreme at higher doses), and abdominal discomfort. For constipation management, see our dedicated guide on tirzepatide constipation treatment.
Fatigue and tiredness on tirzepatide is common, particularly during the early weeks and after dose increases. This is where the B12 component theoretically helps. If some of the fatigue is related to developing B12 insufficiency from altered gastric function, the injected B12 may mitigate this specific contributor to tiredness.
Headaches affect some patients, particularly during the titration phase. Our tirzepatide headache guide covers causes and management strategies.
Injection site reactions including redness, swelling, or mild pain at the injection site can occur with any injectable medication. Proper technique and injection site rotation minimize this risk.
B12-specific considerations
Vitamin B12 in injectable form is generally considered very safe. It is water-soluble, meaning excess amounts are excreted rather than stored. Serious adverse reactions to injectable cyanocobalamin are rare but include allergic reactions (extremely rare), acne-like skin eruptions at very high doses, and mild diarrhea.
One concern specific to compounded formulations is that the interaction between tirzepatide and B12 in the same solution has not been formally studied. While there is no pharmacological reason to expect a negative interaction (they work through completely different mechanisms), the lack of formal stability and compatibility studies means there is a small but non-zero unknown risk.
Safety of compounded versus brand-name products
The FDA has raised concerns about compounded tirzepatide products in general, not specifically about the B12 component. Issues identified by the FDA include variations in potency (some compounded products contained less tirzepatide than labeled), sterility concerns, and quality control inconsistencies. These risks are not unique to tirzepatide/B12 formulations but apply to all compounded products.
To minimize risk when using compounded tirzepatide with B12:
Verify that your compounding pharmacy holds proper licensing (503A or 503B designation). Ask about their quality testing procedures, including potency verification and sterility testing. Ensure the product comes with clear labeling showing all ingredients and concentrations. Store the product according to pharmacy instructions, typically refrigerated between 36 and 46 degrees Fahrenheit.
The regulatory landscape: what happened and where things stand
The regulatory history of compounded tirzepatide is one of the most complex and rapidly evolving stories in pharmaceutical law. For patients using or considering compounded tirzepatide with B12, understanding this landscape is critical because it directly affects availability, legality, and your options going forward.
The shortage era
In late 2022, the FDA placed tirzepatide on the drug shortage list due to overwhelming demand that Eli Lilly could not meet. Under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act, compounding pharmacies can produce versions of commercially available drugs when those drugs are in shortage. This opened the floodgates. Dozens, then hundreds, of compounding pharmacies began producing tirzepatide, often at a fraction of the brand-name cost.
During this period, the B12 addition was not strictly necessary from a regulatory standpoint. The shortage exemption allowed pharmacies to produce essentially identical products. But many pharmacies added B12 (and glycine, and niacinamide) anyway, partly for therapeutic reasons and partly as a hedge against the eventual resolution of the shortage.
The shortage resolution
In October 2024, the FDA announced that the tirzepatide shortage had been resolved, and Eli Lilly could meet market demand. This triggered a transition period during which compounding pharmacies were expected to wind down production of products that were essentially copies of FDA-approved tirzepatide. For 503A pharmacies, the transition deadline was March 2025. For 503B outsourcing facilities, the deadline extended to May 2025.
Multiple lawsuits challenged the FDA determination, with compounding pharmacies and telehealth companies arguing that the shortage was not truly resolved. In May 2025, a federal court in the Northern District of Texas upheld the FDA position, dealing a significant blow to compounders who wanted to continue producing copy versions.
Where things stand now
The regulatory landscape remains in flux. Routine compounding of products that are "essentially copies" of FDA-approved tirzepatide is no longer permitted under the shortage-related exception. However, several pathways for compounding still exist:
Non-copy formulations. Compounded tirzepatide with B12 may qualify as a non-copy formulation because the B12 component makes it materially different from Mounjaro and Zepbound. This is the primary legal basis under which many pharmacies continue to offer tirzepatide/B12 products. The FDA has not provided definitive guidance on whether adding B12 is sufficient to establish non-copy status, and this remains an area of active legal debate.
Patient-specific medical need. Under 503A regulations, compounding pharmacies can prepare medications for individual patients when a prescriber determines that the commercially available product does not meet the patient specific medical need. Examples might include a documented allergy to an inactive ingredient in brand-name tirzepatide or a need for a dosage form not commercially available.
Ongoing litigation. Several legal challenges to the FDA position continue to work through the courts. The regulatory environment could shift if appellate courts overturn lower court decisions or if Congress acts to change the legal framework for compounding.
For patients currently using compounded tirzepatide with B12, the practical implication is uncertainty. Your pharmacy may continue to provide the product if it qualifies as a non-copy formulation. But availability may vary by state, pharmacy, and the evolving interpretation of federal regulations. SeekPeptides maintains updated information on access pathways and regulatory developments affecting the peptide community.
How to store compounded tirzepatide with B12 properly
Proper storage directly affects the potency and safety of your compounded tirzepatide with B12. Unlike brand-name products that come in single-use autoinjectors, compounded formulations in multi-dose vials require more careful handling.
Refrigeration requirements
Most compounded tirzepatide with B12 should be stored in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). The tirzepatide component is a peptide that degrades faster at room temperature, and the B12 component (particularly if methylcobalamin) is light-sensitive. Refrigeration slows both degradation processes significantly.
Never freeze compounded tirzepatide. Freezing can damage the peptide structure and alter the properties of the solution. If your vial accidentally freezes, do not use it. Contact your pharmacy for a replacement. For detailed storage guidance, refer to our comprehensive guide on how long compounded tirzepatide can be out of the fridge.
Light protection
Both tirzepatide and B12 (especially methylcobalamin) can degrade when exposed to light. Keep your vial in its original packaging or a container that blocks light. If your pharmacy provides the product in a clear vial without light protection, store it inside a box or wrapped in aluminum foil. This is particularly important for multi-dose vials that remain in use for weeks.
Multi-dose vial handling
Every time you access a multi-dose vial with a needle, you introduce a small risk of contamination. Follow these practices to maintain sterility: Always clean the rubber stopper with an alcohol swab before inserting the needle. Use a new, sterile needle and syringe for each dose. Never touch the rubber stopper with your fingers after cleaning it. Store the vial upright to keep the solution away from the stopper.
Most compounding pharmacies assign a beyond-use date of 28 to 42 days for multi-dose vials once they have been accessed. This date may differ from the expiration date on the vial. Always use the shorter date as your guide. After the beyond-use date, discard remaining solution even if it appears unchanged. Our guide on tirzepatide expiration explains these timelines in more detail.
Travel considerations
Traveling with compounded tirzepatide with B12 requires planning. The medication needs to stay cold, so you will need an insulated travel case with ice packs or a portable cooler. TSA allows injectable medications through security with documentation (a prescription label on the vial or a letter from your prescriber). Keep your medication in its original pharmacy-labeled vial. Our guide to traveling with injectable peptides covers the logistics in detail, and the same principles apply to compounded tirzepatide.
Who benefits most from the B12 formulation
Not everyone needs B12 in their tirzepatide. Understanding who benefits most helps you make an informed decision about whether to seek out a compounded formulation with B12 or whether separate B12 supplementation (or no supplementation at all) makes more sense for your situation.
People with documented B12 deficiency or insufficiency
If your bloodwork shows low B12 levels (below 200 pg/mL) or borderline levels (200 to 300 pg/mL), the injectable B12 in compounded tirzepatide provides a convenient correction. You receive your B12 alongside your weekly tirzepatide dose without needing a separate injection or daily oral supplement. This is especially relevant for individuals who absorb oral B12 poorly, such as those with pernicious anemia, atrophic gastritis, or a history of gastrointestinal surgery.
Long-term GLP-1 users
The risk of B12 depletion increases with the duration of GLP-1 therapy. If you plan to use tirzepatide for six months or longer (and most weight management protocols are intended for long-term use), the cumulative effect of reduced B12 absorption becomes more significant. Starting with a B12-inclusive formulation from the beginning may prevent deficiency from developing in the first place.
People with limited dietary B12 intake
B12 occurs naturally only in animal products: meat, fish, eggs, and dairy. Vegetarians and especially vegans are at higher risk for B12 deficiency even without GLP-1 therapy. When you add the appetite suppression and reduced food intake caused by tirzepatide, the risk compounds. For plant-based eaters using tirzepatide, the B12 injection provides a reliable source that does not depend on dietary intake. Check our guide on what to eat on tirzepatide for dietary guidance during treatment.
Patients experiencing significant fatigue
If fatigue is a primary complaint during your tirzepatide treatment, and other causes have been evaluated, a B12-inclusive formulation may help, particularly if blood testing reveals low-normal or below-normal B12 levels. However, fatigue during GLP-1 therapy has many potential causes including caloric deficit, dehydration, electrolyte imbalances, and the medication effects on the central nervous system. B12 supplementation only helps if B12 deficiency is contributing to the fatigue.
Who does not need the B12 formulation
If your B12 levels are consistently in the normal range (above 400 pg/mL), you eat a varied diet including animal products, and you do not experience symptoms of B12 deficiency, the B12 in compounded tirzepatide is unlikely to provide meaningful additional benefit. You are essentially paying for an ingredient that your body will excrete unused. In this case, standard tirzepatide (whether brand-name or compounded without B12) may be the more practical choice.
Compounded tirzepatide with B12 versus other combination formulations
B12 is not the only additive compounding pharmacies include with tirzepatide. Understanding how different formulations compare helps you evaluate which option might be most appropriate for your situation. SeekPeptides tracks the evolving landscape of peptide formulations to help researchers make informed decisions.
Tirzepatide with B12 versus tirzepatide with niacinamide
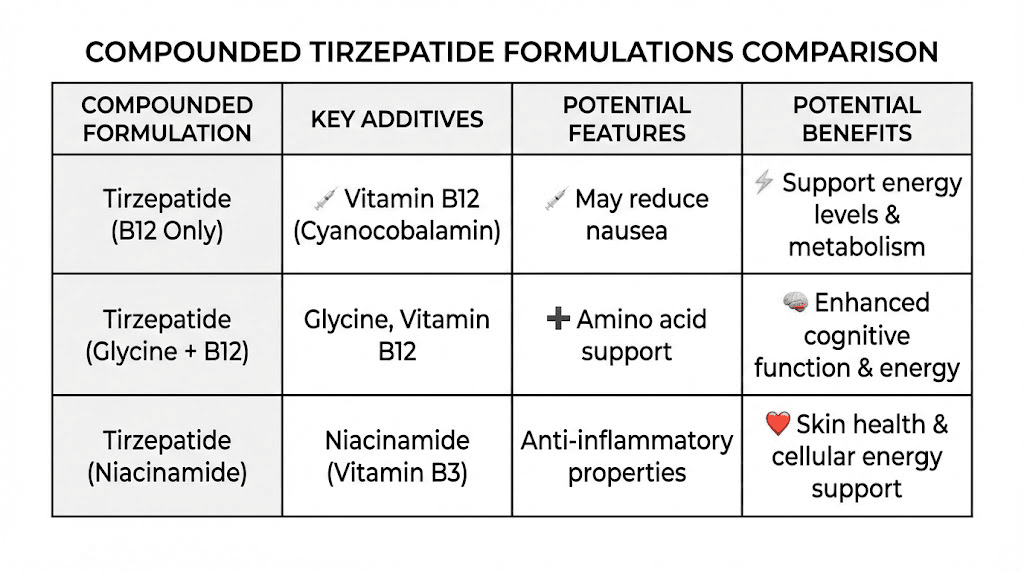
Niacinamide (vitamin B3) is the other common additive in compounded tirzepatide formulations. Some pharmacies, notably Empower Pharmacy, have offered tirzepatide/niacinamide formulations. Niacinamide supports skin health, cellular energy production, and may help with some of the skin changes associated with rapid weight loss. For a detailed comparison, see our tirzepatide with niacinamide guide.
The choice between B12 and niacinamide formulations depends on individual needs. B12 is more relevant for patients at risk of deficiency and those experiencing fatigue. Niacinamide may be more relevant for patients concerned about skin health during weight loss. Neither additive has been proven to enhance the weight loss efficacy of tirzepatide itself.
Tirzepatide with B12 and glycine versus B12 alone
The triple combination of tirzepatide, glycine, and B12 is the most common compounded formulation. Glycine serves dual purposes: it acts as a stabilizer that may improve the shelf life and consistency of the compounded product, and it provides supplemental amino acid that supports muscle preservation during weight loss. For patients concerned about losing lean mass during their weight loss journey, the glycine addition has some supporting evidence, though the amounts included in most formulations may be too small to produce clinically meaningful effects. Our tirzepatide glycine B12 compound guide covers this triple formulation in depth.
How compounded formulations compare to brand-name alternatives
For patients who cannot access or afford brand-name tirzepatide (Mounjaro or Zepbound), compounded formulations with B12 represent one option. For a broader look at cost considerations, see our guide on affordable compounded tirzepatide. It is also worth understanding how tirzepatide compares to other GLP-1 options. Our semaglutide versus tirzepatide comparison covers the key differences between these two leading weight loss peptides.
What to look for in a compounding pharmacy
If you decide that compounded tirzepatide with B12 is the right choice for your situation, selecting the right pharmacy is critical. Not all compounding pharmacies are equal in quality, compliance, or reliability. Here is what to evaluate.
Licensing and accreditation
At minimum, the pharmacy should hold a valid state pharmacy license and either a 503A or 503B federal designation. 503B outsourcing facilities are subject to more stringent oversight, including regular FDA inspections and mandatory adverse event reporting. They also operate under cGMP requirements similar (though not identical) to commercial manufacturers. If given the choice, a 503B facility generally offers more quality assurance.
Third-party accreditation from organizations like the Pharmacy Compounding Accreditation Board (PCAB) or the Accreditation Commission for Health Care (ACHC) provides an additional layer of verification. These accreditations require pharmacies to meet specific quality standards and undergo regular audits.
Testing and quality assurance
Ask whether the pharmacy performs third-party testing on their compounded tirzepatide with B12. Key tests include potency verification (confirming the actual amount of tirzepatide matches the labeled amount), sterility testing, endotoxin testing, and beyond-use date testing. Pharmacies that invest in these tests demonstrate a commitment to quality that goes beyond minimum regulatory requirements.
Transparency about formulation
A reputable pharmacy should be willing to disclose the complete formulation of their product, including the exact form and concentration of B12, the source of their tirzepatide raw material, and any additional inactive ingredients. They should provide clear labeling on the vial itself and include storage instructions, beyond-use dating, and concentration information.
Provider relationship
Compounded tirzepatide with B12 requires a prescription from a licensed healthcare provider. Be cautious of pharmacies or telehealth platforms that make the prescription process feel automatic or perfunctory. A legitimate prescriber should review your medical history, discuss potential contraindications, order baseline bloodwork (including B12 levels), and establish a monitoring plan that includes follow-up appointments and lab work.
Monitoring your health on compounded tirzepatide with B12
Regular monitoring is essential for anyone using tirzepatide, and the compounded B12 formulation does not eliminate the need for medical oversight. Here is what your monitoring plan should include.
Baseline bloodwork
Before starting compounded tirzepatide with B12, your provider should order a comprehensive metabolic panel including fasting glucose and HbA1c, lipid panel, complete blood count, B12 level, thyroid function tests, kidney function markers, and liver function tests. These baselines allow your provider to track changes over time and identify any emerging concerns early.
Ongoing monitoring schedule
During the titration phase (the first 16 to 20 weeks), check in with your provider at each dose increase. Lab work is typically repeated every three to six months once you reach a stable maintenance dose. B12 levels should be checked even though you are receiving injectable B12, because absorption and utilization can vary between individuals.
Weight monitoring is obvious but should be done consistently. Weekly weigh-ins at the same time of day, on the same scale, provide the most reliable trend data. Do not panic over daily fluctuations, which are normal. Track weekly averages instead. If you are not losing weight on tirzepatide, several factors could be at play, and our troubleshooting guide covers the most common causes.
When to contact your provider
Seek medical attention for severe or persistent nausea and vomiting that prevent you from keeping down fluids, signs of pancreatitis (severe abdominal pain radiating to the back, often accompanied by vomiting), symptoms of allergic reaction (difficulty breathing, severe rash, facial swelling), gallbladder symptoms (sharp right upper abdominal pain, especially after meals), or signs of thyroid changes (lump or swelling in the neck, difficulty swallowing, persistent hoarseness).
Also contact your provider if you experience symptoms of B12 deficiency despite the injectable supplementation, including persistent fatigue, tingling or numbness in hands and feet, difficulty with balance, or cognitive changes. While unlikely with injectable B12, individual absorption and utilization can vary.
Combining compounded tirzepatide with B12 with other supplements and medications
Many patients on tirzepatide take additional supplements to support their health during weight loss. Understanding which combinations are safe and which require caution helps you optimize your protocol without introducing unnecessary risks.
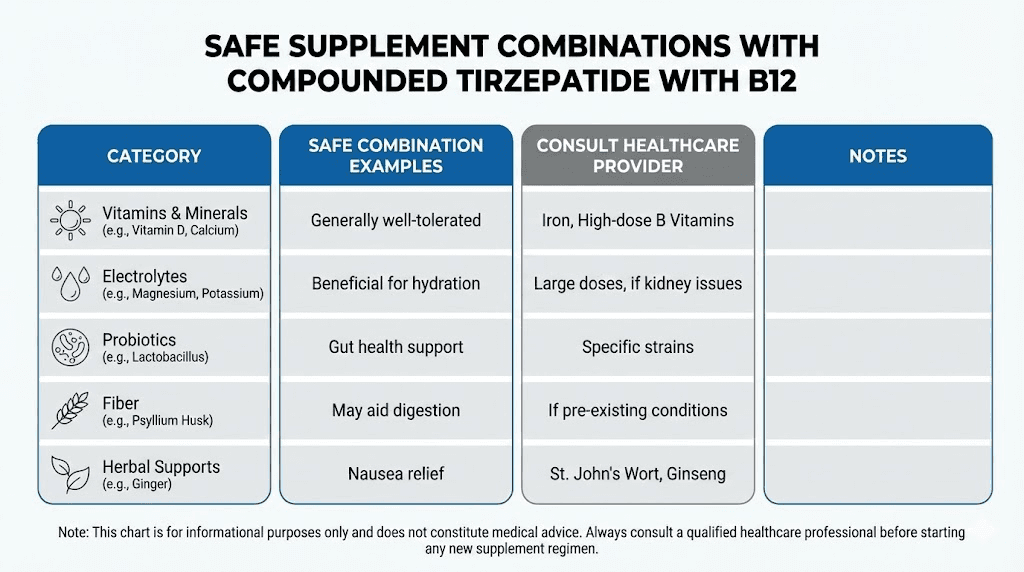
Generally safe combinations
Multivitamins are generally safe and often recommended during significant weight loss to prevent micronutrient deficiencies from reduced food intake. The B12 in your compounded tirzepatide covers that specific vitamin, so look for a multivitamin without additional B12 to avoid excessive intake (though excess B12 is excreted and poses minimal risk).
Protein supplements help maintain lean muscle mass during weight loss, which is a genuine concern with any GLP-1 therapy. Whey protein, collagen peptides, and plant-based protein powders are all compatible with tirzepatide. Adequate protein intake (0.8 to 1.2 grams per kilogram of body weight) is one of the most important dietary considerations during treatment.
Electrolyte supplements can help with dehydration symptoms, especially during the early weeks when nausea may reduce fluid intake. Look for electrolyte products without excessive sugar or calories.
Fiber supplements may help with the constipation that many patients experience on tirzepatide, though they should be taken at a different time than other medications to avoid interfering with absorption. For more on managing this common side effect, see our GLP-1 constipation treatment guide.
Combinations requiring medical guidance
Metformin and tirzepatide can be used together under medical supervision, and this combination is common in type 2 diabetes management. However, both medications affect GI function, and the side effects can compound. Notably, metformin is one of the drugs most strongly associated with B12 malabsorption, making the B12 in compounded tirzepatide particularly relevant for patients on both medications.
Phentermine and tirzepatide is a combination some providers prescribe for enhanced appetite suppression, though it is not FDA-approved as a combination. Learn more about this pairing in our phentermine and GLP-1 combination guide.
Other GLP-1 medications should never be combined with tirzepatide. Using semaglutide and tirzepatide together would double the GLP-1 receptor activation and dramatically increase the risk of severe side effects including pancreatitis.
Alcohol and tirzepatide
Moderate alcohol consumption is generally considered safe on tirzepatide, but there are important caveats. Tirzepatide affects blood sugar regulation, and alcohol can cause unpredictable blood sugar fluctuations. Many patients also report increased alcohol sensitivity on GLP-1 medications. B12 is depleted more rapidly by heavy alcohol use, which is another consideration. For a detailed analysis, see our guide on drinking on tirzepatide.
Expected results and realistic timelines
Setting realistic expectations for compounded tirzepatide with B12 helps prevent discouragement during the early weeks and provides benchmarks for evaluating your progress over time.
What to expect week by week
Week 1: Reduced appetite is often noticeable within the first few days. Nausea is common. Weight loss may begin, typically 1 to 3 pounds, though some of this is water weight. The B12 component begins building levels immediately if you were previously deficient.
Weeks 2 through 4: Appetite suppression becomes more consistent. GI side effects may improve or fluctuate. Weight loss continues at 1 to 2 pounds per week. Energy levels may stabilize as your body adjusts and B12 levels normalize. How fast tirzepatide works varies significantly between individuals.
Weeks 5 through 8 (5mg dose): This is where most patients see the treatment really take hold. Appetite suppression strengthens noticeably. Weight loss often accelerates to 2 to 3 pounds per week. Cravings diminish significantly. Blood sugar improvements become measurable on lab work.
Weeks 9 through 16 (7.5 to 10mg): Consistent, meaningful weight loss. Many patients report losing 10 to 15% of their starting body weight by this point. Energy levels often improve as the body adapts and the B12 supports metabolic function. This is the period where before and after tirzepatide results become visually apparent.
Months 4 through 6 (10 to 15mg): Continued weight loss, though the rate may slow as you approach a new metabolic equilibrium. Total weight loss of 15 to 20% of starting body weight is achievable for many patients. Blood pressure, cholesterol, and other metabolic markers typically show significant improvement.
Beyond 6 months: Maintenance phase for many patients. Some continue losing weight at a slower rate. Others find a stable weight and maintain it with ongoing treatment. The decision to continue, reduce dose, or discontinue should be made with your provider based on your individual goals and health markers.
The B12 component and your energy timeline
If you start compounded tirzepatide with B12 while B12-deficient, you may notice energy improvements within one to two weeks as B12 levels begin to normalize. The full correction of B12 deficiency typically takes four to six weeks of consistent supplementation. If your B12 levels were already normal when you started, you are unlikely to notice any energy changes attributable to the B12 component specifically.
When results plateau
Weight loss plateaus are normal and expected on any weight loss medication. They do not mean the medication has stopped working. Plateaus often indicate that your metabolism has adjusted to your lower caloric intake and new body weight. Strategies for breaking through include confirming adequate protein intake, adding or increasing resistance training, evaluating your caloric intake (which can drift upward even on tirzepatide), and discussing a dose adjustment with your provider. Our guide on tirzepatide not working anymore provides comprehensive troubleshooting.
Nutrition and lifestyle optimization while on compounded tirzepatide with B12
The B12 in your compounded tirzepatide covers one piece of the nutritional puzzle, but successful long-term outcomes depend on a broader approach to nutrition and lifestyle during treatment. SeekPeptides members access comprehensive protocols for optimizing outcomes during GLP-1 therapy, including personalized dietary guidance and progress tracking tools.
Dietary priorities
Protein is the single most important macronutrient to prioritize while on tirzepatide. The medication dramatically reduces appetite and total food intake, and without deliberate effort, protein intake often drops below the threshold needed to preserve lean muscle mass. Aim for 0.8 to 1.2 grams of protein per kilogram of body weight daily, front-loading protein at each meal to ensure you consume it before appetite suppression limits your total intake.
Hydration matters more than most patients realize. Tirzepatide reduces food intake, and a significant portion of daily fluid intake comes from food. Dehydration contributes to headaches, fatigue, constipation, and dizziness. Aim for at least 64 ounces of water daily, more if you exercise or live in a hot climate.
Micronutrient density becomes critical when you are eating less food overall. Every meal should provide maximum nutritional value. Prioritize lean proteins, vegetables, fruits, whole grains, and healthy fats. Minimize processed foods, which provide calories without proportionate nutrition. Our tirzepatide diet plan provides a comprehensive framework, and the foods to eat on GLP-1 medications guide offers specific meal ideas.
Exercise considerations
Resistance training is non-negotiable during significant weight loss on tirzepatide. Without it, approximately 30 to 40% of weight lost comes from lean muscle mass rather than fat. Resistance training shifts that ratio dramatically in favor of fat loss while preserving metabolically active muscle tissue. Even two to three sessions per week of basic compound movements (squats, deadlifts, rows, presses) can make a meaningful difference.
Cardiovascular exercise supports heart health, mood, and caloric expenditure. However, on a significantly reduced caloric intake, intense cardio can exacerbate fatigue and hunger. Moderate intensity activities like walking, swimming, or cycling are typically better tolerated than high-intensity interval training during the initial months of treatment.
Sleep and stress management
Poor sleep increases ghrelin (the hunger hormone) and decreases leptin (the satiety hormone), partially counteracting the appetite-suppressing effects of tirzepatide. Aim for seven to nine hours of quality sleep. Address sleep issues proactively, as they can significantly impact weight loss outcomes.
Chronic stress elevates cortisol, which promotes fat storage, particularly in the abdominal area, and can increase cravings for high-calorie comfort foods. While tirzepatide helps suppress these cravings pharmacologically, managing stress through exercise, mindfulness, or other strategies enhances the overall treatment effect.
How compounded tirzepatide with B12 compares to semaglutide formulations
Many patients considering compounded tirzepatide with B12 are also evaluating semaglutide-based alternatives, including compounded semaglutide and semaglutide with B12. Understanding the key differences helps inform your decision.
Mechanism differences
Tirzepatide activates both GLP-1 and GIP receptors (dual agonist). Semaglutide activates only the GLP-1 receptor (single agonist). This dual mechanism is why tirzepatide has consistently produced greater weight loss in head-to-head clinical trials. The SURMOUNT trials showed approximately 21% body weight reduction with tirzepatide, compared to approximately 15% with semaglutide in the STEP trials. For a detailed side-by-side analysis, see our semaglutide versus tirzepatide side effects comparison.
B12 formulation availability
Both compounded tirzepatide and compounded semaglutide are available with B12 additions. The rationale for B12 is similar in both cases: GLP-1 receptor activation slows gastric emptying, which may impair B12 absorption over time. Compounded semaglutide with methylcobalamin is another popular formulation that uses the active form of B12 rather than cyanocobalamin.
Regulatory status
The regulatory landscape for compounded semaglutide differs slightly from compounded tirzepatide. Semaglutide was also placed on the FDA shortage list, removed, and subject to similar compounding restrictions. The legal arguments for non-copy formulations (such as adding B12) apply to both compounds. However, the specific enforcement actions and court rulings have followed different timelines and affected different pharmacies.
Switching considerations
Some patients switch between semaglutide and tirzepatide, either for efficacy reasons or because one becomes unavailable through their pharmacy. If you are considering a switch, the semaglutide to tirzepatide conversion chart provides guidance on dose equivalencies. Note that there is no direct milligram-to-milligram conversion between the two medications because they have different potencies and mechanisms.
Frequently asked questions
Is compounded tirzepatide with B12 FDA approved?
No. Compounded tirzepatide with B12 is not FDA approved. The FDA-approved forms of tirzepatide (Mounjaro and Zepbound) contain only tirzepatide as the active ingredient and do not include B12. Compounded formulations are prepared by licensed pharmacies under different regulatory frameworks (503A or 503B) and have not undergone the clinical trial process required for FDA approval. This does not mean they are unsafe, but it does mean the specific combination has not been studied in large-scale clinical trials.
Can I get compounded tirzepatide with B12 without a prescription?
No. Tirzepatide is a prescription medication regardless of whether it is brand-name or compounded. You must have a valid prescription from a licensed healthcare provider. Many telehealth platforms now facilitate the prescribing process, but a legitimate medical evaluation must occur before a prescription is issued. Be wary of any service that offers tirzepatide without a proper medical consultation.
Does the B12 in compounded tirzepatide actually make a difference?
It depends on your individual B12 status. If you are B12 deficient or at risk of deficiency (due to diet, age, GI conditions, or long-term medication use), the injectable B12 provides genuine benefit by bypassing digestive absorption issues. If your B12 levels are already normal and you eat a varied diet, the B12 component provides minimal additional benefit. The most honest answer is that the B12 is a reasonable addition with legitimate rationale but modest clinical impact for many patients.
What is the difference between compounded tirzepatide with B12 and the tirzepatide glycine B12 compound?
The tirzepatide glycine B12 compound includes glycine as a third ingredient alongside tirzepatide and B12. Glycine serves as a stabilizer for the formulation and provides supplemental amino acid for muscle preservation. The term "compounded tirzepatide with B12" may or may not include glycine depending on the specific pharmacy formulation. Most compounding pharmacies now include glycine in their standard tirzepatide/B12 preparations.
How should I inject compounded tirzepatide with B12?
Compounded tirzepatide with B12 is typically injected subcutaneously (into the fatty tissue just beneath the skin) in the abdomen, thigh, or upper arm. Rotate injection sites each week to prevent tissue irritation. Our GLP-1 injection guide provides step-by-step instructions for proper technique, including site selection, needle angle, and post-injection care.
Can I switch from brand-name tirzepatide to compounded tirzepatide with B12?
Yes, switching is possible with provider guidance. The tirzepatide component works the same way regardless of the source. The main considerations are ensuring the compounded product concentration allows for accurate dosing of your prescribed amount and understanding that you will be switching from an autoinjector pen to manual syringe injection from a multi-dose vial. Your provider should supervise the transition.
What happens if I stop taking compounded tirzepatide with B12?
Discontinuing tirzepatide typically results in a return of appetite, gradual weight regain, and reversal of metabolic improvements over a period of months. This is not unique to the compounded B12 formulation but applies to all forms of tirzepatide. Studies show that patients who discontinue tirzepatide regain approximately two-thirds of lost weight within one year. For information on GLP-1 discontinuation effects, see our detailed guide. The B12 supplementation would also stop, so if you were previously deficient, you would need an alternative B12 source.
Is compounded tirzepatide with B12 safe during pregnancy or breastfeeding?
No. Tirzepatide has not been adequately studied in pregnant or breastfeeding women and is not recommended during either condition. If you become pregnant while using tirzepatide, discontinue immediately and contact your healthcare provider. The B12 component alone is safe during pregnancy and breastfeeding, but the tirzepatide is not. For related information, see our guide on GLP-1 and breastfeeding.
External resources
For researchers serious about optimizing their weight management protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, personalized protocol builders, and a community of thousands who have navigated these exact questions about compounded tirzepatide formulations and GLP-1 therapy.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay potent, your energy stay steady, and your progress stay consistent.