Mar 24, 2026

If you have lupus and you are considering tirzepatide, stop scrolling through Reddit threads and half-baked blog posts. The stakes are too high for guesswork. Systemic lupus erythematosus already attacks your body from the inside. The medications that keep it in check often cause weight gain that makes everything worse. And now there is a class of medications that might actually help with both the weight and the inflammation, but the research is still catching up to the excitement.
Here is what makes this situation uniquely complicated. Lupus patients do not just need to lose weight. They need to lose weight without triggering a flare, without interfering with immunosuppressants, and without adding another layer of side effects to an already complex medication regimen.
Tirzepatide, the dual GIP and GLP-1 receptor agonist behind brand names like Mounjaro and Zepbound, has shown remarkable results in the general population. But lupus is not the general population.
This guide covers everything currently known about tirzepatide and lupus, from the emerging research on GLP-1 medications and autoimmune conditions to practical drug interaction considerations, monitoring protocols, and what the actual clinical data shows about safety in SLE patients. Whether your rheumatologist brought this up or you are bringing it to them, you will leave this page with the complete picture.
Understanding lupus and the weight challenge
Lupus is not one disease. It is a spectrum.
Systemic lupus erythematosus affects virtually every organ system in the body. The immune system, instead of protecting you, turns against your own tissues. Joints, skin, kidneys, heart, lungs, brain, blood cells. Nothing is off limits. And while the disease itself creates chronic inflammation that disrupts metabolism, the treatments often make weight management even harder.
Approximately 76% of lupus patients experience weight gain after starting corticosteroids. That is not a typo. Three out of four people on prednisone or prednisolone will gain weight, sometimes 10 pounds, sometimes closer to 30. The mechanism is straightforward but cruel: corticosteroids increase appetite while simultaneously slowing metabolism and promoting fat storage, particularly around the abdomen, face, and back. The very medications keeping lupus under control are actively working against body composition.
Why weight matters more with lupus
For someone without an autoimmune condition, carrying extra weight is a health concern. For someone with lupus, it is a disease amplifier. Adipose tissue is not inert storage. It is an active endocrine organ that produces inflammatory cytokines, the same types of molecules that drive lupus flares. More body fat means more baseline inflammation, which means a lower threshold for disease activation.
Research shows that women with lupus experience the negative effects of obesity at a lower BMI than the general population. Where a BMI of 30 typically marks the obesity threshold, lupus patients may see complications starting at a BMI of 26.8 or higher. This means that even moderate weight gain carries disproportionate risk, affecting joint pain severity, cardiovascular health, kidney function, and overall disease activity.
The prevalence of obesity in SLE patients ranges from 28% to 50%, similar to or higher than the general population. But the consequences are amplified. Lupus patients already face elevated cardiovascular risk, a two to three fold increase compared to age-matched controls. Adding obesity to that equation compounds the danger significantly. This is why finding safe, effective weight management strategies for lupus patients is not vanity. It is disease management.
The exercise paradox
Tell someone to exercise more and eat less. Simple advice. Terrible for lupus patients.
Joint pain makes movement difficult. Fatigue, one of the most debilitating and underrecognized symptoms of lupus, makes it hard to get through a normal day, let alone add a workout. Depression, which affects up to 40% of SLE patients, undermines motivation and decision-making around food. During flares, metabolism slows dramatically as the body diverts energy to fighting the autoimmune response. And then the corticosteroid dose gets increased to manage the flare, restarting the weight gain cycle all over again.
This is the trap. Lupus causes inflammation. Inflammation causes weight gain. Weight gain causes more inflammation. Corticosteroids manage the inflammation but cause more weight gain.
Traditional weight loss approaches fail because lupus undermines the fundamental assumptions behind them, that the body will respond normally to calorie restriction and exercise. Understanding this trap is what makes tirzepatide and its benefits beyond weight loss particularly interesting for this population.
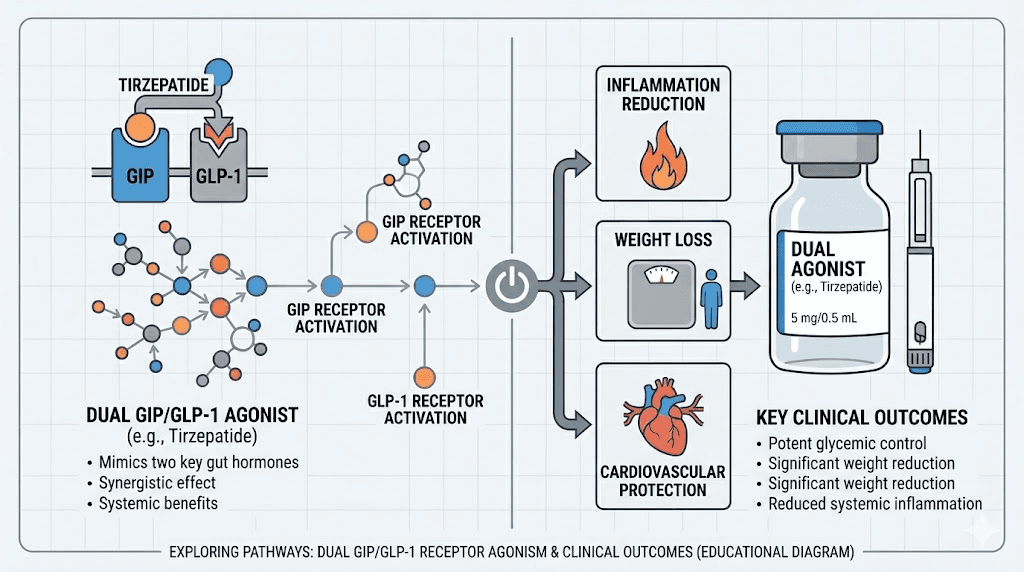
How tirzepatide works and why it matters for autoimmune conditions
Tirzepatide is not just another weight loss drug. It represents a fundamentally different approach to metabolic regulation, and that distinction matters enormously for lupus patients.
Most GLP-1 receptor agonists like semaglutide target a single receptor. Tirzepatide targets two: the GLP-1 receptor and the GIP receptor. This dual mechanism, sometimes called a "twincretin," produces effects that go well beyond appetite suppression. The comparison between semaglutide and tirzepatide becomes especially relevant when evaluating options for complex medical situations.
The GLP-1 pathway
GLP-1, or glucagon-like peptide-1, is a hormone your gut naturally produces after eating. It signals the brain to reduce hunger, slows gastric emptying so you feel full longer, and improves insulin sensitivity. When tirzepatide activates GLP-1 receptors, it amplifies these natural signals far beyond what food alone triggers.
But GLP-1 receptors are not just in the gut and brain. They exist on immune cells, in the kidneys, in the heart, and throughout the vascular system. This widespread distribution is exactly why GLP-1 receptor agonists show effects that extend far beyond weight management. For lupus patients, these extra-metabolic effects may be just as important as the number on the scale. Understanding how peptides work in the body provides additional context for these mechanisms.
The GIP pathway
Glucose-dependent insulinotropic polypeptide, or GIP, adds another dimension. GIP receptors are found on adipose tissue, bone, and the cardiovascular system. Activating these receptors improves fat metabolism, may protect bone density (a critical concern for lupus patients on corticosteroids), and provides additional cardiovascular benefits.
The combination of GLP-1 and GIP activation is what makes tirzepatide produce faster and more significant results than single-target GLP-1 medications. In clinical trials for weight management, tirzepatide produced average weight loss of 20-25% of body weight, roughly double what semaglutide achieves at comparable endpoints. For lupus patients trapped in the inflammation-weight-corticosteroid cycle, that magnitude of effect could be transformative.
Tirzepatide and inflammation: what the research shows
This is where things get genuinely exciting for anyone managing an autoimmune condition.
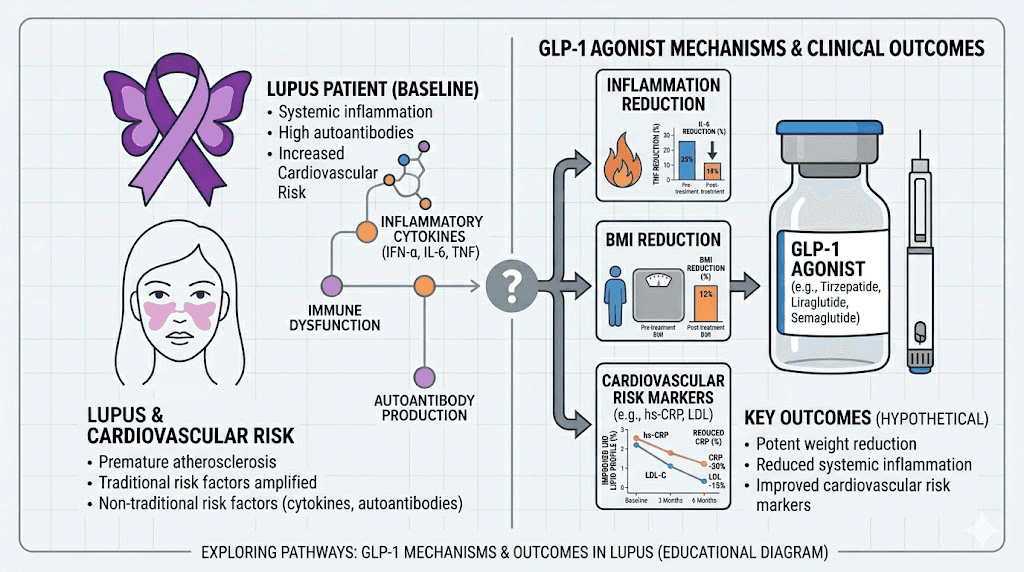
A systematic review and meta-analysis examining tirzepatide across seven randomized clinical trials and one observational study found something remarkable. Tirzepatide use is associated with a significant reduction in inflammatory markers, regardless of the population studied or treatment regimen. Not a small reduction. Not a marginal trend. A statistically robust, dose-dependent decrease in the very biomarkers that drive autoimmune disease activity.
C-reactive protein reduction
High-sensitivity C-reactive protein, or hsCRP, is one of the primary markers rheumatologists use to track inflammation in lupus patients. Elevated hsCRP correlates with disease activity, cardiovascular risk, and kidney involvement. The meta-analysis found that tirzepatide reduced hsCRP compared to placebo with a mean difference of -32.9 (95% CI: -33.6 to -32.2). The effect was dose-dependent.
At the 5mg dose, the reduction was -20.3. At 10mg, it dropped to -33.9. At 15mg, it reached -32.9 with remarkably low heterogeneity between studies. These are not subtle shifts. For a lupus patient whose hsCRP runs chronically elevated, this kind of reduction could meaningfully affect disease trajectory. Tracking these markers alongside standard tirzepatide blood work monitoring becomes especially important.
Interleukin-6 reduction
IL-6 is perhaps even more relevant to lupus than CRP. Interleukin-6 is a pro-inflammatory cytokine directly implicated in SLE pathogenesis. It drives B cell hyperactivity, promotes autoantibody production, and contributes to the renal, cardiovascular, and neuropsychiatric complications of lupus. Reducing IL-6 does not just lower a number on a lab report. It potentially addresses one of the upstream drivers of the disease itself.
Tirzepatide reduced IL-6 compared to placebo with a mean difference of -17.8 (95% CI: -24.3 to -11.3). This effect was consistent across all doses tested. At 5mg: -18.8. At 10mg: -17.9. At 15mg: -16.8. The consistency across doses suggests this is not simply a downstream effect of weight loss but may represent a direct pharmacological action on inflammatory pathways.
For lupus patients, who often have chronically elevated IL-6 and TNF-alpha levels, these findings suggest tirzepatide may offer anti-inflammatory benefits that complement rather than conflict with existing immunomodulatory therapy. Research suggests GLP-1 medications may actually help regulate immune responses rather than suppress them, potentially inhibiting the NF-kB pathway, one of the central signaling cascades in autoimmune inflammation.
What this means practically
Two important caveats. First, reducing inflammatory markers is not the same as treating lupus. Tirzepatide is not and should not be considered a disease-modifying antirheumatic drug. Second, the anti-inflammatory effects observed in these studies occurred primarily in populations without autoimmune diseases, mostly people with type 2 diabetes or obesity. Whether the same magnitude of inflammation reduction translates to lupus patients with different baseline immune profiles remains to be established.
That said, the direction of the evidence is encouraging. A medication that helps with weight loss AND reduces systemic inflammation AND improves cardiovascular markers addresses multiple aspects of the lupus burden simultaneously. That is a rare combination, and it is why rheumatologists are paying attention even though no randomized controlled trials specifically evaluating tirzepatide for autoimmune conditions exist yet.
Clinical evidence: GLP-1 medications in lupus patients
The research specifically examining GLP-1 receptor agonists in lupus patients is limited but growing. Two studies in particular deserve close examination because they represent the best available evidence for making clinical decisions.
The NYU lupus cohort study
Published in Rheumatology (Oxford) by Carlucci and colleagues, this retrospective evaluation examined GLP-1 receptor agonist use within the NYU Lupus Cohort, a prospective longitudinal convenience cohort initiated in 2014 with 1,211 SLE patients.
Of those 1,211 patients, 24 had used a GLP-1 receptor agonist. After excluding six with insufficient documentation, 18 patients formed the analysis group. The demographics: median age 50 years, 94% female, 50% White, 39% Black, 11% Hispanic. All 18 used GLP-1 RAs for weight loss. Only one had type 2 diabetes.
The results were cautiously encouraging.
At baseline, 14 of 18 patients (78%) were in DORIS remission, the Definition Of Remission In SLE. During follow-up on GLP-1 therapy, no patients accumulated new SLE criteria. One mild-to-moderate flare was observed at 8 months. One patient developed recurrent leukopenia at 1-4 months. Three patients (27%) developed low complement and/or anti-dsDNA antibodies at 6-10 months. Two patients (18%) developed alopecia at 6-10 months.
The weight loss results were significant. Median BMI reduction at 1-4 months was 3% (P = 0.002). At 6-10 months, the reduction reached 13% (P = 0.001), with an absolute reduction of 5.6 kg/m2. These numbers are meaningful for a population that traditionally struggles with weight management due to medications and disease activity.
The study concluded that GLP-1 RAs did not trigger new clinical SLE manifestations above expected background rates while producing significant weight loss benefits. But the authors appropriately noted limitations: the retrospective design, small sample size, and the fact that most patients were already in remission when starting therapy.
The Jorge cardiovascular and kidney outcomes study
A larger study published in Arthritis and Rheumatology by Jorge and colleagues in 2026 took a different and arguably more impactful approach. Instead of looking at disease activity, they examined hard cardiovascular and kidney outcomes in lupus patients using GLP-1 receptor agonists versus DPP-4 inhibitors.
The findings were striking. GLP-1 receptor agonists were associated with markedly lower risks of major adverse cardiovascular events, progression of kidney disease, venous thromboembolism, and all-cause mortality compared to DPP-4 inhibitor therapy. Similar reductions were observed among patients with lupus nephritis specifically.
This matters enormously because cardiovascular disease and kidney damage are the leading causes of morbidity and mortality in lupus. If GLP-1 medications reduce these risks, even partially, the implications for long-term lupus management are profound. The 2025 EULAR recommendations for SLE with kidney involvement now acknowledge that EAS-guided prescriptions of GLP-1 receptor agonists could reduce the burden of cardiovascular disease in SLE populations.
Safety considerations: can lupus patients take tirzepatide?
The short answer is: likely yes, with appropriate medical supervision. The longer answer requires understanding several interconnected safety concerns.
Flare risk
The primary fear for any lupus patient starting a new medication is triggering a flare. The NYU study provides the most direct evidence available, and it is reassuring. Of 18 SLE patients on GLP-1 medications, only one experienced a mild-to-moderate flare during follow-up. This rate does not exceed what would be expected as background flare activity in a lupus cohort, even without any new medication.
However, three patients did develop serological changes (low complement and/or positive anti-dsDNA) at 6-10 months without corresponding clinical symptoms. This is important because serological activity can precede clinical flares by weeks or months. It underscores the need for more frequent monitoring when starting tirzepatide, a point we will return to in the monitoring section.
Drug-induced lupus concern
One case report from 2023 described a potential link between semaglutide and drug-induced lupus. Drug-induced lupus is a distinct entity from SLE. It is caused by medication exposure and typically resolves when the offending drug is discontinued. This single case does not establish causation, but it raises a flag that clinicians should be aware of, particularly in patients with pre-existing SLE or lupus-like features.
No similar reports have been published specifically for tirzepatide. The dual mechanism may or may not carry different immunological implications than single-target GLP-1 agonists. Without dedicated research, this remains an area of uncertainty. If you are considering tirzepatide versus semaglutide side effect profiles, this distinction becomes relevant for autoimmune patients.
Immune regulation versus immune suppression
A critical distinction that often gets lost in discussions about GLP-1 medications and autoimmunity. These drugs do not suppress the immune system the way corticosteroids, methotrexate, or mycophenolate do. Research suggests GLP-1 medications may help regulate immune responses rather than suppress them.
The proposed mechanism involves inhibition of the NF-kB pathway, a master regulator of inflammatory gene expression. Rather than broadly dampening immune function (which would increase infection risk), this pathway-specific effect may selectively reduce the aberrant inflammatory signaling that drives autoimmune disease. This is a fundamentally different approach from traditional immunosuppression and one that could theoretically complement existing lupus therapies.
Understanding how peptides interact with autoimmune conditions broadly provides useful context for evaluating these mechanisms.
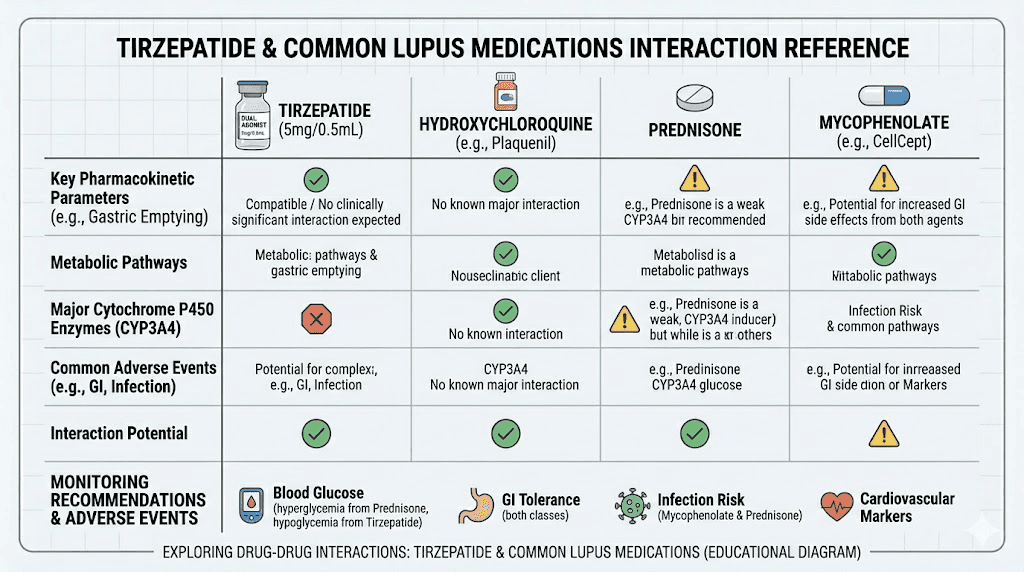
Drug interactions with common lupus medications
Lupus patients rarely take just one medication. A typical regimen might include an antimalarial, a corticosteroid, an immunosuppressant, and possibly a biologic. Each of these needs to coexist safely with tirzepatide.
Hydroxychloroquine (Plaquenil)
Hydroxychloroquine is the backbone of lupus treatment. Nearly every SLE patient takes it, and guidelines recommend it be continued indefinitely. The interaction with tirzepatide is theoretical but worth noting: both medications can potentially affect cardiac rhythm. Hydroxychloroquine carries a small risk of QT prolongation, and while tirzepatide has not shown direct cardiac conduction effects, the combination warrants awareness.
The more practical concern is absorption. Tirzepatide delays gastric emptying, which is how it helps control appetite. This delay can theoretically affect the absorption timing of oral medications. Hydroxychloroquine has a long half-life (40-50 days) and a wide therapeutic window, so minor absorption delays are unlikely to be clinically significant. But if you notice changes in lupus symptoms after starting tirzepatide, altered hydroxychloroquine absorption is one possibility to discuss with your rheumatologist.
Corticosteroids (prednisone, prednisolone)
This is actually one of the more promising combinations. Corticosteroids cause weight gain through increased appetite and metabolic disruption. Tirzepatide directly counteracts the appetite-stimulating effects of corticosteroids while simultaneously improving the metabolic dysfunction they create. Multiple clinicians have noted that the appetite suppression from GLP-1 medications effectively manages steroid-induced hunger in a way that willpower alone cannot.
There are no known direct pharmacological interactions between tirzepatide and prednisone. Both affect glucose metabolism, but in complementary directions: corticosteroids raise blood sugar while tirzepatide lowers it. This could theoretically allow for better glycemic control in lupus patients requiring steroid therapy, though blood glucose monitoring is advisable when combining them.
Mycophenolate (CellCept/Myfortic)
Mycophenolate is widely used for lupus nephritis and moderate-to-severe systemic disease. It is absorbed in the upper GI tract, making it potentially sensitive to changes in gastric emptying. Since tirzepatide slows gastric emptying, there is a theoretical concern about delayed mycophenolate absorption. However, no clinical studies have documented this interaction specifically, and mycophenolate does not have the narrow therapeutic index that would make absorption timing critical.
If you are on mycophenolate and starting tirzepatide, your rheumatologist may want to check mycophenolate levels more frequently during the initial titration period. Tracking what to expect after your first tirzepatide dose helps establish a baseline for distinguishing drug effects from disease effects.
Methotrexate
No direct interaction exists between tirzepatide and methotrexate. Both medications can affect liver function, so more frequent liver enzyme monitoring may be prudent when using them together. Methotrexate nausea and tirzepatide nausea could potentially compound each other, particularly during the early titration phase. If tirzepatide-related GI symptoms become problematic, timing the medications on different days may help.
Azathioprine (Imuran)
Azathioprine has no known direct interaction with tirzepatide. The primary concern is additive nausea, as both medications list nausea as a common side effect. Spacing administration times can help. Azathioprine requires monitoring of complete blood counts, and tirzepatide does not affect hematological parameters, so no additional blood work beyond standard monitoring should be necessary.
Belimumab (Benlysta) and other biologics
Belimumab, the first biologic approved specifically for lupus, targets B-lymphocyte stimulator. There are no known interactions with GLP-1 receptor agonists. Both medications modulate immune function through entirely different pathways. The same applies to anifrolumab (Saphnelo), voclosporin (Lupkynis), and rituximab, all of which are used in various lupus presentations.
The lack of pharmacological overlap between tirzepatide and lupus biologics is actually one of the strongest arguments for its safety in this population. Tirzepatide does not undergo cytochrome P450 metabolism, which eliminates an entire category of potential drug interactions that plague other medications.
Dosing considerations for lupus patients
Standard tirzepatide dosing may not be the right approach for everyone with lupus. The principle of "start low, go slow" applies with particular force here.
Standard titration versus conservative approach
The standard tirzepatide titration starts at 2.5mg weekly for 4 weeks, then increases to 5mg, with subsequent increases to 7.5mg, 10mg, 12.5mg, and potentially 15mg at 4-week intervals. For someone without autoimmune considerations, this schedule is well-established and generally well-tolerated.
For lupus patients, many clinicians recommend a more conservative approach. This might mean extending the time at each dose level to 6-8 weeks instead of 4, allowing more time to assess both tolerability and disease activity at each level. Some may start with even lower doses if compounded formulations are available. The compounded tirzepatide starting dose guide provides more detail on flexible dosing options.
The rationale is straightforward: each dose increase changes the metabolic and inflammatory environment. Lupus patients need time at each level to ensure their disease remains stable before adding more metabolic disruption. A tirzepatide dosing guide in units can help with precise dose calculations.
The microdosing approach
Some clinicians are exploring microdosing of GLP-1 medications specifically for autoimmune patients. The concept involves using doses significantly below the standard therapeutic range, not primarily for weight loss but for the anti-inflammatory and immune-regulatory effects.
The theory is that lower doses may provide the inflammatory marker reductions documented in research while minimizing GI side effects and metabolic disruption. For lupus patients who do not necessarily need dramatic weight loss but could benefit from reduced inflammation and improved cardiovascular markers, microdosing may offer a favorable risk-benefit ratio. Detailed protocols are available in our guide to microdosing tirzepatide and the microdosing tirzepatide chart.
Important: microdosing for autoimmune conditions is not established by clinical trials. This is an off-label application based on mechanistic reasoning and preliminary observations. It should only be considered under medical supervision with appropriate monitoring.
Finding the right maintenance dose
Not every lupus patient needs to reach the maximum 15mg dose. The goal is to find the lowest effective dose that achieves the desired outcomes, whether that is primarily weight loss, inflammation reduction, or both. Some patients find their optimal balance at 5mg or 7.5mg without needing higher doses. Understanding GLP-1 maintenance dosing principles helps inform this decision.
Factors that influence the right dose include baseline body weight, corticosteroid dose (higher steroid doses may require higher tirzepatide doses to overcome steroid-induced appetite stimulation), disease activity level, and individual tolerability. The tirzepatide dosage calculator can assist with initial dose planning.
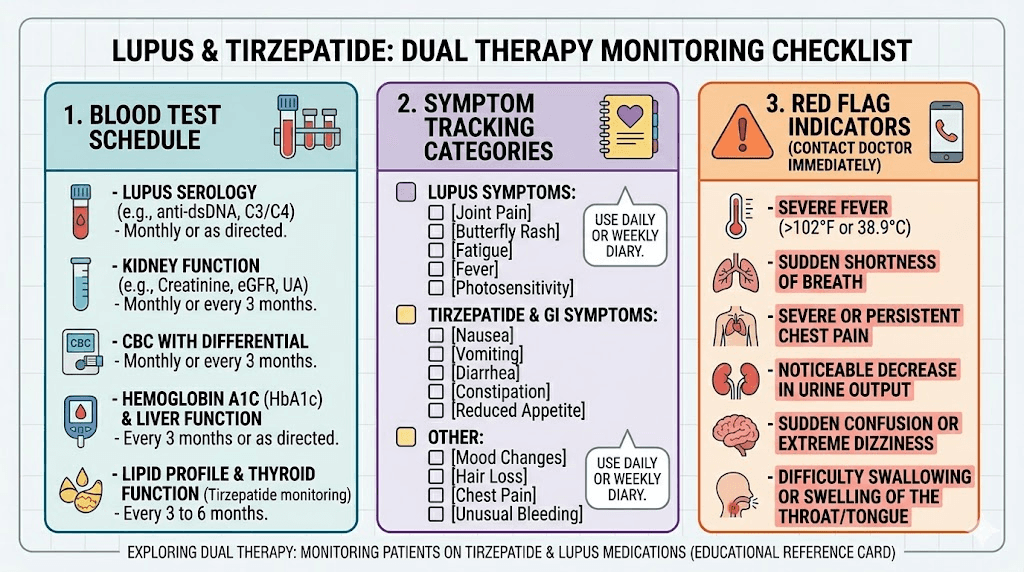
Monitoring protocols for lupus patients on tirzepatide
Standard tirzepatide monitoring is not sufficient for lupus patients. The overlap between medication side effects and disease symptoms demands a more comprehensive approach.
Recommended monitoring schedule
For the first three months, blood work every 2-3 months is advisable rather than the standard 3-6 month interval. After disease stability is confirmed on tirzepatide, the interval can potentially extend to match standard lupus monitoring schedules.
The monitoring panel should include:
Standard lupus labs: Complete blood count with differential, comprehensive metabolic panel, urinalysis with protein-to-creatinine ratio, complement levels (C3, C4), anti-dsDNA antibodies, ESR, and hsCRP. These track disease activity and catch early signs of flare or organ involvement.
Metabolic labs: Fasting glucose, HbA1c, lipid panel, liver enzymes (ALT, AST). Tirzepatide affects glucose metabolism significantly, and corticosteroids do as well. Monitoring both sides of this equation is important.
Thyroid function: TSH at baseline and periodically. GLP-1 receptor agonists carry a theoretical risk of thyroid C-cell effects, and lupus patients have higher rates of thyroid autoimmunity. The relationship between tirzepatide and thyroid health deserves attention in this context.
Kidney function: Estimated GFR, BUN, creatinine. Lupus nephritis affects up to 50% of SLE patients at some point. Tirzepatide appears to have kidney-protective effects based on the Jorge study, but monitoring ensures this holds true individually.
Symptom tracking
This is where lupus makes tirzepatide monitoring uniquely challenging. Many common tirzepatide side effects overlap with lupus symptoms.
Fatigue from GLP-1 medications versus lupus fatigue? Hard to distinguish. Joint pain from tirzepatide versus lupus arthritis? Requires clinical judgment. Hair loss on GLP-1 therapy versus lupus alopecia? Both are common. Nausea from tirzepatide versus lupus-related GI issues? The differential diagnosis gets complex quickly.
Keeping a detailed symptom diary is not optional for lupus patients on tirzepatide. It is essential. Record daily energy levels, joint symptoms, skin changes, GI symptoms, and any new or worsening complaints. Note when they occur relative to your tirzepatide injection day. Over time, patterns emerge that help distinguish drug effects from disease activity.
Cardiovascular and kidney protection
If you have lupus, your cardiovascular risk is not hypothetical. It is one of the most significant threats to your long-term health, and it is where tirzepatide may offer its most meaningful benefits beyond weight loss.
The cardiovascular burden of lupus
SLE patients face a two to three fold increased risk of cardiovascular events compared to the general population. The reasons are multifactorial: chronic inflammation damages blood vessel walls, corticosteroid use raises cholesterol and blood pressure, lupus-specific antibodies (particularly antiphospholipid antibodies) increase clotting risk, and the metabolic disruption caused by both the disease and its treatments creates a perfect storm for atherosclerosis.
Traditional cardiovascular risk calculators underestimate risk in lupus patients because they were not designed for populations with chronic systemic inflammation. A 35-year-old woman with lupus may have the cardiovascular risk profile of a 55-year-old without it. This is not an exaggeration. It is reflected in mortality data showing that cardiovascular disease is a leading cause of death in SLE, particularly in patients who survive the first five years after diagnosis.
How tirzepatide addresses cardiovascular risk
The Jorge 2026 study demonstrated that GLP-1 receptor agonists were associated with significantly lower risks of major adverse cardiovascular events in lupus patients compared to DPP-4 inhibitors. This included reductions in:
Major adverse cardiovascular events (heart attack, stroke, cardiovascular death). Venous thromboembolism, a particularly relevant risk for lupus patients with antiphospholipid antibodies. All-cause mortality.
These are not surrogate endpoints. They are the outcomes that matter most. And while the study compared GLP-1 RAs broadly (not tirzepatide specifically), tirzepatide is a GLP-1 RA with additional GIP activity that may confer extra cardiovascular benefit. The metabolic improvements from tirzepatide contribute to this cardiovascular protection through multiple pathways.
Kidney protection in lupus nephritis
Lupus nephritis, inflammation of the kidneys caused by lupus, affects up to 50% of SLE patients. It is one of the most serious organ manifestations and a leading cause of kidney failure in young adults. Managing kidney disease requires a delicate balance: enough immunosuppression to control inflammation, but not so much that infection risk becomes unacceptable.
The Jorge study found that GLP-1 receptor agonists were associated with reduced progression of kidney disease in SLE patients, including those with lupus nephritis. Preliminary real-world data suggests that these medications may reduce progression to chronic kidney disease, potentially by inhibiting the NF-kB inflammatory pathway that drives renal damage.
This is significant because current lupus nephritis management relies heavily on immunosuppressants (mycophenolate, cyclophosphamide, voclosporin) and biologics (belimumab, rituximab). Adding a medication that provides renal protection through an entirely different mechanism, without additional immunosuppression, could meaningfully improve outcomes. The broader role of peptides in lupus management is an evolving field worth following.
Side effects: what lupus patients should watch for
Every medication has side effects. But lupus patients need to view tirzepatide side effects through a different lens than the general population because some effects that are merely annoying for healthy individuals could signal something more significant in SLE.
Gastrointestinal effects
Nausea is the most common tirzepatide side effect, affecting 12-18% of users in clinical trials. For lupus patients, GI symptoms deserve extra scrutiny because lupus can independently cause GI inflammation, gastroparesis, and serositis. If nausea is new or significantly worse than expected, consider whether it could represent lupus GI involvement rather than a simple medication side effect.
Constipation and diarrhea are also common. Tirzepatide slows gastric emptying, which can worsen constipation, a side effect already seen with some lupus medications. Lupus can also cause a form of gastroparesis by affecting the nerves controlling digestive movement. If you already have lupus-related GI motility issues, tirzepatide may exacerbate them. The GLP-1 constipation relief guide offers practical management strategies.
Fatigue and energy changes
Tiredness on tirzepatide is reported by many users, particularly during caloric restriction. For lupus patients, fatigue is already one of the most common and debilitating symptoms. Adding medication-related fatigue to lupus-related fatigue could significantly impact quality of life.
However, some patients report the opposite effect. As weight drops and metabolic health improves, overall energy increases. The question of whether tirzepatide gives you energy varies by individual. For lupus patients, the trajectory of energy changes can help determine whether the medication is helping or hindering overall disease management.
Musculoskeletal effects
Body aches and muscle pain are reported side effects of tirzepatide. In the general population, these are usually transient and mild. In lupus patients, they could be confused with disease flares. Arthralgia and myalgia are hallmark lupus symptoms, and distinguishing medication-related musculoskeletal complaints from disease activity requires careful clinical assessment.
The key differentiator: tirzepatide-related body aches tend to be diffuse, bilateral, and not associated with joint swelling or serological changes. Lupus arthritis typically involves specific joints (hands, wrists, knees), may show visible swelling, and often correlates with changes in complement levels or anti-dsDNA titers. When in doubt, check labs.
Hair loss
Weight loss from any cause can trigger telogen effluvium, a form of diffuse hair shedding that occurs 2-4 months after a physiological stress like rapid weight loss. GLP-1 medications, including tirzepatide, have been associated with this effect. But lupus also causes alopecia, both the diffuse type and a more concerning scarring variant.
If hair loss develops while on tirzepatide, it is crucial to determine whether it is telogen effluvium (non-scarring, diffuse, reversible) or lupus-related alopecia (which may be scarring and requires different management). A dermatology referral and possibly a scalp biopsy may be warranted. The GLP-1 hair loss guide covers management strategies for medication-related shedding.
Injection site reactions
Injection site reactions with tirzepatide include redness, itching, and localized swelling. For lupus patients, skin reactions can be more significant because lupus itself causes various skin manifestations. A new rash at or near an injection site could be a simple local reaction, but it could also represent a lupus skin flare triggered by tissue trauma (the pathergy phenomenon). If injection sites become persistently red and itchy, consider rotating sites more frequently and consulting your rheumatologist.
Choosing optimal injection sites and following proper injection technique can minimize local reactions. The thigh injection site may be preferable for patients who experience abdominal skin sensitivity from lupus.
Practical protocols: starting tirzepatide with lupus
If you and your medical team decide to proceed with tirzepatide, here is a practical framework based on current evidence and clinical experience.
Pre-start checklist
Before the first injection, establish comprehensive baseline data. This is not the time to wing it.
Baseline labs: CBC with differential, CMP, urinalysis with protein/creatinine ratio, C3 and C4 complement, anti-dsDNA, ESR, hsCRP, HbA1c, fasting glucose, lipid panel, TSH, liver enzymes. These serve as the reference point for all future monitoring.
Disease activity assessment: Have your rheumatologist document your current SLEDAI or BILAG score. This objective measure of disease activity at baseline lets you track whether tirzepatide is associated with any changes in lupus activity over time.
Medication reconciliation: List every medication, supplement, and over-the-counter product you take. Discuss timing with your prescriber, especially for medications with narrow therapeutic windows. The supplements to take with tirzepatide guide includes compatibility considerations for various combinations.
Weight and body composition: Record baseline weight, BMI, and waist circumference. If possible, get a body composition assessment (DEXA scan or bioelectrical impedance). Lupus patients on corticosteroids often have disproportionate visceral fat even at lower body weights, and tracking composition changes is more informative than tracking weight alone.
Week 1-4: initiation phase
Start at 2.5mg weekly. This is the standard starting dose and it is primarily a tolerability assessment, not a therapeutic dose.
During this phase, monitor GI symptoms daily. Some nausea is expected and usually peaks 24-48 hours after injection. Track energy levels, joint symptoms, and any skin changes. Take your tirzepatide injection on the same day each week at a consistent time to establish a routine.
If GI symptoms are severe, review our foods to avoid on tirzepatide guide. Eating smaller, more frequent meals and avoiding high-fat foods in the hours after injection can help. The tirzepatide diet plan provides structured meal guidance.
Week 5-12: titration phase
For lupus patients, consider extending to 6-8 weeks at each dose level instead of the standard 4 weeks. This provides more time to assess disease stability before adding metabolic stress.
At week 6-8, repeat lupus-specific labs (complement, anti-dsDNA, CBC, urinalysis) even if you feel fine. Serological changes can precede clinical symptoms by weeks. If labs are stable, proceed to the next dose increase. If complement levels drop or anti-dsDNA rises, hold the current dose and repeat labs in 4 weeks. Do not increase the dose until serological stability is confirmed.
During titration, weight loss timelines may differ from what the general population experiences. Corticosteroid-induced weight can be stubborn, and the anti-inflammatory benefits may precede visible weight changes. Patience during this phase is critical. Your first month results might be modest, especially if you are on prednisone.
Month 3+: maintenance and optimization
By month 3, you should have a clearer picture of how tirzepatide interacts with your disease. Most patients settle into a maintenance dose between 5mg and 10mg, though some may benefit from higher doses.
This is when the anti-inflammatory benefits become more apparent. The meta-analysis data showed CRP and IL-6 reductions at study endpoints that were typically 12-26 weeks. If your rheumatologist observes decreasing inflammatory markers alongside weight loss, there may be an opportunity to reduce corticosteroid doses, which would further enhance weight loss in a positive feedback loop.
The question of maintaining weight loss after tirzepatide is relevant for long-term planning, as is understanding how to wean off tirzepatide if discontinuation becomes necessary.
Nutrition strategies for lupus patients on tirzepatide
Diet matters for everyone on tirzepatide. It matters doubly for lupus patients because food choices affect both medication tolerance and disease activity.
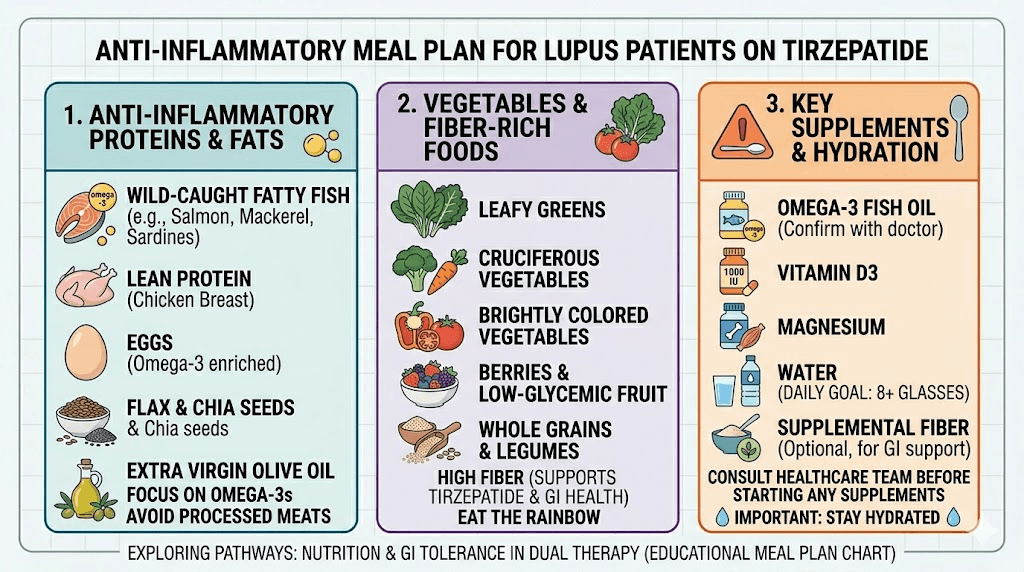
Anti-inflammatory eating
The Mediterranean diet pattern has the strongest evidence base for reducing inflammation in autoimmune conditions. Rich in omega-3 fatty acids, polyphenols, and fiber, it complements the anti-inflammatory effects of tirzepatide. For lupus patients specifically, fish, vegetables, olive oil, nuts, and whole grains form the foundation.
Certain foods can trigger lupus flares in susceptible individuals. Alfalfa sprouts contain L-canavanine, which has been shown to activate lupus in animal models and case reports. Excessive garlic can stimulate immune cells. High-sodium foods worsen fluid retention and blood pressure, already concerns with both lupus and corticosteroid use. The what to eat on tirzepatide guide provides a foundation that lupus patients can adapt.
Protein requirements
Adequate protein intake becomes critical when losing weight on tirzepatide. The appetite suppression can make it easy to under-eat, particularly protein. For lupus patients, protein is especially important because corticosteroids cause muscle wasting, and losing weight without adequate protein intake risks losing muscle mass rather than fat.
Aim for 0.8-1.0 grams of protein per pound of target body weight daily. Use protein shakes designed for GLP-1 users to supplement if solid food intake is limited by nausea. Front-load protein in the morning when appetite tends to be slightly better, and follow GLP-1 breakfast ideas that prioritize protein-dense options.
Nutrient considerations
Lupus patients are frequently deficient in vitamin D, which worsens both bone health and immune regulation. Corticosteroids further deplete vitamin D and calcium. Tirzepatide-induced caloric restriction can compound these deficiencies if supplementation is inadequate.
Essential supplements for lupus patients on tirzepatide include vitamin D (typically 2,000-5,000 IU daily, guided by blood levels), calcium (1,000-1,200mg daily from food and supplements combined), omega-3 fatty acids (for additional anti-inflammatory effect), and a quality multivitamin. The supplements to take with GLP-1 medications guide covers additional options. A fiber supplement can also help manage the constipation that sometimes accompanies tirzepatide use.
Meal planning and timing
Reduced appetite on tirzepatide makes meal planning more important, not less. Without intentional planning, it is easy to eat too little, skip meals, or rely on whatever requires the least effort. For lupus patients managing fatigue, meal prep on good energy days and freezing portions is a practical strategy.
Our tirzepatide meal plan can be adapted for anti-inflammatory eating. The GLP-1 dinner ideas and GLP-1 recipes for weight loss provide additional inspiration. Focus on nutrient-dense foods that address lupus-specific needs: fatty fish for omega-3s, leafy greens for folate and iron, and colorful vegetables for antioxidants.
The corticosteroid reduction opportunity
This may be the single most compelling reason for lupus patients to consider tirzepatide.
Corticosteroids are both the most effective and most damaging long-term medications in lupus management. They control inflammation rapidly but cause weight gain, bone loss, diabetes, cataracts, skin thinning, muscle wasting, mood changes, and increased infection risk. Every rheumatologist aims to minimize corticosteroid exposure. Every lupus patient knows the frustration of needing a medication that causes as many problems as it solves.
Tirzepatide creates a potential path to corticosteroid reduction through two mechanisms. First, the weight loss itself reduces inflammatory burden, which may allow lower steroid doses to maintain disease control. Second, the direct anti-inflammatory effects (reduced CRP, IL-6) provide an additional buffer against disease activity as steroid doses decrease.
This is not theoretical. Clinicians managing lupus patients on GLP-1 medications have reported successful prednisone tapers that were previously difficult or impossible. The pattern typically unfolds over months: tirzepatide produces weight loss and inflammation reduction, which allows a gradual steroid taper, which further improves weight and metabolic health, which may allow additional steroid reduction. It is a virtuous cycle that directly reverses the vicious cycle of steroids causing weight gain causing more inflammation causing higher steroid doses.
Any steroid taper must be done gradually under rheumatological supervision. Abrupt corticosteroid reduction can cause adrenal crisis and lupus flare. But the possibility of safely reducing long-term steroid exposure is a significant potential benefit of tirzepatide for this population.
Bone health considerations
Lupus patients on corticosteroids are at elevated risk for osteoporosis and fractures. Tirzepatide introduces both potential benefits and concerns in this area that deserve careful consideration.
The risk side
Rapid weight loss from any cause can reduce bone mineral density. Weight-bearing loads from body weight contribute to bone maintenance, and losing significant weight removes some of that mechanical stimulus. Combined with corticosteroid-induced bone loss, rapid weight loss could theoretically accelerate osteoporosis progression.
The question of whether tirzepatide causes bone loss is actively being studied. Current evidence is mixed, with some studies showing minimal bone density changes and others showing modest reductions in certain populations.
The benefit side
GIP receptor activation, the feature that distinguishes tirzepatide from single-target GLP-1 agonists like semaglutide, may actually benefit bone health. GIP receptors exist on osteoblasts (bone-building cells) and osteoclasts (bone-resorbing cells). Activating GIP receptors appears to promote bone formation and inhibit bone resorption, at least in preclinical studies.
Additionally, if tirzepatide enables corticosteroid dose reduction, the bone-protective effects of lower steroid exposure could outweigh any bone density loss from weight reduction. This is another area where the net effect depends on the individual patient and the magnitude of steroid taper achieved.
Practical bone protection
Regardless of the theoretical debates, lupus patients on tirzepatide should prioritize bone protection. Adequate calcium and vitamin D supplementation, weight-bearing exercise as tolerated (even walking counts), and baseline and follow-up DEXA scans to track bone density objectively. If osteopenia or osteoporosis is already present, bisphosphonate or denosumab therapy should be discussed with your medical team.
Mental health and tirzepatide with lupus
Lupus affects the brain. This is not metaphorical. Neuropsychiatric lupus is a recognized disease manifestation that can cause cognitive dysfunction, mood disorders, anxiety, and psychosis. Adding tirzepatide to this picture requires attention to mental health impacts.
Some patients on GLP-1 medications report mood changes, including anxiety and depressive symptoms. Whether these are direct pharmacological effects or secondary to caloric restriction, metabolic changes, or body image adjustment is not fully established. For lupus patients already managing neuropsychiatric symptoms, any new mood disturbance should be evaluated carefully.
On the positive side, weight loss and improved metabolic health often enhance mood, self-esteem, and energy in lupus patients. The psychological burden of corticosteroid-induced weight gain, the "moon face" and central obesity that many patients find distressing, can be partially reversed by tirzepatide. This cosmetic and functional improvement should not be underestimated as a quality-of-life factor.
If you take medications for depression or anxiety alongside lupus medications, discuss potential interactions with your prescriber. The wellbutrin and tirzepatide guide covers one common combination. Insomnia on tirzepatide is another potential issue worth monitoring, as sleep disruption compounds lupus fatigue.
Special populations within lupus
Not all lupus patients are the same, and certain subgroups deserve specific consideration.
Lupus nephritis
Patients with active or historical lupus nephritis represent the highest-stakes subgroup. The Jorge study specifically found cardiovascular and kidney benefits in this population, which is encouraging. However, kidney disease can affect drug metabolism and elimination, and tirzepatide is primarily eliminated via proteolytic degradation rather than renal clearance, which means dose adjustments for kidney function are generally not needed.
That said, monitoring kidney function more frequently during tirzepatide initiation is prudent. Any increase in proteinuria or decline in GFR should prompt evaluation for lupus nephritis flare versus coincidental change.
Antiphospholipid syndrome
Many lupus patients have comorbid antiphospholipid syndrome (APS), which increases clotting risk. The Jorge study found that GLP-1 RAs were associated with reduced venous thromboembolism risk in lupus patients, which is particularly relevant for those with APS. However, APS patients are often on anticoagulation therapy, and tirzepatide delayed gastric emptying could theoretically affect oral anticoagulant absorption. If you take warfarin, more frequent INR monitoring during tirzepatide initiation is advisable.
Women of reproductive age
Lupus disproportionately affects women of childbearing age. Tirzepatide is contraindicated in pregnancy and should be discontinued at least 2 months before planned conception (its half-life is approximately 5 days, but a safety margin is standard). GLP-1 medications can also improve fertility in women with obesity-related anovulation, meaning pregnancy could occur unexpectedly.
For lupus patients, unplanned pregnancy carries additional risks because certain lupus medications (methotrexate, mycophenolate, cyclophosphamide) are teratogenic. The guide on pregnancy and tirzepatide and tirzepatide effects on menstrual cycles provide important context. Reliable contraception while on tirzepatide is essential for lupus patients on teratogenic medications.
Patients on multiple immunosuppressants
Some lupus patients require combination immunosuppressive therapy, such as mycophenolate plus belimumab, or azathioprine plus hydroxychloroquine plus low-dose prednisone. Adding tirzepatide to a complex medication regimen increases the total pill burden and the number of potential interactions to track.
In these cases, the conservative approach becomes even more important. Start at the lowest dose, titrate slowly, monitor frequently, and maintain close communication between all prescribers. The risk of a missed interaction or a subtle disease change going unnoticed increases with medication complexity.
Comparing options: tirzepatide versus other GLP-1 medications for lupus
Tirzepatide is not the only GLP-1 medication available. How does it compare to alternatives specifically for lupus patients?
Tirzepatide versus semaglutide
The semaglutide versus tirzepatide comparison takes on specific dimensions for lupus. Tirzepatide produces greater weight loss in head-to-head trials, which matters for patients needing significant weight reduction. The dual GIP/GLP-1 mechanism may provide additional anti-inflammatory and bone-protective effects through GIP receptor activation. However, semaglutide has a longer track record and more safety data in broader populations.
The single case of drug-induced lupus reported was with semaglutide, not tirzepatide. While this is a single case and cannot determine class effects, it is a data point worth noting. The three-way comparison with retatrutide provides additional perspective on the evolving landscape of GLP-1 medications.
Oral versus injectable options
For lupus patients dealing with injection fatigue from other medications, oral tirzepatide options may be appealing. The oral versus injection comparison shows that oral formulations may have different absorption profiles, which could be relevant for patients on medications that affect GI motility. Tirzepatide drops and orally disintegrating tablets represent emerging delivery options.
Insurance and access challenges
The NYU study noted that 50% of lupus patients were initially denied insurance coverage for GLP-1 medications. This reflects a broader access problem that disproportionately affects lupus patients because their indication for these medications (managing corticosteroid-induced weight gain and reducing cardiovascular risk in an autoimmune condition) does not fit neatly into approved indication categories.
None of the GLP-1 medications prescribed in the NYU cohort were prescribed by rheumatologists. They came from weight management (33%), endocrinology (39%), bariatrics (11%), and primary care (11%) providers. This fragmented prescribing pattern means lupus patients often need to coordinate between multiple specialists, which creates additional barriers.
Strategies for improving access include having your rheumatologist document the medical necessity (cardiovascular risk reduction, corticosteroid reduction), working with an endocrinologist or weight management specialist who can prescribe, exploring affordable tirzepatide options including compounded formulations from 503B compounding pharmacies, and appealing insurance denials with supporting documentation.
Knowing what to say when requesting GLP-1 options from healthcare providers can help navigate these conversations more effectively. The qualification process for GLP-1 medications varies by insurer and indication.
Storage and handling for immunocompromised patients
Lupus patients on immunosuppressive medications have compromised immune function. This makes proper medication handling and sterile injection technique even more important than for the general population.
Tirzepatide must be refrigerated at 2-8 degrees Celsius until use. Understanding shelf life and how long it lasts in the fridge prevents using compromised medication. If your tirzepatide is exposed to temperature excursions, our guide on what happens if tirzepatide gets warm explains when the medication is still usable.
For lupus patients who travel, maintaining the cold chain is essential. Our tirzepatide travel guide covers transportation methods and TSA considerations. The GLP-1 travel case guide recommends specific carrying solutions.
Proper injection technique with a syringe minimizes infection risk at injection sites. For compounded tirzepatide, understanding reconstitution procedures and proper mixing ratios ensures consistent dosing and sterility.
What your medical team needs to know
Communication between your rheumatologist, prescribing physician, and any other specialists is not optional. It is the foundation of safe tirzepatide use with lupus.
What to tell your rheumatologist
If you are considering tirzepatide, bring this topic to your rheumatologist first. Share the key research points from this article: the NYU cohort data showing no increased flare risk, the anti-inflammatory marker reductions, the cardiovascular and kidney outcome data from the Jorge study. Many rheumatologists are aware of the emerging data but have not been directly asked about it by patients.
Ask specifically about your disease stability, which medications might interact, how often they would want to monitor you, and whether they would coordinate with the prescribing physician. Not all rheumatologists will be comfortable with this addition to your regimen, and their concerns should be taken seriously.
What to tell your prescribing physician
Whoever prescribes the tirzepatide, whether it is an endocrinologist, weight management specialist, or primary care physician, needs to know your complete lupus history. This includes current medications, disease activity level, history of nephritis or other organ involvement, and any history of medication sensitivities. They should understand that standard titration may not apply and that more frequent monitoring is needed.
SeekPeptides provides comprehensive resources for understanding peptide and GLP-1 medication protocols, including detailed guides on tirzepatide dosing, safety considerations, and inflammation management with peptides.
The future of GLP-1 medications in autoimmune disease
The research landscape is moving quickly. Several developments are worth tracking.
Dedicated clinical trials examining GLP-1 receptor agonists specifically in autoimmune populations are in various stages of planning and execution. The preliminary data from observational studies like the NYU cohort and the Jorge outcomes study provide the justification for these trials, which will produce the higher-quality evidence needed for guideline recommendations.
The 2025 American College of Rheumatology guidelines for SLE treatment and the 2025 EULAR recommendations for lupus nephritis both acknowledge the potential role of metabolic therapies including GLP-1 medications. While they stop short of specific recommendations due to limited evidence, the inclusion signals that the rheumatology community is taking this seriously.
Newer GLP-1 medications in development, including triple agonists that target GLP-1, GIP, and glucagon receptors, may offer even broader metabolic and anti-inflammatory effects. The field is evolving rapidly, and SeekPeptides members access the latest research updates and evidence reviews as they become available.
For researchers serious about optimizing their peptide protocols and staying current with the evolving intersection of GLP-1 science and autoimmune disease management, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
Exercise and physical activity on tirzepatide with lupus
Weight loss medications work best when combined with physical activity, but lupus makes exercise complicated. The right approach balances the metabolic benefits of movement with the reality of living with an unpredictable disease.
Low-impact movement during stable periods
When disease activity is low, lupus patients on tirzepatide should aim for regular low-impact exercise. Walking, swimming, cycling, yoga, and tai chi provide cardiovascular benefits and help preserve muscle mass during weight loss without placing excessive stress on joints. The average weight loss per month on GLP-1 medications improves significantly with consistent physical activity.
Start with 10-15 minutes of gentle activity and build gradually. On tirzepatide, reduced caloric intake means you have less available energy for intense exercise, so moderate effort is more appropriate than high-intensity training. Swimming is particularly beneficial for lupus patients because water supports joint-stressed bodies while providing resistance for muscle maintenance. Avoid outdoor exercise during peak UV hours, as sun exposure is a well-documented lupus flare trigger.
Resistance training to prevent muscle loss
This deserves emphasis. Weight loss from GLP-1 medications does not distinguish between fat and muscle. Without resistance training, a significant portion of lost weight comes from lean tissue. For lupus patients already dealing with corticosteroid-induced muscle wasting, losing additional muscle mass is particularly problematic.
Two to three resistance training sessions per week, using body weight exercises, resistance bands, or light weights, can substantially reduce muscle loss during weight loss. Focus on major muscle groups: legs, back, chest, and core. If joint inflammation limits certain movements, work around affected joints rather than skipping resistance training entirely. A physical therapist familiar with autoimmune conditions can design a program that respects your limitations while providing the stimulus muscles need.
Flare-day modifications
On days when lupus symptoms are elevated, do not push through. Rest is not failure. It is disease management. Gentle stretching, breathing exercises, or a short walk may be appropriate if tolerated, but forcing a workout during a flare risks extending the flare duration and depleting the energy reserves your immune system needs.
The consistency of tirzepatide injections continues regardless of activity level. The medication provides metabolic and appetite benefits whether you exercise or not. On bad days, let the medication do more of the work. On good days, amplify its effects with physical activity. This flexible approach respects the unpredictable nature of lupus while maintaining forward progress.
Frequently asked questions
Is tirzepatide safe to take with lupus?
Based on current evidence, tirzepatide appears safe for lupus patients when used under appropriate medical supervision. The NYU Lupus Cohort study of 18 SLE patients on GLP-1 medications found no increase in disease flares above expected background rates. However, more frequent monitoring through blood work is recommended, particularly during the first three months of treatment.
Can tirzepatide reduce lupus inflammation?
Tirzepatide has been shown to significantly reduce inflammatory markers including hsCRP and IL-6 in clinical studies. These are the same markers elevated in active lupus. However, tirzepatide has not been studied specifically as an anti-inflammatory treatment for SLE, and it should not replace prescribed autoimmune disease treatments. The anti-inflammatory effects are a promising additional benefit, not a primary treatment.
Will tirzepatide interact with hydroxychloroquine?
There are no known direct pharmacological interactions between tirzepatide and hydroxychloroquine. Tirzepatide does not undergo cytochrome P450 metabolism, which eliminates most drug-drug interaction pathways. The theoretical concern about delayed absorption from slowed gastric emptying is unlikely to be clinically significant for hydroxychloroquine due to its very long half-life of 40-50 days.
Can tirzepatide help me reduce my prednisone dose?
This is a possibility but not a guarantee. By promoting weight loss and reducing inflammatory markers, tirzepatide may create conditions that allow for a gradual corticosteroid taper. Any dose reduction must be done gradually under your rheumatologist supervision. Abrupt steroid reduction can cause adrenal crisis and lupus flare regardless of other medications.
Should I start tirzepatide if my lupus is currently active?
Most experts would recommend waiting until your disease is stabilized or in remission before starting tirzepatide. In the NYU study, 78% of patients were in DORIS remission at baseline. Starting a new medication during active disease makes it harder to distinguish medication effects from disease activity and could potentially complicate flare management.
How often should I get blood work while on tirzepatide with lupus?
Every 2-3 months during the first 6 months is recommended, which is more frequent than the standard 3-6 month interval for either lupus or tirzepatide monitoring alone. After disease stability on tirzepatide is confirmed, the interval can be extended to match your regular lupus monitoring schedule. Labs should include standard lupus markers (complement, anti-dsDNA, CBC, urinalysis) plus metabolic markers (glucose, HbA1c, lipid panel, liver enzymes).
Does tirzepatide affect lupus kidney disease?
The Jorge 2026 study found that GLP-1 receptor agonists were associated with reduced progression of kidney disease in lupus patients, including those with lupus nephritis. This is encouraging, but the data is observational rather than from controlled trials. Managing lupus with peptide approaches requires careful renal monitoring regardless of the promising direction of evidence.
Can I take tirzepatide if I also have antiphospholipid syndrome?
There are no specific contraindications, and the reduced venous thromboembolism risk seen in the Jorge study is particularly relevant for APS patients. If you take oral anticoagulants like warfarin, more frequent INR monitoring during tirzepatide initiation is advisable due to the potential for slowed absorption. DOAC medications (apixaban, rivarelbanan) are less affected by absorption timing changes.
External resources
PMC: Retrospective evaluation of GLP-1 receptor agonists in SLE patients
Arthritis and Rheumatology: GLP-1 RA cardiovascular and kidney outcomes in SLE
PubMed: Anti-inflammatory effects of tirzepatide systematic review and meta-analysis
American College of Rheumatology: 2025 guideline for SLE treatment
In case I do not see you, good afternoon, good evening, and good night. May your inflammation stay managed, your protocols stay balanced, and your health stay on an upward trajectory.