Feb 26, 2026

At the cellular level, semaglutide triggers a cascade of metabolic events that suppress appetite, slow gastric emptying, and improve insulin sensitivity. But the peptide itself does not address two critical problems that emerge during rapid weight loss: muscle wasting and debilitating nausea. That is why compounding pharmacies developed the semaglutide glycine B12 blend, a three-component formulation designed to fill the gaps that standalone semaglutide leaves wide open.
The concept is straightforward. Semaglutide handles the heavy lifting of GLP-1 receptor activation and appetite regulation. Glycine, a conditionally essential amino acid, activates mTORC1 signaling pathways that protect lean muscle tissue during caloric restriction. And vitamin B12 (cyanocobalamin) targets the central nervous system mechanisms responsible for the nausea that drives up to 50% of patients to reduce or abandon their compounded semaglutide protocols entirely.
Three ingredients. Three distinct mechanisms. One injection.
This guide breaks down exactly how the semaglutide glycine B12 blend works, what the research says about each component, how compounding pharmacies formulate it, and what you need to know about dosing protocols, side effects, storage, and regulatory status. Whether you are already using semaglutide and considering an upgrade, or you are evaluating compound formulations for the first time, this is the most comprehensive resource available on the triple blend. SeekPeptides created this guide to give you the depth of information that surface-level articles simply do not provide.
What is the semaglutide glycine B12 blend
The semaglutide glycine B12 blend is a compounded injectable medication that combines three active ingredients into a single subcutaneous injection administered once weekly. The typical formulation contains semaglutide at 5 mg/mL, glycine at 5 mg/mL, and cyanocobalamin (vitamin B12) at 1 mg/mL. Some compounding pharmacies adjust these concentrations based on prescriber preferences, but the 5/5/1 ratio represents the most common configuration available through major compounding pharmacies.
This is not a brand-name product. It is not FDA-approved as a combination.
Compounding pharmacies create this blend by combining pharmaceutical-grade semaglutide with glycine and cyanocobalamin under strict sterile conditions. The rationale behind the formulation addresses three specific challenges that patients encounter during GLP-1 therapy: the appetite suppression and metabolic benefits of semaglutide, the muscle-preserving properties of glycine during rapid weight loss, and the nausea-reducing potential of B12 conjugation with GLP-1 receptor agonists.
Each component serves a purpose that the others cannot fill. Semaglutide alone does not protect muscle. B12 alone does not suppress appetite. Glycine alone does not activate GLP-1 receptors. Together, they target the three biggest obstacles to successful long-term weight management with GLP-1 medications: efficacy, tolerability, and body composition.
Understanding why each component matters requires examining them individually before looking at how they work as a system.
How semaglutide works as a GLP-1 receptor agonist
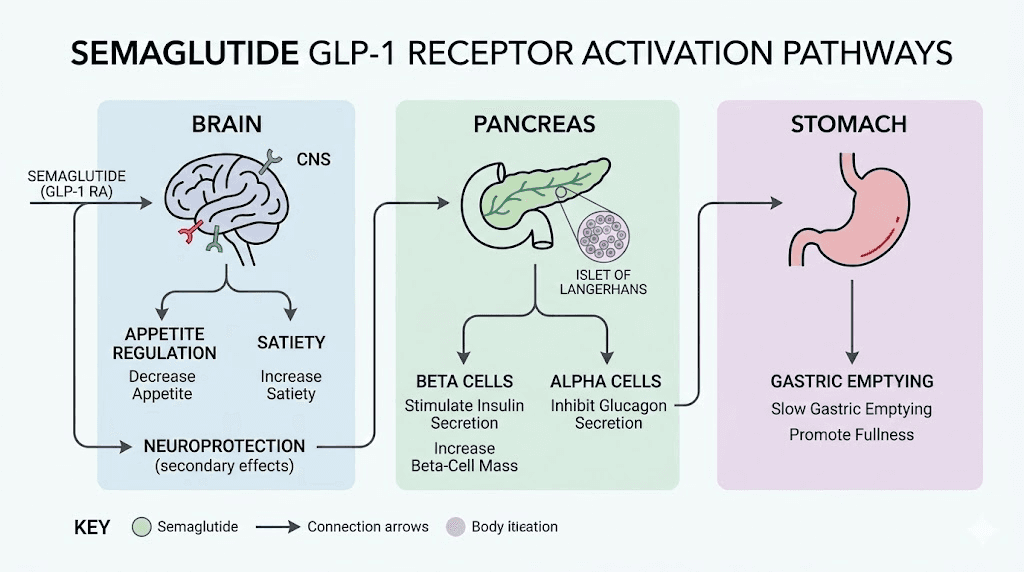
Semaglutide is a synthetic analogue of human glucagon-like peptide-1 (GLP-1), a hormone naturally produced in the gut after eating. When you eat a meal, your intestinal L-cells release GLP-1, which signals the pancreas to produce insulin, tells the liver to reduce glucose output, and communicates with the brain to register satiety. The problem is that natural GLP-1 breaks down within minutes. Semaglutide is engineered to resist that degradation, extending its half-life to approximately seven days, which is why it only requires once-weekly dosing.
The mechanism operates through multiple pathways simultaneously. In the pancreas, semaglutide enhances glucose-dependent insulin secretion, meaning it only triggers insulin release when blood sugar is elevated. This reduces the risk of hypoglycemia that plagues older diabetes medications. In the brain, semaglutide crosses the blood-brain barrier and activates receptors in the hypothalamus and brainstem that regulate hunger, reward-seeking behavior, and food intake. The result is a profound reduction in appetite that most patients describe as simply not thinking about food anymore.
Semaglutide also delays gastric emptying. Food sits in the stomach longer, which extends feelings of fullness and reduces the volume of food consumed at each meal. This effect contributes to both weight loss and the gastrointestinal side effects that make the first weeks on semaglutide challenging for many patients.
Clinical trials demonstrate average weight loss of 10 to 15% of total body weight over 68 weeks at the 2.4 mg weekly dose. Those numbers are impressive. But they come with a caveat that rarely gets the attention it deserves: up to 30 to 40% of that weight loss can come from lean muscle mass rather than fat tissue. That is where glycine enters the equation.
Why glycine is added to the compound
Glycine is the simplest amino acid in human biology. It has no side chain, just a hydrogen atom where other amino acids carry complex molecular structures. But simple does not mean unimportant. Glycine participates in more biochemical reactions than almost any other amino acid, including collagen synthesis, glutathione production, creatine formation, bile acid conjugation, and neurotransmitter regulation.
In the context of GLP-1 therapy, glycine addresses the single biggest concern that clinicians and patients share: losing muscle along with fat.
When semaglutide suppresses appetite and reduces caloric intake by 20 to 40%, the body enters a state of energy deficit. It needs fuel. And while the goal is for that fuel to come from stored fat, the body does not always cooperate. Skeletal muscle tissue becomes a target for breakdown, particularly in patients who are not consuming adequate protein or engaging in resistance training. Research shows that protein intake during GLP-1 therapy is critical, but glycine adds an additional layer of protection at the cellular level.
Compounding pharmacies add glycine to semaglutide formulations because it acts as a metabolic buffer. It provides the amino acid substrate needed for collagen turnover and connective tissue maintenance while simultaneously activating anabolic signaling pathways that resist muscle catabolism. The addition does not interfere with the weight loss effects of semaglutide. Instead, it shifts the ratio of fat loss to muscle loss in a more favorable direction.
Glycine also serves a practical function in the compounding process itself. It acts as a stabilizer for the semaglutide molecule in solution, helping maintain potency and shelf life. This dual purpose, both therapeutic and pharmaceutical, makes it an elegant addition to the formulation.
The science behind glycine and muscle preservation
The evidence for glycine and muscle protection is not theoretical. Multiple peer-reviewed studies demonstrate specific mechanisms through which glycine preserves lean tissue during caloric restriction.
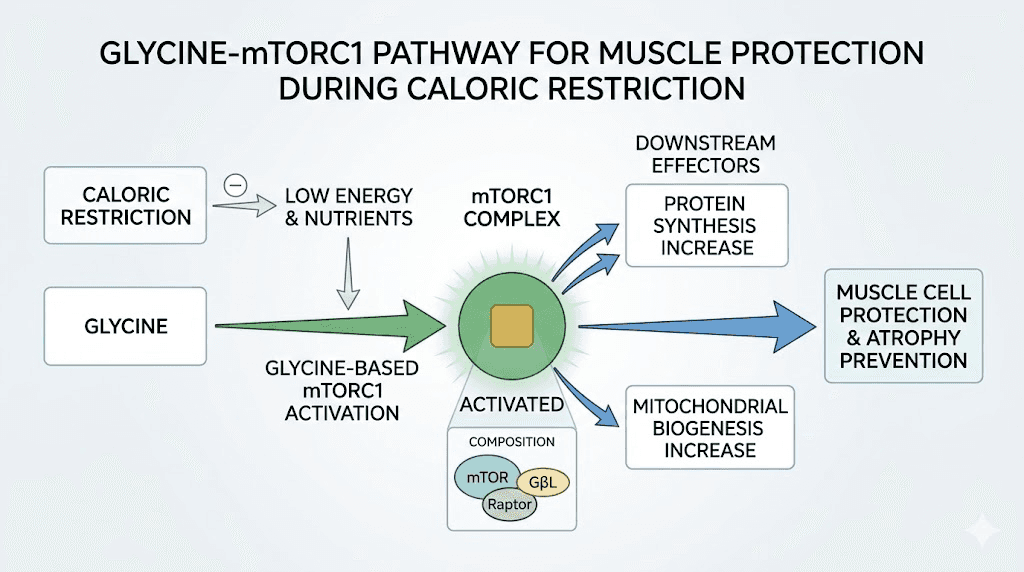
A study published in Frontiers in Nutrition (2019) showed that glycine-treated muscle cells (myotubes) were up to 20% larger compared to control cells in two different models of muscle wasting. The researchers identified the mechanism: glycine activates the mTORC1 signaling pathway, the master switch for muscle protein synthesis. When they blocked mTORC1 with the inhibitor rapamycin, the protective effect of glycine disappeared entirely. This confirmed that mTORC1 activation is the primary route through which glycine preserves muscle mass.
That finding matters enormously for GLP-1 patients.
A separate study in Clinical Nutrition (2016) tested glycine supplementation during calorie restriction in obese mice. The results were striking. The glycine group lost 21% of body weight, reduced whole-body fat mass by 49%, and restored glucose tolerance, all while preserving more lean tissue than the control group. The study concluded that glycine supplementation during calorie restriction accelerates fat loss and protects against further muscle loss.
At the molecular level, glycine works through several complementary pathways. Research from the American Journal of Physiology (2016) demonstrated that glycine maintains Akt-mTOR-FOXO1 signaling, the anabolic cascade that drives muscle protein synthesis, while simultaneously suppressing TLR4 and NOD2 signaling pathways associated with inflammation-driven muscle breakdown. In practical terms, glycine tells your muscles to keep building while telling the inflammatory signals to stop destroying.
Additional research in the Journal of Animal Science showed that glycine increased protein synthesis rates by 20 to 80% and inhibited protein degradation rates by 15 to 30% in a concentration-dependent manner. Higher glycine concentrations produced stronger protective effects, which supports the inclusion of meaningful glycine doses in compounded formulations rather than token amounts.
For patients using the semaglutide glycine B12 blend, this research translates to a practical advantage. The glycine component works alongside proper supplementation, adequate protein-rich nutrition, and resistance training to minimize the lean mass losses that accompany rapid weight reduction on GLP-1 medications.
People who are already experiencing muscle-related side effects from GLP-1 therapy may find the glycine component particularly relevant. The amino acid not only preserves existing tissue but supports the recovery processes that intense training demands.
Why vitamin B12 is included in the formulation
Vitamin B12 (cyanocobalamin) serves two distinct purposes in the semaglutide glycine B12 blend. The first is addressing a nutrient absorption concern. The second is directly targeting the nausea that makes GLP-1 therapy difficult for many patients.
GLP-1 receptor agonists like semaglutide slow gastric emptying and may reduce stomach acid production. Both of these effects can impair vitamin B12 absorption. B12 requires stomach acid and a protein called intrinsic factor to be properly absorbed in the small intestine. When semaglutide slows the entire digestive process and potentially reduces acid secretion, the conditions for optimal B12 uptake become compromised.
Research published in Science Direct found that nutritional deficiencies were diagnosed in 12.7% of patients within six months of starting GLP-1 receptor agonist therapy, and that number climbed to 22.4% within twelve months. B12 deficiency was among the nutrients of concern, alongside iron, calcium, magnesium, and vitamins D and K. For patients on long-term semaglutide protocols spanning months or years of treatment, this cumulative deficiency risk is not trivial.
By including B12 directly in the injection, the formulation bypasses the digestive system entirely. Subcutaneous B12 delivery achieves near-complete bioavailability because it enters the bloodstream directly rather than relying on the compromised gastrointestinal absorption pathway. This is a meaningful advantage over oral B12 supplements that must navigate the same slowed, acid-depleted environment that semaglutide creates.
But the nutrient replacement rationale is only half the story. The more compelling reason for B12 inclusion involves its potential to reduce one of the most common and most disruptive side effects of GLP-1 therapy.
How B12 reduces GLP-1 nausea at the molecular level
Nausea affects up to 50% of patients taking GLP-1 receptor agonists. It is the number one reason people discontinue treatment, reduce their doses below therapeutic levels, or fail to progress through the standard titration schedule. Understanding why B12 may help requires understanding why GLP-1 medications cause nausea in the first place.
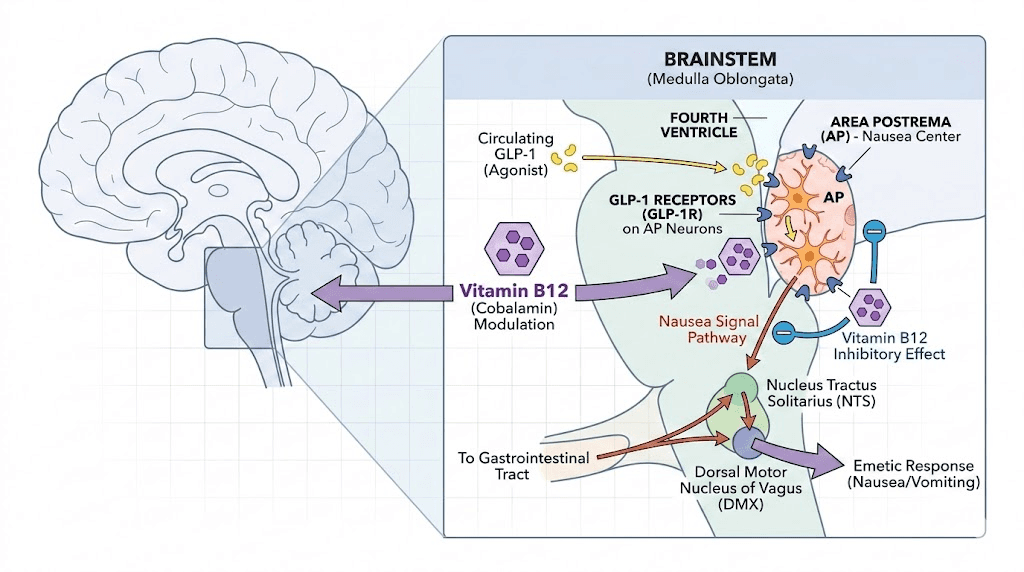
GLP-1 receptor agonists trigger nausea through activation of central GLP-1 receptors in the brainstem, specifically in the area postrema and the nucleus of the solitary tract. These brain regions serve as the body chemoreceptor trigger zone, the vomiting center. When semaglutide crosses the blood-brain barrier and activates receptors in these areas, nausea is a predictable consequence.
Here is where the science gets interesting.
Research published in the British Journal of Pharmacology demonstrated that molecular conjugation of B12 (specifically the corrin ring structure found in cyanocobalamin) with GLP-1 receptor agonists produced a compound that was not as readily absorbed into the brainstem areas responsible for triggering nausea. The B12-conjugated GLP-1 agonist maintained comparable blood sugar control but with significantly better tolerance, less nausea, and fewer emetic episodes in both rat and mammalian models.
The researchers identified this approach as "corrination," the attachment of corrin ring-containing compounds to GLP-1 agonists. The corrin ring structure, which is the molecular backbone of vitamin B12, appears to alter the pharmacokinetic profile in a way that reduces central nervous system exposure to the emetogenic (nausea-causing) effects while preserving the metabolic benefits.
Now, an important distinction. The clinical studies on B12-GLP-1 conjugation used chemically bonded molecular compounds, not simply mixed solutions like those found in compounding pharmacies. The semaglutide glycine B12 blend is a physical mixture, not a chemical conjugate. Whether the B12 in a mixed formulation provides the same level of nausea reduction as a true molecular conjugate remains an open question.
That said, clinical observations from prescribers and patients using the blend are consistently positive. Many practitioners report that patients on the triple blend experience less gastrointestinal distress during dose escalation compared to those on standalone semaglutide. Whether this results from the B12 component, the glycine component (which has its own anti-inflammatory properties), or a placebo effect is difficult to determine without controlled trials.
What is clear: the theoretical mechanism is sound, the preclinical data supports the approach, and the clinical experience from thousands of patients has been favorable enough that compounding pharmacies have made the triple blend one of their most popular formulations.
The synergy of all three components together
Understanding each ingredient individually is important. Understanding how they interact as a system is more important.
The semaglutide glycine B12 blend creates what pharmacologists call complementary targeting. Each component addresses a different limitation of GLP-1 monotherapy, and the combination produces an effect profile that none of the three could achieve alone. Semaglutide provides the core metabolic benefits: appetite suppression, improved insulin sensitivity, reduced glucagon secretion, and delayed gastric emptying. These effects drive the weight loss results that patients seek.
Glycine addresses the body composition problem. Without it, the caloric deficit created by semaglutide pulls energy from both fat stores and muscle tissue. With glycine activating mTORC1 pathways and supporting protein synthesis, the body receives a signal to preferentially break down fat while maintaining lean mass. This does not eliminate muscle loss entirely, nothing can during significant caloric restriction, but it tilts the balance meaningfully.
B12 addresses the tolerability problem. By potentially modulating the central nausea response and simultaneously preventing the nutrient deficiencies that GLP-1 therapy can create, B12 helps patients stay on their prescribed dosing schedule without the interruptions and dose reductions that nausea typically forces.
The practical result is a formulation that patients are more likely to tolerate at therapeutic doses, more likely to maintain for the full duration of treatment, and more likely to achieve favorable body composition outcomes compared to semaglutide alone. Higher tolerance means better adherence. Better adherence means better results. Better body composition means healthier long-term outcomes.
This synergistic logic explains why the triple blend has become one of the most commonly prescribed compounded semaglutide formulations, not because any single addition is revolutionary, but because the combination addresses the three biggest failure points in GLP-1 therapy simultaneously.
Dosing protocols and titration schedules
The semaglutide glycine B12 blend follows the same fundamental titration philosophy as standalone semaglutide: start low, increase gradually, and let the body adapt to each dose level before moving higher. Rushing the escalation is the single most common mistake that leads to severe gastrointestinal side effects and treatment discontinuation.
The standard titration protocol for a 5 mg/mL semaglutide concentration with glycine and B12 typically follows this schedule:
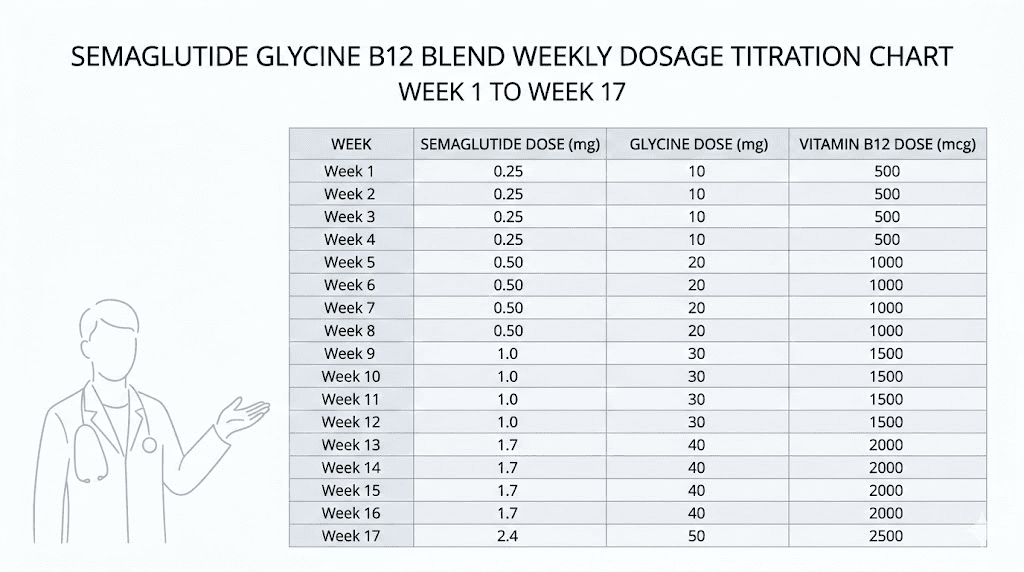
Weeks 1 through 4: 4 units (0.04 mL) once per week. This delivers approximately 0.2 mg of semaglutide, well below the therapeutic threshold but enough to begin receptor sensitization and assess initial tolerance.
Weeks 5 through 8: 8 units (0.08 mL) once per week. This approximately doubles the semaglutide dose to 0.4 mg, moving closer to the first clinically significant dose level. Most patients begin noticing appetite changes during this phase.
Weeks 9 through 12: 18 units (0.18 mL) once per week. This represents a significant jump to approximately 0.9 mg, crossing into the range where meaningful metabolic effects begin. The larger increase is intentional, as the body has had eight weeks to adapt to GLP-1 receptor activation.
Weeks 13 through 16: 30 units (0.30 mL) once per week. At approximately 1.5 mg of semaglutide, this dose matches the standard therapeutic level for type 2 diabetes management and begins approaching the weight management dose range.
Week 17 and beyond: 44 units (0.44 mL) once per week. This delivers approximately 2.2 mg of semaglutide, near the maximum therapeutic dose for weight management. Some prescribers cap at this level while others adjust based on individual response and tolerance.
These numbers can vary. Some prescribers use different concentrations, which changes the unit calculations entirely. The 5 mg/mL concentration described above is the most common, but formulations at 2.5 mg/mL and other concentrations exist. Always verify your specific vial concentration before calculating doses. Our semaglutide dosage calculator can help you determine exact measurements for your specific formulation.
One critical point that many guides overlook: the glycine and B12 components scale proportionally with the semaglutide dose. As you inject more volume to increase your semaglutide dose, you automatically receive more glycine and more B12. This means the protective effects of both additives increase in lockstep with the metabolic intensity of the semaglutide.
If you experience significant nausea during dose escalation, the standard approach is to hold at the current dose for an additional two to four weeks before attempting the next increase. Do not drop back to a lower dose unless symptoms are severe. The body needs time to adjust at each level, and extending the adaptation period is almost always preferable to resetting progress.
Concentrations and formulations from compounding pharmacies
Not all semaglutide glycine B12 blends are created equal. Compounding pharmacies have flexibility in how they formulate the product, and the differences matter more than most patients realize.
The most common formulation is the 5/5/1 blend: semaglutide 5 mg/mL, glycine 5 mg/mL, and cyanocobalamin 1 mg/mL. This concentration allows for a full titration protocol from starting dose through maintenance dose within a single vial, depending on vial size. Empower Pharmacy, one of the largest 503B compounding facilities, has offered semaglutide-cyanocobalamin combinations, though formulation specifics vary over time.
Some pharmacies offer lower concentrations, particularly 2.5 mg/mL semaglutide blends. These require larger injection volumes to achieve the same dose, which some patients prefer (easier to measure precisely with an insulin syringe) and others dislike (more fluid volume under the skin). The syringe measurements change accordingly, so always confirm your concentration before drawing up a dose.
A few things to look for when evaluating compounding pharmacy formulations:
Sterility testing. Any reputable 503B pharmacy performs batch sterility and endotoxin testing before releasing product. Ask for certificates of analysis. If a pharmacy cannot or will not provide them, that is a red flag.
Beyond-use dating. Compounded products have shorter shelf lives than manufactured drugs. Typical beyond-use dates for compounded semaglutide blends range from 30 to 90 days depending on the pharmacy and storage conditions. Understanding how long your vial lasts prevents waste and ensures potency.
Source ingredient quality. The semaglutide used in compounding should come from FDA-registered facilities with drug master files. The glycine and B12 should be USP-grade. Some pharmacies use methylcobalamin instead of cyanocobalamin, which is a different form of B12 with slightly different pharmacokinetics. Our guide on semaglutide with methylcobalamin covers the differences in detail.
Vial size. Common vial sizes include 2 mL, 3 mL, and 5 mL. Larger vials offer more doses per vial but must be used within the beyond-use dating period. If you are on a lower dose, a smaller vial may be more practical to avoid waste.
Price varies significantly between pharmacies. The peptide cost calculator can help you compare per-dose costs across different vial sizes and concentrations. Do not assume the cheapest option is the worst or the most expensive is the best. Evaluate based on testing documentation, pharmacy accreditation, and prescriber recommendations.
How to administer the semaglutide glycine B12 injection
Administration of the semaglutide glycine B12 blend follows the same subcutaneous injection technique used for standalone semaglutide. The process is straightforward once you learn it, but precision matters, particularly with dose measurement.
Step 1: Gather supplies. You need your vial of semaglutide glycine B12 blend, an insulin syringe (typically 0.5 mL or 1 mL with half-unit markings), alcohol swabs, and a sharps container. If your vial has been stored in the refrigerator, let it sit at room temperature for 10 to 15 minutes before injecting. Cold injections are more uncomfortable and may cause increased injection site reactions.
Step 2: Clean and draw. Wipe the vial stopper with an alcohol swab. Draw air into the syringe equal to your dose volume, insert the needle through the stopper, push the air in, then invert the vial and draw out your prescribed dose. Remove any air bubbles by tapping the syringe gently and pushing them back into the vial.
Step 3: Choose your injection site. The recommended injection sites include the abdomen (at least two inches from the belly button), the front of the thighs, and the back of the upper arms. Rotate sites with each injection to prevent lipodystrophy, the hardening of tissue that repeated injections in the same spot can cause.
Step 4: Inject. Pinch a fold of skin at the chosen site. Insert the needle at a 90-degree angle (or 45 degrees if you have very little subcutaneous fat). Push the plunger slowly and steadily. Hold for 5 to 10 seconds after the plunger is fully depressed before withdrawing the needle. Do not rub the injection site afterward. For a more detailed walkthrough, our complete semaglutide injection guide covers every step with visual references.
Step 5: Dispose safely. Place the used syringe immediately into a sharps container. Never recap needles. Never throw syringes in regular trash.
Inject on the same day each week. Consistency matters for maintaining steady blood levels. If you miss your scheduled day, take it as soon as you remember, as long as there are at least two days before your next scheduled dose. Our guide on the best time to take your GLP-1 shot covers timing optimization in more detail.
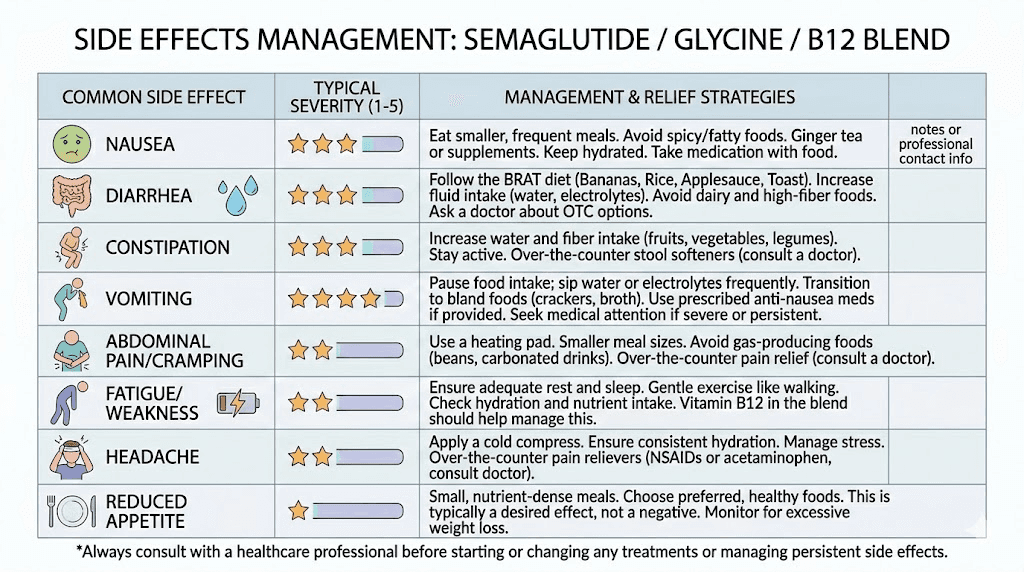
Side effects and how to manage them
The side effect profile of the semaglutide glycine B12 blend is primarily driven by the semaglutide component. The glycine and B12 additions are generally well-tolerated at the doses used in these formulations and rarely contribute their own adverse effects. That said, patients need to know what to expect and how to respond.
Nausea remains the most common side effect, even with the B12 addition. It typically peaks during the first one to two weeks at each new dose level and gradually diminishes as the body adapts. Eating smaller, more frequent meals helps. Avoiding greasy, heavy, or overly sweet foods reduces the severity. The complete list of foods to avoid on semaglutide is essential reading for anyone starting the blend.
Constipation affects a significant percentage of patients. Semaglutide slows the entire digestive tract, not just the stomach. Adequate hydration, fiber intake, and in some cases magnesium supplementation can address this. Our semaglutide constipation treatment guide provides evidence-based strategies.
Fatigue is common, particularly in the early weeks. Reduced caloric intake combined with metabolic changes can leave patients feeling drained. This typically improves as the body adapts to lower caloric intake and begins efficiently accessing fat stores for energy. Understanding why semaglutide fatigue happens and having a management plan makes a significant difference.
Dizziness occurs in some patients, often related to dehydration or rapid blood pressure changes associated with weight loss. Staying well-hydrated and rising slowly from seated or lying positions helps. Our semaglutide dizziness guide covers this in detail.
Injection site reactions include redness, itching, or mild swelling at the injection site. These are usually minor and resolve within a few hours. Rotating injection sites consistently and allowing the solution to reach room temperature before injecting minimizes these reactions.
Headaches can occur, particularly during the initial titration phase or during rapid weight loss. Dehydration and blood sugar fluctuations are the usual culprits. The GLP-1 headache guide provides comprehensive management strategies.
Serious but rare side effects include pancreatitis (severe abdominal pain that radiates to the back), gallbladder issues, and acute kidney injury. The semaglutide glycine B12 blend carries the same FDA black box warning as all GLP-1 receptor agonists regarding medullary thyroid carcinoma risk in rodent studies. Patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 should not use this medication.
Hair thinning is reported by some patients during rapid weight loss phases. This is generally related to the caloric deficit rather than the medication itself, and it typically resolves as caloric intake stabilizes. Our GLP-1 hair loss guide covers prevention and treatment strategies.
For most patients, side effects are manageable and temporary. The addition of glycine and B12 to the formulation may reduce the severity and duration of gastrointestinal symptoms compared to standalone semaglutide, though individual experiences vary.
How the blend compares to standalone semaglutide
The question every patient asks: is the triple blend actually better than regular semaglutide? The honest answer requires separating what we know from what we suspect.
What we know: Standalone semaglutide is the most studied component. Decades of clinical trials support its efficacy for weight loss and glycemic control. The semaglutide in the blend is the same molecule. It works through the same receptors. It produces the same core effects.
What we know about glycine: Peer-reviewed research confirms that glycine activates mTORC1 signaling, protects muscle cells from wasting, accelerates fat loss during caloric restriction, and has anti-inflammatory properties. These effects have been demonstrated in cell cultures, animal models, and limited human studies. The specific combination of glycine with semaglutide has not been tested in large randomized controlled trials.
What we know about B12: The nutrient absorption concern with long-term GLP-1 use is documented. The nausea-reducing mechanism of B12 conjugation is supported by preclinical research. Whether B12 in a mixed solution (rather than a chemical conjugate) provides the same benefit is unconfirmed in controlled human studies.
Here is a practical comparison:
Factor | Standalone semaglutide | Semaglutide glycine B12 blend |
|---|---|---|
Weight loss efficacy | Well-established (10-15%) | Same core mechanism |
Muscle preservation | No built-in protection | Glycine supports mTORC1 |
Nausea management | Standard side effect profile | B12 may reduce severity |
Nutrient protection | Requires separate supplementation | B12 included in injection |
FDA approval | Yes (as Ozempic/Wegovy) | No (compounded product) |
Clinical trial data | Extensive | Limited for combination |
Cost | Varies by source | Typically lower than brand-name |
Convenience | Single ingredient | Three ingredients, one injection |
If you are already using compounded semaglutide without glycine and B12, switching to the triple blend is straightforward since the semaglutide component remains the same. You do not need to restart your titration. If you are comparing semaglutide with B12 alone (without glycine), our semaglutide with B12 guide covers that specific formulation. And for a deep dive into the glycine component by itself, the semaglutide with glycine article provides focused coverage.
The tirzepatide equivalent of this formulation, the tirzepatide glycine B12 compound, follows similar logic but uses a dual GIP/GLP-1 agonist as the base instead. Patients considering a switch between the two base medications should consult our switching guide and side effect comparison.
Storage, handling, and shelf life
Proper storage of the semaglutide glycine B12 blend directly affects potency, safety, and the cost-effectiveness of your treatment. Peptides are fragile molecules. Mistreat them and you lose both money and therapeutic benefit.
Refrigerate at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). This is non-negotiable. Store your vial upright in the main body of the refrigerator, not in the door (temperature fluctuations) and not in the back near the cooling element (risk of freezing). Frozen semaglutide is destroyed semaglutide. The molecular structure denatures irreversibly when ice crystals form. If your vial freezes, discard it. No exceptions.
Our guide on whether compounded semaglutide needs refrigeration covers the science behind storage requirements in detail. Spoiler: yes, it absolutely does.
Brief room temperature exposure is acceptable. If your vial sits out for an hour while you prepare and administer your injection, that is fine. If you accidentally leave it out overnight, the answer depends on ambient temperature and duration. Below 77 degrees Fahrenheit for up to 24 hours is generally acceptable according to most compounding pharmacy guidelines, though potency may be slightly reduced.
Extended heat exposure is destructive. If your vial spends time in a hot car, near a window in direct sunlight, or in any environment above 86 degrees Fahrenheit, the semaglutide begins degrading. The glycine and B12 components are more heat-stable, but the semaglutide, the most important ingredient, is not.
Beyond-use dating. Most compounded semaglutide glycine B12 blends have a beyond-use date of 30 to 90 days from the date of compounding, assuming proper refrigeration. This is shorter than the shelf life of brand-name Ozempic pens because compounded products do not contain the same preservative systems. Understanding when compounded semaglutide expires prevents you from using degraded product.
For patients who travel, traveling with semaglutide requires a small insulated bag with ice packs (not direct contact with the vial) to maintain cold chain integrity. Plan ahead for airport security, hotel refrigerators, and time zone changes that may affect your dosing schedule.
Visual inspection. Before each use, examine your vial. The solution should be clear and colorless to slightly yellow. Cloudiness, particles, discoloration, or any change from the original expected appearance indicates potential degradation or contamination. When in doubt, do not inject it.
FDA regulatory status and what it means for patients
The regulatory landscape for compounded semaglutide, including the glycine B12 blend, has been turbulent. Understanding the current status helps patients make informed decisions.
Semaglutide as an active ingredient is FDA-approved under the brand names Ozempic (for type 2 diabetes) and Wegovy (for weight management). However, the compounded semaglutide glycine B12 blend is not FDA-approved as a combination product. It never has been, and it is unlikely to be, because FDA approval requires hundreds of millions of dollars in clinical trial investment that no compounding pharmacy can justify for a generic combination.
Compounding pharmacies have historically been allowed to produce custom formulations under section 503A (individual prescriptions) and section 503B (outsourcing facilities with batch production) of the Federal Food, Drug, and Cosmetic Act. The legal basis for compounding semaglutide specifically relates to the FDA shortage list. When a drug is listed on the official FDA drug shortage list, compounding pharmacies are permitted to produce copies, including enhanced formulations with additional ingredients like glycine and B12.
Semaglutide was on the FDA shortage list for several years, allowing widespread compounding. The regulatory environment continues to evolve, and patients should stay informed about current availability through their prescribers and pharmacies.
What this means practically: the semaglutide glycine B12 blend is legal when properly prescribed and compounded, but it does not undergo the same level of FDA oversight as brand-name products. Quality depends heavily on the compounding pharmacy. Choosing a pharmacy with PCAB accreditation (Pharmacy Compounding Accreditation Board), full sterility testing, and transparent quality documentation is not optional. It is essential.
Patients who are uncomfortable with compounded products can discuss brand-name alternatives with their prescribers, though the brand-name versions do not include glycine or B12 and require separate supplementation to address those concerns.
Who is a good candidate for this formulation
The semaglutide glycine B12 blend is not for everyone. Understanding who benefits most helps you determine whether this specific formulation matches your situation.
Strong candidates include:
Patients who have experienced significant nausea on standalone semaglutide and want a formulation that may improve tolerability. If nausea has prevented you from reaching therapeutic doses, the B12 component may help you progress through the titration schedule more comfortably.
Patients concerned about muscle loss during rapid weight loss. If you are not currently doing resistance training and high-protein diets (ideally 1.0 to 1.6 grams of protein per kilogram of body weight daily), the glycine component provides an additional layer of lean mass protection. This is especially relevant for older patients and those with significant weight to lose.
Patients already on compounded semaglutide who want to consolidate their supplementation. If you are taking separate B12 supplements and glycine capsules alongside your semaglutide injections, the triple blend simplifies the regimen into a single weekly injection.
Patients on long-term GLP-1 therapy. The longer you use semaglutide, the more relevant the B12 deficiency risk becomes and the more cumulative muscle loss affects your metabolic rate and physical function. The triple blend addresses both long-term concerns proactively.
Less ideal candidates include:
Patients who are doing well on standalone semaglutide without significant nausea or body composition concerns. If your current protocol is working, there is no compelling reason to switch. Do not fix what is not broken.
Patients with allergies or sensitivities to cyanocobalamin or glycine. While rare, these do exist. Methylcobalamin-based formulations are available for those who cannot tolerate cyanocobalamin. Our methylcobalamin guide covers alternatives.
Patients with medullary thyroid carcinoma, Multiple Endocrine Neoplasia syndrome type 2, or a history of pancreatitis should not use any semaglutide formulation, including this blend.
Optimizing your results on the semaglutide glycine B12 blend
The blend gives you better tools. But tools without strategy produce mediocre results.
Prioritize protein intake. Glycine supports muscle preservation at the cellular level, but it cannot replace adequate total protein consumption. Aim for 1.0 to 1.6 grams of protein per kilogram of body weight daily. Front-load protein at breakfast and lunch when appetite is highest. The semaglutide diet plan and our guide on the best foods to eat on semaglutide provide meal-by-meal frameworks.
Resistance training is non-negotiable. No supplement, peptide, or compound formulation can fully replace the anabolic stimulus of progressive resistance training. Even two to three sessions per week of basic compound movements (squats, deadlifts, presses, rows) send powerful muscle-preservation signals that synergize with the glycine component. Patients who combine the triple blend with resistance training consistently achieve superior body composition outcomes compared to those relying on the medication alone.
Stay hydrated. Dehydration amplifies every side effect: nausea, constipation, dizziness, fatigue, and headaches. Aim for at least 64 ounces of water daily, more if you are active or in a warm climate. Electrolyte supplementation may be warranted during the initial phases of significant caloric reduction.
Track your body composition, not just your weight. A scale cannot tell you whether you are losing fat or muscle. Body composition tools, whether DEXA scans, bioelectrical impedance scales, or even simple waist-to-hip ratio measurements, provide the insight you need to evaluate whether the glycine component is working. If your weight is dropping but your lean mass is holding steady, the protocol is working as intended.
Do not skip doses. Consistency is the single biggest predictor of success with GLP-1 therapy. The semaglutide glycine B12 blend works best when blood levels remain steady through regular weekly dosing. Missed doses create troughs that can trigger rebound hunger and disrupt the metabolic adaptations you have built over weeks of consistent use.
If you hit a weight loss plateau, that does not mean the medication stopped working. Plateaus are a normal part of the process and often resolve with dietary adjustments, increased physical activity, or a prescriber-supervised dose modification.
SeekPeptides members access detailed protocol builders, body composition tracking tools, and community support from experienced researchers who have navigated these exact optimization challenges. The platform provides the strategic framework that turns a good medication into exceptional results.
Comparing the semaglutide blend to tirzepatide formulations
Semaglutide is not the only GLP-1 medication available in compounded triple blends. Tirzepatide, a dual GIP/GLP-1 receptor agonist, also comes in glycine and B12 formulations. Understanding the differences helps patients make informed choices.
Tirzepatide activates both the GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors, whereas semaglutide targets only GLP-1. In clinical trials, tirzepatide demonstrated average weight loss of up to 22.5% of body weight, exceeding the 15% typically seen with semaglutide at maximum doses. The full semaglutide vs tirzepatide comparison covers all the differences in mechanism, efficacy, and side effects.
The glycine and B12 rationale applies equally to both medications. Both cause appetite suppression that risks muscle loss. Both cause nausea that limits dose escalation. Both may impair nutrient absorption long-term. The tirzepatide with glycine combination and the compounded tirzepatide with B12 formulation follow the same therapeutic logic as the semaglutide triple blend.
For patients who have plateaued on semaglutide or who respond better to dual-agonist therapy, transitioning to a tirzepatide glycine B12 blend may provide the additional efficacy needed. The three-way comparison of semaglutide, tirzepatide, and retatrutide offers comprehensive guidance for patients evaluating all available options.
Next-generation compounds like survodutide, CagriSema, and orforglipron are also entering the landscape, though most are not yet available in compounded formulations. The field is moving fast, and staying informed matters.
Common mistakes to avoid with the semaglutide glycine B12 blend
Knowing what not to do is sometimes more valuable than knowing what to do.
Mistake 1: Escalating doses too quickly. The titration schedule exists for a reason. Each dose level requires two to four weeks of adaptation. Jumping ahead because you want faster results almost always backfires with severe nausea, vomiting, and treatment interruptions that set you further back than patience would have.
Mistake 2: Neglecting protein intake. Glycine helps preserve muscle, but it is not magic. Without adequate dietary protein, even glycine-enhanced mTORC1 signaling cannot fully prevent lean mass loss. Calculate your protein needs and hit them daily. Use protein shakes designed for GLP-1 patients if solid food intake is difficult during periods of strong appetite suppression.
Mistake 3: Ignoring hydration. This sounds basic because it is basic. And yet dehydration remains the root cause of most manageable side effects. Water, electrolytes, and consistent intake throughout the day prevent more problems than most patients realize.
Mistake 4: Improper storage. Leaving the vial on the bathroom counter, storing it in a car, or placing it too close to the refrigerator freezing element all compromise potency. Degraded semaglutide does not announce itself. You simply stop getting results and waste money.
Mistake 5: Using the wrong syringe or miscalculating doses. With compounded products, the concentration determines how many units equal how many milligrams. A 5 mg/mL vial requires different measurements than a 2.5 mg/mL vial. Double-check your concentration against your prescribed dose every single time. The semaglutide conversion chart and dosage calculator prevent these errors.
Mistake 6: Expecting the blend to replace lifestyle changes. The semaglutide glycine B12 blend is a powerful tool, but it is not a standalone solution. Results without exercise are possible but suboptimal. Results without dietary awareness are possible but less impressive. The patients who achieve the best outcomes treat the blend as one component of a comprehensive approach.
Mistake 7: Stopping abruptly. When you decide to discontinue semaglutide, gradual tapering is preferable to sudden cessation. Abrupt discontinuation can trigger rebound hunger and rapid weight regain. Our guide on semaglutide withdrawal symptoms covers what to expect and how to manage the transition.
What to expect week by week on the blend
Setting realistic expectations prevents frustration and premature discontinuation. Here is what the typical patient experience looks like across the first four months of therapy.
Weeks 1 through 4 (starting dose). Most patients notice minimal appetite changes at this dose. Some feel a slight decrease in hunger. Nausea, if it occurs, is mild. The first week may bring mild gastrointestinal awareness, a sense that something has changed in your digestive system without dramatic symptoms. Weight loss is typically 0 to 2 pounds, mostly water balance shifts rather than meaningful fat loss. This phase is about tolerance building, not results.
Weeks 5 through 8 (second dose level). Appetite suppression becomes more noticeable. Portions shrink naturally. Food noise, the constant background chatter of cravings and food thoughts, begins to quiet. Nausea may increase slightly as the dose doubles, but the B12 component and slow escalation help manage it. Weight loss of 2 to 5 pounds during this phase is typical. The glycine component is working silently in the background, supporting muscle protein synthesis even as caloric intake drops.
Weeks 9 through 12 (third dose level). This is where the medication starts earning its reputation. Appetite suppression is significant. Many patients report difficulty eating enough to hit their protein targets, which is where choosing the right foods becomes critical. Weight loss of 3 to 6 pounds during this phase is common. Some patients lose more. The most common side effects, nausea, constipation, and fatigue, are most prominent during this transition.
Weeks 13 through 16 (fourth dose level). The body has adapted significantly by this point. Side effects typically stabilize or improve. Weight loss continues at a steady pace. Cumulative results from the full treatment period become visible in the mirror, in clothing fit, and in metabolic markers. Energy levels often improve as the body becomes more efficient at accessing fat stores.
Week 17 and beyond (maintenance dose). At the highest prescribed dose, appetite suppression is maximized. Weight loss continues but may slow as you approach a new equilibrium. This is normal and does not indicate treatment failure. The long-term phase is about sustaining results, optimizing body composition, and maintaining the metabolic improvements gained during the initial months.
Drug interactions and safety considerations
The semaglutide glycine B12 blend interacts with medications and supplements in ways that patients must understand before starting treatment.
Other GLP-1 medications. Never combine the semaglutide glycine B12 blend with any other GLP-1 receptor agonist, including Ozempic, Wegovy, liraglutide, or tirzepatide. Stacking GLP-1 agonists increases the risk of severe gastrointestinal events and hypoglycemia without proportional benefits.
Insulin and sulfonylureas. Patients taking insulin or sulfonylureas alongside semaglutide face increased hypoglycemia risk. Dose adjustments of these medications are typically necessary. Work closely with your prescriber to monitor blood glucose levels during the semaglutide titration phase.
Oral medications. Semaglutide slows gastric emptying, which can delay the absorption of oral medications. This is particularly relevant for medications with narrow therapeutic windows, like warfarin, thyroid hormones, and oral contraceptives. Discuss timing adjustments with your prescriber.
Phentermine. Some prescribers combine semaglutide with phentermine for enhanced weight loss, though this combination requires careful monitoring and is not universally recommended.
Alcohol. Drinking on semaglutide is not contraindicated but carries additional risks. Alcohol sensitivity often increases dramatically on GLP-1 therapy. Smaller amounts produce stronger effects. The delayed gastric emptying means alcohol stays in the stomach longer before absorption, which alters the expected timeline of intoxication.
Supplements. The GLP-1 companion supplement guide covers recommended additions during therapy, including magnesium (for constipation), electrolytes (for hydration), and additional protein supplementation.
Frequently asked questions
Is the semaglutide glycine B12 blend FDA approved?
No. The individual ingredient semaglutide is FDA-approved under the brand names Ozempic and Wegovy, but the compounded triple blend is not an FDA-approved combination product. It is prepared by compounding pharmacies under their regulatory framework, typically section 503A or 503B of the Federal Food, Drug, and Cosmetic Act.
Can I switch from regular semaglutide to the glycine B12 blend without restarting?
Yes, in most cases. If you are already on compounded semaglutide, switching to the triple blend is straightforward because the semaglutide component and concentration remain the same. Confirm the concentration matches your current formulation and continue at your current dose level. Consult your prescriber before making any changes.
How long does a vial of the blend last?
This depends on your dose level, the vial size, and the beyond-use dating from the pharmacy. A 5 mL vial at the starting dose of 0.04 mL per week would last over 100 weeks mathematically, but beyond-use dates of 30 to 90 days mean you will likely not use the entire vial before it expires. At higher maintenance doses of 0.44 mL weekly, a 5 mL vial lasts approximately 11 weeks. Understanding semaglutide shelf life prevents waste.
Does the glycine in the blend have any side effects?
Glycine at the doses used in compounded semaglutide formulations (typically 5 mg/mL) is extremely well-tolerated. Glycine is a naturally occurring amino acid present in virtually all protein-containing foods. Side effects specific to the glycine component are essentially nonexistent at these concentrations. Some patients report improved sleep quality, which is consistent with the known neurological effects of glycine at higher supplemental doses.
Can I take additional B12 supplements while using the blend?
Additional oral or sublingual B12 supplementation is generally safe alongside the blend, though likely unnecessary for most patients since the injection provides direct B12 delivery. Patients with documented B12 deficiency or absorption disorders may benefit from additional supplementation under medical guidance.
Is the blend safe during pregnancy or breastfeeding?
No semaglutide formulation, including the glycine B12 blend, should be used during pregnancy. Semaglutide should be discontinued at least two months before planned conception. For breastfeeding considerations, our guide on GLP-1 and breastfeeding covers the current safety data. Patients who become pregnant unexpectedly while on GLP-1 therapy should discontinue immediately and contact their healthcare provider.
How does the blend compare to semaglutide with just B12 (no glycine)?
Semaglutide with B12 alone addresses nausea management and nutrient protection but does not include the muscle-preserving benefits of glycine. The semaglutide with B12 dosage chart covers that specific formulation. The triple blend adds the glycine component for patients who want comprehensive coverage of all three GLP-1 therapy concerns: efficacy, tolerability, and body composition.
What if I experience severe nausea even on the blend?
While the B12 component may reduce nausea severity, it does not eliminate it entirely. If nausea is severe, hold at your current dose for an additional two to four weeks. Eat smaller meals. Avoid trigger foods. Ginger tea and dietary modifications can help. If nausea persists at the starting dose, consult your prescriber about alternative approaches or dose adjustments.
For researchers serious about optimizing their GLP-1 protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed formulation comparisons, and a community of thousands who have navigated these exact questions.
External resources
Glycine Protects Muscle Cells From Wasting via mTORC1 Signaling (Frontiers in Nutrition)
Glycine Supplementation During Calorie Restriction (Clinical Nutrition)
GLP-1 in Diabetes Care: Nausea and Vomiting Mechanisms (British Journal of Pharmacology)
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay potent, your muscle mass stay protected, and your protocols stay consistent.