Feb 10, 2026

That vial sitting on your bathroom counter right now is losing potency. Every minute compounded tirzepatide spends outside the fridge, chemical reactions chip away at the peptide bonds holding the molecule together. And most people have no idea how fast it happens.
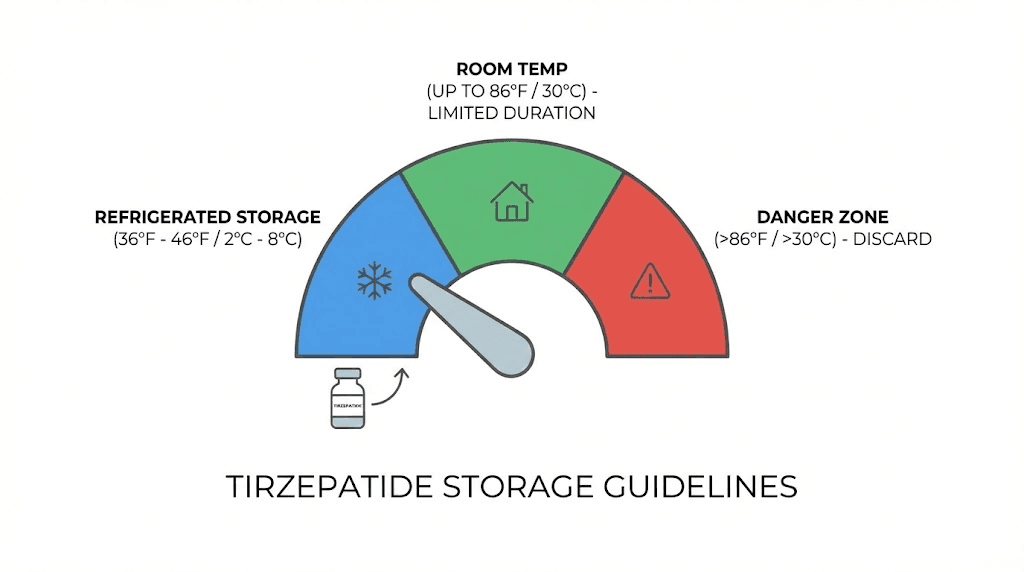
Here is the short answer. Compounded tirzepatide can remain at room temperature for up to 21 days, provided the ambient temperature stays below 86 degrees Fahrenheit (30 degrees Celsius). But that number comes with caveats that most storage guides conveniently ignore. The formulation matters. The preservatives matter. Whether you have already punctured the vial matters. And the temperature in your bathroom, kitchen, or car during summer is almost certainly higher than you think.
This guide breaks down the real science behind tirzepatide storage requirements, the degradation mechanisms that destroy potency at room temperature, the differences between compounded and brand-name formulations, and exactly what to do if you accidentally left your vial out overnight. Whether you are traveling, managing a weekly injection schedule, or simply forgot to put it back after your last dose, you will find specific answers here. Not generic advice. Real protocols backed by pharmaceutical stability data and peptide storage science.
SeekPeptides has compiled everything researchers need to know about maintaining tirzepatide potency outside refrigeration, including the storage mistakes that waste the most medication and the simple fixes that prevent them.
The 21-day rule and why it is more complicated than it sounds
Every pharmacy label, every storage guide, and every FAQ page gives you the same number. Twenty-one days. That is how long compounded tirzepatide can sit at room temperature before you should discard it. Simple enough.
Except it is not simple at all.
The 21-day guideline originates from stability data on brand-name tirzepatide products like Mounjaro and Zepbound. Eli Lilly conducted extensive stability testing on their proprietary formulations, which use specific buffers, stabilizers, and delivery systems designed to protect the peptide at controlled room temperature. Compounded tirzepatide is a different product entirely. It may use different excipients, different concentrations, and different preservatives. Compounding pharmacies assign their own beyond-use dates based on USP 797 guidelines, and those dates can range from 28 to 90 days when refrigerated, depending on the pharmacy and the specific formulation.
So where does the 21-day room temperature limit come from for compounded versions? In most cases, pharmacies default to the manufacturer data for the branded product and apply it broadly. Some pharmacies conduct their own stability testing. Others follow conservative USP guidelines that may actually give you less time than 21 days, depending on the formulation category.
The practical takeaway is straightforward. Check your pharmacy label first. If it specifies a room temperature duration, follow that number exactly. If it does not, the 21-day limit is a reasonable starting point, but only if your room temperature genuinely stays below 86 degrees Fahrenheit.
Temperature is the variable most people underestimate
When pharmacists say "room temperature," they mean a controlled environment between 68 and 77 degrees Fahrenheit (20 to 25 degrees Celsius), with allowable excursions up to 86 degrees Fahrenheit (30 degrees Celsius). That last number is the critical threshold. Above 86 degrees, degradation accelerates significantly.
Think about where you actually store your medication. A bathroom counter during a hot shower can exceed 86 degrees easily. A kitchen counter near a stove or dishwasher runs warm. A car parked in direct sunlight can reach 140 degrees Fahrenheit in minutes. Even a nightstand in a room without air conditioning during summer months can push past the safe limit.
The problem is cumulative. Every hour above 86 degrees does not just pause the 21-day clock. It accelerates degradation exponentially. Two hours at 95 degrees might cause more damage than two full days at 72 degrees. Researchers who understand tirzepatide shelf life in refrigeration sometimes assume room temperature follows the same linear pattern, but heat-driven degradation does not work that way.
What happens at the molecular level when tirzepatide warms up
Tirzepatide is a dual GIP and GLP-1 receptor agonist, a 39-amino-acid peptide with a fatty acid chain attached. That fatty acid chain, specifically a C20 fatty diacid, is what gives tirzepatide its extended half-life in the body. But it also makes the molecule sensitive to temperature-driven degradation through several pathways.
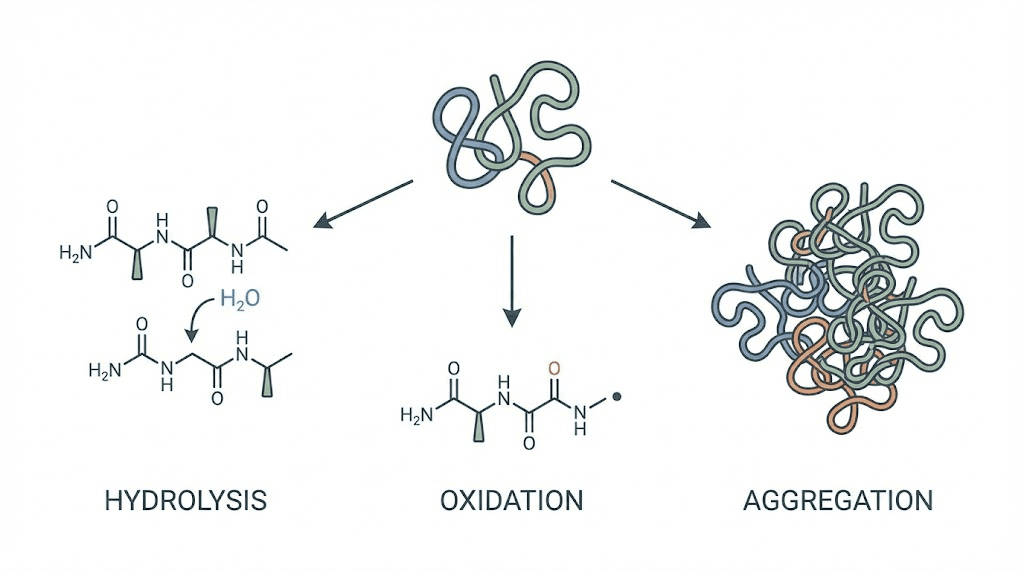
Aggregation is the primary concern. As temperature rises, peptide molecules begin to unfold from their native conformation. These unfolded molecules stick together, forming clumps that reduce the effective dose and can potentially cause injection site reactions. You might notice this as slight cloudiness in a solution that was previously clear.
Oxidation attacks specific amino acid residues. Methionine and tryptophan residues in the peptide chain are particularly vulnerable. Oxidized peptides retain their structure but lose biological activity, meaning your dose looks fine but does not work as expected. This is why some researchers report stalled weight loss on tirzepatide after storage issues without realizing the connection.
Hydrolysis breaks peptide bonds directly. Moisture in the air, combined with warmth, accelerates this reaction. Each broken bond reduces the intact peptide concentration in your vial. Unlike aggregation, hydrolysis fragments are too small to see.
Deamidation alters asparagine and glutamine residues, changing the peptide sequence itself. This reaction is temperature-dependent and essentially irreversible once it occurs. The resulting modified peptide may bind to receptors differently or not at all.
None of these processes announce themselves with a dramatic color change or obvious warning sign. That is what makes temperature excursions so dangerous. The medication looks identical but performs differently.
Compounded versus brand-name tirzepatide storage differences
This distinction matters more than most people realize. Brand-name Mounjaro and Zepbound are manufactured in FDA-inspected facilities with precise formulation control. The excipients, buffers, and preservatives in each pen are identical batch to batch, tested extensively for stability at various temperatures. When Eli Lilly says 21 days at room temperature, that claim is backed by accelerated stability studies and real-time data submitted to the FDA.
Compounded tirzepatide operates under different rules.
Compounding pharmacies prepare tirzepatide formulations according to their own protocols, which must comply with USP 795 (non-sterile) or USP 797 (sterile compounding) guidelines. The beyond-use date assigned to your specific vial depends on the compounding category, the sterility testing performed, and the stability data available for that particular formulation. Some pharmacies invest in proprietary stability testing. Others rely on published literature and USP default BUD assignments.
Key differences that affect room temperature stability
Preservatives vary between pharmacies. Brand-name tirzepatide pens contain m-cresol as a preservative in multi-dose formulations. Compounded versions may use benzyl alcohol, methylparaben, or different preservative systems entirely. Each preservative has its own stability profile at elevated temperatures. Some degrade faster than others when warm, leaving the peptide solution more vulnerable to microbial contamination.
Concentration affects stability. Compounded tirzepatide often comes in different concentrations than brand-name products. Higher concentrations generally show better stability because the peptide-to-excipient ratio is different, but they can also be more prone to aggregation. If your compounding pharmacy provides tirzepatide at custom concentrations, the room temperature stability window may differ from the standard 21-day guideline.
Container type matters. Brand-name tirzepatide comes in single-dose pens or pre-filled syringes with minimal air exposure. Compounded versions typically come in multi-dose vials that you puncture repeatedly with a needle. Each puncture introduces a tiny amount of air and potential contaminants. The more times you access the vial, the shorter its effective room temperature life, regardless of what the label says.
Reconstitution introduces variables. If your compounded tirzepatide arrives as a lyophilized (freeze-dried) powder that requires reconstitution with bacteriostatic water, the reconstituted solution has different stability characteristics than the original powder. The clock starts the moment you add water, and room temperature exposure during reconstitution counts toward your total out-of-fridge time.
What your pharmacy label actually tells you
Read the label carefully. It contains three critical pieces of information.
First, the beyond-use date (BUD). This is the last date you should use the medication, assuming proper storage conditions. For refrigerated compounded tirzepatide, this typically ranges from 28 to 90 days after compounding. Once you remove it from the fridge, a separate clock begins.
Second, storage instructions. These should specify the required refrigeration temperature (usually 36 to 46 degrees Fahrenheit, or 2 to 8 degrees Celsius) and any room temperature allowances. If your label says "refrigerate at all times," that overrides the general 21-day guideline.
Third, the date of compounding. This tells you how old the product already is. A vial compounded six weeks ago has less remaining stability than one compounded yesterday, even if both have the same BUD. This matters when calculating how much room temperature time you can safely afford.
If your label lacks any of these details, contact your compounding pharmacy directly. Do not assume the standard 21-day room temperature guideline applies to your specific formulation without confirmation.
Signs your compounded tirzepatide has degraded
The challenge with peptide degradation is that early stages are invisible. By the time you notice obvious changes, significant potency loss has already occurred. But there are warning signs worth checking before every injection.
Visual inspection checklist
Cloudiness or haziness. Fresh compounded tirzepatide should be clear and colorless. Any cloudiness, even slight, indicates protein aggregation. Hold the vial up to a light source and look for any particles floating in the solution or a general haziness that was not present when you first opened it. This is the most reliable visual indicator of degradation.
Small particles. Tiny specks visible against a light background suggest peptide fragments or aggregated protein. These should not be present in a properly stored solution. Some people confuse tiny air bubbles with particles, so let the vial sit motionless for 30 seconds before inspecting.
Color changes. Any yellowing, browning, or other discoloration means the peptide has undergone chemical modification. A slight yellow tint is often the first visible sign of oxidation. Once you see color changes, the peptide has lost significant biological activity.
Increased viscosity. If the solution seems thicker when drawn into a syringe compared to when it was fresh, aggregation has likely progressed substantially. The "feel" of drawing the solution through the needle is actually a useful diagnostic tool that experienced researchers learn to trust.
Performance indicators of degraded tirzepatide
Visual inspection catches advanced degradation, but performance changes reveal earlier stages. If you are using tirzepatide for weight loss at established doses, pay attention to these changes.
Reduced appetite suppression. Tirzepatide primary mechanism involves GLP-1 and GIP receptor activation, which suppresses appetite. If your usual dose suddenly feels less effective at controlling hunger, storage-related potency loss is a possibility. Many researchers attribute this to tolerance when the real culprit is a degraded vial.
Changes in injection site response. Aggregated peptide can cause more injection site irritation than intact peptide. If you notice increased redness, swelling, or discomfort at injection sites compared to your normal experience, the solution may contain aggregated protein.
Return of symptoms. For those using tirzepatide to manage blood sugar or metabolic markers, unexplained worsening of these markers after stable control could indicate potency loss from improper storage. This is particularly relevant for researchers monitoring tirzepatide appetite suppression timelines.
Side effects without benefits. Degraded tirzepatide may still cause some side effects (particularly GI effects from partially active fragments) while providing reduced therapeutic benefit. If you experience nausea without the expected appetite suppression, degradation is worth investigating.

The accidentally-left-out scenario and what to do
You forgot. It happens. You pulled the vial out for your injection, got distracted by a phone call, and found it sitting on the counter eight hours later. Or you traveled and your cooler pack ran out. Or the power went out and your fridge warmed up overnight. Whatever the reason, your compounded tirzepatide spent time outside its recommended temperature range, and now you need to make a decision.
Do not panic. But do not ignore it either.
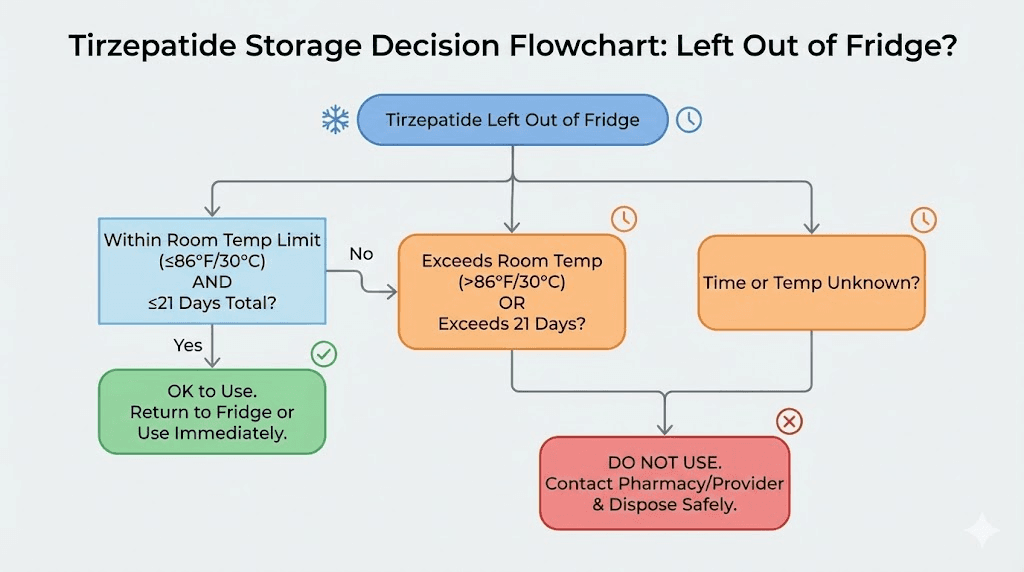
Short exposure: under 4 hours at room temperature
If your vial sat at normal room temperature (below 77 degrees Fahrenheit) for less than 4 hours, the impact on potency is minimal. This falls well within the allowable excursion parameters that pharmaceutical stability testing accounts for. Return the vial to the refrigerator and continue using it as normal.
One important caveat. Do not repeatedly cycle the vial between fridge and room temperature. Each warming and cooling cycle stresses the peptide more than continuous storage at either temperature. If you are taking the vial out for each injection, try to minimize the time outside the fridge to under 15 minutes per dose.
Moderate exposure: 4 to 24 hours at room temperature
A full night on the counter. A day in your bag during travel. These scenarios are common and usually recoverable, provided the temperature stayed below 86 degrees Fahrenheit. The vial has now consumed roughly half a day to one full day of its 21-day room temperature budget.
Return it to the fridge immediately. Mark the date and duration of exposure on the vial with a marker or adhesive label. Subtract the exposure time from your remaining 21-day allowance. If the vial was already partially through its room temperature budget from previous exposures, account for the cumulative total.
Inspect the solution before your next use. Check for any cloudiness, particles, or color changes that were not present before the exposure. If everything looks clear, you can continue using it.
Extended exposure: 24 hours to 7 days at room temperature
This is where judgment calls become necessary. One to seven days at controlled room temperature (below 86 degrees Fahrenheit) consumes a significant portion of the 21-day allowance but does not automatically render the medication unusable.
The key question is temperature. If the environment stayed below 77 degrees consistently, the peptide has likely retained most of its potency. If temperatures fluctuated above 86 degrees for any portion of that time, degradation may be more significant than the time alone would suggest.
Use the vial promptly. Do not push it further by allowing additional room temperature time. Keep it continuously refrigerated between doses and plan to use the remaining contents within the next week or two rather than stretching it out.
Extreme exposure: above 86 degrees Fahrenheit for any duration
High heat is the deal-breaker. If your compounded tirzepatide was exposed to temperatures above 86 degrees Fahrenheit, even for a few hours, the degradation rate accelerates dramatically. A car dashboard in summer, a hot bathroom during showers, proximity to a heater or oven, these scenarios can push temperatures well beyond what the peptide can tolerate.
Brand-name tirzepatide (Mounjaro/Zepbound) has demonstrated stability for up to 7 days at temperatures up to 104 degrees Fahrenheit (40 degrees Celsius) based on Eli Lilly stability data for travel conditions. But this is specific to the branded formulation with its optimized stabilizer system. Compounded versions may not share this resilience.
When in doubt after heat exposure, contact your compounding pharmacy. Describe the conditions and duration. They can advise based on their specific formulation stability profile. If you cannot reach them and the medication shows any visual changes, discard it and request a replacement.
What about freezing?
Never use tirzepatide that has been frozen. Freezing causes ice crystal formation within the solution, which physically shears peptide molecules and denatures the protein irreversibly. Unlike heat exposure, which causes gradual degradation, freezing can destroy a significant percentage of active peptide in a single event.
This matters for travel. Cargo holds on aircraft can reach freezing temperatures. Ice packs placed directly against a vial can freeze the adjacent liquid. Even a refrigerator set too cold can freeze medication stored against the back wall where temperatures are lowest. Keep your tirzepatide between 36 and 46 degrees Fahrenheit, and never let it freeze. If it does freeze accidentally, do not thaw and use it. Discard the vial.
Understanding proper peptide storage after reconstitution helps prevent both heat and freezing mistakes.
Travel storage solutions for compounded tirzepatide
Traveling with compounded tirzepatide requires more planning than most people expect. The challenge is maintaining a temperature between 36 and 46 degrees Fahrenheit during transit without accidentally freezing the medication. Here is what actually works and what does not.
Medical-grade insulated travel cases
Purpose-built medication travel cases are the gold standard for trips longer than a few hours. These cases use pre-conditioned gel packs or phase-change materials that maintain temperatures in the refrigeration range for 12 to 48 hours depending on the product and ambient conditions.
The key features to look for include a temperature indicator or built-in thermometer, insulation rated for your travel duration, separation between gel packs and medication (direct contact can cause localized freezing), and enough capacity for your full supply plus syringes and alcohol swabs.
For trips under one week in temperate climates, a quality insulated case with two gel packs can maintain safe temperatures without access to a refrigerator. For longer trips or hot climates, plan refrigerator access at your destination.
Short trips under 3 days
For weekend getaways or short business trips, a basic insulated lunch bag with one or two gel packs works adequately. Wrap the vial in a cloth or paper towel to prevent direct contact with the gel pack, which can freeze the outer layer of solution. Place the bag in your carry-on luggage, never in checked bags where cargo hold temperatures are unpredictable.
At your destination, refrigerate the vial immediately. If no refrigerator is available, keep the insulated bag closed as much as possible and replace gel packs with fresh ice wrapped in a barrier (like a Ziploc bag inside a towel) if the original packs warm up.
Extended travel: 4 to 14 days
Longer trips need a real strategy. Options include booking accommodations with refrigerators (most hotels provide mini-fridges on request), using the 21-day room temperature allowance strategically, or carrying a portable injection system with proper cooling.
If you plan to use the room temperature allowance, bring only enough medication for the trip duration plus a small buffer. Leave your main supply refrigerated at home. This limits your financial exposure if something goes wrong with storage during travel.
For hot-climate destinations, consider portable USB-powered medication coolers. These small devices maintain refrigeration temperatures using thermoelectric cooling and can run off a portable battery bank for 8 to 12 hours between charges. They cost $40 to $80 and provide peace of mind that passive cooling cannot match.
Air travel specific considerations
Carry your medication in your carry-on bag. Always. Checked luggage is subject to temperature extremes in cargo holds, physical impacts during handling, and potential delays or loss. TSA allows injectable medications through security with appropriate documentation.
Bring your pharmacy label or a letter from your prescribing provider confirming the medication. While TSA agents typically recognize medication vials without issue, documentation prevents delays. Syringes and needles are permitted when accompanied by the medication they are intended for.
Gel packs for medication cooling are allowed through TSA security regardless of the liquid restrictions. They can be frozen solid when you go through the checkpoint. Declare them at the screening point to avoid confusion.
During the flight, your carry-on under the seat in front of you is the best location. The overhead bin can get warmer than the cabin, especially during long flights. If your flight is over 6 hours, the cabin temperature is typically maintained around 68 to 72 degrees Fahrenheit, which is well within the safe range even without active cooling.
What to avoid when traveling with tirzepatide
Do not leave the medication in a parked car. Interior temperatures in direct sunlight can exceed 140 degrees Fahrenheit within 30 minutes, even in mild weather. This is the single most common cause of medication spoilage during travel.
Do not store it in the glove compartment or center console. These compartments trap heat and provide no airflow. A cooler in the back seat or trunk (in mild weather) is better, but the best option is always to take it with you.
Do not put gel packs directly against the vial. Wrap the vial in fabric first. Direct contact with a frozen gel pack can freeze the solution at the contact point, damaging that portion of the medication even if the overall temperature seems fine.
Do not assume your hotel room refrigerator maintains proper temperatures. Many hotel mini-fridges run warmer than a standard household refrigerator. If possible, check the internal temperature with a thermometer or use the coldest setting available. Avoid the freezer compartment, which can over-freeze medications placed too close.
Storage best practices that preserve maximum potency
Beyond the "keep it cold" basics, several less obvious practices significantly extend the usable life of your compounded tirzepatide.
Minimize light exposure
Light, particularly UV light, accelerates peptide degradation through photolysis. Store your vial in its original box or a dark container when refrigerated. When the vial is out for injection, minimize the time it spends in direct light. This is especially important for clear glass vials that provide no UV protection. Amber glass vials offer better light protection but are less common for compounded formulations.
Researchers who understand general peptide refrigeration guidelines know that light exposure compounds the effects of temperature stress.
Reduce air exposure
Every time you puncture the vial stopper with a needle, you introduce a small volume of air into the headspace. Oxygen in this air promotes oxidation reactions. While the preservative system provides some protection, accumulated air exposure over many punctures reduces stability.
Practical steps to minimize air exposure include keeping the vial upright so the stopper stays above the liquid, using the smallest gauge needle that works for your injection (smaller punctures seal better), and not removing the stopper or cap unnecessarily.
For multi-dose vials that will last weeks, some researchers use proper reconstitution techniques and sterile technique consistently to prevent contamination that accelerates degradation.
The right spot in your refrigerator
Where you place the vial in your fridge matters more than you might think.
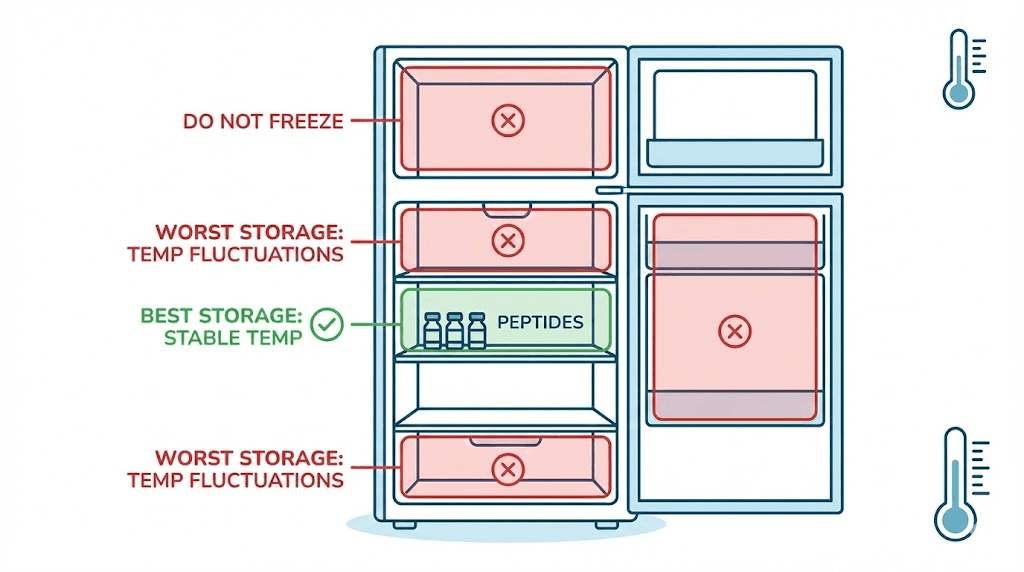
The door shelf is the worst location. It experiences the largest temperature swings every time the door opens. Temperatures on door shelves can fluctuate 10 to 15 degrees with regular use throughout the day.
The back wall is risky for a different reason. Many refrigerators have cooling coils behind the back wall, creating cold spots that can approach or reach freezing temperatures. If your lettuce occasionally freezes in the back of your fridge, your medication could too.
The ideal location is the middle shelf, toward the center of the refrigerator. This zone maintains the most consistent temperature with the least variation from door openings or cooling cycles. Store the vial in its original box or a small container to add a thermal buffer against rapid temperature changes.
Consider placing a small thermometer near your medication storage spot. Cheap refrigerator thermometers cost a few dollars and provide confirmation that your storage temperature stays between 36 and 46 degrees Fahrenheit. If you notice readings outside this range, adjust your fridge settings before trusting your medication to that location.
How compounded tirzepatide storage compares to other peptides
Understanding where tirzepatide falls on the peptide stability spectrum helps contextualize these storage guidelines. Not all peptides are equally fragile, and knowing the relative sensitivity helps calibrate your storage practices.
Tirzepatide versus semaglutide storage
Semaglutide and tirzepatide share similar storage requirements. Both can remain at room temperature for up to 21 days (for branded formulations). Both degrade through similar mechanisms. The primary structural difference, tirzepatide dual GIP/GLP-1 activity versus semaglutide single GLP-1 activity, does not significantly affect storage stability.
Compounded versions of both peptides follow the same general principles. Compounded semaglutide fridge storage timelines and semaglutide refrigeration requirements parallel those for tirzepatide closely. If you have experience storing one, the same practices apply to the other.
Tirzepatide versus shorter peptides
Shorter peptides like BPC-157 (15 amino acids) are generally more stable than tirzepatide (39 amino acids) at room temperature. Fewer amino acids mean fewer potential degradation sites and simpler folding that is more resistant to thermal denaturation. BPC-157 in reconstituted form can tolerate brief room temperature exposure better than tirzepatide, though refrigeration is still recommended for both.
TB-500 (43 amino acids) has a similar size to tirzepatide and comparable stability concerns. The same storage practices that protect tirzepatide apply to TB-500 and other medium-length therapeutic peptides.
General peptide storage hierarchy
As a general rule, peptide room temperature stability follows a pattern:
Most stable at room temperature: Short peptides (under 15 amino acids) without complex modifications. Small, simple structures resist thermal degradation better.
Moderately stable: Medium-length peptides (15 to 50 amino acids) including tirzepatide, semaglutide, and most therapeutic peptides. These require refrigeration but tolerate brief room temperature exposure.
Least stable: Large peptides and complex proteins with extensive post-translational modifications. Growth factors, antibodies, and heavily modified peptides degrade fastest outside refrigeration.
Tirzepatide falls squarely in the moderate category. Its fatty acid modification (the C20 diacid) adds some bulk and hydrophobic character that can promote aggregation at elevated temperatures, but the overall stability profile is manageable with basic refrigeration practices.
For detailed protocols on lyophilized peptide storage and reconstituted peptide shelf life, these dedicated guides provide additional context.
Common storage mistakes and how to fix them
After reviewing thousands of peptide storage questions, certain mistakes appear repeatedly. Most are easy to fix once you know about them.
Mistake 1: using the door shelf
Already covered above, but it bears repeating because it is the single most common storage error. The door shelf is convenient. It is also the worst spot in your fridge for temperature-sensitive medications. Move your tirzepatide to the middle shelf and this mistake is permanently solved.
Mistake 2: not tracking cumulative room temperature time
Many people treat the 21-day limit as a single continuous event. In reality, room temperature exposure is cumulative. Fifteen minutes out for each injection, twice a week, adds up. Over 10 weeks, those brief exposures total about 5 hours, negligible in the context of a 21-day allowance. But if you also left the vial out for a full day during travel and another 8 hours when you forgot it on the counter, your cumulative total is closer to 37 hours, still manageable but worth tracking.
Keep a simple log. Note the date and approximate duration each time the vial spends time outside the fridge beyond routine injections. This log becomes critical if you need to assess whether a vial is still usable after an unplanned exposure event.
Mistake 3: returning warm medication to the fridge repeatedly
Thermal cycling, repeatedly warming and cooling a medication, stresses peptide molecules more than continuous storage at a stable temperature. Each cycle forces the peptide through conformational changes that can accelerate aggregation. While occasional returns to the fridge are fine (and better than leaving it out), try to minimize the number of warming cycles.
The practical solution is to take the vial out, prepare your injection efficiently, and return it to the fridge within 5 to 10 minutes. Do not leave it out "to warm up for comfort." Cold injections are slightly less comfortable but do not affect the medication efficacy, while warm storage definitively reduces it.
Mistake 4: ignoring the beyond-use date
The BUD on your label is not a suggestion. It represents the last date your compounding pharmacy can guarantee the medication meets potency and sterility standards. Using medication past its BUD means you are injecting a product with unknown potency and potentially compromised sterility.
If you find yourself with unused medication past the BUD, discard it. The cost of a replacement vial is always less than the risks of using degraded or contaminated medication. Track your usage patterns and order quantities that match your actual consumption to minimize waste.
Researchers familiar with expired semaglutide safety concerns understand that the same principles apply to expired tirzepatide. Once past the BUD, potency cannot be guaranteed.
Mistake 5: storing near strong odors or chemicals
This one surprises people. Rubber vial stoppers are not perfectly impermeable. Volatile compounds from cleaning products, perfumes, or other chemicals stored nearby can potentially permeate the stopper over extended storage periods and interact with the medication. Keep your medication in a clean area of the refrigerator, away from strong-smelling foods or chemicals.
Temperature monitoring tools worth considering
For researchers managing valuable peptide medications, a few inexpensive tools provide significant peace of mind.
Refrigerator thermometers
A basic refrigerator thermometer costs $5 to $15 and provides at-a-glance confirmation that your storage temperature is in range. Digital versions with min/max memory are particularly useful because they record the highest and lowest temperatures reached since the last reset, catching overnight fluctuations or power outages you might otherwise miss.
Place the thermometer near where you store your medication, not on the door or near the back wall. Check it weekly and after any power outages.
Data logging thermometers
For more detailed monitoring, data loggers record temperatures at set intervals (typically every 15 to 60 minutes) and store the data for later review. Some models connect via Bluetooth to a smartphone app, providing alerts if the temperature exceeds preset limits. These run $20 to $50 and are particularly valuable for researchers managing expensive peptide inventories or those with unreliable refrigerators.
Smart home temperature alerts
If you use a smart home system, temperature sensors placed in your refrigerator can send push notifications to your phone when temperatures deviate from your set range. This provides real-time alerts for power outages, door-ajar situations, or thermostat failures that could compromise your medication before you discover the issue visually.
Combining proper monitoring with comprehensive peptide storage protocols creates a system where temperature excursions are caught early, before they cause significant damage.
Specific scenarios and storage timelines
Let us address the most common real-world situations with specific guidance.
Scenario 1: weekly injection from a multi-dose vial
You take the vial from the fridge, draw your dose, and return it within 10 minutes. Over a typical 4-week vial life, total room temperature exposure is about 40 minutes. This is negligible and well within safe limits. No special precautions needed beyond basic refrigeration between uses.
Scenario 2: traveling for a weekend
Remove the vial Friday evening, use an insulated bag with gel packs for transport, inject Saturday or Sunday, and return to refrigeration Sunday night. Total room temperature exposure is approximately 48 to 60 hours. Well within the 21-day limit. No concerns.
Scenario 3: two-week vacation without reliable refrigeration
This uses most of your 21-day room temperature budget. Plan carefully. Bring only the doses you need (pre-calculated using a dosage calculator for your tirzepatide dose in units). Use a portable cooler with replaceable gel packs. Seek refrigeration wherever possible (hotel reception desks, pharmacy cold storage, restaurant kitchens, or convenience stores will sometimes hold medication briefly). Mark the vial with the date it left refrigeration and the cumulative hours at room temperature. Use the remaining doses within 7 days of returning home.
Scenario 4: power outage during summer
A closed refrigerator maintains safe temperatures for approximately 4 hours. An unopened freezer holds temperature for 24 to 48 hours. During an extended power outage, your medication starts warming gradually, so the clock does not start immediately.
If the power was out less than 4 hours, your refrigerator likely maintained acceptable temperatures. Continue using the medication normally after confirming the fridge has cooled back down.
If the power was out 4 to 12 hours in a warm environment, your medication spent time in an uncertain temperature zone. Check the fridge internal temperature when power returns. If it stayed below 46 degrees Fahrenheit, the medication is fine. If it warmed above that, treat it as room temperature exposure and subtract those hours from your 21-day allowance.
If the power was out longer than 12 hours during warm weather, consider the medication as having been at room temperature for the duration. Apply the cumulative exposure rules above.
Scenario 5: medication delivered and left on the porch
Mail-order compounded tirzepatide should arrive in insulated packaging with cold packs. But delivery timing is unpredictable. If your package sat in the sun for hours, the internal temperature may have exceeded safe limits.
When you receive a delivery, check the cold packs. If they are still partially frozen or cold to the touch, the medication likely stayed in range. If the cold packs are room temperature and the outer package is warm, the medication experienced a temperature excursion. Contact your pharmacy, as many will replace shipments that experienced verified temperature problems during transit.
To minimize this risk, schedule deliveries for times when someone can receive the package promptly, use signature-required shipping when possible, and request that the pharmacy ship earlier in the week so packages do not sit in a warehouse over the weekend.
The relationship between storage and dosage effectiveness
Storage quality directly affects dosing accuracy. This connection is not always obvious but is critically important for researchers trying to optimize their tirzepatide dosing protocols.
Consider this. If improper storage has degraded 15% of the active peptide in your vial, every dose you draw contains 15% less active ingredient than intended. For someone on a 5mg weekly dose, that means each injection delivers only 4.25mg of functional tirzepatide. The remaining 0.75mg is degraded peptide that occupies volume but provides no therapeutic benefit.
This has cascading effects. Appetite suppression may seem inadequate, prompting dose increases. Weight loss plateaus, causing frustration. Side effects may decrease (because the effective dose is lower), giving a false impression that tolerance has developed. Researchers who track their tirzepatide response timelines may attribute variable results to biological factors when storage was actually the issue.
The solution is simple. Maintain proper storage consistently, and you eliminate a major variable from your dosing equation. Every injection delivers what it should. Results become more predictable. And dose adjustments are based on genuine biological response rather than storage artifacts.
For researchers who are not seeing expected results, storage evaluation should be the first troubleshooting step, before considering dose changes, switching medications, or adding supplementary compounds like B12 formulations or niacinamide combinations.
Understanding beyond-use dates for compounded medications
The beyond-use date system for compounded medications is fundamentally different from manufacturer expiration dates, and understanding this difference helps contextualize room temperature storage decisions.
How BUDs are assigned
Compounding pharmacies assign BUDs based on one of three approaches, listed from most conservative to most permissive.
USP default assignments. When no stability data exists for a specific formulation, USP chapters 795 and 797 provide default BUD values. These are conservative, typically 24 to 48 hours for sterile preparations at room temperature without stability testing. Pharmacies using these defaults are being cautious, which benefits patient safety but may be unnecessarily restrictive for well-characterized formulations.
Literature-based assignments. Pharmacies can extend BUDs beyond USP defaults by referencing published stability studies for similar formulations. If peer-reviewed research demonstrates that tirzepatide in a specific buffer system maintains potency for 90 days refrigerated, a pharmacy using the same buffer can reference that data to support a longer BUD.
Proprietary stability testing. The most reliable approach involves the pharmacy conducting its own stability testing using validated analytical methods. This testing measures peptide potency, purity, and sterility at various time points and storage conditions, producing data specific to their exact formulation. Pharmacies that invest in this testing can assign longer, more accurate BUDs.
What BUDs do not tell you
BUDs assume proper storage conditions throughout the product life. They do not account for the time you leave the vial on your counter, the temperature swings in your car during transport, or the brief periods of warming during each injection. These real-world conditions mean the actual usable life of your medication may be shorter than the BUD suggests.
Think of the BUD as a best-case expiration date under ideal conditions. Your real expiration date is the BUD minus any accumulated storage stress from temperature excursions, light exposure, and repeated vial punctures.
Frequently asked questions
Can I put compounded tirzepatide back in the fridge after it has been at room temperature?
Yes, you can return it to refrigeration after room temperature exposure. This does not "reset" the degradation clock, but it does stop further room temperature degradation. The peptide retains whatever potency it had when you returned it to the fridge. Avoid repeated cycling between warm and cold temperatures, as thermal stress accelerates degradation. If the vial was at room temperature for less than 24 hours and stayed below 86 degrees Fahrenheit, it is generally safe to continue using.
Does compounded tirzepatide go bad faster than brand-name Mounjaro?
It depends on the specific compounded formulation. Brand-name Mounjaro benefits from extensive stability testing, optimized preservative systems, and consistent manufacturing conditions. Some compounded formulations match or approach this stability, while others may degrade somewhat faster due to different excipients or preservative concentrations. Follow your pharmacy specific storage instructions rather than assuming brand-name guidelines apply exactly.
What temperature is too hot for compounded tirzepatide?
Above 86 degrees Fahrenheit (30 degrees Celsius), degradation accelerates significantly. Temperatures above 104 degrees Fahrenheit (40 degrees Celsius) can cause rapid potency loss within hours. Never expose tirzepatide to temperatures above 86 degrees for extended periods. Brief exposure during normal handling (removing the vial cap, drawing a dose) is acceptable as long as the overall environment stays below 86 degrees.
Can I tell if my tirzepatide has lost potency just by looking at it?
You can detect advanced degradation visually. Look for cloudiness, particles, color changes (especially yellowing), or changes in viscosity. However, early-stage potency loss (5 to 20%) is invisible. The solution can look perfectly clear and colorless while containing significantly degraded peptide. Visual inspection catches severe problems but cannot confirm that the medication retains its full labeled potency.
How many times can I take tirzepatide out of the fridge for injections?
There is no strict limit on the number of times you remove the vial, as long as each exposure is brief (under 15 minutes) and the cumulative room temperature time stays well within the 21-day allowance. For a typical 4-week vial with weekly injections, four brief exposures totaling about 40 to 60 minutes is completely normal and has minimal impact on potency. Minimize each exposure duration rather than worrying about the count.
Should I warm my tirzepatide before injecting?
No. While warmer injections may feel slightly more comfortable, the brief warming period does not improve absorption or effectiveness. The comfort benefit is marginal and not worth the additional room temperature exposure. If injection comfort is a concern, consider using a finer gauge needle or applying the injection to a different site rather than warming the medication.
What happens if my compounded tirzepatide was shipped without cold packs?
Contact your pharmacy immediately. Compounded sterile medications should be shipped with appropriate cold chain packaging. If the medication arrived at room temperature without cold packs and the transit time exceeded a few hours, the pharmacy should replace it at no charge. Do not use medication that may have been at uncontrolled temperatures for an unknown duration during shipping.
Is lyophilized (powder) tirzepatide more stable than the liquid form?
Yes, significantly. Lyophilized peptides are much more stable than reconstituted solutions because the removal of water eliminates hydrolysis and slows most other degradation pathways. Lyophilized tirzepatide can be stored at room temperature for extended periods (months in many cases) without significant potency loss. However, once reconstituted with bacteriostatic water, the reconstituted solution follows the same storage rules as pre-made liquid formulations. For detailed guidance on handling powder forms, see our guide on peptide powder storage and stability.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols for members. Whether you are managing a single vial or an extensive research inventory, SeekPeptides offers the evidence-based guidance and community support that turns storage uncertainty into confident practice.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your storage practices stay consistent.