Mar 3, 2026

Experienced researchers know something that most beginners miss about compounded tirzepatide. The peptide itself is only half the equation. What gets combined with it, and why, can shift the entire trajectory of a weight loss protocol.
Levocarnitine is one of those additions. Not flashy. Not heavily marketed. But backed by a cellular mechanism that addresses one of the biggest blind spots in GLP-1 therapy: what happens to stored fat once appetite drops.
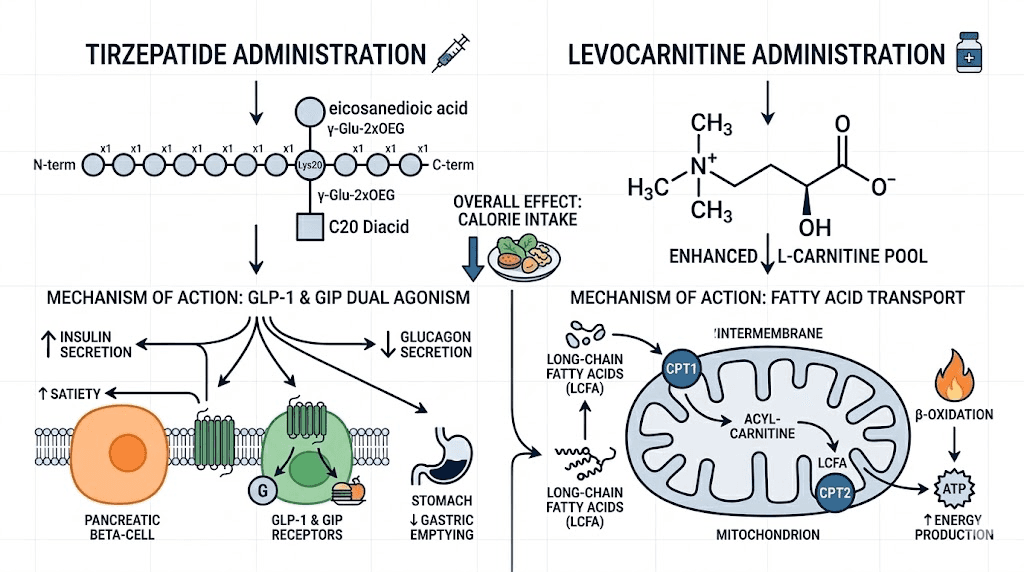
Here is the problem most people do not think about. Tirzepatide suppresses appetite effectively. Calorie intake falls. The body enters a deficit. But a caloric deficit alone does not guarantee efficient fat burning. The body needs a functioning transport system to shuttle fatty acids into the mitochondria where they actually get oxidized for energy. Without adequate carnitine levels, that process slows. Fat sits in storage even when the body needs fuel. Energy drops. Fatigue sets in. And a significant portion of the weight lost comes from lean tissue instead of adipose tissue.
That is where levocarnitine enters the picture. This guide covers everything about the tirzepatide levocarnitine compound, from the cellular mechanisms that make the combination logical, to the dosing protocols compounding pharmacies use, to the safety considerations that every researcher needs to understand before starting. Whether you are evaluating this blend for the first time or trying to determine if it makes sense for your current tirzepatide protocol, this is the comprehensive reference you need.
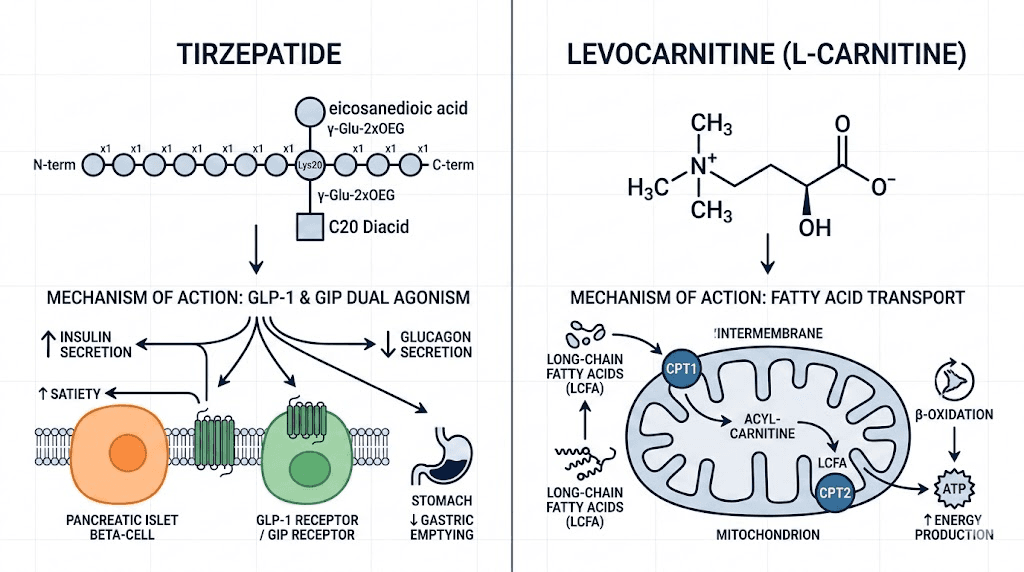
What is tirzepatide levocarnitine?
Tirzepatide levocarnitine is a compounded injectable formulation that combines two distinct compounds into a single weekly injection. The first component, tirzepatide, is a dual GIP and GLP-1 receptor agonist that reduces appetite, slows gastric emptying, and improves insulin sensitivity. The second component, levocarnitine (also called L-carnitine), is a naturally occurring amino acid derivative that serves as the primary transport molecule for long-chain fatty acids into the mitochondria.
Think of it this way. Tirzepatide creates the caloric deficit. Levocarnitine optimizes what happens with the fat that needs to be burned during that deficit.
Compounding pharmacies prepare this blend in various concentrations, with the most common formulations containing tirzepatide at 10 mg/mL alongside levocarnitine at 100 mg/mL. Some pharmacies offer higher tirzepatide concentrations, such as 17 mg/mL, while maintaining the same levocarnitine concentration. The resulting injection delivers both compounds subcutaneously in a single weekly dose, eliminating the need for separate administrations.
This is not an FDA-approved combination. No clinical trial has evaluated this specific blend as a unified product. However, both individual components have substantial research behind them. Tirzepatide received FDA approval for type 2 diabetes (as Mounjaro) and for chronic weight management (as Zepbound). Levocarnitine has FDA approval as a standalone treatment for primary and secondary carnitine deficiency. The compound formulation brings these two evidence-based ingredients together based on a theoretical rationale about complementary mechanisms, not on direct combination studies.
Understanding that distinction matters. The individual components work. The theory behind combining them is sound. But the specific synergy has not been proven in controlled human trials. That context should inform every decision you make about this blend.
How levocarnitine works at the cellular level
Levocarnitine is not a peptide. It is a quaternary ammonium compound that the body synthesizes from the amino acids lysine and methionine, primarily in the liver, kidneys, and brain. A 70 kg person produces approximately 11 to 34 mg of levocarnitine per day through endogenous synthesis. The rest comes from dietary sources, predominantly red meat, dairy products, and fish.
Its primary function is elegantly specific. Levocarnitine acts as a shuttle.
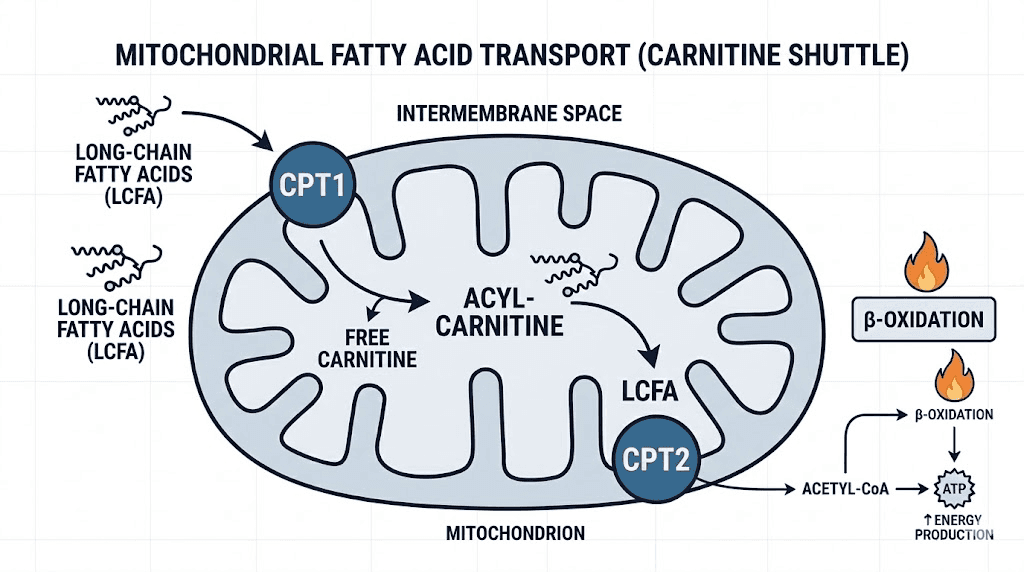
Long-chain fatty acids cannot cross the inner mitochondrial membrane on their own. They require a transport system called the carnitine shuttle. Here is how it works: the enzyme carnitine palmitoyltransferase I (CPT-I) on the outer mitochondrial membrane converts fatty acyl-CoA to fatty acylcarnitine by attaching it to a carnitine molecule. This complex then crosses the inner membrane via a translocase protein. Once inside the mitochondrial matrix, carnitine palmitoyltransferase II (CPT-II) removes the carnitine, releasing the fatty acid for beta-oxidation and energy production.
Without this shuttle, fat oxidation stalls. The fatty acids accumulate. Energy production shifts toward glucose and amino acid metabolism instead, which has significant implications for both energy levels and muscle preservation.
Beyond simple fat transport, levocarnitine influences several metabolic pathways. It modulates the ratio of acetyl-CoA to CoA in the mitochondria, which affects pyruvate dehydrogenase activity and glucose oxidation. It supports the removal of short and medium-chain fatty acid metabolites that would otherwise accumulate and become toxic. And it plays a role in buffering the mitochondrial acyl-CoA pool, helping maintain metabolic flexibility, the ability to switch between fat and carbohydrate as fuel sources depending on demand.
Research has demonstrated that levocarnitine supplementation improves insulin sensitivity. A study published in the Journal of Physiology found that carnitine supplementation elevated pyruvate dehydrogenase (PDH) flux and enhanced tissue metabolic flexibility. For someone on tirzepatide, which already improves glycemic control through GIP and GLP-1 receptor activation, this additional metabolic support from levocarnitine creates a more complete picture of metabolic optimization.
The kidneys play a critical role in carnitine homeostasis. They reabsorb approximately 95% of filtered carnitine under normal conditions. This efficient reabsorption, combined with endogenous synthesis, typically prevents deficiency in healthy individuals. However, several factors can compromise carnitine status: caloric restriction (which reduces dietary intake), certain medications, chronic kidney disease, and strict vegetarian or vegan diets. When someone on tirzepatide is eating significantly less food, dietary carnitine intake naturally drops alongside overall caloric intake, making supplementation potentially more relevant.
Why compounding pharmacies combine tirzepatide with levocarnitine
The rationale is straightforward, even if the clinical proof is still developing.
Tirzepatide works primarily by reducing how much a person eats. It activates both GIP and GLP-1 receptors, which decreases appetite, slows gastric emptying, and improves the hormonal environment around glucose metabolism. Clinical trials demonstrated weight loss of 15% to 22.5% of body weight over 72 weeks. That is remarkable. But it raises a question that the trials themselves highlighted: of the weight lost, 26% to 40% came from lean tissue, not fat.
That ratio concerns researchers and clinicians alike.
Levocarnitine addresses this concern from two angles. First, by enhancing fatty acid transport into the mitochondria, it theoretically ensures that a greater proportion of the energy deficit is filled by fat oxidation rather than muscle protein breakdown. Second, by supporting overall energy production from fat stores, it may help reduce the fatigue and low energy that many people experience during rapid weight loss on GLP-1 medications.
Compounding pharmacies also have a practical motivation. With the compounded GLP-1 market becoming increasingly competitive, pharmacies differentiate their products by adding value-added ingredients. Levocarnitine, vitamin B12, niacinamide, glycine, and NAD+ are among the most common additions. Each targets a different concern: B12 for energy and nerve health, niacinamide for skin and metabolic support, glycine for GI tolerability, and levocarnitine for fat metabolism optimization.
Among these additions, levocarnitine has perhaps the most mechanistically coherent rationale for inclusion. The connection between caloric restriction, increased fat mobilization demand, and the need for efficient mitochondrial fatty acid transport is well-established in biochemistry. Whether that translates to clinically meaningful improvements in body composition outcomes when added to tirzepatide specifically is what remains unproven.
Some compounding pharmacies have recently moved away from the levocarnitine combination, preferring tirzepatide with glycine and B12 instead. This shift appears driven more by market demand and GI tolerability concerns than by any negative research findings about levocarnitine itself.
Tirzepatide levocarnitine dosing protocols
Dosing for the tirzepatide levocarnitine combination follows the standard tirzepatide titration schedule, with the levocarnitine component delivered proportionally based on the formulation concentration.
Standard titration schedule
The starting dose is 2.5 mg of tirzepatide per week, consistent with the approved tirzepatide dose chart. This initial dose allows the body to adjust to GLP-1 receptor activation and minimizes the risk of severe gastrointestinal side effects. After four weeks at 2.5 mg, the dose increases to 5 mg per week. Subsequent increases of 2.5 mg occur every four weeks as tolerated, up to a maximum of 15 mg per week.
With a standard 10 mg/mL tirzepatide and 100 mg/mL levocarnitine formulation, each dose delivers levocarnitine proportionally. At the 2.5 mg tirzepatide starting dose (0.25 mL), the injection delivers 25 mg of levocarnitine. At 5 mg tirzepatide (0.5 mL), it delivers 50 mg. At the maximum 15 mg dose (1.5 mL), the injection delivers 150 mg of levocarnitine.
These levocarnitine doses are modest compared to what oral supplementation studies have tested. Most clinical research on levocarnitine for weight management used doses of 1,000 to 3,000 mg daily. The injectable route does offer better bioavailability since it bypasses first-pass metabolism in the gut and liver, but the total weekly dose from a compounded tirzepatide injection remains well below what studies have used to demonstrate significant metabolic effects.
Understanding the unit conversion
Compounded tirzepatide is typically measured using insulin syringes marked in units. This creates a common source of confusion that requires careful attention. For a 10 mg/mL concentration, 2.5 mg equals 25 units. At 5 mg, that is 50 units. At 7.5 mg, it is 75 units. And at higher doses, the math scales linearly.
The compounded tirzepatide dosage calculator on SeekPeptides can help eliminate confusion around these conversions. Getting the dose right is critical because both under-dosing (reduced efficacy) and over-dosing (increased side effects) create problems that compound over time.
If your pharmacy provides a 17 mg/mL concentration, the unit math changes entirely. Always verify the specific concentration on your vial before drawing any dose. A 2.5 mg dose from a 17 mg/mL vial requires only approximately 14.7 units, not 25. This is one of the most common and most dangerous mistakes in compounded peptide protocols.
Injection timing and frequency
Tirzepatide levocarnitine is administered once weekly, on the same day each week. The specific day does not matter, but consistency does. Some researchers prefer injecting in the morning to align the metabolic effects with daytime activity. Others prefer evening administration to let the appetite-suppressing effects carry through the next day. The timing question that applies to GLP-1 medications broadly applies here as well, and the answer is largely personal preference.
Subcutaneous injection sites include the abdomen (at least 2 inches from the navel), the front of the thighs, and the back of the upper arms. Rotate injection sites with each dose to prevent lipodystrophy and ensure consistent absorption. For detailed guidance on where to inject GLP-1 medications and proper injection technique, those dedicated guides provide step-by-step protocols.
Fat metabolism and the mitochondrial connection
To understand why levocarnitine matters during tirzepatide therapy, you need to understand what happens inside the cell when the body enters a caloric deficit.
When calorie intake drops, as it reliably does on tirzepatide, the body mobilizes stored triglycerides from adipose tissue. Hormone-sensitive lipase breaks these triglycerides into free fatty acids and glycerol, releasing them into the bloodstream. So far, so good. The fat is being mobilized. But mobilization is not the same as oxidation.
Those free fatty acids need to reach the mitochondria, enter the matrix, and undergo beta-oxidation to produce ATP. The rate-limiting step in this process is the carnitine shuttle, specifically the CPT-I enzyme that attaches carnitine to long-chain fatty acids so they can cross the inner mitochondrial membrane.
When carnitine availability is adequate, this process runs efficiently. Fat gets burned. Energy production stays high. The body preferentially uses adipose stores rather than breaking down muscle protein for fuel.
When carnitine availability is low, the bottleneck tightens. Fatty acids accumulate in the cytoplasm. The body compensates by increasing glucose oxidation and, more problematically, by increasing amino acid catabolism, which means breaking down muscle protein for energy. This is precisely the mechanism that contributes to the lean mass losses observed in GLP-1 clinical trials.
A systematic review and meta-analysis of 37 randomized controlled trials found that levocarnitine supplementation produced modest but statistically significant reductions in body weight, BMI, and fat mass, particularly among adults with overweight or obesity. The dose-response analysis suggested that doses exceeding 1,000 mg per day were most effective. Importantly for the tirzepatide context, the effects on fat mass were more pronounced than the effects on total body weight, suggesting that levocarnitine may indeed shift the composition of weight loss toward greater fat loss.
A fascinating finding from mouse studies adds another layer. Researchers discovered that mice lacking a mitochondrial protein controlling carnitine production were cold-intolerant and resistant to weight loss from GLP-1 drugs. This suggests that carnitine biosynthesis may play a functional role in how GLP-1 receptor agonists produce their weight loss effects, not just in how the body handles the resulting caloric deficit.
For researchers tracking their tirzepatide before and after results, understanding this mitochondrial connection helps explain why some people lose weight more efficiently than others on the same dose. Individual variations in carnitine status, mitochondrial density, and metabolic flexibility all influence outcomes.
Muscle preservation during GLP-1 weight loss
This is where the tirzepatide levocarnitine combination makes its strongest theoretical case.
The lean mass problem with GLP-1 receptor agonists is real and well-documented. In the SURMOUNT clinical trials for tirzepatide, lean soft tissue loss comprised 26% to 40% of total weight lost. A study published in Circulation noted that while these reductions in muscle volume appear proportionate to the weight loss achieved, the concern remains that excessive lean mass loss could lead to sarcopenia, reduced metabolic rate, and poorer long-term outcomes.
Preserving muscle matters. A lot.
Muscle is metabolically active tissue. Each pound of muscle burns approximately 6 to 7 calories per day at rest, compared to roughly 2 calories per pound of fat. Losing significant muscle mass during weight loss reduces basal metabolic rate, making it harder to maintain weight loss long-term and easier to regain weight once medication is discontinued. For people who experience withdrawal effects or weight regain after stopping GLP-1 therapy, the amount of muscle preserved during treatment directly influences their trajectory.
Levocarnitine may help through several mechanisms. By enhancing fatty acid oxidation, it reduces the need for the body to catabolize amino acids for energy. When fat burning runs efficiently, muscle protein is spared. Additionally, some research suggests that levocarnitine supports exercise tolerance and muscle recovery. For someone on tirzepatide who is adjusting their diet and increasing physical activity, these effects could meaningfully improve body composition outcomes.
The Massachusetts General Hospital published guidance emphasizing that the most effective strategy for preserving lean body mass during GLP-1 therapy involves combining a high-protein diet with consistent resistance exercise. This outperformed diet alone and high-protein diet alone. Levocarnitine supplementation could theoretically enhance this approach by ensuring that the increased fat mobilization from exercise and caloric restriction is efficiently processed in the mitochondria.
People tracking tirzepatide results who incorporate both levocarnitine and resistance training would be optimizing from multiple angles simultaneously. The protein intake preserves muscle directly. The resistance training stimulates muscle protein synthesis. And the levocarnitine ensures efficient fat oxidation so the body does not need to break down muscle for energy.
That said, no study has directly tested this three-way combination. The logic is strong. The individual components are supported. But the specific synergy remains theoretical. Researchers should understand that distinction clearly when making protocol decisions.
Liver health and metabolic benefits
One of the more compelling research areas for levocarnitine involves its effects on liver health, particularly in the context of non-alcoholic fatty liver disease (NAFLD), a condition that affects a substantial portion of people with obesity and metabolic syndrome.
A systematic review and meta-analysis published in the World Journal of Hepatology examined the effects of levocarnitine supplementation in NAFLD populations. The pooled results from five randomized trials found significant reductions in liver fat content, serum ALT levels (a marker of liver cell damage), AST levels, and triglycerides. Levocarnitine supplementation also reduced HOMA-IR, a measure of insulin resistance.
The mechanism involves multiple pathways. Levocarnitine promotes mitochondrial lipid oxidation in liver cells, reducing the accumulation of fat within hepatocytes. It upregulates PPAR-gamma, which has anti-inflammatory effects in the liver. And by improving the ratio of fatty acid oxidation to fatty acid synthesis, it helps restore metabolic balance in an organ that becomes overwhelmed by excess lipid storage during obesity.
Tirzepatide itself demonstrates significant benefits for liver health. Clinical data show reductions in liver fat and improvements in metabolic markers. When combined with levocarnitine, the theoretical benefit is an additive or even synergistic effect on hepatic fat reduction: tirzepatide reduces the caloric load and improves systemic insulin sensitivity, while levocarnitine enhances the liver ability to oxidize existing fat stores and reduce inflammation.
For researchers concerned about fatigue during GLP-1 therapy, liver function is worth considering. An overloaded, fatty liver processes nutrients less efficiently, contributes to systemic inflammation, and can worsen the energy dips that many people report during the initial weeks of treatment. Supporting liver health through levocarnitine supplementation may address some of these symptoms at their metabolic root rather than merely managing them symptomatically.
The insulin sensitivity improvements from levocarnitine also complement tirzepatide mechanisms. Tirzepatide improves glycemic control primarily through GIP and GLP-1 receptor activation, which enhances insulin secretion and reduces glucagon levels. Levocarnitine improves insulin sensitivity at the cellular level by modulating acetyl-CoA/CoA ratios and supporting mitochondrial function. These are distinct but complementary pathways that together create a more comprehensive metabolic profile than either compound alone.
The TMAO concern and cardiovascular considerations
No honest discussion of levocarnitine supplementation can skip this topic. It deserves careful attention.
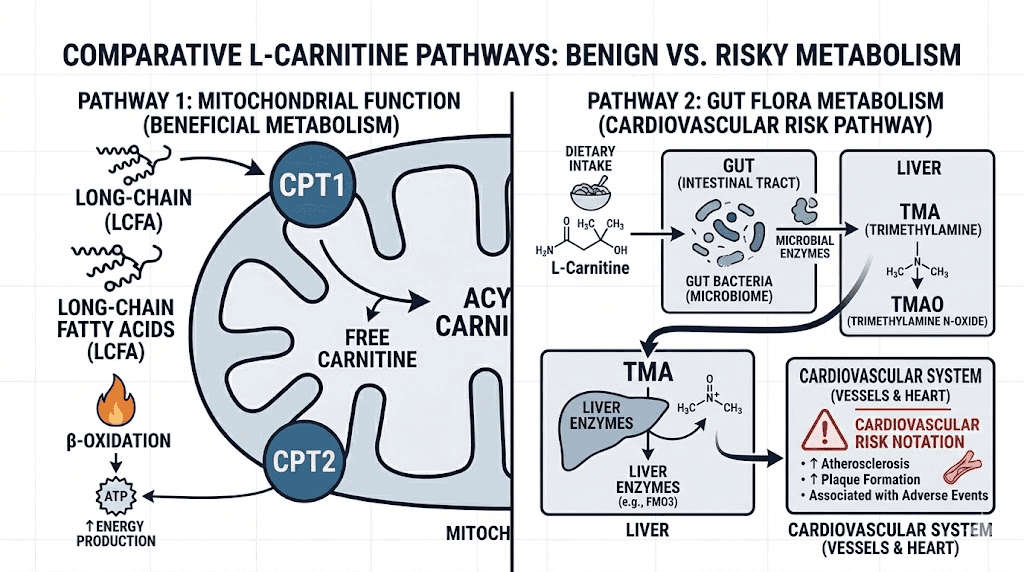
Trimethylamine N-oxide (TMAO) is a metabolite produced when gut bacteria metabolize dietary compounds including levocarnitine and choline. The process works like this: intestinal microbiota convert levocarnitine to trimethylamine (TMA), which then travels to the liver where the enzyme FMO3 oxidizes it to TMAO. Elevated TMAO levels have been associated with increased risks of atherosclerosis, cardiovascular disease, and higher all-cause mortality in multiple epidemiological studies.
This association is what gives some researchers pause about levocarnitine supplementation.
However, the relationship is more nuanced than headlines suggest. A study in Nature Medicine found that plasma levocarnitine levels predicted increased cardiovascular risk only among subjects with concurrently high TMAO levels. In other words, levocarnitine itself is not the problem. The conversion to TMAO, which depends entirely on gut microbiome composition, is what creates the potential risk.
Several factors influence how much TMAO a person produces from levocarnitine supplementation. Omnivores with gut microbiomes adapted to regular meat consumption tend to produce more TMA from carnitine than vegetarians or vegans. The specific bacterial species present in the gut, particularly those in the genera Clostridium, Escherichia, and Proteus, determine conversion rates. Antibiotic use, probiotic supplementation, and dietary patterns all influence gut microbiome composition and therefore TMAO production.
The injectable route of administration used in compounded tirzepatide levocarnitine may partially mitigate this concern. When levocarnitine is injected subcutaneously, it enters systemic circulation directly, bypassing the gastrointestinal tract where bacterial conversion to TMA occurs. This does not eliminate TMAO production entirely, as some circulating levocarnitine can be excreted into the gut, but it likely reduces the total TMAO burden compared to equivalent oral doses.
For researchers concerned about cardiovascular risk, practical strategies to minimize TMAO production include supporting a healthy gut microbiome through prebiotic and probiotic use, monitoring TMAO levels if testing is available, and keeping total levocarnitine exposure within studied safety ranges. The doses delivered through compounded tirzepatide formulations (25 to 150 mg per week) are substantially lower than the doses associated with significant TMAO elevation in studies (typically 1,000 to 3,000 mg daily).
This does not mean the concern is irrelevant. It means the risk profile differs depending on the route, dose, and individual factors. Researchers with existing cardiovascular disease, elevated baseline TMAO levels, or significant cardiovascular risk factors should discuss this specific concern with their healthcare provider before starting a levocarnitine-containing compound.
Side effects and tolerability of the combination
Side effects from the tirzepatide levocarnitine combination come from both components, and the overlap between them is the primary tolerability concern.
Tirzepatide side effects
The most common adverse effects of tirzepatide are gastrointestinal: nausea, vomiting, diarrhea, constipation, abdominal pain, and decreased appetite. These effects are dose-dependent and typically most pronounced during the initial weeks and after each dose escalation. Most people find that symptoms improve as the body adapts, particularly when following a slow and steady titration schedule.
Other reported effects include headaches, fatigue, injection site reactions, hair thinning, anxiety, and muscle pain. More serious but rare adverse events include pancreatitis, gallbladder problems, and changes in kidney function.
Levocarnitine side effects
At oral doses of approximately 3,000 mg per day, levocarnitine can cause nausea, vomiting, abdominal cramps, and diarrhea. Lower doses are generally well-tolerated. The injectable route typically produces fewer gastrointestinal effects since the compound bypasses the digestive system.
A distinctive side effect of levocarnitine supplementation is a fishy body odor, caused by the excretion of trimethylamine through sweat and urine. This is more commonly associated with oral supplementation at higher doses but can theoretically occur with any route of administration.
The overlap problem
Here is where the combination gets complicated. Both tirzepatide and levocarnitine can cause nausea, diarrhea, and abdominal discomfort. When combined, these overlapping side effect profiles may worsen GI tolerability, particularly during the dose escalation phase when tirzepatide GI effects are already at their peak.
This is one reason some compounding pharmacies have shifted away from the levocarnitine combination in favor of glycine-based formulations, which may actually reduce GI side effects rather than potentially adding to them. Glycine has a calming effect on the digestive system and may improve tirzepatide tolerability, while levocarnitine may worsen it in sensitive individuals.
For researchers experiencing significant constipation or nausea on tirzepatide alone, adding levocarnitine may not be the best choice. Those who tolerate tirzepatide well with minimal GI issues are better candidates for the levocarnitine combination.
Managing side effects requires attention to diet and lifestyle. Following a structured meal plan that emphasizes easily digestible foods, staying hydrated, and eating appropriate foods can help minimize GI symptoms. If side effects remain problematic, specific supplements may help manage individual symptoms.
Who may benefit most from tirzepatide levocarnitine
Not everyone on tirzepatide needs levocarnitine. And for some, it may not be the best compound addition. Understanding who benefits most helps researchers make informed decisions rather than following trends.
Strong candidates
People who are physically active and want to maximize fat loss while preserving lean mass represent the strongest case for this combination. When someone exercises regularly while on tirzepatide, the demand for fatty acid oxidation increases substantially. Levocarnitine ensures the mitochondria can meet that increased demand efficiently.
Those who eat a primarily plant-based diet while on tirzepatide may benefit as well. Vegetarians and vegans have lower dietary carnitine intake and reduced carnitine stores. When combined with the reduced food intake from tirzepatide, carnitine levels could drop below optimal thresholds for efficient fat metabolism.
Researchers with metabolic syndrome, elevated liver enzymes, or suspected NAFLD have a metabolic profile that aligns well with the dual benefits of tirzepatide (glycemic control, appetite reduction) and levocarnitine (enhanced hepatic fat oxidation, improved insulin sensitivity at the cellular level).
People experiencing significant fatigue on GLP-1 medications who have already addressed basic factors like hydration, protein intake, and electrolytes may find that levocarnitine helps by improving mitochondrial energy production from fat stores.
Weaker candidates
People with significant GI sensitivity to tirzepatide who are already struggling with nausea, diarrhea, or abdominal pain should probably avoid adding another compound that can worsen those symptoms. A glycine-based compound would be a better fit.
Those with existing cardiovascular disease or elevated TMAO levels should discuss the theoretical TMAO concern with their provider before starting levocarnitine supplementation.
Researchers who are sedentary and not planning to incorporate exercise may see limited benefit from levocarnitine. The enhanced fat oxidation capacity matters most when there is a metabolic demand for it, and physical activity creates that demand much more effectively than caloric restriction alone.
If someone is already taking oral levocarnitine or L-carnitine supplements alongside their tirzepatide, adding injectable levocarnitine through the compound may create redundancy and unnecessarily increase total carnitine exposure.
Storage, handling, and administration
Compounded tirzepatide levocarnitine requires proper storage to maintain potency and safety. Like all compounded peptide formulations, temperature control is critical.
Refrigeration requirements
Tirzepatide requires refrigeration at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). The levocarnitine component does not change this requirement. Store the vial upright in the main body of the refrigerator, not in the door where temperature fluctuations are greater and not in the freezer. The same storage principles that apply to standard compounded tirzepatide shelf life apply to the levocarnitine combination.
If the vial needs to be out of the refrigerator temporarily for administration, brief periods at room temperature are acceptable. However, prolonged exposure to temperatures above 77 degrees Fahrenheit (25 degrees Celsius) degrades both the tirzepatide and levocarnitine components. Never leave the vial in direct sunlight, in a hot car, or near a heat source.
Beyond-use dating for compounded formulations varies by pharmacy but typically ranges from 30 to 90 days when properly refrigerated. Check your specific vial label for the expiration date assigned by your compounding pharmacy. Using expired or improperly stored medication risks both reduced efficacy and potential safety concerns from degradation products. Our guide on tirzepatide expiration covers these considerations in detail.
Administration technique
Draw the prescribed number of units using a sterile insulin syringe. Remove any air bubbles by tapping the syringe gently and pushing them out before injecting. Clean the injection site with an alcohol swab and allow it to dry completely before administration.
Insert the needle at a 45 to 90 degree angle depending on the amount of subcutaneous tissue at the injection site. Inject slowly and steadily. After injecting the full dose, hold the needle in place for 5 to 10 seconds before withdrawing to ensure complete delivery. Apply gentle pressure with a clean cotton ball or gauze. Do not rub the injection site.
For detailed reconstitution and preparation guidance specific to compounded tirzepatide formulations, the tirzepatide reconstitution guide provides comprehensive step-by-step instructions. If your formulation arrives as a lyophilized powder requiring reconstitution, that guide covers the exact process including how much bacteriostatic water to use.
Needle and syringe selection
Most researchers use 29 to 31 gauge insulin syringes with 0.5 inch needles for subcutaneous tirzepatide injection. Thinner needles (31 gauge) cause less discomfort but may require slower injection. Thicker needles (29 gauge) allow faster injection but may cause slightly more sensation at the injection site. The choice is personal preference, and consistency matters more than the specific gauge selected.
Use a new, sterile syringe for every injection. Never reuse needles or syringes, as this introduces contamination risk and dulled needles cause more tissue damage and injection site irritation.
Comparing tirzepatide levocarnitine to other compound blends
Compounding pharmacies offer tirzepatide with various additions beyond levocarnitine. Understanding how these options compare helps researchers select the formulation that best matches their specific goals and tolerability profile.
Tirzepatide with B12
Tirzepatide with vitamin B12 (typically as cyanocobalamin or methylcobalamin) is the most widely available compound combination. B12 supports energy metabolism, nerve function, and red blood cell production. It does not directly enhance fat oxidation the way levocarnitine does, but it addresses the B12 deficiency risk that increases with reduced food intake on GLP-1 therapy. For researchers primarily concerned about energy levels and not specifically about fat metabolism optimization, B12 may be the simpler and better-tolerated choice.
Tirzepatide with glycine
Tirzepatide with glycine targets GI tolerability. Glycine is an amino acid with calming effects on the digestive system, and some evidence suggests it reduces the nausea and GI distress associated with GLP-1 receptor agonists. For researchers who struggle with GI side effects, glycine offers a direct benefit that levocarnitine does not.
Tirzepatide with glycine and B12
The triple compound of tirzepatide, glycine, and B12 has become increasingly popular as a balanced formulation. It addresses energy (B12), tolerability (glycine), and basic nutritional support without the TMAO concerns or GI overlap risks of levocarnitine. This has emerged as the preferred option at many clinics.
Tirzepatide with niacinamide
Tirzepatide with niacinamide targets metabolic health and skin support. Niacinamide (vitamin B3) supports NAD+ production, which is involved in cellular energy metabolism and DNA repair. It addresses a different concern than levocarnitine and may be preferred by researchers focused on longevity and skin health alongside weight management.
Head-to-head comparison
Compound blend | Primary benefit | GI tolerability | Fat oxidation | Best for |
|---|---|---|---|---|
Tirzepatide + Levocarnitine | Enhanced fat burning | May worsen | Strong | Active researchers, muscle preservation |
Tirzepatide + B12 | Energy, nerve support | Neutral | None | Fatigue, B12 deficiency risk |
Tirzepatide + Glycine | GI comfort | Improves | None | GI-sensitive individuals |
Tirzepatide + Glycine + B12 | Balanced support | Improves | None | Most people (general-purpose) |
Tirzepatide + Niacinamide | Metabolic + skin | Neutral | Indirect | Metabolic health focus |
The choice between these formulations should match individual priorities. Someone who is physically active, tolerates tirzepatide well, and wants to maximize fat loss composition would lean toward levocarnitine. Someone who struggles with nausea would lean toward glycine. And someone who just wants solid all-around support would choose the glycine and B12 combination. SeekPeptides provides detailed guides on each of these compound blends to help researchers navigate the decision.
What the research actually says (and does not say)
Intellectual honesty requires separating what is proven from what is plausible. This section does exactly that.
What is proven
Tirzepatide produces significant weight loss (15% to 22.5% of body weight in clinical trials). This is established fact, supported by the SURMOUNT trial series with thousands of participants. The timeline and magnitude of results are well-characterized.
Levocarnitine is essential for mitochondrial fatty acid transport. This biochemistry is settled science, confirmed across decades of research.
Levocarnitine supplementation at doses above 1,000 mg daily produces modest improvements in body weight, BMI, and fat mass in overweight and obese adults. A meta-analysis of 37 randomized controlled trials established this with reasonable confidence.
Levocarnitine supplementation improves liver function markers and reduces liver fat in NAFLD populations. Multiple systematic reviews and meta-analyses support this finding.
Levocarnitine supplementation improves insulin sensitivity as measured by HOMA-IR. This is supported by clinical data across multiple study populations.
What is plausible but unproven
Adding levocarnitine to tirzepatide produces better fat loss outcomes than tirzepatide alone. No clinical trial has tested this specific question. The mechanism is sound. The individual components work. But the additive benefit of combining them remains theoretical.
Levocarnitine preserves lean mass during GLP-1-mediated weight loss. The mechanism supporting this is logical (enhanced fat oxidation reduces reliance on amino acid catabolism), but no direct evidence exists from studies combining these specific interventions.
Injectable levocarnitine produces less TMAO than oral levocarnitine. The pharmacokinetic reasoning supports this (bypassing gut bacteria), but head-to-head comparisons of TMAO levels between routes have not been conducted in the context of compounded tirzepatide.
What is not supported
There is no established medical guideline recommending the combination of tirzepatide with levocarnitine. No major medical society or regulatory body has endorsed this practice. The FDA has not evaluated the specific combination for safety or efficacy.
Claims that levocarnitine dramatically accelerates tirzepatide weight loss are not supported by any published data. The effects of levocarnitine on body composition are modest, and it would be misleading to suggest otherwise.
For researchers who value evidence-based decision-making, this honest assessment should inform expectations. The combination may help. It probably does not hurt (at the doses used in compounded formulations). But anyone marketing it as a proven enhancement to tirzepatide efficacy is getting ahead of the science.
Cost considerations and access
Compounded tirzepatide with levocarnitine typically costs more than standard compounded tirzepatide due to the additional ingredient and the slightly more complex formulation process. However, the price differential is usually modest, often ranging from $10 to $30 per vial depending on the pharmacy.
When evaluating affordable tirzepatide options, the compound blend should be assessed on a cost-per-benefit basis. If levocarnitine supplementation is genuinely desired, getting it combined in the injection is typically more convenient and often less expensive than purchasing separate oral L-carnitine supplements alongside standard compounded tirzepatide.
Access varies by region and pharmacy. Not all compounding pharmacies offer the levocarnitine combination. Some that previously offered it have discontinued the formulation in favor of glycine or B12 blends. Pharmacies like Empower Pharmacy and others in the compounding space offer various tirzepatide formulations, though specific availability changes frequently with market conditions and regulatory developments.
The peptide cost calculator on SeekPeptides can help researchers compare the total cost of different compound formulations and assess value across providers. Given that compounded peptide pricing can vary significantly between pharmacies, comparison shopping is essential.
Practical protocol for tirzepatide levocarnitine
For researchers who decide to proceed with the tirzepatide levocarnitine compound, here is a structured approach based on available evidence and best practices.
Protocol 1: Standard weight management
Goal: Optimize fat loss while minimizing lean mass loss during tirzepatide therapy.
Formulation: Tirzepatide 10 mg/mL + Levocarnitine 100 mg/mL
Schedule:
Weeks 1 to 4: 2.5 mg tirzepatide (25 units) once weekly, delivering 25 mg levocarnitine
Weeks 5 to 8: 5 mg tirzepatide (50 units) once weekly, delivering 50 mg levocarnitine
Weeks 9 to 12: 7.5 mg tirzepatide (75 units) once weekly, delivering 75 mg levocarnitine
Weeks 13+: 10 mg tirzepatide (100 units) once weekly, delivering 100 mg levocarnitine, with potential titration to 12.5 mg or 15 mg based on response and tolerability
Supporting measures:
Protein intake: 0.7 to 1.0 g per pound of body weight daily
Resistance training: 3 to 4 sessions per week minimum
Hydration: minimum 64 oz water daily
Monitor body composition (not just scale weight) monthly
Protocol 2: Enhanced fat oxidation for active researchers
Goal: Maximize fatty acid oxidation capacity during concurrent exercise programs.
Formulation: Tirzepatide 10 mg/mL + Levocarnitine 100 mg/mL
Schedule: Same titration as Protocol 1, with an additional oral L-carnitine supplement (500 to 1,000 mg, taken 60 to 90 minutes before exercise sessions) to boost total carnitine availability on training days. Keep total daily levocarnitine intake below 2,000 mg from all sources.
Exercise timing: Schedule the weekly injection on a rest day or light activity day. Heavy training sessions are best performed 2 to 3 days after the injection when GI side effects have typically subsided.
What to monitor
Track the following metrics to assess whether the levocarnitine addition is providing value:
Body composition changes (use DEXA, InBody, or similar if accessible)
Energy levels and exercise performance
GI symptoms (compare to baseline on tirzepatide alone if possible)
Liver function markers (ALT, AST) if baseline values were elevated
General well-being and recovery quality
If after 8 to 12 weeks the levocarnitine addition has not produced noticeable improvements in energy, exercise performance, or body composition compared to tirzepatide alone, consider adjusting your protocol or switching to a different compound blend. Not every addition works for every person, and forcing a protocol that is not producing results wastes time and money.
Frequently asked questions
Is tirzepatide levocarnitine FDA approved?
No. The combination of tirzepatide with levocarnitine is a compounded formulation prepared by pharmacies under a prescription from a licensed healthcare provider. While tirzepatide is FDA-approved individually (as Mounjaro and Zepbound) and levocarnitine is FDA-approved for carnitine deficiency (as Carnitor), the specific combination has not been evaluated by the FDA for safety or efficacy. All compounded tirzepatide formulations fall outside FDA approval.
Does levocarnitine actually improve tirzepatide weight loss results?
No clinical trial has directly tested whether adding levocarnitine to tirzepatide produces greater weight loss than tirzepatide alone. The rationale is based on the known biochemistry of fatty acid oxidation and the established (though modest) effects of levocarnitine supplementation on body composition in separate studies. Researchers should view the combination as mechanistically plausible rather than clinically proven.
Can I take oral L-carnitine instead of the compounded injection?
Yes. Oral L-carnitine supplements are widely available and have been used in most clinical studies. The typical effective dose is 1,000 to 2,000 mg daily. Oral supplementation has lower bioavailability than injectable forms but may produce more TMAO due to gut bacteria exposure. The injectable form in compounded tirzepatide offers convenience (one injection instead of daily pills) and potentially reduced TMAO production, but the trade-off is a lower total weekly dose of levocarnitine.
What is the difference between levocarnitine and L-carnitine?
They are the same compound. Levocarnitine is the pharmaceutical name for L-carnitine, which refers to the biologically active (levo-rotatory) isomer. The prefix "levo" indicates the specific stereochemistry. D-carnitine, the mirror image, is biologically inactive and potentially harmful, as it can competitively deplete L-carnitine stores. Compounding pharmacies use pharmaceutical-grade levocarnitine to ensure purity and correct stereochemistry.
Will levocarnitine cause a fishy body odor?
It can, particularly at higher doses. The odor comes from trimethylamine (TMA) excretion through sweat and urine. This is more common with oral supplementation at doses above 2,000 mg daily. The lower doses delivered through compounded tirzepatide injections (25 to 150 mg weekly) are unlikely to cause noticeable odor in most people, though individual sensitivity varies.
Should I stop taking levocarnitine if I experience more nausea on the compound?
If GI symptoms worsen significantly after starting the levocarnitine compound compared to standard tirzepatide, discussing a formulation change with your provider is reasonable. Glycine-based compounds may actually improve GI tolerability rather than worsening it. However, first rule out other causes of increased nausea, such as recent dose escalation, dietary changes, or dehydration, before attributing the change to levocarnitine.
How does tirzepatide levocarnitine compare to semaglutide with L-carnitine?
The levocarnitine component works identically regardless of which GLP-1 agonist it is paired with. The difference lies in the base peptide. Tirzepatide activates both GIP and GLP-1 receptors, producing greater average weight loss in head-to-head comparisons with semaglutide. The choice between them should be based on the GLP-1 agonist comparison, not the levocarnitine addition. Use the tirzepatide vs semaglutide dosage chart for detailed comparison.
Can I use tirzepatide levocarnitine while breastfeeding?
Tirzepatide is not recommended during pregnancy or breastfeeding due to insufficient safety data. The levocarnitine component does not change this recommendation. For detailed guidance on GLP-1 medications and breastfeeding, consult your healthcare provider and review the available safety information.
Can I drink alcohol while on tirzepatide levocarnitine?
Alcohol can worsen the GI side effects of tirzepatide (nausea, vomiting, abdominal pain) and may interfere with metabolic processes that levocarnitine is intended to support. The levocarnitine component does not create any unique interaction with alcohol, but the general guidance for alcohol and tirzepatide applies: moderation at most, and awareness that tolerance may change significantly while on GLP-1 therapy.
What if tirzepatide levocarnitine is not working for me?
If you are not seeing expected results after 8 to 12 weeks, evaluate the following: Are you tracking body composition (not just scale weight)? Is your protein intake adequate? Are you incorporating resistance exercise? Is the tirzepatide dose at a therapeutic level (most people need 7.5 mg or higher for significant weight loss)? Our guide on what to do when tirzepatide stops working and troubleshooting stalled weight loss covers these scenarios in detail.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your fat oxidation stay efficient, your lean mass stay preserved, and your protocols stay evidence-based.