Mar 10, 2026

Conflicting information about compounded semaglutide storage is everywhere. One source says 24 hours outside the refrigerator. Another claims 56 days. A third suggests 60 days is perfectly fine. You left your vial on the counter overnight, and now you do not know if it is safe to use or if you just wasted hundreds of dollars worth of medication.
The confusion is understandable.
Brand-name formulations like Ozempic and Wegovy come with clear manufacturer guidelines backed by extensive stability testing. Compounded semaglutide does not. Every compounding pharmacy uses a different formulation, different stabilizers, different concentrations, and different preservation methods. The storage rules change depending on who made your medication, what they put in it, and how they prepared it. That makes a simple question surprisingly complicated, and getting the answer wrong means either wasting perfectly good medication or injecting something that has lost its therapeutic potency. Neither outcome is acceptable when you are investing significant time, money, and effort into your weight loss protocol. Researchers working with SeekPeptides understand that proper storage is not just about following rules. It is about protecting your investment and ensuring every dose delivers the results you expect.
This guide breaks down exactly how long compounded semaglutide can remain at room temperature, what factors affect that timeframe, how to tell if your medication is still effective, and what to do if you accidentally left it out. Every recommendation here comes from pharmaceutical stability data, compounding pharmacy guidelines, and published research on compounded semaglutide degradation. No guesswork. No conflicting advice. Just the specific numbers you need to make the right decision about your medication.
The short answer: how long compounded semaglutide can stay out
Before diving into the details, here is what you need to know right now. If your compounded semaglutide has been sitting on the counter and you are anxious about whether it is still good, this section gives you the quick reference points.
Most compounded semaglutide formulations can safely remain at room temperature, defined as at or below 77 degrees Fahrenheit (25 degrees Celsius), for a limited period. The exact timeframe depends on your specific formulation, but general guidelines from compounding pharmacies and stability studies suggest a range of 14 to 60 days at controlled room temperature. Some pharmacies are more conservative, recommending their formulations stay out no longer than 14 days. Others have conducted stability testing showing their specific product remains viable for up to 56 or even 60 days below 77 degrees Fahrenheit.
That is a massive range. And it matters.
The critical variable is what your compounding pharmacy put in the vial. Different excipients, different pH buffers, different preservatives, and different concentrations all affect how quickly the active peptide degrades outside refrigeration. This is fundamentally different from brand-name GLP-1 medications that have standardized formulations with published stability data. If you are working with a compounded product and need to understand proper dosing protocols, storage knowledge becomes even more critical because incorrect storage can alter the effective dose you receive.
The safest approach is always to check the documentation that came with your specific medication or call your compounding pharmacy directly. They conducted beyond-use date testing for their exact formulation and can tell you precisely how long their product remains stable at room temperature.
How compounded semaglutide differs from brand-name versions
Understanding why storage guidelines vary so much requires understanding what makes compounded semaglutide fundamentally different from brand-name products. This is not just a labeling difference. The actual physical and chemical properties of the medication change depending on how it was made.
Brand-name semaglutide storage rules
Ozempic, the diabetes formulation of semaglutide, has been through rigorous stability testing by Novo Nordisk. The manufacturer has tested the exact formulation at multiple temperatures over extended periods and published clear results. Before first use, Ozempic must stay refrigerated between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). After the first injection from a pen, Ozempic can remain at room temperature, specifically below 86 degrees Fahrenheit (30 degrees Celsius), for up to 56 days.
Wegovy, the weight management version, has slightly different rules. It can stay at room temperature for up to 28 days after first use, again below 86 degrees Fahrenheit. The difference between 56 days and 28 days comes from differences in concentration and formulation between the two products, even though both contain the same active ingredient. Individuals tracking their semaglutide timeline and results need to understand these storage differences to ensure consistent outcomes throughout their protocol.
These numbers come from extensive pharmaceutical testing. Novo Nordisk tested thousands of units under controlled conditions and published the results in regulatory filings reviewed by the FDA. The data is solid. The guidelines are clear.
Why compounded semaglutide is different
Compounded semaglutide exists in a completely different regulatory framework. Compounding pharmacies create custom formulations based on a prescriber order, and each pharmacy may use different processes, different inactive ingredients, and different quality testing standards. Some key differences include formulation variability, where one pharmacy might use a liquid solution while another provides a lyophilized (freeze-dried) powder that requires reconstitution before use.
Concentration differences also play a role. Compounded vials come in various concentrations, commonly 2mg/mL, 5mg/mL, or 10mg/mL, and higher concentrations may have different stability profiles. The pH of the solution matters significantly too. Research published in the Journal of Peptide Science found that semaglutide shows increased degradation between pH 4.5 and 5.5, near its isoelectric point of pH 5.4. Formulations buffered above pH 7.0 demonstrate better thermal stability. Different pharmacies use different buffer systems.
Then there are preservatives and stabilizers. Some pharmacies add bacteriostatic water with benzyl alcohol. Others use sterile water. Some add stabilizing agents. Each choice affects how long the final product remains potent outside refrigeration. If you are mixing your own compounded semaglutide, understanding the reconstitution process and the type of water used becomes essential for predicting storage stability.
The beyond-use date factor
Every compounding pharmacy assigns a beyond-use date (BUD) to their products. This is different from a manufacturer expiration date. The BUD represents the date after which the pharmacy cannot guarantee the product meets potency and sterility standards. For compounded semaglutide, BUDs typically range from 30 to 90 days when refrigerated, depending on the pharmacy formulation and testing data.
The BUD for room temperature storage is always shorter than the refrigerated BUD, sometimes dramatically so. A product with a 90-day refrigerated BUD might only have a 14-day room temperature allowance. Others might extend to 28 or even 60 days at room temperature. The only way to know your specific product BUD at room temperature is to ask the pharmacy that made it. If you received compounded semaglutide with refrigerator storage guidelines, those guidelines apply to refrigerated storage only, not room temperature exposure.
What happens to semaglutide at room temperature
Peptides are not small molecule drugs. They are chains of amino acids folded into specific three-dimensional structures, and that structure is what makes them biologically active. When the structure changes, the peptide stops working. Temperature is one of the primary forces that drives structural change in peptides like semaglutide.
The science of peptide degradation
Semaglutide is a 31-amino-acid peptide analog of human GLP-1 (glucagon-like peptide-1). It works by binding to GLP-1 receptors in the pancreas, gut, and brain to regulate blood sugar and reduce appetite. That binding depends on the peptide maintaining its correct three-dimensional shape. Understanding how semaglutide suppresses appetite helps illustrate why maintaining structural integrity matters so much.
At elevated temperatures, several degradation processes accelerate. Deamidation is one, where asparagine and glutamine residues lose their amide groups. Oxidation is another, where methionine residues react with oxygen. Aggregation happens when unfolded peptide chains clump together. Each of these processes reduces the amount of active, properly structured semaglutide in your vial.
The rate of these reactions follows well-established chemical kinetics. For every 10 degree Celsius increase in temperature, chemical reaction rates roughly double. This means semaglutide at 86 degrees Fahrenheit (30 degrees Celsius) degrades roughly twice as fast as semaglutide at 68 degrees Fahrenheit (20 degrees Celsius). At 95 degrees Fahrenheit (35 degrees Celsius), degradation accelerates further.
A preformulation study published in the Journal of Peptide Science examined semaglutide stability across multiple temperatures, from 5 degrees Celsius through 25, 40, 60, and 80 degrees Celsius. The results showed that while semaglutide remained remarkably stable at refrigerated temperatures, degradation increased progressively at higher temperatures. At 80 degrees Celsius, the peptide remained stable for approximately 3 hours before significant degradation occurred, demonstrating that extreme heat causes rapid breakdown of the molecular structure.
Temperature thresholds that matter
Not all temperature exposures are equal. Here is a practical breakdown of what different temperature ranges mean for your compounded semaglutide, and how they connect to real-world storage scenarios.
Below 46 degrees Fahrenheit (8 degrees Celsius) is the refrigerated zone where degradation occurs at the slowest possible rate. This is where your semaglutide should spend most of its life. Between 46 and 77 degrees Fahrenheit (8 to 25 degrees Celsius) is the controlled room temperature range. Degradation occurs faster than refrigerated but remains within acceptable limits for the timeframes specified by your pharmacy. Most indoor environments with climate control fall in this range.
Between 77 and 86 degrees Fahrenheit (25 to 30 degrees Celsius), you enter the caution zone. Degradation accelerates noticeably. Many pharmacy guidelines use 86 degrees Fahrenheit as the absolute upper limit for any room temperature storage. Above 86 degrees Fahrenheit (30 degrees Celsius) is the danger zone. Peptide degradation becomes rapid enough that potency loss can occur within hours to days rather than weeks. A car dashboard in summer, a windowsill in direct sunlight, or a non-climate-controlled garage can easily exceed this threshold.
Below 32 degrees Fahrenheit (0 degrees Celsius), freezing causes irreversible damage. Ice crystal formation physically disrupts the peptide structure and destroys the protein. Never use semaglutide that has been frozen, even if it looks normal after thawing. This same principle applies to tirzepatide storage and other GLP-1 receptor agonists that share similar peptide stability characteristics.
Why you cannot see potency loss
This is perhaps the most important thing to understand about semaglutide degradation. You cannot detect it visually.
When semaglutide degrades through deamidation or oxidation, the solution typically remains clear and colorless. There is no color change. No cloudiness. No visible particles. The liquid in your vial can look exactly the same as the day you received it while containing 50 percent less active peptide.
Visible changes like cloudiness, discoloration, or floating particles indicate severe degradation or contamination. By the time you can see something wrong, the medication is far past the point of being usable. But the absence of visible changes does not guarantee potency. This is why following time-based storage guidelines is so critical. You cannot rely on visual inspection alone to determine if your compounded semaglutide is still effective.
The practical implication is straightforward. Track how long your vial has been out of the refrigerator. Write the date and time on the vial or a sticky note when you remove it. Do not guess. Do not rely on memory. The stakes are too high, especially when you are following a carefully calibrated semaglutide dosage protocol that depends on receiving the full intended dose each injection.
Room temperature storage guidelines by scenario
Different situations call for different responses. Here is exactly what to do in each common scenario, with specific guidance based on compounding pharmacy recommendations and stability data.
Left out overnight by accident
This is the most common scenario. You took your vial out for your evening injection, got distracted, and found it on the counter the next morning. Eight to twelve hours at room temperature.
The good news: your semaglutide is almost certainly fine.
Even the most conservative compounding pharmacy guidelines allow for at least 14 days at room temperature. A single overnight exposure of 8 to 12 hours represents a fraction of that allowance. As long as your home was at normal room temperature (below 77 degrees Fahrenheit) and the vial was not sitting in direct sunlight or next to a heat source, the medication retains its potency.
What to do: inspect the vial for any visible changes. If the liquid is clear and free of particles, return it to the refrigerator immediately. Note the time it was out. Subtract those hours from your total room temperature allowance. Continue using as normal. There is no need to contact your pharmacy or discard the vial over a single overnight exposure. However, if this happens repeatedly, the cumulative time outside refrigeration adds up. Keep a running total.
People often panic in this situation, but the same principle applies to compounded tirzepatide left out of the fridge. Both peptide medications have enough stability buffer to handle occasional brief room temperature exposure without meaningful potency loss.
Out for a few hours during your injection routine
Taking your vial out of the refrigerator 15 to 30 minutes before injection is actually recommended by many pharmacists. Cold injections can be more uncomfortable and may cause more injection site reactions. Allowing the medication to reach closer to room temperature before injecting reduces discomfort and can improve absorption at the injection site.
A few hours outside the refrigerator during your injection routine is completely inconsequential for stability. Even 2 to 4 hours at room temperature represents a negligible fraction of the total allowable exposure time. The medication will not degrade in any measurable way during this brief period.
Best practice: take the vial out 15 to 20 minutes before your scheduled injection. Prepare your dose using your semaglutide dosage calculator to confirm the correct amount. Inject. Return the vial to the refrigerator immediately after. This simple routine minimizes total room temperature exposure while maximizing injection comfort.
Traveling with compounded semaglutide
Travel creates the most challenging storage situations. Flights, road trips, hotel rooms, and unfamiliar refrigerators all introduce variables that can affect your medication.
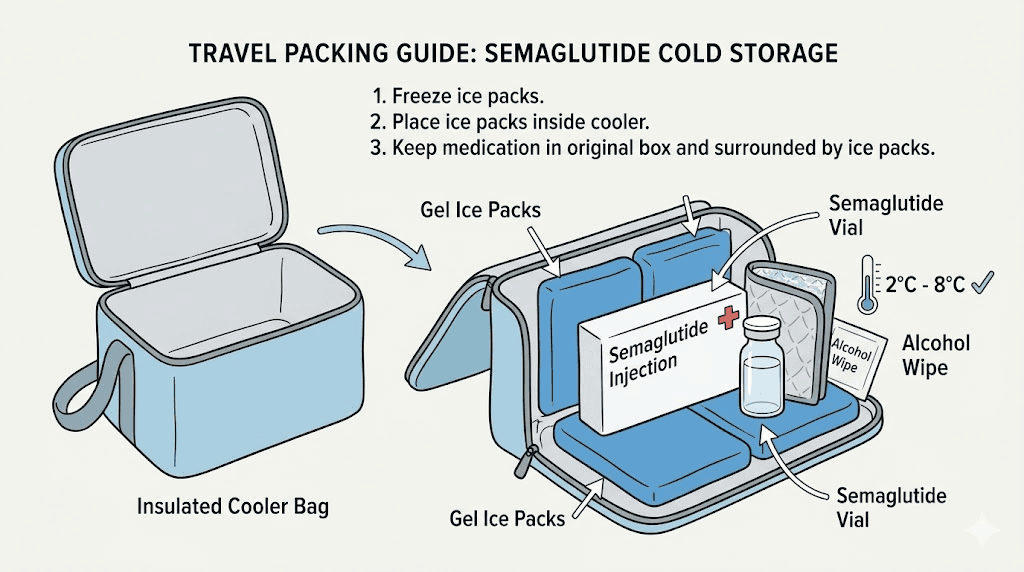
For short trips of 1 to 3 days, a medical-grade insulated cooler bag with ice packs provides adequate temperature control. These bags are specifically designed for medication transport and can maintain refrigerator temperatures for 12 to 24 hours depending on ambient conditions. Pack the ice packs around the vial but use a barrier (like a cloth or paper towel) between the ice pack and the medication to prevent accidental freezing. Direct contact with a frozen ice pack can freeze the medication, causing the irreversible damage discussed earlier.
For longer trips, plan for refrigerator access at your destination. Most hotels have mini-fridges. If yours does not, request one when booking or call ahead to confirm availability. Airport security allows injectable medications through checkpoints. Carry your medication in your carry-on luggage, never in checked bags. Cargo holds can reach temperatures well below freezing during flight, which would permanently destroy the peptide.
If you are on a road trip, keep the medication in the insulated bag inside the passenger cabin. Never leave it in the trunk or glove compartment, where temperatures can exceed 120 degrees Fahrenheit in summer conditions. The same refrigeration principles apply whether you are at home or traveling. The peptide does not care about your location. It only responds to temperature.
Power outage or refrigerator malfunction
Power outages present a unique challenge because you may not know exactly when the temperature in your refrigerator started rising. A closed refrigerator maintains safe food temperatures for approximately 4 hours. The same general timeline applies to your medication.
If the power was out for less than 4 hours and you did not open the refrigerator door, the internal temperature likely stayed within acceptable range. Your medication is fine. For outages lasting 4 to 12 hours, the refrigerator interior temperature gradually rises to room temperature. Your medication transitioned from refrigerated to room temperature storage, starting whatever room temperature clock your pharmacy specifies.
For extended outages lasting more than 12 hours, particularly in summer when ambient temperatures can be high, the situation becomes more concerning. If your home temperature exceeded 86 degrees Fahrenheit for an extended period, the medication may have been exposed to conditions that accelerate degradation beyond what normal room temperature guidelines account for.
The best approach during a power outage is to place your semaglutide vial inside a cooler with ice if you have one available. If not, keep the refrigerator closed as much as possible. After power returns, note how long the outage lasted and check the temperature inside the refrigerator with a thermometer if possible. If you are uncertain about the exposure conditions, contact your compounding pharmacy with the details. They can advise whether the medication is likely still viable based on their stability testing data for your specific formulation.
Comparing storage requirements across GLP-1 medications
Understanding how compounded semaglutide compares to other GLP-1 medications helps put storage requirements in context. Each medication has a different stability profile based on its molecular structure, concentration, and formulation.
Medication | Type | Refrigerated shelf life | Room temp allowance | Max temp |
|---|---|---|---|---|
Ozempic | Brand semaglutide (diabetes) | Until expiration date | 56 days | 86F (30C) |
Wegovy | Brand semaglutide (weight loss) | Until expiration date | 28 days | 86F (30C) |
Compounded semaglutide | Pharmacy-compounded | 30-90 days (BUD) | 14-60 days (varies) | 86F (30C) |
Compounded tirzepatide | Pharmacy-compounded | 30-90 days (BUD) | 14-28 days (varies) | 86F (30C) |
Retatrutide (research) | Investigational | Varies by source | Limited data | 77F (25C) |
Several observations stand out from this comparison. Brand-name products consistently have longer room temperature allowances because they have undergone extensive formal stability testing. The 56-day allowance for Ozempic versus the variable 14-to-60-day range for compounded semaglutide reflects the difference in testing rigor, not necessarily a difference in actual stability.
Compounded tirzepatide generally has shorter room temperature allowances than compounded semaglutide. If you are comparing the two medications, our guides on semaglutide versus tirzepatide side effects and the dosage comparison chart provide additional context. For those considering switching between the two medications, storage requirements should factor into your decision.
The maximum temperature threshold of 86 degrees Fahrenheit is consistent across all formulations. This number comes from standard pharmaceutical stability testing protocols that define "room temperature" as up to 25 degrees Celsius (77 degrees Fahrenheit) and "controlled" conditions as up to 30 degrees Celsius (86 degrees Fahrenheit). Exceeding 86 degrees Fahrenheit takes the medication outside the bounds of any published stability data.
How compounding pharmacy formulations affect stability
Not all compounded semaglutide is created equal. The specific choices a pharmacy makes during the compounding process directly impact how stable the final product is at room temperature. Understanding these differences helps explain why storage guidelines vary so dramatically between providers.
Lyophilized versus liquid formulations
Some compounding pharmacies ship semaglutide as a lyophilized (freeze-dried) powder that you reconstitute at home with bacteriostatic water. Others ship it as a ready-to-use liquid solution.
Lyophilized powder, before reconstitution, is significantly more stable than liquid formulations. The absence of water dramatically slows all degradation reactions. A properly stored lyophilized peptide can remain potent for months or even years at room temperature. However, once you add water to reconstitute it, the clock starts ticking at the same rate as any liquid formulation.
Liquid formulations have the advantage of convenience. No mixing required. But they begin degrading from the moment they are compounded, which is why their beyond-use dates tend to be shorter. If you received a liquid formulation, proper refrigeration becomes even more important because the degradation process is already underway.
For those who reconstitute at home, the mixing process itself matters. Using the correct ratio of bacteriostatic water to semaglutide affects both the concentration and the stability of the final solution. Too much water dilutes the preservative (benzyl alcohol) in bacteriostatic water, potentially reducing its antimicrobial effectiveness. Too little water creates an overly concentrated solution that may have different stability characteristics than what the pharmacy tested. Following the exact reconstitution instructions is critical for predictable storage behavior.
The role of pH in stability
The pH of the solution is one of the most significant factors affecting semaglutide stability at room temperature. Research from the Journal of Peptide Science demonstrated that semaglutide shows its highest degradation rate between pH 4.5 and 5.5. This range includes the peptide isoelectric point (pH 5.4), where the molecule carries no net electrical charge and is most susceptible to aggregation and structural collapse.
Formulations buffered above pH 7.0 demonstrate substantially better thermal stability. This means that a compounded semaglutide solution made with an alkaline buffer system will generally tolerate room temperature exposure better than one made without appropriate pH control. Unfortunately, you cannot easily test the pH of your medication at home. This is another reason why the specific pharmacy you use matters, and why their storage instructions should take precedence over generic guidelines.
Preservatives and stabilizers
Bacteriostatic water, the most common solvent for compounded semaglutide, contains 0.9 percent benzyl alcohol as a preservative. This serves two purposes. It prevents microbial growth in the vial across multiple uses, and it provides some additional stabilization to the peptide in solution.
Some pharmacies add additional stabilizers like mannitol, trehalose, or other excipients that protect the peptide structure at elevated temperatures. These additions can significantly extend room temperature stability. A formulation with optimized stabilizers might remain potent at room temperature for 60 days, while a simple semaglutide-in-bacteriostatic-water preparation might only last 14 to 21 days under the same conditions. This variability is not something you can determine by looking at the vial. It requires asking your pharmacy about their specific formulation and testing data.
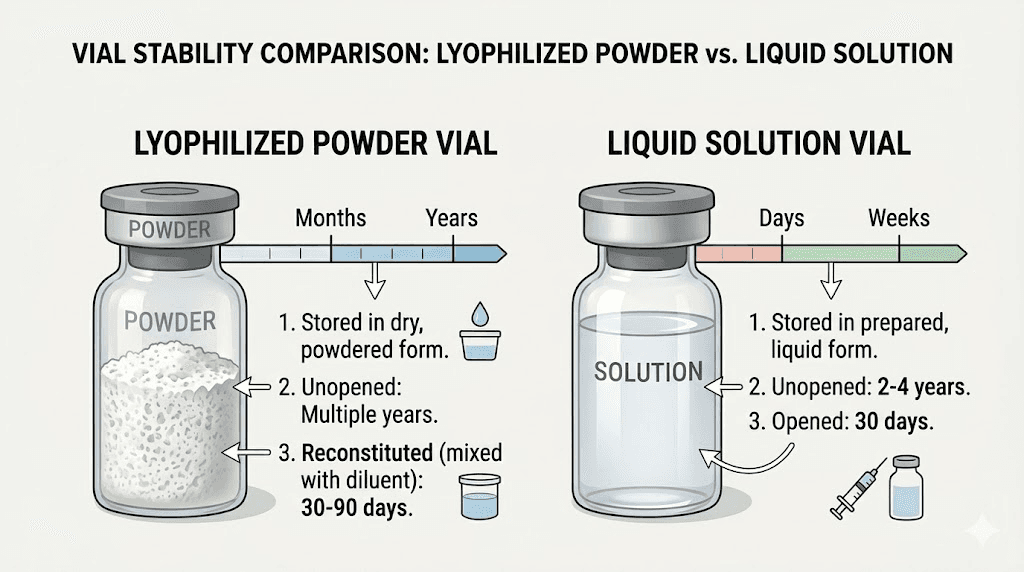
Common storage mistakes and how to avoid them
After analyzing thousands of storage-related questions from peptide researchers, clear patterns emerge. The same mistakes happen repeatedly, and most of them are easily preventable. Knowing what other people get wrong helps you avoid the same pitfalls with your own semaglutide protocol.
Mistake 1: storing in the refrigerator door
The door is the warmest part of your refrigerator. Every time you open the door, the items stored there experience a temperature spike. For food, this barely matters. For temperature-sensitive peptide medications, repeated temperature cycling accelerates degradation faster than a steady temperature at the same average.
Store your semaglutide on a middle or upper shelf toward the back of the refrigerator. This location experiences the least temperature fluctuation. Avoid placement near the freezer compartment, where accidental freezing can occur. A simple thermometer placed near your medication can help you verify the actual temperature in that specific location, which is especially useful if you have an older refrigerator with less precise temperature control.
Mistake 2: assuming re-refrigeration resets the clock
This is a critical misconception. Once compounded semaglutide has been at room temperature for a period, returning it to the refrigerator does not undo the degradation that occurred. It slows future degradation back to the refrigerated rate, but it does not reverse what already happened.
Think of it like a countdown timer, not a reset button. If your medication was at room temperature for 48 hours, those 48 hours are permanently subtracted from the total room temperature allowance, regardless of whether you put it back in the refrigerator afterward. The total cumulative time at room temperature is what matters, not the most recent storage condition.
This principle applies equally to tirzepatide storage and expired semaglutide questions. Degradation is a one-way process that cannot be reversed by returning the medication to ideal conditions.
Mistake 3: relying on visual inspection alone
As discussed earlier, you cannot see most forms of peptide degradation. Clear solution does not mean potent solution. Researchers who skip their scheduled doses or report no weight loss after several weeks may unknowingly be injecting degraded medication that has lost a significant portion of its activity.
Use time-based tracking, not visual inspection, as your primary guide for determining medication viability. Visual inspection should serve as an additional safety check, not the primary one. If you see any changes in clarity, color, or particulates, discard the medication immediately. But clear medication that has exceeded its room temperature allowance should also be discarded.
Mistake 4: leaving medication in a car
Car interiors reach extreme temperatures remarkably fast. Even on a mild 75 degree Fahrenheit day, a car interior in direct sunlight can reach 120 degrees Fahrenheit within 30 minutes. On a 90 degree day, interior temperatures can exceed 140 degrees Fahrenheit. These temperatures cause rapid and severe peptide degradation.
Never leave compounded semaglutide in a car, even briefly. Not in the glove compartment. Not in the center console. Not under a seat. If you must transport medication in a vehicle, use an insulated bag with ice packs and keep it in the climate-controlled passenger compartment. When you arrive at your destination, bring the medication inside immediately.
Mistake 5: not tracking cumulative room temperature exposure
Many people track individual instances of room temperature exposure but fail to account for cumulative time. Every time you take the vial out for an injection, it spends 5 to 15 minutes at room temperature. Over the course of a month with weekly injections, that adds up to 20 to 60 minutes. Not significant. But if you also left it out overnight once (8 hours) and had a brief power outage (3 hours), the cumulative total is now over 11 hours.
For most formulations with 14-to-60-day room temperature allowances, 11 hours is inconsequential. But tracking it ensures you always know where you stand, especially if your pharmacy has a more conservative allowance. This careful tracking becomes even more relevant when you are following precise dosage conversion protocols where consistent potency is essential for predictable results.
What to do if your semaglutide was left out too long
Sometimes the worst-case scenario happens. You find your vial on the kitchen counter after a weekend away. It has been at room temperature for 3 days in summer. Or you discover the refrigerator was accidentally unplugged. Here is how to assess the situation and make an informed decision.
Step 1: determine the duration and conditions
Figure out exactly how long the medication was at room temperature. Check your last injection date. Think about when you last opened the refrigerator. If a power outage occurred, check with your utility company for the exact timeline. Also estimate the ambient temperature. Was the air conditioning running? Was it summer with no AC? The combination of duration and temperature determines the severity of the exposure.
Step 2: compare against your pharmacy guidelines
Check the storage instructions from your compounding pharmacy. If the total room temperature exposure falls within their specified allowance (typically 14 to 60 days below 77 degrees Fahrenheit), the medication is likely still acceptable. If the exposure exceeded their guidelines, the medication should be discarded.
Step 3: inspect visually
Look at the solution carefully. Check for cloudiness, which indicates protein aggregation. Check for discoloration (any yellow, brown, or other color change). Check for particles or floating matter. Check for unusual consistency changes. If any visible changes are present, discard immediately regardless of time exposure. If the solution looks normal, it may still be viable, but remember that visual inspection cannot detect chemical degradation.
Step 4: contact your pharmacy
When in doubt, call your compounding pharmacy. Describe exactly what happened, including the duration of exposure, the estimated temperature, and any visual observations. They have stability data specific to their formulation and can make an informed recommendation. Many pharmacies will offer a replacement vial at a reduced cost if the exposure clearly exceeded safe parameters. Some pharmacies that serve the compounded semaglutide market have customer support teams specifically trained to handle storage questions.
Step 5: monitor your response
If you decide to continue using a vial that had borderline temperature exposure, pay close attention to your results. If you typically experience strong appetite suppression within a day or two of injection and that effect is noticeably reduced, the medication may have lost potency. Similarly, if you are tracking weight and notice a plateau or reversal in progress that coincides with the exposure event, that is another signal.
However, be cautious about attributing changes to a single variable. Many factors affect how quickly semaglutide works and how strong its effects feel on any given week. Diet changes, stress, sleep, hydration, and hormonal fluctuations can all influence your subjective experience of the medication effects.
Proper storage best practices
Prevention is always better than remediation. Following proper storage practices from the start minimizes the chance you will ever need to worry about room temperature exposure degrading your medication.
Refrigerator setup
Designate a specific spot in your refrigerator for medication storage. The ideal location is a middle shelf, away from both the freezer compartment and the door. Place the vial in a small container or bag to prevent it from being knocked over or pushed to the back where it might freeze against the cooling element. Set your refrigerator temperature between 36 and 40 degrees Fahrenheit (2 to 4 degrees Celsius). This keeps the medication well within the safe range while providing a buffer above the freezing point.
Consider placing a small refrigerator thermometer near your medication. This costs a few dollars and gives you peace of mind that the actual temperature, not just the thermostat setting, is within the correct range. Older refrigerators can have significant temperature variations between zones, and the thermostat setting may not accurately reflect the actual internal temperature.
Injection routine
Develop a consistent injection routine that minimizes room temperature exposure. Remove the vial from the refrigerator. Set a timer on your phone for 15 minutes. When the timer goes off, prepare your injection using the correct unit conversion for your dose. Inject following proper GLP-1 injection technique. Return the vial to the refrigerator immediately.
Choosing the best time of day for your injection and sticking to it consistently helps build a routine that becomes automatic. When the process is habitual, you are less likely to forget the vial on the counter.
Labeling and tracking
Write the following information on each vial or on a label attached to it. Include the date received, the date first opened (if lyophilized, the date reconstituted), the beyond-use date from the pharmacy, and any cumulative room temperature exposure time.
A simple system works. Keep a small notepad or use your phone notes app. Each time the vial is out of the refrigerator for more than your injection routine, note the date, time out, time back in, and estimated temperature. This creates a clear record you can reference if questions arise later. SeekPeptides members have access to protocol tracking tools that can help organize this information alongside your dosing schedule and progress data.
Seasonal storage considerations
Storage challenges change with the seasons, and being aware of these patterns helps you prepare in advance rather than react to problems after they occur.
Summer challenges
Summer creates the highest risk for room temperature excursions. Higher ambient temperatures mean your vial reaches dangerous temperature thresholds faster if left out. Power outages from summer storms are more common. Shipping conditions are more extreme, with delivery trucks and doorsteps reaching temperatures that can easily exceed 100 degrees Fahrenheit.
During summer months, request temperature-controlled shipping from your pharmacy. Many pharmacies include cold packs and insulated packaging during warm months. If your medication arrives and the cold packs are completely melted and warm, contact the pharmacy. Some, like those discussed in our compounding pharmacy reviews, have policies for replacing medication that may have been compromised during shipping.
If you live in a region with frequent summer power outages, keep a backup plan ready. A small cooler with reusable ice packs, stored in your freezer and ready to go, can protect your medication during extended outages. This same preparation helps if you need to evacuate for weather events.
Winter challenges
Winter presents the opposite risk: freezing. If your medication is shipped during winter, there is a real possibility it could freeze in transit, especially if left on an unheated porch or in a mailbox overnight. Frozen semaglutide is permanently destroyed. The ice crystal formation physically shears peptide bonds and disrupts the three-dimensional structure that makes the molecule biologically active.
During winter, try to be home for medication deliveries. If you cannot, arrange for the package to be left in a sheltered location or held at the shipping facility for pickup. Check with your pharmacy about winter shipping protocols. Quality pharmacies adjust their shipping methods seasonally, using insulated packaging and heat packs during cold months to prevent freezing.
Indoor freezing risk also increases in winter. If your refrigerator is in an unheated garage or basement, the ambient temperature around the appliance can cause the internal temperature to drop below freezing, especially near the freezer compartment. Monitor the temperature in your specific storage location during cold months.
Understanding your compounding pharmacy beyond-use date testing
The beyond-use date (BUD) assigned to your compounded semaglutide is not an arbitrary number. It comes from specific testing, and understanding that testing helps you interpret storage guidelines more accurately.
How pharmacies determine BUDs
Compounding pharmacies determine BUDs through stability testing of their specific formulation. They prepare batches under their standard conditions, then store samples at various temperatures (refrigerated, room temperature, and sometimes accelerated conditions at higher temperatures). At regular intervals, they test the samples for potency, sterility, pH, and other quality markers.
The BUD is set at the point where all tested parameters remain within acceptable specifications with a safety margin. If testing shows the formulation remains 90 percent potent at room temperature for 60 days, the pharmacy might set the BUD at 45 or 50 days to provide a safety buffer. This conservative approach means your medication likely remains effective for some time beyond the stated BUD, but using it past that date introduces uncertainty.
Why BUDs vary between pharmacies
Different pharmacies get different BUDs because they use different formulations and different testing protocols. A pharmacy using a sophisticated buffer system with optimized stabilizers will likely achieve longer stability than one using a simpler formulation. Similarly, a pharmacy that conducts more extensive testing at more time points and conditions generates more data to support a longer BUD.
This does not necessarily mean that the pharmacy with the longer BUD makes a better product. It might simply mean they invested more in stability testing or use more excipients. The pharmacy with a shorter BUD might have a perfectly effective product that they chose to be more conservative about. When evaluating compounding pharmacy options, BUD length is one factor to consider alongside potency, purity testing, customer support, and overall reputation.
What the BUD does and does not guarantee
The BUD guarantees that the medication meets potency and sterility specifications when stored under the specified conditions for the specified duration. It does not guarantee potency if storage conditions were different from those specified. It does not mean the medication instantly becomes unsafe or ineffective at midnight on the BUD date. It does not account for cumulative temperature excursions that the pharmacy could not predict.
Think of the BUD as a guarantee period, not an expiration switch. On day one past the BUD, the medication has not magically transformed. But the pharmacy can no longer assure you it meets their quality standards. The practical risk increases gradually with each additional day past the BUD, especially if storage conditions were not ideal. For guidance on using semaglutide past its expiration, our detailed guide covers the specific considerations involved.
Special considerations for different semaglutide concentrations
Compounded semaglutide comes in several common concentrations, and concentration can affect stability behavior at room temperature.
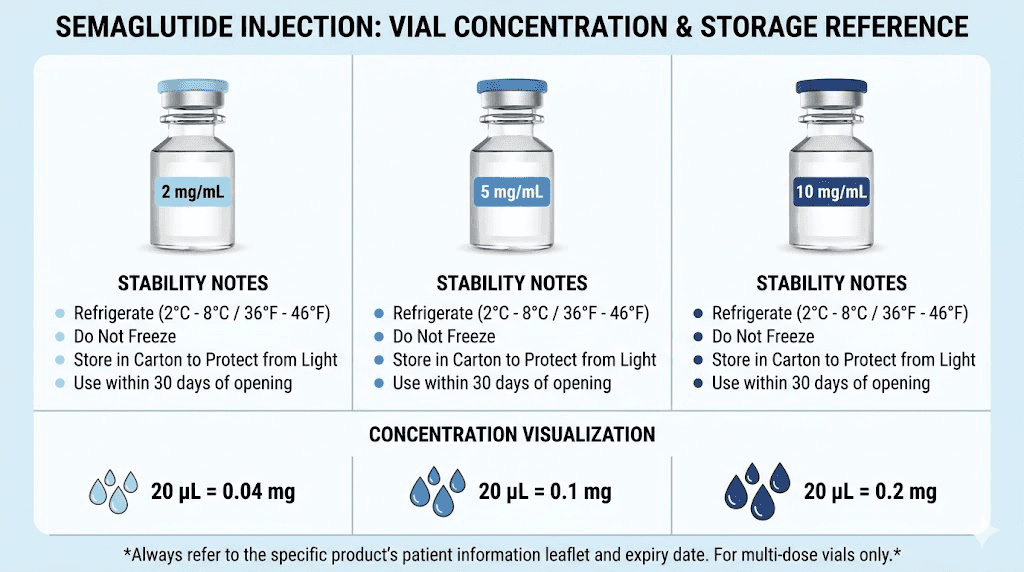
Low concentration (1-2mg/mL)
Lower concentrations have more water relative to peptide content. This means the degradation reactions have more solvent to operate in, but the percentage of peptide affected by degradation at any given time may be proportionally higher. For practical purposes, low-concentration formulations should follow the same storage guidelines as other concentrations, but they may be slightly less forgiving of temperature excursions.
If you are using a low-concentration formulation, the volume you inject per dose is larger, which means you may go through the vial faster. This actually works in your favor for storage, since faster use means less total time the medication exists before it is consumed. Researchers working with these concentrations benefit from understanding unit-to-milliliter conversions to ensure accurate dosing from the larger injection volumes.
Standard concentration (5mg/mL)
This is the most common compounded concentration and the one most pharmacy BUD testing is based on. If your pharmacy provides a standard 5mg/mL semaglutide formulation, their storage guidelines are directly applicable without adjustment. Most of the published stability data and general guidelines in this article are based on concentrations in this range.
High concentration (10mg/mL and above)
Higher concentrations pack more peptide into less solution. This can actually provide some stability advantages because the higher peptide concentration creates a more viscous solution that may be slightly more resistant to some degradation pathways. However, high-concentration solutions are also more susceptible to aggregation, where peptide molecules clump together and become inactive.
If you are using a 10mg/mL semaglutide formulation, pay particular attention to visual inspection for cloudiness or particles, which indicate aggregation. These formulations may need stricter temperature control than standard concentrations. Follow your pharmacy specific guidelines closely, as their BUD testing accounts for the unique stability profile of their high-concentration product.
How room temperature exposure affects your treatment results
The practical consequence of improper storage is not just wasted medication. It can directly impact your treatment outcomes in ways that are difficult to diagnose without knowing about the storage issue.
Gradual potency loss
Peptide degradation at room temperature does not happen all at once. It is a gradual process. A vial that was at room temperature for two weeks might retain 95 percent of its potency. The same vial at four weeks might be at 85 percent. At six weeks, maybe 70 percent. You would still be injecting semaglutide, and it would still have some effect, but the dose you think you are taking and the dose you are actually receiving would be increasingly different.
This gradual potency loss can mimic other problems. If you are experiencing fatigue on semaglutide or noticing that your appetite suppression is weakening, degraded medication is one possible explanation alongside dose tolerance, dietary changes, and metabolic adaptation. Without knowing the storage history of your vial, it is impossible to rule this out as a contributing factor.
Inconsistent results week to week
If your storage practices are inconsistent, with the vial sometimes left out for hours and other times handled perfectly, you may experience inconsistent results from week to week. One injection from a properly stored portion of the vial works great. The next, from a section that degraded more due to the vial being left out, is noticeably weaker.
This inconsistency is particularly frustrating because it makes it difficult to calibrate your protocol. If you are trying to determine whether your current dosage is correct, inconsistent medication potency introduces a variable that confuses the picture entirely. You might increase your dose thinking you need more, when the real issue is storage related potency loss. People working through a first week on semaglutide or adjusting their ongoing protocol need reliable medication to make accurate assessments about what is working.
The financial impact
Compounded semaglutide represents a significant investment. Discarding a vial due to improper storage means paying for a replacement. But using a degraded vial might be even more expensive in the long run if it leads to poor results that you try to solve by increasing doses, switching medications, or adding other interventions.
Proper storage costs nothing. It requires only attention and habit. The return on that small investment of attention is consistent medication potency, predictable results, and no wasted product. For those managing costs carefully, our guide on compounded semaglutide options covers additional ways to optimize the value of your treatment.
The role of your injection schedule in storage management
Your injection frequency directly affects how long each vial needs to remain viable. This relationship between dosing schedule and storage duration is worth considering when planning your protocol.
Semaglutide is typically injected once weekly. A vial containing enough medication for 4 to 8 weeks of treatment needs to maintain potency across that entire period. If the vial is refrigerated throughout, this is rarely an issue since most compounded semaglutide BUDs extend to 30 to 90 days refrigerated.
But if you are traveling for a week and the vial will be at room temperature during that time, you need to consider both the travel period and the remaining weeks of use after you return home. A vial that spent one week at room temperature during a trip, then returned to the refrigerator for the remaining three weeks of use, will likely be fine. A vial that spent four weeks at room temperature because you did not realize it needed refrigeration is a very different situation, especially if your pharmacy BUD only allows 14 days at room temperature.
For individuals on specific dosing schedules, our unit conversion guides and the semaglutide dosage calculator help ensure you are drawing the correct amount from your vial each week, which in turn helps you predict exactly how long each vial needs to last and plan storage accordingly.
Compounded semaglutide storage versus other peptides
If you work with multiple peptides, understanding how semaglutide storage compares to others helps you develop efficient storage routines for all your research compounds.
Semaglutide is actually one of the more stable peptide compounds at room temperature. Its molecular structure includes a C-18 fatty acid chain that enhances albumin binding in the body, and this same structural feature provides some additional stability in solution. Many other research peptides, particularly smaller ones like BPC-157 or TB-500, can be more sensitive to temperature excursions.
GLP-1 receptor agonists as a class tend to have reasonable room temperature stability. Tirzepatide, which targets both GIP and GLP-1 receptors, has a similar stability profile, though specific compounded formulations may vary. Our guide on compounded tirzepatide room temperature storage covers the specific considerations for that medication.
If you are also working with other compounds, the same general principles apply. Keep everything refrigerated. Minimize room temperature exposure. Track cumulative time outside the refrigerator. Use your peptide reconstitution calculator to ensure correct mixing ratios, and follow the specific storage guidelines for each compound. The differences between research and pharmaceutical grade peptides also affect storage requirements, with pharmaceutical grade products generally having more reliable stability data.
When to contact your healthcare provider or pharmacy
While this guide provides comprehensive storage information, certain situations warrant direct professional consultation. Knowing when to reach out saves time and prevents potentially harmful decisions.
Contact your compounding pharmacy if the medication was at room temperature beyond their specified allowance. Contact them if the medication was exposed to temperatures above 86 degrees Fahrenheit for any extended period. Reach out if you notice any visual changes in the solution, if you suspect the medication may have been frozen at any point, or if you are experiencing a sudden decrease in medication effectiveness that could indicate potency loss.
Contact your prescribing healthcare provider if you have been using medication that you now suspect was degraded, if you want to discuss whether to continue a vial with borderline temperature exposure, if you need guidance on restarting your protocol after a storage-related interruption, or if you experience any unusual side effects that could be related to degraded medication. Managing side effects like constipation or withdrawal symptoms is much easier when your healthcare provider has complete information about your medication storage history.
For researchers serious about optimizing their peptide protocols, SeekPeptides provides comprehensive storage guides, stability databases, and community support where experienced members share practical solutions to common storage challenges. The membership includes protocol tracking tools that help you monitor both your dosing schedule and storage conditions in one place.
Frequently asked questions
Can I use compounded semaglutide that was left out overnight?
Yes. A single overnight exposure of 8 to 12 hours at normal room temperature (below 77 degrees Fahrenheit) falls well within the acceptable range for virtually all compounded semaglutide formulations. Inspect the vial visually, return it to the refrigerator, and note the cumulative time out. Continue using as normal. This brief exposure will not cause meaningful potency loss.
Does putting semaglutide back in the fridge after being left out restore its potency?
No. Returning semaglutide to the refrigerator slows future degradation but does not reverse degradation that has already occurred. Think of it as pausing a countdown timer rather than resetting it. The cumulative time at room temperature still counts against the total allowance, regardless of current storage conditions. Our guide on semaglutide refrigeration requirements covers this topic in additional detail.
How can I tell if my compounded semaglutide has gone bad?
Visible signs of degradation include cloudiness, discoloration, particles floating in the solution, or changes in consistency. However, most potency loss from temperature exposure is invisible. The solution can look perfectly normal while containing significantly less active peptide. Use time-based guidelines from your pharmacy rather than visual inspection alone to determine if the medication is still viable.
Is compounded semaglutide less stable than Ozempic or Wegovy?
Not necessarily. Compounded semaglutide may be equally stable or even more stable depending on the formulation. The difference is that brand-name products have published stability data from extensive manufacturer testing, while compounded products rely on each pharmacy individual testing. Some compounded formulations with optimized buffers and stabilizers demonstrate excellent room temperature stability. The lack of standardized data, not inherently worse stability, is the primary concern.
What happens if my compounded semaglutide freezes?
Freezing permanently destroys semaglutide. Ice crystal formation physically disrupts the peptide structure, causing irreversible damage. Never use semaglutide that has been frozen, even if it looks normal after thawing. The damage is at the molecular level and cannot be detected visually. If you suspect freezing occurred during shipping or storage, discard the medication and obtain a replacement.
Can I travel with compounded semaglutide on a plane?
Yes. Carry injectable medications in your carry-on luggage with a prescription label or letter from your prescriber. Use an insulated bag with ice packs to maintain temperature. TSA allows medically necessary liquids and syringes through security checkpoints. Never pack medication in checked luggage, where cargo hold temperatures can drop well below freezing during flight. Our guide on GLP-1 injection procedures covers additional travel considerations for maintaining your protocol on the road.
Does the concentration of my compounded semaglutide affect how long it can be out?
Concentration can have a minor effect on stability, but the primary factors are the formulation excipients, pH, and preservatives used by your pharmacy. Higher concentrations may be slightly more prone to aggregation at elevated temperatures. Lower concentrations may degrade proportionally faster. For practical purposes, follow the specific storage guidelines provided by your pharmacy for your exact product, regardless of concentration.
How long does compounded semaglutide last in the fridge?
Most compounded semaglutide has a refrigerated beyond-use date of 30 to 90 days, depending on the pharmacy and formulation. This is the date assigned by the compounding pharmacy based on their stability testing. Always use the medication before this date. For detailed guidance, see our complete guide on how long compounded semaglutide lasts in the fridge.
External resources
Influence of Buffering Capacity, pH, and Temperature on Semaglutide Stability (PubMed)
Journal of Peptide Science: Semaglutide Preformulation Study
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your results stay consistent.