Feb 27, 2026

You pull the vial from the refrigerator and something looks different. The liquid inside is not what you expected. Maybe it is pink. Maybe it has a yellowish tint. Maybe it looks completely clear and you are wondering if it should have some color at all.
That moment of uncertainty is more common than most people realize. Tirzepatide comes in multiple formulations, from multiple sources, and the color of the solution varies dramatically depending on what you are holding. Brand-name Mounjaro looks nothing like a compounded tirzepatide with B12 and glycine. A lyophilized powder looks nothing like a pre-filled pen. And some color changes signal genuine safety concerns while others are completely normal.
This guide breaks down exactly what every formulation of tirzepatide should look like, what each color means, and when a color change means you need to throw the vial away. Whether you are using FDA-approved tirzepatide or a compounded version, understanding the expected appearance of your medication is one of the simplest safety checks available. And it takes about three seconds. SeekPeptides members consistently report that visual inspection is their first line of defense before every injection, and this article will show you exactly how to do it right.
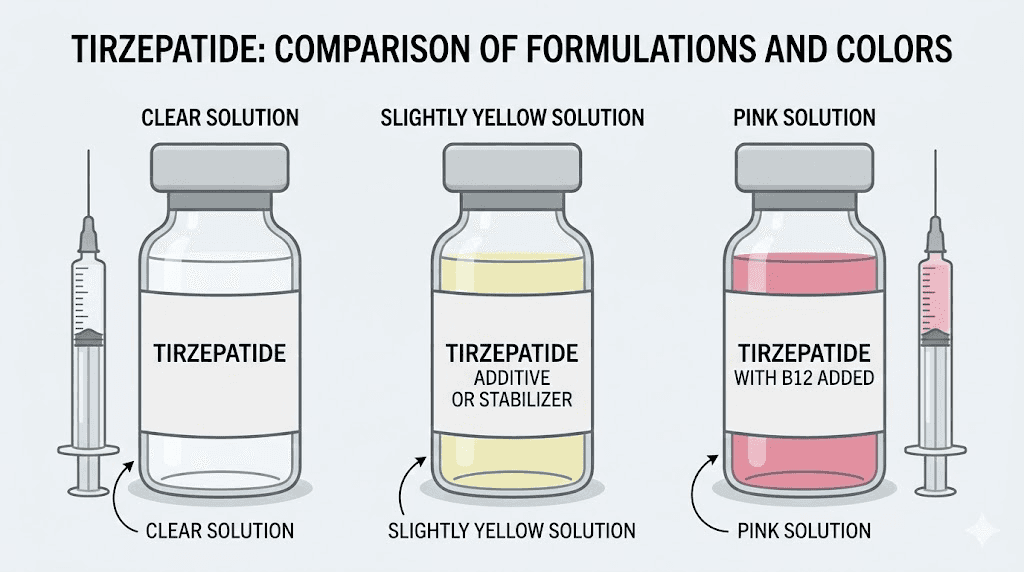
What color is FDA-approved tirzepatide
The simplest answer first. FDA-approved tirzepatide, sold as Mounjaro for type 2 diabetes and Zepbound for weight management, is a clear, colorless to slightly yellow solution. That is the official description straight from the prescribing information. Clear means you can see through it without any cloudiness, haziness, or floating particles. Colorless to slightly yellow means the solution ranges from perfectly transparent to having a faint warm tint.
Both products contain the exact same active ingredient. The difference is the FDA-approved indication, not the formulation. So whether you are holding a Mounjaro pen or a Zepbound pen, the liquid inside should look identical.
Here is what normal looks like in practice. Hold the pen or vial up to a light source. The solution should be transparent. You should be able to read text through the vial if you hold it in front of printed words. There should be no cloudiness, no haziness, no swirling particles, no sediment at the bottom. A very faint yellow tint is perfectly normal and does not indicate any quality issue whatsoever. Think of it like water with the slightest hint of warmth. Some batches appear completely colorless while others have that subtle yellow quality, and both are within the acceptable range established during clinical trials.
Why the variation? Tirzepatide is a 39-amino-acid peptide with a C20 fatty diacid moiety attached via a linker. The slight yellow coloration comes from the peptide itself and minor variations in manufacturing batches. It is not a sign of degradation or contamination. The FDA specifically accounts for this range in their quality standards, which is why the label says "colorless to slightly yellow" rather than just "colorless."
Mounjaro pen color coding by dose
There is an important distinction between the color of the solution inside the pen and the color of the pen itself. Eli Lilly designed Mounjaro KwikPens with a color-coded system to help users identify their correct dose at a glance. Each dose strength has a unique color on the pen label and packaging.
Here is the complete Mounjaro pen color chart:
Dose | Pen color | Concentration |
|---|---|---|
2.5 mg | Gray | 2.5 mg/0.5 mL |
5 mg | Purple | 5 mg/0.5 mL |
7.5 mg | Green | 7.5 mg/0.5 mL |
10 mg | Yellow | 10 mg/0.5 mL |
12.5 mg | Blue | 12.5 mg/0.5 mL |
15 mg | Orange | 15 mg/0.5 mL |
The color appears as a prominent band or triangle shape on the pen label and the outer carton packaging. Every pen also has a gray, triangle-shaped safety cover on one end and a purple injector button on the other end, regardless of dose strength. The dose-identifying color is on the label itself.
This system exists because dosing errors with injectable medications can be serious. If you are starting at 2.5 mg and accidentally grab a 15 mg pen, that is a six-fold overdose. The color system provides a visual safety check, but it should never replace reading the actual milligram strength printed on the pen. Always confirm the number, not just the color.
A common point of confusion involves the 5 mg pen and the injector button. The 5 mg pen has a purple label, and every pen has a purple injector button. These are two different purples for two different purposes, but new users sometimes mix them up. The label purple identifies the dose. The button purple is just the standard design across all pens.
If you are titrating through multiple dose levels, you will notice the pen packaging color changing at each step. Gray to purple to green to yellow to blue to orange. This visual progression can actually help track your titration journey, and many users report that the color change at each new dose level feels like a milestone.

Zepbound pen colors
Zepbound is the weight management version of tirzepatide, also made by Eli Lilly. The same dose strengths are available: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg. Each Zepbound pen contains 0.5 mL of solution, just like Mounjaro.
Zepbound uses its own distinct color-coding system on the packaging and pen labels, though the solution inside is identical, clear and colorless to slightly yellow. The Zepbound pens are designed to be visually distinct from Mounjaro pens so that users and pharmacists do not accidentally confuse the two products, even though the active ingredient is the same. If you are switching medications or managing multiple prescriptions, this visual distinction becomes especially important.
Regardless of which brand you use, always verify the milligram strength printed on the pen label before every injection. Color coding is a helpful safety feature, not a replacement for reading the label. And the solution inside should always appear clear, not cloudy.
What color is compounded tirzepatide
This is where things get interesting. And sometimes confusing.
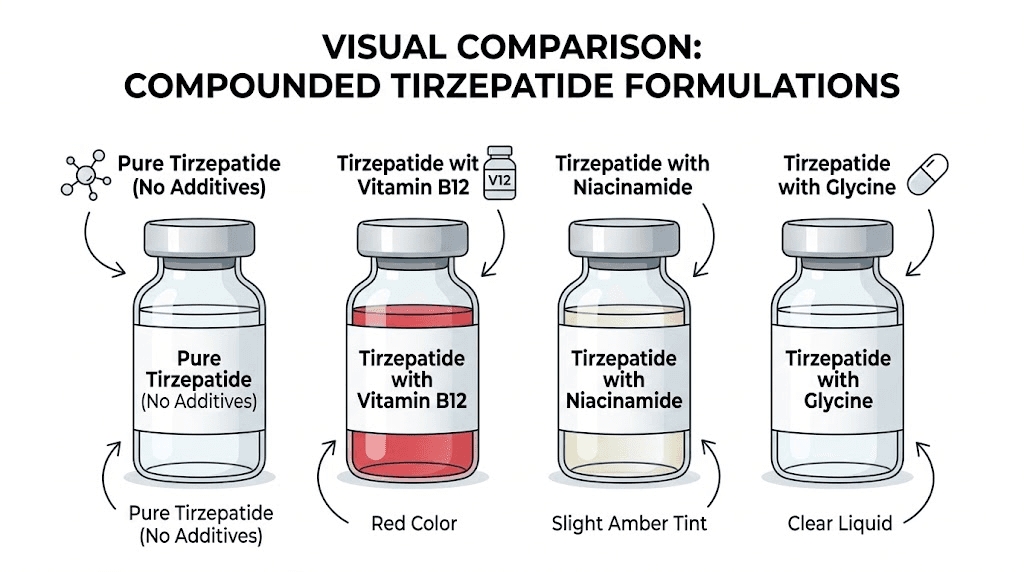
Compounded tirzepatide can look dramatically different from the FDA-approved version. The color depends entirely on what the compounding pharmacy adds to the formulation. Pure compounded tirzepatide without any additives should appear clear and colorless, similar to the brand-name product. But most compounding pharmacies add one or more additional ingredients, and those additives change the color.
The most common compounded tirzepatide formulations include combinations with vitamin B12 (cyanocobalamin or methylcobalamin), niacinamide, glycine, L-carnitine, or some combination of these. Each additive can affect the solution color differently, and the concentration of each ingredient further influences the exact shade you see.
Before we dive into each formulation, here is a quick reference:
Formulation | Expected color | Why |
|---|---|---|
Pure tirzepatide | Clear to slightly yellow | Peptide itself |
Tirzepatide + B12 | Pink to light red | Cyanocobalamin pigment |
Tirzepatide + niacinamide | Clear to slightly yellow | Niacinamide is colorless |
Tirzepatide + glycine | Clear to slightly yellow | Glycine is colorless |
Tirzepatide + glycine + B12 | Pink to light red | B12 dominates color |
Tirzepatide + L-carnitine | Clear to slightly yellow | L-carnitine is colorless |
The pattern is simple. If B12 is in the formulation, expect pink. If B12 is not in the formulation, expect clear to slightly yellow. Everything else is a red flag worth investigating.
Tirzepatide with B12 color
The most dramatic color difference comes from tirzepatide compounded with vitamin B12. Cyanocobalamin, the most common form of supplemental B12, has a deep reddish-pink color in solution. When mixed with tirzepatide, it turns the entire vial pink to light red. The exact shade depends on the B12 concentration.
At lower B12 concentrations, the solution appears pale pink. Almost like rose water. At higher concentrations, it deepens to a more vivid pink or even a light cherry red. Both are normal. The color should be uniform throughout the vial with no layering, separation, or floating particles.
For a detailed breakdown of B12-specific color variations, see our complete guide on what color tirzepatide with B12 should be.
Why do pharmacies add B12 to tirzepatide? Several reasons. GLP-1 receptor agonists like tirzepatide can reduce B12 absorption over time by slowing gastric motility. Adding B12 directly to the injection helps prevent or address deficiency. Some users also report that the B12 helps with the fatigue that sometimes accompanies tirzepatide use, though this is anecdotal rather than clinically established.
The key safety point: if your pharmacy told you to expect a pink solution because it contains B12, then pink is normal. If you receive a pink solution when you did not order a B12 formulation, that is a problem. Always verify what your prescription should contain before your first injection.
Tirzepatide with niacinamide color
Tirzepatide compounded with niacinamide should appear clear to slightly yellow. Niacinamide (also called nicotinamide, a form of vitamin B3) is colorless in solution at the concentrations used in compounded formulations, typically around 2 mg/mL. It does not change the appearance of the tirzepatide in any visible way.
Niacinamide serves as a stabilizer in compounded tirzepatide formulations. It helps prevent peptide aggregation, the process where tirzepatide molecules clump together and lose their effectiveness. This is why pharmacies like Empower Pharmacy include it in their formulations. The stabilization benefit extends the shelf life and helps maintain potency throughout the use period.
If your tirzepatide-niacinamide formulation appears anything other than clear to slightly yellow, something is wrong. Pink, red, brown, or cloudy appearances in a niacinamide-only formulation (no B12) warrant contacting your pharmacy immediately.
Tirzepatide with glycine color
Tirzepatide with glycine should also appear clear to slightly yellow. Glycine is a simple amino acid that dissolves completely and colorlessly in solution. Like niacinamide, it serves primarily as a stabilizer, helping to maintain the structural integrity of the tirzepatide peptide and prevent degradation during storage.
The glycine formulation is popular because glycine has additional biological benefits. It supports collagen production, liver function, and sleep quality. Some researchers appreciate getting these secondary benefits alongside their tirzepatide protocol, though the amounts included in compounded formulations are typically modest.
A clear, colorless solution is what you want to see. No exceptions for this formulation.
Tirzepatide with glycine and B12 color
The triple-compound formulation of tirzepatide with glycine and B12 follows the B12 color rule. B12 dominates. Expect pink to light red, with the glycine contributing no visible color change.
This is one of the most common compounded formulations available, combining the stabilizing benefits of glycine with the nutritional support of B12. The color should be consistent throughout the vial. If you notice the pink color fading over time or the solution becoming cloudy, these are signs of potential degradation that warrant a call to your pharmacy.
Tirzepatide with L-carnitine color
L-carnitine is another additive sometimes compounded with tirzepatide for its role in fat metabolism. In solution, L-carnitine is colorless. A tirzepatide and L-carnitine formulation should appear clear to slightly yellow, identical to pure tirzepatide or the niacinamide formulation.
Any color deviation from clear or slightly yellow in an L-carnitine formulation, if it does not also contain B12, should be investigated before use.
Lyophilized tirzepatide powder color
Not all tirzepatide comes as a ready-to-inject liquid. Lyophilized tirzepatide arrives as a freeze-dried powder that must be reconstituted before use. The appearance of this powder follows specific standards that help you assess quality before you even add the bacteriostatic water.
A properly lyophilized tirzepatide vial should contain a white to off-white powder or cake. The powder might appear as a solid disc, a fluffy cake, or a loose powder depending on the specific lyophilization process used. All of these forms are acceptable as long as the color remains in the white to off-white range.
What you do not want to see: yellow powder, brown powder, powder with dark specks, or a collapsed or sticky-looking cake. These can indicate degradation during manufacturing, improper storage during shipping, or contamination. If the powder looks off, do not reconstitute it. Contact the supplier.
The difference between lyophilized and liquid peptides matters here because lyophilized forms are generally more stable during shipping. The powder form is less susceptible to temperature fluctuations that can cause degradation in liquid formulations. This is one reason some researchers prefer the powder form despite the extra step of reconstitution.
What reconstituted tirzepatide should look like
After adding bacteriostatic water to lyophilized tirzepatide, the powder should dissolve completely within 30 to 60 seconds of gentle swirling. Do not shake vigorously. Shaking creates foam and can damage the peptide bonds. Gentle swirling is all that is needed.
The resulting solution should be clear, colorless, and free of any visible particles. If you see floating specks, cloudiness, or the powder does not fully dissolve after two minutes of gentle swirling, something is wrong. Either the powder has degraded, the bacteriostatic water is contaminated, or the reconstitution ratio is off.
For detailed reconstitution ratios and step-by-step instructions, our guide on how to reconstitute tirzepatide covers everything from water volume calculations to proper technique. You can also use the peptide reconstitution calculator to determine exact volumes based on your vial concentration.
What each tirzepatide color means
Now for the practical part. You are looking at your tirzepatide and it is a specific color. What does it mean? Let us go through every possibility.
Clear and colorless
This is the ideal appearance for pure tirzepatide, FDA-approved or compounded without B12. It indicates proper manufacturing, appropriate storage, and no visible degradation. If your formulation does not contain B12 and the solution is clear and colorless, everything checks out visually.
Clear and colorless is also what you should see after reconstituting lyophilized tirzepatide. The powder should dissolve completely, leaving no trace of cloudiness.
Slightly yellow
A faint yellow tint is within the normal range for tirzepatide. This applies to brand-name Mounjaro, Zepbound, and compounded formulations without B12. The yellow comes from the tirzepatide peptide itself and is specifically listed as acceptable in the FDA prescribing information.
However, there is a difference between "slightly yellow" and "noticeably yellow." Slightly yellow means you can barely detect the tint. You might need to hold it against a white background to even notice it. If the yellow is immediately obvious, deep, or approaching amber, that crosses the line into potential degradation territory. Think lemonade versus apple juice. Lemonade-level yellow is fine. Apple juice yellow is not.
A deepening yellow color over time often indicates oxidation. Tirzepatide, like many peptides, is susceptible to oxidative degradation, particularly at certain pH levels and when exposed to temperatures above the recommended storage range. If your vial was clear last week and is noticeably more yellow this week, the peptide may be breaking down. Research shows tirzepatide is most prone to oxidation at pH 5, even at refrigerated temperatures of 5 degrees Celsius.
Pink to light red
Pink or light red means one thing in almost every case: vitamin B12. If your compounded tirzepatide contains cyanocobalamin or methylcobalamin, pink is the expected color. Completely normal. Not a safety concern.
The shade varies. Pale pink at low B12 concentrations. Deeper pink or light red at higher concentrations. Both are fine as long as the color is uniform and the solution is clear (transparent, not cloudy).
But here is the critical question: did you order a formulation with B12? If yes, pink is exactly right. If no, pink in your tirzepatide is a major red flag. It could mean the pharmacy made a compounding error, you received the wrong medication, or something has contaminated the vial. Do not use it until you verify with your pharmacy.
For more on B12 formulations specifically, read our deep dive on tirzepatide with B12 color. And if you are curious about whether the same color rules apply to semaglutide, our guide on why semaglutide turns red covers the parallel situation.
Bright red or dark red
Bright red or dark red is not normal for any tirzepatide formulation. Even B12-containing compounds should only reach a light red or vivid pink, not a deep or dark red. A dark red solution could indicate excessive B12 concentration beyond what was intended, degradation of the B12 itself, contamination with blood or other biological material, or a compounding error.
Do not use dark red tirzepatide. Contact your pharmacy.
Brown or amber
Brown or amber coloration is a strong indicator of advanced oxidative degradation. The peptide has broken down significantly, and the degradation byproducts are creating the darker color. This typically happens when tirzepatide has been exposed to elevated temperatures for an extended period, has been stored outside the refrigerator for longer than the recommended timeframe, or has been exposed to direct light.
Discard immediately. Brown tirzepatide has lost significant potency and may contain degradation products that are potentially harmful.
Cloudy or milky
Cloudiness is one of the most concerning visual changes. A cloudy or milky appearance typically indicates one of three things: peptide aggregation, where the tirzepatide molecules have clumped together into larger structures that scatter light; bacterial or fungal contamination, where microbial growth is creating turbidity; or improper reconstitution, where the powder did not fully dissolve or the wrong diluent was used.
None of these scenarios are safe for injection. Aggregated peptides may not work as intended and can trigger injection site reactions. Contaminated solutions can cause serious infections. And improperly reconstituted solutions have unpredictable concentrations.
Discard cloudy tirzepatide. No exceptions.
Particles or flakes visible
Visible particles floating in the solution, whether they look like tiny crystals, flakes, or specks, mean the tirzepatide should not be used. Particles can form from peptide crystallization out of solution, which happens when the formulation is frozen, contamination during the reconstitution or drawing process, degradation products that have precipitated out of solution, or incompatibility between the tirzepatide and the vial or stopper material.
Before every injection, hold the vial up to a light source and look carefully. Tilt it gently and watch for any particles that might be settled at the bottom or floating through the solution. This takes five seconds and can prevent injecting compromised medication.

How storage affects tirzepatide color
Storage is the single biggest factor that influences whether your tirzepatide maintains its proper color throughout its use period. Get storage right and the color stays consistent. Get it wrong and you will watch your medication change color in real time.
Temperature and color stability
Tirzepatide, like most peptides, is a delicate molecule. The peptide bonds that hold its 39 amino acids together are susceptible to hydrolysis at elevated temperatures. As these bonds break, the degradation products can change the color of the solution, usually pushing it toward yellow and eventually brown.
The recommended storage temperature for both FDA-approved and compounded tirzepatide is 2 to 8 degrees Celsius, which is standard refrigerator temperature. At this range, the degradation rate is minimized and the solution should maintain its original color throughout the labeled shelf life.
But life happens. Pens and vials come out of the fridge for injections. Shipments get delayed. Refrigerators have warm spots. How much does this matter?
FDA-approved Mounjaro and Zepbound can be stored at room temperature (below 30 degrees Celsius or 86 degrees Fahrenheit) for up to 21 days. After that, the pen must be discarded even if there is medication remaining. Compounded formulations may have different room-temperature stability windows depending on the pharmacy, the specific formulation, and the preservative system used. Always check with your compounding pharmacy for their specific beyond-use dating.
If your tirzepatide has been out of the fridge for longer than recommended and you notice any color change, err on the side of caution and replace it.
Light exposure and color changes
UV light accelerates peptide degradation. This is why Mounjaro and Zepbound come in opaque packaging with specific instructions to store the pens in the original carton. The carton is not just packaging. It is a light shield.
Compounded tirzepatide in clear glass vials is more vulnerable to light-induced degradation. If your vial is clear glass, store it in a dark area of the refrigerator, away from the light that turns on when you open the door. Some researchers wrap their vials in aluminum foil for additional protection, though this is not strictly necessary if the vial stays in the fridge with the door closed most of the time.
Light-induced degradation can cause yellowing, which is why a vial that was perfectly clear when it arrived might develop a yellow tint over weeks if repeatedly exposed to light during each injection session.
Freezing and color effects
Never freeze tirzepatide. Freezing causes ice crystal formation within the solution, and those crystals can physically damage the peptide structure. When the solution thaws, you may see particles, cloudiness, or both. These are signs that the peptide has been structurally compromised.
Frozen tirzepatide may appear normal after thawing in some cases, but the damage is invisible at the molecular level. The peptide may have lost potency or formed aggregates that look clear but behave unpredictably. This is why the manufacturer explicitly states that frozen pens should be discarded.
If you are traveling with tirzepatide, use a temperature-controlled cooler bag. Do not put it directly on ice packs, as direct contact with frozen surfaces can freeze the medication even in a cooler.
When to discard tirzepatide based on appearance
Making the discard decision is not always easy. Medication costs money. Nobody wants to throw away a vial that might still be fine. But the risk-reward calculation here is clear: using compromised medication ranges from ineffective to dangerous, while replacing a questionable vial is just a phone call to your pharmacy.
Here is a definitive guide:
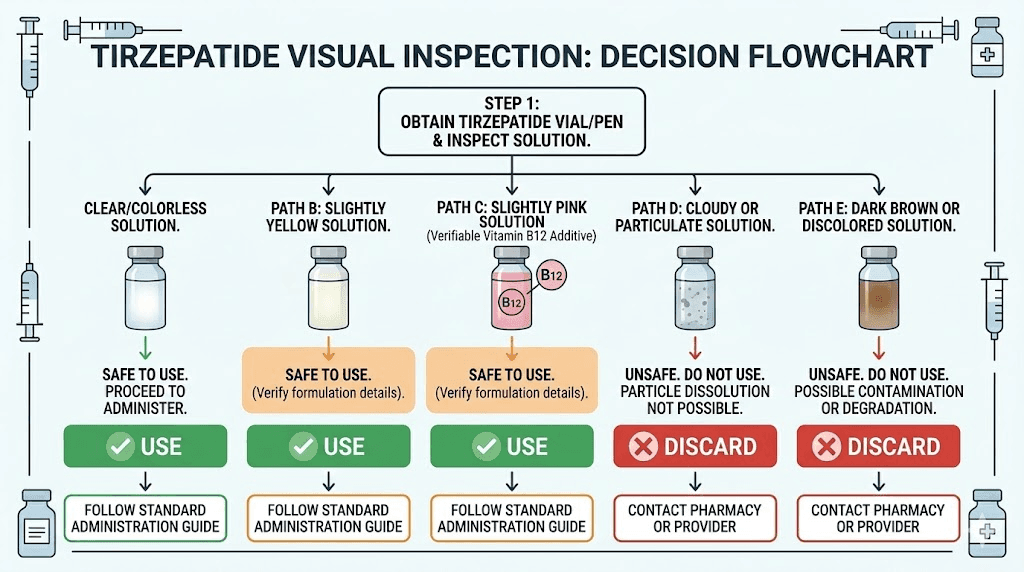
Always discard if you see:
Cloudiness or haziness (any degree)
Visible particles, flakes, or sediment
Brown, amber, or dark yellow color
Dark red color (even in B12 formulations)
Any color you were not told to expect
Layering or separation within the solution
Sticky or viscous consistency (should flow freely like water)
Safe to use:
Clear and colorless (all formulations)
Clear with slight yellow tint (all formulations)
Clear pink to light red (B12 formulations only)
Uniform color throughout
No visible particles
Contact pharmacy before using if:
Color seems different from the last vial of the same formulation
Yellow tint has deepened since you started using the vial
The solution looks different from what your pharmacy described
You are unsure whether the product contains B12
When in doubt, throw it out. Or at minimum, call your pharmacy. This applies whether you are using brand-name Mounjaro, Zepbound, or any compounded formulation. The visual inspection takes seconds. The consequences of injecting compromised medication can take much longer to resolve.
How to properly inspect tirzepatide before each injection
Visual inspection should become automatic. Like checking your mirrors before driving. It takes seconds, costs nothing, and can prevent real problems. Here is the step-by-step process that SeekPeptides members follow before every injection.
Step 1: Check the temperature
If the vial or pen has been in the refrigerator, let it warm to room temperature for 15 to 30 minutes before inspecting. Cold solutions can sometimes appear slightly different from room temperature solutions, and small bubbles from temperature changes can be mistaken for particles.
Step 2: Hold against a light source
Hold the vial or pen up to a well-lit area. A window with natural light works well. The goal is to see the solution clearly. Look through the solution, not at the outside of the vial.
Step 3: Check for clarity
Can you see clearly through the solution? Any cloudiness or haziness means discard. This check is easiest with clear glass vials. With pens, you may need to look through the viewing window on the cartridge.
Step 4: Check for particles
Gently tilt the vial or pen back and forth a few times. Watch for any specks, flakes, or crystals that might be floating in the solution or settling at the bottom. Even tiny particles that you can barely see are grounds for discarding.
Step 5: Verify the color
Does the color match what you expect for your specific formulation? Clear to slightly yellow for non-B12 formulations. Pink to light red for B12 formulations. Compare to what the solution looked like when you first received it. Any significant change in color since then is a warning sign.
Step 6: Check for changes from last use
If this is a multi-dose vial, compare today to your last injection. Has anything changed? Gradual yellowing, new cloudiness, or any particles that were not there before all warrant investigation. Keeping the vial in the same spot in the refrigerator and checking it under the same light conditions each time makes comparison easier.
This entire process takes 15 to 30 seconds. Build it into your injection routine and it becomes second nature.
Tirzepatide color versus semaglutide color
If you have used semaglutide before switching to tirzepatide, or if you are comparing the two medications, the color expectations are very similar. Both are clear, colorless to slightly yellow in their pure forms. Both turn pink or red when compounded with B12. Both should never appear cloudy or contain particles.
The main visual differences are in the delivery devices rather than the solutions themselves. Ozempic pens look different from Mounjaro pens. Wegovy pens look different from Zepbound pens. But pop the solutions out and line them up, and you would have a hard time telling them apart based on appearance alone.
For a detailed comparison of how the two medications differ in ways that actually matter, like mechanism of action, dosing protocols, and side effect profiles, see our comprehensive semaglutide vs tirzepatide comparison.
One notable difference: the semaglutide with B12 and semaglutide with methylcobalamin formulations follow the exact same color rules as their tirzepatide counterparts. Pink means B12. Clear means no B12. If you ever read about red semaglutide, the same explanations apply to tirzepatide.
Counterfeit tirzepatide and color
With the explosive popularity of tirzepatide for weight loss, counterfeit products have become a growing concern. Color can be one indicator of a counterfeit product, though it is far from the only one.
Counterfeit tirzepatide may appear in unusual colors that do not match any legitimate formulation. A green tint, a blue tint, or an unusual shade of yellow that does not match the expected slightly yellow range could indicate a fake product. But sophisticated counterfeits can look identical to the real thing. Color alone cannot confirm authenticity.
Other signs of counterfeit tirzepatide include packaging that looks slightly off (fonts, colors, logos that do not match official packaging), spelling errors on labels or packaging, missing or incorrect lot numbers and expiration dates, pens or vials that feel different in weight or construction, unusually low pricing from unverified sources, and sellers who cannot provide proper documentation or licensing.
The safest approach is to obtain tirzepatide only through verified pharmacies, whether that means a traditional pharmacy dispensing brand-name Mounjaro or Zepbound, or a licensed compounding pharmacy operating under proper oversight. If the price seems too good to be true, it probably is.
Color changes during the tirzepatide dose titration
As you titrate through dose levels, the color of the solution itself should not change. The 2.5 mg solution looks the same as the 15 mg solution inside the pen. Tirzepatide concentration varies between doses, but the concentration difference does not create a visible color change.
What does change is the pen packaging color, as discussed in the Mounjaro pen color coding section. The solution inside always remains clear to slightly yellow regardless of whether you are at the starting 2.5 mg dose or the maximum 15 mg dose.
For compounded formulations, the same principle applies. If you increase your dose from 5 mg to 10 mg but stay with the same pharmacy and formulation, the vial should look the same. Different concentration, same appearance. If the new vial looks different from the previous one, ask your pharmacy whether they changed their formulation.
Oral tirzepatide color
Oral tirzepatide is available in different forms than the injectable version, and the color expectations differ accordingly. Tablet forms of tirzepatide have their own specific appearance based on the inactive ingredients, coatings, and dyes used in manufacturing.
Liquid oral tirzepatide drops from compounding pharmacies may appear clear to slightly yellow, similar to injectable formulations, though the specific excipients used for oral formulations can sometimes add subtle color variations. Always check with your pharmacy for the expected appearance of your specific oral formulation.
The color rules for oral formulations differ enough from injectable that you should not compare between the two. An oral formulation that looks different from an injectable formulation is not necessarily a problem. They are different products with different excipients.
Impact of additives and compounding on color
Understanding why compounding pharmacies add specific ingredients to tirzepatide helps make sense of the color variations you might encounter. Each additive serves a purpose, and knowing that purpose helps you understand whether the color you see makes sense.
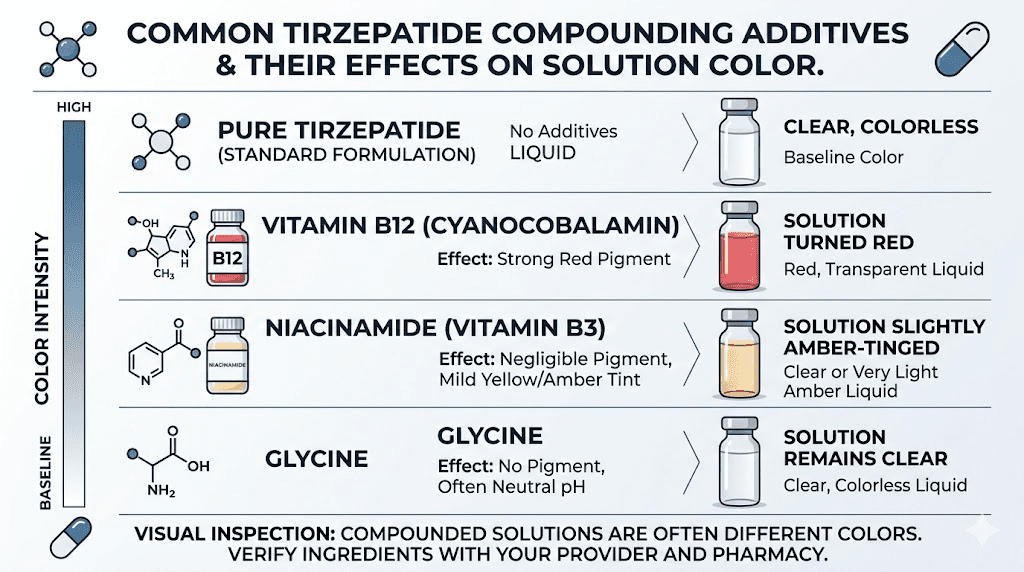
Why B12 is added and what it does to color
Vitamin B12 (cyanocobalamin) is a cobalt-containing molecule. The cobalt ion at its center gives it a distinctive red color. When dissolved in solution, this red color manifests as pink at lower concentrations and deeper red at higher concentrations. The amount of B12 typically added to compounded tirzepatide ranges from 1 to 2 mg per mL, which produces a visible pink coloration.
B12 is added because GLP-1 receptor agonists slow gastric emptying, which can reduce B12 absorption from food over time. Long-term users of tirzepatide and semaglutide may develop B12 deficiency, so including it in the injection bypasses the absorption issue entirely. The B12 also helps address GLP-1 related fatigue in some users.
Why niacinamide is added and its color impact
Niacinamide (vitamin B3) acts as a stabilizer that helps prevent tirzepatide from aggregating, which is the molecular equivalent of the peptide molecules sticking together and forming clumps. Aggregation reduces effectiveness and can cause injection site reactions. Niacinamide helps keep the tirzepatide molecules evenly distributed in solution.
At the concentrations used (typically 2 mg/mL), niacinamide is invisible in solution. It adds no color whatsoever. If your tirzepatide-niacinamide formulation has any color beyond clear to slightly yellow, the color is not coming from the niacinamide.
Why glycine is added and its color impact
Glycine is the simplest amino acid and serves multiple roles in compounded tirzepatide. It acts as a buffering agent to help maintain optimal pH, serves as a cryoprotectant in lyophilized formulations (protecting the peptide during freeze-drying), and provides structural stabilization to the tirzepatide molecule.
Like niacinamide, glycine is colorless in solution. It contributes nothing to the visual appearance of the formulation. The tirzepatide-glycine compound should look identical to pure tirzepatide.
Batch-to-batch variation in compounded formulations
One thing that surprises many users is that compounded tirzepatide can vary slightly in appearance from one batch to the next, even from the same pharmacy. Unlike FDA-approved medications that undergo extremely tight manufacturing controls, compounded formulations have more inherent variability. Slightly different shade of pink between B12 batches, or a marginally different degree of yellow tint, can occur without indicating any quality problem.
The key is distinguishing between minor batch variation and genuine quality issues. Minor: the pink is slightly lighter this time. Concerning: the solution was pink last time and clear this time (or vice versa). If the fundamental character of the solution changes, not just a subtle shade variation, contact your pharmacy before using it.
Storing tirzepatide to maintain proper color
Proper storage is the most effective way to ensure your tirzepatide maintains its expected color throughout its use period. Every color problem described in this guide, from yellowing to cloudiness to particle formation, can be traced back to some form of storage failure.
Refrigerator storage best practices
Refrigeration at 2 to 8 degrees Celsius is the gold standard. But not all spots in your refrigerator are equal. The back of the main compartment tends to be coldest, sometimes dipping below 2 degrees and risking freezing. The door shelves are warmest and experience the most temperature fluctuation every time you open the door. The middle shelves, away from the back wall, typically maintain the most consistent temperature.
Ideal placement: middle shelf, toward the center, in the original packaging. Not in the door. Not touching the back wall. Not next to the freezer vent if your model has one.
For compounded tirzepatide in clear vials, keep the vial in the pharmacy bag or a small opaque container to protect from light exposure. Every time the refrigerator light turns on, any unprotected vials get a dose of light that, over weeks, contributes to oxidative degradation.
Room temperature guidelines
Sometimes refrigeration is not possible. During travel, at work, or during a move, your tirzepatide might need to be at room temperature. The rules differ by product.
FDA-approved Mounjaro and Zepbound: up to 21 days at room temperature below 30 degrees Celsius (86 degrees Fahrenheit). After 21 days at room temperature, discard regardless of expiration date.
Compounded tirzepatide: varies by pharmacy. Some formulations have shorter room temperature stability, as few as 3 to 7 days. Others may be stable for 14 days or more. Always check your pharmacy label or call to confirm. For guidance on compounded storage specifically, see our guide on how long compounded tirzepatide can be out of the fridge.
What NOT to do with tirzepatide storage
Do not freeze it. Do not store it in direct sunlight. Do not leave it in a hot car. Do not store it in the bathroom (humidity and temperature fluctuations). Do not transfer it between containers. Do not pre-draw syringes and store them for later use unless your pharmacy specifically approves this practice. Each of these mistakes can cause color changes and degradation that compromise both effectiveness and safety.
Common tirzepatide color questions people ask
Beyond the formal FAQ section below, there are several scenarios that come up repeatedly in the SeekPeptides community. These are real situations researchers encounter and the practical answers they need.
My tirzepatide was clear when I got it but now looks yellow
A gradual shift from clear to slightly yellow over the use period can be normal, especially for compounded formulations. The critical questions are: how much yellow, and how fast? A subtle tint developing over several weeks of proper storage is usually within acceptable range. A noticeable yellowing within days, or a deep yellow developing at any point, suggests degradation. Compare to a fresh vial from the same pharmacy if possible.
I switched pharmacies and the color changed
Different compounding pharmacies use different formulations, even for the "same" medication. One pharmacy might include B12 while another uses only niacinamide. One might use cyanocobalamin (pink B12) while another uses methylcobalamin (which can be slightly different in hue). Before assuming something is wrong, confirm the complete formulation list with both pharmacies.
One vial from my order looks different from the others
If you received multiple vials and one looks different, do not use the odd one out until you contact your pharmacy. It could be a batch variation (minor difference in shade) or a compounding error (wrong formulation). The pharmacy can verify whether the vials should all look identical or whether some variation is expected.
My tirzepatide has tiny bubbles
Small air bubbles are normal. They are not particles. Air bubbles rise to the surface when the vial sits undisturbed for a few minutes. Particles sink or float but do not disappear. If you are unsure whether you are seeing bubbles or particles, set the vial down on a flat surface and wait two to three minutes. Bubbles will rise and pop at the surface. Particles will remain suspended or settle to the bottom.
Bubbles are especially common immediately after reconstitution or after drawing from the vial. They do not affect the medication quality. For injection technique that minimizes air bubbles, see our guide on injection best practices.
What to do if your tirzepatide looks wrong
You have inspected your tirzepatide and something does not look right. Here is your action plan, step by step.
Step 1: Do not inject it. This is the most important step. When in doubt, skip the dose rather than inject a questionable product. One missed dose is far better than injecting contaminated or degraded medication.
Step 2: Take a photo. Document what you see. Take a clear, well-lit photo of the vial or pen. This gives your pharmacy a reference point when you call them.
Step 3: Note the details. When did you receive this vial? How has it been stored? Is this the first use from this vial or have you been using it? Has it been out of the fridge recently? What formulation did you order?
Step 4: Contact your pharmacy. Call, do not email. Describe what you see and send the photo. The pharmacist can tell you whether the appearance matches what they compounded and whether it is safe to use.
Step 5: Request a replacement if needed. If the pharmacy confirms the product looks abnormal, request a replacement. Reputable pharmacies will replace compromised medication. If they refuse or dismiss your concern, that tells you something about the pharmacy.
Step 6: Report serious concerns. If you suspect contamination, counterfeiting, or a significant compounding error, report it to the FDA MedWatch program. This helps protect other users and holds pharmacies accountable.
For guidance on choosing reliable sources for your medication, our guides on Empower Pharmacy, Lavender Sky, and Peptide Sciences compare specific compounding pharmacies in detail.
Color as part of a broader quality check
Visual inspection is just one part of ensuring your tirzepatide is safe and effective. Color tells you about certain types of degradation and contamination, but it cannot tell you everything. A clear, colorless solution could still have issues that are invisible to the naked eye, like subtle potency loss from improper storage or bacterial contamination below the visible threshold.
A comprehensive quality check includes visual inspection (what we have covered in this guide), verifying the expiration or beyond-use date, confirming proper storage temperature throughout the supply chain, checking that the product matches your prescription, and ensuring the correct concentration and dose for your protocol.
Researchers who get the best results from their tirzepatide protocols tend to be methodical about quality checks. They store correctly. They inspect every time. They track their results so they can detect potency issues early if the medication is not working as expected. And they do not hesitate to discard a questionable vial.
That discipline makes the difference between optimal results and frustrating inconsistency. Color is your first checkpoint. Make it a habit.
For researchers serious about getting the most from their tirzepatide protocol, SeekPeptides provides comprehensive storage guides, quality verification checklists, and a community of experienced researchers who have navigated these exact questions. The platform offers evidence-based protocols for every step of the process, from reconstitution to injection technique to long-term maintenance.
Frequently asked questions
What color should tirzepatide be?
FDA-approved tirzepatide (Mounjaro and Zepbound) should be clear and colorless to slightly yellow. Compounded tirzepatide without B12 should also be clear to slightly yellow. Compounded tirzepatide with B12 should be pink to light red. The solution should always be transparent with no cloudiness or particles.
Why is my tirzepatide pink?
Pink tirzepatide almost always contains vitamin B12 (cyanocobalamin), which has a natural reddish-pink color. This is normal for B12-containing formulations. If you did not order a B12 formulation, contact your pharmacy before using it.
Is yellow tirzepatide safe to use?
A slight yellow tint is normal and listed as acceptable in the FDA prescribing information. However, a deep or darkening yellow can indicate oxidative degradation, especially if the color has changed since you first received the vial. Slightly yellow is fine. Noticeably yellow warrants contacting your pharmacy.
Can I use cloudy tirzepatide?
No. Cloudy tirzepatide should never be used. Cloudiness indicates peptide aggregation, contamination, or improper reconstitution. Discard the vial and contact your pharmacy for a replacement.
Does tirzepatide color change over time?
Properly stored tirzepatide should maintain its color throughout the use period. Gradual yellowing can occur with suboptimal storage conditions and indicates degradation. If you notice progressive color changes, your storage conditions may need improvement. Check our guide on tirzepatide refrigerator storage.
What color is the Mounjaro pen for each dose?
Mounjaro pens use color-coded labels: gray for 2.5 mg, purple for 5 mg, green for 7.5 mg, yellow for 10 mg, blue for 12.5 mg, and orange for 15 mg. The solution inside all pens is the same clear to slightly yellow color regardless of dose. For dose-specific information, see our dosing guides.
Should I worry about air bubbles in my tirzepatide?
No. Small air bubbles are normal and do not indicate a quality problem. Bubbles rise to the surface and disappear within a few minutes. They are different from particles, which remain suspended or sink. If you are unsure, let the vial sit undisturbed for two to three minutes and observe whether the spots disappear.
Does compounded tirzepatide look different from Mounjaro?
It depends on the formulation. Pure compounded tirzepatide looks identical to Mounjaro (clear to slightly yellow). But compounded versions with additives like B12 will look pink or red. Compounded tirzepatide also comes in vials rather than pre-filled pens, so the packaging looks very different even when the solution appears similar.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay clear, your storage stay cold, and your protocols stay consistent.