Feb 6, 2026

You inject tirzepatide on day one. Nothing happens. Day two passes. Still nothing visible. By day seven, you feel something different but cannot quite name it. Maybe less hunger. Maybe just placebo. You wonder how long this actually takes to work. You wonder if your body is responding at all. You wonder if the investment, the injections, the commitment will actually produce the results everyone talks about.
The answer is more specific than most sources admit. Tirzepatide begins working at the cellular level within hours of your first injection, influencing GLP-1 receptor pathways and GIP receptor activity that control gastric emptying, insulin secretion, and hunger signaling. But those molecular changes take time to translate into appetite reduction you can feel, weight changes you can measure, and metabolic improvements that show up in lab work. The gap between cellular activation and visible results creates confusion, frustration, and unnecessary doubt about whether the medication is working at all.
This guide provides the complete timeline backed by clinical research. You will learn when appetite changes typically begin, when weight loss becomes measurable, when peak effects emerge, and what factors accelerate or delay your individual response. You will understand why some people see changes within days while others wait weeks, what early response predicts about long-term outcomes, and how to optimize your protocol for the fastest results your biology allows. No vague reassurances about patience. No generic timelines that ignore individual variation. Just the specific data you need to set accurate expectations and identify if your response falls within normal ranges or signals a problem that needs adjustment.
SeekPeptides members access personalized tirzepatide protocols with week-by-week progress tracking, dose optimization guidance based on individual response patterns, and detailed troubleshooting for delayed or suboptimal results. The platform provides the roadmap most researchers need but cannot find in generic prescribing information or basic online guides.
The pharmacological timeline: what happens from injection to cellular effect
Understanding when tirzepatide works requires separating pharmacokinetics (what your body does to the drug) from pharmacodynamics (what the drug does to your body). These processes operate on different timelines. Tirzepatide reaches peak plasma concentration approximately 8-72 hours after subcutaneous injection, with steady-state concentrations achieved after 4 weeks of once-weekly dosing. The medication has a half-life of approximately 5 days, allowing for sustained receptor activation between weekly doses.
But plasma levels tell only part of the story.
The drug must bind to receptors, trigger intracellular signaling cascades, alter gene expression, and modify cellular behavior before you experience any functional changes. GLP-1 receptor binding occurs rapidly, within hours of administration. GIP receptor activation follows a similar timeline. These dual receptor interactions initiate the downstream effects that eventually produce appetite suppression, improved glycemic control, and weight reduction. The cascade from receptor binding to functional outcome takes days to weeks depending on which effect you are measuring.
Immediate effects: hours to 72 hours post-injection
The first measurable changes occur at the level of gastric motility and insulin secretion. Tirzepatide slows gastric emptying within hours of administration, though you may not consciously notice this effect immediately. The drug enhances glucose-dependent insulin secretion and suppresses inappropriate glucagon release, stabilizing blood sugar fluctuations that drive hunger and energy swings. These metabolic adjustments begin within the first 24-48 hours but produce subtle effects most people do not recognize as drug action.
Some users report early appetite changes within the first 24-72 hours. These early responders typically describe reduced interest in food, earlier satiety during meals, or decreased cravings for high-calorie foods. Others notice nothing at all during this initial window. Both responses are normal. The drug is working at the receptor level regardless of whether you feel subjective appetite changes this early. Individual variation in receptor sensitivity explains much of this difference.
Blood glucose changes appear faster than appetite effects in most users. If you monitor glucose continuously or check fasting and postprandial levels, you may see improvements within the first 48-72 hours, particularly if you started with elevated baseline glucose. Improved glycemic control contributes to appetite regulation and metabolic optimization but does not directly cause weight loss. The weight reduction comes from sustained caloric deficit driven by appetite suppression and potentially increased energy expenditure, effects that take longer to establish.
Week one: initial adaptation and side effect emergence
Most users begin noticing functional changes during week one, though the nature and intensity of these changes vary considerably. The most common first-week experience is reduced hunger between meals and faster satiety during eating. You may find yourself satisfied with smaller portions, less interested in snacking, or able to resist previously irresistible food cues. These changes reflect the drug reaching effective concentrations and producing meaningful receptor activation in your hypothalamus and gastrointestinal tract.
But week one is also when common side effects typically emerge. Nausea, particularly after meals, affects a significant proportion of users during initial dosing. Some experience mild gastrointestinal discomfort, changes in bowel habits, or increased fullness that borders on uncomfortable. These effects reflect the drug's mechanism, slowed gastric emptying and altered gut hormone signaling, and generally diminish as your body adapts. Managing early side effects improves adherence and comfort without compromising effectiveness.
Weight changes during week one are usually minimal. Most users lose 1-3 pounds, primarily water weight and glycogen depletion rather than fat mass. Some lose nothing at all. The absence of dramatic week-one weight loss does not predict poor long-term response. Early weight loss patterns correlate only weakly with 12-week or 6-month outcomes. The drug is establishing metabolic changes that will drive weight reduction over subsequent weeks.
Weeks two through four: appetite suppression solidifies
By week two, most users report consistent appetite suppression. Hunger signals diminish. Cravings for hyperpalatable foods decrease. Portion control becomes easier. These changes reflect sustained receptor activation and metabolic adaptation to the medication. Appetite regulation through GLP-1 and GIP pathways stabilizes, producing reliable daily effects rather than the variable responses some users experience during week one.
This is also when weight loss accelerates for most people. The typical pattern shows 3-6 pounds lost during weeks two through four, depending on starting weight, baseline caloric intake, and adherence to dietary modifications. Users who combine tirzepatide with structured caloric restriction and increased physical activity see faster results during this phase. The drug makes caloric restriction easier by reducing hunger and increasing satiety, but it does not produce weight loss without a caloric deficit.
Week four represents an important milestone. By this point, you should notice clear functional differences in your hunger, satiety, and eating behavior. If you feel no appetite changes by week four, this suggests either inadequate dosing, unusual metabolic factors, or potential issues with medication storage or administration. Troubleshooting delayed response becomes important at this stage to identify correctable factors.
The appetite suppression timeline: when hunger changes become reliable
Clinical trial data shows that most participants experience noticeable appetite reduction within 1-2 weeks of initiating tirzepatide. But noticeable does not mean maximal. The intensity and consistency of appetite suppression increase as you titrate to higher doses and as metabolic adaptation continues. Understanding the progression from initial appetite changes to peak suppression helps set appropriate expectations and guides dose escalation decisions.
First two weeks: variable and establishing effects
The first two weeks produce the most variable appetite responses. Some users report dramatic hunger reduction from day three onward. Others notice subtle changes they would not have recognized without deliberately paying attention. Still others feel no clear difference during this initial period. All three response patterns can lead to successful long-term outcomes.
The variability reflects several factors. Individual receptor expression and sensitivity varies significantly in the population. Some people have naturally high receptor density or enhanced signaling efficiency, producing robust responses to low doses. Others require higher concentrations to achieve comparable effects. Starting doses are intentionally conservative to minimize side effects, which means some users begin below their therapeutic threshold for maximal appetite suppression.
Baseline eating patterns also influence early perception of appetite changes. Users with strong physiological hunger signals and frequent eating episodes notice suppression more clearly than those who eat primarily on schedule regardless of hunger. If you rarely experience strong hunger even before starting tirzepatide, you may not perceive early appetite effects as dramatically as someone who previously dealt with constant food preoccupation. The drug is still working, it has less subjective contrast to create.
Weeks three through eight: reliable daily appetite control
Most users achieve consistent, reliable appetite suppression between weeks three and eight as doses escalate and metabolic adaptation continues. This is the window where eating behavior changes become effortless rather than requiring conscious effort. Food thoughts decrease. Meal planning simplifies. Portion control happens automatically because satiety signals arrive earlier and more forcefully.
The mechanisms underlying this progression involve more than simple receptor activation. Tirzepatide influences leptin sensitivity, reduces ghrelin secretion, and alters central nervous system processing of food reward signals. These changes take time to fully develop. Early dosing initiates the process. Continued exposure and dose escalation strengthen and consolidate the effects. By weeks five through eight, most users describe appetite control as the new normal rather than something they actively notice.
This is also when weight loss typically accelerates. With appetite suppression well-established and eating behavior modified, caloric deficits become consistent rather than intermittent. Users who struggled to maintain restriction on previous diet attempts often find adherence dramatically easier during this phase. The drug is doing what it is designed to do, making caloric restriction tolerable and sustainable.
Week twelve and beyond: peak appetite suppression and maintenance
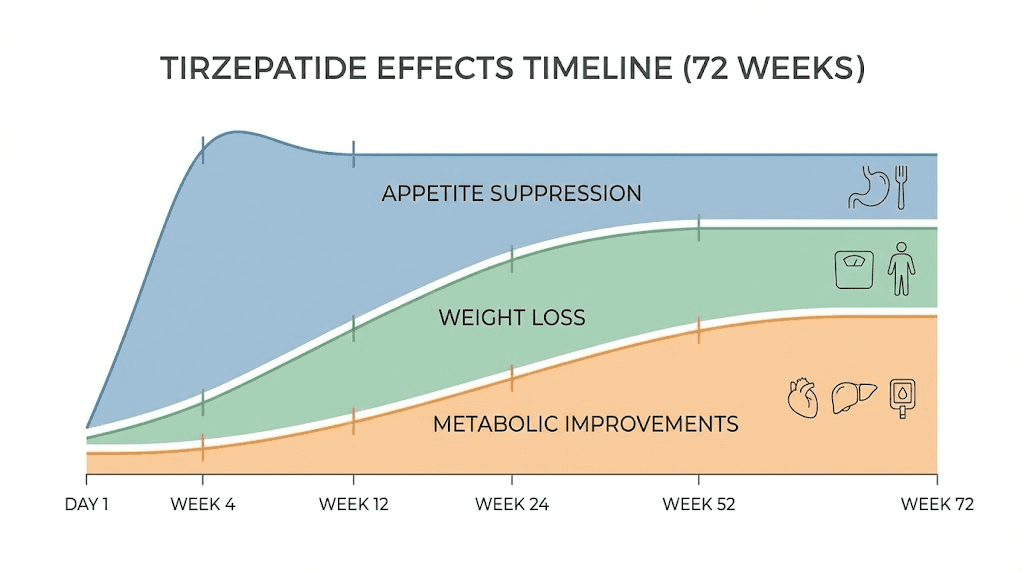
Clinical trials typically assess outcomes at week 12, 24, and longer intervals. The 12-week mark represents an important milestone. By this point, users should be on or approaching their maintenance dose, appetite suppression should be robust and consistent, and weight loss should be clearly established. Participants in the SURMOUNT trials achieved approximately 15-21% total body weight reduction over 72 weeks, with the trajectory of weight loss established by week 12.
Appetite suppression reaches its maximum intensity around the three-month mark for most users. Beyond this point, the effects plateau rather than continuing to increase. Some users report gradual adaptation over many months, where appetite suppression feels slightly less pronounced than during the first three months. This potential tolerance development does not typically compromise weight loss outcomes if you continue treatment and maintain behavioral modifications.
Long-term maintenance requires ongoing medication. Clinical data shows that discontinuing tirzepatide leads to weight regain in most users, with appetite returning toward baseline levels within weeks to months after stopping. The drug provides a tool for sustainable weight management, not a permanent reset of appetite regulation. Long-term GLP-1 use strategies focus on finding the minimum effective dose that maintains results while minimizing side effects and cost.
The weight loss timeline: when changes become measurable and visible
Appetite suppression drives weight loss, but the two timelines do not align perfectly. You may notice appetite changes before any weight reduction appears on the scale. You may see scale changes before physical appearance shifts noticeably. Understanding the progression from metabolic changes to measurable fat loss to visible physical transformation helps set appropriate expectations for each phase.
Weeks one through four: initial losses and metabolic shifts
The first month typically produces 4-8 pounds of weight loss in clinical trial populations, though individual results range from minimal losses to 12+ pounds depending on starting weight, compliance, and individual factors. Much of the early weight reduction comes from water loss, glycogen depletion, and reduced food volume in the digestive tract rather than fat mass reduction. These changes appear on the scale but do not yet reflect significant body composition improvements.
This phase establishes the caloric deficit that will drive fat loss over subsequent weeks. As appetite suppresses and caloric intake drops, your body initially mobilizes easily accessible energy stores, glycogen with its associated water weight. True fat oxidation increases gradually as glycogen stores deplete and metabolic adaptation continues. By week four, most users are losing primarily fat rather than water, though the scale may not distinguish between these components.
Physical appearance changes during this phase are usually subtle. Close friends and family may notice nothing. You might see minor differences in how clothing fits, particularly around the waist where visceral fat reduction begins early. Body composition changes during GLP-1 treatment show that fat loss occurs preferentially from abdominal and visceral depots during early treatment, which improves metabolic health even before visible aesthetic changes appear.
Weeks five through twelve: consistent weekly losses
This window represents the most consistent phase of weight reduction for most users. Clinical data shows average losses of 1-2 pounds per week during this period in participants who respond well to treatment. The cumulative effect by week 12 typically ranges from 10-18 pounds in trial populations, translating to roughly 5-8% total body weight reduction for most starting weights.
But averages obscure substantial individual variation. Some users achieve 12-15% weight reduction by week 12. Others lose only 3-5%. The SURMOUNT-1 trial data shows that approximately 80-85% of participants achieved at least 5% weight loss by week 12, defining them as early responders. The remaining 15-20% were late responders who achieved meaningful weight reduction more slowly but eventually reached comparable outcomes with continued treatment.
Physical changes become noticeable during this phase. Most users report looser-fitting clothes, visible facial changes, and comments from friends or colleagues who notice weight loss. The psychological impact of visible progress improves motivation and reinforces behavioral changes. Tracking progress beyond the scale using measurements, photos, and body composition analysis provides more complete feedback during this phase.
Weeks thirteen through seventy-two: continued reduction and long-term outcomes
Clinical trials extending to 72 weeks show that weight loss continues well beyond the first three months, though the rate of loss typically slows. The SURMOUNT-1 trial demonstrated mean weight reductions of 15-20.9% at 72 weeks depending on dose (10mg or 15mg), with maximal effects observed around months 9-12. Most weight loss occurs during the first 6-9 months, with a maintenance or slight continued reduction phase beyond that point.
Long-term success requires sustained treatment. Discontinuation studies show that stopping tirzepatide leads to substantial weight regain, with approximately 50% or more of lost weight returning within 12 months after cessation. This pattern reflects the fact that tirzepatide treats the symptom (excessive appetite and caloric intake) rather than permanently correcting the underlying regulation of body weight. Maintaining weight loss after GLP-1 peptides requires either continued medication or exceptional adherence to behavioral modifications that most people cannot sustain without pharmacological support.
Physical transformation during the 6-12 month window is typically substantial. Users often describe the experience as life-changing, not just in terms of weight but in mobility, energy, confidence, and health markers. Blood pressure normalizes. Lipid panels improve. Glucose control optimizes. Inflammatory markers decrease.
The benefits extend well beyond aesthetics to genuine health improvements that reduce long-term disease risk.
The metabolic timeline: blood sugar, lipids, blood pressure, and inflammation
Weight loss captures most of the attention in tirzepatide discussions, but the metabolic improvements occur on their own timeline and provide health benefits independent of weight reduction. Understanding when these changes emerge and how they progress helps assess the full therapeutic value of treatment.
Glucose control: hours to weeks
Blood glucose improvements appear fastest among tirzepatide's metabolic effects. Users with type 2 diabetes or prediabetes often see meaningful reductions in fasting glucose within the first week of treatment. Postprandial glucose spikes diminish as gastric emptying slows and insulin secretion improves. Glucose-dependent insulin secretion means the drug enhances insulin release when blood sugar is elevated but does not cause hypoglycemia when glucose levels are normal or low.
Hemoglobin A1c, which reflects average blood glucose over the previous 2-3 months, takes longer to show improvement. Meaningful A1c reductions typically appear by week 12 and continue improving through week 24-40 in clinical trials. The SURPASS trials in patients with type 2 diabetes demonstrated A1c reductions of 1.5-2.5% from baseline, with maximal glycemic effects achieved around 40 weeks of treatment. This represents substantial improvement in diabetes control that reduces long-term complications risk.
For users without diabetes, glucose optimization may be less dramatic but still beneficial. Improved insulin sensitivity, reduced glucose variability, and enhanced metabolic flexibility contribute to long-term metabolic health even if baseline glucose was not clinically elevated. Continuous glucose monitoring during tirzepatide treatment often reveals improved postprandial responses and reduced time in elevated glucose ranges.
Lipid improvements: weeks to months
Lipid profile changes appear more gradually than glucose improvements but can be substantial. Tirzepatide reduces triglycerides, increases HDL cholesterol, and modestly reduces LDL cholesterol in most users. These changes reflect multiple mechanisms including weight loss, improved insulin sensitivity, reduced hepatic lipogenesis, and direct effects on lipoprotein metabolism.
Clinical trials typically assess lipids at week 12, 24, and beyond. Meaningful improvements usually appear by week 12 and continue improving as weight loss progresses. Triglyceride reductions can be dramatic in users with baseline hypertriglyceridemia, sometimes dropping by 30-50% from elevated baselines. HDL increases tend to be modest but consistent. LDL changes are variable, with some users seeing reductions and others experiencing slight increases.
The cardiovascular benefit of these lipid changes extends beyond raw numbers. Tirzepatide reduces atherogenic lipoprotein particles, improves apolipoprotein ratios, and decreases inflammatory markers associated with cardiovascular risk. Cardiovascular benefits of GLP-1 and GIP agonism include outcomes that matter more than isolated lipid values, reductions in heart attack, stroke, and cardiovascular death demonstrated in large outcome trials.
Blood pressure normalization: weeks to months
Users with elevated blood pressure often see gradual reductions as weight loss progresses and metabolic health improves. The effect is not immediate like some pharmaceutical antihypertensives but develops over weeks to months as multiple beneficial factors accumulate. Weight reduction decreases cardiac workload. Improved insulin sensitivity enhances vascular function. Reduced inflammation promotes endothelial health. The cumulative effect produces meaningful blood pressure reductions in many users.
Clinical trial data shows systolic blood pressure reductions of 5-10 mmHg in participants with baseline hypertension, with maximal effects appearing around months 3-6. Some users see enough improvement to reduce or discontinue blood pressure medications under medical supervision. Blood pressure optimization during weight loss requires monitoring and potential medication adjustment as improvements occur.
Inflammatory markers: weeks to months
Chronic low-grade inflammation contributes to metabolic disease, cardiovascular risk, and accelerated aging. Tirzepatide reduces multiple inflammatory markers including C-reactive protein, IL-6, and TNF-alpha. These changes reflect weight loss, improved metabolic health, and potentially direct anti-inflammatory effects of GLP-1 and GIP receptor signaling.
Inflammatory marker improvements typically parallel weight loss and metabolic optimization, appearing gradually over weeks to months rather than acutely. By 6-12 months, many users show substantial reductions in inflammatory biomarkers that correlate with reduced long-term disease risk. Anti-inflammatory effects of metabolic peptides contribute to benefits that extend beyond weight and glucose control.
Factors that accelerate or delay response to tirzepatide
Not everyone follows the average timeline. Some users see dramatic appetite suppression within days and rapid weight loss from week two onward. Others experience minimal changes during the first month and require extended time or dose escalation to achieve meaningful results. Understanding the factors that influence response speed helps optimize your protocol and manage expectations.
Starting dose and escalation protocol
Tirzepatide treatment typically begins with 2.5mg weekly for four weeks, increasing to 5mg for at least four weeks before considering further escalation to 7.5mg, 10mg, 12.5mg, or the maximum 15mg dose. This conservative approach minimizes side effects but means many users spend the first 8-12 weeks on doses below their optimal therapeutic level. Your individual optimal dose depends on factors including body weight, metabolic rate, receptor sensitivity, and therapeutic goals.
Users who respond robustly to low doses may achieve excellent results at 5-7.5mg without needing higher doses. These individuals typically notice appetite suppression within the first week or two and see consistent weight loss on lower doses. Escalating unnecessarily increases side effect risk and cost without improving outcomes. Finding your optimal tirzepatide dose involves balancing effectiveness, tolerability, and practical considerations.
Late responders often need higher doses to achieve effects comparable to early responders at lower doses. If you notice minimal appetite or weight changes on 2.5-5mg, continuing escalation to 7.5-10mg or higher may be necessary. Clinical trial data shows that higher doses produce greater weight loss on average, but the relationship is not linear, doubling the dose does not double the results. Dose-response relationships for GLP-1/GIP agonists show diminishing returns at higher doses, though individual variation means some users need maximal dosing for optimal effects.
Baseline body composition and metabolic health
Starting weight and body composition influence both the rate and magnitude of weight loss. Users with higher baseline BMI tend to lose more absolute weight but similar or lower percentages of total body weight compared to those starting at lower BMI. Metabolic health at baseline also matters. Users with insulin resistance, metabolic syndrome, or type 2 diabetes often see robust metabolic improvements but may experience slower weight loss compared to metabolically healthy individuals at similar weights.
Visceral fat distribution predicts faster early response to tirzepatide than peripheral fat distribution. Abdominal fat is more metabolically active and responds more quickly to caloric deficit and metabolic improvements. Users carrying weight primarily in the abdomen often report faster visible changes than those with more peripheral (hip, thigh, arm) fat distribution. Body fat distribution and peptide response explains some of the visible transformation differences between users losing similar amounts of weight.
Dietary compliance and protein intake
Tirzepatide makes caloric restriction easier but does not eliminate the need for appropriate nutrition. Users who maintain adequate protein intake during weight loss preserve more lean mass and often achieve better body composition outcomes than those who drastically reduce protein as overall calories drop. Target protein intake typically ranges from 0.7-1.0 grams per pound of ideal body weight, adjusted based on activity level and muscle preservation goals.
Some users inadvertently reduce caloric intake so aggressively that metabolic adaptation slows weight loss. Very low calorie intakes (below 800-1000 calories daily) trigger adaptive responses that reduce metabolic rate and slow fat loss despite continued caloric deficit. Optimizing caloric intake during GLP-1 treatment involves finding the deficit that maximizes fat loss without triggering excessive metabolic adaptation.
Physical activity and resistance training
Exercise does not drive weight loss as powerfully as caloric restriction, but it influences body composition, metabolic health, and long-term weight maintenance. Users who incorporate resistance training during tirzepatide treatment preserve significantly more lean mass than those relying on diet changes alone. Maintaining or building muscle during weight loss improves metabolic rate, physical function, and aesthetic outcomes.
Cardiovascular exercise contributes modest additional caloric expenditure and provides cardiovascular health benefits independent of weight loss. Exercise programming during GLP-1 therapy should balance energy expenditure with recovery capacity and avoid excessive volumes that increase hunger or compromise adherence.
Sleep quality and stress management
Chronic sleep deprivation and elevated stress impair weight loss through multiple mechanisms including increased cortisol, altered hunger hormones, reduced insulin sensitivity, and impaired fat oxidation. Users who prioritize sleep optimization during weight loss typically see faster results and better adherence than those dealing with chronic sleep restriction.
High stress levels increase cortisol, which promotes visceral fat deposition and interferes with fat mobilization. Stress management during metabolic optimization improves both the rate of weight loss and overall well-being during treatment. Techniques including meditation, yoga, breathing exercises, and time management reduce physiological stress and support optimal metabolic function.
Medication interactions and concurrent treatments
Certain medications influence tirzepatide effectiveness or weight loss outcomes. Insulin and sulfonylureas increase hypoglycemia risk and may require dose reduction as glucose control improves. Beta-blockers and some antidepressants can slow weight loss or promote weight gain, partially offsetting tirzepatide's effects. Hormonal contraceptives and hormone replacement therapy influence fluid retention and body composition changes.
Users taking multiple medications should work with prescribers to optimize the full medication regimen rather than relying solely on tirzepatide.
Sometimes adjusting or discontinuing weight-promoting medications produces benefits as significant as starting weight loss medications. Medication review during weight loss treatment identifies opportunities to remove barriers to progress.
Early response predicts long-term outcomes: what week 12 results mean
Clinical research has identified that response by week 12 strongly predicts long-term weight loss outcomes. This finding has important implications for managing expectations, guiding dose adjustments, and identifying users who may need alternative or combination approaches.
The early responder advantage
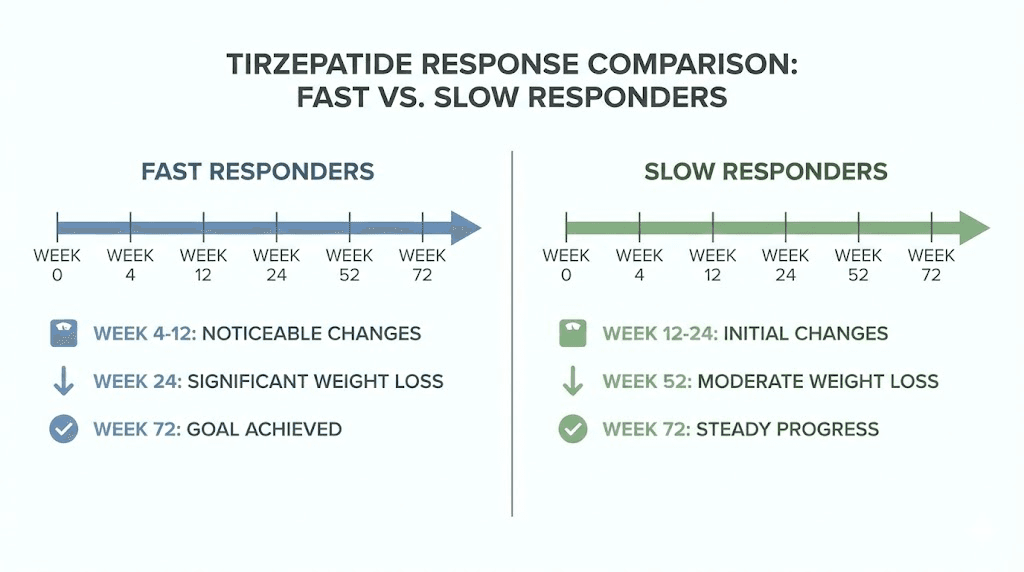
The SURMOUNT-1 trial post-hoc analysis categorized participants as early responders (achieving at least 5% weight reduction by week 12) or late responders (less than 5% weight reduction by week 12). Approximately 80-85% of participants qualified as early responders. These individuals ultimately achieved greater total weight loss at 72 weeks, with mean reductions exceeding 20% in the highest dose groups.
Early responders typically notice appetite suppression within the first two weeks, see consistent weekly weight loss by week four, and achieve the 5% threshold by weeks 10-12. Their trajectory establishes early and continues on a predictable path. The biological mechanisms underlying early response likely include favorable receptor genetics, optimal baseline metabolic health, and strong adherence to protocol recommendations.
Late responders can still achieve excellent outcomes
Late responders, comprising approximately 15-20% of participants, achieve less than 5% weight reduction by week 12 but can still reach clinically significant weight loss with continued treatment. The average time to reach 5% weight reduction in late responders was approximately 25 weeks. Many late responders eventually achieved 15-20% weight reduction by 72 weeks, demonstrating that delayed initial response does not preclude successful long-term outcomes.
Late response patterns suggest several possible explanations. Some users require higher doses to achieve therapeutic effects, meaning they spend more time below their optimal dose during the escalation phase. Others may have metabolic or behavioral factors that slow initial response but do not prevent eventual success. Identifying and optimizing late responder protocols involves systematic troubleshooting to identify correctable barriers.
Using week 12 data to guide decisions
Assessing your response at week 12 provides valuable information for protocol optimization. If you have achieved 5% or greater weight reduction and notice consistent appetite suppression, your current dose and protocol are working well. Continuing the current approach while potentially escalating to the next dose tier if you tolerate the current dose well represents a reasonable strategy.
If you have lost less than 5% by week 12 despite good adherence, several options deserve consideration. Escalating to a higher dose tier often produces better results in late responders. Reviewing and optimizing dietary protein intake, meal timing, sleep quality, stress management, and exercise protocols can remove barriers to response. Checking thyroid function, reviewing medications that may impair weight loss, and assessing for conditions like sleep apnea that interfere with metabolic optimization helps identify treatable factors.
In rare cases, week 12 assessment may reveal minimal response (less than 2-3% weight loss) despite maximal doses and optimal adherence. These individuals may benefit from combination approaches incorporating additional medications or alternative treatment strategies. Genetic variation in drug metabolism, receptor expression, or metabolic pathways may explain rare cases of poor response to otherwise highly effective medications.
When tirzepatide stops working: adaptation, tolerance, and solutions
Some users report that tirzepatide becomes less effective over time, with appetite suppression diminishing or weight loss plateauing despite continued treatment. Understanding the mechanisms underlying these experiences and evidence-based solutions helps maintain long-term success.
True tolerance versus weight loss plateau
Distinguishing between true pharmacological tolerance and expected weight loss plateau is critical. All weight loss eventually plateaus as metabolic rate adapts to lower body weight. A 200-pound person who loses 40 pounds now maintains homeostasis at 160 pounds, where the caloric intake that produced weight loss now matches caloric expenditure at the new weight. This is not tolerance to the medication, it is reaching a new equilibrium.
True tolerance would involve the drug losing effectiveness at suppressing appetite and regulating glucose despite stable dosing and continued use. Limited evidence suggests some users experience gradual adaptation where appetite suppression feels less pronounced after many months of treatment. However, clinical trial data extending to 72 weeks shows sustained weight loss without obvious tolerance development in most participants. Mechanisms of GLP-1 tolerance remain incompletely understood and may affect only a subset of users.
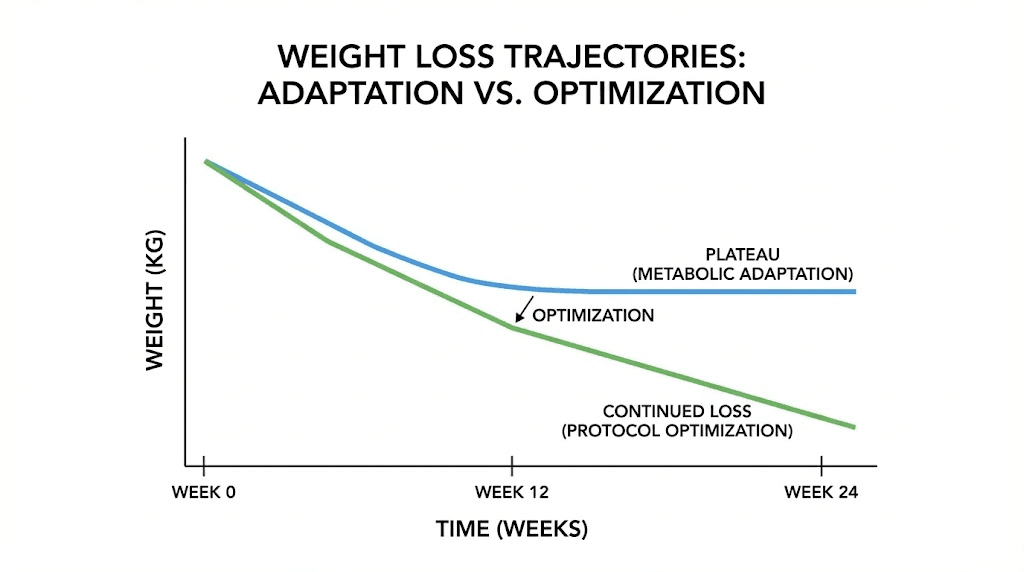
Metabolic adaptation and the weight loss plateau
As you lose weight, your body reduces metabolic rate to match the lower energy requirements of reduced body mass. This adaptive response is normal physiology, not a problem to be fixed. A 160-pound person burns fewer calories at rest and during activity than a 200-pound person. To continue losing weight, caloric intake must decrease further or activity must increase to re-establish a deficit.
Many users interpret this natural plateau as medication failure. The drug is still working, appetite suppression continues, it is simply maintaining the current weight rather than producing continued loss. Breaking through a plateau requires re-establishing a caloric deficit through modest further calorie reduction, increased activity, or both. Strategies for breaking weight loss plateaus focus on sustainable adjustments that restart progress without triggering excessive metabolic adaptation.
Increasing dose to overcome plateaus
Escalating to the next dose tier can restart weight loss in some users who plateau at lower doses. The higher dose may strengthen appetite suppression, allowing for a larger caloric deficit without increased hunger or cravings. However, dose escalation has limits. Once you reach the maximum dose (15mg weekly), further increases are not recommended and unlikely to provide benefit.
Some users experiment with off-label higher doses or more frequent dosing, but evidence supporting these approaches is limited. Side effects typically increase with higher doses, and the risk-benefit balance becomes less favorable. Dose optimization at plateaus involves systematic assessment of whether increasing dose is likely to help based on your current dose, tolerance, and response pattern.
Diet breaks and reverse dieting
Extended caloric restriction triggers metabolic adaptations including reduced metabolic rate, decreased spontaneous activity, increased hunger hormones, and changes in thyroid and reproductive hormones. Temporary diet breaks, periods of eating at maintenance calories, can partially reverse these adaptations and restore metabolic function. After a 7-14 day diet break, resuming restriction often produces renewed weight loss.
Implementing diet breaks during GLP-1 treatment requires careful planning to avoid excessive overeating during the break. The goal is maintenance calories, not unrestricted eating. Many users find that tirzepatide helps maintain control during diet breaks, preventing the overeating that sometimes sabotages these periods. Reverse dieting, gradually increasing calories to rebuild metabolic rate before attempting further weight loss, represents another strategy for users who have been in aggressive deficits for extended periods.
Addressing behavioral factors
Caloric intake often gradually increases during treatment as users become accustomed to the appetite suppression and relax dietary vigilance. Small incremental increases in portion sizes, snacking frequency, or calorie-dense food choices can eliminate the caloric deficit without conscious awareness. Periodic tracking of actual intake helps identify whether behavioral factors contribute to plateaus.
Psychological and emotional factors also influence eating behavior. Stress eating, boredom eating, social eating, and emotional eating can persist despite appetite suppression if these behaviors are driven by factors other than physiological hunger. Addressing non-hunger eating during GLP-1 treatment often requires behavioral strategies beyond pharmaceutical intervention.
Combining tirzepatide with other peptides and medications for enhanced results
Some users explore combination approaches to accelerate results, overcome plateaus, or address multiple health goals simultaneously. Understanding evidence-based combinations and avoiding problematic interactions optimizes safety and effectiveness.
Tirzepatide plus metformin
Metformin, a first-line diabetes medication, is commonly combined with tirzepatide for enhanced metabolic benefits. Metformin improves insulin sensitivity, reduces hepatic glucose production, and may contribute modest weight loss independent of other effects. The combination provides complementary mechanisms, GLP-1/GIP agonism plus improved insulin action producing better glycemic control than either medication alone.
Metformin may also provide benefits for users without diabetes. Some research suggests metformin activates AMPK pathways involved in cellular energy metabolism, potentially supporting longevity and metabolic health beyond glucose control. The combination is generally well-tolerated, though gastrointestinal side effects may be additive in sensitive individuals.
Tirzepatide plus growth hormone peptides
Growth hormone secretagogues like ipamorelin, CJC-1295, and tesamorelin promote growth hormone release, potentially supporting lean mass preservation during weight loss. The theoretical benefit is maintaining or building muscle while tirzepatide drives fat loss, optimizing body composition changes.
Limited evidence directly evaluates this combination, and individual responses vary. Some users report improved energy, better recovery from training, and enhanced body composition outcomes when combining growth hormone peptides with tirzepatide. Others notice minimal additive benefit. Growth hormone peptides during weight loss may be most beneficial for users prioritizing muscle preservation and those with evidence of growth hormone deficiency.
Tirzepatide plus thyroid optimization
Subclinical hypothyroidism slows weight loss and contributes to metabolic dysfunction. Users with borderline low thyroid function, elevated TSH, low free T3, or symptoms of hypothyroidism may benefit from thyroid hormone optimization alongside tirzepatide. Even users with technically normal thyroid labs sometimes see improved weight loss with thyroid medication, though this approach requires careful medical supervision to avoid hyperthyroidism.
Monitoring thyroid function during significant weight loss is important because metabolic changes can alter thyroid hormone needs. Some users require dose adjustments as weight decreases and metabolic rate shifts. Thyroid monitoring during metabolic interventions ensures optimal function throughout treatment.
Combinations to avoid
Combining tirzepatide with other GLP-1 agonists (semaglutide, liraglutide) provides no additional benefit and increases side effect risk. The medications work through overlapping mechanisms and are not intended for concurrent use. Similarly, combining tirzepatide with other appetite-suppressing medications like phentermine may increase cardiovascular risk and is not routinely recommended without specific medical justification.
Insulin and sulfonylureas require careful dose adjustment when combined with tirzepatide to avoid hypoglycemia. SGLT2 inhibitors can be combined but require monitoring for additive effects and potential volume depletion. Medication interactions with GLP-1 agonists should be reviewed with prescribers before starting combination approaches.
Monitoring progress: metrics beyond the scale
Weight change captures only one dimension of tirzepatide's effects. Comprehensive progress tracking incorporates multiple markers that provide fuller understanding of health improvements and help maintain motivation during periods when scale weight plateaus.
Body composition analysis
DEXA scans, bioelectrical impedance, or other body composition assessment methods distinguish between fat mass, lean mass, and bone density changes. Some users lose fat while maintaining or gaining muscle, producing modest scale changes that underestimate true progress. Tracking body composition monthly or quarterly provides data that the scale cannot.
Particularly during later stages of weight loss, body composition changes often continue even when total weight plateaus. Recomposition, simultaneous fat loss and muscle gain, produces dramatic improvements in physique and metabolic health without corresponding scale changes. Users focused exclusively on weight may miss these positive developments.
Measurements and photos
Waist circumference, hip circumference, neck circumference, and other measurements track fat loss patterns and provide feedback independent of weight. Visceral fat reduction appears in waist measurements before it shows on the scale. Progress photos taken monthly under consistent lighting and positioning reveal changes that you may not notice in daily mirror checks. Comprehensive progress tracking methods combine multiple data points for complete assessment.
Metabolic health markers
Lab work including fasting glucose, hemoglobin A1c, lipid panel, liver enzymes, inflammatory markers, and hormone levels provides objective data about metabolic health improvements. Many users see dramatic lab improvements that correlate poorly with weight changes. A 10-pound weight loss might produce A1c reduction of 1.5%, normalize triglycerides, and drop C-reactive protein by 70%. These changes matter more for long-term health than the raw weight number.
Metabolic health markers to track should be assessed at baseline and periodically during treatment. Improvements in these markers demonstrate that treatment is working even during frustrating plateaus when weight is not budging. Insurance typically covers these labs when medically indicated, making comprehensive monitoring accessible for most users.
Functional capacity and quality of life
Daily function improves dramatically with weight loss and metabolic optimization. Energy increases. Joint pain decreases. Sleep quality improves. Mood stabilizes. Exercise capacity expands. These functional improvements may matter more for quality of life than achieving specific weight goals. Quality of life improvements during weight loss should be systematically assessed and celebrated as important outcomes.
Structured questionnaires or simple journaling about energy, sleep, mood, pain, exercise performance, and daily activities provides qualitative data that complements quantitative measurements. Many users report that functional improvements appear before major weight changes, providing early positive feedback that supports adherence.
Frequently asked questions
How quickly will I notice appetite suppression on tirzepatide?
Most users notice some appetite reduction within the first 1-2 weeks of treatment, though the intensity varies considerably. Early responders may feel dramatic hunger suppression within 3-5 days. Others notice subtle changes they would not recognize without deliberately paying attention. By week 4, appetite suppression should be clearly noticeable if you are responding to treatment. If you feel no appetite changes by week 4-6, consider whether dose escalation, protocol optimization, or troubleshooting is needed. Optimizing early tirzepatide response addresses factors that influence initial effectiveness.
When should I see weight loss results with tirzepatide?
Measurable weight loss typically begins by weeks 2-4 and becomes consistently noticeable by weeks 4-8. Clinical trial data shows average losses of 5-8% total body weight by week 12 in early responders, though individual results range from minimal losses to 12-15% depending on starting weight, compliance, and metabolic factors. If you have lost less than 3-5% by week 12 despite good adherence, reviewing dose adequacy, dietary protein intake, sleep quality, and potential medication interactions helps identify opportunities for optimization. Expected weight loss timelines vary based on individual factors but follow predictable patterns in most users.
What if tirzepatide is not working for me?
If you notice minimal appetite or weight changes after 8-12 weeks on tirzepatide, systematic troubleshooting helps identify the cause. First, verify proper medication storage and administration technique. Tirzepatide requires refrigeration before use, and improper storage degrades potency. Proper tirzepatide storage and injection technique ensures you receive the full dose. Second, assess whether your current dose is adequate. Many users need 7.5-10mg or higher to achieve optimal effects. Third, review dietary patterns, sleep quality, stress levels, and medications that may interfere with weight loss. Comprehensive troubleshooting for delayed response systematically addresses common barriers to success.
Can I speed up tirzepatide results?
The pharmacological timeline follows biological processes that cannot be dramatically accelerated, but you can optimize factors that influence response speed. Adequate protein intake (0.7-1.0g per pound ideal body weight) supports lean mass preservation and metabolic function. Resistance training maintains muscle during caloric deficit, preventing metabolic slowdown. Sleep optimization (7-9 hours nightly) improves hormone balance and fat loss. Stress management reduces cortisol-driven fat storage. These factors do not dramatically accelerate results but optimize the timeline your biology allows.
Do I need to stay on tirzepatide forever?
Clinical data shows that discontinuing tirzepatide typically leads to weight regain, with approximately 50% or more of lost weight returning within 12 months after stopping. This pattern reflects the fact that the medication treats symptoms (excessive appetite and caloric intake) rather than permanently correcting underlying body weight regulation. Most users require ongoing treatment to maintain results. Some successfully transition to lower maintenance doses that preserve results while reducing cost and side effects. Long-term maintenance strategies help identify the minimum effective approach for sustained success. A subset of users successfully discontinue medication after establishing stable behavioral and metabolic changes, but this outcome is less common than continued treatment requirement.
What happens if I miss a dose of tirzepatide?
If you miss a dose and remember within 4 days (96 hours), administer the missed dose as soon as possible and return to your regular weekly schedule. If more than 4 days have passed, skip the missed dose and resume your regular schedule with the next planned dose. Do not double dose to make up for missed injections. Missing occasional doses typically does not dramatically compromise results, though consistent adherence produces optimal outcomes. Managing missed doses of weight loss peptides provides guidance for maintaining progress despite occasional schedule disruptions.
External resources
Knowing exactly when tirzepatide works removes the uncertainty that undermines adherence. The drug begins affecting your metabolism within hours, suppresses appetite within days to weeks, and produces measurable weight loss within weeks to months.
Peak effects emerge around months 3-6 and sustain with continued treatment. Individual timelines vary based on dose, metabolic health, adherence, and optimization of supporting factors including diet, sleep, stress management, and exercise.
For researchers seeking structured guidance through every phase of tirzepatide treatment, SeekPeptides provides personalized protocols that account for individual response patterns, week-by-week progress tracking tools, detailed troubleshooting for delayed or suboptimal results, and comprehensive support that extends well beyond generic prescribing information.
In case I don't see you, good afternoon, good evening, and good night. Join here.