Feb 28, 2026

Before you panic about that red bump forming around your injection site, take a breath. That welt, that itch, that warm circle of pink skin spreading outward from where the needle went in, it happens. It happens more often than most guides admit. And in the vast majority of cases, it resolves on its own within 24 to 48 hours without any intervention at all.
But not always.
Some reactions linger. Some grow. Some develop into firm nodules that sit beneath the skin for days. A small number signal something that demands immediate medical attention, and the difference between a normal reaction and a dangerous one often comes down to knowing exactly what to look for, exactly when to worry, and exactly what to do. That is what this guide covers. Every type of GLP-1 injection site reaction documented in clinical trials, every cause researchers have identified, every treatment option that actually works, and the clear warning signs that mean you need to call your doctor today. Whether you are using semaglutide, tirzepatide, retatrutide, or any other GLP-1 receptor agonist, the principles remain the same. SeekPeptides has compiled the research, the clinical data, and the practical guidance into one comprehensive resource so you do not have to piece it together from scattered forum posts and conflicting medical pages.
What a GLP-1 injection site reaction actually is
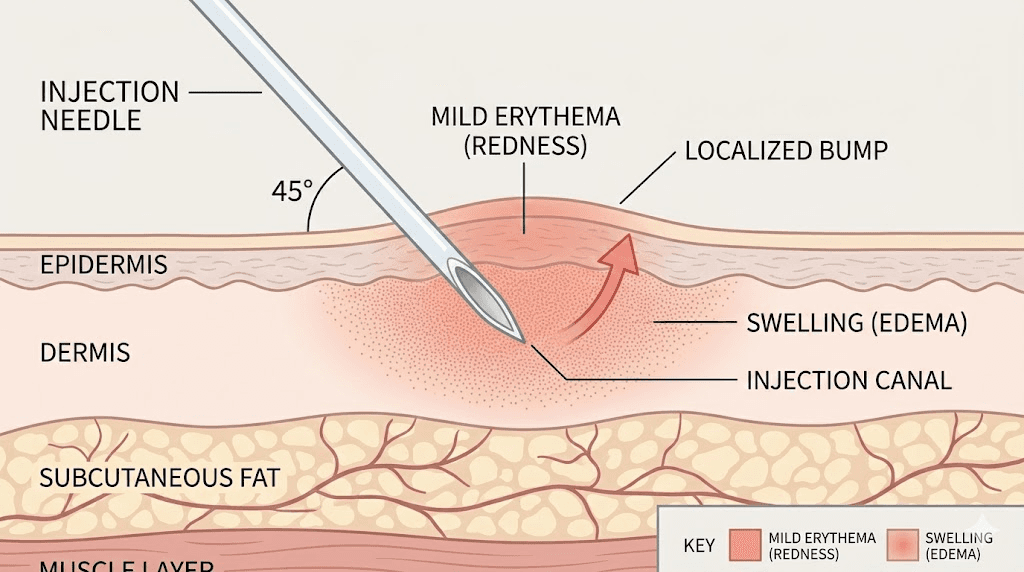
An injection site reaction is a localized inflammatory response that occurs in the tissue where a subcutaneous injection was administered. The key word is localized. These reactions happen at the injection site itself, not systemically throughout the body. They represent the immune system responding to the physical trauma of needle penetration, the chemical properties of the medication, or both.
The mechanism is straightforward. When a needle pierces the skin and deposits medication into subcutaneous fat, several things happen simultaneously. Tissue gets displaced. Blood vessels may rupture. Foreign substances enter a space the immune system monitors closely. Mast cells in the surrounding tissue detect these changes and begin releasing histamine and other inflammatory mediators. Blood flow to the area increases. White blood cells arrive. The area becomes red, warm, and sometimes swollen.
This is not a flaw in the medication. It is biology working exactly as designed.
Every injectable medication carries some risk of injection site reactions. Insulin users have dealt with them for decades. Peptide injections of all types produce them. The difference with GLP-1 receptor agonists is the specific reaction patterns they produce, which vary based on the medication formulation, concentration, injection technique, and individual immune response.
Clinical trials across all commercially available GLP-1 receptor agonists, including semaglutide, tirzepatide, liraglutide, exenatide, dulaglutide, and lixisenatide, have documented injection site reactions as a recognized adverse effect. The reported rates range from 1.4% for semaglutide to 16% for exenatide extended-release, depending on the specific formulation and study population. But these clinical trial numbers likely undercount the true frequency because many mild reactions go unreported. Real-world estimates suggest approximately 50% of GLP-1 users experience some form of localized reaction during their treatment course.
Understanding the difference between expected reactions and concerning reactions is the first step toward managing them effectively. Most reactions fall firmly in the expected category. A handful do not.
The six types of injection site reactions
Not all injection site reactions look the same. Not all feel the same. And they certainly do not all require the same response. The research identifies six distinct types, each with its own presentation timeline, underlying mechanism, and treatment approach.
Type 1: Erythema (redness)
The most common reaction by far. A circular area of pink or red skin appears at the injection site within minutes to hours after administration. The affected area typically measures 1 to 3 centimeters in diameter and feels warm to the touch. No raised borders. No significant pain. Just color change and warmth.
Erythema occurs because histamine release causes local blood vessels to dilate, increasing blood flow to the area. This is the same mechanism behind any minor skin irritation, from a mosquito bite to a scratch. In the context of GLP-1 injections, it indicates the tissue is responding to the needle penetration and medication deposit, nothing more.
Timeline: appears within 1 to 6 hours, peaks at 12 to 24 hours, resolves completely within 24 to 48 hours. If erythema persists beyond 72 hours or expands beyond 5 centimeters, it warrants medical evaluation.
Type 2: Pruritus (itching)
Itching at the injection site often accompanies erythema but can also occur independently. The sensation ranges from mild tingling to intense, persistent itching that makes it difficult to ignore. Some users describe it as a crawling sensation beneath the skin surface.
The itch comes from histamine activating sensory nerve endings in the dermis. People who experience injection site itching with one GLP-1 medication often experience it with others as well, suggesting individual histamine sensitivity plays a significant role. Scratching provides temporary relief but can worsen the reaction by causing additional tissue damage and histamine release.
Timeline: onset within 30 minutes to 4 hours, duration of 24 to 72 hours. Itching that persists beyond one week or spreads significantly beyond the injection site suggests a possible allergic component rather than a simple irritant response.
Type 3: Induration (hardening)
A firm, sometimes tender area develops beneath the skin at the injection site. The texture feels distinctly different from surrounding tissue, like a small marble or rubbery disc embedded under the skin. Induration indicates a more significant inflammatory response than simple erythema, with cellular infiltration creating a palpable mass.
This reaction occurs more frequently with higher concentration formulations and with medications that have longer local residence times. Compounded semaglutide preparations, which may have different concentration profiles than brand-name formulations, sometimes produce more pronounced induration. The response typically reflects the body attempting to wall off and process the medication depot.
Timeline: develops within 12 to 48 hours, may persist for 3 to 10 days. Induration lasting beyond two weeks, especially if accompanied by increasing pain or warmth, requires medical evaluation to rule out abscess formation.
Type 4: Injection site nodules
Nodules represent a step beyond simple induration. These are discrete, palpable lumps that form at the injection site, often described as quarter-sized, hard, erythematous, and pruritic. The first published case report of semaglutide-associated injection site nodules documented a 75-year-old patient who developed these nodules within minutes of injection at the 1 mg dose, with each nodule taking 2 to 4 days to resolve completely.
When the semaglutide dose was reduced from 1 mg to 0.5 mg, the same patient found that nodules subsided within 24 hours instead of 2 to 4 days, suggesting a dose-dependent relationship. Switching to a different GLP-1 receptor agonist eliminated the nodules entirely over a five-month follow-up period.
Injection site nodules have been reported most frequently with exenatide extended-release, where clinical trials documented them in 17.1% of participants. For other GLP-1 receptor agonists including semaglutide and tirzepatide, the risk remains significantly lower.
Type 5: Bruising
A discoloration ranging from purple to green to yellow that develops when the needle nicks a small blood vessel during injection. Unlike the other reaction types, bruising is purely mechanical, not immunological. The blood that leaks from the damaged vessel spreads through surrounding tissue and changes color as hemoglobin breaks down over the following days.
Bruising occurs more frequently in certain injection locations, with the thigh injection site tending to bruise more readily than the abdomen in many users. People taking blood thinners, aspirin, or fish oil supplements experience larger and longer-lasting bruises. The bruise itself is harmless but can make subsequent injections in that area more uncomfortable until it resolves.
Timeline: appears immediately to 24 hours after injection, changes color over 5 to 14 days, resolves completely without treatment.
Type 6: Lipodystrophy
The most concerning long-term reaction. Lipodystrophy involves actual changes to the fat tissue at frequently used injection sites. It presents in two forms. Lipohypertrophy is the accumulation of fat tissue into a rubbery lump, caused by repeated insulin or peptide injections in the same location over weeks to months. Lipoatrophy is the opposite, a depression or loss of fat tissue at the injection site, which represents an immune-mediated destruction of adipocytes.
Lipodystrophy has been extensively documented in insulin therapy, where prevalence ranges from 20% to 70% depending on injection technique and rotation habits. While less studied in GLP-1 populations specifically, the mechanism remains identical. Repeated subcutaneous injections in the same spot cause cumulative tissue changes. Poor GLP-1 injection technique and inadequate site rotation are the primary risk factors.
Timeline: develops gradually over weeks to months of repeated injection in the same area. Partially reversible if the area is rested for an extended period, though severe cases may cause permanent tissue changes.
What causes GLP-1 injection site reactions
Knowing the types matters. But knowing the causes matters more, because causes are what you can actually control.
Five distinct mechanisms drive injection site reactions with GLP-1 receptor agonists, and most reactions involve more than one mechanism operating simultaneously. Understanding which mechanism dominates your particular reaction pattern determines which treatment and prevention strategy will work best.
Mechanical tissue trauma
Every injection creates a small wound channel through the dermis and into subcutaneous fat. The needle displaces tissue, ruptures capillaries, and creates a deposit space for the medication. This physical disruption triggers the inflammatory cascade regardless of what medication is being injected.
Needle gauge and length affect the severity of mechanical trauma. Thinner needles create less tissue disruption. Shorter needles reduce the depth of penetration. The angle of insertion matters too. A 90-degree angle, which is standard for subcutaneous injection with peptide injection pens and syringes, creates the most efficient wound channel. Angled insertions can drag through more tissue and create larger disruption zones.
If you have accidentally injected into muscle rather than subcutaneous fat, the reaction is typically more pronounced because muscle tissue has greater blood supply and immune cell density than fat. The medication absorbs faster and the inflammatory response escalates.
Chemical irritation from the medication
GLP-1 receptor agonists are not pH-neutral. The formulation chemistry, including buffers, preservatives, and stabilizers, can irritate surrounding tissue upon contact. This is why some medications burn on injection while others do not. The chemical composition interacts with local tissue, and if the pH deviates significantly from physiological norms, it triggers pain receptors and inflammatory mediators.
Temperature amplifies chemical irritation significantly. Injecting medication straight from the refrigerator causes vasoconstriction and slows the dispersion of the medication, keeping the concentrated solution in contact with a smaller tissue area for longer. This is why virtually every injection guide recommends allowing the medication to reach room temperature before administration. Fifteen to thirty minutes at room temperature makes a measurable difference in injection comfort and reaction severity.
Compounded formulations may use different excipient profiles than brand-name products. These differences in preservatives, concentrations, and buffers can produce different reaction patterns. A person who tolerates brand-name semaglutide without any site reactions might experience reactions with a compounded version, or vice versa. The active ingredient is the same, but the supporting ingredients affect local tissue tolerability.
Immune-mediated responses
The immune system can develop antibodies against GLP-1 receptor agonists, particularly those based on non-human peptide sequences. Exenatide, derived from the venom of the Gila monster lizard, is the most immunogenic GLP-1 agonist. Research shows that injection site reactions occurred in 14.2% of antibody-positive patients compared to only 3.1% of antibody-negative patients treated with exenatide extended-release.
This antibody-driven mechanism explains why some users develop injection site reactions only after weeks or months of treatment, long after the initial adaptation period should have passed. The immune system needs time to recognize the medication as foreign, produce antibodies, and mount a localized response upon subsequent exposures.
Semaglutide and tirzepatide, engineered with human peptide backbones, produce significantly fewer antibody-mediated reactions than exenatide. But they are not immune to this mechanism entirely. Individual variation in immune response means some users will develop antibodies against any peptide-based medication, regardless of how closely it matches human sequences.
Interestingly, research has revealed a paradoxical relationship between GLP-1 receptors and mast cells. Mast cells appear to have GLP-1 receptors on their surface, and when GLP-1 or its analogues bind to those receptors, they may actually send a calming signal that reduces mast cell degranulation. This partly explains why side effects including injection site reactions often diminish over time rather than worsen.
Alcohol swab reactions
An overlooked cause that accounts for a surprising number of injection site reactions. Cleaning the injection site with an alcohol swab is standard practice. But if the alcohol has not fully evaporated before the needle enters the skin, it gets carried into the subcutaneous tissue along with the medication. Alcohol in subcutaneous fat produces a burning, stinging sensation and can trigger a localized inflammatory response that mimics a medication reaction.
The solution is simple but frequently ignored. After cleaning the injection site with an alcohol swab, wait 30 to 60 seconds for complete evaporation before inserting the needle. The skin should feel completely dry to the touch. Many people rush this step, and the result is a reaction they attribute to the medication itself rather than the alcohol contamination.
Formulation-specific factors
Different GLP-1 formulations use different delivery technologies that produce unique reaction patterns. Exenatide extended-release uses biodegradable polymer microspheres that create a slow-release depot under the skin. These microspheres themselves trigger a foreign body inflammatory response independent of the medication, which is why exenatide extended-release has the highest injection site reaction rate of any GLP-1 agonist at 16%.
The concentration of the solution also matters. Higher concentration formulations deposit more active ingredient in a smaller tissue volume, creating a more concentrated local effect. This is one reason why tirzepatide injection site reactions can vary between different dose levels, even when the injection volume changes to accommodate the dose increase.
For researchers working with reconstituted semaglutide or reconstituted tirzepatide, the concentration after reconstitution directly affects injection site tolerability. Using too little bacteriostatic water creates a higher concentration solution that is more likely to produce local irritation than a properly diluted preparation.
How reaction rates differ between GLP-1 medications
Not all GLP-1 receptor agonists produce injection site reactions at the same rate. The differences are significant enough that reaction history with one medication should inform decisions about switching to another.
Clinical trial data across phase II and phase III studies reveals a clear pattern. Long-acting formulations produce more injection site reactions than short-acting ones. And formulations using depot technologies produce more reactions than simple solution injections.
GLP-1 Medication | Formulation Type | Injection Site Reaction Rate | Most Common Reaction | Antibody Formation Rate |
|---|---|---|---|---|
Exenatide ER (Bydureon) | Microsphere depot | 16% | Nodules, induration | 45-64% |
Albiglutide | Protein fusion | 15% | Erythema, induration | 3-5% |
Taspoglutide (withdrawn) | Zinc-based depot | 10.9% | Nodules, erythema | 49% |
Exenatide IR (Byetta) | Simple solution | 5.1% | Erythema, pruritus | 38-44% |
Lixisenatide | Simple solution | 3.9% | Erythema, pruritus | 56-70% |
Dulaglutide | Simple solution | 1-2% | Erythema | 1-2% |
Semaglutide | Simple solution | 1.4% | Erythema, pruritus | 1-2% |
Simple solution | 2-3% | Erythema, pruritus | 2-5% |
The table tells a clear story. Semaglutide and tirzepatide, the two most commonly used GLP-1 receptor agonists today, have among the lowest injection site reaction rates in their class. Semaglutide at 1.4% is barely above the placebo rate of 1.0% seen in clinical trials. Tirzepatide sits slightly higher at 2 to 3% based on SURPASS trial program data.
But these clinical trial numbers underrepresent real-world experience. Clinical trials use brand-name formulations administered by trained nurses under controlled conditions. Real-world use involves compounded formulations, self-administration with varying technique quality, and conditions that clinical trials do not replicate. The actual reaction rate among self-administering users is likely 3 to 5 times higher than clinical trial figures suggest.
Antibody formation correlates with injection site reactions but does not guarantee them. Lixisenatide produces antibodies in 56 to 70% of users but injection site reactions in only 3.9%. The relationship between antibodies and local reactions depends on antibody type, concentration, and individual immune response characteristics.
For users who experience significant injection site reactions with one GLP-1 medication, switching between GLP-1 medications often resolves the problem. The first published tirzepatide injection site reaction case involved a patient who had tolerated dulaglutide for years without reactions, then developed a severe reaction covering the entire lower abdomen after switching to tirzepatide. Individual drug susceptibility varies independently of GLP-1 receptor agonist class.
Step-by-step treatment for each type of reaction
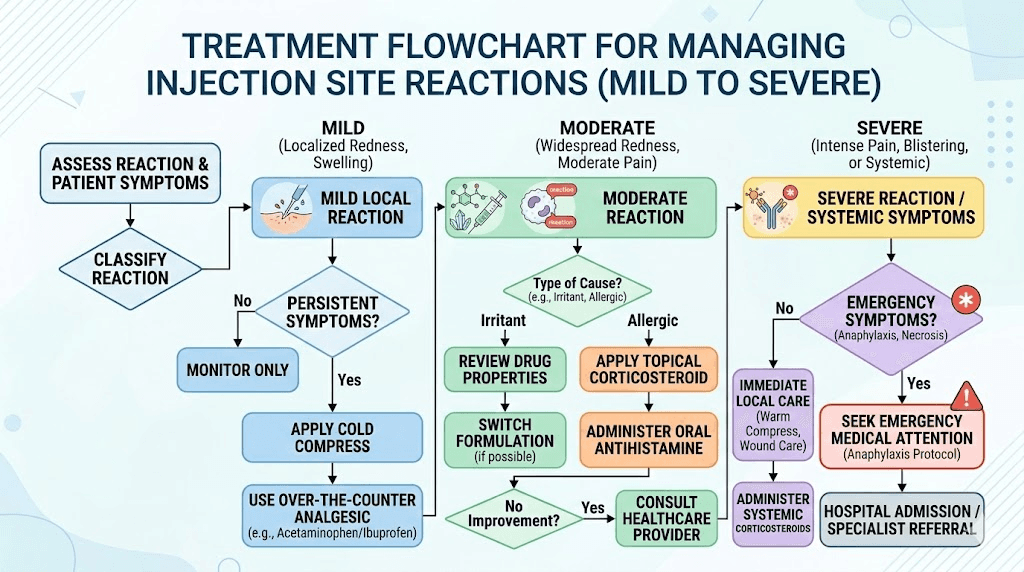
Treatment depends on what type of reaction you are dealing with. The approaches range from doing absolutely nothing for mild erythema to seeking immediate medical care for signs of infection or anaphylaxis.
Treating mild erythema and warmth
Most mild redness requires no treatment at all. The reaction resolves on its own within 24 to 48 hours. If the appearance bothers you or if the warmth is uncomfortable, a cold compress applied for 10 to 15 minutes provides immediate relief by constricting blood vessels and reducing histamine activity at the site.
Do not apply heat. Heat dilates blood vessels and increases blood flow to the area, which makes redness worse and can prolong the reaction. Some well-meaning guides suggest warm compresses for injection site reactions, but for erythema specifically, cold is the correct choice.
Continue your regular GLP-1 injection schedule without modification. Mild erythema does not indicate a problem with the medication and is not a reason to skip or delay doses. Simply choose a different injection site for your next administration.
Treating pruritus and itching
Itching responds to antihistamines. An over-the-counter oral antihistamine like cetirizine (Zyrtec), loratadine (Claritin), or diphenhydramine (Benadryl) reduces histamine activity systemically and can relieve injection site itching within 30 to 60 minutes. Cetirizine and loratadine are non-drowsy options suitable for daytime use. Diphenhydramine causes significant drowsiness but may be preferred for nighttime dosing if itching disrupts sleep.
Topical treatment works directly on the affected area. A thin layer of 1% hydrocortisone cream applied to the injection site reduces both itching and inflammation. Calamine lotion provides a cooling, soothing effect. Some users find that applying aloe vera gel offers relief, though the evidence for this is anecdotal rather than clinical.
The critical instruction is simple. Do not scratch. Scratching damages tissue, releases more histamine, and extends the duration and severity of the reaction. If injection site itching is severe enough that you cannot resist scratching, apply a cold compress to numb the area and take an oral antihistamine to address the underlying histamine response.
Treating induration and hardening
Firm, hardened areas beneath the skin require patience more than active treatment. The induration represents an organized inflammatory response that will resolve as the body processes the medication depot and clears inflammatory cells from the area. Typical resolution takes 3 to 10 days.
Warm compresses can help with induration, unlike erythema where cold is preferred. Warmth increases blood flow, which accelerates the clearing of inflammatory mediators and cellular debris from the hardened area. Apply a warm, damp cloth for 15 to 20 minutes, two to three times daily until the induration softens and resolves.
Gentle massage of the indurated area may help disperse the inflammatory infiltrate, but should be done carefully. Excessive pressure can worsen discomfort and potentially push medication deeper into tissue. Light, circular motions applied with moderate pressure are sufficient.
Do not attempt to inject through or near an existing induration. Allow the area to resolve completely before using it as an injection site again. Mark mentally or physically which areas need rest.
Treating injection site nodules
Nodules require more careful management because they indicate a more significant tissue response. The dose-dependent nature of semaglutide-associated nodules, documented in the first published case report, suggests that dose reduction may resolve the problem when nodules occur repeatedly.
If nodules appear consistently at the same dose level, discuss with your healthcare provider whether a temporary dose reduction is appropriate. In the published case, reducing from 1 mg to 0.5 mg semaglutide shortened nodule duration from 2 to 4 days to less than 24 hours. When the problem persisted even at the reduced dose, switching to dulaglutide eliminated nodules entirely.
Persistent nodules, meaning those lasting beyond two weeks, should be evaluated medically. While most are benign inflammatory responses, persistent subcutaneous nodules occasionally represent granulomatous reactions or other conditions that require diagnostic evaluation.
Treating bruising
Bruising requires no medical treatment. The discoloration follows a predictable progression from purple-red to green to yellow as the extravasated blood is metabolized and cleared. The entire process takes 7 to 14 days depending on bruise size and individual healing rates.
Cold compresses applied within the first 24 hours of bruise formation can limit the extent of blood spread by constricting damaged vessels. After 24 hours, switch to warm compresses to accelerate blood reabsorption. Over-the-counter arnica gel, applied topically, may speed bruise resolution, though clinical evidence for this is modest.
If you are taking blood thinners, aspirin, or supplements that affect clotting (fish oil, vitamin E, ginkgo biloba), expect larger bruises. This is a medication interaction effect, not a problem with the GLP-1 injection itself. Consider adjusting the timing of these supplements relative to injection day, with guidance from your healthcare provider.
Addressing lipodystrophy
Lipodystrophy requires a fundamentally different approach because it represents cumulative tissue damage rather than an acute reaction. The treatment is prevention through rigorous injection site rotation.
If lipohypertrophy has already developed, the primary intervention is complete rest of the affected area. Do not inject into or near the affected tissue. Given enough time, typically several months to a year, the fatty buildup can partially or fully resolve. Using a systematic rotation protocol ensures no single site receives repeated injections.
For insulin users with decades of injection experience, the research on lipodystrophy management is extensive. Those principles apply directly to GLP-1 administration. Palpate injection areas regularly for changes in tissue texture. Any area that feels different, harder, softer, or lumpier than surrounding tissue needs to be rested immediately.
How to prevent injection site reactions
Prevention is more effective than treatment for every type of injection site reaction. The strategies below, drawn from clinical research and decades of subcutaneous injection best practice, reduce both the frequency and severity of reactions.
Master the rotation protocol
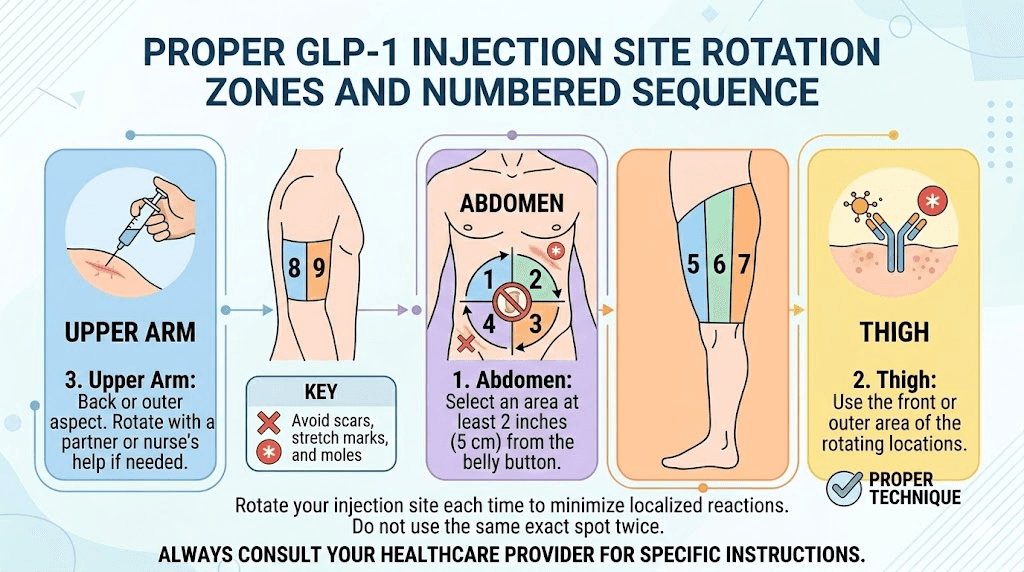
Site rotation is the single most important prevention strategy. Using the same injection location repeatedly is the fastest path to reactions, tissue damage, and altered medication absorption. Every GLP-1 injection guide emphasizes rotation, but few explain how to do it systematically.
The standard rotation uses three primary zones: abdomen, upper thigh, and upper arm. Within each zone, multiple specific sites exist. The abdomen alone provides at least 10 to 12 distinct injection locations when you divide it into quadrants and use locations at least 2.5 centimeters (1 inch) apart. The best GLP-1 injection site for most users is the abdomen because it offers the most surface area, consistent subcutaneous fat depth, and convenient access.
A practical weekly rotation for once-weekly GLP-1 injections might follow this pattern. Week one: right side of abdomen, upper quadrant. Week two: left side of abdomen, upper quadrant. Week three: right thigh, outer surface. Week four: left thigh, outer surface. Week five: right side of abdomen, lower quadrant. Week six: left side of abdomen, lower quadrant. Then repeat. This ensures no single spot is used more than once every six weeks.
Within each zone, maintain at least a 1-inch gap between consecutive injection sites. Some practitioners recommend using a slightly larger spacing of 2 inches to provide even more tissue recovery time. Mark previous injection sites mentally or use a simple diagram to track your rotation.
Let the medication reach room temperature
Cold medication causes more injection site reactions than room-temperature medication. This finding is consistent across virtually all subcutaneous injectable medications. The mechanism involves both vasoconstriction, where cold temperatures narrow blood vessels and slow medication dispersal, and direct tissue irritation from the temperature differential.
Remove the semaglutide or tirzepatide from the refrigerator 15 to 30 minutes before your planned injection time. Place it on a counter at room temperature. Do not use a microwave, warm water bath, or any external heat source to speed the process. These methods can denature the peptide and destroy the medication. Let it warm naturally.
Proper storage practices matter here too. If your medication is stored at the correct refrigeration temperature of 2 to 8 degrees Celsius, it needs adequate time to warm. If it has been stored at room temperature already (which is acceptable for certain formulations for limited periods), this step becomes less critical.
Perfect your injection technique
Technique errors cause or worsen the majority of injection site reactions. The key elements are angle, depth, speed, and needle quality.
Angle: Insert the needle at a 90-degree angle to the skin surface. This creates the shortest wound channel and deposits medication squarely in subcutaneous fat. An angled approach drags the needle through more tissue and increases trauma.
Depth: The needle should reach subcutaneous fat, not muscle. For most adults, standard pen needles and insulin syringes achieve this depth automatically at 90 degrees. Very lean individuals may need to pinch the skin to create a fold of subcutaneous fat before inserting the needle. Injecting too deeply into muscle tissue increases reaction severity and alters absorption kinetics.
Speed: Inject the medication slowly and steadily. Rapid injection creates hydraulic pressure that tears through tissue and disperses medication unevenly. Slow injection allows the medication to spread gently into the subcutaneous space. After completing the injection, hold the needle in place for 5 to 10 seconds before withdrawing to ensure complete delivery and minimize leakback.
Needle quality: Never reuse needles. Used needles have bent tips, microscopic barbs, and bacterial contamination that dramatically increase tissue trauma and infection risk. Each injection gets a fresh, sterile needle.
Let the alcohol dry completely
Clean the injection site with an alcohol swab. Then wait. Count to 30. Check that the skin feels completely dry. If any moisture remains, wait another 15 to 20 seconds. The extra 30 to 60 seconds of drying time prevents alcohol from being carried into subcutaneous tissue, eliminating one of the most common and most preventable causes of injection site burning and stinging.
Consider antihistamine pretreatment
For users who consistently experience mild injection site reactions despite proper technique and rotation, taking a non-drowsy antihistamine 30 to 60 minutes before injection can reduce or prevent histamine-mediated reactions. This approach works for erythema and pruritus specifically, as these reactions are driven by local histamine release.
Cetirizine 10 mg or loratadine 10 mg, taken orally before injection, provides sufficient antihistamine activity to blunt the histamine response at the injection site. This is a reasonable strategy for users who find injection site reactions bothersome but not severe enough to warrant dose adjustment or medication changes.
Members of SeekPeptides often discuss prevention strategies in detail, sharing what has worked for their specific situations and which approaches made the biggest difference in reducing injection site discomfort.
When a reaction means something serious
The vast majority of injection site reactions are nuisances, not emergencies. But a small percentage of reactions indicate something that requires prompt medical attention, and knowing the difference could prevent a minor problem from becoming a major one.
Signs of infection
Infection at an injection site is uncommon when proper aseptic technique is used, but it does occur. The distinguishing features of infection versus simple inflammation are progressive worsening beyond 48 hours, spreading redness with clear borders that expands outward, red streaks radiating from the injection site (indicating lymphangitis), warmth that increases rather than decreases over time, pus or cloudy discharge from the site, and fever above 100.4 degrees Fahrenheit (38 degrees Celsius).
A simple inflammatory reaction improves over 24 to 48 hours. An infection worsens. That trajectory is the key distinction.
If infection is suspected, do not attempt to treat it with topical antibiotics alone. Subcutaneous infections may require oral or intravenous antibiotics and sometimes surgical drainage. Contact your healthcare provider promptly.
Signs of allergic reaction
True allergic reactions to GLP-1 receptor agonists are rare, occurring in less than 0.5% of users across clinical studies. But when they occur, they can progress rapidly. The mechanism typically involves IgE-mediated hypersensitivity, particularly with exenatide-based medications that have a non-human peptide origin.
Localized allergic signs include injection site reactions that worsen with each successive dose rather than improving over time, spreading hives (urticaria) extending well beyond the injection site, and severe swelling (angioedema) at the injection site that does not resolve within 48 hours.
Systemic allergic signs that require emergency medical attention include widespread hives across multiple body areas, swelling of the face, lips, tongue, or throat, difficulty breathing or swallowing, rapid heartbeat or feeling faint, and dizziness or confusion.
Anaphylaxis, while extremely rare with GLP-1 medications, is a medical emergency. If you experience difficulty breathing, throat swelling, or cardiovascular symptoms after a GLP-1 injection, call emergency services immediately. Published case reports of GLP-1-associated anaphylaxis confirm that cross-reactivity between different GLP-1 agents can occur, meaning a person allergic to exenatide might also react to lixisenatide but tolerate liraglutide or semaglutide, which have different molecular structures.
Signs of abscess formation
An abscess is a walled-off collection of pus within tissue. At an injection site, it presents as a progressively enlarging, extremely tender, warm, fluctuant (soft and compressible) mass. The overlying skin may become taut, shiny, and dark red or purple. Fever and malaise often accompany abscess formation.
Abscesses require medical drainage. They do not resolve with oral antibiotics alone because the walled-off nature of the collection prevents adequate antibiotic penetration. If you develop a soft, expanding, painful mass at an injection site, seek medical evaluation within 24 hours.
The 48-hour rule
A practical rule for evaluating injection site reactions. Any reaction that is clearly improving at 48 hours is almost certainly benign and will resolve without intervention. Any reaction that is clearly worsening at 48 hours needs medical evaluation. Any reaction that is unchanged at 48 hours warrants monitoring for another 24 to 48 hours, and medical evaluation if it persists beyond 96 hours without improvement.
This simple framework prevents both unnecessary worry about benign reactions and dangerous complacency about concerning ones.
Compounded versus brand-name GLP-1: does the source affect reactions?
This is a question that comes up constantly, and the honest answer is nuanced. Both compounded and brand-name GLP-1 formulations can produce injection site reactions, but the reaction profiles may differ because the supporting ingredients differ.
Brand-name semaglutide (Ozempic, Wegovy) and brand-name tirzepatide (Mounjaro, Zepbound) use proprietary formulation buffers, preservatives, and pH adjusters that have been optimized through extensive pharmaceutical development. These formulations undergo rigorous testing for injection site tolerability as part of the FDA approval process.
Compounded semaglutide and compounded tirzepatide may use different excipients, different concentrations, and different preservative systems. Bacteriostatic water containing benzyl alcohol as a preservative is commonly used in compounded preparations, and benzyl alcohol itself can cause local tissue irritation in sensitive individuals.
The concentration matters too. A compounded preparation reconstituted with less bacteriostatic water will have a higher peptide concentration per unit volume. Higher concentrations mean more active ingredient deposited in a smaller tissue area, which increases the probability and severity of local irritation. Following proper reconstitution protocols ensures the correct concentration.
Compounding pharmacy quality also varies. Pharmacies operating under 503B regulations follow stricter manufacturing standards than 503A pharmacies. The sterility, purity, and consistency of the preparation all affect injection site reaction risk. Reputable compounding pharmacies like Empower Pharmacy and others maintain quality standards that minimize these risks.
If you experience injection site reactions with one source of semaglutide or tirzepatide, switching sources is a reasonable troubleshooting step. The active medication may be identical, but the formulation differences can meaningfully affect local tissue tolerability.
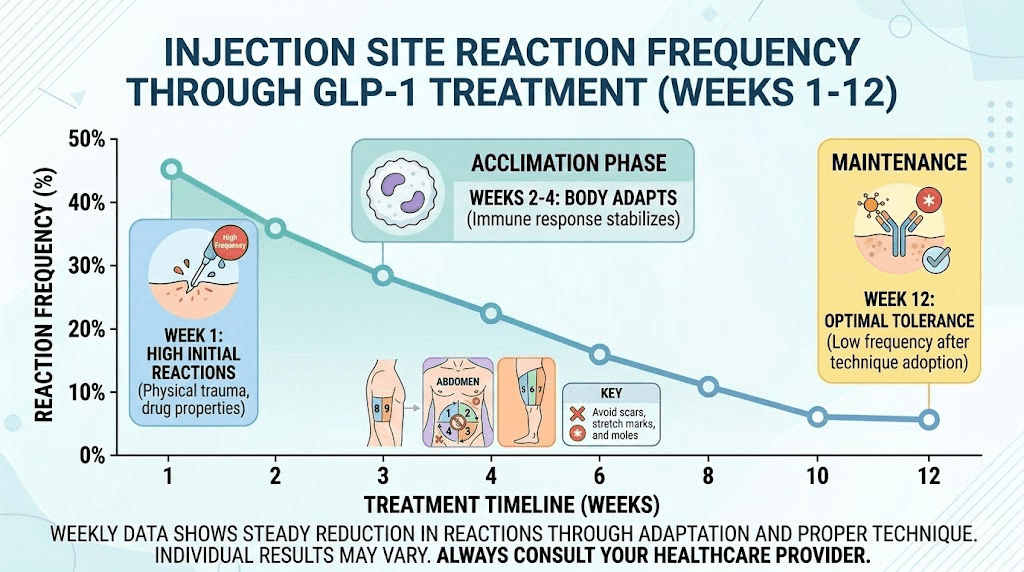
Reaction patterns across your treatment timeline
Injection site reactions do not follow a random pattern. They follow a predictable arc over the course of GLP-1 treatment that, once understood, removes much of the anxiety associated with them.
The first four weeks
Injection site reactions are most common during the first few weeks of treatment. Your tissue has never encountered this medication before. The immune system is encountering a new peptide. Your injection technique may still be developing. And the starting doses, while lower, are being injected into tissue that has not adapted to regular subcutaneous medication administration.
During this period, mild erythema and itching after your first semaglutide injection or first tirzepatide dose are extremely common and entirely expected. These reactions typically become less pronounced with each subsequent injection as the tissue adapts.
Weeks four through eight
Most users who experienced initial injection site reactions notice significant improvement during this period. The immune system develops tolerance to the medication. Injection technique improves with practice. The tissue adapts to regular subcutaneous deposits. Clinical data confirms that injection site reactions are most often transient and generally diminish as the body adjusts to the medication over 4 to 8 weeks.
During dose escalation
Dose escalation can temporarily increase injection site reactions. Moving from a lower dose to a higher dose of semaglutide or tirzepatide means more active medication is being deposited at the injection site. The tissue that had adapted to the lower concentration now encounters a higher one. Reactions during dose escalation are transient and typically follow the same adaptation pattern seen during initial treatment, resolving within 2 to 4 weeks at the new dose level.
Long-term maintenance
On stable long-term dosing, injection site reactions should be rare. If reactions persist or increase in frequency after months of stable dosing, this warrants investigation. Possible explanations include deteriorating injection technique (rushing, poor rotation), medication quality changes (different lot, different pharmacy), development of anti-drug antibodies, or onset of a skin condition unrelated to the injection itself.
Consistent reactions during maintenance therapy that resist prevention strategies may benefit from a medication switch. Discuss with your healthcare provider whether transitioning to a different GLP-1 receptor agonist is appropriate, as cross-reactivity is not guaranteed and many users who react to one medication tolerate another without difficulty.
Special considerations for specific medications
While the general principles apply across all GLP-1 receptor agonists, each medication has unique characteristics that affect injection site reaction patterns.
Semaglutide-specific considerations
Semaglutide has among the lowest clinical trial injection site reaction rates at 1.4%, making it one of the best-tolerated GLP-1 agonists in this regard. Its human GLP-1 backbone with 94% structural homology to native human GLP-1 produces minimal immunogenic response. The fatty acid chain modification that extends its half-life to approximately one week does not appear to contribute to local reactions.
When reactions do occur with semaglutide, they are predominantly mild erythema and pruritus. The published case of semaglutide injection site nodules remains notable precisely because it was the first such report, highlighting how uncommon this presentation is.
Users of oral semaglutide formulations and sublingual semaglutide bypass injection site reactions entirely, which represents one advantage of non-injectable delivery routes. The trade-off is typically lower bioavailability and different absorption kinetics compared to subcutaneous injection.
For those combining semaglutide with other compounds like B12, glycine, or L-carnitine, the additional ingredients in combined preparations may contribute to or modify injection site reaction patterns. Each added compound introduces its own chemical properties that interact with local tissue.
Tirzepatide-specific considerations
Tirzepatide operates as a dual GIP/GLP-1 receptor agonist, which means it has a different molecular structure than pure GLP-1 agonists. The SURPASS trial program documented injection site reactions as generally mild to moderate in severity, with most reactions resolving spontaneously without intervention.
The first published case of a significant tirzepatide injection site reaction described a progressive rash that enlarged over several days after administration, eventually covering the entire lower abdomen before resolving approximately one month after discontinuation. This case is notable because the patient had previously tolerated dulaglutide without any injection site reactions, emphasizing that tolerance to one GLP-1 agonist does not predict tolerance to another.
Tirzepatide formulations that include additional compounds like glycine and B12, niacinamide, or levocarnitine introduce additional variables that may affect local tolerability. When troubleshooting tirzepatide injection site reactions, consider whether the reaction is from the tirzepatide itself or from an additive in the compounded preparation.
Retatrutide considerations
Retatrutide, the triple agonist targeting GLP-1, GIP, and glucagon receptors, is still in clinical trials but early data provides insight into its injection site reaction profile. As a larger, more complex molecule than semaglutide or tirzepatide, its immunogenicity profile may differ. Users transitioning from tirzepatide to retatrutide should monitor for new or different injection site reaction patterns during the switch.
Proper injection technique for retatrutide follows the same principles as other GLP-1 agonists. Site rotation, room temperature administration, complete alcohol drying, and slow injection speed all apply. The retatrutide injection process should not present unique challenges for users already experienced with semaglutide or tirzepatide administration.
Troubleshooting persistent reactions
When injection site reactions continue despite proper technique and prevention measures, a systematic troubleshooting approach helps identify the root cause.
Step 1: Verify your technique
Have someone observe your injection process, or record a video for self-review. Common technique errors that persist undetected include inserting the needle at an angle rather than 90 degrees, injecting too quickly, not pinching skin when needed for lean body composition, and withdrawing the needle before the full dose is delivered. Each of these errors increases tissue trauma and reaction severity.
Step 2: Evaluate your reconstitution
If using compounded medication that requires reconstitution, verify the concentration. Adding too little bacteriostatic water creates an overly concentrated solution. Measure carefully. Use the exact volume recommended for your vial size. The semaglutide dosage calculator and peptide reconstitution calculator on SeekPeptides can help verify your calculations are correct.
Step 3: Check medication storage
Improperly stored medication can produce more injection site reactions. Verify your medication is stored at the correct temperature. Semaglutide storage requirements and tirzepatide refrigeration guidelines specify temperature ranges for a reason. Medication that has been exposed to excessive warmth or temperature excursions may have partially degraded, producing breakdown products that irritate tissue.
Check for visible changes. Semaglutide should appear clear and colorless. Tirzepatide has its own expected appearance. Cloudiness, particulates, or unexpected color changes (such as red discoloration in semaglutide) indicate potential degradation and the medication should not be used.
Check expiration. Expired tirzepatide and expired semaglutide may produce breakdown products that increase local irritation. Always use medication within its recommended shelf life.
Step 4: Try a different source
If reactions persist with one brand or pharmacy, switch to a different source. Different compounding pharmacies use different excipient systems. A different brand-name formulation (switching between pen devices) may have subtle formulation differences. The peptide itself may be identical, but the surrounding ingredients matter.
Step 5: Consider a medication change
Persistent, significant injection site reactions that do not respond to technique optimization and source changes may represent individual drug intolerance. The side effect profiles of semaglutide and tirzepatide differ enough that switching between them is a reasonable strategy. The conversion charts available can help guide dose equivalency when transitioning.
For some users, switching to an oral GLP-1 formulation eliminates injection site reactions entirely. Oral tirzepatide options, tirzepatide drops, and tablet formulations provide the same active medication without the subcutaneous delivery that causes local reactions. The GLP-1 patch format represents another alternative delivery route worth discussing with your provider.
Managing reactions during dose changes and medication switches
Dose escalation and medication transitions are the two highest-risk periods for injection site reactions outside of initial treatment. Understanding what to expect during these transitions helps separate expected reactions from concerning ones.
When increasing your tirzepatide dose or semaglutide dose, expect the possibility of temporary injection site reactions even if you had none at the lower dose. The tissue is adapting to a higher medication concentration. These escalation reactions typically resolve within 2 to 3 doses at the new level.
Some researchers use microdosing strategies or split dosing approaches to manage dose-escalation side effects. While these strategies are primarily used for gastrointestinal side effects, they can also reduce injection site reaction severity by delivering smaller volumes per injection.
When switching from tirzepatide to semaglutide or in the other direction, treat the new medication as if you are starting fresh from an injection site reaction perspective. Monitor for reactions at the new medication as you did during initial treatment. The fact that you tolerated one GLP-1 agonist does not guarantee tolerance of another, as the published tirzepatide case report clearly demonstrated.
Frequently asked questions
How long does a GLP-1 injection site reaction typically last?
Most mild reactions, including redness, warmth, and minor itching, resolve within 24 to 48 hours. Induration and hardening may persist for 3 to 10 days. Nodules can last 2 to 4 days at standard doses. Bruising follows its own 7 to 14 day timeline. Any reaction that worsens after 48 hours or persists beyond two weeks should be evaluated by a healthcare provider. The general trend is improvement over the first weeks of GLP-1 treatment as the body adapts.
Can I still take my GLP-1 medication if I have an injection site reaction?
For mild reactions (redness, itching, small bruises), yes. Continue your regular injection schedule and simply choose a different injection site. For moderate reactions (significant induration, large nodules), continue medication but use a completely different body zone for the next injection. For severe reactions (spreading redness beyond 5 centimeters, signs of infection, breathing difficulty), hold the medication and contact your healthcare provider before the next scheduled dose.
Are injection site reactions worse with compounded GLP-1 medications?
Not necessarily, but they can differ. Compounded formulations use different excipients than brand-name products, which means the local tissue reaction profile may be different. Some users tolerate compounded versions better, others worse. The quality of the compounding pharmacy matters significantly, with 503B facilities generally producing more consistent formulations. If reactions occur with one source, trying a different compounding pharmacy or switching to brand-name medication is a reasonable troubleshooting step.
Does the injection site location affect reaction severity?
Yes. The abdomen tends to produce milder reactions than the thigh for most users, likely because abdominal subcutaneous fat provides a more consistent depot environment with fewer blood vessels. Upper arm injections may produce more bruising due to thinner subcutaneous tissue in some individuals. Individual variation is significant, and the best approach is to monitor your own reaction patterns across different sites and favor the locations that produce the mildest responses.
Should I take an antihistamine before every GLP-1 injection?
Not routinely. Antihistamine pretreatment is a targeted strategy for users who consistently experience histamine-mediated reactions (erythema, pruritus) despite proper technique. If your reactions are minimal or absent, there is no benefit to routine antihistamine use. If reactions are bothersome, trying a single dose of cetirizine or loratadine 30 to 60 minutes before injection is a reasonable low-risk strategy. Discuss with your healthcare provider if you find yourself needing antihistamines before every injection.
Can injection site reactions indicate the medication is not working?
No. Injection site reactions have no correlation with medication efficacy. A medication that causes significant local reactions can still be fully effective systemically. Similarly, a medication that produces no injection site reactions at all is not necessarily working better. The local tissue response and the systemic metabolic effects operate through completely different mechanisms. Judge effectiveness by metabolic outcomes like appetite suppression, weight loss, and glycemic control, not by injection site appearance.
Will injection site reactions get worse as I increase my dose?
They can temporarily increase during dose escalation, as higher doses mean more concentrated medication at the injection site. But this effect is transient. Most users find that injection site reactions during dose escalation follow the same diminishing pattern they experienced during initial treatment, peaking during the first 1 to 2 injections at the new dose and resolving by the third or fourth injection at that level. Proper injection technique and prevention strategies become especially important during dose transitions.
External resources
Injection-Site Nodules Associated With Once-Weekly Subcutaneous Administration of Semaglutide (PMC)
Tirzepatide-Induced Injection Site Reaction Case Report (PMC)
For researchers serious about optimizing their peptide protocols and managing every aspect of GLP-1 administration safely, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your injection sites stay calm, your reactions stay mild, and your protocols stay consistent.