Feb 23, 2026

Before you dismiss that red, itchy bump as nothing, read this. Injection site reactions from tirzepatide catch thousands of researchers off guard every month. Some reactions fade in hours. Others spread across the abdomen over days, turning into something that looks far worse than it actually is. And in rare cases, a reaction signals something that genuinely needs medical attention.
The confusing part is telling the difference.
Clinical trials show that tirzepatide side effects at the injection site affect roughly 2 to 6 percent of users, depending on the study and the dose. That number climbs to 4.6 percent among those who develop anti-tirzepatide antibodies. Most of these reactions are mild. They resolve on their own. But when you are staring at a growing welt on your stomach at 10 PM, mild is not the word that comes to mind. You want answers. You want to know whether to push through, switch sites, apply ice, take an antihistamine, or call your doctor. This guide covers all of it. From the clinical data behind tirzepatide body responses to the practical treatments that work, from prevention strategies that actually reduce reaction frequency to the warning signs that mean it is time to seek help. Whether you are on your first injection or your twentieth, the information here will help you manage injection site reactions with confidence rather than guesswork.
What does a tirzepatide injection site reaction look like
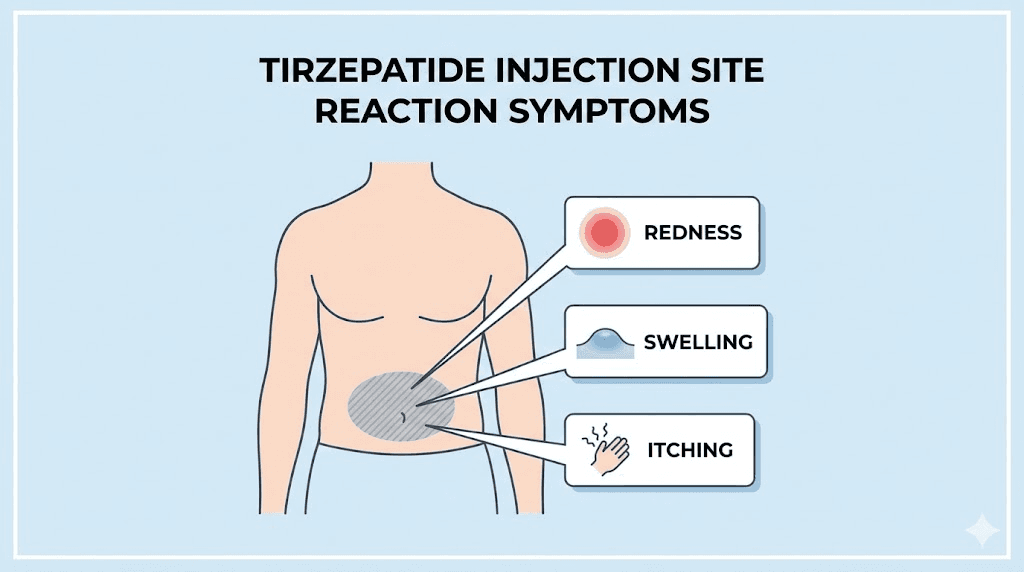
The first thing most people notice is redness. A circular patch of pink or red skin appears around the needle entry point, usually within the first few hours after injection. The size varies. Some people see a dime-sized spot. Others develop a reaction the size of a baseball, warm to the touch and slightly raised above the surrounding skin.
Swelling comes next. The tissue around the injection site puffs up, creating a small bump or a broader area of induration. This firmness under the skin can feel like a mosquito bite at its mildest or a deep bruise at its most uncomfortable. The swelling typically peaks somewhere between 12 and 48 hours after the injection.
Then there is the itching. For many tirzepatide users, the itch is more bothersome than the pain. It ranges from a mild tingle to a persistent, almost maddening urge to scratch. Scratching makes it worse. The area becomes more inflamed, the redness spreads, and recovery takes longer. Understanding proper tirzepatide injection technique helps prevent some of these reactions from occurring in the first place.
Pain and tenderness round out the most common symptoms. Some people describe a burning sensation during and immediately after injection. Others feel a dull ache that develops hours later and lingers for a day or two. The pain is usually proportional to the visible reaction. A bigger red patch generally means more discomfort. A small spot typically means minimal soreness.
Less common but worth knowing about are nodules. Small, firm lumps that form under the skin at the injection site, sometimes lasting several days before dissolving. These represent localized deposits of the medication or minor tissue reactions. They are not dangerous, but they can be alarming if you do not expect them. If you have experienced tirzepatide muscle pain alongside these injection site symptoms, the combination can feel particularly uncomfortable during the first few weeks.
Bruising is another possibility, especially for people who are new to subcutaneous injections or who have thinner skin. A small purple or yellow mark at the injection site is cosmetically annoying but medically insignificant. It indicates that a tiny blood vessel was nicked during the injection process, not that anything went wrong with the medication itself.
The timeline matters too. Most genuine injection site reactions appear within the first 1 to 6 hours after injection. They peak within 24 to 48 hours. And they resolve completely within 3 to 7 days. Anything that persists beyond a week or worsens after the first 48 hours warrants a closer look, which we will cover in detail later in this guide.
Why tirzepatide causes injection site reactions
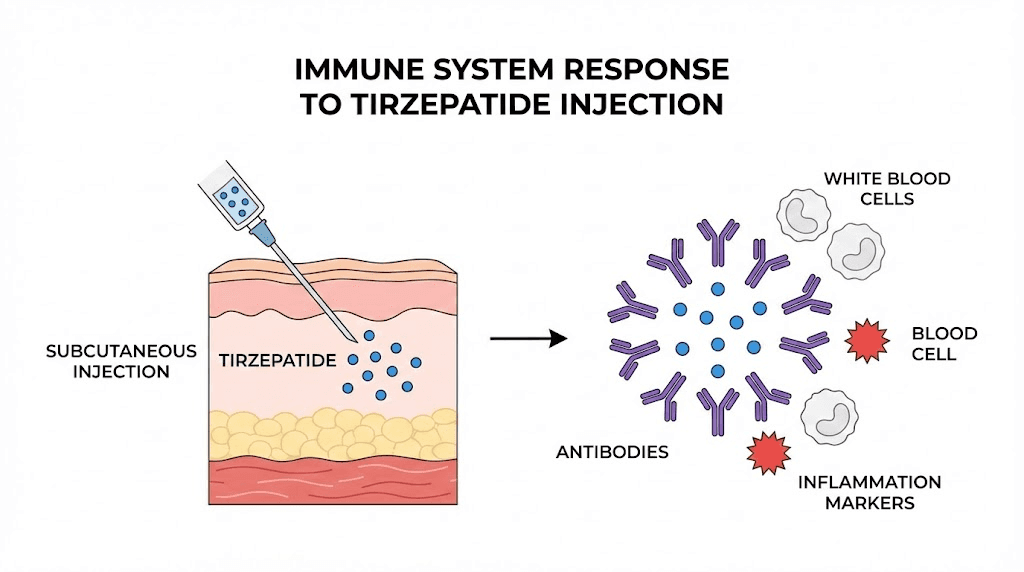
Understanding the mechanism helps. When you inject tirzepatide subcutaneously, you are depositing a synthetic peptide into the layer of fat just beneath the skin. Your immune system has a job to do. It evaluates everything that enters the body, decides whether it belongs, and mounts an appropriate response.
Tirzepatide is a dual GIP and GLP-1 receptor agonist. It is a synthetic molecule, which means the immune system does not recognize it as something the body produced naturally. In many cases, the immune system tolerates the peptide without issue. The medication gets absorbed, enters the bloodstream, and does its work on metabolic pathways without triggering a local inflammatory response.
But in some people, the immune system reacts differently.
The injection itself causes minor tissue trauma. A needle penetrating the skin and subcutaneous layer creates a small wound. This activates local inflammatory mediators, the chemical signals that bring immune cells to the area to assess and repair the damage. In most cases, this response is so mild that you never notice it. But when combined with the immune system encountering the tirzepatide molecule, the response can amplify into a visible, palpable injection site reaction.
Anti-drug antibodies play a significant role. Clinical data from the SURPASS Phase 3 trials revealed that treatment-emergent anti-drug antibodies developed in 51.1 percent of tirzepatide-treated patients. That is more than half of all users. The immune system literally creates antibodies against the tirzepatide molecule, tagging it as something foreign. Among patients who developed these antibodies, injection site reactions occurred in 4.6 percent compared to just 0.7 percent among those who did not develop antibodies. The connection between immune responses to tirzepatide and local skin reactions is well documented in the research.
Here is the reassuring part. Despite the high rate of antibody formation, these antibodies do not appear to reduce the effectiveness of tirzepatide. Neutralizing antibodies against GIP receptor activity were observed in only 1.9 percent of patients. Against GLP-1 receptor activity, 2.1 percent. And less than 1 percent had cross-reactive neutralizing antibodies against native GIP or native GLP-1. The antibodies are there, but they are not blocking the medication from working. Your tirzepatide weight loss timeline remains on track even if you develop mild injection site reactions.
The formulation itself also matters. Compounded tirzepatide may contain different excipients, preservatives, or stabilizers than brand-name Mounjaro or Zepbound. These inactive ingredients can occasionally trigger localized reactions independent of the tirzepatide molecule itself. Bacteriostatic water used in tirzepatide reconstitution contains benzyl alcohol as a preservative, which some people find mildly irritating to subcutaneous tissue. If you are reconstituting your own vials, the concentration you prepare using a tirzepatide reconstitution chart affects how much volume you inject, and larger volumes can cause more tissue displacement and a bigger local response.
Temperature also plays a role in the mechanism. Injecting cold medication straight from the refrigerator causes vasoconstriction at the injection site. The blood vessels narrow, the medication gets absorbed more slowly, and the concentrated peptide sitting in the tissue has more time to trigger an inflammatory response. This is why one of the most effective prevention strategies, letting the medication warm to room temperature before injecting, actually works. Proper tirzepatide storage and pre-injection warming make a measurable difference.
Individual physiology rounds out the picture. People with more reactive immune systems, those with a history of allergies or skin sensitivities, and those with certain autoimmune conditions tend to experience injection site reactions more frequently. Body composition matters too. Injecting into very lean tissue (too little subcutaneous fat) increases the chance of hitting muscle, which causes more pain and a bigger inflammatory response. Understanding proper GLP-1 injection technique reduces this risk significantly.
How common are tirzepatide injection site reactions
Numbers help put things in perspective. Across the SURPASS clinical trial program, injection site reactions with tirzepatide were consistently reported in approximately 2 to 6 percent of patients. Compare that to less than 1 percent with placebo. Compare it to the side effect profiles of other GLP-1 medications, and tirzepatide falls in a similar range.
The specific numbers by study paint a more detailed picture. In SURPASS-1 through SURPASS-6, injection site reactions ranged from less than 1 percent to 7 percent, depending on the trial and dosage tier. The higher incidence rates tended to cluster at higher doses, which makes biological sense. More medication means more foreign protein for the immune system to respond to, more volume displacing tissue, and more potential for a localized reaction.
In the pooled analysis of seven clinical trials, here is where the antibody data gets specific. Among 2,570 tirzepatide-treated patients who developed anti-tirzepatide antibodies, 119 experienced injection site reactions. That is 4.6 percent. Among 2,455 patients who did not develop antibodies, only 18 reported injection site reactions. That is 0.7 percent. The difference is striking. Antibody development increases the risk of injection site reactions by roughly six times.
But context matters enormously here.
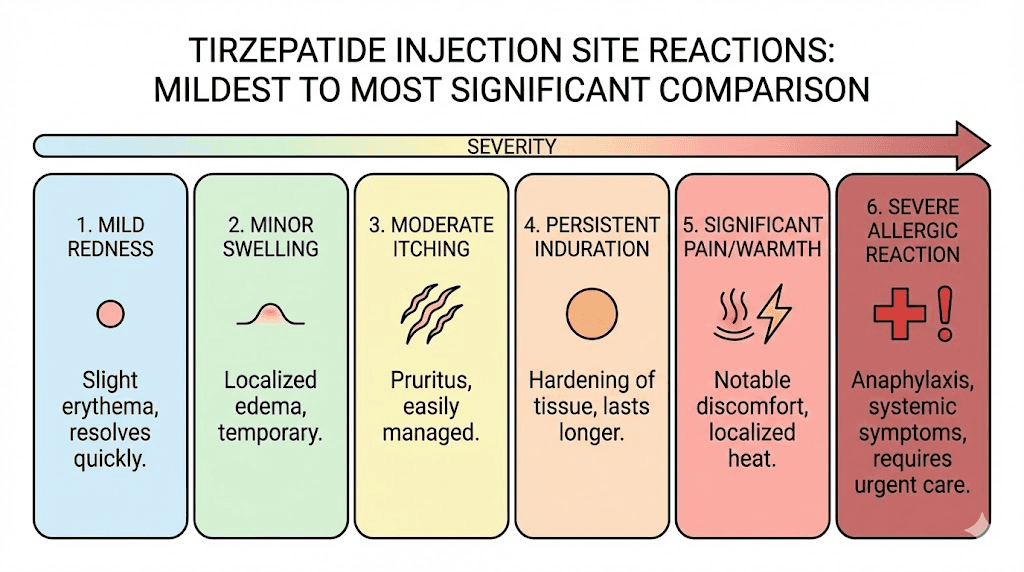
No severe injection site reactions were reported across the entire SURPASS clinical trial program. Zero. Every single documented reaction was classified as mild or moderate. None led to hospitalization. None caused lasting tissue damage. And many resolved without any treatment at all. The overwhelming majority of users on tirzepatide dosing protocols either never experience a reaction or experience one so mild that it barely registers.
Real-world data from the FDA Adverse Event Reporting System (FAERS) provides additional perspective. While injection site reactions appear in the FAERS database for tirzepatide, they are far less frequently reported than gastrointestinal side effects like constipation, diarrhea, and nausea. Injection site reactions rank as one of the less common side effects in the overall tirzepatide safety profile.
One published case report from 2023 in the journal Cureus described a 76-year-old male who developed a progressively enlarging rash across his entire lower abdomen after his first tirzepatide injection. He had previously tolerated dulaglutide without any issues. The rash was characterized by dull pain, burning, and soreness that spread over 10 days. After discontinuing tirzepatide, his skin returned to normal within a month. This case was notable precisely because it was unusual, representing the first formally documented case of tirzepatide-induced injection site reaction severe enough to warrant a published case report.
What does this tell you? Injection site reactions are real. They are not rare enough to dismiss. But they are overwhelmingly mild, self-limiting, and manageable. The statistics should reassure rather than alarm.
If you are weighing whether to start tirzepatide and injection site reactions are a concern, the data says that more than 94 percent of users never experience one at all. Those who do almost always find them easy to manage. For a broader view of what to expect from tirzepatide treatment, understanding both the benefits and the minor inconveniences provides a realistic picture.
The real-world experience matches the clinical data closely. Online communities, forums, and patient support groups consistently report that injection site reactions are among the mildest and most manageable tirzepatide side effects. Compared to the nausea that many people experience during dose escalation, or the dietary adjustments required when appetite changes dramatically, a red bump that fades in a few days barely registers on the inconvenience scale. Most experienced researchers consider it background noise in an otherwise transformative treatment. People who track their tirzepatide weight loss results quickly realize that the outcomes far outweigh the minor hassle of occasional injection site management. The results speak for themselves, and a temporary skin reaction does not change that equation.
Types of tirzepatide injection site reactions
Not all reactions are the same. Understanding the different types helps you respond appropriately to each one.
Localized erythema (redness)
The most common type. A patch of redness centered on the injection site that appears within hours and fades within 1 to 3 days. No treatment needed in most cases. If the redness bothers you, a cold compress handles it. This type of reaction rarely interferes with ongoing tirzepatide use, and most researchers simply note it and move on with their dosing schedule.
Indurated papule (firm bump)
A firm, slightly raised bump at the injection site. Typically pea-sized to marble-sized. Can be tender to the touch. This is the body walling off the area where medication was deposited, creating a small inflammatory pocket. It resolves within 3 to 7 days as the medication fully absorbs. Alternating injection sites per your injection protocol prevents these from accumulating in one area.
Subcutaneous nodule (deeper lump)
A deeper, firmer lump that forms below the skin surface. These can take 1 to 2 weeks to fully resolve. They represent a more significant tissue reaction to the deposited medication. While uncomfortable, they are not dangerous. Warm compresses can help speed absorption. If you notice nodules forming regularly, reviewing your reconstitution concentration and injection depth is worth considering.
Injection site pruritus (itching)
Itching without significant redness or swelling. This is often a histamine-mediated response, meaning the body is releasing histamine at the injection site as part of a mild allergic-type reaction. An oral antihistamine like cetirizine (10 mg) taken 30 to 60 minutes before injection can prevent this entirely. Many users who experience this find that it diminishes over weeks as the body becomes accustomed to regular tirzepatide doses.
Injection site bruising
Purple or yellow discoloration from a nicked blood vessel. Entirely mechanical, not related to the medication itself. More common in people with thinner skin, those on blood thinners, or those new to self-injection. Improving injection technique with syringes reduces bruising frequency significantly. Pinch the skin firmly, insert at a 45 to 90 degree angle depending on body composition, and avoid areas where veins are visible.
Injection site rash (broader reaction)
A more extensive skin reaction that spreads beyond the immediate injection site. This is the type described in the published case report, where redness and inflammation can spread several inches from the injection point. These broader reactions suggest a more significant immune response and may warrant a conversation with your healthcare provider about whether to continue, adjust the dose, or switch medications. Understanding the full range of tirzepatide side effects helps you distinguish between normal and concerning reactions.
Delayed hypersensitivity reaction
A reaction that appears 24 to 72 hours after injection rather than within the first few hours. These delayed reactions involve a different arm of the immune system (T-cell mediated rather than immediate histamine-mediated) and can sometimes be more persistent. They may recur with subsequent injections even when injection sites are rotated. If you experience delayed reactions consistently, discussing alternatives with your prescriber is a reasonable step. Some people find that switching to semaglutide from tirzepatide resolves the issue entirely.
How to treat tirzepatide injection site reactions
Treatment depends on the severity. Most reactions respond well to simple home measures. Here is what works, organized from first-line approaches to more involved interventions.
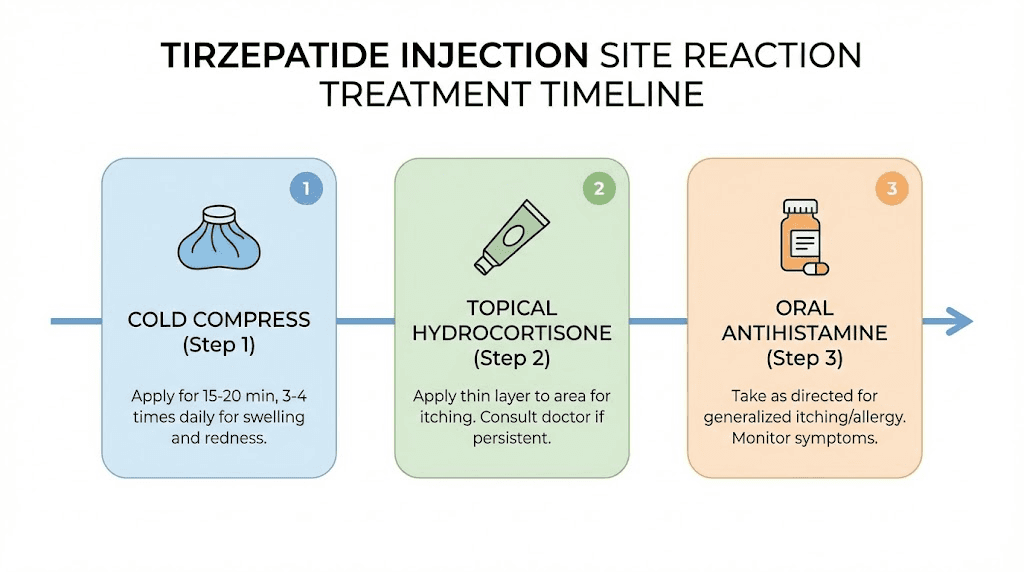
Cold compresses (first-line treatment)
Apply a cold compress or ice pack wrapped in a thin cloth to the injection site for 10 to 15 minutes, several times per day. Cold reduces inflammation, constricts blood vessels to limit swelling, and numbs the area to reduce pain and itching. This is the single most effective immediate treatment for mild to moderate injection site reactions.
Do not apply ice directly to skin. Wrap it in a cloth or use a commercial cold pack. Apply within the first hour after injection if you notice a reaction starting. Continue applying 3 to 4 times daily until the reaction resolves. Many experienced researchers apply cold routinely after every injection as a preventive measure, not just as treatment.
Topical hydrocortisone cream
Over-the-counter hydrocortisone cream (0.5 to 1 percent) applied twice daily to the reaction site reduces inflammation and itching. This is a mild topical steroid that works by suppressing the local immune response. It is safe for short-term use on injection sites and provides noticeable relief within hours of application.
Apply a thin layer directly to the affected area. Do not bandage it unless the area is weeping. Use for up to 7 days. If the reaction persists beyond a week of hydrocortisone use, escalate to medical evaluation. For those managing multiple aspects of their tirzepatide protocol, adding hydrocortisone cream to the supply kit is a practical step.
Oral antihistamines
Cetirizine (10 mg) or loratadine (10 mg) taken orally can significantly reduce itching, redness, and swelling. These non-drowsy antihistamines block histamine receptors, preventing the allergic-type component of the injection site reaction from amplifying.
For best results, take the antihistamine 30 to 60 minutes before your injection. This preemptive approach is far more effective than treating a reaction after it has already developed. Some people take a daily antihistamine throughout their entire tirzepatide treatment course and report dramatically fewer injection site issues. This approach pairs well with other management strategies for tirzepatide-related discomfort.
If non-drowsy options do not provide enough relief, diphenhydramine (25 mg) is more potent but causes drowsiness. Taking it at bedtime on injection day can help manage nighttime itching while you sleep.
Warm compresses (for nodules)
While cold works best for acute inflammation, warm compresses are better for resolving nodules and deeper lumps. Apply a warm (not hot) cloth to the area for 10 to 15 minutes, 2 to 3 times daily. The warmth increases blood flow to the area, speeding up absorption of the deposited medication and resolution of the inflammatory pocket.
Use warm compresses starting 48 hours after the injection if a nodule has formed. The first 48 hours favor cold (to limit initial inflammation), and the period after 48 hours favors warmth (to promote resolution). This two-phase approach mirrors standard sports medicine practice for managing soft tissue injuries.
Topical antihistamine gel
Diphenhydramine gel applied directly to the injection site provides localized itch relief without the drowsiness of oral diphenhydramine. Apply a thin layer to the affected area up to 4 times daily. This is particularly useful for people who experience primarily itching rather than pain or swelling.
Aloe vera gel
Pure aloe vera gel has natural anti-inflammatory properties and provides a cooling, soothing sensation when applied to irritated skin. It will not resolve a significant immune-mediated reaction, but for mild redness and discomfort, it offers relief without any medication at all. Keep it refrigerated for an added cooling effect.
When home treatment is not enough
If injection site reactions are severe, persistent, or worsening despite the measures above, medical intervention may include prescription-strength topical steroids (triamcinolone 0.1 percent), oral corticosteroid courses for widespread reactions, or evaluation for potential allergy to a specific component of the formulation. Your healthcare provider may also suggest adjusting your tirzepatide starting dose or changing the formulation you are using.
How to prevent tirzepatide injection site reactions
Prevention beats treatment every time. These strategies, backed by clinical evidence and real-world experience, significantly reduce the frequency and severity of injection site reactions.
Let the medication reach room temperature
This is the single most impactful prevention strategy. Remove the vial or pen from the refrigerator 30 minutes before injection. Cold medication causes vasoconstriction, slows absorption, and increases the concentration of peptide sitting in one spot for longer, all of which amplify the local immune response.
Room temperature medication flows more easily, distributes more evenly through the subcutaneous tissue, and absorbs more quickly. Many users who initially experienced reactions found them completely eliminated by this one change alone. Understanding proper tirzepatide refrigeration requirements and temperature management is essential for both storage safety and injection comfort.
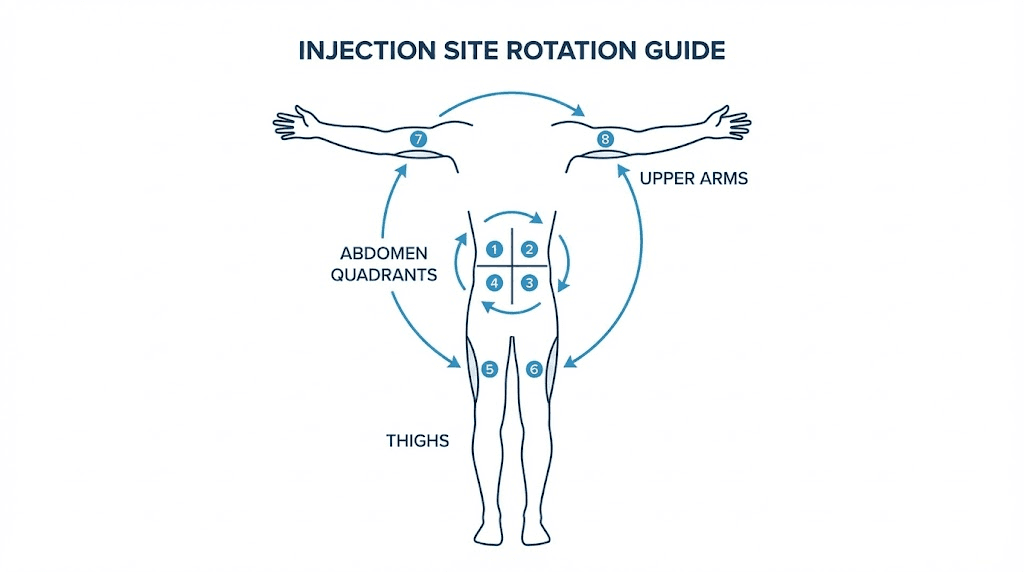
Systematic injection site rotation
Rotate injection sites in a planned pattern. The abdomen, thighs, and upper arms each offer large surface areas for subcutaneous injection. Within each region, maintain at least 1 inch (2.5 cm) of distance between consecutive injection sites.
A practical rotation system: divide the abdomen into four quadrants (upper left, upper right, lower left, lower right). Rotate through the quadrants weekly. Within each quadrant, move the injection point at least an inch from the previous week. This gives each site a full month of recovery before you return to it. The complete guide to GLP-1 injection sites covers rotation patterns in more detail.
Some people keep a simple log noting which site they used each week. Others mark the site with a small dot from a washable marker. Whatever system you use, consistency in rotation prevents tissue fatigue and significantly reduces the chance of reactions developing in overused areas.
Proper injection technique
Technique matters more than most people realize. Clean the injection site with an alcohol swab and let it dry completely before injecting. Injecting into wet alcohol-prepped skin causes stinging and can irritate the subcutaneous tissue. The alcohol needs a full 30 seconds to evaporate.
Pinch a fold of skin to lift the subcutaneous tissue away from the muscle layer. Insert the needle at the appropriate angle (45 degrees for shorter needles, 90 degrees for standard insulin syringes with adequate skin fold). Inject the medication slowly and steadily rather than pushing the plunger quickly. A slow injection allows the medication to distribute more evenly, reducing the concentrated bolus that triggers localized reactions.
After injection, hold the needle in place for 5 to 10 seconds before withdrawing. This prevents medication from leaking back along the needle track, which can cause superficial skin irritation. The complete stomach injection guide walks through these steps in detail with visual references.
Avoid damaged or sensitive skin
Never inject into skin that is sunburned, scarred, bruised, inflamed, or has stretch marks. These areas have altered tissue architecture and blood flow patterns that increase the risk of abnormal reactions. Choose skin that looks healthy, has adequate subcutaneous fat, and is free from any pre-existing irritation.
Consider the injection volume
Larger injection volumes cause more tissue displacement and can increase reaction frequency. If you are reconstituting compounded tirzepatide, the concentration you prepare determines how much volume you inject per dose. A more concentrated solution means a smaller injection volume, which generally produces less local reaction. Review your bacteriostatic water mixing ratios to optimize concentration while maintaining accurate dosing.
However, very high concentrations can also be irritating. The sweet spot is a concentration that keeps the injection volume between 0.1 and 0.5 mL for most doses. Your peptide reconstitution calculator can help determine the ideal ratio.
Pre-treat with antihistamines
If you consistently experience injection site reactions despite other prevention strategies, taking a non-drowsy antihistamine (cetirizine 10 mg or loratadine 10 mg) 30 to 60 minutes before injection provides a preventive buffer. This blocks histamine receptors before the injection triggers histamine release, essentially intercepting the reaction before it can start.
Many long-term tirzepatide users incorporate this into their injection routine as standard practice. Take the antihistamine, set a 30-minute timer, prepare your injection supplies, and inject once the antihistamine has had time to take effect.
Injection speed and needle selection
Injecting too quickly forces the medication into the tissue faster than it can distribute, creating a pressurized pocket that triggers more inflammation. Aim for a slow, steady push that takes at least 5 to 10 seconds for the full dose. Some users find that splitting the injection into two smaller doses at separate sites further reduces reactions, though this adds complexity to the dosing protocol.
Needle gauge also matters. Finer needles (30 or 31 gauge) cause less tissue trauma than thicker needles (27 or 28 gauge). The trade-off is that finer needles require slightly more pressure and take slightly longer to inject the same volume. For tirzepatide specifically, 30-gauge needles offer the best balance of minimal tissue trauma and practical injection speed.
When to worry about a tirzepatide injection site reaction
Most injection site reactions are nuisances, not emergencies. But some signs should prompt immediate action.
Red flags that require medical attention
Spreading redness. If the red area continues to expand after the first 48 hours, growing larger rather than smaller, this could indicate a secondary infection rather than a simple inflammatory reaction. Normal reactions peak and then shrink. Infections expand.
Purulent drainage. Any pus, cloudy fluid, or greenish discharge from the injection site signals infection. This requires medical evaluation and likely antibiotic treatment. Do not attempt to drain or squeeze the area yourself.
Fever. A temperature above 100.4 degrees Fahrenheit (38 degrees Celsius) developing after an injection site reaction suggests systemic infection. Simple injection site reactions do not cause fever. If you have a fever alongside a worsening injection site, seek medical care promptly.
Symptoms lasting beyond 7 days. A reaction that has not improved at all after a full week of appropriate home treatment needs professional evaluation. Persistent reactions can indicate an abscess forming, a foreign body reaction, or a hypersensitivity that home measures cannot resolve.
Signs of allergic reaction (seek emergency care)
These symptoms require immediate medical attention. Hives (urticaria) appearing beyond the injection site, spreading across the body. Facial swelling, particularly around the eyes, lips, or tongue. Difficulty breathing or a sensation of throat tightening. Dizziness, rapid heartbeat, or feeling like you might pass out. These are signs of anaphylaxis or a severe allergic reaction, and they constitute a medical emergency.
True allergic reactions to tirzepatide are extremely rare. The SURPASS trials documented very few cases, and none were fatal. But rare does not mean impossible. Knowing the warning signs and having a plan, including knowing the location of the nearest emergency department, is responsible preparation. This is especially important for people with a history of medication allergies. SeekPeptides emphasizes the importance of safety awareness throughout all peptide protocols, and injection site monitoring is a fundamental part of safe research practices.
Reactions that worsen with dose escalation
Tirzepatide treatment typically involves gradual dose increases. If injection site reactions consistently worsen with each dose escalation, this may indicate dose-dependent immunogenicity, meaning your body produces more antibodies against higher concentrations of the peptide. Discuss this pattern with your prescriber. Options include slower dose escalation, longer intervals between dose increases, or switching to an alternative medication. The microdosing tirzepatide approach offers a way to escalate more gradually for people with sensitive responses.
Repeated reactions at the same site
If one particular injection area (say, the left lower abdomen) consistently produces worse reactions than other areas, stop using that site. Some individuals develop localized hypersensitivity in specific anatomical regions. Avoiding that area entirely while continuing to rotate through other sites often resolves the problem. Review alternative injection sites to expand your rotation options.
Tirzepatide vs semaglutide injection site reactions
Many researchers wonder whether switching medications would eliminate injection site issues. The comparison between these two GLP-1 medications reveals some interesting differences.
Semaglutide injection site reactions occur in approximately 1.4 percent of patients on Ozempic and 2.4 percent on Wegovy. Tirzepatide injection site reactions occur in approximately 2 to 6 percent of patients across the SURPASS trials. On pure numbers, semaglutide appears to cause fewer injection site reactions. But the comparison is not straightforward.
Tirzepatide is a larger, more complex molecule than semaglutide. It targets both GIP and GLP-1 receptors, while semaglutide targets only GLP-1. The dual-agonist structure means a larger synthetic peptide, which may explain the higher rate of antibody formation and subsequent injection site reactions. A detailed semaglutide versus tirzepatide side effects comparison covers the full spectrum of differences beyond just injection site reactions.
However, the clinical significance of this difference is debatable. Both medications produce overwhelmingly mild, self-limiting injection site reactions. Neither medication produces severe reactions at clinically meaningful rates. And for many people, the superior efficacy of tirzepatide for weight loss and metabolic outcomes outweighs the slightly higher chance of a temporary skin reaction.
The formulation matters too. Semaglutide in pre-filled pens (Ozempic, Wegovy) uses a different formulation than compounded semaglutide, which uses different excipients than compounded tirzepatide. People who react to one formulation may not react to another. If you have tried compounded semaglutide without issues, it does not guarantee you will tolerate compounded tirzepatide, because the active molecules, concentrations, and excipients differ.
Some researchers who experience injection site reactions on tirzepatide try switching to semaglutide and find the problem resolves. Others find the opposite, that semaglutide causes reactions and tirzepatide does not. Individual immune responses vary dramatically. The guide to switching from tirzepatide to semaglutide covers the practical aspects of making this transition, including dose conversion charts to maintain therapeutic equivalence.
For those considering alternatives to injections entirely, oral and sublingual formulations are emerging options. Oral tirzepatide versus injectable comparisons, tirzepatide drops, and orally disintegrating tablets bypass the injection site entirely, eliminating local reactions as a concern. These formulations come with their own trade-offs in terms of bioavailability and absorption, but for people who consistently struggle with injection site reactions, they represent a viable alternative path.

Do injection site reactions mean tirzepatide is not working
No. This is one of the most important points in this entire guide.
The presence or absence of injection site reactions has no correlation with the effectiveness of tirzepatide. The Phase 3 trial data is unequivocal on this point. Anti-drug antibodies, which are strongly associated with injection site reactions, had no measurable effect on the pharmacokinetics, efficacy, or weight loss outcomes of tirzepatide treatment.
Patients with antibodies lost the same amount of weight. Their HbA1c improvements were the same. Their metabolic outcomes were identical to patients without antibodies. The immune system can tag tirzepatide as foreign, create antibodies against it, trigger local skin reactions, and the medication still works exactly as intended on GIP and GLP-1 receptors.
Think of it this way. The injection site reaction is a local event. The therapeutic effect is a systemic event. The medication leaving the injection site and entering the bloodstream is what produces weight loss, appetite suppression, and metabolic improvements. A red bump at the injection site does not prevent the medication from reaching the bloodstream and doing its job.
If you are experiencing injection site reactions but your appetite suppression is kicking in, your weight loss is progressing on schedule, and your metabolic markers are improving, the injection site reaction is a nuisance to manage, not a sign of treatment failure. Continue your protocol while implementing the prevention and treatment strategies outlined above.
If, on the other hand, you are experiencing injection site reactions AND your treatment is not producing expected results, the injection site reaction is probably coincidental to the lack of results. Look at other factors instead. Review your troubleshooting guide for when tirzepatide stops working. Check your diet plan and food choices. Evaluate whether your dose needs adjustment. The injection site reaction and the treatment efficacy are independent variables.
Injection site reactions and compounded tirzepatide
Compounded tirzepatide introduces variables that brand-name formulations do not. Understanding these differences matters for managing injection site reactions effectively.
Brand-name Mounjaro and Zepbound are manufactured under strict pharmaceutical controls with consistent formulations, concentrations, and delivery devices. Compounded tirzepatide, by contrast, varies by pharmacy. Different compounding pharmacies use different excipients, different concentrations, different vial sizes, and different grades of bacteriostatic water. Each of these variables can influence injection site reaction frequency.
The reconstitution process itself adds a variable. When you mix lyophilized tirzepatide with bacteriostatic water, the concentration you achieve depends on how much water you add. A 10 mg vial reconstituted with 1 mL of water produces a 10 mg/mL solution. The same vial with 2 mL produces 5 mg/mL. The more dilute solution requires a larger injection volume to deliver the same dose, and larger volumes tend to cause more tissue displacement and more reaction at the injection site.
Benzyl alcohol, the preservative in bacteriostatic water, is a known skin irritant for some individuals. People who react to benzyl alcohol may find that switching to sterile water (without preservative) for single-use reconstitution eliminates their injection site reactions entirely. The trade-off is that sterile water lacks the antimicrobial preservative, so multi-dose vials become a contamination risk without benzyl alcohol. Single-use reconstitution with sterile water solves both problems but requires more preparation.
Pharmacy-specific formulations also matter. Some compounding pharmacies like Empower Pharmacy offer tirzepatide combined with additional ingredients such as glycine and B12, niacinamide, or methylcobalamin. These combination products contain additional ingredients that could independently trigger or amplify injection site reactions. If you experience reactions on a combination product, switching to plain tirzepatide (without additives) can help determine whether the tirzepatide itself or the added compounds are responsible.
Storage handling also affects compounded tirzepatide in ways that influence injection site reactions. Improper temperature management can cause peptide degradation, creating breakdown products that may be more immunogenic than intact tirzepatide. Expired or improperly stored tirzepatide has a higher chance of causing injection site issues. Always verify that your compounded product has been stored properly and is within its expiration date.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers comprehensive resources for understanding compounding variables, storage requirements, and reconstitution best practices. Their evidence-based guides help members navigate the complexities that generic resources often overlook.
Injection site reactions during dose escalation
Dose escalation is a standard part of tirzepatide treatment. Most protocols start at 2.5 mg and gradually increase to 5 mg, 7.5 mg, 10 mg, 12.5 mg, or 15 mg over weeks to months. Each dose increase changes the dynamics of the injection site reaction equation.
Higher doses mean more peptide per injection. More peptide means a stronger potential immune response at the injection site. Some researchers notice that injection site reactions they had not experienced at lower doses begin appearing at higher doses. Others find the opposite, that their body adapts over time and reactions that occurred at 2.5 mg disappear by the time they reach 7.5 mg.
The relationship between dose and reaction is not linear. It depends on antibody development, injection technique consistency, site rotation discipline, and individual immune system behavior. Following a structured dosing progression chart with adequate time at each dose level gives your body the best chance to adapt.
If reactions worsen significantly with each dose increase, consider the microdosing approach. Instead of jumping from 5 mg to 7.5 mg in one step, some researchers use intermediate doses (5.5 mg, 6 mg, 6.5 mg, 7 mg) to give the body more time to adjust. This requires compounded tirzepatide with flexible dosing, which is another advantage of the compounded tirzepatide dosage calculator approach over fixed-dose pens.
The escalation timeline matters too. Standard protocols allow 4 weeks at each dose before increasing. If you are experiencing injection site reactions at a given dose, extending that period to 6 or 8 weeks before escalating gives your body additional time to develop tolerance. There is no clinical penalty for a slower escalation. The full effects of tirzepatide develop over months regardless of escalation speed, and a slower climb often means a more comfortable experience.
The psychological impact of injection site reactions
This section gets overlooked, but it matters. Injection site reactions can create anxiety about future injections. The cycle goes like this: you experience a reaction, you dread the next injection, the dread causes you to tense up during injection, the tension causes poor technique, and the poor technique causes a worse reaction. A self-fulfilling prophecy.
Breaking this cycle starts with perspective. Remember the data. Over 94 percent of tirzepatide users never experience injection site reactions. Among those who do, the reactions are mild and self-limiting. No severe reactions were documented in the clinical trials. The injection site bump will resolve. It always does.
Practical steps help too. Inject in front of a mirror so you can see what you are doing. Use the pinch technique to lift skin away from muscle. Breathe slowly during the injection. Take your time. Rushing leads to mistakes, and mistakes lead to worse reactions. Following a consistent injection schedule at the same time each week helps build routine and reduces injection-related anxiety.
Some people find it helpful to apply a numbing cream (lidocaine 4 percent) to the injection site 30 minutes before injecting. This does not prevent the delayed reaction, but it eliminates the immediate sting, which for many people is the trigger for injection anxiety. If the injection itself is painless, the anticipation and dread diminish dramatically.
Context helps with anxiety too. Remind yourself that you are experiencing the same reactions that millions of GLP-1 users manage every week without issue. The forums and communities are full of stories from people who panicked about their first injection site reaction, implemented the management strategies, and forgot it was ever a concern within a few weeks. Connecting with others on the same journey through communities like SeekPeptides provides reassurance that comes from shared experience rather than clinical data alone.
Reframing also matters. An injection site reaction is not your body rejecting the medication. It is your immune system doing its job, identifying something new and mounting a proportional response. That same immune system will often calm down within weeks as it becomes familiar with the weekly tirzepatide dose. The temporary inconvenience of a red, itchy bump is a small price for the metabolic benefits that tirzepatide delivers. Keeping a record of your before and after results alongside your injection site notes creates a powerful visual reminder of why the minor discomfort is worth it.
Partner or friend support can make a practical difference as well. If you have someone who can administer the injection for you, the transfer of control can significantly reduce anxiety. You do not need to watch.
You do not need to push the plunger yourself. Just pinch the skin, look away, and let someone else handle the mechanical part. This approach is especially helpful during the first few weeks when both the medication and the injection process are new. Once confidence builds and you see that the reactions are manageable, self-injection becomes routine. Many researchers who began their tirzepatide journey with significant injection anxiety report that it disappeared entirely by the second month of treatment.
If injection anxiety is severe enough to interfere with your treatment adherence, discuss it with your healthcare provider. Anxiety about injections is common and nothing to be embarrassed about. Options include switching to non-injection formulations, working with a nurse educator to improve technique and confidence, or exploring the oral tirzepatide option. SeekPeptides members have access to community support where experienced researchers share practical tips for overcoming injection challenges.
Long-term injection site management
For people on tirzepatide for months or years, long-term injection site health becomes an important consideration. Chronic subcutaneous injection at the same sites can lead to lipohypertrophy (thickening of fatty tissue) or lipoatrophy (loss of fatty tissue), both of which affect medication absorption and increase reaction risk.
Lipohypertrophy creates lumpy, rubbery areas of thickened fat. Medication injected into these areas absorbs unpredictably, sometimes too fast (causing stronger systemic effects), sometimes too slowly (reducing efficacy). The affected tissue is also more prone to injection site reactions because its structure is altered.
Prevention is simple: rigorous site rotation. The rotation pattern described earlier (four abdominal quadrants plus thighs and upper arms, with at least 1 inch between adjacent injections) prevents lipohypertrophy when followed consistently. Examine your injection sites monthly by running your fingers over the areas you commonly use. If you feel any unusual thickening or lumps that were not there before, avoid that area for at least 3 months.
Scar tissue buildup is another long-term concern. Each injection creates a tiny wound that heals with a microscopic amount of scar tissue. Over hundreds of injections, this can accumulate. Scar tissue does not absorb medication well, leading to higher local concentrations, slower absorption, and more frequent reactions. Again, rotation is the prevention. If you have been on long-term GLP-1 treatment, expanding your injection site repertoire to include upper thighs and upper arms in addition to the abdomen provides more recovery time for each individual site.
Skin health maintenance also plays a role. Keep injection area skin moisturized and healthy. Dry, cracked skin is more reactive to injections. Avoid shaving directly at injection sites on injection day, as micro-abrasions from shaving can amplify the skin response to the injection. General skin health supports injection site tolerance over the long term.
Tracking your injection site history over months reveals patterns that single observations cannot. Some people find that certain abdominal quadrants consistently produce milder reactions. Others discover that thigh injections cause less redness but more bruising. These individual patterns are only visible with consistent tracking. Keeping a simple spreadsheet or journal entry with the date, site used, dose, and reaction severity (none, mild, moderate) gives you actionable data after 8 to 12 weeks. This data-driven approach to tirzepatide protocol optimization separates experienced researchers from those who are guessing.
Seasonal factors deserve mention too. Skin hydration varies with weather and climate. Winter months with dry indoor air tend to produce drier skin, which may be slightly more reactive to injections. Summer months with higher humidity and sweat can introduce irritants at the injection site. Adapting your skin prep and post-injection care to seasonal conditions is a small detail that matters over long treatment courses.
Finally, overall immune health influences injection site reactivity. Periods of illness, high stress, poor sleep, or significant dietary changes can temporarily alter immune function, making injection site reactions more or less pronounced. If you notice that injection site reactions worsen during stressful periods or when you are sleep-deprived, the connection is real. Supporting general immune health through adequate sleep, stress management, and good nutrition, including the right supplements alongside tirzepatide, creates the best conditions for minimal injection site reactivity.
Injection site reactions and other tirzepatide side effects
Injection site reactions rarely occur in isolation. Most tirzepatide users experience a combination of side effects, particularly during the first few weeks. Understanding how injection site reactions fit into the broader side effect landscape helps you manage everything more effectively.
Gastrointestinal side effects are far more common than injection site reactions. Nausea affects roughly 12 to 18 percent of tirzepatide users, depending on dose. Constipation and diarrhea each affect 5 to 10 percent. These GI side effects tend to be most pronounced during the first 2 to 4 weeks and during dose escalation periods. Injection site reactions follow a similar pattern, peaking early and diminishing with continued use.
Headaches from tirzepatide are another common companion to injection site reactions. Some researchers report a pattern where the injection site flares up, a mild headache develops within the first 24 hours, and both resolve by day 3. This combination likely reflects the systemic inflammatory response that tirzepatide triggers as the body adjusts, not just a local skin event. The broader GLP-1 headache pattern is well documented and usually temporary.
Fatigue can overlap with injection site discomfort, particularly during the first day after injection. The immune response that causes the local injection site reaction also uses metabolic energy. Combined with the caloric deficit that tirzepatide often induces through appetite suppression, some users feel genuinely depleted on injection day. The GLP-1 fatigue guide covers management strategies in detail. Staying hydrated, maintaining adequate protein intake, and timing injections for evenings so you can sleep through the initial response are all practical approaches.
Body aches and muscle pain round out the common early-treatment side effects. These systemic symptoms can make a simultaneous injection site reaction feel worse than it actually is. When your whole body aches and your injection site is red and itchy, the cumulative effect on your comfort level is significant. But it is temporary. Most people find that the combination of side effects peaks around weeks 2 to 3 and steadily improves from there.
For women specifically, changes in menstrual cycles can add another variable. Hormonal fluctuations during certain phases of the cycle may influence immune reactivity, potentially making injection site reactions more or less pronounced depending on timing. Some women report consistently worse injection site reactions during specific phases of their cycle. Adjusting injection timing relative to the menstrual cycle is a personalized strategy worth experimenting with.
Hair changes are reported by some tirzepatide users, though the connection is primarily related to rapid weight loss and caloric deficit rather than injection site immunology. However, if you are experiencing both injection site reactions and hair concerns, the combination can amplify worry about whether the medication is safe for you. It is. Both side effects are well-documented, temporary in most cases, and manageable with appropriate interventions.
The key insight is that injection site reactions exist within a constellation of early treatment effects. They are not a separate warning signal. They are part of the normal adaptation process as your body adjusts to a new medication. Managing them alongside other side effects, rather than in isolation, produces better outcomes and less anxiety. SeekPeptides provides comprehensive side effect management resources that address all of these interconnected responses as part of a holistic approach to peptide protocols.
Injection site care routine for tirzepatide users
Building a consistent injection site care routine minimizes reactions and maintains skin health over the long term. Here is a practical protocol that experienced researchers follow.
Pre-injection preparation (30 minutes before)
Remove the medication from the refrigerator and place it at room temperature. Take your antihistamine if you use one preventively. Gather supplies: alcohol swabs, syringe, sharps container, cold pack (for after). Choose your injection site using your rotation log. The stomach injection guide walks through site selection for abdominal injections specifically.
Injection protocol
Clean the site with an alcohol swab using circular motions moving outward from the center. Wait 30 seconds for the alcohol to dry completely. Pinch a skin fold. Insert the needle at the appropriate angle. Inject slowly over 5 to 10 seconds. Hold the needle in place for 5 to 10 seconds. Withdraw and apply gentle pressure with a clean gauze pad. Do not massage the injection site, as this can spread the medication laterally and increase the reaction area.
Post-injection care (first 2 hours)
Apply a cold compress to the injection site for 10 minutes immediately after injection. Monitor the site. If redness or swelling appears, reapply the cold compress every 30 to 60 minutes. Note the injection site location in your rotation log. If you are using a dosing chart to track your protocol, add the injection site notation to the same record for easy reference.
Days 1 through 3 post-injection
Monitor the injection site daily. Apply hydrocortisone cream twice daily if itching or redness develops. Continue cold compresses as needed. Avoid tight clothing that rubs against the injection site. Do not scratch, even if the itching is intense. Scratching breaks skin barrier integrity and can introduce bacteria, turning a simple reaction into an infection.
Days 4 through 7
If a nodule has formed, switch from cold to warm compresses at this point. The initial inflammatory phase is over, and warmth now promotes absorption and resolution. Continue monitoring. By day 7, the reaction should be completely or nearly resolved. If it is not, evaluate whether medical attention is needed.
This routine takes minimal time and effort but produces consistently better outcomes than handling reactions reactively. Prevention and proactive care always beat scrambling to treat a reaction that has already spiraled. Understanding your injection timing flexibility also helps if you need to adjust your schedule based on reaction patterns.
Frequently asked questions
How long does a tirzepatide injection site reaction last?
Most reactions resolve within 3 to 7 days. Mild redness and itching often clear within 24 to 48 hours. Deeper nodules or lumps may take up to 2 weeks to fully dissolve. If a reaction persists beyond 7 days without improvement, consult your healthcare provider. The timeline is similar to semaglutide injection site reactions in duration and severity.
Can I still take tirzepatide if I get injection site reactions?
In most cases, yes. Mild to moderate injection site reactions are not a reason to discontinue tirzepatide. Implement the prevention strategies (temperature management, site rotation, antihistamines) and continue your dosing protocol. Only discontinue if you experience severe allergic reactions, spreading infection, or if your healthcare provider recommends stopping.
Does the injection site reaction get better over time?
For many people, yes. The body often develops tolerance to tirzepatide over the first 4 to 8 weeks of treatment. Reactions that were noticeable during the early doses frequently diminish or disappear entirely with continued use. This is consistent with the immune system becoming accustomed to the regular presence of the peptide. The early weeks of tirzepatide treatment are typically when reactions are most frequent.
Is it normal for the injection site to itch for days?
Itching lasting 1 to 3 days is within the normal range for histamine-mediated injection site reactions. Itching beyond 3 to 4 days is less common and may warrant additional treatment (topical hydrocortisone, oral antihistamines). Persistent itching combined with spreading redness or warmth should be evaluated by a healthcare provider to rule out secondary infection. Taking appropriate supplements alongside tirzepatide and managing inflammation can help reduce discomfort.
Can I ice the injection site before injecting to reduce pain?
Yes, applying ice for 1 to 2 minutes before injection can numb the skin and reduce immediate injection pain. However, icing before injection does not prevent the delayed inflammatory reaction that causes redness, swelling, and itching hours later. For the best overall strategy, ice before injection (for pain) and apply cold compresses after (for reaction management).
Should I switch from tirzepatide to semaglutide if I keep getting injection site reactions?
Not necessarily as a first step. Try all prevention strategies first (temperature management, site rotation, antihistamines, technique adjustment). If reactions persist despite these measures, switching to semaglutide is a reasonable option. Some people react to one medication but not the other. The conversion chart helps ensure therapeutic equivalence during any switch.
Do injection site reactions mean I am allergic to tirzepatide?
Injection site reactions and true allergies are different. An injection site reaction is a localized inflammatory response at the injection point. A true allergy produces systemic symptoms (widespread hives, facial swelling, breathing difficulty). Most injection site reactions involve mild local immune activation, not full allergic sensitization. However, if you notice reactions getting progressively worse with each injection or developing systemic symptoms, allergic sensitization should be evaluated by an allergist.
Can tirzepatide injection site reactions cause permanent scarring?
Standard mild to moderate injection site reactions do not cause permanent scarring. Repeated injection into the same site without adequate rotation can cause tissue changes (lipohypertrophy or lipoatrophy), which may be long-lasting but are preventable with proper rotation. Following a systematic approach to injection site management prevents any permanent skin or tissue changes.
Are injection site reactions worse with compounded tirzepatide than brand-name Mounjaro?
Not necessarily, but the variables differ. Compounded tirzepatide may contain different excipients that some individuals react to. The reconstitution process introduces variables like concentration and preservative content. Some people react to the benzyl alcohol in bacteriostatic water rather than to the tirzepatide itself. If you experience reactions with compounded product, trying a different pharmacy formulation or switching reconstitution methods may resolve the issue. Compounding pharmacies like Priority Meds and ProRX each use slightly different formulations.
Can I drink alcohol while managing a tirzepatide injection site reaction?
Alcohol is a vasodilator, meaning it widens blood vessels and increases blood flow. Consuming alcohol within the first 24 hours of an injection site reaction could theoretically increase redness and swelling by increasing blood flow to the affected area. More importantly, alcohol impairs immune function and can slow the resolution of inflammatory responses. If you are managing an active injection site reaction, avoiding alcohol for the first 2 to 3 days is prudent. The complete guide to alcohol and tirzepatide covers the broader interaction between alcohol and GLP-1 treatment.
What is the best body area for tirzepatide injection to minimize reactions?
The abdomen is the most commonly used and best-studied injection site for subcutaneous GLP-1 medications. It typically offers the most consistent absorption and the most subcutaneous tissue for comfortable injection. However, some people find that the outer thigh produces fewer reactions than the abdomen, particularly if they have less abdominal fat. The upper arm is another option, though it can be more difficult to self-inject. Experimentation with different anatomical regions, while maintaining good injection site rotation, helps identify your personal sweet spot.
External resources
Tirzepatide-Induced Injection Site Reaction - PMC/Cureus Case Report
Mayo Clinic - Tirzepatide Side Effects and Dosage Information
Tirzepatide Immunogenicity Analysis - Journal of Clinical Endocrinology and Metabolism
Real-World Data Analysis of Tirzepatide in FDA Adverse Event Reporting System
For researchers serious about optimizing their peptide protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Their detailed safety guides, interaction databases, and expert-reviewed content help members approach peptide research with confidence and precision.
In case I do not see you, good afternoon, good evening, and good night. May your injection sites stay calm, your reactions stay mild, and your protocols stay effective.