Feb 8, 2026

Experienced researchers know something beginners often miss about compounded tirzepatide formulations. The addition of niacinamide is not just an arbitrary choice. It represents a deliberate formulation strategy that aims to stabilize the peptide molecule while potentially enhancing metabolic outcomes. When you see tirzepatide niacinamide listed on a compounding pharmacy label, you are looking at a combination designed to address both the practical challenges of peptide stability and the theoretical opportunity for synergistic metabolic benefits.
This is not the same as pharmaceutical-grade Mounjaro or Zepbound. It is a compounded variation that pairs the dual GIP/GLP-1 receptor agonist tirzepatide with vitamin B3 in its non-flushing form, niacinamide. The rationale extends beyond simple formulation chemistry into the realm of NAD+ precursor biology, mitochondrial function, and adipose tissue metabolism.
Understanding what this combination actually does, how the two molecules interact, and whether the theoretical benefits translate into real-world outcomes requires looking past marketing claims and into the biochemistry.
For researchers exploring peptides for fat loss or those comparing semaglutide vs tirzepatide, the niacinamide addition introduces a new variable worth understanding in depth.
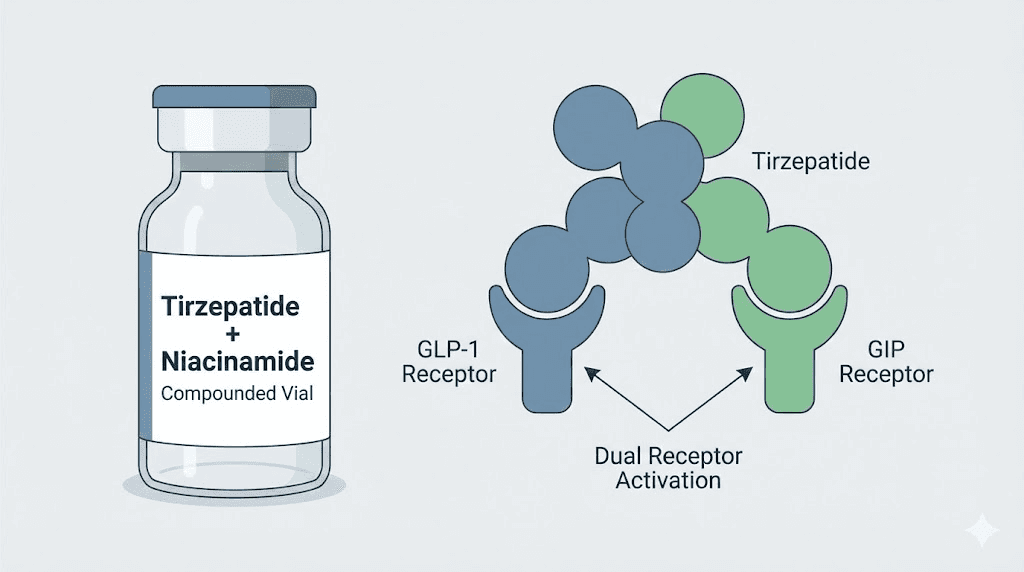
What is tirzepatide niacinamide?
Tirzepatide niacinamide is a compounded formulation that combines tirzepatide, a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, with niacinamide, the amide form of vitamin B3. This is not an FDA-approved combination. It is produced by 503A compounding pharmacies that customize peptide formulations based on prescriber specifications. The typical formulation includes 17mg/mL tirzepatide with 2mg/mL niacinamide in 4mL vials, though concentrations vary. Some pharmacies offer 8mg/mL tirzepatide with 2mg/mL niacinamide in 2.5mL vials. The addition of niacinamide serves multiple purposes within the formulation matrix.
First, niacinamide acts as a stabilizing agent. Peptides in solution can degrade over time through oxidation, aggregation, or hydrolysis. The presence of niacinamide may slow these degradation pathways, extending the viable shelf life of the reconstituted peptide. Second, niacinamide is a precursor to nicotinamide adenine dinucleotide (NAD+), a coenzyme central to cellular energy metabolism, mitochondrial function, and DNA repair. Researchers using peptide calculator tools to plan their protocols often encounter this combination and wonder whether the niacinamide component adds measurable benefit beyond stabilization. The answer is nuanced.
Tirzepatide itself is a powerful metabolic agent. Approved as Mounjaro for type 2 diabetes and Zepbound for chronic weight management, it demonstrated remarkable efficacy in the SURMOUNT-1 clinical trial, where participants achieved 16 to 22.5 percent body weight loss over 72 weeks depending on dose. The drug works by activating both GIP and GLP-1 receptors, a dual mechanism that reduces appetite, slows gastric emptying, improves insulin sensitivity, and enhances fat metabolism. GIP receptor activation appears to amplify the metabolic benefits beyond what GLP-1 agonism alone can achieve, which is why tirzepatide consistently outperforms semaglutide in head-to-head comparisons.
Niacinamide, on the other hand, does not directly activate incretin receptors. Its role is metabolic and cellular. As an NAD+ precursor, niacinamide supports the function of sirtuins, enzymes involved in metabolic regulation, inflammation control, and longevity pathways. NAD+ levels decline with age and metabolic dysfunction, and restoring them through supplementation has shown promise in animal models for improving insulin sensitivity, enhancing mitochondrial biogenesis, and promoting fat oxidation. In one study published in Molecular Nutrition and Food Research, nicotinamide supplementation protected mice against diet-induced obesity, increased energy expenditure, and induced the beiging of white adipose tissue, a process that converts energy-storing fat cells into energy-burning cells.
The combination of tirzepatide and niacinamide in a single injectable formulation is therefore an attempt to merge direct incretin receptor activation with broader metabolic support. Researchers exploring tirzepatide dosing for weight loss in units or reviewing tirzepatide dose chart protocols should recognize that compounded versions with additives like niacinamide are not identical to pharmaceutical formulations. The pharmacokinetics may differ slightly.
The side effect profile could be altered. And most importantly, there are no clinical trials specifically validating the efficacy or safety of this combination in humans.
Compounding pharmacies began offering tirzepatide niacinamide as an alternative when pharmaceutical-grade tirzepatide faced supply shortages and cost barriers. The FDA allows compounding of drugs during shortage periods under specific conditions. However, tirzepatide was removed from the FDA drug shortage list, triggering restrictions on compounded versions. Patients and researchers relying on compounded tirzepatide formulations faced transitions back to pharmaceutical products or continued use under narrower legal frameworks. Understanding the regulatory landscape is essential for anyone considering peptide sourcing or comparing injectable vs oral peptides.
How tirzepatide and niacinamide work together
The interaction between tirzepatide and niacinamide is not a direct biochemical synergy in the sense of one molecule enhancing the receptor affinity or signaling cascade of the other. Instead, the relationship is complementary.
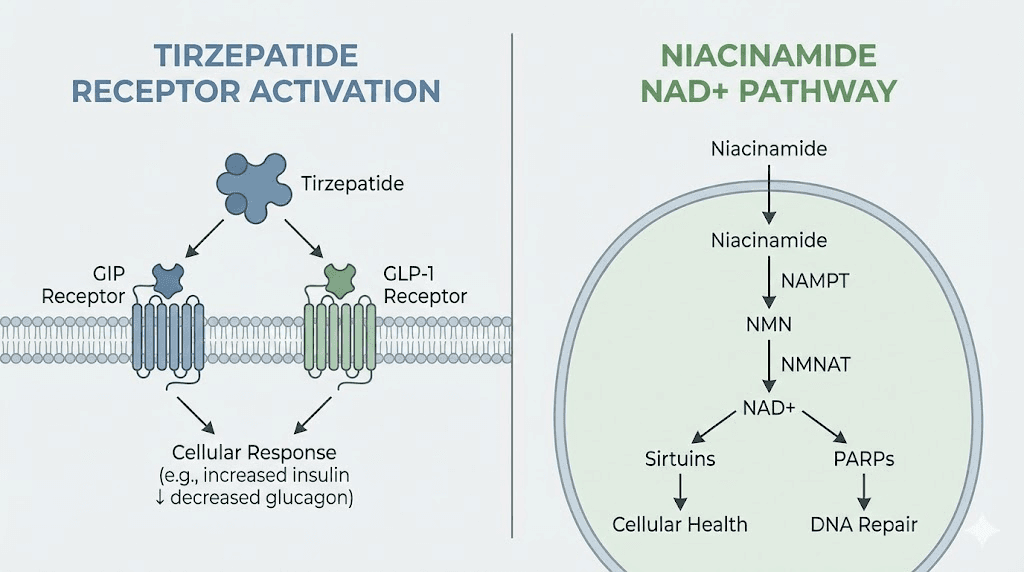
Tirzepatide operates through incretin receptor activation. It binds to both GIP and GLP-1 receptors, which are G-protein-coupled receptors expressed in pancreatic beta cells, the gastrointestinal tract, adipose tissue, and the central nervous system. When tirzepatide activates GLP-1 receptors, it stimulates glucose-dependent insulin secretion, suppresses glucagon release, slows gastric emptying, and reduces appetite through central pathways in the hypothalamus. GIP receptor activation adds an additional layer: it enhances insulin secretion, modulates fat metabolism in adipocytes, and may improve the body response to dietary fat intake. The dual agonism is why tirzepatide produces greater weight loss than semaglutide, a selective GLP-1 agonist, as seen in trials comparing the two agents.
Niacinamide works through entirely different pathways. Once absorbed, it is converted to NAD+ via the salvage pathway, primarily in the liver and other tissues. NAD+ is a coenzyme required for glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation. It is also a substrate for enzymes like sirtuins and poly(ADP-ribose) polymerases (PARPs), which regulate gene expression, DNA repair, and cellular stress responses. In the context of metabolism, NAD+ availability influences mitochondrial function, fat oxidation, and insulin sensitivity. Low NAD+ levels are associated with metabolic syndrome, obesity, and type 2 diabetes. Restoring NAD+ through precursors like niacinamide reactivates sirtuins and can reverse some of these metabolic deficits.
The potential synergy arises from the fact that tirzepatide and niacinamide target overlapping metabolic dysfunctions through different mechanisms. Tirzepatide reduces caloric intake and improves insulin signaling via incretin pathways. Niacinamide supports the cellular machinery that processes those calories, enhances mitochondrial efficiency, and may reduce inflammation in adipose tissue. In theory, this could lead to greater fat loss, improved energy levels, and fewer metabolic side effects compared to tirzepatide alone. However, this is speculation. No randomized controlled trial has tested tirzepatide with niacinamide against tirzepatide alone in humans.
Animal data provides some support for the concept. The study in Molecular Nutrition and Food Research demonstrated that nicotinamide supplementation in mice on a high-fat diet reduced body weight gain, increased energy expenditure, and upregulated genes involved in thermogenesis and fat oxidation, including PPARalpha and PGC1alpha.
The mice also showed increased mitochondrial protein expression in white adipose tissue, suggesting a shift toward a more metabolically active phenotype. If similar effects occur in humans, adding niacinamide to a tirzepatide regimen could theoretically amplify fat loss and improve metabolic flexibility. Researchers exploring peptides for belly fat or fat burning peptides for men often seek combinations that address multiple pathways simultaneously.
Another consideration is the impact on gastrointestinal side effects. Tirzepatide commonly causes nausea, vomiting, diarrhea, and constipation, particularly during dose escalation. These side effects are thought to result from slowed gastric emptying and direct effects on the gastrointestinal tract. Niacinamide has been studied for its anti-inflammatory properties in the gut. Some researchers hypothesize that it could mitigate GI distress by supporting mucosal integrity and reducing inflammation.
However, there is no clinical evidence supporting this claim in the context of tirzepatide use. Researchers comparing semaglutide with B12 or oral tirzepatide formulations may find similar additive strategies aimed at tolerability.
Absorption kinetics is another area where niacinamide could play a role. Some formulation scientists suggest that niacinamide may improve the subcutaneous absorption of tirzepatide by altering the local tissue environment or enhancing solubility. This is plausible but unproven. Pharmaceutical-grade tirzepatide is formulated with excipients carefully selected to optimize bioavailability. Whether compounded formulations with niacinamide achieve equivalent or superior absorption is unknown. Researchers using peptide reconstitution calculator tools should note that altering the formulation can affect reconstitution protocols and storage requirements.
It is also important to address a potential concern. Niacin, a closely related form of vitamin B3, is known to transiently impair insulin sensitivity and raise blood glucose levels. In clinical trials, niacin therapy increased fasting glucose by 4 to 11 mg/dL and hemoglobin A1c by 0.2 to 0.3 percent. This effect is thought to result from increased free fatty acid release from adipose tissue and hepatic glucose production. Niacinamide, the form used in compounded tirzepatide, does not cause the same degree of insulin resistance as niacin, but high doses can still affect glucose metabolism. When combined with tirzepatide, which improves insulin sensitivity, the net effect is likely neutral or positive, but this has not been formally studied. Researchers tracking how long does it take for tirzepatide to work should monitor glucose levels closely if using a niacinamide-containing formulation.
The science behind niacinamide in compounded tirzepatide
Understanding why niacinamide is included in compounded tirzepatide requires exploring its biochemical roles and the preclinical evidence suggesting metabolic benefits.
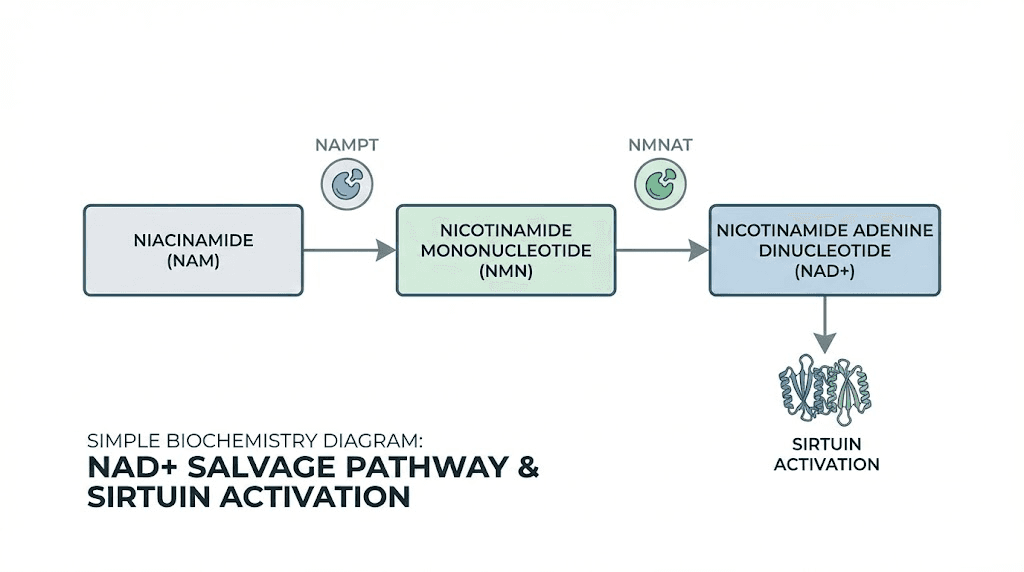
Niacinamide is one of two major forms of vitamin B3, the other being niacin (nicotinic acid). Both are precursors to NAD+, but they differ in side effect profiles and metabolic pathways. Niacin causes vasodilation and flushing due to activation of the GPR109A receptor, while niacinamide does not. This makes niacinamide the preferred form for long-term supplementation and compounding applications. Once ingested or injected, niacinamide is converted to nicotinamide mononucleotide (NMN) and then to NAD+ through the salvage pathway. NAD+ is essential for cellular respiration, with roles in glycolysis, the Krebs cycle, and the electron transport chain. It is also a substrate for sirtuins, a family of NAD+-dependent deacetylases that regulate metabolism, inflammation, and aging.
Sirtuins, particularly SIRT1 and SIRT3, have been extensively studied for their roles in metabolic health. SIRT1 activation improves insulin sensitivity, enhances fat oxidation, and promotes mitochondrial biogenesis. SIRT3, localized to mitochondria, regulates oxidative metabolism and protects against metabolic dysfunction. NAD+ availability is a limiting factor for sirtuin activity. As NAD+ levels decline with age, obesity, and metabolic disease, sirtuin function diminishes, contributing to insulin resistance, mitochondrial dysfunction, and fat accumulation. Restoring NAD+ through precursors like niacinamide reactivates sirtuins and can reverse some of these metabolic deficits.
The study in Molecular Nutrition and Food Research is one of the most relevant pieces of preclinical evidence for niacinamide use in metabolic contexts. Researchers fed mice a high-fat diet and supplemented one group with nicotinamide in drinking water. The supplemented mice gained significantly less weight, had lower fat mass, and showed increased energy expenditure compared to controls. Gene expression analysis revealed upregulation of thermogenic markers in white adipose tissue, including uncoupling protein 1 (UCP1), which is characteristic of brown adipose tissue. This process, known as beiging, converts white fat cells into cells capable of burning energy as heat rather than storing it. The study also found increased expression of PPARalpha and PGC1alpha, transcription factors involved in fat oxidation and mitochondrial biogenesis. These findings suggest that niacinamide supplementation can shift adipose tissue toward a more metabolically active state.
Another line of evidence comes from studies on NAD+ and insulin resistance. In obese and insulin-resistant rodents, NAD+ levels are reduced in liver and adipose tissue. Supplementation with NAD+ precursors, including niacinamide and nicotinamide riboside, has been shown to improve glucose tolerance, reduce liver fat, and enhance mitochondrial function.
A study in Diabetes found that nicotinamide riboside supplementation in humans with obesity and prediabetes improved insulin sensitivity in skeletal muscle, though the effects were modest and variable. This suggests that NAD+ repletion can have metabolic benefits in humans, though the magnitude depends on baseline NAD+ status and the specific precursor used.
The relevance to tirzepatide lies in the potential for additive or synergistic effects. Tirzepatide improves insulin sensitivity primarily through incretin receptor-mediated pathways, reducing glucagon, enhancing insulin secretion, and slowing gastric emptying. Niacinamide could enhance these effects by improving mitochondrial function and cellular energy metabolism, allowing tissues to respond more effectively to the metabolic signals initiated by tirzepatide. For researchers using tirzepatide dosage in units protocols or tracking tirzepatide before and after results, the addition of niacinamide could theoretically improve outcomes, though this remains unproven in controlled trials.
Niacinamide also has anti-inflammatory properties. Chronic low-grade inflammation in adipose tissue is a hallmark of obesity and insulin resistance. Inflammatory cytokines like TNF-alpha and IL-6 interfere with insulin signaling and promote fat accumulation. Niacinamide has been shown to reduce inflammation in various models, potentially through inhibition of NF-kappaB signaling and modulation of immune cell function. If these effects occur in adipose tissue during tirzepatide therapy, they could reduce the inflammatory burden that hinders fat loss and metabolic improvement. Researchers exploring peptides for autoimmune diseases or peptides for hormone balance often seek agents with anti-inflammatory profiles.
One potential concern is the dose. Compounded tirzepatide typically contains 2mg/mL niacinamide. A standard weekly injection of 10mg tirzepatide from a 17mg/mL formulation would deliver approximately 1.2mg of niacinamide. This is far below the doses used in most supplementation studies, which range from 100mg to 1000mg daily. Whether such a low dose provides meaningful metabolic benefit is questionable. However, subcutaneous injection may result in different tissue-level concentrations compared to oral supplementation, and even small amounts of NAD+ precursor could have local effects in adipose tissue near the injection site.
Researchers using peptide injection pen tools should consider whether the niacinamide dose in their formulation is pharmacologically relevant.
Another consideration is the potential for niacinamide to affect glucose metabolism. While niacinamide is generally considered neutral with respect to insulin sensitivity, high doses can increase hepatic glucose production and free fatty acid levels. In the context of tirzepatide therapy, which strongly improves insulin sensitivity, this is unlikely to be a problem, but it is worth monitoring in individuals with diabetes or prediabetes. Researchers comparing mazdutide vs tirzepatide or retatrutide vs semaglutide should be aware that different peptide formulations may have unique metabolic effects when combined with additives.
Tirzepatide niacinamide dosage and titration protocol
Dosing tirzepatide niacinamide follows the same titration schedule used for pharmaceutical-grade tirzepatide, with adjustments based on formulation concentration and individual tolerance.
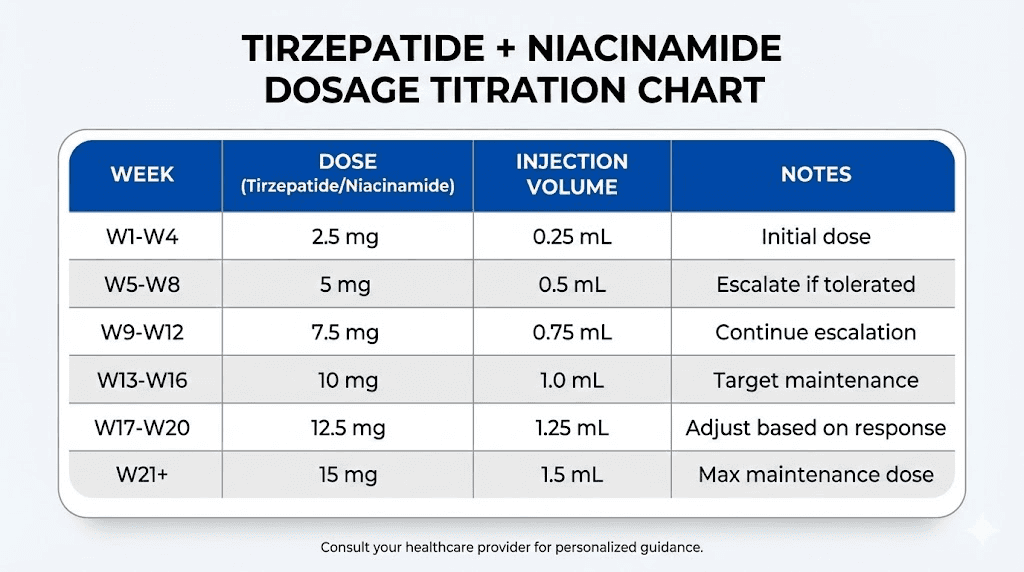
The standard starting dose is 2.5mg of tirzepatide administered subcutaneously once weekly. This dose is maintained for at least four weeks to allow the body to adapt to the medication and to minimize gastrointestinal side effects. After four weeks, the dose is increased to 5mg weekly. Subsequent increases occur every four weeks, moving to 7.5mg, 10mg, 12.5mg, and finally 15mg, which is the maximum approved dose for weight management. The pace of titration can be slowed if side effects are problematic or if weight loss is satisfactory at a lower dose. Some individuals achieve their goals at 5mg or 7.5mg and do not require further escalation.
For a compounded formulation containing 17mg/mL tirzepatide and 2mg/mL niacinamide, the injection volumes are as follows. A 2.5mg dose requires 0.15mL. A 5mg dose requires 0.29mL. A 7.5mg dose requires 0.44mL. A 10mg dose requires 0.59mL. A 12.5mg dose requires 0.74mL. A 15mg dose requires 0.88mL. These volumes are small and should be administered using an insulin syringe with 0.01mL graduations for accuracy. Researchers using semaglutide dosage calculator or cjc-1295 dosage calculator tools will find similar precision required for peptide dosing.
If using an 8mg/mL tirzepatide formulation, the volumes differ. A 2.5mg dose requires 0.31mL. A 5mg dose requires 0.63mL. A 7.5mg dose requires 0.94mL. A 10mg dose requires 1.25mL. This formulation is less concentrated, so injection volumes are larger. Some individuals prefer lower concentration formulations to reduce injection site discomfort, while others prefer higher concentrations to minimize injection volume. The niacinamide concentration is typically 2mg/mL regardless of tirzepatide concentration, so the total niacinamide dose per injection increases proportionally with tirzepatide dose.
Injections are administered on the same day each week. Consistency is important for maintaining stable drug levels and predictable appetite suppression. If a dose is missed, it should be taken within four days of the scheduled dose. If more than four days have passed, skip the missed dose and resume the schedule with the next planned injection. Do not double doses. Researchers tracking how long does it take for tirzepatide to suppress appetite should note that missing doses can disrupt appetite control and lead to rebound hunger.
The injection site should be rotated to prevent lipohypertrophy or lipoatrophy, which are changes in subcutaneous fat that can affect absorption. Common injection sites include the abdomen (at least two inches away from the navel), the front or side of the thighs, and the back of the upper arms. The abdomen is generally preferred because absorption is most consistent in this area. Avoid injecting into areas with scars, bruises, or visible blood vessels. Clean the skin with an alcohol swab and allow it to dry before injection. Pinch the skin to create a fold, insert the needle at a 90-degree angle, and inject slowly. After injecting, release the skin fold and withdraw the needle. Do not rub the injection site, as this can increase absorption variability.
Some individuals experience injection site reactions, including redness, swelling, or itching. These are usually mild and resolve within a few days. If reactions are persistent or severe, consider changing injection sites more frequently or using a different formulation. Niacinamide is generally well tolerated at the doses present in compounded tirzepatide, but some individuals may experience mild flushing or warmth shortly after injection.
This is not dangerous and typically subsides within minutes. Researchers comparing how to mix peptides with bac water or how much bacteriostatic water to add to peptides should follow proper reconstitution protocols to minimize contamination and ensure accurate dosing.
Dose adjustments should be based on efficacy and tolerability. If weight loss stalls at a given dose, increasing the dose may restart progress. However, if side effects are limiting, staying at the current dose or reducing the dose is appropriate. The goal is sustainable weight loss with tolerable side effects, not maximal doses at all costs. Some individuals find that microdosing tirzepatide protocols, which involve smaller, more frequent doses, provide better side effect control while maintaining efficacy. However, this approach is off-label and not supported by clinical trial data.
Duration of therapy is another consideration. Clinical trials of tirzepatide extended up to 72 weeks, with continued weight loss throughout the study period. Real-world use often extends beyond this timeframe, particularly for individuals with significant obesity or metabolic dysfunction. Long-term safety data beyond two years is limited. Some prescribers recommend periodic breaks or dose reductions after achieving weight loss goals, while others advocate for continuous therapy to prevent weight regain. Researchers exploring peptides for menopause weight loss or why not losing weight on semaglutide should consider that peptide therapy is most effective when combined with lifestyle modification and continued long-term.
How to inject tirzepatide niacinamide
Proper injection technique ensures consistent absorption, minimizes discomfort, and reduces the risk of complications.
Gather your supplies before beginning. You will need the vial of compounded tirzepatide niacinamide, an insulin syringe (typically 0.5mL or 1mL with 0.01mL graduations), alcohol swabs, and a sharps disposal container. If the vial has been refrigerated, allow it to reach room temperature for 30 to 60 minutes before injecting. Cold injections can be uncomfortable and may reduce absorption slightly. Wash your hands thoroughly with soap and water.
Choose your injection site. The abdomen is the most common choice. Select a spot at least two inches away from the navel and avoid areas with visible veins, scars, or previous injection sites. The front or outer thighs are another option, though absorption may be slightly slower in this area. The back of the upper arms can be used if you have assistance or are flexible enough to reach comfortably. Rotate injection sites with each dose to prevent tissue changes that can affect absorption. Researchers using BPC-157 dosage calculator or TB-500 dosage calculator tools for other peptides will find similar rotation strategies beneficial.
Clean the injection site with an alcohol swab using a circular motion, starting at the center and moving outward. Allow the skin to air dry completely. Injecting into wet skin can cause stinging. While the skin dries, prepare the syringe. Clean the rubber stopper on the vial with a fresh alcohol swab. Draw air into the syringe equal to the volume of tirzepatide you plan to inject. Insert the needle into the vial through the rubber stopper and push the air in. This equalizes pressure and makes drawing the liquid easier. Invert the vial so the needle tip is submerged in the liquid. Slowly pull back on the plunger to draw the correct volume. Check for air bubbles. If bubbles are present, tap the syringe gently to move them to the top, then push them back into the vial and redraw if necessary. Verify the dose by checking the syringe markings carefully.
Remove the needle from the vial and replace the needle cap if needed. Pinch the skin at the injection site between your thumb and forefinger to create a fold of tissue about one to two inches wide. This ensures you inject into subcutaneous fat rather than muscle. Hold the syringe like a pencil or dart, with your dominant hand. Insert the needle at a 90-degree angle to the skin with a quick, smooth motion. The needle should penetrate fully into the subcutaneous tissue. Do not angle the needle unless you are very lean, in which case a 45-degree angle may be more appropriate. Once the needle is inserted, release the skin fold if desired, though some individuals prefer to maintain the fold throughout injection.
Push the plunger slowly and steadily to inject the medication. Aim for a duration of five to ten seconds for the full injection. Injecting too quickly can cause discomfort and may increase the risk of leakage. Once the plunger is fully depressed, pause for a few seconds before withdrawing the needle. This allows the medication to disperse into the tissue and reduces leakage. Withdraw the needle at the same angle it was inserted. Do not rub the injection site, as this can alter absorption and cause irritation. If a small drop of liquid or blood appears, gently blot it with a clean tissue or gauze.
Dispose of the used syringe immediately in a sharps container. Never recap needles, as this increases the risk of needlestick injury. Sharps containers are available at pharmacies and should be sealed and disposed of according to local regulations when full. Do not dispose of needles in regular trash or recycling bins. Researchers tracking how to store peptides after reconstitution should follow similar safety protocols for all injectable peptides.
After injection, monitor for any unusual reactions. Mild redness, swelling, or itching at the injection site is common and typically resolves within 24 to 48 hours. Applying a cool compress can reduce discomfort. If you experience severe pain, significant swelling, or signs of infection such as warmth, redness spreading beyond the injection site, or fever, contact a healthcare provider. Allergic reactions to tirzepatide are rare but can include rash, hives, difficulty breathing, or swelling of the face or throat. These require immediate medical attention.
Some individuals experience a brief sensation of warmth or flushing after injecting tirzepatide niacinamide. This is likely due to the niacinamide component and is not harmful. It typically subsides within minutes. If flushing is bothersome, try injecting at a slightly slower rate or using a colder injection site. Researchers using sublingual peptides or integrative peptides BPC-157 oral supplement formulations may find subcutaneous injection more predictable in terms of bioavailability and side effects.
Storage and handling for tirzepatide niacinamide
Proper storage is essential to maintain peptide stability and efficacy.
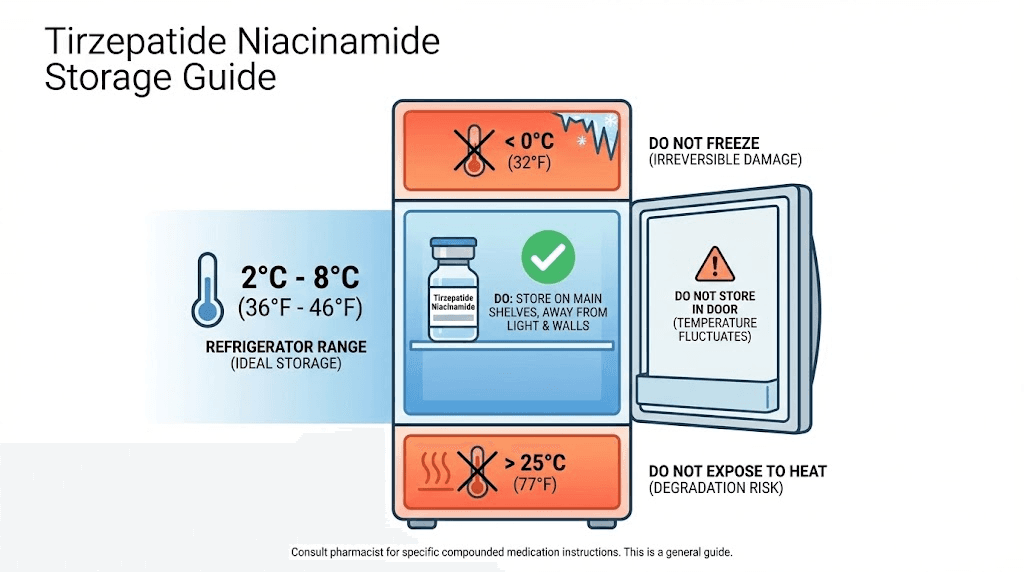
Compounded tirzepatide niacinamide should be stored in a refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) immediately upon receipt. Do not freeze the medication. Freezing can cause the peptide to denature, rendering it ineffective. If the vial has been frozen accidentally, discard it and obtain a replacement. Store the vial in its original packaging to protect it from light. Peptides are sensitive to photodegradation, and exposure to light can reduce potency over time. Keep the vial in the back of the refrigerator, away from the door, to minimize temperature fluctuations.
Once a vial is in use, it can be kept at room temperature (up to 77 degrees Fahrenheit or 25 degrees Celsius) for short periods, such as during travel or immediately before injection. However, prolonged exposure to room temperature should be avoided. If you plan to travel with tirzepatide niacinamide, use an insulated cooler with ice packs to maintain a cool temperature. Avoid direct contact between the vial and ice packs, as extreme cold can damage the peptide. During security screening at airports, inform personnel that you are carrying temperature-sensitive medication and keep it in your carry-on luggage to prevent exposure to cargo hold temperatures. Researchers exploring how long does tirzepatide last in the fridge should note that compounded formulations may have shorter stability periods than pharmaceutical-grade products.
Check the expiration date on the vial. Compounded peptides typically have a shorter shelf life than mass-produced pharmaceuticals, often ranging from 30 to 90 days from the date of compounding. Do not use the medication after the expiration date, as potency cannot be guaranteed. If the expiration date is not clearly marked, contact the compounding pharmacy for clarification. Some pharmacies include a beyond-use date rather than a traditional expiration date. This date reflects the maximum time the compounded preparation is expected to remain stable under proper storage conditions.
Inspect the vial before each use. The solution should be clear and colorless or slightly yellow. If you notice cloudiness, particulate matter, discoloration, or any change in appearance, do not use the medication. These changes may indicate contamination or degradation. Contact the pharmacy for a replacement. Do not shake the vial, as vigorous agitation can cause the peptide to aggregate or denature. If the solution needs to be mixed, gently swirl the vial rather than shaking it.
Researchers using lyophilized vs liquid peptides should be aware that reconstituted lyophilized peptides have different storage requirements and may require refrigeration immediately after reconstitution.
Do not transfer the medication to another container. Use the vial as provided by the pharmacy. Transferring to a different container can introduce contamination and makes it difficult to track the beyond-use date. If you need to carry a dose with you, draw it into a syringe immediately before leaving and keep the syringe refrigerated or in a cooler until use. Pre-filled syringes should be used within 24 hours to minimize the risk of contamination or degradation.
Once a vial is punctured with a needle, the risk of contamination increases. Compounding pharmacies typically recommend discarding multi-dose vials after 28 days of first use, even if medication remains. This reduces the risk of bacterial growth in the vial. If you are using a single-dose vial, discard any unused portion after drawing your dose. Do not save partial vials for future use unless the pharmacy specifically indicates the vial is designed for multi-dose use and includes a preservative like benzyl alcohol. Researchers comparing research vs pharmaceutical peptides should note that pharmaceutical-grade products often have more robust preservative systems and longer shelf lives.
If you are storing multiple vials, label them clearly with the date of first use and the discard date. This prevents confusion and ensures you use the oldest vials first. Keep vials out of reach of children and pets. In case of accidental ingestion or exposure, contact a poison control center or healthcare provider immediately.
Researchers tracking peptide cost calculator data should factor in the cost of discarded medication due to expiration when budgeting for peptide protocols.
Tirzepatide niacinamide side effects and safety
Tirzepatide niacinamide carries the same side effect profile as pharmaceutical-grade tirzepatide, with the addition of potential niacinamide-related effects.
The most common side effects are gastrointestinal. Nausea occurs in approximately 20 to 30 percent of individuals during dose escalation. It is usually mild to moderate and improves with continued use. Eating smaller, more frequent meals and avoiding high-fat or spicy foods can reduce nausea. Vomiting is less common, occurring in 5 to 10 percent of users. Diarrhea and constipation are also frequent, with each affecting 10 to 20 percent of individuals. These symptoms are thought to result from slowed gastric emptying and altered gut motility. Increasing fluid intake, consuming fiber-rich foods, and taking over-the-counter anti-diarrheal or laxative medications as needed can help manage these symptoms. Researchers exploring peptides for gut health or does tirzepatide make you tired should note that gastrointestinal side effects can impact quality of life and adherence.
Abdominal pain or discomfort is another common complaint. This is usually transient and resolves within a few days of dose escalation. If abdominal pain is severe, persistent, or accompanied by fever, it could indicate pancreatitis, a rare but serious complication. Acute pancreatitis has been reported in clinical trials of tirzepatide, though the incidence is low, approximately 0.2 percent. Symptoms include severe upper abdominal pain that may radiate to the back, nausea, vomiting, and fever. If these symptoms occur, discontinue tirzepatide and seek medical attention immediately. Individuals with a history of pancreatitis should use tirzepatide with caution or consider alternative therapies.
Injection site reactions are common but usually mild. Redness, swelling, itching, or bruising at the injection site occur in 5 to 10 percent of users. These reactions typically resolve within a few days and do not require treatment. Rotating injection sites and using proper technique can minimize these effects. In rare cases, individuals may develop lipohypertrophy (thickening of subcutaneous fat) or lipoatrophy (thinning of subcutaneous fat) at injection sites. These changes can affect absorption and should be reported to a healthcare provider if they occur.
Fatigue is reported by some individuals, particularly during the first few weeks of therapy. This may be related to reduced caloric intake, altered metabolism, or direct effects of the medication. Ensuring adequate sleep, staying hydrated, and consuming sufficient protein can help mitigate fatigue. If fatigue is severe or persistent, consider reducing the dose or investigating other potential causes such as thyroid dysfunction or anemia. Researchers tracking peptides for depression and anxiety should be aware that mood changes and fatigue can overlap with metabolic adjustments.
Hypoglycemia (low blood sugar) is uncommon in individuals not taking insulin or sulfonylureas. Tirzepatide is glucose-dependent in its mechanism, meaning it stimulates insulin secretion only when blood glucose is elevated. However, in individuals with diabetes who are taking other glucose-lowering medications, the risk of hypoglycemia increases. Symptoms include shakiness, sweating, confusion, dizziness, and rapid heartbeat. If hypoglycemia occurs, consume 15 grams of fast-acting carbohydrates such as glucose tablets, juice, or candy, and recheck blood glucose after 15 minutes. Dose adjustments to other diabetes medications may be necessary when starting tirzepatide.
Tirzepatide carries a boxed warning for thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). This warning is based on animal studies showing an increased incidence of thyroid tumors in rodents exposed to GLP-1 receptor agonists. The relevance to humans is unclear, as humans have fewer thyroid C-cells than rodents. However, as a precaution, tirzepatide is contraindicated in individuals with a personal or family history of MTC or multiple endocrine neoplasia syndrome type 2 (MEN 2). Individuals should be counseled to report symptoms of thyroid tumors, such as a lump in the neck, difficulty swallowing, or persistent hoarseness.
Gallbladder disease, including cholelithiasis (gallstones) and cholecystitis (inflammation of the gallbladder), has been reported in individuals using tirzepatide. Rapid weight loss is a known risk factor for gallstone formation, as it alters the balance of bile components. Symptoms of gallbladder disease include right upper abdominal pain, nausea, vomiting, and fever. If these symptoms occur, imaging studies such as ultrasound may be needed to evaluate the gallbladder. In some cases, surgical removal of the gallbladder (cholecystectomy) is required. Researchers exploring peptides for menopause weight loss or peptides for fat loss should be aware that rapid weight loss increases gallstone risk regardless of the method used.
Kidney function should be monitored in individuals with pre-existing kidney disease. Severe gastrointestinal side effects leading to dehydration can worsen kidney function. Ensuring adequate hydration and managing side effects aggressively can mitigate this risk. If kidney function declines, dose reduction or discontinuation may be necessary.
Niacinamide at the doses present in compounded tirzepatide (approximately 1 to 2 mg per injection) is generally well tolerated. However, some individuals may experience mild flushing or warmth shortly after injection. This is not dangerous and typically resolves within minutes. At higher supplemental doses (500 to 3000 mg daily), niacinamide can cause nausea, liver enzyme elevation, or glucose intolerance, but these effects are unlikely at the low doses used in compounded formulations. Researchers using vitamin C and peptides or exploring other additive strategies should consider dose-dependent effects when evaluating safety.
Pregnancy and breastfeeding are contraindications. Tirzepatide has not been studied in pregnant or breastfeeding women, and animal studies suggest potential fetal harm. Women of childbearing potential should use effective contraception during therapy. If pregnancy occurs, discontinue tirzepatide immediately and consult a healthcare provider. Weight loss during pregnancy is not recommended, as it can deprive the fetus of essential nutrients. Researchers comparing semaglutide withdrawal symptoms should note that discontinuing GLP-1 or GIP/GLP-1 agonists can lead to rebound appetite and weight regain if not managed carefully.
Tirzepatide niacinamide vs other compounded formulations
Compounding pharmacies offer tirzepatide in combination with various additives, each with distinct rationales and potential benefits.
Tirzepatide with B12 is one of the most common alternatives. Vitamin B12 (cyanocobalamin or methylcobalamin) is often included to address fatigue and support energy metabolism. Individuals with obesity or insulin resistance frequently have marginal B12 deficiency, particularly if they have been on metformin, which interferes with B12 absorption. Adding B12 to a weight loss regimen can improve energy levels, reduce brain fog, and support red blood cell production. B12 does not directly enhance the weight loss effects of tirzepatide, but it may improve the overall experience by mitigating fatigue, a common side effect. Researchers exploring semaglutide with B12 will find similar logic applied to semaglutide formulations.
Tirzepatide with glycine is another option. Glycine is a non-essential amino acid with roles in collagen synthesis, neurotransmitter function, and metabolic regulation. Some compounding pharmacies suggest that glycine may reduce nausea, improve sleep quality, and support tissue repair during weight loss. Glycine has been shown in animal studies to improve insulin sensitivity and reduce inflammation in metabolic syndrome models. However, clinical evidence supporting glycine as an adjunct to tirzepatide is lacking. The doses used in compounded formulations are typically low, and whether they provide meaningful benefit is unclear. Researchers using regenerative peptides or peptides for bone healing protocols may find glycine useful for tissue support during intensive training or recovery.
Tirzepatide with L-carnitine is formulated with the goal of enhancing fat oxidation. L-carnitine is an amino acid derivative that transports long-chain fatty acids into mitochondria for oxidation. It is popular in weight loss and athletic performance contexts. However, clinical studies of L-carnitine supplementation for weight loss have shown modest and inconsistent results.
Endogenous carnitine synthesis is usually sufficient, and supplementation primarily benefits individuals with true carnitine deficiency, which is rare. Whether adding L-carnitine to tirzepatide provides additional fat loss is unproven. Researchers exploring peptides for athletic performance or peptides for muscle growth may find L-carnitine useful in specific contexts, but not necessarily as a tirzepatide additive.
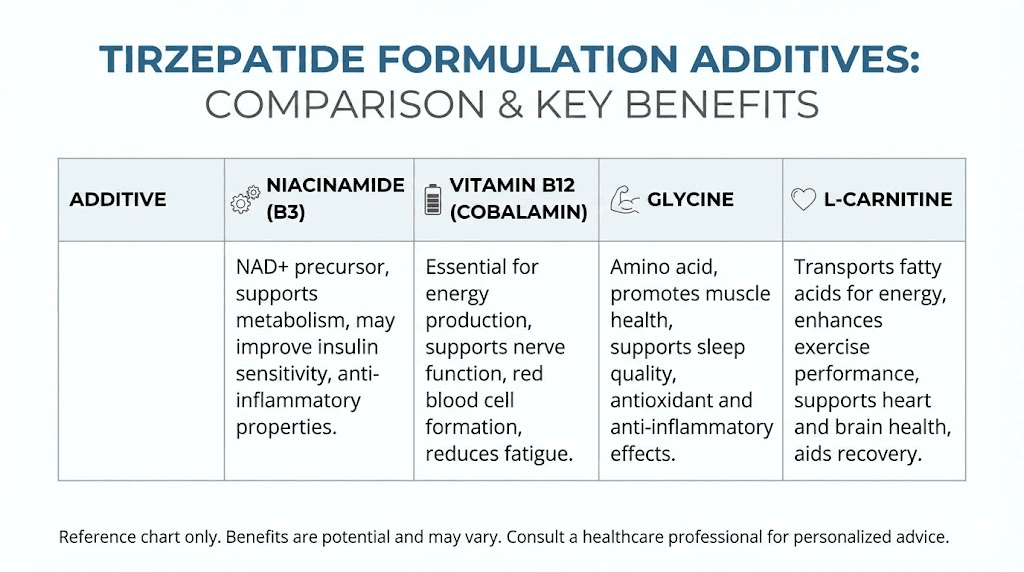
Tirzepatide with NAD+ or NAD+ precursors such as nicotinamide riboside (NR) or nicotinamide mononucleotide (NMN) is a more advanced formulation. These compounds directly raise NAD+ levels, supporting mitochondrial function, sirtuin activity, and cellular repair. NAD+ supplementation has shown promise in preclinical models for improving insulin sensitivity, enhancing fat oxidation, and promoting longevity. The rationale for combining NAD+ precursors with tirzepatide is similar to that for niacinamide, but with potentially greater potency due to higher NAD+ bioavailability. However, injectable NAD+ precursors are more expensive and less widely available than niacinamide. Researchers exploring khavinson peptides or klotho peptide longevity protein may be interested in NAD+ strategies for longevity and metabolic health.
Plain compounded tirzepatide without additives is also available. This is the simplest formulation and the closest to pharmaceutical-grade products. For individuals who tolerate tirzepatide well and do not require additional metabolic support, plain tirzepatide may be the best choice. It eliminates the risk of interactions or side effects from additives and is often less expensive. Researchers comparing semaglutide vs tirzepatide or retatrutide vs semaglutide should consider that formulation differences can affect outcomes, tolerability, and cost.
The choice between tirzepatide niacinamide and other formulations should be based on individual needs, side effect profiles, and cost. If fatigue is a concern, tirzepatide with B12 may be preferable. If nausea is problematic, tirzepatide with glycine could be worth trying, though evidence is limited. If metabolic optimization and mitochondrial support are priorities, tirzepatide niacinamide or NAD+ precursor formulations are logical choices. For simplicity and cost-effectiveness, plain tirzepatide is a solid option. Researchers using peptide stack calculator tools to plan complex regimens should evaluate the additive value of each component and avoid unnecessary complexity.
What to expect week by week on tirzepatide niacinamide
The timeline of effects with tirzepatide niacinamide follows a predictable pattern, though individual variation is substantial.
Week 1 to 4: Initial adaptation. During the first month on the 2.5mg starting dose, appetite suppression is noticeable but not overwhelming. Many individuals report reduced hunger between meals and earlier satiety during meals. Cravings, particularly for high-fat or high-sugar foods, often diminish. Weight loss during this period is typically 2 to 4 pounds, primarily from reduced caloric intake and initial fluid shifts. Gastrointestinal side effects such as mild nausea, bloating, or changes in bowel habits may occur. These are usually tolerable and improve as the body adapts. Energy levels may fluctuate as the metabolism adjusts. Researchers tracking how long does it take for tirzepatide to suppress appetite should note that appetite suppression begins within days but becomes more pronounced over weeks.
Week 5 to 8: First dose increase. The dose is increased to 5mg weekly. Appetite suppression intensifies. Some individuals describe feeling genuinely uninterested in food, a stark contrast to their baseline state. Weight loss accelerates, with an average of 1 to 2 pounds per week. This phase often includes visible changes in body composition, particularly around the abdomen and face. Gastrointestinal side effects may worsen temporarily after the dose increase but typically stabilize within a week. Fatigue or mild dizziness may occur, especially if caloric intake is too low. Ensuring adequate protein and micronutrient intake is important during this phase. Researchers exploring before and after tirzepatide weight loss results often see the most dramatic changes beginning in this period.
Week 9 to 12: Continued dose escalation. The dose is increased to 7.5mg weekly. Appetite remains well-controlled. Weight loss continues at a rate of 1 to 2 pounds per week. Total weight loss by the end of 12 weeks is typically 8 to 15 pounds, though some individuals lose significantly more. Muscle preservation becomes a concern during this phase. Resistance training and adequate protein intake (at least 1 gram per pound of ideal body weight) are essential to minimize muscle loss. Some individuals experience hair thinning, a common side effect of rapid weight loss due to disrupted hair growth cycles. This is usually temporary and resolves with continued therapy or dose stabilization. Researchers using tirzepatide before and after tracking tools should document changes in body composition, not just scale weight.
Week 13 to 24: Mid-phase adaptation. Doses continue to escalate to 10mg and 12.5mg weekly. Weight loss remains steady, averaging 1 to 1.5 pounds per week.
By six months, total weight loss is typically 15 to 25 pounds, with higher losses in individuals who started at higher body weights. Appetite suppression is strong, and many individuals need to consciously ensure they meet minimum caloric and nutrient requirements. Gastrointestinal side effects generally subside, though occasional nausea or constipation may persist. Energy levels stabilize or improve as the body adapts to lower body weight and improved insulin sensitivity. Metabolic markers such as fasting glucose, A1c, and lipid panels often show significant improvement. Researchers tracking how long does it take for tirzepatide to work should note that metabolic benefits often precede visible weight loss.
Week 25 to 52: Long-term phase. The dose reaches the maintenance level, typically 12.5mg or 15mg weekly. Weight loss continues but at a slower rate, averaging 0.5 to 1 pound per week. By one year, total weight loss is typically 15 to 30 percent of starting body weight, depending on adherence and baseline characteristics. Appetite suppression remains effective, though some individuals report slight tolerance development. Adjusting the dose or incorporating periodic breaks may help maintain efficacy. Body composition changes become more pronounced, with substantial fat loss and improved muscle definition if resistance training is maintained. Metabolic health markers continue to improve, with many individuals achieving normal glucose levels, reduced blood pressure, and improved lipid profiles. Researchers exploring why not losing weight on semaglutide or similar challenges should consider that weight loss plateaus are common after six to nine months and may require dose adjustments or lifestyle modifications.
Beyond one year: Maintenance and sustainability. Long-term data from tirzepatide trials extend to 72 weeks, showing continued weight loss with minimal plateauing. Real-world use beyond this timeframe is becoming more common, with many individuals maintaining therapy for years. The goal shifts from weight loss to weight maintenance and continued metabolic health. Some individuals reduce the dose to the minimum effective level, while others maintain higher doses to prevent weight regain. Periodic reassessment of goals, side effects, and metabolic markers is essential. Researchers exploring peptides vs TRT or peptides vs SARMs should note that peptide-based therapies are generally safer and more sustainable long-term compared to anabolic agents.
Maximizing results with tirzepatide niacinamide
While tirzepatide is highly effective on its own, optimizing lifestyle factors can enhance outcomes and improve long-term sustainability.
Protein intake is the most critical dietary consideration. Adequate protein preserves lean muscle mass during weight loss, supports satiety, and enhances metabolic rate. Aim for at least 1 gram of protein per pound of ideal body weight daily. High-quality sources include lean meats, fish, eggs, dairy, legumes, and protein supplements. Distribute protein intake across meals rather than consuming it all at once, as this maximizes muscle protein synthesis. Researchers exploring peptides for muscle growth or peptides for athletic performance will find similar protein recommendations for optimizing body composition.
Resistance training is essential. Losing weight without resistance training results in proportional loss of fat and muscle. Preserving muscle mass maintains metabolic rate, improves body composition, and supports long-term weight maintenance. Train at least three times per week, focusing on compound movements such as squats, deadlifts, presses, and rows. Progressive overload, gradually increasing weight or resistance over time, is key to stimulating muscle growth and preservation. Researchers using peptides for shoulder pain or fast injury healing protocols should ensure training is balanced and does not exacerbate existing injuries.
Cardiovascular exercise supports calorie expenditure and cardiovascular health but should not replace resistance training. Moderate-intensity activities such as walking, cycling, or swimming for 30 to 60 minutes most days of the week are sufficient. Avoid excessive cardio, as it can increase muscle loss and elevate cortisol, which may hinder weight loss.
Sleep quality and duration are often overlooked but critical. Poor sleep disrupts hunger hormones, increases cortisol, and impairs insulin sensitivity. Aim for seven to nine hours of quality sleep per night. Maintain a consistent sleep schedule, minimize screen time before bed, and create a cool, dark sleeping environment. If sleep disturbances persist, consider evaluating for sleep apnea or other sleep disorders. Researchers exploring pineal peptide for sleep or other sleep-enhancing interventions may find peptides useful adjuncts to standard sleep hygiene practices.
Stress management is another key factor. Chronic stress elevates cortisol, which promotes fat storage, particularly in the abdominal region, and interferes with appetite regulation. Practices such as meditation, yoga, deep breathing, or regular physical activity can reduce stress. Addressing underlying psychological factors such as emotional eating or food addiction may require professional support. Researchers tracking peptides for depression and anxiety should recognize that mental health and metabolic health are deeply interconnected.
Hydration is simple but important. Aim for at least 64 ounces of water daily, more if exercising or in hot climates. Adequate hydration supports digestion, reduces constipation, and helps distinguish hunger from thirst. Some individuals find that drinking water before meals enhances satiety and reduces caloric intake.
Micronutrient intake should not be neglected. Rapid weight loss and reduced caloric intake can lead to deficiencies in vitamins and minerals. Consider a high-quality multivitamin, along with specific supplements such as vitamin D, magnesium, and omega-3 fatty acids. Regular blood work to assess nutrient status, thyroid function, and metabolic markers is advisable. Researchers exploring vitamin C and peptides or other micronutrient strategies should tailor supplementation to individual needs based on testing.
Meal timing and structure can influence outcomes. Some individuals find that intermittent fasting or time-restricted eating enhances weight loss and simplifies meal planning. Others prefer smaller, more frequent meals to manage appetite and energy levels. Experiment to find what works best for you. Avoid grazing or mindless snacking, as this can undermine the appetite-suppressing effects of tirzepatide. Researchers comparing microdosing tirzepatide protocols should consider meal timing as a variable that can affect side effects and efficacy.
Monitoring progress is essential. Track weight weekly, but also measure body composition using methods such as skinfold calipers, bioelectrical impedance, or DEXA scans. Take progress photos and measurements of waist, hips, chest, and limbs. Monitor how clothing fits. These objective measures provide a more complete picture than weight alone. If weight loss stalls for more than two weeks, reassess caloric intake, activity levels, and adherence. Consider a brief diet break or a temporary increase in calories to reset metabolism. Researchers using peptide cost calculator tools should also track cost per pound of weight lost to evaluate value.
Regulatory status and sourcing
The regulatory environment for compounded tirzepatide has shifted significantly and continues to evolve.
Tirzepatide was approved by the FDA as Mounjaro for type 2 diabetes and later as Zepbound for chronic weight management. These are pharmaceutical-grade products manufactured by Eli Lilly. During periods of drug shortage, the FDA permits 503A compounding pharmacies to produce versions of the drug to meet patient demand. This allowance is based on the premise that patients cannot access the FDA-approved product due to supply constraints. Compounded versions are not FDA-approved and have not undergone the same rigorous testing for efficacy, safety, and quality. They are produced on a patient-specific basis based on a prescription from a licensed healthcare provider.
Tirzepatide was removed from the FDA drug shortage list. This action was based on manufacturer reports indicating that supply had stabilized and that the drug was available in sufficient quantities. The removal triggered a wind-down period during which compounding pharmacies could continue to produce tirzepatide for existing patients but were required to cease new compounding after the period ended. Patients relying on compounded tirzepatide faced transitions to pharmaceutical-grade products or sought alternative therapies. Some compounding pharmacies challenged the FDA decision, arguing that local or regional shortages persisted despite the national shortage designation being lifted. The legal and regulatory landscape remains fluid, and policies may change. Researchers exploring retatrutide buy complete guide or other emerging peptides should stay informed about regulatory developments.
Compounded tirzepatide is only legal when prescribed by a licensed healthcare provider and produced by a registered 503A compounding pharmacy. Purchasing peptides from unregulated sources, including online vendors without prescriber oversight, is illegal and dangerous. Products from such sources may be contaminated, improperly dosed, or counterfeit. There have been reports of individuals receiving vials labeled as tirzepatide that contained semaglutide or other substances. Testing for identity and purity is not routinely performed on compounded peptides, unlike pharmaceutical-grade products. Researchers comparing research vs pharmaceutical peptides should understand the quality and safety differences between regulated and unregulated sources.
Sourcing legitimate compounded tirzepatide requires finding a provider who works with reputable compounding pharmacies. Telemedicine platforms have made this process more accessible, with many offering consultations, prescriptions, and pharmacy coordination. Costs for compounded tirzepatide typically range from $200 to $500 per month, depending on dose and pharmacy. This is significantly less expensive than pharmaceutical-grade products, which can cost $1,000 to $1,500 per month without insurance. However, insurance does not cover compounded medications, so patients pay out of pocket.
Pharmaceutical-grade tirzepatide is covered by many insurance plans for approved indications. For type 2 diabetes, Mounjaro is widely covered. For weight management, Zepbound coverage is more variable, with some insurers requiring prior authorization or step therapy. Patient assistance programs are available for individuals who meet financial and medical criteria. The FDA-approved products have the advantage of consistent quality, established supply chains, and comprehensive safety monitoring. For individuals who can access pharmaceutical-grade tirzepatide affordably, it is generally the preferred option. Researchers comparing semaglutide vs tirzepatide should consider both efficacy and accessibility when choosing a therapy.
International sourcing is another consideration. Some individuals purchase tirzepatide from international pharmacies or research chemical suppliers. This practice carries significant risks. Product quality is uncertain, legality varies by jurisdiction, and there is no recourse if the product is defective or harmful. Importing peptides across borders may violate customs regulations and result in confiscation or legal penalties. Researchers exploring lyophilized vs liquid peptides or other formulation types should prioritize sourcing from regulated entities within their country.
Frequently asked questions
What is the difference between tirzepatide niacinamide and regular tirzepatide?
Tirzepatide niacinamide is a compounded formulation that combines the dual GIP/GLP-1 receptor agonist tirzepatide with niacinamide, the amide form of vitamin B3. Regular tirzepatide refers to pharmaceutical-grade products like Mounjaro or Zepbound, or plain compounded tirzepatide without additives. The niacinamide is included for potential stabilization benefits and metabolic support, though clinical evidence for added efficacy is limited. Researchers comparing tirzepatide dosage in units protocols should note that formulation differences do not typically require dose adjustments.
Can I use tirzepatide niacinamide if I have diabetes?
Yes, tirzepatide is approved for type 2 diabetes management. The addition of niacinamide does not contraindicate use in diabetes, though monitoring blood glucose closely is important. Niacin, a related form of vitamin B3, can transiently raise glucose levels, but niacinamide is generally considered neutral in this regard. If you are taking other glucose-lowering medications such as insulin or sulfonylureas, dose adjustments may be needed to prevent hypoglycemia. Researchers tracking how long does it take for tirzepatide to work should monitor glucose levels weekly during titration.
How much weight can I expect to lose on tirzepatide niacinamide?
Weight loss varies widely based on starting weight, adherence, lifestyle factors, and dose. Clinical trials of tirzepatide showed 16 to 22.5 percent body weight loss over 72 weeks at doses of 10mg to 15mg weekly. For a 200-pound individual, this translates to 32 to 45 pounds. Real-world results may differ. Some individuals lose more, others less. Compounded formulations with niacinamide have not been specifically studied, so whether the additive provides additional weight loss is unknown. Researchers exploring before and after tirzepatide weight loss results should track individual progress and adjust expectations based on response.
Do I need to refrigerate tirzepatide niacinamide?
Yes, compounded tirzepatide niacinamide should be stored in a refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Do not freeze. The vial can be kept at room temperature for short periods, such as during travel or immediately before injection, but prolonged exposure to room temperature should be avoided. Researchers tracking how long does tirzepatide last in the fridge should note that compounded formulations may have shorter stability periods than pharmaceutical-grade products, typically 30 to 90 days from compounding.
Can I take tirzepatide niacinamide if I am on other supplements?
Most supplements are safe to take with tirzepatide niacinamide. Common supplements like multivitamins, vitamin D, omega-3 fatty acids, and protein powders do not interact with tirzepatide. However, supplements that affect blood sugar, such as berberine or chromium, should be used with caution and monitored closely. High-dose niacin supplements should be avoided, as they can impair insulin sensitivity and raise glucose levels, potentially counteracting the benefits of tirzepatide. Researchers exploring vitamin C and peptides or other supplement strategies should consult a healthcare provider before combining multiple agents.
What should I do if I miss a dose of tirzepatide niacinamide?
If you miss a dose and it has been less than four days since your scheduled injection, take the missed dose as soon as you remember. If more than four days have passed, skip the missed dose and resume your regular schedule with the next planned injection. Do not double doses. Missing doses can disrupt appetite control and lead to rebound hunger, so maintaining consistency is important.
Is tirzepatide niacinamide safe for long-term use?
Clinical trials of tirzepatide have extended up to 72 weeks, with no major safety concerns emerging. Real-world use often extends beyond this timeframe, with many individuals maintaining therapy for years. Long-term safety data beyond two years is limited. The main concerns for extended use include gallbladder disease, potential thyroid effects, and unknown impacts of chronic appetite suppression. Regular monitoring of metabolic markers, kidney function, and overall health is advisable. Niacinamide at the doses used in compounded tirzepatide is considered safe long-term, as it is a naturally occurring vitamin.
Can I drink alcohol while on tirzepatide niacinamide?
Moderate alcohol consumption is generally safe with tirzepatide, though it may exacerbate gastrointestinal side effects or increase the risk of hypoglycemia in individuals with diabetes. Alcohol is calorie-dense and can hinder weight loss, so limiting intake is advisable. Some individuals find that tirzepatide reduces their desire for alcohol, a benefit reported in GLP-1 receptor agonist research. Heavy alcohol use should be avoided, as it increases the risk of pancreatitis, a rare but serious side effect of tirzepatide. Researchers exploring peptides for gut health should recognize that alcohol can disrupt gut microbiota and impair recovery from metabolic dysfunction.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available for evidence-based peptide education, advanced dosing calculators, and detailed protocol guides. Members gain access to continuously updated content on emerging peptides, comparative analyses of formulations like tirzepatide niacinamide versus standard options, and practical tools for tracking progress. Whether you are navigating compounded formulations, comparing retatrutide vs semaglutide, or seeking protocols for peptides for hormone balance, SeekPeptides provides the depth and precision that serious researchers require.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay stable, your titrations stay smooth, and your metabolic health stay on track.