Feb 27, 2026

Experienced researchers know something that most newcomers to the GLP-1 space completely miss. The pharmacy filling your tirzepatide prescription is not just a pharmacy. It operates under a specific federal designation that determines everything from how the medication is manufactured, to how it is tested, to whether the FDA has ever set foot inside the building. That designation, 503A or 503B, shapes the quality, availability, and legality of every compounded vial you receive.
And right now, the distinction matters more than ever.
The FDA pulled tirzepatide off its drug shortage list in December 2024, triggering a cascade of enforcement deadlines, court battles, and regulatory chaos that left patients, providers, and pharmacies scrambling to understand what comes next. 503B outsourcing facilities, the large-scale compounders that supplied millions of doses during the shortage, suddenly found themselves on the wrong side of federal law. Some shut down production. Others pivoted to alternative formulations. A few are fighting the FDA in court.
This guide covers everything you need to understand about 503B compounding pharmacies and tirzepatide, from what these facilities actually do, to why the FDA created them, to how the current regulatory landscape affects your ability to access compounded tirzepatide through legitimate channels. Whether you are currently using a compounded product, considering switching from brand-name to compounded, or simply trying to make sense of the headlines, the answers are here. SeekPeptides has tracked these developments from the beginning, and this is the complete picture.
What is a 503B compounding pharmacy
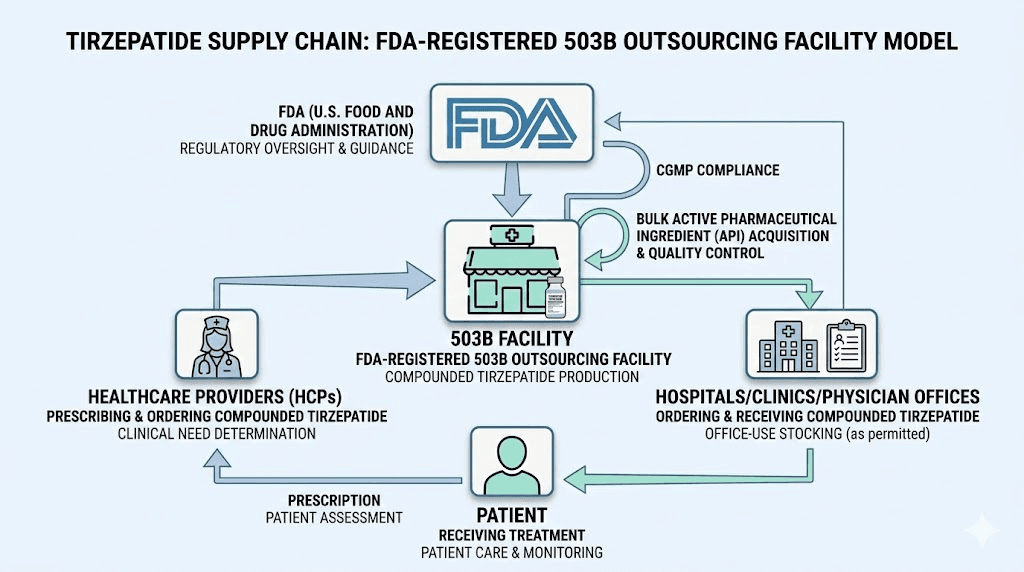
A 503B compounding pharmacy is not your neighborhood drugstore. It is a federally registered outsourcing facility that manufactures compounded medications in bulk, without requiring individual patient prescriptions. The name comes from Section 503B of the Federal Food, Drug, and Cosmetic Act, which Congress added through the Drug Quality and Security Act (DQSA) of 2013.
The DQSA emerged from tragedy.
In 2012, the New England Compounding Center in Framingham, Massachusetts shipped contaminated steroid injections that caused a nationwide fungal meningitis outbreak. Seventy-six people died. Over 750 were sickened across 20 states. The facility had been operating in a regulatory gray zone, not quite a traditional pharmacy, not quite a manufacturer, and not adequately overseen by either state or federal authorities.
Congress responded by creating two distinct categories of compounding pharmacies. Section 503A covered traditional pharmacies, the ones that fill individual prescriptions under state oversight. Section 503B created an entirely new entity, the outsourcing facility, designed for large-scale compounding under direct FDA supervision. These facilities would follow the same Current Good Manufacturing Practice (cGMP) standards required of major pharmaceutical companies like Eli Lilly and Novo Nordisk.
The distinction matters because 503B facilities can do something 503A pharmacies cannot. They can manufacture compounded medications in advance, in large batches, without waiting for a specific patient prescription. Hospitals can stock them. Clinics can keep them on hand. Physicians can administer them directly in their offices. This capability made 503B facilities critical during the tirzepatide and semaglutide shortages, when brand-name supply could not keep up with exploding demand.
How 503B facilities operate day to day
Think of a 503B outsourcing facility as a miniature pharmaceutical manufacturer. The facility maintains cleanroom environments with controlled air quality, temperature, and humidity. Every batch of medication undergoes sterility testing, potency testing, and endotoxin screening before it ships. Staff work under the direct supervision of licensed pharmacists, though the facility itself does not need to hold a pharmacy license.
The FDA inspects these facilities on a risk-based schedule, typically every 12 to 24 months. Inspectors examine everything from storage conditions to quality control documentation to the actual compounding process itself. If they find problems, they issue Form 483 observations, warning letters, or in severe cases, pursue enforcement actions that can shut the facility down entirely.
This level of oversight separates 503B facilities from the compounding pharmacies most patients encounter. A 503B operation like Empower Pharmacy in Houston maintains FDA-registered cGMP facilities in multiple states, producing thousands of units per batch. Compare that to a local 503A pharmacy that might compound a few dozen vials per week based on individual prescriptions.
What 503B facilities can and cannot compound
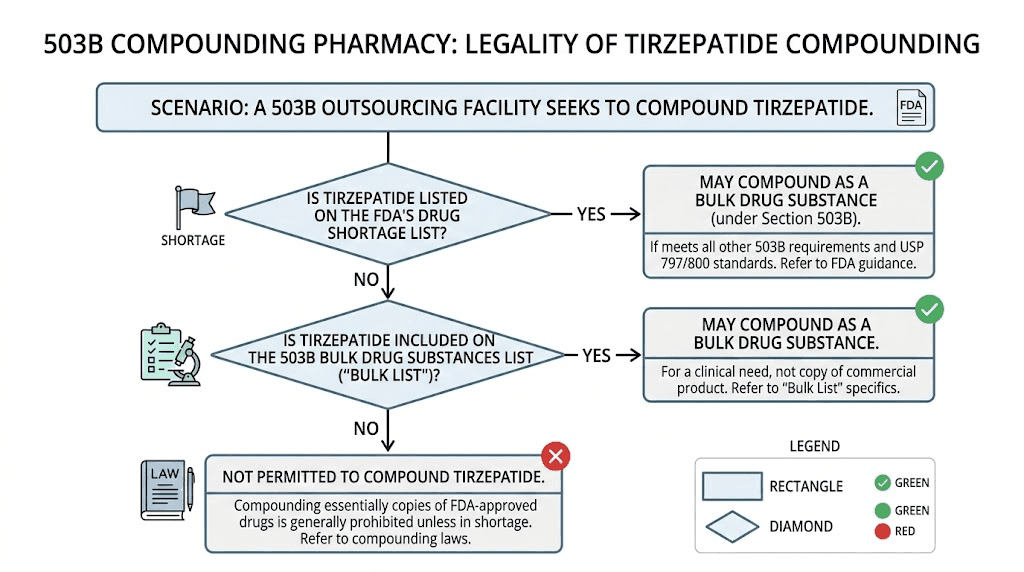
Here is where it gets complicated. 503B facilities cannot simply manufacture any medication they want. Federal law imposes specific restrictions.
They cannot produce drugs that are "essentially copies" of commercially available FDA-approved medications, unless those drugs appear on the FDA drug shortage list. They can only use bulk active pharmaceutical ingredients (APIs) that appear on the FDA Category 1 list or have been nominated through the formal review process. And every product must be compounded under cGMP conditions with full batch testing and documentation.
For tirzepatide specifically, neither the active ingredient nor the finished product appears on the Category 1 bulk drug substances list. This means 503B facilities have historically relied on the shortage exception to justify compounding tirzepatide, an exception that no longer applies now that the FDA has declared the shortage resolved.
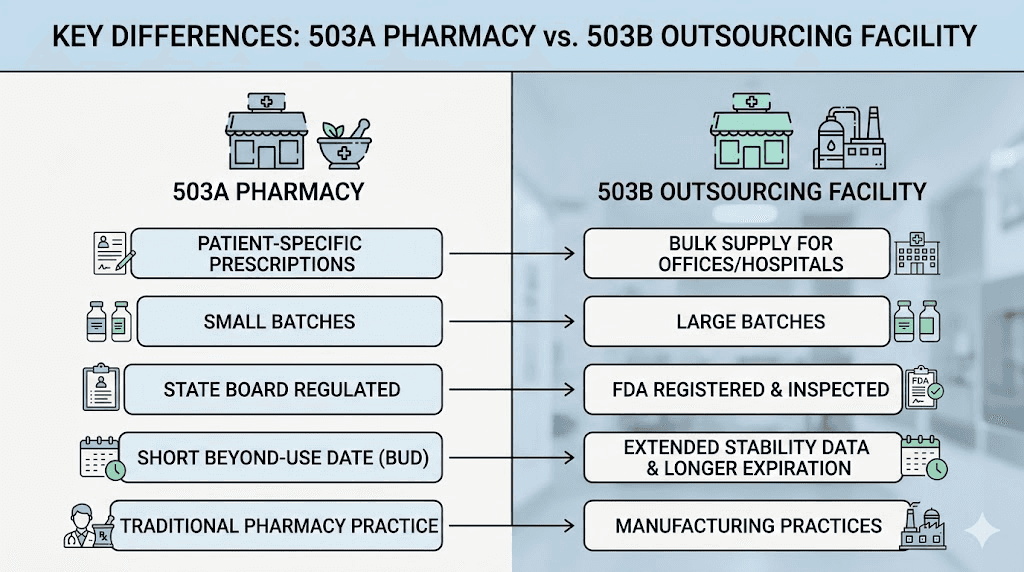
503A vs 503B: the differences that actually matter
The distinction between 503A and 503B pharmacies is not academic. It directly affects the quality, cost, legality, and availability of the compounded tirzepatide you receive. Understanding these differences is essential for anyone using or considering compounded tirzepatide.
Factor | 503A pharmacy | 503B outsourcing facility |
|---|---|---|
Regulation | State pharmacy boards | FDA (federal oversight) |
Prescription required | Yes, patient-specific | No, can compound in advance |
Manufacturing standards | USP 795/797/800 | cGMP (21 CFR 210/211) |
FDA inspection | Not routine | Every 12-24 months |
Batch size | Small (individual orders) | Large (bulk production) |
Sterility testing | Process-based | Full batch testing required |
Adverse event reporting | Not required to FDA | Required to FDA |
Distribution | Within state (mostly) | Can ship across state lines |
Office use allowed | Limited | Yes, clinics can stock |
Regulatory oversight: state vs federal
This is the fundamental divide. A 503A pharmacy answers to its state board of pharmacy. Standards vary. Inspection frequency varies. Enforcement varies. A 503A pharmacy in Texas might face different requirements than one in California or Florida. Some states have rigorous compounding oversight programs. Others do not.
A 503B facility answers to the FDA. Period. The standards are uniform, the inspections are rigorous, and the consequences of noncompliance are severe. The FDA publishes inspection findings, warning letters, and enforcement actions on its website for anyone to review. This transparency does not exist for most state-regulated 503A pharmacies.
Does this mean 503B products are always safer? Not necessarily. The FDA has found serious safety issues at some 503B facilities, including sterility failures, subpotent products, and inadequate quality controls. But at least the inspection framework exists to catch these problems. With 503A pharmacies, problems may go undetected for years.
Prescription requirements and patient access
503A pharmacies compound medications only after receiving a valid prescription for a specific patient. Your doctor writes a prescription, the pharmacy fills it, you pick it up. Simple.
503B facilities work differently. They produce compounded medications in bulk, then distribute them to healthcare facilities, clinics, hospitals, and sometimes directly to patients through licensed healthcare providers. A weight loss clinic can order hundreds of vials of compounded tirzepatide from a 503B facility and keep them in stock for patient visits. This model is why telehealth companies were able to scale GLP-1 programs so rapidly during the shortage.
The tradeoff is personalization. A 503A pharmacy can tailor a formulation to your exact needs. If you have an allergy to a specific inactive ingredient, they can leave it out. If you need an unusual concentration, they can make it. A 503B facility produces standardized formulations in large batches, which means less flexibility but more consistency across units.
Quality standards: USP vs cGMP
503A pharmacies follow United States Pharmacopeia (USP) chapters 795 (non-sterile), 797 (sterile), and 800 (hazardous drugs). These standards cover environmental controls, beyond-use dating, and personnel training. They are rigorous but less comprehensive than what 503B facilities face.
503B facilities must comply with 21 CFR Parts 210 and 211, the same Current Good Manufacturing Practice regulations that govern major pharmaceutical companies. This includes detailed requirements for facility design, equipment qualification, process validation, in-process testing, stability studies, CAPA (Corrective and Preventive Actions) systems, and data integrity controls.
In practical terms, a 503B facility performs sterility testing on every batch before release. A 503A pharmacy typically relies on process validation and environmental monitoring rather than testing each individual batch. Both approaches can produce safe, sterile products, but the 503B model provides an additional layer of verification through direct product testing.

The regulatory timeline: what happened to tirzepatide compounding
The story of 503B tirzepatide compounding is really the story of a shortage that created a billion-dollar market, and what happened when that shortage ended.
The shortage era: when compounding was legal
Tirzepatide, sold under the brand names Mounjaro and Zepbound by Eli Lilly, experienced severe supply constraints beginning in late 2022. Demand for GLP-1 receptor agonists exploded as clinical data showed remarkable weight loss results, and manufacturing could not keep pace. The FDA added tirzepatide to its official drug shortage list, which triggered an important legal exception.
Under federal law, compounding pharmacies (both 503A and 503B) are generally prohibited from producing "essentially copies" of commercially available FDA-approved drugs. But when a drug appears on the FDA shortage list, that prohibition lifts. Compounders can step in to fill the gap.
And step in they did. 503B outsourcing facilities began producing compounded tirzepatide at scale. Empower Pharmacy, Olympia Pharmaceuticals, and dozens of other 503B facilities ramped up production. The compounded versions cost a fraction of brand-name Mounjaro and Zepbound, typically $200 to $500 per month versus $1,000 or more for the brand-name products without insurance.
Millions of patients gained access to tirzepatide through compounding. Telehealth platforms built entire businesses around connecting patients with compounding pharmacies. The market grew rapidly.
December 2024: the FDA declares the shortage resolved
On December 19, 2024, the FDA made a determination that would reshape the entire compounding landscape. It declared that the tirzepatide injection shortage was resolved. Eli Lilly had expanded manufacturing capacity, and the agency concluded that commercial supply could now meet demand.
This declaration started the clock on enforcement deadlines. The FDA gave compounders a grace period to wind down operations.
503A pharmacies had until February 18, 2025 to stop compounding tirzepatide injections under the shortage exception. 503B outsourcing facilities had until March 19, 2025. After those dates, the FDA could take enforcement action against any compounder still producing essentially identical copies of commercially available tirzepatide.
The reaction was immediate and intense. Patients panicked. Compounding pharmacies scrambled. And the Outsourcing Facilities Association filed a lawsuit.
The court battle: OFA v. FDA
The Outsourcing Facilities Association (OFA), a trade group representing 503B facilities, sued the FDA in the US District Court for the Northern District of Texas. They argued that the shortage was not truly resolved, that patients still faced access barriers due to cost and insurance coverage, and that the FDA had overstepped its authority in making the determination.
On March 5, 2025, Judge Pittman denied OFA's request for a preliminary injunction. The court concluded that the FDA acted within its statutory authority in determining that tirzepatide was no longer in shortage. The decision emphasized the agency's discretion in managing the drug shortage list and found that the plaintiffs had not demonstrated sufficient harm to warrant judicial intervention.
On May 7, 2025, the court issued its full decision upholding the FDA's removal of tirzepatide from the shortage list. OFA filed a notice of interlocutory appeal on March 10, 2025, taking the fight to the Fifth Circuit Court of Appeals.
The legal battle continues. But for now, the FDA's position stands. Compounded tirzepatide in its standard injection form is no longer authorized under the shortage exception.
Current legal status: can 503B facilities still compound tirzepatide
The short answer is complicated. The practical answer depends on what exactly the 503B facility is compounding and why.
Standard tirzepatide injections: no
As of March 19, 2025, 503B outsourcing facilities cannot legally compound standard tirzepatide injections that are essentially copies of Mounjaro or Zepbound. The shortage exception no longer applies, the drug does not appear on the Category 1 bulk drug substances list, and courts have upheld the FDA's authority to make these determinations.
Any 503B facility still producing standard compounded tirzepatide injections after this date is operating outside the legal framework and risks FDA enforcement action, including warning letters, injunctions, and potential criminal prosecution.
Clinically different formulations: possibly
Here is where the nuance lives. Federal law prohibits compounding "essentially copies" of commercially available drugs. But if a compounded product is meaningfully different from the commercial version, it may not qualify as an essentially copy.
What counts as meaningfully different? The FDA has provided some guidance. A compounded formulation that combines tirzepatide with another active ingredient, such as niacinamide, glycine, vitamin B12, or levocarnitine, may be considered clinically different because the commercial product does not contain those additional ingredients.
This is exactly why you have seen an explosion of tirzepatide combination products from compounding pharmacies. Tirzepatide with glycine and B12 blends. Tirzepatide with methylcobalamin formulations. These combinations may provide a legal pathway for continued compounding, but the FDA has not provided definitive clarity on where the line falls.
The risk is real. The FDA could determine that adding a small amount of B12 to tirzepatide does not make it meaningfully different from the commercial product. Compounders operating in this gray zone are making a calculated bet that the FDA will not take that position, or that courts will side with them if the agency does.
Patient-specific clinical need: limited applicability for 503B
503A pharmacies have another pathway. They can compound medications for individual patients who have a documented clinical need that the commercial product cannot meet. For example, a patient with a documented allergy to an inactive ingredient in Mounjaro could receive a compounded version that omits that ingredient.
This pathway is largely unavailable to 503B facilities. Because 503B operations compound in bulk without patient-specific prescriptions, they cannot document individual clinical needs in the same way that 503A pharmacies can. A 503B facility compounding thousands of vials cannot claim that each one addresses a specific patient allergy.
This structural limitation is why the post-shortage landscape is particularly challenging for 503B facilities. 503A pharmacies retain some ability to compound tirzepatide on a case-by-case basis. 503B facilities, by design, cannot operate on a case-by-case basis.
Safety and quality: what to look for in a 503B pharmacy
Not all 503B facilities are created equal. The designation means the facility has registered with the FDA and agreed to follow cGMP standards. It does not guarantee flawless execution. Understanding what separates a high-quality 503B facility from a problematic one is critical, especially when dealing with injectable medications like tirzepatide.
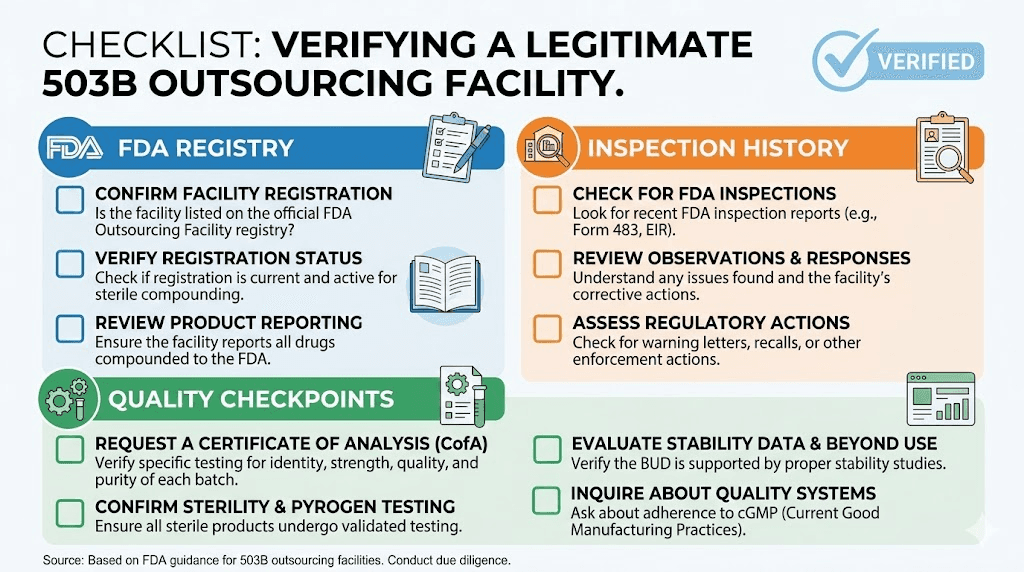
FDA inspection history matters
The FDA publishes inspection results for 503B facilities. Before using any 503B compounder, check their inspection history. Look for Form 483 observations, which document specific violations found during inspection. Minor observations about documentation are common and not necessarily alarming. Observations about sterility failures, inadequate testing, or contaminated products are red flags that should give you serious pause.
You can search for inspection results on the FDA's Compounding Inspections, Recalls, and Other Actions page. If a facility has received a warning letter, the full text is typically available online. Read it. The details tell you exactly what the FDA found wrong.
Sterility testing and potency verification
Every batch of injectable medication from a 503B facility should undergo sterility testing before release. This is not optional under cGMP. Ask the facility, or your provider, whether the specific lot you are receiving passed both sterility and potency testing.
The FDA has documented alarming failures in this area. One facility released a batch of tirzepatide with glycine and methylcobalamin that failed sterility testing after it had already been distributed. Potency testing at another facility found tirzepatide injections at 86.8% and 89.1% of labeled strength, meaning patients received significantly less active ingredient than they thought.
These are not abstract risks. A subpotent injection means your tirzepatide dosage is lower than expected, which affects efficacy and makes dose titration unreliable. A non-sterile injection can cause infections at the injection site, abscesses, or potentially life-threatening systemic infections like sepsis.
Source of active pharmaceutical ingredients
Where does the 503B facility get its tirzepatide API? Both Eli Lilly and Novo Nordisk have publicly stated they do not supply compounders with tirzepatide or semaglutide. This means every compounded version uses API from third-party sources, often overseas manufacturers.
Legitimate 503B facilities source API from FDA-registered establishments and maintain complete documentation of the supply chain. Facilities that cut corners may use API from unregistered sources, which introduces risks around purity, identity, and potency. FDA inspectors have found facilities using bulk drug substances from sources that are not FDA-registered, a serious violation that calls into question every product made with that material.
You should feel comfortable asking your provider or the facility itself about their API sourcing. A reputable 503B facility will have documentation readily available. Reluctance to share this information is a warning sign.
Adverse event reporting
503B facilities are legally required to report adverse events to the FDA. This requirement does not apply to most 503A pharmacies. As of November 30, 2024, the FDA had received over 200 reports of adverse events from compounded tirzepatide products, some potentially related to dosing errors, contamination, or subpotency issues.
The adverse event reporting requirement means that problems with 503B products are more likely to be captured in the FDA's database. This is both a safety advantage (problems get reported) and a source of information for consumers and providers evaluating different compounding facilities.
Known 503B pharmacies and tirzepatide
Several major 503B outsourcing facilities have been prominent in the tirzepatide compounding space. Understanding who the major players are helps you evaluate your options, even as the regulatory landscape shifts.
Empower Pharmacy
Empower Pharmacy operates FDA-registered cGMP-compliant 503B outsourcing facilities in Houston, Texas and East Windsor, New Jersey. They also maintain a 503A pharmacy operation, giving them flexibility to operate under both regulatory frameworks. Empower has been one of the most visible compounders of GLP-1 medications, offering both tirzepatide formulations and semaglutide formulations.
Empower has adapted to the post-shortage environment by offering alternative formulations, including tirzepatide ODT (orally disintegrating tablets) and combination products that may not qualify as essentially copies of the commercial injection.
Olympia Pharmaceuticals
Olympia Pharmaceuticals is another major 503B outsourcing facility that has been active in the GLP-1 compounding space. 503B pharmacies like Olympia are the only compounding pharmacies allowed to provide office-use medications, meaning clinics and medical practices can stock their products for direct patient administration.
Other notable facilities
The 503B landscape extends beyond these two names. Dozens of registered outsourcing facilities have participated in GLP-1 compounding. Some have clean inspection histories and transparent operations. Others have faced FDA scrutiny, warning letters, or voluntary recalls.
Several other compounding operations serve the tirzepatide market through various pharmacy models. Red Rock Pharmacy, Southend Pharmacy, South Lake Pharmacy, and ProRx Pharmacy have all been reviewed within the compounded tirzepatide space. Some operate as 503A pharmacies rather than 503B facilities, so it is important to verify which designation applies when evaluating your options.
Cost considerations: 503B vs brand-name tirzepatide
Cost has been the primary driver behind the compounded tirzepatide market. The difference between brand-name and compounded pricing is not subtle. It is dramatic.
Brand-name pricing
Mounjaro and Zepbound, the two FDA-approved brand-name tirzepatide products, carry list prices exceeding $1,000 per month without insurance. Even with insurance, many patients face substantial copays or prior authorization requirements that delay or prevent access. Some insurance plans do not cover GLP-1 medications for weight loss at all, limiting coverage to the diabetes indication.
The result is a two-tier system. Patients with generous insurance coverage can access brand-name tirzepatide relatively affordably. Patients without coverage face a financial barrier that puts the medication out of reach.
Compounded tirzepatide pricing
Compounded tirzepatide from 503B facilities has typically cost between $200 and $500 per month, depending on the dosage, formulation, and provider. Some telehealth platforms bundled the medication with consultation fees, bringing total monthly costs to $300 to $600. Still dramatically less than brand-name pricing.
This cost advantage is the reason compounded tirzepatide became so popular so quickly. It is also why the FDA's decision to end the shortage exception has generated such intense reaction. For many patients, compounded tirzepatide was not a second choice. It was the only financially viable option.
What happens to pricing now
With the shortage resolved and compounding restricted, patients face difficult choices. Brand-name tirzepatide remains expensive. Some compounding pharmacies continue to offer combination formulations (tirzepatide with niacinamide, B12, or glycine) that may fall outside the "essentially copy" prohibition, but the legal status of these products remains uncertain.
Eli Lilly has introduced some patient assistance programs and coupons to address affordability concerns. Whether these programs adequately bridge the cost gap for patients who relied on compounded versions remains an open question.
Peptide cost calculators can help you compare the true monthly cost of different options, factoring in concentration, dosage requirements, and frequency.
503B compounded tirzepatide formulations
One of the most significant developments in the post-shortage landscape has been the proliferation of tirzepatide combination formulations. These products add additional active ingredients to the base tirzepatide, potentially creating a product that is not an "essentially copy" of the commercial version.
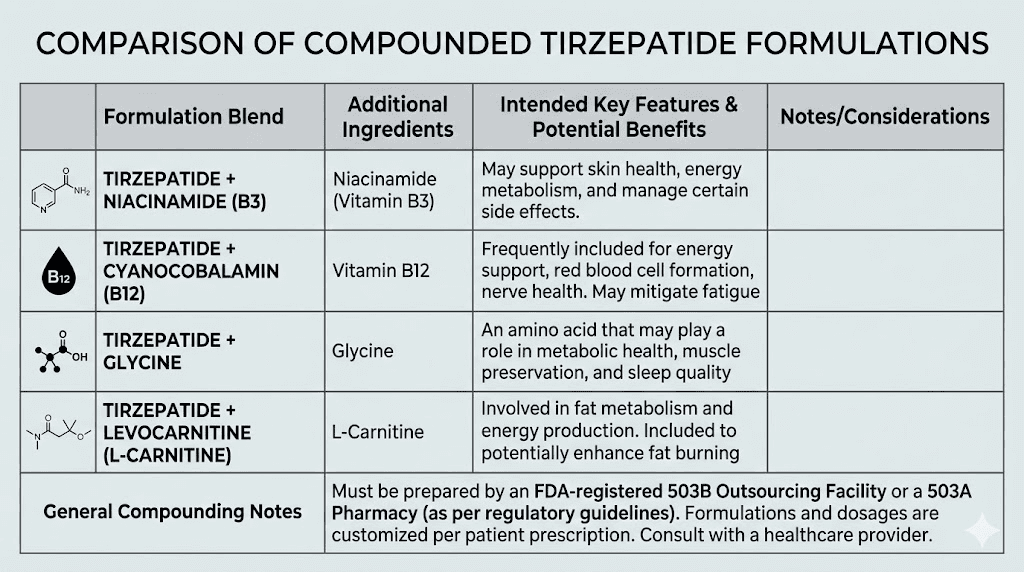
Tirzepatide with niacinamide
Tirzepatide combined with niacinamide (vitamin B3) has been one of the most common combination formulations. Empower Pharmacy notably offered this combination. Niacinamide may support metabolic health and has been studied for potential benefits related to insulin sensitivity. Whether the addition of niacinamide creates a sufficiently different product to avoid the "essentially copy" designation is a legal question that has not been definitively resolved.
Tirzepatide with B12 and glycine
Tirzepatide compounded with glycine and vitamin B12 (often as methylcobalamin) represents another popular combination. B12 supplementation is common among GLP-1 users because these medications can affect nutrient absorption. Glycine, an amino acid, is often added as a stabilizer and may provide its own metabolic benefits. Compounded tirzepatide with B12 formulations have been widely available through both 503A and 503B channels.
Tirzepatide with levocarnitine
Tirzepatide combined with levocarnitine (L-carnitine) targets enhanced fat metabolism. L-carnitine plays a role in transporting fatty acids into mitochondria for energy production. The combination appeals to patients who want to maximize the fat-burning potential of their tirzepatide protocol, and similar semaglutide-levocarnitine combinations have also gained popularity.
Alternative delivery forms
Some 503B facilities have moved beyond injectable tirzepatide entirely, creating alternative delivery forms that are inherently different from the commercially available injection products. Tirzepatide ODT (orally disintegrating tablets) dissolve on the tongue without water. Tirzepatide drops offer another oral option. Oral versus injectable tirzepatide represents a genuine difference in dosage form, which strengthens the argument that these products are not essentially copies of the FDA-approved injections.
The legal strength of this argument varies by product. An ODT or sublingual formulation represents a fundamentally different delivery mechanism and pharmacokinetic profile. A combination injectable that simply adds a small amount of B12 to the same injection is a more tenuous distinction.
How to verify a 503B pharmacy is legitimate
Whether you are evaluating a 503B facility for the first time or verifying the credentials of one you already use, there are specific steps you should follow. Given the regulatory uncertainty around tirzepatide compounding, due diligence is more important than ever.
Step 1: check the FDA outsourcing facility registry
The FDA maintains a public list of all registered outsourcing facilities. If a facility claims to be a 503B outsourcing facility but does not appear on this list, it is either not registered (which makes it illegal) or misrepresenting its status. The registry is available on the FDA website and is updated regularly.
Step 2: review inspection history
Once you confirm a facility is registered, check its inspection history. The FDA Compounding Inspections, Recalls, and Other Actions page provides details on inspection outcomes, warning letters, and enforcement actions. Pay attention to the severity of any findings and whether the facility has taken corrective action.
Key red flags include repeat observations for the same issue across multiple inspections, failure to respond adequately to Form 483 observations, any finding related to sterility testing failures, subpotency, or contamination, and use of bulk drug substances from unregistered sources.
Step 3: verify state licensing
503B facilities must also comply with state pharmacy board requirements in the states where they operate and distribute products. Check with your state board of pharmacy to confirm the facility holds appropriate licenses. Some states have additional requirements beyond the federal framework.
Step 4: ask about testing and quality control
A legitimate 503B facility should be able to provide certificates of analysis (COA) for the specific batch of medication you receive. These certificates document potency, sterility, endotoxin levels, and other quality parameters. If a facility cannot or will not provide a COA, consider that a significant red flag.
Step 5: evaluate the provider relationship
How are you accessing compounded tirzepatide? Through a telehealth platform? Through your personal physician? Through a clinic? The legitimacy of the prescribing relationship matters. A provider who conducts a thorough medical evaluation, monitors your progress with regular follow-ups, and adjusts your starting dose based on your response is operating responsibly. A platform that rubber-stamps prescriptions with minimal evaluation is putting you at risk regardless of how good the pharmacy is.
What patients need to know right now
The regulatory landscape for compounded tirzepatide is evolving rapidly. What was legal six months ago may not be legal today, and what is legal today could change with the next court ruling or FDA guidance document. Here is what matters most for patients navigating this uncertainty.
If you are currently using compounded tirzepatide
Ask your provider whether the product you are using is still being compounded legally. Specifically ask whether it is a combination formulation (which may have a legal pathway) or a standard tirzepatide injection (which likely does not). Ask for documentation of the facility's 503B registration and the batch testing results for your specific product.
Do not assume that because your provider is still offering compounded tirzepatide, it is necessarily legal. Some providers may be sourcing from facilities that are continuing to compound despite the enforcement deadline. Others may have transitioned to combination formulations or alternative delivery forms. Knowing which situation applies to you is important for both legal and safety reasons.
If you are considering switching from brand-name to compounded
The financial incentive to switch remains powerful. But the risk profile has changed. During the shortage, compounded tirzepatide was explicitly authorized by the legal framework. Now, the authorization has expired for standard formulations, and the legal status of combination products is uncertain.
Evaluate the specific product being offered. Is it a combination formulation? What additional ingredients does it contain? Is the compounding pharmacy a registered 503B facility or a 503A pharmacy operating under the patient-specific clinical need exception? What dose will you be receiving, and has the potency been verified by batch testing?
These are questions you should ask before making any switch. A provider who cannot answer them clearly should give you pause.
If your compounding pharmacy has stopped offering tirzepatide
Some 503B facilities have voluntarily ceased tirzepatide compounding in response to the FDA's enforcement timeline. If your pharmacy has stopped, you have several options. You can transition to brand-name Mounjaro or Zepbound, though cost may be prohibitive without insurance coverage. You can seek a 503A pharmacy that compounds tirzepatide under the patient-specific clinical need exception, if you have a documented clinical reason. You can explore alternative delivery forms like ODT or oral drops that may be legally distinct from the commercial injection.
You can also discuss alternative GLP-1 medications with your provider. Compounded semaglutide has faced a similar but distinct regulatory trajectory, and newer agents like retatrutide are in development. Understanding how semaglutide and tirzepatide compare can help you evaluate whether a switch makes sense for your situation.
The future of 503B tirzepatide compounding
Predicting the regulatory future is risky, but the key factors shaping the landscape are identifiable.
The appeal and ongoing litigation
The Outsourcing Facilities Association's appeal to the Fifth Circuit Court of Appeals could change the calculus. If the appellate court reverses the district court and finds that the FDA improperly removed tirzepatide from the shortage list, compounding could resume under the shortage exception. This outcome is not guaranteed, but it is possible, and the Fifth Circuit has historically been willing to check federal agency authority.
Congressional action
Several members of Congress have expressed concern about patient access to compounded GLP-1 medications. Proposed legislation could create new legal pathways for compounding even when a drug is not on the shortage list, particularly for patients who cannot afford the brand-name version. Whether any such legislation passes is uncertain, but the political pressure is real and growing.
FDA guidance evolution
The FDA could issue new guidance clarifying the "essentially copy" doctrine as it applies to GLP-1 combination products. This guidance could either expand or restrict the current gray zone that combination formulations occupy. Compounders and their legal teams are watching closely for any signals from the agency.
Market dynamics
Eli Lilly has an obvious interest in protecting its tirzepatide franchise. The company has pursued both legal and regulatory strategies to limit compounding. At the same time, the sheer size of the demand, millions of patients who cannot afford brand-name pricing, creates sustained political and market pressure for accessible alternatives.
New GLP-1 agents entering the market could also shift the landscape. Retatrutide, a triple-agonist peptide in development, could provide new options if approved. Oral tirzepatide formulations from Eli Lilly itself could address some access concerns by offering a different product form.
For researchers and patients navigating the current uncertainty, SeekPeptides provides the most comprehensive and up-to-date guidance available. Members access detailed protocol databases, dosing guides, and community knowledge that helps cut through the regulatory noise and focus on what actually works.
Storage and handling of 503B compounded tirzepatide
Compounded tirzepatide from a 503B facility requires proper storage and handling to maintain potency and sterility. The rules are similar to brand-name products but with some important differences.
Refrigeration requirements
Most compounded tirzepatide must be refrigerated between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Unlike the commercial pen devices, compounded vials are often multi-dose, meaning you will draw from the same vial multiple times. Each time you access the vial, you introduce a small contamination risk, making proper refrigeration and storage essential.
If your compounded tirzepatide is left out of the fridge, the time and temperature it was exposed to matters. Brief excursions at room temperature are usually acceptable, but extended exposure can degrade the peptide and compromise sterility. When in doubt, contact the compounding pharmacy for guidance specific to their formulation. Understanding what happens if tirzepatide gets warm can help you make informed decisions about product viability.
Shelf life and expiration
503B compounded products must include beyond-use dating based on stability data. This is different from the expiration date on a brand-name product, which is determined through formal FDA-required stability studies. Beyond-use dates for compounded products are typically shorter, often 30 to 90 days when refrigerated, depending on the formulation and testing data.
Check the expiration date on your vial and plan your dosing schedule accordingly. Using compounded tirzepatide beyond its beyond-use date is not recommended, as both potency and sterility may be compromised. The shelf life of tirzepatide varies by formulation and manufacturer.
Reconstitution considerations
Some 503B compounded tirzepatide ships as a lyophilized (freeze-dried) powder that must be reconstituted with bacteriostatic water before use. The amount of bacteriostatic water you add determines the concentration, which directly affects your dosing. Using our peptide reconstitution calculator ensures you get the math right every time.
Proper reconstitution technique matters. Contamination during reconstitution can introduce bacteria into an otherwise sterile product. Always use a new sterile needle, swab the vial stopper with alcohol, and inject the bacteriostatic water slowly to avoid damaging the peptide. Roll the vial gently, never shake it, to dissolve the powder completely.
Injection guidance for compounded tirzepatide
Whether your tirzepatide comes from a 503B facility or any other source, proper injection technique is essential for safety and efficacy.
Choosing an injection site
Subcutaneous injection sites for tirzepatide include the abdomen (at least 2 inches from the navel), the front of the thigh, and the back of the upper arm. Rotating injection sites helps prevent lipodystrophy and injection site reactions. Many users find that abdominal injections provide the most consistent absorption, though individual experiences vary.
Drawing from a multi-dose vial
Unlike brand-name tirzepatide pens, compounded products from 503B facilities typically come in multi-dose vials. This means you need to know how to draw the correct dose using an insulin syringe. Understanding your tirzepatide syringe dosage in units is critical for accuracy.
Each time you draw from a multi-dose vial, clean the stopper with an alcohol swab. Use a new sterile needle for each injection. Draw air equal to your dose volume, inject it into the vial to equalize pressure, then draw your dose. Check for air bubbles and tap them out before injecting.
If you are new to self-injection, our guides on how to inject GLP-1 medications and where to inject tirzepatide provide step-by-step instructions with visual references.
Managing side effects from compounded tirzepatide
The side effect profile of compounded tirzepatide is generally similar to the brand-name product, though the addition of other active ingredients in combination formulations may introduce their own effects. Common side effects include gastrointestinal symptoms that most users experience to some degree.
Gastrointestinal effects
Nausea is the most commonly reported side effect, particularly during the initial weeks and with each dose increase. Diarrhea and constipation can occur, sometimes alternating. Proper dietary management, including avoiding high-fat and heavily processed foods, can help minimize these symptoms. A well-planned tirzepatide diet plan makes a significant difference in tolerability.
Fatigue and energy changes
Fatigue on tirzepatide is commonly reported, particularly in the first few weeks. This can be related to reduced caloric intake, metabolic adjustments, or the medication itself. Some compounded formulations that include B12 or energy-supporting compounds may help offset this effect. Ensuring adequate protein intake and staying hydrated are practical strategies that help.
Other reported effects
Headaches, insomnia, body aches, anxiety, and muscle pain have all been reported by tirzepatide users. These effects are typically dose-dependent and often improve over time as the body adjusts. Starting with a conservative starting dose and titrating slowly is the most reliable strategy for minimizing side effects.
Tracking your response, including both benefits and side effects, helps your provider optimize your protocol. SeekPeptides members use detailed tracking tools and community insights to fine-tune their approaches based on real-world experience from thousands of other researchers.
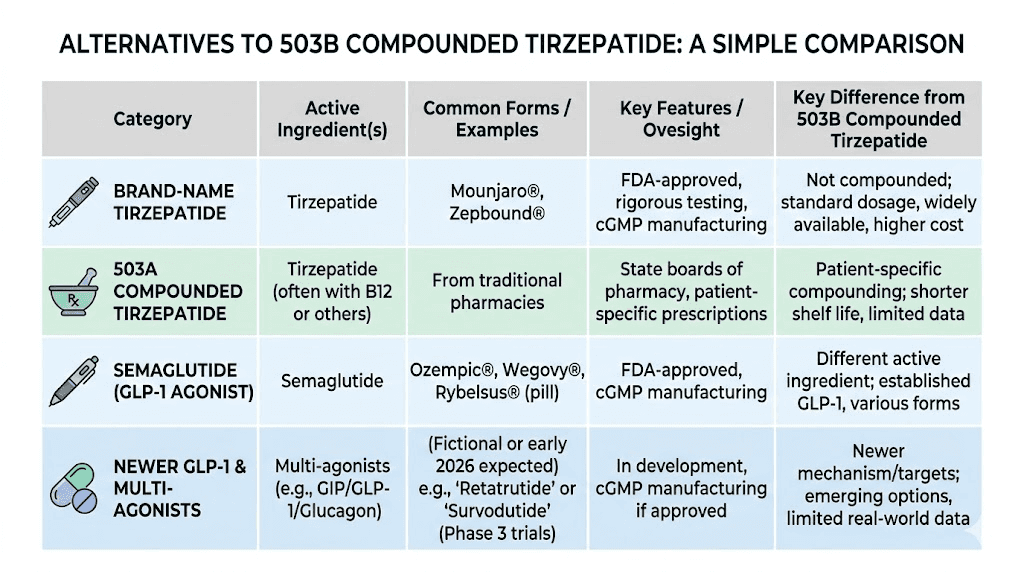
Alternatives if 503B compounded tirzepatide is unavailable
If 503B compounding of tirzepatide becomes fully restricted or you prefer options with clearer legal standing, several alternatives exist.
503A compounding with documented clinical need
503A pharmacies can still compound tirzepatide for individual patients who have a documented clinical need that the commercial product cannot address. The most common qualifying scenario involves allergies to inactive ingredients in the brand-name formulation. If you have a documented allergy, discuss this pathway with your healthcare provider.
Brand-name Mounjaro or Zepbound
The FDA-approved products offer guaranteed quality, potency, and sterility. They are also significantly more expensive. However, Eli Lilly's savings programs, manufacturer coupons, and some insurance plans can reduce the out-of-pocket cost substantially. It is worth exploring all available financial assistance options before ruling out brand-name products entirely.
Compounded semaglutide
Compounded semaglutide has faced its own regulatory journey but with some differences from tirzepatide. The side effect profiles differ between the two medications, and comparing semaglutide and tirzepatide head-to-head can help you decide whether switching makes sense. Our semaglutide dosage calculator helps with dose conversions if you make the switch.
Newer GLP-1 agents
Retatrutide, a triple-receptor agonist targeting GLP-1, GIP, and glucagon receptors, is in late-stage clinical trials with promising results. Survodutide and mazdutide represent additional next-generation options being evaluated. While these are not yet widely available, the GLP-1 pipeline is robust and growing.
Lifestyle optimization
For patients who cannot access any GLP-1 medication, optimizing diet, exercise, and metabolic health through other means remains important. Understanding the dietary principles that support GLP-1 therapy can provide benefits even without the medication, and strategic supplementation may help bridge the gap.
Frequently asked questions
What is the difference between a 503A and 503B compounding pharmacy?
A 503A pharmacy is a traditional state-regulated compounding pharmacy that fills individual patient prescriptions. A 503B outsourcing facility is an FDA-registered, federally regulated operation that can produce compounded medications in bulk without patient-specific prescriptions. 503B facilities follow cGMP manufacturing standards and undergo regular FDA inspections, while 503A pharmacies are primarily overseen by state pharmacy boards.
Can 503B pharmacies still legally compound tirzepatide?
Standard tirzepatide injections that are essentially copies of Mounjaro or Zepbound cannot be legally compounded by 503B facilities since the FDA declared the shortage resolved in December 2024, with the enforcement deadline passing in March 2025. However, combination formulations that are meaningfully different from the commercial product, such as tirzepatide with niacinamide or glycine and B12, may have a legal pathway, though this remains legally uncertain.
Is compounded tirzepatide from a 503B facility safe?
503B facilities operate under FDA oversight with cGMP standards, which provides a higher level of quality assurance than many other compounding models. However, the FDA has documented sterility failures, subpotency issues, and contamination at some 503B facilities. Always verify the facility's registration and inspection history, and request certificates of analysis for your specific batch.
How much does compounded tirzepatide cost compared to brand name?
Compounded tirzepatide from 503B facilities has typically cost between $200 and $500 per month, while brand-name Mounjaro and Zepbound exceed $1,000 per month without insurance coverage. Cost varies based on dosage, formulation, and the specific pharmacy or telehealth platform used.
What happened in the Outsourcing Facilities Association v. FDA case?
The OFA sued the FDA after it removed tirzepatide from the drug shortage list. On May 7, 2025, the US District Court for the Northern District of Texas upheld the FDA's decision, finding the agency acted within its statutory authority. OFA has filed an appeal to the Fifth Circuit Court of Appeals, and the case remains ongoing.
Should I switch from compounded tirzepatide to brand name?
This depends on your specific situation, including cost, insurance coverage, and whether your current compounded product is being produced legally. If your 503B pharmacy has ceased production, transitioning to brand-name or exploring alternative GLP-1 options may be necessary. Consult your healthcare provider for guidance tailored to your circumstances.
How do I find a legitimate 503B outsourcing facility?
Check the FDA's public registry of registered outsourcing facilities. Review inspection histories for any warning letters or serious violations. Verify state licensing, ask for certificates of analysis, and confirm that the facility sources its API from FDA-registered establishments. A facility that cannot provide transparent documentation about its operations and testing should be avoided.
What formulations of compounded tirzepatide are still available?
Combination formulations that differ from the commercially available product may still be available. These include tirzepatide with B12, tirzepatide with glycine, and tirzepatide ODT (orally disintegrating tablets). The legal status of these products varies, and patients should verify the legal basis with their provider.
External resources
For researchers serious about staying ahead of the regulatory curve, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access real-time updates on compounding regulations, detailed provider evaluations, and protocol databases that reflect the latest developments.
In case I do not see you, good afternoon, good evening, and good night. May your pharmacies stay legitimate, your formulations stay potent, and your protocols stay effective.