Feb 12, 2026

People use the terms interchangeably. Doctors sometimes do too. GLP-1, Ozempic, semaglutide, they all get thrown into the same bucket as if they mean the same thing. They do not. One is a hormone your body produces naturally every time you eat a meal. The other is a pharmaceutical product designed by Novo Nordisk, sold under a specific brand name, prescribed for specific conditions. The confusion is understandable because Ozempic works by mimicking that natural hormone, but understanding the difference between what your body already makes and what a medication does changes everything about how you approach weight loss peptides, diabetes management, and metabolic health in general.
This distinction matters more than most people realize. When someone asks their doctor about "getting on GLP-1," they might mean Ozempic specifically, or they might mean any medication in the entire class of GLP-1 receptor agonists. The answer they get depends entirely on which meaning their doctor assumes. And that gap between what people say and what they mean has real consequences for treatment decisions, expectations, and outcomes. This guide will clarify exactly what GLP-1 is, what Ozempic is, how they relate to each other, and why the difference matters for anyone considering these options for weight management or blood sugar control.
What GLP-1 actually is
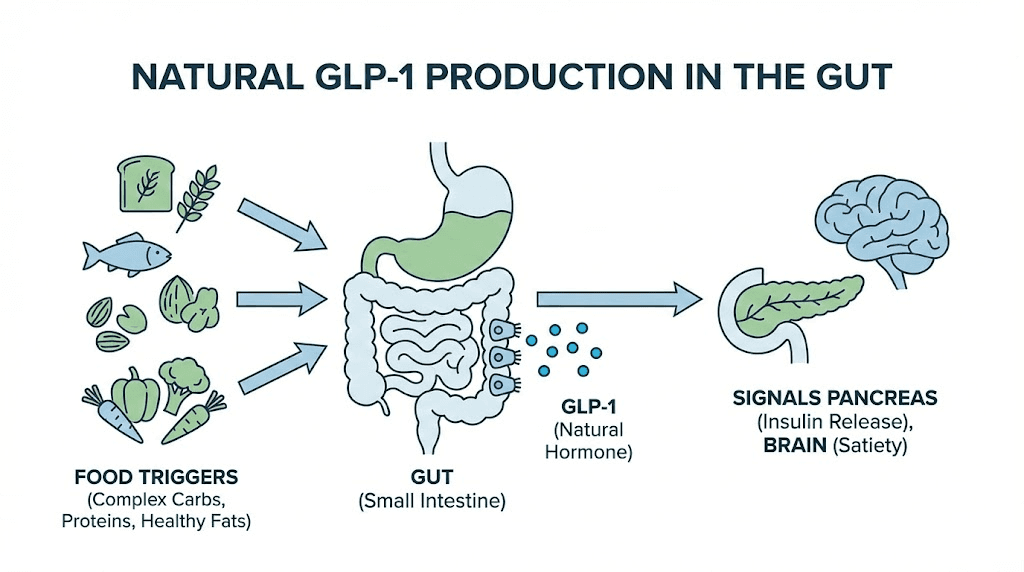
GLP-1 stands for glucagon-like peptide-1. It is a hormone. Your body makes it. Specifically, L-cells in your small intestine produce GLP-1 and release it into your bloodstream after you eat. This is not a drug, not a treatment, not something you buy at a pharmacy. It is a naturally occurring peptide that plays several critical roles in how your body processes food, manages blood sugar, and regulates appetite.
Think of it this way.
Every time you eat a meal, your gut sends chemical signals to your brain and your pancreas. GLP-1 is one of the most important of those signals. It tells your pancreas to release insulin, which brings blood sugar down. It tells your liver to stop dumping glucose into your bloodstream. It tells your stomach to slow down, keeping food in there longer so you feel full. And it communicates directly with your brain through the hypothalamus, reducing hunger and dampening cravings for high calorie foods.
The problem is timing. Natural GLP-1 has a circulating half-life of only 1 to 2 minutes. Your body produces it, it does its job, and then an enzyme called DPP-4 breaks it down almost immediately. This rapid breakdown is fine for healthy metabolic function, but it means you cannot simply inject natural GLP-1 and expect lasting effects. The molecule disappears too quickly to be useful as a medication.
That limitation is exactly why pharmaceutical companies developed synthetic versions. They needed something that acted like GLP-1 but lasted long enough to be practical as a treatment.
The roles GLP-1 plays in your body
Understanding what GLP-1 does helps clarify why so many medications now target this pathway. The hormone influences at least four major systems simultaneously, and each one contributes to metabolic health in a different way.
Insulin secretion. GLP-1 stimulates your pancreas to release insulin, but only when blood sugar is elevated. This glucose-dependent mechanism is important because it means GLP-1 does not cause dangerous blood sugar drops the way some older diabetes medications can. Your body already has this safety mechanism built in. The pancreatic peptide system responds to rising glucose levels and GLP-1 amplifies that response without overriding it.
Glucagon suppression. While GLP-1 increases insulin, it simultaneously reduces glucagon, a hormone that raises blood sugar by telling your liver to release stored glucose. This dual action, boosting insulin while suppressing glucagon, creates a powerful blood sugar lowering effect that works from two angles at once.
Gastric emptying. GLP-1 slows the rate at which food moves from your stomach into your small intestine. Slower gastric emptying means glucose enters your bloodstream more gradually after meals, preventing the sharp spikes that contribute to insulin resistance over time. It also means you feel physically full for longer after eating, which naturally reduces caloric intake without conscious effort.
Appetite regulation. Signals from GLP-1 travel to the brain through both the bloodstream and the vagus nerve. Once they reach the hypothalamus, these signals reduce hunger, decrease the desire for high calorie foods, and increase the sensation of satisfaction after eating. This central nervous system effect is arguably the most powerful mechanism behind the weight loss seen with GLP-1 based medications.
How your body naturally produces GLP-1
Your L-cells respond to specific nutritional triggers. Protein is one of the strongest stimulators of GLP-1 release, which partly explains why high protein diets tend to reduce appetite and improve blood sugar control. Healthy fats, particularly monounsaturated fats and omega-3 fatty acids, also trigger significant GLP-1 release from gut cells.
Fiber matters too. Fermentable fibers found in foods like artichokes, asparagus, lentils, beans, and bananas feed gut bacteria that produce short-chain fatty acids, which in turn stimulate L-cells to release more GLP-1. This is one reason why researchers often recommend eating protein and fiber before carbohydrates at meals, because doing so primes the GLP-1 response before glucose hits your bloodstream.
Exercise amplifies GLP-1 production as well. Both moderate and high intensity exercise increase GLP-1 levels regardless of the type of movement, and the combination of aerobic and resistance training appears to be especially effective. Some studies suggest that exercise-induced GLP-1 elevations persist for 24 hours after a workout, providing extended metabolic benefits beyond the exercise session itself.
Even simple habits make a difference. Eating slowly, taking smaller bites, and spacing meals throughout the day all support healthier GLP-1 secretion patterns. Chronic stress, on the other hand, increases cortisol and impairs GLP-1 release, creating a metabolic environment that promotes overeating and insulin resistance. Managing stress through sleep, movement, and recovery is not just good advice. It directly supports the hormonal systems that regulate your weight and blood sugar.
What Ozempic actually is
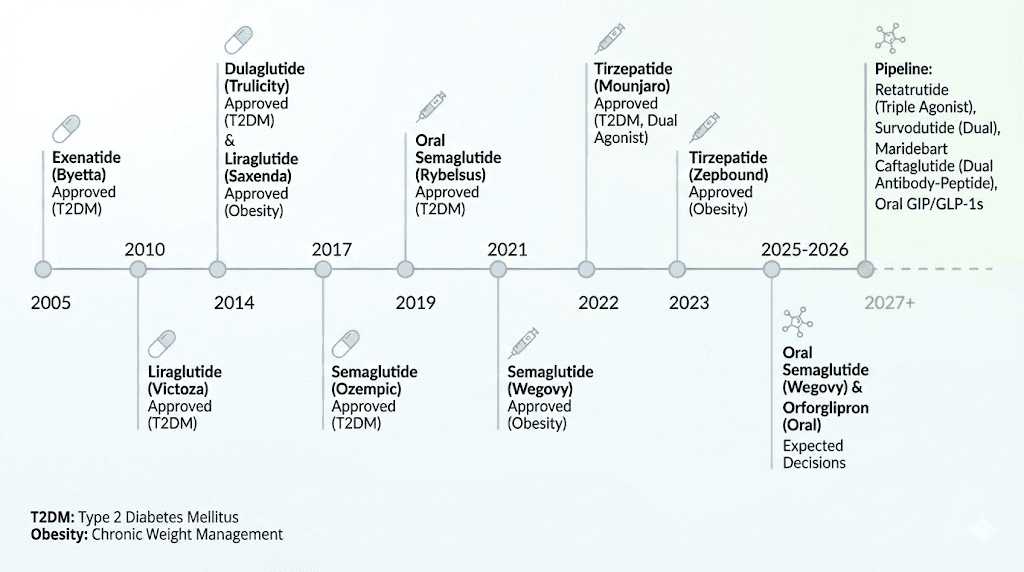
Ozempic is a brand name. The active ingredient is semaglutide, a synthetic molecule designed to mimic the effects of natural GLP-1 while lasting dramatically longer in the body. Novo Nordisk developed semaglutide and received FDA approval for Ozempic in December 2017 as a treatment for type 2 diabetes.
Semaglutide shares 94% structural similarity with human GLP-1. That 6% difference is what makes it useful. Researchers modified the amino acid sequence and attached a fatty acid chain that allows semaglutide to bind to albumin in your blood, shielding it from the DPP-4 enzyme that destroys natural GLP-1 within minutes. The result is a molecule with a half-life of approximately one week instead of one to two minutes. That extended duration means a single weekly injection provides continuous GLP-1 receptor activation for seven days.
This is the core innovation. Same receptor targets. Same downstream effects. Massively different duration of action. And that duration changes everything about how effectively the medication works for blood sugar control and weight loss.
Ozempic is just one brand name for semaglutide
Here is where the confusion multiplies. Semaglutide is not only sold as Ozempic. The same active molecule appears under three different brand names, each approved for different uses and sold at different doses.
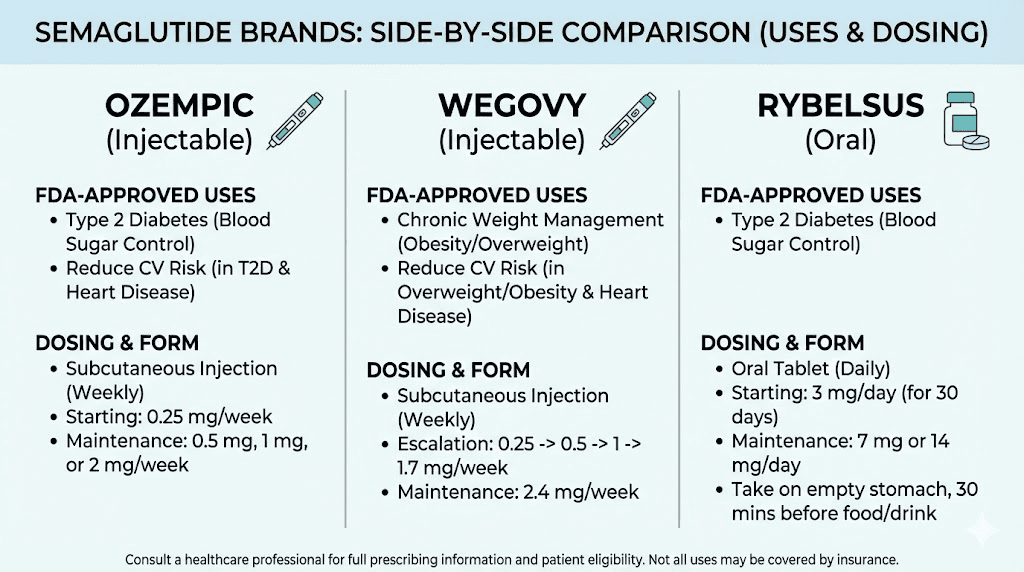
Ozempic is the injectable form approved for type 2 diabetes management. It comes in doses of 0.25 mg, 0.5 mg, 1 mg, and 2 mg, administered once weekly by subcutaneous injection. The primary indication is blood sugar control, though weight loss occurs as a significant secondary effect.
Wegovy uses the same semaglutide molecule but at a higher dose, up to 2.4 mg weekly, and is specifically approved for chronic weight management in adults with obesity or overweight with at least one weight-related condition. The weight loss protocol with semaglutide involves gradual dose escalation over 16 to 20 weeks to minimize gastrointestinal side effects.
Rybelsus is the oral tablet form of semaglutide, taken daily instead of weekly, and approved for type 2 diabetes. It represents the first and currently only GLP-1 receptor agonist available in pill form, using an absorption enhancer called SNAC to help the peptide survive the digestive system. The concept of oral peptide delivery continues to evolve as researchers develop more options for people who prefer pills over injections.
So when someone says they are taking "Ozempic for weight loss," they might actually be on Wegovy, since that is the semaglutide product specifically indicated for weight management. Or they might genuinely be using Ozempic off-label for weight loss, which is common but technically not the approved use. The active ingredient is the same. The dose and indication are different.
How Ozempic works in the body
Ozempic activates the same GLP-1 receptors that your natural hormone targets, but it does so continuously for a full week rather than for a few minutes after meals. This sustained activation amplifies every effect that natural GLP-1 produces.
The semaglutide dosing protocol starts low and increases gradually. Patients typically begin at 0.25 mg weekly for the first four weeks, then move to 0.5 mg. From there, the dose can increase to 1 mg and eventually 2 mg depending on the clinical response and tolerance. This titration strategy exists because the gastrointestinal side effects, particularly nausea, are dose-dependent and tend to diminish as the body adjusts.
In clinical trials, semaglutide at the 2.4 mg weekly dose (Wegovy) produced an average placebo-corrected weight loss of approximately 12 to 15% of body weight over 68 weeks. For someone weighing 220 pounds, that translates to roughly 26 to 33 pounds of weight loss beyond what placebo participants lost. The appetite suppression from semaglutide typically becomes noticeable within the first few weeks, with significant weight loss accumulating over months of consistent use.
The relationship between GLP-1 and Ozempic
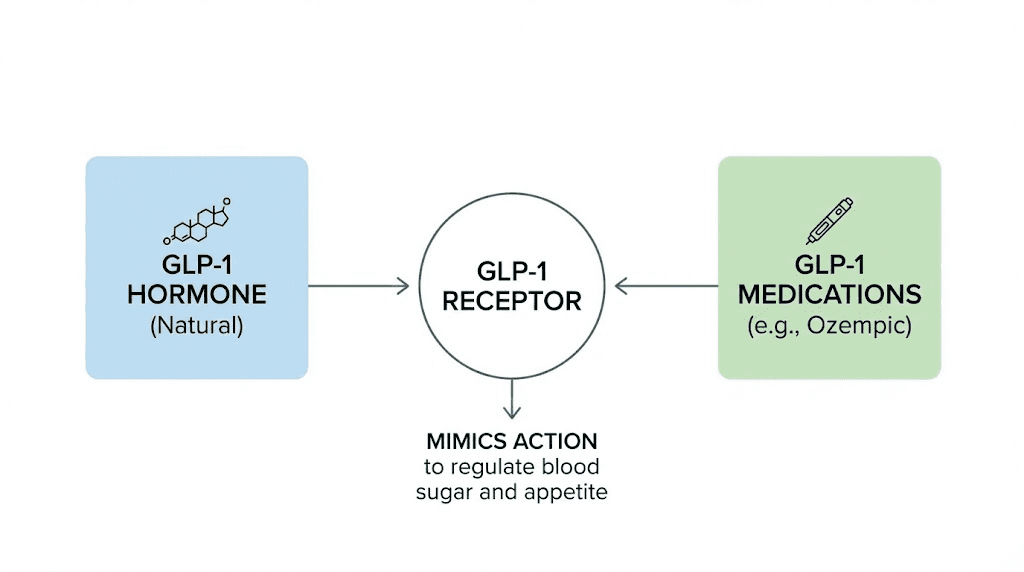
Now that both terms are clear individually, the relationship between them becomes straightforward. GLP-1 is the natural hormone. Ozempic is a medication that mimics it. Ozempic belongs to a class of drugs called GLP-1 receptor agonists, which means it activates the same receptors that GLP-1 activates, producing the same downstream effects but with much greater intensity and duration.
The analogy works like this. Imagine your natural GLP-1 as a match that lights briefly after each meal and then goes out. Ozempic is like a blowtorch that stays lit for an entire week. Same type of flame. Same receptors being activated. Vastly different intensity and duration. That sustained activation is what produces the dramatic effects on weight and blood sugar that have made these medications so popular.
But Ozempic is not the only medication in this class. Not even close.
The full GLP-1 receptor agonist family
Multiple GLP-1 receptor agonists exist, each with different dosing schedules, durations of action, and clinical profiles. Understanding the full family helps people make informed decisions rather than defaulting to whichever brand name they have heard most often.
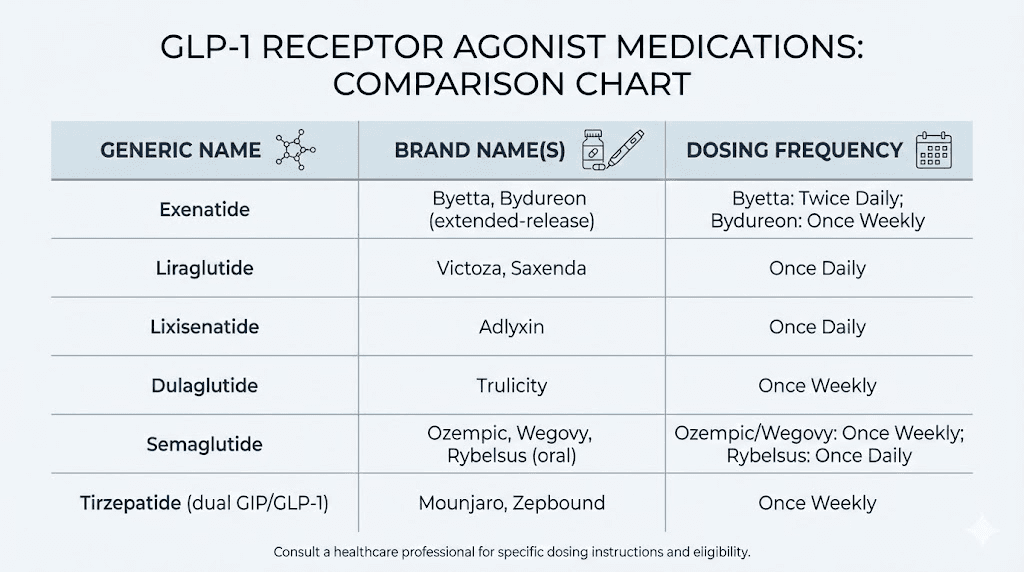
Semaglutide (Ozempic, Wegovy, Rybelsus) is a once-weekly injectable or daily oral medication. It shares 94% homology with natural GLP-1. Clinical trials showed placebo-corrected weight loss of approximately 12 to 15% with the weight management dose. Discontinuation typically leads to weight regain, which is an important consideration for anyone starting treatment.
Liraglutide (Victoza, Saxenda) was one of the earlier GLP-1 receptor agonists, approved for diabetes as Victoza in 2010 and for weight management as Saxenda in 2014. It requires daily injection and produces more modest weight loss than semaglutide, with placebo-corrected losses averaging around 5%. Both products are manufactured by Novo Nordisk.
Tirzepatide (Mounjaro, Zepbound) represents the next evolution. Rather than targeting GLP-1 receptors alone, tirzepatide activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors simultaneously. This dual mechanism produces stronger effects on both blood sugar and body weight. In clinical trials, tirzepatide at the highest dose produced average weight loss of approximately 20.2% of body weight over 72 weeks. For comparison, tirzepatide results consistently outperform semaglutide in head-to-head comparisons, though both are effective.
Dulaglutide (Trulicity) is another once-weekly injectable GLP-1 receptor agonist, approved for type 2 diabetes. Manufactured by Eli Lilly and approved in 2014, it produces moderate weight loss and blood sugar improvement but is not specifically indicated for weight management.
Exenatide (Byetta, Bydureon) was the first GLP-1 receptor agonist approved in the United States, with Byetta receiving approval in 2005 as a twice-daily injection and Bydureon later available as a once-weekly formulation. It remains in use but has largely been superseded by newer agents with better efficacy profiles.
Each of these medications targets GLP-1 receptors. Each mimics the natural GLP-1 hormone. None of them IS GLP-1. They are all synthetic drugs designed to replicate and extend its effects.
Why people confuse GLP-1 with Ozempic
The confusion is not random. Several factors drive it, and understanding them helps clarify the conversation.
Media coverage. News outlets frequently use "GLP-1" and "Ozempic" interchangeably because Ozempic became the most recognized brand name in the class. When a news story says "GLP-1 drugs like Ozempic," readers often collapse both terms into a single concept. Over time, GLP-1 becomes synonymous with Ozempic in the public consciousness, even though GLP-1 refers to the hormone (or the entire drug class) while Ozempic refers to one specific product.
Doctor shorthand. Healthcare providers sometimes say "I am going to put you on a GLP-1" when they mean a GLP-1 receptor agonist medication. The specific product they prescribe might be Ozempic, Wegovy, Mounjaro, or something else entirely. Patients hear "GLP-1" and assume it means Ozempic because that is the name they recognize.
Social media. On platforms like TikTok and Instagram, people share their experiences with "GLP-1" or "Ozempic" without distinguishing between the two, creating a massive volume of content where both terms appear to mean the same thing.
The practical impact of this confusion shows up in clinical encounters. A patient might tell their doctor they want Ozempic when they actually want any GLP-1 medication that helps with weight loss. Or they might refuse a prescription for Wegovy because they wanted "the real thing" (Ozempic), not realizing both contain the same active ingredient. Some people even confuse tirzepatide products with semaglutide products because everything gets lumped under the "GLP-1" umbrella.
Ozempic vs other GLP-1 medications
Choosing between GLP-1 medications involves understanding real differences in mechanism, efficacy, side effects, and approved indications. This is not a situation where all options are interchangeable.
Semaglutide vs tirzepatide
The most common comparison people encounter is between semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). These represent two different approaches to targeting metabolic pathways.
Semaglutide activates GLP-1 receptors only. Tirzepatide activates both GLP-1 and GIP receptors, making it a dual-agonist. That second pathway, GIP receptor activation, enhances insulin secretion through an additional mechanism and appears to contribute to greater fat burning effects. The clinical data reflects this difference clearly.
Factor | Semaglutide (Ozempic/Wegovy) | Tirzepatide (Mounjaro/Zepbound) |
|---|---|---|
Mechanism | GLP-1 receptor agonist | Dual GLP-1/GIP receptor agonist |
Weight loss | ~13-15% body weight | ~20% body weight |
Dosing | Once weekly injection | Once weekly injection |
Diabetes indication | Ozempic (type 2 diabetes) | Mounjaro (type 2 diabetes) |
Weight loss indication | Wegovy (obesity/overweight) | Zepbound (obesity/overweight) |
Manufacturer | Novo Nordisk | Eli Lilly |
Both medications produce significant weight loss and blood sugar improvement. Tirzepatide generally produces more weight loss in clinical trials, but individual responses vary. Some people respond better to one than the other for reasons researchers do not fully understand yet. The process of switching between them is something many patients navigate as they find the option that works best for their body.
Diabetes brands vs weight loss brands
A critical distinction that many people miss is the separation between diabetes indications and weight loss indications.
Ozempic and Mounjaro are FDA-approved for type 2 diabetes. They help manage blood sugar levels and carry indications specific to metabolic disease management. Weight loss happens as a secondary benefit, often significant, but the drugs are technically marketed and prescribed for glucose control.
Wegovy and Zepbound are FDA-approved for chronic weight management. They are indicated for adults with a BMI of 30 or greater (obesity) or 27 or greater (overweight) with at least one weight-related comorbidity. The doses are often higher than their diabetes counterparts, optimized for maximum weight loss rather than blood sugar management alone.
Same active ingredients. Different doses. Different indications. Different insurance coverage pathways. Someone trying to get tirzepatide for weight loss specifically would need Zepbound, not Mounjaro, unless their doctor prescribes Mounjaro off-label.
Understanding compounded versions
The conversation gets more complicated with compounded semaglutide and compounded tirzepatide. During periods of drug shortages, compounding pharmacies have been allowed to produce versions of these medications. Compounded semaglutide contains the same active molecule but is manufactured by compounding pharmacies rather than by Novo Nordisk.
People researching compounded options often search for information about compounded semaglutide storage, refrigeration requirements, and compounded tirzepatide pricing. The reconstitution process for compounded semaglutide differs from using a prefilled pen, requiring knowledge of proper mixing techniques and bacteriostatic water ratios.
How GLP-1 medications actually work in your body
Whether someone takes Ozempic, Wegovy, Mounjaro, or any other GLP-1 receptor agonist, the fundamental mechanism is the same. The medication activates GLP-1 receptors throughout the body, producing amplified and extended versions of the same effects your natural GLP-1 hormone creates.
The brain connection
GLP-1 receptors exist in the hypothalamus, the region of your brain that controls hunger, satiety, and food reward. When natural GLP-1 reaches these receptors after a meal, it reduces hunger for a brief period. When a synthetic GLP-1 agonist like semaglutide continuously activates these receptors for a full week, the hunger suppression becomes constant and dramatic.
People on these medications consistently describe a fundamental shift in their relationship with food. The constant background noise of cravings, the pull toward snacking, the difficulty stopping after one serving, it all quiets down. This is not willpower. It is neurochemistry. The medication is activating receptors in the brain that control these drives, producing a state of reduced food interest that most people have never experienced before.
This brain effect is also why people sometimes experience what researchers call "food noise reduction." That mental chatter about food, the planning of the next meal while eating the current one, the difficulty walking past a bakery without stopping, diminishes significantly on GLP-1 therapy. For many people, this psychological shift is even more impactful than the direct metabolic effects.
The gut connection
Slowed gastric emptying is one of the most noticeable effects. Food stays in the stomach longer, creating prolonged feelings of fullness and reducing the desire to eat again quickly. This delayed emptying also smooths out blood sugar responses after meals, preventing the sharp spikes and crashes that drive further eating.
However, this same mechanism is responsible for some of the most common side effects. Nausea, fatigue, and digestive discomfort occur precisely because the stomach is emptying more slowly than the body expects. Most people find these symptoms manageable and temporary, typically improving within the first few weeks of each dose increase as the body adapts to the new rate of gastric emptying.
The pancreas connection
Enhanced insulin secretion in response to glucose is the original therapeutic purpose of GLP-1 medications. For people with type 2 diabetes, this effect directly addresses the core problem of inadequate insulin response. The glucose-dependent nature of this mechanism means insulin is only released when blood sugar is actually elevated, reducing the risk of hypoglycemia that comes with some older diabetes medications.
At the same time, GLP-1 receptor activation suppresses inappropriate glucagon release from the pancreas. Glucagon normally tells the liver to release stored glucose, raising blood sugar between meals. In type 2 diabetes, glucagon release is often poorly regulated, contributing to high fasting blood sugar levels. GLP-1 medications help correct this imbalance.
The cardiovascular connection
Emerging research suggests that GLP-1 receptor agonists provide cardiovascular benefits beyond what would be expected from improved blood sugar and weight loss alone. The SELECT trial with semaglutide demonstrated a 20% reduction in major adverse cardiovascular events in people with established cardiovascular disease and obesity but without diabetes. These findings have expanded the potential clinical applications of these medications beyond metabolic disease into cardiovascular risk reduction.
Side effects of GLP-1 medications
Every medication that mimics GLP-1 shares a similar side effect profile because they all activate the same receptors. Understanding these effects helps set realistic expectations and prepare for management strategies.
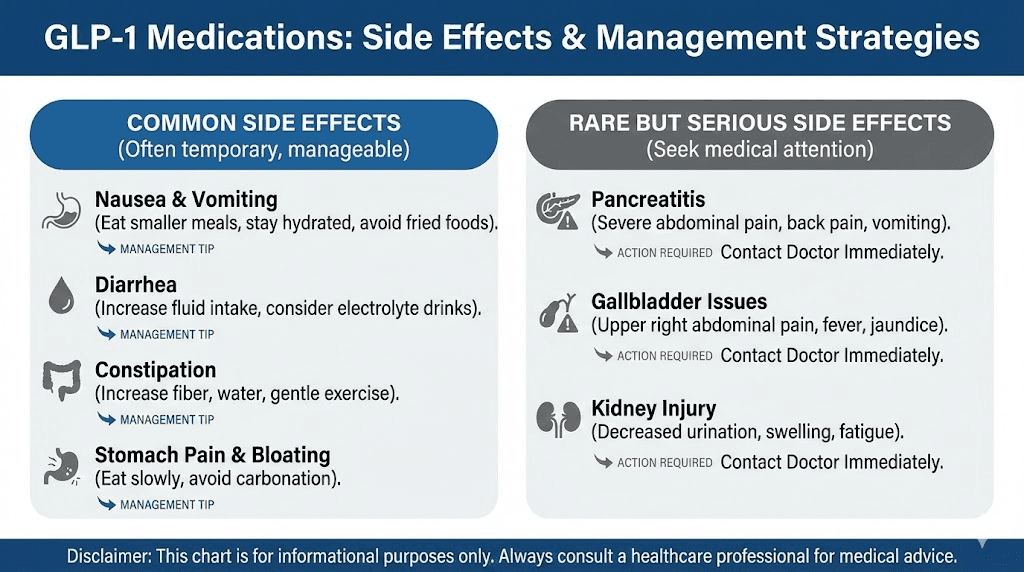
Common gastrointestinal effects
Nausea is the most frequently reported side effect across all GLP-1 receptor agonists. Studies consistently show that 15 to 44% of patients experience nausea, with higher rates during dose escalation periods. The nausea typically peaks during the first week or two at each new dose level and then subsides as the body adjusts.
Constipation affects a significant percentage of users, and dietary adjustments often help manage it. Diarrhea, vomiting, and abdominal discomfort are also reported, though less commonly than nausea. Most gastrointestinal side effects are classified as mild to moderate and do not lead to discontinuation of the medication.
Practical management strategies include eating smaller meals more frequently, avoiding high fat or very rich foods, staying well hydrated, and eating slowly. Many physicians recommend starting at the lowest available dose and increasing gradually over weeks to months, giving the body time to adapt at each level.
Other effects to know about
Hair loss is a concern that many people research before starting GLP-1 therapy. The hair thinning that some users experience is generally attributed to rapid weight loss rather than to the medication itself, a phenomenon called telogen effluvium that can occur with any significant caloric deficit or weight reduction.
Anxiety and mood changes have been reported by some users, though the relationship between GLP-1 medications and mental health is still being studied. The brain contains GLP-1 receptors in regions involved in mood regulation, which provides a plausible mechanism for these reports, but large-scale clinical trials have not consistently identified increased anxiety as a common side effect.
Fatigue during the initial weeks of treatment is common and likely reflects the combination of reduced caloric intake, metabolic adjustment, and the body adapting to lower blood sugar levels. Most users report that energy levels normalize or even improve once they adjust to the medication and establish new eating patterns.
Rare but serious potential risks include pancreatitis, gallbladder problems, and in animal studies, thyroid tumors. These risks are monitored through clinical trials and post-marketing surveillance, and they represent a small fraction of overall users.
GLP-1 medications beyond weight loss and diabetes
The story of GLP-1 receptor agonists is expanding rapidly beyond their original indications. Research into new applications has accelerated dramatically, revealing that activating GLP-1 receptors produces benefits across multiple organ systems.
Cardiovascular protection
The cardiovascular benefits of GLP-1 receptor agonists have become one of the most compelling areas of research. Beyond the SELECT trial results mentioned earlier, multiple studies have demonstrated reduced rates of heart attack, stroke, and cardiovascular death in patients using semaglutide and liraglutide. These benefits appear to be partly independent of weight loss and blood sugar improvement, suggesting direct cardiovascular protective effects from GLP-1 receptor activation.
For people with existing heart disease and obesity, this dual benefit of weight loss and cardiovascular protection makes GLP-1 medications particularly attractive compared to other weight loss interventions.
Liver disease
Metabolic dysfunction-associated steatohepatitis (MASH), previously known as nonalcoholic steatohepatitis, affects millions of people with obesity and insulin resistance. GLP-1 medications have shown promising results in reducing liver fat, inflammation, and fibrosis in patients with MASH. Semaglutide has received regulatory interest for this indication, and survodutide, a newer dual GLP-1/glucagon receptor agonist currently in clinical trials, has shown particularly strong results in MASH treatment.
Kidney protection
The FLOW trial demonstrated that semaglutide significantly reduced the risk of kidney disease progression in people with type 2 diabetes and chronic kidney disease. This renal protective effect represents another dimension of benefit beyond metabolic management.
Addiction and substance use
Some of the most surprising emerging research involves GLP-1 receptor agonists and addictive behaviors. Animal studies and early human data suggest that activating GLP-1 receptors in the brain may reduce cravings for alcohol, nicotine, and other addictive substances. While clinical trials specifically designed to test these effects are still in early stages, the preliminary findings have generated significant scientific interest.
The connection makes mechanistic sense. GLP-1 receptors exist in brain reward circuits that overlap with addiction pathways. If these medications can modulate reward-seeking behavior for food, they may also influence reward-seeking behavior for other substances.
The next generation of GLP-1 drugs
The pharmaceutical pipeline for GLP-1 based therapies is expanding rapidly, with several new medications in late-stage clinical trials that promise greater efficacy, more convenient delivery, or both.
Retatrutide
Retatrutide is a triple-agonist developed by Eli Lilly that activates GLP-1, GIP, and glucagon receptors simultaneously. Early clinical trial data shows exceptional weight loss, with participants losing up to an average of 71 pounds over the treatment period. The addition of glucagon receptor activation promotes fat burning and energy expenditure beyond what dual-agonists achieve. The availability of retatrutide is projected around 2028, with cost and pricing details still being determined.
The retatrutide dosage protocol in clinical trials has involved gradual titration similar to existing GLP-1 medications. The dosing schedule follows a weekly injection pattern, and early data suggests the side effect profile is comparable to existing options. For those comparing options, the retatrutide vs semaglutide comparison highlights the potential advantages of triple versus single receptor targeting.
Orforglipron
Also developed by Eli Lilly, orforglipron represents a different kind of innovation. Rather than improving on existing injectable peptides, orforglipron is a non-peptide oral GLP-1 receptor agonist taken once daily. Unlike Rybelsus (oral semaglutide), which is a peptide that requires special absorption enhancers, orforglipron is a small molecule that can be absorbed without those limitations. This potentially means more reliable absorption, fewer dietary restrictions around dosing, and easier manufacturing.
An FDA decision on orforglipron is expected in early to mid-2026, and if approved, it would significantly expand access to effective GLP-1 therapy by removing the injection requirement entirely.
Survodutide
Developed by Boehringer Ingelheim and Zealand Pharma, survodutide is a dual-agonist that targets GLP-1 and glucagon receptors rather than GLP-1 and GIP. This different combination produced mean weight loss of approximately 14.9% over 49 weeks in clinical trials. The glucagon component promotes fat burning and energy expenditure through a different pathway than GIP activation, and survodutide has shown particular promise for liver disease treatment.
What the pipeline means for patients
The rapid expansion of GLP-1 based therapies means that people who do not respond well to current options will likely have more choices in coming years. Different receptor combinations, oral versus injectable delivery, and varying side effect profiles will allow for more personalized treatment approaches. The overall trend is toward more effective medications with fewer side effects and more convenient dosing.
SeekPeptides tracks these developments closely, providing members with updated information on new GLP-1 research, emerging medications, and evolving treatment protocols as the field advances.
Dosing and administration of GLP-1 medications
Understanding how these medications are dosed helps illustrate why professional guidance matters and why the right medication choice depends on individual circumstances.
Semaglutide dosing (Ozempic and Wegovy)
The semaglutide dosing schedule follows a gradual escalation designed to minimize side effects. For Ozempic, the typical progression is 0.25 mg weekly for 4 weeks, then 0.5 mg for 4 weeks, then potentially 1 mg and 2 mg depending on clinical response. For Wegovy, the escalation continues up to 2.4 mg weekly over 16 to 20 weeks.
Injection technique matters. Semaglutide is administered subcutaneously in the abdomen, thigh, or upper arm. Rotation of injection sites helps prevent lipodystrophy at any single location. The prefilled pens used for brand-name products simplify the process compared to compounded versions that require reconstitution from vials.
For those using compounded semaglutide, understanding the semaglutide reconstitution process is essential. The concentration of the solution determines how many units of the syringe correspond to each milligram of medication, and getting this calculation wrong can result in significantly over or under-dosing. The semaglutide dosage calculator helps with these conversions.
Tirzepatide dosing (Mounjaro and Zepbound)
Tirzepatide follows a similar escalation pattern but with different dose levels. The starting dose is 2.5 mg weekly for 4 weeks, increasing to 5 mg, then 7.5 mg, 10 mg, 12.5 mg, and potentially 15 mg based on clinical response and tolerance. The tirzepatide dosing in units depends on the concentration of the solution being used.
For compounded tirzepatide, the reconstitution process requires careful attention to mixing ratios. The compounded tirzepatide dosage chart provides reference values, and proper storage is critical for maintaining potency. Tirzepatide is typically stored refrigerated, with specific shelf life guidelines that vary depending on whether the product is reconstituted or prefilled.
Practical injection guidance
Regardless of which GLP-1 medication someone uses, several practical considerations apply across the board. Timing of the weekly injection does not need to be precisely the same day each week, though consistency helps establish routine. The injection should be subcutaneous, meaning the needle goes into the fat layer just under the skin, not into the muscle.
Common injection sites include the abdomen (at least 2 inches from the navel), the front of the thigh, and the back of the upper arm. Rotating between sites and within each site area reduces the risk of local reactions. The peptide injection process is straightforward once learned, but proper technique ensures consistent absorption and minimal discomfort.
For people who prefer not to inject, oral options are becoming more available. Rybelsus (oral semaglutide) is currently the only FDA-approved oral GLP-1 medication, though orforglipron may soon join it. The sublingual delivery route is also being explored for various peptides, potentially expanding options for needle-averse patients.
When to consider GLP-1 therapy
GLP-1 medications are not appropriate for everyone, and understanding the criteria helps set appropriate expectations.
Type 2 diabetes
GLP-1 receptor agonists have become first-line or second-line therapy for many patients with type 2 diabetes, particularly those who also have obesity or established cardiovascular disease. The combination of blood sugar improvement, weight loss, and cardiovascular protection makes them uniquely suitable for patients with multiple metabolic risk factors.
The American Diabetes Association guidelines recommend considering GLP-1 receptor agonists early in the treatment algorithm, especially when weight management is a treatment goal alongside blood sugar control.
Obesity and weight management
Wegovy and Zepbound are indicated for adults with a BMI of 30 or greater, or 27 or greater with at least one weight-related condition such as high blood pressure, high cholesterol, type 2 diabetes, or obstructive sleep apnea. These medications are intended as adjuncts to lifestyle modifications, not replacements for them.
People often wonder why they are not losing weight on semaglutide or why tirzepatide results are not matching expectations. Several factors can influence response, including starting dose, duration of treatment, concurrent medications, dietary patterns, physical activity levels, and underlying medical conditions. The medications work best when combined with appropriate nutrition and movement strategies.
What GLP-1 therapy is not
These medications are not short-term solutions. Research consistently shows that discontinuing GLP-1 therapy leads to regain of two-thirds or more of the lost weight within a year. They are currently understood as chronic therapy for a chronic condition, similar to how blood pressure medications work. This means the decision to start involves committing to long-term use, with all the financial, logistical, and medical considerations that entails.
They are also not substitutes for fundamental lifestyle factors. Exercise, nutrition, sleep, and stress management remain important for overall health regardless of whether someone uses GLP-1 therapy. The medications reduce appetite and improve metabolic parameters, but they do not replace the need for adequate protein intake, resistance training to preserve muscle mass, or cardiovascular exercise for heart health.
Natural strategies to support GLP-1 function
Whether or not someone uses GLP-1 medications, supporting the body natural GLP-1 production through lifestyle and dietary strategies provides metabolic benefits. These approaches can complement medication therapy or serve as standalone strategies for people who prefer non-pharmaceutical options.
Dietary strategies
Protein prioritization. Protein is one of the strongest dietary triggers for GLP-1 release. Aiming for 25 to 35 grams of protein per meal supports both natural GLP-1 production and muscle preservation, which matters especially during weight loss. Good sources include poultry, fish, eggs, Greek yogurt, legumes, and whey protein.
Fiber-rich foods. Fermentable fibers feed gut bacteria that produce short-chain fatty acids, which stimulate L-cells to release more GLP-1. High fiber foods include artichokes, asparagus, lentils, beans, oats, and most vegetables. Adding fiber to each meal creates consistent GLP-1 stimulation throughout the day.
Healthy fats. Monounsaturated fats (olive oil, avocados, nuts) and omega-3 fatty acids (fatty fish, flaxseed) both trigger GLP-1 release. Including healthy fats at meals slows gastric emptying naturally and enhances the satiety response.
Meal sequencing. Eating protein and fiber before carbohydrates at each meal primes the GLP-1 response before glucose hits the bloodstream. This simple habit can meaningfully reduce post-meal blood sugar spikes and enhance feelings of fullness.
Exercise and movement
Both aerobic exercise and resistance training increase GLP-1 levels. The combination appears most effective, with some studies showing GLP-1 elevations persisting for 24 hours after a workout. Regular physical activity also improves insulin sensitivity independently of GLP-1 effects, creating complementary benefits for metabolic health.
For people on GLP-1 medications, resistance training is particularly important. These medications can cause loss of lean muscle mass along with fat, and progressive resistance exercise helps preserve muscle tissue during weight loss. Maintaining muscle mass supports metabolism, functional capacity, and long-term weight management success.
Sleep and stress management
Chronic stress elevates cortisol, which impairs GLP-1 secretion and promotes insulin resistance. Sleep deprivation produces similar metabolic disruption, reducing GLP-1 levels and increasing ghrelin (the hunger hormone). Prioritizing 7 to 9 hours of quality sleep and implementing stress reduction practices supports healthier hormonal patterns across the board.
These are not minor factors. Research suggests that inadequate sleep can reduce GLP-1 levels significantly enough to impair glucose tolerance and increase appetite the following day. For someone using GLP-1 medications, poor sleep can partially counteract the benefits of the drug itself.
The cost question
GLP-1 medications are expensive, and this financial reality shapes many people decisions about which product to use and whether to pursue treatment at all.
Brand-name semaglutide (Ozempic or Wegovy) carries a list price of approximately $900 to $1,400 per month without insurance. Brand-name tirzepatide (Mounjaro or Zepbound) falls in a similar range. Insurance coverage varies dramatically depending on the carrier, plan, and indication. Many plans cover these medications for diabetes but not for weight loss, creating a frustrating gap for patients whose primary goal is weight management.
Compounded versions offer a lower-cost alternative during shortage periods, though availability, quality, and regulatory status can vary. Understanding the differences between compounded and brand-name products, including storage requirements, reconstitution calculations, and potency verification, becomes important for anyone considering compounded options.
SeekPeptides provides tools including the peptide cost calculator to help people understand and compare the financial aspects of different peptide therapy options, along with resources for making informed decisions about which approach best fits individual needs and budgets.
Common misconceptions about GLP-1 and Ozempic
Clearing up the most persistent misunderstandings helps people make better decisions and have more productive conversations with their healthcare providers.
Misconception: GLP-1 is a drug
GLP-1 is a hormone, not a drug. Your body makes it every time you eat. When people say "I am taking GLP-1," what they usually mean is they are taking a GLP-1 receptor agonist, which is a drug that mimics the hormone. The distinction matters because understanding that GLP-1 is a natural part of your metabolism helps contextualize what these medications actually do. They are amplifying and extending a process your body already performs.
Misconception: Ozempic is the only GLP-1 drug
Ozempic is one brand of one drug (semaglutide) within a class of multiple drugs. Other GLP-1 receptor agonists include liraglutide, tirzepatide, dulaglutide, and exenatide, each sold under their own brand names. Someone who "tried GLP-1 and it did not work" may have only tried one specific medication at one specific dose, and a different product might produce entirely different results.
Misconception: all GLP-1 medications are the same
While they share a basic mechanism, differences in receptor selectivity, duration of action, delivery method, and dosing produce meaningfully different clinical outcomes. Tirzepatide versus semaglutide comparisons show that the dual-agonist approach produces greater weight loss than single-receptor targeting. Liraglutide requires daily injections while semaglutide requires weekly. Oral and injectable options absorb differently. These are not interchangeable products.
Misconception: GLP-1 drugs are just for weight loss
While weight loss has dominated public attention, these medications were originally developed for type 2 diabetes and carry significant benefits for cardiovascular health, kidney protection, and potentially liver disease. Reducing these medications to "weight loss drugs" undersells their therapeutic breadth and contributes to stigma around their use.
Misconception: you can just stop taking them when you reach your goal weight
Current evidence strongly suggests that GLP-1 medications require ongoing use to maintain their benefits. Stopping the medication typically results in return of appetite, regain of weight, and deterioration of metabolic improvements. This does not mean the medications "do not work." It means obesity is a chronic condition that requires chronic management, similar to hypertension requiring ongoing blood pressure medication.
Misconception: GLP-1 medications are dangerous
Like all medications, GLP-1 receptor agonists carry risks and side effects. But the clinical trial data across multiple large studies consistently shows a favorable benefit-to-risk ratio for indicated populations. The cardiovascular protective effects actually appear to reduce overall mortality risk in people with obesity and established heart disease. Serious adverse events like pancreatitis and gallbladder disease do occur but at low rates, and they are monitored through ongoing surveillance.
The safety profile of peptide therapies depends heavily on proper use, appropriate medical supervision, and understanding of individual risk factors. SeekPeptides members access detailed safety guides that cover these considerations comprehensively.
Making sense of GLP-1 terminology
The vocabulary around these medications trips people up constantly. Here is a quick reference guide to the terms that matter most.
GLP-1 (glucagon-like peptide-1): A natural hormone produced by L-cells in your small intestine after eating. It regulates blood sugar, slows digestion, and reduces appetite. Half-life of 1 to 2 minutes.
GLP-1 receptor agonist (GLP-1 RA): A class of medications that activate GLP-1 receptors, mimicking the natural hormone but lasting much longer. This is the drug class. Ozempic, Wegovy, Mounjaro, and others all belong to this class.
Semaglutide: A specific GLP-1 receptor agonist molecule (the generic drug name). Sold as Ozempic (diabetes), Wegovy (weight loss), and Rybelsus (oral diabetes).
Tirzepatide: A dual GLP-1/GIP receptor agonist molecule. Sold as Mounjaro (diabetes) and Zepbound (weight loss).
Incretin mimetics: Another name for GLP-1 receptor agonists, referencing the fact that GLP-1 is an incretin hormone (a gut hormone that stimulates insulin release).
DPP-4 inhibitors: A separate class of diabetes medications that work by blocking the enzyme (DPP-4) that breaks down natural GLP-1. Rather than adding synthetic GLP-1, these medications allow your own GLP-1 to last longer. They are less effective than GLP-1 receptor agonists for both blood sugar control and weight loss.
GIP (glucose-dependent insulinotropic polypeptide): Another incretin hormone that works alongside GLP-1. Tirzepatide targets both GIP and GLP-1 receptors, which is why it is sometimes called a dual-agonist or "twincretin."
Compounded: Medications prepared by compounding pharmacies, typically during periods of branded drug shortages. Compounded semaglutide and compounded tirzepatide contain the same active molecules but are manufactured differently than the brand-name products.
Frequently asked questions
Is GLP-1 a drug or a hormone?
GLP-1 is a hormone that your body produces naturally in the small intestine after eating. It is not a drug. However, the term "GLP-1" is commonly used as shorthand for GLP-1 receptor agonist medications like Ozempic, Wegovy, and Mounjaro, which are drugs designed to mimic the effects of the natural hormone.
Is Ozempic the best GLP-1 medication?
Ozempic is one effective GLP-1 medication, but "best" depends on individual goals, tolerance, and medical history. For weight loss specifically, tirzepatide (Mounjaro/Zepbound) has shown greater average weight loss than semaglutide in clinical trials. For diabetes management, multiple options may be equally appropriate. The right choice depends on your specific situation and should be discussed with a healthcare provider.
Can I boost my GLP-1 naturally without medication?
Yes. Dietary strategies including high protein intake, fiber-rich foods, healthy fats, and meal sequencing (eating protein before carbohydrates) can all increase natural GLP-1 production. Exercise, adequate sleep, and stress management also support GLP-1 levels. These strategies produce more modest effects than medications but carry no side effects and provide broad metabolic benefits.
What is the difference between Ozempic and Wegovy?
Both contain semaglutide. Ozempic is FDA-approved for type 2 diabetes at doses up to 2 mg weekly. Wegovy is FDA-approved for weight management at a dose of 2.4 mg weekly. The active ingredient is identical, but the dose, indication, and insurance coverage differ. The dosage chart for semaglutide shows the differences in dosing schedules between the two products.
Do GLP-1 medications work for everyone?
Response varies significantly between individuals. Most people experience meaningful weight loss and blood sugar improvement, but the degree of response differs based on genetics, baseline metabolic status, dose, diet, exercise, sleep, and other factors. Some people who do not respond to one GLP-1 medication may respond to another with a different mechanism, such as switching from semaglutide to tirzepatide or vice versa.
Are there oral GLP-1 medications available?
Currently, Rybelsus (oral semaglutide) is the only FDA-approved oral GLP-1 medication. It is taken as a daily tablet on an empty stomach with a small amount of water. Orforglipron, a non-peptide oral GLP-1 receptor agonist from Eli Lilly, is expected to receive an FDA decision in early to mid 2026 and could significantly expand oral GLP-1 options.
How long do GLP-1 medications take to work?
Appetite suppression typically becomes noticeable within the first 1 to 2 weeks. Measurable weight loss usually appears within the first month, with maximum effects developing over 6 to 12 months of treatment. Blood sugar improvements in diabetic patients often occur within the first few weeks. The full timeline depends on the specific medication, dose escalation schedule, and individual response. See our guides on how fast semaglutide works and how fast tirzepatide works for detailed timelines.
Will I regain weight if I stop taking a GLP-1 medication?
Current evidence suggests that most people regain a significant portion of lost weight within 12 months of stopping GLP-1 therapy. This is consistent with the understanding that obesity is a chronic condition requiring ongoing management. Duration of treatment decisions should factor in this reality, and lifestyle strategies including exercise and dietary changes can help mitigate rebound weight gain.
External resources
Cleveland Clinic - GLP-1 Agonists: What They Are, How They Work, and Side Effects
National Library of Medicine - Glucagon-Like Peptide-1 Receptor Agonists
Harvard Health - How Does Ozempic Work? Understanding GLP-1s
For researchers serious about understanding the full GLP-1 landscape, from natural hormone function to medication protocols to emerging therapies, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, dosing calculators, and a community of thousands who navigate these exact questions daily.
In case I do not see you, good afternoon, good evening, and good night. May your hormones stay balanced, your appetite stay regulated, and your health decisions stay informed.