Feb 6, 2026

Your doctor hands you a tirzepatide prescription. You see numbers. Weekly doses. Escalation schedules. Different concentrations. And one critical question sits unanswered in the back of your mind: what dose actually works, and how do you get there safely?
The stakes are higher than most realize. Start too aggressively and gastrointestinal side effects can become debilitating. Progress too cautiously and you waste months on suboptimal doses that deliver minimal results. Jump between doses erratically and your body never adapts properly. The difference between effective weight loss and a miserable experience often comes down to following the right dosing progression at the right pace.
This complexity multiplies when you consider that tirzepatide dose charts vary significantly between pharmaceutical brands like Mounjaro and Zepbound, compounded formulations, and off label protocols. Each requires different approaches to dose escalation, timing adjustments, and maintenance strategies. Understanding which chart applies to your specific situation, and how to navigate the progression from start to maintenance, determines whether your protocol succeeds or fails.
This guide provides complete tirzepatide dose charts for every protocol type, walks through proper escalation timing, explains how to identify your optimal maintenance dose, covers what to do when side effects emerge, details how to adjust based on individual response, and outlines the critical safety considerations that most simplified charts completely ignore. Whether you are starting your first dose or troubleshooting an existing protocol, these evidence based charts give you the framework for effective tirzepatide use.
Understanding tirzepatide dose escalation principles
Tirzepatide dose escalation is not arbitrary. The standard progression, starting at 2.5 mg and increasing by 2.5 mg increments every 4 weeks, emerged from extensive clinical trials designed specifically to balance efficacy against tolerability. This careful titration schedule allows your body to adapt to the medication while minimizing the gastrointestinal side effects that plague aggressive dosing approaches.
The biological reasoning centers on receptor adaptation.
Tirzepatide activates both GIP and GLP-1 receptors, triggering changes in gastric emptying, insulin secretion, and appetite regulation. These physiological shifts take time. Your digestive system needs weeks to adjust to delayed gastric emptying. Your pancreatic beta cells require time to optimize their insulin response patterns. Your hypothalamic appetite centers must recalibrate their signaling thresholds.
Rush this adaptation process and your body rebels with severe nausea, vomiting, diarrhea, and constipation. The clinical trial data proves this conclusively. In the SURMOUNT-1 study, which established the standard escalation protocol, gastrointestinal side effects occurred primarily during dose escalation periods, not during maintenance phases. Most adverse events were mild to moderate precisely because the 4 week intervals allowed physiological adaptation.
Why 4 week intervals matter
The 4 week escalation interval was not chosen randomly. Earlier phase 2 trials tested different timing protocols, including 2 week escalations and faster progressions to higher doses. The results were clear. Participants who escalated too quickly experienced significantly higher rates of treatment discontinuation due to intolerable side effects.
Four weeks provides sufficient time for several critical adaptations. Your stomach adjusts to slower emptying rates. Your intestinal transit normalizes despite altered motility. Your appetite regulation centers recalibrate their baseline thresholds. Your pancreatic function optimizes insulin secretion patterns in response to the dual receptor activation.
Clinical data from multiple trials shows that extending beyond 4 weeks offers no additional tolerability benefit while unnecessarily delaying therapeutic effect. Shortening below 4 weeks consistently increases adverse event rates without improving outcomes. The 4 week interval represents the optimal balance between rapid therapeutic escalation and physiological adaptation time.
Understanding this principle helps explain why jumping doses or rushing escalation consistently produces poor outcomes, even when the same final maintenance dose would be well tolerated with proper progression.
Dose response relationships
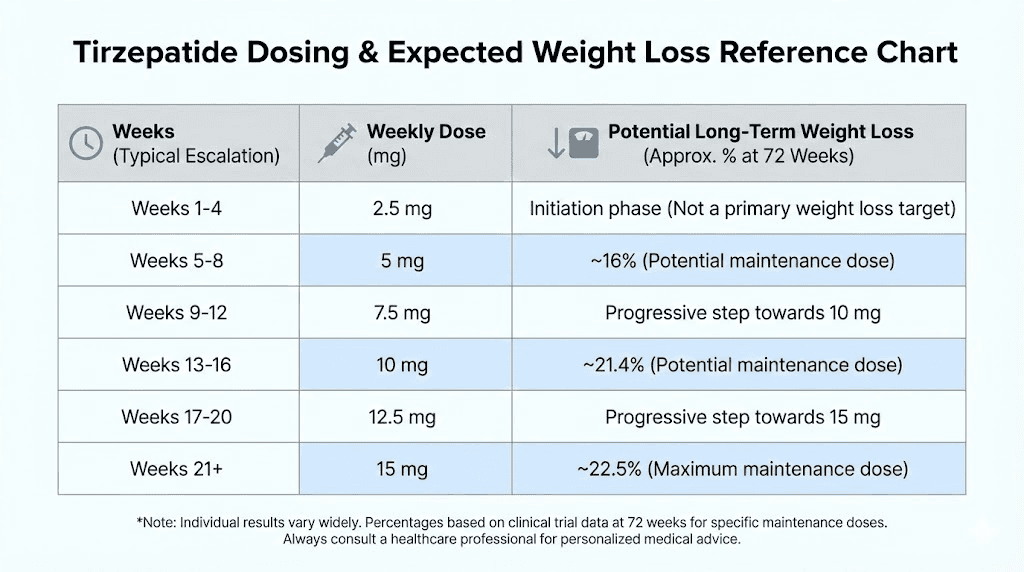
Tirzepatide demonstrates clear dose dependent efficacy for weight loss. In SURMOUNT-1, the largest tirzepatide obesity trial, participants taking 5 mg lost approximately 15 percent of baseline body weight over 72 weeks. Those on 10 mg achieved roughly 20 percent reduction. The 15 mg group reached approximately 21 percent weight loss.
This dose response curve is not linear. The jump from 5 mg to 10 mg produces substantial additional efficacy. The increase from 10 mg to 15 mg yields more modest incremental benefit. This pattern suggests that for many individuals, 10 mg may represent the optimal balance between efficacy and side effect burden, though individual responses vary considerably.
The dose response extends beyond weight loss to metabolic parameters. Higher doses produce greater improvements in glycemic control, larger reductions in hemoglobin A1c, and more substantial decreases in fasting glucose levels. Lipid parameters, including triglycerides and LDL cholesterol, show similar dose dependent improvements.
However, side effects also follow dose dependent patterns. Nausea, vomiting, and diarrhea increase in frequency and severity with higher doses. This creates an individualized optimization challenge: finding the lowest dose that produces satisfactory results while minimizing adverse effects.
For some individuals, 5 mg provides sufficient weight loss with minimal side effects, making higher doses unnecessary. Others tolerate 15 mg well and benefit from the additional efficacy. The dose chart provides the framework, but individual optimization requires attention to both efficacy markers and tolerability signals.
Individual variation factors
Significant individual variation affects optimal tirzepatide dosing beyond what standardized charts can capture. Body weight, though not directly factored into dosing algorithms, influences pharmacokinetics. Larger individuals may achieve therapeutic concentrations at different rates than smaller individuals on identical doses.
Metabolic status at baseline matters substantially. Individuals with severe insulin resistance may require higher doses to achieve comparable metabolic improvements compared to those with milder metabolic dysfunction. Pre-existing gastrointestinal conditions, particularly gastroparesis or irritable bowel syndrome, significantly affect tolerability and may necessitate slower escalation or lower maintenance doses.
Genetic factors influence GLP-1 receptor responsiveness, though routine genetic testing for these variants is not standard practice. Some individuals are naturally more or less sensitive to GLP-1 agonist effects, which translates to varying dose requirements for equivalent responses.
Concurrent medications can affect tirzepatide pharmacodynamics. Medications that slow gastric emptying, including certain antidepressants and opioids, may amplify gastrointestinal side effects. Drugs affecting insulin secretion or glucose metabolism may interact with tirzepatide metabolic effects.
Diet composition and meal timing patterns influence how individuals experience tirzepatide effects. Those consuming high fat, high volume meals while on tirzepatide typically experience more severe gastrointestinal symptoms than those adjusting to smaller, more frequent, lower fat meals.
These individual factors mean that while dose charts provide essential frameworks, successful tirzepatide use requires individualized adjustments based on response monitoring and tolerability assessment.
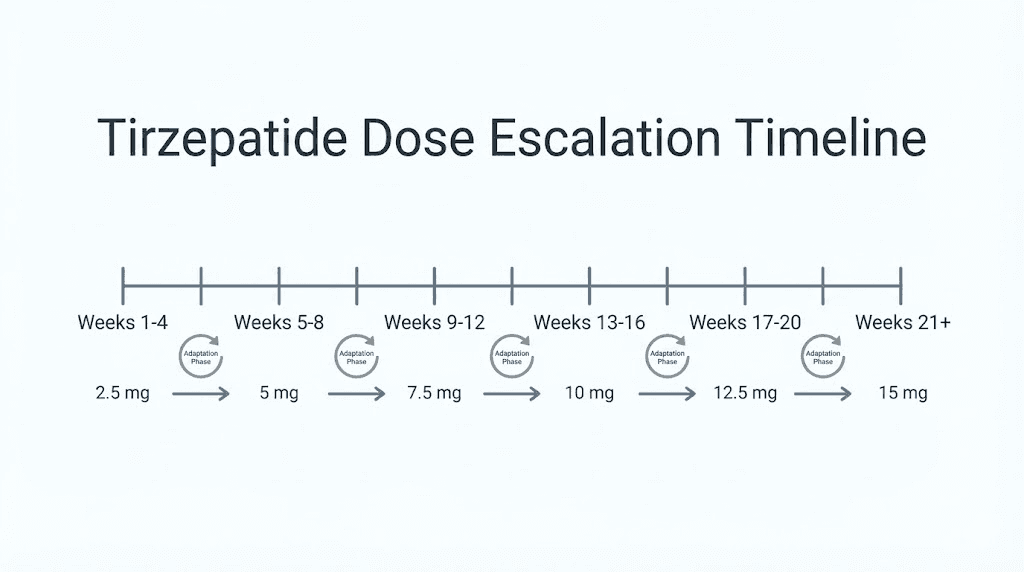
Standard pharmaceutical tirzepatide dose chart
The FDA approved tirzepatide dosing schedule, used for both Mounjaro for type 2 diabetes and Zepbound for chronic weight management, follows an identical escalation pattern. This standardization emerged from the pivotal clinical trials that established both efficacy and safety profiles.
Complete dose progression table
Weeks 1 through 4: 2.5 mg once weekly. This starter dose initiates receptor activation and begins the physiological adaptation process. Most individuals experience minimal side effects at this dose, though some notice mild nausea or decreased appetite. Weight loss during this phase is typically modest, averaging 2 to 4 percent of baseline body weight.
Weeks 5 through 8: 5.0 mg once weekly. The first dose increase doubles your exposure and typically produces the first substantial weight loss acceleration. Nausea and gastrointestinal symptoms peak during the first 1 to 2 weeks after this increase, then gradually improve as adaptation occurs. Average weight loss reaches 5 to 7 percent of baseline by week 8.
Weeks 9 through 12: 7.5 mg once weekly. This intermediate dose provides a stepping stone for individuals progressing toward higher maintenance doses. Some protocols skip this dose and move directly from 5 mg to 10 mg, particularly for individuals tolerating the 5 mg dose well with minimal side effects. Weight loss continues accelerating, typically reaching 8 to 12 percent of baseline by week 12.
Weeks 13 through 16: 10 mg once weekly. For many individuals, this becomes the maintenance dose, balancing substantial efficacy against manageable side effects. Clinical trial data shows this dose produces approximately 20 percent average weight loss over 72 weeks. Gastrointestinal side effects at this dose are generally well tolerated by individuals who progressed gradually through lower doses.
Weeks 17 through 20: 12.5 mg once weekly. This optional intermediate step exists primarily for individuals who tolerate 10 mg well but have not achieved desired weight loss goals. Not all protocols include this dose level, and many physicians progress directly from 10 mg to 15 mg if further escalation is warranted.
Week 21 and beyond: 15 mg once weekly. The maximum approved dose provides the highest efficacy, producing approximately 21 percent average weight loss in clinical trials. Individual responses range from 15 to 30 percent weight loss, with higher responders typically being those who implement comprehensive lifestyle modifications alongside medication use.
This progression takes 20 to 24 weeks to reach maximum dose, though many individuals achieve satisfactory results at lower maintenance doses and never progress to 15 mg.
Dose holding and skipping guidelines
If you miss a dose and fewer than 4 days have passed since your scheduled injection day, administer the missed dose as soon as possible. Then resume your regular weekly schedule. If more than 4 days have passed, skip the missed dose entirely and administer your next dose on the regularly scheduled day. Never double dose to make up for a missed injection.
Temporary dose holds become necessary when side effects become intolerable despite conservative management strategies. Severe persistent nausea, repeated vomiting, or signs of dehydration warrant holding your next dose and consulting your prescriber. Do not resume at the same dose level that caused severe symptoms. Drop back to the previous dose level and extend the duration at that dose before attempting escalation again.
If you need to hold doses for more than 2 weeks due to illness, surgery, or other medical reasons, discuss with your prescriber whether dose reduction is appropriate when resuming. Extended breaks can reset some of the physiological adaptations you achieved during escalation.
Some individuals find alternating between two dose levels helpful when the lower dose feels too weak but the higher dose produces excessive side effects. For example, alternating between 7.5 mg and 10 mg weekly can provide an effective average dose of approximately 8.75 mg. Discuss this approach with your prescriber before implementing, as it represents off label modification of standard protocols.
Maintenance phase considerations
Once you reach your maintenance dose, whether that is 5 mg, 10 mg, or 15 mg, consistency becomes paramount. Weekly administration on the same day helps maintain stable therapeutic levels and reinforces the habit of regular dosing.
Maintenance efficacy continues beyond initial weight loss. The SURMOUNT-4 trial demonstrated that participants who achieved significant weight loss on tirzepatide and then continued treatment maintained their weight loss, while those switched to placebo regained substantial weight. This underscores that tirzepatide is a long term maintenance therapy, not just a short term weight loss tool.
Some individuals experience weight loss plateaus during maintenance. These plateaus are normal and do not necessarily indicate that your dose is inadequate. Plateaus typically last 2 to 4 weeks before weight loss resumes, assuming caloric intake remains controlled and physical activity continues. If plateaus extend beyond 4 to 6 weeks, evaluate diet and activity patterns before considering dose increases.
Maintenance phases also provide opportunities to optimize injection timing and technique. Some individuals find morning injections preferable to minimize daytime nausea. Others prefer evening injections to sleep through peak side effect periods. Rotating injection sites between abdomen, thigh, and upper arm prevents lipohypertrophy and maintains consistent absorption.
Long term maintenance on tirzepatide requires monitoring metabolic parameters every 3 to 6 months, including fasting glucose, hemoglobin A1c if diabetic, lipid panel, liver enzymes, and kidney function tests. These assessments ensure that beneficial metabolic effects persist and no adverse metabolic changes emerge.
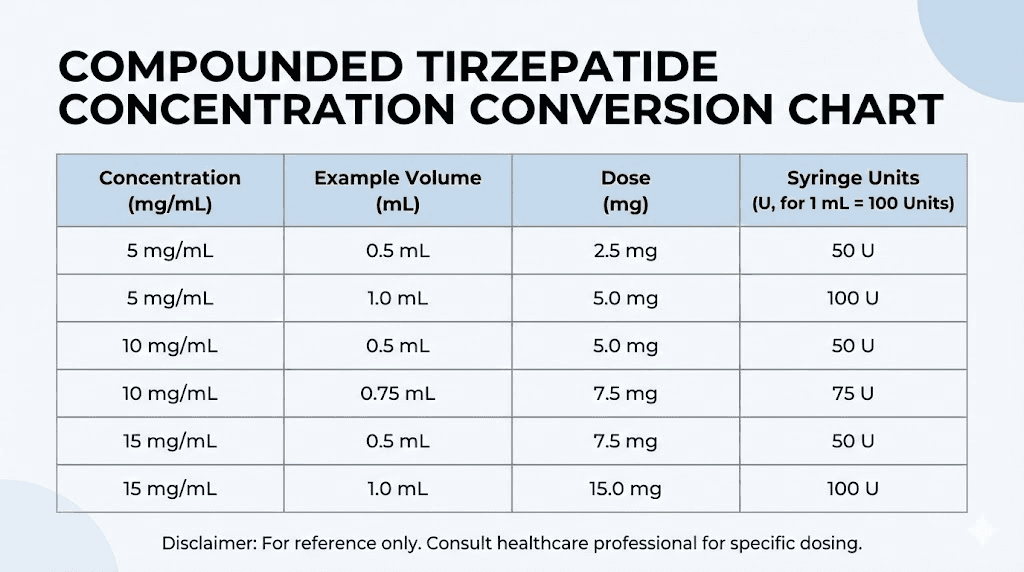
Compounded tirzepatide dosing adjustments
Compounded tirzepatide introduces significant complexity to dosing charts because concentrations vary between compounding pharmacies and even between batches from the same pharmacy. Unlike pharmaceutical products with standardized concentrations, compounded formulations may range from 2.5 mg/mL to 10 mg/mL or higher.
Concentration conversion calculations
Understanding your specific vial concentration is absolutely critical before drawing any dose. The prescription label should state both the total vial content and the concentration. For example, a label might read 5 mg/mL concentration in a 2 mL vial, meaning the vial contains 10 mg total tirzepatide.
To calculate the volume you need to draw for any given dose, use this formula: desired dose in mg divided by concentration in mg per mL equals volume in mL. If you want a 5 mg dose from a 5 mg/mL vial, you would calculate 5 mg divided by 5 mg/mL equals 1.0 mL. If you want that same 5 mg dose from a 10 mg/mL vial, you would calculate 5 mg divided by 10 mg/mL equals 0.5 mL.
These calculations become more complex when converting to insulin syringe units. Most insulin syringes measure in units, where 100 units equals 1.0 mL. To convert mL to units, multiply by 100. Using our previous example, 0.5 mL equals 50 units on a standard insulin syringe.
Our tirzepatide dosage calculator handles these conversions automatically, accounting for vial concentration, desired dose, and syringe type. This eliminates calculation errors that frequently occur with manual math, particularly when dealing with decimal values or unit conversions.
Always verify your calculation before drawing. A simple check: if your concentration is 10 mg/mL and you need 5 mg, your volume should be less than 1 mL. If your concentration is 2.5 mg/mL and you need 5 mg, your volume should be more than 1 mL. This directional check catches most calculation errors before they result in incorrect dosing.
Vial longevity and dosing schedules
Compounded vial longevity depends on both total vial content and your dose. A 10 mg vial provides four doses at 2.5 mg, two doses at 5 mg, or one dose at 10 mg. Planning your refills requires understanding these relationships to avoid running out mid cycle.
Most compounded tirzepatide vials contain between 10 mg and 30 mg total peptide. Calculate your vial longevity by dividing total vial content by your dose. A 20 mg vial lasts 8 weeks at 2.5 mg weekly, 4 weeks at 5 mg weekly, or 2 weeks at 10 mg weekly.
This math creates practical challenges during dose escalation. If you start with a 10 mg vial for your 2.5 mg starter dose, that single vial lasts 4 weeks. When you escalate to 5 mg at week 5, you need a fresh vial that will last only 2 weeks. Planning ahead prevents gaps in treatment that can reset your adaptation progress.
Some compounding pharmacies offer multi vial kits designed to support complete escalation schedules. These kits typically include appropriately sized vials for each dose tier, eliminating the waste that occurs when partial vials remain after dose escalation. Ask your compounding pharmacy if such kits are available.
Reconstitution requirements add another variable. Some compounded tirzepatide arrives lyophilized and requires reconstitution with bacteriostatic water. The volume of bacteriostatic water you add determines your final concentration, which then affects your dosing volume calculations. Always follow your pharmacy specific reconstitution instructions precisely, as these vary between formulations.
Quality and consistency considerations
Compounded tirzepatide lacks the rigorous manufacturing controls and batch to batch consistency guarantees of pharmaceutical products.
This introduces potential variability in actual peptide content compared to stated concentrations.
Reputable compounding pharmacies conduct certificate of analysis testing on their formulations, verifying that actual peptide content matches labeled concentrations. Request these certificates from your pharmacy. They should show testing results within 90 to 110 percent of stated concentration. Results outside this range suggest quality control problems.
Potency degradation occurs more rapidly with compounded formulations than pharmaceutical products. While Mounjaro and Zepbound remain stable for extended periods when properly stored, compounded tirzepatide typically maintains full potency for only 28 to 60 days after reconstitution or compounding, depending on formulation and storage conditions.
Monitor your response patterns when switching between compounding pharmacies or starting new vials. If efficacy suddenly decreases or side effects change substantially despite using the same dose, suspect potency issues. Subpotent formulations waste both money and time, delaying progress toward your goals.
The cost savings from compounded tirzepatide, often 60 to 80 percent lower than pharmaceutical prices, come with these consistency and quality trade offs. For individuals who cannot afford pharmaceutical products, compounded options provide access. For those who can afford pharmaceutical products, the guaranteed consistency and quality may justify the additional cost.
Understanding these compounded specific factors helps you navigate dosing challenges that standard pharmaceutical dose charts do not address.
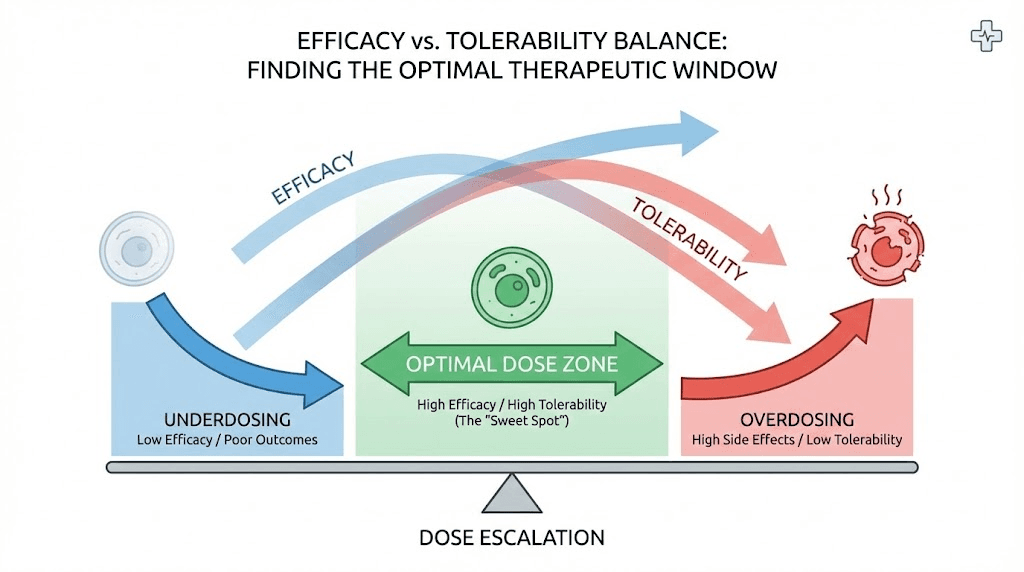
Identifying your optimal maintenance dose
Finding your optimal maintenance dose involves balancing efficacy, tolerability, and sustainability. The maximum dose is not automatically the best dose. Many individuals achieve excellent results on moderate doses while avoiding the increased side effects that higher doses produce.
Efficacy markers to track
Weight loss rate provides the most obvious efficacy signal. A sustainable rate of 0.5 to 1 percent of body weight per week indicates effective dosing. Slower rates suggest potential underdosing, assuming dietary compliance remains strong. Faster rates, while sometimes achievable short term, rarely sustain long term and may indicate excessive caloric restriction that will trigger metabolic adaptation.
Beyond scale weight, body composition changes matter substantially. Measuring body fat percentage, lean mass, and waist circumference every 4 weeks provides more complete efficacy assessment than weight alone. Effective tirzepatide use preserves lean mass while reducing fat mass. If you are losing substantial lean mass alongside fat loss, evaluate whether your protein intake and resistance training are adequate.
Metabolic markers offer objective efficacy measures independent of scale weight. Fasting glucose should normalize or approach normal ranges within 8 to 12 weeks. Hemoglobin A1c improvements of 1 to 2 percent occur in individuals with elevated baseline values. Lipid improvements, including triglyceride reductions of 20 to 30 percent and modest LDL decreases, typically manifest by 16 to 20 weeks.
Subjective markers also provide valuable efficacy signals. Appetite suppression should feel substantial but not uncomfortable. You should feel satisfied with smaller meals without experiencing hunger that interferes with daily function. Energy levels should remain stable or improve as weight loss progresses. Persistent fatigue despite adequate sleep and nutrition suggests potential underdosing or inadequate caloric intake.
Cardiovascular improvements, including blood pressure reductions and resting heart rate decreases, emerge gradually over months. These changes correlate with improved metabolic health independent of weight loss magnitude.
Side effect severity assessment
Mild nausea that resolves within 2 to 3 days after each injection represents expected, tolerable side effects. Severe nausea persisting for 4 to 5 days post injection suggests your dose exceeds your current tolerance level. Vomiting more than once per week indicates clear overdosing for your individual physiology.
Gastrointestinal side effects should decrease over time at any given dose as adaptation occurs. If nausea, diarrhea, or constipation worsen rather than improve over 4 to 6 weeks at a stable dose, suspect issues beyond normal adaptation, including dietary factors, concurrent illness, or medication interactions.
The relationship between meal timing and nausea provides important dosing insights. If nausea emerges only with large or high fat meals, your dose may be appropriate but your dietary choices need adjustment. If nausea occurs even with small, bland meals, your dose likely exceeds your tolerance.
Fatigue patterns help distinguish appropriate dosing from excessive dosing. Mild fatigue on injection day that resolves within 24 hours represents normal response. Persistent fatigue throughout the week suggests inadequate caloric or nutrient intake relative to your dose induced metabolic changes.
Injection site reactions, including redness, swelling, or itching, are usually unrelated to dose level and instead reflect injection technique issues or localized sensitivity. However, systemic allergic reactions necessitate immediate discontinuation regardless of efficacy.
Finding your dose sweet spot
The optimal dose produces substantial, sustainable weight loss with manageable side effects that do not significantly impair quality of life. For many individuals, this occurs at 7.5 to 10 mg weekly. Some require 12.5 or 15 mg to achieve adequate appetite suppression and weight loss. A smaller subset responds excellently to 5 mg and sees no benefit from higher doses.
Individual optimization often requires systematic experimentation within the safe dosing range. If 10 mg produces excellent efficacy but borderline intolerable nausea, try 7.5 mg for 6 to 8 weeks to assess whether efficacy remains adequate with improved tolerability. If 10 mg is well tolerated but weight loss has plateaued after 6 months, trial 12.5 mg for 8 to 12 weeks to determine whether additional dose produces additional benefit.
Maintenance dose is not necessarily permanent. Some individuals escalate dose when weight loss plateaus, then reduce dose once new lower weight stabilizes. Others find their tolerance improves over months, allowing dose increases that were previously intolerable. Conversely, some individuals who initially tolerated high doses find that after substantial weight loss, lower doses provide adequate maintenance with fewer side effects.
Cost considerations factor into optimization for most individuals. If 7.5 mg produces 90 percent of the benefit of 10 mg at 75 percent of the cost, the lower dose may represent better value. Pharmaceutical tirzepatide costs approximately $1000 per month regardless of dose since pens contain fixed amounts. Compounded tirzepatide costs scale directly with dose, making lower maintenance doses significantly more affordable.
Working with experienced prescribers who understand individual optimization rather than simply following maximum dose protocols helps you find your personal sweet spot faster and with fewer trial and error cycles.
SeekPeptides members access detailed dose optimization protocols that account for individual response patterns, side effect profiles, and long term sustainability considerations most standardized approaches miss.
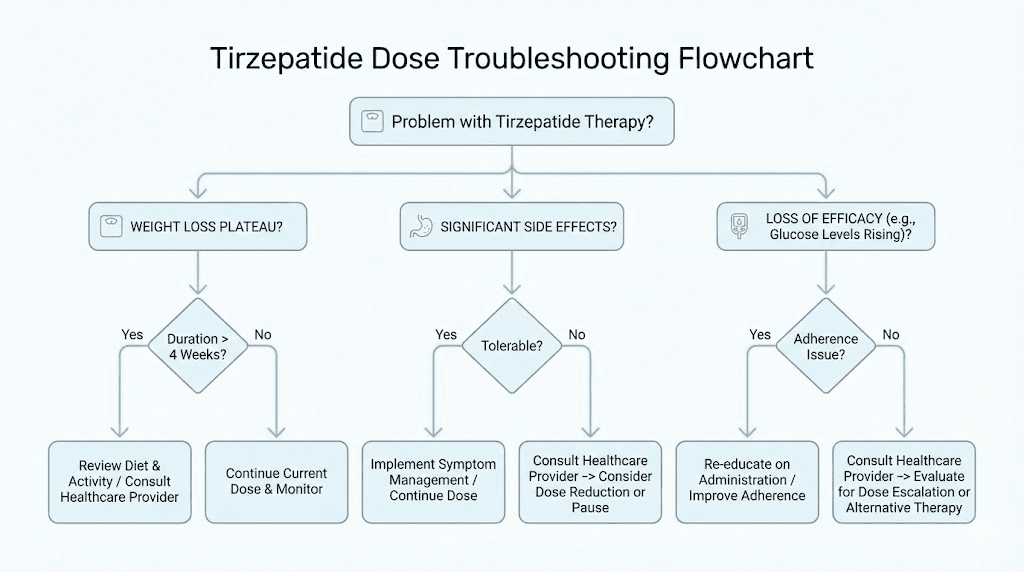
Troubleshooting dose related challenges
Even with perfect adherence to standard dose charts, challenges emerge that require systematic troubleshooting. Understanding common patterns and their solutions prevents unnecessary protocol abandonment.
Plateaus and stalls
Weight loss plateaus occur in virtually everyone using tirzepatide long term. These plateaus typically last 2 to 6 weeks before weight loss resumes, assuming nothing else has changed. The physiological explanation involves metabolic adaptation, where your body adjusts energy expenditure to match reduced caloric intake, temporarily halting weight loss despite continued caloric deficit.
True plateaus occur at stable weight. Fluctuations of 2 to 3 pounds up and down throughout a week while trending nowhere represent normal variation, not true plateaus. Track your weight daily but evaluate weekly averages to distinguish true plateaus from normal fluctuation.
Before considering dose increases in response to plateaus, systematically evaluate non dose factors. Dietary compliance often degrades gradually as initial enthusiasm wanes. Food tracking for one week during a plateau frequently reveals caloric creep where portion sizes expanded or frequency of indulgences increased without conscious awareness.
Physical activity reductions commonly coincide with plateaus. Weight loss produces fatigue that unconsciously reduces non exercise activity thermogenesis, the calories burned through daily movement outside structured exercise. Tracking daily steps often reveals substantial activity decreases during plateau periods.
Stress and sleep disruptions affect weight loss independent of diet and activity. Elevated cortisol from chronic stress promotes fat retention, particularly visceral fat. Sleep deprivation disrupts hunger hormones, increasing ghrelin and decreasing leptin, which undermine tirzepatide appetite suppression effects. Addressing these lifestyle factors often breaks plateaus without dose adjustments.
If plateaus persist beyond 6 weeks despite optimized diet, activity, stress, and sleep, consider whether your current dose remains adequate. Some individuals experience reduced efficacy over time, requiring dose increases to maintain suppression of appetite and metabolic effects. However, this tachyphylaxis is relatively uncommon with tirzepatide compared to older weight loss medications.
Tolerance loss and tachyphylaxis
True tachyphylaxis, where a previously effective dose loses efficacy despite unchanged circumstances, is uncommon but possible with tirzepatide. Distinguishing tachyphylaxis from other explanations requires systematic evaluation.
Document whether appetite suppression diminished, whether the same dose produces fewer gastrointestinal effects than previously, and whether metabolic parameters began deteriorating. True tachyphylaxis affects all these domains simultaneously. If only weight loss stalled while appetite suppression remains strong, the issue is likely plateau, not tolerance loss.
Some individuals experience apparent tolerance loss that actually reflects changes in compounded peptide potency.
If you switched vials, changed compounding pharmacies, or have been using the same reconstituted vial for over 30 days, suspect potency degradation rather than true tolerance.
Testing with fresh pharmaceutical grade product can distinguish between these possibilities.
Medication interactions can produce apparent tolerance loss. Starting new medications that affect glucose metabolism, appetite, or gastrointestinal function may counteract tirzepatide effects. Review any medication changes that coincided with apparent efficacy loss.
If genuine tachyphylaxis is confirmed, strategies include dose escalation to the next tier, brief treatment breaks of 2 to 4 weeks to allow receptor resensitization, or switching to alternative GLP-1 agonists like semaglutide to provide different receptor activation patterns.
Managing severe adverse reactions
Severe nausea and vomiting that prevents adequate fluid intake constitutes a medical emergency requiring immediate attention. Dehydration from GLP-1 agonist induced vomiting can progress to acute kidney injury, particularly in individuals with pre existing kidney disease or those taking medications affecting kidney function.
Warning signs of dangerous dehydration include dark urine, dizziness when standing, inability to keep down fluids for more than 12 hours, decreased urination, extreme fatigue, confusion, or rapid heartbeat. These symptoms require emergency medical evaluation, not home management.
Less severe but still problematic gastrointestinal reactions warrant dose holds and systematic side effect management. Hold your next dose if you experience vomiting more than twice in one week, if nausea prevents you from eating adequate nutrition for more than 2 days, or if diarrhea becomes severe enough to cause dehydration concerns.
When resuming after a dose hold, drop back to the previous dose level and maintain that level for an additional 4 weeks before attempting escalation again. Some individuals require 6 to 8 weeks at intermediate doses rather than the standard 4 weeks to achieve adequate tolerance.
Pancreatitis represents a rare but serious adverse reaction. Symptoms include severe abdominal pain that radiates to the back, nausea and vomiting, fever, and rapid pulse. Suspected pancreatitis requires immediate medical evaluation and permanent tirzepatide discontinuation. Do not resume tirzepatide after pancreatitis without explicit approval from a gastroenterologist familiar with GLP-1 agonist associated pancreatitis.
Gallbladder problems, including cholecystitis and cholelithiasis, occur with increased frequency during rapid weight loss regardless of method. Sharp right upper abdominal pain, particularly after fatty meals, suggests gallbladder issues. These require medical evaluation and may necessitate temporary dose reduction or discontinuation pending resolution.
Hypoglycemia, though uncommon with tirzepatide monotherapy, can occur in individuals taking concurrent diabetes medications, particularly insulin or sulfonylureas. Symptoms include shakiness, sweating, confusion, rapid heartbeat, and extreme hunger. Mild hypoglycemia resolves with 15 grams of fast acting carbohydrate. Severe hypoglycemia requires emergency glucagon administration and medical attention. Concurrent diabetes medications often require dose reductions when starting or escalating tirzepatide.
Allergic reactions, including rash, itching, swelling, or difficulty breathing, necessitate immediate discontinuation and emergency medical attention. True allergy to tirzepatide is rare but represents an absolute contraindication to continued use.
Pregnancy and breastfeeding considerations
Tirzepatide is contraindicated during pregnancy and should be discontinued at least 2 months before planned conception due to the long washout period. The medication crosses the placenta and animal studies showed fetal harm, though human data remains limited.
If pregnancy occurs while taking tirzepatide, discontinue immediately and inform your obstetrician. The critical period for potential fetal effects is first trimester organogenesis, making early discontinuation important.
Breastfeeding safety remains unknown as tirzepatide presence in human milk has not been studied. Given the molecular weight and structure, some excretion into breast milk likely occurs. Most experts recommend against tirzepatide use during breastfeeding until safety data becomes available.
Women of childbearing potential should use reliable contraception while taking tirzepatide. The weight loss itself improves fertility in individuals with obesity related infertility, potentially leading to unplanned pregnancy. The appetite suppression may reduce absorption of oral contraceptives taken around the time of tirzepatide injection, though this interaction has not been formally studied.
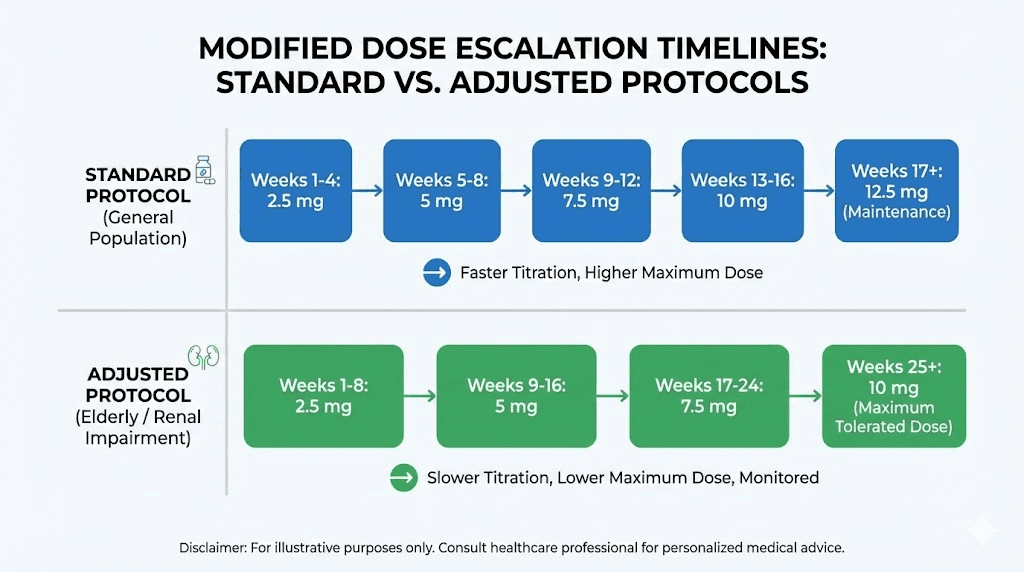
Special population dosing modifications
Standard dose charts assume otherwise healthy adults with obesity. Specific populations require modified approaches that balance efficacy against increased risk profiles.
Elderly patients
Individuals over 65 often require more conservative escalation schedules. Age related decreases in renal function affect tirzepatide clearance, potentially increasing exposure at standard doses. Reduced gastric acid production and altered gastrointestinal motility make elderly patients more susceptible to gastrointestinal side effects.
Starting elderly patients at 2.5 mg remains appropriate, but extending each dose tier to 6 to 8 weeks rather than 4 weeks improves tolerability. Maximum doses of 10 mg often provide adequate efficacy with better tolerability than 15 mg in elderly populations.
Malnutrition risk increases in elderly patients taking tirzepatide due to the combination of age related appetite decline and medication induced appetite suppression. Monitoring protein intake, muscle mass, and functional status becomes critical. Weight loss in elderly individuals should preserve muscle mass and function. Loss of lean mass or functional decline suggests that nutritional support is inadequate relative to weight loss rate.
Polypharmacy concerns are more prominent in elderly patients. Many take multiple medications that may interact with tirzepatide effects. Medications affecting gastric emptying, including anticholinergics, opioids, and certain antidepressants, amplify gastrointestinal side effects. Diabetes medications require closer monitoring and more aggressive dose reductions when starting tirzepatide in elderly patients.
Renal impairment
Mild to moderate renal impairment does not require dose adjustments, as tirzepatide is primarily metabolized through peptidase degradation rather than renal elimination. However, severe renal impairment and end stage renal disease lack sufficient safety data.
Individuals with estimated glomerular filtration rate below 30 mL per min should use tirzepatide with caution, starting at 2.5 mg and escalating more slowly while monitoring renal function closely. The dehydration risk from gastrointestinal side effects poses particular concern in patients with compromised renal function.
Volume status monitoring becomes critical. Even mild dehydration can precipitate acute kidney injury in patients with chronic kidney disease. Emphasize adequate hydration, monitor for signs of volume depletion, and check renal function at baseline, 4 weeks, 12 weeks, and any time side effects worsen.
Patients on dialysis represent particularly complex cases. Tirzepatide is not removed by dialysis, so standard pharmacokinetics apply. However, these patients often have severe comorbidities, altered nutritional status, and multiple medication interactions that complicate tirzepatide use. Specialist consultation with nephrology and endocrinology is appropriate for dialysis patients considering tirzepatide.
Hepatic impairment
Mild to moderate hepatic impairment does not require dose adjustments. Severe hepatic impairment lacks adequate safety data, making tirzepatide use in these patients rely on careful individual benefit risk assessment.
Patients with non alcoholic fatty liver disease, which is common in obesity, often experience substantial liver enzyme improvements on tirzepatide. ALT and AST reductions of 20 to 40 percent occur commonly, reflecting improved hepatic steatosis and inflammation. These improvements represent beneficial effects, not toxicity.
However, monitor liver enzymes at baseline and periodically during treatment to distinguish beneficial reductions from rare cases of drug induced liver injury. Elevations of liver enzymes to more than 3 times the upper limit of normal warrant dose hold and hepatology consultation.
Type 2 diabetes specific considerations
Individuals with type 2 diabetes starting tirzepatide often require adjustments to their existing diabetes medications. Insulin doses typically need reduction of 20 to 50 percent when initiating tirzepatide to prevent hypoglycemia. Sulfonylureas also require dose reduction or discontinuation.
The glucose lowering effects of tirzepatide exceed those of most other GLP-1 agonists due to the additional GIP receptor activation. Hemoglobin A1c reductions of 2 to 2.5 percent are common, particularly in individuals with baseline A1c above 8 percent.
Diabetic patients require more intensive glucose monitoring during tirzepatide initiation and escalation. Check fasting glucose daily for the first 2 weeks after starting or increasing dose. Monitor for hypoglycemia symptoms, particularly if taking concurrent insulin or sulfonylureas.
Diabetic gastroparesis represents a relative contraindication. Tirzepatide further slows gastric emptying, which can exacerbate existing gastroparesis and make blood sugar control more erratic. If gastroparesis is present, start with 2.5 mg and escalate only if gastrointestinal symptoms remain tolerable.
Diabetic kidney disease patients require the renal monitoring discussed above. The cardiovascular benefits of tirzepatide, including blood pressure reductions and improved lipid profiles, are particularly valuable in diabetic populations with elevated cardiovascular risk. However, this population also has higher baseline risk for complications, necessitating closer monitoring than non diabetic individuals.
For researchers managing diabetes with peptides, understanding these diabetes specific considerations prevents dangerous hypoglycemia while maximizing metabolic benefits.
Combining tirzepatide with other interventions
Tirzepatide works most effectively as part of comprehensive weight management rather than monotherapy. Strategic combinations with diet, exercise, and other interventions maximize results while potentially allowing lower maintenance doses.
Dietary approaches
High protein diets complement tirzepatide particularly well. Protein intakes of 1.2 to 1.6 grams per kilogram body weight help preserve lean mass during weight loss while supporting satiety beyond what tirzepatide provides alone. The combination often allows adequate results at lower doses than standard protocols.
Meal timing strategies affect side effect severity substantially. Smaller, more frequent meals of 300 to 400 calories produce less nausea than standard three meal patterns. Many individuals find that eating 4 to 5 small meals daily dramatically improves tolerability at higher doses.
Low fat approaches reduce nausea severity. Tirzepatide induced delayed gastric emptying affects fatty foods most dramatically. Limiting fat to 20 to 30 percent of calories, particularly in the 24 hours after injection, minimizes gastrointestinal distress.
Fiber intake requires balance. Adequate fiber prevents constipation, a common tirzepatide side effect. However, excessive fiber combined with delayed gastric emptying can worsen bloating and discomfort. Targeting 25 to 35 grams of fiber daily from varied sources usually provides the right balance.
Hydration becomes critical on tirzepatide. The appetite suppression often reduces thirst drive alongside hunger drive. Dehydration worsens side effects, particularly nausea, fatigue, and constipation. Aim for 3 to 4 liters of fluid daily, more if exercising significantly or in hot environments.
Exercise integration
Resistance training during tirzepatide use preserves muscle mass that would otherwise be lost. Two to three weekly resistance sessions targeting all major muscle groups maintains lean mass while allowing fat loss to proceed. This muscle preservation improves long term metabolic rate and functional outcomes.
Cardiovascular exercise enhances weight loss beyond medication effects alone. Moderate intensity cardio of 150 to 300 minutes weekly produces additional energy expenditure that compounds tirzepatide effects. However, avoid excessive cardio that creates unsustainable energy deficits which trigger metabolic compensation.
Timing exercise relative to tirzepatide injections affects tolerability. Some individuals find that intense exercise within 24 hours of injection worsens nausea. Others report no issues. Experiment with timing to find your optimal pattern.
Recovery considerations change during weight loss. Caloric deficits impair recovery from intense training. Adjust training volume and intensity to match your energy availability. Pushing through severe fatigue produces suboptimal results and increases injury risk.
Supplement considerations
Multivitamin supplementation becomes important during rapid weight loss. Micronutrient deficiencies develop when food intake decreases substantially. A comprehensive multivitamin providing 100 percent of daily values for key vitamins and minerals prevents deficiency related complications.
Electrolyte supplementation helps manage some tirzepatide side effects. Sodium, potassium, and magnesium support hydration status and may reduce fatigue, muscle cramps, and headaches. Those experiencing significant gastrointestinal side effects benefit from electrolyte replacement beyond what diet provides.
Protein supplementation simplifies meeting elevated protein targets while managing reduced appetite. Whey or plant based protein powders provide concentrated nutrition without excessive volume that triggers nausea.
Fiber supplements, particularly psyllium husk or methylcellulose, help manage constipation when dietary fiber proves insufficient. Start with small doses and increase gradually while maintaining excellent hydration to prevent worsening constipation.
Probiotic supplementation may reduce gastrointestinal side effects, though evidence remains limited. The altered gastric emptying and intestinal transit changes microbiome composition. Supplementing with broad spectrum probiotics might help maintain gut health during this transition.
Peptide combinations
Some individuals combine tirzepatide with other peptides for synergistic effects. BPC-157 may help manage any gastrointestinal side effects through its protective effects on gut mucosa. Standard BPC-157 dosing of 250 to 500 micrograms twice daily is typical.
CJC-1295 and ipamorelin combinations with tirzepatide may help preserve lean mass during weight loss by supporting growth hormone release. However, evidence for this combination remains anecdotal rather than clinically validated.
Thyroid peptides like thymosin alpha-1 are sometimes combined with tirzepatide by individuals focused on metabolic optimization. These combinations lack clinical validation and require careful monitoring for adverse interactions.
Always discuss peptide combinations with knowledgeable prescribers before implementation. While combination approaches may offer theoretical benefits, they also increase complexity, cost, and potential for unexpected interactions.
SeekPeptides provides comprehensive protocols for safely combining tirzepatide with complementary interventions, helping members maximize results while minimizing risks through evidence based approaches most resources completely ignore.
Long term dosing and discontinuation planning
Tirzepatide is designed for long term use, but understanding maintenance strategies and eventual discontinuation planning helps prevent weight regain and metabolic deterioration that commonly follows cessation.
Maintenance duration considerations
Clinical trial data demonstrates that tirzepatide efficacy persists for at least 72 weeks, the duration of the longest published trials. Real world data suggests efficacy continues beyond this timeframe in most individuals who maintain treatment.
The SURMOUNT-4 trial specifically examined what happens when individuals who achieved significant weight loss on tirzepatide either continued treatment or switched to placebo. Those who continued tirzepatide maintained their weight loss and even experienced modest additional reductions. Those switched to placebo regained substantial weight, averaging 14 percent regain over the subsequent 52 weeks.
This data underscores that tirzepatide is not a short term solution but rather long term metabolic management. The physiological changes that produced obesity, including altered appetite regulation, impaired satiety signaling, and metabolic adaptations, do not resolve permanently with weight loss alone. Discontinuing tirzepatide allows these underlying factors to reassert themselves.
Some individuals require indefinite treatment. Others may maintain results with dose reductions after sustained weight loss stabilization. Individual factors, including the severity of underlying metabolic dysfunction, lifestyle modification sustainability, and weight loss magnitude, influence whether long term treatment remains necessary.
Dose reduction strategies
After maintaining goal weight for 6 to 12 months at a given dose, some individuals successfully transition to lower maintenance doses. The weight loss itself improves metabolic function, potentially reducing the dose required to maintain results.
Systematic dose reduction involves decreasing by one tier every 8 to 12 weeks while closely monitoring weight, appetite, and metabolic parameters. If weight remains stable, appetite stays controlled, and metabolic markers remain improved, continue reducing. If weight increases by more than 5 percent or appetite becomes difficult to manage, return to the previous higher dose.
Some individuals find that alternating between two dose levels provides effective maintenance at lower average doses than continuous higher dosing. For example, alternating weekly between 7.5 mg and 5 mg provides an average dose of 6.25 mg, which may maintain results that previously required continuous 7.5 mg dosing.
Dose reduction attempts should occur only after weight has been stable for several months. Attempting reductions while still actively losing weight increases risk of premature weight regain.
Discontinuation protocols
When discontinuation becomes necessary, whether due to side effects, cost, pregnancy planning, or personal preference, abrupt cessation causes rapid weight regain in most individuals. Gradual tapering with intensive lifestyle focus provides the best chance for maintaining some of the weight loss achieved.
A reasonable discontinuation protocol involves reducing dose by one tier every 4 to 6 weeks while intensifying dietary control and physical activity. Move from 10 mg to 7.5 mg for 6 weeks, then 7.5 mg to 5 mg for 6 weeks, then 5 mg to 2.5 mg for 6 weeks before complete discontinuation. This 18 week taper allows gradual metabolic adjustment rather than abrupt change.
During tapering, expect appetite to increase and weight loss to slow or reverse. Proactive dietary restriction and increased physical activity help counteract these changes. Many individuals need to reduce caloric intake by 300 to 500 calories below weight maintenance levels during tapering to prevent rapid regain.
After complete discontinuation, most individuals regain some weight. Regaining 25 to 50 percent of lost weight within one year is common. Maintaining the remaining 50 to 75 percent of weight loss represents a significant achievement that still provides substantial health benefits.
Some individuals discontinue with plans to restart if regain becomes excessive. This intermittent use pattern, while not extensively studied, may provide a practical approach for individuals who cannot afford or tolerate continuous long term treatment.
Alternative maintenance strategies
Transitioning from tirzepatide to other GLP-1 agonists represents one alternative maintenance strategy. Semaglutide provides similar though slightly less potent effects than tirzepatide. Some individuals transition to semaglutide for cost reasons or better insurance coverage.
Transitioning from injectable tirzepatide to oral semaglutide or upcoming oral tirzepatide formulations may improve long term adherence. While oral formulations are generally less potent than injectable versions, they may provide sufficient maintenance effects after initial weight loss on injectable medications.
Combining lower dose GLP-1 agonist with other weight management medications represents another approach. Adding metformin, topiramate, or naltrexone-bupropion to low dose tirzepatide may allow maintenance at lower tirzepatide doses than monotherapy requires.
Very low calorie diets periodically, meal replacement programs, or intermittent fasting approaches may help some individuals maintain weight loss after tirzepatide discontinuation. These intensive dietary interventions can help counteract the metabolic adaptations and appetite increases that follow cessation.
Lifestyle medicine programs focusing on behavior change, stress management, sleep optimization, and social support improve long term maintenance rates. While medication provides the initial weight loss, sustainable lifestyle changes determine whether results persist after discontinuation.
Frequently asked questions
How long does it take to reach maximum tirzepatide dose?
Following standard escalation protocols, reaching the maximum 15 mg dose takes 20 weeks from initiation. However, many individuals achieve adequate results at lower doses and never progress to 15 mg. The time to reach your personal optimal dose varies based on individual tolerability and efficacy. Our tirzepatide dosing guide details personalized escalation approaches.
Can I stay at 2.5 mg if it is working well?
While 2.5 mg may produce initial appetite suppression and modest weight loss, clinical data shows this dose provides suboptimal long term efficacy. Most individuals benefit from escalation to at least 5 to 10 mg for sustained results. The initial response at 2.5 mg often diminishes over time, making escalation necessary to maintain efficacy. Discuss with your prescriber whether your results at 2.5 mg justify remaining at this dose or whether escalation would benefit you.
What happens if I miss multiple doses?
Missing one dose has minimal impact if you resume your regular schedule the following week. Missing 2 to 3 weeks may reduce efficacy as blood levels decline. If you miss more than 4 weeks, restart at a lower dose than where you left off, typically dropping back one or two dose tiers to allow readaptation. Extended breaks may necessitate repeating portions of the escalation schedule. Our peptide protocol guide covers restart strategies in detail.
Should I increase dose if weight loss slows?
Not necessarily. Weight loss naturally slows as you approach healthier body weight. Plateaus lasting 2 to 6 weeks are normal and do not indicate dose inadequacy. Evaluate diet and activity patterns before considering dose increases. If plateaus extend beyond 6 to 8 weeks despite optimized lifestyle factors, discuss dose escalation with your prescriber. Many plateaus break spontaneously without dose changes.
Can I split my weekly dose into twice weekly injections?
Tirzepatide is designed and studied as once weekly dosing. Splitting into twice weekly injections has not been studied and may alter efficacy and side effect profiles unpredictably. Some individuals experiment with this approach to reduce peak side effects, but this represents off label use without clinical validation. Discuss any dosing modifications with your prescriber before implementation.
How do I convert between Mounjaro and Zepbound doses?
Mounjaro and Zepbound contain identical tirzepatide formulations at the same dose levels. A 10 mg Mounjaro dose equals a 10 mg Zepbound dose. The only differences are the indications each is approved for, not the medication itself. Transitioning between brands requires no dose adjustment. Insurance coverage and cost often differ between the two brands despite identical formulations.
Will I regain weight if I stop tirzepatide?
Most individuals regain some weight after discontinuation. The SURMOUNT-4 trial showed average regain of 14 percent of body weight within one year after stopping. However, this still leaves most individuals substantially below their starting weight. The amount of regain varies considerably based on lifestyle modifications maintained after discontinuation. Gradual tapering combined with intensified diet and exercise provides the best chance for maintaining more of your weight loss. Our weight maintenance strategies help maximize retention of results.
Can I use tirzepatide while taking other diabetes medications?
Yes, but dose adjustments to your other diabetes medications are usually necessary. Insulin doses typically require reduction of 20 to 50 percent when starting tirzepatide to prevent hypoglycemia. Sulfonylureas also frequently need reduction or discontinuation. Metformin can generally continue unchanged and may provide additive benefits. Always coordinate tirzepatide initiation with your prescriber to ensure appropriate medication adjustments. Our medication interaction guide covers comprehensive management strategies.
What is the difference between pharmaceutical and compounded tirzepatide dosing?
Pharmaceutical tirzepatide from Mounjaro or Zepbound pens contains standardized concentrations and follows established dose escalation protocols. Compounded tirzepatide varies in concentration between pharmacies and batches, requiring careful calculation to ensure you are drawing the correct volume for your intended dose. Both follow the same dose progression of 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg, but compounded formulations require more attention to concentration conversions. Our compounding guide explains these differences comprehensively.
External resources
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers the most comprehensive resource available, with evidence based dosing strategies, individualized escalation approaches, troubleshooting protocols for challenging situations, and access to a community of thousands who have navigated these exact dosing questions successfully.
In case I don't see you, good afternoon, good evening, and good night. May your doses stay optimal, your side effects stay manageable, and your results stay sustainable. Join us.