Feb 10, 2026

Before you draw up your next dose, check your vial. Is it sitting on the counter? Has it been out of the fridge for hours? Days? If you are not sure how long your compounded semaglutide has been at room temperature, you might already have a problem. And the worst part is that degraded semaglutide does not always look different. It does not smell off. It does not change color until significant breakdown has occurred. You could be injecting a solution that has lost 30%, 50%, or even more of its potency without knowing it. That means your carefully calculated dose is delivering a fraction of what you think. Your weight loss stalls, your appetite suppression weakens, and you start wondering if the medication even works at all.
It does work. But only when stored correctly.
Compounded semaglutide is not the same as brand-name Ozempic or Wegovy when it comes to storage. The formulations differ. The stabilizers differ. The shelf life differs. And the consequences of poor storage can range from reduced effectiveness to complete waste of your investment. This guide covers everything you need to know about refrigerating compounded semaglutide, including the exact temperature ranges that matter, what happens when those ranges are exceeded, how to travel safely with your medication, and the specific warning signs that tell you a vial has gone bad. Whether you are new to semaglutide or months into your protocol, getting storage right is one of the simplest ways to protect both your results and your budget. SeekPeptides members consistently report that proper storage habits are one of the most overlooked factors in successful protocols, and the data backs them up.
The short answer: yes, compounded semaglutide must be refrigerated
There is no ambiguity here. Compounded semaglutide requires refrigeration at all times when not actively being used for an injection. The recommended storage temperature is between 36 and 46 degrees Fahrenheit, which translates to 2 to 8 degrees Celsius. This is the standard peptide storage range that applies to most reconstituted or liquid peptide formulations, and semaglutide is no exception.
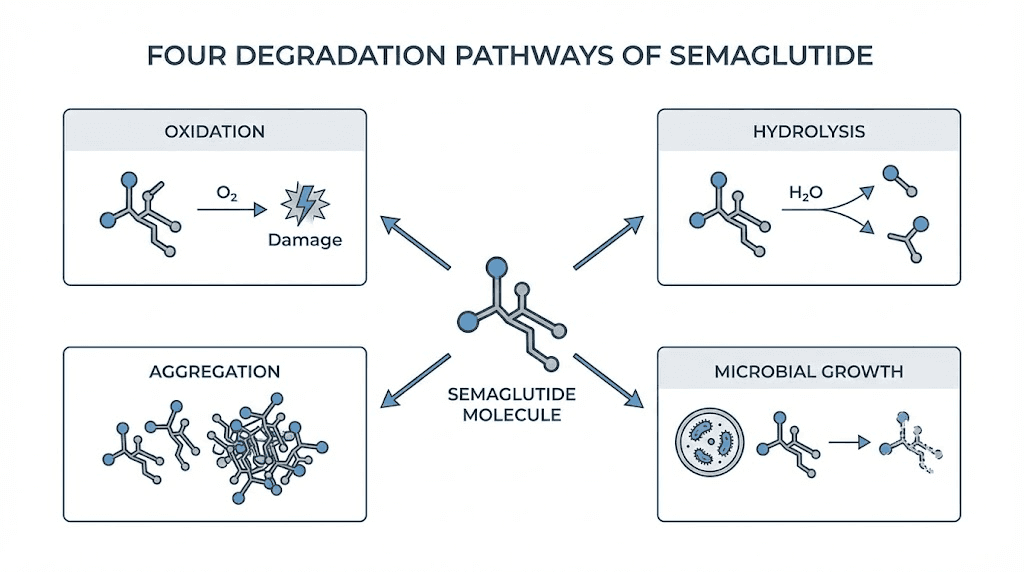
Why does this matter so much? Semaglutide is a 31-amino-acid peptide analog of human GLP-1. Like all peptides, its structure is held together by bonds that are sensitive to heat, light, and pH changes. When you store it outside the recommended temperature range, those bonds begin to break down through processes called oxidation, hydrolysis, and aggregation. The peptide literally falls apart at the molecular level.
This is not theoretical. Research published on semaglutide stability demonstrates that temperature is one of the primary factors driving degradation. Studies using liquid chromatography high-resolution mass spectrometry have identified multiple degradation pathways that accelerate at higher temperatures. At the ideal storage pH of above 7.0, semaglutide shows maximum stability, but temperature excursions can override even optimal pH conditions. The isoelectric point of semaglutide sits around pH 5.4, and degradation accelerates significantly between pH 4.5 and 5.5, which means that any formulation changes caused by temperature exposure can compound the problem by shifting the solution toward less stable conditions.

Compounded semaglutide vs. brand-name products: storage differences that matter
One of the biggest mistakes people make is assuming that compounded semaglutide follows the same storage rules as Ozempic or Wegovy. It does not. The differences are significant, and misunderstanding them can cost you potency, money, or both.
Brand-name storage allowances
Ozempic (the brand-name pen for diabetes) can be stored at room temperature, defined as 59 to 86 degrees Fahrenheit (15 to 30 degrees Celsius), for up to 56 days after first use. That is nearly two months at room temperature. This generous allowance exists because Novo Nordisk, the manufacturer, includes proprietary stabilizers, preservatives, and buffering agents that protect the peptide from degradation. The pre-filled pen design also limits exposure to contaminants and light.
Wegovy (the brand-name pen for weight loss) has tighter room-temperature limits. Once removed from refrigeration, it can remain at temperatures between 46 and 86 degrees Fahrenheit (8 to 30 degrees Celsius) for up to 28 days. After that, any unused medication must be discarded. Even with commercial-grade stabilizers, Wegovy cannot match Ozempic for room-temperature endurance because the formulations and delivery systems differ slightly.
Compounded semaglutide: a different situation entirely
Compounded semaglutide operates under fundamentally different rules. Compounding pharmacies create custom formulations based on individual prescriptions, and these formulations typically lack the proprietary stabilizers found in brand-name products. Many compounded versions do not contain preservatives at all. This means the peptide is more vulnerable to temperature fluctuations, microbial contamination, and chemical degradation from the moment it is prepared.
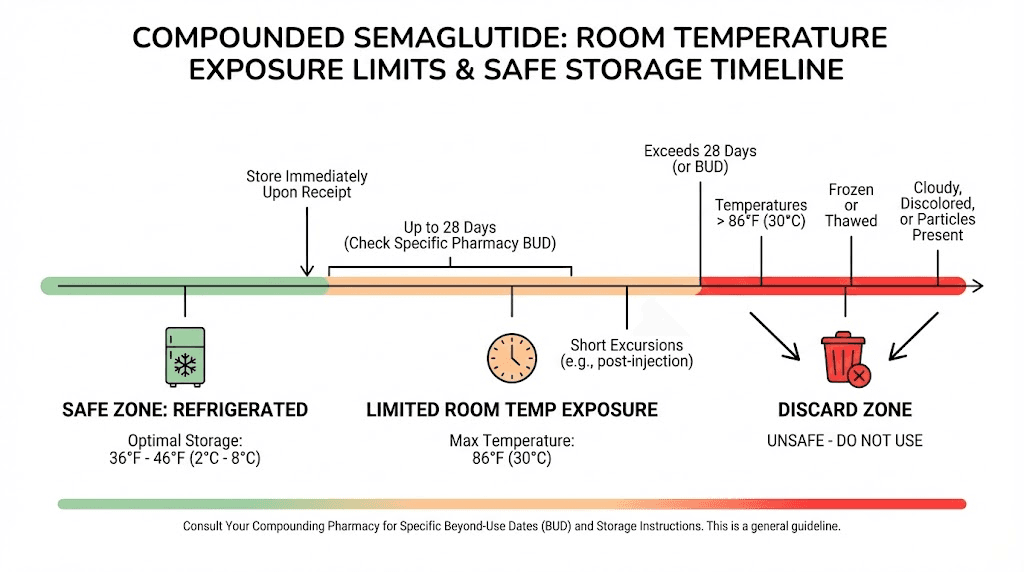
The practical result? Compounded semaglutide should be refrigerated continuously and only removed from the fridge for the brief time needed to prepare and administer an injection. We are talking minutes, not hours. Some pharmacies assign a Beyond-Use Date (BUD) of 30 to 90 days from the date of compounding, but this timeline assumes constant refrigeration. Leave your vial on the counter for a few hours on a warm day, and that BUD becomes meaningless.
The contrast is stark. Ozempic gives you 56 days at room temperature. Compounded semaglutide gives you minutes. Understanding this difference is critical for anyone switching from a brand-name product to a compounded formulation.
Product | Refrigerated shelf life | Room temp allowance | Stabilizers | Format |
|---|---|---|---|---|
Ozempic | Until expiration date | 56 days (15-30C) | Yes (proprietary) | Pre-filled pen |
Wegovy | Until expiration date | 28 days (8-30C) | Yes (proprietary) | Pre-filled pen |
Compounded semaglutide | 30-90 days (BUD) | Minutes only | Varies (often none) | Multi-dose vial |
What actually happens when compounded semaglutide gets warm
Understanding the science behind degradation helps you appreciate why the temperature rules exist. When compounded semaglutide is exposed to temperatures above 46 degrees Fahrenheit (8 degrees Celsius), several destructive processes begin simultaneously.
Oxidation
The amino acid residues in the semaglutide molecule, particularly methionine and tryptophan, are susceptible to oxidation. Heat accelerates this process dramatically. Oxidized semaglutide still looks like semaglutide under basic visual inspection, but its ability to bind to GLP-1 receptors is compromised. You get a molecule that resembles the active peptide but does not function like it.
Hydrolysis
Water molecules in the solution attack the peptide bonds holding the amino acid chain together. At refrigerator temperatures, this process happens slowly enough that the peptide remains effective for weeks or months. At room temperature, the rate increases. At elevated temperatures, above 86 degrees Fahrenheit (30 degrees Celsius), hydrolysis can become significant within hours. The peptide chain literally gets cut into fragments that have no therapeutic value.
Aggregation
This is perhaps the most dangerous degradation pathway. When semaglutide molecules begin to unfold due to heat exposure, they can stick together and form clumps called aggregates. These aggregates reduce the amount of active, available semaglutide in solution. Worse, protein aggregates can sometimes trigger immune reactions at the injection site, leading to redness, swelling, or hard lumps under the skin. If you notice injection site reactions that seem worse than usual, temperature-induced aggregation could be the culprit.
Microbial growth
Compounded semaglutide vials are typically multi-dose, meaning the rubber stopper gets punctured multiple times. Each puncture creates a potential entry point for bacteria. At refrigerator temperatures, bacterial growth is suppressed. At room temperature, bacteria can multiply rapidly, especially in formulations without preservatives. Using a contaminated vial does not just waste medication. It creates a genuine infection risk at the injection site.
Stress stability studies on semaglutide have demonstrated that the peptide remains relatively stable for 28 days at temperatures up to 25 degrees Celsius and shows significant degradation at 40 degrees Celsius within the same timeframe. At 60 degrees Celsius, breakdown products become detectable within days. At 80 degrees Celsius, semaglutide maintained stability for only about 3 hours before significant decomposition occurred.
The exact refrigeration protocol for compounded semaglutide
Knowing that refrigeration is required is not enough. How you refrigerate matters almost as much as whether you refrigerate.
Temperature sweet spot
The target range is 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Within this range, aim for the middle, around 40 to 42 degrees Fahrenheit (4 to 5 degrees Celsius). This gives you a buffer on both sides. If your refrigerator temperature fluctuates slightly during door openings (which happens dozens of times daily), you stay within the safe zone.
Where to place the vial
Not all spots in your refrigerator maintain the same temperature. The door shelves experience the most temperature fluctuation because they are directly exposed to warm air every time the door opens. The back of the top shelf or the center of a middle shelf provides the most consistent temperature. If you have a dedicated medication area, even better.
Keep the vial away from the refrigerator walls and cooling elements. Direct contact with the coldest surfaces can cause localized freezing, which is just as damaging as heat exposure. Freezing semaglutide causes the peptide molecules to form ice crystals that physically shear the molecular structure apart. Once frozen and thawed, the damage is irreversible.
Light protection
UV and visible light accelerate photo-degradation of semaglutide. Research using UV stability chambers has confirmed that light exposure drives additional degradation pathways beyond what temperature alone causes. Keep your vial in its original box or wrap it in aluminum foil if the box has been discarded. Many compounding pharmacies ship semaglutide in amber vials specifically to reduce light exposure, but additional protection does not hurt.
Handling protocol
When preparing your injection, remove the vial from the refrigerator. Allow it to warm slightly in your hand for 30 to 60 seconds. Cold injections can cause more discomfort at the injection site. Draw your dose using proper reconstitution and dosing techniques. Return the vial to the refrigerator immediately after use. The entire process should take less than 5 minutes from fridge to fridge.
Never leave the vial on a bathroom counter while you shower. Do not set it on a kitchen counter and forget it while cooking dinner. Do not toss it in your bag for a gym session and remember it hours later. Each of these common scenarios exposes the peptide to conditions that accelerate every degradation pathway described above.
How long can compounded semaglutide be out of the fridge?
Accidents happen. You forget to put it back. The power goes out. A delivery arrives during a heat wave. Understanding the real-world limits helps you make informed decisions about whether a vial is still usable.
The conservative guideline
Most compounding pharmacies recommend that compounded semaglutide not exceed room temperature exposure of more than a few minutes at a time. This is the safest guidance. If you follow this, you eliminate virtually all storage-related potency concerns.
The practical reality
Third-party stability studies suggest that semaglutide (in certain formulations) can maintain reasonable stability at temperatures up to 86 degrees Fahrenheit (30 degrees Celsius) for up to 24 hours. Some data suggests stability extends to 28 days at controlled room temperature for specific brand-name formulations, but remember, those formulations contain stabilizers that compounded versions typically lack.
For compounded semaglutide specifically, the practical guidance breaks down like this:
Under 30 minutes at room temperature (below 77F/25C): Almost certainly fine. Return to the fridge and continue using normally.
30 minutes to 2 hours at room temperature: Likely still usable, but some potency reduction is possible depending on the specific formulation and ambient conditions. Return to the fridge immediately.
2 to 8 hours at room temperature: Gray area. Contact your compounding pharmacy. The answer depends on the specific formulation, whether preservatives were included, and the ambient temperature during exposure.
8 to 24 hours at room temperature: Significant potency loss is likely. If the ambient temperature was below 77 degrees Fahrenheit, some pharmacies may say it is still usable. If the temperature was higher, or if you are unsure, replacing the vial is the safer choice.
Over 24 hours at room temperature: Discard the vial. The risk of degradation, contamination, and reduced effectiveness is too high to justify continued use.
Any exposure above 86F (30C): Discard immediately. Even brief exposure to high heat causes rapid degradation that cannot be reversed by returning the vial to the fridge.
Any freezing: Discard immediately. Ice crystal formation physically destroys the peptide structure. Thawing does not restore functionality.
When in doubt, contact your pharmacy. They know the specific formulation, the stabilizers (or lack thereof), and can give you guidance based on the exact product you received. But as a general rule, if you have to ask whether your compounded semaglutide is still good, the safest answer is usually to replace it. The cost of a new vial is far less than the cost of weeks on a degraded medication that is not delivering results.
Lyophilized powder vs. liquid formulation: different storage rules
Compounded semaglutide comes in two primary forms, and the storage requirements differ significantly between them. Understanding which form you have changes everything about how you handle it.
Lyophilized (freeze-dried) powder
Some compounding pharmacies provide semaglutide as a lyophilized powder that you reconstitute yourself using bacteriostatic water or another diluent. In this form, the peptide is remarkably stable. Properly stored lyophilized semaglutide can maintain potency for up to 36 months at refrigerator temperatures (2 to 8 degrees Celsius). Some research suggests it can even tolerate room temperatures up to 30 degrees Celsius for up to 14 days without significant degradation.
However, once you add bacteriostatic water and reconstitute the powder into a liquid solution, the clock starts ticking. Reconstituted semaglutide must be refrigerated immediately and used within 28 days. Some pharmacies recommend even shorter windows, as low as 14 days, depending on the specific formulation and diluent used.
If you receive lyophilized semaglutide, keep the unreconstituted vials in the refrigerator. Only reconstitute what you need for your current dosing cycle. This approach maximizes the overall lifespan of your supply and minimizes waste. Use our semaglutide mixing guide for the correct bacteriostatic water ratios.
Pre-mixed liquid formulation
Most compounded semaglutide arrives as a pre-mixed liquid solution ready for injection. This is the more convenient option, but it comes with stricter storage requirements. Pre-mixed formulations have already been reconstituted by the pharmacy, which means the 28-day (or longer, depending on the BUD) countdown began the moment it was compounded, not when you received it.
Shipping time matters here. If your medication spent two days in transit without proper cold-chain shipping, those are two days of potential degradation that count against your usable window. Reputable compounding pharmacies ship with ice packs and insulated packaging specifically to minimize temperature excursions during delivery. When your package arrives, check the ice packs. If they are completely melted and the medication is warm to the touch, contact the pharmacy immediately. They should replace the shipment.
Form | Before reconstitution | After reconstitution | Room temp tolerance |
|---|---|---|---|
Lyophilized powder | Up to 36 months (2-8C) | 14-28 days (2-8C) | 14 days at up to 30C (powder only) |
Pre-mixed liquid | N/A (already mixed) | 30-90 days BUD (2-8C) | Minutes at room temp |
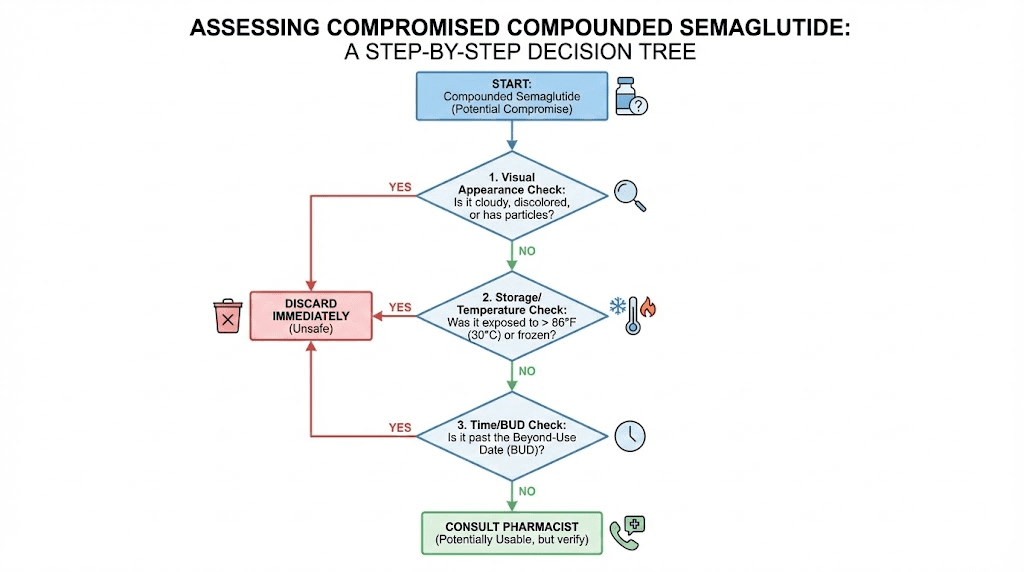
Signs your compounded semaglutide has gone bad
Degraded semaglutide does not always announce itself. In the early stages of breakdown, the solution can look perfectly normal while delivering significantly reduced potency. But as degradation progresses, visible and functional signs emerge.
Visual indicators
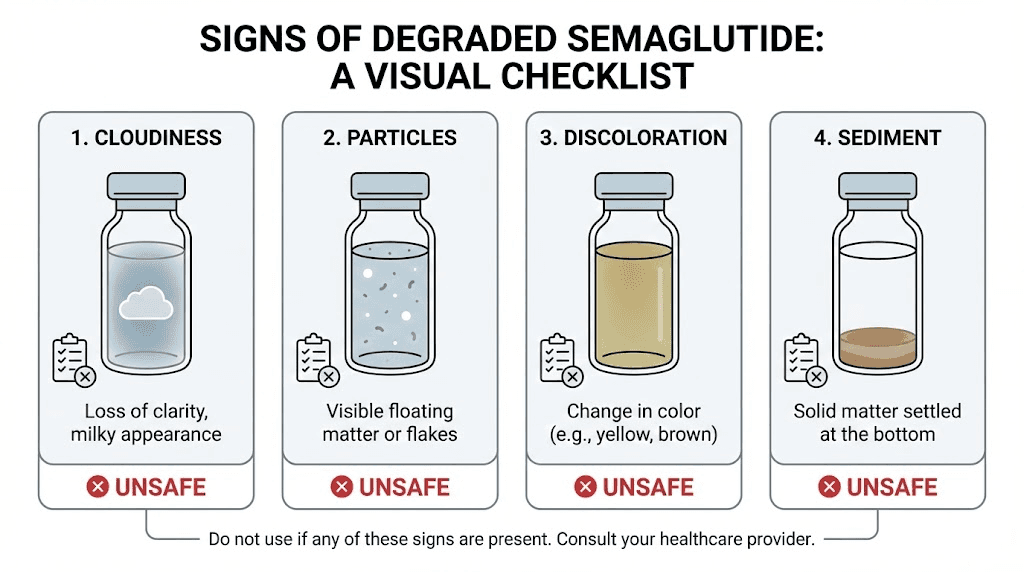
Cloudiness or haziness. Fresh compounded semaglutide should be clear and colorless, like water. Any cloudiness, haziness, or opalescence indicates aggregation or contamination. This is the most obvious visual sign of degradation. Do not use a cloudy vial under any circumstances.
Floating particles. Hold the vial up to a light source and look carefully. Tiny floating particles, fibers, or specks indicate either contamination or peptide aggregation. Either way, the vial is compromised.
Color changes. Fresh semaglutide solution is colorless. Any yellow, brown, or amber discoloration suggests oxidative degradation. The darker the color, the more extensive the breakdown. Even slight yellowing warrants caution.
Sediment at the bottom. If you see material settled at the bottom of the vial that does not dissolve with gentle swirling, the peptide has aggregated beyond recovery.
Functional indicators
Sometimes the vial looks fine, but the medication is not working as expected. Watch for these patterns:
Reduced appetite suppression. If you have been on a stable dose and suddenly notice your appetite returning stronger than usual, storage-related potency loss could be the cause. Before assuming the medication stopped working, check your storage practices.
Weight loss plateau or reversal. A sudden stall after consistent progress, particularly one that coincides with a new vial or a storage incident, suggests the new vial may have degraded peptide. Compare your results between vials. If each new vial from a different batch performs differently, storage during shipping could be the issue.
Changed side effect profile. Paradoxically, degraded semaglutide might produce different side effects than the intact molecule. Aggregated peptide fragments can trigger injection site reactions that the native peptide does not cause. New lumps, redness, or itching at the injection site could indicate you are injecting degradation products rather than active semaglutide.
Unusual odor. Semaglutide solution should be essentially odorless. Any unusual smell, particularly anything musty, sour, or chemical, indicates bacterial contamination or chemical degradation. Discard immediately.
Traveling with compounded semaglutide: the complete protocol
Travel presents the biggest storage challenge for compounded semaglutide users. Whether you are flying across the country, driving to a weekend destination, or spending a week at the beach, maintaining proper cold-chain storage requires planning. Here is how to handle every scenario.
Air travel
Always carry your medication in your carry-on bag. Never check it. Checked luggage compartments can experience extreme temperature swings, from freezing at altitude to baking on a hot tarmac. Neither is safe for compounded semaglutide. TSA allows medications in carry-on luggage, including syringes and needles, as long as the medication is clearly labeled.
Use an insulated medical travel pouch or a small insulated lunch bag with a frozen gel pack. Wrap the gel pack in a thin cloth or paper towel to prevent direct contact with the vial, which could cause freezing. The goal is to maintain temperatures between 36 and 46 degrees Fahrenheit throughout the journey. For flights longer than 4 to 6 hours, consider using two gel packs and swapping them if one warms up.
Bring documentation. A copy of your prescription or a letter from your compounding pharmacy helps if TSA questions the medication. While most agents are familiar with injectable medications, having documentation eliminates potential delays.
Car travel
Never store your medication in the trunk, especially during summer months. Trunk temperatures can exceed 140 degrees Fahrenheit (60 degrees Celsius) in direct sunlight. That is a temperature at which semaglutide degradation happens in hours, not days. Use the same insulated pouch approach as air travel, and keep it in the passenger compartment with climate control.
For longer road trips, a small plug-in cooler that runs off the car power outlet provides the most reliable temperature control. These devices can maintain refrigerator temperatures indefinitely, eliminating the worry about gel packs melting.
Hotel stays
Most hotel rooms have a mini-fridge. Place your semaglutide inside immediately upon arrival. If the mini-fridge does not have adjustable temperature settings, test it with a glass of water first to make sure it is actually cold enough without freezing. Some hotel mini-fridges run warm (more like 50 degrees Fahrenheit) while others can freeze items placed in certain spots.
If your accommodation does not have a refrigerator, call ahead and request one. Most hotels will provide a small fridge for medical needs at no charge. As a backup, you can ask the hotel restaurant or bar to store your medication in their walk-in cooler.
Beach, camping, and outdoor activities
For outdoor situations where standard refrigeration is not available, a quality insulated cooler with gel packs is your best option. High-end coolers can maintain cold temperatures for 24 to 48 hours. Keep the cooler in the shade, avoid opening it unnecessarily, and replace gel packs from a nearby source whenever possible.
If you are camping for multiple days without access to refrigeration or ice, consider adjusting your dosing schedule to take your weekly injection before you leave and after you return, rather than trying to maintain cold-chain in wilderness conditions.
Beyond-use dates: what they mean and why they matter
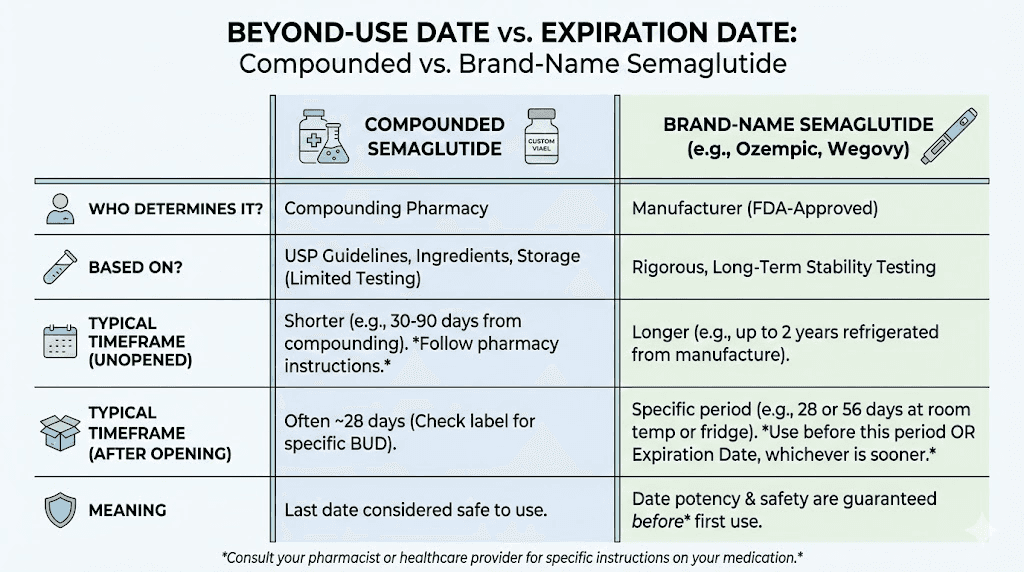
Unlike FDA-approved medications that carry manufacturer-assigned expiration dates backed by years of stability testing, compounded semaglutide comes with a Beyond-Use Date (BUD). Understanding the difference helps you make informed decisions about your medication.
What a BUD actually represents
A Beyond-Use Date is the date after which a compounded medication should not be used. It is assigned by the compounding pharmacist based on available stability data, USP guidelines, and the specific formulation. The BUD is typically more conservative than a traditional expiration date because compounded medications have not undergone the extensive stability testing that commercial products require.
Most compounded semaglutide formulations receive a BUD of 30 to 90 days from the date of compounding. Pharmacies that conduct their own rigorous stability testing may assign longer BUDs, sometimes up to 120 days. Pharmacies that rely on external stability data or general USP guidelines tend toward shorter BUDs, often 14 to 28 days.
The BUD assumes proper storage
This is the critical point that many people miss. A BUD of 90 days means 90 days under continuous refrigeration at 2 to 8 degrees Celsius. Every hour spent at room temperature effectively shortens that BUD. A vial with a 90-day BUD that spent 3 days in transit without proper cooling, then sat on your counter for 6 hours before you noticed, no longer has a real BUD of 90 days. Its effective BUD is shorter, possibly much shorter, and there is no easy way to calculate exactly how much shorter.
How to check your BUD
Your compounding pharmacy is required to print the BUD on the vial label. Look for a date marked as "BUD," "Use By," "Expiration," or "Discard After." If you cannot find a date on your vial, contact the pharmacy immediately. Using compounded medication without a clear BUD is risky and generally inadvisable.
Keep a log of when you received the medication and any storage incidents. This helps you make informed decisions about whether to continue using a vial that has experienced temperature excursions. SeekPeptides provides detailed tracking tools for members who want to monitor their medication storage and usage patterns systematically.
Common storage mistakes that destroy your semaglutide
Even people who know that refrigeration is required make mistakes that compromise their medication. Here are the most common ones, along with the fix for each.
Mistake 1: storing in the refrigerator door
The refrigerator door is the warmest, most temperature-variable location in any fridge. Every time the door opens, items on the door shelves experience a blast of warm air. For food, this barely matters. For peptides, it creates repeated micro-temperature excursions that accelerate degradation over time.
Fix: Store your vial in the center or back of a middle shelf, ideally in a small container or ziplock bag for additional protection.
Mistake 2: placing the vial directly against the back wall
The back wall of many refrigerators, especially older models, contains the cooling element. Temperatures directly against this wall can drop below freezing, even if the overall fridge temperature is set correctly. A vial touching the back wall can experience localized freezing that destroys the peptide in that portion of the solution.
Fix: Leave at least two inches between your vial and the back wall. Use a small box or container as a buffer.
Mistake 3: forgetting the vial on the counter after injection
This is by far the most common mistake. You take the vial out, prepare your injection, administer it, clean up, and then get distracted. An hour later, you realize the vial is still sitting next to the bathroom sink. Maybe two hours. Maybe you go to bed and find it in the morning.
Fix: Create a rigid habit. The vial goes back in the fridge the moment you finish drawing your dose, before you inject. Set an alarm or a visual reminder near your injection supplies. Some people place their car keys or phone next to the vial as a forced reminder to return it.
Mistake 4: ignoring shipping conditions
Your vial arrives in the mail. The box is warm. The ice packs are liquid. You shrug and put it in the fridge, assuming it will "recover." It will not. Once degradation has occurred, refrigeration does not reverse it. Refrigeration only slows further degradation.
Fix: When your shipment arrives, immediately check the ice packs and the vial temperature. If the ice packs are warm and the medication feels room temperature or above, contact your pharmacy before using it. Reputable pharmacies will replace shipments that were compromised during transit.
Mistake 5: stockpiling vials without checking BUDs
Some people order multiple vials at once and stack them in the fridge without checking the Beyond-Use Dates. Three months later, they reach for the oldest vial and discover it expired weeks ago. The refrigerator kept it cold, but it still degraded over time as chemical processes continued slowly.
Fix: Mark each vial with the date received and the BUD. Use a first-in, first-out system. Always use the oldest vial first.
Mistake 6: using a bathroom medicine cabinet
Bathrooms are the worst location for any medication that requires temperature control. Steam from showers creates humidity and temperature spikes that can reach 90 degrees Fahrenheit or higher multiple times daily. A medicine cabinet in a frequently used bathroom provides some of the harshest storage conditions in any home.
Fix: Never store compounded semaglutide anywhere other than a refrigerator. The medicine cabinet is for aspirin and bandages, not temperature-sensitive peptides.
Power outages and emergency situations
When the power goes out, your refrigerator becomes a slowly warming box. Knowing what to do during a power outage can save your medication.
Short outages (under 4 hours)
A well-stocked refrigerator maintains safe temperatures for approximately 4 hours if the door remains closed. Keep the fridge door shut. Do not open it to check on your medication. Every door opening releases cold air and accelerates warming. If the outage ends within 4 hours, your medication is almost certainly fine.
Extended outages (4 to 24 hours)
After 4 hours, interior temperatures begin rising above the safe zone. If you have advance warning (hurricane, planned maintenance), transfer your medication to a cooler with ice or frozen gel packs before the power goes out. A good cooler can maintain refrigerator temperatures for 12 to 24 hours.
If you did not prepare and the power has been out for more than 4 hours, open the fridge quickly and check the internal temperature if you have a thermometer. If the temperature is still below 46 degrees Fahrenheit (8 degrees Celsius), the medication is fine. If it has risen above that threshold, note the time and temperature. The longer it stays above 46 degrees, the more degradation occurs.
Multi-day outages
For outages lasting more than 24 hours, find alternative refrigeration. A neighbor with power, a local pharmacy willing to store your medication, or a store-bought bag of ice in a cooler can all work. If none of these options are available and the medication has been at room temperature for more than 24 hours, plan to get a replacement vial once services are restored.
How proper storage affects your results
Storage is not just a technical detail. It directly impacts the outcomes you are working toward. Here is why getting this right matters for your actual results.
Dose consistency
Your dosage protocol is calibrated based on the assumption that each injection delivers the full amount of active semaglutide. If degradation has reduced the potency by 20%, your 2.5mg dose is actually delivering 2mg. Your body does not know the label says 2.5mg. It only responds to what it actually receives. This creates the illusion that the dose is too low, potentially leading you to increase the dose unnecessarily, which creates problems when you eventually switch to a properly stored vial that delivers the full amount.
Appetite suppression consistency
One of the primary benefits of semaglutide is appetite suppression through GLP-1 receptor activation. When potency fluctuates due to inconsistent storage, your appetite suppression becomes unpredictable. Some weeks it works great. Other weeks, you are hungry all day. This inconsistency makes it nearly impossible to establish stable eating patterns, which is critical for sustained weight loss.
Side effect management
Many semaglutide side effects, particularly gastrointestinal symptoms, are dose-dependent. When degradation reduces potency, you might experience fewer side effects. Then, when you switch to a properly stored vial, the full-potency dose hits harder than expected. This roller coaster makes side effect management difficult and can discourage people from continuing their protocol.
Cost efficiency
Compounded semaglutide represents a significant financial investment. A vial that degrades due to improper storage is money thrown away. Even partial degradation means you are paying full price for reduced effectiveness. Proper storage is the simplest way to protect your investment and ensure you get every milligram of value from each vial.
Refrigerator thermometer: the $10 investment that protects your semaglutide
Most people have no idea what temperature their refrigerator actually maintains. The dial inside that says "1 through 5" or "Min to Max" gives you no useful information about actual temperature. A dedicated refrigerator thermometer costs less than $10 and provides critical data.
Why you need one
Consumer refrigerators are designed to keep food safe, not to maintain pharmaceutical-grade temperature control. The internal temperature can vary by 5 to 10 degrees Fahrenheit depending on location within the fridge, time since the last door opening, and how full the fridge is. A thermometer placed next to your medication vial tells you exactly what temperature your semaglutide is experiencing.
What to look for
Digital thermometers with min/max memory are ideal. They record the lowest and highest temperatures reached since the last reset, which tells you about temperature fluctuations even when you are not looking. Some models include alarms that alert you when the temperature exceeds your set range. For medication storage, set the high alarm at 46 degrees Fahrenheit and the low alarm at 35 degrees Fahrenheit to catch both overheating and near-freezing conditions.
Calibration
Test your thermometer in ice water (should read 32 degrees Fahrenheit / 0 degrees Celsius) to verify accuracy. If it is off by more than 2 degrees, adjust accordingly or replace it. An inaccurate thermometer is worse than no thermometer because it gives false confidence.
Frequently asked questions
Can I freeze compounded semaglutide to make it last longer?
No. Freezing destroys the peptide structure through ice crystal formation. Once frozen and thawed, compounded semaglutide loses effectiveness and should be discarded. This applies to both liquid formulations and reconstituted solutions. Lyophilized (freeze-dried) powder is the only form that can tolerate sub-zero temperatures, and that is because the water has already been removed.
My vial was left out overnight. Is it still good?
It depends on the ambient temperature and the specific formulation. If your room stays below 77 degrees Fahrenheit (25 degrees Celsius) and the vial was out for less than 8 hours, it may still be usable. Contact your compounding pharmacy with the specific details. If the room was warm, above 80 degrees, or the vial was in direct sunlight, the safer choice is to discard it. Degradation cannot be reversed by returning the vial to the refrigerator.
Does compounded semaglutide expire faster than Ozempic?
Yes. Compounded formulations typically have Beyond-Use Dates of 30 to 90 days, while unopened Ozempic pens remain stable until their manufacturer-printed expiration date (often 18 to 24 months from production). Once opened, Ozempic is good for 56 days at room temperature. Compounded semaglutide cannot match these timelines because it lacks the proprietary stabilizers in brand-name products.
What temperature should my fridge be set to for semaglutide storage?
Set your refrigerator to maintain a temperature between 36 and 40 degrees Fahrenheit (2 to 4 degrees Celsius). This puts you comfortably in the middle of the recommended 36 to 46 degree range with buffer on the warm side. Use a dedicated thermometer to verify the actual temperature rather than relying on the refrigerator dial setting.
Is it safe to use semaglutide that looks slightly yellow?
No. Fresh compounded semaglutide should be clear and colorless. Any discoloration, even slight yellowing, indicates oxidative degradation. The peptide has broken down and should not be injected. Using degraded medication wastes your dose, delivers reduced results, and potentially exposes you to degradation byproducts.
Can I transfer compounded semaglutide to a different container for travel?
No. Keep the medication in the original vial from the compounding pharmacy. Transferring to another container introduces contamination risk, exposes the medication to uncontrolled conditions, and removes it from the labeled packaging that includes dosing information and the Beyond-Use Date.
How do I store semaglutide during a long flight?
Use an insulated medical travel pouch with a frozen gel pack. Place a cloth barrier between the gel pack and the vial to prevent freezing. Keep it in your carry-on, not checked baggage. For flights longer than 6 hours, bring an extra gel pack. Most airline staff will not provide refrigeration, so plan to be self-sufficient.
Does the needle puncture affect storage life?
Each needle puncture through the rubber stopper creates a tiny channel that bacteria can potentially enter. This is why multi-dose vials have shorter use windows than sealed vials. Always swab the stopper with an alcohol wipe before each use, use a new sterile needle for every draw, and never touch the stopper with your fingers. These practices minimize contamination risk and help your vial remain usable throughout its BUD.
Building a proper storage system at home
The best storage system is one you do not have to think about. Here is a simple setup that eliminates decision-making and protects your medication automatically.
The dedicated medication zone
Designate a specific spot in your refrigerator for medications. A small, clear plastic container works well. Place it on a middle shelf, away from the back wall and door. This container becomes the one place your semaglutide always lives when not in your hand. Building this habit eliminates the "I just set it down for a second" problem that leads to hours-long counter exposure.
The injection station
Set up your injection supplies near the refrigerator, not in the bathroom. Keeping your injection supplies in the kitchen or wherever your fridge is located means the vial never has to travel far. Alcohol wipes, syringes, and a sharps container can all sit in a small kit near the fridge. You take the vial out, prepare and administer at the counter, and return the vial, all within a few feet of the refrigerator.
The tracking system
Keep a simple log, either a sticky note on the fridge or a note on your phone, with three pieces of information for each vial: the date you received it, the Beyond-Use Date, and any storage incidents (left out, power outage, etc.). This log helps you make informed decisions about whether a vial is still reliable and when to reorder. SeekPeptides members have access to protocol tracking tools that make this process even more streamlined.
What to do if you suspect your semaglutide has degraded
If you have reason to believe your compounded semaglutide has been compromised, whether through a storage incident, visual changes, or unexpected loss of effectiveness, here is your action plan.
Step 1: inspect the vial
Hold it up to a bright light. Look for cloudiness, particles, color changes, or sediment. If any of these are present, stop using the vial immediately.
Step 2: review your storage history
Think back over recent days. Was the vial left out? Did the power go out? Was it shipped during a heat wave? Did you notice the fridge feeling warm? Any of these events could explain reduced potency.
Step 3: contact your pharmacy
Describe what happened. Give them specifics: how long the vial was at room temperature, the approximate ambient temperature, and any visual changes. They can advise whether the vial is still usable based on their knowledge of the specific formulation.
Step 4: replace if uncertain
When in doubt, replace. A new vial costs money, but using a degraded vial costs you weeks of suboptimal results. The math favors replacement every time. If you have been struggling with plateau issues and suspect storage problems, a fresh vial with confirmed cold-chain handling is the fastest way to get back on track.
Step 5: fix your storage system
Whatever went wrong, prevent it from happening again. Move the vial to a better spot in the fridge. Set a phone alarm to remind you to return it after injections. Get a thermometer. Create the habit system described above. Prevention is always cheaper than replacement.
The relationship between storage and your weight loss timeline
People want to know how long semaglutide takes to work. They research dosing charts. They track their weekly progress. But many overlook the fact that inconsistent storage creates inconsistent results that distort their entire timeline assessment.
Consider this scenario. You start your protocol with a properly stored vial and see great results for the first four weeks. You reorder, and the new vial arrives during a summer heat wave. It sat on a delivery truck for 8 hours in 95-degree heat before you got it home. You put it in the fridge, but the damage is done. Weeks 5 through 8 show dramatically reduced results. You start questioning whether semaglutide is still working for you. Maybe you think you have hit tolerance. Maybe you think the dose needs adjusting.
But the real problem was storage. Fix the storage, and the results return.
This pattern repeats across the semaglutide community more often than most people realize. Before making any changes to your dosing protocol, rule out storage issues first. It is the simplest variable to control and the easiest to fix.
Comparing storage requirements across GLP-1 medications
If you are using or considering other GLP-1 receptor agonist medications, here is how their storage requirements compare. Understanding these differences is especially important if you are switching between medications.
Medication | Refrigerated storage | Room temp tolerance | BUD/Expiration | Freezing tolerance |
|---|---|---|---|---|
Compounded semaglutide | Required (2-8C) | Minutes only | 30-90 day BUD | None |
Ozempic (pen) | Before first use | 56 days (15-30C) | Mfr. expiration | None |
Wegovy (pen) | Before first use | 28 days (8-30C) | Mfr. expiration | None |
Compounded tirzepatide | Required (2-8C) | Minutes only | 30-90 day BUD | None |
Mounjaro (pen) | Before first use | 21 days (up to 30C) | Mfr. expiration | None |
Notice the pattern. Brand-name products with commercial stabilizers offer generous room-temperature windows. Compounded formulations, whether semaglutide or tirzepatide, require strict refrigeration. This is not a quality issue. It is a formulation issue. Compounding pharmacies are creating effective, legitimate medications, but those medications need more careful handling than their brand-name counterparts.
If you are comparing semaglutide to tirzepatide or evaluating options like retatrutide, storage requirements should factor into your decision. Medications you can store at room temperature are more convenient for travel and daily life. Medications requiring continuous refrigeration demand more planning but often come at a lower cost point. Both approaches work, as long as storage protocols are followed.
The cost of improper storage: doing the math
Here is a practical calculation that puts storage mistakes into financial perspective.
A typical compounded semaglutide vial costs between $100 and $400 depending on the pharmacy, concentration, and quantity. Let us use $200 as a moderate estimate for a 4-week supply.
Scenario 1: vial left on counter for 6 hours in a 78-degree kitchen. Estimated potency loss: 5 to 15%. You are now getting $170 to $190 worth of medication from your $200 vial. Over a year, that is $120 to $360 wasted.
Scenario 2: vial arrives warm from shipping and you use it anyway. Estimated potency loss: 15 to 40% depending on transit conditions. You are getting $120 to $170 of value from your $200 vial. Over a year, if this happens with every shipment, that is $360 to $960 wasted.
Scenario 3: vial freezes against the back wall of the fridge. Complete loss. $200 gone. If this happens once per year, that is the cost of a nice dinner out. If it happens repeatedly because the problem is not fixed, it adds up fast.
Compare these losses to the cost of prevention: a $10 thermometer, a $5 plastic container for the fridge, and 30 seconds of attention each time you use your vial. The return on investment for proper storage is enormous.
Storage tips from experienced users
The SeekPeptides community has collectively handled thousands of compounded semaglutide vials. Here are the most practical tips that experienced users share:
Label every vial with the date received. The pharmacy label shows the BUD, but not necessarily when you got it. Knowing both dates helps you track cold-chain gaps.
Take a photo of the vial when it arrives. Note the clarity, color, and any markings. If the solution changes later, you have a reference point for comparison.
Use a small rubber band around the vial. The band prevents it from rolling in the fridge and ending up against the back wall where freezing can occur.
Order replacement vials before your current one runs out. This gives you a buffer if a shipment is delayed or arrives compromised. Running out and then waiting for delivery creates pressure to use potentially degraded medication.
Keep a backup gel pack in the freezer. If the power goes out, you can immediately transfer your medication to an insulated container with the gel pack. Having this ready to go eliminates scrambling during emergencies.
Clean the rubber stopper every time. This has less to do with storage temperature and more to do with contamination prevention, but it extends the safe-use window of your vial. An alcohol wipe before every draw takes three seconds and significantly reduces contamination risk.
For researchers looking to optimize every aspect of their semaglutide protocol, from storage to dosing to expected timelines, SeekPeptides provides the most comprehensive resource available. Members access detailed protocols, expert guidance, and a community of thousands who have navigated these exact questions.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your results stay consistent.