Feb 26, 2026

Your joints ache. Your rings feel tighter than they should. And that low-grade fatigue that never quite goes away? It has a name. Chronic inflammation sits behind more health problems than most people realize, quietly driving joint pain, metabolic dysfunction, cardiovascular damage, and autoimmune flares. For years, the standard approach meant corticosteroids, NSAIDs, or heavy immunosuppressants. Effective, sometimes. But blunt instruments with side effects that stack up fast.
Then something unexpected happened.
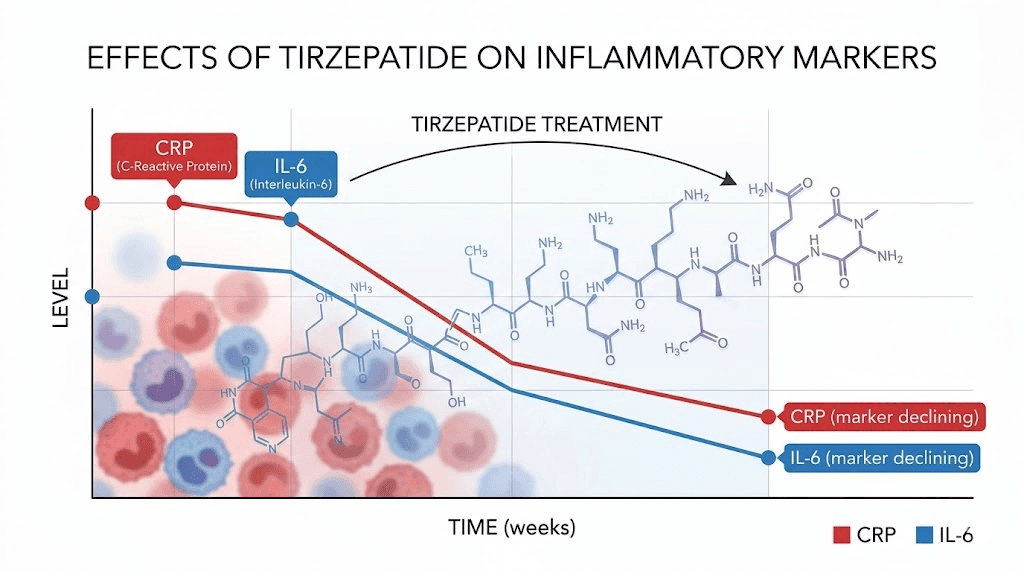
Researchers studying tirzepatide for weight loss started noticing inflammatory markers dropping, and not just because participants were losing weight. The dual GLP-1/GIP receptor agonist appeared to be doing something to inflammation itself, something beyond what weight reduction alone could explain. A systematic review and meta-analysis published in Reviews in Endocrine and Metabolic Disorders confirmed it: tirzepatide reduced high-sensitivity C-reactive protein (hsCRP) by a mean difference of -32.9 and interleukin-6 (IL-6) by -17.8 compared to placebo across multiple clinical trials. Those are not small numbers.
Now a growing community of practitioners and researchers is asking a different question entirely. What if you do not need the full weight-loss dose? What if microdosing tirzepatide, using carefully calibrated lower doses, could target inflammation specifically without the dramatic weight changes and gastrointestinal side effects that higher doses bring? This guide examines everything the research shows about microdosing tirzepatide for inflammation, from the hard data on inflammatory biomarkers to the practical protocols practitioners are exploring, the mechanisms that make it work, and the honest limitations you need to understand before considering this approach.
How tirzepatide fights inflammation at the molecular level
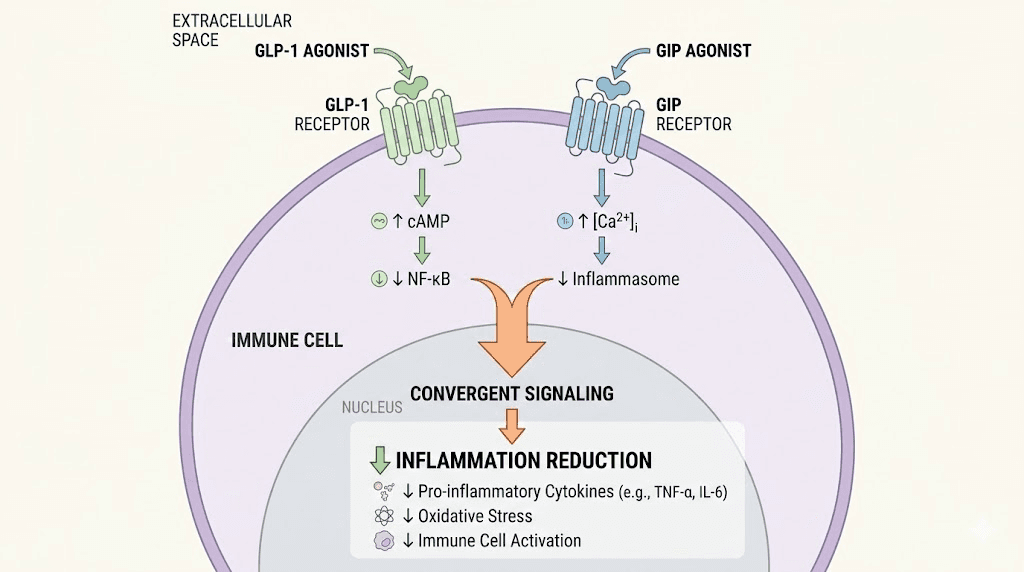
Understanding why tirzepatide reduces inflammation requires looking at its unique dual-agonist mechanism. Unlike semaglutide and other pure GLP-1 receptor agonists, tirzepatide activates both the GLP-1 and GIP receptors simultaneously. Both of these receptors exist on immune cells, not just metabolic tissues, and that distinction matters enormously for inflammation.
The GLP-1 pathway and immune modulation
GLP-1 receptors are expressed on monocytes, macrophages, and lymphocytes throughout the body. When tirzepatide binds to these receptors, it triggers a cascade that goes far beyond blood sugar regulation. Research published in the Journal of Clinical Investigation demonstrates that GLP-1-based therapies inhibit nuclear factor kappa B (NF-kB) signaling, the master switch that turns on inflammatory gene expression in virtually every tissue. This means less production of pro-inflammatory cytokines like tumor necrosis factor-alpha (TNF-alpha), interleukin-1-beta (IL-1-beta), and interleukin-6 (IL-6).
In animal studies, GLP-1 analogs reduced macrophage infiltration in blood vessels. Fewer macrophages means less inflammatory signaling. Less inflammatory signaling means less tissue damage. The effects ripple outward from the molecular level to the systemic level, influencing everything from vascular health to joint integrity.
The GIP pathway adds a second anti-inflammatory layer
Here is where tirzepatide separates itself from semaglutide and other GLP-1-only drugs. The glucose-dependent insulinotropic polypeptide (GIP) receptor carries its own anti-inflammatory properties. GIP receptor signaling reduces oxidative stress in endothelial cells, decreases inflammatory cytokine secretion, and improves blood flow while mitigating the infiltration of pro-inflammatory immune cells into tissues.
Research on tirzepatide specifically shows that it promotes the apoptosis (programmed death) of M1-type macrophages, the inflammatory phenotype, while reducing their inflammatory factor secretion by inhibiting ERK phosphorylation. At the same time, it restores the PI3K/Akt/GSK3-beta pathway. Why does that matter? Because sustained Akt activation facilitates the transition from inflammation to tissue repair while simultaneously reducing pro-inflammatory cytokines. Essentially, tirzepatide helps flip the switch from destruction to healing at the cellular level.
The combination of both GLP-1 and GIP receptor activation may produce greater anti-inflammatory effects than either hormone alone. This dual mechanism is what makes tirzepatide particularly interesting for inflammation research, and why some practitioners believe lower doses might still deliver meaningful anti-inflammatory benefits even without the aggressive dose escalation used for weight management.
Weight-dependent versus weight-independent inflammation reduction
A critical finding from the SURMOUNT trial post-hoc analyses clarifies something important. Early changes in hsCRP at week 24 were weight reduction-unassociated. Read that again. The inflammation dropped before the weight loss fully kicked in. This suggests tirzepatide has direct anti-inflammatory effects that operate independently of its metabolic benefits, at least in the initial weeks of treatment.
At week 72, the picture shifted. Changes in IL-6 and hsCRP were mainly weight reduction-associated, with 78% of IL-6 changes and 57% of hsCRP changes linked to body weight reduction in SURMOUNT-2 participants. This dual timeline, early weight-independent effects followed by longer-term weight-dependent effects, has major implications for microdosing strategies. If you are using lower doses that produce minimal weight change, you may still capture some of the direct anti-inflammatory benefits while foregoing the weight-mediated inflammation reduction that comes with higher doses.
What the clinical data actually says about tirzepatide and inflammation
Anecdotes are easy to find. Hard data is what matters. Here is what the research actually shows.
The meta-analysis numbers
The 2025 systematic review and meta-analysis analyzed 7 randomized clinical trials and 1 observational study (6 eligible for meta-analysis) examining tirzepatide and inflammatory markers. The results were consistent across populations and treatment regimens.
hsCRP reduction by dose:
5 mg tirzepatide: Mean difference of -20.3 compared to placebo (95% CI: -35.2 to -5.3)
10 mg tirzepatide: Mean difference of -33.9 compared to placebo (95% CI: -50.3 to -17.6)
15 mg tirzepatide: Mean difference of -32.9 compared to placebo (95% CI: -33.6 to -32.2)
Notice something? Even the lowest studied dose of 5 mg produced statistically significant CRP reduction. The jump from 5 mg to 10 mg was substantial, but the difference between 10 mg and 15 mg was minimal for hsCRP. This dose-response curve matters for anyone considering whether lower doses can still deliver anti-inflammatory benefits.
IL-6 reduction by dose:
5 mg tirzepatide: Mean difference of -18.8 (95% CI: -32.9 to -4.6)
10 mg tirzepatide: Mean difference of -17.9 (95% CI: -28.2 to -7.7)
15 mg tirzepatide: Mean difference of -16.8 (95% CI: -31.1 to -2.6)
The IL-6 data tells an even more interesting story. The reduction was remarkably similar across all three dose levels. The 5 mg dose actually showed the largest numerical reduction in IL-6, though the confidence intervals overlap enough that dose-dependent differences were not statistically significant. For inflammation, more is not necessarily better.
SURMOUNT trial inflammatory biomarker data
The SURMOUNT program, the largest clinical trial program for tirzepatide in obesity, provided additional inflammatory biomarker data through post-hoc analysis. A total of 700 participants were randomly selected from SURMOUNT-1 and SURMOUNT-2 (100 participants from each treatment arm). The findings confirmed that tirzepatide reduces inflammation in people with overweight or obesity regardless of whether they also had type 2 diabetes.
The relative contribution of weight reduction-dependent effects was more prominent in participants without type 2 diabetes compared to those with it. This suggests that in metabolically healthier individuals, a larger proportion of the anti-inflammatory benefit comes through weight loss rather than direct drug effects. For metabolic health applications, this distinction helps frame realistic expectations.
SURPASS trial cardiovascular biomarker improvements
Post-hoc analyses from the SURPASS clinical trial program (focused on type 2 diabetes) also demonstrated reductions in hsCRP levels. A study published examining cardiovascular risk biomarkers showed improvements across multiple inflammatory and cardiovascular markers, reinforcing the consistent anti-inflammatory signal observed across tirzepatide research.
The SUMMIT trial, studying tirzepatide in heart failure with preserved ejection fraction, found additional CRP reductions, extending the evidence base beyond obesity and diabetes into cardiovascular disease specifically.
What microdosing tirzepatide means and who is doing it
Microdosing GLP-1 medications has become one of the most discussed trends in metabolic medicine. But the term itself remains frustratingly vague.
Defining the microdose
There is no universal definition of what constitutes a microdose of tirzepatide. The FDA-approved starting dose is 2.5 mg weekly, intended as a 4-week initiation dose before escalating to 5 mg. Standard therapeutic doses range from 5 mg to 15 mg weekly for weight management and diabetes control.
In practice, microdosing generally refers to one of three approaches:
Sub-therapeutic dosing: Using doses below 2.5 mg weekly (often 0.5 mg to 1.5 mg)
Extended low-dose maintenance: Staying at the 2.5 mg initiation dose indefinitely rather than escalating
Split dosing: Dividing a standard dose into smaller, more frequent administrations (such as splitting a weekly dose into twice-weekly injections)
Each approach produces different pharmacokinetic profiles and potentially different anti-inflammatory effects. No clinical trials have specifically studied tirzepatide at sub-2.5 mg doses for any indication, which means the evidence base for true microdosing relies heavily on extrapolation from standard-dose research and practitioner observations.
Who is using microdose tirzepatide for inflammation
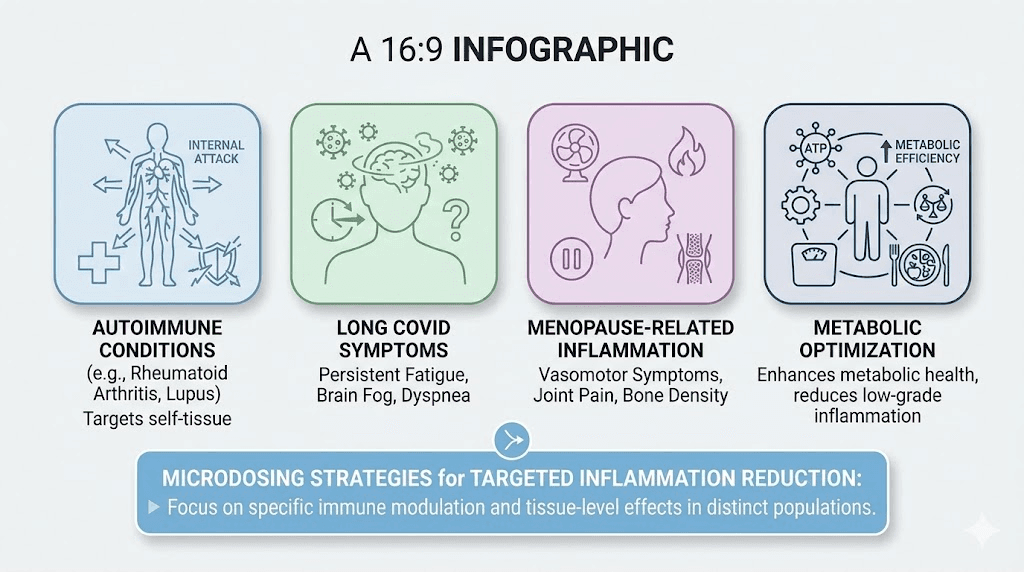
Several distinct groups have gravitated toward microdosing tirzepatide specifically for inflammation rather than weight loss.
Autoimmune patients. People with conditions like rheumatoid arthritis, lupus, Hashimoto thyroiditis, and inflammatory bowel disease have reported improvements in inflammation markers and symptoms at lower doses. The appeal is clear: reduce systemic inflammation without the significant weight loss that already-thin autoimmune patients cannot afford. Our guide on microdosing GLP-1 for autoimmune conditions covers this population in detail.
Long COVID sufferers. The Long COVID Treatment Trial-Tirzepatide (LoCITT-T) at Scripps Research is actively investigating whether tirzepatide can reduce neuroinflammation and systemic inflammation in long COVID patients. Some patients with ME/CFS, fibromyalgia, and long COVID have reported significant improvements in cognitive function and fatigue at lower doses, though responses vary considerably.
Perimenopause and menopause patients. Women experiencing the inflammatory surge that accompanies hormonal transitions are another growing population. The metabolic inflammation associated with declining estrogen, including increased visceral adiposity and elevated inflammatory markers, appears responsive to GLP-1/GIP therapy even at lower doses.
Metabolic health optimization seekers. Normal-weight individuals with elevated inflammatory markers, sometimes called "metabolically unhealthy normal weight," represent a population that does not need weight loss but may benefit from the direct anti-inflammatory effects. This group is perhaps the most natural fit for microdosing since they specifically need the anti-inflammatory effects without substantial body composition changes.
Microdosing protocols practitioners are exploring
Important disclaimer: these protocols are based on practitioner observations and extrapolation from clinical trial data. No randomized controlled trials have specifically evaluated tirzepatide microdosing for inflammation. Every protocol should involve medical supervision and regular monitoring of inflammatory biomarkers.
Protocol 1: Conservative sub-therapeutic approach
Goal: Target inflammation with minimal metabolic impact
Starting dose: 0.5 to 1.0 mg tirzepatide weekly
Escalation: Increase by 0.25 to 0.5 mg every 4 to 6 weeks based on inflammatory marker response
Target maintenance: The lowest dose that produces measurable CRP/IL-6 reduction
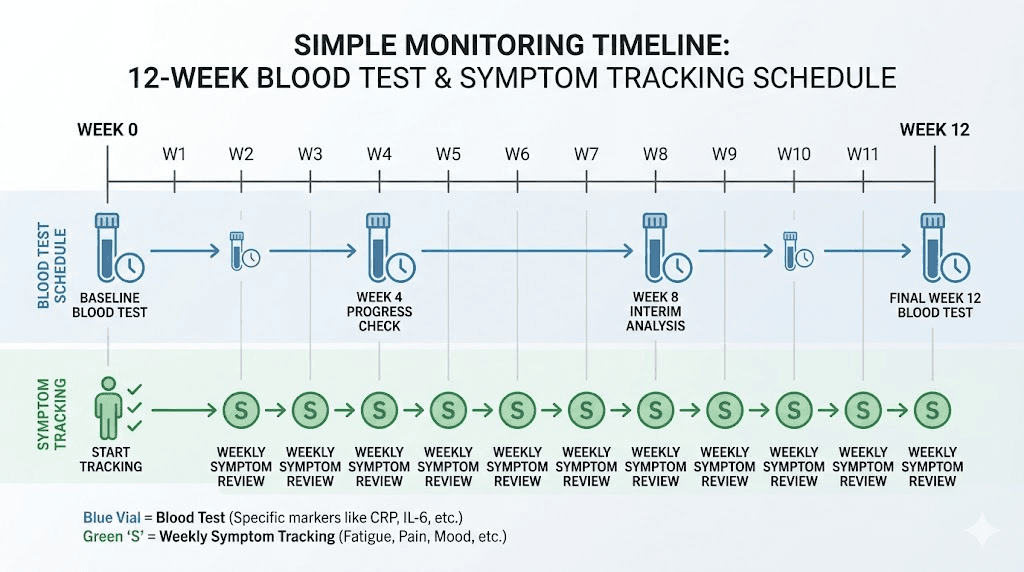
Monitoring schedule:
Baseline: hsCRP, IL-6, complete metabolic panel, body weight
Week 4: hsCRP, symptom assessment
Week 8: hsCRP, IL-6, body weight
Every 8 weeks thereafter: full inflammatory panel
Duration: Minimum 12 weeks to assess response, with ongoing reassessment
Best for: Normal-weight individuals with elevated inflammatory markers, autoimmune patients who cannot afford weight loss
This protocol uses the compounded tirzepatide dosage calculator approach since standard vials are not designed for sub-2.5 mg dosing. Compounding pharmacies can prepare lower-concentration formulations that allow more precise low-dose administration.
Protocol 2: Extended initiation dose maintenance
Goal: Capture anti-inflammatory benefits at the lowest FDA-studied dose
Dose: 2.5 mg tirzepatide weekly, maintained indefinitely without escalation
Rationale: The meta-analysis showed even 5 mg produced significant CRP and IL-6 reduction. While 2.5 mg was not analyzed separately (it is an initiation dose in trials), it represents the lowest standard dose with established safety data.
Monitoring schedule:
Baseline: hsCRP, IL-6, fasting insulin, HbA1c, body weight
Week 4: hsCRP, symptom assessment, body weight
Week 12: Full panel including IL-6
Every 12 weeks thereafter
Duration: Ongoing with quarterly reassessment
Best for: Individuals who want some anti-inflammatory benefit with an evidence-supported dose, patients who experience gastrointestinal side effects at higher doses
Protocol 3: Split-dose inflammatory targeting
Goal: Maintain steadier drug levels for more consistent anti-inflammatory coverage
Dose: 1.25 mg twice weekly (total 2.5 mg weekly) or 0.8 mg three times weekly (total approximately 2.5 mg weekly)
Rationale: Splitting the dose creates more consistent plasma levels throughout the week, potentially providing steadier anti-inflammatory signaling rather than the peak-and-trough pattern of once-weekly dosing. Some practitioners report this approach reduces GI side effects while maintaining efficacy.
Monitoring: Same as Protocol 2
Best for: Patients who experience end-of-week symptom return, those sensitive to peak-dose side effects
What to monitor and when
Regardless of which protocol is used, tracking the right biomarkers separates informed practice from guesswork.
Primary inflammatory markers:
hsCRP: The most accessible and well-studied marker. Target reduction of 20% or more from baseline within 8 to 12 weeks.
IL-6: More specific to the inflammatory pathways tirzepatide targets. Less commonly tested but more informative for this specific application.
Secondary markers worth tracking:
TNF-alpha: Key inflammatory cytokine, especially relevant for joint inflammation
Fasting insulin: Insulin resistance drives inflammation; tracking this helps assess metabolic inflammation separately
ESR (erythrocyte sedimentation rate): Broader inflammation marker useful as a cross-reference
Fibrinogen: Acute-phase protein that responds to inflammatory changes
Symptom tracking:
Joint stiffness (morning duration, severity 1 to 10)
Fatigue levels (daily 1 to 10 scale)
Ring tightness or peripheral swelling
Cognitive clarity (especially relevant for long COVID patients)
Pain levels at specific sites
The combination of objective biomarker data and subjective symptom tracking gives the most complete picture of whether microdosing is producing meaningful anti-inflammatory effects.
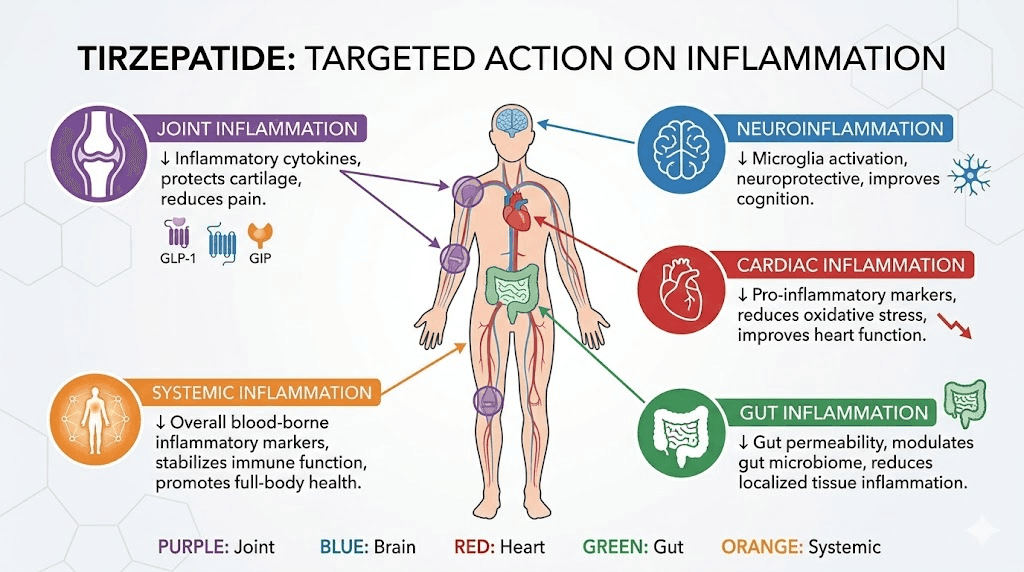
Tirzepatide for specific inflammatory conditions
The anti-inflammatory potential of tirzepatide, even at lower doses, has drawn interest across a range of conditions where chronic inflammation drives disease progression.
Joint inflammation and osteoarthritis
Research shows that people taking tirzepatide had a significantly lower risk of osteoarthritis compared to those using other medications, even after accounting for age and overall weight loss. The mechanism likely involves both direct anti-inflammatory effects on joint tissue and indirect benefits from reduced mechanical load and improved metabolic health.
For musculoskeletal symptoms, the anti-inflammatory pathway through macrophage modulation is particularly relevant. Inflammatory macrophages in synovial tissue drive much of the cartilage destruction in osteoarthritis. Tirzepatide promotes M1 macrophage apoptosis, potentially slowing this destructive process. At lower doses, patients report reduced morning stiffness and joint swelling without the muscle loss concerns that can accompany rapid weight reduction at higher doses.
Some researchers are exploring tirzepatide for lipedema, a condition involving painful fat deposits with significant inflammatory and fibrotic components. A narrative review published in the International Journal of Molecular Sciences discussed tirzepatide as a potential disease-modifying therapy, bridging metabolism, inflammation, and fibrosis in lipedema management.
Neuroinflammation and cognitive function
The brain has GLP-1 receptors. This is not a peripheral-only story.
GLP-1 receptor activation in the central nervous system modulates microglial activity, the brain immune cells responsible for neuroinflammation. When microglia become chronically activated, they produce inflammatory mediators that damage neurons and impair cognitive function. This process underlies symptoms in conditions ranging from long COVID brain fog to Alzheimer disease progression.
The Long COVID Treatment Trial (LoCITT-T) at Scripps Research specifically targets this mechanism with tirzepatide, hypothesizing that reducing neuroinflammation could alleviate the cognitive and fatigue symptoms that define long COVID for many patients. While results are pending, the biological rationale is compelling. Some patients using tirzepatide for energy and cognitive benefits report improvements at doses lower than those used for weight management, though this remains anecdotal.
Cardiovascular inflammation
Inflammation in blood vessel walls drives atherosclerosis, the buildup of plaques that causes heart attacks and strokes. The SUMMIT trial demonstrated that tirzepatide reduced CRP levels in patients with heart failure with preserved ejection fraction, a condition where cardiac inflammation plays a central role.
The cardiovascular benefits extend beyond CRP reduction. Tirzepatide decreases oxidative stress in endothelial cells through the GIP receptor pathway, reduces inflammatory macrophage infiltration in blood vessels, and improves endothelial function. These effects have led researchers to investigate tirzepatide as having vasculoprotective and anti-atherosclerotic potential beyond its metabolic effects, as outlined in a review published in the International Journal of Molecular Sciences.
For microdosing specifically, the cardiovascular inflammation data is encouraging because the early (week 24) CRP reduction in SURMOUNT trials was weight-independent. This suggests that even doses insufficient for meaningful weight loss might still provide some cardiovascular inflammatory protection.
Gut inflammation
A review in the Journal of Crohn and Colitis examined GLP-1 receptor agonists in inflammatory bowel disease (IBD), identifying several mechanisms by which these drugs reduce gut inflammation. GLP-1 receptors are expressed throughout the intestinal tract, and their activation reduces local inflammatory cytokine production while supporting gut barrier integrity.
For individuals dealing with gastrointestinal symptoms from tirzepatide, this creates an interesting paradox. The drug may help gut inflammation while simultaneously causing GI side effects through its motility-slowing properties. Microdosing potentially resolves this paradox by providing enough receptor activation to reduce gut inflammation while minimizing the motility effects that cause nausea, constipation, and other digestive complications.
Acute pancreatitis inflammation
Animal model research published in Animal Models and Experimental Medicine assessed tirzepatide for modulating inflammatory responses and mitigating acute pancreatitis. While this is preclinical data and not directly applicable to human microdosing protocols, it demonstrates the breadth of inflammatory conditions where tirzepatide shows therapeutic potential and reinforces the drug direct anti-inflammatory mechanisms.
The honest limitations of microdosing for inflammation
The hype around microdosing GLP-1 medications has outpaced the evidence. Significantly. Anyone considering this approach needs to understand what we do not know, which is substantial.
No clinical trials on sub-therapeutic doses for inflammation
Zero randomized controlled trials have evaluated tirzepatide at doses below 2.5 mg for any indication. The meta-analysis data on CRP and IL-6 reduction comes from standard doses (5 mg, 10 mg, 15 mg). Extrapolating those findings to 0.5 mg or 1.0 mg doses involves significant assumptions that have not been validated.
A STAT News investigation highlighted that microdosing aims to extend the lifespan of the GLP-1 compounding market, with physicians and researchers noting there is no clinical evidence that tirzepatide is effective at very small doses. The drugs are not proven to help patients with many of the symptoms being marketed around microdosing.
The dose-response question is unanswered
While the meta-analysis showed that 5 mg produced significant CRP reduction, we do not know where the minimum effective anti-inflammatory dose falls. It could be 2.5 mg. It could be lower. Or the dose-response curve could have a threshold below which anti-inflammatory effects are negligible. Without dedicated research, we are guessing.
The IL-6 data showing similar reductions across 5 mg, 10 mg, and 15 mg suggests the dose-response curve for some inflammatory markers may plateau early. This is genuinely encouraging for lower-dose approaches. But "the curve plateaus between 5 mg and 15 mg" does not tell us anything definitive about what happens below 5 mg.
Individual variation is enormous
Not everyone responds the same way. Some patients on standard doses show dramatic CRP drops while others show minimal change. The factors driving this variation, genetics, baseline inflammation levels, gut microbiome composition, concurrent medications, sleep quality, dietary patterns, all complicate predictions about microdose efficacy.
This is why monitoring is absolutely essential for anyone trying microdosing for inflammation. Without objective biomarker data, you cannot distinguish a genuine anti-inflammatory response from placebo effect, natural symptom fluctuation, or lifestyle changes made alongside the medication.
Direct effects versus weight-mediated effects
The SURMOUNT data showed that at week 72, the majority of inflammatory marker improvement was weight-reduction-associated. This is important. If you are microdosing specifically to avoid weight loss, you may be foregoing the larger component of tirzepatide anti-inflammatory effects. You might capture the direct, weight-independent portion, the early effect seen at week 24, but miss the more substantial weight-dependent portion.
For someone with significant excess weight driving their inflammation, standard dosing that produces weight loss may actually be more anti-inflammatory than microdosing. The right approach depends entirely on the individual clinical picture.
It is not a replacement for disease-specific treatments
GLP-1 and GLP-1/GIP medications are not disease-modifying for autoimmune disorders. If someone has rheumatoid arthritis, lupus, or inflammatory bowel disease, tirzepatide, at any dose, does not replace methotrexate, biologics, or other targeted immunotherapies. It may serve as an adjunctive approach to reduce overall inflammatory burden, but positioning it as a primary treatment for serious inflammatory conditions is irresponsible.
The same applies to cardiovascular inflammation. Statins, blood pressure medications, and lifestyle interventions remain the evidence-based foundation. Tirzepatide anti-inflammatory effects are a potential bonus, not a substitute.
How microdosing tirzepatide compares to other anti-inflammatory approaches
Putting microdose tirzepatide in context requires comparing it to established anti-inflammatory strategies. No approach exists in a vacuum.
Approach | Mechanism | CRP reduction | Evidence level | Side effects | Best for |
|---|---|---|---|---|---|
Microdose tirzepatide | Dual GLP-1/GIP receptor, NF-kB inhibition | Extrapolated from 5 mg data (~20% reduction) | Low (no direct trials) | Mild GI, minimal weight change | Metabolic inflammation, adjunctive use |
Standard dose tirzepatide | Same + weight reduction | ~33% reduction (meta-analysis) | High (RCTs) | Significant GI effects, substantial weight loss | Obesity-driven inflammation |
Standard dose semaglutide | GLP-1 receptor only | ~20-30% reduction | High (RCTs) | GI effects, weight loss | Weight + inflammation |
NSAIDs | COX inhibition | Variable | High | GI bleeding, cardiovascular risk | Acute inflammation, pain |
Low-dose methotrexate | Immune suppression | Significant | High | Liver toxicity, immune suppression | Autoimmune inflammation |
Mediterranean diet | Multiple anti-inflammatory pathways | ~20-40% over months | High | None | Everyone |
Regular exercise | Anti-inflammatory myokines | ~20-30% | High | Injury risk if excessive | Everyone |
The table reveals a nuanced picture. Microdose tirzepatide sits in a unique position: potentially more targeted than lifestyle approaches alone, with fewer side effects than pharmaceutical anti-inflammatories, but with a significantly weaker evidence base. For supplementary inflammation management, it may fill a gap. As a primary anti-inflammatory strategy, the evidence simply is not there yet.
Combining microdose tirzepatide with other approaches
The most rational approach to inflammation rarely involves a single intervention. Practitioners exploring microdose tirzepatide for inflammation typically combine it with:
Dietary optimization. An anti-inflammatory diet rich in omega-3 fatty acids, polyphenols, and fiber amplifies tirzepatide effects. The tirzepatide diet plan principles apply even at microdoses, emphasizing whole foods that support the anti-inflammatory cascade rather than working against it. Certain foods that worsen inflammation should be minimized regardless of medication status.
Targeted supplementation. Omega-3 fish oil (2 to 4 grams daily), curcumin (500 to 1000 mg with piperine), and vitamin D (maintaining levels above 40 ng/mL) all have independent anti-inflammatory evidence. Key supplements to pair with tirzepatide can potentially enhance the anti-inflammatory effects beyond what either approach achieves alone.
B vitamin optimization. Tirzepatide with B12 and methylcobalamin formulations address a practical concern: GLP-1 medications can affect B vitamin absorption. B12 deficiency itself drives inflammation, so maintaining optimal levels supports both the anti-inflammatory goal and overall metabolic health. Some compounded formulations include glycine and B12 alongside tirzepatide.
Glycine supplementation. Glycine combined with tirzepatide has gained attention because glycine itself has anti-inflammatory properties. The amino acid inhibits NF-kB activation through a different pathway than tirzepatide, potentially creating complementary anti-inflammatory effects. Glycine formulations with tirzepatide are available through some compounding pharmacies.
Regular movement. Exercise produces anti-inflammatory myokines that work synergistically with GLP-1/GIP signaling. Even moderate activity, 150 minutes per week of walking, produces measurable CRP reduction. For patients who struggle with exercise, the anti-inflammatory benefits of microdosing may provide enough symptom relief to make physical activity more accessible.
Sleep optimization. Poor sleep drives inflammation through elevated cortisol and disrupted immune function. Sleep disturbances on tirzepatide should be addressed aggressively when using the drug for inflammation, since poor sleep could negate the anti-inflammatory benefits.
Practical considerations for getting started
If you and your healthcare provider decide to explore microdosing tirzepatide for inflammation, several practical details determine success or failure.
Sourcing and preparation
Standard tirzepatide formulations (Mounjaro, Zepbound) come in pre-filled pens designed for specific dose increments. These are not easily adapted for sub-2.5 mg dosing. Most practitioners using microdose protocols work with compounded tirzepatide, which can be prepared at lower concentrations allowing more precise small-dose administration.
If using compounded tirzepatide vials, proper reconstitution technique becomes critical. The accuracy of your dose depends entirely on proper mixing. Use our compounded tirzepatide dosage calculator to determine exact drawing volumes based on your vial concentration. Bacteriostatic water ratios must be precise to ensure consistent dosing.
Injection technique at low doses
Smaller doses mean smaller injection volumes, which introduces precision challenges. When drawing 0.5 mg from a reconstituted vial, even small measurement errors represent a large percentage of the intended dose. Insulin syringes with half-unit markings provide the most precise measurement for microdose volumes.
Proper injection technique matters regardless of dose. Subcutaneous injection into the abdomen, thigh, or upper arm remains standard. Rotation of injection sites prevents lipodystrophy even at lower doses, since injection site reactions and localized inflammation can occur at any dose level.
Storage requirements
Proper refrigeration remains essential for maintaining potency. Compounded tirzepatide is typically less stable than commercial formulations, making storage even more critical. Keep reconstituted vials at 36 to 46 degrees Fahrenheit. Temperature excursions can degrade the peptide and reduce its effectiveness. If you are traveling with tirzepatide, insulated transport containers are non-negotiable.
Compounded tirzepatide shelf life is generally shorter than commercial products. Most compounded formulations should be used within 28 to 42 days of reconstitution, depending on the pharmacy specifications. Check your specific product expiration guidance and never use degraded product, as peptide degradation products could potentially trigger immune responses that work against your anti-inflammatory goals.
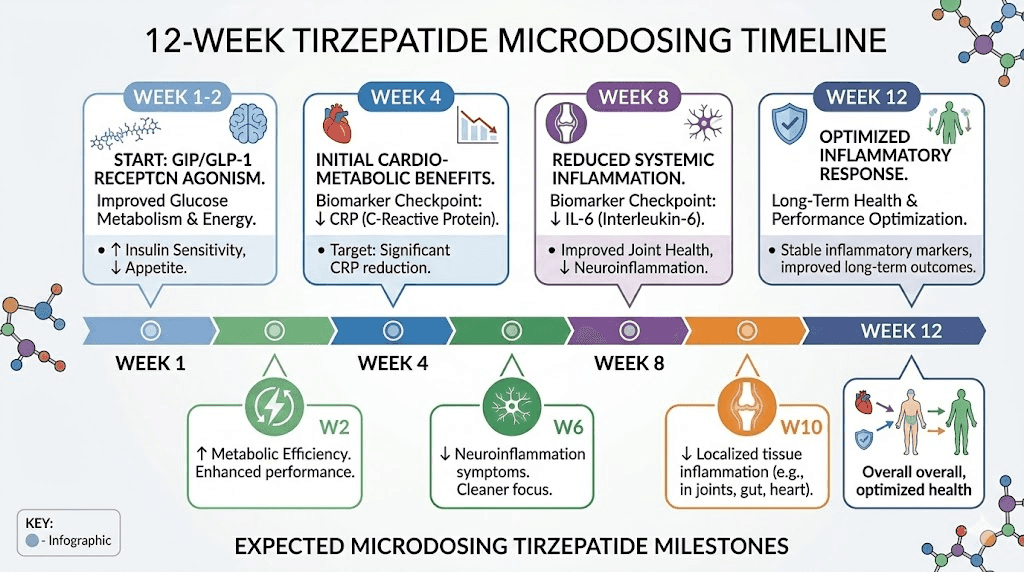
Timeline for seeing results
Do not expect overnight transformation. Based on the clinical trial data and practitioner observations:
Week 1 to 2: Most patients notice no significant changes. The drug is reaching steady state.
Week 2 to 4: Some patients report subtle improvements in joint stiffness, ring fit, and energy. These are subjective and may reflect placebo effect. First blood draw for comparison.
Week 4 to 8: If direct anti-inflammatory effects are going to manifest, hsCRP should show measurable change by this point. This aligns with the weight-independent CRP reduction seen at week 24 in SURMOUNT trials (accounting for the faster pharmacokinetic response at steady-state dosing).
Week 8 to 12: Clearer picture of response. IL-6 should be retested. Symptom patterns stabilize enough to distinguish drug effects from natural variation.
Beyond week 12: Long-term assessment. If no measurable improvement in inflammatory markers by week 12, the microdose approach may not be producing meaningful effects for that individual.
Patience is essential, but so is objectivity. If your CRP and IL-6 have not budged after 12 weeks of microdosing, continuing the same approach without dose adjustment is not evidence-based, it is wishful thinking.
Side effects at microdoses versus standard doses
One of the primary appeals of microdosing is the theoretically better side effect profile. The logic is straightforward: lower doses should produce fewer and milder side effects while potentially retaining some therapeutic benefit.
Gastrointestinal effects
The most common side effects of standard-dose tirzepatide, nausea, vomiting, diarrhea, and constipation, are dose-dependent. Clinical trials show clear increases in GI side effect incidence as doses escalate from 5 mg to 15 mg. At microdoses below 2.5 mg, practitioners report significantly lower rates of GI complaints, with many patients experiencing no gastrointestinal effects whatsoever.
However, "significantly lower" does not mean zero. Some individuals are highly sensitive to GLP-1/GIP receptor activation and may experience nausea or changes in bowel habits even at very low doses. GI symptoms that persist beyond the first 2 to 3 weeks at a given microdose may require dose reduction or discontinuation.
Fatigue and energy effects
Standard doses of tirzepatide can cause fatigue, particularly during dose escalation phases. At microdoses, fatigue is less commonly reported. Paradoxically, some patients report improved energy at low doses, possibly due to reduced inflammatory burden rather than a direct stimulatory effect. Our guide on tirzepatide and energy explores this complex relationship.
Headaches
Headaches are another dose-dependent side effect. Tirzepatide-related headaches occur more frequently at higher doses and during escalation. At stable microdoses, headache incidence appears minimal based on practitioner reports.
Mood and cognitive effects
Anxiety on tirzepatide has been reported at standard doses. At microdoses, mood effects are less documented. Some patients report improved mood, possibly related to reduced neuroinflammation, while others note no psychological effects. Sleep disruption should be monitored since it can independently worsen both mood and inflammation.
Weight change
By definition, microdosing aims to minimize weight change. Most practitioners report that patients on sub-2.5 mg doses lose little to no weight, which is the specific goal for this population. However, some individuals are highly responsive even to small doses and may experience unwanted weight reduction. Regular weight monitoring helps catch this early. For those who do lose weight, understanding weight maintenance strategies becomes important if they eventually discontinue.
Who should NOT microdose tirzepatide for inflammation
Microdosing tirzepatide is not appropriate for everyone, even among those with chronic inflammation.
People with active pancreatitis or history of severe pancreatitis. GLP-1/GIP medications carry pancreatic safety concerns at any dose. While animal research suggests potential pancreatitis benefits, clinical reality demands caution.
People with medullary thyroid carcinoma risk factors. GLP-1 receptor agonists carry a boxed warning regarding thyroid C-cell tumors. This applies regardless of dose.
Pregnant or planning pregnancy. Tirzepatide is not approved for use during pregnancy. Unplanned pregnancy on tirzepatide requires immediate discontinuation and medical consultation. Breastfeeding mothers should also avoid tirzepatide.
People who need aggressive inflammation control now. If you have an active autoimmune flare requiring immediate intervention, microdose tirzepatide is too slow and too uncertain. Use proven immunomodulatory therapies first, then consider tirzepatide as adjunctive therapy once the flare is controlled.
People with severe gastroparesis. Even microdoses slow gastric emptying to some degree. Patients with existing gastroparesis risk worsening their condition.
People who cannot afford regular monitoring. Without blood work to track inflammatory markers, you have no way to know if microdosing is working. Subjective symptom improvement alone is not sufficient given the strong potential for placebo effects in this context.
The future of tirzepatide for inflammation research
The scientific landscape around GLP-1/GIP therapy and inflammation is evolving rapidly. Several developments will shape the evidence base in coming years.
The LoCITT-T trial. The Scripps Research long COVID trial using tirzepatide will provide the first controlled data on tirzepatide for a condition defined primarily by inflammation rather than metabolic dysfunction. Results will help clarify whether the anti-inflammatory effects are clinically meaningful as a primary therapeutic target.
Expanded inflammatory bowel disease research. The published review in Journal of Crohn and Colitis has opened the door for clinical trials examining GLP-1 receptor agonists specifically in IBD populations. If positive, this would provide the first direct evidence for using these drugs to treat inflammatory conditions.
Oral formulations. Oral tirzepatide and orally disintegrating tablet formulations may make lower-dose administration easier and more precise than injectable approaches. Tirzepatide drops and tablet formulations could particularly benefit the microdosing population by eliminating injection-related barriers.
Dual and triple agonists. Retatrutide, a triple GLP-1/GIP/glucagon agonist, and other next-generation compounds may offer even more potent anti-inflammatory profiles. The mazdutide versus tirzepatide and survodutide versus tirzepatide comparisons will reveal whether different receptor activation profiles produce different anti-inflammatory outcomes.
Dedicated microdosing trials. The gap in evidence is obvious enough that research groups are beginning to design trials specifically examining low-dose GLP-1/GIP therapy for non-weight-loss indications. Until those results arrive, the evidence base remains limited.
For researchers serious about staying informed on peptide inflammation research and optimizing their protocols, SeekPeptides provides evidence-based guides, detailed protocols, and a community of experienced researchers who navigate exactly these questions. The SeekPeptides platform tracks emerging research and translates complex findings into practical guidance, particularly valuable when the science is moving as fast as it is in this space.
Building your anti-inflammatory protocol around microdose tirzepatide
For those who have discussed microdosing with their healthcare provider and decided to proceed, building a comprehensive anti-inflammatory protocol, not just relying on the medication alone, maximizes the probability of meaningful results.
Step 1: Establish your baseline
Before the first dose, get comprehensive blood work. At minimum: hsCRP, IL-6 (if your lab offers it), fasting insulin, HbA1c, complete metabolic panel, thyroid function, vitamin D level, and a complete blood count. This baseline lets you objectively assess any changes.
Document your symptoms. Rate joint stiffness, fatigue, cognitive clarity, pain levels, and overall wellbeing on a 1 to 10 scale. Do this daily for at least one week before starting to establish your natural variation pattern.
Step 2: Start low and track aggressively
Begin with the lowest practical dose your provider recommends. Track symptoms daily. Get blood work at 4 weeks. Resist the urge to increase the dose before you have objective data on whether the initial dose is working.
Use the peptide calculator to ensure dosing accuracy. Small errors at microdose levels represent large percentage deviations from your intended dose.
Step 3: Layer in complementary approaches
During the first 4 weeks, optimize the factors that independently reduce inflammation:
Clean up your diet following anti-inflammatory eating principles
Start or maintain regular physical activity
Optimize sleep hygiene (7 to 9 hours, consistent schedule)
Add evidence-based anti-inflammatory supplements (our supplement guide covers what pairs well with GLP-1 therapy)
Address stress management, cortisol drives inflammation directly
Step 4: Assess and adjust at week 8
Repeat full inflammatory panel. Compare to baseline. If hsCRP has dropped 20% or more, the protocol is showing objective results. If IL-6 has decreased, even better, that is more specific to the pathways tirzepatide targets.
If markers have not changed, discuss with your provider whether to:
Increase the dose modestly (0.25 to 0.5 mg increment)
Add a split-dosing approach for steadier levels
Focus on optimizing complementary factors before further dose changes
Consider whether standard dosing might be more appropriate for your situation
Step 5: Long-term monitoring and adjustment
If the protocol is working, continue with quarterly blood work and ongoing symptom tracking. Inflammation is dynamic, and what works initially may need adjustment as your body adapts or as life circumstances change.
Practitioners who work with SeekPeptides members frequently report that the combination of objective monitoring, evidence-based protocols, and community support produces better outcomes than any single intervention alone. SeekPeptides members access detailed protocol builders, biomarker tracking tools, and expert guidance specifically designed for researchers navigating complex applications like inflammation management.
Frequently asked questions
Does microdosing tirzepatide actually reduce inflammation or is it just weight loss?
Both, but the mechanisms operate on different timelines. Post-hoc analysis from the SURMOUNT trials showed that early CRP reduction (around week 24) was not associated with weight loss, suggesting direct anti-inflammatory effects. However, at week 72, the majority of inflammatory marker improvements were weight-dependent (78% of IL-6 changes, 57% of CRP changes). Microdosing may capture the direct effects while minimizing weight change, but this has not been confirmed in dedicated trials.
What is the lowest effective dose of tirzepatide for inflammation?
Nobody knows yet. The lowest dose studied in clinical trials for inflammatory markers was 5 mg, which produced significant CRP reduction (mean difference -20.3 versus placebo) and IL-6 reduction (mean difference -18.8). The 2.5 mg initiation dose was not analyzed separately in the meta-analysis. Sub-2.5 mg doses have no published data for any outcome, including inflammation. Practitioners using lower doses are working from extrapolation, not evidence.
How long does it take for microdose tirzepatide to reduce inflammation?
Based on clinical trial data at standard doses, measurable CRP changes occurred by week 24 (about 6 months). At steady-state microdosing, some practitioners report seeing hsCRP changes within 4 to 8 weeks. However, this timeline is based on observations, not controlled studies. Plan for at least 8 to 12 weeks before concluding whether microdosing is producing anti-inflammatory effects for you. Use our tirzepatide timeline guide for additional context on response timeframes.
Can I microdose tirzepatide alongside my autoimmune medications?
Potentially, but this absolutely requires medical supervision. Tirzepatide does not replace disease-modifying therapies for autoimmune conditions. It may serve as an adjunctive approach. Drug interactions with common autoimmune medications (methotrexate, biologics, JAK inhibitors) have not been specifically studied, though no major pharmacokinetic interactions are expected based on tirzepatide metabolism pathway. Always inform all treating physicians about all medications you are taking. See our complete guide to microdosing GLP-1 for autoimmune conditions.
Is microdosing tirzepatide better than microdosing semaglutide for inflammation?
The theoretical advantage of tirzepatide is its dual GLP-1/GIP receptor activation. Since both GIP and GLP-1 have independent anti-inflammatory properties, the combination may produce greater inflammation reduction than GLP-1-only drugs like semaglutide. The meta-analysis data supports this, showing robust inflammatory marker reductions. However, no head-to-head comparison of microdose tirzepatide versus microdose semaglutide for inflammation has been conducted. The semaglutide versus tirzepatide comparison page covers broader differences between these two medications.
Will microdosing tirzepatide help with long COVID inflammation?
The Scripps Research LoCITT-T trial is investigating exactly this question. Preliminary anecdotal reports from patients and providers suggest some improvement in fatigue, cognitive function, and pain in long COVID patients using GLP-1/GIP medications. The proposed mechanism involves reducing neuroinflammation through central GLP-1 receptor activation. However, controlled trial data is not yet available, and individual responses vary significantly.
What blood tests should I get before starting microdose tirzepatide for inflammation?
At minimum: hsCRP (most accessible inflammatory marker), fasting insulin, HbA1c, complete metabolic panel, and body weight. Ideally also: IL-6, TNF-alpha, ESR, vitamin D, and thyroid function. These establish your inflammatory baseline and help rule out other conditions driving elevated markers. Repeat testing at 4 weeks (hsCRP minimum), 8 to 12 weeks (full panel), and quarterly thereafter.
Can I use niacinamide or glycine alongside microdose tirzepatide for enhanced anti-inflammatory effects?
Both compounds have independent anti-inflammatory evidence. Niacinamide (vitamin B3) inhibits inflammatory pathways and is commonly combined with tirzepatide in compounded formulations. Glycine inhibits NF-kB through a different mechanism than tirzepatide, potentially creating complementary effects. Glycine-B12-tirzepatide compounds are available through select compounding pharmacies. No controlled studies have evaluated these specific combinations for inflammation outcomes.
External resources
Anti-inflammatory effects of tirzepatide: a systematic review and meta-analysis (PubMed)
LoCITT-T: Long COVID Treatment Trial with Tirzepatide (Scripps Research)
Glucagon-like peptide-1: a multi-faceted anti-inflammatory agent (PMC)
For researchers committed to evidence-based peptide protocols, SeekPeptides offers the most comprehensive resource available. Members access detailed anti-inflammatory protocol guides, biomarker tracking frameworks, and a community of thousands who have navigated these exact decisions with data rather than guesswork.
In case I do not see you, good afternoon, good evening, and good night. May your inflammation markers stay low, your protocols stay evidence-based, and your recovery stay steady.