Feb 12, 2026

What if the reason your semaglutide results are inconsistent has nothing to do with the peptide itself? What if the problem started before you ever drew your first dose, right there at the mixing stage, with a reconstitution error so subtle you never even noticed it?
That is the reality for a surprising number of researchers. They follow dosing charts to the letter. They store their vials at the right temperature. They inject on schedule. And yet, results vary wildly from week to week. The concentration is off. The math does not add up. The solution looks cloudy or foamy. And all of it traces back to one moment: the moment bacteriostatic water met lyophilized powder.
Reconstituting semaglutide is not difficult. But it demands precision. The difference between a perfectly mixed vial and a compromised one comes down to details that most guides either skip entirely or bury under walls of generic advice. How fast you inject the water matters. Where you aim the stream matters. How you mix afterward matters. Even the type of water you choose changes how long your solution remains viable, from 24 hours to 28 days.
This guide covers every vial size you will encounter, from 2mg to 10mg, with exact water volumes, concentration charts, unit conversions, and step-by-step instructions built for researchers who want accuracy on the first attempt. Whether you are reconstituting semaglutide for the first time or refining a protocol you have used for months, the information here will eliminate guesswork and protect your investment. SeekPeptides members already know how critical this step is, and now you will too.
What reconstitution actually means and why it matters
Semaglutide arrives as a lyophilized powder. That is a freeze-dried form designed for stability during shipping and long-term storage. In this state, the peptide can survive months or even years at controlled temperatures without degrading. But you cannot inject powder. You need to dissolve it in a sterile liquid to create an injectable solution, and that process is called reconstitution.
Simple enough concept. The execution is where things get tricky.
When you add bacteriostatic water to a vial of semaglutide, you are creating a solution with a specific concentration measured in milligrams per milliliter. That concentration determines everything downstream: how many units you draw for each dose, how many doses you get per vial, and ultimately whether your protocol delivers the results the research suggests it should. Get the concentration wrong, and every single dose that follows will be wrong too.
This is not like mixing a protein shake where close enough works fine. A 10% error in reconstitution volume creates a 10% error in every dose. Over weeks of a protocol, that compounds into significant under-dosing or over-dosing, either of which undermines your research.
Lyophilized powder versus pre-mixed solutions
Some semaglutide products arrive pre-mixed in liquid form from compounding pharmacies. These do not require reconstitution. The pharmacy has already dissolved the powder, set the concentration, and packaged it ready to use. If your vial already contains a clear liquid, skip the reconstitution steps entirely and move straight to the dosage chart section.
Lyophilized vials contain a white or off-white powder cake or loose powder at the bottom of the vial. Sometimes the powder appears as a solid disc. Other times it looks like fine particles clinging to the glass. Both are normal. What is not normal is discoloration. If the powder looks yellow, brown, or has visible specks of foreign material, do not use it.
Why the type of water you use changes everything
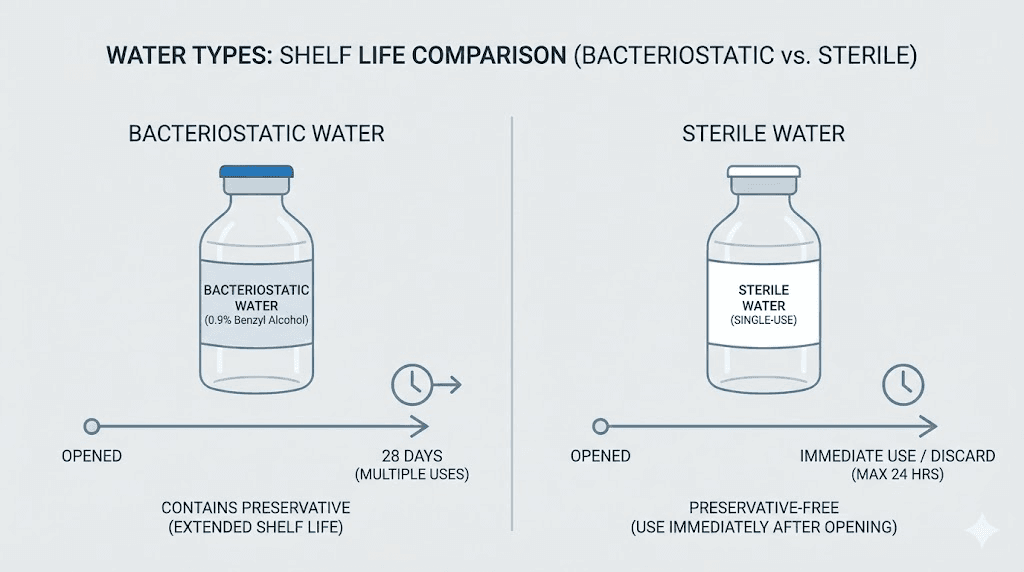
You have two options for reconstitution: bacteriostatic water and sterile water for injection. This is not a preference. It is a critical decision that determines how long your reconstituted semaglutide remains usable.
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative. This preservative prevents bacterial growth each time you puncture the vial stopper with a needle. With bacteriostatic water, your reconstituted semaglutide remains viable for up to 28 days when refrigerated properly. For multi-dose protocols, this is the only practical choice.
Sterile water for injection contains no preservative. It is pure water, nothing else. Without that antimicrobial agent, bacteria can colonize the solution after the first needle puncture. Sterile water reconstitutions should be used within 24 hours. Period. If your protocol involves drawing multiple doses over days or weeks, sterile water is not an option.
For virtually every semaglutide protocol, bacteriostatic water is the correct choice. The peptide reconstitution calculator on our site assumes bacteriostatic water as the default, and for good reason.
Supplies you need before starting
Gather everything before you begin. Stopping mid-reconstitution to find a missing syringe or alcohol swab creates contamination risk and breaks your sterile workflow. Here is what you need, nothing more and nothing less.
Essential supplies checklist
Semaglutide lyophilized vial. Common sizes include 2mg, 3mg, 5mg, and 10mg. Check the label carefully. The vial size determines how much bacteriostatic water you add and what concentration you create. Mixing up a 5mg vial procedure on a 10mg vial doubles your concentration and every dose with it.
Bacteriostatic water. A 30mL vial is standard and provides enough water for multiple reconstitutions. Make sure it has not expired. Check that the rubber stopper shows no signs of repeated punctures beyond what you have personally done. If you are buying bacteriostatic water, look for USP-grade from reputable suppliers. Learn more about how much bacteriostatic water to add to peptides across different compounds.
Insulin syringes. Use 1mL (100 unit) insulin syringes with 29-31 gauge needles. These fine-gauge needles minimize damage to the vial stopper and reduce the risk of coring, where a needle punches through and deposits a tiny rubber fragment into your solution. Some researchers prefer 27 gauge for drawing from bacteriostatic water vials and 29-31 gauge for the semaglutide vial itself.
Alcohol swabs. 70% isopropyl alcohol prep pads. You will use these to sterilize vial tops before every needle puncture. Not optional. Not sometimes. Every single time.
A clean, flat work surface. Wipe it down with disinfectant spray or alcohol before you begin. A clean kitchen counter works. A dusty desk does not.
Good lighting. You need to see inside the vial clearly to confirm the powder has fully dissolved and no particles remain.
Supplies to avoid
Do not use regular tap water or distilled water from the grocery store. These are not sterile. Do not use saline solution unless specifically directed by a protocol. Do not use syringes larger than 1mL for the actual injection, as the larger barrel makes precise measurements difficult. And do not reuse syringes. Ever. A fresh syringe for every vial puncture is the standard.
Step-by-step reconstitution process
This process applies to every vial size. The only variable is how much bacteriostatic water you add, which we cover in the vial-specific sections below. Master these steps once and they apply universally, not just to semaglutide but to tirzepatide reconstitution, GHK-Cu reconstitution, and essentially any lyophilized peptide you work with.
Step 1: wash your hands thoroughly
Soap and water. Twenty seconds minimum. Scrub between fingers, under nails, around wrists. Dry with a clean paper towel, not a cloth towel that has been hanging in the bathroom collecting bacteria all week. This sounds basic because it is basic, and yet contamination from unwashed hands remains the most common source of infection in home reconstitution.
Step 2: prepare your workspace
Wipe your work surface with 70% isopropyl alcohol or a disinfectant spray. Let it air dry completely. Lay out all your supplies: semaglutide vial, bacteriostatic water vial, insulin syringe (still in its sterile packaging), and alcohol swabs. Having everything within reach means you will not need to leave your sterile workspace mid-process.
Step 3: inspect the semaglutide vial
Look at the powder. Is it white or off-white? Good. Is it a compact cake or loose powder? Both are fine. Is there any discoloration, moisture, or foreign particles? If yes, do not use it. Check the expiration date on the label. Confirm the dosage printed matches what you intended to reconstitute. A 5mg vial and a 10mg vial can look identical from the outside.
Step 4: clean both vial tops
Take an alcohol swab and firmly wipe the rubber stopper on your semaglutide vial. Use a circular motion, starting from the center and moving outward. Let it air dry for 10 seconds. Repeat with the bacteriostatic water vial. Do not blow on them to speed drying. Your breath carries bacteria.
Step 5: draw bacteriostatic water into the syringe
Remove the syringe from its sterile packaging. Pull the plunger back to draw air into the syringe equal to the volume of water you plan to withdraw. Insert the needle into the bacteriostatic water vial through the center of the rubber stopper. Push the air in. This equalizes pressure inside the vial, making it easier to draw liquid. Then invert the vial and slowly pull the plunger to draw the exact amount of bacteriostatic water you need.
Check for air bubbles. If you see any, tap the syringe barrel gently with your finger to move bubbles toward the needle end, then push the plunger slightly to expel them back into the vial. Re-draw to the correct volume. Precision here matters. If your target is 2mL and you draw 2.1mL, your entire concentration calculation shifts.
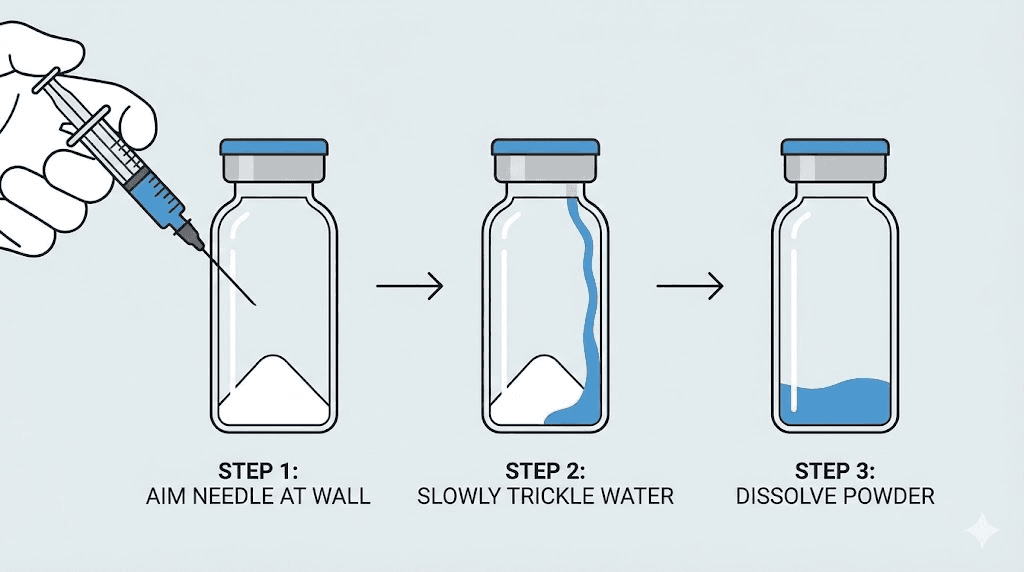
Step 6: inject water into the semaglutide vial (the critical step)
This is where most reconstitution errors happen. Insert the needle into the semaglutide vial through the rubber stopper. Now, here is what separates a good reconstitution from a bad one.
Aim the needle at the glass wall of the vial, not directly at the powder.
Push the plunger slowly. Let the water trickle down the inside wall of the vial. The entire injection should take 15 to 20 seconds for volumes under 2mL, and 30 seconds or more for larger volumes. You are not trying to blast the powder apart. You are gently introducing water so it flows over and around the lyophilized cake without creating foam or turbulence.
Why does this matter? Direct force on the powder can denature the peptide. Semaglutide is a protein. Proteins can unfold and lose their biological activity when subjected to mechanical stress. Foam means air has been incorporated into the solution, which introduces oxidation and makes accurate volume measurements impossible until the foam settles. And aggressive mixing breaks molecular bonds that you cannot repair.
Slow and gentle. Wall of the vial. Every time.
Step 7: mix gently
After all the water is in the vial, do not shake it. Shaking creates foam and mechanical stress on the peptide. Instead, use one of these gentle mixing techniques.
Swirl method: Hold the vial between your thumb and fingers and rotate your wrist in a slow circular motion. Imagine you are swirling wine in a glass. Continue for 30 to 60 seconds.
Roll method: Place the vial between your palms and gently roll it back and forth. This is particularly effective for stubborn powder that does not dissolve easily with swirling alone.
Most semaglutide powder dissolves quickly, often within 30 to 60 seconds of gentle swirling. If small particles remain, set the vial down for 5 minutes and try again. Do not add more water. Do not shake harder. Patience beats force every time.
Step 8: inspect the solution
Hold the vial up to a light source. The solution should be perfectly clear and colorless. No cloudiness. No visible particles. No foam (a few tiny bubbles on the surface are acceptable and will dissipate). If the solution appears cloudy or you can see undissolved particles after several minutes of gentle mixing, something went wrong. Do not inject a cloudy solution.
If the solution is clear, your reconstitution is complete. Label the vial with today's date, the concentration (we will calculate this next), and the expiration date (28 days from today if using bacteriostatic water).
Reconstitution charts by vial size
Here is where generic guides fail. They give you one ratio and assume every vial is the same. But the amount of bacteriostatic water you add depends on three factors: the amount of semaglutide in the vial, the concentration you want to achieve, and the syringe markings you will use for dosing. Different concentrations work better for different dose ranges and syringe types.
The following charts cover the most common vial sizes with the most practical concentration options. For vial-specific deep dives, check our dedicated guides for 5mg semaglutide reconstitution and 10mg vial dosage charts.
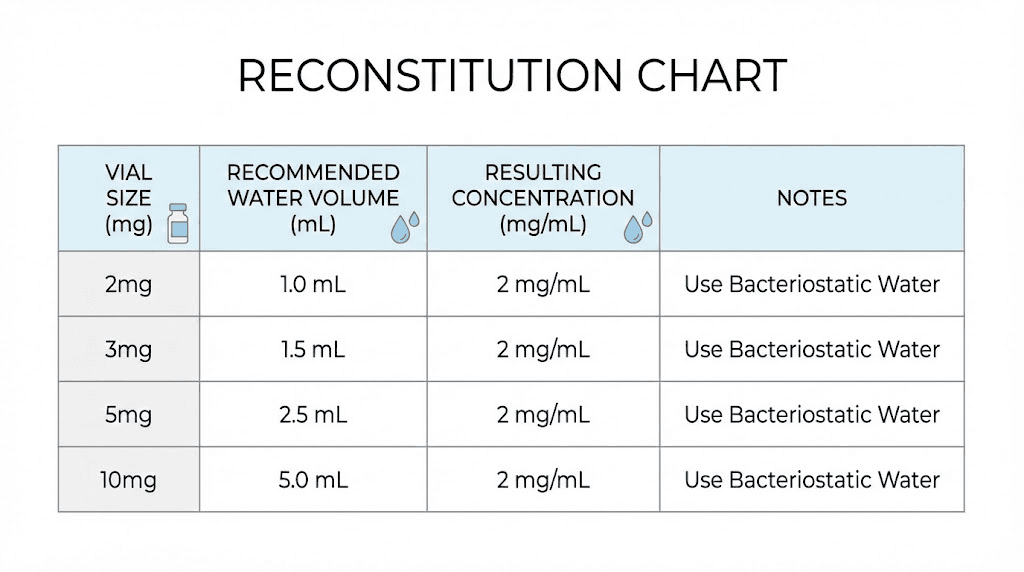
2mg semaglutide vial
The 2mg vial is the smallest commonly available size. It is ideal for researchers starting at low doses or those who want to minimize waste during short protocols.
BAC Water Added | Concentration | 0.25mg Dose | 0.5mg Dose | Total Doses at 0.25mg |
|---|---|---|---|---|
0.5mL | 4mg/mL | 6.25 units | 12.5 units | 8 doses |
0.8mL | 2.5mg/mL | 10 units | 20 units | 8 doses |
1.0mL | 2mg/mL | 12.5 units | 25 units | 8 doses |
2.0mL | 1mg/mL | 25 units | 50 units | 8 doses |
For a 2mg vial, adding 0.8mL of bacteriostatic water creates a 2.5mg/mL concentration. This puts the common 0.25mg starting dose at exactly 10 units on an insulin syringe, which is the easiest measurement to draw accurately. If you are comfortable with smaller volume measurements, 0.5mL of water creates a more concentrated solution that preserves syringe volume for larger future doses.
3mg semaglutide vial
The 3mg vial offers a middle ground. Not too concentrated, not too dilute, with enough peptide for a meaningful protocol run.
BAC Water Added | Concentration | 0.25mg Dose | 0.5mg Dose | 1mg Dose |
|---|---|---|---|---|
1.0mL | 3mg/mL | 8.3 units | 16.7 units | 33.3 units |
1.2mL | 2.5mg/mL | 10 units | 20 units | 40 units |
1.5mL | 2mg/mL | 12.5 units | 25 units | 50 units |
3.0mL | 1mg/mL | 25 units | 50 units | 100 units |
The sweet spot for 3mg vials is 1.2mL of bacteriostatic water. This gives you a clean 2.5mg/mL concentration where standard doses land on easy-to-read syringe markings. Our semaglutide dosage calculator can verify these numbers for any custom concentration you prefer.
5mg semaglutide vial
The 5mg vial is the most popular size. It provides enough peptide for a complete escalation protocol from the typical 0.25mg starting dose through maintenance doses of 1mg or higher. Here is our complete 5mg semaglutide mixing guide for even more detail.
BAC Water Added | Concentration | 0.25mg Dose | 0.5mg Dose | 1mg Dose | 2.5mg Dose |
|---|---|---|---|---|---|
1.0mL | 5mg/mL | 5 units | 10 units | 20 units | 50 units |
2.0mL | 2.5mg/mL | 10 units | 20 units | 40 units | 100 units |
2.5mL | 2mg/mL | 12.5 units | 25 units | 50 units | N/A (over 100u) |
5.0mL | 1mg/mL | 25 units | 50 units | 100 units | N/A (over 100u) |
Two concentrations stand out for 5mg vials. Adding 2mL creates a 2.5mg/mL solution where the 0.25mg starting dose equals exactly 10 units and the maximum common dose of 2.5mg fills the entire 100-unit syringe. Clean numbers across the board. Alternatively, 1mL creates a 5mg/mL concentration that works well if you plan to stay at lower doses and want maximum doses per vial. The 5mg dosage chart and 5mg/mL dosage chart break down unit conversions for every common dose at these concentrations.
10mg semaglutide vial
The 10mg vial is the most cost-effective option for longer protocols or higher maintenance doses. It requires the most bacteriostatic water, which means the most precision during reconstitution. See our dedicated 10mg semaglutide mixing guide and 10mg dosage chart for comprehensive breakdowns.
BAC Water Added | Concentration | 0.25mg Dose | 0.5mg Dose | 1mg Dose | 2.5mg Dose |
|---|---|---|---|---|---|
2.0mL | 5mg/mL | 5 units | 10 units | 20 units | 50 units |
3.0mL | 3.33mg/mL | 7.5 units | 15 units | 30 units | 75 units |
4.0mL | 2.5mg/mL | 10 units | 20 units | 40 units | 100 units |
5.0mL | 2mg/mL | 12.5 units | 25 units | 50 units | N/A |
For 10mg vials, the 4mL option creates that same clean 2.5mg/mL concentration with easy unit markings. However, 3mL is popular among experienced researchers because the 3.33mg/mL concentration provides a good balance between dose precision and total injectable volume. The 10mg mixing chart provides visual references for these concentrations.
The concentration formula explained
If you want to calculate any concentration yourself rather than relying on pre-made charts, here is the math. It is simple division.
Concentration (mg/mL) = Total semaglutide (mg) / Total bacteriostatic water added (mL)
So for a 5mg vial with 2mL of water: 5 / 2 = 2.5mg/mL.
For a 10mg vial with 3mL of water: 10 / 3 = 3.33mg/mL.
Once you know the concentration, calculating your dose in syringe units is equally straightforward.
Dose in units = (Desired dose in mg / Concentration in mg/mL) x 100
Want 0.5mg from a 2.5mg/mL solution? That is (0.5 / 2.5) x 100 = 20 units.
Want 1mg from a 5mg/mL solution? That is (1 / 5) x 100 = 20 units.
This formula works universally. Write it down. Tape it to your refrigerator. Bookmark the peptide calculator page. Whatever keeps you from doing mental math when precision matters. For detailed semaglutide dosage conversions in units, check our complete conversion charts.
Why unit measurements exist
Insulin syringes are calibrated in units, not milliliters. One hundred units equals one milliliter. This is a legacy of insulin dosing that the peptide community has adopted because insulin syringes are the most precise, affordable, and widely available option for subcutaneous injections. When someone says they inject 20 units of semaglutide, they are injecting 0.2mL of whatever concentration their vial contains. The actual milligram dose depends entirely on the concentration, which is why getting your reconstitution right matters so much.
A 20-unit injection from a 2.5mg/mL vial delivers 0.5mg of semaglutide. The same 20-unit injection from a 5mg/mL vial delivers 1mg. Double the dose, identical syringe measurement. This is where dosing errors happen, and it is entirely preventable with correct reconstitution math. See our guides on how many mg is 40 units of semaglutide and how many mg is 50 units of semaglutide for more unit-to-milligram conversions.
Choosing the right concentration for your protocol
Not all concentrations are equally practical. The best concentration for your vial depends on your dosing range, how long you plan to use the vial, and your comfort with small volume measurements on an insulin syringe.
Low-dose protocols (0.25mg to 0.5mg)
If you are starting a semaglutide protocol at the standard 0.25mg weekly dose and plan to escalate slowly, a lower concentration like 1mg/mL or 2mg/mL works best. These put your dose at 25 units or 12.5 units respectively, both easy to read on a standard insulin syringe. The trade-off is that higher doses later in your protocol will require drawing larger volumes, potentially exceeding the 100-unit syringe capacity at doses above 1mg to 2.5mg depending on concentration.
Full-range protocols (0.25mg to 2.5mg)
For protocols that escalate through the full dose range, 2.5mg/mL is the goldilocks concentration. The starting 0.25mg dose is 10 units. The common maintenance dose of 1mg is 40 units. Even the maximum 2.5mg dose is exactly 100 units, filling the syringe but not exceeding it. Every standard dose lands on a clean, readable syringe marking. This is why 2.5mg/mL is the most recommended concentration across most semaglutide dosage charts.
High-dose or high-volume protocols
Researchers working with larger vials who want to minimize the number of reconstitutions might prefer 5mg/mL. This concentration means smaller injection volumes (5 units for 0.25mg), which some find more comfortable. But it demands extremely precise syringe measurements. The difference between 5 units and 6 units is a 20% dose difference. For experienced researchers comfortable with fine measurements, this works. For beginners, the margin of error is too tight. Understanding semaglutide dosing in units becomes critical at higher concentrations.
Common reconstitution mistakes and how to avoid them
After analyzing hundreds of forum posts, community discussions, and protocol reports, the same mistakes appear over and over. Each one is entirely preventable.
Mistake 1: spraying water directly onto the powder
This is the single most common error. Researchers insert the needle, point it straight at the powder cake, and push the plunger. The force of the water stream blasts apart the lyophilized cake, creating foam, potentially denaturing the peptide, and making it nearly impossible to determine if the powder fully dissolved or if you are just looking at foam.
Fix: Aim the needle at the inside wall of the vial. Push the plunger slowly. Let gravity do the work as water trickles down the glass and gently contacts the powder from below and around the sides.
Mistake 2: shaking the vial
Aggressive shaking introduces air into the solution, creates foam that traps peptide molecules, and generates mechanical forces that can denature proteins. Once a protein is denatured, it does not un-denature. That biological activity is gone permanently.
Fix: Swirl gently or roll between your palms. If the powder has not dissolved after 60 seconds of swirling, set it down for 5 minutes and try again. Some peptides take a few minutes to fully dissolve, and that is completely normal.
Mistake 3: using the wrong amount of water
This one cascades through every subsequent dose. Adding 3mL to a 5mg vial when you meant to add 2mL creates a 1.67mg/mL concentration instead of 2.5mg/mL. Your 10-unit dose now delivers 0.167mg instead of 0.25mg, a 33% underdose. Multiply that across weeks and your entire protocol is compromised.
Fix: Decide your target concentration before you start. Write it down. Calculate the exact water volume. Draw it carefully into the syringe. Double-check the syringe markings before inserting into the semaglutide vial. Once the water is in, there is no taking it back. Use the reconstitution calculator to double-check your math.
Mistake 4: using sterile water for multi-dose vials
Sterile water contains no preservative. Each needle puncture introduces a pathway for bacteria. By the second or third dose drawing, your solution may already be compromised. And you will not see, smell, or taste bacterial contamination until it is far too late.
Fix: Always use bacteriostatic water for any vial you plan to draw from more than once. The 0.9% benzyl alcohol preservative maintains sterility across multiple punctures for up to 28 days. Learn more about proper peptide storage techniques.
Mistake 5: not cleaning the vial stopper
The rubber stopper on your vial has been exposed to air, shipping environments, warehouse dust, and your hands. Inserting a needle without alcohol sterilization transfers whatever is on that stopper directly into your solution.
Fix: Alcohol swab. Every vial. Every time. Before every single needle insertion. This applies to both the semaglutide vial and the bacteriostatic water vial.
Mistake 6: confusing vial sizes
A 5mg vial and a 10mg vial of semaglutide can look nearly identical. The difference is printed on a small label that is easy to misread or overlook. Reconstituting a 10mg vial with the water volume intended for a 5mg vial doubles your concentration and every dose that follows.
Fix: Read the label twice before adding water. Confirm the milligram amount printed on the vial matches your calculation. If you work with multiple vial sizes, consider labeling them with colored tape or keeping them in separate storage locations. The semaglutide reconstitution chart provides a quick reference for each size.

Storage after reconstitution
You have reconstituted your semaglutide. The solution is clear. The label is applied. Now what? Storage determines whether your solution remains potent for the full 28 days or degrades within a week.
Temperature requirements
Reconstituted semaglutide must be refrigerated at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit). This is the standard refrigerator temperature range. Place the vial toward the back of the refrigerator, not in the door. Door compartments experience temperature fluctuations every time you open the fridge, and those fluctuations accelerate degradation.
Do not freeze reconstituted semaglutide. Freezing can cause the peptide to precipitate out of solution, forming particles that do not re-dissolve upon thawing. If your reconstituted vial accidentally freezes, discard it. For more on this topic, read our guide about whether compounded semaglutide needs refrigeration.
How long reconstituted semaglutide lasts
With bacteriostatic water and proper refrigeration, reconstituted semaglutide maintains its potency for up to 28 days. Some researchers report usable potency beyond 28 days, but the safety margin narrows significantly after that point. The 0.9% benzyl alcohol in bacteriostatic water is effective at preventing bacterial growth, but its preservative action does have limits.
With sterile water, discard within 24 hours regardless of refrigeration. No exceptions.
Mark the reconstitution date on the vial or a piece of tape attached to it. Set a calendar reminder for 28 days later. When in doubt, discard and reconstitute fresh. The cost of a new vial is trivial compared to the risk of injecting a contaminated or degraded solution. Our detailed guide on how long compounded semaglutide lasts in the fridge covers this topic in depth.
Light and vibration exposure
Semaglutide, like most peptides, degrades when exposed to light and vibration. Store your vial in a dark area of the refrigerator. Some researchers wrap vials in aluminum foil for extra light protection. Avoid storing near the compressor or any vibrating component of the refrigerator.
Transportation also matters. If you need to travel with reconstituted semaglutide, use an insulated cooler with ice packs. Keep the vial upright and cushioned. Minimize transit time. And never leave it in a hot car, even for a few minutes.
Room temperature windows
Brief room temperature exposure during dose preparation is fine. Taking the vial out of the fridge, drawing your dose, and returning it within 10 to 15 minutes will not meaningfully impact potency. But do not leave reconstituted semaglutide at room temperature for hours. After 30 minutes outside the fridge, return it immediately. Extended room temperature exposure accelerates degradation exponentially.
Drawing your dose from a reconstituted vial
Reconstitution is complete. Now you need to draw accurate doses from the vial. This is where your concentration math becomes practical, and where syringe skills matter.
Step-by-step dose drawing
1. Clean the vial stopper. Alcohol swab. Wait 10 seconds. Yes, again, even though you cleaned it during reconstitution.
2. Draw air into a fresh syringe. Pull the plunger to the unit marking that matches your dose. If your dose is 20 units, draw 20 units of air.
3. Inject air into the vial. Push the needle through the center of the stopper and inject the air. This equalizes pressure and makes drawing liquid much easier.
4. Invert the vial. Turn it upside down so the stopper faces the floor and the liquid covers the needle tip completely.
5. Slowly pull the plunger. Draw slightly more than your target dose. If you need 20 units, draw to 22 or 23.
6. Remove bubbles. While the needle is still in the vial, tap the syringe barrel to move any air bubbles toward the needle end. Push the plunger slowly to expel air and excess liquid back into the vial until you hit your exact target marking.
7. Remove the needle and cap immediately. If you are not injecting right away, recap the needle to maintain sterility. Use the peptide injection guide for proper injection technique.
Reading insulin syringe markings accurately
Standard 1mL insulin syringes have markings every 2 units, with numbers printed every 10 units (10, 20, 30, 40, 50, 60, 70, 80, 90, 100). The flat bottom of the plunger's rubber tip is your reference point. Not the top of the rubber. Not the side. The flat bottom, where it meets the barrel wall.
Hold the syringe at eye level with the markings facing you. Parallax (looking from above or below) creates reading errors that seem tiny on the syringe but translate to meaningful dose differences. A 2-unit error on a 10-unit dose is a 20% difference. Check the semaglutide syringe dosage chart for visual guides on reading markings correctly.
What to do about dead space
Every syringe has dead space, the small volume trapped in the needle hub that does not get injected. For insulin syringes, this is approximately 0.5 to 1 unit. Over the course of a vial, dead space losses can add up. A 5mg vial providing 20 doses loses approximately 10 to 20 units total to dead space, which is 0.1 to 0.2mL or roughly one extra dose worth of semaglutide.
Some researchers account for this by adding slightly more bacteriostatic water during reconstitution. Others simply accept the minor loss. If you are using low-dead-space syringes (designed for insulin), the loss is minimal and typically not worth worrying about.
Semaglutide reconstitution versus other peptides
If you work with multiple peptides, you may wonder whether semaglutide reconstitution differs from other compounds. The core technique is identical. Bacteriostatic water, aim at the wall, swirl gently, refrigerate. But there are a few semaglutide-specific considerations.
Semaglutide dissolves faster than most peptides
Compared to peptides like BPC-157 or tirzepatide, semaglutide lyophilized powder tends to dissolve more readily in bacteriostatic water. You should see full dissolution within 60 seconds of gentle swirling. If it takes significantly longer, the powder may have been exposed to moisture or temperature extremes before reconstitution.
Semaglutide is more fragile than some smaller peptides
Semaglutide is a larger molecule than peptides like BPC-157 or KPV. Its complex structure makes it more susceptible to denaturation from mechanical stress. This is why the slow-injection and no-shaking rules are especially important for semaglutide. Smaller peptides have some resilience against rough handling. Semaglutide does not. Similar fragility applies to tirzepatide, which shares structural complexity as a fellow GLP-1 receptor agonist.
Concentration ranges differ from other peptides
Many peptides use much lower concentrations than semaglutide. BPC-157, for example, is typically reconstituted at much higher dilutions because the doses are measured in micrograms rather than milligrams. Semaglutide concentrations between 1mg/mL and 5mg/mL are standard, which falls in the mid-range for peptide reconstitution. If you are used to reconstituting other peptides, double-check your math before applying the same water volumes. Our complete guide to mixing peptides with bacteriostatic water covers this comparison in detail.
Escalation protocols and how reconstitution supports them
Semaglutide protocols typically involve dose escalation. You start low and gradually increase over weeks or months. Understanding how your reconstitution concentration interacts with this escalation is critical for maintaining consistent, accurate dosing throughout.
Standard escalation timeline
The typical semaglutide escalation follows a pattern like this:
Weeks | Dose | Units at 2.5mg/mL | Units at 5mg/mL |
|---|---|---|---|
1-4 | 0.25mg | 10 units | 5 units |
5-8 | 0.5mg | 20 units | 10 units |
9-12 | 1.0mg | 40 units | 20 units |
13-16 | 1.75mg | 70 units | 35 units |
17+ | 2.5mg | 100 units | 50 units |
Notice how the 2.5mg/mL concentration provides clean, whole-number measurements at every escalation point. At 5mg/mL, the 0.25mg starting dose is only 5 units, which is difficult to measure precisely. This is why choosing the right concentration before you reconstitute affects your entire protocol, not just the first dose. For a complete overview of how fast semaglutide works, check our timeline guide. Understanding how long semaglutide takes to work helps set realistic expectations alongside proper reconstitution.
When to reconstitute a new vial during escalation
A 5mg vial at 2.5mg/mL (2mL of water) contains 200 total units of solution. Here is how many weekly doses you get at each escalation level:
0.25mg (10 units): 20 doses = 20 weeks
0.5mg (20 units): 10 doses = 10 weeks
1.0mg (40 units): 5 doses = 5 weeks
2.5mg (100 units): 2 doses = 2 weeks
But remember, reconstituted semaglutide only lasts 28 days. A 5mg vial at the 0.25mg starting dose provides 20 weeks of doses, but the solution expires after 4 weeks. You will only use 4 of those 20 potential doses before the vial expires.
This is why vial size planning matters. For the first 4 weeks at 0.25mg, a 2mg vial is sufficient and wastes less peptide than a 10mg vial. As doses increase, larger vials become more economical because you use them faster and waste less to expiration. Our peptide cost calculator can help you model the most cost-effective vial sizes for your specific escalation plan. For details on how long to stay on semaglutide, we have a dedicated guide.
Reconstituting compounded semaglutide specifically
Compounded semaglutide comes from compounding pharmacies and may differ from research-grade lyophilized powder in several ways. If you have received compounded semaglutide, here is what to know.
Pre-mixed versus lyophilized compounded products
Many compounding pharmacies ship semaglutide already reconstituted in liquid form. These vials contain a pre-mixed solution at a specified concentration (often 5mg/mL or 2.5mg/mL). If your compounded semaglutide arrives as a liquid, do not add bacteriostatic water. It is ready to use. Simply store it according to the pharmacy's instructions and draw your doses directly.
Some compounding pharmacies do ship lyophilized powder, especially for higher-concentration products or specialty formulations. These follow the same reconstitution process described above. The pharmacy should include specific reconstitution instructions, and those instructions supersede any general guide, including this one. The compounded semaglutide dose chart covers the most common pharmacy formulations.
Compounded semaglutide with B12 or other additives
Some compounded formulations include vitamin B12 (cyanocobalamin), L-carnitine, glycine, or other additives. These are already mixed into the powder or liquid by the pharmacy. Do not add your own supplements to the vial. Adding anything beyond bacteriostatic water changes the solution's pH, osmolality, and stability in unpredictable ways. For information on these combination products, check our guides on semaglutide with B12 and semaglutide with glycine.
Concentration differences from compounding pharmacies
Compounding pharmacies may use concentrations that differ from the standard research-grade options. A pharmacy might send a 10mg/2mL pre-mixed vial (5mg/mL) or a 12.5mg/2.5mL vial (also 5mg/mL) or any number of custom concentrations. Always check the label. Always verify the math. The dosing chart from one pharmacy does not apply to a different pharmacy's product if the concentrations differ. Reference the semaglutide dosage chart in mL for pharmacy-specific conversions.
Troubleshooting reconstitution problems
Even with careful technique, problems can arise. Here is how to handle the most common issues without panicking or wasting product.
The powder will not dissolve
If gentle swirling for 60 seconds leaves visible particles, do not add more water. Set the vial down on a flat surface and wait 5 to 10 minutes. Sometimes the lyophilized cake needs time to absorb water from the edges inward. After waiting, try swirling again.
If particles persist after 15 minutes, the powder may have been compromised before reconstitution. Exposure to moisture during storage, temperature excursions during shipping, or an expired product can all prevent proper dissolution. In these cases, do not use the product. Contact your supplier.
The solution is cloudy
A properly reconstituted semaglutide solution should be crystal clear and colorless. Cloudiness indicates either incomplete dissolution (wait and swirl more) or protein aggregation (the peptide has been damaged). If cloudiness persists after 15 minutes of gentle intermittent mixing, the vial is likely compromised. Do not inject cloudy solutions.
There is foam on top
A small amount of surface foam from gentle mixing is normal and will dissipate within a few minutes. Excessive foam, thick enough that you cannot see through it, suggests the water was injected too forcefully. Set the vial upright and wait 10 to 15 minutes for the foam to settle. Once it clears, check if the solution underneath is clear. If yes, the vial is likely still usable. If the solution under the foam is cloudy, the peptide may have been denatured by the force.
You added too much water
Once bacteriostatic water is in the vial, you cannot remove it without contamination risk. The solution is now at a lower concentration than intended. Your options: recalculate your dose based on the actual concentration, or discard and start fresh with a new vial.
For example, if you meant to add 2mL to a 5mg vial (2.5mg/mL) but accidentally added 3mL (1.67mg/mL), your 0.25mg dose is now 15 units instead of 10. Adjust your dosing chart accordingly. The solution itself is fine. Only the concentration changed.
You added too little water
Same principle, opposite direction. The concentration is higher than intended. You can add more bacteriostatic water to bring it to the correct volume, but you need to know exactly how much you originally added. If you are uncertain, your best option is to recalculate based on an estimated concentration and proceed cautiously, starting with lower unit amounts. Check the 5mg reconstitution chart or 10mg reconstitution chart for quick reference.
The rubber stopper keeps coring
Coring happens when the needle punches through the rubber stopper instead of cleanly piercing it, depositing a tiny rubber fragment into the solution. Using finer gauge needles (29-31 gauge), inserting at a 45-degree angle initially before going vertical, and rotating slightly during insertion all reduce coring risk. If you notice rubber fragments in your solution, do not use it.
Advanced tips for experienced researchers
Once you have the basics down, these refinements optimize your reconstitution workflow and improve consistency over time.
Pre-warm the bacteriostatic water slightly
Bacteriostatic water stored in the refrigerator is colder than room temperature, which can slow dissolution. Taking the BAC water out 10 to 15 minutes before reconstitution allows it to approach room temperature, which helps the semaglutide powder dissolve faster and more completely. Do not warm it beyond room temperature. Do not microwave it. Just let it sit on the counter briefly. For comprehensive bacteriostatic water mixing guidelines, see our detailed resource.
Use a consistent reconstitution schedule
If your protocol uses a new vial every 4 weeks, reconstitute on the same day each month. This creates a predictable rhythm where you always know how old your current vial is, reducing the risk of using expired reconstituted product. Some researchers reconstitute on the same day they take their weekly dose, making it part of their routine.
Keep a reconstitution log
Track every reconstitution: date, vial size, amount of BAC water added, calculated concentration, and discard date. This log protects against dosing errors, helps identify trends if you notice inconsistencies, and provides a reference if you need to verify a past reconstitution. SeekPeptides members use protocol tracking tools that make this logging automatic and accessible.
Consider syringe filtering for research applications
For researchers who want an additional safety layer, passing the reconstituted solution through a 0.22-micron syringe filter removes any particulate matter or potential contaminants. This step is not required for properly reconstituted semaglutide from reputable sources, but it adds an extra margin of safety. Syringe filters are inexpensive and widely available from laboratory supply companies.
Multi-vial reconstitution batching
If you have multiple vials to reconstitute, prepare all your supplies at once and work through them in sequence. Use a fresh syringe for each vial. Label each vial immediately after reconstitution, before moving to the next one. Working in batches reduces setup time while maintaining sterile technique. Just never lose track of which vial received how much water. Learn more about storing peptides after reconstitution to keep everything organized.
How reconstitution connects to your broader semaglutide protocol
Reconstitution is step one. What follows determines whether semaglutide delivers the results the research supports.
Injection site selection
After reconstitution, semaglutide is typically administered as a subcutaneous injection. The most common injection sites are the abdomen (at least 2 inches from the navel), the front of the thigh, and the upper outer arm. Rotate injection sites weekly to prevent lipodystrophy (changes in fat tissue under the skin). Our guide on where to inject GLP-1 peptides covers site selection and rotation strategies in detail. For step-by-step injection technique, see the peptide injection guide.
Timing and frequency
Standard semaglutide protocols call for once-weekly subcutaneous injection. Choose the same day each week for consistency. Morning or evening does not significantly impact efficacy based on available research, though some users report better appetite suppression when injecting in the morning. The key is consistency. Same day. Same approximate time. Every week.
Diet and lifestyle during a semaglutide protocol
Reconstitution and injection technique are important, but they exist within a broader protocol context. Semaglutide diet plans that emphasize protein intake, adequate hydration, and nutrient-dense foods support better outcomes than protocols focused solely on the peptide. Understanding semaglutide fatigue and managing side effects proactively also contributes to protocol success. And yes, researchers often ask about alcohol consumption during semaglutide use, which we address in a dedicated article.
What to do if semaglutide does not seem to be working
Before blaming the peptide, check your reconstitution. Was the concentration calculated correctly? Was the water volume accurate? Has the vial been stored properly? Is it within the 28-day window? Reconstitution errors are the most common and most easily fixed reason for inconsistent results. If reconstitution checks out, our guide on why semaglutide is not working for weight loss covers the other common causes.
Comparing semaglutide to tirzepatide reconstitution
Many researchers use both semaglutide and tirzepatide, or switch between them. The reconstitution process is essentially identical, but there are a few practical differences worth noting.
Factor | Semaglutide | Tirzepatide |
|---|---|---|
Dissolution speed | Fast (30-60 seconds) | Moderate (60-120 seconds) |
Common vial sizes | 2mg, 3mg, 5mg, 10mg | 5mg, 10mg, 15mg, 30mg |
Popular concentration | 2.5mg/mL | 2.5mg/mL or 5mg/mL |
Starting dose | 0.25mg weekly | 2.5mg weekly |
Shaking sensitivity | High | High |
Storage after reconstitution | 28 days refrigerated | 28 days refrigerated |
The biggest practical difference is dose range. Tirzepatide starts at 2.5mg weekly, ten times higher than semaglutide's 0.25mg starting dose. This means tirzepatide vials are generally larger, concentrations may be higher, and the unit volumes per dose are correspondingly different. Check our tirzepatide reconstitution chart for specific details. If you are considering a switch, our guide on semaglutide vs tirzepatide dosage charts maps equivalent doses between the two compounds.
Special considerations for specific semaglutide formulations
Semaglutide with B12
Compounded semaglutide with vitamin B12 (cyanocobalamin) is one of the most common combination formulations. The B12 is already incorporated into the lyophilized powder or pre-mixed solution. Reconstitute using the same technique as standard semaglutide. The solution may have a slight pink or reddish tint from the B12, which is completely normal. A pink tint is not discoloration. Our semaglutide B12 dosage chart covers dosing specifics for these combination products. More details in our compounded semaglutide with B12 dosage chart.
Oral semaglutide drops
The growing interest in oral semaglutide drops has created confusion about reconstitution. Oral formulations are designed for sublingual absorption and do not require reconstitution in the traditional sense. They arrive as ready-to-use liquid drops. If your product is specifically labeled for oral or sublingual use, follow the manufacturer's instructions rather than the injectable reconstitution process described here.
Higher concentration vials for experienced researchers
Some suppliers offer concentrated semaglutide vials of 15mg or 20mg. These require proportionally more bacteriostatic water but follow the same reconstitution process. The risk with larger vials is greater financial loss if reconstitution goes wrong, so these are best reserved for researchers who have already mastered the technique on standard 5mg or 10mg vials. The semaglutide mixing chart covers these larger vial sizes.
When to discard reconstituted semaglutide
Knowing when to throw away a reconstituted vial is as important as knowing how to mix one. Injecting degraded or contaminated semaglutide is worse than no semaglutide at all.
Mandatory discard situations
Discard immediately if you observe any of the following.
The solution has turned cloudy or hazy after being clear at reconstitution. This indicates protein aggregation or bacterial contamination.
You see visible particles, fibers, or floating material in the solution. Any particulate matter means the solution is compromised.
The solution has changed color. Semaglutide solutions should be clear and colorless (or slightly pink if B12 is added). Yellow, brown, or any other discoloration means degradation.
The vial was left at room temperature for more than 2 hours. Extended temperature excursions may compromise both the peptide and the preservative system.
It has been more than 28 days since reconstitution with bacteriostatic water, or more than 24 hours with sterile water.
You detect an unusual smell when opening or drawing from the vial. Normal reconstituted semaglutide has very little odor. A strong or unusual smell may indicate contamination.
The rubber stopper shows excessive puncture damage. After many draws, the stopper can develop channels that compromise the seal. Our guide on using expired semaglutide goes deeper on this topic.
Cost optimization through proper reconstitution
Semaglutide is not cheap. Proper reconstitution minimizes waste and maximizes the value of every vial.
Matching vial size to protocol phase
During the 0.25mg escalation phase (typically weeks 1-4), you only need 1mg of semaglutide total (0.25mg x 4 weeks). A 2mg vial is more than sufficient and wastes less to expiration than a 10mg vial would. As you escalate to higher doses, larger vials become more cost-effective because you consume them faster, often within the 28-day window.
Minimizing waste from expired reconstituted product
The 28-day expiration after reconstitution means you need to plan vial usage carefully. If your weekly dose is 0.25mg from a 10mg vial, you will only use 1mg of the 10mg before the reconstituted solution expires. That is 9mg wasted, potentially 90% of the vial.
Better approach: use a 2mg vial for the first 4 weeks (1mg used, 1mg wasted). Then switch to a 5mg vial for weeks 5-8 at 0.5mg weekly (2mg used, 3mg wasted). As doses increase, waste percentage drops. By the time you are at 2.5mg weekly using a 10mg vial, you use all 10mg in exactly 4 weeks with zero waste. The peptide cost calculator can model these scenarios for any protocol timeline.
Sharing vials (and why you should not)
Multi-dose vials technically contain enough semaglutide for multiple people at lower doses. But sharing introduces contamination risk from additional needle punctures, liability and legal issues, and dosing complications. Each person should use their own vials with their own syringes. Period.
Frequently asked questions
Can I use saline instead of bacteriostatic water to reconstitute semaglutide?
Normal saline (0.9% sodium chloride) can technically dissolve semaglutide, but it is not recommended for multi-dose vials. Like sterile water, standard saline solutions do not contain a preservative. Bacteriostatic sodium chloride does exist and contains a preservative, but bacteriostatic water remains the standard and most widely available option. Unless a specific protocol explicitly calls for saline, stick with bacteriostatic water.
How do I know if my semaglutide has been denatured during reconstitution?
Unfortunately, you cannot tell just by looking at the solution. Denatured semaglutide may appear perfectly clear and colorless. The most reliable indicator is reduced or absent effects at your expected dose. If your reconstituted semaglutide suddenly seems less effective despite proper dosing and storage, denaturation during reconstitution or storage is a possibility. This is why technique matters for every single vial.
Can I reconstitute semaglutide at a higher concentration and dilute it later?
Yes, but this adds a dilution step that introduces additional error potential. It is simpler and safer to reconstitute at your target concentration directly. The only scenario where reconstituting concentrated and diluting makes practical sense is when you need very low concentrations that would require adding more water than the vial can hold.
What happens if I accidentally inject air bubbles?
Small subcutaneous air bubbles are harmless but irritating. They do not cause air embolism (that risk applies only to intravenous injection of large air volumes). However, an air bubble in your syringe means the liquid volume is less than what the markings indicate. A 20-unit draw with a 3-unit air bubble only delivers approximately 17 units of actual semaglutide solution. Remove bubbles before injection for accurate dosing.
Should I warm the reconstituted semaglutide before injecting?
Some researchers find cold injections mildly uncomfortable and prefer to hold the drawn syringe between their fingers for 30 to 60 seconds before injecting. This slightly warms the solution and may reduce injection site discomfort. Do not warm the entire vial, only the drawn dose in the syringe, and do not exceed body temperature.
Can I reconstitute semaglutide and freeze it for later use?
Do not freeze reconstituted semaglutide. Freezing causes ice crystal formation that can damage the peptide structure and create aggregates that do not re-dissolve upon thawing. Freeze-thaw cycles are one of the most destructive things you can do to a reconstituted protein. Store lyophilized (unreconstituted) semaglutide in the freezer for long-term storage. Store reconstituted semaglutide in the refrigerator for short-term use only.
How many times can I puncture the vial stopper before it becomes contaminated?
With bacteriostatic water, the preservative helps maintain sterility across multiple punctures. Most researchers draw from a single vial 4 to 10 times over its 28-day life without issues. Using fine-gauge needles (29-31 gauge) and proper alcohol swabbing before each puncture minimizes contamination risk. If the stopper becomes visibly damaged or starts to core (releasing rubber fragments), stop using the vial regardless of how many days remain.
Is there a difference between reconstituting research-grade and pharmaceutical-grade semaglutide?
The reconstitution technique is identical. The difference lies in the manufacturing quality of the powder itself. Pharmaceutical-grade semaglutide undergoes stricter quality control, may dissolve more consistently, and comes with precise reconstitution instructions from the pharmacy. Research-grade products vary by supplier. Both require the same careful technique. Learn about these distinctions in our compounding pharmacy vs research peptide analysis.
External resources
For researchers serious about optimizing every aspect of their semaglutide protocol, from reconstitution through results tracking, SeekPeptides offers the most comprehensive resource available. Members access detailed protocols, concentration calculators, community support from experienced researchers, and evidence-based guidance that goes far beyond what any single article can cover.
In case I do not see you, good afternoon, good evening, and good night. May your reconstitutions stay sterile, your concentrations stay accurate, and your protocols stay consistent.