Mar 20, 2026

Tirzepatide and retatrutide look nearly identical on paper. Both are injectable peptides. Both target GLP-1 receptors. Both produce dramatic weight loss in clinical trials. And both come from the same pharmaceutical company, Eli Lilly.
But the similarities mask a fundamental difference that changes everything about how these compounds work inside the body. One activates two receptors. The other activates three. That single extra receptor, the glucagon receptor, creates a cascade of metabolic effects that separates these two peptides in ways most comparison guides completely ignore.
Researchers who understand this distinction make better decisions about protocols, dosing, and expectations. Those who do not often waste months on the wrong compound for their specific goals. The clinical data tells a clear story. Tirzepatide delivers proven, FDA-approved results with years of safety data behind it. Retatrutide shows potentially superior weight loss numbers but remains investigational, with Phase 3 trials still underway. Choosing between them requires understanding not just what each compound does, but how those mechanisms translate into real-world outcomes for different types of researchers.
This guide breaks down every meaningful difference between tirzepatide and retatrutide, from receptor pharmacology to clinical trial results, dosing protocols, side effect profiles, and practical availability. SeekPeptides has compiled the most comprehensive comparison available, drawing on Phase 2 and Phase 3 trial data, published research, and the practical experience of thousands of researchers navigating this exact decision.
How tirzepatide and retatrutide actually work
Understanding the difference between these two peptides starts at the receptor level. Every metabolic peptide works by binding to specific receptors on cell surfaces, triggering downstream effects that influence appetite, insulin release, fat metabolism, and energy expenditure. The receptors each compound targets determine its entire profile of benefits and limitations.
This is not a minor technical detail. It is the single most important factor separating tirzepatide from retatrutide.
The dual agonist approach: tirzepatide
Tirzepatide activates two receptors simultaneously. The first is the GLP-1 receptor, which most people recognize from semaglutide and other incretin-based therapies. GLP-1 receptor activation suppresses appetite, slows gastric emptying, and enhances insulin secretion in a glucose-dependent manner. These effects account for the appetite reduction and blood sugar improvements that tirzepatide users report within the first few weeks.
The second receptor is GIP, or glucose-dependent insulinotropic polypeptide. This is where tirzepatide diverges from older GLP-1 agonists like semaglutide. GIP receptor activation improves how the body processes and stores fat, enhances insulin sensitivity beyond what GLP-1 alone achieves, and appears to reduce the inflammatory signaling associated with excess adipose tissue.
The dual mechanism explains why tirzepatide produces greater weight loss than pure GLP-1 agonists. In the SURMOUNT-1 clinical trial, participants on the highest dose achieved 22.5% body weight reduction over 72 weeks. Compare that to semaglutide, which typically produces 15-17% weight loss at comparable timepoints. The GIP component accounts for much of that additional efficacy.
The combined action of GLP-1 and GIP also produces more favorable effects on body composition. Research suggests tirzepatide preserves lean muscle mass more effectively than GLP-1-only compounds, though this remains an active area of investigation. For researchers concerned about losing muscle alongside fat, this dual mechanism offers meaningful advantages over single-receptor approaches.
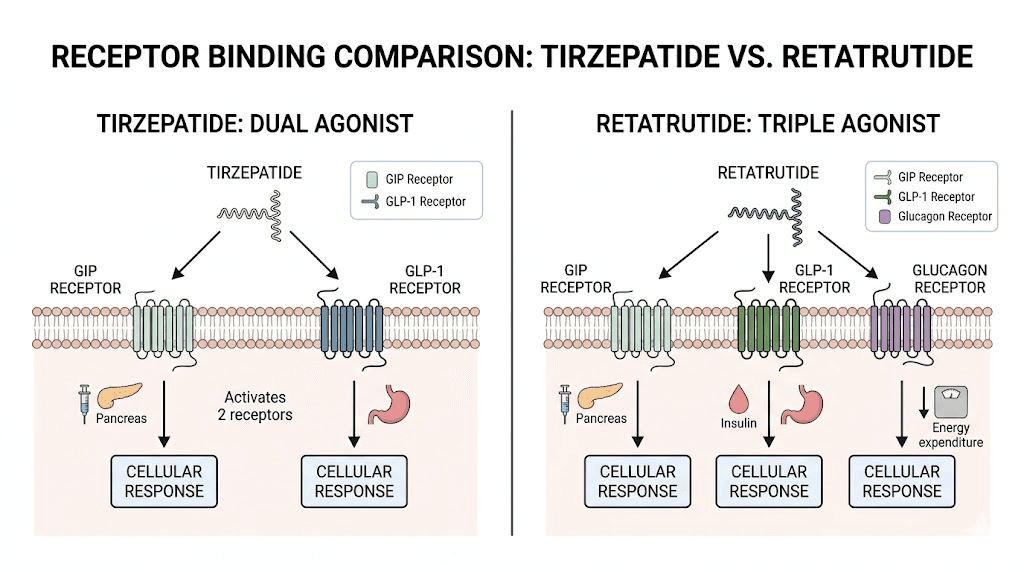
The triple agonist approach: retatrutide
Retatrutide does everything tirzepatide does. And then it does more.
Like tirzepatide, retatrutide activates both GLP-1 and GIP receptors, producing the same appetite suppression, insulin enhancement, and fat metabolism improvements. But retatrutide adds a third target: the glucagon receptor. This makes it the first triple-agonist peptide to reach advanced clinical development.
Glucagon is traditionally viewed as the opposite of insulin, a hormone that raises blood sugar by stimulating the liver to release stored glucose. So activating the glucagon receptor might seem counterproductive for metabolic health. It is not. The reality is more nuanced and considerably more interesting.
Glucagon receptor activation increases energy expenditure directly. The body burns more calories at rest. It also promotes hepatic fat oxidation, meaning the liver breaks down stored fat more aggressively. For researchers dealing with significant fat accumulation, particularly visceral and liver fat, this third mechanism adds a powerful tool that tirzepatide simply does not have.
The triple agonist approach creates what researchers describe as a metabolic triple threat. GLP-1 reduces food intake. GIP improves fat processing and insulin sensitivity. Glucagon increases calorie burning and fat breakdown. Together, these three mechanisms attack excess body weight from multiple angles simultaneously, which explains the remarkable weight loss numbers emerging from retatrutide clinical trials.
Why the glucagon receptor changes everything
The glucagon component is not just an incremental improvement. It fundamentally changes the metabolic equation.
With tirzepatide, weight loss occurs primarily through reduced calorie intake (GLP-1) and improved fat metabolism (GIP). The body takes in less energy and processes stored energy more efficiently. These are powerful mechanisms, but they both operate on the "input" side of the energy balance equation.
Retatrutide adds the "output" side. Glucagon receptor activation increases basal metabolic rate, meaning the body actively burns more calories throughout the day. Early research suggests this effect accounts for 2-4% of additional weight loss beyond what dual agonism achieves, a number that compounds significantly over a 48 to 68 week treatment period.
The liver fat reduction is equally significant. In Phase 2 trials, retatrutide achieved up to 82% reduction in liver fat, a number that caught hepatologists by surprise. Tirzepatide reduces liver fat as well, but not to the same degree. For researchers with metabolic dysfunction-associated steatotic liver disease (formerly called NAFLD), this difference matters enormously.
Weight loss results compared: clinical trial data
Numbers tell the story more clearly than mechanisms. Both tirzepatide and retatrutide have undergone rigorous clinical trials, and the weight loss data from these studies provides the most reliable comparison available. The differences are significant, though context matters when interpreting them.
Tirzepatide: SURMOUNT trial results
The SURMOUNT program represents the largest clinical trial dataset for tirzepatide weight loss. These Phase 3 trials enrolled thousands of participants across multiple studies, providing robust evidence for efficacy and safety.
SURMOUNT-1 was the landmark study. Over 72 weeks, participants without type 2 diabetes achieved the following weight loss results:
5 mg dose: 16.0% average body weight reduction
10 mg dose: 21.4% average body weight reduction
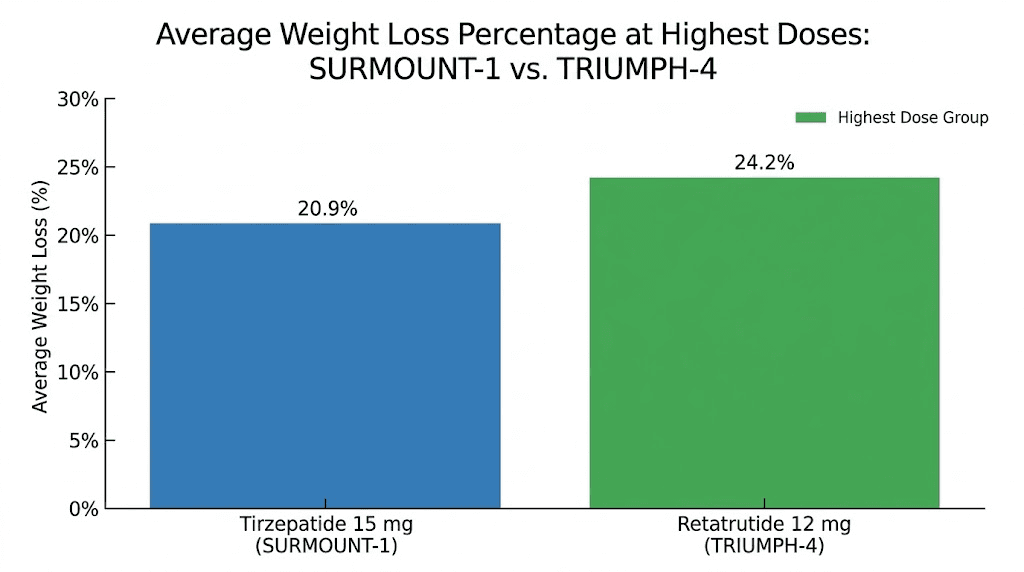
15 mg dose: 22.5% average body weight reduction
Placebo: 2.4% average body weight reduction
At the highest dose, 92.7% of participants achieved at least 5% weight loss, 84.4% achieved at least 10%, 73.6% achieved at least 15%, 56.1% achieved at least 20%, and 38.7% achieved at least 25% weight loss. These numbers established tirzepatide as the most effective weight loss peptide available at the time of approval.
SURMOUNT-4 looked at weight maintenance. After a 36-week lead-in period with tirzepatide treatment, participants who continued on the compound for an additional 52 weeks achieved 25.8% total weight loss. Those switched to placebo regained weight steadily, confirming that ongoing treatment is necessary to maintain results.
The SURMOUNT data also showed impressive metabolic improvements. Blood pressure dropped. Triglycerides fell. Inflammatory markers improved. Insulin sensitivity increased substantially across all dose groups. These benefits extend well beyond the scale, addressing the metabolic dysfunction that accompanies excess body weight.
Retatrutide: Phase 2 and TRIUMPH results
Retatrutide clinical data comes from two major sources: the Phase 2 obesity trial published in the New England Journal of Medicine, and the recently announced Phase 3 TRIUMPH-4 results.
The Phase 2 trial enrolled participants with obesity and evaluated multiple dose levels over 48 weeks:
1 mg dose: 8.7% average body weight reduction
4 mg dose (escalated from 2 mg): 17.1% average body weight reduction
8 mg dose (escalated from 2 mg): 22.8% average body weight reduction
12 mg dose (escalated from 2 mg): 24.2% average body weight reduction
Placebo: 2.1% average body weight reduction
Those Phase 2 numbers were striking enough. Then the TRIUMPH-4 Phase 3 data arrived in late 2025 and exceeded expectations.
Over 68 weeks, with a starting dose of 2 mg escalated every 4 weeks:
9 mg dose: 26.4% average body weight reduction (29.1 kg / 64.2 lbs)
12 mg dose: 28.7% average body weight reduction (32.3 kg / 71.2 lbs)
Placebo: 2.1% average body weight reduction
The milestone achievements tell an even more compelling story. Among participants on the 12 mg dose, 58.6% achieved at least 25% weight loss, 39.4% achieved at least 30% weight loss, and 23.7% achieved at least 35% weight loss. Nearly one in four participants lost more than a third of their body weight. These are numbers that would have seemed impossible just five years ago.
Head-to-head comparison table
No direct head-to-head trial between tirzepatide and retatrutide exists yet. The comparisons below draw from separate clinical trials with different designs, durations, and participant populations. Network meta-analyses help bridge this gap, but direct comparison data would be more definitive.
Factor | Tirzepatide (15 mg) | Retatrutide (12 mg) |
|---|---|---|
Max weight loss | 22.5% (72 weeks) | 28.7% (68 weeks) |
Absolute weight loss | ~11.8 kg average | ~32.3 kg average |
Achieved 25%+ loss | 38.7% | 58.6% |
Achieved 30%+ loss | Not reported | 39.4% |
Trial phase | Phase 3 (completed) | Phase 3 (ongoing) |
Receptor targets | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
FDA approved | Yes | No |
The weight loss advantage of retatrutide over tirzepatide appears to be approximately 6 percentage points at the highest doses, a clinically meaningful difference. A network meta-analysis published in the Journal of the Endocrine Society found retatrutide achieved 23.77% weight reduction compared to 16.79% for tirzepatide when standardized across trials, with absolute weight loss of 16.34 kg versus 11.82 kg. These numbers should be interpreted cautiously given the different trial designs, but the trend is consistent across multiple analyses.
Dosing protocols and escalation schedules
How you take a peptide matters almost as much as which peptide you choose. Both tirzepatide and retatrutide use gradual dose escalation strategies to minimize gastrointestinal side effects, but their specific protocols differ in ways that affect the practical experience of using each compound.
Tirzepatide dosing protocol
Tirzepatide follows a well-established escalation schedule refined through multiple clinical trials and years of real-world use:
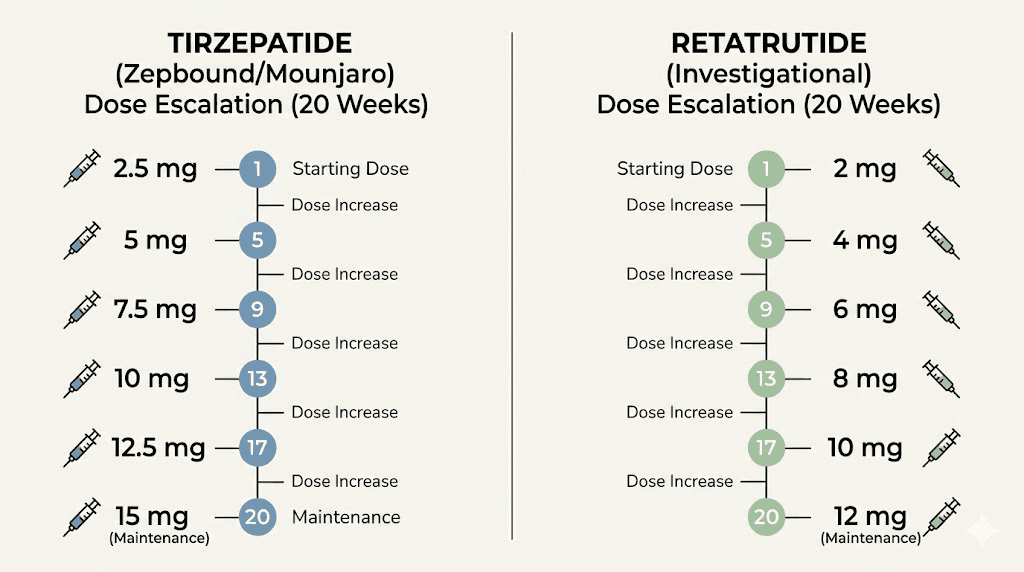
Weeks 1-4: 2.5 mg once weekly (starting dose)
Weeks 5-8: 5 mg once weekly
Weeks 9-12: 7.5 mg once weekly (optional intermediate step)
Weeks 13-16: 10 mg once weekly
Weeks 17-20: 12.5 mg once weekly (optional intermediate step)
Week 21+: 15 mg once weekly (maximum dose)
The starting dose of 2.5 mg is intentionally subtherapeutic. Its purpose is tolerance building, not weight loss. Most researchers begin noticing appetite suppression at the 5 mg level, with significant weight loss typically beginning around weeks 8-12 as doses increase.
For those using compounded formulations, understanding the unit-to-milligram conversions is essential. A 10 mg/mL concentration means 50 units equals 5 mg, while 100 units equals 10 mg. Getting these conversions wrong is one of the most common mistakes researchers make, and it leads to either underdosing (wasted time) or overdosing (excessive side effects).
The compounded tirzepatide dosage chart varies by concentration. Always verify your vial concentration before calculating injection volumes. Use the tirzepatide dosage calculator to determine exact volumes based on your specific concentration.
Retatrutide dosing protocol
Retatrutide uses a similar escalation approach but with different dose levels. The TRIUMPH-4 trial protocol provides the best reference for effective dosing:
Weeks 1-4: 2 mg once weekly (starting dose)
Weeks 5-8: 4 mg once weekly
Weeks 9-12: 6 mg once weekly
Weeks 13-16: 8 mg once weekly
Week 17+: 9 mg or 12 mg once weekly (maintenance dose)
The escalation happens every 4 weeks, which is slightly faster than many tirzepatide protocols. This aggressive but controlled ramp-up helps the body adjust to the triple receptor activation while minimizing the gastrointestinal distress that comes with too-rapid dose increases.
For researchers working with reconstituted retatrutide, the math requires careful attention. Vial concentrations for research-grade retatrutide vary significantly between suppliers. The retatrutide dosage chart provides concentration-specific guidance, and the peptide reconstitution calculator can determine exact water-to-peptide ratios.
Practical dosing comparison
Factor | Tirzepatide | Retatrutide |
|---|---|---|
Starting dose | 2.5 mg weekly | 2 mg weekly |
Maximum dose | 15 mg weekly | 12 mg weekly |
Escalation pace | Every 4 weeks | Every 4 weeks |
Time to max dose | ~20 weeks | ~16 weeks |
Injection frequency | Once weekly | Once weekly |
Injection site | Subcutaneous (abdomen, thigh, arm) | Subcutaneous (abdomen, thigh, arm) |
Both compounds use once-weekly subcutaneous injection. The injection technique is identical for both, and the same injection sites work for either compound. Researchers familiar with GLP-1 injection protocols will find the transition between compounds straightforward from a practical administration standpoint.
One notable difference is the time required to reach maximum dose. Tirzepatide takes approximately 20 weeks to reach 15 mg if following standard escalation with intermediate steps, while retatrutide reaches 12 mg in about 16 weeks. This faster escalation means peak effectiveness may arrive sooner with retatrutide, though the GI side effects during escalation tend to be more pronounced.
Side effects and safety profiles
Every effective metabolic peptide comes with trade-offs. Both tirzepatide and retatrutide produce gastrointestinal side effects as their primary adverse reactions, but the frequency, severity, and specific patterns differ between the two compounds. Understanding these differences helps researchers set realistic expectations and develop management strategies before problems arise.
Common side effects of tirzepatide
Tirzepatide has the advantage of extensive real-world safety data. Millions of people have used it since FDA approval, and the side effect profile is well-characterized.
The most common adverse effects from the SURMOUNT trials include:
Nausea: 24-33% of participants (dose-dependent)
Diarrhea: 18-25% of participants
Constipation: 13-17% of participants
Vomiting: 8-12% of participants
Decreased appetite: 10-15% of participants
Most of these effects are mild to moderate and occur primarily during dose escalation periods. They typically resolve or become manageable within 2-4 weeks at each new dose level. The body adapts. Patience during escalation is critical.
Less common but notable side effects include headaches, muscle pain, fatigue, anxiety, sleep disturbances, injection site reactions, and body aches. Some researchers also report effects on menstrual cycles and joint discomfort.
The serious adverse event rate with tirzepatide is low. No cases of medullary thyroid carcinoma or pancreatitis were confirmed in the SURMOUNT-4 trial. Discontinuation due to adverse events runs approximately 4-7% across studies, which is comparable to other GLP-1 receptor agonists.
Common side effects of retatrutide
Retatrutide shares the same GI side effect profile as tirzepatide but at higher rates. The TRIUMPH-4 Phase 3 data provides the most current safety information:
Nausea: 38.1% (9 mg) to 43.2% (12 mg) versus 10.7% placebo
Diarrhea: 34.7% (9 mg) to 33.1% (12 mg) versus 13.4% placebo
Constipation: 21.8% (9 mg) to 25.0% (12 mg) versus 8.7% placebo
Vomiting: 20.4% (9 mg) to 20.9% (12 mg) versus 0% placebo
Decreased appetite: 19.0% (9 mg) to 18.2% (12 mg) versus 9.4% placebo
The pattern is clear. Retatrutide produces approximately 30-50% more frequent GI side effects than tirzepatide at comparable efficacy doses. The nausea rate of 43.2% at 12 mg is substantially higher than tirzepatide at 15 mg. The vomiting rate of approximately 21% is particularly notable, as it approaches one in five users.
Discontinuation rates due to adverse events were 12.2% at the 9 mg dose and 18.2% at the 12 mg dose in TRIUMPH-4. These numbers are meaningfully higher than tirzepatide discontinuation rates and represent an important consideration for researchers planning long-term protocols.
The dysesthesia question
One side effect unique to retatrutide deserves special attention. Dysesthesia, an abnormal sensation often described as tingling, burning, or numbness in the skin, appeared in 8.8% of participants on the 9 mg dose and 20.9% on the 12 mg dose, compared to just 0.7% on placebo.
This is a new safety signal that emerged from the TRIUMPH-4 data. It does not appear in the tirzepatide side effect profile at comparable rates. The mechanism is not yet fully understood, but it may relate to the glucagon receptor activation or the broader metabolic changes that the triple agonist produces. Researchers considering retatrutide should be aware of this potential effect and monitor for unusual skin sensations during dose escalation.
The dysesthesia signal does not appear to be permanent. Most cases resolved during the study period. But for some researchers, this novel side effect may influence the risk-benefit calculation when choosing between the two compounds.
Managing GI side effects with either compound
Regardless of which peptide a researcher chooses, GI management strategies remain similar.
Diet plays a major role. Eating smaller, more frequent meals reduces nausea. Avoiding high-fat and greasy foods during dose escalation helps significantly. The foods to avoid on tirzepatide list applies equally to retatrutide, as both compounds slow gastric emptying through the same GLP-1 mechanism.
Staying hydrated is critical. Both compounds can cause fluid shifts, and dehydration worsens nausea, headaches, dizziness, and constipation. Aim for adequate water intake throughout the day, not just around meals.
The dietary approach matters too. High-protein foods tend to be better tolerated than carbohydrate-heavy meals during the adjustment period. The tirzepatide diet plan and meal planning resources provide structured approaches that work for either compound.
Certain supplements may help as well. B12 supplementation is particularly relevant because GLP-1 agonists can reduce B12 absorption over time. Many compounded formulations include B12, glycine, or niacinamide to offset this effect and improve tolerability.
Beyond weight loss: metabolic benefits compared
Weight loss grabs headlines. But the metabolic improvements these compounds produce may be even more significant for long-term health outcomes. Both tirzepatide and retatrutide influence blood sugar regulation, liver health, cardiovascular markers, and inflammatory pathways, though their relative strengths in each area differ.
Blood sugar control and type 2 diabetes
Tirzepatide received its first FDA approval specifically for type 2 diabetes management, marketed as Mounjaro. Its glycemic control is exceptional. In the SURPASS clinical trial program, tirzepatide reduced HbA1c by 2.0-2.4% depending on dose, bringing the majority of participants to target levels below 7.0%.
Retatrutide shows comparable glycemic improvements. In Phase 2 trials involving participants with type 2 diabetes, HbA1c improved by up to 2.2%, with 82% of participants reaching HbA1c levels at or below 6.5%. The triple agonist mechanism appears to match or slightly exceed tirzepatide for blood sugar management, likely because the glucagon component, while it can raise blood sugar in isolation, works synergistically with GLP-1 and GIP activation to produce a net positive effect on glucose regulation.
For researchers whose primary goal is metabolic health improvement rather than maximum weight loss, both compounds deliver exceptional results. The choice between them for glycemic control alone would likely favor tirzepatide simply because it is available and proven, while retatrutide remains investigational.
Liver fat reduction
This is where retatrutide pulls decisively ahead.
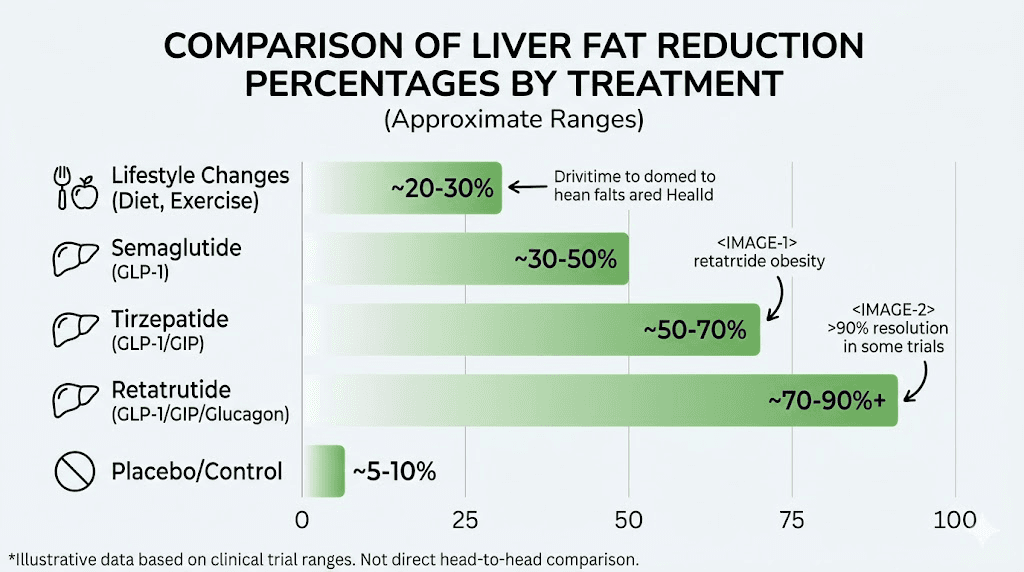
In a Phase 2a trial specifically evaluating liver outcomes, retatrutide achieved an average 82% reduction in liver fat content. That number is extraordinary. To put it in context, lifestyle interventions typically achieve 20-30% liver fat reduction, while semaglutide produces approximately 40-50% reduction in comparable studies.
Tirzepatide also reduces liver fat, with studies showing meaningful improvements, but the magnitude does not approach what retatrutide achieves. The glucagon receptor activation is the key differentiator. Glucagon directly stimulates hepatic fat oxidation, essentially telling the liver to burn through its stored fat reserves. This mechanism has implications for the estimated 100 million Americans with fatty liver disease, many of whom have no effective pharmacological treatment options.
A dedicated Phase 3 trial evaluating retatrutide specifically for metabolic dysfunction-associated steatotic liver disease is currently underway. If the Phase 2a results hold, retatrutide could become the first truly effective pharmaceutical treatment for this condition.
Cardiovascular and kidney outcomes
Tirzepatide has cardiovascular outcome data from the SURPASS-CVOT study, which showed favorable effects on major adverse cardiovascular events in people with type 2 diabetes. Compared to other weight loss approaches, tirzepatide demonstrates meaningful reductions in blood pressure, triglycerides, and inflammatory markers associated with cardiovascular risk.
Retatrutide does not yet have dedicated cardiovascular or renal outcome trials, though Phase 3 studies evaluating these endpoints are currently enrolling participants with expected completion in 2026. The early data from obesity trials shows favorable trends in cardiovascular risk factors, but definitive outcome data remains forthcoming.
For researchers who prioritize cardiovascular safety data, tirzepatide currently offers more reassurance simply because the data exists. Retatrutide may ultimately prove equal or superior, but we do not have that evidence yet.
Joint pain and osteoarthritis
The TRIUMPH-4 trial produced an unexpected but welcome finding regarding joint health. Participants with knee osteoarthritis experienced dramatic pain reduction alongside their weight loss.
Retatrutide reduced WOMAC pain scores by up to 4.5 points (75.8% improvement) at the 9 mg dose and 4.4 points (74.3% improvement) at the 12 mg dose. More remarkably, 14.1% of participants on the 9 mg dose and 12.0% on the 12 mg dose were completely free of knee pain by the end of the trial. For context, only 4.2% of placebo participants achieved complete pain freedom.
Much of this pain reduction likely stems from the substantial weight loss reducing mechanical stress on joints. However, some researchers speculate that the glucagon receptor activation may also reduce joint inflammation through pathways independent of weight loss. This hypothesis requires further investigation.
Tirzepatide has not been studied specifically for osteoarthritis outcomes in a dedicated trial, though weight loss from any intervention typically improves joint pain. The magnitude of improvement seen with retatrutide, driven by its greater weight loss, represents a potentially significant advantage for researchers dealing with obesity-related joint conditions.
Availability, approval status, and cost
The practical reality of accessing these compounds differs dramatically. One is available at any pharmacy with a prescription. The other exists primarily in clinical trials and research supply chains. This accessibility gap is the most immediate factor for most researchers choosing between the two.
Tirzepatide: FDA approved and widely available
Tirzepatide received FDA approval for type 2 diabetes (as Mounjaro) in 2022 and for chronic weight management (as Zepbound) in 2023. It is available through standard pharmacy channels with a prescription.
Affordability varies significantly depending on insurance coverage, pharmacy choice, and formulation type. Brand-name Mounjaro and Zepbound carry substantial list prices, though manufacturer coupons and insurance coverage can reduce out-of-pocket costs considerably.
Compounded tirzepatide represents an alternative that many researchers explore. Compounded formulations are available through compounding pharmacies at significantly lower price points. Popular options include formulations from Empower Pharmacy, ProRx, Orderly Meds, and Southend Pharmacy. The compounded versions often include beneficial additions like B12, glycine, and B12 combinations, or methylcobalamin.
Storage requirements for tirzepatide are straightforward. The compound needs refrigeration for long-term storage, though it can tolerate limited time at room temperature. Understanding shelf life in the fridge and expiration considerations helps researchers maximize potency and avoid waste.
Tirzepatide is also available in oral formulations, sublingual drops, and orally disintegrating tablets, giving researchers multiple administration options beyond traditional injection. The oral versus injection comparison and tablets versus injections analysis can help researchers determine which format best fits their needs.
Retatrutide: still in clinical trials
Retatrutide has not received FDA approval for any indication. It remains an investigational compound in Phase 3 clinical development. The TRIUMPH program includes eight Phase 3 trials evaluating retatrutide for obesity, type 2 diabetes, osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular outcomes, and liver disease. Seven of these trials are expected to report results in 2026.
The current availability of retatrutide is limited to clinical trial participation and research-grade supply through peptide suppliers. Purchasing retatrutide for research requires navigating the research peptide market, where quality, purity, and reliability vary significantly between vendors.
For researchers seeking research-grade retatrutide, several factors matter: third-party purity testing, proper administration guidance, clear injection protocols, and reliable cold-chain shipping. The online purchasing guide and bulk retatrutide information provide detailed guidance for researchers evaluating their options.
The cost of retatrutide through research suppliers currently runs higher per milligram than compounded tirzepatide, largely because of limited manufacturing scale and the absence of compounding pharmacy competition. If and when retatrutide receives FDA approval, compounding pharmacy availability would likely drive costs closer to current tirzepatide pricing.
Assuming Eli Lilly follows a similar timeline to tirzepatide, the earliest possible FDA approval for retatrutide would likely occur in late 2027 or 2028, depending on Phase 3 trial results and regulatory review timelines.
Cost comparison
Factor | Tirzepatide | Retatrutide |
|---|---|---|
FDA approved | Yes (Mounjaro, Zepbound) | No (investigational) |
Pharmacy availability | Widely available | Research suppliers only |
Compounded options | Multiple pharmacies | Not yet compounded |
Oral options | Drops, ODT, tablets | Injectable only |
Insurance coverage | Possible (varies) | None |
Estimated approval | Already approved | Late 2027-2028 (estimated) |
Switching between tirzepatide and retatrutide
Some researchers begin with tirzepatide and later consider transitioning to retatrutide as it becomes more accessible. Others may move in the opposite direction. Understanding the practical considerations of switching helps prevent common mistakes during transitions.
When switching from tirzepatide to retatrutide makes sense
Several scenarios might prompt a researcher to consider the transition:
Weight loss has plateaued on tirzepatide. Some researchers reach a point where tirzepatide stops producing results even at maximum doses. The additional glucagon receptor activation from retatrutide could break through this plateau by adding increased energy expenditure to the equation.
Liver health is a primary concern. If liver fat reduction is a key objective, retatrutide offers substantially greater hepatic fat clearance than tirzepatide, as discussed above.
Maximum weight loss is the goal. For researchers who need or want the greatest possible weight reduction, the approximately 6 percentage point advantage of retatrutide represents a meaningful difference.
Researchers who have used tirzepatide before starting retatrutide report that the transition requires careful dose management. Starting retatrutide at full maintenance doses after being on high-dose tirzepatide can lead to excessive GI side effects because of the additional glucagon receptor stimulation. Most experienced researchers recommend dropping back to a lower escalation dose and working up gradually, even with prior GLP-1 agonist experience.
When staying with tirzepatide makes more sense
Tirzepatide remains the better choice in several scenarios.
Safety data matters to you. Tirzepatide has years of real-world safety data and completed Phase 3 programs across multiple indications. Retatrutide is still building its safety profile. For risk-averse researchers, proven safety carries significant weight.
Accessibility and consistency are priorities. Tirzepatide is available through multiple channels with predictable quality and supply. Retatrutide availability remains limited and variable.
Side effect tolerance is low. If a researcher struggles with GI effects on tirzepatide, retatrutide will likely be worse, not better. The higher adverse event rates mean that researchers with sensitive GI systems may find retatrutide intolerable at effective doses.
The results are already satisfactory. If tirzepatide is producing the desired outcomes, there is no compelling reason to switch to a less proven compound with more side effects. Researchers who are responding well to tirzepatide, losing weight consistently, and tolerating the medication, should seriously consider staying the course.
The conversion guidance that exists for transitioning between other GLP-1 agonists can provide some framework for thinking about dosing during transitions, though no specific tirzepatide-to-retatrutide conversion protocol has been established in clinical literature.
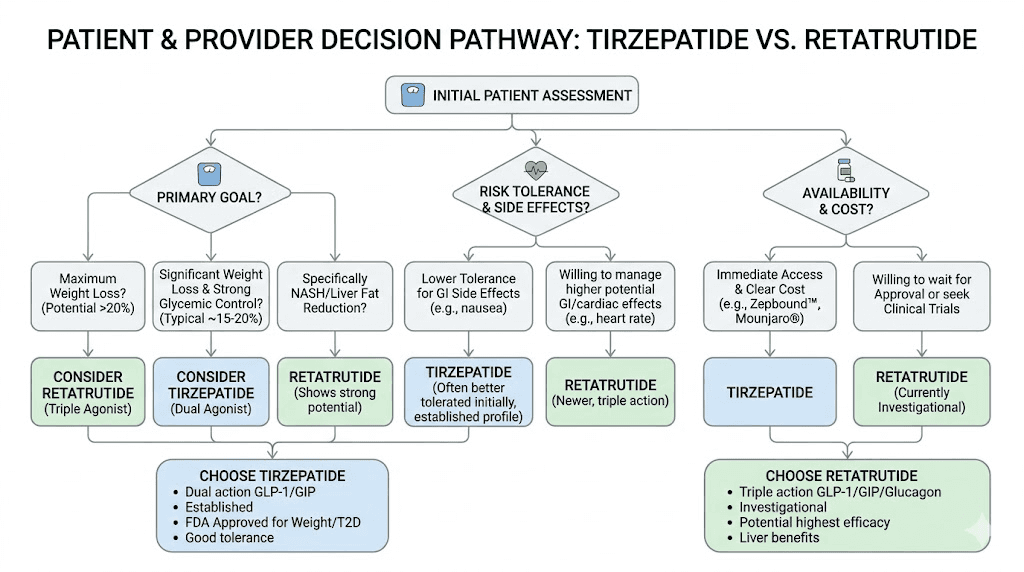
Who should consider each compound
The right choice depends on individual circumstances, goals, risk tolerance, and practical constraints. Neither compound is universally superior. Each has a profile that matches certain researcher needs better than others.
Best candidates for tirzepatide
Tirzepatide is the stronger choice for researchers who:
Want a proven, FDA-approved compound with extensive safety data
Need reliable, consistent access through established supply chains
Prefer multiple administration options (injectable, oral drops, tablets)
Have lower side effect tolerance and want a more predictable GI experience
Are achieving satisfactory weight loss goals (15-22% range)
Want insurance coverage possibilities
Are new to GLP-1 agonist therapy and want to start with the established option
Need to travel with their medication and want pharmacy accessibility
The practical advantages of tirzepatide cannot be overstated for many researchers. Having a compound that is legally prescribed, covered by some insurance plans, available at local pharmacies, and backed by years of post-market surveillance data provides a level of confidence and convenience that research-grade compounds simply cannot match.
Best candidates for retatrutide
Retatrutide makes more sense for researchers who:
Need maximum possible weight loss and have not achieved adequate results with dual agonists
Have significant liver fat accumulation as a primary concern
Have plateaued on tirzepatide or stopped responding
Are comfortable with investigational compounds and research-grade sourcing
Have higher side effect tolerance
Want the most comprehensive metabolic intervention currently available
Are experienced with GLP-1 agonist protocols and understand escalation management
Have access to medical monitoring during the protocol
Retatrutide is not a beginner compound. Researchers who have never used a GLP-1 agonist should generally start with tirzepatide or even semaglutide to understand their individual response to incretin therapy before attempting a triple agonist with higher side effect rates.
Decision framework
Your situation | Best choice | Why |
|---|---|---|
First time using GLP-1 therapy | Tirzepatide | Proven safety, established protocols, accessible |
Plateaued on tirzepatide | Retatrutide | Additional mechanism may break plateau |
Significant liver fat | Retatrutide | 82% liver fat reduction in trials |
Need max weight loss | Retatrutide | 28.7% vs 22.5% at highest doses |
Low side effect tolerance | Tirzepatide | Lower adverse event rates |
Need pharmacy access | Tirzepatide | FDA approved, widely available |
Budget conscious | Tirzepatide | Compounded options more affordable |
Type 2 diabetes primary | Tirzepatide | Approved indication, proven outcomes |
Obesity with osteoarthritis | Retatrutide | TRIUMPH-4 showed significant pain reduction |
Want insurance coverage | Tirzepatide | Only approved option |
SeekPeptides members access detailed protocol guides for both tirzepatide and retatrutide, including personalized dosing calculators, side effect management strategies, and transition protocols. The platform provides evidence-based guidance that helps researchers navigate this decision with confidence, backed by the experience of thousands of members who have evaluated both compounds.
The future landscape: what comes next
The comparison between tirzepatide and retatrutide is a snapshot of a rapidly evolving field. Several developments will reshape this landscape within the next 12-24 months.
Seven additional Phase 3 trials for retatrutide are expected to report results in 2026. These will provide data on long-term safety, cardiovascular outcomes, renal effects, and efficacy in populations with type 2 diabetes, sleep apnea, and chronic back pain. Each readout will add important information to the comparison. Some may reveal advantages for retatrutide that are not yet apparent. Others may identify limitations.
Other compounds are also entering the picture. Survodutide, another GLP-1/glucagon dual agonist from Boehringer Ingelheim, is advancing through clinical trials. CagriSema, a combination of cagrilintide and semaglutide from Novo Nordisk, represents yet another approach to multi-receptor targeting. Mazdutide and its comparison with retatrutide adds another dimension to the evolving landscape of metabolic peptide therapy.
The competitive environment benefits researchers. More compounds mean more options, more data, and ultimately better outcomes. Whether tirzepatide or retatrutide proves to be the optimal choice for a given individual will depend on factors that continue to clarify as clinical evidence accumulates.
For researchers making decisions today, the calculus is relatively straightforward. Tirzepatide is the proven, accessible, well-characterized option that delivers exceptional results for most people. Retatrutide is the promising, potentially superior option that carries more uncertainty but offers unique advantages for specific use cases. Both represent transformative advances in metabolic health.
Practical tips for researchers evaluating both compounds
Making an informed choice requires more than comparing clinical trial data. These practical considerations often prove decisive in real-world decision making.
Start with proper tools
Whichever compound you choose, accurate dosing matters enormously. The peptide calculator helps determine exact doses based on concentration and target amount. The reconstitution calculator ensures proper water-to-peptide ratios when working with lyophilized powders. The cost calculator helps compare the financial implications of different compounds, concentrations, and vendors.
For tirzepatide specifically, the semaglutide dosage calculator provides a useful reference point for understanding GLP-1 agonist dosing principles, while the syringe dosage guide covers the mechanics of drawing accurate doses.
Understand reconstitution requirements
Research-grade peptides arrive as lyophilized powders requiring reconstitution with bacteriostatic water. The process is identical for both compounds. Understanding how to reconstitute tirzepatide gives you the skills needed for retatrutide reconstitution as well. Key resources include the bacteriostatic water mixing guide for tirzepatide and the equivalent guide for retatrutide.
Sterile technique matters. Contaminated vials can cause injection site infections and render expensive peptides useless. Always use alcohol swabs on vial stoppers, draw bacteriostatic water with a sterile syringe, and inject it gently down the side of the vial rather than directly onto the powder.
Storage is not optional
Both tirzepatide and retatrutide require refrigeration after reconstitution. Unreconstituted lyophilized powder is more stable but still benefits from cold storage. Understanding refrigeration requirements prevents potency loss and wasted product.
If you accidentally leave your peptide out, do not panic. The room temperature tolerance guide and overnight temperature exposure guide provide specific timelines for how long peptides remain viable outside refrigeration.
Monitor your progress systematically
Track weight, body measurements, energy levels, appetite changes, side effects, and any relevant lab values throughout your protocol. Systematic tracking allows you to identify when a compound is working, when it is not, and when adjustments are needed.
The weight loss timeline for tirzepatide provides a useful benchmark. Most researchers see initial results within 4-8 weeks, with the most dramatic changes occurring between weeks 12-24. Retatrutide appears to follow a similar trajectory based on clinical trial data, though individual responses vary.
If results stall, the troubleshooting guide for tirzepatide and early plateau guidance offer specific strategies. Common culprits include inadequate protein intake, insufficient sleep, excessive stress, and medication interactions.
Consider combination approaches
Some researchers explore stacking additional compounds with their primary GLP-1 agonist. Creatine helps preserve muscle mass during rapid weight loss. Phentermine combinations have been used for enhanced appetite suppression, though this requires medical supervision. The peptide stack calculator can help evaluate potential combinations.
For researchers already on tirzepatide who want to understand how it compares to the broader landscape, the phentermine versus GLP-1 comparison provides useful context, as does the Ozempic versus retatrutide analysis.
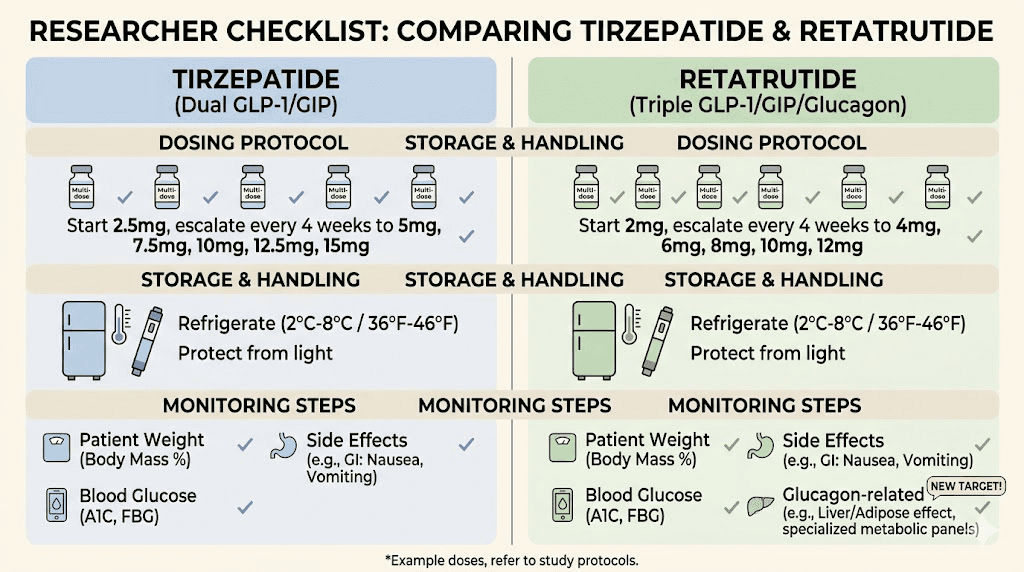
Special considerations and populations
Certain populations and situations require additional thought when choosing between these compounds.
Women of reproductive age
Both GLP-1 agonists can affect menstrual cycles and hormone levels. Weight loss itself alters estrogen metabolism and can change cycle timing. Some researchers have reported unexpected pregnancies while on tirzepatide, likely because improved metabolic health can restore ovulation in women with weight-related anovulation.
Neither compound is recommended during pregnancy or breastfeeding. Researchers in these situations should discontinue use and consult with healthcare providers.
Alcohol consumption
Both compounds interact with alcohol metabolism in ways that matter. GLP-1 agonists slow gastric emptying, which can change how quickly alcohol absorbs and how it affects the body. The tirzepatide and alcohol guide and semaglutide and alcohol guidance provide specific recommendations that apply to retatrutide as well.
Surgical planning
Researchers who may need surgery should understand that GLP-1 agonists slow gastric emptying, which can increase aspiration risk during anesthesia. Understanding when to resume after surgery and how long before surgery to pause the compound is critical for safety.
Pre-existing GI conditions
Researchers with existing gastrointestinal issues, gastroparesis, inflammatory bowel disease, or chronic constipation, should exercise particular caution. Both compounds worsen GI symptoms, but retatrutide does so more frequently. Starting with tirzepatide at conservative doses provides a safer entry point for this population.
The constipation management guide and fatigue management approaches offer practical solutions for researchers dealing with these common challenges regardless of which compound they choose.
Frequently asked questions
Is retatrutide stronger than tirzepatide?
In terms of weight loss, yes. Clinical trial data shows retatrutide produces approximately 28.7% weight loss at the highest dose compared to 22.5% for tirzepatide. However, "stronger" also means more frequent side effects. Retatrutide has higher rates of nausea, diarrhea, and vomiting than tirzepatide. The additional glucagon receptor activation adds both efficacy and adverse events.
Can I switch from tirzepatide to retatrutide?
Yes, though the transition requires careful dose management. Do not start retatrutide at your tirzepatide maintenance dose. Begin at a lower escalation dose and work up gradually to allow your body to adjust to the third receptor target. The transition timeline guide provides specific recommendations.
When will retatrutide be FDA approved?
The earliest realistic approval timeline is late 2027 to 2028, assuming positive Phase 3 trial results and a standard FDA review process. Seven Phase 3 trials are expected to complete in 2026, with regulatory submission likely following shortly after. Check the availability updates page for the latest status.
Which compound is better for type 2 diabetes?
Currently, tirzepatide is the clear choice for type 2 diabetes because it has FDA approval for this indication and extensive clinical evidence. Retatrutide shows comparable HbA1c improvements in Phase 2 trials, but it lacks the comprehensive diabetes-specific trial data that tirzepatide possesses through the SURPASS program.
Does retatrutide work for liver fat better than tirzepatide?
Significantly so. Retatrutide achieved 82% liver fat reduction in Phase 2a trials, far exceeding what tirzepatide or semaglutide achieve. The glucagon receptor activation drives hepatic fat oxidation in ways that dual agonists do not replicate. A dedicated Phase 3 liver disease trial for retatrutide is currently underway.
Are the side effects of retatrutide manageable?
For most researchers, yes, though they require more active management than tirzepatide side effects. Gradual dose escalation, dietary modifications, adequate hydration, and supportive supplements help significantly. The dysesthesia side effect (tingling or numbness) at higher doses is unique to retatrutide and resolves for most researchers, but it is an additional consideration.
Can I use tirzepatide and retatrutide together?
No. Using two incretin-based compounds simultaneously would create overlapping receptor activation at the GLP-1 and GIP sites, dramatically increasing side effect risk without proportional efficacy gains. Researchers should use one compound at a time.
How do I know which dose of retatrutide equals my tirzepatide dose?
There is no established dose equivalence table between these compounds because they have different receptor profiles and potencies. A researcher on 10 mg tirzepatide should not assume that 10 mg retatrutide produces equivalent effects. The compounds work differently enough that direct dose translation is not valid. Work with the retatrutide dose guide to determine appropriate dosing independently.
For researchers serious about making the most informed choice between these compounds, SeekPeptides offers the most comprehensive resource available. Members access detailed comparison tools, personalized protocol builders, and a community of experienced researchers who have navigated this exact decision, with evidence-based guides updated as new clinical data emerges.
External resources
New England Journal of Medicine: Retatrutide Phase 2 Trial for Obesity
PMC: Comparative Efficacy of Tirzepatide vs Retatrutide Meta-Analysis
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your protocols stay optimized, and your results stay consistent.