Feb 8, 2026

Experienced researchers know something beginners do not about tirzepatide formulations. The pink-tinted solution arriving in compounded vials contains more than just the dual-agonist peptide itself. When you see that distinctive color, you are looking at cyanocobalamin, a specific form of vitamin B12 deliberately added to the formulation. This is not decorative. The compounding pharmacies include B12 for physiological reasons related to how GLP-1 receptor agonists affect your digestive system. The absorption of this essential vitamin decreases when gastric emptying slows dramatically. Your body processes food differently on tirzepatide. The addition of B12 addresses this metabolic shift directly.
The combination works through multiple pathways. Tirzepatide dosing protocols typically start at low levels and increase gradually over months. During this titration period, your stomach empties food at approximately 40-50% of its normal rate. Nutrient absorption changes. B12 absorption depends heavily on intrinsic factor binding in the stomach and transit time through the small intestine. When food sits in your stomach for extended periods, the normal B12 absorption cascade faces disruption. Studies tracking GLP-1 users found that 12.7% developed nutritional deficiencies within six months. By twelve months, that number climbed to 22.4%. These are not trivial percentages. Resources like SeekPeptides have documented these patterns extensively, providing researchers with tools to navigate nutrient management during GLP-1 therapy.
The SURMOUNT-1 trial established tirzepatide efficacy with remarkable clarity. Participants on the 5mg maintenance dose lost an average of 16% body weight. Those on 10mg lost 21.4%. The 15mg group achieved 22.5% weight loss.
More impressive: 91% of those on 15mg lost at least 5% of their starting weight, and 57% crossed the 20% threshold. These numbers represent significant metabolic changes happening rapidly in your body. When you lose weight this quickly, nutrient demands shift. Your reduced caloric intake, which drops by 16-39% on GLP-1 medications according to metabolic ward studies, creates potential gaps. B12 helps fill one of those gaps.
Most compounded formulations also include glycine. This amino acid serves a distinct purpose from B12. While cyanocobalamin addresses nausea and vitamin status, glycine targets muscle preservation. During aggressive weight loss, your body does not discriminate perfectly between fat and muscle tissue. The addition of glycine provides building blocks for protein synthesis at a time when dietary protein intake often decreases. This three-component formulation addresses the realities of rapid weight loss in ways that single-ingredient preparations cannot.
What compounded tirzepatide with B12 is and how it differs from brand formulations
Compounded tirzepatide with B12 represents a lyophilized powder that requires reconstitution before use. The vial contains tirzepatide as the active peptide, cyanocobalamin (vitamin B12) typically at concentrations around 0.5-1mg per vial, and often glycine as a third component. You receive this as a white or off-white powder. After adding bacteriostatic water, the solution turns pink or light red. That color comes entirely from the cyanocobalamin. Pure tirzepatide solutions without B12 appear clear or slightly opalescent.
The formulation differs fundamentally from Mounjaro or Zepbound, the FDA-approved brand versions. Those products contain only tirzepatide as the active ingredient, along with pharmaceutical excipients for stability and pH buffering. They arrive in pre-filled pens at specific, fixed doses. Semaglutide vs tirzepatide comparisons often focus on efficacy, but formulation differences matter equally. Compounded versions allow flexible dosing adjustments that pre-filled pens cannot match. You can increase your dose by 10 units instead of waiting for the next standard tier. This granular control helps some users manage side effects more effectively.
Compounding pharmacies create these formulations under Section 503A or 503B of the Federal Food, Drug, and Cosmetic Act. They source pharmaceutical-grade tirzepatide powder, combine it with B12 and other excipients, and perform sterile compounding in controlled environments. The resulting product undergoes testing for potency, sterility, and endotoxins. However, compounded medications do not receive the same FDA approval process as brand drugs. This distinction carries both advantages and limitations. The advantage: customization and often lower cost. The limitation: less regulatory oversight compared to branded pharmaceuticals.
The inclusion of B12 specifically addresses nausea, one of the most common side effects reported in tirzepatide clinical trials. In SURMOUNT-1, nausea occurred in 24.6% of participants on 5mg, 33.3% on 10mg, and 31.0% on 15mg. Most cases were mild to moderate and decreased over time. But for some users, nausea becomes dose-limiting. Cyanocobalamin has established antiemetic properties. While the exact mechanism remains debated, clinical observations suggest that B12 supplementation reduces nausea severity in a subset of users. The compounding pharmacies add it for this practical reason, not merely to increase ingredient count.
Glycine serves muscle preservation. During the rapid weight loss phase, your body mobilizes both adipose tissue and lean mass. Best peptides for muscle growth typically focus on anabolic signals, but anti-catabolic support matters equally during caloric restriction. Glycine provides substrate for collagen synthesis and supports protein balance. While not a complete solution to muscle loss, it offers some protective effect when combined with adequate dietary protein and resistance training.
The concentration of tirzepatide in compounded vials varies. Common formulations include 10mg, 12mg, 15mg, or 20mg per vial. You reconstitute these with 1mL, 2mL, or sometimes 3mL of bacteriostatic water depending on desired final concentration. peptide reconstitution calculator tools help determine the exact volume needed to achieve specific dosing in units on an insulin syringe. If you have a 12mg vial and add 2mL of bacteriostatic water, you create a 6mg/mL solution. Each 10 units on a U-100 insulin syringe then delivers 0.6mg of tirzepatide.
Storage requirements differ slightly from brand formulations due to the reconstituted nature. Lyophilized powder before mixing can often be stored at room temperature, though refrigeration extends shelf life. After reconstitution, the solution requires refrigeration at 2-8°C. How long peptides last in the fridge depends on several factors including bacteriostatic water quality and sterile technique during reconstitution. Most compounded tirzepatide with B12 remains stable for 28-42 days when refrigerated properly.
The pink color serves as a visual indicator. If your reconstituted solution appears clear instead of pink, the vial likely does not contain B12.
Some compounding pharmacies offer tirzepatide without B12 as a separate option. Others include it by default. Always verify with your pharmacy what components your specific formulation contains. The presence or absence of B12 affects both your supplementation strategy and your interpretation of injection site reactions or systemic responses.
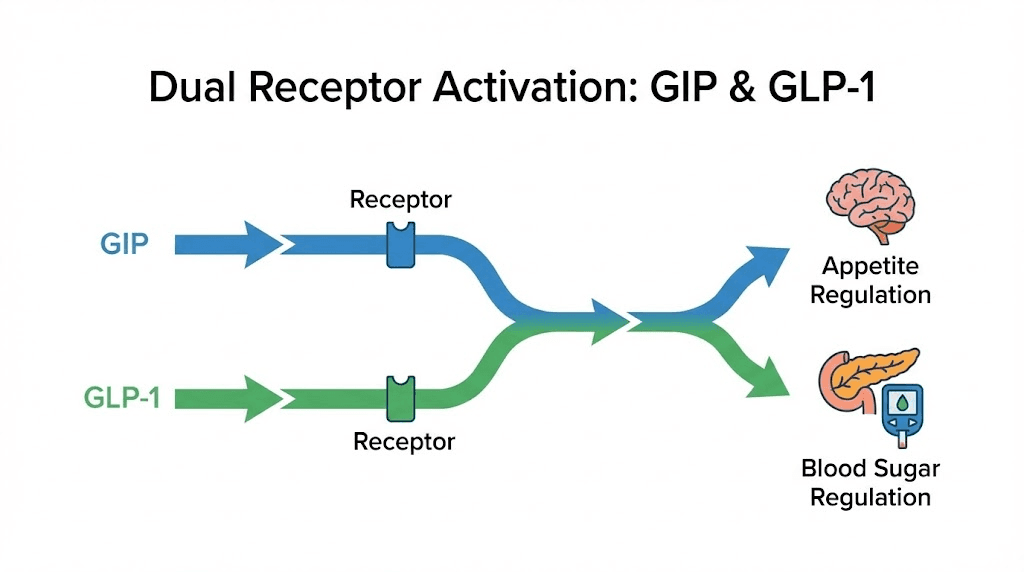
How tirzepatide works through dual GIP and GLP-1 receptor activation
Tirzepatide activates two distinct incretin receptors simultaneously. It binds to glucose-dependent insulinotropic polypeptide (GIP) receptors and glucagon-like peptide-1 (GLP-1) receptors. This dual mechanism distinguishes it from semaglutide with B12, which targets only GLP-1 receptors. The GIP component adds metabolic effects that single-agonist drugs cannot produce.
GLP-1 activation decreases appetite through multiple pathways. The peptide crosses the blood-brain barrier and acts on hypothalamic nuclei that regulate hunger. It slows gastric emptying significantly, which prolongs satiety after meals. It also modulates reward pathways in the brain, reducing food cravings particularly for high-fat and high-sugar foods. Clinical observations show that users report thinking about food less frequently. Meal planning becomes less mentally consuming. This is not willpower. This is direct neurohormonal modulation.
The GIP receptor activation contributes differently. Early research suggested GIP opposed weight loss by promoting fat storage. More recent evidence indicates that sustained GIP receptor agonism in the presence of GLP-1 activation enhances insulin sensitivity and may preferentially reduce visceral adipose tissue. The combination produces greater weight loss than GLP-1 agonism alone. SURMOUNT-1 demonstrated this clearly when comparing tirzepatide results to historical semaglutide data. At comparable timepoints, tirzepatide showed 3-5 percentage points greater weight loss across dose ranges.
Both receptors influence glucose metabolism. When blood glucose rises after a meal, GLP-1 and GIP both stimulate insulin secretion from pancreatic beta cells. This is glucose-dependent, meaning the effect only occurs when blood sugar is elevated. At normal or low glucose levels, the peptides do not trigger insulin release. This safety feature reduces hypoglycemia risk compared to insulin or sulfonylurea medications. The glucose-lowering effect proved so robust that tirzepatide earned FDA approval for type 2 diabetes management before its weight loss indication.
Gastric emptying slows dramatically under tirzepatide. Studies using scintigraphy to track food transit show that stomach emptying rates decrease by 40-60% at therapeutic doses. This means food sits in your stomach substantially longer. From a satiety perspective, this helps. From a nutrient absorption perspective, this complicates things. Vitamins and minerals that require specific absorption windows in the small intestine may not encounter those windows at optimal times. B12 absorption specifically depends on several coordinated steps that rely on normal gastric acid production and transit timing.
The peptide also affects energy expenditure, though this effect is modest. Some studies show a small increase in resting metabolic rate, possibly mediated by improved insulin sensitivity and reduced lipotoxicity in tissues.
However, this is not the primary mechanism of weight loss. The dominant driver is reduced caloric intake. Tirzepatide appetite suppression timeline typically shows noticeable effects within the first week, with maximum suppression reached after 4-8 weeks on a stable dose.
Receptor binding kinetics matter. Tirzepatide has a half-life of approximately 5 days, allowing for once-weekly dosing. The peptide structure includes modifications that resist degradation by dipeptidyl peptidase-4 (DPP-4), an enzyme that rapidly breaks down natural GLP-1. This extended half-life keeps receptor activation constant throughout the week. Blood levels peak around 1-2 days post-injection and decline slowly. Even at trough, just before your next dose, receptor activation remains sufficient to maintain appetite suppression.
The molecular structure includes a C20 fatty acid chain attached to the peptide backbone. This lipid modification allows binding to albumin in the bloodstream. The albumin-bound peptide acts as a reservoir, slowly releasing active drug over days. This pharmaceutical design principle appears in several modern peptide therapeutics. How peptides work often involves these modifications to overcome the short half-lives of natural peptides.
Tissue distribution favors areas with high GLP-1 and GIP receptor expression: pancreas, gut, brain, and adipose tissue. The peptide does not significantly cross into most other tissues. This targeted distribution contributes to the favorable side effect profile relative to systemic weight loss interventions like stimulants. You are not activating adrenergic receptors throughout your body. You are specifically modulating incretin pathways that naturally regulate feeding behavior and glucose metabolism.
Why compounding pharmacies add vitamin B12 to tirzepatide formulations
The addition of B12 to compounded tirzepatide addresses two primary concerns: nausea management and potential vitamin deficiency during treatment. Nausea represents the most commonly reported side effect in clinical trials. While often mild and self-limiting, it causes some users to discontinue therapy or struggle with dose escalation. Cyanocobalamin has documented antiemetic properties, though the exact mechanism remains incompletely understood. Some researchers propose that B12 influences neurotransmitter metabolism in the chemoreceptor trigger zone of the brainstem. Others suggest it modulates gastric motility through effects on myenteric plexus neurons. Regardless of mechanism, clinical observations support a nausea-reducing effect in a subset of users.
The vitamin deficiency concern arises from how GLP-1 receptor agonists alter gastric physiology. B12 absorption requires multiple coordinated steps. Food-bound B12 must first be released by gastric acid and pepsin. Free B12 then binds to R-proteins secreted by salivary glands. In the duodenum, pancreatic enzymes cleave B12 from R-proteins, allowing binding to intrinsic factor produced by gastric parietal cells. The B12-intrinsic factor complex travels to the terminal ileum where specific receptors mediate absorption. This entire process depends on adequate gastric acid, proper enzyme secretion, and appropriate transit time through the GI tract.
Tirzepatide disrupts this cascade at multiple points. Delayed gastric emptying extends the time B12 spends in the acidic stomach environment, which might initially seem beneficial. However, prolonged gastric retention also means delayed arrival at the intrinsic factor binding sites and the ileal absorption receptors. Additionally, some research suggests chronic GLP-1 agonist use may reduce gastric acid secretion over time, though this finding remains controversial. The combination of altered transit and potentially reduced acid creates conditions where B12 absorption efficiency could decline.
Population studies of GLP-1 users show measurable nutritional deficiencies developing over time. A retrospective analysis of patients on semaglutide or liraglutide found that 12.7% developed at least one nutritional deficiency within six months of starting therapy. By twelve months, this increased to 22.4%. The most common deficiencies involved vitamin B12, vitamin D, and folate. While not every user becomes deficient, the risk is substantial enough to warrant proactive supplementation, particularly during aggressive weight loss phases.
The reduced caloric intake on tirzepatide compounds the problem. Why not losing weight on semaglutide discussions often overlook the opposite problem: losing weight too rapidly. When users cut their intake by 30-40%, they dramatically reduce their consumption of B12-rich foods like meat, fish, eggs, and dairy. Even with normal absorption efficiency, low intake creates deficiency risk. Combine low intake with impaired absorption, and deficiency becomes highly probable without supplementation.
Compounding pharmacies recognized these patterns and began including B12 as a preventive measure. The typical concentration ranges from 0.5mg to 1mg per vial. Semaglutide with B12 formulations use similar concentrations. This provides 500-1000mcg of B12 with each weekly injection, well above the recommended daily allowance of 2.4mcg for adults. The high dose compensates for potentially impaired absorption and ensures adequate tissue stores.
Cyanocobalamin specifically appears in most formulations rather than methylcobalamin, adenosylcobalamin, or hydroxocobalamin. This choice reflects stability considerations. Cyanocobalamin demonstrates superior stability in solution, particularly when exposed to light or temperature fluctuations. Methylcobalamin degrades more rapidly. For a medication that users store in home refrigerators for weeks after reconstitution, stability matters. The body readily converts cyanocobalamin to active forms through normal metabolic pathways, so bioavailability differences between B12 forms are minimal in individuals with intact conversion capacity.
The pink color serves a practical purpose beyond aesthetics. It provides immediate visual confirmation that the vial contains B12. If you reconstitute a powder and see clear solution instead of pink, you know immediately that either the B12 was omitted or the concentration is extremely low. This visual check prevents confusion when managing multiple vial types. Peptide storage guide recommendations emphasize labeling, but color coding adds an additional safety layer.
Some users report that B12-containing formulations produce less nausea than pure tirzepatide. This observation lacks rigorous controlled trial data but appears consistently in user reports. The mechanism might involve direct antiemetic effects of B12, or it might reflect improved overall metabolic function when vitamin status is maintained. Nausea and fatigue often overlap. If B12 prevents fatigue by supporting cellular energy metabolism, users might better tolerate the gastric slowing effects of tirzepatide. Disentangling these effects requires controlled studies that have not yet been conducted.
Tirzepatide with B12 dosing protocols and titration schedules
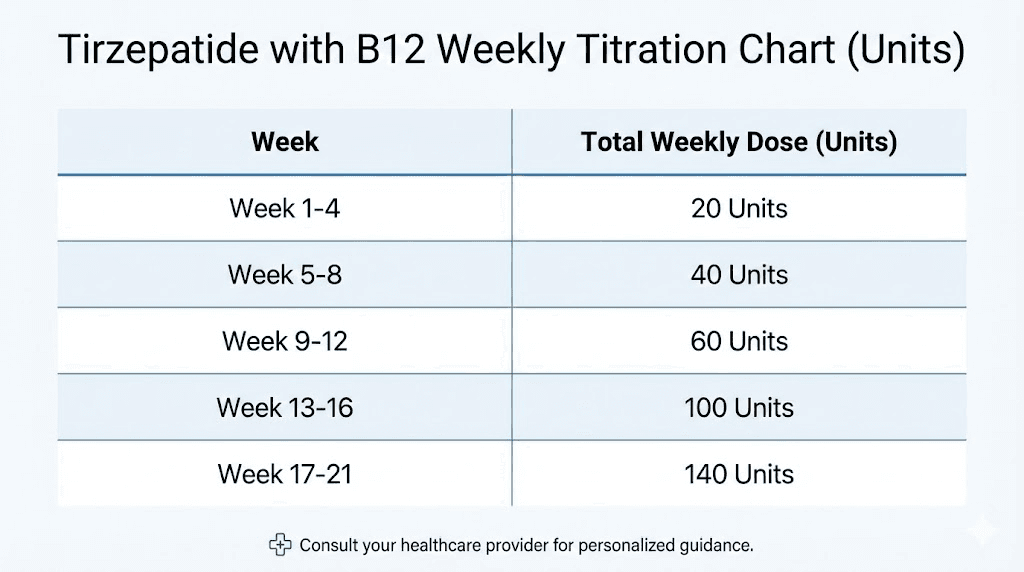
The standard titration protocol for tirzepatide with B12 follows a stepwise escalation over 20-24 weeks. You do not start at the maximum dose. Rapid escalation increases side effect severity, particularly nausea and gastrointestinal distress. The protocol balances effectiveness against tolerability. Tirzepatide dosing guide recommendations typically outline this schedule:
Weeks 1-4: 2.5mg (0.25mL of a 10mg/mL solution, or 25 units on a U-100 syringe). This initiation dose establishes baseline receptor activation. Most users notice some appetite reduction but not the dramatic suppression seen at higher doses. Side effects are generally minimal at this level.
Weeks 5-8: 5mg (0.5mL or 50 units). Appetite suppression becomes more noticeable. Weight loss typically accelerates during this phase. Nausea may emerge, usually mild and often resolving within 1-2 weeks as your body adapts.
Weeks 9-12: 7.5mg (0.75mL or 75 units). This dose produces significant appetite suppression in most users. Food thoughts decrease. Portion sizes naturally shrink. Nausea risk peaks during this transition for users who will experience it.
Weeks 13-16: 10mg (1.0mL or 100 units). Clinical trial data shows substantial weight loss at this level. Many users remain at 10mg as their maintenance dose rather than escalating further.
Weeks 17-20: 12.5mg (1.25mL or 125 units). This intermediate step helps some users transition to the maximum dose without overwhelming side effects. Not all protocols include this tier.
Week 21+: 15mg (1.5mL or 150 units). The maximum approved dose produces the greatest weight loss in clinical trials but also the highest side effect incidence. Not every user requires this dose to achieve their goals.
Dosing flexibility represents one advantage of compounded formulations. If you experience intolerable side effects when jumping from 5mg to 7.5mg, you can try 6mg or 6.5mg instead. Microdosing tirzepatide chart approaches allow even finer adjustments, though evidence supporting extremely low doses remains limited. The insulin syringe allows precise measurement in 1-unit increments, giving exceptional control over your actual dose.
Injection timing follows a weekly schedule. Most users inject on the same day each week to maintain consistent drug levels. The specific day does not matter physiologically. Choose a day that fits your routine. Some users prefer weekends when they can rest if side effects emerge. Others prefer weekdays to maintain normal eating patterns on weekends. How long tirzepatide takes to work is typically 1-2 weeks for appetite effects and 4-8 weeks for measurable weight loss.
If you miss a dose by more than 4 days, the manufacturer recommends skipping that dose and resuming on your next scheduled day. Do not double up. The long half-life means you will still have drug on board even several days late. Doubling doses increases side effect risk substantially. If you miss by less than 4 days, take the missed dose as soon as you remember and continue your regular schedule.
Injection sites include the abdomen, thigh, and upper arm. Rotate sites weekly to prevent lipohypertrophy or lipoatrophy. These injection site reactions occur when you repeatedly inject into the same small area. Tissue changes develop over months. The abdomen offers the most surface area for rotation. Divide it into quadrants and rotate through them. Peptide injections guide techniques emphasize clean site selection at least 2 inches from previous injection points.
Subcutaneous injection depth targets the fat layer under skin but above muscle. Use a short needle, typically 4mm to 8mm length. Insert at a 90-degree angle in areas with sufficient subcutaneous fat like the abdomen. In leaner areas like the thigh, a 45-degree angle may be more appropriate. Pinch the skin to create a fold, insert the needle, inject slowly, wait 5-10 seconds before withdrawing. This waiting period prevents medication from leaking back out.
Some users split their weekly dose into two smaller injections. For example, instead of 10mg once weekly, they inject 5mg twice weekly. The pharmacokinetic rationale is questionable given the 5-day half-life, but some users report better side effect tolerance with split dosing. No clinical trial data supports this approach, and it increases your injection frequency. However, if it improves your adherence and tolerability, the theoretical pharmacokinetic disadvantage may not matter practically.
Dose adjustments during maintenance depend on individual response. If weight loss plateaus at 10mg and you tolerate the medication well, escalating to 12.5mg or 15mg may restart progress. Conversely, if you reach your goal weight at 7.5mg, there is no reason to escalate further. Tirzepatide dose chart tools help track your progression and plan future adjustments based on your response pattern.
Coming off tirzepatide requires planning. Abrupt discontinuation often leads to rapid weight regain as appetite returns to baseline or even overshoots temporarily. Gradual dose reduction over 8-12 weeks may help transition to weight maintenance without medication. However, many users require indefinite therapy to maintain weight loss. This reflects the underlying biology of obesity as a chronic condition requiring ongoing management. Semaglutide withdrawal symptoms apply similarly to tirzepatide: increased appetite, potential weight regain, and sometimes temporary metabolic slowdown.
Clinical benefits of combining tirzepatide with vitamin B12
The weight loss results from tirzepatide with B12 mirror the SURMOUNT clinical trial outcomes. At 72 weeks, participants on 15mg lost an average of 22.5% body weight compared to 2.4% in the placebo group. This represents a 20-percentage-point difference. For a 100kg individual, that translates to 22.5kg lost versus 2.4kg. The difference is substantial and clinically meaningful. Over 90% of participants on the highest dose achieved at least 5% weight loss, the threshold where metabolic improvements begin. More than half lost over 20%, the level where obesity-related complications improve dramatically.
The B12 component specifically addresses quality of life during weight loss. Energy maintenance matters. When you cut calories by 30-40%, fatigue often follows. B12 supports mitochondrial function and cellular energy production through its role in fatty acid oxidation and the citric acid cycle. Adequate B12 status helps maintain energy levels even during caloric restriction. Users report feeling less exhausted during the initial weeks of therapy when B12 is included compared to anecdotal reports from those using tirzepatide alone.
Nausea reduction improves medication adherence. If you feel severely nauseous every week after your injection, you are less likely to continue long-term therapy. The inclusion of B12 appears to blunt nausea severity for some users. While not every person benefits, and while nausea often resolves on its own after 2-3 weeks on a stable dose, any intervention that improves tolerability increases the likelihood that users remain on treatment long enough to achieve meaningful results. Does tirzepatide make you tired is a common concern that B12 may help mitigate through metabolic support.
Metabolic improvements extend beyond weight loss. Tirzepatide significantly improves glycemic control in individuals with type 2 diabetes or prediabetes. HbA1c reductions of 1.5-2.5 percentage points are common. Fasting glucose drops. Insulin resistance improves. These effects stem from the peptide itself, not the B12. However, maintaining B12 status during aggressive weight loss prevents the potential complication of developing new deficiencies while correcting obesity-related metabolic dysfunction. You are optimizing one system without degrading another.
Cardiovascular risk markers improve on tirzepatide. Blood pressure decreases by an average of 5-10 mmHg systolic. Triglycerides drop. LDL cholesterol often improves, though changes vary by individual. Inflammation markers like high-sensitivity C-reactive protein decline. These improvements correlate with weight loss magnitude and appear within months of starting therapy. Peptides for anti-aging applications often focus on these metabolic parameters as key longevity markers.
The combination may support cognitive function during dieting. Severe caloric restriction sometimes impairs concentration and mental clarity, likely due to low glucose availability and potential micronutrient deficiencies. B12 supports myelin synthesis and neurotransmitter production. Maintaining adequate levels theoretically preserves cognitive function during weight loss. While no studies have specifically examined this in tirzepatide users, the biological plausibility is sound. Users anecdotally report clear thinking and sustained focus, contrasting with the brain fog sometimes experienced on very low-calorie diets.
Mood stabilization represents another potential benefit. B12 deficiency associates with depression, irritability, and mood disturbances. Rapid weight loss, hormonal fluctuations, and caloric restriction all stress mood regulatory systems. Ensuring adequate B12 removes one potential contributor to mood problems during this challenging period. Combined with the appetite suppression that reduces food-related stress and decision fatigue, many users report improved mood on tirzepatide despite lower caloric intake.
Body composition changes favor fat loss over muscle loss when compared to equivalent weight loss through diet alone. While some lean mass loss inevitably occurs during significant weight reduction, the rate is typically 20-25% of total weight lost on tirzepatide compared to 30-40% with diet-only approaches. The inclusion of glycine in many formulations may contribute to muscle preservation. Best peptides for muscle growth and preservation require adequate protein intake and resistance training, but the metabolic environment created by tirzepatide appears more muscle-sparing than traditional caloric restriction.
Liver health improves dramatically. Non-alcoholic fatty liver disease (NAFLD) affects a majority of individuals with obesity. Tirzepatide reduces hepatic fat content by 30-50% in most users within 6 months. Liver enzymes normalize. Fibrosis markers improve. This represents reversal of a progressive disease that can lead to cirrhosis and liver failure. The metabolic shifts induced by GLP-1 and GIP activation create conditions where the liver preferentially mobilizes stored triglycerides.
Sleep apnea severity decreases as weight drops. While not a primary endpoint in most trials, observational data shows that many users experience reduced apnea-hypopnea index scores. Sleep quality improves. Daytime sleepiness decreases. These changes correlate with weight loss magnitude, particularly reduction in visceral adiposity and neck circumference. Better sleep further supports metabolic health through improved insulin sensitivity and appetite hormone regulation.
Joint stress reduction occurs mechanically as body weight decreases. Knee and hip pain improve significantly in users with obesity-related osteoarthritis. Every kilogram of weight loss removes approximately 4kg of stress from knee joints during walking. For someone losing 20kg, that represents 80kg less force with each step. This mechanical unloading allows damaged cartilage to recover to some degree and reduces inflammation. Best peptides for joint pain often include BPC-157 and TB-500, but weight reduction through tirzepatide provides complementary benefits through mechanical unloading.
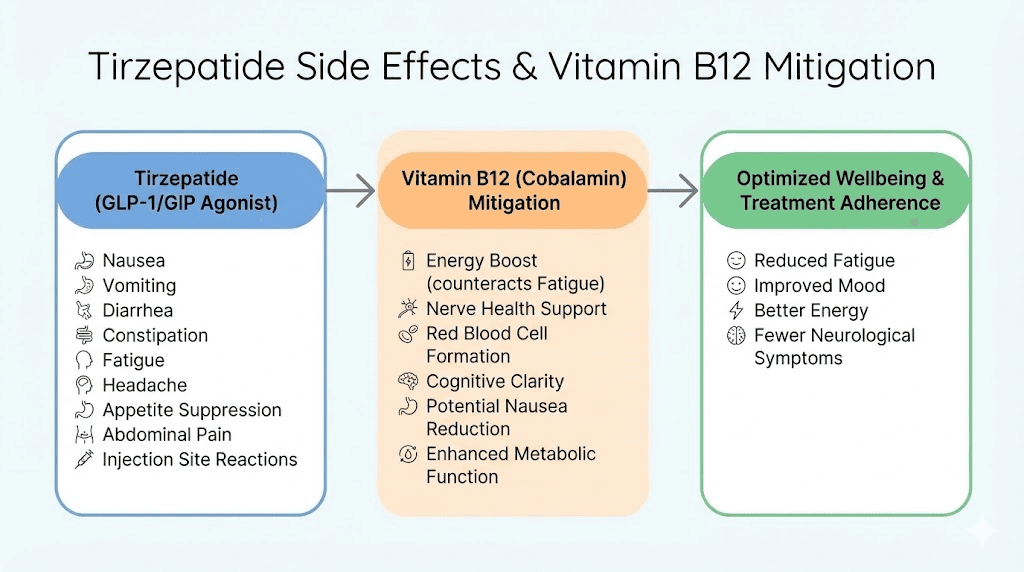
Side effects of tirzepatide with B12 and management strategies
Gastrointestinal side effects dominate the adverse event profile. Nausea occurs in 25-35% of users during dose escalation phases. Most cases rate as mild to moderate. Severe nausea requiring medication discontinuation affects approximately 2-4% of users. The nausea typically emerges within 1-2 days of injection and resolves within 48-72 hours. It tends to decrease with subsequent injections at the same dose as your body adapts. The addition of B12 may reduce severity, though individual responses vary.
Management strategies for nausea include slower titration, eating smaller frequent meals, avoiding trigger foods, staying hydrated, and ginger supplementation. Some users benefit from over-the-counter antiemetics like dimenhydrinate or meclizine taken prophylactically before injections. Prescription options include ondansetron or promethazine for more severe cases. If nausea persists beyond 3-4 weeks on a stable dose, consider reducing to the previous dose level and attempting re-escalation more gradually after 4-6 weeks.
Diarrhea and constipation both occur, sometimes alternating in the same individual. Approximately 15-20% of users experience significant changes in bowel habits. The slowed gastric emptying combined with altered gut motility creates unpredictable effects on colonic transit. Some users develop chronic constipation requiring fiber supplementation, increased water intake, or occasional use of osmotic laxatives like polyethylene glycol. Others experience loose stools or diarrhea, particularly after high-fat meals. Peptides for gut health like BPC-157 target different mechanisms but may complement GLP-1 therapy in managing GI symptoms.
Abdominal pain or discomfort affects 10-15% of users. This manifests as cramping, bloating, or vague upper abdominal fullness. The delayed gastric emptying means food sits in your stomach longer, creating physical distension. Eating smaller portions helps. Avoiding carbonated beverages reduces additional gas volume. If pain becomes severe or persistent, evaluation for other causes like gallstones becomes necessary, as rapid weight loss increases cholecystitis risk.
Fatigue appears in 8-12% of users, though distinguishing medication-induced fatigue from diet-related energy deficits proves difficult. The dramatic reduction in caloric intake necessarily decreases available energy. B12 inclusion theoretically mitigates this, but it cannot completely compensate for a 500-1000 calorie daily deficit. Ensuring adequate protein intake, maintaining hydration, and preserving sleep quality help manage fatigue. If fatigue persists or worsens over time, checking B12 levels, thyroid function, and other nutritional markers is warranted.
Injection site reactions include redness, swelling, or itching at the injection location. These typically resolve within 24-48 hours. True allergic reactions are rare but possible. The pink color from B12 sometimes stains skin temporarily at the injection site.
This is harmless and washes off. Rotating injection sites prevents chronic irritation. If you develop persistent nodules or hard areas under the skin, you may be injecting too superficially or too deeply. Adjust your technique or consult with someone experienced in subcutaneous injections.
Gallbladder issues increase during rapid weight loss regardless of method. Tirzepatide users show elevated risk of cholecystitis and cholelithiasis compared to baseline. The rapid mobilization of cholesterol from adipose stores saturates bile. When bile becomes supersaturated with cholesterol, gallstones precipitate. Symptoms include right upper quadrant pain, particularly after fatty meals, nausea, and sometimes jaundice. If these develop, ultrasound evaluation is necessary. Some practitioners prescribe ursodeoxycholic acid prophylactically during aggressive weight loss to reduce gallstone risk.
Hypoglycemia risk is low in individuals without diabetes not taking other glucose-lowering medications. The glucose-dependent mechanism of tirzepatide means insulin secretion only increases when blood sugar is elevated. However, if you take insulin, sulfonylureas, or meglitinides concurrently, hypoglycemia risk increases substantially. Dose adjustments of these medications are usually necessary when starting tirzepatide. Monitor blood glucose closely during the initiation period if you have diabetes.
Pancreatitis represents a rare but serious potential complication. Lipase elevations occur in some users without symptoms. True acute pancreatitis develops in approximately 0.2-0.4% of users based on clinical trial data. Symptoms include severe persistent abdominal pain radiating to the back, nausea, vomiting, and elevated pancreatic enzymes. If these occur, discontinue tirzepatide immediately and seek medical evaluation. History of pancreatitis represents a relative contraindication to GLP-1 receptor agonist use.
Thyroid concerns arose during drug development due to C-cell hyperplasia and medullary thyroid carcinoma seen in rodent studies. However, humans have far fewer GLP-1 receptors in thyroid C-cells than rodents. Post-marketing surveillance has not identified increased thyroid cancer rates in human users. Nevertheless, personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 (MEN 2) represents an absolute contraindication to tirzepatide use. Routine thyroid monitoring is not required in users without risk factors.
Hair loss sometimes occurs 3-6 months into therapy. This represents telogen effluvium triggered by rapid weight loss and potential nutritional deficiencies, not a direct drug effect. The hair follicles shift from growth phase to resting phase synchronously, leading to increased shedding. GLP-1 hair loss complete guide discusses this phenomenon in detail. Ensuring adequate protein, iron, zinc, and biotin intake helps minimize hair loss. The shedding is usually temporary, with regrowth occurring once weight stabilizes.
Mood changes, including irritability, anxiety, or depressive symptoms, affect a small subset of users. Distinguishing medication effects from psychological responses to dietary restriction and body changes is challenging. If mood disturbances are severe or persistent, evaluation by a mental health professional is appropriate. Some users benefit from continuing therapy with adjunctive psychological support. Others may need to discontinue if symptoms are intolerable.
The role of glycine in tirzepatide formulations
Glycine appears in many compounded tirzepatide formulations as a third active component alongside tirzepatide and B12. This simplest amino acid serves multiple purposes in the context of aggressive weight loss. As a protein building block, glycine supports muscle protein synthesis at a time when dietary protein intake often decreases and catabolic pressures increase. The metabolic state created by significant caloric restriction activates pathways that break down muscle tissue to provide amino acids for gluconeogenesis and other essential processes.
The body can synthesize glycine endogenously from serine and threonine. Under normal conditions, production meets demands. During rapid weight loss, however, demand may exceed endogenous synthesis capacity. Glycine requirements increase because it serves as a precursor for collagen, the most abundant protein in the body. Skin, tendons, ligaments, and connective tissues all require constant collagen turnover. When losing significant weight, skin must remodel and tighten. Providing supplemental glycine theoretically supports this adaptation.
Typical formulations include 50-200mg of glycine per vial. This amount is modest compared to dietary intake from protein-rich foods or standalone glycine supplements, which often use 3-5 grams daily. The inclusion in compounded tirzepatide provides a small but consistent weekly dose. Whether this amount produces meaningful physiological effects compared to placebo has not been rigorously tested. The rationale rests more on theoretical benefit and observed clinical patterns than controlled trial evidence.
Glycine exhibits inhibitory neurotransmitter activity in the central nervous system. It binds to glycine receptors in the spinal cord and brainstem, producing calming effects. Some users report improved sleep quality on formulations containing glycine compared to those without it. The dose in weekly tirzepatide injections is far below the amounts used in sleep studies, however. Those studies typically use 3 grams taken before bed. Whether 50-200mg weekly produces any CNS effects is doubtful from a pharmacological perspective.
Glutathione synthesis depends on glycine availability. Glutathione represents the body primary antioxidant defense system. It neutralizes reactive oxygen species and supports detoxification in the liver. During weight loss, oxidative stress increases as adipose tissue releases stored toxins and cellular turnover accelerates. Supporting glutathione production through glycine supplementation may help manage this increased oxidative burden. Again, the dose in compounded formulations is small, and whether it meaningfully affects glutathione status remains speculative.
Collagen production requires glycine in a 1:3 ratio with other amino acids in the collagen structure. Every third amino acid in collagen is glycine. This creates a unique demand that cannot be met by other amino acids. When skin must contract and tighten after major weight loss, collagen remodeling accelerates. Adequate glycine availability theoretically supports this process. Copper peptides GHK-CU similarly target collagen synthesis through different mechanisms and are sometimes used alongside GLP-1 therapy for skin quality.
The safety profile of glycine supplementation is excellent. As a naturally occurring amino acid consumed in food daily, toxicity concerns are minimal. Even mega-doses in the range of 15-20 grams daily show no serious adverse effects in most individuals. The small amounts in compounded tirzepatide pose essentially no risk. The worst plausible outcome is no benefit, not harm.
Some formulations omit glycine entirely. Whether its inclusion provides meaningful advantage over formulations containing only tirzepatide and B12 remains uncertain. No head-to-head comparison studies exist. User reports suggest that some individuals perceive better muscle preservation and skin quality with glycine-containing formulations, while others notice no difference. Individual variability in glycine metabolism, baseline dietary intake, and genetic factors likely influence response.
The cost difference between formulations with and without glycine is typically negligible. Glycine is an inexpensive ingredient. Its inclusion does not substantially increase manufacturing costs or retail pricing. For most users, choosing a formulation with glycine poses no downside and offers potential upside. If your pharmacy offers both options at similar prices, the formulation containing glycine represents the more comprehensive choice.
Comparing cyanocobalamin and methylcobalamin in tirzepatide formulations
Cyanocobalamin dominates compounded tirzepatide formulations, but methylcobalamin appears in some preparations. These represent different forms of vitamin B12 with distinct chemical structures and metabolic fates. Cyanocobalamin contains a cyanide group attached to the cobalamin molecule. Methylcobalamin contains a methyl group. This structural difference affects stability, bioavailability, and conversion requirements in the body.
Cyanocobalamin demonstrates superior stability in solution and during storage. It resists degradation from light exposure, temperature fluctuations, and oxidation. This stability makes it the preferred form for injectable formulations that users store in home refrigerators for weeks. Methylcobalamin degrades more rapidly under the same conditions. After reconstitution, a methylcobalamin-containing solution loses potency faster than cyanocobalamin. For a weekly injection schedule where vials remain in use for 4-10 weeks, stability matters significantly.
The cyanide component of cyanocobalamin raises concerns for some users. Each molecule contains one cyanide group that is cleaved during metabolism. The amount of cyanide released from B12 supplementation is minuscule compared to dietary exposure from foods like almonds, cassava, and cruciferous vegetables. A 1mg dose of cyanocobalamin releases approximately 20 micrograms of cyanide. Your body detoxifies this easily through rhodanese enzyme systems that convert cyanide to thiocyanate for urinary excretion. Toxicity from B12-derived cyanide has never been documented even with mega-dose supplementation.
Methylcobalamin requires no conversion to become biologically active. It directly participates in methionine synthase reactions that convert homocysteine to methionine. Cyanocobalamin must first be converted to hydroxocobalamin, then to either methylcobalamin or adenosylcobalamin depending on tissue needs. This conversion requires several enzymatic steps. In individuals with genetic polymorphisms affecting these enzymes, methylcobalamin theoretically offers an advantage by bypassing the conversion requirement.
However, healthy individuals with intact B12 metabolism convert cyanocobalamin efficiently. Blood tests show equivalent B12 status whether supplementing with cyanocobalamin or methylcobalamin at equal doses. The bioavailability difference that some supplement marketers emphasize appears minimal in practice for most people. The exception involves individuals with specific genetic variants in the MTHFR, MTR, or MTRR genes that impair B12 metabolism. For these individuals, methylcobalamin may provide superior results.
Cost differs between the two forms. Methylcobalamin costs 3-5 times more than cyanocobalamin in pharmaceutical-grade form. This price difference reflects more complex manufacturing and lower stability requiring careful handling. Compounding pharmacies pass this cost to consumers. If two formulations are otherwise identical except for B12 form, the methylcobalamin version will be more expensive. Whether that premium is justified depends on individual factors.
Sublingual absorption of B12 bypasses some digestive factors that GLP-1 agonists disrupt. Some users choose to take additional B12 sublingually rather than relying solely on the injected form in their tirzepatide. For sublingual use, methylcobalamin may offer advantages because it is absorbed directly into the bloodstream without requiring conversion. Cyanocobalamin taken sublingually still requires conversion after absorption. However, for injected B12, this distinction is less relevant because both forms enter circulation directly.
Anecdotal reports suggest that some users feel more energized on methylcobalamin compared to cyanocobalamin. These subjective reports are impossible to verify without controlled trials. Placebo effects, expectation bias, and individual variability all confound interpretation. Some users may indeed respond better to one form based on genetic or metabolic factors. Others likely experience no difference. Determining your personal response requires trial, ideally with some attempt at blinding to reduce bias.
Adenosylcobalamin represents a third B12 form that rarely appears in tirzepatide formulations. It functions as a cofactor for methylmalonyl-CoA mutase in mitochondrial metabolism. Some supplement proponents argue for combining methylcobalamin and adenosylcobalamin to cover both major B12-dependent pathways. However, the body readily converts one form to another as needed. Providing only one form still supports all B12-dependent processes in individuals with normal conversion capacity.
For most users, cyanocobalamin in compounded tirzepatide provides adequate B12 supplementation. Its stability, lower cost, and extensive safety record make it the practical default choice. If you have known MTHFR mutations, a history of poor response to cyanocobalamin, or strong preference for methylcobalamin, seeking a formulation with that form is reasonable. Otherwise, the form of B12 should not be a primary factor in choosing a tirzepatide preparation. Concentration, total formulation composition, pharmacy reputation, and cost matter more for most users.
Storage and handling requirements for tirzepatide with B12
Lyophilized tirzepatide with B12 powder before reconstitution requires cool, dry storage away from light. Most manufacturers recommend refrigeration at 2-8°C even for unopened vials, though some formulations remain stable at room temperature for short periods. The lyophilization process removes water to create a stable powder, but temperature still affects long-term stability. Freezing should be avoided as ice crystal formation can damage the peptide structure. If a vial accidentally freezes, discard it rather than attempting to use it after thawing.
After reconstitution with bacteriostatic water, the solution must be refrigerated continuously. Tirzepatide fridge storage typically maintains potency for 28-42 days. Beyond this window, degradation accelerates even under refrigeration. The bacteriostatic water contains 0.9% benzyl alcohol to prevent bacterial growth, but this does not prevent peptide degradation. The B12 component is highly stable, but tirzepatide itself slowly degrades in solution.
Temperature excursions compromise potency. If your vial sits at room temperature for several hours, some potency loss occurs. A single brief excursion during travel or a power outage is probably acceptable. Repeated or prolonged warm exposure degrades the peptide significantly. Do peptides expire covers degradation kinetics in detail. For tirzepatide, every 10°C increase in temperature roughly doubles the degradation rate.
Light exposure, particularly UV light, degrades both tirzepatide and B12 to some degree. Store vials in the original packaging or wrap them in aluminum foil if the vial is clear glass. The pink color from cyanocobalamin actually provides some photoprotection by absorbing certain wavelengths. Nevertheless, minimizing light exposure extends shelf life. Keep vials in the refrigerator vegetable drawer or another dark location rather than on a brightly lit shelf.
Sterile technique during reconstitution and withdrawal is critical. Compounded formulations are not preservative-free; the bacteriostatic water contains benzyl alcohol. However, minimizing bacterial introduction still matters. Wipe the vial stopper with an alcohol swab before each needle puncture. Use sterile syringes and needles. Never reuse a syringe. These practices prevent contamination that could lead to injection site infections or systemic illness.
When reconstituting, inject the bacteriostatic water slowly down the side of the vial rather than directly onto the powder. Let the liquid flow gently to dissolve the powder. Swirl gently; do not shake vigorously. Vigorous shaking creates foam and may denature the peptide through mechanical stress. The solution should appear clear to slightly hazy and pink or light red in color. If you see particulates, cloudiness, or discoloration other than pink, do not use the vial.
Drawing up your dose requires care to avoid introducing air bubbles or contaminating the solution. Insert the needle through the stopper, invert the vial, and pull back the plunger to the desired volume. Tap the syringe to move air bubbles toward the needle, then push them back into the vial. Withdraw the needle and check that you have the correct volume. Peptide reconstitution calculator tools help you determine the exact volume needed for your dose based on vial concentration.
Traveling with tirzepatide requires planning. The medication must remain refrigerated, so a cooler with ice packs is necessary for trips longer than a few hours. Medical coolers designed for insulin work well. Include a temperature monitoring device if traveling for extended periods. TSA allows injectable medications in carry-on luggage. Bring a prescription or pharmacy label to avoid questions at security checkpoints. Do not pack tirzepatide in checked luggage where temperature cannot be controlled.
If refrigeration is unavailable during travel, tirzepatide tolerates room temperature for up to 21 days according to manufacturer data for branded products. Compounded formulations may have different stability, so verify with your pharmacy. Once exposed to room temperature for extended periods, the vial should not be returned to refrigerated storage and used beyond the 21-day window. Track your storage conditions carefully when traveling.
Disposal of used vials and syringes follows standard sharps safety protocols. Needles and syringes go into a puncture-resistant sharps container. When the container is three-quarters full, seal it and dispose according to local regulations. Many pharmacies accept sharps containers for disposal. Never throw loose needles in household trash. Empty vials can be disposed in regular trash after ensuring no medication remains and the vial is sealed in a bag to prevent glass breakage.
Expiration dates on vials indicate the manufacturer guaranteed potency window. Using medication slightly past expiration is probably safe but may be less effective due to degradation. Using medication significantly past expiration is inadvisable. Potency decreases over time even under optimal storage. If you have old vials, the worst likely outcome is reduced effectiveness rather than toxicity, but the economic waste and suboptimal results make using expired peptides a poor choice.
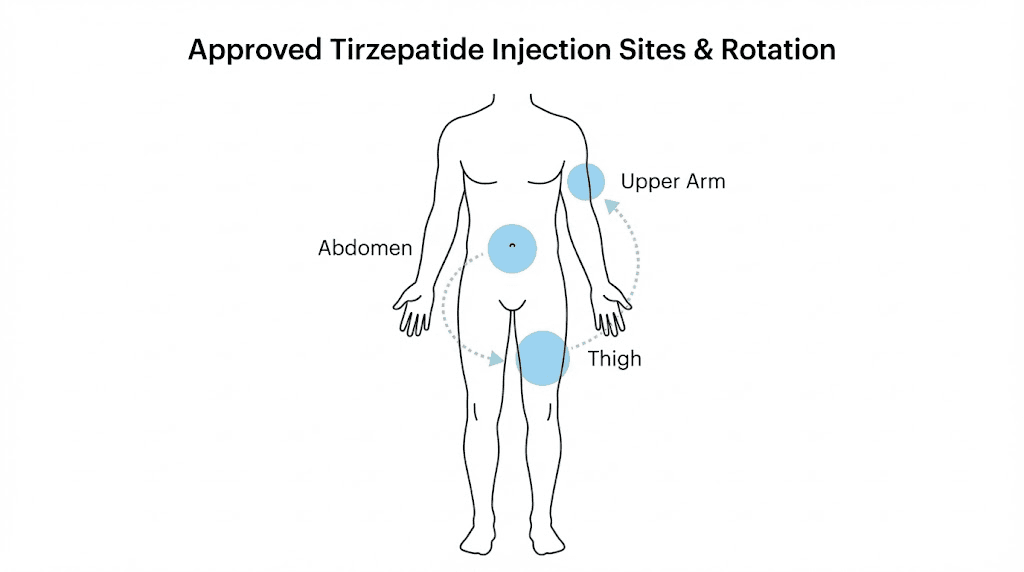
Injection technique and site rotation strategies
Subcutaneous injection places medication into the fat layer between skin and muscle. This route allows slow, sustained absorption into the bloodstream. The technique differs from intramuscular injection, which uses longer needles and deeper penetration. For tirzepatide, subcutaneous administration is required. Intramuscular injection would alter absorption kinetics and is not studied or approved.
Selecting an injection site involves identifying areas with adequate subcutaneous fat. The abdomen works well for most users. Avoid the area within 2 inches of the navel. The outer abdomen, particularly the lower quadrants, contains substantial fat even in relatively lean individuals. The thigh, specifically the front and outer portions of the upper leg, provides another option. Avoid the inner thigh where large blood vessels run near the surface. The upper arm, specifically the back or outer portion where you cannot easily reach, is technically suitable but difficult to self-inject without assistance.
Site rotation prevents lipohypertrophy and lipoatrophy. Lipohypertrophy involves fat accumulation at injection sites due to the lipogenic effects of repeated injections in the same small area. The tissue becomes lumpy and firm. Lipoatrophy is the opposite: fat tissue breakdown creating divots or depressions. Both conditions are unsightly and can affect medication absorption. Rotating sites prevents these complications. Peptide injections guide recommendations typically suggest at least 1 inch separation from previous injection points and systematic rotation through multiple sites.
A practical rotation strategy divides the abdomen into quadrants: upper right, upper left, lower right, lower left. Inject in a different quadrant each week, cycling through all four before returning to the first. This provides 4 weeks between injections in the same area. For users injecting more frequently or wanting more sites, subdivide further or alternate between abdomen and thigh. Mark injection sites on a body diagram or use a tracking app to maintain systematic rotation.
Preparing the injection site involves cleaning with an alcohol swab. Wipe the area and allow it to air dry completely. Alcohol remaining on the skin causes stinging when the needle penetrates. The drying process takes 30-60 seconds. During this time, prepare your syringe, verify the dose, and perform a final visual check of the solution for particles or discoloration.
Pinching the skin creates a fold that ensures subcutaneous rather than intramuscular injection. Use your non-dominant hand to grasp a fold of skin and fat. Pinch firmly enough to create a distinct fold but not so hard that it hurts. The fold should feel soft and mobile, confirming you have grasped fat rather than muscle. If the area feels firm and resistant, you may be pinching muscle; choose a different site with more subcutaneous tissue.
Needle insertion uses either a 90-degree angle or 45-degree angle depending on the amount of subcutaneous fat. For areas with substantial fat like the abdomen in most adults, insert at 90 degrees straight down. For leaner areas or very thin individuals, a 45-degree angle prevents penetrating through the fat layer into muscle. Insert with a quick, smooth motion. Hesitating or inserting very slowly increases discomfort. The needle should slide in easily with minimal resistance.
Aspiration is not necessary for subcutaneous injections. Older injection techniques taught pulling back the plunger to check for blood before injecting. Current guidelines indicate this is unnecessary for subcutaneous administration in areas like the abdomen and thigh. The risk of inadvertent intravenous injection is extremely low in these locations. Aspirating may actually increase pain by creating tissue movement around the needle.
Injecting the medication should proceed slowly and steadily. Push the plunger smoothly over 5-10 seconds. Rapid injection increases discomfort and may cause the medication to leak back out of the injection site. Some users report a burning or stinging sensation during injection, particularly with larger volumes. This is normal and resolves within seconds after completing the injection.
Waiting before withdrawing the needle prevents medication leakage. After fully depressing the plunger, count to 10 slowly while keeping the needle in place. This allows time for the injected solution to disperse into the surrounding tissue rather than tracking back up the needle path. After waiting, withdraw the needle smoothly at the same angle you inserted it. Do not twist or angle the needle during withdrawal.
Applying gentle pressure to the injection site with a clean cotton ball or gauze manages any bleeding. Minor bleeding is normal and typically stops within seconds. Do not rub the area vigorously, as this may increase bruising. If bleeding continues beyond 1-2 minutes, maintain pressure longer. Persistent bleeding or expanding bruising may indicate that you hit a small blood vessel, which is generally harmless but can be minimized with proper technique.
Needle length selection affects injection depth. Common lengths for subcutaneous injection range from 4mm to 8mm. Most users do well with 6mm needles, which penetrate deep enough to reach subcutaneous fat but not so deep as to risk intramuscular injection in typical sites. Very lean individuals may prefer 4mm needles. Those with substantial subcutaneous fat can use 8mm needles safely. How to reconstitute peptides and administer them properly includes needle selection based on body composition.
Needle gauge affects injection comfort. Higher gauge numbers indicate thinner needles. A 30-gauge needle is thinner than a 27-gauge needle. Thinner needles cause less discomfort during insertion. For tirzepatide, 27-31 gauge needles work well. Insulin syringes typically come in 28-31 gauge, all suitable for comfortable subcutaneous injection. Thinner needles require slightly more pressure to push through skin but cause less tissue trauma.
Who should consider tirzepatide with B12 and who should avoid it
Ideal candidates for tirzepatide with B12 include adults with obesity (BMI ≥30) or overweight with comorbidities (BMI ≥27 with conditions like type 2 diabetes, hypertension, or dyslipidemia). The FDA approval criteria for branded tirzepatide weight loss indication follow these parameters. Compounded versions are prescribed under similar guidelines, though individual practitioners may vary. The medication works best when combined with dietary modifications and increased physical activity. It is not a standalone solution but rather a tool that makes behavioral changes more achievable by reducing appetite and cravings.
Individuals with type 2 diabetes benefit particularly from tirzepatide due to its dual effects on weight and glycemic control. If you are struggling to achieve HbA1c targets despite metformin and lifestyle modifications, tirzepatide offers substantial improvement potential. The weight loss secondarily improves insulin sensitivity, creating a beneficial cycle. Many users reduce or eliminate other diabetes medications as their metabolic health improves on tirzepatide.
Those who have struggled with repeated diet attempts and weight regain are good candidates. The appetite suppression breaks the cycle of restriction followed by rebound overeating. The medication provides a neurohormonal reset that allows you to adhere to lower caloric intake without the constant struggle against hunger that characterizes traditional dieting. Best peptides for weight loss comparisons show tirzepatide at the top for magnitude of effect and quality of evidence.
People with cardiovascular risk factors see meaningful improvements. If you have elevated blood pressure, unfavorable lipid profiles, or concerning inflammatory markers, tirzepatide addresses multiple risk factors simultaneously through weight reduction and direct metabolic effects. Cardiovascular outcome trials for tirzepatide are ongoing, but the favorable effects on intermediate markers suggest probable long-term cardiovascular benefit.
Individuals concerned about nutritional deficiencies during weight loss benefit from the B12 inclusion. If you have a history of B12 deficiency, malabsorption issues, or are vegetarian/vegan with marginal B12 status, the weekly B12 injection provides protection during the high-risk period of rapid weight loss. The guaranteed delivery via injection bypasses the absorption issues that tirzepatide creates in the GI tract.
People who experience significant nausea on GLP-1 receptor agonists may tolerate tirzepatide with B12 better than formulations without it. If you previously tried semaglutide and discontinued due to nausea, the addition of B12 might allow successful treatment. Switching from tirzepatide to semaglutide or vice versa is common when optimizing individual response and tolerability.
Absolute contraindications include personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 (MEN 2). The black box warning on GLP-1 receptor agonists stems from animal studies showing C-cell hyperplasia and medullary thyroid cancer in rodents. While human risk appears minimal based on post-marketing surveillance, the contraindication remains. If you or a first-degree relative has been diagnosed with these conditions, do not use tirzepatide.
Pregnancy and breastfeeding represent contraindications. Tirzepatide crosses the placenta and may appear in breast milk. Animal studies show embryo-fetal toxicity. The weight loss effects are inappropriate during pregnancy and lactation when adequate nutrition is critical. Women of childbearing age should use contraception while on tirzepatide and discontinue the medication at least 2 months before attempting conception to allow complete washout given the long half-life.
Active or recent pancreatitis contraindicates use. While causality remains debated, GLP-1 receptor agonists associate with increased pancreatitis risk in some populations. If you have a history of pancreatitis, the risk-benefit calculation changes unfavorably unless other factors strongly favor use and careful monitoring is in place. Discuss this thoroughly with your prescribing physician before proceeding.
Severe gastroparesis or other serious GI motility disorders make tirzepatide problematic. The medication further slows gastric emptying, which could exacerbate pre-existing motility issues. If you already struggle with delayed emptying, adding a drug that intensifies this is likely to worsen symptoms. Peptides for gut health like BPC-157 target different mechanisms and might be more appropriate for individuals with existing GI pathology.
History of severe hypersensitivity reactions to tirzepatide or any formulation component requires avoidance. If you experienced anaphylaxis, angioedema, or other serious allergic reactions during previous exposure, do not attempt rechallenge. Mild injection site reactions are not contraindications, but systemic allergic responses are.
End-stage renal disease or severe hepatic impairment requires caution. While tirzepatide does not undergo significant renal clearance, limited safety data exist in severe kidney disease. Similarly, data in advanced liver disease are sparse. If you have cirrhosis or creatinine clearance below 15 mL/min, alternative weight loss approaches may be safer pending more robust safety data in these populations.
Active eating disorders represent a relative contraindication. Bulimia nervosa or binge eating disorder may worsen in some individuals on appetite-suppressing medications. The altered relationship with food and hunger cues can be destabilizing. If you have a history of eating disorders, pursue tirzepatide only with concurrent psychological support and close monitoring for disorder recurrence.
Age considerations include lack of approval for individuals under 18. Pediatric obesity is common, but safety and efficacy data for tirzepatide in children and adolescents are limited. Off-label use in this population should involve pediatric endocrinologists with expertise in weight management. For adults over 65, no dose adjustment is required, but careful monitoring for adverse effects is prudent as older adults often have multiple comorbidities and medications that increase complication risk.

Monitoring nutrient status while on tirzepatide with B12
Baseline laboratory assessment before starting tirzepatide should include B12 levels, complete blood count (CBC), comprehensive metabolic panel (CMP), lipid panel, and HbA1c if you have diabetes or prediabetes. These baseline values provide comparison points for monitoring changes during therapy. If you have pre-existing deficiencies, addressing them before starting aggressive weight loss improves outcomes and reduces complication risk.
B12 monitoring during therapy ensures the supplementation is adequate. Most practitioners check B12 levels at 3-6 months after initiating tirzepatide and then every 6-12 months during maintenance. Normal B12 ranges typically fall between 200-900 pg/mL, though functional B12 status is better assessed by methylmalonic acid (MMA) and homocysteine levels. If B12 is low-normal (<400 pg/mL) despite weekly B12 injections, increasing the B12 dose or adding oral supplementation may be necessary.
Complete blood count tracks for anemia related to nutritional deficiencies. B12 deficiency causes macrocytic anemia with elevated mean corpuscular volume (MCV). Iron deficiency from reduced meat intake or increased menstrual losses in women losing weight rapidly causes microcytic anemia with low MCV. Checking CBC every 6 months identifies these issues before symptoms develop. Early intervention prevents progression to severe anemia with fatigue and other complications.
Vitamin D levels often decline during weight loss despite sun exposure because vitamin D is fat-soluble and sequesters in adipose tissue. As you lose fat mass rapidly, stored vitamin D may be mobilized but also excreted or metabolized. Check 25-hydroxyvitamin D levels at baseline and every 6 months. Supplement with vitamin D3 to maintain levels above 30 ng/mL, ideally 40-60 ng/mL for optimal health.
Folate deficiency can develop alongside B12 deficiency or independently. Reduced intake of folate-rich vegetables and legumes during caloric restriction increases risk. Folate and B12 work together in one-carbon metabolism. Deficiency in either causes similar hematologic changes. Check red blood cell folate or serum folate at baseline and during follow-up. Supplementation is inexpensive and safe if levels drop.
Liver function tests monitor for rare hepatotoxicity and track improvements in fatty liver disease. Tirzepatide typically improves liver enzymes as hepatic fat content decreases.
If ALT or AST rise during therapy, investigate other causes like medication interactions or viral hepatitis. Worsening liver function on tirzepatide is unusual and warrants thorough evaluation.
Lipase monitoring remains controversial. Some practitioners check lipase periodically to screen for subclinical pancreatitis. Elevations up to 3 times the upper limit of normal without symptoms are common and usually benign. However, elevations above this threshold or any elevation accompanied by abdominal pain requires investigation. If you have risk factors for pancreatitis like gallstones or heavy alcohol use, more frequent monitoring may be appropriate.
Thyroid function testing is not routinely necessary unless you have pre-existing thyroid disease or develop symptoms suggestive of thyroid dysfunction. TSH and free T4 at baseline provide a reference. Repeat testing only if symptoms develop. Despite theoretical concerns from rodent studies, tirzepatide does not appear to affect thyroid function in humans at therapeutic doses.
Iron studies including serum iron, ferritin, and transferrin saturation identify iron deficiency before anemia develops. Reduced meat consumption and the period of rapid weight loss both increase iron deficiency risk, particularly in menstruating women. Check iron studies at baseline and every 6-12 months. If ferritin falls below 30 ng/mL, consider iron supplementation even if hemoglobin remains normal.
Calcium and magnesium levels sometimes decline during aggressive weight loss. Reduced dairy intake affects calcium. GI side effects causing diarrhea can deplete magnesium. Check these minerals if you develop symptoms like muscle cramps, palpitations, or tetany. Supplementation is straightforward and effective if deficiencies are identified.
Protein status can be assessed indirectly through albumin and prealbumin levels. Significant drops suggest inadequate protein intake relative to losses during rapid weight loss. Safest peptides for muscle growth and preservation require adequate dietary protein, typically 1.2-1.6 g/kg of ideal body weight during aggressive weight loss. If laboratory markers suggest protein deficiency, increasing intake is critical to prevent muscle wasting.
Bone density monitoring via DEXA scan is appropriate for individuals at risk of osteoporosis. Rapid weight loss, particularly in postmenopausal women, accelerates bone density loss. Baseline DEXA before starting tirzepatide and follow-up after significant weight loss (>20%) helps assess bone health. If density declines significantly, interventions include calcium and vitamin D optimization, weight-bearing exercise, and sometimes pharmacologic therapy.
Tracking these parameters requires coordination with your healthcare provider. SeekPeptides members access detailed monitoring protocols and interpretation guides to discuss with their physicians. Regular laboratory assessment transforms tirzepatide therapy from a simple weight loss intervention into a comprehensive metabolic optimization program that addresses nutritional status, metabolic health, and long-term wellbeing simultaneously.
Comparing tirzepatide with B12 to other GLP-1 formulations
Tirzepatide with B12 differs from semaglutide with B12 primarily through the dual GIP/GLP-1 mechanism. Semaglutide with B12 complete guide details that formulation, which uses a single GLP-1 receptor agonist approach. Head-to-head trials show tirzepatide producing 3-5 percentage points greater weight loss at comparable timepoints. SURMOUNT-1 data with tirzepatide show 22.5% weight loss at 72 weeks on the highest dose. STEP trial data with semaglutide show approximately 15-17% weight loss at similar duration on 2.4mg weekly.
The side effect profiles differ slightly between the two medications. Nausea incidence is similar, affecting 25-35% of users on both drugs. However, some users report better tolerability with tirzepatide, possibly due to the GIP component modulating GLP-1 effects. Others tolerate semaglutide better. Individual variation dominates, making it difficult to predict which medication a specific person will tolerate best. If one causes intolerable side effects, trying the other is reasonable. Switching from tirzepatide to semaglutide guidelines help navigate transitions between medications.
Dosing schedules differ. Semaglutide reaches maximum approved dose (2.4mg) faster than tirzepatide (15mg). The semaglutide escalation typically completes in 16-20 weeks. Tirzepatide escalation takes 20-24 weeks. However, the slower tirzepatide titration may improve tolerability by allowing more gradual adaptation. Some practitioners extend titration even further for particularly sensitive individuals.
Cost comparisons vary by source. Brand name versions (Wegovy for semaglutide, Zepbound for tirzepatide) both list at approximately $1,000-1,300 per month without insurance. Compounded versions of both are substantially cheaper, typically $200-400 per month depending on pharmacy and dose. The cost difference between compounded semaglutide and compounded tirzepatide is usually minimal. Given the superior efficacy of tirzepatide, it represents better value at similar prices.
Retatrutide represents a newer option that adds glucagon receptor agonism to GLP-1 and GIP activation, creating a triple agonist. Early trial data show even greater weight loss than tirzepatide, approaching 24-25% at 48 weeks. However, retatrutide is not yet FDA approved and availability is limited to clinical trials and research settings. Retatrutide vs semaglutide comparisons highlight the progression toward more potent multi-agonist approaches.
Cagrilintide combined with semaglutide (marketed as CagriSema) represents another emerging option. Cagrilintide is an amylin analog that complements GLP-1 effects. The combination shows approximately 15-18% weight loss, falling between semaglutide alone and tirzepatide. Cagrilintide and semaglutide data suggest the combination is well-tolerated, though the convenience of a single-agent therapy like tirzepatide may appeal to many users.
Oral formulations of GLP-1 agonists exist but show reduced efficacy compared to injectable versions. Oral tirzepatide complete guide discusses the challenges of delivering peptides through the GI tract. Bioavailability of oral formulations is typically 1-3% compared to near 100% for subcutaneous injection. To achieve comparable effects, oral doses must be 30-50 times higher, creating cost and side effect challenges. Injectable remains the standard route for tirzepatide.
The addition of B12 to either semaglutide or tirzepatide formulations provides similar benefits: nausea reduction and deficiency prevention. Whether you choose semaglutide with B12 or tirzepatide with B12, the B12 component serves the same purposes. The choice between medications should rest primarily on efficacy data, tolerability, availability, and cost rather than the B12 inclusion, which is valuable in both formulations.
Some compounded formulations include additional ingredients beyond B12 and glycine. Niacinamide appears in some preparations.
Tirzepatide niacinamide complete guide explains the rationale: niacinamide (vitamin B3) supports cellular energy metabolism and may enhance insulin sensitivity independently. The evidence for meaningful additional benefit from niacinamide inclusion is limited, but like glycine, it adds minimal cost and no significant risk.
Brand versus compounded formulations involve tradeoffs. Brand products undergo extensive FDA review and quality control. Compounded products offer customization and lower cost but less regulatory oversight. For most users, reputable compounding pharmacies produce formulations that are bioequivalent to brand products. However, the possibility of underdosing, contamination, or formulation errors exists in compounded products. Choose pharmacies with good reputations, third-party testing, and proper licensing.
Frequently asked questions about tirzepatide with B12
How long does it take for tirzepatide with B12 to start working for weight loss?
Appetite suppression typically begins within 1-2 weeks of your first injection. The effect intensifies as you escalate doses over the following months. Measurable weight loss usually becomes apparent within 4-6 weeks. The rate of loss accelerates during the dose escalation phase and typically peaks around weeks 16-32 when you reach higher maintenance doses. How long tirzepatide takes to suppress appetite varies by individual, but most users report noticeable hunger reduction within the first two injections.
Can I take additional B12 supplements while using tirzepatide with B12?
Yes. B12 supplementation is extremely safe with no established upper limit for toxicity. The amount in compounded tirzepatide (typically 0.5-1mg weekly) is well above the RDA but may not fully compensate for absorption issues and increased demands during rapid weight loss. Adding oral B12, sublingual B12, or additional B12 injections poses no risk. If laboratory testing shows low or low-normal B12 levels despite weekly injections, additional supplementation is appropriate and beneficial.
Why is the tirzepatide solution pink instead of clear?
The pink or light red color comes from cyanocobalamin (vitamin B12). Pure tirzepatide solutions without B12 are clear or slightly cloudy. The pink color serves as a visual indicator that your formulation contains B12. If you reconstitute a vial and see clear solution, either the B12 was omitted or the concentration is extremely low. The intensity of the pink color correlates with B12 concentration.
Do I need to refrigerate tirzepatide with B12 after mixing?
Yes. After reconstitution, the solution must be stored in a refrigerator at 2-8°C. Tirzepatide refrigeration requirements are non-negotiable for maintaining potency. The peptide degrades rapidly at room temperature. Brief temperature excursions during transport from the refrigerator to injection are fine, but the vial should not sit at room temperature for extended periods. Use a medical cooler when traveling.
Can I use tirzepatide with B12 if I have the MTHFR gene mutation?
Yes. MTHFR mutations affect folate metabolism and can impact methylation pathways including B12 activation. If you have MTHFR mutations and previous poor response to cyanocobalamin supplements, you might prefer a formulation containing methylcobalamin instead. However, most individuals with MTHFR variants still convert cyanocobalamin adequately. If you know you have MTHFR mutations, discuss B12 form with your prescriber and consider methylcobalamin if available.
How do I know if the B12 in my tirzepatide is working?
Laboratory testing provides objective confirmation. Check B12 levels at 3-6 months after starting therapy. Levels should be in the normal or high-normal range if supplementation is adequate. Subjectively, adequate B12 manifests as maintained energy levels, absence of neurological symptoms like numbness or tingling, and normal cognitive function. If you develop fatigue, memory problems, or neuropathy despite weekly B12 injections, check B12, methylmalonic acid, and homocysteine levels to assess functional B12 status.
Can I split my weekly tirzepatide dose into two smaller injections?
While not studied in clinical trials, some users report better tolerability splitting their weekly dose. For example, 7.5mg could be divided into two 3.75mg injections 3-4 days apart. The pharmacokinetic rationale is questionable given tirzepatide 5-day half-life. Blood levels remain relatively stable throughout the week even with once-weekly dosing. However, if split dosing improves your adherence and reduces side effects, the practical benefit may outweigh theoretical pharmacokinetic concerns. Discuss with your prescriber before modifying the standard weekly schedule.
What should I do if I miss a dose of tirzepatide?
If you remember within 4 days of your scheduled injection, take the dose as soon as possible and resume your regular weekly schedule. If more than 4 days have passed, skip that dose and inject on your next scheduled day. Do not double up to make up for a missed dose. The long half-life means you will still have therapeutic drug levels even several days late. Semaglutide dosage calculator principles apply similarly to tirzepatide regarding missed dose management.
Is glycine necessary in tirzepatide formulations or just B12?
Glycine is not necessary in the same way B12 is. While B12 directly addresses absorption issues created by tirzepatide effects on the GI tract, glycine provides theoretical muscle preservation benefits. Many successful formulations contain only tirzepatide and B12 without glycine. If your pharmacy offers both options at similar prices, choosing the formulation with glycine adds potential benefit without downside. If glycine inclusion increases cost significantly, it is probably not worth the premium given the limited evidence for meaningful benefit at the doses used.
How long can I stay on tirzepatide with B12 safely?
Clinical trial data extend to 2-3 years of continuous use with no significant safety concerns emerging. Real-world use of similar GLP-1 receptor agonists like liraglutide and semaglutide extends beyond 5 years in some patients. For weight management, many users require indefinite therapy to maintain weight loss. This reflects obesity as a chronic metabolic condition rather than an acute problem that resolves permanently. Long-term safety monitoring includes periodic laboratory testing, but the medication appears safe for extended use in appropriate candidates.
Will I regain weight if I stop taking tirzepatide with B12?
Most users experience some weight regain after discontinuation. The magnitude varies based on lifestyle factors and individual biology. On average, users regain approximately 50-70% of lost weight within 12 months of stopping. Some maintain their weight through sustained dietary and exercise modifications. Others regain all the weight lost. This pattern is not unique to tirzepatide but reflects the biology of obesity and metabolic adaptation. Gradual dose reduction rather than abrupt cessation may improve weight maintenance, though evidence is limited. Many users choose to continue therapy long-term at maintenance doses rather than discontinuing.
Can I drink alcohol while taking tirzepatide with B12?
Moderate alcohol consumption is generally acceptable. However, alcohol increases nausea risk, and the combination with tirzepatide might be poorly tolerated, particularly during the first few weeks on a new dose. Alcohol provides empty calories that work against weight loss goals. Additionally, tirzepatide may affect how quickly alcohol is absorbed due to delayed gastric emptying, potentially altering your tolerance. Some users report feeling intoxicated more quickly than before starting therapy. Limit alcohol to minimal amounts and pay attention to your individual response.
Does tirzepatide with B12 interact with other medications?
Tirzepatide delays gastric emptying, which can affect the absorption of oral medications. Medications requiring rapid absorption like pain relievers may work more slowly. Medications with narrow therapeutic windows may need dose adjustments. If you take oral contraceptives, antibiotics, or other time-sensitive medications, discuss timing with your prescriber. The most significant interaction involves other glucose-lowering drugs. If you take insulin, sulfonylureas, or meglitinides, your doses will likely require reduction to prevent hypoglycemia. Monitor blood glucose closely when starting tirzepatide if you have diabetes.
External resources for tirzepatide and B12 research
Clinical trial data for tirzepatide weight loss efficacy comes primarily from the SURMOUNT trial series published in The New England Journal of Medicine. The SURMOUNT-1 trial compared tirzepatide at 5mg, 10mg, and 15mg doses against placebo in adults with obesity. Results showed dose-dependent weight loss reaching 22.5% at the highest dose over 72 weeks. Access the full publication through NEJM.org for detailed methodology, adverse event data, and subgroup analyses.
The American Diabetes Association publishes comprehensive guidelines on GLP-1 receptor agonist use for type 2 diabetes management. These guidelines synthesize evidence from multiple trials and provide recommendations on patient selection, dosing, monitoring, and combination therapy strategies. The Standards of Medical Care in Diabetes, updated annually, includes dedicated sections on incretin-based therapies relevant for understanding tirzepatide pharmacology and clinical application.
PubMed provides access to hundreds of research papers examining B12 absorption, deficiency risk factors, and supplementation strategies during weight loss. Search terms like "vitamin B12 GLP-1 agonist," "cyanocobalamin absorption gastric emptying," and "B12 deficiency obesity surgery" yield relevant papers. While much research focuses on bariatric surgery patients, the mechanisms of B12 deficiency during rapid weight loss overlap significantly with GLP-1 therapy.
The FDA website hosts prescribing information for Mounjaro (tirzepatide for diabetes) and Zepbound (tirzepatide for weight loss). These documents detail clinical pharmacology, indications, contraindications, warnings, adverse reactions, and drug interactions based on the complete clinical development program. While compounded formulations differ from branded products, the core safety and efficacy data derive from these FDA-reviewed documents.
Obesity Medicine Association resources include position statements on pharmacotherapy for obesity, practice guidelines, and continuing education materials. The organization focuses specifically on obesity as a disease requiring medical management. Their guidelines help contextualize where tirzepatide fits within comprehensive obesity treatment algorithms including lifestyle intervention, behavioral therapy, pharmacotherapy, and bariatric surgery.
The Endocrine Society publishes clinical practice guidelines on obesity management that include sections on incretin-based therapies. These guidelines address patient selection, expected outcomes, monitoring parameters, and integration with other obesity treatments. The evidence-based approach synthesizes data from multiple drug classes to provide practical clinical recommendations.
Understanding the metabolic changes during tirzepatide therapy
Your body undergoes profound metabolic shifts during tirzepatide therapy. The initial weeks involve adaptation to reduced caloric intake and altered hunger signaling. Ghrelin, the primary hunger hormone, typically rises during caloric restriction. This drives increased appetite and slowed metabolism, making sustained weight loss difficult through diet alone. Tirzepatide suppresses this compensatory ghrelin increase through central nervous system effects, breaking the typical hunger rebound cycle.
Insulin sensitivity improves rapidly as weight loss begins. Even modest weight reduction of 5-7% significantly enhances insulin receptor function in muscle and liver tissue. As you continue losing weight, improvements compound. Many users with prediabetes see glucose tolerance normalize. Those with type 2 diabetes often reduce medication requirements or achieve remission. The GLP-1 and GIP effects on pancreatic beta cells may also improve insulin secretion capacity beyond the effects of weight loss alone.
Leptin levels decline as adipose tissue mass decreases. Leptin signals energy sufficiency to the brain. During traditional dieting, falling leptin triggers hunger and metabolic slowdown. The GLP-1 receptor activation partially bypasses leptin pathways, maintaining appetite suppression even as leptin drops. This represents a key mechanism by which tirzepatide produces greater weight loss than diet alone despite similar caloric restriction.
Resting metabolic rate typically decreases during weight loss as your body adapts to lower energy intake and reduced body mass. A smaller body requires fewer calories to maintain. Additionally, adaptive thermogenesis slows metabolism beyond what body composition changes predict. Tirzepatide does not prevent this metabolic adaptation entirely, but the degree of slowdown may be less than with diet-only weight loss. Preserving lean body mass through adequate protein and resistance training helps maintain metabolic rate.
Fat oxidation pathways shift during the aggressive weight loss phase. Your body mobilizes stored triglycerides from adipose tissue at an accelerated rate. Free fatty acids enter circulation and are taken up by liver, muscle, and other tissues for beta-oxidation. This process requires adequate B vitamins including B12 for proper function. The mitochondrial machinery that burns fat depends on B vitamin cofactors. Supplementation ensures this system operates efficiently during peak fat mobilization.
Ketone production may increase moderately even if you are not following a ketogenic diet. The combination of reduced insulin levels and increased fat oxidation creates conditions favorable for hepatic ketogenesis. Most users do not enter deep ketosis, but mild elevation of beta-hydroxybutyrate is common. This provides an alternative fuel source for the brain and may contribute to appetite suppression. Testing ketones is not necessary unless you are specifically pursuing ketogenic dieting alongside tirzepatide therapy.
Adipokine profiles improve as visceral adiposity decreases. Adiponectin, an anti-inflammatory adipokine, typically rises. This improves insulin sensitivity and cardiovascular health. Inflammatory adipokines like TNF-alpha and IL-6 decline as dysfunctional adipose tissue is metabolized. The reduction in chronic low-grade inflammation explains many of the metabolic health improvements seen beyond simple weight loss numbers. Inflammation peptides target similar pathways through different mechanisms.
Gut microbiome composition changes in response to altered diet and medication effects. GLP-1 receptor agonists modify gut motility, which affects bacterial populations. The dietary changes you make, typically involving more protein and fiber and less simple carbohydrates, further shift the microbiome. These changes may contribute to metabolic improvements independently of weight loss. Probiotic supplementation during tirzepatide therapy is not specifically studied but may support healthy microbiome adaptation.
Hormonal changes extend beyond metabolic hormones. In women, estrogen metabolism shifts as adipose tissue decreases. Fat tissue contributes to estrogen production, particularly after menopause. Significant fat loss can affect estrogen levels. Some women experience changes in menstrual patterns, hot flashes, or mood during aggressive weight loss. Peptides for menopause symptom management may complement tirzepatide therapy in perimenopausal and postmenopausal women.
In men, testosterone levels often improve as weight decreases. Obesity associates with reduced testosterone due to increased aromatase activity in adipose tissue converting testosterone to estrogen. As fat mass decreases, this conversion declines and testosterone levels normalize. Many men report improved energy, libido, and muscle mass maintenance as testosterone rises during tirzepatide therapy. Peptides for men often target testosterone optimization through different mechanisms.
Thyroid hormone conversion may be affected by caloric restriction. T3 (the active thyroid hormone) production can decrease during prolonged dieting as a metabolic adaptation. While tirzepatide does not directly affect thyroid function, the caloric deficit it produces creates similar metabolic pressure. Monitoring thyroid function through TSH and free T3 testing helps identify whether thyroid supplementation might benefit users experiencing persistent fatigue or metabolic slowdown despite appropriate tirzepatide dosing.
Cortisol patterns sometimes change during weight loss. Chronic caloric restriction can elevate cortisol as a stress response. However, the improved metabolic health and reduced body weight often normalize cortisol levels that were elevated due to obesity. The net effect on cortisol varies by individual. If you develop signs of high cortisol like central weight gain despite overall weight loss, morning headaches, or mood disturbances, testing cortisol through saliva or 24-hour urine collection may be appropriate.
Sleep architecture improves in many users as weight decreases. Obstructive sleep apnea severity reduces. Time in restorative deep sleep increases. These changes support metabolic health, hormone regulation, and recovery. Sleep quality represents an underappreciated factor in successful weight management. Users who prioritize sleep hygiene alongside tirzepatide therapy often achieve better results than those who neglect sleep.
The future of combination peptide therapies for metabolic health
Tirzepatide represents current state-of-the-art dual-agonist therapy, but the field continues evolving rapidly. Retatrutide, the triple agonist targeting GLP-1, GIP, and glucagon receptors, shows approximately 24% weight loss in phase 2 trials. If approved, it would surpass tirzepatide efficacy. Retatrutide dosage chart data remain preliminary as the drug progresses through clinical development.
Mazdutide represents another triple agonist combining GLP-1, glucagon, and GIP activity. Early trials show significant weight loss and metabolic improvements. Mazdutide vs tirzepatide comparisons await head-to-head trials, but both represent the trend toward multi-receptor targeting for enhanced efficacy.
CagriSema combines semaglutide with cagrilintide, an amylin analog. CagriSema dosing protocols show that combining medications targeting different pathways may achieve results exceeding either component alone. This combination approach allows lower doses of each component, potentially reducing side effects while maintaining efficacy.
Oral GLP-1 formulations continue development despite bioavailability challenges. Convenience drives research into transdermal patches, sublingual formulations, and improved oral delivery systems. GLP-1 patches represent one approach to eliminate injections while maintaining therapeutic drug levels. If effective patch systems reach market, patient acceptance may increase dramatically.
Personalized approaches using genetic testing to predict GLP-1 agonist response may emerge. Polymorphisms in GLP-1 receptor genes, incretin pathway enzymes, and appetite regulation circuits influence individual response. Identifying responders before treatment could improve outcomes and reduce failed treatment attempts. SeekPeptides tracks emerging research on pharmacogenomics applications in peptide therapy.
Combination protocols pairing metabolic peptides with other interventions show promise. Using tirzepatide alongside growth hormone secretagogues like ipamorelin or CJC-1295 may preserve lean mass better during aggressive fat loss. Combining with tissue repair peptides like BPC-157 might support gut health during the GI stress of GLP-1 therapy. Evidence for these combinations remains limited to case reports and small case series, but interest is growing.
Nutrigenomics approaches tailoring diet to individual genetics may complement peptide therapy. Understanding your response to different macronutrient ratios, meal timing, and specific foods could optimize the weight loss achieved on tirzepatide. Some users thrive on higher protein approaches. Others do better with moderate carbohydrate. Personalized nutrition based on genetic testing, microbiome analysis, and metabolic phenotyping represents a frontier in obesity medicine.
Microbiome modulation through targeted prebiotics, probiotics, or fecal transplantation might enhance GLP-1 agonist response. Specific bacterial strains influence GLP-1 secretion from enteroendocrine cells. Manipulating the microbiome to favor GLP-1-producing bacteria could amplify tirzepatide effects. While speculative, early research suggests microbiome composition influences metabolic health outcomes independently and may be modifiable to improve treatment response.
Long-acting formulations extending beyond once-weekly dosing could improve adherence further. Monthly or even quarterly depot injections of GLP-1 agonists are in development. These would provide steady drug levels without weekly injection burden. Trade-offs include less flexibility in dose adjustment and prolonged exposure if adverse effects develop. For stable responders on established maintenance doses, extended-release formulations offer convenience advantages.
The integration of continuous glucose monitors (CGM) with peptide therapy allows real-time feedback on metabolic responses. Seeing how specific foods affect your glucose and adjusting accordingly optimizes metabolic control. Some users on tirzepatide combine CGM data with their dosing schedule to fine-tune timing and dose. This data-driven approach to peptide therapy represents precision medicine applied to weight management.
Artificial intelligence and machine learning algorithms may predict individual response to tirzepatide based on baseline characteristics. Age, sex, BMI, diabetes status, genetic markers, microbiome composition, and lifestyle factors all influence outcomes. AI models integrating these variables could forecast expected weight loss and side effect risk, guiding treatment selection and dose titration. Peptide research and studies increasingly incorporate machine learning for pattern recognition in complex datasets.
The field moves rapidly toward more effective, better-tolerated, and more personalized metabolic therapies. Tirzepatide with B12 represents current best practice for many users. Future innovations will build on this foundation, offering even better tools for managing obesity and metabolic disease. Staying informed about emerging options positions you to benefit from advances as they transition from research to clinical availability.
For researchers serious about optimizing their tirzepatide protocols, SeekPeptides offers the most comprehensive resource available. Members access evidence-based guides, proven protocols, detailed dosing calculators, and a community of thousands who have navigated these exact questions about B12 formulations, titration schedules, and side effect management.
In case I do not see you, good afternoon, good evening, and good night. May your tirzepatide stay potent, your B12 levels stay optimal, and your weight loss results stay consistent.