Feb 12, 2026

Of the thousands of researchers who start a GLP-1 protocol each month, roughly half will experience side effects that make them question everything. Was this the right medication? Should I have picked the other one? Is this normal, or is something wrong?
Those questions deserve real answers. Not vague reassurances. Not marketing copy dressed up as medical guidance. Actual data from actual clinical trials, broken down so you can compare semaglutide and tirzepatide side by side and make a decision grounded in evidence rather than anxiety.
Here is what most comparison guides get wrong. They list side effects for each drug separately, as if you are reading two different pamphlets stapled together. That does not help. What helps is knowing that nausea hits 44% of people on high-dose semaglutide but only 17-22% on tirzepatide at comparable efficacy. What helps is knowing that tirzepatide causes more injection site reactions but less gastroparesis. What helps is understanding which side effects are transient annoyances that resolve in weeks and which ones require genuine medical attention.
This guide compares every documented side effect between semaglutide and tirzepatide using data from the STEP, SUSTAIN, SURPASS, and SURMOUNT clinical trial programs. We cover the common gastrointestinal effects, the rare but serious risks, the cosmetic concerns like hair loss, and the emerging data on mental health. Along the way, you will find specific management strategies for every side effect discussed, because knowing about a problem is only useful if you also know how to handle it.
SeekPeptides has tracked side effect patterns across thousands of research protocols, and the patterns tell a consistent story. Both medications work. Both cause side effects. But the type, severity, and duration of those side effects differ in ways that matter enormously for choosing the right protocol.
How semaglutide and tirzepatide work differently
Understanding why these two medications cause different side effects starts with understanding how they work. Semaglutide is a GLP-1 receptor agonist. It mimics a single gut hormone called glucagon-like peptide-1, which slows gastric emptying, reduces appetite, and improves insulin sensitivity. One mechanism. One receptor.
Tirzepatide does something different.
It activates two receptors simultaneously. The GLP-1 receptor and the GIP receptor. This dual mechanism is why tirzepatide often produces greater weight loss, but it also explains some differences in the side effect profile. The GIP receptor activation adds metabolic effects that pure GLP-1 agonists do not provide, including potential benefits for bone formation and gut motility that may actually buffer against certain gastrointestinal side effects.
This matters for side effects in three important ways. First, the GIP component may explain why tirzepatide produces comparable or even lower rates of nausea despite stronger weight loss. Second, dual receptor activation changes how the body processes the medication, affecting how long side effects last during dose escalation. Third, the two medications reach peak blood levels at different rates, which influences the timing and intensity of symptoms.
Both semaglutide and tirzepatide are administered as weekly subcutaneous injections. Both require dose escalation, meaning you start at a low dose and increase gradually. And both cause the majority of their side effects during these escalation periods, not during maintenance dosing. That last point is critical and often overlooked. The side effects you experience during your first month are not necessarily the side effects you will live with long term.
For a detailed comparison of semaglutide versus tirzepatide beyond side effects, including efficacy, dosing, and weight loss data, our dedicated comparison page covers the full picture.
Gastrointestinal side effects: the numbers that matter
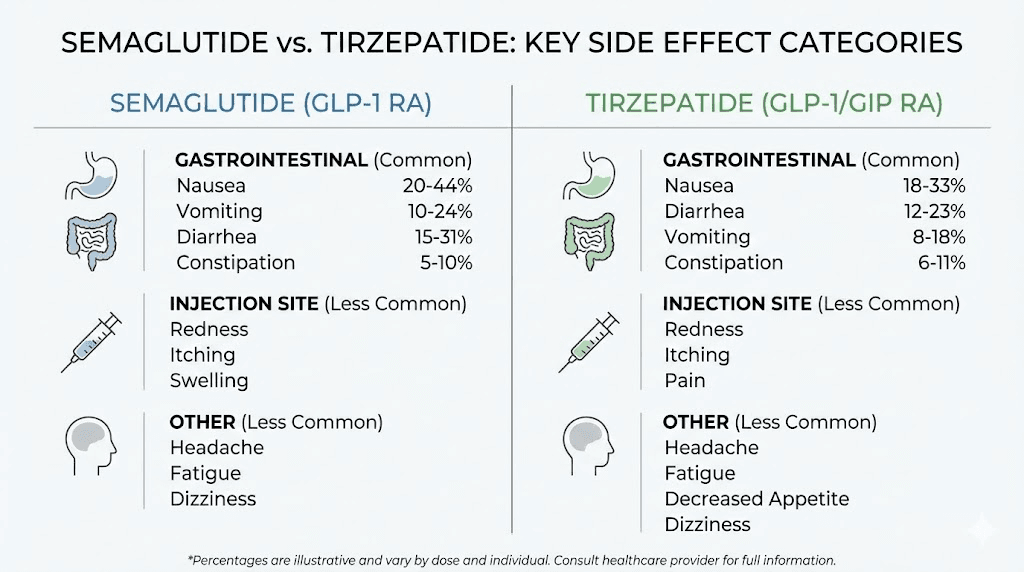
Let us start with the side effects everyone asks about first. The stomach issues. Nausea, vomiting, diarrhea, and constipation are the most commonly reported adverse events for both medications, and the clinical trial data gives us precise numbers to compare.
Nausea
Nausea is the single most reported side effect for both drugs, but the rates differ significantly depending on the dose and the trial context.
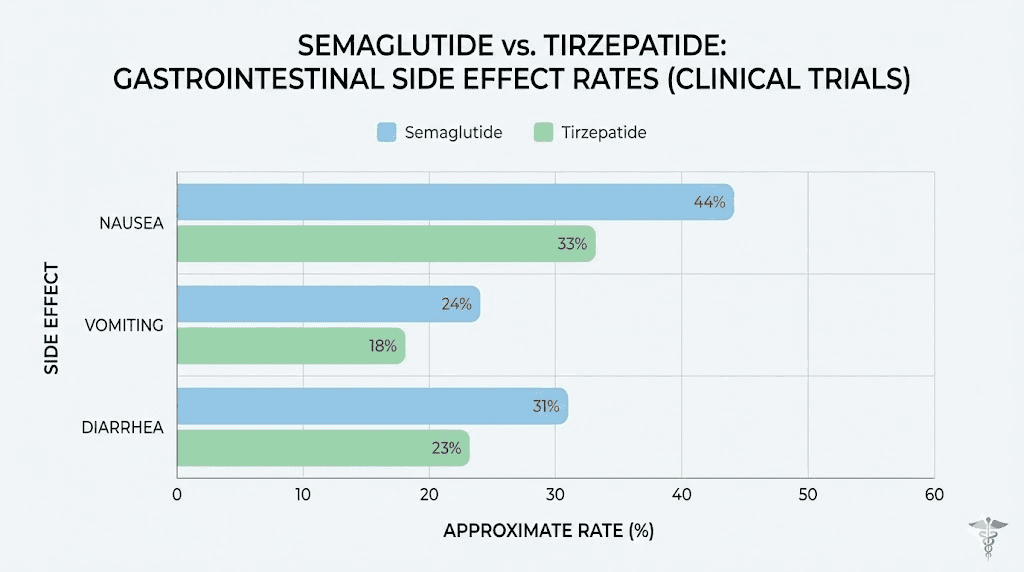
In the STEP trials, which tested semaglutide 2.4 mg for weight management, nausea affected 43.9% of participants compared to 16.1% on placebo. That is a substantial number. Nearly half of everyone on high-dose semaglutide experienced nausea at some point during the trial.
Tirzepatide tells a different story. In the SURPASS-2 trial, which directly compared tirzepatide to semaglutide 1 mg for type 2 diabetes, nausea rates were 17-22% for tirzepatide across all three dose levels (5 mg, 10 mg, and 15 mg) versus 18% for semaglutide 1 mg. At comparable doses in a head-to-head setting, the nausea rates were remarkably similar.
But here is the nuance that changes everything.
The semaglutide dose in SURPASS-2 was 1 mg, the diabetes dose. The weight loss dose is 2.4 mg, more than double. When you compare the weight loss doses, semaglutide at 2.4 mg causes significantly more nausea than tirzepatide at any dose, despite tirzepatide producing greater weight loss. This suggests that the GIP receptor activation in tirzepatide may provide some protection against the nausea that pure GLP-1 stimulation causes.
For researchers exploring how long semaglutide takes to suppress appetite, the nausea timeline closely mirrors the appetite suppression timeline. Both typically begin within the first week and peak during dose escalation periods.
Vomiting
Vomiting follows a similar pattern but at lower rates. In the STEP trials, semaglutide 2.4 mg caused vomiting in 24.5% of participants versus 6.3% on placebo. In SURPASS-2, tirzepatide caused vomiting in 6-10% of participants across dose levels, while semaglutide 1 mg caused vomiting in 8%.
Again, the dose comparison matters. At the weight management dose of 2.4 mg, semaglutide causes roughly 2.5 to 4 times more vomiting than tirzepatide at its highest dose. This is one of the most clinically meaningful differences between the two medications.
Vomiting is more disruptive than nausea. It interferes with nutrition. It can cause dehydration. It affects medication absorption if you are taking oral medications. For researchers who have a history of emetic sensitivity, this difference alone might influence which protocol they choose. Understanding proper tirzepatide dosing for weight loss and semaglutide dosing is essential for minimizing these effects through proper escalation.
Diarrhea
Diarrhea affects about 29.7% of semaglutide 2.4 mg users in the STEP trials versus 15.9% on placebo. In SURPASS-2, tirzepatide caused diarrhea in 13-16% of participants, while semaglutide 1 mg caused it in 12%.
The rates are closer here than with nausea and vomiting. Both medications stimulate the GLP-1 receptor, which increases intestinal water secretion and speeds colonic transit. The difference narrows further when you consider that diarrhea with both medications tends to be mild to moderate and resolves as the body adjusts.
What does make a practical difference is the timing. Semaglutide-related diarrhea tends to persist slightly longer during dose escalation, likely because the higher weight-loss dose takes longer to reach steady state. Tirzepatide users often report that diarrhea resolves faster, typically within the first two to three weeks at each new dose level.
For dietary strategies that help manage this side effect, our guides on semaglutide diet planning and tirzepatide diet planning cover evidence-based approaches to reducing GI distress through food choices.
Constipation
On the opposite end, constipation affected 24.2% of semaglutide 2.4 mg users versus 11.1% on placebo in the STEP trials. Tirzepatide data from SURMOUNT-1 showed constipation in roughly 10-17% of participants depending on dose.
This might seem contradictory. How can a medication cause both diarrhea and constipation? The answer lies in the slowed gastric emptying. GLP-1 receptor activation slows the entire digestive tract, which can swing between loose and hard stools depending on hydration, diet, and individual physiology. Some people experience both symptoms at different points in their treatment.
Constipation tends to be more persistent than diarrhea with both medications. It often requires active management through increased fiber intake, adequate hydration, and sometimes stool softeners. The key difference is that semaglutide users at the 2.4 mg dose report higher rates and longer duration of constipation compared to tirzepatide users, even at the highest 15 mg dose.
Head-to-head GI comparison table
Side effect | Semaglutide 2.4 mg (STEP) | Semaglutide 1 mg (SURPASS-2) | Tirzepatide 5-15 mg (SURPASS-2) | Tirzepatide 5-15 mg (SURMOUNT-1) |
|---|---|---|---|---|
Nausea | 43.9% | 18% | 17-22% | 24-33% |
Vomiting | 24.5% | 8% | 6-10% | 9-13% |
Diarrhea | 29.7% | 12% | 13-16% | 17-23% |
Constipation | 24.2% | Not reported separately | Not reported separately | 10-17% |
The pattern is clear. At equivalent diabetes doses, the two medications produce very similar GI side effect rates. At weight loss doses, semaglutide produces notably higher rates of every major GI symptom. And across all comparisons, most GI events are mild to moderate in severity, transient, and concentrated during dose escalation periods.
Gastroparesis and stomach motility concerns
Beyond ordinary nausea and vomiting, a more serious gastrointestinal concern has emerged with both medications. Gastroparesis, sometimes called stomach paralysis, involves severely delayed gastric emptying that goes beyond the normal slowing these drugs produce.
Both semaglutide and tirzepatide slow gastric emptying as part of their mechanism. That is by design. Slower emptying means you feel full longer, eat less, and lose weight. But in some cases, this slowing becomes excessive, leading to food sitting in the stomach for hours longer than normal, causing severe bloating, pain, and vomiting of undigested food.
The data here actually favors tirzepatide.
A comparative analysis found that non-diabetic patients with obesity on tirzepatide had a 64% lower risk of developing new-onset gastroparesis compared to those on semaglutide. GLP-1 agonists overall were associated with a 3.67 times higher risk of developing gastroparesis compared to non-users, but within that elevated risk category, tirzepatide appears significantly safer than semaglutide.
Why the difference? The GIP receptor component of tirzepatide may actually counterbalance some of the extreme gastric slowing that pure GLP-1 stimulation causes. GIP has prokinetic effects in the upper GI tract that could prevent the stomach from grinding to a complete halt. This theory is supported by the clinical data showing that tirzepatide produces comparable appetite suppression with less severe gastric emptying delay.
Both drug labels now include ileus, a condition where normal intestinal muscle contractions stop entirely, as a potential adverse event. While the available data on the association between bowel obstruction and GLP-1 medications remains inconsistent, the reports exist and researchers should be aware.
If you experience severe abdominal pain, inability to keep any food down, or vomiting of food eaten many hours earlier, seek medical attention immediately. These symptoms could indicate gastroparesis or ileus rather than ordinary GI side effects. For researchers managing semaglutide fatigue alongside GI symptoms, the combination can sometimes indicate that the dose escalation is moving too quickly.
Injection site reactions
Here is one area where tirzepatide clearly comes out worse.
Injection site reactions occurred in 1.9-4.5% of tirzepatide recipients compared to just 0.2% of semaglutide recipients in the SURPASS-2 trial. That is roughly 10 to 22 times more frequent with tirzepatide. The reactions include redness, swelling, itching, and small bruises at the injection site.
The good news is that these reactions are almost universally mild to moderate and resolve within 24 to 48 hours. They do not indicate an allergic reaction in most cases. About 51% of tirzepatide participants developed antidrug antibodies, and those with antibodies had higher rates of injection site reactions, but these were non-serious and transient with no observed differences in drug efficacy.
Practical management tips that work for most researchers include rotating injection sites between the abdomen, thigh, and upper arm. Allowing the medication to reach room temperature before injection reduces the sting. Using an ice pack on the area for 30 seconds before injection can numb the skin. And injecting slowly rather than pushing the plunger quickly tends to reduce local irritation.
For detailed guidance on injection technique, our guide on where to inject GLP-1 medications covers best practices for minimizing discomfort and reactions. Understanding the proper peptide injection technique makes a significant difference in how your body responds at the injection site.
If you are someone who is particularly sensitive to injection site reactions and that is a major concern for your protocol selection, semaglutide has a clear advantage here. But if you can tolerate mild, temporary skin reactions at the injection site, this should not be a primary deciding factor between the two medications.
Hair loss: what the data actually shows
Hair loss has become one of the most discussed side effects of GLP-1 medications on forums and social media. The actual clinical data tells a more nuanced story than the headlines suggest.
In clinical trials, hair loss was reported in 4-5% of people using tirzepatide (Zepbound) compared to 1% on placebo. For semaglutide (Wegovy), about 3% reported hair loss compared to 1% on placebo. So tirzepatide has a slightly higher reporting rate, but both are in the low single digits.
Here is the important context that changes how you should think about this.
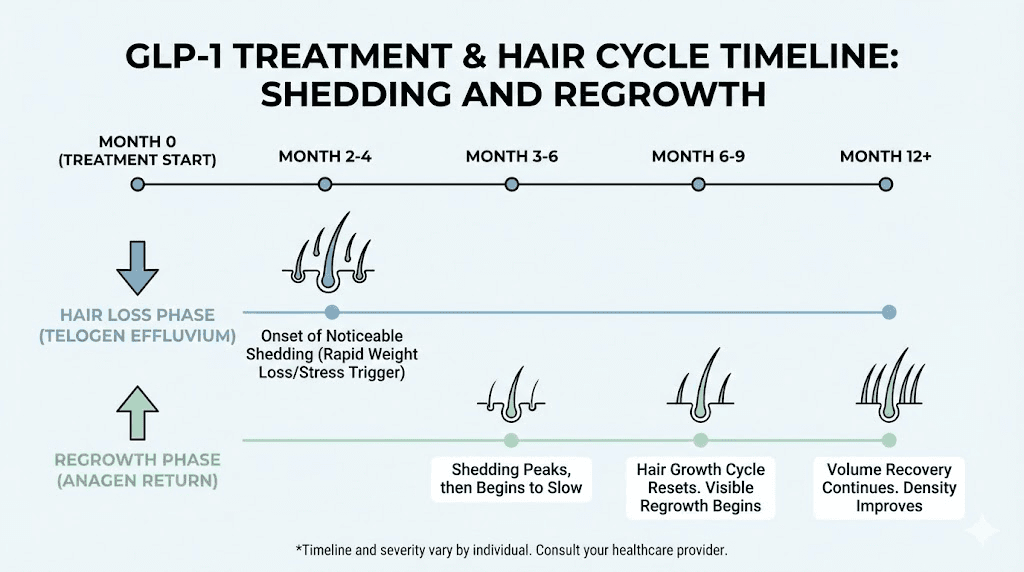
The hair loss associated with both medications is almost always telogen effluvium, a temporary and reversible form of hair shedding triggered by rapid weight loss and nutritional stress. It is not the medication directly damaging hair follicles. It is the body responding to significant caloric deficit, protein shifts, and changes in micronutrient levels including iron, zinc, biotin, and vitamin D.
This means the hair loss correlates more strongly with the amount and speed of weight lost than with which specific medication caused it. Since tirzepatide typically produces greater weight loss (20-25% body weight versus 15-17% with semaglutide at weight loss doses), more tirzepatide users may experience telogen effluvium simply because they lost more weight faster.
The hair typically regrows once weight stabilizes. Most researchers report that shedding peaks around months 3-6 of treatment and resolves by months 9-12. Supporting hair health during this period with adequate protein intake (at least 60-80 grams daily), iron and zinc supplementation if levels are low, and biotin supplementation can accelerate recovery.
For a comprehensive look at this topic, our guide on GLP-1 and hair loss covers prevention strategies, supplementation protocols, and when to seek dermatological evaluation. For tirzepatide users specifically, understanding peptide-related hair loss provides additional context about the mechanisms involved.
Alt: Hair loss timeline during semaglutide and tirzepatide treatment showing recovery phases
Lean muscle mass loss: a critical comparison
This is arguably the most important side effect comparison that most guides barely mention. Weight loss is the goal, but not all weight loss is created equal. Losing fat is desirable. Losing muscle is not. And the two medications differ meaningfully here.
The data from clinical trials paints a clear picture.
In the STEP-1 trial, semaglutide 2.4 mg produced a total lean mass loss of 6.92 kg, which represented 13.2% of baseline lean mass. Of the total weight lost, approximately 45.2% came from lean mass rather than fat. That means for every kilogram of weight lost on semaglutide, nearly half was muscle rather than fat.
Tirzepatide performs measurably better here. In the SURMOUNT-1 trial at the highest dose, lean mass decreased by 5.67 kg or 10.9% from baseline, with only 25.7% of total weight lost coming from lean mass. Tirzepatide users lost more total weight but preserved a greater proportion of their muscle.
Why does this matter?
Muscle mass drives your resting metabolic rate. Lose too much muscle and your metabolism slows, making weight regain more likely when treatment ends. Muscle loss also affects strength, mobility, bone density, and overall health outcomes. For older adults especially, preserving lean mass during weight loss is not optional. It is a health imperative.
The GIP receptor activation in tirzepatide may contribute to its superior muscle preservation. Research suggests GIP signaling has anabolic effects on bone and muscle that pure GLP-1 stimulation lacks. This is one of the most compelling arguments in tirzepatide's favor beyond simple weight loss numbers.
Mitigation strategies work for both medications. Researchers who engaged in resistance training 3-5 days per week with high protein intake saw dramatically better lean mass preservation. One case series documented a patient losing only 8.7% of total weight as lean tissue when combining tirzepatide with structured resistance exercise and protein intake above 1.2 grams per kilogram of body weight daily. That is a fraction of the lean mass loss seen in sedentary trial participants.
For researchers concerned about body composition, our guides on peptides for muscle growth and the best peptides for muscle growth explore complementary strategies. The muscle growth category page provides a broader overview of how to optimize body composition during weight loss protocols.
Muscle preservation comparison
Metric | Semaglutide 2.4 mg (STEP-1) | Tirzepatide 15 mg (SURMOUNT-1) |
|---|---|---|
Total weight loss | -15.3 kg (-14.9%) | -22.1 kg (-20.9%) |
Lean mass loss | -6.92 kg (-13.2%) | -5.67 kg (-10.9%) |
% of weight lost from lean mass | 45.2% | 25.7% |
Fat mass loss | ~54.8% of weight loss | ~74.3% of weight loss |
The difference is striking. Tirzepatide produces 40% more total weight loss while preserving significantly more muscle. For researchers prioritizing body composition over pure scale numbers, this data is compelling.
Gallbladder and biliary side effects
Gallbladder problems have emerged as a notable difference between the two medications, and this is an area where semaglutide performs worse.
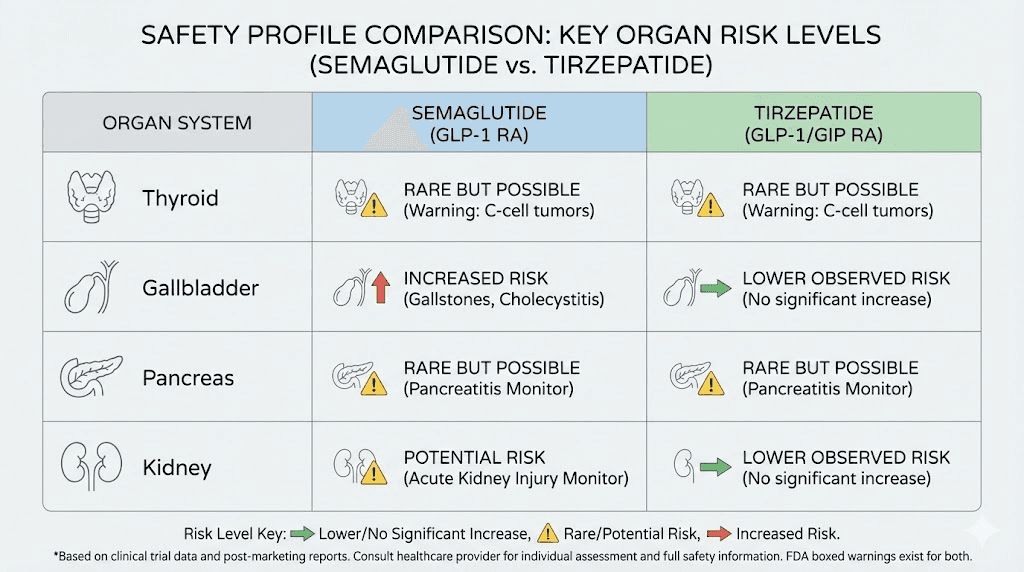
Meta-analysis data shows that semaglutide increases gallbladder-related disorders, particularly cholelithiasis (gallstones), by over 2.6 times compared to placebo. Tirzepatide, by contrast, showed no significant increase in biliary risk in the available data.
Rapid weight loss is a known risk factor for gallstone formation regardless of the method. When you lose weight quickly, the liver secretes more cholesterol into bile, and the gallbladder contracts less frequently because you are eating less. These two factors create the perfect conditions for cholesterol to crystallize into stones. Both medications cause rapid weight loss, so some gallbladder risk exists with both.
But the degree of risk differs.
Tirzepatide data from the SURPASS trials showed gallbladder events (cholelithiasis, biliary colic, and cholecystectomy) in approximately 0.6% of treated patients versus none in placebo groups. While this is a real increase, it is a fraction of the gallbladder risk reported with semaglutide.
For researchers with a history of gallbladder problems, existing gallstones, or other biliary risk factors like rapid previous weight loss episodes or high BMI, this difference should factor into their medication choice. Simple risk reduction strategies include maintaining adequate fat intake (at least 20-30% of daily calories) to stimulate regular gallbladder contraction, staying well hydrated, and reporting any right upper abdominal pain promptly to a healthcare provider.
The foods to avoid on tirzepatide guide covers dietary strategies that also help minimize gallbladder stress. Similarly, understanding optimal nutrition on semaglutide can help reduce this risk.
Thyroid concerns and cancer risk
Both semaglutide and tirzepatide carry a black box warning about medullary thyroid carcinoma (MTC) risk. This warning stems from animal studies showing thyroid C-cell tumors in rodents exposed to GLP-1 receptor agonists. But what does the human data show?
The answer is reassuring, though nuanced.
Clinical analyses of semaglutide did not show increased risk for thyroid or pancreatic cancers compared to placebo or other diabetes medications. Similarly, a meta-analysis of tirzepatide-only data reported no excess risk for thyroid cancer compared to control groups.
However, there is one detail worth noting. Higher doses of tirzepatide showed increased serum calcitonin levels compared to placebo. Calcitonin is a tumor marker for medullary thyroid carcinoma, so elevated levels theoretically warrant attention. That said, no increased risk of papillary thyroid carcinoma, which is the most common form of thyroid cancer, was found with tirzepatide use.
The practical guidance is straightforward. Both medications are contraindicated in people with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). For everyone else, the current evidence does not support an elevated thyroid cancer risk with either medication in humans. Monitoring thyroid function during treatment is reasonable but not mandated by current clinical guidelines.
For researchers who want to understand the broader safety landscape, our peptide safety and risks guide provides comprehensive coverage. The benefits and risks of peptides article offers additional context on weighing therapeutic benefits against potential concerns.
Pancreatitis risk
Pancreatitis, inflammation of the pancreas, has been a concern with all incretin-based therapies since the earliest GLP-1 drugs reached market. The concern is valid, given that these medications directly affect pancreatic function.
The clinical trial data for both medications is reassuring on this front. Neither semaglutide nor tirzepatide significantly increased hepatic or pancreatic adverse events in their respective trial programs. A systematic review specifically examining tirzepatide found no significantly increased risk of pancreatitis compared to control groups including placebo, basal insulin, and selective GLP-1 receptor agonists.
Real-world pharmacovigilance data is broadly consistent with these findings, though isolated case reports exist for both medications. The background rate of pancreatitis in the general population, especially among people with obesity and type 2 diabetes who use these medications, is already elevated by the underlying conditions themselves. Separating medication-caused pancreatitis from coincidental pancreatitis in a high-risk population remains methodologically challenging.
Warning signs that require immediate medical attention include severe, persistent abdominal pain that radiates to the back, nausea and vomiting that do not improve with usual anti-nausea strategies, and abdominal tenderness that worsens over hours rather than improving. If you experience these symptoms, stop the medication and contact your healthcare provider immediately. Do not assume it is ordinary GI side effects and wait it out.
For researchers managing tirzepatide headaches or other side effects, distinguishing between common adverse events and those requiring urgent medical care is essential for safe protocol management.
Kidney and liver safety
The kidney and liver data actually presents one of the most interesting differences between the two medications.
Kidney effects
Both medications can be used in patients with any level of kidney function, as neither drug relies on renal clearance. Renal impairment does not meaningfully impact the pharmacokinetics of either semaglutide or tirzepatide.
But here is where it gets interesting. Tirzepatide appears to have protective kidney effects. In a post-hoc analysis of the SURPASS-4 trial, tirzepatide decreased albuminuria (protein in urine, a marker of kidney damage) and nearly halved the risk of a composite kidney endpoint compared to insulin glargine. Tirzepatide at 10 mg and 15 mg doses was superior to placebo in reducing urine albumin-to-creatinine ratio.
Comparative safety data shows acute kidney injury was listed in 0.47% of tirzepatide adverse event reports versus 1.07% of semaglutide reports in the FDA Adverse Event Reporting System (FAERS), with the reporting odds ratio favoring tirzepatide. The dehydration risk from severe vomiting and diarrhea, which can lead to acute kidney injury, may partially explain this difference, as semaglutide causes more severe GI events at weight loss doses.
For researchers interested in the renal aspects, staying well hydrated during GLP-1 therapy is not just good advice. It is a genuine safety measure. Dehydration from GI side effects is the most common pathway to medication-related kidney problems.
Liver effects
Both medications have been linked to rare instances of liver enzyme elevations and isolated cases of clinically apparent liver injury. The rates are very low for both, and neither has been associated with a systematic pattern of hepatotoxicity.
Tirzepatide has shown a slightly higher rate of acute gallbladder disease (0.6% versus essentially zero in placebo groups), which can secondarily affect liver function through biliary obstruction. However, direct liver toxicity from either medication remains extremely rare.
Understanding how your body responds to these medications over time is part of responsible protocol management. SeekPeptides provides comprehensive safety monitoring guides that help researchers track relevant biomarkers and know when laboratory testing is warranted during treatment.
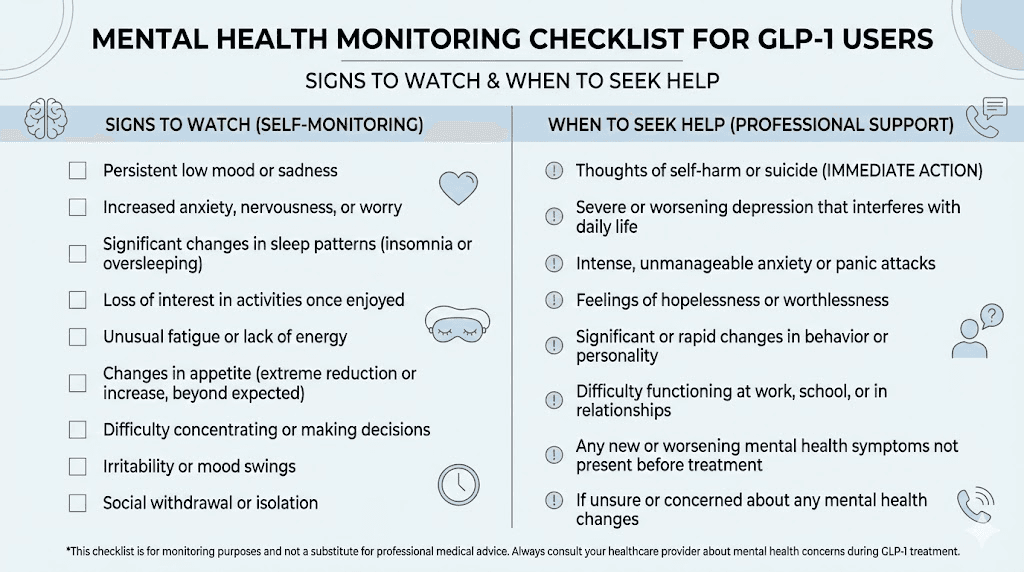
Mental health side effects: emerging data
This is one of the most actively researched areas in GLP-1 safety, and the data is evolving rapidly. Reports of depression, anxiety, and suicidal ideation have attracted significant regulatory and media attention.
A pharmacovigilance analysis of the EudraVigilance database recorded 102 suicidal events across all GLP-1 medications. Of these, 50% were associated with semaglutide and only 3.9% with tirzepatide. Depression was the most commonly reported psychiatric adverse event (50.3% of psychiatric reports), followed by anxiety (38.7%) and suicidal ideation (19.6%).
A more recent FAERS analysis found significant disproportionality signals only for semaglutide in both depression and suicide/self-injury events, while tirzepatide did not show significant signals. The reporting odds ratio for semaglutide was 1.87 for depression and 1.73 for suicide/self-injury events.
But context matters enormously here.
Pharmacovigilance databases capture reports, not confirmed causal relationships. Semaglutide has been on the market significantly longer and is prescribed far more widely than tirzepatide, which naturally produces more reports. When clinical trial data is examined directly, semaglutide-treated participants did not show increased risk of developing moderately severe depression symptoms or suicidal thoughts compared to placebo. Only 1% or fewer of participants reported suicidal ideation or behavior during treatment.
In fact, some research suggests the opposite. An NIH-funded study found that people taking semaglutide actually had a lower risk of suicidal thoughts compared to non-users. Weight loss itself can improve mental health, and the metabolic improvements from these medications may have neuroprotective benefits.
The practical takeaway is this. Screening for pre-existing psychiatric conditions before starting either medication is wise. Monitoring mental health during treatment is reasonable. But current evidence does not support avoiding these medications purely out of mental health concern, especially in patients without pre-existing psychiatric conditions.
For researchers exploring the relationship between peptides and mental health, our guides on peptides for depression and anxiety and peptides for brain function provide relevant context on how different peptides interact with neurological systems. The peptides for anxiety guide covers additional mental health considerations for peptide researchers.
Fatigue and energy levels
Fatigue is a commonly reported side effect with both medications that rarely appears in the clinical trial headlines but significantly affects quality of life for many researchers.
Semaglutide users frequently report fatigue, especially during the first few weeks and during dose escalation. Our comprehensive guide on semaglutide fatigue covers this in detail. The fatigue typically stems from three sources: reduced caloric intake (you are eating less, which means less fuel), GI discomfort (nausea and stomach issues are exhausting), and metabolic adjustment as your body adapts to new hormone signaling patterns.
Tirzepatide users also report fatigue, though anecdotally the reports tend to be less severe. This may relate to tirzepatide's potentially better muscle preservation and its different impact on glucose metabolism through dual receptor activation. When your body preserves more lean mass and maintains more stable blood sugar, subjective energy levels tend to remain higher.
The fatigue typically improves as the body adjusts, usually resolving significantly by weeks 4-8 at any given dose. Strategies that help include eating adequate protein even when appetite is suppressed, staying hydrated, maintaining light physical activity (which paradoxically fights fatigue better than rest), and ensuring sufficient sleep.
Understanding why tirzepatide makes you tired can help researchers plan around the most fatiguing periods of their protocol. For those on semaglutide exploring why weight loss stalls, fatigue-driven inactivity is sometimes the hidden factor.
Headaches and dizziness
Headaches affect a meaningful percentage of users on both medications. Dizziness, while less common, also appears in the clinical trial data. In SURPASS-2, dizziness was listed among the commonly reported adverse events for both tirzepatide and semaglutide groups.
The mechanisms behind GLP-1-related headaches are not fully understood, but likely involve changes in blood sugar levels (especially during early treatment when insulin sensitivity improves rapidly), dehydration from reduced fluid intake or GI losses, and the central nervous system effects of GLP-1 receptor activation, since GLP-1 receptors exist in the brain.
Tirzepatide may cause slightly more headaches initially due to its dual receptor activation affecting both GLP-1 and GIP pathways in the brain simultaneously, though the data does not show a statistically significant difference between the two medications.
Management is straightforward. Stay hydrated. Eat regularly even if appetite is reduced. Monitor blood sugar if diabetic, as hypoglycemia can cause both headaches and dizziness. Most headaches resolve within the first 2-4 weeks at any given dose level.
For tirzepatide users specifically, our headache management guide provides detailed protocols. Researchers experiencing headaches alongside anxiety on tirzepatide should discuss both symptoms with their healthcare provider, as the combination may indicate dose adjustment needs.
Cardiovascular considerations
This is one area where semaglutide currently has a regulatory advantage, though tirzepatide data continues to accumulate.
Semaglutide is FDA approved to help reduce the risk of serious cardiovascular events, including heart attacks, strokes, and cardiovascular death, in people with established heart disease who are overweight or obese. This approval came from the SELECT trial, which demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide compared to placebo.
Tirzepatide does not yet have this cardiovascular indication. The SURPASS-CVOT trial examining tirzepatide's cardiovascular outcomes is ongoing, and results are expected to inform future labeling. Early data from existing trials is promising, showing improvements in cardiovascular risk factors like blood pressure, lipid profiles, and inflammatory markers, but the definitive cardiovascular outcomes data is not yet available.
This does not mean tirzepatide is cardiovascularly unsafe. It means we have less definitive evidence for tirzepatide's cardiovascular benefits compared to semaglutide. For researchers with existing cardiovascular disease, this regulatory distinction may be clinically relevant and worth discussing with their healthcare provider.
Our cardiogen peptide benefits guide and vesugen peptide benefits article cover cardiovascular-supportive peptides for researchers interested in heart health optimization.
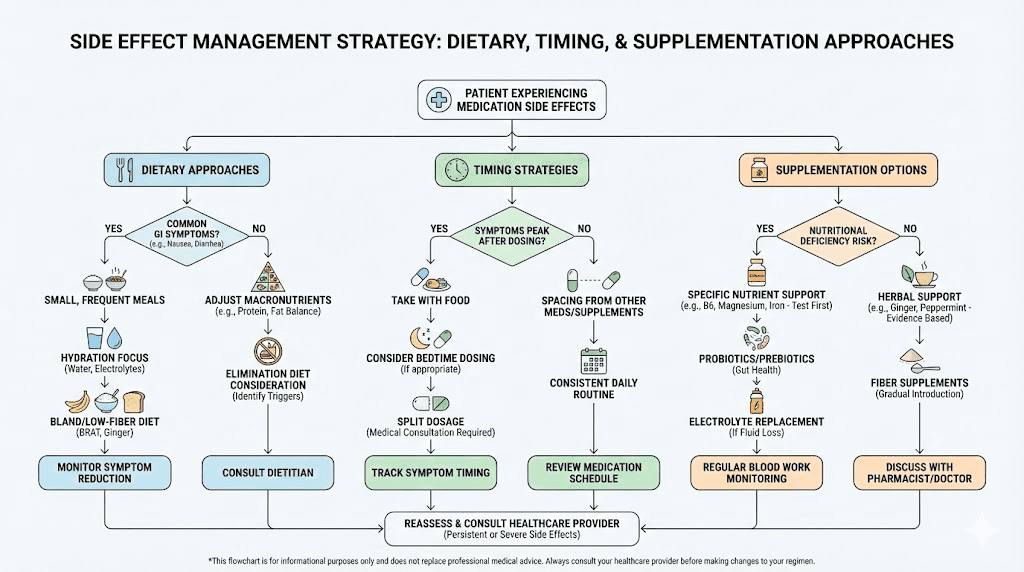
Side effect management strategies that work for both medications
Regardless of which medication you choose, the management strategies for common side effects are largely the same. Here are evidence-based approaches that clinical experience supports.
Slow dose escalation
This is the single most effective strategy for reducing side effects with both medications. The standard dose escalation schedules in the prescribing information are starting points, not rigid requirements. Many clinicians and researchers extend the time at each dose level if side effects are bothersome.
For semaglutide, the standard escalation is 0.25 mg for 4 weeks, then 0.5 mg for 4 weeks, then increases every 4 weeks up to 2.4 mg. Extending any step to 8 weeks instead of 4 significantly reduces GI side effects for most people.
Tirzepatide follows a similar pattern starting at 2.5 mg for 4 weeks, then 5 mg, then increasing by 2.5 mg increments every 4 weeks up to 15 mg. Again, extending the time at each dose allows the body more time to adjust.
Our microdosing tirzepatide chart provides an alternative escalation approach that some researchers find dramatically reduces side effects. For semaglutide, the dosage chart in units helps researchers fine-tune their escalation.
Dietary strategies
Eating smaller, more frequent meals reduces the burden on a stomach that is already emptying slowly. Avoiding high-fat, greasy, or heavily spiced foods during dose escalation helps significantly. Prioritizing protein at every meal supports both muscle preservation and satiety. Ginger tea, peppermint, and small sips of carbonated water can help manage nausea.
For specific food guidance, tirzepatide foods to avoid and the semaglutide diet plan provide detailed nutritional frameworks. Both guides emphasize that what you eat matters more than usual when your GI system is adapting to these medications.
Hydration
Many side effects, including headaches, dizziness, constipation, and fatigue, worsen with dehydration. Both medications reduce appetite for food and fluids, making it easy to underhydrate without realizing it. Aim for at least 64 ounces (approximately 2 liters) of water daily, more if you are experiencing diarrhea or vomiting.
Supplementation
Some researchers using semaglutide with B12 or tirzepatide with B12 report reduced fatigue and improved energy. Other supportive supplements include glycine, which some researchers combine with their protocol. Both semaglutide with glycine and tirzepatide with glycine combinations have their own evidence base worth exploring. Tirzepatide with niacinamide is another combination that researchers use for complementary support.
Timing and storage
Proper medication storage affects efficacy, which indirectly affects side effects. If your medication loses potency due to improper storage, you may need higher doses to achieve the same effect, which means more side effects. Understanding how long tirzepatide lasts in the fridge, how long compounded semaglutide lasts refrigerated, and whether tirzepatide needs refrigeration is essential for maintaining medication integrity.
For compounded formulations, our tirzepatide reconstitution guide and semaglutide reconstitution guide cover proper handling to preserve potency and minimize side effects from degraded medication.
When to consider switching between medications
Sometimes the best response to intolerable side effects is not pushing through. It is switching. Here are the clinical scenarios where switching from one medication to the other makes sense.
Switch from semaglutide to tirzepatide when
Nausea and vomiting remain severe beyond 8-12 weeks at the same dose. Since tirzepatide often produces less nausea at equivalent efficacy, switching may provide the same weight loss with better GI tolerance. Gallbladder symptoms develop, as tirzepatide has a lower biliary risk profile. Weight loss plateaus despite maximum dose, since tirzepatide's dual mechanism may overcome semaglutide resistance. Muscle loss is a significant concern, given tirzepatide's better lean mass preservation data.
Our detailed guide on switching between these medications covers the practical aspects of transitioning. The semaglutide to tirzepatide conversion chart provides dose equivalency guidance for making the switch smoothly.
Switch from tirzepatide to semaglutide when
Injection site reactions are persistent and bothersome, since semaglutide has dramatically lower injection site reaction rates. Cardiovascular risk reduction is a primary treatment goal, given semaglutide's FDA-approved cardiovascular indication. Cost or access issues make tirzepatide impractical, as semaglutide has broader insurance coverage and more compounding options. The tirzepatide to semaglutide conversion chart helps with this transition.
Regardless of which direction you switch, do not simply stop one medication and start the other at a full dose. Transition gradually. Most clinicians recommend starting the new medication at its lowest dose and escalating even if you were on a high dose of the previous medication, because receptor sensitivity and side effect profiles differ.
Special population considerations
Side effect profiles can vary based on individual factors. Here are the key populations where the comparison shifts.
People with type 2 diabetes
Both medications can cause hypoglycemia when combined with insulin or sulfonylureas. The risk is similar for both, though tirzepatide may have slightly better glucose control, which paradoxically increases hypoglycemia risk with concurrent insulin use. Monitor blood sugar closely during dose escalation and work with your prescriber to adjust concurrent diabetes medications.
Women and hormonal considerations
Both medications affect hormonal balance through weight loss. Rapid fat loss releases stored estrogen, which can cause menstrual irregularities, mood changes, and other hormonal symptoms. These effects are proportional to weight loss speed and amount, making them slightly more common with tirzepatide due to greater average weight loss.
For women specifically, our guides on peptides for weight loss in women, best peptides for women, and peptides for menopause weight loss address gender-specific considerations. The peptides for hormone balance guide covers broader hormonal optimization during weight loss.
Older adults
Muscle preservation becomes even more critical in adults over 60, making tirzepatide's superior lean mass preservation data more clinically significant. Dehydration risk is also higher in older adults, which amplifies the kidney safety advantage of tirzepatide (lower AKI reporting rate). The peptides for men guide and peptides for women over 40 guide address age-specific considerations.
Breastfeeding
For women considering GLP-1 medications while breastfeeding, our guide on taking GLP-1 while breastfeeding covers the current safety data and recommendations for both medications.
Cost and access: the indirect side effect
While not a pharmacological side effect, the cost of these medications creates real-world consequences that affect adherence and outcomes. Inconsistent use due to cost barriers often leads to yo-yo dosing, which worsens side effects when restarting.
Semaglutide currently has broader availability through compounding pharmacies and more insurance coverage options. Tirzepatide is newer and may have more limited access depending on location and insurance. The affordable tirzepatide guide covers strategies for managing costs. For semaglutide, resources like the compounded semaglutide guide and cheap compounded tirzepatide article explore cost-effective alternatives.
For researchers using compounded formulations, understanding proper preparation is crucial. Our peptide reconstitution calculator and peptide calculator tools help ensure accurate dosing with compounded products. The compounded tirzepatide dosage calculator guide provides specific instructions for compounded formulations.
Complete side effect comparison table
Here is the comprehensive comparison across all major side effect categories, summarizing the data covered throughout this guide.
Side effect category | Semaglutide | Tirzepatide | Better option |
|---|---|---|---|
Nausea (weight loss dose) | 43.9% | 24-33% | Tirzepatide |
Vomiting (weight loss dose) | 24.5% | 9-13% | Tirzepatide |
Diarrhea | 29.7% | 17-23% | Tirzepatide |
Constipation | 24.2% | 10-17% | Tirzepatide |
Injection site reactions | 0.2% | 1.9-4.5% | Semaglutide |
Gastroparesis risk | Higher | 64% lower risk | Tirzepatide |
Gallbladder risk | 2.6x increase | No significant increase | Tirzepatide |
Hair loss | ~3% | 4-5% | Similar (weight-loss driven) |
Lean mass loss (% of weight lost) | 45.2% | 25.7% | Tirzepatide |
Acute kidney injury reports | 1.07% | 0.47% | Tirzepatide |
Mental health signals | Significant signals | No significant signals | Tirzepatide (limited data) |
Cardiovascular benefit | FDA approved (SELECT) | Not yet proven | Semaglutide |
Pancreatitis | No significant increase | No significant increase | Similar |
Thyroid cancer | No human evidence | No human evidence | Similar |
The pattern that emerges from this comprehensive comparison is that tirzepatide generally has a more favorable side effect profile for most categories, with the notable exceptions of injection site reactions and hair loss (which is weight-loss driven rather than drug-specific). Semaglutide's main advantages are its proven cardiovascular benefit and lower injection site reaction rate.
Making the decision: which side effect profile fits your situation
After reviewing all this data, the question remains. Which medication should you choose based on side effects?
The honest answer is that it depends entirely on your individual risk factors, priorities, and health history. But the data does allow us to create decision frameworks.
Choose semaglutide if: You have existing cardiovascular disease and want FDA-backed cardiovascular risk reduction. You are extremely sensitive to injection site reactions. You need broader access and insurance coverage. You are already tolerating it well and do not want to switch.
Choose tirzepatide if: GI tolerance is a primary concern and you want lower nausea and vomiting rates. Muscle preservation matters significantly (older adults, athletes, people with sarcopenia risk). You have gallbladder risk factors. You want maximum weight loss with proportionally fewer GI side effects. Kidney protection is important for your health profile.
Neither medication is the "safe" choice. Both carry real side effects. The question is which set of trade-offs aligns better with your specific situation. Discussing these trade-offs with a knowledgeable healthcare provider who understands your complete medical history is always the best approach.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members get access to detailed side effect tracking tools, personalized protocol recommendations, and expert guidance for managing adverse events effectively.
Frequently asked questions
Which has fewer side effects overall, semaglutide or tirzepatide?
Based on clinical trial data, tirzepatide generally produces lower rates of gastrointestinal side effects at weight loss doses despite causing greater weight loss. Tirzepatide has lower rates of nausea (24-33% vs 43.9%), vomiting (9-13% vs 24.5%), and constipation compared to semaglutide at its weight loss dose. However, tirzepatide causes more injection site reactions. The overall side effect burden tends to be lower with tirzepatide for most researchers.
Do the side effects go away over time?
Yes, for the vast majority of people. Clinical data from both the STEP and SURMOUNT trials shows that GI side effects are concentrated during dose escalation and resolve for most participants within 4-8 weeks at any given dose level. About 99.5% of GI adverse events with semaglutide were non-serious and 98.1% were mild to moderate. The same pattern holds for tirzepatide. Most researchers who make it through the initial escalation period find long-term side effects manageable.
Can I reduce side effects by starting at a lower dose?
Absolutely. Slower dose escalation is the most effective single strategy for reducing side effects with both medications. Extending the time at each dose from 4 weeks to 6-8 weeks gives your body more time to adjust. Some researchers use microdosing approaches to create even more gradual escalation. The trade-off is that slower escalation means slower weight loss initially, but the improved tolerability often leads to better long-term adherence and outcomes.
Is hair loss permanent with these medications?
No. The hair loss associated with GLP-1 medications is telogen effluvium, a temporary, reversible shedding triggered by rapid weight loss. Hair typically begins regrowing once weight stabilizes, usually within 6-12 months. Adequate protein intake, iron and zinc supplementation, and biotin can support faster recovery. The hair loss is driven by the weight loss itself rather than the specific medication.
Which medication is safer for long-term use?
Both medications have demonstrated safety profiles across multi-year clinical trials. Semaglutide has more long-term data available simply because it has been on the market longer. Tirzepatide generally shows favorable safety signals including lower GI event rates, better muscle preservation, and protective kidney effects. For long-term use of semaglutide, cardiovascular benefits are well established. Long-term tirzepatide cardiovascular data is still being collected.
Can switching from one to the other reduce side effects?
Yes, switching is a legitimate strategy. Some researchers who do not tolerate semaglutide well find tirzepatide more manageable, and vice versa. The semaglutide to tirzepatide conversion chart and tirzepatide to semaglutide conversion chart provide dose equivalency guidance. Always start the new medication at the lowest dose and escalate gradually regardless of what dose you were on previously.
Do compounded versions have different side effects than brand-name versions?
The active ingredient is the same, so the core side effect profile should be identical. However, compounded versions may have different concentrations, which affects dosing precision. Improper reconstitution or storage can degrade the peptide, potentially reducing efficacy (requiring higher doses and more side effects) or introducing contaminants. Using proper reconstitution techniques and sourcing from reputable compounding pharmacies helps ensure equivalent safety.
What should I do if side effects are unbearable?
Do not push through severe side effects in silence. Contact your healthcare provider. Options include holding the dose and not escalating further, dropping back to the previous dose, switching to the other medication, adding supportive medications for specific symptoms, or in rare cases, discontinuing treatment entirely. Our peptide safety guide covers when to seek immediate medical attention versus when to manage at home.
External resources
SURPASS-2 Trial: Tirzepatide vs Semaglutide in Type 2 Diabetes (NEJM)
Body Weight Reducing Effects of Tirzepatide: Efficacy and Adverse Effects Review (PMC)
In case I do not see you, good afternoon, good evening, and good night. May your side effects stay mild, your dose escalation stay smooth, and your protocols stay effective.