Feb 17, 2026

Experienced researchers know something beginners do not about tirzepatide formulations. The peptide itself is only part of the equation. What compounding pharmacies add to that vial matters just as much as the active ingredient, sometimes more. And the combination that has generated the most attention among informed users is the triple compound: tirzepatide with glycine and B12 in a single formulation.
This is not theoretical.
Compounding pharmacies across the country now offer tirzepatide/glycine/B12 as a premixed injectable, typically in concentrations like 30/5/0.5 mg/mL delivered in 2 mL vials. The formulation exists because each ingredient addresses a specific limitation of tirzepatide alone. Glycine protects muscle tissue during rapid weight loss. B12 combats the nutrient depletion that GLP-1 medications cause while simultaneously reducing nausea through a fascinating pharmacological mechanism most users have never heard of. Together, they transform a weight loss peptide into something closer to a comprehensive metabolic support system.
But here is where it gets complicated. There are already separate guides covering tirzepatide with glycine and tirzepatide with B12 individually. This guide is different. This guide focuses specifically on the combined formulation, because the three ingredients together create synergies that neither glycine nor B12 achieve alone. The interaction effects, the dosing considerations, the practical realities of using a triple compound, these require their own deep analysis. That is exactly what SeekPeptides provides here: the most thorough breakdown of the tirzepatide/glycine/B12 compound available anywhere.
Whether you are evaluating compounding pharmacy options, trying to understand why your provider prescribed this specific formulation, or researching how to optimize your tirzepatide protocol for both fat loss and muscle preservation, this guide covers every angle with specific numbers, research citations, and actionable protocols.
What the tirzepatide glycine B12 compound actually is
The tirzepatide/glycine/B12 compound is a single injectable formulation that combines three distinct ingredients into one vial. It is not three separate injections. It is not a DIY mixture you create at home. It is a pharmacy-compounded product prepared under sterile conditions, with each ingredient measured to specific concentrations and verified for stability and compatibility. If you are new to the world of peptide-based therapeutics, understanding the basics first will make everything that follows much clearer.
Understanding what each component does individually is essential before examining how they work together.
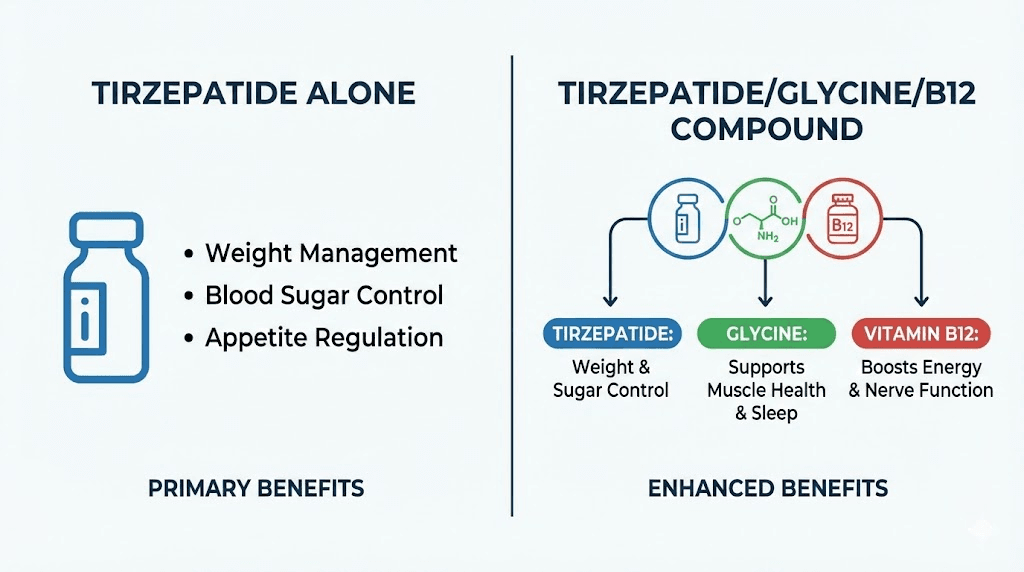
Tirzepatide: the dual-action foundation
Tirzepatide is a dual GLP-1/GIP receptor agonist. That means it activates two different incretin receptors simultaneously, which is what separates it from single-action GLP-1 agonists like semaglutide. Understanding how peptides work at the receptor level helps explain why dual activation produces superior results. The GLP-1 activation slows gastric emptying, reduces appetite, and improves insulin sensitivity. The GIP activation enhances fat metabolism and provides additional glycemic control through a complementary pathway.
The clinical results speak for themselves. The SURMOUNT-1 trial demonstrated weight reductions of 16.0% on 5 mg, 21.4% on 10 mg, and 22.5% on 15 mg over 72 weeks. Those numbers are remarkable. The SURPASS trials confirmed significant A1C reductions alongside weight loss, establishing tirzepatide as one of the most effective metabolic medications available. For a detailed comparison with other options, the semaglutide versus tirzepatide comparison breaks down the differences comprehensively.
But tirzepatide has limitations.
Nausea affects up to 20% of users. Between 40% and 70% of people on GLP-1 medications experience nausea at some point during treatment. Weight loss is not purely fat loss either. Research shows that approximately 75% of weight lost on tirzepatide is fat mass, while 25% is lean mass. In some trials, lean mass loss accounts for 26% to 40% of total weight reduction. For someone losing 50 pounds, that could mean losing 12 to 20 pounds of muscle. Anyone exploring peptides for weight loss needs to understand this trade-off. These limitations are exactly what glycine and B12 are designed to address.
Glycine: the muscle protector
Glycine is the simplest amino acid. It is also one of the most underestimated. In the context of a tirzepatide compound, glycine serves three critical functions: muscle preservation, collagen support, and metabolic enhancement.
Research published in PMC6871541 demonstrated that glycine activates mTORC1 signaling in muscle cells. This is significant because mTORC1 is the master regulator of muscle protein synthesis. When mTORC1 is activated, muscle cells increase protein production, resist catabolism, and maintain size even under metabolic stress. In the study, glycine-treated muscle cells were up to 20% larger compared to control groups. During the caloric restriction that tirzepatide naturally creates, this mTORC1 activation becomes a protective mechanism against the lean mass loss that plagues GLP-1 therapy.
A separate study (PubMed 26431812) examined glycine supplementation during calorie restriction specifically. The findings showed that glycine accelerated fat loss while simultaneously protecting muscle tissue. That is the exact combination tirzepatide users need: more fat loss, less muscle loss. Understanding how peptides support muscle growth and preservation provides additional context for why glycine matters so much in this formulation.
Glycine also constitutes roughly one-third of all amino acids in collagen. During rapid weight loss, skin elasticity becomes a significant concern. Research indicates that adequate glycine intake can improve skin elasticity by up to 20% within 8 to 12 weeks. For tirzepatide users losing substantial weight quickly, this collagen support is not cosmetic vanity. It is a practical measure against the loose skin that can follow dramatic weight reduction.
B12 (methylcobalamin): the nausea reducer and nutrient restorer
Vitamin B12 in the tirzepatide compound is typically methylcobalamin, the bioactive form that the body can use immediately without conversion. It serves two purposes in this formulation, and most people only know about one of them.
The obvious purpose is addressing B12 deficiency. GLP-1 medications reduce gastric acid production and slow gastric emptying, both of which impair B12 absorption from food. Over months of treatment, B12 levels can decline significantly, leading to fatigue, cognitive fog, nerve tingling, and mood changes. By including B12 directly in the injection, the compound bypasses the digestive absorption problem entirely. The B12 goes straight into subcutaneous tissue and then into the bloodstream, no stomach acid required.
The less obvious purpose is nausea reduction.
A study published in PMC5899935 examined B12 conjugation with GLP-1 receptor agonists and found something fascinating. B12 reduces the brain penetration of GLP-1 compounds. Since nausea from GLP-1 medications is primarily mediated by central nervous system receptors, reducing how much of the drug reaches the brain reduces the nausea signal without significantly impacting the peripheral metabolic effects. The weight loss still happens. The blood sugar control still works. But the brain receives less of the nausea-triggering stimulation. For anyone who has read about semaglutide with B12, this mechanism applies similarly across GLP-1 class medications.
This is why the compound exists. Not as a marketing gimmick or an unnecessary add-on, but as a pharmacologically rational combination where each ingredient solves a specific problem that tirzepatide alone creates. For anyone just getting started with peptides, the compound formulation represents one of the more sophisticated applications of multi-ingredient design in the GLP-1 space.
How the three ingredients work together synergistically
Individual benefits are one thing. Synergy is another.
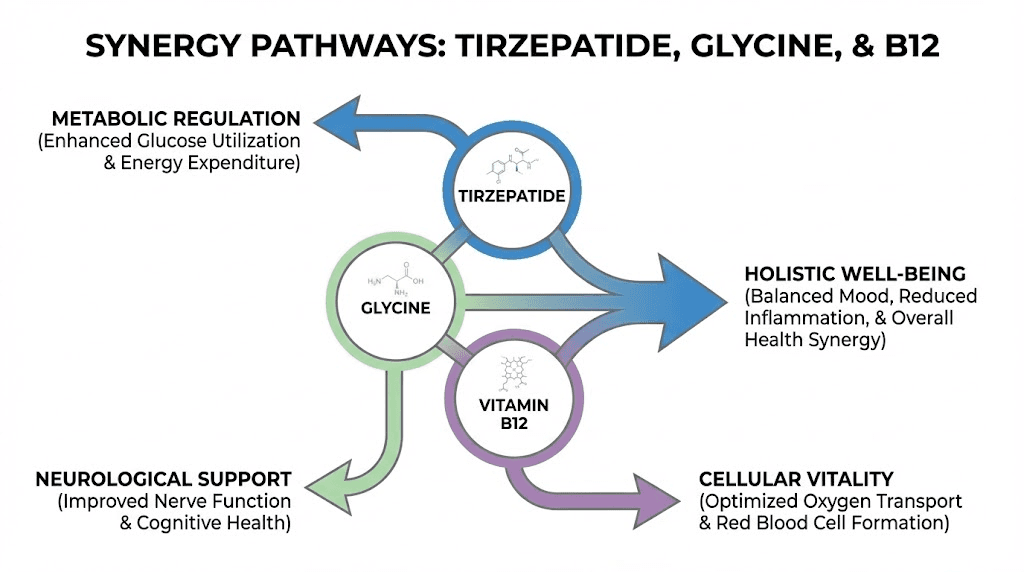
The tirzepatide/glycine/B12 compound is more than the sum of its parts because the three ingredients interact in ways that amplify each other while counteracting the weaknesses inherent in tirzepatide monotherapy. Understanding these synergies is what separates informed users from those who simply follow whatever their provider prescribes without asking questions. The peptide stacking guide covers the general principles of combining compounds for enhanced effects.
Synergy one: enhanced fat-to-muscle loss ratio
Tirzepatide alone produces weight loss that is roughly 75% fat and 25% lean mass. That ratio is already better than most interventions, but it still means significant muscle loss during extended treatment. The goal of the compound formulation is to push that ratio closer to 85% or even 90% fat loss while preserving lean tissue.
Glycine contributes to this shift through mTORC1 activation, as discussed above. But B12 plays a supporting role that is often overlooked. Adequate B12 status is essential for energy metabolism. When B12 is depleted, which happens gradually on GLP-1 medications, the body becomes less efficient at metabolizing fat and more likely to break down muscle for energy. By maintaining B12 levels, the compound helps ensure that the metabolic machinery responsible for fat oxidation operates at full capacity.
The practical result is that users of the combined formulation often report better body composition outcomes than those using tirzepatide alone. This is not just about the number on the scale. It is about what that number represents. Losing 30 pounds of mostly fat versus losing 30 pounds that includes 8 to 10 pounds of muscle represents dramatically different outcomes for metabolic health, physical function, and long-term weight maintenance. The resources at peptides for fat loss explore how different compounds approach this fat-versus-muscle balance.
Synergy two: nausea management that preserves efficacy
This is the synergy that researchers find most compelling.
One of the biggest challenges with tirzepatide is that the doses required for maximum weight loss are also the doses most likely to cause nausea. The standard approach is to titrate slowly, starting at 2.5 mg and increasing by 2.5 mg every four weeks. Even with this careful approach, many users hit a nausea wall at higher doses that either forces them to plateau or abandon the medication entirely.
B12 in the compound addresses nausea at the pharmacological level by reducing brain penetration. Glycine adds a complementary effect through its role as an inhibitory neurotransmitter. Glycine has calming effects on the central nervous system and has been shown to reduce gastric inflammation. These two mechanisms attack nausea through different pathways simultaneously.
The practical implication is significant. Users who tolerate the compound formulation better can titrate to higher doses faster and maintain those doses longer, leading to greater total weight loss. A user who could only tolerate 7.5 mg of tirzepatide alone might comfortably reach 10 mg or even 15 mg with the glycine/B12 compound. The difference between 7.5 mg and 15 mg over a full treatment course is substantial. The SURMOUNT data shows the gap clearly: 16% body weight loss on 5 mg versus 22.5% on 15 mg. The best peptides for weight loss ranking consistently places tirzepatide at the top, and the compound formulation pushes that advantage further. For anyone struggling with side effects when comparing GLP-1 medications, the compound approach offers a potential path forward.
Synergy three: collagen and metabolic recovery
Rapid weight loss stresses the body in ways that go beyond simple caloric deficit. Skin loses elasticity. Connective tissue remodels. Metabolic pathways shift. The combination of glycine and B12 addresses these recovery processes simultaneously.
Glycine is the primary amino acid in collagen. About one-third of all collagen amino acids are glycine. During weight loss, the body needs adequate glycine to remodel skin and connective tissue as fat deposits shrink. Without sufficient glycine, skin elasticity suffers, and the loose skin that many people fear becomes more likely.
B12, meanwhile, supports the methylation cycle that drives cellular repair and regeneration. Methylation is the process by which cells repair DNA, produce new proteins, and maintain healthy function. When B12 is deficient, methylation slows, and cellular repair processes become less efficient at exactly the time the body needs them most.
Together, glycine providing structural building materials and B12 supporting the cellular machinery that uses those materials, the compound creates better conditions for the body to adapt to rapid weight change. This is why providers who specialize in GLP-1 therapy increasingly recommend compound formulations over plain tirzepatide. The science supports the combination, and the clinical observations align with the research. Reviewing the broader peptide dosage chart library helps contextualize how compound dosing fits within the larger peptide landscape. For a broader understanding of how different peptides support anti-aging processes, the collagen connection provides important context.
Synergy four: sleep quality and recovery
This synergy is less discussed but clinically relevant.
Glycine has demonstrated sleep-improving properties in multiple studies. Research shows that 3 grams of glycine taken before bed improves subjective sleep quality, reduces time to fall asleep, and improves next-day cognitive function. While the glycine in a tirzepatide compound is delivered via injection rather than oral supplementation, the systemic effects on sleep-promoting pathways still apply.
Why does sleep matter for tirzepatide users specifically? Because sleep deprivation increases hunger hormones, reduces insulin sensitivity, and promotes muscle catabolism. Poor sleep essentially counteracts many of the benefits tirzepatide provides. By improving sleep quality through glycine, the compound may indirectly enhance the metabolic benefits of the tirzepatide component. Users who report feeling tired on GLP-1 medications, a topic covered extensively in the tirzepatide fatigue guide, may find that the glycine component provides meaningful relief.
B12 supports this synergy by maintaining healthy nerve function and energy metabolism. Low B12 contributes to fatigue and poor sleep architecture. By maintaining adequate B12 levels, the compound helps ensure that users get the restorative sleep their bodies need during a demanding weight loss protocol.
Typical formulation concentrations and what they mean
Not all tirzepatide/glycine/B12 compounds are created equal. Different compounding pharmacies use different concentrations, and understanding what those numbers mean is essential for proper dosing and setting realistic expectations about what you are actually injecting.
Standard concentration: 30/5/0.5 mg/mL
The most common formulation is 30/5/0.5 mg/mL, which means each milliliter contains 30 mg of tirzepatide, 5 mg of glycine, and 0.5 mg (500 mcg) of vitamin B12. This is typically supplied in 2 mL vials, giving a total vial content of 60 mg tirzepatide, 10 mg glycine, and 1 mg B12.
At a starting dose of 2.5 mg tirzepatide, you would draw approximately 0.083 mL (or about 8.3 units on an insulin syringe). That same injection would deliver approximately 0.42 mg of glycine and 0.042 mg of B12. At the maximum dose of 15 mg tirzepatide, you would draw 0.5 mL, delivering 2.5 mg glycine and 0.25 mg B12.
This means the glycine and B12 doses scale proportionally with the tirzepatide dose. Higher tirzepatide doses automatically deliver more glycine and B12. This is intentional, because the side effects and muscle loss risks also increase with dose, so the supportive ingredients scale to match. Using a compounded tirzepatide dosage calculator can help you determine exactly how much of each ingredient you receive at any given dose.
Other common concentrations
Compounding pharmacies also produce tirzepatide/glycine/B12 at other tirzepatide concentrations. Here is how the math works across common options:
10 mg/mL tirzepatide formulations: These lower-concentration vials require larger injection volumes. A 2.5 mg dose requires 0.25 mL. A 15 mg dose requires 1.5 mL. The larger volumes can be less comfortable for injection but provide more glycine and B12 per tirzepatide dose (assuming proportional compounding). The tirzepatide 10 mg dosage chart provides detailed volume calculations for this concentration.
15 mg/mL tirzepatide formulations: A middle-ground option. Injection volumes are moderate, and the glycine/B12 ratios provide adequate support at most dose levels. This concentration works well for users in the 5 mg to 10 mg tirzepatide range who want reasonable injection volumes.
20 mg/mL tirzepatide formulations: Higher concentration means smaller injection volumes, which many users prefer. However, the glycine and B12 per injection may be lower unless the pharmacy adjusts the ratios. The tirzepatide 20 mg/mL dosing chart details exact calculations for this concentration.
Reading your vial label
Your compound vial label should list all three ingredients with their concentrations. A typical label reads something like:
Tirzepatide 30 mg/mL, Glycine 5 mg/mL, Cyanocobalamin 0.5 mg/mL
Note that some pharmacies use cyanocobalamin (the synthetic form of B12) while others use methylcobalamin (the bioactive form). Methylcobalamin is generally preferred because it does not require hepatic conversion, but both forms are effective. The methylcobalamin guide covers the differences between B12 forms in detail.
Always verify that the vial label matches what was prescribed. Compounding errors, while rare, do occur. If the concentrations do not match your prescription or if you see any ingredient you do not recognize, contact the pharmacy before using the product. The reconstituting tirzepatide chart and printable reconstitution reference can be kept alongside your vial for quick verification during each preparation.
Vial size and number of doses
Most tirzepatide/glycine/B12 compounds come in 2 mL vials. At a 30 mg/mL concentration, a single 2 mL vial contains 60 mg of tirzepatide total. The number of weekly doses you can draw from one vial depends on your current tirzepatide dose:
At 2.5 mg per week, one vial lasts 24 weeks. At 5 mg per week, one vial lasts 12 weeks. At 7.5 mg per week, one vial lasts 8 weeks. At 10 mg per week, one vial lasts 6 weeks. At 15 mg per week, one vial lasts 4 weeks. The 2.5 mg unit conversion guide can help verify your exact injection volume at the starting dose.
However, once reconstituted (if applicable) or once the vial is first punctured, stability becomes a factor. Most compounding pharmacies recommend using vials within 28 to 60 days of first puncture when stored properly in the refrigerator. Understanding how long tirzepatide lasts in the fridge is critical for users at lower doses who will be using the same vial for weeks. The tirzepatide storage requirements apply to compound formulations just as they do to plain tirzepatide.
Complete dosing protocol for the compound formulation
Dosing a triple compound requires understanding all three components simultaneously. The tirzepatide dose follows standard titration schedules, but the glycine and B12 doses are determined by the fixed ratios in the compound. This section walks through the complete protocol from start to maintenance.
Standard titration schedule
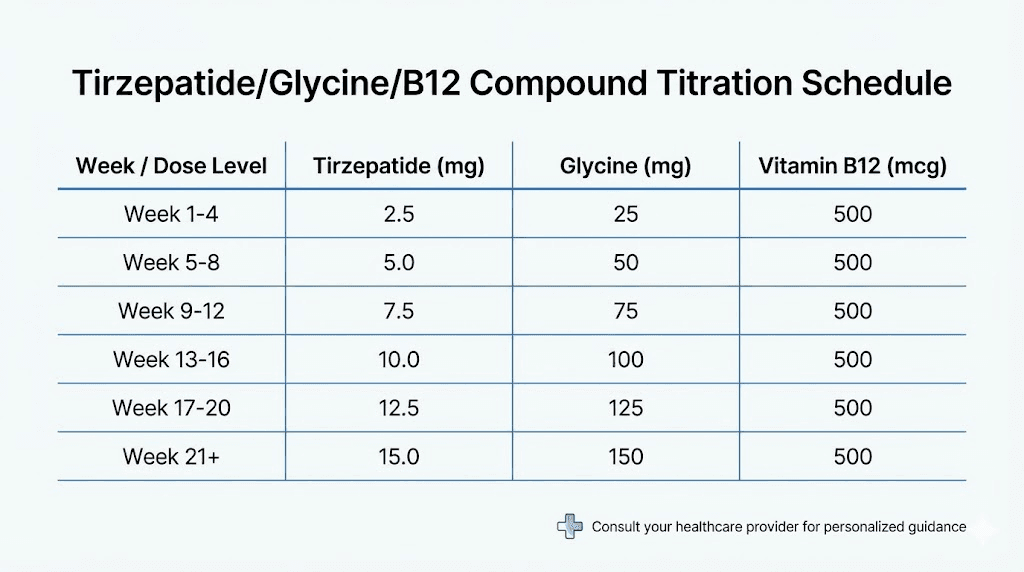
The recommended approach follows the FDA-approved tirzepatide titration, adapted for compounded formulations:
Weeks 1 through 4: 2.5 mg tirzepatide per week. This starting dose allows the body to adjust to GLP-1/GIP receptor activation with minimal side effects. At 30/5/0.5 mg/mL concentration, draw 0.083 mL (approximately 8 units on a standard insulin syringe). This delivers roughly 0.42 mg glycine and 42 mcg B12.
Weeks 5 through 8: 5 mg tirzepatide per week. The first titration increase. Draw 0.167 mL (approximately 17 units). Delivers roughly 0.83 mg glycine and 83 mcg B12. Most users tolerate this increase well if they managed the starting dose without significant issues. For detailed unit calculations, the 5 mg unit conversion guide provides exact numbers.
Weeks 9 through 12: 7.5 mg tirzepatide per week. Draw 0.25 mL (25 units). Delivers 1.25 mg glycine and 125 mcg B12. This is where many users begin to experience more noticeable appetite suppression and, potentially, more GI side effects. The increased glycine and B12 from the higher dose help offset these effects. Check the 7.5 mg unit guide for precise measurements.
Weeks 13 through 16: 10 mg tirzepatide per week. Draw 0.333 mL (approximately 33 units). Delivers 1.67 mg glycine and 167 mcg B12. For many users, 10 mg represents the sweet spot where weight loss is robust and side effects are manageable.
Weeks 17 through 20: 12.5 mg tirzepatide per week. Draw 0.417 mL (approximately 42 units). Delivers 2.08 mg glycine and 208 mcg B12. Not all users need to go this high. If weight loss is progressing well at 10 mg with acceptable side effects, staying at that dose is perfectly reasonable.
Week 21 and beyond: 15 mg tirzepatide per week (maximum). Draw 0.5 mL (50 units). Delivers 2.5 mg glycine and 250 mcg B12. This is the highest approved dose and produces the maximum weight loss effect. The complete dosing in units guide covers all dose levels with exact calculations. You can also reference the tirzepatide dosage in units overview and the dosage chart in units for visual reference at each dose level.
Slower titration option
Some providers recommend extending each dose level to six or eight weeks instead of four. This gives the body more time to adapt and reduces the severity of side effects at each new dose. The compound formulation makes this approach even more viable because the glycine and B12 are already helping manage side effects, so the urgency to push through a difficult dose transition is reduced.
The microdosing tirzepatide chart provides an alternative approach for users who want even more gradual titration, with dose increases of 1.25 mg at a time rather than the standard 2.5 mg jumps.
Drawing the correct volume
Accuracy matters with compound formulations because you are dosing three ingredients simultaneously. An error in injection volume affects all three components proportionally. Use an insulin syringe with clear unit markings. For most tirzepatide compound formulations, a 0.5 mL or 1 mL insulin syringe with half-unit markings provides sufficient precision. The 20 units conversion guide, 40 units guide, and 50 units guide provide quick references for common syringe readings.
The process follows standard tirzepatide preparation procedures. If the compound requires reconstitution, follow the pharmacy instructions exactly. Many compound formulations come pre-mixed and ready to draw, which eliminates the reconstitution step entirely. The peptide reconstitution calculator can verify volumes if needed, and the tirzepatide reconstitution chart provides a quick reference.
Administration technique
The tirzepatide/glycine/B12 compound is administered as a once-weekly subcutaneous injection. The technique is identical to standard tirzepatide injection. Choose a site on the abdomen (at least two inches from the navel), the front of the thigh, or the back of the upper arm. Rotate injection sites weekly to prevent lipodystrophy and irritation.
Inject slowly. Compound formulations sometimes have slightly different viscosity than plain tirzepatide due to the additional ingredients. Rushing the injection can cause more injection site discomfort. A steady 10-second injection is generally sufficient for the small volumes involved. For complete injection guidance, the GLP-1 injection site guide and the injection technique guide cover proper methods in detail.
Why glycine matters more than most people realize
Glycine deserves more attention than it typically receives in discussions about tirzepatide compounds. Most conversations focus on the tirzepatide itself and maybe mention B12 as a bonus. Glycine gets treated as an afterthought. That is a mistake.
The muscle preservation crisis in GLP-1 therapy
Here is the uncomfortable truth about GLP-1 medications. They work extraordinarily well for weight loss. Too well, in some cases. The aggressive caloric deficit they create, through both appetite suppression and metabolic effects, puts muscle tissue at risk. And muscle loss during weight loss is not a cosmetic concern. It is a metabolic disaster.
Muscle is metabolically active tissue. Each pound of muscle burns roughly 6 to 7 calories per day at rest, compared to about 2 calories per pound for fat tissue. Lose 10 pounds of muscle, and your resting metabolic rate drops by approximately 60 to 70 calories per day. Over a year, that deficit adds up to more than 7 pounds of potential fat regain. This is one of the primary mechanisms behind the weight regain that occurs when people stop GLP-1 medications, a pattern also explored in the GLP-1 discontinuation guide. They lost muscle during treatment, their metabolism slowed, and when they resume normal eating patterns, they gain fat back faster than someone who preserved their muscle mass.
The problem is not small. Clinical trials show lean mass loss accounting for 26% to 40% of total weight reduction on GLP-1 medications. For someone losing 60 pounds, that could mean losing 15 to 24 pounds of lean tissue. Understanding the relationship between peptides and muscle growth provides important context for why preserving muscle during weight loss protocols matters so much for long-term outcomes.
How glycine activates muscle protection
Glycine activates the mTORC1 pathway in muscle cells. mTORC1 (mechanistic target of rapamycin complex 1) is the central regulator of muscle protein synthesis. When mTORC1 is active, muscle cells build and maintain protein. When it is suppressed, muscle cells shift toward catabolism.
During caloric restriction, mTORC1 activity naturally decreases. The body senses reduced energy availability and downregulates growth processes, including muscle maintenance. This is a survival mechanism. But in the context of intentional weight loss with adequate protein intake, this default response is counterproductive. We want to lose fat while maintaining muscle, and the body default programming does not distinguish between these goals.
Glycine pushes back against this catabolic shift. Research shows that glycine-treated muscle cells maintain mTORC1 activity even under nutrient-restricted conditions. The muscle cells in glycine-treated groups were up to 20% larger than control groups under identical caloric conditions. That is a meaningful difference that translates directly to better body composition outcomes during GLP-1-mediated weight loss.
For researchers interested in the broader role of amino acids and peptides in muscle maintenance, the safest peptides for muscle growth guide provides additional options and considerations.
Glycine and collagen: the skin elasticity connection
Rapid weight loss creates a practical problem that goes beyond the scale. Skin elasticity.
When fat deposits shrink quickly, the overlying skin must remodel and tighten to match the reduced volume. This process requires collagen, specifically new collagen production to replace the stretched fibers. Glycine is the most abundant amino acid in collagen, comprising approximately one-third of the total amino acid content. Without adequate glycine, collagen synthesis slows, and skin remodeling cannot keep pace with fat loss.
Studies show that glycine supplementation can improve skin elasticity by up to 20% within 8 to 12 weeks. For someone losing weight rapidly on tirzepatide, starting glycine supplementation (via the compound) from day one means the collagen support is in place throughout the entire weight loss phase, not added after the damage is done. The collagen peptide guide explores different forms of collagen support in greater detail.
Glycine anti-inflammatory and metabolic benefits
Glycine is a potent anti-inflammatory agent. It suppresses inflammatory cytokines, reduces oxidative stress, and supports healthy immune function. These properties are particularly relevant during weight loss because adipose tissue (especially visceral fat) releases inflammatory signals as it shrinks. The inflammatory response to fat mobilization can cause joint pain, fatigue, and general malaise that many tirzepatide users experience, sometimes described as GLP-1 flu.
By dampening this inflammatory response, glycine in the compound may reduce the general discomfort that accompanies rapid weight loss. Users who report muscle pain on tirzepatide or headaches may find some relief from the anti-inflammatory properties of the glycine component.
Glycine also improves insulin sensitivity through mechanisms independent of the tirzepatide itself. Research shows that glycine supplementation enhances glucose tolerance and reduces fasting blood sugar. In combination with tirzepatide dual GLP-1/GIP activation, glycine provides a complementary metabolic benefit that may enhance overall glycemic control. For users whose primary goal is metabolic health rather than weight loss alone, this additional benefit is significant.
Why B12 is not just a nice to have in the compound
Vitamin B12 might seem like the least important ingredient in a tirzepatide/glycine/B12 compound. It is a vitamin. You can get it from food or a cheap supplement. Why include it in an expensive compounded injection?
Because the research shows it does something in this context that a standalone B12 supplement cannot replicate.
The nausea reduction mechanism explained
This is the finding that changed how compounding pharmacists think about GLP-1 formulations.
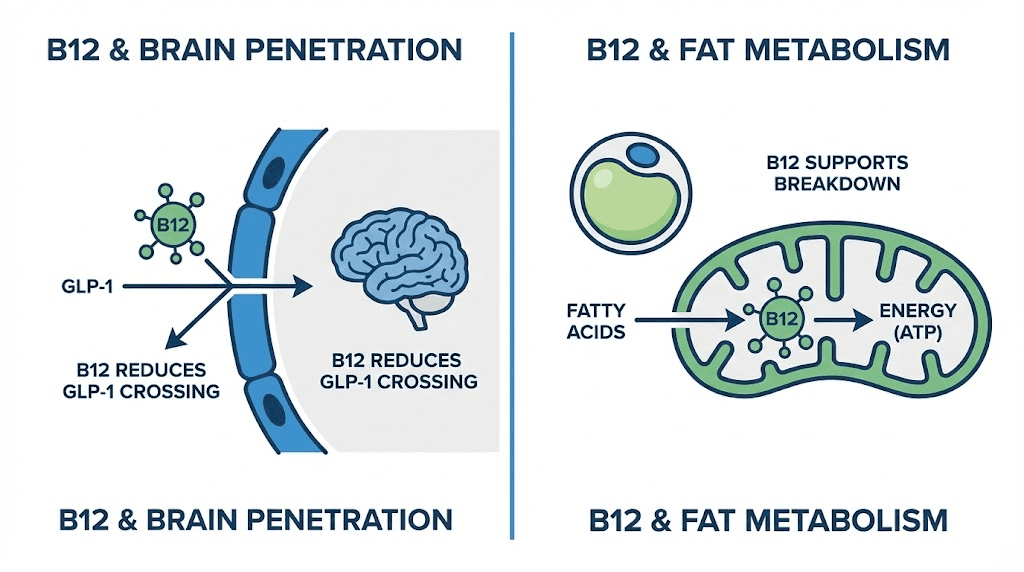
The PMC5899935 study examined what happens when B12 is conjugated with GLP-1 receptor agonists. The researchers found that B12 acts as a molecular shield, reducing the ability of GLP-1 compounds to cross the blood-brain barrier. The GLP-1 compound still circulates systemically. It still activates peripheral GLP-1 receptors in the gut, pancreas, and adipose tissue. But less of it reaches the central nervous system.
Why does this matter for nausea? Because the nausea response to GLP-1 medications is mediated primarily by brainstem receptors, specifically the area postrema and nucleus tractus solitarius. These brain regions detect GLP-1 signaling and trigger the nausea response as a protective mechanism. By reducing how much tirzepatide reaches these brain centers, B12 attenuates the nausea signal without meaningfully reducing the metabolic effects that occur peripherally.
Think of it this way. The weight loss effects of tirzepatide happen primarily outside the brain, in the gut, pancreas, fat tissue, and liver. The nausea happens primarily inside the brain. B12 reduces brain penetration. So you get to keep most of the weight loss while losing some of the nausea. That is a trade most users are happy to make.
This mechanism is distinct from simply taking B12 separately. The B12 needs to be present in the same formulation as the tirzepatide to exert this brain-penetration effect. Taking a B12 pill alongside a plain tirzepatide injection does not produce the same result. The physical co-formulation matters. This distinction is critical for understanding why the compound exists as a single product rather than as separate components.
Addressing GLP-1-induced B12 deficiency
The second role of B12 in the compound is more straightforward but equally important over the long term.
GLP-1 medications impair B12 absorption through multiple mechanisms. They reduce gastric acid production (intrinsic factor activation requires acid). They slow gastric emptying (reducing the time food spends in the optimal absorption zone). And they alter gut microbiome composition (certain bacteria produce and facilitate B12 absorption).
Over months to years of treatment, these effects can lead to clinically significant B12 deficiency. Symptoms include fatigue, numbness and tingling in the hands and feet (peripheral neuropathy), cognitive difficulties, mood changes, and in severe cases, irreversible nerve damage. The GLP-1 fatigue guide discusses how nutrient depletion contributes to the tiredness many users experience.
By delivering B12 directly through injection, the compound bypasses the digestive absorption pathway entirely. The B12 enters systemic circulation directly from the subcutaneous injection site, no stomach acid, no intrinsic factor, no gut microbiome involvement required. This is the same principle behind B12 injections that have been used for decades to treat pernicious anemia and other B12 malabsorption conditions.
B12 and energy metabolism during caloric restriction
B12 is a cofactor in two critical metabolic reactions. First, it is required for the conversion of methylmalonyl-CoA to succinyl-CoA, a step in fatty acid metabolism. Second, it is necessary for the conversion of homocysteine to methionine, a key step in the methylation cycle.
During the caloric restriction that tirzepatide creates, efficient fat metabolism is essential. If B12 is deficient, the body cannot process fatty acids as efficiently, which can lead to the fatigue, brain fog, and metabolic sluggishness that some GLP-1 users report. By maintaining optimal B12 levels through the compound, users ensure that their bodies can efficiently metabolize the fat being released from adipose tissue.
This has practical implications for energy levels and daily function. Many tirzepatide users report a period of adjustment where energy levels dip before stabilizing. B12 in the compound may shorten this adjustment period and reduce its severity. Users who have experienced fatigue on semaglutide and switched to a tirzepatide/B12 compound often report improved energy, though individual responses vary. For anyone asking whether GLP-1 medications can actually boost energy, the semaglutide energy guide provides relevant context.
How the compound compares to plain tirzepatide
The question everyone asks: is the compound formulation actually better than plain tirzepatide? The honest answer is that it depends on your priorities, your tolerance profile, and your specific health situation. But the comparison reveals clear advantages in several key areas.
Side effect profile comparison
Plain tirzepatide and the compound formulation share the same base medication, so the fundamental side effect profile is similar. However, the compound formulation appears to modify the severity and frequency of several common side effects:
Nausea: The most common tirzepatide side effect, affecting 40% to 70% of users at some point. The B12 component reduces brain penetration of the GLP-1 compound, potentially lowering nausea severity. Users and providers report that compound formulations with B12 tend to produce less nausea than equivalent doses of plain tirzepatide, though controlled head-to-head studies in humans are limited.
Fatigue: Common during the first weeks at each new dose. B12 in the compound helps maintain energy metabolism, and glycine anti-inflammatory properties may reduce the general malaise that accompanies early treatment. The combination of both ingredients addresses fatigue through complementary mechanisms.
Muscle pain and weakness: Reported by a subset of tirzepatide users, particularly at higher doses. Glycine mTORC1 activation and anti-inflammatory effects may provide some protection against the muscle-related side effects that concern many users.
Constipation: Still occurs with the compound formulation because it is driven by the GLP-1 component slowdown of gastric emptying. The glycine and B12 do not specifically address constipation. Users who experience this side effect still need the same management strategies described in the tirzepatide constipation treatment guide.
Injection site reactions: The compound formulation may have slightly different injection site characteristics due to the additional ingredients. Some users report less injection site irritation with compounds that include glycine (which has pH-buffering properties), while others notice no difference.
Body composition outcomes
This is where the compound formulation shows its most compelling theoretical advantage.
Plain tirzepatide produces weight loss that is approximately 75% fat mass and 25% lean mass. The glycine component in the compound formulation, through mTORC1 activation and anti-catabolic effects, may improve this ratio. While large-scale clinical trials comparing plain tirzepatide to the compound formulation have not been conducted, the mechanistic evidence is strong, and provider observations generally support better muscle preservation with the compound.
The practical difference matters enormously for long-term outcomes. Someone who maintains more muscle during weight loss will have a higher resting metabolic rate afterward, making weight maintenance easier. They will have better physical function, better insulin sensitivity per pound of body weight, and a lower risk of the metabolic slowdown that leads to weight regain. The tirzepatide before and after results page and the men before and after gallery show real outcomes, and compound users often report being particularly satisfied with body composition changes rather than just scale weight. The comprehensive results gallery demonstrates the range of transformations possible.
Cost comparison
Compound formulations typically cost more than plain compounded tirzepatide. The additional ingredients, the more complex compounding process, and the quality testing required for multi-ingredient formulations all contribute to higher prices. However, the difference is often less than users expect.
A plain compounded tirzepatide vial might cost 20% to 40% less than an equivalent compound vial with glycine and B12. But consider the total cost picture. If you would otherwise be purchasing glycine supplements, B12 injections, or spending money on managing side effects that the compound helps prevent, the compound may actually be more economical overall. Resources like the peptide cost calculator and the affordable tirzepatide guide can help evaluate the total cost picture. For a broader view of compounding pharmacy pricing, the compounded tirzepatide cost guide provides current market context.
When plain tirzepatide might be the better choice
The compound is not universally superior. Plain tirzepatide may be preferable if you are sensitive to B12 supplementation (rare but possible), if you prefer to control glycine and B12 supplementation separately for precise dosing, if cost is the primary concern and you are tolerating tirzepatide well without additional support, or if your provider has specific reasons for preferring a single-ingredient formulation. Understanding the landscape of compounded GLP-1 medications helps contextualize the compound versus plain debate.
Optimizing results on the tirzepatide glycine B12 compound
The compound formulation provides a better foundation than plain tirzepatide, but the formulation alone does not guarantee optimal results. What you do alongside the compound matters enormously. Nutrition, exercise, supplementation, and lifestyle factors all interact with the three ingredients to determine your actual outcomes.
Protein intake: the non-negotiable foundation
Research consistently shows that protein intake of 1.2 to 1.6 grams per kilogram of body weight per day helps preserve lean mass during caloric restriction. For a 200-pound (91 kg) person, that translates to 109 to 145 grams of protein daily. For a 150-pound (68 kg) person, 82 to 109 grams daily.
The glycine in the compound supports muscle preservation through mTORC1 activation, but it works best when combined with adequate total protein intake. mTORC1 activation without sufficient amino acid building blocks is like turning on a construction crew without providing materials. The signal to build is there, but the resources are not. Make protein your priority at every meal. The tirzepatide nutrition guide and the foods to avoid guide provide meal-specific recommendations.
When appetite suppression makes eating enough protein difficult (as it often does on tirzepatide), prioritize protein-dense foods over everything else. Lean meats, eggs, Greek yogurt, cottage cheese, and protein shakes can help you meet your targets even when total food intake is dramatically reduced. The GLP-1 diet planning guide offers meal templates that ensure adequate protein intake at every calorie level.
Resistance training: activating what glycine protects
Glycine activates mTORC1 signaling. Resistance training also activates mTORC1 signaling. The two together produce a synergistic effect that is greater than either alone.
Without exercise, glycine helps preserve existing muscle. With resistance training, glycine helps build new muscle while also preserving what is already there. The compound formulation is designed to be used alongside physical activity, not as a replacement for it.
For tirzepatide users, the resistance training recommendation is straightforward. Train each major muscle group at least twice per week with moderate to heavy loads. Compound movements like squats, deadlifts, bench press, rows, and overhead press are the most efficient choices. Each session does not need to be long. Three to four sets per exercise, 8 to 12 repetitions, three to four sessions per week is sufficient. For men specifically, the fat burning peptides for men guide discusses how to combine GLP-1 therapy with resistance training for maximum body recomposition. Women can find targeted guidance in the peptides for weight loss in women resource.
The critical point is that resistance training during GLP-1 therapy is not optional for anyone who cares about body composition outcomes. It is the single most important lifestyle factor for muscle preservation, and it works synergistically with the glycine component of the compound. Resources at peptides for muscle growth explore how various compounds interact with exercise to enhance body composition.
Additional supplementation considerations
The compound provides glycine and B12, but there are other supplements that complement the formulation:
Creatine monohydrate (3 to 5 grams daily): Supports muscle cell hydration and energy production. Well-studied, safe, and particularly beneficial during caloric restriction. Works synergistically with glycine mTORC1 activation.
Vitamin D (2,000 to 5,000 IU daily): Supports calcium absorption, immune function, and muscle protein synthesis. Many people are deficient, and GLP-1 medications may further impair absorption. Get levels checked and supplement accordingly.
Magnesium (200 to 400 mg daily): Supports muscle function, sleep quality, and bowel regularity. The laxative effect of magnesium citrate or magnesium oxide can help counteract tirzepatide-related constipation.
Omega-3 fatty acids (2 to 4 grams daily): Anti-inflammatory properties complement glycine, and omega-3s support cardiovascular health during the metabolic changes that accompany weight loss.
Collagen peptides (10 to 15 grams daily): While the compound includes glycine (a key collagen amino acid), additional collagen supplementation provides the other amino acids needed for complete collagen synthesis. The glutamine peptides guide covers another amino acid that supports gut health and recovery during GLP-1 therapy.
For a comprehensive look at what to combine with GLP-1 medications, the supplements to take with tirzepatide guide covers the full range of evidence-based options.
Timing your injection for best results
The once-weekly injection should be given on the same day each week. Most users find that injecting in the morning works best because any initial side effects (mild nausea, reduced appetite) align with daytime hours when they can be managed actively.
Some users prefer injecting in the evening, reasoning that any nausea will be slept through. This approach can work, but note that glycine promotes sleep quality, so an evening injection may actually provide a dual benefit: the nausea reduction from the B12 component plus the sleep-promoting effects of the glycine component.
There is no definitive evidence that one timing is superior. The best timing is whatever allows you to be consistent. Understanding how long tirzepatide takes to work and when appetite suppression kicks in helps you plan meals and activities around your injection day. Some users find guidance in the best time of day for GLP-1 medications resource, and the appetite suppression timeline sets realistic expectations for how quickly the medication takes effect.
Compounding pharmacy considerations
Not all compounding pharmacies are equal, and the quality of your tirzepatide/glycine/B12 compound depends entirely on the pharmacy that produces it. This section covers what to look for, what to avoid, and how to evaluate the product you receive.
Pharmacy accreditation and quality markers
The single most important quality indicator for a compounding pharmacy is PCAB (Pharmacy Compounding Accreditation Board) accreditation. PCAB-accredited pharmacies undergo rigorous inspections and must meet standards that exceed basic state requirements. They must demonstrate proper sterile compounding techniques, quality testing procedures, and staff training.
Beyond accreditation, look for pharmacies that provide certificates of analysis (COA) for each batch. A COA confirms that the compound has been tested for potency (correct concentration of each ingredient), sterility (no microbial contamination), endotoxin levels (within safe limits), and pH (within stability range). If a pharmacy cannot or will not provide a COA, that is a red flag. The Peptide Sciences review is one example of how different vendors approach quality documentation.
Several well-known compounding pharmacies produce tirzepatide compounds. The Empower pharmacy guide (with its dosage chart), Olympia pharmacy guide, Southend pharmacy guide, and Lavender Sky guide provide detailed information about specific pharmacy options and their formulations. The compound dosage chart and compound dosing chart offer pharmacy-agnostic references that work across vendors.
FDA status and regulatory context
It is important to understand the regulatory landscape. Compounded tirzepatide/glycine/B12 formulations are NOT FDA-approved products. The FDA approves the brand-name tirzepatide product (Mounjaro for diabetes, Zepbound for weight loss), but compounded versions are produced under a different regulatory framework.
Compounding pharmacies operate under Section 503A or 503B of the FDA, FD&C Act. Section 503A pharmacies compound medications based on individual prescriptions and are primarily regulated by state boards of pharmacy. Section 503B pharmacies (outsourcing facilities) are registered with the FDA and subject to more stringent federal oversight, including regular FDA inspections.
For tirzepatide compounds, 503B outsourcing facilities generally provide a higher level of quality assurance due to the federal oversight and batch testing requirements. However, high-quality 503A pharmacies with PCAB accreditation can also produce excellent products. The key is doing your due diligence regardless of the pharmacy classification. Resources like the injectable peptides directory and best peptides for fat loss guide provide additional context for evaluating products and their sources. Understanding the broader context of peptide safety and risks helps you make informed decisions about sourcing.
Storage and handling of the compound
The tirzepatide/glycine/B12 compound requires refrigeration, typically between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). The glycine and B12 components do not significantly alter the storage requirements compared to plain tirzepatide, but the multi-ingredient nature of the formulation may affect stability timelines.
Most pharmacies assign a beyond-use date (BUD) of 28 to 60 days from the date of compounding (or from the date of first puncture, whichever is shorter). This varies by pharmacy and depends on the specific formulation and any stability testing they have performed. Always check the label for the BUD and do not use the product beyond that date.
Brief temperature excursions (a few hours outside the refrigerator during shipping or daily use) are generally tolerable but should be minimized. The guide to tirzepatide temperature exposure covers the limits in detail, and the tirzepatide expiration guide addresses shelf life questions. For general best practices, the peptide storage guide provides comprehensive storage protocols. Related questions about storing peptides after reconstitution and peptide refrigerator shelf life are also worth reviewing.
Evaluating your compound upon receipt
When you receive your tirzepatide/glycine/B12 compound, inspect it before use. The solution should be clear and colorless to very slightly yellow. There should be no visible particles, cloudiness, or discoloration. The vial should be intact with no cracks, and the rubber stopper should be sealed and undamaged.
Check the label against your prescription. Verify that the concentrations match, the pharmacy name and license number are listed, the beyond-use date is within an acceptable range, and storage instructions are included. If anything seems off, contact the pharmacy before using the product. The peptide vendor evaluation guide provides broader context for assessing quality across all peptide products. For handling the mixing process specifically, the peptide reconstitution guide, the bacteriostatic water guide, and the mixing instructions cover the fundamentals.
Managing side effects specific to the compound
While the compound formulation is designed to reduce certain side effects compared to plain tirzepatide, side effects still occur. Understanding which side effects are common, which are concerning, and how to manage each one is essential for a successful treatment course.
Gastrointestinal effects
Nausea, diarrhea, constipation, and decreased appetite remain the most common side effects even with the compound formulation. The B12 component helps reduce nausea severity, but it does not eliminate it entirely. Most GI effects are dose-dependent and improve with time.
Nausea management beyond B12: If nausea persists despite the B12 in the compound, additional strategies include eating smaller, more frequent meals, avoiding high-fat and greasy foods, staying upright for 30 minutes after eating, ginger tea or ginger supplements, and ensuring adequate hydration. The tirzepatide diet plan provides meal-specific guidance designed to minimize nausea.
Constipation management: The glycine and B12 in the compound do not specifically address the constipation caused by GLP-1-mediated slowing of gut motility. Standard constipation management, including increased fiber, adequate hydration, magnesium supplementation, and regular physical activity, remains necessary. For severe cases, osmotic laxatives like polyethylene glycol (Miralift) are generally safe and effective. The complete tirzepatide constipation treatment guide covers every evidence-based approach.
Diarrhea: Less common than constipation but more disruptive when it occurs. The glycine component may help here, as glycine has anti-inflammatory effects on the gut lining. Stay hydrated, avoid dairy if it worsens symptoms, and contact your provider if diarrhea persists for more than a few days or is severe.
Fatigue and energy fluctuations
Some users experience fatigue, particularly during the first weeks at a new dose level. The B12 in the compound should help maintain energy metabolism, but the dramatic caloric reduction that tirzepatide creates can still cause transient energy dips.
The glycine component may actually help here as well. Glycine improves sleep quality, and better sleep translates to better daytime energy. Users who inject in the evening sometimes report that sleep quality improves from the first dose. Over the first few weeks, as the body adapts to the new caloric intake pattern, energy levels typically stabilize and often improve beyond baseline as excess weight comes off. The comprehensive GLP-1 fatigue guide covers the full range of energy management strategies.
Anxiety and mood changes
A subset of tirzepatide users report anxiety or mood changes. The mechanisms are not fully understood but may relate to hormonal shifts during weight loss, blood sugar fluctuations, or the psychological adjustment to dramatically reduced eating. Some users wonder if tirzepatide causes anxiety, and while it is not a common side effect in clinical trials, real-world reports are frequent enough to warrant attention.
Glycine, as an inhibitory neurotransmitter, may provide modest anxiolytic effects. It has calming properties that complement its other functions in the compound. B12 also supports neurological function and mood regulation. However, if anxiety or mood changes are significant, these should be discussed with a healthcare provider rather than managed solely through the compound ingredients.
Hair loss concerns
Telogen effluvium (temporary hair shedding) occurs in some GLP-1 users, typically beginning 2 to 4 months after starting treatment. This is related to the metabolic stress of rapid weight loss and caloric restriction, not to the medication itself directly. The GLP-1 hair loss guide provides detailed information on prevention and management.
The compound formulation may offer some protection against hair loss. B12 deficiency is a known contributor to hair loss, and maintaining adequate B12 through the compound addresses this risk factor. Glycine supports keratin production (hair protein) and collagen in the hair follicle matrix. Neither ingredient is a guaranteed hair loss prevention measure, but they address two of the nutritional factors that contribute to the problem.
When to contact your healthcare provider
Most side effects of the tirzepatide/glycine/B12 compound are manageable and temporary. However, certain symptoms require immediate medical attention.
Seek emergency care for severe abdominal pain that does not resolve (possible pancreatitis), signs of allergic reaction such as swelling, difficulty breathing, or widespread hives, persistent vomiting that prevents keeping down any fluids, or symptoms of thyroid problems such as neck swelling, difficulty swallowing, or voice changes.
Contact your provider promptly for side effects that worsen rather than improve over 2 to 3 weeks, any neurological symptoms like numbness, tingling, or vision changes, significant mood changes or depression, or unexplained skin rashes or injection site reactions that spread or worsen.
Comparing the compound to other tirzepatide formulations
The tirzepatide/glycine/B12 compound is not the only enhanced tirzepatide formulation available. Understanding how it compares to other options helps you and your provider make the best choice for your situation.
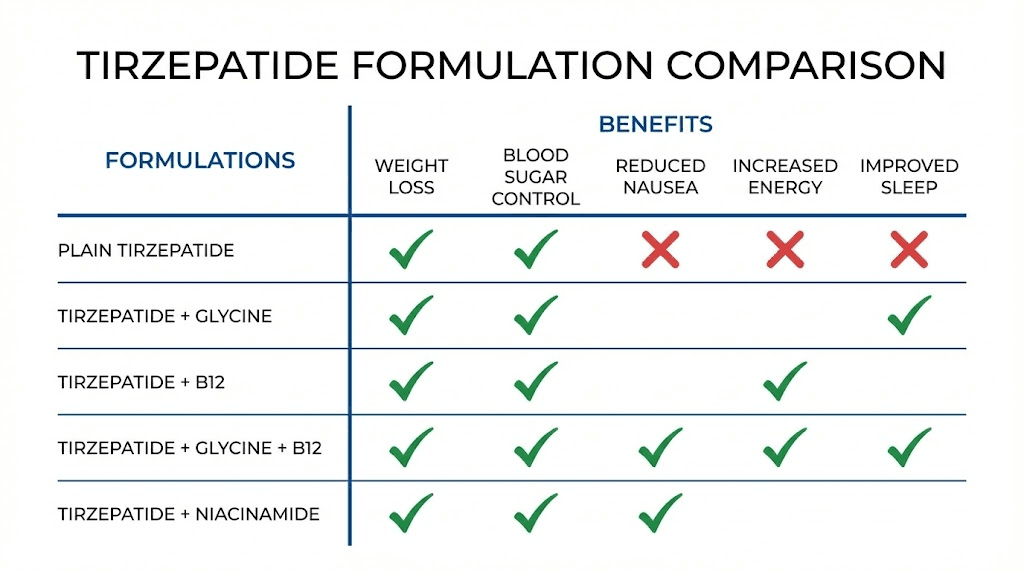
Tirzepatide/glycine (without B12)
Some compounding pharmacies offer tirzepatide with glycine alone, without B12. This formulation provides the muscle preservation and collagen benefits of glycine but misses the nausea reduction and B12 deficiency prevention that the full triple compound offers. The tirzepatide glycine guide covers this formulation in depth.
The glycine-only compound may be appropriate for users who already receive B12 supplementation through other means (such as separate B12 injections or sublingual B12), or for those who are primarily concerned about muscle preservation and collagen support rather than nausea management.
Tirzepatide/B12 (without glycine)
The tirzepatide/B12 formulation focuses on nausea reduction and nutrient support without the muscle preservation benefits of glycine. This might be preferred by users who are already getting adequate glycine through diet or supplementation, or who have a specific medical reason to avoid additional glycine. The tirzepatide with B12 guide provides comprehensive coverage of this option.
Tirzepatide/niacinamide
A newer formulation that combines tirzepatide with niacinamide (vitamin B3) instead of, or in addition to, glycine and B12. Niacinamide supports skin health and has anti-inflammatory properties, making it an interesting alternative for users whose primary concern is skin quality during weight loss rather than muscle preservation. The tirzepatide niacinamide guide explores this formulation and its unique benefits.
Semaglutide compound alternatives
For users considering their options across the GLP-1 class, semaglutide compounds with similar additions exist. Semaglutide with glycine and semaglutide with B12 formulations offer analogous benefits for the semaglutide platform. The semaglutide to tirzepatide conversion chart helps users considering a switch between the two base medications, and the switching guide covers the practical considerations involved. Pharmacy-specific dosing guides for semaglutide compounds, including those from Empower, Olympia, and Direct Meds, provide detailed comparison points.
Plain compounded tirzepatide
The simplest option is compounded tirzepatide without any additions. This is the least expensive compounded option and appropriate for users who tolerate tirzepatide well and prefer to manage supplementation separately. Dosing charts for plain compounded tirzepatide are available for various concentrations, including the 30 mg chart, general compound chart, and compound dosing chart.
Brand-name tirzepatide
Mounjaro (for type 2 diabetes) and Zepbound (for weight loss) are FDA-approved tirzepatide products from Eli Lilly. They do not contain glycine or B12. They are more expensive than compounded options but carry the full weight of FDA approval, including standardized manufacturing, extensive clinical trial data, and regulatory oversight. The semaglutide versus tirzepatide comparison provides context for both brand-name and compounded versions.
Delivery methods: injection versus oral options
The standard tirzepatide/glycine/B12 compound is delivered via subcutaneous injection. But the GLP-1 market is evolving rapidly, and alternative delivery methods are emerging that deserve discussion in the context of compound formulations.
Subcutaneous injection (standard)
The injectable form remains the gold standard for the compound. Subcutaneous injection ensures reliable bioavailability of all three components, consistent dosing accuracy, and the specific B12 co-formulation benefits (reduced brain penetration) that only occur when B12 is physically present with the tirzepatide in the same solution.
Injection technique for the compound is the same as for any subcutaneous peptide injection. The peptide injection guide covers the fundamentals, and the GLP-1 injection guide addresses specifics for this medication class.
Oral tirzepatide: emerging options
Oral tirzepatide formulations are in development. However, oral delivery presents unique challenges for a triple compound. Glycine is well-absorbed orally, B12 requires intrinsic factor for oral absorption (which GLP-1 medications impair), and tirzepatide oral bioavailability is still being optimized. The oral tirzepatide guide and the oral versus injection comparison discuss the current state of oral GLP-1 delivery.
For compound formulations specifically, injectable delivery is likely to remain the preferred route for the foreseeable future because it preserves the B12 nausea-reduction mechanism and ensures reliable delivery of all three ingredients. The injectable versus oral peptides comparison provides broader context for this delivery method debate.
Sublingual drops
Some compounding pharmacies offer tirzepatide as sublingual drops. The tirzepatide drops guide covers this option. Currently, tirzepatide/glycine/B12 as a sublingual formulation is not widely available because the volume required for effective dosing and the stability of the triple formulation in a sublingual vehicle present technical challenges. However, this may change as compounding technology evolves. The broader conversation about GLP-1 classification and brand-name products and bioactive precision peptide formulations provides additional context for how delivery methods are evolving across the entire class.
Real-world protocols and expected timelines
Theory is one thing. Practice is another. This section provides practical protocols that integrate the compound formulation into a complete treatment plan, along with realistic timelines for what to expect.
Protocol one: standard weight loss (moderate approach)
Goal: 15% to 20% body weight reduction over 6 to 9 months with maximum muscle preservation.
Compound: Tirzepatide/glycine/B12 at 30/5/0.5 mg/mL.
Titration: Standard four-week intervals. Start at 2.5 mg, increase to target dose of 10 to 15 mg over 16 to 20 weeks. Use the dosage chart in units and the tirzepatide dose chart for precise injection volumes.
Nutrition: Protein target of 1.4 g/kg body weight daily. Focus on lean proteins and vegetables. Minimize processed foods and added sugars. Follow the recommended foods list (most principles apply across GLP-1 medications).
Exercise: Resistance training 3 to 4 times per week. Moderate cardio 2 to 3 times per week. Walking 8,000 to 10,000 steps daily.
Supplementation: Creatine 5 g daily, vitamin D 3,000 IU daily, magnesium 300 mg daily, omega-3 3 g daily.
Expected timeline:
Weeks 1 through 4: Initial adaptation. Weight loss of 2 to 5 pounds. Some nausea possible but typically milder than plain tirzepatide. Appetite suppression begins. Glycine and B12 begin systemic effects.
Weeks 5 through 12: Acceleration phase. Weight loss of 1 to 2 pounds per week. Appetite suppression fully established. Energy levels stabilize. Body composition starting to improve visibly.
Weeks 13 through 24: Peak weight loss. At optimal dose, weight loss continues at 1 to 2 pounds per week. Muscle preservation noticeable compared to expected lean mass loss. Skin elasticity maintained by glycine collagen support.
Weeks 25 through 36: Maintenance transition. Weight loss slows as the body approaches a new set point. Focus shifts from weight loss to body composition and maintenance. Some users reduce dose at this phase.
Protocol two: aggressive fat loss with muscle priority
Goal: Maximum fat loss with minimal muscle loss. For users who are already experienced with resistance training and nutrition optimization.
Compound: Tirzepatide/glycine/B12 at 30/5/0.5 mg/mL.
Titration: Standard four-week intervals up to 15 mg. No extended titration delays.
Nutrition: Protein target of 1.6 g/kg body weight daily (upper end). Caloric intake tracked precisely. Carbohydrate timing around workouts. Strategic refeed days every 10 to 14 days.
Exercise: Resistance training 4 to 5 times per week with progressive overload. Minimal cardio (walking only) to preserve muscle. The peptide stacking for weight loss guide discusses how some users combine GLP-1 therapy with other compounds for enhanced results.
Expected timeline:
Months 1 through 2: Initial response. 8 to 12 pounds total loss, predominantly fat. Strength maintained or slightly reduced.
Months 3 through 4: Acceleration. 12 to 20 pounds additional loss. Body composition changes clearly visible. Strength stabilizes. The glycine effect on muscle preservation becomes most apparent during this period.
Months 5 through 6: Continued progress. 10 to 15 additional pounds. Total loss of 30 to 47 pounds with significantly better lean mass preservation than plain tirzepatide protocols. Tirzepatide before and after results at this stage often show dramatic body recomposition beyond what the scale indicates.
Protocol three: metabolic health focus (diabetes or prediabetes)
Goal: A1C reduction and metabolic health improvement with weight loss as a secondary benefit.
Compound: Tirzepatide/glycine/B12 at 30/5/0.5 mg/mL.
Titration: Extended six-week intervals between increases for more gradual adaptation. Target dose of 5 to 10 mg (may not need maximum dose for metabolic goals).
Nutrition: Low glycemic index focus. Protein target of 1.2 g/kg body weight. Fiber intake of 30 to 35 grams daily. Mediterranean-style eating pattern. The glycine in the compound provides additional insulin-sensitizing effects that complement the dietary approach.
Exercise: Mix of resistance training (2 to 3 times per week) and moderate cardio (3 to 4 times per week). Daily walking 7,000+ steps.
Expected timeline:
Months 1 through 3: A1C typically drops 0.5% to 1.5% depending on starting level. Weight loss of 5 to 15 pounds. Fasting glucose improvement often visible within the first 2 to 4 weeks.
Months 4 through 6: Continued A1C improvement. Many users reach target A1C levels. Weight loss continues gradually. Energy levels significantly improved as metabolic health normalizes.
Months 7 through 12: Maintenance and optimization. A1C stabilized at target. Weight at or near goal. Glycine anti-inflammatory effects supporting long-term metabolic health. B12 levels maintained throughout, avoiding the deficiency that can occur with long-term GLP-1 use.
What to do when results plateau
Weight loss plateaus are normal and expected, typically occurring after 3 to 6 months of treatment. Before attributing a plateau to the medication, verify that you are still in a caloric deficit (appetites can adapt), that protein intake remains adequate, that exercise intensity has not decreased, and that the compound is stored properly and not expired.
If the plateau persists despite all variables being optimized, discuss dose adjustment with your provider. Some users benefit from a brief dose increase followed by a return to maintenance dose. Others find that a temporary diet break at maintenance calories for 1 to 2 weeks can reset metabolic adaptations. The troubleshooting guide for tirzepatide plateaus covers every scenario in detail. For users in the early stages who are still waiting for the medication to take effect, the early plateau guide addresses this specific frustration (the principles apply across GLP-1 medications).
Safety, contraindications, and important warnings
Every discussion of the tirzepatide/glycine/B12 compound must include a thorough safety section. The compound formulation is generally well-tolerated, but there are specific contraindications, warnings, and safety considerations that every user must understand.
Absolute contraindications
Do NOT use the tirzepatide/glycine/B12 compound if you have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). Tirzepatide, like all GLP-1 receptor agonists, carries a boxed warning regarding thyroid C-cell tumors. In animal studies, GLP-1 receptor agonists caused thyroid tumors. While human relevance has not been established, anyone with these risk factors must not use the medication.
A history of pancreatitis is a relative contraindication. GLP-1 medications have been associated with acute pancreatitis in rare cases. If you have had pancreatitis previously, discuss the risks carefully with your provider before using any tirzepatide formulation.
Known hypersensitivity to tirzepatide or any component of the formulation is an absolute contraindication. This includes allergies to glycine supplements or B12 in any form (though true B12 allergy is extremely rare).
Drug interactions to be aware of
Tirzepatide slows gastric emptying, which can affect the absorption of oral medications. Medications that require precise timing for absorption, such as oral contraceptives, certain antibiotics, and thyroid medications, may need dose adjustment or timing changes when used alongside tirzepatide.
Diabetes medications, particularly insulin and sulfonylureas, may need dose reduction when combined with tirzepatide to prevent hypoglycemia. The tirzepatide component improves insulin sensitivity and stimulates insulin release, so adding it to existing diabetes medications can create a risk of blood sugar dropping too low.
Alcohol use with tirzepatide is a common question. The alcohol and tirzepatide guide covers this topic in depth. Generally, moderate alcohol consumption is possible but should be approached cautiously, especially during the early titration phase when GI side effects are most common. Alcohol can worsen nausea, dehydration, and blood sugar fluctuations.
The compound is not FDA-approved
This point bears repeating clearly. The tirzepatide/glycine/B12 compound is a pharmacy-compounded product. It is NOT FDA-approved. This means the specific combination has not undergone the rigorous clinical trial process required for FDA approval. The individual ingredients (tirzepatide, glycine, and B12) have extensive research supporting their safety and efficacy, but the combination in a single injection has not been evaluated in the same way.
This does not mean the compound is unsafe. Compounding has been a legitimate and essential part of pharmacy practice for over a century. But it does mean that users should understand what they are using, ensure they obtain it from a reputable compounding pharmacy, and maintain regular follow-up with a healthcare provider who can monitor their response.
Monitoring recommendations
Regular lab work is important for anyone on long-term GLP-1 therapy, and the compound formulation is no exception. Recommended monitoring includes:
Every 3 months: Comprehensive metabolic panel (kidney and liver function), A1C (even for non-diabetic users, to monitor metabolic improvement), and lipid panel (cholesterol, triglycerides).
Every 6 months: Complete blood count, B12 levels (even though the compound provides B12, monitoring confirms adequate levels), thyroid function tests, and vitamin D levels.
Body composition assessment: If available, DEXA scans at baseline and every 3 to 6 months provide the most accurate measurement of fat versus lean mass changes, allowing you to quantify the muscle preservation benefits of the glycine component.
Common mistakes users make with the compound
Even with the best formulation, users make mistakes that undermine their results. Knowing what to avoid is just as valuable as knowing what to do. The common peptide mistakes guide covers general errors, but compound formulations have their own specific pitfalls.
Mistake one: assuming the compound eliminates all side effects
The compound reduces nausea and supports muscle preservation. It does not eliminate side effects entirely. Users who start the compound expecting zero nausea, zero constipation, and zero fatigue are setting themselves up for disappointment. The compound makes the treatment more tolerable, not effortless.
Mistake two: neglecting protein because glycine protects muscle
The glycine in the compound activates mTORC1 signaling, but mTORC1 needs amino acid building blocks to actually synthesize muscle protein. If your total protein intake is inadequate, glycine mTORC1 activation is like an engine running without fuel. You still need 1.2 to 1.6 g/kg of protein daily. The glycine enhances the effect of adequate protein intake. It does not replace it.
Mistake three: skipping resistance training
Similar to the protein mistake, the glycine in the compound works best when combined with resistance training stimulus. Without mechanical loading on muscles, the mTORC1 activation from glycine preserves existing muscle but does not stimulate growth. With resistance training, the combination of glycine mTORC1 activation and exercise-induced mTORC1 activation produces superior results.
Mistake four: improper storage destroying the compound
Multi-ingredient compounds can be more sensitive to temperature excursions than single-ingredient formulations. Leaving your vial out of the refrigerator for extended periods, exposing it to direct sunlight, or using it past the beyond-use date can degrade one or more components without obvious visual changes. The tirzepatide might still work, but the glycine or B12 could be degraded, eliminating the benefits you are paying for.
Mistake five: titrating too fast because the compound is easier to tolerate
Some users, finding that the compound formulation produces less nausea than they expected, decide to titrate faster than the recommended schedule. This is risky. The reduced nausea does not mean the body has fully adapted to the metabolic effects. Too-rapid titration can cause rebound GI symptoms, pancreatitis risk, or gallbladder issues that the nausea signal would normally warn you about. Stick to the recommended titration schedule regardless of how well you feel. The dosing guide exists for safety reasons. Understanding the proper peptide dosing principles and following a cycle planning approach helps avoid this trap.
Mistake six: not planning for the end of treatment
Every GLP-1 protocol should include an exit strategy. What happens when you reach your goal weight? Abruptly stopping tirzepatide can lead to rapid weight regain, particularly if the metabolic and behavioral changes have not been solidified. The GLP-1 withdrawal guide discusses tapering strategies and maintenance planning. The compound formulation does not change the need for a thoughtful discontinuation plan.
The future of tirzepatide compound formulations
The tirzepatide/glycine/B12 compound represents the current state of the art in enhanced GLP-1 formulations. But the field is evolving rapidly, and understanding where things are heading provides context for current decisions.
Next-generation GLP-1 compounds
Research into triple agonists (GLP-1/GIP/glucagon receptor agonists) like retatrutide suggests even more effective weight loss medications are on the horizon. As these new molecules enter clinical practice, compounding pharmacies will likely develop similar enhanced formulations that include supportive ingredients like glycine and B12. The mazdutide comparison, cagrilintide combination guide, and cagrilintide safety profile preview some of these emerging options. Even comparisons with non-peptide alternatives like phentermine versus semaglutide and questions about whether phentermine qualifies as a GLP-1 help map the evolving weight loss medication landscape.
Novel delivery systems
Transdermal patches for GLP-1 delivery are in development. The GLP-1 patch guide covers the current state of this technology. If transdermal delivery becomes practical for tirzepatide, the question of how to incorporate glycine and B12 into a patch format will be an interesting pharmaceutical challenge.
Personalized compounding
The future likely includes more personalized compound formulations where the ratios of glycine and B12 are adjusted based on individual lab results, side effect profiles, and treatment goals. A user with low B12 levels and high nausea might receive a formulation with higher B12 concentration. A user primarily concerned about muscle preservation might receive higher glycine. Personalization is already happening to some degree, and it will become more sophisticated as understanding of these interactions deepens.
For researchers and users who want to stay at the cutting edge of peptide science and formulation optimization, SeekPeptides provides ongoing coverage of new developments, evidence-based protocols, and community insights from thousands of experienced users. Use the peptide stack calculator to explore how different compounds interact, and check peptide expiration guidelines and expected timelines for peptide effects to set realistic expectations for any protocol.
Frequently asked questions
Is the tirzepatide glycine B12 compound the same as Mounjaro or Zepbound?
No. Mounjaro and Zepbound are FDA-approved brand-name tirzepatide products manufactured by Eli Lilly. They contain only tirzepatide. The tirzepatide/glycine/B12 compound is a pharmacy-compounded product that adds glycine and vitamin B12 to the base tirzepatide. It is not FDA-approved as a combination product. The tirzepatide component functions the same way, but the added ingredients provide additional benefits for how the formulation works in your body.
Can I add glycine and B12 separately instead of using the compound?
You can supplement glycine orally and take B12 injections or sublingual tablets separately. However, the nausea-reduction benefit of B12 specifically requires co-formulation with tirzepatide in the same injection. Taking a B12 pill alongside a plain tirzepatide injection does not produce the same pharmacological effect on brain penetration. Oral glycine supplementation does work for muscle preservation, but the compound ensures consistent delivery with every dose.
How do I calculate my dose from the compound vial?
For a 30/5/0.5 mg/mL formulation, divide your target tirzepatide dose by 30 to get the injection volume in mL. For example, 5 mg divided by 30 equals 0.167 mL, which is approximately 17 units on an insulin syringe. The peptide calculator and tirzepatide conversion chart can help with precise calculations.
Does the compound need to be refrigerated?
Yes. Store the compound between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Keep it in the refrigerator when not in use. Brief room temperature exposure during injection preparation is fine, but prolonged heat exposure can degrade the ingredients. The tirzepatide refrigeration guide covers storage specifics.
Will the glycine in the compound prevent all muscle loss?
No. Glycine reduces muscle loss but does not eliminate it entirely. It activates mTORC1 signaling which promotes muscle protein synthesis, and research shows treated muscle cells were up to 20% larger than controls. But optimal muscle preservation also requires adequate protein intake (1.2 to 1.6 g/kg daily) and regular resistance training. The compound is one tool in a comprehensive strategy, not a standalone solution. The muscle preservation guide covers the full approach.
How much does the compound cost compared to plain tirzepatide?
The tirzepatide/glycine/B12 compound typically costs 20% to 40% more than plain compounded tirzepatide. The exact price varies by pharmacy, concentration, and location. Use the peptide cost calculator to estimate costs, and review the peptide pricing guide for broader market context, and the semaglutide versus tirzepatide dosage chart for comparison pricing between the two main GLP-1 platforms.
Can I use the compound if I am not trying to lose weight?
Tirzepatide is FDA-approved for type 2 diabetes (as Mounjaro) where the primary goal is blood sugar control rather than weight loss. The glycine and B12 additions provide benefits (anti-inflammatory effects, metabolic support, B12 maintenance) that are relevant regardless of whether weight loss is the primary goal. Discuss your specific situation with your healthcare provider. The semaglutide timeline guide provides comparable data for those weighing their GLP-1 options for metabolic health rather than weight loss.
What if I experience side effects that the compound is supposed to prevent?
The compound reduces certain side effects but does not guarantee elimination. If you experience significant nausea despite the B12 component, muscle loss despite the glycine, or other concerning symptoms, discuss these with your provider. Dose adjustment, timing changes, or additional supportive measures may be needed. The peptide safety guide covers when to seek help.
Is the tirzepatide glycine B12 compound safe for long-term use?
The individual components, tirzepatide, glycine, and B12, all have extensive safety data supporting long-term use. Tirzepatide has been studied in trials lasting up to 72 weeks. Glycine is a naturally occurring amino acid with an excellent safety profile. B12 is a water-soluble vitamin with no known toxicity at supplemental doses. However, the specific combination has not been studied in long-term clinical trials as a single product, so ongoing monitoring with your healthcare provider is important. Planning for the long term, including potential discontinuation, is covered in the peptide cycle planning guide. The mixing water guide and tirzepatide speed of action resource also provide helpful context for establishing safe, effective protocols.
External resources
For researchers committed to getting the most from their tirzepatide protocol, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, protocol databases, expert-reviewed dosing tools, and a community of thousands who navigate these exact decisions every day. Whether you are choosing between formulations, optimizing your compound protocol, or troubleshooting side effects, the SeekPeptides membership provides the guidance that makes the difference between adequate results and exceptional ones.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay potent, your muscle stay preserved, and your protocols stay optimized.