Feb 12, 2026

The baby weight will not budge. You have tried calorie counting, walking with the stroller at dawn, and every postpartum diet plan your mother-in-law forwarded. Meanwhile, your social media feed keeps showing GLP-1 transformations. Semaglutide. Tirzepatide. Retatrutide. The results look real. The science looks solid. But you are breastfeeding, and that one detail changes everything.
Here is the tension that millions of new mothers face right now. GLP-1 receptor agonists represent the most effective weight loss peptides developed in decades, with clinical trials showing 15 to 22 percent body weight reduction in some participants. These are not fringe compounds. They are FDA-approved medications reshaping how medicine approaches obesity. And for postpartum women carrying 20, 30, or 50 extra pounds, the appeal is obvious.
But the question is not whether GLP-1 medications work. They do. The question is whether they are safe for a nursing infant whose entire nutritional world flows through your body.
That question has a complicated answer. Not because the data is alarming, but because the data is thin. Only a handful of human studies exist. The FDA labels are cautious by design. And the stakes, a developing infant, are as high as they get.
This guide breaks down every piece of evidence available. We will cover what the actual studies found when researchers measured semaglutide and tirzepatide in breast milk, what the FDA prescribing labels say, how molecular weight affects milk transfer, what risks remain theoretical versus proven, and what alternatives exist for mothers who want to lose weight without risking their milk supply. No hype. No shortcuts. Just the research.
How GLP-1 receptor agonists work in the body
Before evaluating safety during lactation, you need to understand what these molecules actually do. GLP-1 receptor agonists mimic a natural hormone called glucagon-like peptide-1. Your gut produces this hormone after eating. It signals your brain that food arrived, slows stomach emptying, and triggers insulin release from the pancreas. The result is reduced appetite, better blood sugar regulation, and a cascade of metabolic effects that promote weight loss.
Simple concept. Powerful execution.
Semaglutide, the active ingredient in Ozempic and Wegovy, is a modified version of human GLP-1. Researchers altered the amino acid sequence and attached a fatty acid chain so the molecule survives longer in circulation. Instead of the natural hormone lasting minutes, semaglutide persists for roughly a week. That is why it works as a once-weekly injection.
Tirzepatide, found in Mounjaro and Zepbound, goes further. It activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors simultaneously. This dual mechanism produces even stronger appetite suppression and weight loss results in clinical trials. The SURMOUNT trials demonstrated up to 22.5 percent body weight reduction with tirzepatide at the highest doses.
Both compounds share a critical characteristic relevant to breastfeeding. They are large protein-based molecules. Semaglutide has a molecular weight of approximately 4,114 Daltons. Tirzepatide weighs approximately 4,813 Daltons. This matters because molecular size directly affects whether a drug can cross from blood into breast milk.
Most drugs that easily transfer into milk weigh less than 500 Daltons. At over 4,000 Daltons, both semaglutide and tirzepatide face significant biological barriers to milk transfer. This does not guarantee safety, but it provides the theoretical foundation for why researchers suspected these medications might be compatible with breastfeeding long before human studies confirmed it.
Understanding this mechanism is essential. It separates GLP-1 receptor agonists from smaller molecules like medications that readily cross into milk and create clear risks for nursing infants.
What the FDA prescribing labels actually say
Let us start with what the regulatory authorities have on record. The FDA labels for both semaglutide and tirzepatide address lactation, but the language reveals more about the state of research than the state of risk.
Semaglutide (Ozempic and Wegovy)
The FDA prescribing information states plainly that there are no data on the presence of semaglutide in human milk, the effects on the breastfed infant, or the effects on milk production. It notes that semaglutide was present in the milk of lactating rats, but adds that due to species-specific differences in lactation physiology, the clinical relevance of these data is not clear.
In practical terms, the label says we do not know. Not that it is dangerous. Not that it is safe. That the data did not exist when the label was written.
The label also recommends that the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for the medication and any potential adverse effects on the breastfed infant.
Tirzepatide (Mounjaro and Zepbound)
The tirzepatide label uses nearly identical language. No data on presence in human or animal milk. No data on effects on breastfed infants. No data on effects on milk production. The same recommendation to weigh breastfeeding benefits against clinical need.
One additional detail. The tirzepatide label for Zepbound specifically notes that weight loss offers no benefit to a pregnant patient and may cause fetal harm. Women should discontinue the medication when pregnancy is recognized. For lactation, the language is less restrictive but still cautious.
Here is what matters. These labels were written before the human lactation studies were published. The FDA label approval process does not update in real time as new research emerges. The most relevant data, the actual measurements of these drugs in breast milk, came after the labels were finalized.
That does not mean you should ignore the labels. They represent the formal regulatory position. But understanding that the labels reflect an absence of data rather than the presence of danger is important context for any mother evaluating her options with her healthcare provider.
The semaglutide breast milk study: what researchers actually found
The most important piece of evidence in this entire conversation is a study published in the journal Nutrients in September of a recent year. Researchers measured semaglutide concentrations in breast milk from eight nursing mothers who were taking subcutaneous semaglutide injections weekly.
Study design and participants
Eight lactating women participated. They were taking subcutaneous semaglutide at doses ranging from 0.25 mg to 1 mg per week, which covers the standard semaglutide dosage range used clinically. Their infants ranged from 4 to 23 months old. The mothers had been breastfeeding while taking semaglutide for 3 to 9 weeks at the time of sample collection.
Researchers collected milk samples at three time points: immediately before the weekly dose (time zero), 12 hours after the dose, and 24 hours after the dose. These windows capture the peak absorption period when milk concentrations would theoretically be highest.
Results
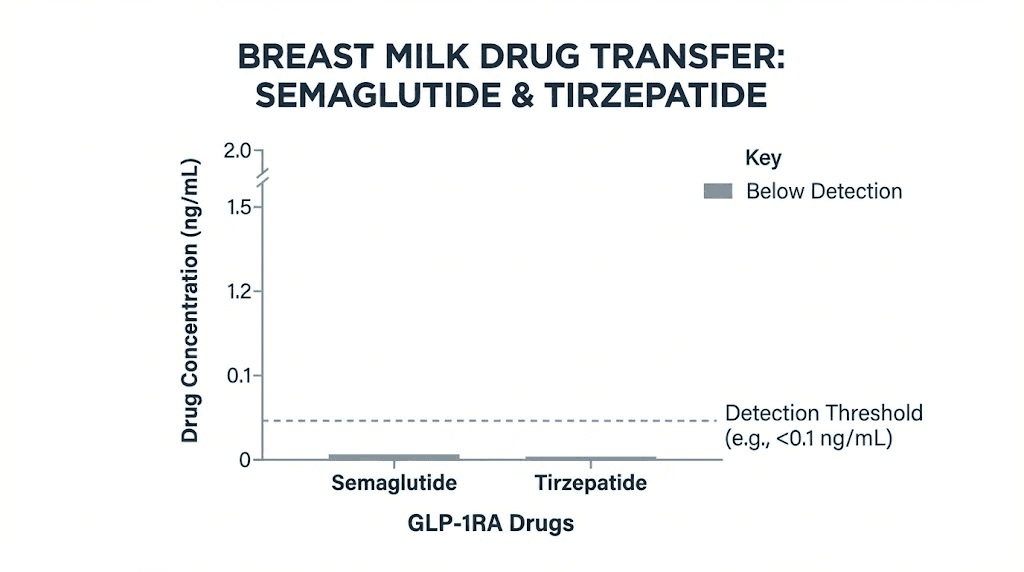
None of the samples contained any measurable semaglutide. The detection limit was 1.7 micrograms per liter, and every single sample came back below that threshold. Zero detectable semaglutide in breast milk across all eight mothers at all three time points.
That is worth repeating. Not low levels. Not trace amounts. Undetectable.
The researchers then calculated what the relative infant dose would be if semaglutide were present at the detection limit, the worst-case scenario. That number came to approximately 1.12 percent of the maternal weight-adjusted dose. For context, the standard threshold for considering a medication compatible with breastfeeding is less than 10 percent. Semaglutide falls at roughly one-ninth of that threshold in the absolute worst case.
And there is another layer of safety. Even if tiny amounts of semaglutide did reach the infant through milk, the oral bioavailability of semaglutide in adults maxes out at approximately 1 percent. That means 99 percent of any orally ingested semaglutide gets destroyed in the digestive tract before reaching the bloodstream. An infant's digestive system would further reduce any absorption.
Infant outcomes
All eight infants were reported to have normal growth and development throughout the study period. No adverse effects were documented. No changes in feeding behavior, weight gain, or developmental milestones.
This is a small study. Eight mothers. That is the honest limitation. But what it found was not ambiguous. The drug simply was not getting into the milk in measurable quantities.
What this means for nursing mothers considering semaglutide
The study provides the first direct human evidence that subcutaneous semaglutide does not appear to transfer into breast milk at clinically meaningful levels. Combined with the molecular weight argument (semaglutide is too large to easily cross biological barriers into milk) and the poor oral bioavailability (any swallowed semaglutide would be destroyed in the infant gut), the safety profile looks reassuring.
But reassuring is not the same as proven safe. The study was small. It measured a single formulation (subcutaneous injection). And it did not examine long-term outcomes for the infants. These limitations matter, and they are why the formal recommendation remains cautious.
Mothers interested in starting semaglutide while nursing should discuss these specific findings with their physician. The conversation is different now than it was before this data existed.
The tirzepatide breast milk study: newer data, similar findings
Tirzepatide data emerged more recently, and the findings mirror what researchers found with semaglutide.
The manufacturer study
Eli Lilly conducted a study involving 11 lactating women who received a single 5 mg subcutaneous dose of tirzepatide. Researchers collected 171 milk samples over 28 days and measured tirzepatide concentrations in each one.
The results. Tirzepatide was undetectable (below 4 micrograms per liter) in 164 of 171 samples. That is a 95.9 percent undetectable rate. In the seven samples where tirzepatide was above the detection limit, the cumulative amount was less than 0.02 percent of the maternal dose. The last measurable concentrations appeared at 5 days post-dose, after which the drug disappeared entirely from milk.
Less than 0.02 percent. Even when it was detectable, the amount was negligible.
The ongoing real-world study
A separate study followed five women receiving weekly tirzepatide doses ranging from 0.5 to 5 mg. Milk samples showed a few readings below the limit of detection (0.7 micrograms per liter), and no samples exceeded the limit of quantification (2.38 micrograms per liter). No adverse effects were reported in the breastfed infants.
Why tirzepatide may be even less likely to enter milk
Tirzepatide has a molecular weight of approximately 4,813 Daltons, roughly 700 Daltons heavier than semaglutide. Larger molecules face greater difficulty crossing biological membranes. Combined with the study data showing near-complete absence in milk, the evidence suggests tirzepatide has a similarly favorable profile during lactation.
The LactMed database, maintained by the National Library of Medicine, now states that tirzepatide is usually undetectable in breastmilk with subcutaneous doses of up to 5 mg and that absorption by the infant is unlikely because it is probably partially destroyed in the infant's gastrointestinal tract and poorly absorbed orally. It further notes that if a mother requires tirzepatide, it is not a reason to discontinue breastfeeding.
That last statement is significant. LactMed is one of the most conservative, evidence-based resources for drug safety during lactation. When it says a medication is not a reason to stop breastfeeding, physicians pay attention.
For mothers who have been researching tirzepatide results, this data provides important context for informed conversations with their healthcare providers.
The critical distinction: injectable versus oral GLP-1 formulations
This is the section that could prevent a serious mistake. Not all GLP-1 formulations carry the same safety profile during breastfeeding. The difference between injectable and oral forms is not subtle. It is potentially dangerous to ignore.
Why injectable forms are considered safer
Injectable semaglutide (Ozempic, Wegovy) and injectable tirzepatide (Mounjaro, Zepbound) are administered subcutaneously. The drug enters the bloodstream directly, bypasses the digestive tract, and circulates at therapeutic levels. When researchers measured milk levels, they used mothers taking injectable forms. The reassuring data applies specifically to injections.
Why oral semaglutide is different
Oral semaglutide (Rybelsus) presents a unique problem. The pill contains semaglutide plus an absorption enhancer called salcaprozate sodium (SNAC). This chemical is necessary because without it, the digestive system would destroy semaglutide before it could reach the bloodstream. SNAC temporarily opens tight junctions in the stomach lining, allowing the peptide to pass through.
Here is the concern. Salcaprozate sodium may transfer into breast milk. Unlike the large semaglutide molecule, SNAC is a smaller chemical that could potentially cross into milk more easily. If it accumulates in breast milk, it could theoretically affect the infant's gut barrier in ways that have not been studied.
The LactMed database explicitly warns that some brands of oral semaglutide contain the absorption enhancer salcaprozate sodium, which may enter milk and accumulate in infants. This warning does not apply to injectable forms.
The bottom line on formulations
If you are breastfeeding and your physician approves GLP-1 therapy, only injectable formulations should be considered. Oral versus injectable is not a preference issue during lactation. It is a safety issue.
This distinction extends to compounded formulations as well. Sublingual tirzepatide drops and other oral preparations may contain absorption enhancers similar to SNAC. Without specific safety data on these formulations during breastfeeding, they should be avoided.
How molecular weight determines breast milk transfer
Understanding why some drugs pass into milk and others do not requires a basic understanding of molecular weight and biological barriers. This is not abstract chemistry. It directly explains why GLP-1 receptor agonists appear safer during lactation than many smaller medications.
The size threshold
Breast milk is produced in the mammary glands through a process that involves filtering substances from maternal blood. Small molecules pass relatively easily through the epithelial cells lining the milk ducts. Larger molecules face increasing resistance.
The general threshold used in lactation pharmacology is approximately 500 Daltons. Molecules below this weight tend to transfer into milk more readily. Molecules above 800 to 1,000 Daltons transfer poorly. At 4,114 Daltons (semaglutide) and 4,813 Daltons (tirzepatide), GLP-1 agonists are 8 to 10 times larger than the typical threshold for easy milk transfer.
Protein degradation in the infant gut
Even if trace amounts of a GLP-1 agonist did enter breast milk, the infant would need to absorb it orally for any systemic effect. Protein-based molecules are broken down by proteases in the stomach and small intestine. This is why insulin, another peptide hormone, cannot be taken as a pill. The digestive system destroys it.
The same principle applies to semaglutide and tirzepatide in breast milk. Any amount ingested by a nursing infant would face enzymatic degradation in the gut. The oral bioavailability of semaglutide in adults is approximately 1 percent even with the absorption enhancer SNAC. Without SNAC, it is essentially zero. An infant ingesting trace amounts through milk would absorb virtually nothing.
This creates a double safety barrier. First, the drug barely gets into milk. Second, any drug in milk barely gets into the infant's bloodstream. The actual systemic exposure to the infant approaches zero through two independent mechanisms.
Comparison with other medications
For perspective, acetaminophen (Tylenol) has a molecular weight of 151 Daltons and transfers into breast milk at a relative infant dose of 6 to 12 percent. Ibuprofen weighs 206 Daltons and transfers at less than 1 percent. Semaglutide, at 4,114 Daltons and a worst-case relative infant dose of 1.12 percent (which in practice was undetectable), is comparable to or better than many medications routinely considered safe during breastfeeding.
This molecular weight context helps explain why the lactation studies found what they found. The biology predicted the outcome before the studies confirmed it.
Risks that remain real even if the drug does not transfer
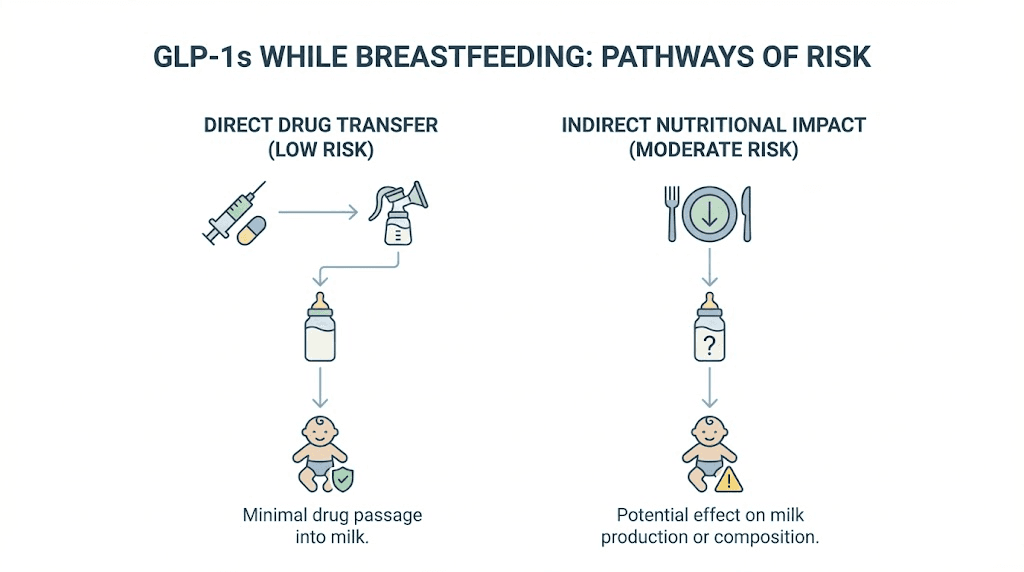
Drug transfer into milk is only one piece of the puzzle. Even if semaglutide and tirzepatide do not meaningfully enter breast milk, GLP-1 therapy during breastfeeding carries indirect risks that deserve serious attention.
Reduced caloric intake and milk supply
GLP-1 receptor agonists work by suppressing appetite. That is their primary mechanism for weight loss. But breastfeeding demands significant caloric expenditure. Lactating women need approximately 450 to 500 extra calories daily to maintain adequate milk production.
If a GLP-1 medication causes significant appetite reduction, a nursing mother may eat far less than her body requires to sustain milk production. The consequences are predictable. Reduced milk volume. Altered milk composition. Nutritional deficits for the infant.
This is not a theoretical concern. Nausea and fatigue are common side effects of GLP-1 therapy, especially during the dose titration phase. A mother experiencing persistent nausea may struggle to eat enough calories regardless of her intentions. Dietary restrictions combined with appetite suppression could push caloric intake dangerously low.
Some researchers argue this indirect risk may be more clinically relevant than the drug itself entering milk. A well-nourished mother taking semaglutide may pose minimal risk to her infant. A malnourished mother on any weight loss medication, GLP-1 or otherwise, poses significant risk.
Rapid weight loss and milk quality
When the body loses weight rapidly, it mobilizes stored fat. Fat tissue stores environmental toxins, heavy metals, and lipophilic chemicals accumulated over years. Rapid fat mobilization releases these stored substances into the bloodstream, and some can transfer into breast milk.
This concern applies to any rapid weight loss method during breastfeeding, not just GLP-1 medications. But because GLP-1 agonists can produce substantial weight loss (sometimes 1 to 2 pounds per week or more), the rate of fat mobilization could be significant enough to affect milk quality.
Most lactation experts recommend that breastfeeding mothers limit weight loss to approximately 1 to 1.5 pounds per week. Aggressive GLP-1 dosing that produces faster weight loss may need to be moderated during the breastfeeding period.
Nutrient depletion
Breastfeeding already draws heavily on maternal nutrient stores. Calcium, iron, vitamin D, omega-3 fatty acids, and B vitamins all flow from mother to infant through milk. Reduced food intake from GLP-1 therapy can compound nutrient depletion that breastfeeding naturally causes.
Mothers considering GLP-1 medications while breastfeeding should work with their physician and a nutritionist to ensure adequate supplementation. B12 supplementation is particularly relevant, as some compounded semaglutide formulations already include B12 for this reason.
Dehydration
GLP-1 medications can cause gastrointestinal side effects including nausea, vomiting, and diarrhea, especially in the early weeks. These side effects increase fluid loss. Breastfeeding also requires significant fluid intake, approximately 128 ounces (one gallon) daily for many lactating women.
Dehydration directly reduces milk production. A mother experiencing GLP-1 side effects while breastfeeding faces compounded dehydration risk that could significantly impact her milk supply.
Semaglutide while breastfeeding: a detailed safety breakdown
Let us examine semaglutide specifically, since it is the most widely prescribed GLP-1 receptor agonist and the one with the most lactation data.
What we know
The human study of eight breastfeeding mothers found undetectable levels in all milk samples. The maximum theoretical relative infant dose is 1.12 percent, well below the 10 percent safety threshold. The molecular weight of 4,114 Daltons makes milk transfer unlikely. Oral bioavailability without an absorption enhancer is negligible, meaning any trace amounts in milk would not be absorbed by the infant.
All eight infants in the study showed normal growth and development with no adverse effects during 3 to 9 weeks of maternal semaglutide use.
What we do not know
No studies have examined semaglutide exposure through breast milk for more than 9 weeks. No studies have measured infant blood levels to confirm zero systemic exposure. No studies have examined effects on infant metabolism, pancreatic development, or appetite regulation long-term. No studies have specifically evaluated higher semaglutide doses (above 1 mg weekly) during breastfeeding. And no studies have examined the effect of compounded semaglutide formulations, which may contain different excipients.
The formulation warning
Oral semaglutide (Rybelsus) should not be used during breastfeeding. The absorption enhancer salcaprozate sodium poses theoretical risks to the nursing infant. Only injectable subcutaneous semaglutide should be considered.
The dose consideration
Most semaglutide dosing protocols start at 0.25 mg weekly and titrate up to 2.4 mg weekly for weight loss. The human lactation study only evaluated doses up to 1 mg. Higher doses have not been specifically studied in breastfeeding women.
This does not mean higher doses are dangerous. The molecular weight barrier and oral bioavailability limitation apply regardless of dose. But the absence of data at higher doses is a legitimate gap that physicians should consider when prescribing to lactating women.
Practical guidance for mothers and physicians
Based on current evidence, a reasonable approach for a lactating woman who requires semaglutide might include starting at the lowest effective dose. Using only the injectable subcutaneous formulation. Monitoring caloric intake to ensure adequate nutrition for milk production. Watching for gastrointestinal side effects that could affect hydration. Tracking infant weight gain and development closely. And maintaining regular communication with both the prescribing physician and the pediatrician.
This is not a DIY decision. The conversation between mother and doctor should include these specific study findings, the formulation distinction, and a plan for monitoring both maternal nutrition and infant development.
Tirzepatide while breastfeeding: what the newer data shows
Tirzepatide has less published data than semaglutide but the existing evidence points in the same direction.
Current evidence summary
In 11 lactating women given 5 mg subcutaneous tirzepatide, 95.9 percent of milk samples showed undetectable levels. The cumulative amount in the seven detectable samples was less than 0.02 percent of the maternal dose over 28 days. Five women on weekly doses up to 5 mg showed no quantifiable tirzepatide in milk.
No adverse effects were reported in any breastfed infants across both studies. The LactMed database states that tirzepatide is not a reason to discontinue breastfeeding.
Why tirzepatide data matters separately
Even though semaglutide and tirzepatide are both GLP-1 receptor agonists, they are different molecules. Tirzepatide also activates GIP receptors. It has a higher molecular weight (4,813 versus 4,114 Daltons). It has different pharmacokinetics. These differences mean that semaglutide safety data cannot be automatically applied to tirzepatide and vice versa.
Having separate lactation studies for each drug strengthens the overall confidence in GLP-1 agonist safety during breastfeeding. Both drugs, through independent studies, showed the same pattern: essentially no transfer into breast milk.
Dosing considerations for tirzepatide
The tirzepatide dosing schedule typically starts at 2.5 mg weekly and can increase to 15 mg weekly. The lactation data covers doses up to 5 mg. Like semaglutide, higher therapeutic doses have not been specifically studied in nursing mothers.
For breastfeeding women who require tirzepatide, starting at 2.5 mg and titrating slowly while monitoring milk supply and infant development represents the most conservative approach. The dosing protocol can be adjusted based on individual response and tolerance.
The dual-agonist question
One theoretical question that researchers have not fully addressed is whether tirzepatide's GIP activation creates any additional considerations during breastfeeding. GIP receptors exist throughout the body, including in the developing digestive system. Whether trace exposure through breast milk could affect infant GIP signaling is unknown but considered unlikely given the undetectable milk levels.
Mothers who are comparing different GLP-1 medications should weigh this uncertainty as part of their decision, though the practical significance is likely minimal based on current understanding.
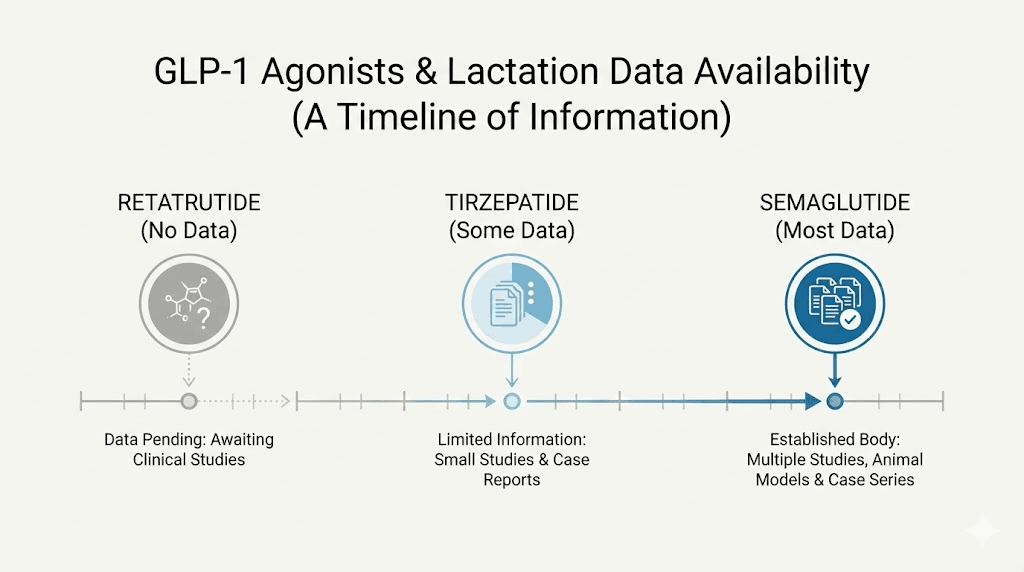
Retatrutide and other newer GLP-1 agonists: no lactation data yet
The GLP-1 landscape is expanding rapidly. Retatrutide, a triple agonist that activates GLP-1, GIP, and glucagon receptors simultaneously, is in advanced clinical trials. Other compounds like mazdutide and survodutide are progressing through development pipelines.
None of these newer agents have any lactation data. Zero human studies. Zero animal lactation studies (publicly available). Extrapolating from semaglutide and tirzepatide data is reasonable given shared molecular characteristics (large protein-based structures, poor oral bioavailability), but it is not the same as having direct evidence.
Breastfeeding mothers interested in newer GLP-1 agonists should wait for specific safety data before considering them. The availability of retatrutide and other next-generation compounds will likely expand in coming years, and lactation studies will hopefully follow.
For now, semaglutide and tirzepatide remain the only GLP-1 receptor agonists with any human breast milk data.
Animal studies: what they showed and why they matter less than you think
The FDA labels reference animal lactation data, and some physicians cite animal studies when advising against GLP-1 use during breastfeeding. These studies deserve context.
What the animal studies found
In lactating rats, semaglutide was detected in breast milk. The FDA label notes this finding but immediately qualifies it by stating that due to species-specific differences in lactation physiology, the clinical relevance is unclear.
A systematic review published in Frontiers in Endocrinology examined GLP-1 agonist and SGLT2 inhibitor use during pregnancy and lactation in animal models. GLP-1 agonists in these studies were found to be excreted in breast milk and affected growth in offspring.
Why animal data does not directly apply
Rat mammary gland physiology differs significantly from human mammary gland physiology. The mechanisms that filter maternal blood into milk, the composition of the milk itself, and the maturity of the neonatal rat gut all differ from their human counterparts.
More importantly, the human studies that now exist directly contradict the animal findings. Semaglutide was detectable in rat milk but undetectable in human milk. This discrepancy illustrates exactly why the FDA label warns against over-interpreting animal data.
Animal studies serve an important role in identifying potential concerns that warrant human investigation. That investigation has now occurred (at least in preliminary form), and the human results are more reassuring than the animal data suggested.
The practical takeaway
If your physician cites animal studies as the primary reason to avoid GLP-1 therapy while breastfeeding, ask about the human lactation data that has since been published. The conversation should be informed by both the animal data (which raised the question) and the human data (which provided a more relevant answer).
The caloric math: breastfeeding, weight loss, and nutrition
Forget the drug transfer question for a moment. The most practical concern for any breastfeeding mother considering GLP-1 therapy is whether she can maintain adequate nutrition while experiencing appetite suppression.
What breastfeeding demands
Exclusive breastfeeding requires approximately 450 to 500 additional calories per day above a non-lactating woman's maintenance needs. For a woman who maintains weight at 2,000 calories, breastfeeding demands 2,450 to 2,500 calories daily. For a woman who maintains at 2,200, the target is 2,650 to 2,700.
These are not aspirational numbers. They are biological requirements. Dip significantly below them, and milk production drops. The body prioritizes survival over lactation.
What GLP-1 therapy does to appetite
GLP-1 receptor agonists can reduce daily caloric intake by 20 to 40 percent in some patients. A woman who normally eats 2,500 calories might find herself comfortably eating only 1,500 to 2,000 calories on semaglutide or tirzepatide. That might be an appropriate deficit for a non-breastfeeding woman pursuing weight loss. For a lactating woman, it could mean a 500 to 1,000 calorie shortfall relative to what her body needs.
The math does not work unless managed carefully.
How to manage the balance
Mothers who use GLP-1 therapy during breastfeeding (with physician approval) need a nutrition strategy that accounts for both the appetite-suppressing effects of the medication and the caloric demands of lactation. This might include eating on a schedule rather than waiting for hunger cues, since GLP-1 therapy specifically blunts those cues. Prioritizing calorie-dense, nutrient-rich foods at every meal. Supplementing with a comprehensive prenatal or postnatal vitamin that includes B12. Monitoring milk output for signs of decreased production. Weighing the infant regularly to confirm adequate growth.
A structured diet plan becomes essential rather than optional. Random eating will not cut it when appetite suppression meets lactation demands.
Protein requirements
Breastfeeding women need approximately 71 grams of protein daily, which is significantly higher than the 46 grams recommended for non-pregnant, non-lactating women. GLP-1 therapy can make eating adequate protein challenging because the feeling of fullness arrives faster and lasts longer.
Protein-rich foods like eggs, Greek yogurt, lean meats, and legumes should be consumed first at each meal, before carbohydrates and fats fill the limited appetite. Some mothers find that protein shakes help them meet targets on days when solid food feels overwhelming due to GLP-1 side effects.
A tirzepatide-specific diet plan designed for breastfeeding would emphasize higher caloric targets than standard GLP-1 diet plans, with protein prioritization and micronutrient supplementation built in.
What about compounded GLP-1 peptides and breastfeeding?
The lactation safety data applies specifically to FDA-approved, pharmaceutical-grade semaglutide and tirzepatide. Compounded versions introduce additional variables.
The compounding difference
Compounded tirzepatide and compounded semaglutide are prepared by compounding pharmacies and may contain different excipients (inactive ingredients), different concentrations, and different purity profiles than FDA-approved formulations. The lactation studies used pharmaceutical-grade products. Whether compounded versions behave identically in terms of milk transfer has not been studied.
This does not mean compounded formulations are dangerous during breastfeeding. The active ingredient is the same molecule. But excipients matter, and without specific testing, an additional layer of uncertainty exists.
Reconstitution and storage considerations
Compounded GLP-1 peptides require proper reconstitution and storage, and errors in either process could affect the drug's molecular integrity. For breastfeeding mothers, this means the already small theoretical risk could be compounded (no pun intended) by degradation products that have not been characterized.
If you are using reconstituted tirzepatide and breastfeeding, ensuring proper storage in the fridge and temperature management becomes even more important. Degraded peptides could theoretically produce smaller fragments with different milk transfer properties.
The conservative approach
For breastfeeding mothers who have physician approval for GLP-1 therapy, FDA-approved formulations provide the highest level of confidence. If compounded formulations are used due to cost or availability, sourcing from a reputable pharmacy and following strict storage protocols is essential.
Alternatives to GLP-1 therapy for postpartum weight loss while breastfeeding
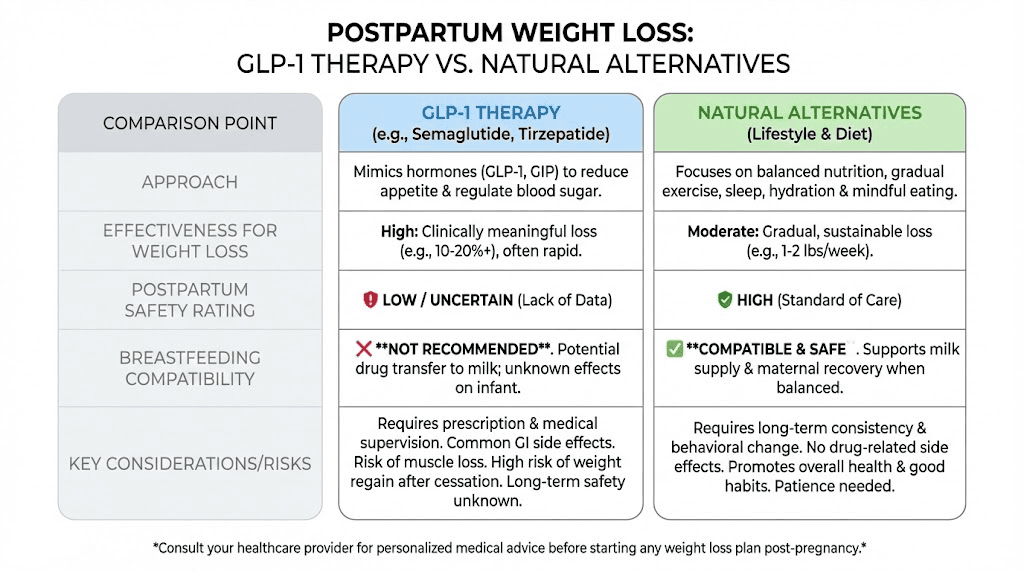
For mothers who decide that GLP-1 therapy is not right during breastfeeding, or whose physicians recommend waiting until weaning, effective alternatives exist. None produce the dramatic results of semaglutide or tirzepatide, but several are considered safe during lactation and can produce meaningful weight loss.
Breastfeeding itself
This sounds reductive, but it is evidence-based. Exclusive breastfeeding burns 450 to 500 extra calories daily. Over six months, that adds up to approximately 82,000 extra calories burned, equivalent to roughly 23 pounds of fat at 3,500 calories per pound. Research consistently shows that longer breastfeeding duration is associated with reduced postpartum weight retention and lower rates of type 2 diabetes.
The catch is that breastfeeding can increase appetite to compensate for the caloric expenditure. Without mindful eating, the weight loss benefit of breastfeeding may be offset by increased food intake.
Metformin
Metformin is one of the few medications considered generally safe during breastfeeding that also supports weight management. It improves insulin sensitivity, modestly reduces appetite, and produces a small amount of weight loss (typically 2 to 5 percent of body weight). The LactMed database reports minimal transfer into breast milk, and it has decades of safety data in lactating women.
For postpartum women with insulin resistance, PCOS, or gestational diabetes that persists after delivery, metformin may serve double duty by managing blood sugar and supporting weight loss.
Structured nutrition and exercise
The least exciting answer is often the most appropriate. A structured nutrition plan designed for breastfeeding, one that creates a modest 300 to 500 calorie daily deficit below maintenance plus lactation needs, can produce safe weight loss of approximately 0.5 to 1 pound per week.
This approach preserves milk production, provides adequate nutrients for the infant, and avoids the risks associated with any medication. It requires patience. Postpartum weight loss through nutrition and exercise alone typically takes 6 to 12 months to reach pre-pregnancy weight.
Resistance training, when cleared by a physician (typically 6 to 8 weeks postpartum), helps preserve lean muscle mass during weight loss and supports metabolic health. Walking is safe almost immediately after delivery and remains one of the most effective, lowest-risk forms of exercise for new mothers.
Behavioral approaches
Cognitive behavioral therapy, mindful eating programs, and structured support groups can address the psychological components of postpartum weight retention. Emotional eating, sleep deprivation (which disrupts hunger hormones), and the stress of new parenthood all contribute to weight gain that medications alone do not solve.
These approaches can be used alongside any other strategy, including GLP-1 therapy after weaning, and often improve long-term outcomes by addressing root causes rather than just symptoms.
The timeline: when can you start GLP-1 therapy after breastfeeding?
For mothers who decide to wait until weaning, the next question is timing. When is it safe to start semaglutide or tirzepatide after stopping breastfeeding?
After full weaning
Once breastfeeding has completely stopped and milk production has fully ceased, there is no waiting period required before starting GLP-1 therapy. The medication cannot enter breast milk if there is no breast milk. Mothers can begin treatment as soon as their physician confirms they are appropriate candidates based on BMI and health status.
During partial weaning
Many mothers wean gradually, reducing feeding sessions over weeks or months. During this transition, the breast milk safety question still applies. The lactation studies suggest minimal risk from injectable semaglutide and tirzepatide, but the safest approach during partial weaning depends on how much the infant is still relying on breast milk.
A baby who nurses once daily and eats solid foods for the other meals has minimal breast milk exposure. A baby who still gets 80 percent of nutrition from breast milk has significant exposure. The risk calculus changes accordingly.
The two-month pre-pregnancy rule
One important detail for women who plan to conceive again. The FDA recommends discontinuing semaglutide at least two months before a planned pregnancy due to the drug's long half-life. Semaglutide remains in the body for several weeks after the last dose, and animal studies showed potential fetal risks.
If you are breastfeeding your current child while planning another pregnancy, this creates an additional timeline consideration. Starting GLP-1 therapy only to stop it two months before trying to conceive may not provide a long enough treatment window to achieve meaningful results.
Realistic expectations after weaning
Mothers who begin GLP-1 therapy after weaning can expect the standard weight loss trajectory. With semaglutide, most patients notice appetite suppression within the first few weeks, with measurable weight loss beginning by weeks 4 to 8. Tirzepatide tends to work slightly faster in some patients, with noticeable effects within 2 to 4 weeks.
The full dose titration period takes 16 to 20 weeks for semaglutide and 20 weeks for tirzepatide, meaning maximum weight loss velocity typically begins 4 to 5 months after starting treatment. Mothers planning their postpartum GLP-1 strategy should factor in this ramp-up period.
The decision framework: a structured approach for breastfeeding mothers
Given the complexity of this decision, a structured approach can help mothers and their physicians evaluate whether GLP-1 therapy during breastfeeding is appropriate for their specific situation.
Step 1: Assess clinical need
How urgent is the weight loss? Postpartum weight retention that resolves gradually with lifestyle changes may not warrant medication. But for women with a BMI over 30, weight-related comorbidities (hypertension, type 2 diabetes, sleep apnea), or psychological impacts of significant weight gain, the clinical need may be substantial.
Step 2: Evaluate the infant's age and feeding status
An older infant (over 6 months) who receives significant nutrition from solid foods faces less breast milk exposure than a newborn who depends entirely on breast milk. The theoretical risk decreases as the infant's diet diversifies.
For mothers of newborns or premature infants, the LactMed database specifically recommends extra caution with tirzepatide pending additional data.
Step 3: Choose the formulation
Only injectable subcutaneous formulations should be considered. No oral semaglutide. No sublingual drops. No compounded oral preparations. This is non-negotiable based on current evidence.
Step 4: Start low and monitor
Begin with the lowest available dose. For semaglutide, that means 0.25 mg weekly. For tirzepatide, 2.5 mg weekly. Titrate more slowly than the standard schedule if needed, prioritizing milk supply maintenance over aggressive weight loss.
Step 5: Monitor aggressively
Track caloric intake daily. Weigh the infant weekly. Watch for changes in milk volume or the infant's feeding behavior. Report any infant symptoms, no matter how mild, to the pediatrician immediately.
Step 6: Set clear stop criteria
Establish specific triggers for discontinuing GLP-1 therapy. These might include any decline in infant weight gain percentile. A significant drop in milk production. Inability to consume adequate calories despite dietary strategies. Any adverse symptoms in the infant. Or the mother's own clinical judgment that the risks outweigh the benefits.
Having these criteria defined before starting treatment removes the pressure of making judgment calls in the moment.
Common questions nursing mothers ask about GLP-1 medications
The questions below reflect what real mothers searching this topic want to know. Each answer draws on the evidence reviewed throughout this guide.
Does semaglutide pass into breast milk?
Based on the only human study available, subcutaneous semaglutide was undetectable in breast milk from eight nursing mothers at doses up to 1 mg weekly. The large molecular weight (4,114 Daltons) and poor oral bioavailability make significant milk transfer biologically unlikely. However, the study was small, and larger confirmatory studies have not been conducted.
Does tirzepatide pass into breast milk?
In two studies, tirzepatide was undetectable in approximately 96 percent of milk samples and present only in negligible amounts (less than 0.02 percent of the maternal dose) in the remaining samples. The LactMed database states that tirzepatide use is not a reason to discontinue breastfeeding.
Can GLP-1 medications reduce my milk supply?
Indirectly, yes. GLP-1 receptor agonists suppress appetite, and if a breastfeeding mother does not consume enough calories (approximately 450 to 500 extra daily), milk production can decline. This is a nutritional issue rather than a direct pharmacological effect on the mammary glands. Careful attention to dietary intake can mitigate this risk.
Is oral semaglutide safe while breastfeeding?
No. Oral semaglutide (Rybelsus) contains the absorption enhancer salcaprozate sodium, which may transfer into breast milk and accumulate in the infant. Only injectable subcutaneous formulations of semaglutide should be used during breastfeeding.
Can I take compounded semaglutide or tirzepatide while breastfeeding?
The lactation safety data applies to pharmaceutical-grade injectable formulations. Compounded versions may contain different excipients, and their milk transfer properties have not been specifically studied. If physician-approved, using a reputable pharmacy and following strict storage and handling protocols is essential.
How long should I wait after weaning to start GLP-1 therapy?
Once breastfeeding has fully stopped and milk production has ceased, there is no required waiting period before starting semaglutide or tirzepatide. Consult with your physician to confirm you meet the criteria for GLP-1 therapy.
Will taking semaglutide while breastfeeding affect my baby's appetite or metabolism?
There is no evidence that semaglutide affects infant appetite or metabolism through breast milk exposure. The undetectable milk levels suggest the infant receives no meaningful pharmacological exposure. However, long-term studies on infant metabolic outcomes have not been conducted.
What is the safest GLP-1 medication for breastfeeding mothers?
Based on current evidence, injectable semaglutide and injectable tirzepatide have the most lactation safety data. Both show undetectable or negligible milk levels. Tirzepatide has a slightly higher molecular weight, which theoretically makes it even less likely to transfer into milk, but both appear similarly safe based on available studies.
Can I take Wegovy or Ozempic while breastfeeding?
Both are injectable semaglutide formulations. The lactation study used injectable semaglutide and found undetectable milk levels. Any decision to use Wegovy (the weight loss formulation) or Ozempic (the diabetes formulation) during breastfeeding should be made with your physician, considering the evidence reviewed in this guide.
Should I pump and dump after taking my GLP-1 injection?
The current evidence does not support pumping and dumping after a semaglutide or tirzepatide injection. Since the drug was undetectable in milk at all time points tested (0, 12, and 24 hours post-dose for semaglutide), there is no peak concentration period to avoid. Pumping and dumping would unnecessarily waste breast milk without providing additional safety.
What the medical community recommends right now
The formal medical consensus remains cautious. Most professional organizations and regulatory bodies recommend against GLP-1 use during breastfeeding, not because of proven danger, but because of insufficient data to confirm safety definitively.
The FDA position
The FDA labels for both semaglutide and tirzepatide neither recommend nor prohibit use during lactation. They note the absence of data and recommend that physicians weigh the benefits of breastfeeding against the clinical need for the medication.
The CMAJ guidance
A guidance article published in the Canadian Medical Association Journal (CMAJ) on glucagon-like peptide-1 receptor agonists during pregnancy and lactation concluded that while the safety data is limited, the pharmacological properties of GLP-1 agonists (large molecular weight, poor oral bioavailability) suggest low theoretical risk during breastfeeding.
The LactMed position
LactMed, the most widely referenced database for medication safety during breastfeeding, provides the most specific guidance. For semaglutide, it notes the undetectable milk levels and states that only injectable forms should be used. For tirzepatide, it explicitly states that the medication is not a reason to discontinue breastfeeding.
The MotherToBaby position
MotherToBaby, a service of the Organization of Teratology Information Specialists, notes that while studies are limited, the available data on injectable semaglutide and tirzepatide suggest low risk during breastfeeding. They recommend consulting with a healthcare provider for individualized guidance.
Where the recommendations may be heading
The direction of the evidence is clear. Each new study adds to the picture that injectable GLP-1 receptor agonists do not transfer into breast milk in meaningful amounts. As more data accumulates, formal guidelines will likely become more permissive. But medicine moves slowly when infant safety is involved, and that caution, while frustrating for mothers who want definitive answers, is appropriate.

The emotional reality: why this question matters so much
Behind every search for "can you take GLP-1 while breastfeeding" is a mother dealing with competing pressures. The pressure to lose weight. The pressure to breastfeed. The pressure to be healthy for her child. The pressure to feel like herself again.
Postpartum weight retention is not vanity. Carrying significant extra weight increases the risk of type 2 diabetes, cardiovascular disease, and future pregnancy complications. The psychological impact of not recognizing your own body, of clothes that do not fit, of a world that celebrates "bouncing back" while offering no meaningful help, is real and documented.
At the same time, the desire to breastfeed is equally valid. The health benefits for infants, reduced infections, improved immune function, potential long-term metabolic benefits, are well established. The bonding experience matters. And the social pressure to breastfeed, while sometimes excessive, reflects genuine evidence about infant health.
Telling a mother to "just wait until you wean" dismisses the urgency of her situation. Telling her to "just take the medication" dismisses the importance of breastfeeding. Neither response respects the complexity of her position.
The most helpful thing anyone can do for a mother in this situation is give her the actual evidence, clearly presented, so she can make an informed decision with her physician. That is what this guide attempts to provide.
The research gaps that still need to be filled
Honest reporting requires acknowledging what we do not know. The current evidence is promising but incomplete. Several critical research gaps remain.
Sample size
The semaglutide study included eight mothers. The tirzepatide studies included 11 and 5 mothers respectively. These are tiny sample sizes by medical research standards. Rare adverse events or idiosyncratic reactions could be missed entirely with groups this small.
Dose range
Semaglutide was studied at doses up to 1 mg weekly. The therapeutic dose for weight loss goes up to 2.4 mg. Tirzepatide was studied at up to 5 mg weekly. The therapeutic range extends to 15 mg. Higher dose data is absent.
Duration
The longest observation period was 9 weeks for semaglutide. Many women breastfeed for 12 months or longer. Whether milk transfer characteristics change with prolonged use is unknown.
Long-term infant outcomes
No study has followed exposed infants for years to assess potential effects on growth, metabolism, appetite regulation, or pancreatic development. While the undetectable milk levels make significant effects unlikely, formal long-term safety data does not exist.
Milk composition
No study has examined whether GLP-1 therapy affects the nutritional composition of breast milk itself, independent of drug transfer. Whether macronutrient ratios, micronutrient concentrations, or bioactive factors in milk change during maternal GLP-1 therapy is an open question.
Compounded formulations
All lactation data comes from FDA-approved pharmaceutical products. The rapidly growing market for compounded GLP-1 preparations has generated no breastfeeding safety data.
These gaps are not reasons to panic. They are reasons to be informed, cautious, and engaged with your healthcare team. The existing data is reassuring. The missing data is a reminder that reassurance is not the same as certainty.
A practical summary for decision-making
After reviewing all available evidence, here is a distilled summary that breastfeeding mothers can bring to their physician.
What the evidence supports:
Injectable semaglutide was undetectable in breast milk from eight nursing mothers at doses up to 1 mg weekly
Injectable tirzepatide was undetectable in approximately 96 percent of milk samples from 11 nursing mothers at 5 mg doses
Both medications have molecular weights (4,114 and 4,813 Daltons) that make significant milk transfer biologically unlikely
Any trace amounts in milk would have negligible oral bioavailability in the infant
No adverse infant effects were observed in any of the lactation studies
LactMed states tirzepatide is not a reason to discontinue breastfeeding
What the evidence does not support:
Use of oral semaglutide (Rybelsus) during breastfeeding due to the absorption enhancer SNAC
Use of any GLP-1 agonist without physician supervision during lactation
Aggressive weight loss goals (more than 1 to 1.5 pounds per week) while breastfeeding
Assumption that compounded formulations carry the same safety profile as pharmaceutical-grade products without specific data
What remains unknown:
Safety at higher therapeutic doses (semaglutide above 1 mg, tirzepatide above 5 mg)
Long-term infant outcomes from maternal GLP-1 exposure during breastfeeding
Effects on breast milk composition and nutritional quality
Safety of newer GLP-1 agonists like retatrutide
The decision is ultimately yours and your physician's. The evidence presented here is meant to inform that decision, not make it for you. Every mother's health status, infant's age and feeding patterns, and clinical need for weight loss medication creates a unique risk-benefit calculation.
SeekPeptides exists to provide exactly this kind of detailed, evidence-based guidance on peptide-related questions. For mothers navigating the complex intersection of GLP-1 therapy and breastfeeding, having access to comprehensive, up-to-date research, dosage calculators, and protocol guidance can make the difference between informed confidence and anxious guessing.
For those who want deeper support with their peptide research journey, SeekPeptides members access comprehensive protocols, expert-reviewed guides, and a community of researchers who have navigated these exact questions. The postpartum period is challenging enough without facing complex medical decisions alone.
External resources
Subcutaneous Semaglutide during Breastfeeding: Infant Safety (PMC)
GLP-1 Receptor Agonists During Pregnancy and Lactation (CMAJ/PMC)
In case I do not see you, good afternoon, good evening, and good night. May your milk supply stay abundant, your recovery stay steady, and your decisions stay informed.