Feb 13, 2026

Before you blame tirzepatide for your muscle pain, consider this. That ache in your shoulders might not be a side effect at all. It could be dehydration. It could be electrolyte depletion from nausea you barely noticed. It could be your body adjusting to rapid weight loss, or the new walking routine you started because you finally have energy again. The relationship between tirzepatide and muscle pain is more complex than most doctors acknowledge, and understanding the actual mechanisms behind it can mean the difference between stopping a medication that is working and solving a problem that has nothing to do with the drug itself.
Muscle pain on tirzepatide is real, but it is also misunderstood. Clinical trial data shows that musculoskeletal complaints occur in tirzepatide users, but not at rates significantly higher than placebo. Yet postmarketing reviews tell a different story, with up to 15% of users reporting body aches that were not captured in controlled trials. This disconnect matters because it shapes how we approach the problem. If muscle pain were an inherent pharmacological effect of tirzepatide, we would see it uniformly across users at predictable rates. Instead, we see patterns that suggest modifiable factors are at play.
This guide examines every documented mechanism linking tirzepatide to muscle pain, from electrolyte imbalances caused by gastrointestinal side effects to the paradoxical inflammatory responses seen in rare cases. You will learn the exact timeline of when muscle pain typically emerges, how to distinguish normal adaptation from concerning symptoms, and eight evidence-based strategies to prevent or resolve body aches without discontinuing therapy. We will also cover the rare but serious risk of rhabdomyolysis, a condition that has been documented in tirzepatide users and requires immediate medical attention.
What the clinical data actually says about tirzepatide and muscle pain
The SURMOUNT-1 clinical trial, which enrolled over 2,500 participants, documented musculoskeletal complaints including back pain, arthralgia, and myalgia. These were tracked as adverse events. The critical finding was that these symptoms did not occur at statistically significant higher rates in tirzepatide groups compared to placebo groups. This suggests that muscle pain in the controlled trial environment was not driven primarily by the pharmacological action of tirzepatide at standard doses.
Postmarketing surveillance presents a different picture. A review of 500 tirzepatide users found approximately 15% reported muscle aches or body pain that they attributed to the medication. This rate is substantially higher than what clinical trials captured. The discrepancy likely reflects differences in real-world use versus controlled trials. In clinical trials, participants receive structured support, regular monitoring, and often nutritional counseling. They are screened to exclude certain comorbidities. In real-world settings, users may have inadequate hydration, suboptimal protein intake, or underlying conditions that interact with rapid weight loss.
Case reports provide the most detailed mechanistic insights. One published case described a 37-year-old male with class I obesity who developed new-onset neck and back pain when his dose increased from 7.5 mg to 10 mg. Symptoms appeared within 24 hours of injection, peaked at 48 to 72 hours, and occurred consistently with each dose. The pain resolved when he changed his injection site from abdomen to thigh. This suggests a localized inflammatory or tissue response rather than a systemic pharmacological effect.
Another case involved a 43-year-old male with type 2 diabetes and class II obesity who experienced bilateral neck and shoulder pain 24 hours after initiating therapy. Peak pain occurred at 72 hours post-injection. Changing the injection site to his arm resolved the symptoms. These cases point to injection site reactions as a contributing factor in some users, particularly those who experience pain that correlates temporally with injections and anatomically with injection regions.
The most severe documented case involved a 35-year-old female who developed rhabdomyolysis while on tirzepatide. Her creatine kinase (CK) levels reached 57,014 units per liter, compared to a normal reference range of 25 to 192. Muscle biopsy revealed necrotizing myopathy. The Naranjo adverse drug reaction probability scale assigned a score of 7, indicating a probable causal relationship. She recovered after discontinuation of tirzepatide and administration of intravenous fluids. This case is rare, but it establishes that severe muscle pathology can occur in the context of tirzepatide use.
The overall incidence of myalgia in clinical practice appears to be between 2% and 5%, based on aggregated data from multiple sources. Most cases are mild to moderate in severity. They typically emerge within the first four to eight weeks of therapy, often after dose escalation. Resolution occurs within two to four weeks with appropriate intervention in the majority of cases. Understanding this timeline helps distinguish tirzepatide-related muscle pain from unrelated musculoskeletal conditions.
It is also worth noting what tirzepatide does not cause. A study presented at the American College of Rheumatology found that tirzepatide users had a lower risk of joint pain, including low back pain, hip pain, and knee pain, compared to users of other weight loss medications. The mechanism here is straightforward. Weight loss reduces mechanical loading on joints, which decreases pain from osteoarthritis and other degenerative conditions. This protective effect is relevant because it means some users may experience net improvement in musculoskeletal symptoms despite transient muscle aches.
Seven reasons tirzepatide causes muscle pain (and which ones apply to you)
Understanding why muscle pain occurs on tirzepatide requires examining the physiological cascades triggered by GLP-1 and GIP receptor agonism, the metabolic shifts from rapid weight loss, and the behavioral changes that accompany appetite suppression. Each mechanism has distinct characteristics that can help you identify which factors are contributing to your symptoms.
Electrolyte depletion from gastrointestinal side effects
Nausea, vomiting, and diarrhea are among the most common side effects of tirzepatide, occurring in 20% to 30% of users during dose escalation. These gastrointestinal symptoms cause fluid and electrolyte loss. Potassium and magnesium are particularly vulnerable. When potassium levels drop, muscle cells struggle to maintain proper electrical gradients. This leads to cramping, weakness, and soreness. Magnesium deficiency impairs muscle relaxation and increases susceptibility to spasms.
The timeline of electrolyte-related muscle pain often corresponds with periods of intense gastrointestinal symptoms. If you experienced significant nausea or diarrhea in the days before muscle pain began, electrolyte depletion is a likely contributor. This mechanism is supported by the observation that muscle cramps and aches often improve rapidly with electrolyte supplementation. Potassium-rich foods like bananas, potatoes, and spinach can help, as can magnesium supplementation at 300 to 400 mg daily. B vitamin support can also help with overall nutrient balance.
Dehydration compounds the problem. Reduced fluid intake due to nausea, combined with increased fluid loss from vomiting or diarrhea, decreases blood volume. This forces the cardiovascular system to work harder during physical activity, leading to muscle fatigue and soreness. Monitoring urine color provides a simple assessment. Dark yellow urine suggests inadequate hydration. Aim for pale yellow. Maintaining hydration on tirzepatide requires conscious effort, especially during the first weeks after dose increases.
Rapid lean mass loss and muscle catabolism
Weight loss on GLP-1 receptor agonists is not purely fat loss. Studies show that 25% to 40% of weight lost comes from lean body mass, including muscle. This is higher than the typical 20% to 25% lean mass loss seen with caloric restriction alone. The mechanism involves inadequate protein intake relative to the body needs during rapid weight loss. Appetite suppression reduces total food intake, but protein is often disproportionately affected because high-protein foods tend to be more filling.
When protein intake falls below 1.6 grams per kilogram of body weight per day, the body begins catabolizing muscle tissue to meet amino acid requirements for essential processes. This breakdown generates muscle soreness similar to what occurs after intense exercise. The soreness is not from muscle damage caused by mechanical stress, but from the metabolic processes of proteolysis and amino acid mobilization.
Muscle loss also reduces the capacity for physical activity. Weaker muscles fatigue more easily, leading to delayed-onset muscle soreness (DOMS) even from light activities. If you have noticed that activities which were previously easy now leave you sore, and you have lost significant weight quickly, muscle catabolism is likely involved. Strategies to preserve muscle mass during weight loss focus on increasing protein intake to 1.6 to 2.2 grams per kilogram daily and incorporating resistance training.
Protein deficiency and inadequate muscle repair
Closely related to lean mass loss is the issue of protein deficiency. Even if you are not in a severe catabolic state, suboptimal protein intake impairs the repair of normal muscle microtrauma that occurs during daily activities. Muscle tissue undergoes constant turnover, with breakdown and synthesis occurring simultaneously. When synthesis cannot keep pace with breakdown due to insufficient amino acids, cumulative damage manifests as persistent soreness and achiness.
The appetite suppression caused by tirzepatide can make consuming adequate protein challenging. Many users report that they simply do not feel hungry, and when they do eat, protein-rich foods feel particularly heavy or unappealing. This creates a situation where total caloric intake may be adequate for weight loss, but protein intake is inadequate for muscle maintenance. Tracking protein intake for several days using tools like the peptide calculator approach can reveal whether you are meeting minimum requirements.
A practical benchmark is 100 grams of protein daily for most adults on tirzepatide, with higher amounts needed for larger individuals or those engaging in resistance training. If you are consuming less than 80 grams per day, protein deficiency is almost certainly contributing to muscle symptoms. Optimizing macronutrient intake on tirzepatide often requires intentional planning, including protein shakes or other concentrated sources.
Dehydration cascade and reduced muscle perfusion
Dehydration on tirzepatide results from multiple mechanisms operating simultaneously. Reduced fluid intake occurs because appetite suppression extends to thirst. Increased fluid loss happens through gastrointestinal side effects. Some users also report increased urination, possibly related to improved glycemic control and reduced fluid retention. The combined effect can be significant hypovolemia, even if you do not feel particularly thirsty.
Muscle tissue is approximately 75% water. Dehydration reduces muscle cell volume, which impairs contractile function and increases susceptibility to injury. Blood flow to muscles also decreases in hypovolemic states, reducing oxygen and nutrient delivery. This creates an environment where muscles are more prone to fatigue and soreness. The soreness often has a diffuse, achiness quality rather than sharp pain localized to specific muscle groups.
Monitoring hydration status involves more than just drinking when thirsty. By the time thirst is perceived, dehydration is already present. A better approach is to consume a baseline of eight to ten glasses of water daily, with additional intake if you are experiencing gastrointestinal side effects or engaging in physical activity. Electrolyte solutions can enhance hydration more effectively than water alone, particularly if sodium and potassium stores are depleted. Optimizing tirzepatide results includes addressing hydration proactively.
Injection site reactions and localized inflammation
The case reports mentioned earlier highlight a mechanism that is often overlooked. Injection site reactions can manifest as regional muscle pain that radiates from the injection area. Tirzepatide is administered subcutaneously, but the inflammatory response can extend into underlying muscle tissue, particularly if the injection is deeper than intended or if there is individual susceptibility to inflammatory reactions.
Symptoms consistent with injection site-related muscle pain include pain that begins within 24 to 72 hours of injection, pain that is worse on the side of the body where injection occurred, and pain that improves or resolves when injection sites are rotated to different anatomical regions. One user reported in injection versus oral discussions that abdominal injections caused neck and back pain, while thigh injections did not. Another found that arm injections resolved shoulder pain that occurred with abdominal injections.
The mechanism may involve cytokine release at the injection site, with inflammatory mediators diffusing to nearby muscle tissue. Alternatively, subcutaneous injection may trigger referred pain patterns, where pain is perceived in areas distant from the actual site of tissue irritation. Rotating injection sites systematically can help determine if this mechanism is contributing. Proper injection technique for GLP-1 medications includes using different anatomical sites in a structured rotation pattern.
Dose escalation response and metabolic adjustment
Muscle pain frequently emerges after dose increases. This pattern was evident in the case report where pain developed when the dose increased from 7.5 mg to 10 mg. The temporal relationship suggests that higher doses trigger metabolic or inflammatory responses that lower doses do not. This could relate to more pronounced gastrointestinal side effects at higher doses, leading to greater electrolyte and fluid losses. It could also reflect dose-dependent effects on muscle metabolism that are not fully understood.
Tirzepatide activates GLP-1 receptors in skeletal muscle tissue. These receptors mediate glucose uptake through a mechanism that involves phosphoinositide 3-kinase (PI3K) but is independent of protein kinase B (PKB). This pathway influences glycogen synthesis in muscle. Rapid changes in muscle glycogen content can cause transient soreness as cells adjust to altered metabolic states. Higher doses may produce more dramatic shifts in muscle glucose handling, leading to symptoms during the adjustment period.
If your muscle pain began shortly after a dose increase and you had no symptoms at lower doses, dose escalation response is likely involved. The solution may be to slow the titration schedule, extending the time at each dose level from four weeks to six or eight weeks. This allows metabolic adaptation to occur more gradually. Adjusting tirzepatide titration schedules can reduce side effects without compromising long-term efficacy.
Underlying conditions unmasked by weight loss or metabolic changes
Tirzepatide does not cause muscle pain in isolation. It can unmask or exacerbate underlying musculoskeletal conditions that were previously asymptomatic or mild. Rapid weight loss changes body mechanics, altering how forces are distributed across joints and muscles. This can expose weaknesses in posture, gait, or muscle balance that were compensated for when body mass was higher. The result is pain that is temporally associated with tirzepatide use but is mechanistically related to biomechanical changes.
Metabolic improvements from tirzepatide can also affect pain perception. Improved insulin sensitivity and reduced systemic inflammation may alter how the nervous system processes pain signals. Paradoxically, this can sometimes make people more aware of minor aches and pains that were previously drowned out by higher baseline inflammation. This is not a worsening of musculoskeletal health, but a change in perception.
Additionally, increased physical activity is common as users experience more energy and motivation. Starting new exercise routines, increasing walking, or resuming activities that were previously too difficult can cause delayed-onset muscle soreness. This is normal muscle adaptation, not a side effect of tirzepatide. However, because the activity change is enabled by tirzepatide, users may attribute the soreness to the medication. Supporting athletic performance during metabolic changes involves gradual progression in activity levels.
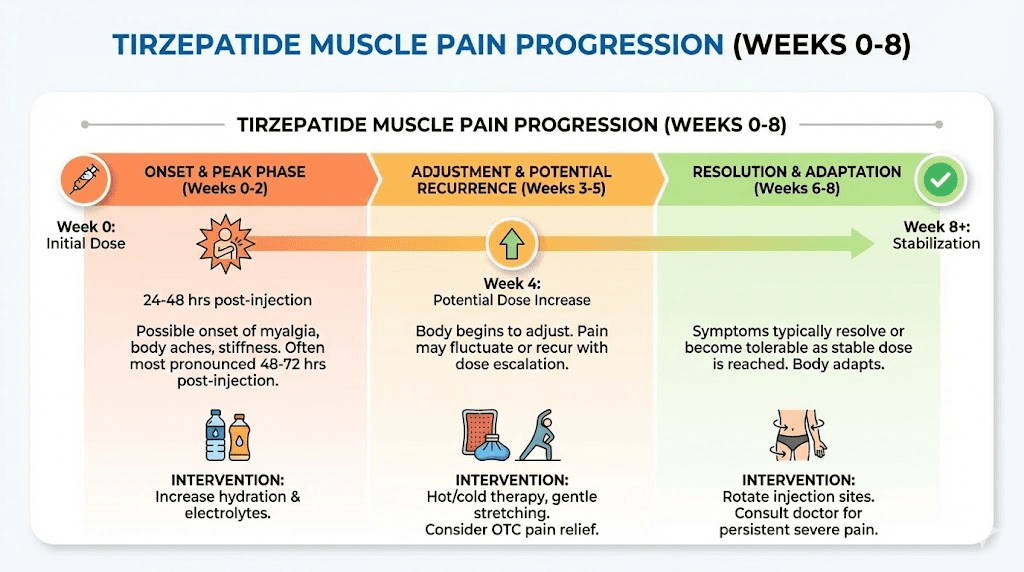
The timeline of tirzepatide muscle pain (when it starts, peaks, and resolves)
Understanding the typical progression of muscle pain on tirzepatide helps distinguish normal adaptation from concerning symptoms that require medical evaluation. Most users who experience muscle pain report onset within the first four to eight weeks of therapy. This window corresponds with the initial dose escalation phase and the period of most rapid metabolic adjustment.
The immediate post-injection period is particularly relevant for injection site-related pain. Symptoms typically begin 24 to 48 hours after administration and peak at 48 to 72 hours. This pattern mirrors the pharmacokinetic profile of tirzepatide, which reaches maximum plasma concentration approximately 24 hours after subcutaneous injection after subcutaneous injection. If pain correlates consistently with this timeline and recurs with each weekly dose, injection site reactions or localized inflammatory responses are likely contributors.
For pain related to electrolyte depletion or dehydration, onset is more variable and depends on the severity of gastrointestinal side effects. Users who experience significant nausea or diarrhea may notice muscle cramps and soreness within days. Those with milder gastrointestinal symptoms may have a more gradual onset over two to three weeks as cumulative deficits in hydration and electrolytes accumulate. This type of pain often improves within three to five days of aggressive rehydration and electrolyte repletion.
Muscle pain from rapid lean mass loss and protein deficiency tends to develop more slowly, typically becoming noticeable in weeks three to six of therapy. This reflects the time required for protein balance to become sufficiently negative that muscle catabolism generates symptoms. The pain is often described as a generalized achiness or heaviness in muscles, rather than sharp or localized pain. It may be accompanied by weakness, particularly noticeable when climbing stairs or lifting objects. Managing body composition changes on tirzepatide can prevent this type of pain from developing.
Peak severity of muscle pain varies widely. In cases related to electrolyte or hydration issues, peak pain may last only a few days before improvement begins. For pain related to muscle catabolism or metabolic adjustment, peak symptoms may persist for two to three weeks. The majority of users report significant improvement or complete resolution within four weeks of symptom onset, provided that contributing factors are addressed.
Resolution accelerates when interventions target the underlying mechanisms. Increasing protein intake to 1.8 to 2.2 grams per kilogram daily can reverse muscle catabolism within one to two weeks. Optimizing hydration and electrolytes often produces noticeable improvement within 48 to 72 hours. Rotating injection sites may eliminate pain immediately if injection site reactions were the primary cause. The key is identifying which mechanisms are operative in your case and intervening appropriately.
Some users experience recurrent muscle pain with each dose escalation. This pattern suggests that metabolic adjustment to higher doses requires time, and that the standard four-week titration schedule may be too aggressive. Extending the titration interval to six or eight weeks allows the body to adapt more fully before introducing additional metabolic stress. Alternative dosing strategies for tirzepatide include slower escalation protocols that reduce the incidence of side effects.
Rhabdomyolysis and tirzepatide: the rare but serious risk you need to understand
Rhabdomyolysis is the breakdown of muscle tissue that releases myoglobin into the bloodstream. Myoglobin is toxic to kidneys and can cause acute kidney injury or failure. The condition is characterized by severe muscle pain, weakness, and dark brown or cola-colored urine. It is rare, but it has been documented in tirzepatide users, making awareness essential.
The case mentioned earlier involved a 35-year-old female who developed necrotizing myopathy with CK levels exceeding 57,000 units per liter. Normal CK levels range from 25 to 192 units per liter for women and 25 to 200 for men. Levels above 1,000 suggest significant muscle injury. Levels above 5,000 indicate severe rhabdomyolysis with high risk of kidney damage. Her levels were more than 200 times the upper limit of normal.
The mechanism linking tirzepatide to rhabdomyolysis is not fully established. Potential contributing factors include severe electrolyte imbalances, particularly hypokalemia and hypophosphatemia, which can occur secondary to gastrointestinal side effects. Dehydration increases the risk by concentrating myoglobin in renal tubules. Drug interactions may also play a role, as certain medications combined with tirzepatide could potentiate muscle toxicity. Statin medications, which are commonly used in patients with metabolic syndrome, are known to increase rhabdomyolysis risk.
Early recognition is critical. Warning signs include muscle pain that is severe and out of proportion to any activity, muscle weakness that makes it difficult to rise from a chair or climb stairs, and any change in urine color to dark brown or red. Swelling of affected muscles, fever, and confusion can also occur in severe cases. If you experience these symptoms, seek medical attention immediately. Rhabdomyolysis is diagnosed with blood tests measuring CK levels and urinalysis detecting myoglobin.
Treatment involves discontinuation of tirzepatide, aggressive intravenous fluid administration to flush myoglobin from the kidneys, and correction of electrolyte abnormalities. Most patients recover fully if treatment is initiated promptly. Delayed treatment can result in permanent kidney damage requiring dialysis. The risk of recurrence with rechallenge is not well characterized, but most clinicians would avoid restarting tirzepatide after a confirmed episode of rhabdomyolysis.
Risk factors for rhabdomyolysis in the context of tirzepatide use include pre-existing kidney disease, use of statin medications, severe gastrointestinal side effects leading to marked dehydration or electrolyte depletion, and intense physical exercise during the early weeks of therapy. If you have multiple risk factors, closer monitoring with periodic CK levels may be warranted, particularly during dose escalation. Recognizing serious side effects on tirzepatide requires attention to symptoms that go beyond typical adjustment effects.
Eight proven strategies to prevent and treat muscle pain on tirzepatide
Preventing muscle pain is more effective than treating it after onset. The following strategies target the primary mechanisms discussed earlier and have documented efficacy in clinical practice. Implementing multiple strategies simultaneously provides the best results, as muscle pain on tirzepatide is typically multifactorial.
Strategy one: aggressive hydration with electrolyte repletion
Hydration is the foundation of preventing muscle pain on tirzepatide. Aim for a minimum of eight to ten glasses of water daily, which translates to approximately two to 2.5 liters. This baseline should increase if you are experiencing gastrointestinal side effects, engaging in physical activity, or living in a hot climate. Monitoring urine color provides real-time feedback. Pale yellow indicates adequate hydration. Dark yellow or amber suggests inadequacy.
Electrolyte supplementation goes beyond drinking water. Sodium, potassium, and magnesium are critical for muscle function. Sodium intake can come from broths, electrolyte drinks, or simply salting food more liberally than usual. Potassium-rich foods include bananas, potatoes, avocados, spinach, and beans. A single medium banana provides approximately 400 mg of potassium. Daily needs range from 2,500 to 3,000 mg for most adults.
Magnesium supplementation at 300 to 400 mg daily is particularly effective for muscle cramps and spasms. Magnesium glycinate or magnesium citrate are well-absorbed forms with lower risk of gastrointestinal side effects compared to magnesium oxide. Magnesium also supports muscle relaxation and recovery. Deficiency is common even in the general population, and tirzepatide can exacerbate it through gastrointestinal losses. Supporting digestive health during GLP-1 therapy can reduce electrolyte losses at the source.
Strategy two: high-protein intake to prevent muscle catabolism
Protein intake of 1.6 to 2.2 grams per kilogram of body weight daily is the target for preserving lean mass during rapid weight loss. For a 90-kilogram individual using dosage calculations, this translates to 144 to 198 grams of protein daily. This level is significantly higher than the general recommendation of 0.8 grams per kilogram and requires intentional planning on tirzepatide, given appetite suppression.
Distributing protein across three to four meals enhances muscle protein synthesis more effectively than consuming the same total amount in one or two meals. Each meal should contain 30 to 50 grams of protein. Protein shakes can fill gaps when whole food intake is challenging. Whey protein isolate provides 25 grams per scoop and is rapidly absorbed. Plant-based options like pea protein or soy protein are viable alternatives.
Prioritizing protein at the start of meals takes advantage of the limited appetite window that many tirzepatide users experience. Eating protein-rich foods first ensures adequate intake before satiety kicks in. Chicken breast, fish, eggs, Greek yogurt, and cottage cheese are nutrient-dense sources. Tracking protein intake for one week using a food diary or app can reveal whether you are meeting targets and where adjustments are needed. Optimizing muscle preservation during weight loss relies heavily on protein sufficiency.
Strategy three: resistance training to stimulate muscle protein synthesis
Resistance training provides an anabolic stimulus that partially counteracts the catabolic effects of caloric restriction and rapid weight loss. It signals the body to prioritize muscle preservation. Two to three sessions per week, targeting all major muscle groups, is sufficient for most individuals. Each session should include six to eight exercises, with two to three sets of eight to twelve repetitions per exercise.
The intensity does not need to be extreme. Moderate loads that challenge muscles without causing excessive fatigue are appropriate, particularly during the initial months on tirzepatide. Progressive overload, gradually increasing weight or repetitions over time, ensures continued adaptation. Bodyweight exercises like squats, push-ups, and planks are effective starting points if gym access is limited.
Resistance training also improves insulin sensitivity and enhances the metabolic benefits of tirzepatide. Muscle is metabolically active tissue that supports long-term weight maintenance. Building or preserving muscle during weight loss improves body composition and functional capacity. Timing protein intake around resistance training sessions, with 20 to 30 grams consumed within two hours post-workout, maximizes muscle protein synthesis. Combining peptides with strength training can amplify muscle preservation effects.
Strategy four: systematic injection site rotation
Rotating injection sites reduces the risk of localized inflammation and tissue irritation that can manifest as muscle pain. The abdomen, thighs, upper arms, and buttocks are all approved injection sites for subcutaneous GLP-1 medications. Using a different site each week and avoiding the same exact spot within a site prevents tissue buildup and inflammatory responses.
A structured rotation schedule might look like this. Week one uses the right abdomen, week two the left abdomen, week three the right thigh, week four the left thigh, week five the right upper arm, week six the left upper arm. After six weeks, the cycle repeats. Within each site, injections should be at least one inch away from previous injection points. Scarring or lipohypertrophy at injection sites impairs medication absorption and increases discomfort.
Proper injection technique also matters. Pinch the skin to create a fold, insert the needle at a 90-degree angle for most individuals or a 45-degree angle for very lean individuals, inject slowly, and hold for five seconds after depressing the plunger before withdrawing the needle. Allow the skin to return to room temperature if refrigerated medication is used, as cold injections are more painful. Proper peptide storage ensures medication quality. Proper handling of compounded tirzepatide includes injection technique considerations.
Strategy five: slower dose titration to allow metabolic adaptation
The standard tirzepatide titration schedule increases the dose every four weeks. Starting at 2.5 mg, the schedule proceeds to 5 mg, 7.5 mg, 10 mg, 12.5 mg, and finally 15 mg. For some users, this pace is too aggressive, leading to recurrent side effects including muscle pain with each escalation. Extending the interval to six or eight weeks at each dose allows more complete metabolic and physiological adaptation.
Slower titration does not compromise long-term efficacy. Weight loss may be slightly slower in the first few months, but total weight loss at one year is comparable. The benefit is reduced side effect burden and improved adherence. Users who experience severe muscle pain after dose increases should discuss extended titration schedules with their prescribing physician. Some clinicians also use smaller dose increments, such as increasing by 1.25 mg every four weeks instead of 2.5 mg.
Another approach is to skip dose levels for individuals who achieve adequate weight loss and metabolic improvement at lower doses. Not everyone needs to reach the maximum dose of 15 mg. If you are losing one to two pounds per week consistently at 7.5 mg and tolerating the medication well, there may be no reason to escalate further. Customizing tirzepatide dosing based on individual response allows for personalized optimization.
Strategy six: gentle stretching and mobility work
Stretching improves muscle flexibility, reduces tension, and enhances blood flow to muscle tissue. These effects can alleviate muscle pain and prevent it from worsening. Gentle static stretching for 10 to 15 minutes twice daily targets major muscle groups including hamstrings, quadriceps, calves, hip flexors, chest, and shoulders. Each stretch should be held for 20 to 30 seconds without bouncing.
Mobility work complements stretching by addressing joint range of motion and movement patterns. Exercises like arm circles, leg swings, and spinal rotations prepare muscles for activity and reduce stiffness. Yoga and tai chi are effective forms of mobility work that also incorporate mindfulness and stress reduction, which can modulate pain perception. Foam rolling or self-myofascial release techniques can reduce muscle tightness and improve recovery.
Avoid intense stretching or deep tissue massage during periods of acute discomfort of acute muscle pain, as aggressive manipulation can worsen inflammation. The goal is gentle, controlled movement that encourages circulation and flexibility without causing additional trauma. Morning stretching routines are particularly beneficial, as muscles are often stiffest upon waking. Accelerating recovery from muscle strain can involve targeted stretching protocols.
Strategy seven: avoid intense exercise during the first month
Starting a new intense exercise program during the first month of tirzepatide therapy is a common contributor to muscle pain. The body is already undergoing significant metabolic stress from appetite suppression, rapid weight loss, and hormonal changes. Adding high-intensity exercise compounds this stress and increases the risk of muscle injury, excessive fatigue, and prolonged soreness.
A better approach is to maintain light to moderate activity during the first four to six weeks. Walking, gentle cycling, swimming, or light yoga support metabolic health without overwhelming the musculoskeletal system. Once adaptation to tirzepatide is evident, typically after the first month, exercise intensity can be gradually increased. Adding one new element at a time, such as increasing duration before increasing intensity, allows the body to adapt systematically.
This recommendation is particularly relevant for individuals who were sedentary before starting tirzepatide and feel motivated to exercise due to increased energy. The enthusiasm is valuable, but pacing is critical. Delayed-onset muscle soreness from unaccustomed exercise can be severe and discouraging, potentially derailing both exercise adherence and medication compliance. Supporting athletic performance during body recomposition involves strategic timing of training intensification.
Strategy eight: monitor and address underlying conditions
If muscle pain persists despite addressing hydration, protein, and activity factors, underlying conditions should be considered. Vitamin D deficiency is common and causes muscle pain and weakness. Serum 25-hydroxyvitamin D levels below 20 ng/mL indicate deficiency. Supplementation with 2,000 to 4,000 IU daily corrects deficiency in most individuals within eight to twelve weeks. Thyroid dysfunction, particularly hypothyroidism, also presents with muscle aches and should be screened for if symptoms persist.
Autoimmune conditions like polymyalgia rheumatica or inflammatory myopathies can be unmasked by metabolic changes or weight loss. These conditions typically present with symmetrical muscle pain, morning stiffness lasting more than 30 minutes, and elevated inflammatory markers like C-reactive protein or erythrocyte sedimentation rate. Rheumatologic evaluation is warranted if these features are present. Managing inflammatory musculoskeletal conditions may require targeted therapies beyond lifestyle interventions.
Medication interactions should also be reviewed. Statins, fibrates, and certain antibiotics can cause myopathy, and their effects may be potentiated by tirzepatide-induced metabolic changes. Discussing all medications and supplements with your physician ensures that interactions are identified and managed appropriately. At SeekPeptides, members have access to detailed protocols for identifying and addressing complex side effect profiles.
Supplements that actually help with tirzepatide muscle pain
Supplement evidence for muscle pain in the context of GLP-1 therapy is limited, but several supplements have mechanisms that address the underlying causes identified earlier. These are not replacements for foundational strategies like hydration and protein intake, but they can provide additional benefit when used appropriately.
Magnesium supplementation at 300 to 400 mg daily is the most evidence-backed intervention. Magnesium is essential for muscle relaxation, and deficiency is associated with cramps and spasms. Magnesium glycinate is preferred for muscle symptoms due to superior absorption and minimal gastrointestinal side effects. Studies in athletes show that magnesium supplementation reduces muscle soreness and improves recovery. The mechanism involves regulation of calcium flux in muscle cells and modulation of inflammatory pathways.
Creatine monohydrate supports muscle preservation during caloric restriction. It increases muscle phosphocreatine stores, which support ATP regeneration during muscle contraction. Supplementation at 5 grams daily has been shown to reduce muscle protein breakdown and support lean mass retention in individuals losing weight. Creatine also draws water into muscle cells, improving muscle hydration and function. It is one of the most well-researched supplements in sports nutrition and has an excellent safety profile. When combined with BPC-157, some researchers explore synergistic tissue support.
Branched-chain amino acids (BCAAs), particularly leucine, stimulate muscle protein synthesis. Supplementation with 5 to 10 grams of BCAAs before or after resistance training can enhance muscle preservation. However, BCAAs are less effective than complete protein sources that provide all essential amino acids. If protein intake from food and shakes is adequate, BCAA supplementation offers minimal additional benefit. They are most useful when total protein intake is suboptimal and cannot be immediately corrected through diet.
Omega-3 fatty acids from fish oil have anti-inflammatory properties that may reduce muscle soreness. Doses of 2 to 3 grams of combined EPA and DHA daily have been shown to reduce delayed-onset muscle soreness and improve recovery in some studies. The mechanism involves modulation of prostaglandin and cytokine pathways. Omega-3s also support cardiovascular health and insulin sensitivity, making them a reasonable addition to a tirzepatide regimen for multiple reasons.
Vitamin D supplementation at 2,000 to 4,000 IU daily corrects deficiency, which is associated with muscle pain and weakness. Vitamin D receptors are present in muscle tissue, and vitamin D plays a role in muscle protein synthesis and muscle function. Correcting deficiency can improve muscle strength and reduce pain, though effects are most pronounced in individuals with baseline deficiency. Testing serum 25-hydroxyvitamin D levels before supplementation ensures appropriate dosing.
Coenzyme Q10 (CoQ10) is sometimes recommended for statin-related myopathy, and some users report benefit for muscle pain on tirzepatide. CoQ10 supports mitochondrial function and energy production in muscle cells. Doses of 100 to 200 mg daily are typical. Evidence for efficacy is mixed, with some studies showing benefit and others showing no effect. It is generally safe, making it a reasonable trial for individuals with persistent symptoms despite other interventions.
Curcumin, the active compound in turmeric, has anti-inflammatory and antioxidant properties. Doses of 500 to 1,000 mg daily, particularly in formulations with enhanced bioavailability like curcumin with piperine or liposomal curcumin, may reduce muscle soreness. Studies in athletes show modest reductions in muscle damage markers and perceived soreness after intense exercise. The effect is not dramatic, but curcumin is well-tolerated and offers other health benefits. Combining supplements with peptide therapy requires attention to interactions and cumulative effects.
When muscle pain on tirzepatide is actually a good sign
Not all muscle pain on tirzepatide is a problem. Some types of muscle soreness indicate positive adaptations or beneficial processes. Distinguishing between problematic pain and adaptive pain helps avoid unnecessary concern or premature discontinuation of therapy.
Delayed-onset muscle soreness from increased physical activity is a normal response to unaccustomed exercise. Many tirzepatide users become more active as they lose weight and gain energy. Walking longer distances, starting resistance training, or resuming sports activities that were previously difficult can cause muscle soreness 24 to 48 hours after the activity. This soreness is benign and typically resolves within three to five days. It indicates muscle adaptation and is associated with strength gains over time.
Muscle soreness from stretching and mobility work can also occur as flexibility improves. Tight muscles being lengthened may feel sore initially, but this discomfort diminishes as range of motion increases. The pain is usually mild and localized to the stretched muscle groups. It should not be confused with the more diffuse achiness associated with electrolyte depletion or protein deficiency.
The weight loss joint relief paradox is particularly relevant here. Studies show that tirzepatide users experience reduced joint pain due to decreased mechanical loading on weight-bearing joints. This is especially pronounced in the lower back, hips, and knees. Some users report that while they have mild muscle soreness, their chronic joint pain has improved significantly. This trade-off is favorable, as joint pain from osteoarthritis is often more limiting and harder to manage than transient muscle soreness.
Muscle fatigue during weight loss can also feel like muscle pain. As the body shifts from using primarily glycogen and glucose for energy to utilizing fat stores, there is an adaptation period where muscles may feel less powerful or more easily fatigued. This is not damage, but metabolic retraining. It typically resolves within two to four weeks as fat oxidation capacity improves. Users often describe it as heaviness rather than sharp pain.
Recognizing these adaptive processes prevents unnecessary alarm and supports adherence to both tirzepatide therapy and beneficial lifestyle changes. The key distinction is that adaptive muscle soreness improves over time, does not interfere significantly with daily activities, and is not accompanied by concerning symptoms like dark urine, severe weakness, or systemic signs like fever. Understanding the full spectrum of peptide effects includes recognizing normal adaptation processes.
Tirzepatide versus semaglutide: which causes more muscle pain
Comparing tirzepatide and semaglutide for muscle pain incidence provides context for understanding whether muscle symptoms are specific to tirzepatide or are a class effect of GLP-1 receptor agonists. Both medications suppress appetite, cause gastrointestinal side effects, and lead to rapid weight loss, so the mechanisms underlying muscle pain are similar. However, there are differences in receptor activity and clinical profiles.
Tirzepatide is a dual GIP and GLP-1 receptor agonist, while semaglutide is a selective GLP-1 receptor agonist. The addition of GIP agonism in tirzepatide may influence muscle metabolism differently. GIP receptors are present in adipose tissue and influence fat storage and lipolysis. Whether GIP receptors in muscle tissue contribute to muscle pain is not well studied. Theoretical considerations suggest that dual agonism could produce more pronounced metabolic shifts, potentially increasing the risk of muscle-related symptoms.
Clinical trial data for semaglutide also reports musculoskeletal complaints, but again, not at rates significantly higher than placebo. Postmarketing reports for semaglutide include muscle pain and weakness, similar to tirzepatide. The incidence appears comparable, in the range of 2% to 5%, though direct head-to-head comparisons are lacking. Users switching from semaglutide to tirzepatide sometimes report new muscle pain, while others report improvement. This variability suggests individual factors are more important than the specific medication.
Gastrointestinal side effects are generally more pronounced with semaglutide than tirzepatide, particularly nausea. This could theoretically lead to greater electrolyte depletion and dehydration, increasing muscle pain risk. However, tirzepatide causes more rapid weight loss in clinical trials, which could lead to greater lean mass loss if protein intake is inadequate. These opposing factors may balance out, resulting in similar overall muscle pain incidence.
One study comparing adverse event profiles found that semaglutide users reported slightly higher rates of fatigue, while tirzepatide users reported slightly higher rates of injection site reactions. Muscle pain specifically was not broken out as a separate category, making direct comparison difficult. Anecdotally, clinicians report that muscle pain complaints are common with both medications and are managed similarly. Comparing side effect profiles of GLP-1 medications helps inform medication selection based on individual tolerance.
For users considering switching medications due to muscle pain, it is important to first address modifiable factors like hydration, protein intake, and injection technique. Switching medications without addressing these factors is unlikely to resolve symptoms. If muscle pain persists despite optimization of all modifiable factors, a trial of semaglutide or another GLP-1 agonist may be reasonable. Individual responses vary, and some users do better on one medication than another for reasons that are not fully predictable. Transitioning between GLP-1 medications requires careful dose adjustment and monitoring.
When to call your doctor about tirzepatide muscle pain
Most muscle pain on tirzepatide is mild and resolves with conservative interventions. However, certain features indicate the need for medical evaluation. Recognizing these red flags ensures that serious conditions are identified and treated promptly.
Severe muscle pain that is out of proportion to any activity you have done is concerning. Pain that prevents you from performing normal daily activities, wakes you from sleep, or is unresponsive to over-the-counter pain relievers warrants evaluation. This level of pain can indicate muscle injury, severe inflammation, or rhabdomyolysis. It is not a normal side effect of tirzepatide and should not be dismissed as an expected adjustment.
Muscle weakness, particularly if it is progressive or asymmetric, requires assessment. Difficulty rising from a chair, climbing stairs, or lifting objects that were previously manageable suggests significant muscle dysfunction. Weakness affecting the neck or swallowing muscles is especially concerning and can indicate inflammatory myopathy. Testing for creatine kinase levels, electrolytes, and inflammatory markers is appropriate when weakness is present.
Dark brown or cola-colored urine is a hallmark sign of rhabdomyolysis. Myoglobin released from damaged muscle tissue is filtered by the kidneys and discolors the urine. This is a medical emergency. Immediate evaluation is necessary to prevent acute kidney injury. If you notice dark urine in the context of muscle pain and weakness, seek care urgently. Do not wait to see if it resolves on its own.
Swelling, redness, or warmth in a muscle group can indicate infection, thrombosis, or severe inflammation. These features are not typical of benign muscle soreness and require evaluation to rule out conditions like deep vein thrombosis, cellulitis, or compartment syndrome. Fever accompanying muscle pain raises concern for infection or inflammatory conditions like viral myositis or drug-induced hypersensitivity reactions.
Muscle pain that persists for more than four weeks despite implementing all the preventive strategies discussed earlier should be evaluated. Prolonged symptoms suggest an underlying condition that needs diagnosis and specific treatment. This could include vitamin deficiencies, thyroid dysfunction, autoimmune myopathy, or medication interactions. Blood work including complete blood count, comprehensive metabolic panel. Users of compounded medications should be especially vigilant, creatine kinase, thyroid function tests, vitamin D levels, and inflammatory markers can identify most causes.
If you have pre-existing risk factors for rhabdomyolysis, such as kidney disease, statin use, or a history of muscle disorders, lower thresholds for evaluation are appropriate. Baseline creatine kinase levels before starting tirzepatide and periodic monitoring during therapy can detect early muscle injury. Discuss these considerations with your prescribing physician before initiating therapy. SeekPeptides members have access to detailed symptom tracking tools and clinical decision support to determine when medical evaluation is necessary.
Frequently asked questions
Does tirzepatide cause permanent muscle damage?
No, tirzepatide does not cause permanent muscle damage in the vast majority of users. Muscle pain associated with tirzepatide is typically related to modifiable factors like dehydration, electrolyte imbalance, or inadequate protein intake. When these factors are addressed, muscle pain resolves without lasting effects. The rare cases of rhabdomyolysis documented in the literature resolved after discontinuation of tirzepatide and supportive treatment, with no reports of permanent muscle damage. Muscle loss during weight loss is a concern, but this is related to caloric deficit and protein insufficiency, not direct muscle toxicity from tirzepatide. Strategies to preserve muscle during weight loss can prevent clinically significant muscle loss.
How long does muscle pain from tirzepatide last?
Muscle pain from tirzepatide typically emerges within the first four to eight weeks of therapy and peaks within two to three weeks of onset. Most cases resolve within four weeks if contributing factors are addressed. For pain related to injection site reactions, improvement may occur within days of changing injection sites. Pain related to electrolyte depletion often improves within 48 to 72 hours of rehydration and electrolyte repletion. Muscle pain from protein deficiency and muscle catabolism may take one to two weeks to improve after protein intake is optimized. Persistent pain beyond four weeks warrants medical evaluation to rule out underlying conditions. Understanding the timeline of tirzepatide effects includes both therapeutic effects and side effect resolution.
Can you continue tirzepatide if you have muscle pain?
Yes, most users can continue tirzepatide despite muscle pain by implementing strategies to address the underlying causes. Increasing hydration, optimizing protein intake, supplementing electrolytes, rotating injection sites, and slowing dose escalation resolve muscle pain in the majority of cases without requiring discontinuation. Muscle pain alone is not an indication to stop tirzepatide unless it is severe, accompanied by weakness, associated with dark urine, or unresponsive to interventions. Consulting with your prescribing physician allows for individualized assessment and management. In rare cases where rhabdomyolysis is confirmed, tirzepatide should be discontinued. Adjusting tirzepatide therapy based on tolerance allows continuation with improved comfort.
What supplements help with tirzepatide muscle pain?
Magnesium glycinate at 300 to 400 mg daily is the most effective supplement for muscle pain on tirzepatide, particularly for cramps and spasms. Creatine monohydrate at 5 grams daily supports muscle preservation and reduces muscle protein breakdown. Omega-3 fatty acids at 2 to 3 grams of combined EPA and DHA daily have anti-inflammatory effects that may reduce muscle soreness. Vitamin D supplementation at 2,000 to 4,000 IU daily corrects deficiency and improves muscle function. Branched-chain amino acids can be helpful if protein intake is inadequate, though complete protein sources are more effective. CoQ10 at 100 to 200 mg daily may benefit some users, though evidence is mixed. Combining supplements with peptide therapy requires attention to dosing and interactions.
Is muscle pain worse with higher doses of tirzepatide?
Some users experience worsening muscle pain with dose escalation, particularly when increasing from 7.5 mg to 10 mg or higher. This pattern suggests dose-dependent effects on metabolism or gastrointestinal side effects that lead to greater electrolyte and fluid losses. Not all users experience this pattern, and some have no muscle pain at any dose. For those who do experience dose-related muscle pain, slowing the titration schedule to six or eight weeks per dose level allows more complete adaptation. Smaller dose increments, such as 1.25 mg increases instead of 2.5 mg, can also reduce the severity of symptoms. Alternative dosing strategies for tirzepatide may reduce dose-related side effects.
Does changing injection sites really help with muscle pain?
Yes, for users whose muscle pain is related to injection site reactions, changing injection sites can produce immediate improvement. Case reports document resolution of neck and back pain when injection sites were moved from abdomen to thigh or arm. The mechanism may involve localized inflammation or referred pain patterns. Systematic rotation of injection sites using the abdomen, thighs, upper arms, and buttocks in a structured pattern prevents tissue irritation and ensures consistent medication absorption. If your muscle pain correlates temporally with injections and is worse on the side of your body where you inject, injection site reactions are likely contributing. Optimal injection techniques for GLP-1 medications include rotation strategies.
Can tirzepatide cause muscle loss?
Tirzepatide does not directly cause muscle loss, but rapid weight loss on tirzepatide can include loss of lean body mass if protein intake is inadequate and resistance training is not incorporated. Studies show that 25% to 40% of weight lost on GLP-1 receptor agonists comes from lean mass, which is higher than typical caloric restriction. This is preventable with protein intake of 1.6 to 2.2 grams per kilogram daily and resistance training two to three times per week. Muscle loss is not an inevitable consequence of tirzepatide therapy, but it requires proactive management. Optimizing body composition during weight loss preserves muscle while maximizing fat loss.
Should you stop exercising if you have muscle pain on tirzepatide?
No, you should not stop exercising entirely, but you may need to modify the intensity and type of exercise. Gentle activities like walking, swimming, and light yoga support metabolic health and muscle function without exacerbating pain. Avoiding high-intensity exercise during the first month of therapy reduces the risk of excessive muscle soreness. Once adaptation to tirzepatide is evident, exercise intensity can be gradually increased. If muscle pain is severe, taking one to two days of rest followed by gentle movement is appropriate. Complete inactivity can lead to deconditioning and worsen muscle weakness. Supporting athletic performance during metabolic changes involves smart training progression.
External resources
For additional information on tirzepatide, muscle pain, and metabolic health, the following authoritative resources provide evidence-based guidance.
Case Reports in Endocrinology: Musculoskeletal Pain Associated with Tirzepatide - Detailed case presentations of injection site-related muscle pain with tirzepatide.
Journal of Medical Case Reports: Necrotizing Myopathy and Rhabdomyolysis in a Patient on Tirzepatide - Clinical documentation of rhabdomyolysis associated with tirzepatide use.
SURMOUNT-1 Trial: Tirzepatide for Weight Reduction in Obesity - Primary clinical trial data on tirzepatide efficacy and adverse events.
Journal of Clinical Endocrinology and Metabolism: Body Composition Changes with GLP-1 Receptor Agonists - Analysis of lean mass loss during weight loss on GLP-1 therapy.
American College of Rheumatology: Tirzepatide and Joint Pain Risk - Study showing reduced joint pain risk with tirzepatide compared to other weight loss medications.
American Family Physician: Rhabdomyolysis - Pathophysiology and Management - Comprehensive review of rhabdomyolysis recognition and treatment.
You notice your shoulders ache on Tuesday. By Wednesday, your thighs feel heavy. Thursday brings stiffness in your lower back. You start to wonder if tirzepatide is worth it, if the weight loss is worth this constant low-grade discomfort. But here is what you might not realize. Your potassium is low because you were too nauseous to eat much on Monday. You are drinking half the water you need because your appetite for fluids disappeared along with your appetite for food. You started walking three miles a day instead of one because you finally have energy, and your muscles are adapting to demands they have not faced in years.
The muscle pain is not random. It is not mysterious. It is a series of solvable problems masquerading as a single side effect. Fix the hydration, and half the cramping disappears. Fix the protein, and the weakness starts to lift. Rotate your injection sites, and the shoulder pain that seemed so tied to the medication vanishes like it was never there. This is not to say muscle pain on tirzepatide is insignificant or should be ignored. It is to say that muscle pain on tirzepatide is almost always addressable, almost always reversible, and almost never a reason to abandon therapy that is otherwise working.
At SeekPeptides, members access detailed protocols for managing every documented side effect of GLP-1 therapy, including tirzepatide muscle pain. You learn which symptoms require urgent evaluation and which respond to simple interventions. You get access to tracking tools that correlate symptoms with hydration, protein intake, injection timing, and activity levels, so you can identify patterns that your doctor might miss in a brief office visit. You discover that what seemed like an inevitable consequence of the medication was actually a constellation of fixable problems, each with its own solution. The question is not whether you can manage muscle pain on tirzepatide. The question is whether you have the information and support to do it effectively.
In case I do not see you, good afternoon, good evening, and good night. May your muscles stay pain-free, your electrolytes stay balanced, and your tirzepatide journey stay on track.