Feb 13, 2026

Conflicting information everywhere. One source says Empower Pharmacy still compounds tirzepatide. Another claims compounding ended months ago. A third talks about oral tablets that dissolve on your tongue, and you are not even sure those are real. All you want is a clear, honest breakdown of what Empower Pharmacy actually offers for tirzepatide, whether it is safe, whether it is legal, and whether it is the right choice for your weight management goals.
That frustration makes sense.
The regulatory landscape around compounded tirzepatide has shifted dramatically. The FDA declared the tirzepatide shortage resolved. Eli Lilly filed lawsuits against compounding pharmacies. New rules changed what pharmacies can and cannot produce. And through all of that chaos, Empower Pharmacy adapted its formulations, launched new delivery methods, and continued serving patients under updated legal frameworks. But understanding what they actually offer now, how it differs from brand-name Mounjaro, and what the real risks are requires sorting through layers of marketing, legal jargon, and contradictory forum posts. This guide does that sorting for you. Every claim is examined. Every product is explained. Every risk is addressed. SeekPeptides has analyzed Empower Pharmacy tirzepatide formulations, regulatory filings, customer reviews, and clinical data to give you the most comprehensive resource available on this topic.
What is Empower Pharmacy and why does it matter for tirzepatide
Empower Pharmacy is a Houston-based compounding pharmacy that operates under both 503A and 503B designations. That distinction matters more than most people realize. A 503A compounding pharmacy creates patient-specific medications based on individual prescriptions. A 503B outsourcing facility can produce larger batches under FDA oversight, following current good manufacturing practice (cGMP) standards similar to traditional pharmaceutical manufacturers.
Empower operates one of the largest compounding operations in the United States. They compound hundreds of different medications across categories including hormone replacement therapy, weight management, longevity, and sexual health. Their tirzepatide products represent just one piece of a much larger catalog.
For the weight management community, Empower became significant because they offered compounded tirzepatide at prices substantially lower than brand-name Mounjaro. During the tirzepatide shortage that began in late 2022, compounding pharmacies like Empower were legally permitted to produce copies of tirzepatide under Section 503A of the Federal Food, Drug, and Cosmetic Act. That shortage exception allowed millions of patients to access tirzepatide who otherwise could not afford the brand-name version.
But things changed.
The FDA declared the tirzepatide shortage resolved in October 2024, and new rules took effect that fundamentally altered what compounding pharmacies can produce. Understanding those changes is essential before ordering anything from Empower or any other compounder.
Empower Pharmacy tirzepatide products explained
Empower currently offers two distinct tirzepatide formulations. Each serves a different purpose and comes with its own set of considerations. Neither is identical to brand-name Mounjaro, and that difference is both intentional and legally necessary.
Tirzepatide/niacinamide injection
The primary tirzepatide product from Empower is a tirzepatide/niacinamide injection. This compounded formulation combines tirzepatide with niacinamide, a form of vitamin B3. The addition of niacinamide is not random. It serves multiple purposes that we will examine in detail below.
Three concentration options are available.
The first is 17 mg/mL tirzepatide with 2 mg/mL niacinamide in a 4 mL vial. This higher-concentration option provides the most medication per vial and works well for patients on higher tirzepatide doses. A single 4 mL vial at 17 mg/mL contains 68 mg total tirzepatide, enough for several weeks of treatment depending on your prescribed dose.
The second option is 8 mg/mL tirzepatide with 2 mg/mL niacinamide in a 2.5 mL vial. This lower concentration is ideal for patients starting treatment or those on smaller maintenance doses. The 2.5 mL vial at 8 mg/mL contains 20 mg total tirzepatide. For someone on a 2.5 mg weekly dose, that provides roughly eight weeks of treatment from a single vial.
The third is 17 mg/mL tirzepatide with 2 mg/mL niacinamide in a 2 mL vial. This smaller high-concentration option contains 34 mg total, offering a middle ground between the other two configurations.
All three require subcutaneous injection once weekly. Recommended injection sites include the abdomen, thigh, or upper arm. Site rotation between injections helps prevent localized reactions and ensures consistent absorption.
Tirzepatide ODT (orally disintegrating tablet)
In a more recent development, Empower launched tirzepatide ODT. These are compounded oral disintegrating tablets that dissolve on the tongue without water. For people who dread needles, this sounds like a dream come true.
Three strengths are available: 3 mg, 4 mg, and 5 mg.
The tablet dissolves within 30 to 60 seconds when placed on the tongue. You do not need water. You should not swallow excess saliva during dissolution. The tablet can be taken with or without food at any time of day, though consistency in timing helps maintain stable blood levels.
Here is the critical caveat. Tirzepatide ODT has not undergone clinical trials. No published studies demonstrate that orally administered tirzepatide achieves the same bioavailability, efficacy, or safety profile as injectable tirzepatide. The oral route faces significant challenges including peptide degradation in the gastrointestinal tract, variable absorption rates, and potential differences in how the body processes the medication compared to subcutaneous delivery.
Shipping restrictions also apply. Empower cannot ship tirzepatide ODTs to California, Wisconsin, Iowa, or Minnesota due to state-specific regulations.
How these differ from brand-name Mounjaro
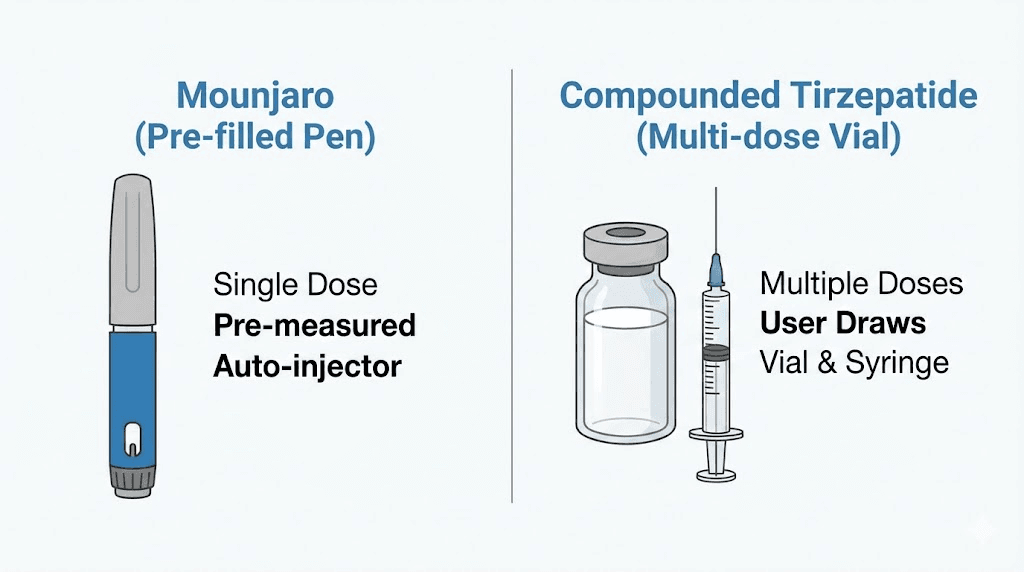
Brand-name Mounjaro (manufactured by Eli Lilly) contains only tirzepatide. No additional ingredients beyond standard pharmaceutical excipients. It comes in pre-filled injection pens at fixed doses: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg.
Empower formulations differ in three fundamental ways.
First, they contain niacinamide (in the injection) or use an entirely different delivery method (the ODT). Second, they come in multi-dose vials rather than pre-filled pens, requiring you to draw up each dose with a syringe and needle. Third, and most importantly, they are compounded medications. That means the FDA has not reviewed them for safety or efficacy. No clinical trials have been conducted on these specific formulations. The manufacturing standards, while regulated, follow different pathways than FDA-approved pharmaceutical production.
That does not automatically make them dangerous. But it does mean the evidence base is different, and you should understand that difference before making decisions about your treatment.
Why niacinamide is added to Empower tirzepatide
The niacinamide question comes up constantly. Why add a B vitamin to a GLP-1 receptor agonist? Is it for health benefits, stability, or just a legal workaround? The answer involves all three, though the proportions depend on who you ask.
Stability and formulation benefits
Niacinamide helps maintain the stability of the tirzepatide molecule in solution. Peptides are fragile. They degrade when exposed to heat, light, agitation, and time. Adding niacinamide to the formulation creates a more stable environment that extends shelf life and reduces the risk of degradation before the medication reaches you.
This is not unique to Empower. Many compounded tirzepatide formulations across different pharmacies include niacinamide or other stabilizing agents for exactly this reason. The concentration of 2 mg/mL niacinamide is relatively low, well within safe supplementation ranges when administered subcutaneously.
Potential metabolic benefits
Niacinamide, also known as nicotinamide, is the amide form of vitamin B3. It plays important roles in cellular energy production through NAD+ metabolism. At the doses present in Empower formulations, niacinamide provides modest supplementation that may support mitochondrial function and energy metabolism.
Some practitioners believe the addition of niacinamide complements tirzepatide action in several ways. It may support fat metabolism by assisting with the conversion of nutrients to cellular energy. It has antioxidant properties that could reduce inflammation associated with metabolic disorders. And some evidence suggests it may help mitigate certain gastrointestinal side effects that tirzepatide commonly causes, though this claim lacks robust clinical evidence specific to this combination.
The honest assessment? The metabolic benefits of niacinamide at these doses are real but modest. You would not take this formulation specifically for the niacinamide. It is a helpful addition, not a primary selling point.
The legal angle
After the FDA declared the tirzepatide shortage resolved, compounding pharmacies could no longer produce "essentially a copy" of commercially available tirzepatide. Adding niacinamide creates a formulation that is "clinically different" from brand-name Mounjaro. This difference allows compounding under Section 503A to potentially continue, provided the prescribing physician documents a specific clinical reason for the compounded version over the commercial product.
This is the elephant in the room. The niacinamide addition, while providing genuine benefits, also serves a regulatory purpose. Without it, Empower could not legally compound tirzepatide injections now that the shortage has been resolved. The same logic applies to other compounders who add B12, glycine, or other ingredients to their tirzepatide formulations.
Whether this regulatory strategy will survive ongoing legal challenges remains uncertain. Eli Lilly has filed lawsuits specifically targeting compounding pharmacies that it believes are producing impermissible copies of its products. The legal landscape continues to evolve.
Regulatory status and legal considerations
Understanding the regulatory framework around compounded tirzepatide requires knowing a few key dates and decisions. This section is dense but essential. Getting this wrong could mean using a product that is not legally compliant or, worse, not properly manufactured.
The shortage timeline
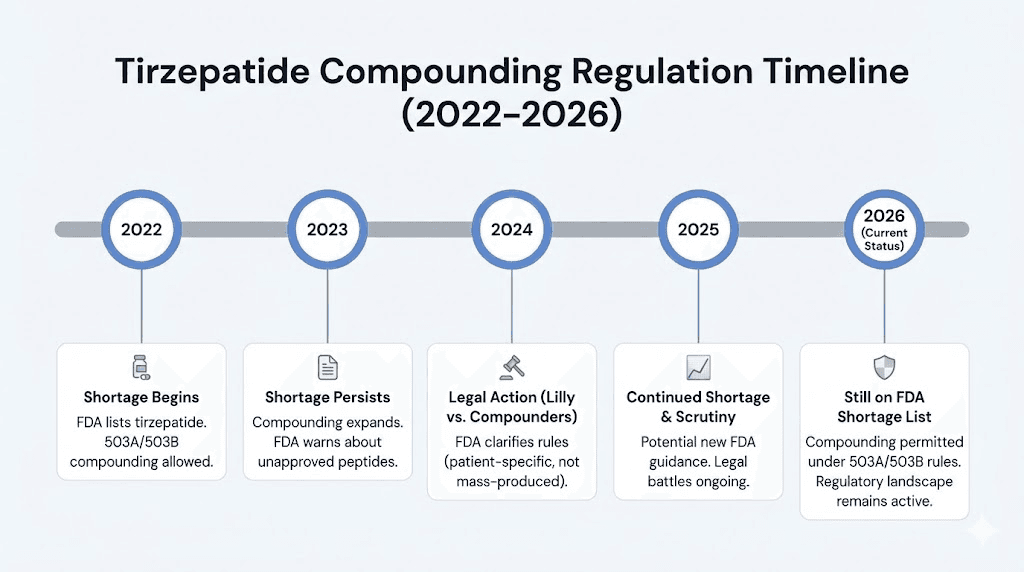
Tirzepatide (Mounjaro) first experienced shortage conditions in late 2022 as demand exploded following clinical trial results showing remarkable weight loss outcomes. The SURMOUNT trials demonstrated average weight loss of 20 to 25 percent of body weight, numbers that exceeded anything previously seen with pharmaceutical interventions.
During the shortage, Section 503A of the FD&C Act permitted compounding pharmacies to produce copies of the drug. This gave millions of patients access to tirzepatide at significantly lower costs. Compounders thrived. Telehealth platforms partnered with compounding pharmacies to create streamlined access. The market exploded.
Then, in October 2024, the FDA declared the tirzepatide shortage resolved.
What "shortage resolved" means for compounding
Once a drug is no longer in shortage, the exception that allowed compounding pharmacies to produce copies evaporates. The transition periods were specific. State-licensed pharmacies operating under Section 503A had until February 18, 2025, to wind down production. Outsourcing facilities operating under Section 503B had until March 19, 2025.
After those dates, compounding pharmacies can only produce tirzepatide formulations that are "not essentially a copy" of the commercially available product. This means the compounded version must differ in a clinically meaningful way, such as containing additional active ingredients (like niacinamide), using a different delivery route (like oral formulations), or addressing a specific documented patient need that the commercial product does not meet.
The Eli Lilly lawsuits
Eli Lilly has been aggressive in protecting its tirzepatide market. In April 2025, the pharmaceutical giant sued Empower Pharmacy specifically, alleging that the company was marketing unapproved tirzepatide compounds using misleading claims. The lawsuit targets what Lilly considers unauthorized manufacturing and deceptive marketing practices.
This legal battle is not just about Empower. It affects the entire compounding industry and every patient who relies on compounded GLP-1 medications. The outcome will likely set precedents for how compounding pharmacies operate in the post-shortage era for all GLP-1 receptor agonists.
Current legal status
As of early 2026, the situation remains fluid. Courts have generally upheld the FDA position that the tirzepatide shortage is resolved. In May 2025, a district court ruled that the FDA acted within its statutory authority in making that determination. However, compounding pharmacies continue to operate under the argument that their modified formulations (tirzepatide with niacinamide, tirzepatide with B12, tirzepatide with glycine) represent clinically different products, not copies.
The legal gray area is real. If your prescriber writes a prescription for compounded tirzepatide/niacinamide and documents a clinical justification, Empower can fill it. Whether that justification will hold up under increasing regulatory scrutiny is the billion-dollar question the entire industry is watching.
How to get Empower Pharmacy tirzepatide
Getting tirzepatide from Empower is not as simple as placing an online order. The process involves several steps, and understanding each one helps you avoid delays, denials, and wasted money.
Prescription requirement
You need a valid prescription from a licensed healthcare provider. This is non-negotiable. Empower operates as a legitimate 503A compounding pharmacy, and every medication they dispense requires a patient-specific prescription based on an established patient-practitioner relationship.
Your prescriber must document the clinical justification for using a compounded tirzepatide formulation instead of brand-name Mounjaro. Common justifications include allergy or sensitivity to an inactive ingredient in the commercial product, need for a specific dose not available commercially, or documented medical need for the additional niacinamide component.
Working with telehealth providers
Many patients access Empower tirzepatide through telehealth platforms that have established relationships with the pharmacy. These platforms typically offer online consultations with licensed prescribers who evaluate your health history, order necessary labs, and write prescriptions if appropriate.
The process usually looks like this. You complete an online health questionnaire. A licensed provider reviews your information and conducts a video or phone consultation. If appropriate, they write a prescription to Empower. The pharmacy compounds your medication and ships it directly to you.
Turnaround times vary. Some patients report receiving their medication within a week. Others describe delays of two to three weeks, particularly during high-demand periods.
Direct prescriber route
If you already have a physician, endocrinologist, or weight management specialist, they can write a prescription to Empower directly. Many clinics and medical spas have established accounts with the pharmacy, streamlining the ordering process.
This route often results in faster fulfillment because the prescriber relationship with Empower is already established. Your doctor handles the clinical justification documentation, and the pharmacy fills the prescription according to the specifications.
Ordering and shipping
Empower ships nationwide through their patient portal system. Orders require login credentials tied to your prescription. They offer overnight and two-day shipping options, with medications packed in temperature-controlled packaging to maintain stability during transit.
A few practical considerations matter here. The injection requires refrigeration upon arrival. Store between 36 and 46 degrees Fahrenheit, protected from light, and never freeze. The ODT tablets store at room temperature (68 to 77 degrees Fahrenheit) in their original packaging to prevent moisture exposure.
If you are wondering about how long tirzepatide lasts in the fridge or how long it can be out of the fridge, those guides cover the specific stability timelines for compounded formulations.
Empower tirzepatide dosing guide
Dosing compounded tirzepatide from a multi-dose vial is fundamentally different from using a pre-filled Mounjaro pen. You need to understand concentrations, calculate volumes, and draw up precise amounts with an insulin syringe. Getting this right is not optional. Underdosing wastes your time and money. Overdosing creates unnecessary risk.
Understanding concentrations
Empower offers two concentrations for the injection: 8 mg/mL and 17 mg/mL. The concentration tells you how many milligrams of tirzepatide are dissolved in each milliliter of solution.
For the 8 mg/mL concentration, each 0.1 mL (or 10 units on an insulin syringe) contains 0.8 mg of tirzepatide. To get a 2.5 mg dose, you would draw up approximately 0.31 mL, or about 31 units on a standard U-100 insulin syringe. For a 5 mg dose, you need roughly 0.63 mL, or 63 units.
For the 17 mg/mL concentration, each 0.1 mL contains 1.7 mg. A 2.5 mg dose requires approximately 0.15 mL (15 units). A 5 mg dose needs about 0.29 mL (29 units). A 10 mg dose requires roughly 0.59 mL (59 units).
The Empower tirzepatide dosage chart provides exact unit calculations for every standard dose at both concentrations. If you prefer a more detailed breakdown, the Empower tirzepatide dosing chart walks through the math step by step.
Standard titration protocol
The FDA-approved titration schedule for brand-name Mounjaro serves as the reference point, though your prescriber may customize it. The standard approach starts at 2.5 mg weekly for four weeks, then increases to 5 mg weekly. After that, increases of 2.5 mg occur no more frequently than every four weeks, up to a maximum of 15 mg weekly.
With compounded tirzepatide, some prescribers modify this protocol. Microdosing approaches start even lower, sometimes at 1 mg or 1.5 mg, to minimize gastrointestinal side effects during the initial adjustment period. Others titrate more slowly, spending six to eight weeks at each dose level rather than four.
The multi-dose vial format actually offers an advantage here. Unlike pre-filled pens locked to specific doses, you can draw up any amount within the vial concentration, allowing truly personalized dose adjustments that would be impossible with commercial products.
Dose calculation quick reference
For the 17 mg/mL concentration (most common Empower formulation):
Desired dose | Volume to draw | Units on U-100 syringe |
|---|---|---|
2.5 mg | 0.147 mL | ~15 units |
5 mg | 0.294 mL | ~29 units |
7.5 mg | 0.441 mL | ~44 units |
10 mg | 0.588 mL | ~59 units |
12.5 mg | 0.735 mL | ~74 units |
15 mg | 0.882 mL | ~88 units |
For precise calculations, the SeekPeptides peptide calculator handles the math automatically. Enter your vial concentration and desired dose, and it provides the exact volume and unit count.
Injection technique
Proper injection technique affects both comfort and absorption. Clean the injection site with an alcohol swab. Pinch a fold of skin at the chosen site. Insert the needle at a 45 to 90 degree angle depending on body composition. Inject slowly. Hold for 5 to 10 seconds before withdrawing. Do not rub the site afterward.
Rotate between at least three injection sites: left abdomen, right abdomen, and alternating thighs or upper arms. Keeping a simple log of which site you used each week prevents accidentally using the same spot too frequently. Repeated injections in the same location can cause lipodystrophy, where the fatty tissue under the skin changes texture and may affect absorption.
Side effects and safety considerations
Every medication has side effects. Tirzepatide is no exception, and compounded versions may carry additional considerations compared to the FDA-approved product.
Common side effects
Gastrointestinal symptoms dominate the side effect profile. This is true for brand-name Mounjaro, compounded tirzepatide from Empower, and every other source. The mechanism is inherent to how tirzepatide works. It slows gastric emptying and affects appetite signaling, which unavoidably impacts the digestive system.
Nausea affects 20 to 33 percent of patients, most commonly during the first few weeks and after dose increases. Headaches occur in roughly 10 to 15 percent of users, typically mild and transient. Diarrhea affects 18 to 23 percent, while constipation impacts a similar proportion. Some patients experience both at different times during treatment. Vomiting occurs in 5 to 12 percent of cases, more commonly at higher doses.
Dietary modifications significantly reduce gastrointestinal symptoms. Eating smaller meals, avoiding high-fat foods, staying hydrated, and following a tirzepatide-specific diet plan helps most patients manage these effects without needing to reduce their dose.
Fatigue and energy
Fatigue is a commonly reported side effect that does not always appear in clinical trial summaries but shows up consistently in patient reports. The caloric restriction that tirzepatide facilitates means your body is receiving less energy from food. Combined with the metabolic changes triggered by dual GIP and GLP-1 receptor activation, tiredness during the first several weeks is expected.
For most patients, energy levels stabilize within four to six weeks as the body adapts. Ensuring adequate protein intake, staying hydrated, and getting sufficient sleep all help. If GLP-1 related fatigue persists beyond two months, discuss dose adjustment with your prescriber.
Anxiety and mood changes
A smaller but significant subset of patients reports mood changes, including increased anxiety, on tirzepatide. The rapid metabolic shifts, reduced caloric intake, and hormonal changes associated with significant weight loss can all contribute to mood fluctuations. These typically improve as the body adapts but should be monitored and discussed with your healthcare provider.
Additional considerations with compounded formulations
Compounded medications carry risks that FDA-approved products do not. The manufacturing environment is different. Quality control processes, while regulated, follow different standards. Potency verification, sterility testing, and stability data may be less extensive than what the FDA requires for approved drugs.
Empower Pharmacy specifically has faced regulatory scrutiny that bears mentioning. The FDA has issued multiple warning letters to Empower over the past several years, documenting concerns about sterile compounding practices, contamination control, and quality assurance procedures. In April 2025, the FDA issued a warning letter documenting positive microbial growth detected during environmental monitoring in a sterile production area. A November 2025 inspection found contamination in a drug-preparation area tied to a batch of testosterone that was subsequently pulled from the market.
These findings do not mean every product Empower produces is unsafe. Many compounding pharmacies receive warning letters and make corrections. But it does mean you should be aware of the company history when making informed decisions about your care. Transparency about these issues matters more than pretending they do not exist.
Contraindications
Tirzepatide, whether compounded or brand-name, should not be used by individuals with a personal or family history of medullary thyroid carcinoma, Multiple Endocrine Neoplasia syndrome type 2, severe gastroparesis or gastrointestinal disease, or during pregnancy. Known hypersensitivity to tirzepatide or any formulation component (including niacinamide in the compounded version) also contraindicates use.
Drug interactions require attention as well. Tirzepatide affects gastric emptying, which can alter the absorption of oral medications including insulin, sulfonylureas, oral contraceptives, and narrow therapeutic index drugs like warfarin. If you take any of these, your prescriber needs to adjust monitoring or timing accordingly.
Empower tirzepatide customer reviews and experiences
Real user feedback tells a more nuanced story than marketing materials ever could. We analyzed reviews across multiple platforms to give you an honest picture of what patients actually experience with Empower tirzepatide products.
Positive experiences
Patients who report positive outcomes typically highlight several themes. The medication works as expected for appetite suppression and weight loss. Shipping is generally reliable with proper cold-chain packaging. The multi-dose vial format allows precise dose customization that pre-filled pens cannot match. And for patients who previously used brand-name Mounjaro but found the cost prohibitive, switching to Empower provided continued access at a manageable price point.
One recurring positive note involves the niacinamide addition. Some patients report that the compounded formulation seems gentler on their stomach compared to their experience with brand-name tirzepatide, though whether this is due to the niacinamide, different excipients, or placebo effect remains unclear.
Negative experiences
The negative reviews cluster around several consistent themes. Customer service responsiveness appears to be a significant pain point. Multiple reviewers describe difficulty reaching the pharmacy by phone, long wait times for prescription processing, and inadequate communication about order status or delays. Trustpilot shows an average rating of 1.9 stars. Yelp reviews average 2.1 stars. These numbers are concerning, though they should be weighted against the reality that dissatisfied customers disproportionately leave reviews.
Some patients report inconsistent effectiveness, suggesting potential variability in compounded product potency. Others describe gastrointestinal problems that seem worse than what they experienced with the brand-name product. Whether this reflects formulation differences, batch variability, or individual sensitivity is difficult to determine without controlled comparison studies.
Shipping delays have been a recurring complaint, particularly during periods of high demand. Temperature control during shipping has also been questioned by some patients, though Empower uses insulated packaging with cold packs for temperature-sensitive medications.
How to interpret reviews
A few important context points. Compounding pharmacies in the GLP-1 space serve an enormous volume of patients, many of whom have strong opinions about cost, access, and regulation. Review platforms tend to amplify extreme experiences, both positive and negative. The medication itself causes side effects in most users regardless of source, so some negative experiences may reflect tirzepatide side effects rather than issues specific to Empower formulations.
The most valuable reviews are those that describe specific, verifiable details: batch numbers, potency concerns backed by lab testing, or documented customer service interactions. General complaints about "it did not work" or "I got nauseous" are less informative because those outcomes occur with brand-name products too.
Empower tirzepatide ODT: the oral option
The tirzepatide ODT deserves its own section because it represents such a fundamentally different approach to GLP-1 delivery. Oral peptide delivery has been one of the holy grails of pharmaceutical development for decades, and Empower entered this space with a compounded product that raises both excitement and legitimate questions.
How the ODT works
The oral disintegrating tablet bypasses the traditional oral route. Instead of being swallowed and absorbed through the gastrointestinal tract (where most peptides get destroyed by digestive enzymes), the tablet dissolves on the tongue and is absorbed through the oral mucosa. This sublingual-like absorption route theoretically allows the peptide to enter the bloodstream more directly, avoiding first-pass metabolism in the liver and enzymatic degradation in the stomach.
At least, that is the theory. Whether this actually achieves clinically meaningful blood levels of tirzepatide comparable to subcutaneous injection has not been demonstrated in published clinical trials. The technology is plausible. Oral semaglutide (Rybelsus) proves that oral GLP-1 delivery is possible with the right formulation. But Rybelsus went through extensive clinical trials to prove its efficacy and required a specialized absorption enhancer (SNAC) to achieve adequate bioavailability.
Empower tirzepatide ODT has not undergone similar validation.
Who might benefit
Despite the uncertainties, certain patients may find the ODT appealing. Those with genuine needle phobia, which affects an estimated 20 to 25 percent of adults to some degree, may find the oral route more sustainable for long-term treatment. Patients with dexterity issues that make injection difficult could benefit from a simpler administration method. And travelers who find injection supplies cumbersome may prefer the portability of tablets.
The question is not whether these benefits exist. They do. The question is whether the ODT delivers enough active tirzepatide to produce the metabolic effects you are paying for. Without clinical trial data, that question remains unanswered.
Practical considerations
Storage differs from the injection. Keep ODT tablets at room temperature (68 to 77 degrees Fahrenheit) in their original packaging. Moisture is the primary enemy of disintegrating tablets, so do not transfer them to pill organizers or other containers. Protect from light and extreme temperatures. These storage requirements are simpler than the refrigeration needed for injectables, which is a practical advantage.
The available doses (3 mg, 4 mg, and 5 mg) represent a relatively narrow range compared to injectable tirzepatide doses of 2.5 mg to 15 mg. This limits titration options and may not provide sufficient dose for patients who require higher amounts. If the ODT works for you at 5 mg but you would benefit from 7.5 mg or more, you have no oral option for escalation and would need to switch to injection.
Empower tirzepatide versus other compounding pharmacies
Empower is far from the only pharmacy compounding tirzepatide formulations. Understanding how they compare helps you make an informed choice. Each compounder has its own formulations, pricing structures, reputation, and regulatory history.
Empower versus Olympia Pharmacy
Olympia Pharmacy is another major player in the compounded GLP-1 space. They offer tirzepatide formulations with their own modifications. The Olympia tirzepatide dosage chart shows their specific concentrations and recommended dosing protocols. Key differences include formulation composition, available concentrations, pricing structure, and customer service reputation. Both pharmacies have large operations, both have faced regulatory scrutiny, and both serve large patient populations.
Empower versus direct pharmacy options
Direct-to-consumer pharmacy models have emerged as alternatives to traditional compounding relationships. These platforms often bundle telehealth consultations, prescriptions, and medication into a single subscription. The convenience factor is high, but the clinical oversight may differ from working with an independent physician who prescribes to a specific compounding pharmacy.
Comparison table
Factor | Empower Pharmacy | Brand-name Mounjaro | Other compounders |
|---|---|---|---|
FDA approved | No (compounded) | Yes | No (compounded) |
Clinical trials | No | Yes (SURMOUNT 1-4) | No |
Formulation | Tirzepatide/niacinamide | Tirzepatide only | Varies (B12, glycine, etc.) |
Delivery options | Injection + ODT | Pre-filled pen injection | Mostly injection |
Dose flexibility | Any amount from vial | Fixed doses only | Any amount from vial |
Prescription required | Yes | Yes | Yes |
Cost | Lower than brand | Highest | Varies |
Nationwide shipping | Yes (some restrictions) | Via pharmacy | Varies by pharmacy |
Storage and handling for Empower tirzepatide
Proper storage is not a minor detail. Tirzepatide needs refrigeration, and compounded formulations may have different stability profiles than the brand-name product. Here is exactly what you need to know.
Injection storage requirements
Store the tirzepatide/niacinamide injection between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). This means the main body of your refrigerator, not the door (which experiences more temperature fluctuation) and definitely not the freezer. Freezing destroys the peptide structure and renders the medication useless.
Protect from light exposure. The vial should stay in its original packaging or a secondary container that blocks light when not in use. UV radiation accelerates peptide degradation, reducing potency over time.
Before each use, visually inspect the vial for particulate matter, discoloration, or cloudiness. Clear, colorless to slightly yellow solution is normal. Any visible particles, unusual color, or turbidity means the vial should not be used.
If the vial has been at room temperature, stability timelines for compounded tirzepatide outside the fridge vary depending on the specific formulation and ambient temperature. Generally, brief excursions to room temperature for dose preparation are fine. Extended periods above 46 degrees Fahrenheit reduce stability and should be avoided.
ODT storage requirements
The oral disintegrating tablets have simpler storage needs. Room temperature between 68 and 77 degrees Fahrenheit (20 to 25 degrees Celsius). Keep them in the original packaging because moisture is their primary degradation pathway. Do not expose to direct sunlight or extreme heat. Do not place in bathroom medicine cabinets where steam and humidity fluctuate.
Shipping and transit
When your order arrives, inspect the packaging immediately. The injectable should arrive cold, with ice packs still partially frozen or at least cold to the touch. If the package arrives warm or the ice packs are completely thawed and warm, contact Empower before using the medication. Potency may be compromised.
For ODT shipments, verify the packaging is intact and the tablets have not been exposed to moisture. Damaged blister packs or visible moisture inside the packaging warrants a replacement request.
Switching to or from Empower tirzepatide
Many patients find themselves switching between tirzepatide sources as availability, pricing, and regulations change. Whether you are moving from brand-name Mounjaro to Empower, from Empower to another compounder, or from tirzepatide to semaglutide, the transition requires careful planning.
From Mounjaro to Empower
If you have been using brand-name Mounjaro and are switching to Empower compounded tirzepatide, keep your dose the same. The active ingredient is the same molecule. Calculate your dose volume based on the Empower vial concentration, verify with your prescriber, and continue your weekly schedule without interruption.
Be aware that some patients report subjective differences in how the compounded version feels compared to the brand-name product. Whether this reflects genuine pharmacological differences, nocebo effects, or batch variation is unclear. If you notice significant changes in appetite suppression, gastrointestinal effects, or weight loss trajectory after switching, document them and discuss with your provider.
From another compounder to Empower
Switching between compounding pharmacies requires attention to concentration differences. If your previous pharmacy used a 10 mg/mL concentration and Empower uses 17 mg/mL, your injection volume will be significantly different for the same dose. Always recalculate before your first injection from a new vial. Use the compound tirzepatide dosage chart as reference.
From Empower injection to Empower ODT
Switching from injection to the oral disintegrating tablet is more complex because bioavailability likely differs between the two routes. A 5 mg injected dose does not necessarily produce the same blood levels as a 5 mg oral dose. Your prescriber should manage this transition with appropriate monitoring, potentially starting at a higher oral dose or expecting some adjustment period.
Dose equivalence considerations
The tirzepatide conversion chart helps when switching between different concentrations or formulations. For those coming from semaglutide, the semaglutide to tirzepatide conversion guide provides approximate equivalence ratios, though individual responses vary significantly.
Weight loss expectations with Empower tirzepatide
Setting realistic expectations prevents both disappointment and unnecessary alarm. Weight loss on tirzepatide, from any source, follows predictable patterns that help you gauge whether your protocol is working.
Typical timeline
During the first two to four weeks on the starting dose of 2.5 mg, most patients lose 2 to 5 pounds. Some lose more, some less. The initial weight loss often includes water weight as tirzepatide reduces food intake and begins altering metabolic function. How fast tirzepatide works depends on individual factors including starting weight, metabolic rate, adherence to dietary recommendations, and activity level.
Significant results typically emerge between weeks 8 and 16, after dose titration reaches the 5 to 10 mg range. By this point, appetite suppression is well established, and the body has adjusted to the metabolic changes. Monthly weight loss of 5 to 10 pounds is common during this active loss phase.
Peak weight loss occurs over 12 to 18 months of treatment. The SURMOUNT clinical trials showed average weight loss of approximately 22.5 percent of body weight at 72 weeks on the 15 mg dose. Individual results with compounded formulations should approximate these outcomes if the active ingredient is equivalent, though individual variation is significant.
Plateau management
If you reach a point where weight loss stalls, several strategies can help. Dose escalation within your prescriber guidelines is the most common intervention. Dietary optimization, particularly increasing protein intake and managing macronutrient balance, supports continued progress. Adding structured physical activity accelerates fat loss while preserving muscle mass. And reviewing your protocol with your provider can identify factors you might be overlooking.
Some patients find they respond better to one tirzepatide source than another. If you have tried Empower and your results have plateaued despite dose optimization and lifestyle modifications, discussing a switch to brand-name Mounjaro or another compounder with your prescriber is reasonable.
Before and after expectations
Real tirzepatide before and after results vary enormously. Factors that predict better outcomes include higher starting BMI, adherence to dietary recommendations, regular physical activity, proper dose titration, and consistent weekly administration. Factors that limit results include inadequate protein intake, metabolic adaptation, medication non-compliance, and unrealistic timelines.
For those curious about gender-specific outcomes, male-specific tirzepatide results tend to show faster initial weight loss due to higher baseline metabolic rates, while long-term outcomes are comparable across genders.
Combining Empower tirzepatide with other treatments
Many patients wonder about adding other medications or supplements to their tirzepatide protocol. While your prescriber should guide these decisions, understanding the options helps you have more productive conversations with your healthcare team.
Tirzepatide and phentermine
Combining GLP-1 agonists with phentermine is a topic that generates significant interest. Phentermine is not a GLP-1. It works through a completely different mechanism, stimulating norepinephrine release to suppress appetite. Some prescribers combine the two for enhanced appetite suppression, particularly during weight loss plateaus. Others advise against it due to limited safety data on the combination.
Supportive supplements
Supplements that support tirzepatide treatment focus on addressing nutritional gaps that reduced food intake creates. Protein supplementation helps preserve lean muscle mass. B-complex vitamins support energy metabolism. Fiber supplements can help with constipation that tirzepatide sometimes causes. Electrolytes prevent dehydration-related issues as fluid intake often decreases alongside food intake.
Alcohol considerations
Alcohol use during tirzepatide treatment requires caution. Tirzepatide slows gastric emptying, which can intensify and prolong the effects of alcohol. Many patients report lower alcohol tolerance and more severe hangovers. The caloric content of alcohol also works against weight loss goals. While moderate consumption is not absolutely contraindicated, most prescribers recommend significant reduction or abstinence during active weight loss phases.
Empower tirzepatide and meal planning
Medication alone does not produce optimal results. What you eat while on tirzepatide matters enormously, and many patients underestimate how dramatically the medication changes their relationship with food.
Tirzepatide reduces appetite and slows digestion. That means every calorie you consume carries more weight (no pun intended) in your nutritional profile. When you can only eat 1,200 to 1,500 calories without feeling uncomfortably full, those calories need to be nutritionally dense. A tirzepatide-specific meal plan prioritizes lean protein sources, complex carbohydrates, healthy fats, and micronutrient-rich vegetables.
Protein deserves particular attention. Weight loss on tirzepatide includes both fat mass and lean mass. Inadequate protein accelerates muscle loss, which lowers metabolic rate and makes long-term weight maintenance harder. Target at least 100 grams of protein daily, ideally split across three to four meals. Protein shakes can help when solid food feels difficult to manage, especially in the first few days after a dose increase.
Hydration is the other pillar that patients consistently neglect. Reduced food intake means less water from food sources. Tirzepatide can also cause mild dehydration through its effects on gastric function. Aim for at least 64 ounces of water daily, more if you exercise or live in a warm climate. Dehydration worsens nearly every tirzepatide side effect, from constipation to headaches to fatigue.
Timing your meals around your injection also helps manage side effects. Many patients find that eating a moderate, protein-rich meal an hour before their weekly injection helps buffer gastrointestinal effects. Avoiding heavy, high-fat meals for 24 to 48 hours after injection minimizes nausea. Timing considerations for GLP-1 medications apply broadly across formulations, though individual patterns vary.
Reconstitution and preparation
Unlike pre-filled Mounjaro pens, Empower multi-dose vials require you to draw up each dose manually. For patients new to injectable medications, this process can feel intimidating. It is actually straightforward once you understand the steps.
The complete tirzepatide reconstitution guide covers every step in detail. Note that Empower tirzepatide/niacinamide injections arrive ready to use, not lyophilized. You do not need to add bacteriostatic water. The vial contains liquid solution at the specified concentration. Simply clean the vial top with an alcohol swab, insert your insulin syringe, draw up the calculated volume, and inject.
Syringe selection matters. U-100 insulin syringes with fine-gauge needles (29 to 31 gauge) minimize injection discomfort. For doses requiring larger volumes (above 0.5 mL), some patients prefer using 1 mL syringes with slightly larger needles for easier aspiration. Your prescriber or pharmacist can recommend the appropriate syringe based on your specific dose and injection preferences.
One common mistake: reusing needles. Every injection should use a fresh, sterile needle. Reusing needles increases infection risk, causes more pain (needle tips deform after a single use), and can introduce contaminants into the multi-dose vial. A box of 100 insulin syringes costs a few dollars. That is not the place to cut corners when dealing with a compounded injectable medication.
For patients managing multiple peptide protocols, keeping your supplies organized prevents mix-ups. Label each vial clearly. Store different medications in separate designated areas of your refrigerator. Keep a written or digital log of dose dates, amounts, and injection sites.
Cost analysis and value assessment
Empower Pharmacy does not publicly list tirzepatide pricing on their website, and specific costs vary based on formulation, concentration, and vial size. However, compounded tirzepatide generally costs significantly less than brand-name Mounjaro, which lists at approximately $1,000 to $1,200 per month without insurance coverage.
Compounded tirzepatide from pharmacies like Empower typically ranges from $200 to $500 per month depending on dose, concentration, and the specific pharmacy pricing structure. The exact cost depends on your prescribed formulation and your provider relationship.
Several factors affect the value calculation beyond sticker price. Multi-dose vials waste less medication than pre-filled pens when patients are titrating or using non-standard doses. Lower cost enables longer treatment duration, which matters because tirzepatide works best with sustained use over 12 to 18 months. And for patients without insurance coverage for Mounjaro, compounded alternatives may be the only financially viable option.
However, lower cost should not come at the expense of safety or efficacy. If a compounded product is significantly cheaper but delivers inconsistent potency, the savings evaporate alongside the results. The cheapest option is not always the best value. Understanding what drives pricing differences helps you distinguish genuine value from false economy.
How Empower compares to brand-name Mounjaro: the complete picture
This comparison deserves nuance that most online discussions lack. Brand-name Mounjaro offers FDA approval, clinical trial validation, consistent manufacturing standards, and pre-filled pen convenience. Empower compounded tirzepatide offers lower cost, dose flexibility, formulation options (injection and ODT), and the addition of niacinamide.
Neither option is categorically "better." The right choice depends on your specific circumstances.
Choose brand-name Mounjaro if: You have insurance coverage that makes it affordable. You prioritize FDA-approved products with extensive clinical trial data. You want the convenience of pre-filled pens. You are uncomfortable with the regulatory uncertainty surrounding compounded products.
Choose Empower compounded tirzepatide if: Cost is a significant barrier to treatment. You need dose flexibility beyond what pre-filled pens offer. You have a documented clinical reason for a modified formulation. You are comfortable with the trade-offs of compounded medications. You understand and accept the regulatory and quality considerations involved.
For researchers evaluating their options, SeekPeptides provides detailed comparison tools, dosing calculators, and evidence-based guides that help you make informed decisions about your protocol. Understanding the full picture, not just the marketing claims, is what separates effective treatment from expensive experimentation.
Understanding GLP-1 receptor agonists and how tirzepatide fits
Before evaluating any specific pharmacy source, understanding what tirzepatide actually does at the molecular level helps you assess claims about different formulations. Tirzepatide is not just another GLP-1 receptor agonist. It is a dual agonist that activates both the GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This dual mechanism is what separates it from single-agonist medications like semaglutide and explains why clinical trial results showed superior weight loss outcomes.
The GLP-1 receptor activation slows gastric emptying, reduces appetite, and improves insulin sensitivity. You feel full sooner, stay full longer, and your body processes glucose more efficiently. The GIP receptor activation adds another layer. GIP affects fat tissue directly, influencing how your body stores and burns fat. It also has effects on bone metabolism and potentially neuroprotection, though these secondary benefits are still being studied.
When you inject Empower compounded tirzepatide with niacinamide, the tirzepatide molecule activates these same receptors. The question is not whether compounded tirzepatide can work. If the molecule is properly synthesized and formulated, it will bind the same receptors and trigger the same physiological responses. The question is whether the compounding process consistently delivers the correct amount of properly folded, biologically active tirzepatide.
That is a manufacturing quality question, not a pharmacology question. And it is the core reason why regulatory oversight, quality control, and pharmacy reputation matter so much when choosing a compounded source.
How tirzepatide differs from semaglutide
Many patients evaluating Empower tirzepatide are also considering Empower semaglutide. The semaglutide versus tirzepatide comparison comes down to mechanism and outcomes. Semaglutide activates only the GLP-1 receptor. Tirzepatide activates both GLP-1 and GIP receptors.
In head-to-head clinical trials, tirzepatide produced greater average weight loss than semaglutide at comparable endpoints. The SURMOUNT-1 trial showed 22.5 percent body weight reduction with 15 mg tirzepatide at 72 weeks, compared to approximately 15 percent with semaglutide 2.4 mg in the STEP trials. Individual results vary significantly, and some patients respond better to one medication than the other.
The side effect profiles differ somewhat. Both cause gastrointestinal symptoms as the most common adverse effects, but the specific pattern and severity can differ. Some patients tolerate tirzepatide better than semaglutide. Others find the opposite. There is no reliable way to predict this without trying.
Cost comparisons between compounded tirzepatide and compounded semaglutide from Empower depend on current pricing, which changes. But historically, compounded semaglutide has been slightly less expensive per month than compounded tirzepatide, reflecting differences in raw material costs and formulation complexity.
Long-term considerations and treatment duration
One of the most important questions patients ask is: how long do you stay on GLP-1 medications? The evidence increasingly suggests that these medications work best as long-term treatments rather than short-term interventions. Weight regain after discontinuation is well documented, with studies showing that most patients regain 50 to 70 percent of lost weight within 12 months of stopping treatment.
This has direct implications for choosing Empower compounded tirzepatide. If you plan to use tirzepatide for 12 to 24 months or longer, the cumulative cost difference between compounded and brand-name products becomes substantial. At $300 per month versus $1,000 per month, a 24-month course costs $7,200 from a compounder versus $24,000 for brand-name. That $16,800 difference is life-changing money for many families.
But long-term use also amplifies quality concerns. A single batch with slightly reduced potency from a compounding pharmacy is a minor inconvenience. Twelve months of potentially variable potency is a significant clinical issue that could affect your weight loss trajectory, metabolic health, and motivation to continue treatment.
The ideal approach balances cost and quality. Many patients use compounded tirzepatide during the active weight loss phase and consider transitioning to brand-name products for long-term maintenance, particularly if insurance coverage becomes available. Others remain on compounded formulations indefinitely and monitor their results closely to catch any potency issues early.
What happens when you stop tirzepatide
Understanding the rebound effect helps you plan your treatment timeline. When you stop tirzepatide, the GLP-1 and GIP receptor activation ceases. Appetite returns to pre-treatment levels, often within days to weeks. Gastric emptying speeds back up. The metabolic advantages that supported weight loss disappear.
The withdrawal experience from GLP-1 medications varies. Some patients describe a gradual return of hunger and cravings over two to four weeks. Others report an almost immediate rebound in appetite that feels overwhelming after months of suppressed hunger. The key predictors of successful weight maintenance after stopping include the amount of muscle mass preserved during weight loss, dietary habits established during treatment, physical activity level, and psychological readiness for increased appetite.
This is why sustainable lifestyle changes during tirzepatide treatment matter as much as the medication itself. Use the appetite suppression window to establish eating patterns, exercise habits, and mindset shifts that can persist even when pharmacological support ends.
Red flags and what to watch for with any compounded tirzepatide
Whether you choose Empower or any other compounding pharmacy, certain warning signs should prompt caution. Knowing these helps protect your health and your investment.
Product quality red flags
Visible particles or cloudiness in a vial that should contain clear solution. Unusual color changes compared to your previous vials. Cracked or compromised vial seals. Expiration dates that seem unusually far in the future. Missing or incomplete labeling. These all warrant contacting the pharmacy before use.
Clinical red flags
Dramatic changes in effectiveness between batches. One vial produces strong appetite suppression and the next produces almost none. This suggests potency inconsistency and should be reported to both your prescriber and the pharmacy. Injection site reactions that go beyond normal mild redness, particularly large welts, persistent pain, or signs of infection, require medical attention.
Unexpected side effects that differ significantly from your established pattern on tirzepatide also deserve investigation. If you have been tolerating the medication well for months and suddenly develop severe nausea or other symptoms after starting a new vial, the formulation may be different or compromised.
Business practice red flags
Pharmacies that do not require prescriptions. Any operation selling tirzepatide without a valid prescription is operating illegally and should be avoided entirely. Pharmacies making specific weight loss guarantees. Compounded medications cannot legally make efficacy claims because they have not undergone the clinical trials necessary to support such claims. Prices that seem too good to be true often indicate corner-cutting on quality, sourcing, or testing.
Pharmacies that pressure you to order large quantities upfront or lock you into long-term contracts deserve scrutiny. Legitimate compounding pharmacies fill prescriptions as written by your prescriber, typically for one to three months at a time.
Empower tirzepatide and specific health conditions
How compounded tirzepatide interacts with specific health conditions matters for patient safety and outcome optimization.
Type 2 diabetes
Tirzepatide has FDA approval (as brand-name Mounjaro) for type 2 diabetes management. For diabetic patients using compounded formulations, blood glucose monitoring becomes especially important. Dose-dependent hypoglycemia risk exists, particularly when combined with insulin or sulfonylureas. Your endocrinologist should be involved in prescribing decisions and monitoring.
Hair loss concerns
A growing concern among tirzepatide users involves GLP-1 related hair loss. Rapid weight loss from any cause, including tirzepatide, can trigger telogen effluvium, a temporary but distressing condition where increased hair shedding occurs two to four months after significant weight reduction. This is not unique to any particular GLP-1 medication or pharmacy source. It is a response to the caloric deficit and metabolic stress of rapid weight loss.
Adequate protein intake (at least 0.8 to 1.0 grams per pound of body weight), micronutrient supplementation, and gradual dose titration can help minimize this effect. Hair typically regrows as the body stabilizes at its new weight.
Gastrointestinal conditions
Patients with pre-existing gastrointestinal conditions need particular caution with tirzepatide. The medication slows gastric emptying significantly, which can worsen conditions like gastroparesis, severe GERD, or inflammatory bowel disease. If you have a complex medical history, comprehensive evaluation by your prescriber before starting any form of tirzepatide is essential, not optional.
The future of compounded tirzepatide
The landscape continues to evolve rapidly. Several factors will determine whether compounded tirzepatide from pharmacies like Empower remains available in its current form.
Legal outcomes from the Eli Lilly lawsuits will set precedents. If courts determine that adding niacinamide or other ingredients does not create a sufficiently "different" product, compounding pharmacies may face existential challenges in this market. If the courts allow modified formulations to continue, the market will likely stabilize and potentially grow.
FDA enforcement priorities matter. The agency has limited resources and must prioritize which compounding violations to pursue. Political pressure from patients who rely on affordable compounded alternatives creates a counterbalance to pharmaceutical industry lobbying. The regulatory outcome will likely land somewhere between full prohibition and unrestricted compounding.
New GLP-1 medications entering the market could change the calculus entirely. Retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, is advancing through clinical trials with even more impressive weight loss data than tirzepatide. Mazdutide represents another next-generation option. Cagrilintide takes yet another approach. As these alternatives become available, they may reduce the demand pressure on compounded tirzepatide, potentially making both brand-name and compounded versions more accessible.
For patients currently using Empower tirzepatide, staying informed about these developments helps you plan ahead. Subscribe to reliable sources of regulatory updates. Maintain an open relationship with your prescriber about treatment continuity plans. And do not assume that current access will continue unchanged. Having a backup plan, whether that involves brand-name products, alternative compounders, or different medications entirely, protects you from disruption.
Making informed decisions about peptide protocols requires more than pharmacy comparisons. SeekPeptides members access comprehensive protocol libraries, cost analysis tools, reconstitution calculators, and community insights from thousands of researchers navigating the same questions. Whether you are starting your first tirzepatide protocol or optimizing an existing one, evidence-based guidance beats guesswork every time.
Frequently asked questions
Is Empower Pharmacy tirzepatide the same as Mounjaro?
No. Empower compounds tirzepatide with niacinamide, creating a formulation that differs from brand-name Mounjaro. The active ingredient (tirzepatide) is the same molecule, but the final product is not FDA-approved, has not undergone clinical trials, and contains an additional ingredient. Manufacturing processes and quality control standards also differ between a compounding pharmacy and a traditional pharmaceutical manufacturer.
Can I still get compounded tirzepatide from Empower after the shortage ended?
Yes, but under different legal conditions. Since the FDA declared the tirzepatide shortage resolved, Empower can only compound tirzepatide formulations that are "not essentially a copy" of the commercial product. Their tirzepatide/niacinamide formulation and tirzepatide ODT are designed to meet this requirement. Your prescriber must provide clinical justification for the compounded version.
How do I know if my Empower tirzepatide is working?
Track three indicators: appetite changes (reduced hunger and earlier satiety), weight loss trajectory (most patients notice effects within 2-4 weeks), and metabolic markers (if your provider monitors labs). If you experience appetite suppression but weight loss has stalled, the medication may be working but other factors need adjustment.
Does Empower tirzepatide need to be refrigerated?
The injection must be stored between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius). Never freeze it. The ODT tablets store at room temperature. For detailed storage guidelines, including stability during temporary temperature excursions, see our complete storage guide.
What is the difference between Empower tirzepatide injection and ODT?
The injection delivers tirzepatide/niacinamide subcutaneously with established absorption pathways. The ODT dissolves on the tongue for oral mucosal absorption. The injection has more predictable bioavailability based on what we know about subcutaneous tirzepatide delivery. The ODT has not been clinically validated for bioequivalence. Our oral versus injection comparison covers the trade-offs in detail.
Are there states where Empower cannot ship tirzepatide?
Empower ships injectable tirzepatide nationwide. However, the tirzepatide ODT cannot be shipped to California, Wisconsin, Iowa, or Minnesota due to state-specific compounding regulations. Check Empower licensure information for the most current shipping availability to your state.
What should I do if my Empower tirzepatide arrives warm?
Contact Empower Pharmacy immediately before using the medication. Temperature excursions during shipping can compromise peptide stability and potency. The pharmacy should provide guidance on whether the specific temperature exposure renders the medication unusable and arrange a replacement if necessary. Do not use a vial that you suspect has been temperature-compromised.
Can I switch between Empower tirzepatide and Mounjaro?
Yes, with your prescriber guidance. The active molecule is the same, so dose conversion is straightforward. Maintain your current dose when switching. Recalculate injection volume if concentrations differ. Some patients notice subjective differences between formulations, but these typically resolve within one to two weeks.
External resources
For researchers serious about optimizing their weight management protocols, SeekPeptides offers the most comprehensive resource available. Members access detailed tirzepatide dosing protocols, precision calculators, expert-reviewed safety guides, and a community of thousands who have navigated these exact decisions. Whether you are evaluating Empower, considering brand-name options, or exploring other compounding sources, SeekPeptides provides the evidence-based guidance that makes informed choices possible.
In case I do not see you, good afternoon, good evening, and good night. May your formulations stay potent, your doses stay accurate, and your progress stay consistent.