Feb 24, 2026

You stored it right. You kept it cold. You followed every instruction on the label. And now you are staring at a vial of tirzepatide, wondering if it is still good. The expiration date is close. Maybe it already passed. Maybe the vial sat out on the counter longer than you planned, and you are not sure if those extra hours matter. They do.
Tirzepatide is not a simple molecule. It is a 39-amino-acid peptide chain with a C20 fatty acid modification, and every link in that chain is vulnerable to heat, light, moisture, and time. When even one bond breaks or one amino acid oxidizes, the entire molecule can lose its ability to activate the GIP and GLP-1 receptors that make it work. That means reduced appetite suppression, diminished weight loss results, and wasted money. But here is what most guides skip over. The shelf life of tirzepatide is not a single number. It depends on whether you have a brand-name pen or a compounded vial, whether the product is refrigerated or sitting at room temperature, and whether it has been reconstituted from lyophilized powder. Each of these variables changes the timeline dramatically.
This guide breaks down every scenario. Brand versus compounded. Refrigerated versus room temperature. Opened versus unopened. Lyophilized powder versus reconstituted solution. You will learn exactly how long tirzepatide lasts under each condition, what accelerates its degradation, how to spot a vial that has gone bad, and what the actual science says about peptide stability. SeekPeptides built this resource because researchers deserve precise, evidence-based answers rather than vague instructions that leave room for error.

Understanding tirzepatide as a peptide molecule
Before you can understand shelf life, you need to understand what tirzepatide actually is at the molecular level. This is not a small chemical compound like aspirin or acetaminophen. Tirzepatide is a peptide, which means it is built from amino acids linked together in a specific sequence. Specifically, it is a 39-amino-acid chain with a synthetic modification: a C20 fatty diacid moiety attached via a linker to the lysine residue at position 20.
That fatty acid attachment is what gives tirzepatide its long half-life in the body. It binds to albumin in the bloodstream, slowing clearance and allowing once-weekly dosing. But that same structural complexity makes the molecule sensitive to environmental stressors.
Peptides degrade through several mechanisms. Hydrolysis breaks peptide bonds when water molecules attack the backbone. Oxidation targets vulnerable amino acids like methionine and tryptophan, altering their chemical structure. Deamidation converts asparagine residues to aspartate, changing the charge profile of the molecule. Aggregation causes individual peptide chains to clump together, forming particles that reduce both potency and safety. Each of these degradation pathways accelerates at higher temperatures, and each one reduces how effectively tirzepatide activates the dual GIP/GLP-1 receptor system that drives its therapeutic effects.
Research on peptide stability published in the Journal of Pharmaceutical Sciences demonstrates that temperature is the single most important variable. For every 10 degrees Celsius increase in storage temperature, the rate of peptide degradation roughly doubles. This is why refrigeration is not just recommended. It is essential.
Brand-name tirzepatide shelf life (Mounjaro and Zepbound)
Let us start with the clearest numbers. Brand-name tirzepatide, sold as Mounjaro for type 2 diabetes and Zepbound for weight management, comes in prefilled pens manufactured by Eli Lilly. These pens contain a ready-to-use liquid formulation that has undergone extensive stability testing under International Council for Harmonisation (ICH) guidelines.
Unopened, refrigerated
Unopened Mounjaro and Zepbound pens have a manufacturer-assigned expiration date, typically 12 to 18 months from the date of manufacture. As long as the pen remains sealed and stored in a refrigerator between 36 and 46 degrees Fahrenheit (2 to 8 degrees Celsius), it will maintain full potency until that expiration date. The pen should stay in its original carton to protect it from light exposure, which can trigger photodegradation of certain amino acid residues.
This is straightforward. Check the date on the box. If the pen is sealed and cold, you are good.
Unopened, room temperature
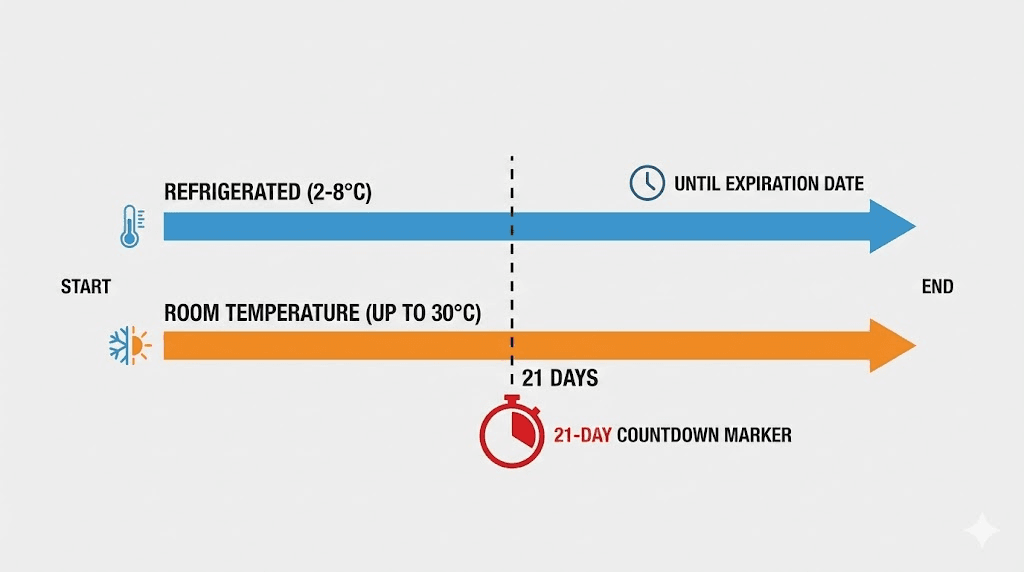
Sometimes refrigeration is not possible. You are traveling. Your pharmacy shipped the pen and it arrived at room temperature. According to Eli Lilly prescribing information, an unopened tirzepatide pen can be stored at room temperature (up to 86 degrees Fahrenheit or 30 degrees Celsius) for a maximum of 21 days. After those 21 days, the pen must be discarded, even if it looks perfectly fine.
Here is a critical detail that many people miss. Once a pen has been stored at room temperature, it cannot go back into the refrigerator. The temperature cycling, moving from cold to warm and back to cold, can accelerate aggregation and destabilize the formulation. The manufacturer explicitly warns against this.
So the 21-day room temperature window is a one-way trip. Use it when you need to. But plan accordingly.
After first use
Once you use a Mounjaro or Zepbound pen for the first time, the same 21-day countdown applies. Whether you keep the opened pen in the refrigerator or at room temperature (below 86 degrees Fahrenheit), any remaining medication must be discarded after 21 days from first use. Since each pen is designed for a single dose, this is rarely an issue with the prefilled pen format. But if you are using the multi-dose vial format, this timeline becomes very important.
Compounded tirzepatide shelf life
This is where things get more complicated. And this is where most people run into trouble.
Compounded tirzepatide is prepared by compounding pharmacies rather than manufactured by Eli Lilly. It may come as a ready-to-inject solution in a multi-dose vial, or as lyophilized (freeze-dried) powder that requires reconstitution before use. The shelf life of compounded tirzepatide depends entirely on the formulation, the compounding conditions, and the specific pharmacy that prepared it.
Beyond-use dates versus expiration dates
Brand-name medications have expiration dates assigned by the manufacturer based on years of stability testing under controlled conditions. Compounded medications have beyond-use dates (BUDs), which are shorter and assigned by the compounding pharmacy based on United States Pharmacopeia (USP) Chapter 797 guidelines for sterile preparations.
These are fundamentally different things.
An expiration date says: this product has been tested and confirmed stable for this long under these conditions. A beyond-use date says: based on available evidence and regulatory guidelines, this preparation should be used before this date. The BUD is typically more conservative because compounded products do not undergo the same extensive long-term stability testing as commercially manufactured drugs.
Ready-to-inject compounded tirzepatide
Most compounded tirzepatide solutions come with a BUD of 28 to 90 days from the date of compounding, depending on the pharmacy. Some pharmacies assign a 28-day BUD as a conservative baseline. Others, after conducting their own stability testing, extend to 60 or even 90 days. A few pharmacies with advanced stability data and cleanroom environments may assign BUDs up to 6 months, though this is less common.
The variation exists because USP 797 allows pharmacies to extend BUDs if they can provide supporting stability data. Without that data, the default BUD for a sterile aqueous preparation stored refrigerated is relatively short.
What does this mean for you? Check the BUD on your vial. That date is the hard deadline.
Lyophilized (freeze-dried) compounded tirzepatide
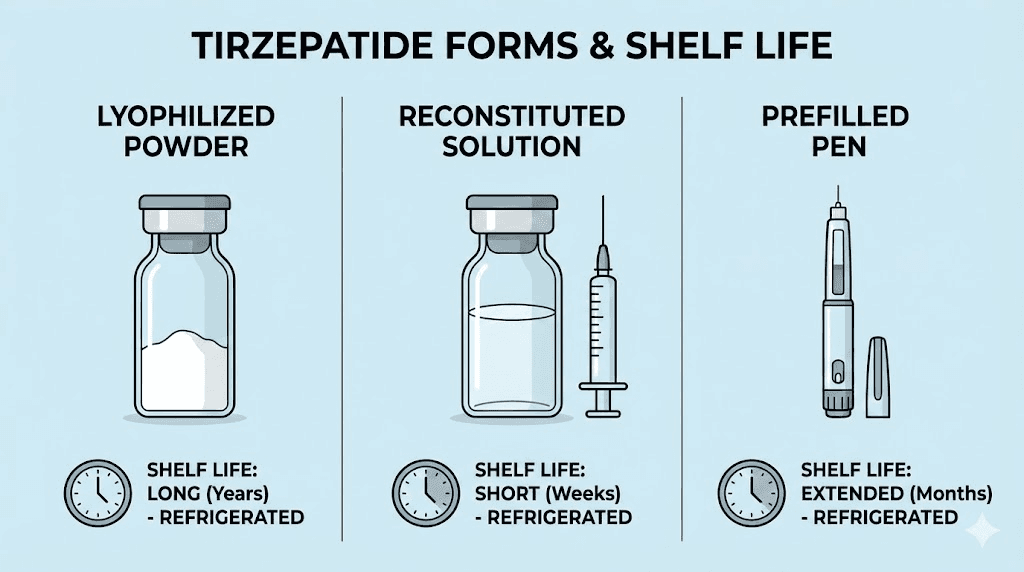
Lyophilized tirzepatide powder is the most stable form of the compound. Without water present, the degradation pathways that destroy peptides in solution, hydrolysis, deamidation, oxidation, are dramatically slowed. Research on peptide stability in powder form shows that lyophilized peptides stored at minus 20 degrees Celsius can remain stable for years.
At refrigerator temperatures (2 to 8 degrees Celsius), lyophilized tirzepatide typically maintains potency for several months to over a year. At room temperature, stability drops but can still extend to several weeks or months depending on the specific formulation and packaging.
However, once you add bacteriostatic water to reconstitute the powder, the clock starts ticking much faster. The reconstituted solution behaves like any other aqueous peptide preparation, subject to all the same degradation forces.
After reconstitution
Reconstituted tirzepatide solution should be stored in the refrigerator at 2 to 8 degrees Celsius and typically remains stable for 7 to 28 days, depending on the diluent used and the concentration achieved. When bacteriostatic water is the diluent, the benzyl alcohol preservative helps prevent microbial growth, which can extend usability compared to sterile water alone.
At room temperature, reconstituted tirzepatide degrades significantly faster. Most sources recommend using reconstituted peptide solutions within 24 to 72 hours if stored at room temperature, though some formulations may tolerate up to 7 days. The safest approach is always to refrigerate immediately after reconstitution and use within the timeframe specified by your pharmacy.
The complete tirzepatide shelf life reference table
Here is everything consolidated into one reference.
Form | Storage condition | Shelf life | Notes |
|---|---|---|---|
Brand pen (unopened) | Refrigerated (36-46F) | Until expiration date (12-18 months) | Keep in original carton |
Brand pen (unopened) | Room temp (below 86F) | 21 days maximum | Cannot return to fridge |
Brand pen (opened) | Refrigerated or room temp | 21 days from first use | Discard after 21 days |
Compounded solution | Refrigerated (36-46F) | 28-90 days (check BUD) | Varies by pharmacy |
Compounded solution | Room temp (below 86F) | Up to 21 days | Degrades faster than refrigerated |
Lyophilized powder | Frozen (-20C) | 1-3+ years | Most stable form |
Lyophilized powder | Refrigerated (2-8C) | Several months to 1 year+ | Keep sealed, dry |
Lyophilized powder | Room temperature | Weeks to months | Less stable than refrigerated |
Reconstituted solution | Refrigerated (2-8C) | 7-28 days | Use bacteriostatic water |
Reconstituted solution | Room temperature | 24-72 hours | Refrigerate immediately after mixing |
Use this table as your quick reference. But remember, the most reliable number is always the one printed on your specific product by the manufacturer or compounding pharmacy.
What destroys tirzepatide potency
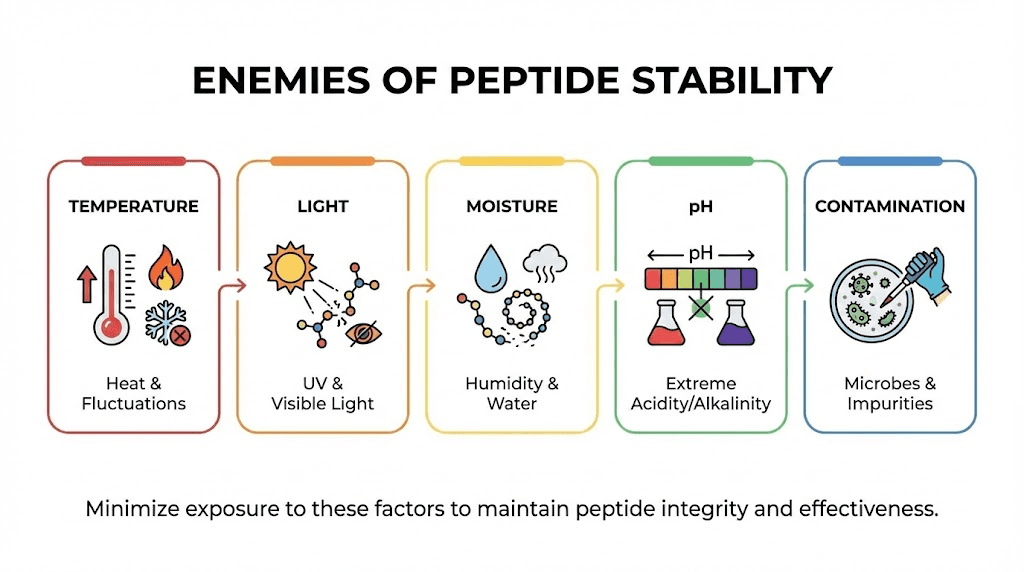
Knowing the timelines is one thing. Understanding why those timelines exist gives you the knowledge to protect your investment. Five primary factors degrade tirzepatide, and each one works differently.
Temperature
Heat is the number one enemy of peptide stability. The Arrhenius equation, a fundamental principle of chemical kinetics, tells us that reaction rates approximately double for every 10 degrees Celsius increase in temperature. For tirzepatide specifically, this means storage at 30 degrees Celsius degrades the peptide roughly twice as fast as storage at 20 degrees Celsius, and roughly four times faster than storage at 10 degrees Celsius.
At refrigerator temperatures (2 to 8 degrees Celsius), the degradation reactions slow to a crawl. Hydrolysis still occurs, but so slowly that the peptide maintains therapeutic potency for months. At room temperature, those same reactions proceed fast enough to noticeably reduce potency within weeks.
What about freezing? This is counterintuitive, but freezing can be worse than refrigeration for ready-to-use liquid formulations. When water freezes, ice crystals form and concentrate the dissolved peptide into smaller pockets of unfrozen solution. This creates localized high concentrations that promote aggregation. The ice crystals themselves can physically damage peptide structure. This is why both Eli Lilly and most compounding pharmacies explicitly warn against freezing tirzepatide liquid formulations.
Lyophilized powder is different. It is already dry, so freezing does not cause ice crystal formation. Frozen storage at minus 20 degrees Celsius is actually ideal for long-term preservation of lyophilized peptides.
Light
Ultraviolet and visible light carry enough energy to trigger photochemical reactions in peptide molecules. Tirzepatide contains tryptophan and tyrosine residues, both of which are particularly susceptible to photo-oxidation. When light strikes these amino acids, it generates reactive oxygen species that can modify the peptide chain and reduce biological activity.
This is why Mounjaro and Zepbound pens come in opaque cartons. The carton is not just packaging. It is a light shield. If you remove a pen from its carton and leave it on a bright countertop or near a window, you accelerate degradation even if the temperature is correct.
Compounded vials are sometimes packaged in amber glass, which filters out the most damaging wavelengths. If your vial is clear glass, keep it in its box or wrapped in foil when not in use.
Moisture and humidity
Water is the medium in which hydrolysis occurs. For liquid formulations, there is obviously no way to avoid water exposure since the peptide is dissolved in an aqueous solution. But for lyophilized powder, moisture is the critical variable.
Lyophilized peptides achieve their exceptional stability precisely because they lack water. If the seal on a lyophilized vial is compromised, or if the vial is stored in a humid environment where moisture can slowly penetrate, the powder begins absorbing water vapor. This reintroduces the hydrolysis pathway and dramatically shortens shelf life.
Keep lyophilized vials sealed until you are ready to reconstitute. Store them in a cool, dry environment. If you notice the powder looks clumpy or wet, the seal may have been compromised.
pH
Tirzepatide formulations are buffered to maintain a specific pH range, typically around pH 4 to 5 for liquid preparations. At this slightly acidic pH, the peptide is most stable. Moving outside this range in either direction accelerates degradation.
This is generally not something you can control with brand-name products since the manufacturer handles formulation. But with compounded tirzepatide, the quality of the buffer system matters. Well-formulated compounded products maintain stable pH throughout their beyond-use date. Poorly formulated ones may drift, accelerating peptide breakdown. This is one reason why choosing a reputable compounding pharmacy matters so much.
Microbial contamination
This is not about chemical degradation. It is about safety. Once a multi-dose vial is punctured with a needle, the sterile barrier is broken. Bacteria can enter the vial through the rubber stopper, and in a nutrient-rich peptide solution, they can multiply rapidly at room temperature.
Bacteriostatic water contains 0.9% benzyl alcohol, which inhibits bacterial growth and provides a critical safety margin for multi-dose vials. Sterile water for injection does not contain a preservative, so vials reconstituted with sterile water have a much shorter usable life. If your compounded tirzepatide uses bacteriostatic water, that preservative is doing real work.
Regardless of the diluent, always swab the vial stopper with an alcohol prep pad before each needle insertion. Use a fresh needle every time. And never share vials between people.
How to tell if your tirzepatide has gone bad
There is no home test kit for peptide potency. But there are warning signs that should stop you from using a questionable vial. Some are obvious. Some are subtle.
Visual inspection
Tirzepatide solution should be clear and colorless to very slightly yellow. Hold the vial up to a light source and look carefully. If you see any of the following, do not use the product.
Cloudiness or haziness. This suggests aggregation, where peptide molecules have clumped together and fallen out of solution. Aggregated peptide is not just less effective. It can trigger injection site reactions or immune responses because the body recognizes aggregated protein as foreign.
Visible particles or floaters. Tiny specks, fibers, or precipitates in the solution indicate either contamination or advanced degradation. Even one visible particle means the vial should be discarded.
Color change. A shift from colorless toward yellow, brown, or pink can indicate oxidation of amino acid residues. Compounded tirzepatide solutions may have a natural very light yellow or slightly pink tint depending on the formulation, but any change from the original appearance is a red flag.
Unusual consistency. If the solution seems thicker or more viscous than when you first received it, this may indicate concentration changes from evaporation or formulation instability.
The silent problem
Here is what makes this tricky. Tirzepatide can lose significant potency without any visible change. A vial can look perfectly clear and colorless while containing peptide that has degraded 30, 40, even 50 percent. The degradation products are often soluble and colorless, making them invisible to the naked eye.
This is why expiration dates and beyond-use dates matter even when the product looks fine. You cannot see potency. You can only trust the timeline.
Performance indicators
If your tirzepatide is still within its labeled date but you suspect degradation (perhaps it was exposed to heat during shipping, or left out of the fridge longer than intended), watch for performance changes.
Reduced appetite suppression. If the appetite-reducing effects you normally experience from tirzepatide start becoming noticeably weaker at the same dose, degradation is one possible explanation.
Return of cravings. Tirzepatide works partly by modulating hunger hormones. Degraded peptide may not suppress cravings as effectively, leading to a return of the food noise that tirzepatide normally quiets.
Slower weight loss. If your weight loss progress stalls or reverses despite no changes in diet or exercise, suspect the medication before blaming your metabolism.
Blood sugar instability. For those using tirzepatide for glycemic control, degraded medication may result in less predictable blood glucose readings.
These symptoms have many possible causes beyond degradation. But if they coincide with a temperature excursion or nearing the end of a vial shelf life, degradation should be high on your list of suspects.
Brand-name versus compounded: shelf life differences explained
The shelf life gap between brand-name and compounded tirzepatide is significant, and understanding why helps you make better decisions about sourcing and storage.
Manufacturing environment
Eli Lilly manufactures Mounjaro and Zepbound in FDA-inspected facilities with rigorous environmental controls. Temperature, humidity, particulate levels, and microbial counts are continuously monitored. The manufacturing process itself is optimized and validated over thousands of batches.
Compounding pharmacies operate under different (though still regulated) standards. They follow USP 797 guidelines for sterile compounding, which specify requirements for personnel training, cleanroom environments, and quality control. However, the scale and resources differ enormously from a major pharmaceutical manufacturer. Some compounding pharmacies operate state-of-the-art facilities with ISO 5 cleanrooms. Others meet the minimum requirements. This variability directly impacts product quality and stability.
Formulation optimization
Eli Lilly spent years optimizing the tirzepatide formulation. They tested different buffer systems, surfactants, pH levels, and concentrations to maximize stability. They conducted accelerated stability testing, exposing the product to elevated temperatures for extended periods and measuring degradation rates, then projected shelf life under normal storage conditions.
Compounding pharmacies typically use simpler formulations. They may not have the resources for extensive stability testing. This is not a criticism. It is a structural reality of compounding versus manufacturing. The result is that compounded products generally have shorter validated shelf lives, even if the actual potency might persist longer.
Stability testing depth
Brand-name stability testing follows ICH Q1A guidelines, involving multiple storage conditions (25 degrees Celsius/60% relative humidity for long-term, 40 degrees Celsius/75% relative humidity for accelerated), multiple time points (0, 3, 6, 9, 12, 18, 24 months), and multiple analytical methods (HPLC for potency, size exclusion chromatography for aggregation, pH measurement, visual inspection, and more).
Compounding pharmacy stability testing, when performed, is typically less extensive. This is why USP 797 assigns conservative default BUDs and requires pharmacies to provide supporting data before extending those dates.
What this means for you
If shelf life is a major concern, brand-name tirzepatide offers the longest validated storage window. If you are using compounded tirzepatide for cost or availability reasons, proper storage becomes even more critical because you are working with a shorter timeline. Every hour at elevated temperature eats into an already limited shelf life.

Temperature excursions: what happens when storage goes wrong
Life is not a laboratory. Deliveries sit on doorsteps in summer heat. Refrigerators malfunction. Power outages happen. Travel plans change. What happens to tirzepatide shelf life when storage conditions are not ideal?
Short-term heat exposure
If your tirzepatide vial or pen was left at room temperature for a few hours, there is likely no significant impact on potency. The 21-day room temperature window exists precisely because the peptide can tolerate moderate temperatures for limited periods. A couple of hours at 75 degrees Fahrenheit will not meaningfully degrade the product.
However, the situation changes if temperatures exceed 86 degrees Fahrenheit (30 degrees Celsius). At 90 to 100 degrees Fahrenheit, degradation rates increase substantially. If your medication sat in a hot car, on a sunny porch, or in a delivery vehicle during summer heat, the damage may already be done even if the exposure was brief.
What about delivery day?
Most pharmacies ship tirzepatide with cold packs in insulated packaging. But these cold packs maintain temperature for a limited time, usually 24 to 48 hours depending on ambient conditions. If your package sits on a hot porch for several hours after the cold packs expire, the medication may have experienced temperatures well above 86 degrees Fahrenheit.
When you receive a tirzepatide shipment, check the cold packs. If they are still partially frozen or cold to the touch, the medication is likely fine. If the cold packs are warm and the insulation feels hot, contact your pharmacy. They may need to replace the shipment.
Freezing incidents
What if your refrigerator was set too cold, or the medication accidentally ended up in the freezer? Freezing is a problem for liquid tirzepatide formulations. Ice crystal formation damages the peptide through several mechanisms.
First, the physical force of ice crystal expansion can mechanically disrupt peptide structure. Second, freeze-concentration effects increase local peptide concentration in the remaining liquid fraction, promoting aggregation. Third, pH shifts occur during freezing because buffer components freeze out of solution at different rates, temporarily creating pockets of extreme pH.
If your liquid tirzepatide froze, even partially, the manufacturer recommends discarding it. You may not see any visual changes, but the structural damage at the molecular level can reduce potency and increase the risk of injection site reactions.
Again, lyophilized powder is the exception. Freezing does not harm dry peptide powder because there is no water to form damaging ice crystals.
Repeated temperature cycling
Taking medication in and out of the refrigerator repeatedly is worse than a single temperature excursion. Each cycle of warming and cooling stresses the peptide formulation. Condensation can form on vial surfaces, potentially introducing moisture. Temperature fluctuations can cause micro-separations in the formulation that may not be visible but still reduce stability.
Pick a storage location and stick with it. If you need room temperature tirzepatide for travel, take it out of the fridge once and use it within the allowed timeframe. Do not cycle between cold and warm storage.
Storing tirzepatide correctly: a practical protocol
Theory is useful. Practice is what keeps your tirzepatide potent. Here is exactly how to store tirzepatide in every common scenario.
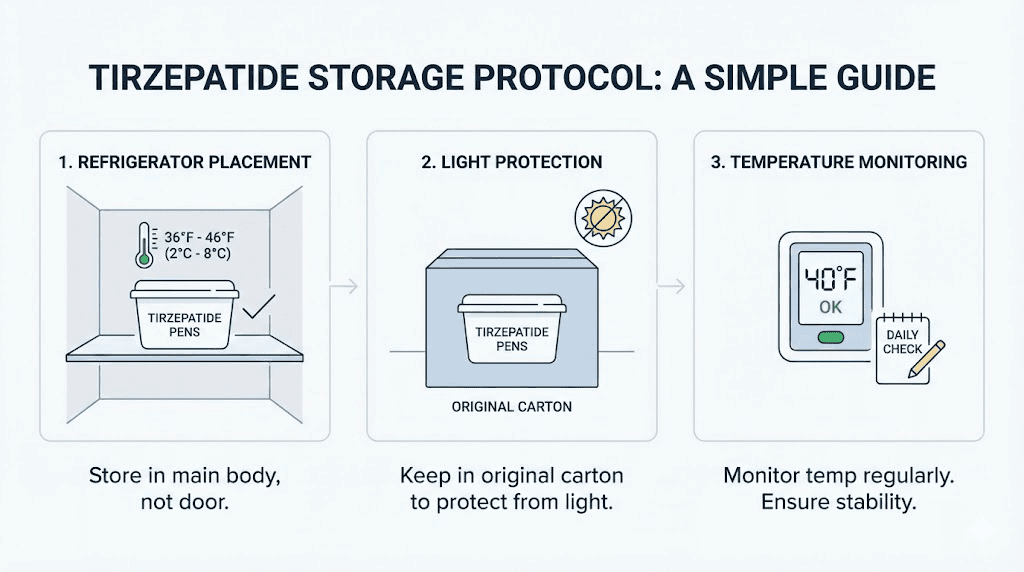
At home (primary storage)
Place the tirzepatide pen or vial in the main body of your refrigerator, not in the door and not against the back wall. The door experiences the most temperature fluctuation from opening and closing. The back wall, especially in older refrigerators, may drop below freezing. The middle of a shelf in the main compartment offers the most consistent temperature.
Keep the product in its original packaging if possible. The carton or box protects against light and provides a buffer against minor temperature changes. If the original packaging is not available, wrap the vial in aluminum foil to block light and place it in a small zip-lock bag to protect against moisture from refrigerator humidity.
Use a refrigerator thermometer. Built-in temperature displays are often inaccurate by several degrees. A separate digital thermometer, placed near your medication, gives you confidence that the temperature is actually in the 36 to 46 degree Fahrenheit range.
During travel
For trips under 21 days, you can bring tirzepatide at room temperature as long as it stays below 86 degrees Fahrenheit. A simple insulated pouch or travel case is adequate for most situations. For hot climates or extended travel, use a medical-grade cooling case with gel packs. Avoid direct sunlight and never leave medication in a parked car.
For longer trips, consider a portable medication cooler that maintains 2 to 8 degrees Celsius. These small electric coolers are available specifically for temperature-sensitive medications and can run on USB power or car adapters.
After reconstitution
If you are working with lyophilized tirzepatide powder, reconstitute only when ready to begin using the vial. Do not reconstitute ahead of time just to have it ready. Every day the reconstituted solution sits in the fridge eats into its limited shelf life.
After adding bacteriostatic water, gently swirl the vial to dissolve the powder. Do not shake vigorously, as this introduces air bubbles and can promote aggregation at the air-liquid interface. Once dissolved, immediately place the vial in the refrigerator. Write the reconstitution date on the vial with a permanent marker so you always know when the clock started.
Dose preparation
When drawing a dose, remove the vial from the refrigerator only long enough to draw the injection. Swab the stopper with alcohol. Draw the dose with a sterile needle and syringe. Return the vial to the fridge. The entire process should take less than five minutes.
Leaving the vial out on the counter while you prep your injection site, gather supplies, or eat breakfast adds unnecessary room temperature exposure. Those minutes add up across dozens of doses.
Maximizing the shelf life of compounded tirzepatide
Because compounded tirzepatide has a shorter baseline shelf life than brand-name products, maximizing every day of potency matters more. Here are specific strategies.
Choose the right pharmacy
Not all compounding pharmacies are equal. Look for pharmacies that are accredited by the Pharmacy Compounding Accreditation Board (PCAB) or licensed by the state board of pharmacy for sterile compounding. Ask about their cleanroom classification, their quality control processes, and whether they perform stability testing on their tirzepatide formulations.
Pharmacies that conduct stability testing can assign longer, data-backed BUDs. Pharmacies that rely on default USP 797 BUDs without testing will assign shorter dates, even if the product would remain stable longer. The testing is what gives confidence.
Request lyophilized when possible
If your pharmacy offers both ready-to-use solution and lyophilized powder, the powder form gives you much more flexibility with shelf life. You can store the sealed powder for months and reconstitute only when you need it, giving you a fresh solution with maximum remaining potency every time.
The trade-off is that reconstitution adds a step and introduces a small risk of contamination or error. But for researchers who plan to store tirzepatide for extended periods, the lyophilized format is clearly superior.
Order the right quantity
Do not order more compounded tirzepatide than you can use within the BUD. If your dosing protocol calls for a certain amount per week, calculate how many weeks of supply you need and order accordingly. Having excess vials expire before you can use them is wasted money.
This is especially relevant for people who are adjusting their dose during the titration phase. If you are on a low starting dose, a multi-dose vial may last longer than its BUD allows. In that case, smaller vials or more frequent orders make more sense.
Monitor your refrigerator
A refrigerator that runs slightly warm, say 50 degrees Fahrenheit instead of the recommended 36 to 46 degrees, will reduce the shelf life of your compounded tirzepatide. Those extra few degrees matter because of the exponential relationship between temperature and degradation rate.
Invest in a min/max thermometer that records the highest and lowest temperatures over a given period. Check it weekly. If your refrigerator is consistently running above 46 degrees Fahrenheit, adjust the settings or consider a dedicated medication refrigerator.
Tirzepatide shelf life versus other GLP-1 medications
How does tirzepatide compare to other GLP-1 receptor agonists when it comes to storage and stability? The comparison reveals some interesting differences.
Medication | Type | Refrigerated shelf life (unopened) | Room temp tolerance | Freezing OK? |
|---|---|---|---|---|
Tirzepatide (Mounjaro/Zepbound) | Dual GIP/GLP-1 | Until expiration (12-18 months) | 21 days below 86F | No (liquid) |
Semaglutide (Ozempic/Wegovy) | GLP-1 | Until expiration (24 months) | 56 days below 86F | No |
Triple agonist | Varies (investigational) | Limited data | No (liquid) | |
Amylin analog | Varies (investigational) | Limited data | No |
Notice that semaglutide has a notably longer room temperature tolerance, 56 days versus tirzepatide at 21 days. This is a meaningful practical difference for people who travel frequently or have limited refrigerator access. The reason likely relates to differences in formulation stability, excipient choice, and the structural properties of the peptide itself.
If you are comparing tirzepatide and semaglutide, shelf life and storage convenience are worth considering alongside efficacy and side effect profiles. For some people, the longer room temperature window of semaglutide is a genuine advantage.
The science behind peptide degradation
For researchers who want to understand the mechanisms at a deeper level, here is how peptide degradation actually works at the molecular scale.
Hydrolysis
The peptide bond, the amide linkage between amino acids, is thermodynamically unstable in the presence of water. Hydrolysis is the process by which water molecules attack and break these bonds, fragmenting the peptide chain into smaller pieces. The rate of hydrolysis depends on temperature, pH, and the specific amino acid sequence. Certain sequences are more susceptible than others, particularly those adjacent to aspartate residues.
For tirzepatide, hydrolysis at critical positions can completely inactivate the molecule by disrupting the binding interface that interacts with GIP and GLP-1 receptors.
Oxidation
Tirzepatide contains methionine and tryptophan residues that are vulnerable to oxidation. Reactive oxygen species, present in trace amounts in all aqueous solutions and generated by light exposure, can modify these amino acids. Methionine sulfoxide formation is one of the most common oxidative modifications in pharmaceutical peptides.
Oxidized tirzepatide may still bind to its target receptors, but with reduced affinity. This manifests as partial loss of potency rather than complete inactivation. You might still get some effect, but less than you should.
Deamidation
Asparagine residues in peptides spontaneously convert to aspartate through a deamidation reaction, particularly at elevated temperatures and at neutral to basic pH. This changes the charge of the amino acid, which can alter the three-dimensional structure of the peptide and reduce its biological activity.
Deamidation is one of the primary degradation pathways studied during pharmaceutical stability testing. It is also one of the reasons that peptide formulations are typically buffered at slightly acidic pH, where deamidation is slower.
Aggregation
Under certain conditions, individual peptide molecules associate with each other to form dimers, oligomers, and eventually larger aggregates. Aggregation is promoted by high concentration, elevated temperature, agitation (shaking), and freeze-thaw cycling.
Aggregated peptide is problematic for two reasons. First, aggregated molecules cannot bind to receptors properly, so potency decreases. Second, protein aggregates can trigger immune responses, potentially causing injection site reactions or, in rare cases, the development of anti-drug antibodies that neutralize future doses.
This is why tirzepatide labels warn against shaking the product. Gentle inversion is fine. Vigorous shaking creates air-liquid interfaces that promote aggregation.
Frequently asked questions
Can I use tirzepatide after the expiration date?
No. The expiration date represents the manufacturer guarantee of potency and safety. After that date, degradation may have reduced the active peptide below therapeutic levels. Using expired tirzepatide means unpredictable dosing, which can stall your progress or cause unexpected side effects from degradation products. Always use a fresh vial.
Does compounded tirzepatide expire faster than brand-name?
Yes, in most cases. Brand-name Mounjaro and Zepbound carry expiration dates of 12 to 18 months from manufacture, while compounded tirzepatide typically has beyond-use dates of 28 to 90 days from compounding. The difference reflects manufacturing standards, formulation optimization, and the extent of stability testing rather than an inherent quality difference in the peptide itself.
What happens if tirzepatide is left out of the fridge overnight?
A single overnight room temperature exposure (8 to 12 hours below 86 degrees Fahrenheit) is unlikely to cause significant degradation. This falls well within the 21-day room temperature allowance. Note the time and factor it into your remaining room temperature budget if you plan to be without refrigeration again.
Can I put tirzepatide back in the fridge after it has been at room temperature?
For brand-name pens, Eli Lilly states that once stored at room temperature, the pen should not be returned to the refrigerator. For compounded tirzepatide, follow your pharmacy instructions. The concern is that temperature cycling promotes condensation and aggregation, reducing stability more than consistent storage at either temperature.
How do I know if my compounding pharmacy is reliable?
Look for PCAB accreditation, state board of pharmacy licensing for sterile compounding, third-party purity testing, clear beyond-use date labeling, and responsive customer service. Ask if they perform stability testing on their tirzepatide formulations. A good pharmacy will be transparent about their processes.
Does the concentration of compounded tirzepatide affect shelf life?
Higher concentrations can actually be slightly more stable because there is a lower ratio of water to peptide, which means fewer water molecules available for hydrolysis per peptide molecule. However, high concentrations also increase the risk of aggregation. Most compounding pharmacies formulate at concentrations that balance these factors.
Is lyophilized tirzepatide better than ready-to-use solution?
For long-term storage, lyophilized is clearly superior. Dry powder at refrigerator or freezer temperatures can maintain potency for months to years. Once reconstituted, it behaves like any solution with a limited shelf life. If you need maximum flexibility and the longest storage window, choose lyophilized.
Can I tell if my tirzepatide has lost potency just by looking at it?
Not reliably. Significant potency loss can occur without visible changes. Cloudiness, particles, or color changes are definitive red flags, but clear, colorless solution is not a guarantee of full potency. Always respect expiration dates and beyond-use dates regardless of appearance.
What should I do with expired tirzepatide?
Do not inject it. Do not flush it down the toilet. Check with your pharmacy or local waste management for medication take-back programs. Many pharmacies accept expired medications for proper disposal. If no take-back program is available, the FDA recommends mixing the medication with an undesirable substance like coffee grounds or dirt, sealing it in a container, and placing it in household trash.
Does tirzepatide shelf life change at different doses?
The shelf life of the product itself does not change based on your dose. However, if you are using a lower dose from a multi-dose vial, the vial may last longer in terms of number of doses, potentially exceeding the beyond-use date before you finish it. In that case, the BUD is the limiting factor, not the number of remaining doses.
External resources
For researchers serious about protecting their investment and optimizing their protocols, SeekPeptides provides comprehensive storage guides, stability databases, and handling protocols for members. Whether you are working with tirzepatide, semaglutide, or any other peptide in your research, the difference between effective and wasted product often comes down to storage. SeekPeptides members access detailed protocols that cover every variable, from optimal storage conditions to reconstitution best practices, ensuring nothing goes to waste.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay cold, your peptides stay potent, and your shelf life never catch you off guard.