Feb 18, 2026
On paper, mazdutide and retatrutide look like they belong in the same category. Both target the glucagon receptor. Both reduce body weight at rates that dwarf first-generation GLP-1 agonists. Both emerged from pipelines designed to push metabolic therapy beyond what semaglutide and tirzepatide could achieve alone. But in practice? The similarities end faster than you might expect.
One is a dual agonist. The other is a triple agonist. One is already approved and available in China. The other is still working through Phase 3 trials with regulatory submission likely a year or more away. One was developed by Innovent Biologics with origins at Eli Lilly. The other remains fully within the Lilly pipeline as potentially the most powerful weight loss compound ever tested in humans.
The differences go deeper than receptor count. They show up in clinical trial design, in tolerability profiles, in the types of metabolic benefits each compound delivers, and in the practical timelines for when researchers might actually be able to access them. This guide breaks down every meaningful comparison between mazdutide and retatrutide, from molecular mechanisms to real trial data, so you can understand exactly how these two next-generation compounds stack up against each other and against the current standard treatments.
SeekPeptides has tracked both of these compounds through every phase of development, and the data tells a more nuanced story than most sources suggest. Let us walk through it.
How mazdutide and retatrutide actually work
Understanding the difference between these two compounds starts at the receptor level. Every peptide in the GLP-1 family works by binding to specific receptors on cells throughout the body. The number and type of receptors a compound targets determines what it can do.
Mazdutide targets two receptors
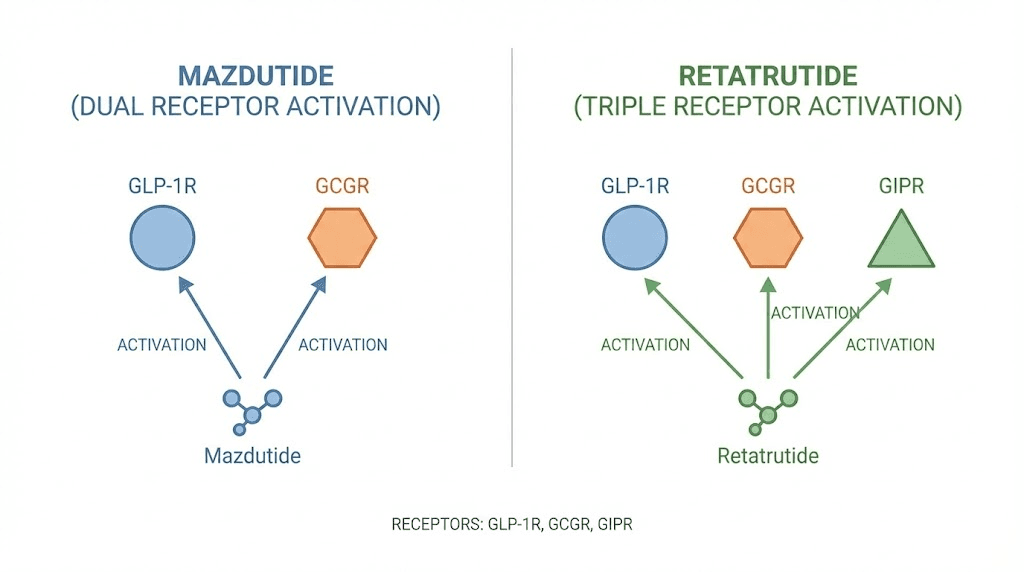
Mazdutide (IBI362) is a synthetic peptide related to mammalian oxyntomodulin. It activates two receptor types simultaneously. The GLP-1 receptor and the glucagon receptor.
GLP-1 receptor activation does the heavy lifting for appetite suppression. It slows gastric emptying, which means food stays in the stomach longer and you feel full sooner. It enhances insulin secretion in response to meals. It signals satiety to the brain through pathways in the hypothalamus. These are the same mechanisms that make semaglutide effective, and mazdutide shares all of them.
The glucagon receptor activation is where things get interesting. Glucagon is the hormone your body uses to mobilize stored energy. When glucagon receptors activate, the liver breaks down glycogen into glucose and ramps up fatty acid oxidation. In practical terms, this means your body burns more fat for fuel even at rest. It also means a higher basal metabolic rate.
This dual mechanism creates a two-pronged approach. Eat less through GLP-1 suppression. Burn more through glucagon activation.
Innovent Biologics attached a fatty acid side chain to mazdutide to extend its half-life, allowing once-weekly dosing. The compound received marketing approval in China in June 2025 under the brand name Xinermei, making it the first domestically approved dual GLP-1/glucagon agonist anywhere in the world.
Retatrutide targets three receptors
Retatrutide (LY3437943) takes the concept further. Developed entirely by Eli Lilly, it activates GLP-1 receptors, glucagon receptors, and GIP (glucose-dependent insulinotropic polypeptide) receptors. This makes it a triple agonist, sometimes called a "triple G" agonist for targeting all three pathways.
The GLP-1 and glucagon mechanisms work the same way they do in mazdutide. Appetite suppression plus enhanced fat oxidation. But the addition of GIP receptor activation adds a third dimension that neither mazdutide nor any dual agonist can replicate.
GIP receptor activation enhances insulin sensitivity at the cellular level. It improves how efficiently your body processes glucose after meals. In animal models, GIP activation has been shown to reduce inflammation in adipose (fat) tissue and potentially improve how the body stores and mobilizes fat. The combination of all three receptors firing simultaneously creates what researchers describe as synergistic metabolic effects, meaning the total impact exceeds what you would get from activating each receptor individually.
Retatrutide has a half-life of approximately 6 days, which supports once-weekly subcutaneous injection. It remains investigational and has not been approved in any country as of early 2026.
Why the third receptor matters
The question everyone asks is simple. Does that third receptor actually make a difference?
The clinical data says yes. Convincingly.
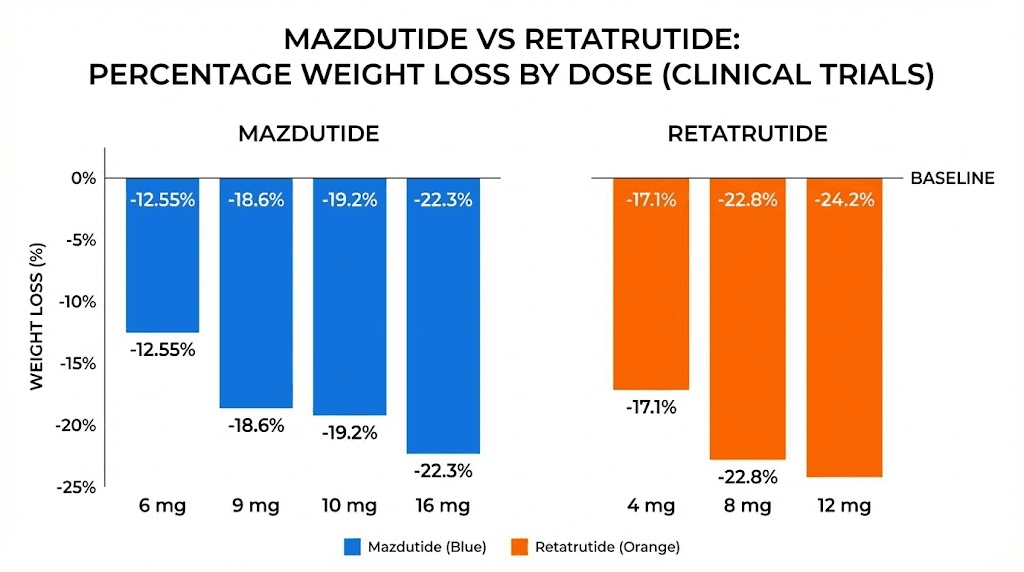
Tirzepatide, which targets GLP-1 and GIP (but not glucagon), produces weight loss results of roughly 20-22% at its highest doses over 72 weeks. Mazdutide, targeting GLP-1 and glucagon (but not GIP), reaches about 14-20% weight loss depending on dose and trial duration. Retatrutide, hitting all three receptors, delivered 24.2% in Phase 2 and up to 28.7% in Phase 3.
That gap between mazdutide and retatrutide is not trivial. For a 250-pound person, the difference between 14% weight loss and 28% weight loss is the difference between losing 35 pounds and losing 70 pounds. In the TRIUMPH-4 trial, participants on the 12 mg dose of retatrutide lost an average of 71.2 pounds over 68 weeks.
The mechanism that likely drives this gap is the combination of GIP and glucagon receptor activation working together. GIP appears to amplify the metabolic benefits of glucagon activation in ways that neither receptor achieves alone. This synergy, combined with powerful GLP-1 appetite suppression, creates the most potent weight loss effect seen in any clinical trial to date.
Clinical trial results compared head to head
Neither Innovent Biologics nor Eli Lilly has run a direct head-to-head trial comparing mazdutide to retatrutide. The data we have comes from separate trial programs with different designs, different patient populations, and different endpoints. That context matters when comparing numbers. Still, the weight loss results tell a clear story when you lay them side by side.
Mazdutide weight loss data
Mazdutide has been studied across multiple Phase 2 and Phase 3 trials, primarily in Chinese populations. Here is what the key trials showed.
The GLORY-1 trial was a Phase 3 study published in the New England Journal of Medicine. At week 48, participants on mazdutide 4 mg lost an average of 11.0% of their body weight. The 6 mg group lost 14.01%. The placebo group gained 0.3%. Nearly half of participants in the 6 mg group (49.5%) achieved at least 15% weight reduction, compared to just 2% on placebo.
The GLORY-2 trial tested the higher 9 mg dose. Results were even more impressive. Participants without type 2 diabetes who received mazdutide 9 mg lost an average of 20.08% of their body weight at week 60. Among these participants, 48.7% achieved at least 20% weight reduction versus 3.1% on placebo. The trial also met all key secondary endpoints, including improvements in waist circumference, systolic blood pressure, triglycerides, non-HDL cholesterol, LDL cholesterol, and serum uric acid.
The DREAMS-3 trial put mazdutide directly against semaglutide in a head-to-head comparison. At 32 weeks, 48.0% of participants on mazdutide 6 mg achieved both HbA1c below 7% and at least 10% body weight reduction, compared to just 21.0% on semaglutide 1 mg. Mean weight loss was 10.29% with mazdutide versus 6.00% with semaglutide. Mazdutide beat semaglutide on both glycemic control and weight loss in the same trial.
A Phase 1 high-dose trial explored mazdutide at 16 mg and found it well tolerated with even greater weight loss, suggesting the ceiling for mazdutide efficacy has not been reached.
Retatrutide weight loss data
Retatrutide has produced the largest weight loss numbers ever recorded in a clinical trial for an anti-obesity medication. The data from both Phase 2 and Phase 3 programs is remarkable.
The Phase 2 trial, published in the New England Journal of Medicine, tested doses from 1 mg to 12 mg over 48 weeks. The results were dose-dependent and dramatic. The 1 mg group lost 8.7% of body weight. The combined 4 mg groups lost 17.1%. The 8 mg groups lost 22.8%. And the 12 mg group lost 24.2%, with participants still losing weight at the end of the trial period, meaning the maximum effect had not been reached.
The TRIUMPH-4 Phase 3 trial delivered even bigger numbers. Over 68 weeks, the 12 mg group lost an average of 28.7% of their body weight, equivalent to an average of 71.2 pounds. The 9 mg group lost 26.4%. These numbers exceeded the Phase 2 results, partly because the trial ran longer (68 weeks versus 48 weeks) and partly because the dose escalation schedule was refined.
TRIUMPH-4 also found significant benefits beyond weight loss. Participants experienced a 75.8% reduction in knee osteoarthritis pain scores. More than 1 in 8 participants treated with retatrutide became completely free of knee pain by the end of the trial. Physical function improved significantly across all measures.
Seven additional Phase 3 trials from the TRIUMPH program are expected to report results throughout 2026, including trials in type 2 diabetes, obstructive sleep apnea, and general obesity without specific comorbidities.
Putting the numbers in context
Here is a side-by-side comparison of peak weight loss results from both compounds.
Measure | Mazdutide (9 mg, GLORY-2) | Retatrutide (12 mg, TRIUMPH-4) |
|---|---|---|
Mean weight loss | 20.08% at 60 weeks | 28.7% at 68 weeks |
Trial population | Chinese adults with obesity | Adults with obesity and knee OA |
Participants achieving 20%+ loss | 48.7% | Not yet reported for TRIUMPH-4 |
Dosing frequency | Once weekly | Once weekly |
Mechanism | Dual (GLP-1 + glucagon) | Triple (GLP-1 + GIP + glucagon) |
Trial phase | Phase 3 (approved in China) | Phase 3 (investigational) |
Direct comparison is imperfect because the trials enrolled different populations, ran for different durations, and used different primary endpoints. The GLORY trials enrolled exclusively Chinese participants, while the TRIUMPH trials enrolled participants primarily in the United States and Europe. Genetic, dietary, and lifestyle differences between these populations could influence outcomes. The TRIUMPH-4 trial also ran 8 weeks longer than GLORY-2, giving retatrutide more time to produce results.
Still, the 8-9 percentage point gap between peak results from each compound is consistent with what you would expect from a dual versus triple agonist approach. Retatrutide consistently produces more weight loss at every comparable timepoint.
Side effects and tolerability compared
Both mazdutide and retatrutide cause gastrointestinal side effects. That is the price of activating GLP-1 receptors. Every compound in this class, from semaglutide to tirzepatide to these newer agents, causes nausea, vomiting, and diarrhea to some degree. The question is how severe, how long, and how many people quit because of them.
Mazdutide tolerability profile
Mazdutide has shown an impressively clean tolerability profile across multiple trials. In the GLORY-1 trial, the most frequently reported adverse events were gastrointestinal and mostly mild to moderate in severity. Nausea, diarrhea, and vomiting were the most common complaints, occurring primarily during dose escalation periods.
Here is the critical finding. In the Phase 1b trial of higher doses (9 mg and 10 mg), 95% of participants experienced at least one adverse event, but only one participant discontinued treatment due to side effects. One person. Out of the entire trial.
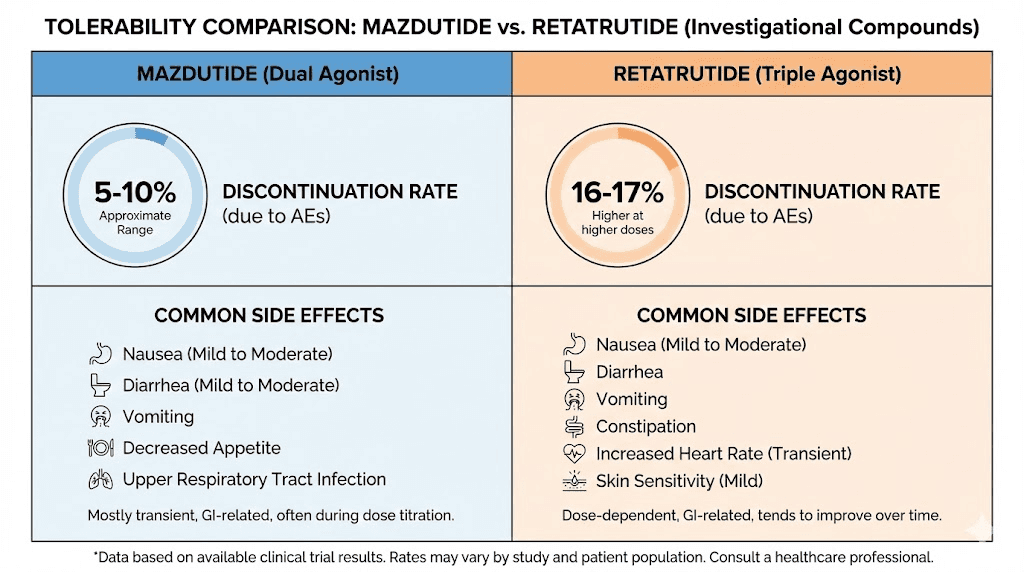
That discontinuation rate is remarkably low for this class of drugs. For context, semaglutide and tirzepatide typically see 4-7% discontinuation rates due to adverse events in their trials. Mazdutide appears to produce side effects that are real but manageable enough that almost nobody stops treatment.
The temporal pattern matters too. Diarrhea, nausea, and vomiting occurred more frequently during dose escalation and declined gradually over time. By the time participants reached their maintenance dose, the worst of the gastrointestinal symptoms had typically resolved. No serious adverse events were reported in the early trials, and the safety profile has remained consistent across the larger Phase 3 programs.
One notable finding is that all participants on mazdutide experienced increased heart rates. This is consistent with glucagon receptor activation, which has known cardiovascular effects. Heart rate increases were not associated with adverse cardiac events in the trials, but it is something researchers monitor closely.
Retatrutide tolerability profile
Retatrutide causes more gastrointestinal side effects than mazdutide, which is consistent with its more potent receptor activation profile. In the TRIUMPH-4 Phase 3 trial, the most common adverse events for the 12 mg dose were nausea (43.2%), diarrhea (33.1%), constipation (25.0%), vomiting (20.9%), and decreased appetite.
The discontinuation rates tell the most important part of the story. In TRIUMPH-4, 18.2% of participants on retatrutide 12 mg discontinued due to adverse events, compared to 12.2% on the 9 mg dose and just 4.0% on placebo. That 18.2% rate is notably higher than what mazdutide produces, and higher than what tirzepatide shows at its highest doses as well.
The gastrointestinal side effects follow a similar temporal pattern to mazdutide, with symptoms being most intense during dose escalation and improving over time. But the peak severity and the dropout rate suggest that the triple receptor activation, while more effective for weight loss, comes at a real cost in terms of tolerability.
A BioSpace report noted that a new safety signal emerged from the TRIUMPH program, though details were limited in the topline data release. Full safety data from all seven TRIUMPH trials will provide a more complete picture as results are published throughout 2026.
Side-by-side tolerability comparison
Side effect | Mazdutide | Retatrutide |
|---|---|---|
Nausea | Common, mild to moderate | 38-43%, moderate to severe possible |
Diarrhea | Common during escalation | 33-35% |
Vomiting | Less common | 20-21% |
Constipation | Less commonly reported | 22-25% |
Discontinuation rate | Very low (near 1% in early trials) | 12-18% depending on dose |
Heart rate increase | Yes, consistent finding | Not prominently reported |
Severity pattern | Worst during escalation, resolves | Worst during escalation, mostly resolves |
The tolerability difference is significant and practical. If you are someone who struggles with gastrointestinal side effects on existing GLP-1 agonists, mazdutide may offer a more comfortable experience. If maximum weight loss is the priority and you can tolerate more side effects, retatrutide delivers substantially better results.
Dosing protocols and administration
Both mazdutide and retatrutide are administered as once-weekly subcutaneous injections. The dosing schedules follow the same general principle used across all GLP-1 receptor agonists: start low, escalate gradually, reach a maintenance dose. But the specific numbers and titration timelines differ.
Mazdutide dosing
Clinical trials have tested mazdutide at several dose levels. The approved doses in China are 4 mg and 6 mg for weight management, with a 9 mg dose under review for moderate-to-severe obesity.
The titration schedule from clinical trials follows a gradual escalation. Participants typically start at a lower dose (1.5 mg or 3 mg in Phase 2 trials) and increase every four weeks until reaching their target dose. The GLORY-1 trial used 4 mg and 6 mg maintenance doses. The GLORY-2 trial used a 9 mg maintenance dose.
The Phase 1 high-dose trial explored doses up to 16 mg, finding that the compound remained well tolerated even at these higher levels. This suggests that future regulatory applications could push the approved dose ceiling higher for patients with severe obesity who need more aggressive intervention.
For researchers interested in reconstitution and preparation, mazdutide follows similar handling requirements to other peptide-based GLP-1 agonists. Proper storage and handling protocols are essential for maintaining peptide integrity.
Retatrutide dosing
Retatrutide has been studied at doses ranging from 0.5 mg to 12 mg in clinical trials. The TRIUMPH Phase 3 program uses 9 mg and 12 mg as the primary therapeutic doses, with a 4 mg maintenance dose being explored for weight maintenance after initial loss.
The titration schedule from TRIUMPH-4 started all participants at 2 mg once weekly. For the 9 mg target, doses increased in steps at 2 mg, 4 mg, and 6 mg, with increases every four weeks until reaching 9 mg. For the 12 mg target, the escalation went through 2 mg, 4 mg, 6 mg, and 9 mg steps before reaching the full 12 mg dose.
Retatrutide has a half-life of approximately 6 days, which supports the once-weekly dosing schedule. The compound is designed for subcutaneous injection, typically administered in the abdomen, thigh, or upper arm, following the same injection site recommendations used for other GLP-1 agonists.
For researchers working with lyophilized peptides, understanding reconstitution ratios for retatrutide is critical. The retatrutide dosage calculator on SeekPeptides can help determine exact amounts based on your vial concentration and target dose.
Dosing comparison
Parameter | Mazdutide | Retatrutide |
|---|---|---|
Route | Subcutaneous injection | Subcutaneous injection |
Frequency | Once weekly | Once weekly |
Starting dose | 1.5-3 mg (trial dependent) | 2 mg |
Therapeutic doses | 4 mg, 6 mg, 9 mg | 9 mg, 12 mg (4 mg maintenance) |
Maximum tested | 16 mg (Phase 1) | 12 mg (Phase 3) |
Escalation interval | Every 4 weeks | Every 4 weeks |
Half-life | Extended via fatty acid chain | ~6 days |
Metabolic benefits beyond weight loss
Weight loss numbers grab headlines, but both of these compounds do far more than reduce body weight. The metabolic benefits they produce have implications for cardiovascular disease, liver health, diabetes management, and more. And here, the differences between dual and triple agonism create meaningfully different benefit profiles.
Mazdutide metabolic benefits
Mazdutide has demonstrated broad metabolic improvements across its trial programs.
In the GLORY-1 and GLORY-2 trials, mazdutide produced beneficial effects on every prespecified cardiometabolic measure. Waist circumference decreased significantly. Systolic blood pressure improved. Triglycerides dropped. Non-HDL cholesterol, LDL cholesterol, and serum uric acid all improved.
The DREAMS-3 head-to-head trial against semaglutide showed mazdutide achieving superior glycemic control. The mean HbA1c reduction from baseline was 2.03% with mazdutide versus 1.84% with semaglutide. For patients with type 2 diabetes, that difference in glucose control translates to meaningful clinical outcomes over time.
An exploratory analysis from the GLORY-1 trial found something particularly noteworthy. Mazdutide produced an 80.2% reduction in liver fat content. This was presented at the American Diabetes Association meeting and suggests that mazdutide, through its glucagon receptor activation, drives significant hepatic fat reduction. This has major implications for patients with metabolic dysfunction-associated steatotic liver disease (MASLD, formerly known as NAFLD).
The glucagon receptor component of mazdutide is likely responsible for much of the liver benefit. Glucagon promotes hepatic fatty acid oxidation, essentially directing the liver to burn its own fat stores. This mechanism operates independently of weight loss, meaning the liver benefits may exceed what you would expect from the degree of weight reduction alone.
Retatrutide metabolic benefits
Retatrutide has demonstrated even more dramatic metabolic benefits, particularly for liver health and insulin sensitivity.
In a dedicated Phase 2a liver fat trial published in Nature Medicine, retatrutide produced dose-dependent reductions in liver fat that are unlike anything previously seen. At 24 weeks, the mean relative change from baseline in liver fat was a 42.9% reduction in the 1 mg group, a 57.0% reduction at 4 mg, an 81.4% reduction at 8 mg, and an 82.4% reduction at the 12 mg dose. The placebo group showed a 0.3% increase.
Even more striking, 86% of participants on the 12 mg dose achieved normal liver fat levels (below 5%) at 24 weeks. At 48 weeks, more than 90% of the 12 mg group had resolved their steatosis entirely. For a condition that affects roughly 30% of the global population and has limited treatment options, these results are extraordinary.
The insulin sensitivity improvements were equally impressive. Retatrutide at doses of 4 mg or higher significantly reduced fasting insulin (by 37% to 71%), HOMA2-IR (a measure of insulin resistance, reduced by 36% to 69%), and increased adiponectin (by 30% to 99%). It also reduced triglycerides by 35-40%, leptin by 29-56%, and FGF-21 by 52-66%.
Beta-hydroxybutyrate, a marker of fatty acid oxidation, increased by 78% to 181%, confirming that retatrutide drives substantial fat burning at the cellular level. These metabolic shifts were significantly related to changes in body weight, abdominal fat, and measures of insulin sensitivity and lipid metabolism.
The combination of GIP and glucagon receptor activation appears to produce additive benefits for liver fat reduction and metabolic health that neither receptor achieves alone. This is one area where the triple agonist approach clearly outperforms dual agonism.
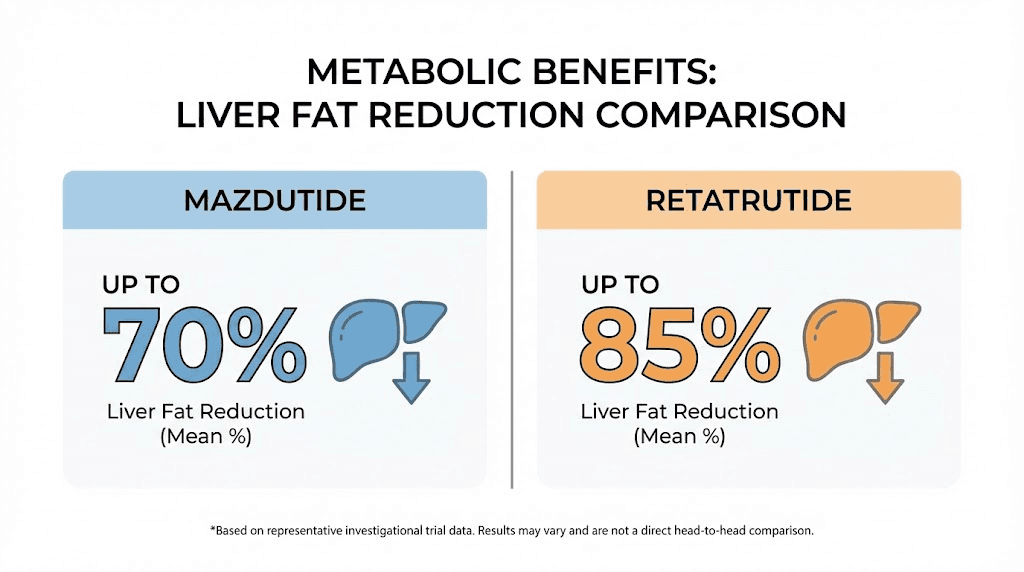
Metabolic benefits summary
Benefit | Mazdutide | Retatrutide |
|---|---|---|
Liver fat reduction | 80.2% (GLORY-1 exploratory) | 82.4% at 24 weeks, 90%+ resolution at 48 weeks |
HbA1c reduction | 2.03% (beat semaglutide) | Significant, data pending full Phase 3 |
Insulin sensitivity | Improved | 37-71% fasting insulin reduction |
Triglycerides | Improved | 35-40% reduction |
Blood pressure | Improved | Improved |
Cardiovascular markers | Broad improvement | Broad improvement |
MASLD/liver disease | Strong evidence | Strongest evidence of any drug tested |
Development timeline and availability
One of the most practical differences between mazdutide and retatrutide is simple: one is already available and the other is not. For researchers and clinicians making decisions today, timeline matters as much as efficacy data.
Mazdutide availability
Mazdutide received approval in China in June 2025, marketed under the brand name Xinermei by Innovent Biologics. The approved doses are 4 mg and 6 mg for chronic weight management. A supplementary application for the 9 mg dose has been accepted for review by China NMPA, based on the GLORY-2 results showing 20% weight loss.
The compound was originally developed by Eli Lilly and licensed to Innovent Biologics for development and commercialization in China. This means mazdutide is currently available only in the Chinese market. There are no active regulatory submissions in the United States, Europe, or other major markets as of early 2026.
For the international research community, this limits direct access to the approved product. However, the compound is available through research peptide suppliers for investigational purposes. Researchers working with mazdutide should ensure they source from vendors with proper third-party testing and quality documentation.
Innovent has ongoing clinical programs that could support regulatory filings in additional markets, but no specific timeline has been announced for US or European submissions.
Retatrutide availability
Retatrutide availability remains limited to clinical trials and the research peptide market. Eli Lilly has not submitted regulatory applications in any country as of early 2026.
The TRIUMPH Phase 3 program includes eight separate trials. TRIUMPH-4 was the first to report positive results in December 2025. Seven additional trials are expected to complete throughout 2026, covering obesity, type 2 diabetes, obstructive sleep apnea, and other indications.
Industry analysts, including GlobalData, predict FDA approval for retatrutide in 2027, with a projected sales forecast of $15.6 billion by 2031. This timeline assumes successful completion of the remaining TRIUMPH trials and a standard FDA review process. If the results are as strong as TRIUMPH-4 suggests, Lilly could potentially seek priority review, which would shorten the timeline by several months.
For researchers who want access now, purchasing retatrutide through the research peptide market is possible but requires careful attention to sourcing, purity testing, and proper handling. The cost of retatrutide varies significantly between vendors, and SeekPeptides provides comparison tools to help navigate options.
Timeline comparison
Milestone | Mazdutide | Retatrutide |
|---|---|---|
Phase 2 completed | Yes | Yes |
Phase 3 completed | Yes (GLORY-1, GLORY-2, DREAMS) | Partially (TRIUMPH-4, others ongoing) |
Approved anywhere | Yes (China, June 2025) | No |
US/EU regulatory filing | No timeline announced | Expected 2026-2027 |
Predicted US approval | Unknown | 2027 (analyst estimate) |
Research availability | Limited, growing | Available from research vendors |
Who developed each compound and why it matters
The corporate backing behind a pharmaceutical compound affects everything from clinical trial quality to eventual pricing and global availability. Understanding who stands behind mazdutide and retatrutide provides important context for evaluating their long-term prospects.
Mazdutide: Innovent Biologics (licensed from Eli Lilly)
Mazdutide was originally discovered by Eli Lilly, then licensed to Innovent Biologics, a Chinese biopharmaceutical company, for development and commercialization in China. Innovent has run all of the GLORY and DREAMS clinical trials, focusing primarily on the Chinese population.
This licensing arrangement has pros and cons. Innovent moved quickly through the Chinese regulatory process and brought mazdutide to market faster than most Western pharmaceutical timelines would allow. China approved the drug in June 2025, and the higher-dose application is already under review.
The downside is that Innovent is a smaller company with less global infrastructure than Eli Lilly. This means international expansion, if it happens, will be slower and may require partnerships with additional pharmaceutical companies in different regions. The clinical trial data is also primarily from Chinese populations, which some Western regulators may want supplemented with multi-ethnic data before granting approval.
Retatrutide: Eli Lilly (fully owned)
Retatrutide is developed and fully owned by Eli Lilly, one of the largest pharmaceutical companies in the world. Lilly also manufactures tirzepatide (Mounjaro/Zepbound), giving them unmatched experience in the GLP-1 agonist space.
Lilly has the resources to run large, global Phase 3 programs across multiple indications simultaneously. The TRIUMPH program includes eight trials enrolling thousands of participants across multiple countries. This scale of development virtually guarantees comprehensive safety and efficacy data that will satisfy regulators worldwide.
Lilly also has the manufacturing capacity and distribution network to launch retatrutide globally once approved. Given the company projected $15.6 billion in annual sales for the compound, the commercialization effort will be massive. This means broader access, more formulation options, and likely competitive pricing strategies designed to capture market share from existing GLP-1 agonists.
Mazdutide and retatrutide compared to current treatments
Neither of these compounds exists in a vacuum. To understand their significance, you need to see how they compare to the treatments already available: semaglutide and tirzepatide.
How mazdutide compares to semaglutide
Mazdutide has been directly compared to semaglutide in the DREAMS-3 head-to-head trial. The results were clear: mazdutide 6 mg produced greater weight loss (10.29% vs 6.00%) and better glycemic control (HbA1c reduction of 2.03% vs 1.84%) than semaglutide 1 mg over 32 weeks.
The mechanism explains why. Semaglutide only activates GLP-1 receptors. Mazdutide adds glucagon receptor activation, which drives additional fat burning and metabolic benefits that semaglutide simply cannot produce. For anyone currently on semaglutide who wants better results, mazdutide represents a mechanistic upgrade.
The caveat is that semaglutide 1 mg is not the highest available dose. Semaglutide 2.4 mg (the Wegovy dose for weight management) produces roughly 15-17% weight loss in clinical trials, which is closer to mazdutide 6 mg results. A fair comparison would require mazdutide to be tested against semaglutide 2.4 mg, which has not happened yet.
How retatrutide compares to tirzepatide
Tirzepatide is a dual agonist targeting GLP-1 and GIP receptors. Retatrutide adds glucagon receptor activation on top of those same two targets. In the SURMOUNT-1 trial, tirzepatide 15 mg produced 22.5% weight loss at 72 weeks. Retatrutide 12 mg produced 28.7% at 68 weeks in TRIUMPH-4.
That 6+ percentage point gap is clinically meaningful. For a 220-pound person, it represents roughly 13 additional pounds of weight loss. And remember, retatrutide achieved its results in fewer weeks. The Phase 2 data showed weight loss curves that had not plateaued, meaning longer treatment durations could push the numbers even higher.
The glucagon component in retatrutide likely accounts for the additional weight loss over tirzepatide. While tirzepatide uses GIP to enhance insulin sensitivity and metabolic efficiency, retatrutide adds direct fat-burning stimulation through glucagon. This creates a three-pronged attack: suppress appetite (GLP-1), improve metabolic efficiency (GIP), and directly stimulate fat oxidation (glucagon).
Where mazdutide sits in the landscape
Mazdutide occupies an interesting middle ground. It targets a different two-receptor combination than tirzepatide (glucagon instead of GIP), which gives it unique metabolic benefits, particularly for liver fat. But it produces less weight loss than retatrutide, which targets all three receptors.
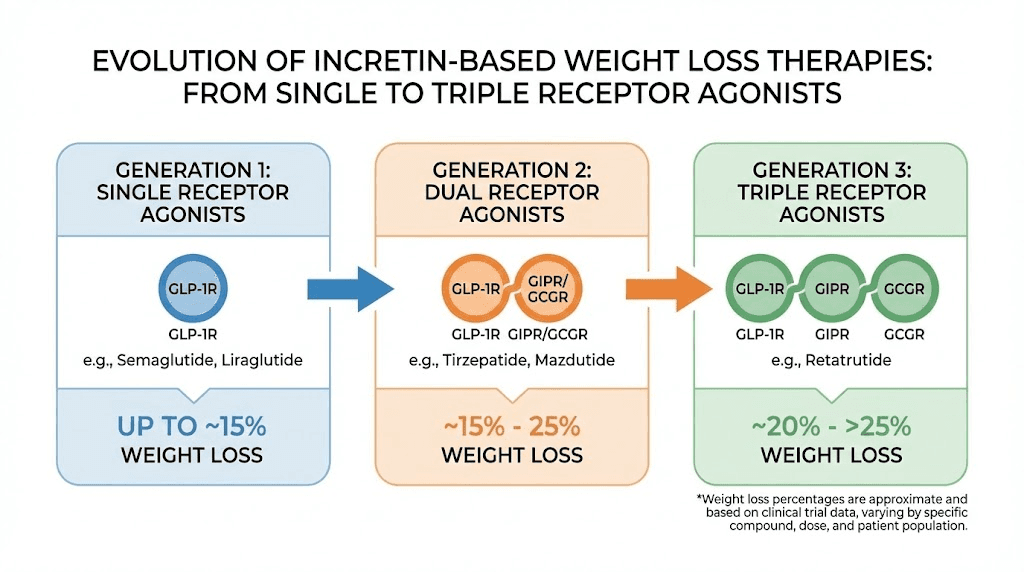
Think of it this way. If semaglutide (single agonist) is first generation, and tirzepatide and mazdutide (dual agonists) are second generation, then retatrutide (triple agonist) is the beginning of the third generation. Each step adds receptor targets and produces incrementally better results.
Compound | Receptors | Peak weight loss | Approval status |
|---|---|---|---|
Semaglutide 2.4 mg | GLP-1 | ~15-17% | Approved globally |
Tirzepatide 15 mg | GLP-1 + GIP | ~22.5% | Approved globally |
Mazdutide 9 mg | GLP-1 + Glucagon | ~20% | Approved in China |
Retatrutide 12 mg | GLP-1 + GIP + Glucagon | ~28.7% | Phase 3 (investigational) |
Liver health and MASLD: a critical differentiator
Both compounds reduce liver fat, but this is one area where the data is detailed enough to make meaningful comparisons. And it is an increasingly important comparison, because metabolic dysfunction-associated steatotic liver disease (MASLD) affects an estimated 1.7 billion people worldwide.
Mazdutide liver benefits
The exploratory analysis from GLORY-1 showed mazdutide producing an 80.2% reduction in liver fat content. This is a substantial finding. Glucagon receptor activation drives hepatic fatty acid oxidation, essentially forcing the liver to burn its stored fat. Since mazdutide activates glucagon receptors as part of its core mechanism, liver fat reduction is a built-in benefit of the compound.
For patients with MASLD, this means mazdutide could serve double duty: treating obesity while simultaneously addressing the liver disease that often accompanies it. No separate medication would be needed. The gut and liver health connection makes this particularly relevant for the growing population dealing with metabolic syndrome.
Retatrutide liver benefits
Retatrutide has the strongest liver fat reduction data of any compound ever tested. In the dedicated MASLD Phase 2a trial, 86% of participants on 12 mg achieved normal liver fat levels at 24 weeks. By 48 weeks, more than 90% had resolved their steatosis entirely.
The mean relative reduction in liver fat at 24 weeks was 82.4% for the 12 mg dose. This was not a secondary endpoint buried in supplementary data. It was a primary finding from a dedicated liver trial published in Nature Medicine. The reduction was consistent across dose levels, with even the 4 mg dose producing a 57% reduction.
Retatrutide also reduced biomarkers associated with liver inflammation and fibrosis progression. The increases in beta-hydroxybutyrate (78-181%) confirmed that the fatty acid oxidation mechanism was driving these changes at the cellular level. The reductions in FGF-21 and improvements in adiponectin further supported the metabolic mechanism behind the liver benefits.
For the millions of people with MASLD who currently have no approved pharmacological treatment, retatrutide represents potentially transformative therapy. Eli Lilly is likely considering MASLD as a separate indication for regulatory submission, which could accelerate the timeline for liver-specific approval.
Cardiovascular considerations
Both mazdutide and retatrutide improve cardiovascular risk markers, but the specifics differ in ways that matter for different patient populations.
Blood pressure effects
Both compounds reduce systolic blood pressure. This is partially mediated by weight loss (less body mass means less cardiovascular work) and partially by direct metabolic effects. Improved insulin sensitivity reduces the inflammatory burden on blood vessels, and reduced triglycerides lower the atherogenic lipid profile.
Lipid improvements
Retatrutide has the more detailed lipid data. Triglycerides decreased by 35-40%, which exceeds the reductions seen with semaglutide or tirzepatide at comparable timepoints. Non-HDL cholesterol and LDL cholesterol both improved in the GLORY trials with mazdutide, and similar improvements are expected (though less specifically reported) with retatrutide.
Heart rate considerations
Mazdutide consistently increases heart rate across all trial participants. This is a known effect of glucagon receptor activation and is being monitored closely. While no adverse cardiac events were attributed to heart rate increases in the trials, long-term cardiovascular outcomes data is needed.
Retatrutide has not prominently reported heart rate increases in its topline data, though the full Phase 3 safety database will provide more detail. The presence of GIP receptor activation in retatrutide may modulate the cardiovascular effects of glucagon differently than in mazdutide, where glucagon acts without GIP counterbalance.
Neither compound has completed dedicated cardiovascular outcomes trials (CVOTs), which are typically required post-approval. The long-term cardiovascular safety profile of both compounds remains to be fully characterized.
Diabetes management potential
Both compounds show strong potential for managing type 2 diabetes alongside obesity. The GLP-1 receptor activation in both drugs improves insulin secretion and glycemic control. But the additional receptor targets create distinct profiles for diabetes management.
Mazdutide for diabetes
Mazdutide has been studied specifically in type 2 diabetes patients through the DREAMS trial series. The compound is approved in China for type 2 diabetes management based on the DREAMS-1 and DREAMS-2 studies.
In DREAMS-3, mazdutide achieved an HbA1c reduction of 2.03%, which was statistically superior to semaglutide 1 mg (1.84% reduction). The dual composite endpoint of HbA1c below 7% plus 10% or more weight loss was achieved by 48% of mazdutide patients versus 21% on semaglutide.
Glucagon receptor activation in mazdutide creates an interesting dynamic for diabetes. Glucagon traditionally raises blood sugar by promoting hepatic glucose production. However, when combined with GLP-1 receptor activation and the resulting insulin enhancement, the net effect is improved glycemic control rather than hyperglycemia. The glucagon component may also contribute to reduced hepatic lipid accumulation, which improves hepatic insulin sensitivity over time.
Retatrutide for diabetes
In the Phase 2 trial in participants with type 2 diabetes, retatrutide produced significant HbA1c reductions and improved glycemic control across all dose levels. The TRIUMPH program includes dedicated diabetes trials that will provide more comprehensive data.
The triple agonist mechanism may offer unique advantages for diabetes management. GIP receptor activation enhances insulin secretion in a glucose-dependent manner, meaning it boosts insulin when blood sugar is high but has minimal effect when blood sugar is normal. This reduces the risk of hypoglycemia, a common concern with diabetes medications.
The addition of glucagon receptor activation in retatrutide, combined with GIP, creates a metabolic environment that simultaneously improves insulin sensitivity, enhances appropriate insulin secretion, and increases energy expenditure. For type 2 diabetes patients with significant obesity, this comprehensive metabolic reset could potentially reduce or eliminate the need for multiple diabetes medications.
The research community perspective
For the research community currently working with peptide compounds, both mazdutide and retatrutide represent significant leaps beyond current options. The practical considerations for researchers differ from the clinical perspective in important ways.
Sourcing and quality
Neither compound is widely available through the established research peptide supply chain that provides compounds like BPC-157, TB-500, or GHK-Cu. Retatrutide is more readily available from research vendors than mazdutide, partly because it has been in the research community longer and partly because Lilly-originated compounds tend to reach research suppliers faster.
Quality verification is essential for both compounds. Third-party testing through accredited laboratories should confirm identity, purity, and absence of contaminants before any research use. Mass spectrometry and HPLC analysis are the standard verification methods for peptide compounds of this complexity.
Reconstitution and storage
Both compounds follow similar reconstitution protocols to other peptide-based GLP-1 agonists. Lyophilized powder is reconstituted with bacteriostatic water, stored refrigerated, and administered via subcutaneous injection.
For retatrutide specifically, the retatrutide dosage calculator can help determine appropriate reconstitution volumes based on vial sizes and target concentrations. The retatrutide reconstitution chart provides visual guidance for common vial configurations.
Proper peptide storage is critical for maintaining compound integrity. Both mazdutide and retatrutide should be stored as lyophilized powder at -20C for long-term storage, or refrigerated at 2-8C after reconstitution. Reconstituted solutions should be used within 28-30 days. Avoid repeated freeze-thaw cycles. Protect from light. Standard post-reconstitution storage protocols apply.
Cost considerations
The cost of retatrutide in the research market varies significantly between vendors. Because neither compound is available as a commercial pharmaceutical product outside China, pricing follows research peptide market dynamics rather than pharmaceutical pricing models.
Researchers should compare prices across multiple verified vendors, factoring in purity levels, testing documentation, and shipping conditions. The peptide cost calculator on SeekPeptides can help standardize cost comparisons across different vendors and vial sizes.
Which compound fits which goals
After examining the clinical data, mechanisms, side effects, and availability, the question becomes practical. Which compound makes more sense for different goals and situations?
Choose mazdutide if
Tolerability is your primary concern. Mazdutide has demonstrated an exceptionally low discontinuation rate, meaning the side effects are real but manageable for nearly everyone. If you have struggled with gastrointestinal side effects on semaglutide or tirzepatide, mazdutide may offer a more comfortable experience with meaningful weight loss and metabolic benefits.
Liver health is a priority. Both compounds reduce liver fat, but mazdutide provides strong hepatic benefits through its glucagon component without the additional gastrointestinal burden of triple agonism. For researchers focused specifically on MASLD, mazdutide offers an excellent risk-benefit profile.
Diabetes management with weight loss is the goal. The DREAMS-3 data showing superiority over semaglutide for the dual endpoint of glycemic control and weight loss makes mazdutide particularly attractive for the diabetes population.
You want an approved compound. Mazdutide is approved and commercially available in China. While access outside China is limited, the regulatory pathway has been established and the safety database is substantial.
Choose retatrutide if
Maximum weight loss is the priority. Nothing else comes close to retatrutide for sheer magnitude of weight reduction. The 28.7% average weight loss at 68 weeks exceeds every other compound that has been tested in rigorous clinical trials. If you need the most aggressive weight loss intervention available, retatrutide is it.
Severe obesity requires maximum intervention. For individuals with BMI above 40 or 50, the difference between 20% and 28% weight loss can be the difference between still being classified as obese and reaching a healthy weight range. The TRIUMPH-4 trial enrolled participants with BMI of 35 or higher, and the average weight loss of 71 pounds demonstrates the compound ability to make transformative changes in severe obesity.
Comprehensive metabolic improvement is needed. The triple agonist mechanism produces the broadest range of metabolic benefits: superior liver fat reduction, dramatic insulin sensitivity improvement, significant lipid improvements, and meaningful blood pressure reduction. For patients with metabolic syndrome affecting multiple organ systems, retatrutide addresses more pathways simultaneously.
You can manage significant gastrointestinal side effects. The 18% discontinuation rate at the highest dose means roughly 1 in 5 people cannot tolerate retatrutide at maximum dose. If you are among the 82% who can, the results are remarkable. But going in with realistic expectations about side effects is important.
Decision matrix
Priority | Better choice | Why |
|---|---|---|
Maximum weight loss | Retatrutide | 28.7% vs 20% peak results |
Best tolerability | Mazdutide | Near-zero discontinuation rate |
Liver fat reduction | Retatrutide (slight edge) | 90%+ resolution at 48 weeks |
Diabetes management | Mazdutide (more data) | Head-to-head superiority vs semaglutide |
Current availability | Mazdutide | Approved in China since June 2025 |
US/EU future access | Retatrutide | Eli Lilly global filing expected 2026-2027 |
Severe obesity (BMI 40+) | Retatrutide | 71 lb average loss in TRIUMPH-4 |
Moderate weight management | Mazdutide | Good results with better tolerability |
Safety considerations researchers should know
Both compounds are newer than established GLP-1 agonists, which means their long-term safety profiles are still being characterized. Researchers should be aware of several important safety considerations.
Gastrointestinal risks
The gastrointestinal side effects of both compounds can lead to dehydration if not managed properly. Nausea, vomiting, and diarrhea are common during dose escalation. Adequate hydration, slow dose titration, and meal timing adjustments are standard mitigation strategies. Some researchers find that starting at even lower doses than the clinical trial protocols and escalating more slowly can reduce the severity of gastrointestinal symptoms.
Pancreatic concerns
All GLP-1 receptor agonists carry theoretical concerns about pancreatitis and pancreatic cancer risk. Clinical trials for both mazdutide and retatrutide have not shown elevated rates of pancreatitis, but the sample sizes and durations are still relatively small compared to the decades of post-market surveillance available for older medications. Monitoring amylase and lipase levels during treatment is a standard precaution.
Gallbladder considerations
Rapid weight loss from any cause increases the risk of gallstone formation. Both mazdutide and retatrutide, by producing substantial weight loss, may increase gallbladder complications. This risk is not unique to these compounds. It occurs with bariatric surgery, very-low-calorie diets, and other GLP-1 agonists as well. Monitoring for gallbladder symptoms during treatment is advisable.
Muscle mass preservation
Significant weight loss typically includes some loss of lean muscle mass. The glucagon receptor activation in both compounds may partially mitigate this through increased protein turnover and metabolic activation, but the extent of muscle preservation has not been specifically studied in dedicated body composition analyses for either compound. Researchers should consider incorporating resistance training and adequate protein intake to support muscle preservation during treatment.
Drug interactions
Both compounds slow gastric emptying, which can affect the absorption of oral medications taken concurrently. This is particularly relevant for medications with narrow therapeutic windows. Timing adjustments for co-administered medications may be necessary. Researchers combining these compounds with other peptide stacks should consider pharmacokinetic interactions carefully.
The future of multi-receptor agonists
Mazdutide and retatrutide represent two branches of the same evolutionary tree in metabolic pharmacology. The success of both compounds, using different combinations of the same three receptor targets, has validated the multi-agonist approach and opened the door for even more combinations.
Novo Nordisk is developing its own triple agonist. Other companies are exploring quadruple agonists that add amylin receptor activation to the mix. CagriSema, combining cagrilintide (an amylin analog) with semaglutide, represents another approach to multi-target therapy. The amylin receptor pathway adds yet another dimension to appetite suppression and metabolic regulation.
The trend is clear. More targets equals more efficacy. But more targets also equals more complexity, more potential side effects, and more unknowns about long-term safety. The field is moving fast, perhaps faster than the safety data can accumulate.
For researchers tracking this space, the key trials to watch in 2026 include the remaining TRIUMPH program readouts for retatrutide, the GLORY-OSA trial for mazdutide in obstructive sleep apnea, and the first Phase 1 data from several quadruple-agonist programs entering clinical development.
SeekPeptides tracks these developments as they happen, providing members with updated protocols, dosing guidance, and evidence-based analysis as new data emerges. For researchers serious about staying ahead of the curve in metabolic peptide research, having access to curated, updated information is essential.
Frequently asked questions
Is mazdutide the same as retatrutide?
No. Mazdutide is a dual agonist targeting GLP-1 and glucagon receptors. Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors. They are made by different companies (Innovent Biologics and Eli Lilly, respectively), have different clinical trial programs, and produce different levels of weight loss. Retatrutide consistently produces greater weight reduction due to its additional receptor target.
Which produces more weight loss, mazdutide or retatrutide?
Retatrutide produces substantially more weight loss. In Phase 3 trials, retatrutide 12 mg achieved 28.7% body weight reduction at 68 weeks, while mazdutide 9 mg achieved 20.1% at 60 weeks. The additional GIP receptor activation in retatrutide likely accounts for this difference by creating synergistic metabolic effects beyond what dual agonism achieves.
Is mazdutide better tolerated than retatrutide?
Yes. Mazdutide has demonstrated remarkably low discontinuation rates (near 1% in early trials) compared to retatrutide (12-18% in Phase 3). Both compounds cause gastrointestinal side effects like nausea and diarrhea, but mazdutide side effects are generally milder and more manageable.
Can you get mazdutide in the United States?
Mazdutide is not FDA-approved and there are no announced timelines for US regulatory submission. It is approved in China under the brand name Xinermei. For US-based researchers, it is available through the research peptide market but not through pharmacies or prescription.
When will retatrutide be approved?
Industry analysts predict FDA approval for retatrutide in 2027. This depends on successful completion of the remaining TRIUMPH Phase 3 trials, most of which are expected to report results throughout 2026. Eli Lilly has not announced a specific regulatory filing timeline.
Does mazdutide work better than semaglutide?
In the head-to-head DREAMS-3 trial, mazdutide 6 mg produced greater weight loss (10.29% vs 6.00%) and better glycemic control than semaglutide 1 mg at 32 weeks. The glucagon receptor activation in mazdutide provides additional fat-burning benefits that semaglutide does not have.
Can you switch from tirzepatide to retatrutide?
Switching from tirzepatide to retatrutide requires a washout period and careful dose titration. Both compounds share GLP-1 and GIP receptor activity, but retatrutide adds glucagon activation. Starting retatrutide at the lowest dose and following the standard escalation protocol is advisable regardless of previous tirzepatide experience.
Which is better for fatty liver disease?
Both compounds significantly reduce liver fat, but retatrutide has stronger data. In a dedicated MASLD trial, retatrutide resolved liver steatosis in more than 90% of participants at 48 weeks. Mazdutide showed 80.2% liver fat reduction in exploratory analyses. Both are substantially better than current treatment options for metabolic liver disease.
External resources
New England Journal of Medicine: GLORY-1 mazdutide Phase 3 results
New England Journal of Medicine: Retatrutide Phase 2 trial for obesity
Nature Medicine: Retatrutide for metabolic liver disease (MASLD)
For researchers serious about optimizing their metabolic peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, dosage calculators, and a community of thousands who navigate these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your protocols stay optimized, and your results stay consistent.