Feb 22, 2026

Some people qualify for GLP-1 medications at a BMI of 27. Others get denied at 35. The difference is not always body weight, and it is not always straightforward. Between FDA labeling, insurance company policies, provider discretion, and the specific GLP-1 medication being prescribed, the BMI threshold you need to clear can shift dramatically depending on who you ask and how you ask them.
This matters because millions of people sit in a gray zone. They know what GLP-1 medications are. They have heard the results. They may have watched friends or family members lose significant weight on semaglutide or tirzepatide. But they do not know if their BMI qualifies them, which comorbidities count, or whether their insurance will actually cover the prescription even if they meet the clinical criteria.
What follows is the most complete breakdown of BMI requirements for GLP-1 eligibility you will find anywhere. Every threshold, every exception, every loophole that matters. From FDA approval criteria and clinical trial enrollment standards to the wildly inconsistent rules insurance companies use to approve or reject coverage, this guide covers the full picture. If you have been wondering whether your body mass index is high enough, low enough, or complicated enough to qualify, you are about to get a definitive answer.
Understanding body mass index and why it matters for GLP-1 eligibility
Body mass index is a simple calculation. Take your weight in kilograms and divide it by your height in meters squared. That is the formula. It produces a single number that places you somewhere on a scale from underweight to severely obese, and that number has become the primary gatekeeper for GLP-1 medication access.
The concept dates back to the 1830s. A Belgian mathematician named Adolphe Quetelet developed it as a population-level statistical tool, not a clinical diagnostic measure. It was never designed to assess individual health. Yet here we are, nearly two centuries later, using it as the single most important number in determining whether someone can access the most effective weight loss medications ever developed.
That tension matters.
BMI does not distinguish between muscle mass and fat mass. It does not account for where your body stores fat, whether viscerally around your organs or subcutaneously beneath your skin. It does not factor in metabolic health markers like insulin resistance, inflammation, or cardiovascular risk. A 220-pound bodybuilder with 12% body fat and a 220-pound sedentary person with 40% body fat can have identical BMIs. Their health profiles could not be more different.
Still, BMI remains the standard. The FDA uses it. Insurance companies use it. Clinical trials use it as enrollment criteria. And until the medical establishment adopts better metrics, understanding exactly where you fall on the BMI scale is the first step toward accessing GLP-1 therapy. The SeekPeptides platform breaks down these thresholds in detail so you know exactly where you stand before ever walking into a provider appointment.
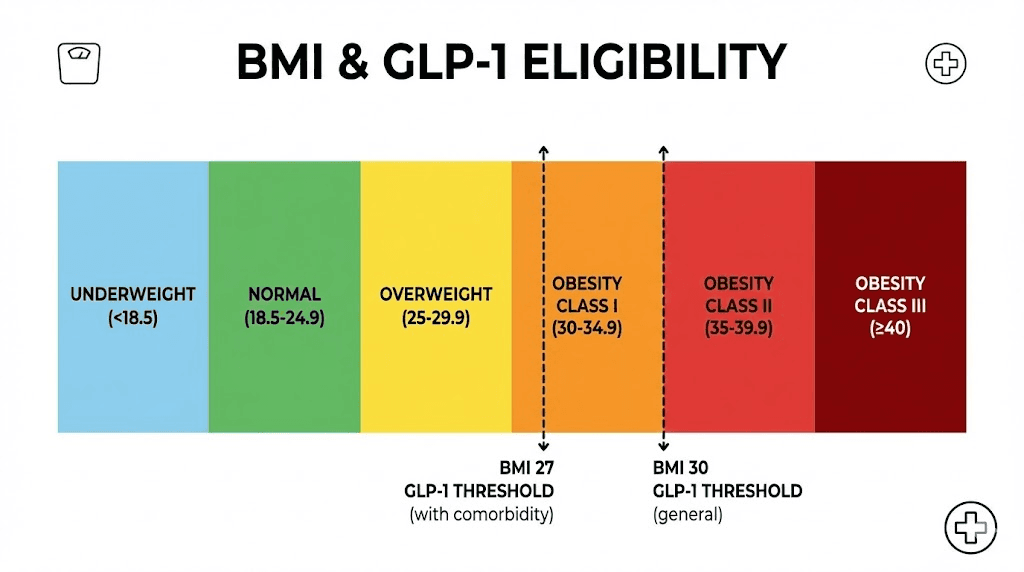
The exact BMI thresholds the FDA sets for GLP-1 medications
The FDA approval language is remarkably consistent across GLP-1 medications approved for weight management. Two pathways exist. Two numbers matter.
Pathway one: BMI of 30 or higher. If your body mass index hits 30 kg/m2 or above, you meet the obesity threshold. No additional conditions required. The FDA considers you clinically obese, and GLP-1 medications are approved for your use regardless of whether you have any other health complications. This applies to both semaglutide formulations and tirzepatide preparations approved for chronic weight management.
Pathway two: BMI of 27 or higher with at least one weight-related comorbidity. This is the alternative pathway. If your BMI falls between 27 and 29.9, you are classified as overweight rather than obese. The FDA still allows GLP-1 prescriptions, but only if you also have at least one qualifying health condition linked to your weight. Type 2 diabetes. Hypertension. Dyslipidemia. Cardiovascular disease. Obstructive sleep apnea. These conditions effectively lower the BMI bar from 30 to 27.
Those two numbers, 27 and 30, form the backbone of every eligibility conversation you will ever have about GLP-1 medications. Everything else, the insurance rules, the provider assessments, the clinical trial criteria, builds on top of that foundation.
But here is what the FDA language does not tell you. It does not mandate that providers follow these exact thresholds in practice. FDA approval means a medication can be prescribed for that indication. It does not mean it must be prescribed, and it does not mean it cannot be prescribed for other reasons. The concept of off-label prescribing exists throughout medicine, and GLP-1 medications are no exception. Some providers will prescribe below BMI 27 based on clinical judgment. Others will refuse to prescribe until a patient has tried and failed other interventions, even when the BMI threshold is clearly met.
The FDA sets the floor. Providers and insurance companies set the actual bar.
BMI requirements for each GLP-1 medication
Semaglutide: Wegovy and Ozempic
Semaglutide operates through two branded formulations with different approved indications, and the BMI requirements vary accordingly.
Wegovy (semaglutide 2.4mg weekly injection) carries the chronic weight management indication. The approved BMI thresholds match the FDA standard: 30 or higher, or 27 or higher with at least one weight-related comorbidity. This is the formulation most commonly discussed in weight loss contexts, and it is the one where BMI eligibility matters most for insurance coverage. If you are tracking how long semaglutide takes to work, the timeline research assumes you meet these baseline criteria.
Ozempic (semaglutide 0.5mg, 1mg, or 2mg weekly injection) is FDA-approved for type 2 diabetes management, not weight loss. However, weight loss is a well-documented secondary effect, and many providers prescribe Ozempic off-label specifically for its weight management benefits. The BMI requirements for Ozempic depend entirely on the prescribing context. For diabetes, the relevant criteria involve blood glucose levels and A1C readings, not BMI. For off-label weight management, providers typically apply the same 30/27 thresholds used for Wegovy.
A newer development is the oral semaglutide pill (Rybelsus for diabetes, and the recently approved oral Wegovy), which brings the same active ingredient in tablet form. The BMI thresholds remain identical to the injectable version. Delivery method changes. Eligibility criteria do not. Our guide on oral semaglutide formulations covers the practical differences between injectable and oral delivery.
Semaglutide dosing follows a gradual escalation schedule that starts at 0.25mg weekly and increases over 16-20 weeks to the maintenance dose. You can use our semaglutide dosage calculator to map out the exact schedule. That escalation timeline is the same regardless of your starting BMI, though appetite suppression onset may vary based on individual factors.
Tirzepatide: Zepbound and Mounjaro
Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it works through two pathways instead of one. The BMI requirements mirror semaglutide for the weight management indication.
Zepbound (tirzepatide for chronic weight management) follows the same 30/27 threshold. BMI of 30 or higher qualifies outright. BMI between 27 and 29.9 qualifies with a weight-related comorbidity. The clinical trial data from SURMOUNT-1 enrolled participants with a mean BMI of 38, and the results showed dramatic weight loss outcomes across dosing tiers.
Mounjaro (tirzepatide for type 2 diabetes) carries the same dynamic as Ozempic. Primary indication is diabetes management based on blood glucose criteria, not BMI. Off-label weight loss prescriptions apply the standard BMI thresholds at provider discretion.
The tirzepatide dosing schedule starts at 2.5mg weekly and escalates to a maximum of 15mg. Our tirzepatide dosage calculator can help you map the specific units and milligram conversions. Some researchers explore microdosing tirzepatide at levels below the standard starting dose, though this approach falls outside typical prescribing guidelines.
One important distinction: tirzepatide consistently produces greater percentage weight loss than semaglutide in head-to-head comparisons. The semaglutide versus tirzepatide comparison shows tirzepatide achieving 20-22.5% body weight reduction at the highest dose versus 14.9% for semaglutide. This matters for BMI discussions because people starting at lower BMIs may reach a normal weight range faster on tirzepatide, which raises questions about treatment duration that our guide on GLP-1 treatment duration addresses in detail.
Retatrutide and next-generation GLP-1 agents
Retatrutide is a triple receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. It is not yet FDA-approved but has shown remarkable weight loss results in phase 2 trials, with participants losing up to 24.2% of body weight at the highest dose over 48 weeks. The retatrutide dosing guide covers the current research protocols.
Clinical trial enrollment for retatrutide follows the same BMI pattern: 30 or higher, or 27 or higher with comorbidities. If and when it receives FDA approval, the labeling will almost certainly adopt identical thresholds. The retatrutide versus semaglutide comparison highlights just how much more aggressive the weight loss trajectory may be, which could have implications for how aggressively providers prescribe to patients at lower BMI ranges.
Other agents in development include mazdutide, cagrilintide, survodutide, and orforglipron (an oral small-molecule GLP-1 agonist). Each will likely follow the same 30/27 BMI framework for weight management approval, though the actual prescribing landscape will depend on individual approval timelines and the specific populations studied in phase 3 trials. Keep an eye on the retatrutide availability timeline for updates on these newer agents.
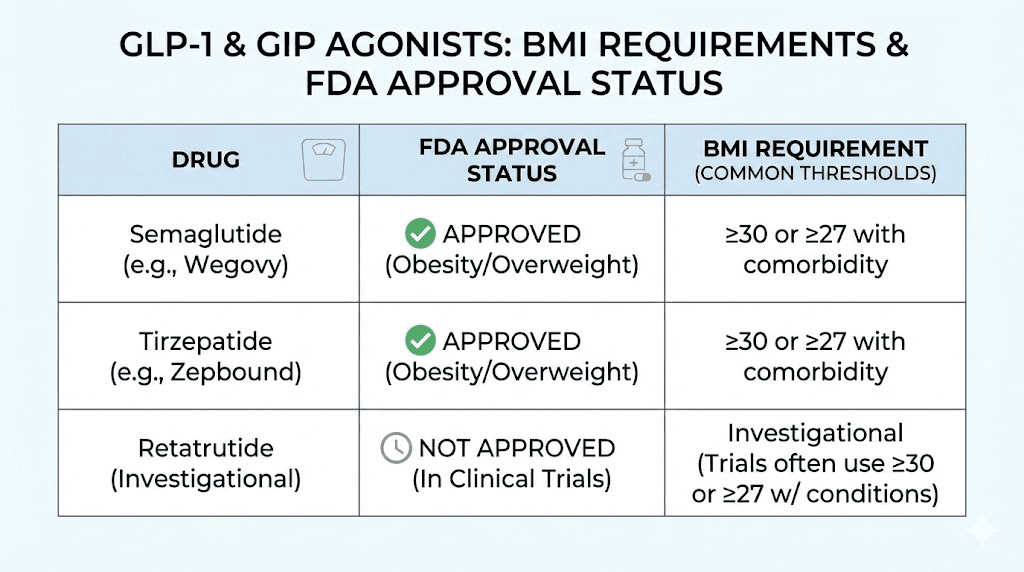
Qualifying health conditions when your BMI falls between 27 and 30
The comorbidity pathway is where most of the confusion lives. You have a BMI of 28. Maybe 29. You do not meet the straight obesity threshold. But you might still qualify if you have the right documented health condition. The question is: which conditions count?
Here is the complete list of weight-related comorbidities recognized by the FDA and most insurance companies for GLP-1 eligibility.
Type 2 diabetes. This is the single most common qualifying condition. If you have been diagnosed with type 2 diabetes, your eligibility for GLP-1 medications is essentially guaranteed regardless of BMI, because these drugs were originally developed and approved for diabetes management. A BMI of 27 with type 2 diabetes is one of the clearest qualification pathways that exists. Understanding the relationship between GLP-1 medications and metabolic function helps explain why this connection is so strong.
Hypertension. High blood pressure is the second most common qualifier. A documented diagnosis of hypertension, especially if you are currently on blood pressure medication, satisfies the comorbidity requirement at most insurance companies. Research shows that even modest weight loss of 5-10% can reduce systolic blood pressure by 5-8 mmHg, which is part of why insurers view GLP-1 therapy as medically necessary for hypertensive patients with elevated BMI.
Dyslipidemia. Abnormal cholesterol levels qualify. This includes elevated LDL cholesterol, elevated triglycerides, or low HDL cholesterol. If you are taking a statin or other lipid-lowering medication, you have documented dyslipidemia by definition. The weight loss effects of GLP-1 medications typically improve lipid panels significantly.
Cardiovascular disease. A history of heart attack, stroke, angina, coronary artery disease, or peripheral arterial disease qualifies. The SELECT trial specifically demonstrated that semaglutide reduces major adverse cardiovascular events by 20% in patients with established cardiovascular disease, regardless of diabetes status. This evidence has made cardiovascular history one of the strongest qualifying conditions for GLP-1 coverage.
Obstructive sleep apnea. Documented sleep apnea, particularly if you use a CPAP machine or have had a positive sleep study, qualifies. Weight loss is one of the primary treatment recommendations for sleep apnea, making GLP-1 therapy a logical medical intervention.
Less commonly recognized but still valid qualifying conditions include prediabetes (elevated fasting glucose or A1C between 5.7-6.4%), polycystic ovary syndrome, non-alcoholic fatty liver disease, osteoarthritis of weight-bearing joints, and chronic kidney disease.
The key takeaway is documentation. Your qualifying condition must be documented in your medical record. An active diagnosis, current medication for the condition, or recent lab work showing the abnormality all count. A provider saying you probably have high cholesterol does not count. A lipid panel showing LDL of 160 mg/dL does.
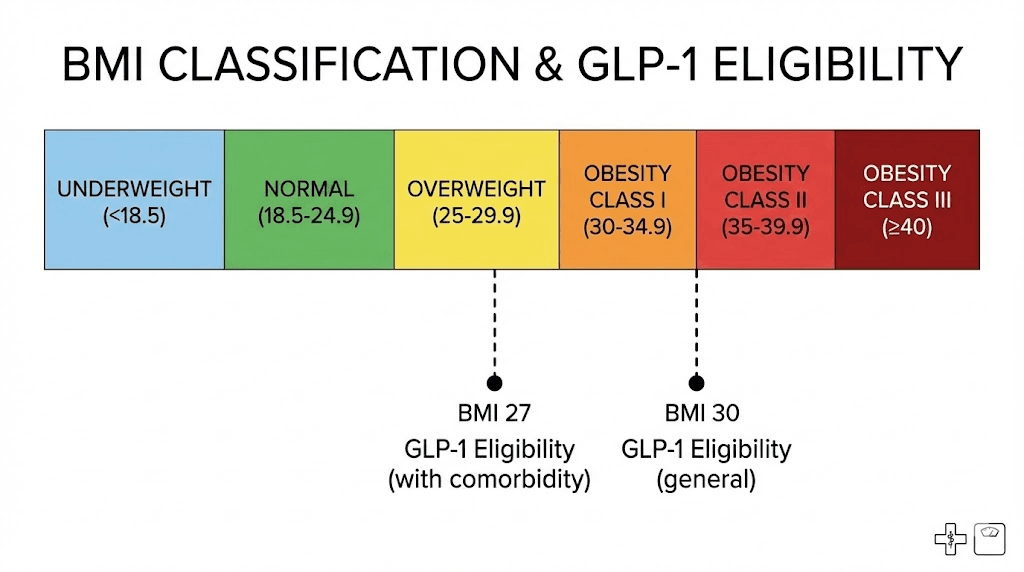
BMI categories and obesity classifications explained
Before diving deeper into eligibility specifics, it helps to understand exactly where each BMI number places you in the medical classification system. These categories determine not just GLP-1 eligibility but also which tier of treatment intensity providers recommend and which insurance coverage pathways open up.
Underweight: BMI below 18.5. GLP-1 medications are not indicated and would be harmful. Weight loss is the opposite of what someone in this category needs.
Normal weight: BMI 18.5 to 24.9. No GLP-1 eligibility for weight management. Some providers may prescribe off-label for specific metabolic conditions, but this remains controversial and is not covered by any standard insurance plan.
Overweight: BMI 25.0 to 29.9. The gray zone. You do not qualify at BMI 25 or 26 under any FDA-approved indication. At BMI 27-29.9 with a qualifying comorbidity, you cross the threshold. This 25-27 gap is where many people find themselves frustrated, knowing they carry excess weight but falling just short of the medical criteria that would unlock treatment.
Class I obesity: BMI 30.0 to 34.9. You qualify for GLP-1 medications under FDA labeling without any additional conditions. Most insurance companies will also cover treatment at this level, though some require documentation of prior weight loss attempts. People in this category typically see significant results. The early weeks on semaglutide may feel slow, but weight loss accelerates as doses increase.
Class II obesity: BMI 35.0 to 39.9. Strong eligibility across all pathways. Insurance coverage is more readily approved. Providers view GLP-1 therapy as a clear medical necessity. At this level, you may also qualify for bariatric surgery referrals, though many patients prefer trying GLP-1 therapy first.
Class III obesity: BMI 40.0 and above. Previously called morbid obesity. Maximum eligibility across every pathway. Every insurance company covers GLP-1 at this level. Providers may recommend more aggressive dosing schedules, and some patients achieve truly transformative results. The before-and-after tirzepatide outcomes in this BMI category are among the most dramatic documented in clinical practice.
Understanding your specific category matters because it shapes every subsequent conversation with providers and insurers. A BMI of 29.5 and a BMI of 30.1 are clinically almost identical, but they sit on different sides of a regulatory line that changes everything about access and coverage.
How insurance companies set their own BMI cutoffs for GLP-1 coverage
Here is where the conversation gets complicated. The FDA says 30 or 27 with comorbidities. But insurance companies are not required to follow FDA labeling for coverage decisions. Many set their own thresholds, and those thresholds can be significantly more restrictive than what the FDA approves.
Private insurance thresholds
Private insurance coverage for GLP-1 weight loss medications varies enormously by carrier, plan type, and even specific employer group.
Some examples of real insurance company BMI thresholds that have been documented in coverage policies:
CVS Caremark has required a BMI of at least 40, or a BMI of 35 with one or more comorbid conditions including cardiovascular disease, hypertension, dyslipidemia, diabetes, elevated waist circumference, or obstructive sleep apnea. That is significantly more restrictive than the FDA threshold.
Other pharmacy benefit managers have set various bars. Some follow the FDA standard at 30/27. Others require BMI 32 or higher. Some demand documentation of failed prior weight loss attempts, including supervised diet programs or other medications like phentermine, before approving GLP-1 coverage regardless of BMI.
The frustrating reality is that two people with identical BMIs, identical health conditions, and identical GLP-1 prescriptions can receive completely different coverage decisions based solely on which insurance company they happen to have. One gets approval. The other gets a denial letter and a suggestion to try diet and exercise first. If your coverage gets denied, understanding affordable tirzepatide options or compounded alternatives becomes critical.
Prior authorization requirements add another layer. Most insurance companies require prior authorization for GLP-1 weight loss medications, meaning your provider must submit clinical documentation proving you meet the plan-specific criteria before the pharmacy will fill the prescription. This process can take days to weeks and may require appeals if initially denied.
Medicare coverage and the BALANCE model
Medicare has historically been prohibited from covering weight loss medications under Part D. This changed with recent legislation and the announcement of the BALANCE (Better Approaches to Lifestyle and Nutrition for Comprehensive health) Model in December 2025, which creates a Medicare GLP-1 payment demonstration scheduled to begin in July 2026.
Under the BALANCE model, Medicare coverage for GLP-1 medications applies to beneficiaries meeting specific criteria that go beyond simple BMI thresholds. The requirements include patients at high metabolic or cardiovascular risk with a BMI greater than 27 with prediabetes or established cardiovascular disease, or a BMI over 30 with uncontrolled hypertension despite treatment, advanced kidney disease, or heart failure.
Beneficiaries who qualify under the expanded criteria will pay approximately 50 dollars per month for their GLP-1 medication, a dramatic reduction from the retail prices that have kept these drugs out of reach for many Medicare enrollees. For context, retail pricing for brand-name GLP-1 medications can exceed 1,000 dollars per month without insurance. The peptide cost calculator can help you estimate expenses based on your specific situation.
Medicaid coverage by state
Medicaid coverage of GLP-1 medications for weight loss is optional for states. Coverage for diabetes indications is required, but the weight management indication is left to individual state discretion.
As of early 2025 data, 11 states covered GLP-1 medications for weight loss under their state employee health plans. Nine states provided coverage under their Medicaid programs. Five states offered coverage under both programs. This means the majority of states still do not cover GLP-1 medications for weight management under Medicaid, even when patients meet every clinical criterion.
The state-by-state variability creates a geographic lottery effect. A person with a BMI of 32 in a state with Medicaid GLP-1 coverage gets their prescription filled. That same person in a neighboring state without coverage does not. Same BMI, same medical need, different zip code, different outcome.
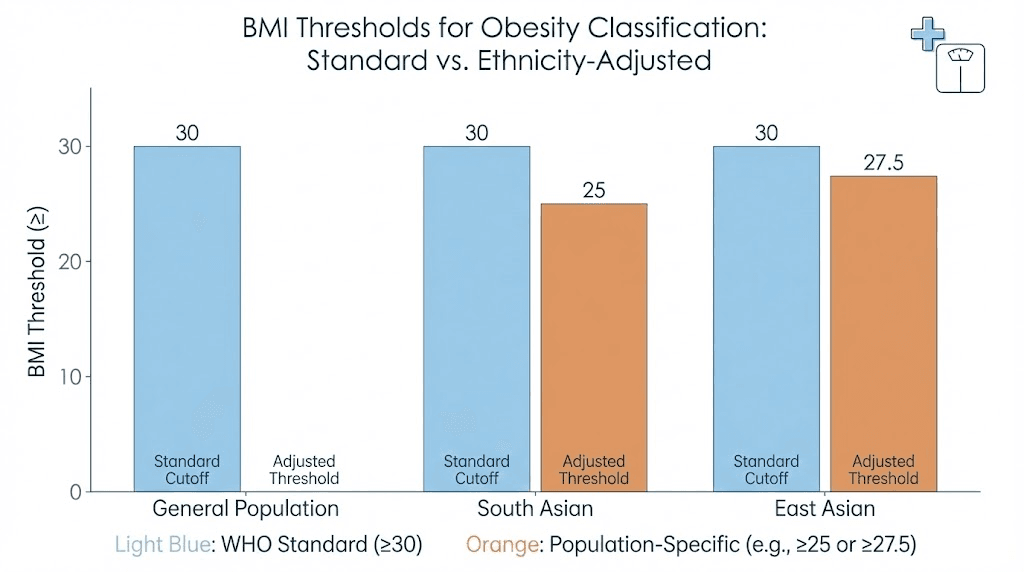
Adjusted BMI thresholds for different ethnic backgrounds
One of the most important but least discussed aspects of BMI and GLP-1 eligibility involves ethnic and racial differences in how BMI correlates with metabolic risk. The standard BMI categories were developed primarily from European population data, and they do not apply equally across all ethnic groups.
The World Health Organization has acknowledged lower BMI cutoffs for Asian populations. Where the standard threshold for obesity is 30, the WHO suggests 27.5 for Asian populations. Where the standard overweight threshold is 25, the WHO suggests 23 for Asian individuals. These adjustments reflect research showing that Asian populations develop obesity-related health complications like type 2 diabetes and cardiovascular disease at significantly lower BMIs than European-descended populations.
Specific research has quantified these differences. A BMI of 30 in a white individual carries equivalent diabetes risk to a BMI of approximately 23.9 in a South Asian individual and 26.9 in a Chinese individual. That gap is enormous. A South Asian person at BMI 24, technically normal weight by standard criteria, faces the same metabolic risk as a white person at BMI 30, who would qualify for GLP-1 medications without question.
The American Diabetes Association has responded by recommending lower BMI cutoffs of 23 or higher for diabetes screening in Asian American populations. However, these adjusted thresholds have not been formally incorporated into FDA labeling for GLP-1 medications, creating a significant gap where Asian patients may have clear metabolic indications for treatment but fail to meet the standard BMI criteria.
Research published in The Lancet examining GLP-1 receptor agonist eligibility across 99 countries found that eligibility varies dramatically based on which BMI thresholds are used. When Asian-adjusted cutoffs were applied, the eligible population increased substantially in Asian regions.
If you are of Asian descent and your BMI falls between 23 and 30, have a detailed conversation with your provider about ethnicity-adjusted risk assessment. A provider who understands these differences may be willing to prescribe based on your actual metabolic risk rather than a standard BMI cutoff that was not designed for your population. Understanding your full metabolic picture, including the role of supporting compounds and supplements alongside GLP-1 therapy, can strengthen your case.
What the clinical trials reveal about BMI and weight loss outcomes
The clinical trial data offers the most rigorous evidence we have about how GLP-1 medications perform across different BMI categories. These trials define both who can access treatment and what results they can expect.
STEP trials: semaglutide
The STEP (Semaglutide Treatment Effect in People with obesity) program is the landmark clinical trial series for semaglutide 2.4mg weekly. Across multiple STEP trials, the enrollment criteria required a BMI of 30 or higher, or 27 or higher with at least one weight-related comorbidity (excluding type 2 diabetes, which was studied separately).
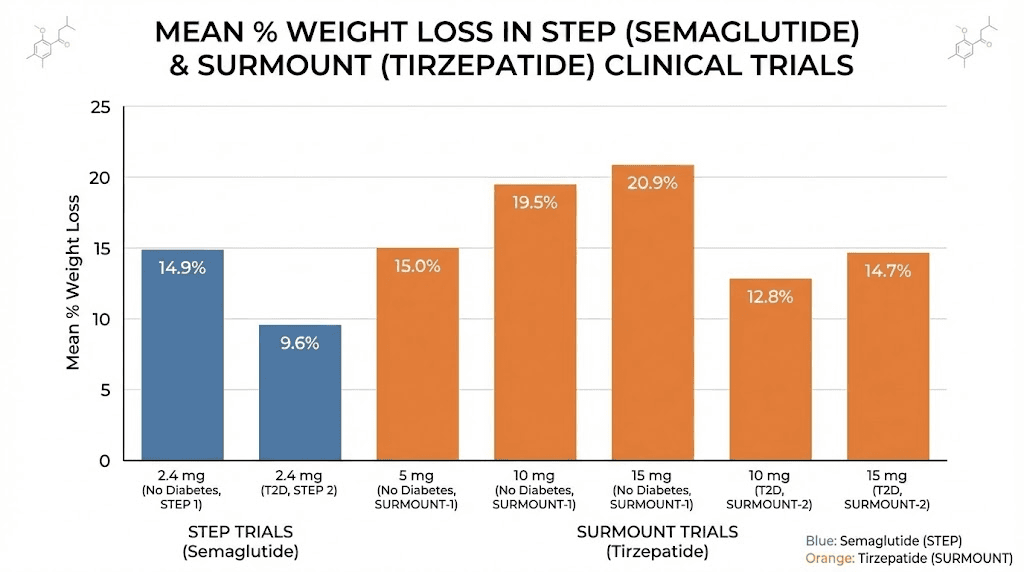
STEP 1 enrolled 1,961 adults. The mean percentage change in body weight from baseline to 68 weeks was 14.9% in the semaglutide group versus 2.4% in the placebo group. The breakdown by weight loss achievement is striking. A full 86.4% of semaglutide participants lost at least 5% of their body weight. 69.1% lost at least 10%. 50.5% lost at least 15%. And 32% lost 20% or more.
STEP 5 examined long-term outcomes over 104 weeks. At the two-year mark, 77.1% of semaglutide participants maintained at least 5% weight loss versus 34.4% on placebo. The durability of results confirms that semaglutide appetite suppression is sustained over extended treatment periods, which matters for people evaluating how long to stay on semaglutide.
An important nuance: while the trials did not publish detailed BMI-stratified results in their primary papers, the data consistently shows that higher baseline BMI correlates with greater absolute weight loss in kilograms, while lower baseline BMI correlates with greater percentage weight loss. Someone starting at BMI 32 might lose a higher percentage of their body weight than someone starting at BMI 42, even though the person at BMI 42 loses more total kilograms. Both outcomes are clinically meaningful, but they illustrate why BMI at treatment initiation shapes expectations about results.
SURMOUNT trials: tirzepatide
The SURMOUNT trials evaluated tirzepatide for weight management. SURMOUNT-1 enrolled 2,539 adults with a mean baseline BMI of 38.0, and 94.5% of participants had a BMI of 30 or higher.
The results were extraordinary. At week 72, mean percentage weight loss was 15.0% with tirzepatide 5mg, 19.5% with 10mg, and 20.9% with 15mg, compared to just 3.1% with placebo. At the highest dose, 63% of participants achieved at least 20% body weight reduction versus 1.3% on placebo. The tirzepatide versus semaglutide dosage comparison puts these numbers in context.
Post-hoc analysis from SURMOUNT-1 examined early responders versus late responders. Participants with class III obesity (BMI 40 or higher) were more likely to be late responders, meaning they needed more time to achieve significant weight loss. This has practical implications. If you start at a very high BMI, do not panic if the first 12 weeks produce modest results. The data shows that even late responders achieved substantial weight loss by week 72. Our guide on what to do when tirzepatide seems to stop working addresses the plateau periods that are common during treatment.
Body composition analysis from SURMOUNT-1 revealed that tirzepatide produces both fat mass and lean mass reduction, though the ratio favors fat loss. This is relevant because muscle-related concerns with tirzepatide are common, and understanding how BMI category affects body composition changes helps set appropriate expectations. Supporting muscle preservation through proper nutrition on tirzepatide and foods to prioritize and avoid becomes especially important at higher starting BMIs where total weight loss is greater.
What happens if your BMI falls below 27
This is the question that generates the most debate. Can you get GLP-1 medications with a BMI below 27? The honest answer is: it depends.
Clinically, GLP-1 medications are not FDA-approved for weight management in people with a BMI below 27. The safety and efficacy data in this population is limited because clinical trials specifically excluded them. There is no robust evidence base guiding treatment in the 25-27 range, and essentially no data for BMIs below 25.
That said, off-label prescribing happens. Some telehealth platforms and cash-pay clinics will prescribe GLP-1 medications to patients below the standard thresholds, particularly if they are paying out of pocket and not seeking insurance coverage. This practice is legal but controversial. Medical societies have expressed concern about the use of these medications for purely cosmetic weight loss in people who are not overweight or only mildly overweight.
The risks for lower-BMI patients are different than for higher-BMI patients. Muscle loss becomes a proportionally larger concern when there is less excess fat to lose. Gastrointestinal side effects like constipation on semaglutide or constipation on tirzepatide can be harder to manage on a lighter frame. The potential for over-restriction and inadequate nutrition increases.
There is also a growing body of discussion around people using GLP-1 medications to maintain a normal weight after previous weight loss. An NPR report from February 2026 explored the benefits and risks of using GLP-1 medications at a normal BMI, noting that the long-term effects of these drugs in normal-weight populations are not well studied. The risk of yo-yo effects, where patients lose muscle on the drug and regain fat after stopping, is a real concern.
If your BMI is below 27, the realistic path forward involves focusing on the health conditions that could qualify you through the comorbidity pathway if your BMI is at least 27, or working with a provider who assesses your metabolic risk beyond the BMI number alone. Waist circumference, body fat percentage, visceral fat imaging, and metabolic blood panels can all paint a more complete picture of whether your weight is actually harming your health. SeekPeptides offers comprehensive resources for understanding these alternative assessment methods and how they factor into treatment decisions.
The WHO December 2025 guideline and what it means for BMI thresholds
In December 2025, the World Health Organization released its first-ever guideline on the use of GLP-1 therapies for treating obesity in adults. This was a landmark moment because it represented global recognition of GLP-1 medications as a legitimate long-term treatment for obesity rather than a temporary or supplementary intervention.
The WHO guideline specifically recommends GLP-1 therapies for adults with obesity, defined as BMI 30 or higher. It covers three agents: liraglutide, semaglutide, and tirzepatide. An important distinction from the FDA criteria is that the WHO guideline does not extend its recommendation to adults with a BMI between 27 and 30 who have obesity-related conditions. The WHO took a more conservative stance, recommending treatment only for clinically obese populations.
This creates an interesting divergence. The FDA approves at 27 with comorbidities. The WHO recommends only at 30. In practice, because the FDA governs drug approvals in the United States and most Western countries follow similar regulatory frameworks, the 27 threshold remains the functional standard for clinical practice. But the WHO guideline matters because it influences global health policy, insurance coverage decisions in some countries, and the general medical consensus about when these drugs are appropriate.
The WHO guideline also emphasized that obesity is a chronic, relapsing disease requiring long-term treatment, not a short-term intervention. This framing supports the case for continued access to GLP-1 medications even after patients achieve their target weight, which relates to concerns about semaglutide withdrawal symptoms and weight regain after stopping treatment.
How to calculate your BMI accurately
Getting your BMI right matters because a small difference, even a fraction of a point, can place you on one side of an eligibility threshold or the other. Here is how to calculate it correctly, avoid common errors, and document it properly for medical purposes.
The standard formula
Metric system: Weight in kilograms divided by height in meters squared. If you weigh 90kg and stand 1.75m tall, that is 90 divided by (1.75 times 1.75), which equals 90 divided by 3.0625, which equals 29.4.
Imperial system: Weight in pounds divided by height in inches squared, then multiplied by 703. If you weigh 200 pounds and stand 5 feet 10 inches tall (70 inches), that is 200 divided by (70 times 70) times 703, which equals 200 divided by 4,900 times 703, which equals 28.7.
Online calculators exist from the CDC, NIH, and countless medical websites. Use them. Manual calculation introduces rounding errors that can shift your result.
Common measurement mistakes
Accuracy starts with accurate inputs. Here are the most common errors people make when calculating their BMI for GLP-1 qualification purposes.
Measuring height incorrectly. Standing against a wall with shoes off and looking straight ahead gives the most accurate result. Many people overestimate their height by an inch or more, which artificially lowers BMI. At a BMI near the threshold, one inch of height difference can shift your result by a full point.
Using outdated weight measurements. Your weight fluctuates daily by 2-4 pounds based on hydration, food intake, and bowel content. Weigh yourself in the morning, after using the bathroom, before eating or drinking, wearing minimal clothing. This is your most consistent and typically lowest weight. For GLP-1 qualification purposes, you want the most accurate current measurement, not one from a doctor visit three months ago.
Time of day matters. Weight measured in the evening after a full day of eating and drinking can be 3-5 pounds higher than morning weight. If the difference between qualifying and not qualifying comes down to a pound or two, when and how you measure makes a difference.
Scale calibration. Bathroom scales drift over time. Compare yours against a known weight or against a medical-grade scale at your provider office. A scale that reads 2 pounds low could be the difference between a BMI of 29.8 and a BMI of 30.1, which matters enormously for eligibility.
The peptide calculator tools on SeekPeptides emphasize precision because small measurement errors compound when calculating dosages, reconstitution volumes, and other values where accuracy determines outcomes. The same principle applies to BMI calculation. Getting the inputs right is not optional.
Working with your healthcare provider to establish eligibility
The clinical encounter where GLP-1 eligibility gets determined is one of the most important medical appointments you will have in your weight management journey. How you prepare for it and what you communicate can meaningfully affect the outcome.
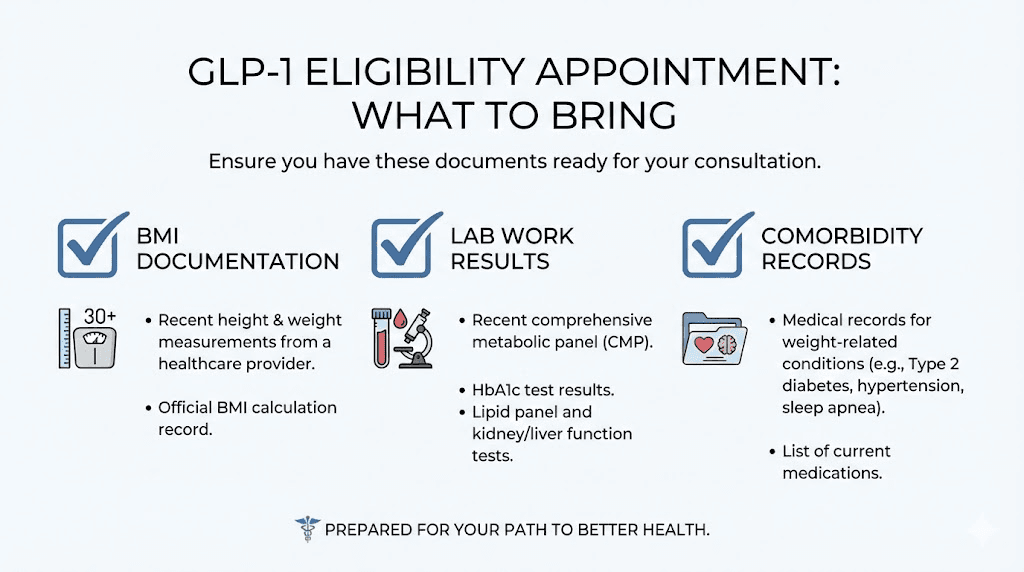
What to bring to the appointment
A recent weight measurement. Ideally from a medical-grade scale. If your home scale and the provider office scale disagree, the provider scale typically wins for documentation purposes.
Your height measured within the past year. Height changes with age, posture, and time of day. If you are over 40, get a fresh measurement rather than relying on what your drivers license says.
Documentation of qualifying comorbidities. If your BMI falls between 27 and 30, this is essential. Bring recent lab work showing elevated blood glucose, A1C, cholesterol, or triglycerides. Bring records of blood pressure readings above 130/80. Bring documentation of sleep apnea diagnosis, CPAP usage, or sleep study results. Bring records of cardiovascular events, procedures, or diagnoses.
Prior weight loss attempt documentation. Many insurance companies require evidence that you have tried and failed other weight loss methods before approving GLP-1 coverage. Document any supervised diet programs, exercise regimens, previous medications like phentermine or other appetite suppressants, and behavioral therapy. Some plans require 3-6 months of documented prior attempts.
Family history. A family history of obesity, type 2 diabetes, cardiovascular disease, or metabolic syndrome can strengthen your case for medical necessity, even if it does not formally change the BMI threshold.
What to discuss with your provider
Be direct about your goals. If you want GLP-1 medication for weight management, say so clearly. Providers are more likely to advocate for your insurance coverage when they understand your specific objectives and can document the medical rationale accordingly.
Ask your provider about which GLP-1 medication they recommend and why. The choice between semaglutide and tirzepatide depends on multiple factors including your specific health profile, insurance coverage, and treatment goals. Our side effect comparison guide can help you prepare for that conversation.
Discuss the treatment timeline. GLP-1 medications are not quick fixes. They require dose escalation over weeks to months, and full therapeutic effects may not appear until you reach the maintenance dose. Understanding how long tirzepatide takes to work or when semaglutide results appear helps set realistic expectations from day one.
Ask about what happens if insurance denies coverage. Your provider may be willing to submit a peer-to-peer review, file an appeal, or suggest alternative pathways. Compounded versions of semaglutide and tirzepatide represent one alternative pathway that some patients explore when brand-name coverage is unavailable. If your provider recommends a compounded formulation, understanding proper semaglutide reconstitution or tirzepatide reconstitution becomes essential.
Common mistakes that prevent people from qualifying
After studying hundreds of GLP-1 eligibility discussions across medical forums and patient communities, clear patterns emerge. The same mistakes show up again and again. Knowing what they are lets you avoid them entirely.
Not checking BMI before the appointment. This sounds obvious, but many patients walk into their provider office assuming they qualify based on how they look or feel without ever calculating their actual BMI. Then the provider weighs them, calculates BMI at 26.3, and the conversation ends before it starts. Know your number before you go. Better yet, know your number and your qualifying comorbidities before you go.
Failing to document comorbidities. Having high blood pressure is not the same as having documented high blood pressure. If your BMI falls in the 27-30 range, your qualifying condition needs to be in your medical record with supporting evidence. Schedule a visit specifically to get labs drawn and conditions documented before your GLP-1 appointment. This is especially important if you have been managing conditions without formal medical follow-up.
Not knowing your insurance policy requirements. Call your insurance company before the appointment. Ask specifically: what are your coverage criteria for Wegovy, Zepbound, or whatever GLP-1 medication you are pursuing? What BMI threshold do they require? What documentation do they need? Do they require prior authorization? Do they require evidence of failed prior weight loss attempts? Do they require a specific type of provider to prescribe? Getting these answers in advance lets you and your provider submit a complete prior authorization on the first attempt rather than going through rounds of denials and appeals.
Timing your weigh-in poorly. If you are borderline, your weight at the provider office matters. Being weighed after a large lunch, while dehydrated from a workout, or in heavy clothing all affect the result. Morning appointments in lightweight clothing after a normal meal the night before give you the most representative and typically highest weight. If you need your BMI to hit 30, every pound counts.
Accepting the first denial. Insurance denials are not final. They are the beginning of a negotiation. Most GLP-1 coverage denials can be appealed successfully with additional documentation, a letter of medical necessity from your provider, or a peer-to-peer review between your provider and the insurance company medical director. The appeal success rate for GLP-1 medications is higher than most people assume, but only for those who actually file appeals.
Overlooking compounding pharmacies. If brand-name GLP-1 medications are not covered by your insurance or are prohibitively expensive, compounding pharmacies like Empower and Empower tirzepatide formulations offer alternatives at significantly lower price points. The BMI requirements for compounded formulations are typically set by the prescribing provider rather than insurance criteria, which can make access easier for borderline cases. Other options include Olympia pharmacy semaglutide and various telehealth platforms.
Alternative metrics: when BMI is not the whole story
BMI is the gatekeeper, but it is not the only measure of whether you would benefit from GLP-1 therapy. A growing number of providers and researchers argue that BMI alone is an inadequate assessment tool, particularly for patients who carry metabolic risk at lower body weights.
Waist circumference measures abdominal fat distribution. A waist circumference above 40 inches in men or 35 inches in women indicates elevated cardiovascular and metabolic risk regardless of BMI. Some insurance companies include elevated waist circumference as a qualifying comorbidity for GLP-1 coverage.
Waist-to-hip ratio refines the waist circumference measurement by accounting for body frame size. A ratio above 0.90 in men or 0.85 in women suggests central obesity and elevated disease risk.
Body fat percentage directly measures what BMI tries to estimate indirectly. DEXA scans, bioelectrical impedance analysis, and hydrostatic weighing can all measure body fat percentage accurately. A body fat percentage above 25% in men or 32% in women indicates excess adiposity even if BMI falls in the normal range.
Metabolic biomarkers tell the story that BMI misses entirely. Fasting insulin levels, HOMA-IR (a measure of insulin resistance), high-sensitivity C-reactive protein (an inflammation marker), liver enzymes (indicators of fatty liver disease), and advanced lipid panels all provide evidence of metabolic dysfunction that weight alone cannot capture.
These alternative metrics cannot override the BMI thresholds set by insurance companies. But they can strengthen a case for medical necessity in borderline situations, support off-label prescribing decisions, and help providers make more nuanced clinical judgments. If your BMI is close to a threshold, gathering these additional data points gives your provider more ammunition to advocate on your behalf. Resources on SeekPeptides help members understand these metrics and how they factor into comprehensive health assessment beyond the single BMI number.
Building your GLP-1 qualification strategy by BMI range
Based on everything covered above, here is a practical strategy guide organized by your current BMI range. Every range requires a different approach.
BMI below 25 (normal weight). GLP-1 medications for weight management are not indicated and would not be covered by any insurance plan. If you have specific metabolic concerns, discuss them with your provider in the context of those conditions rather than weight loss. Focus instead on metabolic optimization through nutrition, exercise, and targeted supplementation.
BMI 25.0 to 26.9 (overweight, below threshold). You are close but do not meet the comorbidity pathway threshold of 27. Options are limited to off-label prescribing through cash-pay or telehealth providers, which can be expensive without insurance coverage. Some people at BMI 26.5 or higher find that a slight increase in documentation weight (morning versus evening measurement, different scale calibration) pushes them across the 27 line. This is not about gaming the system. It is about ensuring you are measured consistently and accurately.
BMI 27.0 to 29.9 (overweight with potential eligibility). This is the action zone for the comorbidity pathway. Your top priority is documenting qualifying health conditions. Schedule blood work to check fasting glucose, A1C, lipid panel, liver enzymes, and blood pressure. If any values come back abnormal, you have your qualifying condition. Many people in this range have undiagnosed prediabetes, subclinical hypertension, or dyslipidemia that would qualify them if it were simply identified and documented.
BMI 30.0 to 34.9 (class I obesity). You meet the FDA threshold without needing comorbidities. Your main challenge is insurance coverage, which some plans restrict to higher BMIs. Be prepared with comorbidity documentation anyway because it strengthens your prior authorization. If your insurance requires BMI 35 or higher, file an appeal with a letter of medical necessity from your provider that includes all qualifying conditions.
BMI 35.0 to 39.9 (class II obesity). Strong eligibility across virtually all pathways. Focus on choosing the right GLP-1 medication for your specific profile. The semaglutide versus tirzepatide comparison is worth studying carefully at this stage. Insurance coverage is usually straightforward, and prior authorization approvals are more likely. Also consider your injection timing, diet planning, and overall protocol optimization to maximize results from day one.
BMI 40.0 and above (class III obesity). Maximum eligibility. Every insurance pathway is open. Providers view GLP-1 therapy as clearly medically necessary. Your focus should be on building a comprehensive treatment plan that includes not just the medication but also nutritional support, physical activity modifications, and monitoring for the more significant side effects that can accompany larger doses. The dietary guidelines while on semaglutide and meal planning on tirzepatide become especially important at higher starting weights.
The future of BMI thresholds for GLP-1 access
The BMI thresholds that define GLP-1 eligibility today are not permanent. Several forces are pushing toward change.
First, the growing recognition that BMI is a flawed metric is driving conversations about alternative eligibility criteria. Medical societies, including the American Medical Association, have formally acknowledged that BMI is an imperfect measure of adiposity and metabolic health. As more nuanced measures gain acceptance, the rigid BMI cutoffs may evolve into more flexible, multi-factor assessments.
Second, the expanding indications for GLP-1 medications beyond weight loss are changing the eligibility landscape. The SELECT trial demonstrated cardiovascular benefits of semaglutide independent of weight loss, leading to a cardiovascular risk reduction indication that does not require a specific BMI. As more non-weight indications emerge, including potential applications for addiction, Alzheimer disease, fatty liver disease, and chronic kidney disease, the relevance of BMI as the primary gatekeeper will diminish.
Third, pricing pressure is changing insurance coverage dynamics. As generic and compounded versions of GLP-1 medications become more available and brand-name prices potentially decrease, insurance companies will face less financial incentive to maintain restrictive BMI thresholds. Lower medication costs mean covering more patients becomes financially feasible.
Fourth, Medicare coverage expansion through the BALANCE model and potential legislative changes could establish new precedents for how BMI thresholds are applied in government health programs. If Medicare successfully demonstrates cost-effective GLP-1 coverage at lower BMI thresholds, private insurers may follow suit.
For now, the 30/27 framework remains the standard. But if history is any guide, access will expand rather than contract as the evidence base grows and medication costs decrease. Staying informed through resources like SeekPeptides ensures you are positioned to take advantage of expanded eligibility as it develops. The platform provides ongoing updates on regulatory changes, new medication approvals, and coverage policy shifts that affect who can access GLP-1 therapy. New delivery methods like GLP-1 patches and oral versus injectable formulations are also changing how these medications reach patients, which could indirectly affect eligibility standards as access models evolve.

Frequently asked questions
What is the minimum BMI to qualify for semaglutide?
The minimum BMI for semaglutide (Wegovy) under FDA labeling is 30 without any additional conditions, or 27 with at least one weight-related comorbidity such as type 2 diabetes, hypertension, or dyslipidemia. Insurance companies may set higher thresholds. Our guide on how semaglutide works covers what to expect once you qualify and begin treatment.
Can I get GLP-1 medications with a BMI of 28?
Yes, if you have a documented weight-related health condition. A BMI of 28 falls within the 27-29.9 overweight range where the comorbidity pathway applies. You will need a diagnosis of type 2 diabetes, hypertension, high cholesterol, cardiovascular disease, or sleep apnea to qualify. Without a documented comorbidity, a BMI of 28 alone does not meet the FDA threshold for weight management indications.
Do BMI requirements differ for tirzepatide versus semaglutide?
No. The FDA-approved BMI thresholds are the same for both medications: 30 or higher, or 27 or higher with at least one weight-related comorbidity. The choice between semaglutide and tirzepatide depends on factors like efficacy preferences, side effect profiles, insurance coverage, and provider recommendation rather than BMI differences.
What if my insurance requires a higher BMI than the FDA threshold?
File an appeal. Include a letter of medical necessity from your provider, documentation of all qualifying comorbidities, evidence of failed prior weight loss attempts, and any supporting metabolic data. Request a peer-to-peer review between your provider and the insurance company medical director. Many initial denials are overturned on appeal. If coverage remains unavailable, explore affordable tirzepatide options or compounded semaglutide as alternatives.
Are BMI cutoffs different for Asian populations?
The WHO recommends lower cutoffs for Asian populations: obesity at BMI 27.5 (versus 30 standard) and overweight at BMI 23 (versus 25 standard). Research shows that South Asian individuals develop equivalent diabetes risk at BMI 23.9 compared to BMI 30 in white individuals. However, these adjusted thresholds have not been formally incorporated into FDA labeling for GLP-1 medications. Discuss ethnicity-adjusted risk assessment with your provider if you are of Asian descent.
Will Medicare cover GLP-1 medications for weight loss?
Medicare coverage for GLP-1 medications is expanding. The BALANCE Model, announced December 2025, creates a Medicare GLP-1 payment demonstration beginning July 2026. Qualifying criteria include BMI greater than 27 with prediabetes or established cardiovascular disease, or BMI over 30 with uncontrolled hypertension, advanced kidney disease, or heart failure. Qualifying beneficiaries will pay approximately 50 dollars per month.
Can I qualify for GLP-1 medications if my BMI is normal but I have metabolic issues?
Currently, the FDA-approved weight management indication requires a BMI of at least 27 with comorbidities. A normal BMI with metabolic issues would not qualify under the weight management pathway. However, if you have type 2 diabetes, you may qualify for GLP-1 medications approved for diabetes management (Ozempic, Mounjaro) regardless of BMI, and weight loss occurs as a secondary benefit.
How often does my BMI need to be measured for GLP-1 eligibility?
Most insurance companies require a BMI measurement documented within the past 3-6 months as part of the prior authorization process. Some require measurement at the prescribing visit itself. Your provider will typically weigh you and record your BMI at the appointment where the prescription is written. For ongoing authorization renewals, periodic BMI documentation may be required to demonstrate continued medical necessity.
External resources
STEP 1 Trial: Semaglutide in Adults with Overweight or Obesity (NEJM)
SURMOUNT-1 Trial: Tirzepatide for Treatment of Obesity (NEJM)
NIH BMI Calculator (National Heart, Lung, and Blood Institute)
For researchers serious about navigating GLP-1 eligibility and optimizing their treatment protocols, SeekPeptides offers the most comprehensive resource available. Evidence-based guides, dosing tools, treatment comparisons, and a community of thousands who have navigated these exact questions make it the go-to platform for peptide education and support.
In case I do not see you, good afternoon, good evening, and good night. May your BMI calculations stay accurate, your provider conversations stay productive, and your treatment outcomes stay exactly what the clinical trials promised.