Feb 19, 2026

On paper, they look like the same thing. Two GLP-1 medications from Eli Lilly. Both targeting weight loss. Both backed by phase 3 clinical trial data. But the moment you look beneath the surface, orforglipron and tirzepatide reveal themselves as fundamentally different tools designed for different situations, different people, and different stages of the weight management journey.
One is a daily pill. The other is a weekly injection. One activates a single receptor. The other hits two. One has been available for years under the brand name Mounjaro and Zepbound, building a massive track record of real-world weight loss results. The other is still working its way through FDA review, with an expected decision date in early-to-mid spring. And the weight loss numbers? They are not even close. Tirzepatide delivers up to 22.5% body weight reduction in clinical trials. Orforglipron maxes out around 12.4%. That is nearly double the efficacy from tirzepatide.
So why would anyone consider orforglipron at all?
Because needles are a dealbreaker for millions of people. Because daily pills fit certain lifestyles better than weekly injections. Because some people have already lost weight on injectable GLP-1 medications and need a maintenance option that does not require ongoing injections. And because the ATTAIN-MAINTAIN trial just proved something remarkable: people who switched from tirzepatide to orforglipron kept their weight off.
This guide breaks down everything you need to know about these two medications. The mechanisms. The clinical data. The side effects. The practical considerations that determine which one actually makes sense for your situation. SeekPeptides has analyzed every major clinical trial and regulatory update to give you the most comprehensive comparison available anywhere online.
No shortcuts. No oversimplifications. Just the evidence.
What is orforglipron and how does it work?
Orforglipron represents a genuinely new approach to GLP-1 receptor activation. Unlike every other GLP-1 receptor agonist on the market, including semaglutide and tirzepatide, orforglipron is not a peptide. It is a small molecule. That distinction matters enormously because peptides get destroyed in the digestive system, which is exactly why most GLP-1 medications require injection. Orforglipron survives oral administration because it is built differently at the molecular level.
The drug works as a potent partial agonist of the GLP-1 receptor. When it binds to the receptor, it triggers cAMP signaling similar to what native GLP-1 produces in the body. But here is where things get interesting. Orforglipron shows what researchers call "biased signaling." It strongly activates the cAMP pathway while producing minimal activation of the beta-arrestin pathway. Beta-arrestin is responsible for receptor internalization, which is essentially the body pulling receptors off the cell surface and reducing their availability.
Less beta-arrestin recruitment could mean less receptor desensitization over time. In theory, this pharmacological profile might allow orforglipron to maintain its effectiveness longer than traditional full GLP-1 receptor agonists. The research on this is still evolving, but the mechanism is promising.
The oral advantage explained
Oral semaglutide already exists as Rybelsus, but it comes with significant limitations. You have to take it on an empty stomach. You cannot eat or drink anything except plain water for 30 minutes afterward. You cannot even take other medications within that window. These restrictions exist because oral semaglutide uses an absorption enhancer called SNAC to get through the stomach lining, and food interferes with that process.
Orforglipron has none of those restrictions. Take it with food or without. Take it in the morning or at night. Take it with your other medications. The half-life sits between 29 and 49 hours, supporting once-daily dosing without the rigid timing requirements that make oral semaglutide inconvenient for many people.
This matters for adherence. Research consistently shows that the easier a medication is to take, the more likely people are to actually take it. And in weight management, consistency over months and years is what produces lasting results. Understanding optimal timing for GLP-1 medications becomes far simpler when the medication itself does not impose strict requirements.
Clinical effects at the biological level
At the physiological level, orforglipron enhances first-phase insulin secretion. This is the rapid burst of insulin your pancreas releases within minutes of eating. People with type 2 diabetes typically lose this first-phase response, which contributes to post-meal blood sugar spikes. Orforglipron helps restore it.
The drug also suppresses inappropriate glucagon secretion. Glucagon tells your liver to release stored glucose into the bloodstream. When glucagon stays elevated after meals, which happens in type 2 diabetes, blood sugar climbs higher than it should. Orforglipron brings glucagon back in line.
All of these effects happen in a glucose-dependent manner. That means orforglipron primarily works when blood sugar is elevated. When blood sugar is normal, the drug steps back. This glucose-dependent mechanism minimizes the risk of hypoglycemia, which is the dangerous drop in blood sugar that some diabetes medications can cause. Understanding how GLP-1 medications work in the body helps put orforglipron unique profile in context.
For appetite suppression, orforglipron slows gastric emptying and acts on brain centers that regulate hunger and satiety. These are the same mechanisms that make injectable GLP-1 medications effective for weight loss, just delivered through a pill instead of a needle.
What is tirzepatide and how does it work?
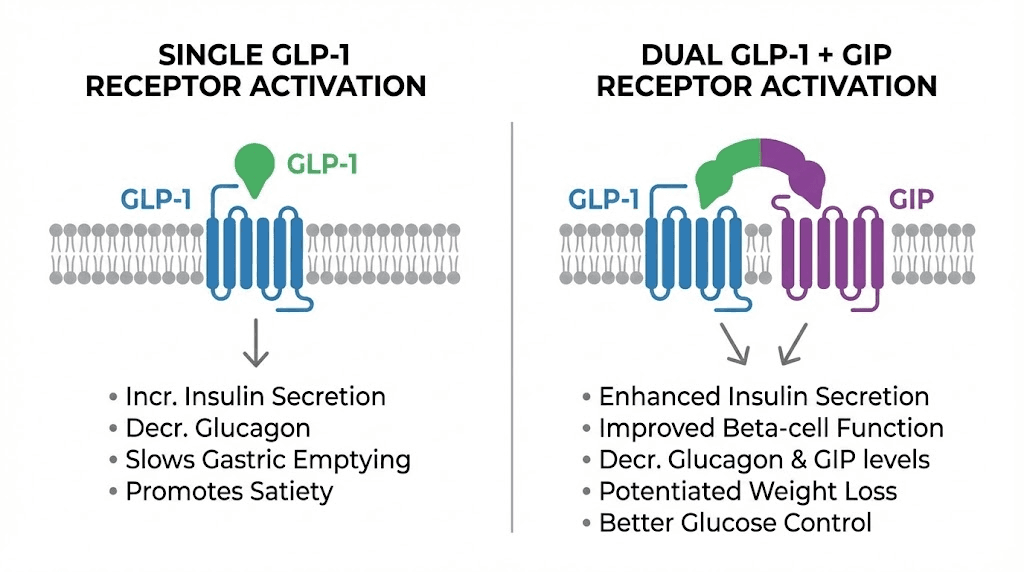
Tirzepatide is a dual-action agonist that activates both the GLP-1 receptor and the glucose-dependent insulinotropic polypeptide (GIP) receptor. This dual mechanism is what sets it apart from every single-receptor GLP-1 medication, including orforglipron, semaglutide, and liraglutide.
The GIP receptor activation adds a layer of metabolic benefit that GLP-1 alone cannot provide. GIP influences fat metabolism, improves insulin sensitivity in adipose tissue, and may play a role in how the body partitions calories between fat storage and energy use. When you combine GLP-1 and GIP receptor activation, the result is more potent appetite suppression, greater improvements in insulin sensitivity, and significantly more weight loss than either receptor activation alone.
Tirzepatide is sold under two brand names. Mounjaro for type 2 diabetes and Zepbound for weight management. The molecule is identical in both products. The difference is purely regulatory and marketing.
Administration and dosing
Tirzepatide is administered as a once-weekly subcutaneous injection using an auto-injector pen. The dosing schedule for tirzepatide follows a gradual titration pattern. Most protocols start at 2.5 mg weekly for the first four weeks, then increase to 5 mg weekly. From there, the dose can be increased in 2.5 mg increments every four weeks based on tolerability and response, up to a maximum of 15 mg weekly.
This titration approach is critical. Jumping straight to higher doses dramatically increases the risk of gastrointestinal side effects. The gradual increase gives the body time to adapt. For researchers trying to understand tirzepatide dosing in units, the conversion depends on the concentration of the specific formulation being used.
The injection itself is straightforward. The auto-injector pen does most of the work. You select the dose, press the pen against your skin, and click the button. The needle is fine gauge, and many people report barely feeling it. Common injection sites include the abdomen, thigh, and upper arm. For detailed guidance on technique, proper injection technique matters for both comfort and absorption.
The dual-receptor advantage
Why does hitting two receptors produce so much more weight loss than hitting one? The answer lies in how GLP-1 and GIP work together.
GLP-1 receptor activation primarily works through appetite suppression, slowed gastric emptying, and improved glucose metabolism. These effects are powerful on their own. Every GLP-1 receptor agonist on the market, from semaglutide to liraglutide to orforglipron, relies on these mechanisms.
GIP receptor activation adds metabolic effects that complement GLP-1. GIP improves insulin sensitivity in fat tissue, influences lipid metabolism, and appears to enhance the body ability to mobilize stored fat for energy. Some research suggests GIP activation also has central nervous system effects that amplify the appetite-suppressing signal from GLP-1.
The combination creates a synergistic effect. The total is greater than the sum of its parts. This is not theoretical. The SURMOUNT trial program proved it with hard data, showing tirzepatide weight loss results that exceeded anything achieved by GLP-1-only medications.
Head-to-head weight loss comparison
This is where the conversation gets real. Numbers do not lie, and the clinical trial data tells a clear story about relative efficacy.
Orforglipron weight loss data
The ATTAIN-1 trial was the pivotal phase 3 study for orforglipron in obesity. It ran for 72 weeks and tested three doses against placebo.
Results at 72 weeks showed mean body weight reductions of 7.5% with the 6 mg dose, 8.4% with the 12 mg dose, and 11.2% with the 36 mg dose. Placebo achieved 2.1% weight loss. The absolute weight loss with the highest dose averaged 27.3 pounds.
Looking at clinically meaningful thresholds: 54.6% of patients on the 36 mg dose achieved at least 10% body weight reduction. About 36% hit the 15% mark. And 18.4% achieved 20% or greater weight loss. These numbers are respectable. They show orforglipron works. But they also reveal its ceiling.
The phase 2 data had been more optimistic, showing up to 14.7% mean weight reduction at 36 weeks. Phase 3 results, which use larger patient populations and longer follow-up periods, came in somewhat lower. This is common in drug development. Phase 2 results often represent a best-case scenario. Phase 3 gives you the real-world picture.
Tirzepatide weight loss data
The SURMOUNT-1 trial tested tirzepatide at 5 mg, 10 mg, and 15 mg weekly doses over 72 weeks. The results were staggering.
Participants on 15 mg achieved an average of 22.5% body weight reduction, translating to approximately 52 pounds of weight loss. At the 10 mg dose, average weight loss was 21.4%. Even the lowest dose of 5 mg produced 16.0% weight loss.
The proportion of patients hitting meaningful thresholds tells an even more dramatic story. On the 15 mg dose, 63% of participants achieved at least 20% body weight reduction. A pooled analysis across all SURMOUNT trials showed that 92.7% achieved at least 5% weight loss, 84.4% hit 10%, 73.6% reached 15%, 56.1% achieved 20%, and 38.7% made it to 25%.
These numbers are unprecedented in obesity pharmacotherapy. No other medication, including surgical options for some patient populations, has consistently demonstrated this level of weight loss in clinical trials. For perspective on what these results look like in practice, real-world tirzepatide transformations confirm what the trials showed.
The direct comparison table
Metric | Orforglipron (36 mg) | Tirzepatide (15 mg) |
|---|---|---|
Mean weight loss at 72 weeks | 11.2% (27.3 lbs) | 22.5% (52 lbs) |
Patients achieving 10%+ loss | 54.6% | 84.4% |
Patients achieving 15%+ loss | 36.0% | 73.6% |
Patients achieving 20%+ loss | 18.4% | 56.1% |
Administration | Daily oral pill | Weekly subcutaneous injection |
Receptor targets | GLP-1 only | GLP-1 + GIP |
Manufacturer | Eli Lilly | Eli Lilly |
The gap is significant. Tirzepatide produces roughly double the weight loss of orforglipron at their respective highest approved or studied doses. This is not a marginal difference. It is a fundamental gap in efficacy driven by the dual-receptor mechanism.
But efficacy is only one variable in the decision. Understanding how different GLP-1 medications compare requires looking at the complete picture, including tolerability, convenience, cost, and individual circumstances.
Side effects and safety comparison
Both medications share the same general side effect profile common to all GLP-1 receptor agonists. The gastrointestinal effects dominate. But there are important differences in severity, timing, and discontinuation rates that matter for real-world use.
Orforglipron side effects
The most common adverse events in orforglipron trials were gastrointestinal: nausea, diarrhea, constipation, and vomiting. These effects were predominantly mild to moderate and occurred most frequently during dose escalation phases. Once patients stabilized on their target dose, GI symptoms generally improved.
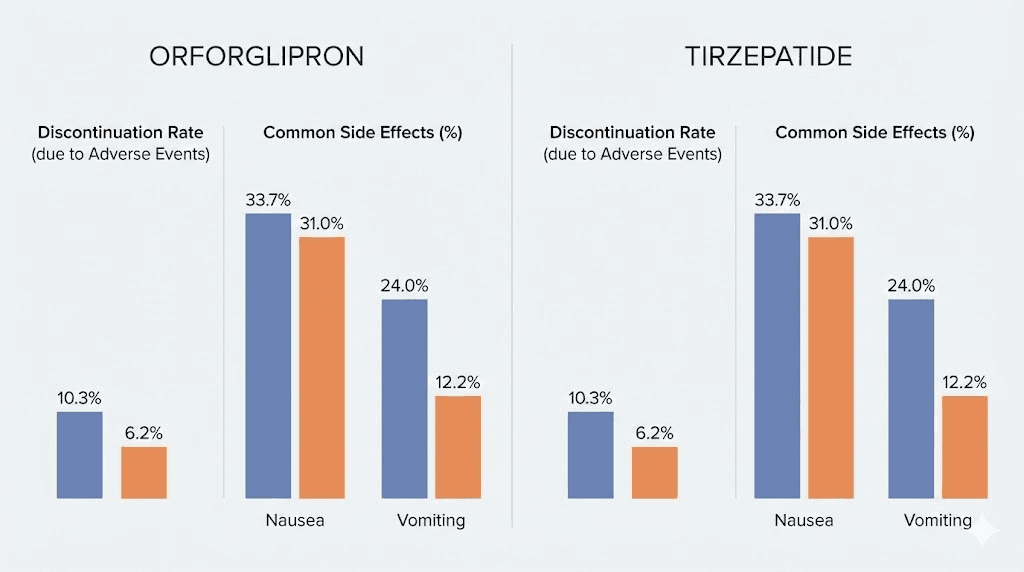
Discontinuation rates due to adverse events ranged from 5.3% to 10.3% across orforglipron dose groups, compared to 2.7% in the placebo group. These rates are within the range seen with other GLP-1 receptor agonists, though slightly higher at the upper doses. The dose-dependent nature of side effects is important: higher doses (36-45 mg daily) produced more GI symptoms than lower doses (6-12 mg daily).
No significant liver problems were observed in clinical trials. Serious side effects like pancreatitis and gallbladder inflammation were reported but remained rare, consistent with the broader GLP-1 class. For context on GI management strategies, many of the approaches used for managing GLP-1 constipation and tirzepatide-specific GI issues apply to orforglipron as well.
Tirzepatide side effects
Tirzepatide shares the same GI side effect profile. Nausea, vomiting, diarrhea, and constipation are the most common adverse events. Like orforglipron, these occur most frequently during dose titration and tend to improve over time.
Where tirzepatide differs is in the breadth of documented side effects from its longer time on market and larger real-world patient population. Reports include fatigue, headaches, muscle pain, anxiety, and injection site reactions. Most of these are mild and manageable. The hair loss concern that some GLP-1 users report appears to be related to rapid weight loss itself rather than the medication directly.
Discontinuation rates in SURMOUNT trials were comparable to other GLP-1 medications. The gradual titration schedule helps minimize early side effects, and most patients who complete the titration phase tolerate the medication well long-term.
Safety comparison table
Safety metric | Orforglipron | Tirzepatide |
|---|---|---|
Most common side effects | Nausea, diarrhea, constipation, vomiting | Nausea, diarrhea, constipation, vomiting |
Severity | Mostly mild to moderate | Mostly mild to moderate |
Discontinuation rate (adverse events) | 5.3-10.3% | 4.3-7.1% |
When side effects peak | During dose escalation | During dose titration |
Hypoglycemia risk | Low (glucose-dependent) | Low (glucose-dependent) |
Injection site reactions | None (oral) | Occasional, mild |
Serious adverse events | Rare (pancreatitis, gallbladder) | Rare (pancreatitis, gallbladder) |
The side effect profiles are remarkably similar. This makes sense because both drugs act through GLP-1 receptor pathways, and GI effects are inherent to that mechanism. The main practical difference is that orforglipron eliminates injection site reactions entirely, while tirzepatide occasionally causes mild redness or discomfort at the injection site.
Managing side effects on either medication follows similar strategies. Eating smaller, more frequent meals helps with nausea. Staying hydrated addresses constipation. Starting at lower doses and titrating slowly gives the body time to adapt. For detailed dietary guidance, understanding what to eat while on GLP-1 medications and foods to avoid can significantly reduce GI discomfort.
The switching study: ATTAIN-MAINTAIN results
Perhaps the most important piece of evidence for understanding how orforglipron and tirzepatide relate to each other is not a head-to-head competition. It is a switching study. The ATTAIN-MAINTAIN trial asked a question nobody had answered before: can people who lose weight on injectable GLP-1 medications maintain that weight loss after switching to an oral pill?
The answer, based on the data, is yes.
Study design
ATTAIN-MAINTAIN enrolled participants who had already completed 72 weeks of treatment with the highest tolerated doses of either semaglutide (Wegovy) or tirzepatide (Zepbound) as part of the SURMOUNT-5 trial. These patients had already achieved significant weight loss. The question was whether transitioning them to orforglipron would preserve those results or whether they would regain the weight.
Participants were randomized to receive either orforglipron or placebo for 52 weeks after stopping their injectable medication. This design directly tested orforglipron ability to serve as a maintenance therapy.
Results from the semaglutide switch
Patients who switched from semaglutide to orforglipron maintained their weight loss effectively. The mean change in body weight from the switch point was minus 0.1 kg with orforglipron, compared to plus 9.4 kg with placebo at 24 weeks. That is a difference of 9.5 kg. The placebo group gained back nearly 21 pounds while the orforglipron group stayed essentially flat.
Over the full study period, the average difference between orforglipron and placebo was 0.9 kg among those switching from semaglutide. For people who had built their weight loss on months of semaglutide therapy, orforglipron successfully preserved those gains.
Results from the tirzepatide switch
The tirzepatide-to-orforglipron switch showed a slightly different pattern. Because tirzepatide produces more weight loss than semaglutide (dual receptor versus single receptor), and because orforglipron is a single GLP-1 receptor agonist, there was an expected gap in potency.
Patients switching from tirzepatide to orforglipron gained an average of 2.6 kg, compared to 9.1 kg with placebo at 24 weeks. So there was some weight regain, about 5.7 pounds on average, but dramatically less than the 20 pounds regained by the placebo group. The overall difference between orforglipron and placebo was approximately 5 kg.
This makes pharmacological sense. Tirzepatide hits two receptors. Orforglipron hits one. Switching from a dual agonist to a single agonist means you lose the GIP component. Some degree of reduced efficacy is expected. But the key finding is that orforglipron still provided dramatically better weight maintenance than stopping treatment entirely.
What this means practically
ATTAIN-MAINTAIN opens up a new treatment paradigm. Someone could use tirzepatide during the active weight loss phase, achieving the maximum possible weight reduction with the dual-receptor mechanism, and then transition to orforglipron for long-term maintenance. This approach would eliminate the need for ongoing injections during the maintenance phase, which could last years or even decades.
The data also supports using orforglipron as a bridge for people who need to temporarily stop injections, whether for travel, surgery, supply issues, or personal preference. Understanding medication transitions in the GLP-1 space is becoming increasingly important as more options become available.
For those concerned about what happens when stopping GLP-1 medications, orforglipron represents a potential off-ramp that avoids the dramatic weight regain typically seen with abrupt discontinuation.
Dosing protocols compared
Understanding the dosing landscape for both medications helps clarify what daily treatment actually looks like in practice.
Orforglipron dosing
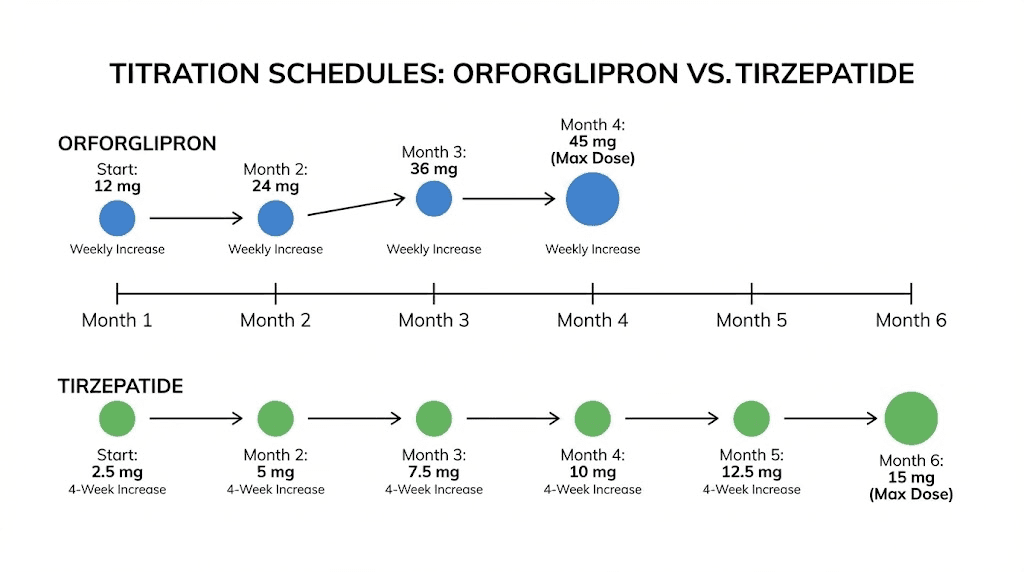
Orforglipron titration typically starts at 3 mg daily for the first two weeks, then increases to 6 mg daily. From there, the dose can be increased to 12 mg, 24 mg, or up to 36 mg daily based on tolerability and response. Some protocols have tested up to 45 mg daily, though 36 mg appears to offer the best balance of efficacy and tolerability.
The optimal doses for weight loss appear to be 24 mg and 36 mg daily based on the phase 3 data. The 12 mg dose provides meaningful but more modest weight loss. The 6 mg dose offers the gentlest introduction with the fewest side effects but produces the smallest weight reduction.
Because orforglipron is a daily medication, the titration happens more quickly than with weekly injectables. You are adjusting the dose every 1-2 weeks rather than every 4 weeks. This can mean reaching the effective dose sooner, but it also means the dose escalation side effects come faster. For researchers familiar with standard GLP-1 titration schedules, orforglipron follows a similar principle of gradual increase but on a compressed timeline.
Tirzepatide dosing
Tirzepatide follows a well-established titration schedule. Start at 2.5 mg weekly for four weeks. Increase to 5 mg weekly for at least four weeks. Then increase by 2.5 mg increments every four weeks as tolerated, up to the maximum of 15 mg weekly.
The full titration from 2.5 mg to 15 mg takes a minimum of 20 weeks. Many clinicians extend this timeline, spending extra weeks at intermediate doses to minimize side effects. There is no rush. The gradual approach produces better tolerability and often better long-term outcomes. Complete tirzepatide dosing charts provide visual reference for the standard schedule.
For compounded versions, the dosage calculator helps determine exact volumes based on concentration and target dose. Understanding the reconstitution process is essential for anyone working with lyophilized formulations.
Dosing comparison table
Parameter | Orforglipron | Tirzepatide |
|---|---|---|
Starting dose | 3 mg daily | 2.5 mg weekly |
Maximum dose | 36-45 mg daily | 15 mg weekly |
Titration interval | 1-2 weeks | 4 weeks |
Time to max dose | 8-12 weeks | 20+ weeks |
Frequency | Once daily | Once weekly |
Food restrictions | None | N/A (injection) |
Half-life | 29-49 hours | 5 days (120 hours) |
The half-life difference is notable. Tirzepatide longer half-life of approximately 5 days is what allows once-weekly dosing. Orforglipron shorter half-life of 29-49 hours requires daily administration. For people who struggle with daily medication adherence, the weekly injection may actually be easier to manage despite requiring a needle.
Oral versus injectable: the practical reality
The pill-versus-needle debate is not purely about preference. It has real implications for treatment adherence, lifestyle compatibility, and long-term outcomes.
Why some people prefer oral medications
Needle phobia affects an estimated 20-25% of adults. For these individuals, injectable medications are not just inconvenient. They are psychologically impossible. The anxiety, avoidance behavior, and stress associated with self-injection can completely prevent treatment adherence. For this population, orforglipron is not just an alternative. It is the only viable GLP-1 option.
Beyond phobia, there are practical considerations. Oral medications do not require refrigeration (orforglipron is stable at room temperature during use). They do not require proper injection technique or disposal of sharps. They do not raise questions during travel through airport security. They do not require the careful travel planning that injectable medications demand.
For people who already take daily medications, adding one more pill is familiar. The behavioral pattern exists. There is no learning curve.
Why some people prefer weekly injections
Counterintuitively, many people find weekly injections easier to manage than daily pills. You do it once, on the same day each week, and you do not think about it again for seven days. With daily medications, every single day requires remembering. Miss a day, and you have a gap in coverage.
Adherence data across medication classes consistently shows that less frequent dosing correlates with better compliance. Taking something once a week is objectively easier to remember than taking something every day. This is why extended-release and weekly formulations have become increasingly popular across medicine.
The injection itself, with modern auto-injector pens, is quick and nearly painless. People who have been properly trained on GLP-1 injection technique report that the process takes less than 30 seconds. Many describe it as less painful than a mosquito bite.
There is also the efficacy factor. If weekly tirzepatide injections produce double the weight loss of daily orforglipron pills, some people will gladly accept the needle. The results justify the method.
Storage and handling differences
Tirzepatide requires refrigeration before first use. After opening, it can be kept at room temperature for a limited time, but the storage requirements add complexity. Understanding temperature management and room temperature stability is essential for maintaining potency.
Orforglipron, as an oral tablet, has much simpler storage requirements. Standard room temperature storage. No cold chain concerns. No worrying about whether your medication got warm during shipping or while sitting in a hot mailbox. This simplicity is a genuine advantage for practical daily use.
FDA status and availability
The regulatory landscape determines when you can actually access these medications, and the timelines are very different.
Tirzepatide availability
Tirzepatide has been FDA-approved and available since 2022 for type 2 diabetes (as Mounjaro) and since 2023 for weight management (as Zepbound). It has an extensive real-world track record with millions of prescriptions filled. The supply chain has stabilized after initial shortage periods, and both branded and compounded formulations are available.
The oral form of tirzepatide is also in development, but it is a separate product from orforglipron. Lilly is pursuing both an oral tirzepatide and orforglipron, which are different molecules with different mechanisms.
Orforglipron availability
Orforglipron is not yet FDA-approved. Eli Lilly submitted a New Drug Application (NDA) for orforglipron for the treatment of obesity in adults. The FDA granted the application a Priority Review designation, and the drug also received a Commissioner National Priority Voucher, which could further accelerate the review process.
The target action date (PDUFA date) was initially set for late March but has shifted to approximately April 10 of this year. Some analysts expect the FDA decision could come in the second quarter. If approved, Lilly would still need to ramp up manufacturing and distribution, meaning widespread availability might not happen until later in the year.
A separate regulatory submission for orforglipron in type 2 diabetes is planned for later this year. So the obesity indication would come first, with diabetes following.
What this means for you
Right now, tirzepatide is the only option of the two that you can actually get. Orforglipron exists in clinical trials and regulatory review. If your weight management goals cannot wait for FDA approval and commercial launch, tirzepatide (or semaglutide) are the available choices.
When orforglipron does become available, it will likely fill a specific niche: people who want an oral option, people transitioning off injectables for maintenance, and people for whom needle-based treatment is not feasible. It probably will not replace tirzepatide for people pursuing maximum weight loss, given the roughly 2x efficacy gap.
Who should consider orforglipron?
Orforglipron is not a universally better or worse option than tirzepatide. It is a different tool with different strengths. Here is who it makes the most sense for.
People with needle phobia or injection aversion
If needles are a hard no, orforglipron removes that barrier entirely. For millions of people, this is the difference between getting treated and not getting treated. No amount of counseling about how small the needle is or how painless modern injection pens are will convince someone with genuine needle phobia. Orforglipron makes GLP-1 therapy accessible to this population for the first time in a meaningful way.
People in the maintenance phase
The ATTAIN-MAINTAIN data specifically supports using orforglipron after achieving weight loss with injectable medications. If you have reached your target weight on tirzepatide or semaglutide and want to maintain those results without continuing injections indefinitely, orforglipron could serve as a step-down therapy. You lose some potency compared to tirzepatide, but you gain the convenience of a daily pill with no injection requirements.
People who prioritize convenience
No refrigeration. No sharps disposal. No injection site rotation. No reconstitution. No supply chain cold storage concerns. For people whose lifestyle makes injectable medication management difficult, whether due to travel, living situation, or personal preference, orforglipron simplifies the daily routine. Understanding these practical differences helps when comparing oral and injectable GLP-1 options.
People with moderate weight loss goals
Not everyone needs to lose 22% of their body weight. Someone with a BMI of 32 who wants to lose 25-30 pounds might find orforglipron perfectly adequate. The 11-12% average weight loss at the highest dose could be exactly what they need, without the intensity of a dual-receptor agonist. For understanding weight management trajectories, how quickly different GLP-1 medications produce results varies based on the specific agent and dose.
Who should consider tirzepatide?
Tirzepatide remains the more potent option for several specific populations.
People with significant weight to lose
If your BMI is 40+ and you need to lose 80, 100, or more pounds, tirzepatide dual-receptor mechanism provides the strongest pharmacological support available. The 22.5% average weight loss, with some patients achieving 25%+ reductions, offers the best chance of reaching a healthy weight range. For this population, the injection requirement is a small price for dramatically superior results. Tracking progress through documented weight loss journeys helps set realistic expectations.
People with type 2 diabetes
Tirzepatide dual mechanism provides additional metabolic benefits beyond weight loss. The GIP receptor activation improves insulin sensitivity in ways that GLP-1-only medications cannot fully replicate. For people managing both obesity and type 2 diabetes, tirzepatide addresses both conditions simultaneously with a single medication. Understanding how tirzepatide affects metabolism helps explain why it is particularly effective in this population.
People who want weekly dosing simplicity
Once a week. Same day. Done. For people who struggle with daily medication adherence but can reliably remember a weekly routine, tirzepatide injection schedule may actually be easier to maintain than orforglipron daily pill. The irony of preferring an injection for simplicity is real but backed by adherence data.
People who have plateaued on other GLP-1 medications
If semaglutide stopped working or never produced adequate results, tirzepatide dual mechanism offers a different pharmacological approach. The addition of GIP receptor activation can restart weight loss that stalled on GLP-1-only therapy. Many people who experience plateaus on one medication find success with tirzepatide different mechanism of action.
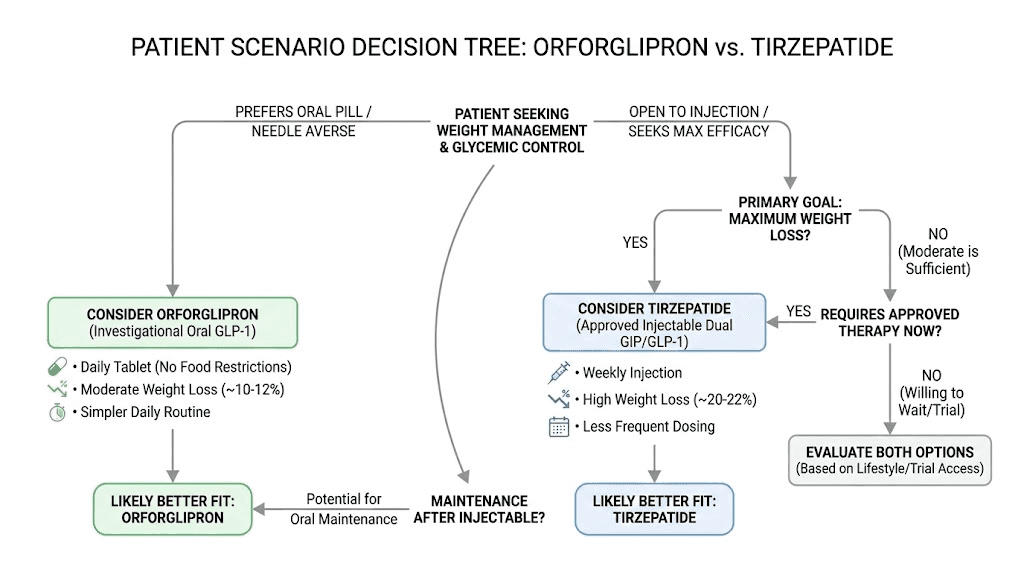
Can you use both? The sequential approach
The ATTAIN-MAINTAIN trial introduces a compelling treatment strategy: using tirzepatide and orforglipron sequentially rather than choosing one or the other.
The two-phase protocol concept
Phase 1 would use tirzepatide for active weight loss. The dual-receptor mechanism produces maximum fat reduction over 6-12 months of treatment. You leverage the most potent tool available during the phase where aggressive weight loss is the primary goal.
Phase 2 would transition to orforglipron for weight maintenance. Once you have reached your target weight, you switch from weekly injections to a daily pill. The ATTAIN-MAINTAIN data shows this transition is feasible with minimal weight regain, particularly for patients who were previously on semaglutide and even for those on tirzepatide (with modest regain of approximately 5 kg).
This approach plays to the strengths of each medication. Tirzepatide for power. Orforglipron for practicality. It also acknowledges that weight management is a lifelong endeavor, and the optimal tool might change as goals and circumstances evolve.
Transition considerations
The switch from tirzepatide to orforglipron does not happen overnight. There is an adjustment period as the body transitions from dual-receptor coverage to single-receptor coverage. The GIP component disappears, and some metabolic effects shift. The ATTAIN-MAINTAIN trial showed that this transition is manageable, but patients should expect some adaptation.
During the transition, maintaining good nutrition is essential. Understanding dietary strategies for GLP-1 therapy helps optimize outcomes regardless of which medication you are on. Supportive supplements can also help bridge any nutritional gaps during the transition period.
The sequential approach makes particular sense for people who plan to be on GLP-1 therapy long-term. The convenience of switching to a daily pill for maintenance could improve quality of life and treatment satisfaction over years of ongoing therapy.
Cost and access considerations
The financial picture for these two medications reflects both their regulatory status and market positioning.
Tirzepatide costs
Branded tirzepatide (Mounjaro and Zepbound) carries a significant list price that puts it out of reach for many patients without insurance coverage. However, the landscape has evolved with various pathways to affordable tirzepatide, including manufacturer savings cards, insurance coverage for qualifying conditions, and compounded alternatives.
Compounded tirzepatide has significantly reduced costs for many patients, though the regulatory landscape around compounding continues to evolve. Several compounding pharmacies have offered tirzepatide at a fraction of the branded price, making the medication accessible to a much broader population. For those exploring this route, understanding how different providers like Empower Pharmacy operate provides useful context.
Orforglipron costs (projected)
Since orforglipron is not yet approved, pricing has not been finalized. However, oral medications are generally cheaper to manufacture than injectable biologics. The small-molecule nature of orforglipron means production costs should be lower than peptide-based injectables. Whether Lilly will pass those savings to consumers remains to be seen.
Industry analysts expect orforglipron to be priced competitively but not dramatically cheaper than existing GLP-1 options, at least initially. The weight loss market supports premium pricing, and Lilly has no financial incentive to undercut its own tirzepatide franchise. The pricing strategy will likely position orforglipron as an alternative option rather than a budget replacement.
One potential cost advantage: oral medications eliminate the expenses associated with injection supplies (needles, syringes, sharps containers, alcohol swabs) that add up over months and years of treatment.
How orforglipron compares to other oral GLP-1 options
Orforglipron is not the only oral GLP-1 option in development or on the market. Understanding where it fits in the broader landscape helps clarify its unique position.
Oral semaglutide (Rybelsus)
Rybelsus has been available for years as an oral GLP-1 option for type 2 diabetes. However, it has significant limitations. The fasting requirement (empty stomach, no food or water for 30 minutes) makes it inconvenient. Its bioavailability is low, meaning most of the drug gets destroyed before reaching the bloodstream. And it has not been approved for weight management at its currently available doses.
Orforglipron addresses all of these limitations. No fasting requirement. Higher bioavailability as a small molecule. And clinical trial data specifically supporting weight management. For anyone currently considering oral semaglutide options, orforglipron will likely represent a significant upgrade once available.
Oral tirzepatide (in development)
Lilly is also developing an oral formulation of tirzepatide itself. This would be a different product from orforglipron, essentially the same dual-receptor molecule but in pill form. If successful, oral tirzepatide could combine the dual-receptor potency of injectable tirzepatide with the convenience of a pill.
However, formulating a peptide for oral delivery is challenging. The bioavailability and efficacy of oral tirzepatide may not match the injectable form. The tablet versus injection comparison for tirzepatide involves trade-offs that are still being defined through clinical trials.
Other emerging oral options
Several other oral GLP-1 receptor agonists are in various stages of development. Danuglipron (from Pfizer) showed mixed results and faced development challenges. Other pharmaceutical companies are pursuing their own small-molecule approaches. The oral GLP-1 space is getting crowded, but orforglipron currently leads the pack in terms of clinical data and regulatory progress.
Metabolic benefits beyond weight loss
Both orforglipron and tirzepatide offer metabolic improvements that extend beyond the number on the scale. Understanding these broader effects helps appreciate the full clinical picture.
Cardiovascular markers
Orforglipron demonstrated significant improvements in waist circumference, systolic blood pressure, triglyceride levels, and non-HDL cholesterol in the ATTAIN-1 trial. These cardiovascular risk factors improved proportionally with weight loss, suggesting that the metabolic benefits are driven at least partly by the weight reduction itself.
Tirzepatide showed similar cardiovascular improvements in the SURMOUNT trials, with the added benefit of greater magnitude due to greater weight loss. The SURPASS-CVOT trial is specifically evaluating tirzepatide cardiovascular outcomes. Given the broader systemic effects of GLP-1 medications, the cardiovascular data is an important piece of the overall value proposition.
Blood sugar control
Both medications improve glycemic control, but through somewhat different mechanisms. Orforglipron enhances first-phase insulin secretion and suppresses glucagon through GLP-1 receptor activation. Tirzepatide does this plus adds GIP-mediated improvements in insulin sensitivity.
For people with type 2 diabetes, tirzepatide has demonstrated superior A1C reduction compared to GLP-1-only medications. The SURPASS trial program showed A1C reductions of up to 2.3% with tirzepatide 15 mg, which is exceptional. Orforglipron, while effective for glycemic control, achieves its blood sugar benefits through a single receptor pathway.
Body composition changes
One concern with rapid weight loss from any mechanism is the loss of lean muscle mass alongside fat. Both medications cause some lean mass loss, but the ratio of fat loss to lean mass loss appears to favor tirzepatide based on available body composition data from the SURMOUNT trials.
The GIP receptor activation in tirzepatide may contribute to better preservation of muscle mass during weight loss, though the evidence is not yet definitive. For anyone concerned about maintaining muscle during GLP-1 therapy, combining medication with adequate protein intake and resistance training is essential regardless of which medication is used.

The bigger picture: GLP-1 landscape evolution
Orforglipron and tirzepatide exist within a rapidly evolving treatment landscape. Understanding where they fit helps make better decisions about current and future options.
The injectable era is not ending
Oral options like orforglipron will not replace injectable GLP-1 medications. They will complement them. Injectable tirzepatide will likely remain the gold standard for maximum weight loss for the foreseeable future, while oral options serve different patient populations and treatment phases.
Other injectable options continue to advance. Retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors, has shown even more impressive weight loss data than tirzepatide in early trials. The comparison between mazdutide and tirzepatide and mazdutide versus retatrutide shows the next generation of multi-receptor agonists pushing efficacy even higher.
Personalized treatment approaches
The future of GLP-1 therapy is not about finding one best medication. It is about matching the right medication to the right patient at the right time. Someone might start with orforglipron as a first-line oral option, switch to tirzepatide if they need more aggressive intervention, add combination therapies like cagrilintide for additional benefit, and eventually step down to oral maintenance therapy.
This kind of personalized, phase-based approach requires understanding the full toolkit. SeekPeptides helps researchers navigate these complex decisions with evidence-based protocols and comprehensive medication guides tailored to individual goals and circumstances.
Compounding and access
The availability of compounded GLP-1 medications has transformed access for many patients. While orforglipron as a small molecule could potentially be compounded after patent considerations, the regulatory landscape around compounding is complex and evolving. For now, branded pharmaceutical products and compounded formulations of existing medications represent the primary access pathways.
Combining GLP-1 therapy with lifestyle optimization
Neither orforglipron nor tirzepatide works optimally in isolation. The medications create a biological environment that supports weight loss, but lifestyle factors determine how much of that potential you actually realize.
Nutrition on GLP-1 therapy
Both medications reduce appetite, which means you naturally eat less. But the quality of what you eat matters enormously when caloric intake drops. Every calorie needs to count. High-protein, nutrient-dense foods become essential for preserving muscle mass, supporting metabolism, and providing the vitamins and minerals your body needs.
Detailed diet plans optimized for GLP-1 therapy focus on protein intake of 1.0-1.2 grams per kilogram of body weight daily, adequate fiber for gut health and satiety, and micronutrient-dense foods to prevent deficiency. The semaglutide diet plan principles apply equally to orforglipron since both work through GLP-1 receptor-mediated appetite suppression.
Exercise and muscle preservation
Resistance training during GLP-1 therapy is not optional. It is essential. Without strength training stimulus, the body will catabolize muscle tissue alongside fat, leading to a worse body composition outcome despite the number on the scale going down. This is particularly important for people on more aggressive protocols where rapid weight loss increases muscle loss risk.
Understanding how GLP-1 medications affect energy levels helps plan exercise around periods of optimal energy. Some people find that their energy fluctuates during dose titration, stabilizing once they reach their maintenance dose.
Supplement support
GLP-1 therapy can create specific nutritional gaps that targeted supplementation addresses. Vitamin B12 supplementation is commonly recommended because GLP-1 medications can reduce absorption of this crucial vitamin. Some formulations, like tirzepatide compounded with glycine and B12, address this directly.
Other supplements worth considering include glycine for metabolic support, semaglutide with glycine combinations, and niacinamide for cellular health. The complete supplement guide for GLP-1 therapy covers the evidence for each option.
Troubleshooting common concerns
Real-world GLP-1 therapy comes with questions that clinical trial summaries do not always address. Here are the most common concerns and evidence-based answers.
What if the medication stops working?
Weight loss plateaus happen with both orforglipron and tirzepatide. This does not mean the medication has failed. It often means the body has reached a new equilibrium at a lower weight. Strategies for breaking through include dose optimization, dietary adjustments, exercise modifications, and in some cases, medication switching. For specific troubleshooting, addressing tirzepatide plateaus and semaglutide non-response guides provide actionable protocols.
Can you drink alcohol on these medications?
Both GLP-1 medications interact with alcohol in ways that warrant caution. Appetite suppression can lead to drinking on an emptier stomach, intensifying alcohol effects. GI side effects can worsen with alcohol consumption. And alcohol is calorie-dense, working against weight loss goals. Detailed guidance on alcohol and tirzepatide and alcohol and semaglutide covers the nuances.
What about combining with other medications?
Questions about combining GLP-1 therapy with other weight loss aids are common. The evidence for phentermine with GLP-1 medications is mixed, and the direct comparison between phentermine and GLP-1 agents generally favors the GLP-1 medications for both efficacy and safety. Always consult a healthcare provider before combining medications.
Timing and scheduling questions
People frequently ask about taking GLP-1 medications early or adjusting their schedule. With orforglipron daily dosing, timing flexibility is built in since there are no food restrictions. With tirzepatide weekly dosing, the long half-life provides some flexibility, though consistency is ideal.
Frequently asked questions
Is orforglipron the same as tirzepatide in pill form?
No. Orforglipron and tirzepatide are completely different molecules. Orforglipron is a small-molecule, non-peptide GLP-1 receptor agonist that only activates the GLP-1 receptor. Tirzepatide is a peptide-based dual agonist that activates both GLP-1 and GIP receptors. Oral tirzepatide is a separate product also in development by Eli Lilly, which would be the actual tirzepatide molecule formulated for oral delivery.
How much weight can you lose on orforglipron versus tirzepatide?
In phase 3 clinical trials, orforglipron at its highest studied dose (36 mg daily) produced an average weight loss of 11.2% (about 27 pounds) over 72 weeks. Tirzepatide at 15 mg weekly produced 22.5% weight loss (about 52 pounds) over the same period. Tirzepatide delivers roughly double the weight loss, driven by its dual-receptor mechanism. Individual results vary significantly based on dose, diet, exercise, and starting weight.
When will orforglipron be available?
The FDA target action date for orforglipron is approximately April 10. If approved, commercial availability would follow, though manufacturing ramp-up could delay widespread access. Tirzepatide is already available as both Mounjaro and Zepbound.
Can you switch from tirzepatide to orforglipron?
Yes. The ATTAIN-MAINTAIN trial demonstrated that patients switching from tirzepatide to orforglipron maintained most of their weight loss, with modest regain of approximately 5 kg on average. This was dramatically better than the placebo group, which regained approximately 9 kg. The switch is feasible and supported by clinical data. For guidance on switching between GLP-1 medications, similar transition principles apply.
Are the side effects of orforglipron worse than tirzepatide?
The side effect profiles are very similar. Both medications primarily cause gastrointestinal effects including nausea, diarrhea, constipation, and vomiting. These are mostly mild to moderate and improve over time. Orforglipron discontinuation rates due to side effects ranged from 5.3-10.3%, while tirzepatide rates were 4.3-7.1%. The main difference is that orforglipron eliminates injection site reactions since it is an oral medication.
Is orforglipron cheaper than tirzepatide?
Pricing for orforglipron has not been announced since it is not yet approved. Small-molecule oral medications are generally cheaper to manufacture than injectable peptide biologics, but market pricing depends on many factors beyond manufacturing cost. Industry analysts do not expect a dramatic price difference initially, as Eli Lilly is unlikely to undercut its own tirzepatide products significantly.
Can you take orforglipron with food?
Yes. Unlike oral semaglutide (Rybelsus), which requires an empty stomach and 30-minute fasting window, orforglipron can be taken with or without food at any time of day. There are no food or water restrictions. This is one of its key advantages over other oral GLP-1 options.
Does orforglipron work for type 2 diabetes?
Yes. Orforglipron has demonstrated statistically significant improvements in blood sugar control in the ATTAIN trials for type 2 diabetes. However, the initial FDA submission is for obesity only. A separate submission for the diabetes indication is expected later. In contrast, tirzepatide is already approved for both obesity (Zepbound) and type 2 diabetes (Mounjaro).
External resources
ATTAIN-1 Trial: Orforglipron for Obesity Treatment (New England Journal of Medicine)
SURMOUNT-1 Trial: Tirzepatide for Obesity (New England Journal of Medicine)
GLP-1 Receptor Agonists for Type 2 Diabetes and Obesity Review (PubMed Central)
For researchers serious about optimizing their GLP-1 protocols and understanding how these medications fit into a comprehensive weight management strategy, SeekPeptides offers the most comprehensive resource available. Evidence-based guides, detailed comparison tools, and a community of experienced researchers who have navigated these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your choices stay informed, your protocols stay optimized, and your results stay lasting.