Feb 12, 2026

Six months ago, a researcher posted a question on a peptide forum that generated over 400 replies. The question was simple. Can semaglutide drops actually work as well as injections? The answers ranged from enthusiastic endorsements to complete dismissal. Some users reported losing 15 pounds in their first month on sublingual drops. Others said they felt nothing at all, not even the nausea that typically signals the peptide is active. The confusion was not surprising. Oral semaglutide drops represent one of the most misunderstood delivery methods in the entire peptide weight loss space, and the gap between doing it right and doing it wrong determines whether the drops work at all.
The core challenge is absorption. Injectable semaglutide delivers nearly 100% of the active compound directly into subcutaneous tissue, where it enters the bloodstream efficiently. Oral semaglutide drops bypass the gastrointestinal tract entirely by absorbing through the mucous membranes under the tongue, but only if you follow the protocol precisely. Hold the drops for 45 seconds instead of the recommended two minutes and you have just swallowed most of your dose into a stomach full of digestive enzymes that will destroy the peptide before it does anything useful. The difference between injectable vs oral peptides comes down to bioavailability, and with oral semaglutide drops, technique is everything.
This guide covers every detail that determines whether oral semaglutide drops will actually work for you. Absorption mechanics. Dosing protocols from starting dose through maintenance. The timing rules that protect bioavailability. Storage requirements that prevent degradation. Side effect management specific to the oral format. And the honest comparison between drops and injections that most sources gloss over. Whether you are considering drops because needles are not your thing or because you want a more convenient daily protocol, this is the reference that separates real results from wasted product. SeekPeptides has tracked thousands of protocols across delivery methods, and the data tells a clear story about when drops work, when they do not, and exactly what makes the difference.
What oral semaglutide drops actually are
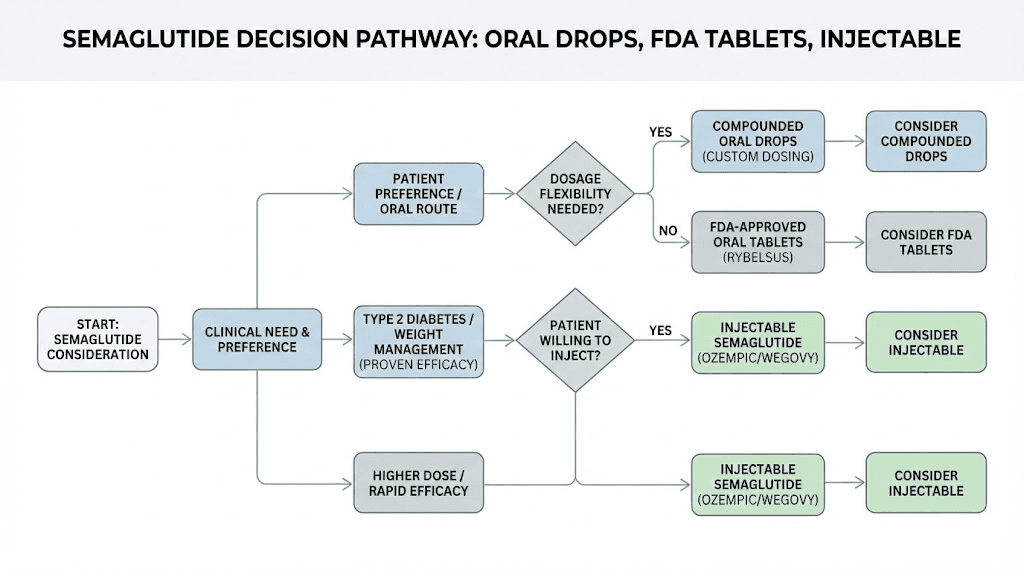
The term oral semaglutide drops can mean two very different things, and confusing them leads to bad decisions. The first is compounded sublingual semaglutide, a liquid formulation prepared by compounding pharmacies that you place under your tongue for absorption through the sublingual mucosa. The second is the FDA-approved oral semaglutide tablet, sold as Rybelsus for type 2 diabetes and more recently as oral Wegovy for weight management. These are completely different products with different mechanisms, different dosing, and different expectations.
Compounded sublingual drops are the focus of this guide. They are not FDA-approved. They are prepared by 503A or 503B compounding pharmacies using semaglutide base powder formulated into a liquid suspension. The liquid typically uses a specialized sublingual vehicle designed to enhance absorption through the tissue under your tongue. Some pharmacies use proprietary bases like SubMagna, which is engineered for delivering high molecular weight peptides through mucosal tissue.
The drops arrive in a small bottle, usually amber or opaque to protect against light degradation. You measure your dose using an oral syringe, place the liquid under your tongue, hold it there for a minimum of two minutes, and then swallow whatever remains. The semaglutide absorbs directly through the sublingual mucosa into the capillary bed beneath the tongue, bypassing the digestive system entirely.
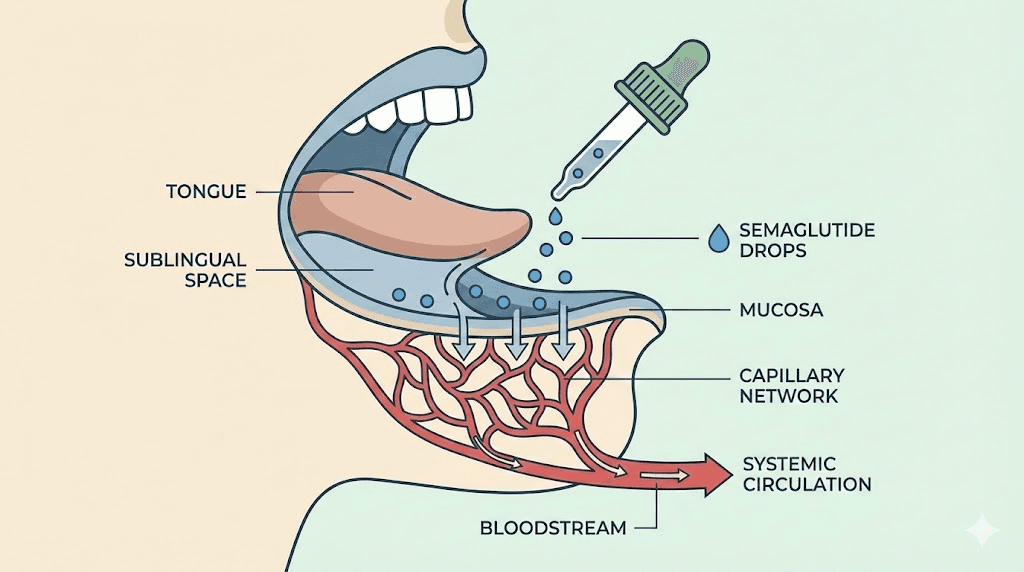
This is fundamentally different from how Rybelsus works. The FDA-approved tablet uses a technology called SNAC, which stands for sodium N-(8-[2-hydroxybenzoyl] amino) caprylate. SNAC creates a localized pH buffer around the tablet as it dissolves in the stomach, protecting the semaglutide from gastric enzymes long enough for a small percentage to absorb through the stomach lining. The bioavailability of Rybelsus is approximately 0.4% to 1%. That means for every 14mg tablet you swallow, only about 0.06mg to 0.14mg actually reaches your bloodstream. The rest gets destroyed by stomach acid and digestive enzymes. If you want to understand the full landscape of how fast semaglutide works, the delivery method is the single biggest variable.
Sublingual drops claim higher absorption than gastric tablets because the sublingual mucosa is thin, highly vascularized, and does not expose the peptide to stomach acid. However, and this is critical, there are no published clinical trials establishing the exact bioavailability of compounded sublingual semaglutide drops. The absorption rate depends on the formulation vehicle, the hold time, the concentration, and individual anatomy. Estimates from compounding pharmacies suggest sublingual bioavailability ranges from 3% to 15%, dramatically higher than the 0.4% to 1% of oral tablets but still far lower than the near-100% bioavailability of semaglutide injections.
How sublingual absorption works at the molecular level
Understanding the science behind sublingual absorption explains why technique matters so much with oral semaglutide drops. The tissue under your tongue is different from the rest of your mouth. It is non-keratinized epithelium, meaning it lacks the tough protective layer that covers your gums and hard palate. This makes it more permeable to molecules.
Beneath this thin tissue lies a dense network of capillaries. When semaglutide molecules pass through the sublingual mucosa, they enter these capillaries and drain into the sublingual vein, which feeds into the internal jugular vein, which goes directly to systemic circulation. This route completely bypasses first-pass metabolism in the liver, a major advantage over drugs that are swallowed and processed through the hepatic portal system. The sublingual peptides guide covers this pathway in more detail across different peptide types.
But semaglutide is a large molecule. It is a 31-amino acid peptide with a molecular weight of approximately 4,113.58 daltons. Most drugs that absorb well sublingually are small molecules under 500 daltons. Nitroglycerin, for example, is only 227 daltons and absorbs almost instantly under the tongue. Semaglutide is roughly 18 times larger. This size creates a fundamental barrier to sublingual absorption.
Compounding pharmacies address this challenge through specialized formulation vehicles. These vehicles typically contain permeation enhancers that temporarily increase the permeability of the sublingual mucosa. They may also include stabilizers that prevent the peptide from aggregating or degrading during the absorption window. The quality of this formulation is the single biggest factor determining whether your drops actually deliver meaningful amounts of semaglutide into your bloodstream.
This is why not all compounded sublingual semaglutide products are equivalent. A pharmacy using a well-designed sublingual vehicle with appropriate permeation enhancers will produce drops that absorb significantly better than a pharmacy that simply dissolves semaglutide in water or a basic solution. The difference can be five-fold or more in actual absorption. When evaluating peptide vendors and pharmacies, the formulation technology matters as much as the semaglutide itself.
Dosing protocols for oral semaglutide drops
Dosing sublingual semaglutide drops requires understanding that the numbers are not directly comparable to injectable doses. Because sublingual bioavailability is lower than injection bioavailability, the actual milligram dose in the drops is higher than the equivalent injectable dose. A sublingual dose of 1mg does not produce the same blood levels as an injectable dose of 1mg.
Most compounding pharmacies structure their sublingual semaglutide protocols in escalating phases, similar to injectable protocols but with different numbers. Here is a typical 13-week escalation protocol based on commonly prescribed dosing schedules.
Phase 1: initiation (weeks 1 to 2)
Concentration: 1mg/mL suspension. Daily dose: 0.5mL (0.5mg semaglutide). Place under tongue once daily in the morning on an empty stomach. Hold for minimum 2 minutes. Do not eat or drink for 30 minutes after dosing.
The initiation phase serves two purposes. First, it allows your GLP-1 receptors to begin upregulating in response to the agonist. Second, it gives your gastrointestinal system time to adjust to the appetite-suppressing and gastric-slowing effects of semaglutide. Starting too high almost always produces severe nausea. The appetite suppression timeline typically begins within the first week even at these starting doses.
Phase 2: early escalation (weeks 3 to 4)
Concentration: 1mg/mL suspension. Daily dose: 1mL (1mg semaglutide). Same administration protocol. The dose doubles from initiation. Most users begin noticing appetite changes during this phase. Mild nausea is common and typically resolves within three to five days of the increase.
Phase 3: mid escalation (weeks 5 to 8)
Concentration: 2mg/mL suspension. Daily dose: 1mL (2mg semaglutide). Your pharmacy will provide a new bottle with the higher concentration. Continue with the same volume but now you are receiving twice the semaglutide per dose. This is the phase where most users report significant appetite reduction and the beginning of measurable weight changes. Side effects like semaglutide-related symptoms should be monitored carefully during dose increases.
Phase 4: therapeutic dose (weeks 9 to 12)
Concentration: 3mg/mL suspension. Daily dose: 1mL (3mg semaglutide). This is the target therapeutic dose for most protocols. At this level, the sublingual delivery should be providing enough systemic semaglutide to produce meaningful GLP-1 receptor activation. Weight loss typically accelerates during this phase.
Phase 5: maintenance (week 13 and beyond)
Concentration: 3mg/mL suspension. Daily dose: 1mL (3mg semaglutide). Continue at therapeutic dose. Some protocols may increase to 4mg or 5mg daily if the response at 3mg is insufficient after four to six weeks. However, increasing beyond the standard maintenance dose significantly increases the risk of gastrointestinal side effects.
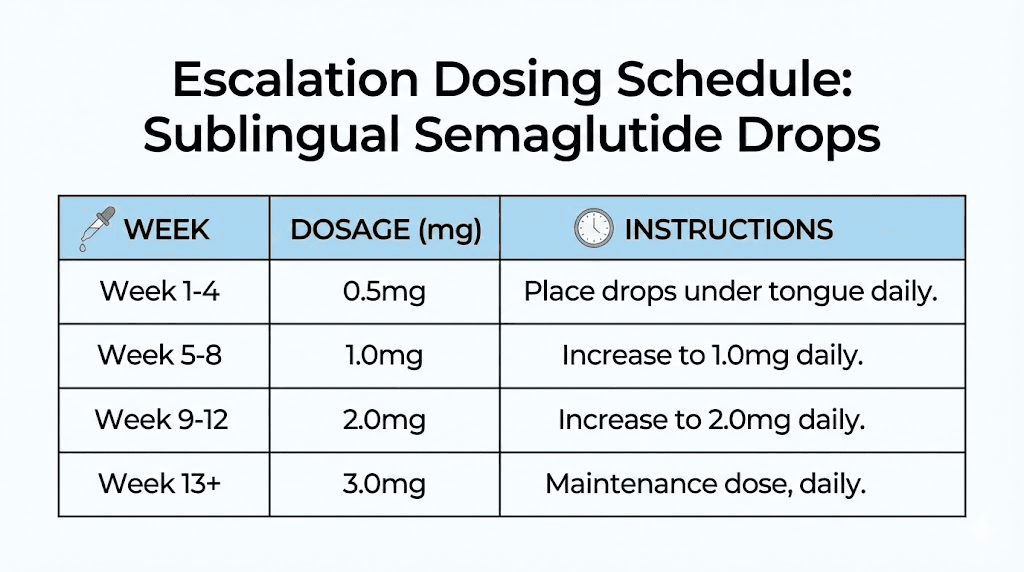
It is worth noting that these protocols vary between providers and pharmacies. Some start at 0.25mg rather than 0.5mg. Others use a faster escalation with dose increases every week instead of every two weeks. The right protocol depends on your tolerance, your response, and your provider's clinical judgment. The semaglutide dosage calculator can help you verify unit conversions if you are working with different concentrations.
The technique that makes or breaks absorption
This section might be the most important in this entire guide. You can have the highest quality compounded semaglutide drops in the world and still get zero results if your administration technique is wrong. The sublingual route is unforgiving. Small mistakes in technique can reduce absorption by 50% to 80%.
Step 1: prepare the bottle
Roll the bottle gently between your palms for 10 to 15 seconds before each use. Semaglutide in suspension can settle at the bottom of the bottle between doses. If you do not mix it before drawing your dose, the concentration in the syringe may be significantly different from what the label says. Do not shake vigorously. Aggressive shaking can create foam and potentially damage the peptide through excessive agitation. Gentle rolling is sufficient.
Step 2: draw the dose
Use the oral syringe provided by your pharmacy. Draw the prescribed volume carefully. Check for air bubbles, which can reduce the actual volume of liquid in the syringe. Expel any bubbles and re-draw if necessary. Accuracy matters. A 0.1mL error in a 1mL dose is a 10% dosing error.
Step 3: place under the tongue
Lift your tongue and place the syringe tip beneath it, as far back as comfortable without triggering a gag reflex. Slowly depress the plunger to release the liquid into the sublingual space. The goal is to pool the liquid directly over the sublingual mucosa, the tissue on the floor of your mouth beneath the tongue.
Step 4: hold for minimum two minutes
This is where most people fail. Two minutes feels much longer than you expect when you have liquid pooled under your tongue. Your instinct will be to swallow. Resist it. Set a timer on your phone. Close your mouth but keep your tongue relaxed and slightly elevated. Do not talk, do not move your tongue around, do not swallow. The liquid needs to stay in contact with the sublingual tissue for as long as possible.
Some protocols recommend holding for up to five minutes. Longer is generally better. Every additional second of contact time means more semaglutide crossing the mucosal barrier. If you can comfortably hold for three to five minutes, do it. The improvement in absorption can be substantial.
Step 5: swallow the remainder
After holding for the minimum two minutes, you can swallow whatever liquid remains in your mouth. Some semaglutide may absorb through the esophageal or gastric mucosa, though this contribution is likely minimal compared to sublingual absorption. Do not spit it out. Do not rinse your mouth.
Step 6: wait before eating or drinking
Do not eat, drink, or take other medications for at least 30 minutes after your dose. Food and beverages in the mouth can dilute any residual semaglutide that has not yet fully absorbed through the mucosa. Drinking water immediately after dosing washes away peptide that was still in contact with the sublingual tissue. This waiting period is similar to the requirements for compounded semaglutide protocols and applies equally to all oral GLP-1 formats.
Critical technique mistakes that kill absorption
Swallowing too early. This is the number one mistake. Even 60 seconds of hold time instead of 120 reduces absorption significantly. The sublingual mucosa needs sustained contact.
Taking drops with food in your stomach. Even if the drops go under your tongue, having food in your stomach changes saliva production and can trigger swallowing reflexes. Always dose on an empty stomach, ideally first thing in the morning before anything else.
Talking or moving the tongue. Any mouth movement redistributes the liquid away from the optimal sublingual contact area. Silence and stillness for two minutes.
Using too much liquid volume. If your dose requires more than 1.5mL, the pool of liquid under your tongue becomes too large to maintain without swallowing. Work with your pharmacy to get a higher concentration so you can use less volume.
Not rolling the bottle. Uneven suspension means inconsistent dosing. One day you get 0.3mg, the next you get 1.7mg. Roll the bottle every single time.
Oral semaglutide drops vs injections: the honest comparison
This comparison needs to be straightforward because too many sources either oversell drops or dismiss them entirely. Both reactions miss the nuance. Drops and injections each have legitimate advantages and meaningful limitations.
Bioavailability
Injectable semaglutide: approximately 89% to 100% bioavailability. Nearly every microgram you inject reaches systemic circulation.
Sublingual semaglutide drops: estimated 3% to 15% bioavailability depending on formulation and technique. The wide range reflects the variability in compounding quality and user compliance with hold-time protocols.
This difference is enormous. It means a 0.25mg weekly injection delivers more systemic semaglutide than a 1mg daily sublingual dose in many cases. When you look at the semaglutide 5mg dosage chart or any injectable dosing reference, you cannot directly translate those numbers to sublingual equivalents.
Dosing frequency
Injectable semaglutide: once weekly. You inject on the same day each week and do not think about it again for seven days.
Sublingual drops: once daily. Every single morning, you need to measure your dose, hold it under your tongue for two to five minutes, and then wait 30 minutes before eating or drinking. Missing doses or inconsistent timing reduces effectiveness. For people already managing complex peptide cycles, adding a daily oral dose may simplify the overall protocol by reducing injection frequency.
Convenience
This is where drops win. No needles. No syringes. No alcohol swabs. No injection site rotation. No explaining needle marks. No sharps disposal. No refrigeration requirements in some cases. For people with needle phobia, which affects an estimated 20% to 25% of the adult population, drops remove the single biggest barrier to starting a GLP-1 protocol.
Travel is also easier with drops. No carrying needles through airport security. No worrying about cold chain maintenance during long flights. No finding a private place to inject. You can dose with a small bottle and an oral syringe anywhere.
Effectiveness for weight loss
Here is the uncomfortable truth. There is no clinical trial data directly comparing compounded sublingual semaglutide drops to injectable semaglutide for weight loss. The effectiveness comparison is based on provider observations, user reports, and pharmacokinetic estimates.
Real-world observations suggest that properly dosed sublingual semaglutide at therapeutic levels can produce meaningful weight loss, typically in the range of 8% to 15% body weight over 12 to 16 weeks. Injectable semaglutide at equivalent therapeutic doses (2.4mg weekly Wegovy) produces average weight loss of 14.9% over 68 weeks in clinical trials. The injectable data is from rigorous randomized controlled trials. The sublingual data is from clinical observation. These are not equivalent levels of evidence.
That said, many providers report that patients who follow the sublingual protocol precisely, correct technique, consistent daily dosing, proper escalation, do achieve clinically meaningful results. Not always as dramatic as injection results, but meaningful enough to warrant the approach for patients who refuse or cannot tolerate injections. If you are comparing your options across the full GLP-1 landscape, the semaglutide vs tirzepatide comparison provides context for both injectable and oral formats.
Cost comparison
Brand-name injectable semaglutide (Wegovy): approximately $199 per month cash price at lower doses, $299 at higher doses. Insurance may cover with prior authorization.
Compounded injectable semaglutide: typically $150 to $350 per month depending on dose and pharmacy.
Compounded sublingual drops: typically $149 to $300 per month through telehealth providers.
FDA-approved oral semaglutide tablets (Rybelsus/oral Wegovy): $149 to $299 per month cash price depending on dose.
Cost is roughly comparable across formats. The drops are not dramatically cheaper or more expensive than injections. The choice between them should be based on preference, compliance likelihood, and clinical factors rather than price.

Side effect profile
Both formats produce similar GLP-1-related side effects because they deliver the same active molecule. Nausea, reduced appetite, constipation, diarrhea, and headache are common with both. However, there are some differences in side effect patterns.
Sublingual drops may cause mouth irritation. Some users report a bitter taste, mild tingling, or slight irritation of the tissue under the tongue. This is usually mild and resolves as tolerance develops. Drops may also cause more variable side effects because daily dosing creates more consistent blood levels than weekly injection peaks. Some users find the steady-state from daily dosing actually produces fewer dramatic side effect episodes compared to the peak-and-trough pattern of weekly injections. Understanding interactions with semaglutide is equally important regardless of delivery format.
Injectable semaglutide can cause injection site reactions including redness, swelling, and pain. It can also cause a more pronounced nausea peak in the first 24 to 48 hours after injection, followed by improvement through the rest of the week. Some users describe this as a weekly cycle of feeling rough for a day or two and then feeling fine for five.
Who should consider oral semaglutide drops
Not everyone is a good candidate for sublingual drops. The best results come from people who fit specific profiles.
Ideal candidates
People with needle phobia or severe needle anxiety. If the choice is between drops and no treatment at all, drops are the clear winner. Some level of GLP-1 activation is infinitely better than none. The getting started with peptides guide addresses common barriers including needle fear.
People who travel frequently and need a portable option without cold chain requirements. Drops in a bag are simpler than needles and vials in a cooler.
People who have difficulty with self-injection due to physical limitations. Arthritis, tremors, or limited dexterity can make drawing up and injecting challenging. An oral syringe under the tongue requires less fine motor control than a subcutaneous injection.
People who prefer daily dosing over weekly injections. Some individuals find daily administration easier to remember and integrate into their routine than weekly injections that require planning.
Poor candidates
People who want maximum effectiveness. If your primary goal is the most aggressive weight loss possible and you can tolerate injections, injectable semaglutide will almost certainly outperform drops at equivalent therapeutic intent.
People who struggle with daily compliance. Missing injectable doses once per week is much harder than missing daily oral doses. If you are the type of person who forgets to take daily medications, weekly injections are actually more forgiving. Building consistent habits around semaglutide duration and dosing schedules matters regardless of format.
People with oral lesions, chronic dry mouth, or conditions affecting the sublingual mucosa. Compromised tissue integrity reduces absorption and can cause pain during administration.
People who cannot commit to the 30-minute fasting window after dosing. If your morning routine does not allow 30 minutes between dosing and breakfast, absorption will be compromised.
Storage and stability of oral semaglutide drops
Proper storage is critical for maintaining potency. Semaglutide is a peptide, and peptides degrade when exposed to heat, light, or contamination.
Temperature requirements
Most compounded sublingual semaglutide drops should be stored at controlled room temperature between 68 and 77 degrees Fahrenheit (20 to 25 degrees Celsius). This is a significant convenience advantage over injectable semaglutide, which typically requires refrigeration at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). However, some formulations may require refrigeration, so always follow the specific instructions from your compounding pharmacy. The semaglutide storage guide covers temperature requirements across different formulations.
The anhydrous nature of many sublingual formulation vehicles contributes to stability at room temperature. Water accelerates peptide degradation through hydrolysis. Formulations that minimize water content tend to have longer shelf lives at ambient temperatures.
Light protection
Semaglutide is sensitive to photodegradation. Always store your drops in the original amber or opaque container provided by the pharmacy. Never transfer drops to a clear container. Do not leave the bottle in direct sunlight, near a window, or under bright artificial light. If you carry the bottle in a bag during the day, keep it in a pocket or pouch that blocks light.
Beyond-use dating
Compounded medications have beyond-use dates (BUDs) rather than expiration dates. The BUD is determined by the compounding pharmacy based on their stability testing and USP guidelines. Typical BUDs for compounded sublingual semaglutide range from 30 to 90 days, though this varies significantly between pharmacies and formulations. Once you open the bottle, the clock starts. Using the drops past the BUD means you may be administering degraded product with reduced potency. The general principles in using expired semaglutide apply here, the peptide does not become dangerous but it loses effectiveness.
Contamination prevention
The oral syringe touches your mouth and then goes back into the bottle for the next dose. This creates a contamination pathway. Some best practices include using a clean syringe for each dose if your pharmacy provides multiples, wiping the bottle opening with a clean tissue after each use, and never allowing the syringe to touch anything besides the inside of the bottle and the space under your tongue. Bacterial contamination accelerates peptide degradation and could theoretically cause infection, though the sublingual vehicle itself often contains antimicrobial properties.
Side effects specific to oral semaglutide drops
While the systemic side effects of semaglutide are similar regardless of delivery route, sublingual drops have some unique considerations.
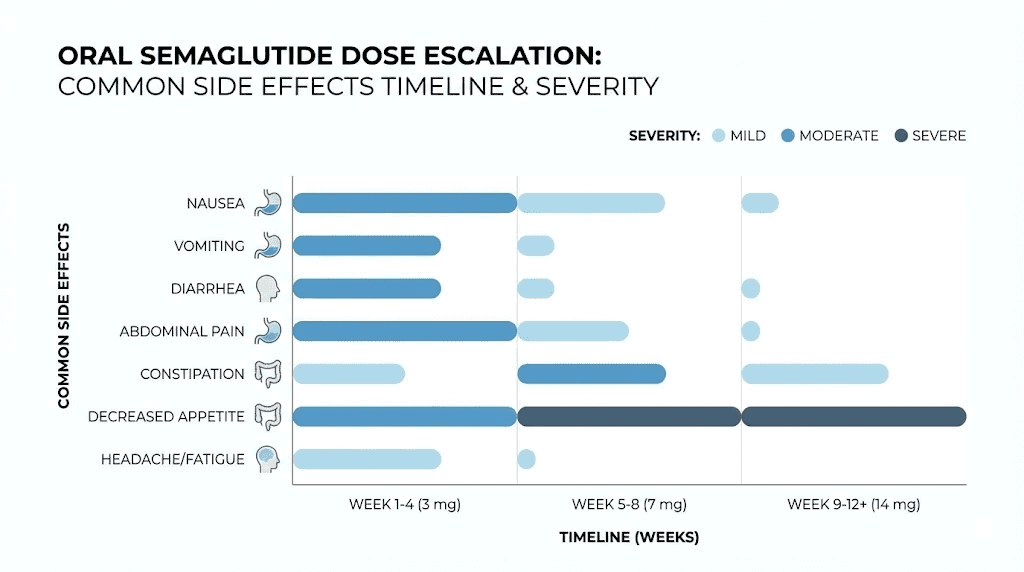
Gastrointestinal effects
Nausea is the most common side effect. It affects approximately 20% to 40% of users during the escalation phase. The nausea from sublingual drops differs from injection nausea in its timing pattern. With weekly injections, nausea tends to peak one to two days post-injection and then subside. With daily sublingual drops, the nausea is typically lower intensity but more consistent, presenting as a mild persistent queasiness rather than acute episodes.
Constipation occurs in roughly 10% to 15% of users. Semaglutide slows gastric emptying, which can reduce bowel movement frequency. Adequate hydration, fiber intake, and movement help mitigate this. Some users find that the gradual onset of slowed motility with daily dosing is actually easier to manage than the abrupt changes from weekly injections.
Diarrhea is less common than constipation but affects some users, particularly during dose escalation. It is usually self-limiting and resolves within a few days of each dose increase.
Local oral effects
Sublingual irritation. Some formulation vehicles cause mild burning, tingling, or discomfort under the tongue. This is usually temporary and resolves within the first week of use. If irritation persists or worsens, contact your pharmacy about alternative formulation options. Persistent irritation can actually reduce absorption because inflamed tissue does not absorb as efficiently as healthy tissue.
Taste changes. Semaglutide itself has a mildly bitter taste. The formulation vehicle may add additional flavoring, but many users report an unpleasant taste during the hold period. This is cosmetic rather than clinical but can affect compliance. Some users find rinsing with water after the 30-minute waiting period helps.
Dry mouth. Some users report increased dry mouth with sublingual dosing. This may be related to the formulation vehicle rather than the semaglutide itself. Staying hydrated throughout the day helps manage this symptom.
Systemic effects
Headache occurs in approximately 10% to 15% of users. It is more common during the first two weeks and during dose escalation. Over-the-counter pain relief is usually sufficient.
Fatigue affects some users, particularly during the first few weeks. The relationship between semaglutide and energy levels is complex. Reduced caloric intake from appetite suppression can contribute to fatigue, as can the metabolic adjustments your body makes in response to GLP-1 receptor activation. The broader topic of semaglutide onset and timeline helps set realistic expectations for when side effects peak and when they resolve.
Dizziness is less common but reported by some users, particularly when combined with rapid dietary changes. Ensuring adequate hydration and not skipping meals entirely can help.
When to stop and seek medical attention
Severe persistent vomiting that prevents adequate hydration.
Signs of pancreatitis including severe upper abdominal pain radiating to the back, especially after eating.
Signs of gallbladder problems including right upper quadrant pain, fever, or yellowing of skin or eyes.
Signs of allergic reaction including rash, swelling of the face or throat, or difficulty breathing.
Significant changes in vision.
These serious side effects are rare but require immediate medical attention regardless of whether you are using drops or injections.
The FDA-approved oral option: Rybelsus and oral Wegovy
While this guide focuses primarily on compounded sublingual drops, understanding the FDA-approved oral alternatives provides important context.
Rybelsus (oral semaglutide for type 2 diabetes)
Rybelsus is the brand name for FDA-approved oral semaglutide tablets containing SNAC permeation enhancer. Available in 3mg, 7mg, and 14mg tablets. Approved for improving blood sugar control in adults with type 2 diabetes. Not formally approved for weight loss, though weight reduction occurs as a secondary effect.
The dosing protocol starts at 3mg daily for 30 days, increases to 7mg daily for 30 days, and then reaches the maintenance dose of 14mg daily. The tablet must be taken on an empty stomach with no more than 4 ounces of plain water. You must wait at least 30 minutes before eating, drinking anything else, or taking other oral medications.
Weight loss with Rybelsus is modest compared to injectable semaglutide. Clinical data shows approximately 5 to 8 pounds of weight loss on Rybelsus 7mg and 14mg respectively, far less than the 30+ pounds average on injectable Wegovy. This gap exists primarily because of the dramatic bioavailability difference.
Oral Wegovy (new 25mg tablet)
In late December 2025, the FDA approved a higher-dose oral semaglutide tablet specifically for chronic weight management and cardiovascular risk reduction. This tablet is available in doses of 1.5mg, 4mg, 9mg, and 25mg. The escalation proceeds through these doses with the 25mg tablet as the target maintenance dose.
This is a significant development because it is the first oral GLP-1 medication with an FDA-approved indication for weight management. The 25mg dose was designed to overcome the bioavailability limitation of oral semaglutide by using a high enough dose that even the small percentage absorbed produces therapeutic blood levels. Cash pricing starts at $149 per month for lower doses and $299 per month for the 25mg dose.
For researchers and users deciding between compounded drops and FDA-approved tablets, the oral Wegovy option changes the calculation. It offers the convenience of oral dosing with the backing of clinical trial data, regulatory oversight, and standardized manufacturing. The trade-off is cost, though the gap between compounded and brand-name pricing has narrowed significantly.
Combining oral semaglutide drops with other protocols
Many users do not take semaglutide in isolation. It is often part of a broader protocol that may include other peptides, supplements, or lifestyle interventions. Understanding how drops interact with these combinations is important.
B12 supplementation
Many compounding pharmacies add methylcobalamin (vitamin B12) to their sublingual semaglutide formulations. This is done for two reasons. First, GLP-1 receptor agonists can reduce B12 absorption over time by altering gastric acid secretion. Second, sublingual B12 is well-absorbed and adding it to the drops is convenient. The semaglutide with B12 combination has become standard in many protocols for this reason.
If your drops do not contain B12, consider supplementing separately. B12 deficiency can develop gradually during long-term GLP-1 agonist use and contributes to fatigue, cognitive fog, and neuropathy.
Glycine addition
Some formulations include glycine alongside semaglutide. Glycine is an amino acid with multiple potential benefits during weight loss protocols including improved sleep quality, anti-inflammatory effects, and support for collagen synthesis during rapid body composition changes. The peptide stacking guide discusses combining multiple compounds in a single protocol.
Timing with other medications
The 30-minute fasting window after sublingual semaglutide dosing means you need to plan around other morning medications. Take semaglutide drops first, wait 30 minutes, then take other oral medications with breakfast. Do not take other sublingual medications at the same time as semaglutide drops, as they may compete for absorption sites and dilute the semaglutide.
Thyroid medications like levothyroxine also require empty stomach dosing. If you take both, establish a schedule with your provider. Some protocols dose semaglutide drops immediately upon waking, wait 30 minutes, then take thyroid medication with a sip of water, and wait another 30 to 60 minutes before breakfast. This requires planning but ensures both medications absorb properly.
Exercise timing
Intense exercise within an hour of sublingual dosing may increase nausea. The combination of GLP-1-mediated gastric slowing and exercise-induced blood flow redistribution away from the gut can produce significant discomfort. Most users find that dosing first thing in the morning and exercising at least 60 to 90 minutes later works well. The broader relationship between weight loss peptide stacks and exercise timing follows similar principles.
Converting between oral drops and injectable semaglutide
Sometimes users need to switch between delivery methods. You might start with drops and want to transition to injections for stronger effects. Or you might be on injections and want to switch to drops for convenience. Either way, you cannot simply use the same milligram dose.
Drops to injection conversion
There is no standardized conversion ratio because the bioavailability of compounded drops varies. As a rough guideline, many providers use a ratio where the daily sublingual dose divided by 7 (to account for daily vs weekly dosing) and then multiplied by a bioavailability correction factor gives the approximate weekly injection dose.
For example, a user on 3mg daily sublingual drops. Total weekly sublingual intake is 21mg. If sublingual bioavailability is approximately 10%, the effective weekly dose is approximately 2.1mg. The equivalent injectable dose would therefore be approximately 2mg to 2.4mg weekly, since injectable bioavailability approaches 100%. This is a rough estimate and should be validated with your prescribing provider.
The semaglutide conversion charts focus on injectable-to-injectable conversions but the principles of dose equivalence apply when switching between routes.
Injection to drops conversion
Going from injections to drops typically requires a higher milligram dose to maintain similar blood levels. A user on 1mg weekly injection would need approximately 1.5mg to 3mg daily sublingual, depending on formulation quality and absorption efficiency. The wide range reflects the uncertainty in sublingual bioavailability.
Most providers recommend overlapping the transition rather than switching abruptly. For example, start the drops at a moderate dose while reducing the injection frequency from weekly to every 10 days, then every 14 days, then discontinuing injections entirely as the drops reach therapeutic dose. This prevents the gap in GLP-1 coverage that can cause rebound appetite and rapid weight regain. Users who have experience switching between GLP-1 formats report that gradual transitions produce much smoother outcomes than abrupt switches.
Quality concerns with compounded oral semaglutide drops
Not all compounded semaglutide drops are created equal. The compounding pharmacy market has exploded with the demand for GLP-1 medications, and quality varies dramatically.
What to look for in a compounding pharmacy
503B outsourcing facility registration. Pharmacies registered as 503B outsourcing facilities are subject to FDA oversight, including inspections and adverse event reporting requirements. This provides a higher level of quality assurance than standard 503A pharmacies.
Certificate of analysis (COA) availability. Reputable pharmacies can provide a COA for each batch of semaglutide drops, confirming the identity, potency, and purity of the active ingredient. Ask for this document. If the pharmacy cannot provide it, that is a red flag.
Accreditation. Look for PCAB (Pharmacy Compounding Accreditation Board) accreditation or similar third-party quality certifications.
Beyond-use date testing. Quality pharmacies conduct stability testing to support their beyond-use dates rather than simply using default USP guidelines. Ask about stability data for the specific sublingual formulation.
Red flags
Prices that seem too good to be true. Quality semaglutide costs real money to produce. If someone is selling sublingual drops at dramatically lower prices than established pharmacies, the product quality is likely compromised.
No prescription required. Legitimate compounded semaglutide requires a valid prescription from a licensed healthcare provider. Any source selling it without a prescription is operating outside legal channels and product quality cannot be verified.
Vague labeling. The bottle should clearly state the semaglutide concentration per milliliter, the total volume, the beyond-use date, the pharmacy name and license number, and the prescribing provider name. Missing information suggests substandard practices. The same quality verification principles in peptide testing and verification apply to compounded oral formulations.
Lack of provider oversight. Telehealth services that prescribe semaglutide drops with minimal evaluation, no follow-up, and no monitoring are providing substandard care. GLP-1 medications require ongoing monitoring for side effects, metabolic changes, and dose adjustments.
The FDA perspective on compounded semaglutide
Understanding the regulatory landscape helps you make informed decisions about using compounded sublingual drops.
The FDA has consistently warned about the risks of compounded semaglutide products. Their concerns center on several issues. First, compounded medications are not FDA-approved, meaning they have not undergone the rigorous testing for safety, efficacy, and manufacturing quality that brand-name drugs have. Second, the FDA has identified instances of compounded semaglutide products that do not contain the correct amount of active ingredient, contain impurities, or are not sterile when they should be. Third, dosing errors are more likely with compounded products because concentrations and formulations vary between pharmacies.
These are legitimate concerns. They do not mean all compounded semaglutide drops are dangerous or ineffective, but they do mean that source quality matters enormously. Choosing a reputable pharmacy with proper accreditation and oversight mitigates many of these risks.
The legal status of compounded semaglutide has been in flux. Compounding is permitted under federal law when there is a shortage of the brand-name drug or when a patient has a medical need for a specific formulation that is not commercially available. The shortage status of semaglutide has changed over time, and different forms may have different availability designations. Check current FDA guidance for the latest regulatory status.
Maximizing results with oral semaglutide drops
The drops alone are a tool. Results come from how you use that tool within a comprehensive protocol.
Dietary considerations
Semaglutide reduces appetite. This is its primary mechanism for weight loss. But reduced appetite does not automatically mean optimal nutrition. In fact, the appetite suppression can be so strong that users under-eat, losing muscle mass along with fat, developing micronutrient deficiencies, and setting themselves up for metabolic adaptation that stalls weight loss.
Prioritize protein intake. Aim for 0.7 to 1 gram of protein per pound of body weight daily. When appetite is suppressed, protein-rich foods should be the first priority at every meal. Muscle preservation during weight loss depends on adequate protein and resistance training.
Track calories loosely even when not hungry. The goal is not starvation. It is a moderate calorie deficit of 300 to 500 calories below maintenance, sustained consistently. Eating too little slows metabolism, depletes energy, and ultimately stalls results. The weight loss and muscle preservation balance is critical during any GLP-1 protocol.
Stay hydrated. Dehydration is common during GLP-1 protocols because reduced food intake means less water from food sources. Aim for a minimum of 64 ounces of water daily, more if you exercise intensely.
Exercise programming
Resistance training is non-negotiable during semaglutide use. The weight you lose should be primarily fat, not muscle. Without resistance training stimulus, your body will cannibalize muscle tissue for energy alongside fat stores. This leaves you lighter but not leaner, a condition sometimes called being skinny fat.
Three to four resistance training sessions per week, focusing on compound movements like squats, deadlifts, presses, and rows, provides sufficient stimulus to preserve and even build muscle during calorie restriction. Cardiovascular exercise supports overall health and accelerates fat loss but should complement, not replace, resistance training.
Sleep optimization
Poor sleep undermines semaglutide effectiveness through multiple pathways. Sleep deprivation increases ghrelin (the hunger hormone), reduces leptin (the satiety hormone), increases insulin resistance, and promotes cortisol-driven visceral fat storage. Aim for seven to nine hours per night. If semaglutide-related nausea disrupts sleep, try dosing in the morning rather than evening and discuss timing adjustments with your provider.
Monitoring and tracking
Weigh yourself at the same time daily, preferably in the morning after using the bathroom. Use a seven-day moving average rather than individual data points. Daily weight fluctuates by two to five pounds due to water, food in the digestive system, and hormonal changes. The trend over weeks is what matters. Comparing your trajectory to expected semaglutide weight loss timelines helps identify whether your protocol needs adjustment.
Track waist circumference monthly. This catches body composition changes that the scale misses. You can lose fat and gain muscle simultaneously, especially early in a resistance training program, which may show minimal scale change despite significant physical transformation.
Get bloodwork before starting and every three months during use. Monitor fasting glucose, HbA1c, lipid panel, B12 levels, thyroid function, liver enzymes, and kidney function. These markers tell you whether the protocol is producing metabolic improvement beyond just weight change.
Common mistakes with oral semaglutide drops
After tracking thousands of protocols, SeekPeptides members report these as the most frequent errors with sublingual semaglutide drops.
Mistake 1: expecting injection-level results
Setting expectations based on injectable semaglutide trial data and then being disappointed with drop results is extremely common. Drops are a lower-bioavailability delivery method. They produce real results but typically not as dramatic as injections. Calibrate your expectations to the format you are using.
Mistake 2: inconsistent daily dosing
Weekly injections are forgiving. Miss your injection day by a day or two and the long half-life of semaglutide maintains reasonable blood levels. Daily drops are not forgiving. Miss three days in a row and your blood levels drop significantly. The appetite suppression wanes. Cravings return. Consistency is the price of convenience with oral dosing.
Mistake 3: inadequate hold time
Already covered extensively, but worth repeating. Most users who report that drops do not work are holding for 30 to 60 seconds instead of the minimum two minutes. This single variable is the difference between absorption and waste.
Mistake 4: taking drops with food or coffee
Morning coffee stimulates saliva production and gastric motility. Both reduce sublingual absorption. Taking drops after breakfast means food particles in the mouth interfere with mucosal contact. Always dose first thing, before coffee, before food, before anything except plain water to take other critical medications.
Mistake 5: skipping the escalation
Jumping straight to a high dose because you want faster results almost always backfires. Severe nausea leads to skipped doses. Skipped doses lead to inconsistent blood levels. Inconsistent levels lead to poor results and frustration. The slow escalation exists for a reason. Follow it. The same principles that guide semaglutide dosing escalation apply regardless of delivery format.
Mistake 6: not adjusting diet to match reduced appetite
Some users let appetite suppression guide their entire dietary approach. They eat 600 calories on some days and 2,000 on others, with no attention to macronutrient balance. This produces muscle loss, metabolic slowdown, and nutrient deficiencies. Use the appetite suppression as a tool, not as your entire strategy. Plan your meals even when you are not hungry.
Mistake 7: stopping abruptly
Discontinuing semaglutide drops cold turkey often leads to rapid appetite rebound and weight regain. If you decide to stop, taper the dose gradually over two to four weeks. This gives your appetite regulation system time to readjust. The semaglutide discontinuation symptoms can be managed much more effectively with a tapering approach.
Oral semaglutide drops vs oral tirzepatide
The GLP-1 landscape is not limited to semaglutide. Tirzepatide, a dual GIP/GLP-1 receptor agonist, is also available in oral and sublingual compounded formats. Understanding how these compare helps with decision-making.
Tirzepatide activates both the GIP receptor and the GLP-1 receptor, while semaglutide is a GLP-1 agonist only. The dual mechanism of tirzepatide tends to produce greater weight loss in clinical trials, approximately 20% to 25% body weight reduction compared to 15% to 17% for semaglutide at maximum doses. The oral tirzepatide guide covers the sublingual format of this alternative in detail.
For users specifically interested in drops, both semaglutide and tirzepatide sublingual formulations are available from compounding pharmacies. The same technique requirements apply to both. The same bioavailability limitations exist. And the same quality considerations are relevant. If you are deciding between the two, the choice typically comes down to whether the GIP component of tirzepatide is worth the potentially higher cost and different side effect profile.
Some users who do not respond adequately to semaglutide drops find better results with tirzepatide drops, and vice versa. Individual variation in receptor sensitivity and metabolic response means there is no universally superior option. Trial and assessment is often necessary. The next-generation GLP-1 comparisons show how this landscape continues to evolve.
The future of oral GLP-1 medications
The oral semaglutide drops space is evolving rapidly. Several developments are worth watching.
Improved formulation technologies
Compounding pharmacies are investing in better sublingual delivery vehicles. Newer formulations use advanced permeation enhancers that may significantly improve bioavailability beyond the current 3% to 15% range. If a formulation can consistently achieve 20% to 30% sublingual bioavailability, the gap between drops and injections narrows considerably.
Novo Nordisk oral pipeline
Novo Nordisk, the manufacturer of Ozempic and Wegovy, continues developing oral semaglutide formulations. The newly approved oral Wegovy at 25mg daily represents the current state of the art in FDA-approved oral GLP-1 therapy. Future iterations may achieve even higher bioavailability through improved SNAC technology or alternative permeation enhancers.
Competing oral GLP-1 products
Multiple pharmaceutical companies are developing oral GLP-1 agonists that may eventually compete with semaglutide. Some of these use entirely different peptide structures that may be better suited to oral delivery. The Onmorlo GLP-1 patch is another non-injection approach entering the market. The GLP-1 patches overview covers this alternative delivery method.
Regulatory evolution
The regulatory framework for compounded GLP-1 medications continues to shift. Changes in shortage designations, compounding regulations, and FDA enforcement priorities all affect the availability and legality of compounded sublingual drops. Staying informed about regulatory changes is important for anyone using or considering these products.
Building a complete oral semaglutide drops protocol
Putting it all together, here is a complete protocol framework for oral semaglutide drops.
Pre-start checklist
Obtain a valid prescription from a licensed healthcare provider who has evaluated your medical history, current medications, and suitability for GLP-1 therapy.
Source drops from a reputable compounding pharmacy with appropriate accreditation and the ability to provide certificates of analysis.
Get baseline bloodwork including fasting glucose, HbA1c, lipid panel, B12, thyroid function, liver enzymes, and kidney function.
Record baseline weight, waist circumference, and body composition measurements.
Set up a consistent morning routine that allows for the dosing and fasting protocol. The peptide dosing fundamentals apply to any sublingual protocol.
Daily protocol
Wake up. Roll the semaglutide bottle gently between palms for 10 to 15 seconds. Draw prescribed dose using oral syringe. Place under tongue. Start timer. Hold for minimum 2 minutes, preferably 3 to 5 minutes. Swallow remainder. Set second timer for 30 minutes. During the 30-minute window, do not eat, drink, or take other oral medications. After 30 minutes, proceed with breakfast and other medications.
Weekly check-ins
Weigh yourself daily and calculate weekly average. Note any side effects, their severity, and whether they are improving or worsening. Track food intake at least three days per week to ensure adequate protein and appropriate calorie levels. Report any concerns to your prescribing provider.
Monthly assessments
Measure waist circumference. Review weight trend. Assess energy levels, sleep quality, and overall wellbeing. Discuss dose adjustments with provider if weight loss has plateaued or side effects are unmanageable. Get follow-up bloodwork every three months.
Protocol adjustments
If nausea is severe, slow the escalation. Stay at the current dose for an additional one to two weeks before increasing. If appetite suppression is insufficient at the current dose, ensure technique is correct before requesting a dose increase.
Poor technique mimics inadequate dosing. If weight loss plateaus for more than three to four weeks at therapeutic dose, reassess dietary intake, exercise programming, sleep quality, and stress management before attributing the plateau to the medication alone.
Frequently asked questions
Do oral semaglutide drops work as well as injections?
Oral semaglutide drops produce meaningful results but typically not at the same magnitude as injections. The bioavailability difference, approximately 3% to 15% for sublingual drops versus near-100% for injections, means less active compound reaches your bloodstream. Proper technique, consistent daily dosing, and adequate hold time maximize what drops can deliver. Many users achieve 8% to 15% body weight loss with drops over three to four months when following the protocol precisely.
How long do I need to hold the drops under my tongue?
The minimum recommended hold time is two minutes. Longer is better. Three to five minutes provides more complete absorption through the sublingual mucosa. Set a timer rather than guessing. Most people significantly underestimate two minutes when they have liquid pooled under their tongue. This single variable has the largest impact on whether drops work effectively.
Can I eat or drink right after taking the drops?
No. Wait at least 30 minutes after dosing before eating, drinking, or taking other oral medications. Food and beverages in the mouth can wash away semaglutide that has not fully absorbed through the sublingual tissue. Coffee and acidic beverages are particularly problematic because they stimulate saliva production and change the pH environment in the mouth.
Do oral semaglutide drops need to be refrigerated?
Most compounded sublingual drops can be stored at controlled room temperature between 68 and 77 degrees Fahrenheit. However, formulations vary. Always follow the specific storage instructions provided by your compounding pharmacy. Keep the bottle in its original light-protected container and away from direct heat or sunlight. The peptide storage guide covers temperature requirements for various peptide formats.
What happens if I miss a dose of oral semaglutide drops?
Take your next scheduled dose the following day. Do not double up to make up for a missed dose. Missing one day is unlikely to significantly affect your results. Missing multiple consecutive days will reduce blood levels and may lead to temporary return of appetite. If you miss three or more consecutive days, you may experience mild nausea when resuming due to reduced tolerance. Consistency is much more important than perfection.
Can I switch from injectable semaglutide to oral drops?
Yes, but the conversion is not one-to-one. Sublingual doses need to be higher than injectable doses to account for lower bioavailability. Work with your prescribing provider to determine the appropriate sublingual starting dose based on your current injection dose. Most providers recommend a gradual transition rather than an abrupt switch to prevent gaps in GLP-1 coverage. The semaglutide dosage conversion reference can help with the math.
Are compounded oral semaglutide drops FDA-approved?
No. Compounded sublingual semaglutide drops are not FDA-approved. They are prepared by compounding pharmacies under a valid prescription. FDA-approved oral semaglutide products include Rybelsus (for type 2 diabetes) and oral Wegovy (for weight management). Compounded products have not undergone the same clinical trials and manufacturing oversight as FDA-approved products. This does not automatically mean they are unsafe, but it does mean quality depends heavily on the compounding pharmacy you choose.
How quickly will I see results with oral semaglutide drops?
Most users notice appetite changes within the first one to two weeks. Measurable weight loss typically begins by week three to four, with the most significant changes occurring after reaching therapeutic dose levels around weeks nine to twelve. Individual variation is substantial. Some users respond quickly while others need the full escalation period before seeing meaningful changes. Track trends over weeks, not days.
External resources
For researchers serious about optimizing their weight management protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides covering every delivery method, detailed protocol builders, and a community of thousands who have navigated these exact questions about oral versus injectable GLP-1 medications. SeekPeptides members get access to dosing calculators, protocol comparisons, and expert guidance that makes the difference between wasting product and achieving real results.
In case I do not see you, good afternoon, good evening, and good night. May your drops stay potent, your hold times stay long, and your results stay consistent.