Feb 19, 2026

You have been on tirzepatide for months. Maybe a year. The 10 mg dose worked beautifully at first, suppressing appetite like clockwork, melting pounds every week. Then 12.5 mg. Then 15 mg. And now? The scale has stopped moving. The appetite suppression that once felt effortless has faded to background noise. You are maxed out on a dual agonist and wondering what comes next.
Retatrutide comes next. But not the way most people think.
The transition from high dose tirzepatide to retatrutide is not a simple dose swap. You cannot take your 15 mg tirzepatide number, run it through a conversion formula, and land on the equivalent retatrutide dose. That formula does not exist. It cannot exist. These compounds operate through fundamentally different receptor profiles, and the retatrutide dosage schedule reflects a level of biological complexity that tirzepatide never touches.
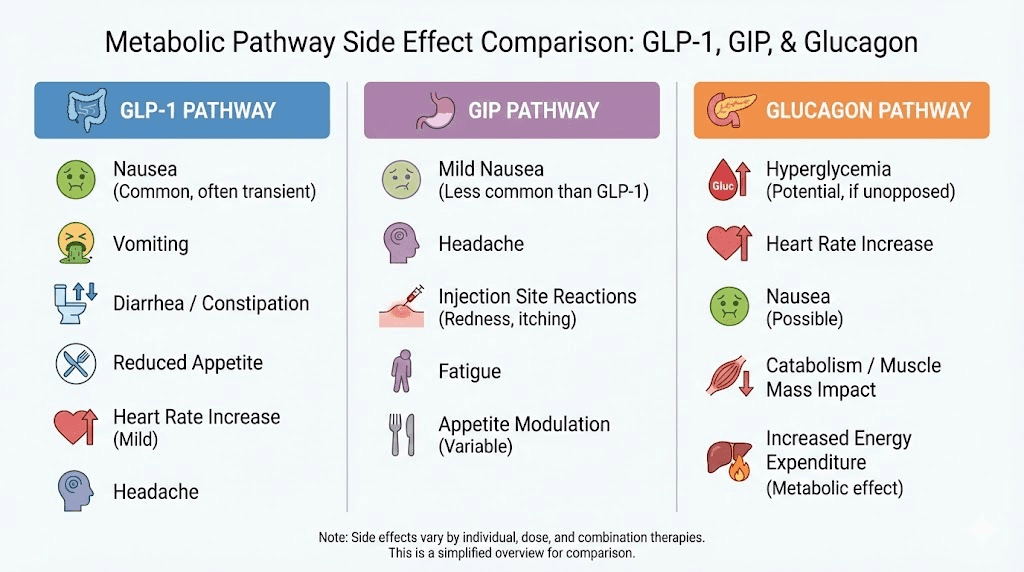
Tirzepatide activates two receptors: GLP-1 and GIP. Your body knows these pathways. It has adapted to them over months of weekly injections. Retatrutide activates three receptors: the same GLP-1, the same GIP, plus glucagon. That third receptor changes everything. Your GLP-1 and GIP tolerance from tirzepatide means nothing to the glucagon pathway. It has never been activated by your medication before. It is completely naive to this stimulation, and it will respond accordingly, with full force GI effects, thermogenic changes, and metabolic shifts you have not experienced even at the highest tirzepatide dose.
This guide covers the exact dosage schedule for making that transition safely. The washout timing. The starting dose rationale. The week by week escalation protocol. The side effects specific to people coming from high dose tirzepatide. And the clinical data driving every recommendation, because this is not guesswork. This is pharmacology. SeekPeptides breaks it down so you can make the switch with precision, not assumptions.
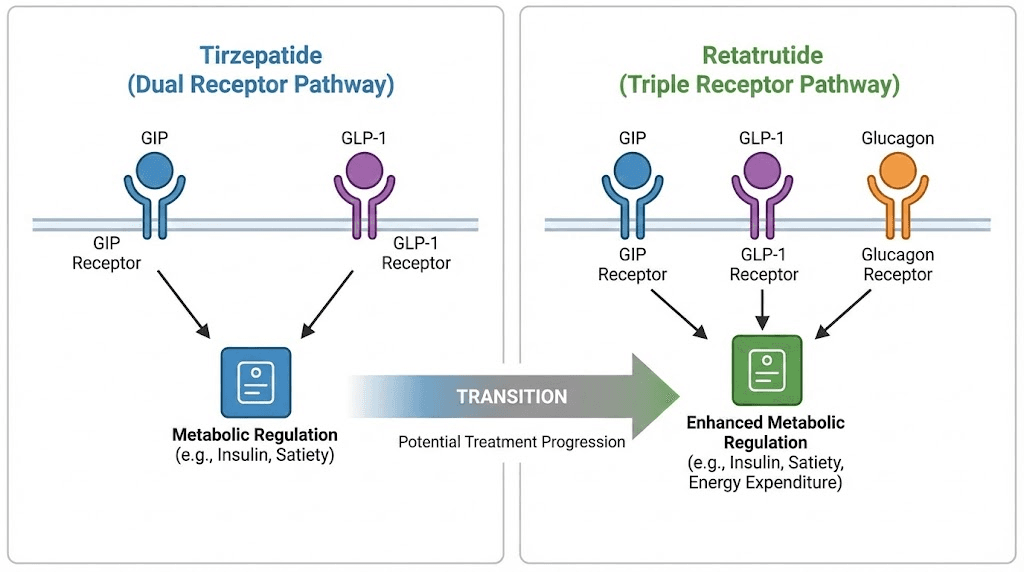
Why people switch from tirzepatide to retatrutide
Nobody switches medications that are working. The decision to move from tirzepatide to retatrutide almost always starts with one of three situations: plateau, insufficient results at maximum dose, or a specific metabolic goal that tirzepatide cannot achieve.
The tirzepatide plateau problem
Weight loss plateaus on tirzepatide are well documented. The SURMOUNT-1 trial data shows that most participants at 15 mg reach their maximum weight loss between weeks 60 and 72, with the curve flattening well before the trial endpoint. At that ceiling, the average body weight reduction sits around 22.5%. For many people, that is life changing. For others, it is not enough.
The plateau happens because tirzepatide reaches its pharmacological ceiling. Two receptor pathways can only do so much. GLP-1 suppresses appetite and slows gastric emptying. GIP enhances insulin response and modulates fat processing. Together, they create powerful metabolic effects. But they do not directly increase energy expenditure. They do not promote thermogenesis. And they do not directly mobilize stored fat through lipolysis the way glucagon receptor activation does.
That is the wall. And retatrutide breaks through it.
The glucagon advantage
Retatrutide adds glucagon receptor agonism to the same GLP-1 and GIP pathways tirzepatide uses. What does glucagon activation actually do?
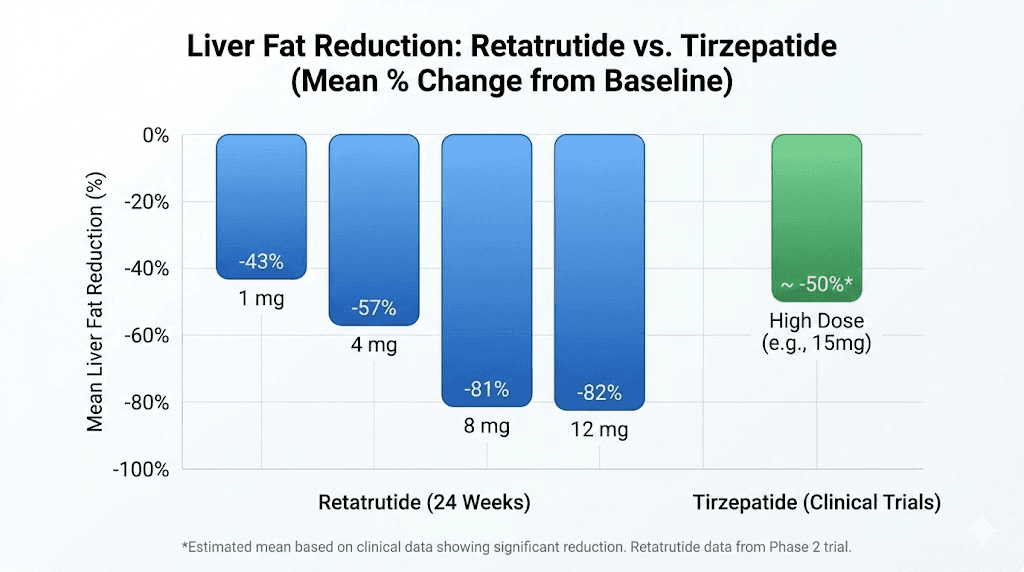
It increases energy expenditure by promoting thermogenesis in brown adipose tissue. Your body burns more calories at rest. It reduces lipogenesis, slowing the creation of new fat deposits. It induces lipolysis, actively breaking down existing fat stores into usable energy. And it has profound effects on liver fat, reducing hepatic steatosis by up to 86% in clinical trials. These are mechanisms tirzepatide does not touch at any dose, not at 5 mg and not at 15 mg. The receptor difference between retatrutide and other GLP-1 agonists is not incremental. It is categorical.
The Phase 3 TRIUMPH-4 trial confirmed what the Phase 2 data suggested: retatrutide at 12 mg delivered 28.7% average body weight reduction at 68 weeks. That is 6.2 percentage points beyond what tirzepatide 15 mg achieves at 72 weeks. And the retatrutide weight loss curve had not plateaued at 68 weeks. The trajectory was still declining. No other weight loss peptide has demonstrated this characteristic in clinical trials.
Metabolic health beyond the scale
Some people switch for reasons the scale does not capture. Fatty liver disease affects roughly 25% of the global population, and many people on tirzepatide have it. While tirzepatide provides moderate liver fat reduction, it cannot match what retatrutide does to hepatic fat stores.
In the Phase 2a MASLD trial, retatrutide at 12 mg reduced liver fat by 86%. At 48 weeks, 93% of participants at the highest dose had liver fat below 5%, effectively resolving their fatty liver disease. Tirzepatide has never demonstrated numbers even close to this. The glucagon receptor pathway directly affects hepatic lipid metabolism in ways that GLP-1 and GIP activation alone cannot replicate.
For anyone whose blood work shows elevated ALT, elevated AST, or imaging confirms hepatic steatosis, the retatrutide dose schedule offers a pathway to resolution that staying on tirzepatide simply will not provide.
The washout period: when to stop tirzepatide
Before starting retatrutide, you need to stop tirzepatide. The timing of that stop matters more than most people realize.
Tirzepatide half life and clearance
Tirzepatide has a half life of approximately 5 days. That means after your last injection, it takes roughly 5 days for the drug concentration in your blood to drop by half. After two half lives (10 days), roughly 25% remains. After three half lives (15 days), about 12.5% lingers. Full clearance takes approximately 25 to 30 days, or about five half lives.
But you do not need to wait for full clearance.
Recommended washout timing
The practical recommendation based on pharmacokinetic data and clinical reasoning is a washout period of 7 to 14 days after your last tirzepatide injection before starting retatrutide. Here is why that range works.
At 7 days (one week), roughly 40% of your last tirzepatide dose remains active. Your GLP-1 and GIP receptors are still partially stimulated. Starting retatrutide at this point means some overlap, which can actually benefit GI tolerance because your receptors are not going through a complete withdrawal and re-stimulation cycle. The transition feels smoother. The appetite rebound that some people experience when stopping GLP-1 agonists abruptly is minimized.
At 14 days (two weeks), approximately 15-20% of tirzepatide remains. Your GLP-1 and GIP receptors have partially reset but are not completely naive. This gives the glucagon receptor a cleaner introduction without the confounding variable of significant tirzepatide overlap.
Both approaches are valid. The choice depends on your tolerance for GI symptoms and your concern about appetite rebound during the gap.
What to expect during the washout
If you have been on high dose tirzepatide (10 mg or higher), the washout period may feel uncomfortable. Not dangerous. Uncomfortable.
Appetite returns. Sometimes aggressively. The GLP-1 mediated appetite suppression fades over 5-7 days, and many people report a rebound hunger that exceeds their pre-tirzepatide baseline. This is temporary. It is your brain recalibrating to the absence of exogenous GLP-1 signaling.
Food volume tolerance increases. Your gastric emptying speeds back up within days of your last dose. Meals that felt enormous on tirzepatide suddenly feel normal again.
Weight may tick up slightly. A pound or two of water and food volume, not fat gain. Do not panic. The retatrutide will address this once you begin.
The key is having your retatrutide reconstituted and ready before you enter the washout window. Preparation eliminates anxiety. You want to start retatrutide the moment your washout period ends, not scramble to mix your retatrutide while your appetite is screaming.
Starting retatrutide after high dose tirzepatide: the critical first dose
This is where most people make their biggest mistake. They assume their tirzepatide tolerance translates to retatrutide tolerance. It does not.
Why you start at 2 mg regardless of your tirzepatide dose
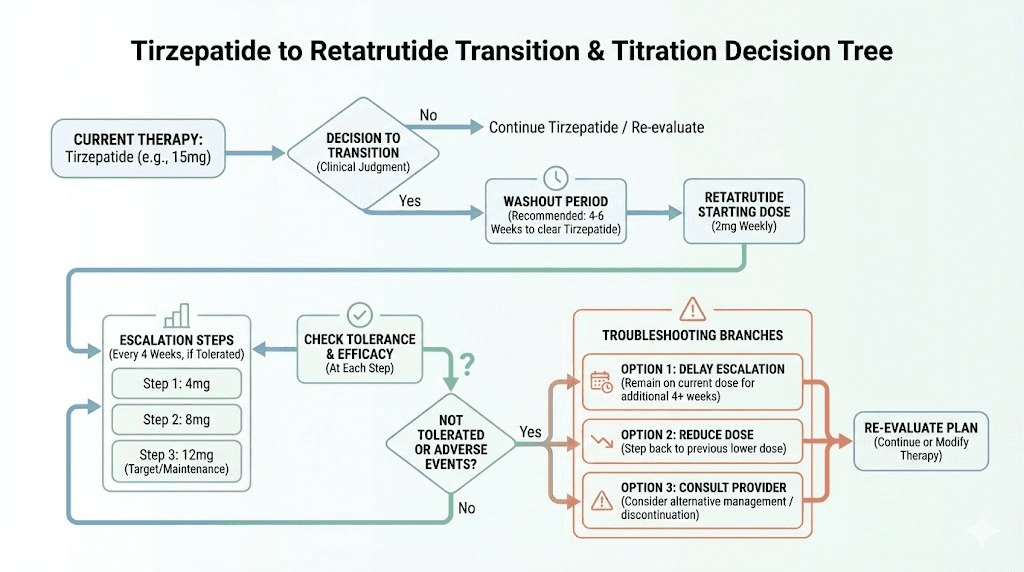
Whether you were on tirzepatide 10 mg, 12.5 mg, or 15 mg, you start retatrutide at 2 mg.
Not 4 mg. Not 8 mg. Two milligrams.
This seems counterintuitive. You have been injecting 15 mg of a powerful incretin agonist weekly. Starting a new compound at 2 mg feels like going backwards. But the pharmacology demands it, and here is why.
Your GLP-1 receptors have tolerance. After months of tirzepatide, these receptors have adapted to sustained agonism. They will handle the GLP-1 component of retatrutide with relative ease. You probably will not get the same intensity of appetite suppression from the GLP-1 portion that a naive user would experience.
Your GIP receptors have tolerance. Same story. Tirzepatide is actually built on a GIP backbone with GLP-1 activity added, so your GIP pathway is thoroughly conditioned.
Your glucagon receptors have zero tolerance. This is the critical point. Tirzepatide does not touch glucagon receptors. Not at any dose. Not at any duration. Your glucagon pathway is completely naive to pharmaceutical stimulation. Starting retatrutide at a high dose would hit these virgin receptors with full force glucagon agonism while simultaneously restimulating your GLP-1 and GIP pathways.
The Phase 2 trial data showed that participants who started at higher doses (4 mg) instead of 2 mg had nearly double the rate of early GI side effects. These were GLP-1 naive participants. For someone coming off high dose tirzepatide with conditioned GLP-1/GIP receptors but naive glucagon receptors, the risk calculus is different but the conclusion is the same: start low.
The 2 mg starting dose lets your glucagon receptors experience activation gradually. The thermogenic effects ramp up slowly. The lipolytic response builds over weeks rather than slamming on at full intensity. And the combined three receptor stimulation integrates into your existing receptor landscape without overwhelming any single pathway.
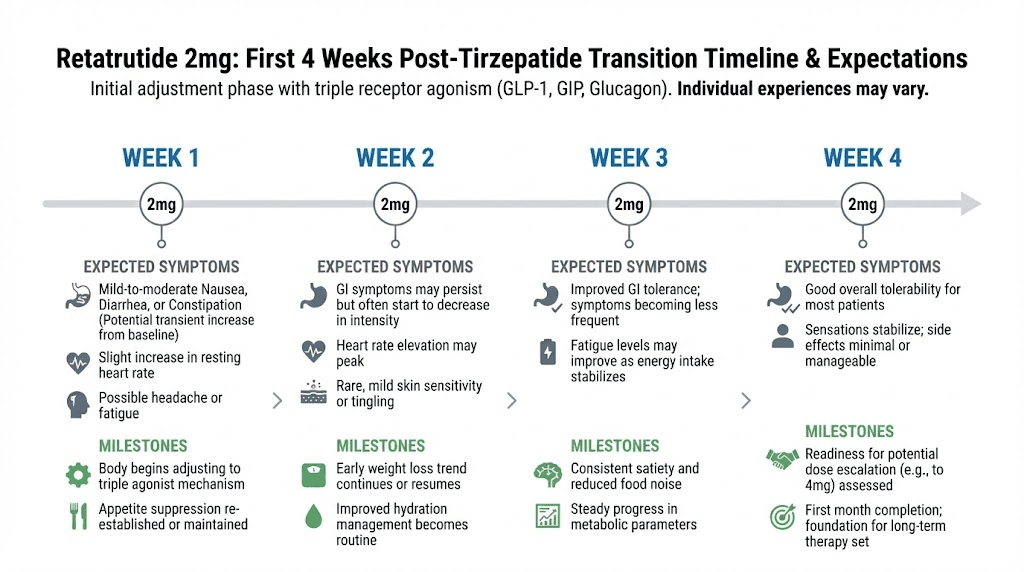
The first four weeks: what to expect at 2 mg
Your experience at 2 mg retatrutide will be different from a first time user. Here is what the tirzepatide background changes.
Appetite suppression may feel weaker than expected. Your GLP-1 receptors are partially desensitized from tirzepatide. The 2 mg retatrutide dose provides less GLP-1 stimulation than even 2.5 mg tirzepatide, and your receptors are calibrated to 10-15 mg. You may feel hungrier than you did on tirzepatide. This is normal and temporary. It improves with each escalation step.
New sensations from glucagon activation. You may notice increased body heat, especially after meals. This is thermogenesis from brown fat activation, a mechanism tirzepatide does not provide. Some people describe it as feeling warmer throughout the day without fever. Others notice increased sweating during exercise. These are signs the glucagon pathway is active and working.
GI effects may be milder than expected. Because your GLP-1 and GIP receptors have tolerance, the gastrointestinal slowdown effect may be less pronounced than a naive user would experience. Your gastric emptying will slow, but from a partially adapted baseline. Some people coming off high dose tirzepatide report smoother GI transitions to retatrutide than they experienced during their initial tirzepatide titration.
Early metabolic changes. Even at 2 mg, research data shows measurable metabolic improvements. In the Phase 2 trial, the 1 mg dose (lower than your starting point) still produced 7.2% weight loss at 24 weeks and notable liver fat reduction. At 2 mg, these effects are more pronounced. Your dosage is sub-therapeutic for maximum weight loss but not sub-therapeutic for beginning metabolic improvement.
Complete retatrutide dosage schedule after high dose tirzepatide
Here is the week by week protocol. This schedule is based on the Phase 2 and Phase 3 clinical trial titration data, adapted for the specific context of transitioning from high dose tirzepatide. It accounts for existing GLP-1/GIP receptor tolerance, naive glucagon receptors, and the pharmacokinetic reality of switching between these compounds.
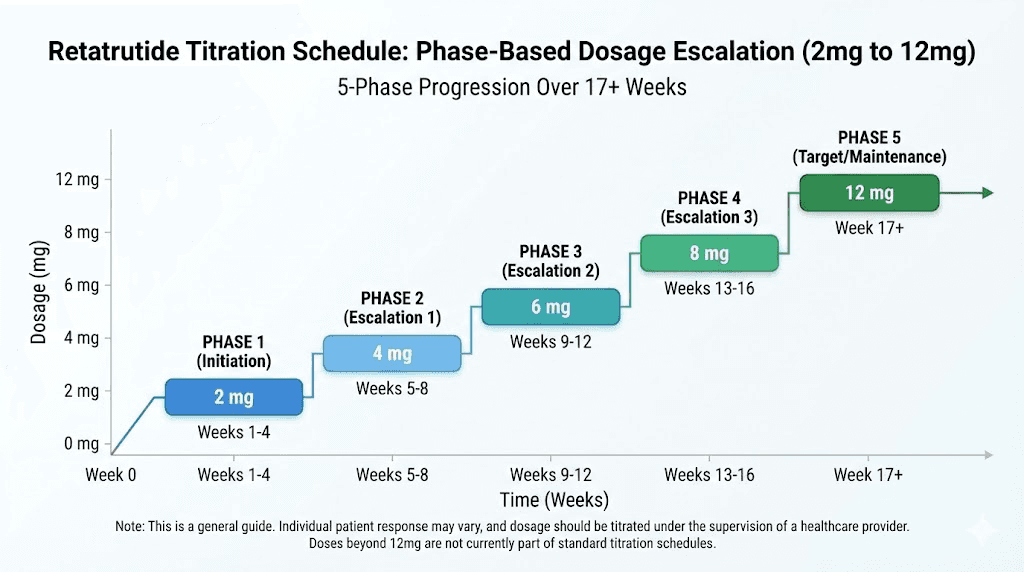
Phase 1: initiation (weeks 1 through 4)
Dose: 2 mg weekly
Goal: Introduce glucagon receptor activation while providing baseline GLP-1/GIP stimulation
Administration: Subcutaneous injection once weekly, same day each week
Injection sites: Rotate between abdomen, thigh, and upper arm. See our guide on where to inject retatrutide for optimal site selection.
Expected effects at 2 mg:
Mild appetite suppression (less than tirzepatide provided)
Subtle thermogenic warmth, especially postprandial
Possible mild nausea during week 1, usually resolving by week 2
Minimal weight loss (0.5 to 1 pound per week typical)
Beginning of liver fat mobilization
Reconstitution: Use our peptide reconstitution calculator to determine exact bacteriostatic water volumes for your vial concentration. Standard retatrutide vials come in 5 mg or 10 mg sizes. Proper reconstitution technique is essential for accurate dosing.
Do not escalate early. Even if you feel fine. Even if you feel undertreated. The glucagon receptor needs four full weeks to establish baseline tolerance before you increase stimulation. Patience here prevents problems at higher doses.
Phase 2: first escalation (weeks 5 through 8)
Dose: 4 mg weekly
Goal: Reach first therapeutic dose level while monitoring glucagon receptor response
The jump from 2 mg to 4 mg doubles your dose. Every receptor pathway doubles its stimulation. For GLP-1 and GIP, this may feel familiar if somewhat weaker than your tirzepatide doses. For glucagon, this is a significant escalation from a still developing baseline.
Expected effects at 4 mg:
Noticeable appetite suppression returns (approaching what low dose tirzepatide provided)
Increased thermogenesis, measurably warmer body temperature
GI effects peak during week 5 and 6, then moderate
Weight loss accelerates to 1 to 2 pounds per week
Phase 2 trial data: 12.9% weight loss at 24 weeks at this dose level
Liver fat reduction becomes measurable
In the Phase 2 trial, the 4 mg dose delivered 17.1% body weight reduction at 48 weeks. For someone with existing GLP-1/GIP tolerance from tirzepatide, the actual weight loss trajectory may differ. Your conditioned receptors may respond less dramatically to the GLP-1/GIP components at this dose, but the glucagon mediated fat mobilization is new territory producing novel metabolic effects.
This is also the dose where you start to see a meaningful difference from your tirzepatide experience. The weight loss mechanism now includes active thermogenesis and lipolysis that tirzepatide never provided. Even if the appetite suppression feels weaker than your old 15 mg tirzepatide dose, the total metabolic effect is building toward something more powerful.
Phase 3: primary therapeutic dose (weeks 9 through 12)
Dose: 8 mg weekly
Goal: Reach the dose that produced 22.8% weight loss in Phase 2 trials
This is the big jump. From 4 mg to 8 mg is a doubling, and it is where many people transitioning from tirzepatide start to feel the full three receptor effect for the first time.
Expected effects at 8 mg:
Strong appetite suppression, likely meeting or exceeding what 15 mg tirzepatide provided
Significant thermogenic effect, possibly uncomfortable warmth in some situations
GI effects during escalation week (week 9), typically resolving by week 11
Weight loss of 2 to 3 pounds per week in many individuals
Phase 2 data: 22.8% body weight reduction at 48 weeks
Liver fat reduction approaching 81% at this dose level
Measurable improvements in blood pressure, triglycerides, and inflammatory markers
At 8 mg, the retatrutide starts outperforming what your maximum tirzepatide dose could achieve. The three receptor synergy is fully online. GLP-1 suppresses appetite and slows digestion. GIP enhances insulin sensitivity and fat processing. Glucagon burns stored fat through thermogenesis and lipolysis while reducing liver fat at rates no dual agonist can match.
For many people transitioning from tirzepatide, 8 mg is the sweet spot. The weight loss exceeds what 15 mg tirzepatide delivered (22.8% versus 22.5%), the metabolic benefits are substantially broader, and the side effect profile, while higher than tirzepatide, is manageable with proper GI side effect management strategies.
Some researchers choose to stay at 8 mg indefinitely. The data supports this decision.
Phase 4: enhanced therapeutic dose (weeks 13 through 16)
Dose: 9 mg weekly
Goal: Incremental escalation toward maximum efficacy
The Phase 3 TRIUMPH-4 trial tested 9 mg as one of the therapeutic doses, alongside 12 mg. This smaller step from 8 mg to 9 mg (only a 12.5% increase) serves as a transitional dose that many clinicians expect to be part of the commercial titration schedule.
Expected effects at 9 mg:
Modestly increased appetite suppression beyond 8 mg
Continued thermogenic effect
TRIUMPH-4 data: 26.4% body weight reduction (average 29.1 kg lost) at 68 weeks
Substantial improvements in knee osteoarthritis pain scores (up to 75.8% reduction in WOMAC pain)
Continued liver fat reduction
The jump from 8 mg to 9 mg is deliberately small. After the large 4 to 8 mg escalation, this more modest increase lets you assess whether the additional stimulation is tolerable and beneficial before committing to the maximum dose. If 9 mg produces adequate weight loss and acceptable side effects, there is no pharmacological requirement to go higher.
Phase 5: maximum dose (weeks 17 and beyond)
Dose: 12 mg weekly
Goal: Maximum weight loss and metabolic improvement
The 12 mg dose is the highest dose tested in retatrutide clinical trials. It delivered the most impressive results in the program, but it also carries the highest side effect burden.
Expected effects at 12 mg:
Maximum appetite suppression across all three receptor pathways
Strong thermogenic effect
TRIUMPH-4 data: 28.7% body weight reduction (average 71.2 pounds lost) at 68 weeks
Weight loss curve still declining at 68 weeks, no plateau reached
Liver fat reduction up to 86%, with 93% of participants achieving normal liver fat levels
Blood pressure reduction of 14.0 mmHg systolic
Significant improvements in non-HDL cholesterol, CRP, and triglycerides
At 12 mg, retatrutide outperforms every other weight loss compound tested in clinical history. The 28.7% average body weight reduction at 68 weeks exceeds tirzepatide 15 mg at 72 weeks (22.5%) by over 6 percentage points. And the curve has not flattened. The trajectory suggests continued weight loss beyond 68 weeks, a characteristic no other peptide-based therapy has demonstrated.
But 12 mg is not for everyone. The side effect data demands consideration.
Complete dosage schedule summary table
Phase | Weeks | Retatrutide dose | Key milestone | Expected weekly weight loss |
|---|---|---|---|---|
Washout | Pre-start | 0 mg (off tirzepatide) | 7-14 days after last tirzepatide dose | Possible 1-2 lb gain (water/food volume) |
Phase 1 | 1-4 | 2 mg | Glucagon receptor introduction | 0.5-1 lb |
Phase 2 | 5-8 | 4 mg | First therapeutic dose | 1-2 lbs |
Phase 3 | 9-12 | 8 mg | Primary therapeutic dose | 2-3 lbs |
Phase 4 | 13-16 | 9 mg | Enhanced therapeutic | 2-3 lbs |
Phase 5 | 17+ | 12 mg | Maximum dose | 2-4 lbs |
Total transition time from last tirzepatide dose to maximum retatrutide dose: approximately 18 to 19 weeks, depending on washout duration.
Important: These weekly weight loss estimates are approximations based on clinical trial data and adjusted for the context of transitioning from tirzepatide. Individual results vary based on starting weight, metabolic health, diet, exercise, and response to glucagon receptor activation. Use our peptide calculator to determine your exact injection volumes at each dose level.
Side effects specific to the tirzepatide to retatrutide transition
People transitioning from high dose tirzepatide experience a different side effect profile than people starting retatrutide from scratch. Understanding these differences helps you prepare and manage them effectively.
GI side effects: potentially milder than expected
This surprises many people. You might expect worse GI effects moving to a more powerful compound. But the clinical reality is nuanced.
Your GLP-1 receptors have adapted to high dose stimulation. The GLP-1 mediated gastric slowing, the primary driver of nausea and vomiting with incretin therapies, may be partially blunted by your existing tolerance. Many people transitioning from tirzepatide report less nausea during retatrutide initiation than they experienced during their original tirzepatide titration years earlier.
The GI effects that do occur tend to be driven more by the glucagon component. Glucagon activation affects gut motility differently than GLP-1. Some people experience increased bowel frequency rather than the constipation that characterizes tirzepatide. Others experience a mixed pattern where the GLP-1 component slows the upper GI tract while glucagon activation affects the lower GI tract differently.
Here is what the clinical data shows for GI side effects at retatrutide doses relevant to the transition:
Side effect | Retatrutide 4 mg | Retatrutide 8 mg | Retatrutide 12 mg | Your likely experience (coming from tirzepatide) |
|---|---|---|---|---|
Nausea | 24% | 35% | 43% | Likely lower due to GLP-1 tolerance |
Diarrhea | 16% | 25% | 33% | May be higher, glucagon-driven effect is new |
Vomiting | 10% | 15% | 21% | Likely lower due to GLP-1 tolerance |
Constipation | 12% | 18% | 25% | Possibly less than naive users |
The percentages above come from Phase 2 trial data where participants were GLP-1 naive. Your experience will differ because two of the three receptor pathways are already familiar to your body.
Dysesthesia: the new side effect
The Phase 3 TRIUMPH-4 trial identified a side effect that tirzepatide users have never encountered: dysesthesia. Approximately 20.9% of patients on the 12 mg dose reported this condition, characterized by skin sensitivity, tingling, or tenderness to touch.
This is not a GI effect. It is a neurological sensation, likely related to the glucagon receptor component or the metabolic changes that three receptor agonism produces. It is not typical of any GLP-1 agonist therapy, and it appears to be relatively unique to retatrutide.
For someone transitioning from tirzepatide, dysesthesia is completely unfamiliar territory. It is not dangerous according to trial data (it was not a major reason for discontinuation), but it can be disconcerting. Monitor for unusual skin sensations, tingling in extremities, or increased sensitivity to touch, particularly during dose escalations to 8 mg and above.
Heart rate changes
Retatrutide increased heart rate by up to 6.7 beats per minute in clinical trials. Tirzepatide also increases heart rate, but typically to a lesser degree. When transitioning from tirzepatide, your cardiovascular system is already adapted to some degree of heart rate elevation from GLP-1 agonism. The additional increment from glucagon activation may push the total heart rate increase slightly higher than either compound alone.
This is worth monitoring, especially during the first few weeks after escalating to 8 mg and above. A resting heart rate increase of 5 to 10 beats per minute is within expected parameters. Increases beyond 15 bpm warrant discussion with your healthcare provider.
Thermogenic effects: a feature, not a side effect
The increased body warmth from glucagon mediated thermogenesis is technically a physiological effect, not a side effect. But it feels like a side effect when you are lying in bed at night feeling uncomfortably warm despite the thermostat being set normally.
This is the glucagon receptor at work. Brown fat activation generates heat as a byproduct of fatty acid oxidation. You are literally burning stored fat for warmth. The effect is most noticeable at doses of 4 mg and above, and it tends to peak during the first two weeks after each dose escalation before stabilizing.
Management strategies include sleeping in a cooler room, wearing lighter clothing, and staying well hydrated. The thermogenesis represents active calorie burning, so while it may be uncomfortable, it is contributing directly to weight loss and metabolic improvement.
Managing the transition: practical protocols
Theory and pharmacology are important. But you also need to know what to do each week, what to eat, how to handle problems, and when to adjust.
Nutrition during the transition
Your dietary approach during the tirzepatide to retatrutide transition matters more than most people think.
During the washout period, appetite rebounds. This is when discipline becomes critical. Do not overeat just because you suddenly can. Maintain the portion sizes you adapted to on tirzepatide. Eat protein rich meals (aim for 0.7 to 1 gram of protein per pound of lean body mass daily) to protect muscle during the transition gap. Use our dietary guidance for GLP-1 users as a framework even during the washout.
During early retatrutide dosing (2 to 4 mg), appetite suppression may be weaker than you are accustomed to. Lean into structured meal planning. Prioritize protein. Eat vegetables. Minimize ultra-processed foods that trigger overeating. The appetite control will strengthen with each dose escalation, but these early weeks require conscious dietary management.
At therapeutic doses (8 mg and above), the appetite suppression becomes profound, often exceeding what tirzepatide provided. This is where the food selection shifts from managing hunger to ensuring adequate nutrition. Many people at high dose retatrutide struggle to eat enough. Focus on calorie dense, nutrient rich foods. Protein shakes become valuable tools for meeting daily protein targets when solid food appetite is minimal. Supplementation helps cover micronutrient gaps.
Exercise during the transition
Your exercise capacity may fluctuate during the transition. Here is what to expect at each phase.
During washout: Energy levels may drop initially as tirzepatide clears. Some people feel sluggish during days 3 through 7 after their last dose. Maintain your routine but reduce intensity by 10 to 20% if needed. This passes quickly.
At 2 to 4 mg retatrutide: You may notice increased warmth during exercise as glucagon driven thermogenesis adds to exercise generated heat. Hydrate more aggressively than you did on tirzepatide. The calorie burn during exercise may increase slightly due to the thermogenic contribution, but the effect is modest at these doses.
At 8 to 12 mg retatrutide: Exercise tolerance can be affected by reduced caloric intake (from strong appetite suppression) and increased resting energy expenditure (from glucagon activation). If you are burning more at rest and eating less, intense workouts may require adjustment. Listen to your body. Strength training remains critical for muscle preservation during rapid weight loss. Consider reducing cardio volume if fatigue becomes an issue and prioritize resistance training instead.
Blood work monitoring schedule
Anyone transitioning between these compounds should track their metabolic markers. Here is a practical monitoring schedule.
Baseline (before starting retatrutide): Complete metabolic panel, HbA1c, liver enzymes (ALT, AST), lipid panel, fasting insulin, C-reactive protein. This gives you a clean comparison point separate from your tirzepatide values.
Week 8 (at 4 mg): Repeat liver enzymes and lipid panel. Retatrutide glucagon mediated liver fat reduction should already show ALT/AST improvements. Lipid changes from the transition may be visible.
Week 16 (at 9 mg): Full panel repeat. By this point, you have been on therapeutic retatrutide doses for 8 weeks. The metabolic picture should show clear differentiation from your tirzepatide baseline, especially in liver fat markers and inflammatory markers.
Week 24 and every 12 weeks after: Ongoing monitoring. Weight, body composition, liver enzymes, lipids, HbA1c if diabetic or pre-diabetic. Track trends, not individual snapshots.
Dose adjustments and troubleshooting
Not everyone follows the standard escalation schedule exactly. Here is how to handle common scenarios that arise during the transition.
Scenario 1: severe GI effects at a dose escalation
If you escalate (for example, from 4 mg to 8 mg) and experience severe nausea, vomiting, or diarrhea that persists beyond 10 to 14 days, do not push through. Drop back to the previous dose for another 2 to 4 weeks, then try the escalation again.
Some clinicians recommend a half step approach: instead of jumping from 4 mg to 8 mg, try 6 mg for 4 weeks first. While 6 mg was not a standard dose in Phase 2 trials, it represents a reasonable intermediate step that reduces the escalation shock to all three receptor pathways. Use your reconstitution calculator to determine the exact injection volume for a 6 mg dose from your specific vial concentration.
Scenario 2: weight loss stalls at a specific dose
If your weight loss stalls at 8 mg for more than 4 weeks despite consistent dosing and reasonable diet, escalating to 9 mg or 12 mg is appropriate. A 4 week stall at sub-maximum doses suggests your body has adapted to that stimulation level and needs additional receptor activation to continue progress.
Before escalating, verify the basics first. Are you actually eating at a deficit? Appetite suppression can make you feel like you are eating less when portion sizes have gradually crept up. Are you tracking your intake? Strong appetite suppression sometimes creates complacency about food choices, leading to caloric maintenance despite feeling like you are restricting.
Scenario 3: the retatrutide is not working as well as tirzepatide did
This happens occasionally, particularly at lower doses. Remember that your GLP-1 and GIP receptors have downregulated from sustained tirzepatide exposure. Retatrutide at 2 to 4 mg provides less GLP-1/GIP stimulation per milligram than tirzepatide did. The appetite suppression may genuinely be weaker at these early doses.
The solution is patience. By 8 mg, the three receptor synergy typically surpasses anything tirzepatide provided. The glucagon component produces metabolic effects that no amount of GLP-1/GIP stimulation alone can replicate. If you are at 12 mg and still not seeing results that exceed your tirzepatide experience, the issue is likely dietary, not pharmacological.
Scenario 4: you want to go back to tirzepatide
It happens. Some people prefer the side effect profile of tirzepatide, or they miss the convenience of pre-filled injection pens, or they find the glucagon mediated effects uncomfortable. Switching between incretin therapies is always an option.
To switch back: stop retatrutide, wait 7 to 14 days, start tirzepatide at 2.5 mg per standard protocol. Your GLP-1 and GIP receptors may actually have partially resensitized during the period of lower GLP-1/GIP stimulation at early retatrutide doses, potentially making tirzepatide feel more effective on the return. Follow the standard tirzepatide dosing schedule rather than jumping back to your previous high dose.
Reconstitution and preparation for the transition
Getting your retatrutide supply ready before the washout period begins eliminates stress and ensures a smooth transition.
What you need
Before your last tirzepatide injection, have the following on hand.
Retatrutide vials: Standard sizes are 5 mg and 10 mg. For the full titration from 2 mg to 12 mg, calculate your total needs. At 2 mg weekly for 4 weeks = 8 mg total. At 4 mg weekly for 4 weeks = 16 mg total. At 8 mg weekly for 4 weeks = 32 mg total. At 9 mg weekly for 4 weeks = 36 mg total. At 12 mg weekly ongoing = 12 mg per week. Total for the first 16 weeks: approximately 92 mg. Source from a verified vendor. Our retatrutide buying guide covers vendor verification and quality considerations.
Bacteriostatic water: Multiple vials. You will need bacteriostatic water for every reconstitution. Have enough for your entire supply.
Insulin syringes: 1 mL (100 unit) syringes with 29 to 31 gauge needles for injection. For reconstitution, you can use larger syringes (3 mL) to transfer bacteriostatic water into the peptide vial.
Alcohol swabs: For cleaning vial tops and injection sites.
Sharps container: For safe needle disposal.
Reconstitution protocol
Reconstitute only what you will use within 28 to 30 days. Do not reconstitute your entire supply at once. Reconstituted peptides have a limited refrigerator shelf life.
Standard approach: Add bacteriostatic water to the vial slowly, angling the stream against the glass wall. Do not squirt directly onto the lyophilized powder. Let the water dissolve the powder naturally. Gently swirl if needed. Never shake. The solution should be clear and colorless. Any cloudiness, particles, or discoloration means the reconstitution failed or the peptide is degraded.
For exact water volumes and resulting concentrations at each dose level, our reconstitution calculator handles the math. Input your vial size (5 mg or 10 mg), desired dose (2, 4, 8, 9, or 12 mg), and preferred injection volume, and it calculates the exact bacteriostatic water amount needed.
Storage: Reconstituted retatrutide must be refrigerated at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit). Do not freeze. Do not leave at room temperature for extended periods. If traveling, use an insulated cooling case. See our guide on traveling with injectable peptides for practical tips that apply to retatrutide as well.
Clinical data driving this protocol
Every recommendation in this guide traces back to published clinical trial data. Here is the evidence base.
Phase 2 trial (NEJM, 2023)
The foundational retatrutide study, published in the New England Journal of Medicine, randomized 338 participants to various doses over 48 weeks. Key findings relevant to this transition protocol include the dose dependent weight loss curve (1 mg: 8.7%, 4 mg: 17.1%, 8 mg: 22.8%, 12 mg: 24.2%), the superiority of the 2 mg starting dose over 4 mg for GI tolerability, and the absence of a weight loss plateau at 48 weeks in the higher dose groups.
The trial also established that GI adverse events occurred primarily during dose escalation, not maintenance, supporting the 4 week stabilization periods between dose increases in this protocol.
Phase 3 TRIUMPH-4 (December 2025)
The first Phase 3 readout confirmed and extended the Phase 2 findings. In participants with obesity and knee osteoarthritis, retatrutide at 12 mg delivered 28.7% average body weight reduction at 68 weeks (approximately 71.2 pounds from a starting weight around 248 pounds). The 9 mg dose delivered 26.4%. Both doses also produced substantial knee pain relief, with WOMAC pain scores dropping up to 75.8%.
Critically, the dysesthesia signal (20.9% at 12 mg) was identified for the first time in this Phase 3 data, adding a new consideration to the side effect management protocol.
Phase 2a MASLD trial (Nature Medicine, 2024)
This trial specifically evaluated retatrutide for liver fat reduction. At 48 weeks, 93% of participants at 12 mg had liver fat below 5%. This data is particularly relevant for tirzepatide transitioners with fatty liver disease, as it quantifies the additional benefit the glucagon pathway provides over and above what tirzepatide can achieve.
Tirzepatide SURMOUNT-1 (NEJM, 2022)
The reference standard for tirzepatide efficacy. This trial established the 22.5% weight loss ceiling at 15 mg over 72 weeks and documented the plateau around weeks 60 to 72. Understanding this ceiling is what motivates most tirzepatide to retatrutide transitions.
Long term considerations after the transition
Once you have completed the titration and stabilized on your maintenance retatrutide dose, several long term factors require attention.
Maintenance dosing strategy
There is no established maintenance dose reduction protocol for retatrutide yet. The Phase 2 trial ran 48 weeks at fixed doses. The Phase 3 TRIUMPH-4 ran 68 weeks. Neither tested dose reduction. This means the current recommendation is to maintain your therapeutic dose indefinitely.
Some researchers are experimenting with dose reduction after reaching their target weight, similar to the approach some clinicians use with tirzepatide (escalating to 10 to 15 mg for active weight loss, then reducing to 5 to 7.5 mg for maintenance). This may work with retatrutide (for example, losing weight at 12 mg then maintaining at 8 mg), but no trial data supports or refutes this approach yet.
What we do know: stopping GLP-1 agonists entirely leads to significant weight regain, typically 50 to 70% of lost weight within 12 months. There is no reason to believe retatrutide will differ. Plan for ongoing maintenance therapy.
Tolerance development
Anecdotal reports suggest some people experience diminishing appetite suppression over time at fixed doses of any incretin therapy. The clinical trial data does not show significant tolerance development at fixed doses over 48 to 68 weeks, but post-marketing experience with tirzepatide suggests some degree of tolerance may occur with longer use.
Retatrutide, with its additional glucagon pathway, may theoretically resist tolerance better than dual agonists. The thermogenic and lipolytic effects of glucagon activation operate through different cellular mechanisms than the satiety signals of GLP-1, and receptor desensitization patterns may differ across these pathways. This is speculative but pharmacologically plausible.
If you notice diminishing effects at a given dose, verify your diet first (caloric creep is far more common than true pharmacological tolerance), then discuss dose adjustment with your healthcare provider.
The possibility of future compounds
The transition from tirzepatide to retatrutide represents a move from dual to triple agonism. The weight loss field continues to evolve. Survodutide, orforglipron, and mazdutide represent other approaches to multi-receptor weight loss therapy. Cagrisema (semaglutide plus cagrilintide) targets different receptor combinations. Each new compound will require its own transition protocol.
For now, the tirzepatide to retatrutide pathway represents the most data-supported transition between a dual and triple agonist. The protocol in this guide will likely remain relevant even as the field evolves, because the pharmacological principles, start low at new pathways while respecting existing tolerance, apply universally.
Frequently asked questions
Can I skip the 2 mg starting dose if I was on 15 mg tirzepatide?
No. Your tirzepatide dose is irrelevant to your starting retatrutide dose. The glucagon receptor pathway that retatrutide activates has never been stimulated by tirzepatide at any dose. Your glucagon receptors are completely naive. Starting at 2 mg protects these receptors from overstimulation while allowing your body to integrate the novel three-receptor activation pattern gradually. The clinical trial data supports starting at 2 mg for all users, regardless of prior GLP-1 exposure.
How long should I wait between stopping tirzepatide and starting retatrutide?
Seven to fourteen days is the recommended washout period. Tirzepatide has a half life of approximately 5 days, so at 7 days roughly 40% of your last dose remains active. At 14 days approximately 15-20% remains. Both timeframes are acceptable. A shorter washout minimizes appetite rebound. A longer washout gives your glucagon receptors a cleaner introduction to retatrutide stimulation.
Will I regain weight during the washout and early dosing?
You may gain 1 to 3 pounds during the washout period, primarily from increased food volume and water retention as gastric emptying normalizes. This is not fat gain. It reverses quickly once retatrutide reaches therapeutic doses. By week 8 (at 4 mg), most people have surpassed their pre-transition weight and are in active weight loss territory again.
What if retatrutide gives me worse side effects than tirzepatide ever did?
The most common issue is during the 4 mg to 8 mg escalation, where all three receptor pathways ramp up significantly. If side effects are intolerable, drop back to the previous dose for 2 to 4 additional weeks. Consider an intermediate dose step (6 mg between 4 mg and 8 mg). The GI management strategies that worked during your tirzepatide titration, smaller meals, adequate hydration, ginger supplements for nausea, will also help during retatrutide escalation.
Is the dysesthesia permanent?
Based on Phase 3 trial data, dysesthesia appears to be dose dependent and not necessarily permanent. Some participants reported resolution during continued treatment. Others experienced it throughout the trial period. If the sensation becomes problematic, a dose reduction from 12 mg to 8 or 9 mg may alleviate it while maintaining substantial weight loss efficacy.
Can I combine retatrutide with BPC-157 for gut healing?
Some researchers combine retatrutide with BPC-157 to manage GI side effects, as BPC-157 has demonstrated gastroprotective properties in animal studies. This is an off-label combination with no clinical trial data supporting the pairing. If you choose this approach, discuss it with your healthcare provider. Our peptide stacking guide covers general principles of combining peptides, and peptides commonly combined with GLP-1 agonists provides additional context.
When will retatrutide be FDA approved?
Based on the Phase 3 TRIUMPH trial timeline, FDA approval is anticipated in late this year or early next year. Seven TRIUMPH trials are expected to complete during the current year. Until approval, retatrutide is available only through research channels. See our guide on retatrutide availability and purchasing options for current access information.
Should I tell my doctor I am making this switch?
Yes. Always. Transitioning between incretin therapies requires medical oversight, particularly for monitoring cardiac effects, liver function, and metabolic markers. Your healthcare provider can order appropriate blood work, monitor for complications, and adjust the protocol based on your individual response. This is especially important because retatrutide is investigational and any use outside clinical trials is off-label.
Who should not make this transition
The tirzepatide to retatrutide transition is not appropriate for everyone. Several situations warrant caution or contraindicate the switch entirely.
If your tirzepatide is still working: There is no reason to switch from a medication that is producing results. If you are still losing weight at your current tirzepatide dose and have not plateaued, stay the course. Switching introduces unnecessary risk, GI disruption, and a dose-titration period where your weight loss temporarily slows.
If you have cardiac concerns: Retatrutide increases heart rate more than tirzepatide (up to 6.7 bpm versus approximately 2 to 4 bpm). For anyone with arrhythmias, tachycardia, or cardiovascular disease, the additional cardiac stimulation from glucagon activation warrants careful evaluation before switching.
If you are pregnant or planning pregnancy: Neither compound is safe during pregnancy. Discontinue all incretin therapies before attempting conception. See our guide on pregnancy and peptide therapy for detailed guidance.
If you have pancreatitis history: Both tirzepatide and retatrutide carry theoretical risks for pancreatitis. Adding glucagon receptor activation to an already at-risk pancreas is not well studied. Extreme caution is warranted.
If you have medullary thyroid carcinoma or MEN2 syndrome: GLP-1 agonists carry a boxed warning for these conditions. Retatrutide, as a GLP-1 agonist (among other things), falls under the same precaution.
For everyone else, particularly those who have plateaued on tirzepatide, have reached maximum dose without sufficient results, or have metabolic goals (especially liver health) that tirzepatide cannot address, this transition represents the most powerful pharmacological upgrade currently available in the weight loss space.
For researchers committed to optimizing their peptide protocols, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact transition decisions. SeekPeptides members access detailed dosage calculators, cycle planning tools, and expert-reviewed protocols that account for the individual nuances a general guide cannot capture.
External resources
Triple-Hormone-Receptor Agonist Retatrutide for Obesity, Phase 2 Trial (NEJM)
Tirzepatide Once Weekly for the Treatment of Obesity, SURMOUNT-1 (NEJM)
Retatrutide for Metabolic Dysfunction-Associated Steatotic Liver Disease (Nature Medicine)
TRIUMPH Clinical Trials Program, Retatrutide (ClinicalTrials.gov)
In case I do not see you, good afternoon, good evening, and good night. May your transitions stay smooth, your glucagon receptors stay responsive, and your weight loss curves stay on the decline.