Feb 20, 2026

Before you adjust another dose or skip another cycle without thinking twice, stop. Something is happening to your period. And if you are taking tirzepatide, there is a very good chance the two are connected.
You are not imagining it. That late period, the heavier flow, the spotting between cycles, the cramps that feel different from anything you have experienced before. Thousands of women on tirzepatide are reporting the same thing. Yet most guides barely mention it. The clinical trials did not even track it systematically. And the prescribing information? It does not list menstrual irregularities as a specific adverse effect.
That does not mean it is not happening.
A 2024 analysis found that roughly 27 percent of women noticed changes in their menstrual cycle after starting tirzepatide treatment. Women with pre-existing hormone sensitivity, polycystic ovary syndrome, or a history of irregular cycles reported the highest rates of disruption. Some saw their periods disappear entirely. Others experienced heavier bleeding than they had dealt with in years. And a surprising number of women with PCOS actually saw their cycles improve for the first time without birth control intervention.
The relationship between tirzepatide and your menstrual cycle is more complex than a simple yes-or-no answer. It involves fat tissue, estrogen production, insulin signaling, the hypothalamic-pituitary-ovarian axis, and the cascade of metabolic changes that accompany significant weight loss on tirzepatide. This guide covers every angle, from the hormonal mechanisms driving these changes to the FDA birth control warning that most women never hear about, to practical management strategies that can help you navigate this period of adjustment without unnecessary worry.
Whether your cycle has gone haywire or you have not started tirzepatide yet and want to know what to expect, the next several thousand words contain the answers that clinical trial data left out.
The short answer: yes, tirzepatide can affect your period
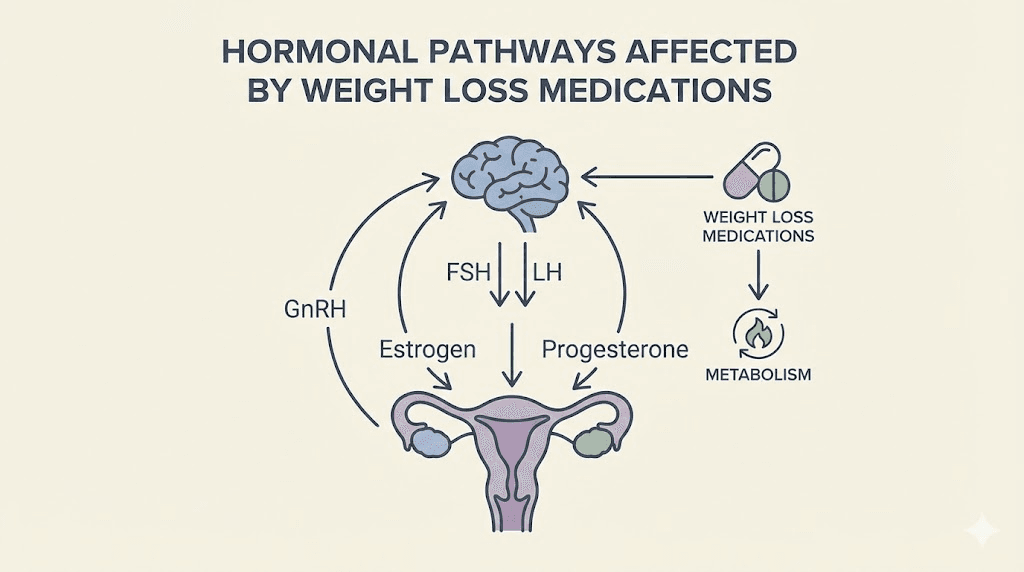
Tirzepatide does not directly target reproductive hormones. It was not designed to interact with estrogen, progesterone, follicle-stimulating hormone, or luteinizing hormone. Its mechanism of action centers on two specific receptors: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). These receptors regulate appetite, blood sugar, insulin secretion, and gastric emptying. Not menstruation.
But the body does not work in isolated compartments.
When tirzepatide triggers significant weight loss, reduces insulin resistance, alters gut hormone signaling, and shifts metabolic function, the downstream effects ripple through every hormonal system in the body. Including the one that controls your menstrual cycle. The changes are indirect but they are real, documented, and for many women, impossible to ignore.
Understanding how quickly tirzepatide works helps explain why menstrual changes often appear within the first few months of treatment. The SURMOUNT-1 trial demonstrated average weight loss of approximately 20.9 percent at the 15 mg dose. That level of body composition change, sometimes happening over just 36 to 72 weeks, creates metabolic shifts that the reproductive system cannot simply ignore.
The short answer is yes. Tirzepatide can and often does affect your period. The longer answer requires understanding exactly how and why, which is what the rest of this guide covers in detail.
How tirzepatide changes your hormonal environment
To understand why your period changes on tirzepatide, you need to understand what tirzepatide actually does to your body beyond suppressing appetite and reducing blood sugar. The mechanisms behind peptide action extend far beyond their primary targets.
Fat tissue is not just storage, it is an endocrine organ
Most people think of body fat as passive storage. It is not. Adipose tissue actively produces and metabolizes hormones, including estrogen. Fat cells contain an enzyme called aromatase that converts androgens into estrogen. The more fat tissue you carry, the more estrogen your body produces through this pathway.
When tirzepatide causes rapid fat loss, two things happen simultaneously. First, you lose the tissue that was producing a significant portion of your circulating estrogen. Second, the estrogen that was stored within fat cells gets released into circulation before eventually declining. This creates a hormonal rollercoaster that your reproductive system was not prepared for.
Women who lose 15 to 25 percent of their body weight on tirzepatide dosing protocols may experience substantial shifts in estrogen levels. The magnitude depends on starting body composition, the speed of weight loss, and individual hormonal sensitivity.
The hypothalamic-pituitary-ovarian axis responds to energy availability
Your menstrual cycle is controlled by a communication chain called the hypothalamic-pituitary-ovarian (HPO) axis. The hypothalamus releases gonadotropin-releasing hormone (GnRH) in pulses. These pulses signal the pituitary gland to produce follicle-stimulating hormone (FSH) and luteinizing hormone (LH). FSH and LH then direct the ovaries to mature eggs and produce progesterone and estrogen in the cyclical pattern that creates your period.
This entire chain is exquisitely sensitive to energy availability.
When your body detects a significant caloric deficit, which tirzepatide creates through profound appetite suppression, the hypothalamus can slow or alter GnRH pulsatility. The logic is evolutionary. If the body does not have enough energy to sustain itself, it certainly does not have enough to sustain a pregnancy. So the reproductive system gets dialed down as a protective mechanism.
Research on functional hypothalamic amenorrhea shows that low energy availability can disturb the pulsatile release of GnRH, suppressing the HPO axis to various degrees. As suppression increases, normal ovulation and estrogen production cannot be fully maintained due to decreases in FSH and LH levels.
Leptin drops with fat loss, and your hypothalamus notices
Leptin is a hormone produced by fat cells that tells your brain how much energy you have stored. It falls in proportion to fat mass. When leptin levels drop below approximately 3 ng/mL, the hypothalamus interprets this as a signal that the body cannot support pregnancy, and it can suppress GnRH pulses accordingly.
Women losing significant weight on tirzepatide will experience declining leptin levels. The speed at which leptin drops matters. Gradual weight loss gives the HPO axis time to adapt. Rapid weight loss, which tirzepatide is capable of producing, can create a sharper leptin decline that the reproductive system struggles to adjust to smoothly.
This is one reason why some women experience fatigue on GLP-1 medications alongside menstrual changes. Both symptoms trace back to the same underlying metabolic shift: the body recalibrating to a new energy balance.
Insulin sensitivity improvements affect ovarian function directly
Tirzepatide dramatically improves insulin sensitivity. This is one of its primary therapeutic benefits and a major reason it works so well for weight loss and type 2 diabetes management. But insulin does more than regulate blood sugar.
Insulin signals directly influence ovarian function. High insulin levels stimulate the ovaries to produce excess androgens (male hormones like testosterone). This is one of the central mechanisms driving polycystic ovary syndrome. When tirzepatide normalizes insulin levels, it changes the hormonal environment within the ovaries themselves.
For women without PCOS, this shift may cause temporary menstrual irregularity as the ovaries adjust to a new signaling environment. For women with PCOS, this same shift can actually restore normal ovulatory function, sometimes for the first time in years.

The most common menstrual changes women report on tirzepatide
Not every woman on tirzepatide experiences period changes. But for the roughly 27 percent who do, the disruptions tend to fall into predictable patterns. Understanding what is normal and what requires medical attention can save you significant anxiety during the adjustment period.
Irregular cycle timing
The most frequently reported change is shifts in cycle length. Periods that arrived predictably every 28 days may start coming every 24 days, or every 35 days, or at seemingly random intervals. Some women report cycles extending beyond 40 days during the first few months of treatment.
This irregularity typically reflects the HPO axis recalibrating to new estrogen levels, new leptin signals, and new insulin sensitivity. It is the same type of irregularity that women experience during any period of significant metabolic change, whether from weight loss surgery, extreme dieting, or hormonal transitions like perimenopause.
Women who track their cycles carefully while monitoring tirzepatide dosing schedules often find that the irregularity follows a pattern. Cycles tend to be most disrupted during dose escalation phases and begin to stabilize once a maintenance dose is reached and weight loss slows.
Changes in flow volume
Some women report significantly heavier periods on tirzepatide. Others notice markedly lighter flow. Both directions are possible, and some women experience both at different points during treatment.
Heavier periods can occur when estrogen levels fluctuate in a way that causes the uterine lining to build up more than usual before shedding. This is more common in the early months of treatment when hormonal shifts are most dramatic. Lighter periods or scanty flow often indicate lower overall estrogen levels, which means the uterine lining is not building as thickly between cycles.
Neither change is inherently dangerous. But women experiencing very heavy bleeding, defined as soaking through a pad or tampon every hour for several consecutive hours, should contact their healthcare provider. That level of bleeding, regardless of the cause, warrants medical evaluation.
Missed or skipped periods
Missing a period entirely is alarming, especially for women who have been regular their entire lives. On tirzepatide, missed periods can happen for several reasons. The caloric deficit may be significant enough to temporarily suppress ovulation. Estrogen levels may have dropped below the threshold needed to trigger a normal cycle. Or the HPO axis may simply be recalibrating.
One critical consideration: tirzepatide may increase fertility in some women, particularly those whose excess weight was previously suppressing ovulation. A missed period on tirzepatide should always prompt consideration of pregnancy, especially given the important interactions between GLP-1 medications and reproductive health.
If you miss three or more consecutive periods that were previously regular, medical evaluation is recommended to rule out pregnancy and assess hormonal status.
Breakthrough bleeding and spotting
Mid-cycle spotting, light bleeding between periods, and unexpected breakthrough bleeding are common during the first three to six months of tirzepatide treatment. These symptoms typically reflect hormonal instability rather than anything structurally wrong.
The endometrium (uterine lining) responds to fluctuating estrogen and progesterone levels. When those levels shift unpredictably, as they do during rapid metabolic change, the lining may partially shed at unusual times. This is similar to the spotting many women experience when starting or changing hormonal birth control.
Persistent intermenstrual bleeding beyond the first six months of treatment, or bleeding that is heavy rather than light spotting, warrants a conversation with your doctor.
Altered premenstrual symptoms
Changes to PMS symptoms are less discussed but widely reported. Women describe their premenstrual experience shifting in unexpected ways. Some report worse cramps, more intense mood changes, or new symptoms like breast tenderness that they never experienced before. Others find that their PMS symptoms actually improve, becoming milder and more manageable.
The direction of change often correlates with the type of hormonal shift occurring. Women whose estrogen levels are fluctuating dramatically tend to experience more intense PMS. Women whose hormonal environment is stabilizing, particularly those with improved insulin sensitivity, often report reduced PMS severity. Some women also notice changes in body aches and discomfort patterns that may overlap with premenstrual symptoms.
Why weight loss disrupts your cycle, with or without medication
Tirzepatide is not unique in its ability to affect menstruation. Any form of significant weight loss can alter your cycle. Understanding this broader context helps separate the effects of the medication itself from the effects of the weight loss it produces.
The energy deficit equation
Your reproductive system requires approximately 30 calories per kilogram of lean body mass per day to function optimally. When energy availability drops below this threshold consistently, the HPO axis begins to downregulate. This is not specific to tirzepatide. It happens with caloric restriction, excessive exercise, bariatric surgery, or any situation where energy output significantly exceeds energy input.
What makes tirzepatide different is the degree and speed of caloric restriction it can create. The profound appetite suppression that makes it so effective for weight loss can also create a caloric deficit severe enough to affect reproductive function. This is why dietary planning on tirzepatide matters so much, not just for weight loss outcomes, but for hormonal health.
Women who experience the most dramatic menstrual changes on tirzepatide often report eating very little, sometimes below 1,000 calories per day, because their appetite is so suppressed. Ensuring adequate caloric intake, even when you do not feel hungry, can help mitigate some of the hormonal disruption.
Cortisol elevation and the stress response
Chronic caloric deficit elevates cortisol, the primary stress hormone. Research has linked cortisol levels above 18 micrograms per deciliter to a 2.4-fold higher odds of functional hypothalamic amenorrhea. Cortisol directly interferes with GnRH pulsatility, adding another pathway through which energy deficit disrupts menstruation.
Tirzepatide users who combine the medication with aggressive exercise programs or highly restrictive diets may compound this cortisol effect. The body reads the combined signal as extreme stress and responds by prioritizing survival functions over reproductive functions.
Managing stress levels and avoiding overtraining while on tirzepatide is not just advice for general wellbeing. It is a practical strategy for minimizing menstrual disruption. Understanding the full spectrum of GLP-1 side effects helps you anticipate and manage these interconnected symptoms.
The body fat threshold for menstruation
Research suggests that most women need a minimum of approximately 17 to 22 percent body fat to maintain regular menstrual cycles. Below this threshold, estrogen production from adipose tissue drops significantly, and the hypothalamus receives inadequate leptin signaling to maintain normal reproductive function.
For women who start tirzepatide at higher body weights, this threshold is unlikely to become relevant. But women who begin treatment closer to normal weight ranges, or who continue aggressive weight loss protocols beyond their initial goals, may approach body fat percentages that compromise menstrual function.
Monitoring body composition rather than just scale weight becomes important for women concerned about menstrual health. A woman at 22 percent body fat losing another 5 percent is in a very different hormonal situation than a woman at 40 percent body fat losing 5 percent, even if the scale shows similar numbers.
Tirzepatide and PCOS: when disruption becomes improvement
For women with polycystic ovary syndrome, the story of tirzepatide and menstruation takes a dramatically different turn. While many women experience temporary cycle disruption, women with PCOS often experience the opposite: their cycles actually become more regular, sometimes for the first time in their adult lives.
The insulin-androgen connection
PCOS is fundamentally a disorder of insulin resistance and androgen excess. High insulin levels drive the ovaries to produce excessive testosterone and other androgens. These elevated androgens prevent normal follicle maturation and ovulation, leading to the irregular or absent periods that define the condition.
Tirzepatide attacks this problem at its root. By dramatically improving insulin sensitivity through dual GIP and GLP-1 receptor activation, it reduces the insulin signal that drives ovarian androgen production. As androgen levels fall, the ovarian environment normalizes, follicles can mature properly, and ovulation can resume.
One study found that irregular menstrual cycles in women with PCOS decreased from 85.7 percent to 32.1 percent following treatment with GLP-1 receptor agonists. Ovarian cyst prevalence dropped from 89.3 percent to 41 percent. These are remarkable improvements that no lifestyle intervention alone typically achieves so quickly.
The weight loss amplifier effect
Weight loss independently improves PCOS symptoms. Even a 5 to 10 percent reduction in body weight can restore ovulatory cycles in some women with the condition. Tirzepatide produces weight loss far beyond this threshold for most users, with the SURMOUNT trials showing average reductions of 20 to 26 percent of body weight in women.
The combination of improved insulin sensitivity and significant weight loss creates a powerful dual mechanism for PCOS improvement. The metabolic changes tirzepatide produces go beyond simple calorie restriction, fundamentally altering the hormonal environment that perpetuates PCOS.
Women with PCOS who have never ovulated regularly may find themselves ovulating for the first time on tirzepatide. This is overwhelmingly positive from a reproductive health standpoint, but it carries an important implication: if you were previously anovulatory and using that as de facto birth control, you may now be fertile. Pregnancy prevention becomes critical, which connects directly to the FDA birth control warning discussed later in this guide.
Clinical trials and PCOS-specific research
Eli Lilly is currently conducting clinical trials specifically testing tirzepatide for reproductive function and metabolic health in women with PCOS who are overweight or obese. The primary endpoint focuses on whether tirzepatide can normalize menstrual cycles, defined as cycle length between 21 and 35 days or more than 8 cycles per year.
Among women with PCOS, prescribing of semaglutide or tirzepatide increased from 2.4 percent in 2021 to 17.6 percent in 2025, a more than 7-fold increase. This reflects growing clinical adoption as physicians recognize the potential of these medications beyond weight management. Comparing semaglutide and tirzepatide for PCOS management is an active area of research, with tirzepatide showing potential advantages due to its dual receptor mechanism.
The timeline: when do period changes happen and when do they resolve?
Understanding the typical timeline of menstrual changes on tirzepatide helps reduce anxiety and set realistic expectations. While individual experiences vary, patterns emerge from the collective data.
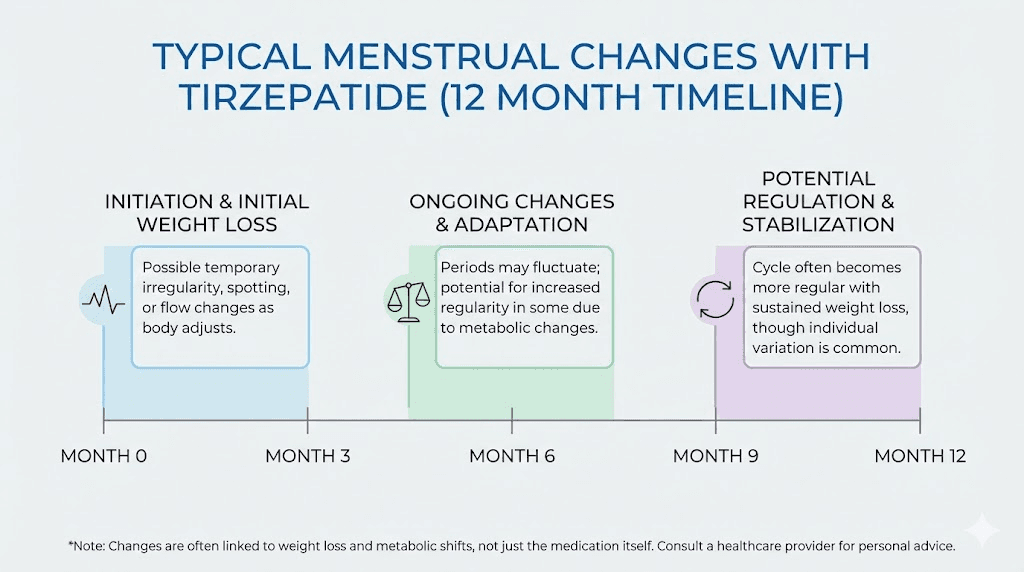
Months 1 through 3: the adjustment phase
The first three months of tirzepatide treatment bring the most dramatic metabolic shifts. You are typically escalating through starting doses during this period, and your body is adjusting to reduced caloric intake, changing blood sugar levels, and initial weight loss.
Menstrual changes during this phase are most common and most unpredictable. Cycles may be shorter than usual, longer than usual, or simply irregular. Flow may change in either direction. Spotting between periods is particularly common during dose escalation. Referring to a tirzepatide dosage guide can help you understand which dose escalation phase you are in and anticipate when changes may occur.
This is the phase where most women first notice something is different. It is also the phase where the changes feel most alarming, because the pattern has not yet been established.
Months 3 through 6: stabilization begins
By months three through six, most women report that their cycles begin to find a new pattern. It may not be identical to your pre-tirzepatide cycle, but it becomes more predictable. The hormonal system is adapting to your new body composition, new insulin sensitivity, and new energy balance.
Women who have reached their target dose and are experiencing steady rather than rapid weight loss tend to stabilize faster. The overall timeline of tirzepatide effectiveness often parallels the timeline of menstrual normalization. As the medication reaches steady state in your system and weight loss velocity decreases, hormonal fluctuations become less dramatic.
Months 6 through 12 and beyond: the new normal
By six to twelve months, the vast majority of women report that their menstrual cycles have stabilized. For most, cycles return to a pattern similar to their pre-treatment norm. For women with PCOS, cycles may actually be more regular than they have been in years.
Research on related interventions suggests that over 80 percent of women regain ovulatory cycles within nine months of achieving stable energy balance, even after extended periods of amenorrhea. This timeline aligns with what tirzepatide users consistently report: the changes are temporary, and the body adapts.
Women who continue to experience significant menstrual irregularity beyond 12 months should seek medical evaluation. At that point, other causes should be explored, including thyroid dysfunction, premature ovarian insufficiency, or other endocrine conditions that may have been masked by previous obesity-related hormonal changes.
Tirzepatide and birth control: the FDA warning you need to know
This section contains information that could prevent an unplanned pregnancy. Read it carefully.
The delayed gastric emptying problem
Tirzepatide slows gastric emptying, the rate at which food and medications move from your stomach into your small intestine for absorption. This is one of the mechanisms through which it promotes satiety and reduces food intake. But it also affects how oral medications are absorbed, including oral hormonal contraceptives.
The FDA prescribing information for both Mounjaro (tirzepatide for diabetes) and Zepbound (tirzepatide for weight management) includes a specific warning about this interaction. After the first dose of tirzepatide, gastric emptying is most substantially delayed. The effect then undergoes tachyphylaxis (diminishes) after subsequent doses, but it recurs with each dose escalation.
The specific numbers
Clinical data shows that when a combined oral contraceptive containing 0.035 mg ethinyl estradiol and 0.25 mg norgestimate was taken alongside a single 5 mg dose of tirzepatide, the absorption of the contraceptive components was significantly reduced. Maximum blood concentration of ethinyl estradiol decreased by 59 percent. Maximum concentration of norgestimate decreased by 66 percent. Maximum concentration of norelgestromin decreased by 55 percent.
These are not subtle reductions. A 59 percent decrease in peak ethinyl estradiol concentration means your oral birth control may not be achieving the blood levels needed for reliable contraceptive effect. This is especially concerning given that tirzepatide can increase fertility in women who were previously anovulatory due to excess weight.
What the FDA recommends
The FDA recommendation is straightforward. Patients using oral hormonal contraceptives should switch to a non-oral contraceptive method, or add a barrier method of contraception, for four weeks after initiation with tirzepatide and for four weeks after each dose escalation.
Non-oral contraceptive methods that are not affected by delayed gastric emptying include IUDs (both hormonal and copper), contraceptive implants, injectable contraceptives, patches, and vaginal rings. These deliver hormones through routes that bypass the GI tract entirely.
Many women taking tirzepatide are not aware of this interaction. If you are relying solely on oral birth control pills while taking tirzepatide, your contraceptive coverage may be compromised. This is not a theoretical risk. The pharmacokinetic data shows substantial reductions in drug absorption that could translate to real-world contraceptive failure.

What the SURMOUNT clinical trials revealed about women
The SURMOUNT program represents the most comprehensive clinical data on tirzepatide for weight management. Understanding what these trials did and did not measure helps contextualize the menstrual effects discussion.
Trial demographics and weight loss results
Approximately 67 percent of SURMOUNT-1 trial participants were women. The trial demonstrated remarkable weight loss across all dose groups. At the 15 mg dose, participants lost an average of 20.9 percent of body weight over 72 weeks, compared to roughly 3 percent in the placebo group.
A post-hoc analysis published in Obesity (the journal of The Obesity Society) specifically examined weight loss by reproductive stage. Among women in the pre-menopausal, peri-menopausal, and post-menopausal subgroups, reductions from baseline in body weight ranging from 24 to 26 percent were significantly greater with tirzepatide compared to placebo. This means the medication works similarly well regardless of reproductive stage.
Women interested in the magnitude of results that tirzepatide produces can explore tirzepatide before and after experiences from real users. The weight loss trajectory directly influences the timeline and severity of menstrual changes.
What the trials did not measure
Here is the critical gap. Menstrual irregularities were not systematically reported as adverse events in published results from the major SURMOUNT trials. The primary endpoints focused on weight loss and glycemic outcomes, with reproductive health not designated as a primary or secondary endpoint.
This does not mean menstrual changes did not occur. It means they were not formally tracked, quantified, or analyzed. The 27 percent figure cited earlier comes from post-market analysis and patient-reported outcomes rather than controlled trial data. Given that two-thirds of trial participants were women who lost an average of 20 to 26 percent of their body weight, it is virtually certain that many experienced menstrual changes that simply were not captured in the published data.
This represents a meaningful gap in our understanding of tirzepatide side effects and highlights why patient-reported experiences are so valuable for filling in what clinical trials missed.
The menopause and hormone therapy connection
Recent research has explored the intersection of tirzepatide and menopausal hormone therapy. A study published in The Lancet found that postmenopausal women using tirzepatide plus menopause hormone therapy achieved a 17 percent reduction in body weight, compared to 14 percent in those using tirzepatide alone. Even more striking, total body weight loss exceeding 20 percent was achieved by 45 percent of patients taking hormone therapy, compared to just 18 percent among non-users.
Preclinical data suggests a synergistic interaction between estrogen and GLP-1 signaling, where estrogen appears to amplify the appetite-suppressing effects of GLP-1. For women navigating menopause and weight loss, this research opens interesting possibilities for optimizing treatment outcomes.
Managing period changes while taking tirzepatide
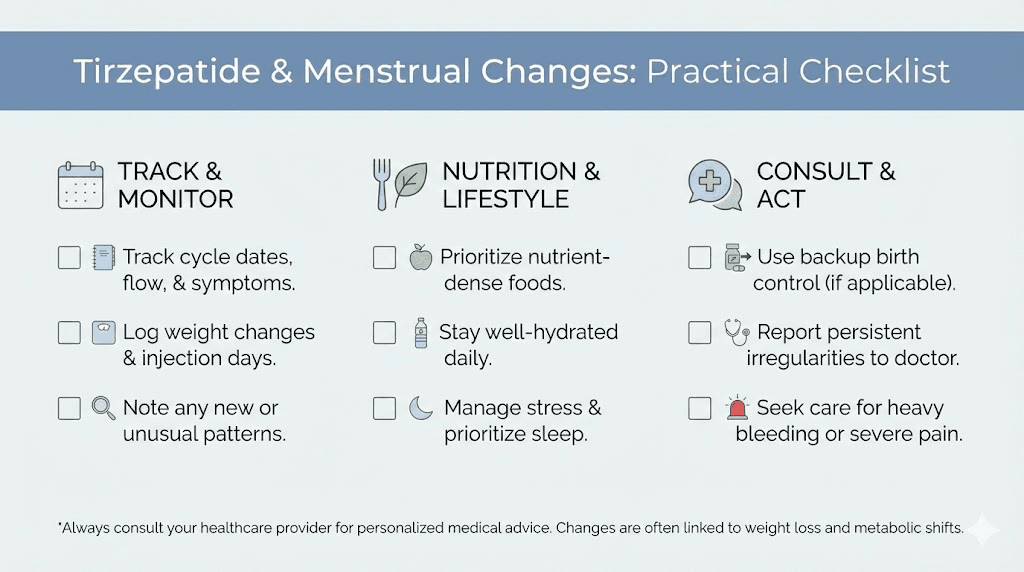
Knowing that menstrual changes are common and usually temporary is reassuring. But practical management strategies make the experience much more manageable. Here is what you can actively do.
Track your cycle from day one
Start tracking your menstrual cycle before you begin tirzepatide, if possible. Record cycle length, flow volume, symptom severity, and any irregularities. Continue tracking throughout treatment. This baseline data becomes invaluable for distinguishing tirzepatide-related changes from other potential causes.
Use a period tracking app or simple calendar method. Note your tirzepatide dose alongside your cycle data so you can identify correlations between dose escalations and cycle changes. Many women discover that their most significant menstrual disruptions coincide with the dosage adjustment periods rather than steady-state phases.
Maintain adequate caloric intake
This is the single most impactful intervention for minimizing menstrual disruption. Tirzepatide suppresses appetite powerfully, and many women find themselves eating far less than their body needs to maintain normal hormonal function. Aim for a minimum of 1,200 calories per day, even on days when you feel no hunger at all.
Focus on foods that support your body on tirzepatide, with particular attention to adequate protein (at least 60 to 80 grams per day) and healthy fats. Fat is essential for hormone production. Cutting dietary fat too aggressively while also losing body fat can compound the estrogen decline that drives menstrual changes.
Following a structured tirzepatide meal plan helps ensure you are meeting minimum nutritional requirements even when appetite is suppressed.
Support nutritional foundations
Several nutrients play direct roles in menstrual health and may need additional attention during tirzepatide treatment. Iron is particularly important for women experiencing heavier periods, as increased blood loss depletes iron stores faster. Women with PCOS often have underlying iron deficiency that worsens with heavier flow.
B vitamins, particularly B6 and B12, support hormonal metabolism and nervous system function. Many tirzepatide formulations now include B12 for this reason. Magnesium supports over 300 enzymatic processes including those involved in hormonal regulation, and supplementation may help with both menstrual cramps and the sleep difficulties some women experience on the medication.
Omega-3 fatty acids from fish oil or algae sources support anti-inflammatory pathways that influence menstrual comfort. Vitamin D plays a role in reproductive hormone regulation and is commonly deficient in women with obesity. A comprehensive approach to supplementation while taking tirzepatide addresses these needs systematically.
Manage stress and exercise appropriately
High-intensity exercise combined with caloric restriction and tirzepatide can create a cortisol environment that compounds menstrual disruption. This does not mean you should avoid exercise. It means you should be strategic about it.
Moderate exercise, such as walking, swimming, yoga, or light resistance training, supports metabolic health without creating excessive stress. Avoid combining tirzepatide with extreme exercise programs, especially during the initial adjustment phase when your body is already managing multiple metabolic changes simultaneously.
Sleep quality matters enormously for hormonal health. Women on tirzepatide who also experience insomnia or poor sleep should address this proactively, as cortisol elevation from sleep deprivation adds another layer of hormonal disruption. Understanding the fatigue patterns on tirzepatide helps you plan appropriate rest and recovery.
Consider your dose escalation timeline
Slower dose escalation may reduce the severity of menstrual disruption. Each dose increase triggers a new wave of metabolic adjustment that can affect cycle regularity. If menstrual changes are severe, discuss with your prescriber whether a more gradual escalation schedule might be appropriate.
Some women find that microdosing approaches to tirzepatide produce fewer side effects overall, including less menstrual disruption, while still achieving meaningful weight loss. The trade-off is a slower rate of weight loss, but for women prioritizing cycle stability, this may be worthwhile.
Tirzepatide vs semaglutide: comparing menstrual effects
Women choosing between tirzepatide and semaglutide often want to know which medication is less likely to affect their cycle. The honest answer is that both can, and the mechanisms are similar, but there are meaningful differences worth understanding.
Mechanism differences that matter
Tirzepatide activates both GLP-1 and GIP receptors. Semaglutide activates only GLP-1 receptors. This dual mechanism gives tirzepatide a greater effect on gastric emptying compared to typical GLP-1 receptor agonists alone. Greater gastric emptying delay means a potentially stronger interaction with oral contraceptives, and the distinct GIP receptor activation creates a different metabolic profile that may influence hormonal systems differently.
Tirzepatide also produces greater average weight loss than semaglutide in head-to-head comparisons. The SURMOUNT trials showed 20 to 26 percent weight loss compared to the roughly 15 percent seen with semaglutide in the STEP trials. Greater weight loss means greater metabolic change, which means a potentially larger impact on the HPO axis and menstrual function.
For a detailed analysis of how the two medications compare across all side effect categories, see our comprehensive semaglutide vs tirzepatide side effects comparison.
What semaglutide users report about periods
Women taking semaglutide report similar menstrual changes to tirzepatide users: irregular timing, altered flow, missed periods, and spotting. The patterns are largely the same because the underlying mechanism, weight loss-mediated hormonal change, is the same.
A meta-analysis of GLP-1 receptor agonist studies in women with PCOS found that these medications improved natural pregnancy rates and menstrual regularity across the class. The benefits appear to be class-wide rather than specific to any single medication. Women who have experienced menstrual disruption on semaglutide and are considering switching between the two medications should expect similar menstrual effects from either option.
Comparison table: menstrual effects
Factor | Tirzepatide | Semaglutide |
|---|---|---|
Average weight loss | 20-26% body weight | 12-17% body weight |
Gastric emptying delay | Greater (dual receptor) | Moderate (GLP-1 only) |
Oral contraceptive interaction | FDA warning, up to 59% reduction | Less studied, likely present |
PCOS improvement potential | Strong evidence | Strong evidence |
Menstrual disruption rate | ~27% reported | Similar rates reported |
Timeline to stabilization | 3-6 months typically | 3-6 months typically |
Both medications work through similar pathways, and neither is clearly better or worse for menstrual health. The choice between them should be based on overall efficacy, side effect profile, and individual response rather than menstrual effects alone. Use the full semaglutide vs tirzepatide comparison to evaluate all factors.
When weight loss medications restore fertility unexpectedly
One of the most significant and underappreciated effects of tirzepatide on reproductive health is its potential to restore fertility in women who were previously subfertile or infertile due to obesity-related hormonal disruption.
The mechanism of restored ovulation
Excess body weight suppresses ovulation through multiple pathways: elevated estrogen from adipose tissue, insulin resistance driving ovarian androgen production, chronic low-grade inflammation, and altered leptin signaling. When tirzepatide resolves these metabolic disturbances, ovulation can resume spontaneously.
For women who have spent years assuming they cannot get pregnant, or who were told their weight was preventing conception, this restoration of fertility can come as a complete surprise. Reports of unplanned pregnancies on tirzepatide and similar medications are accumulating, and physicians are increasingly counseling patients about this possibility.
Women who are sexually active, not using reliable contraception, and experiencing restored menstrual regularity on tirzepatide should consider this a signal that fertility may be returning. A conversation with a reproductive health provider is warranted.
The pregnancy timing consideration
Tirzepatide is not approved for use during pregnancy, and the FDA recommends stopping the medication at least two months before a planned pregnancy. Women who discover they are pregnant while on tirzepatide should discontinue the medication and contact their healthcare provider immediately.
The experiences of women who became pregnant on tirzepatide provide important real-world context for this scenario. While data on tirzepatide exposure during early pregnancy is limited, animal studies have shown adverse developmental effects at supratherapeutic doses, warranting the precautionary approach.
Tirzepatide, menopause, and perimenopause
Women in perimenopause or post-menopause face unique considerations when evaluating menstrual changes on tirzepatide. The hormonal changes of reproductive aging overlap with medication-induced changes in ways that can be confusing.
Perimenopause: separating causes
Perimenopausal women already experience irregular cycles, changing flow, and hormonal fluctuations as part of the natural transition toward menopause. Adding tirzepatide to this equation makes it difficult to attribute any specific menstrual change to the medication versus the underlying perimenopausal process.
The practical approach for perimenopausal women is the same: track cycles carefully, maintain adequate nutrition, and report significant changes to your healthcare provider. Do not assume that new or unusual bleeding patterns are simply perimenopause without medical evaluation, as unexplained bleeding in this age group warrants investigation.
Women navigating perimenopause alongside weight management may benefit from exploring peptide options specifically suited to perimenopause, as different compounds offer different benefits for this life stage.
Postmenopause: the return of bleeding
Postmenopausal women who have not had a period in over 12 months should not experience vaginal bleeding on tirzepatide or any other medication. If bleeding occurs in a postmenopausal woman, it requires prompt medical evaluation regardless of whether she is taking tirzepatide. Postmenopausal bleeding can indicate endometrial hyperplasia, polyps, or other conditions that need investigation.
The relationship between peptides and menopause is an evolving area of research, with some compounds showing promise for managing specific menopausal symptoms.
Hormone therapy interaction and weight loss
The 2025 Lancet study on tirzepatide combined with menopause hormone therapy has important implications. Postmenopausal women who are already on hormone replacement therapy may experience enhanced weight loss outcomes when adding tirzepatide. The 17 percent average weight loss with combined therapy versus 14 percent with tirzepatide alone is clinically meaningful.
For women over 40 exploring peptide-based weight management, understanding these interactions helps optimize treatment strategies. Coordination between your prescribing physician and endocrinologist ensures that hormonal therapies and weight management medications work together rather than at cross-purposes.
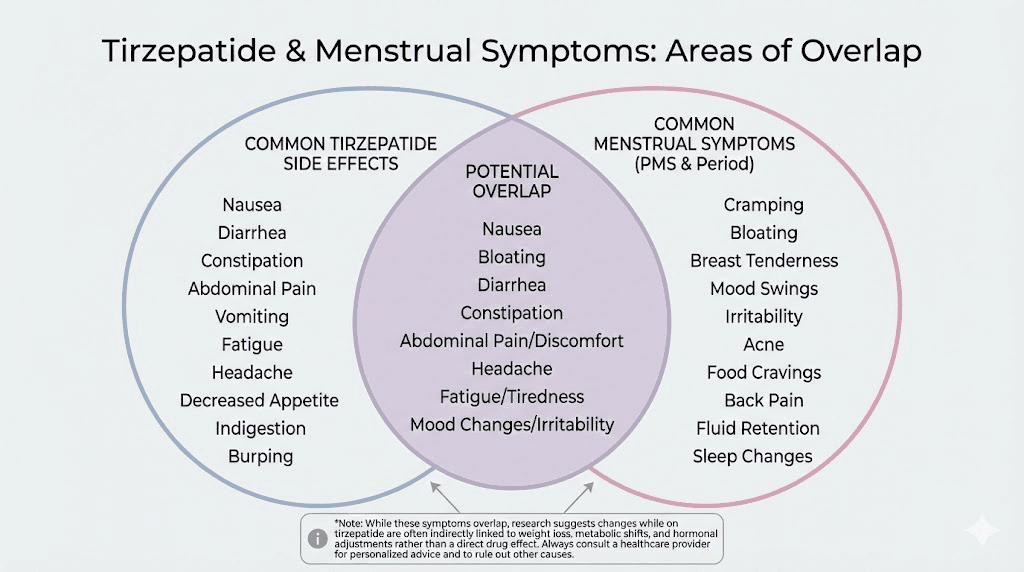
Side effects that overlap with menstrual symptoms
Several common tirzepatide side effects can mimic, worsen, or be confused with menstrual symptoms. Recognizing these overlaps prevents misattribution and ensures appropriate management.
Fatigue and energy changes
Tirzepatide commonly causes fatigue, especially during dose escalation and periods of rapid weight loss. This fatigue can intensify premenstrual tiredness and make the week before your period feel significantly more draining than usual. Women experiencing both energy changes on tirzepatide and menstrual-related fatigue may find the combination challenging.
Distinguishing medication fatigue from premenstrual fatigue helps you manage both effectively. Tracking energy levels alongside your cycle reveals patterns that inform both your medication management and your activity planning.
Gastrointestinal symptoms
Nausea, bloating, and abdominal discomfort are among the most common tirzepatide side effects. These overlap significantly with premenstrual and menstrual symptoms. A woman experiencing bloating and cramping mid-cycle may struggle to determine whether the symptoms are medication-related, menstrual, or both.
Understanding which foods to avoid on tirzepatide can help minimize the gastrointestinal contribution to your symptoms, making it easier to identify which discomfort is menstrual in origin. Managing constipation on tirzepatide is particularly important, as constipation can worsen pelvic pressure and cramping during menstruation.
Headaches
Menstrual migraines and hormonal headaches are common in women, and tirzepatide can independently cause headaches as a side effect. The combination can create headache patterns that are more frequent or severe than either factor alone. Women who track both their cycle and their tirzepatide headache patterns can identify whether headaches cluster around menstrual timing, dose escalation, or both.
Mood changes and anxiety
Hormonal fluctuations affect mood. Tirzepatide-related metabolic changes add another layer of neurochemical adjustment. Some women report heightened anxiety or mood instability that seems to worsen around their period. Understanding whether tirzepatide contributes to anxiety helps you work with your provider to address mood symptoms from the right angle.
Muscle pain and body aches
Menstrual cramps involve prostaglandin-mediated muscle contraction. Muscle pain is a reported tirzepatide side effect in some users. When both occur simultaneously, the pain experience can be more intense than either would be alone. Anti-inflammatory approaches that address both menstrual pain and medication-related muscle discomfort provide the most efficient relief.
Practical protocols for women starting tirzepatide
Based on the evidence and patient experiences, here are specific approaches for women who want to minimize menstrual disruption while maximizing the benefits of tirzepatide treatment.
Protocol 1: pre-treatment preparation
Goal: Establish baseline data and create a supportive foundation before starting tirzepatide.
Steps:
Track 2-3 menstrual cycles before starting treatment, recording length, flow, and symptoms
Get baseline bloodwork including CBC (for iron status), thyroid panel, and reproductive hormone panel (FSH, LH, estradiol, progesterone, testosterone, DHEA-S)
Review contraception with your provider, switching to non-oral methods if using oral birth control
Stock appropriate supplies for potentially heavier or irregular periods (various absorbency products)
Begin a daily multivitamin with iron, B12, and vitamin D at least two weeks before starting
Women preparing to start tirzepatide should also review the most common mistakes beginners make with peptide-based treatments to optimize their experience from the start.
Protocol 2: first three months management
Goal: Navigate the adjustment phase with minimal disruption and maximum awareness.
Steps:
Continue tracking menstrual cycles alongside dose changes and side effects
Eat at least 1,200 calories daily with 60-80g protein and adequate healthy fats
Avoid extreme exercise during dose escalation weeks
Keep a symptom journal noting headaches, fatigue, mood changes, and GI symptoms alongside cycle data
Use backup contraception for four weeks after each dose increase if using oral birth control
Take a pregnancy test if you miss a period, especially if sexually active without reliable contraception
Follow your tirzepatide dosing protocol consistently, as erratic dosing can create additional hormonal instability.
Protocol 3: ongoing optimization
Goal: Support long-term menstrual health while maintaining treatment benefits.
Steps:
Repeat bloodwork at 3 and 6 months to monitor iron status, thyroid function, and metabolic markers
Adjust caloric intake upward as weight loss slows, matching intake to your new energy needs
Consider body composition testing (DEXA scan) if concerned about body fat percentage dropping too low
Report persistent irregularity beyond 6 months to your provider for evaluation
Maintain nutritional supplementation with emphasis on iron (if periods are heavier), B12, magnesium, and omega-3 fatty acids
Working with a qualified peptide therapy provider ensures ongoing monitoring and adjustment based on your individual response.
The emotional side of period changes on tirzepatide
Clinical discussions about menstrual changes tend to focus on mechanisms and management. But the emotional impact deserves acknowledgment.
Your period is personal. It connects to your sense of femininity, fertility, health, and normalcy in ways that clinical data does not capture. When it changes unexpectedly, especially due to a medication, the experience can trigger anxiety, frustration, and uncertainty that goes beyond the physical symptoms.
Some women feel betrayed by a medication that was supposed to improve their health but seems to be disrupting something fundamental. Others feel alarmed by the possibility of pregnancy when they thought they were protected. Some women with PCOS experience complex emotions when their period returns after years of absence, grateful for the sign of improving health but unprepared for the practical reality.
These emotional responses are valid and normal. They are not a reason to stop treatment, but they are a reason to seek support. Talk to your prescriber about what you are experiencing. Connect with other women navigating the same changes. And remember that for the vast majority of women, these changes are temporary. Your body is adjusting to a new metabolic reality, and it will find its balance.
For researchers serious about optimizing their peptide protocols and managing the full spectrum of effects, SeekPeptides offers comprehensive resources including detailed protocol guides, community support from experienced users, and evidence-based management strategies for exactly these kinds of situations.
Long-term reproductive health considerations
Beyond the immediate menstrual changes, women on tirzepatide should consider the broader reproductive health implications of sustained treatment.
Bone health and estrogen
Estrogen plays a critical role in maintaining bone density. Prolonged periods of low estrogen, whether from menopause, hypothalamic amenorrhea, or medication-related hormonal changes, increase the risk of bone loss. Women who experience extended amenorrhea (absent periods) on tirzepatide should discuss bone density monitoring with their provider.
This concern is most relevant for women who lose very large amounts of weight rapidly and experience amenorrhea lasting more than six months. For most women, the temporary menstrual irregularity associated with tirzepatide does not pose significant long-term bone health risks. But awareness of this connection supports informed decision-making.
Fertility planning
Women planning to conceive in the near future should discuss the timing of tirzepatide treatment with their reproductive health provider. The FDA recommends stopping tirzepatide at least two months before a planned pregnancy. However, the improvements in insulin sensitivity, body composition, and hormonal balance that tirzepatide produces may actually optimize fertility potential.
For women with PCOS-related infertility, achieving significant weight loss on tirzepatide before attempting conception may improve pregnancy rates and reduce pregnancy complications. The sequencing of treatment relative to conception plans requires individualized medical guidance.
Thyroid function monitoring
Thyroid disorders can cause menstrual irregularities that overlap with tirzepatide-related changes. The safety considerations for peptide treatments include monitoring thyroid function, especially given the boxed warning on tirzepatide regarding thyroid C-cell tumors observed in animal studies.
Baseline and periodic thyroid testing helps distinguish menstrual changes caused by thyroid dysfunction from those related to weight loss and metabolic adjustment. This is particularly important for women with pre-existing thyroid conditions or family history of thyroid disease.
What other GLP-1 medications do to periods
Tirzepatide is not the only medication in this class that affects menstruation. Understanding the broader pattern helps contextualize the experience.
Semaglutide (Ozempic, Wegovy)
Women on semaglutide report similar menstrual changes to tirzepatide users. A comprehensive look at how semaglutide affects your period reveals the same patterns: irregular timing, altered flow, missed periods, and eventual stabilization. The key difference is that semaglutide typically produces less weight loss than tirzepatide, which may mean slightly less hormonal disruption for some women. Understanding the withdrawal timeline for semaglutide also matters for women considering treatment breaks.
Retatrutide and emerging medications
Retatrutide, a triple agonist (GLP-1, GIP, and glucagon receptor), is showing even greater weight loss potential than tirzepatide in clinical trials. If the relationship between weight loss magnitude and menstrual disruption holds, women on retatrutide may experience even more pronounced cycle changes. Emerging medications like survodutide, mazdutide, and orforglipron also warrant monitoring for menstrual effects as their use expands.
The side effect profiles of newer agents like cagrilintide are still being characterized, and menstrual effects will likely mirror the class pattern observed with tirzepatide and semaglutide.
The class effect pattern
Across all GLP-1 based medications, the pattern is consistent: menstrual changes are common, driven primarily by weight loss rather than direct hormonal interaction, usually temporary, and potentially beneficial for women with PCOS. The specific medication matters less than the magnitude and speed of weight loss it produces.
Women choosing between options should focus on overall efficacy and tolerability rather than menstrual effects alone. A thorough comparison of available options helps inform this decision.
When to see a doctor about period changes on tirzepatide
Most menstrual changes on tirzepatide resolve without medical intervention. But certain situations require prompt evaluation.
Red flags that need immediate attention
Very heavy bleeding: Soaking through a pad or tampon every hour for several consecutive hours
Bleeding lasting more than 7 days: Prolonged menstrual bleeding that does not resolve
Severe cramping: Pain that is significantly worse than your normal menstrual cramps and does not respond to over-the-counter pain medication
Possible pregnancy: Any missed period in a sexually active woman, especially given the contraceptive interaction
Postmenopausal bleeding: Any vaginal bleeding after 12 or more months without a period
Yellow flags that warrant a scheduled appointment
Three or more consecutive missed periods in a woman who was previously regular
Six or more months of amenorrhea in a woman who was previously irregular
New symptoms: Severe mood changes, significant hair loss, extreme fatigue, or other symptoms suggesting broader hormonal disruption
Persistent irregularity beyond 12 months of treatment that shows no trend toward stabilization
Concern about fertility: Any woman planning pregnancy within the next 12 months
Women experiencing hair loss alongside menstrual changes should seek evaluation, as the combination may indicate more significant hormonal disruption that warrants investigation. Similarly, persistent lack of progress on tirzepatide combined with menstrual changes could indicate a hormonal imbalance that is interfering with both weight loss and cycle regularity.
What to tell your doctor
When discussing menstrual changes with your healthcare provider, bring specific data rather than general impressions. Your cycle tracking records, tirzepatide dosing timeline, weight loss trajectory, and symptom journal provide the concrete information your doctor needs to assess your situation accurately.
Key information to communicate includes your last menstrual period date, typical cycle length before and after starting tirzepatide, changes in flow volume or duration, any intermenstrual bleeding, current tirzepatide dose, total weight lost, and any other symptoms that have developed during treatment.
Frequently asked questions
Does tirzepatide directly cause period changes?
No. Tirzepatide does not directly interact with reproductive hormones. The menstrual changes women experience are primarily caused by the metabolic effects of the medication, specifically the weight loss, improved insulin sensitivity, and hormonal shifts that accompany significant changes in body composition. Understanding how peptides work in the body helps clarify why indirect effects can still be significant.
Will my period go back to normal after stopping tirzepatide?
For most women, yes. Menstrual cycles typically return to their pre-treatment pattern once weight stabilizes, whether that happens during continued treatment or after stopping the medication. Over 80 percent of women regain regular ovulatory cycles within nine months of achieving stable energy balance. Women concerned about what happens when stopping GLP-1 medications should discuss a gradual tapering strategy with their provider.
Can tirzepatide make me more fertile?
Yes. Tirzepatide can restore ovulation in women whose excess weight was previously suppressing it. This is particularly relevant for women with PCOS, where improved insulin sensitivity and weight loss can normalize ovarian function. Women who do not want to become pregnant should use reliable contraception throughout treatment, keeping in mind the FDA warning about oral contraceptive interaction.
Should I stop tirzepatide if my period becomes irregular?
In most cases, no. Menstrual irregularity during the first three to six months of treatment is common and usually resolves without intervention. Stopping the medication removes its weight management benefits without necessarily resolving the menstrual changes more quickly, as your body still needs to adjust to its new metabolic state. Discuss concerns with your prescriber before making changes to your treatment plan.
Does tirzepatide affect period cramps?
Some women report changes in cramping severity, both worsening and improvement. Women whose prostaglandin levels shift with changing hormonal balance may experience different cramp intensity. The anti-inflammatory effects of improved metabolic health may actually reduce cramping for some women over time. Managing muscle pain and discomfort on tirzepatide can help address overlapping pain symptoms.
Is it safe to use tirzepatide during my period?
Yes. There is no need to adjust your tirzepatide dosing schedule around your menstrual cycle. The medication is safe to use at all points in your cycle. If you experience worse nausea or GI symptoms during your period while on tirzepatide, timing your injection for a day when you are not menstruating may improve comfort, but this is a preference rather than a medical necessity.
Can I take iron supplements with tirzepatide?
Yes, and women experiencing heavier periods on tirzepatide should consider iron supplementation to prevent deficiency. Take iron supplements at least two hours apart from your tirzepatide injection to optimize absorption, as delayed gastric emptying could affect the timing of nutrient absorption. Including iron-rich foods in your tirzepatide diet plan provides additional support.
Does the dose of tirzepatide affect how much it changes your period?
Higher doses generally produce greater weight loss and more pronounced metabolic changes, which can translate to more significant menstrual effects. Women on the 15 mg dose may experience more cycle disruption than those on 5 mg, though individual variation is substantial. Starting dose selection and escalation speed both influence the menstrual impact.
External resources
The Potential Utility of Tirzepatide for the Management of Polycystic Ovary Syndrome - PMC
Effects of GLP1RAs on Pregnancy Rate and Menstrual Cyclicity in Women with PCOS - PMC
Functional Hypothalamic Amenorrhea: Endocrine Society Clinical Practice Guideline
Body Weight Reduction in Women Treated with Tirzepatide by Reproductive Stage - PMC
For researchers serious about understanding the full spectrum of peptide effects on the body, SeekPeptides provides the most comprehensive resource available, with evidence-based guides, detailed protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your cycles stay regular, your hormones stay balanced, and your health goals stay within reach.