Feb 18, 2026

What if the real question is not how much retatrutide to take, but how much retatrutide YOUR body actually needs? Because those are two very different questions. One gives you a number from a chart. The other gives you a protocol that works.
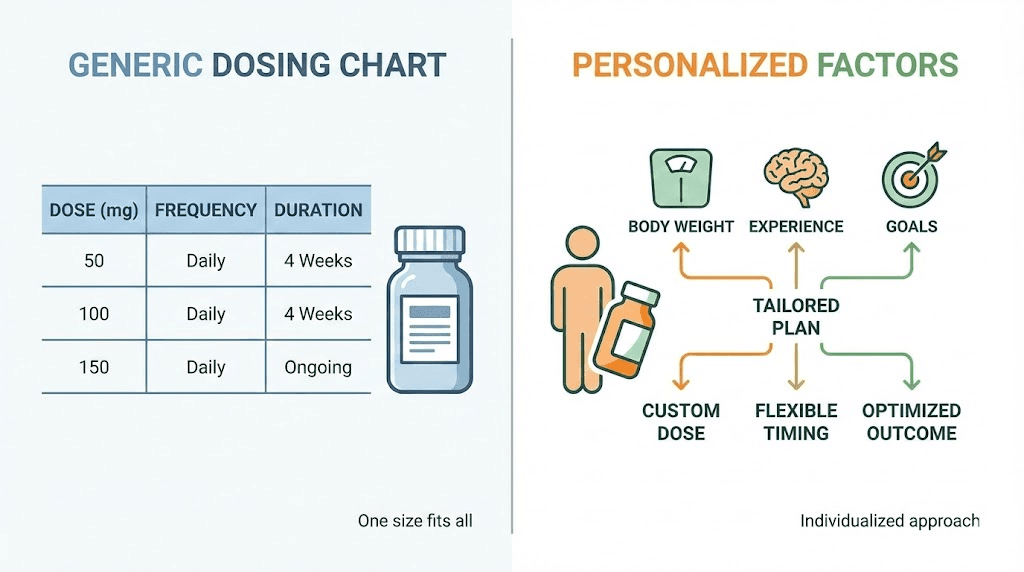
Most people searching for retatrutide dosing information find the same recycled table. Start at 1 mg. Increase every four weeks. Top out at 12 mg. And while that basic framework comes straight from the Phase 2 clinical trial published in the New England Journal of Medicine, it tells you almost nothing about what dose is right for you specifically. Your body weight matters. Your previous experience with GLP-1 receptor agonists matters. Your tolerance for gastrointestinal side effects matters. And your goals, whether that is 15% body weight reduction or 25% or something else entirely, absolutely matter.
Retatrutide is not semaglutide. It is not tirzepatide either. It is the first triple receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. That triple mechanism changes everything about how the compound affects appetite, metabolism, and fat oxidation. It also means the dosing considerations are more nuanced than anything that came before it.
This guide breaks down exactly how much retatrutide to take based on your specific situation. Not a generic chart copied from a clinical trial summary. Actual guidance based on body weight ranges, prior peptide experience, individual goals, and the tolerance patterns observed across hundreds of documented protocols. SeekPeptides members have access to even more detailed protocol builders, but this guide covers everything you need to make an informed decision about your starting dose, your escalation timeline, and your maintenance target.
How retatrutide works and why dosing matters more than you think
Before deciding how much retatrutide to take, you need to understand what makes this compound fundamentally different from every other weight loss peptide on the market. Retatrutide is a triple hormone receptor agonist developed by Eli Lilly. It activates three distinct receptor systems simultaneously. GLP-1 receptors control appetite and gastric emptying. GIP receptors influence insulin secretion and lipid metabolism. And glucagon receptors drive energy expenditure and fat mobilization directly.
That third receptor is the game changer.
Semaglutide hits one receptor. Tirzepatide hits two. Retatrutide hits all three. And the glucagon receptor activation creates something no other approved or investigational weight loss compound currently delivers: direct stimulation of thermogenesis and hepatic fat oxidation. Your body does not just eat less. It actively burns more. The comparison between retatrutide and semaglutide makes this distinction clear in the clinical data.
In the Phase 2 trial published in the New England Journal of Medicine, 338 participants were randomized across multiple dose groups. The results at 48 weeks told a striking story. The 1 mg group lost 8.7% of body weight. The 4 mg group lost 17.1%. The 8 mg group lost 22.8%. And the 12 mg group lost 24.2%, compared to just 2.1% in the placebo group. Those are not small differences between doses. Moving from 4 mg to 12 mg nearly doubled the absolute weight loss percentage. Dose matters enormously with retatrutide because each receptor system has its own dose-response curve, and the glucagon receptor component appears to require higher doses to fully engage.
But here is what the summary data does not show you. The side effect profile also scales with dose. And the rate of dose escalation dramatically affects tolerability. Participants who started directly at 4 mg experienced nearly twice the rate of gastrointestinal symptoms compared to those who titrated up from 2 mg. The how-fast-you-get-there matters just as much as the where-you-end-up.
The three receptor systems and their dose thresholds
Each receptor system in retatrutide has a different activation threshold, and understanding this helps explain why certain doses produce dramatically different results.
GLP-1 receptor activation begins at the lowest doses. Even at 1 mg weekly, most users report noticeable appetite suppression and reduced food noise. This receptor drives the satiety signal that makes you feel full sooner and think about food less often. If you have used semaglutide or tirzepatide before, this effect will feel familiar, though the intensity profile differs because retatrutide has relatively lower GLP-1 potency compared to dedicated GLP-1 agonists.
GIP receptor activation is where retatrutide actually shows its strongest potency relative to the natural hormone. This system enhances insulin sensitivity and shifts lipid metabolism toward utilization rather than storage. The GIP component works synergistically with GLP-1 to amplify appetite control while independently improving how your body processes and distributes dietary fat. You start seeing meaningful GIP-mediated effects around the 2 to 4 mg range, which is why the 4 mg dose group already showed impressive 17.1% weight loss at 48 weeks.
Glucagon receptor activation requires the highest doses to fully engage. This is the unique component that separates retatrutide from everything else. Glucagon receptor agonism increases resting energy expenditure, promotes hepatic fat oxidation, and drives thermogenesis. The jump from 8 mg to 12 mg, where glucagon receptor engagement becomes more complete, produced measurable additional weight loss in the clinical data. It also showed particular benefits for visceral fat reduction and liver fat clearance.
A Phase 2a trial specifically examining metabolic dysfunction-associated steatotic liver disease (MASLD) found that retatrutide at 12 mg reduced liver fat by an extraordinary amount, with some participants achieving complete resolution of fatty liver. That glucagon-driven hepatic effect does not fully activate at lower doses.
Why the standard chart is not enough
Every retatrutide dosing chart you find online shows the same thing: start at 1 mg, go to 2 mg at week 5, then 4 mg, then 8 mg, then 12 mg. And that is a reasonable general framework pulled from the clinical trial protocol. But clinical trials use standardized dosing for statistical analysis. They are not designed to optimize individual outcomes.
The standard retatrutide dosage chart does not account for a 130-pound woman who has never used any GLP-1 compound. It does not account for a 280-pound man who has been on tirzepatide at 15 mg for six months and wants to switch. It does not differentiate between someone targeting 10% body weight loss and someone targeting 25%. These are dramatically different situations that call for dramatically different dosing strategies.
Starting dose: what the research says versus what actually works
The clinical trial starting dose for retatrutide is 1 mg subcutaneously once weekly. Period. Every trial arm except one used this as the entry point, and for good reason. Starting low allows the body to adapt to triple receptor activation gradually. The gastrointestinal system needs time to adjust to slowed gastric emptying. The liver needs time to adapt to enhanced glucagon signaling. And the metabolic shift from storage mode to oxidation mode does not happen overnight.
Some researchers ask whether they can start higher. The answer from the data is clear: do not.
The Phase 2 trial actually tested this directly. Some dose groups started at 2 mg while others started at 4 mg before escalating to their target doses. The groups that started at 4 mg experienced significantly more nausea, vomiting, and diarrhea than those who began at 2 mg. The clinical investigators specifically noted that "gastrointestinal adverse events were partially mitigated by the use of a lower starting dose." Starting at 1 mg, which is what the Phase 3 TRIUMPH trials use, provides even more tolerability protection.
1 mg for four weeks: what to expect
During your first four weeks at 1 mg, the primary effects come from GLP-1 receptor activation. You should notice reduced appetite, some decrease in food noise (the constant background thinking about food), and potentially mild gastrointestinal adjustment. Weight loss during this phase is modest, typically 1 to 3% of body weight over four weeks. Do not be discouraged. This phase is about building tolerance, not maximizing results.
Common experiences at 1 mg include mild nausea in the first 24 to 48 hours after injection, particularly after the first dose. This typically resolves within days. Some users report decreased interest in food that feels subtle rather than dramatic. Energy levels may fluctuate during the first week as your metabolism begins to shift. Mild fatigue is normal and usually resolves by week two.
If you experience zero side effects at 1 mg after four weeks, that is a good sign. It means your body adapted smoothly and you are ready for the next escalation step. If you experience persistent nausea or significant gastrointestinal discomfort even at 1 mg, that is also useful information. It suggests your system is highly sensitive to GLP-1 activation, and you may want to extend the 1 mg phase to six or even eight weeks before moving up.
The exception: people coming from tirzepatide or semaglutide
If you have been on semaglutide or tirzepatide and are transitioning to retatrutide, your body already has significant adaptation to GLP-1 and possibly GIP receptor activation. You will not experience the same initial GI shock that a completely naive user would. However, the glucagon receptor component is entirely new to your system, so starting at 1 mg is still recommended.
The transition from tirzepatide to retatrutide is particularly interesting because tirzepatide already activates both GLP-1 and GIP receptors. The transition timeline from tirzepatide to retatrutide depends on your previous dose and how long you were on it. Some researchers report that the adaptation phase at 1 mg and 2 mg feels like a step backward in appetite suppression because retatrutide has relatively lower GLP-1 potency. The full effects, especially the thermogenic and fat-oxidation benefits from glucagon activation, typically do not manifest until you reach the 4 to 8 mg range.
For people coming from high-dose semaglutide (2.4 mg weekly), the same principle applies. Your GLP-1 receptors are well-adapted, but the additional receptor systems need introduction. Patience through the first eight weeks pays dividends in tolerability at higher doses.
Dose escalation: the four-week framework and when to adjust it
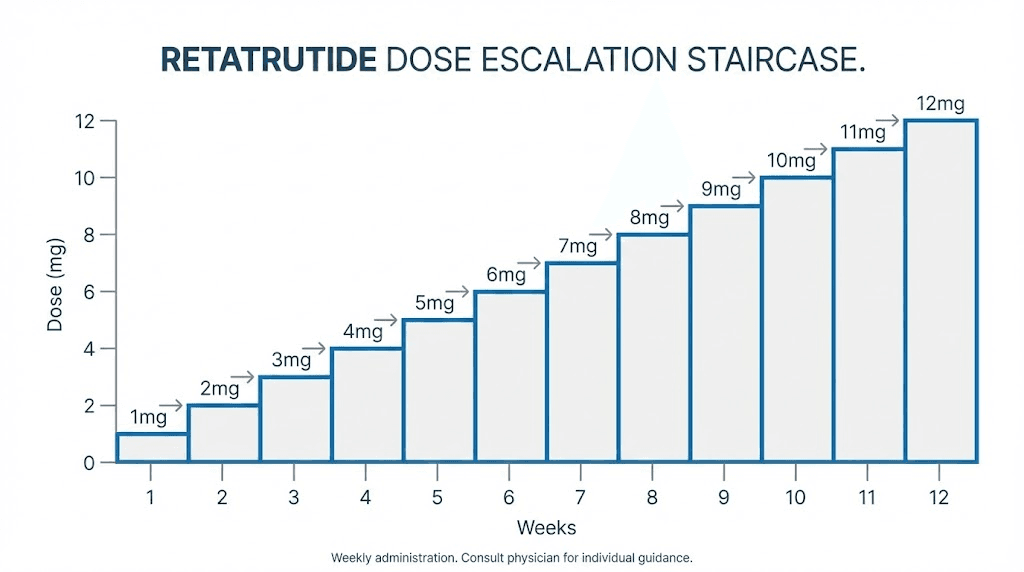
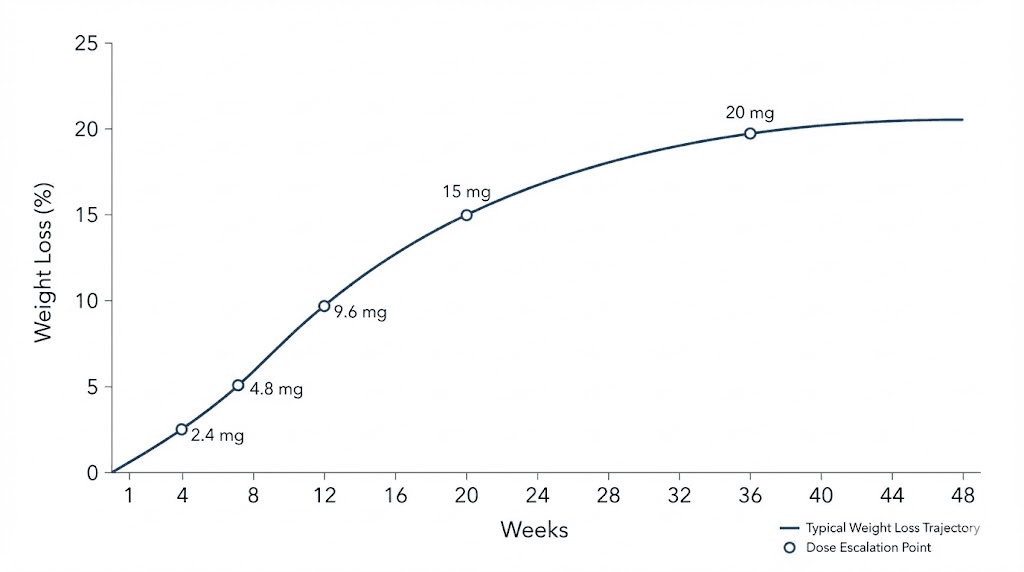
The standard retatrutide escalation protocol increases the dose every four weeks. This comes directly from the clinical trial design, and four weeks is generally sufficient for most people to adapt to each new dose level before moving up. The standard dose schedule looks like this.
Weeks 1 through 4: 1 mg weekly. Weeks 5 through 8: 2 mg weekly. Weeks 9 through 12: 4 mg weekly. Weeks 13 through 16: 8 mg weekly. Weeks 17 onward: 12 mg weekly.
That gives you a 16-week ramp to the maximum studied dose. But not everyone should follow this exact timeline. And not everyone should target 12 mg.
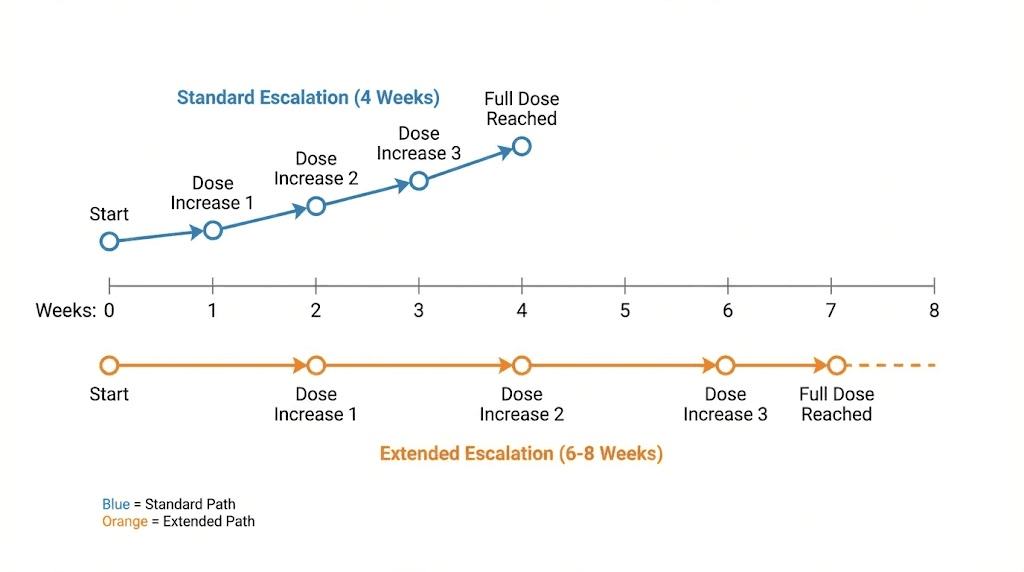
When to slow down the escalation
Slow your escalation if any of these apply. Persistent nausea lasting more than 48 hours after each injection. Vomiting more than once per week. Diarrhea that disrupts daily activities. Significant fatigue or energy crashes that do not resolve by mid-week. Inability to maintain adequate hydration or nutrition.
If you experience moderate to severe gastrointestinal symptoms at any dose, extend that dose level to six or eight weeks before attempting the next increase. There is no penalty for taking longer. The clinical trials used four-week intervals for standardization, not because four weeks is the medically optimal adaptation period for every individual. Some people need six weeks. Some need eight. SeekPeptides members who track their escalation timelines consistently show that patient titration produces better long-term adherence and outcomes than rushing to the maximum dose.
When to speed up (rarely)
Accelerating escalation is almost never appropriate with retatrutide. Even if you tolerate a dose perfectly with zero side effects, the four-week minimum allows for metabolic adaptation that is not always reflected in symptoms. Your liver, pancreas, and metabolic systems are adjusting to triple receptor activation in ways that are not immediately perceptible.
The only scenario where a slightly faster escalation might be considered is for someone transitioning from high-dose tirzepatide (10 to 15 mg) who shows zero GI response at 1 mg and 2 mg. In that case, some protocols shorten the 1 mg phase to three weeks and the 2 mg phase to three weeks, reaching 4 mg by week 7 instead of week 9. This is not a dramatic acceleration, and it should only be considered when the individual has extensive documented tolerance to dual-receptor agonism.
Never skip dose levels entirely. Going from 2 mg directly to 8 mg, or from 4 mg to 12 mg, dramatically increases the risk of severe gastrointestinal events and potential complications. The Phase 2 data showed this directly, and it is one of the clearest safety signals in the entire retatrutide dataset.
How much retatrutide to take based on your body weight
Body weight is one of the most important factors in determining your optimal retatrutide dose, though the relationship is not as simple as a milligram-per-kilogram calculation. The clinical trials used fixed doses (1, 4, 8, and 12 mg) rather than weight-based dosing, which means the same 12 mg dose was given to a 170-pound woman and a 300-pound man. But the pharmacokinetic profile and clinical response differ significantly across body weight ranges.
Under 150 pounds (under 68 kg)
If you weigh under 150 pounds, you are on the lighter end of the clinical trial population, and lower doses may produce proportionally stronger effects. The 4 mg maintenance dose produced 17.1% average weight loss across the full trial population, but lighter individuals within that group often experienced responses closer to 20% or higher. At this body weight, the milligram-per-kilogram exposure is significantly higher than for heavier participants.
Practical considerations for lighter individuals include potentially stronger gastrointestinal side effects at each dose level, faster onset of appetite suppression, and the possibility that 4 to 8 mg may be your effective maintenance range rather than 12 mg. Pay close attention to your body during escalation. If 4 mg produces dramatic appetite suppression and you are losing weight consistently, there may be no need to push to 8 or 12 mg.
150 to 220 pounds (68 to 100 kg)
This range covers the majority of the clinical trial population and is where the standard titration data most directly applies. Most individuals in this weight range will find their optimal maintenance dose between 8 and 12 mg. The 8 mg group in the Phase 2 trial achieved 22.8% average weight loss at 48 weeks, which is extraordinary by any standard.
The key decision for this weight range is whether to target 8 mg or push to 12 mg. If you are tolerating 8 mg well with consistent weight loss of 1 to 2 pounds per week, staying at 8 mg is entirely reasonable. The additional 1.4% average weight loss from 8 to 12 mg comes with additional GI side effect risk. Some researchers in this range find that 8 mg provides the best balance of efficacy and tolerability.
Over 220 pounds (over 100 kg)
Heavier individuals generally need higher doses to achieve equivalent plasma concentrations and clinical effects. The pharmacokinetics of peptide-based medications, including how they distribute through tissue and how quickly they are cleared, are influenced by body mass. If you weigh over 220 pounds, the standard escalation to 12 mg is typically appropriate, and you should expect the full escalation schedule before reaching your maintenance dose.
Interestingly, the Phase 2 data stratified by BMI showed that participants with BMI over 35 achieved greater absolute weight loss in pounds, even though the percentage weight loss was comparable to or slightly lower than lighter participants. A 300-pound person losing 24% of body weight loses 72 pounds. That is a profound transformation. For individuals starting at higher body weights, the retatrutide dosage chart for weight loss provides the visual reference for this standard escalation.
Heavier individuals also tend to tolerate escalation steps more smoothly because the per-kilogram exposure at each dose level is lower. This does not mean side effects will be absent, but the severity is often more manageable compared to lighter individuals taking the same absolute dose.
Weight-based dosing reference
While clinical trials do not use weight-based dosing for retatrutide, researchers often find it helpful to think about their dose in context of body weight. Here is a reference framework based on the available data and documented outcomes.
Body weight range | Likely starting dose | Common maintenance dose | Expected weight loss range |
|---|---|---|---|
Under 150 lbs | 1 mg | 4 to 8 mg | 18 to 26% |
150 to 200 lbs | 1 mg | 8 to 12 mg | 20 to 25% |
200 to 250 lbs | 1 mg | 8 to 12 mg | 20 to 24% |
Over 250 lbs | 1 mg | 12 mg | 18 to 24% |
These ranges reflect documented outcomes from clinical trials and published research protocols. Individual results vary based on diet, exercise, adherence, and metabolic factors. The retatrutide dosage calculator on our site can help you model specific scenarios.
How much retatrutide to take based on your goals
Your target outcome significantly influences the dose you should aim for. Not everyone using retatrutide is pursuing maximum weight loss. Some researchers prioritize metabolic health markers. Others want fat loss with muscle preservation. And some are specifically targeting liver fat reduction. Each goal suggests a different optimal dose range.
Goal: moderate weight loss (10 to 15% of body weight)
If your target is 10 to 15% body weight reduction, you may not need to escalate beyond 4 to 8 mg. The Phase 2 data showed that 92% of participants in the 4 mg group achieved at least 5% weight loss, 75% achieved at least 10%, and 60% achieved at least 15% at 48 weeks. These are remarkable response rates at a relatively moderate dose.
A 4 to 8 mg maintenance dose produces significantly fewer gastrointestinal side effects than 12 mg. For researchers whose primary goal is not maximum weight loss but rather meaningful, sustainable weight reduction with good quality of life during the process, this range represents the sweet spot. You still get substantial GLP-1 and GIP receptor activation with meaningful glucagon engagement. The appetite suppression is strong without being overwhelming. And the metabolic benefits, including improved insulin sensitivity and reduced inflammation, are significant at these doses.
Goal: aggressive weight loss (20% or more of body weight)
If you need to lose 20% or more of your body weight, the data strongly supports escalating to 8 or 12 mg. In the 8 mg group, 100% of participants lost at least 5%, 91% lost at least 10%, and 75% lost at least 15%. In the 12 mg group, 100% lost at least 5%, 93% lost at least 10%, and 83% lost at least 15%. The newest Phase 3 TRIUMPH-4 data showed average weight loss of 28.7% at doses of 9 to 12 mg over 68 weeks.
At these higher doses, the full triple-agonist mechanism is engaged. GLP-1 provides strong appetite suppression. GIP optimizes lipid metabolism. And glucagon drives significant energy expenditure increases. The combination creates a metabolic state that is extremely favorable for rapid, sustained fat loss. But the side effect burden is real. Nausea and headaches are more common. Mood changes can occur. And maintaining adequate nutrition while experiencing strong appetite suppression requires deliberate effort.
Goal: metabolic health improvement without maximum weight loss
Some researchers are primarily interested in retatrutide for metabolic health markers: insulin sensitivity, liver fat reduction, lipid profiles, and inflammatory markers. For these goals, moderate doses of 4 to 8 mg appear highly effective. The MASLD trial data showed dramatic liver fat reduction even at moderate doses, though maximum liver fat clearance was observed at 12 mg.
Fat loss peptides in general show metabolic benefits that often precede significant scale changes. With retatrutide specifically, improvements in fasting glucose, HbA1c, triglycerides, and liver enzymes were observed within the first 12 to 16 weeks of treatment across all dose groups. If metabolic health is your primary goal, you may achieve excellent results at 4 to 8 mg without needing to push to the maximum dose.
Goal: fat loss with maximum muscle preservation
One concern with aggressive weight loss is lean mass loss. A substudy of the Phase 2 trial examining body composition found that retatrutide produced significant fat mass reduction with some accompanying lean mass loss, though the ratio was favorable compared to dietary restriction alone. At higher doses, the glucagon receptor activation may actually help preserve lean mass through increased protein turnover and growth hormone modulation, but the data on this is still emerging.
For researchers prioritizing muscle preservation, a moderate dose of 4 to 8 mg combined with adequate protein intake (1.0 to 1.2 grams per kilogram of body weight) and resistance training appears to optimize the fat-to-lean mass loss ratio. Higher protein intake and structured exercise may be more impactful for muscle preservation than dose selection alone.
The complete week-by-week titration protocol
Here is the detailed week-by-week breakdown of what to expect at each dose level, including what to monitor, when to be concerned, and how to decide whether to proceed to the next step. This goes beyond the standard retatrutide dose chart by adding practical decision points at each stage.
Weeks 1 through 4: 1 mg weekly
What is happening: Initial GLP-1 receptor engagement begins. Mild appetite suppression develops. Gastric emptying starts to slow. Your body is being introduced to triple receptor activation at the gentlest possible level.
What to monitor: Nausea severity and duration after each injection. Changes in appetite and food intake. Energy levels and sleep quality. Bowel habits including constipation or diarrhea. Injection site reactions.
Expected weight change: 1 to 3% of body weight over four weeks (roughly 2 to 6 pounds for a 200-pound person).
Decision point at week 4: If side effects were mild and resolved within 48 hours of each injection, proceed to 2 mg. If nausea or GI symptoms persisted beyond 48 hours or worsened with each weekly dose, extend 1 mg for an additional two to four weeks.
Weeks 5 through 8: 2 mg weekly
What is happening: GLP-1 effects strengthen noticeably. GIP receptor activation becomes more meaningful. Early glucagon receptor engagement begins. This is where many researchers first notice a significant shift in their relationship with food.
What to monitor: The same factors as the 1 mg phase, plus blood glucose patterns if you monitor them, hydration status (GLP-1 agonists can reduce thirst cues), and mood or energy shifts that may indicate metabolic transition.
Expected weight change: An additional 2 to 4% of body weight over these four weeks, bringing cumulative loss to approximately 3 to 7%.
Decision point at week 8: Most people tolerate the 1 to 2 mg transition well because it is a modest absolute increase. If tolerance is good, proceed to 4 mg. This next jump is the first significant escalation, so adequate tolerance at 2 mg is important before moving forward.
Weeks 9 through 12: 4 mg weekly
What is happening: This is where retatrutide starts to differentiate itself from single and dual-agonist compounds. All three receptor systems are now meaningfully engaged. Appetite suppression intensifies. Fat oxidation increases noticeably. Some researchers report feeling warmer, which reflects the thermogenic effect of glucagon receptor activation.
What to monitor: Intensified GI symptoms (the 2 to 4 mg jump is significant). Caloric intake, because strong appetite suppression can lead to underfeeding. Signs of dehydration. Fatigue patterns that might indicate inadequate nutrition rather than medication effects.
Expected weight change: An additional 3 to 5% of body weight, bringing cumulative loss to approximately 6 to 12%. This is where the results start getting exciting.
Decision point at week 12: If your goal is moderate weight loss (10 to 15%), and you are already approaching or within that range, you can stay at 4 mg as your maintenance dose. If you are targeting more aggressive results, assess tolerance carefully before moving to 8 mg. The 4 to 8 mg jump is the largest absolute increase in the standard protocol and carries the highest side effect risk.
Weeks 13 through 16: 8 mg weekly
What is happening: Full triple-agonist effects are now substantially engaged. Glucagon receptor activation is driving meaningful thermogenesis. The metabolic rate increase becomes clinically significant. Appetite suppression is typically strong to very strong. Some researchers report dramatic changes in food preferences, often shifting away from high-fat and high-sugar foods involuntarily.
What to monitor: This is the dose where nutritional adequacy becomes critical. Monitor protein intake (minimum 60 grams daily, ideally 80 to 100 grams). Watch for signs of hair thinning, which can indicate protein or caloric deficit. Monitor hydration aggressively. Track any mood changes or anxiety symptoms.
Expected weight change: An additional 3 to 5% of body weight, bringing cumulative loss to approximately 9 to 17%. At this point, the rate of weight loss is typically 1.5 to 2.5 pounds per week for most individuals.
Decision point at week 16: Many researchers find 8 mg to be their optimal maintenance dose. The clinical data showed 22.8% average weight loss at 48 weeks with 8 mg, which is comparable to the highest tirzepatide doses. If you are losing weight consistently, tolerating the dose well, and feeling good, 8 mg may be where you stay. If you want maximum results and are tolerating 8 mg without significant issues, proceed to 12 mg.
Weeks 17 onward: 12 mg weekly (if indicated)
What is happening: Maximum triple-agonist activation. Every receptor system is fully engaged at clinically studied levels. This is the dose that produced 24.2% average weight loss at 48 weeks in Phase 2 and up to 28.7% in the Phase 3 TRIUMPH-4 trial at 68 weeks.
What to monitor: Everything listed above, plus liver function markers if accessible, heart rate (glucagon can modestly increase heart rate), and overall quality of life. If the side effects at 12 mg are significantly impacting your daily function, reducing to 8 mg is not a failure. It is optimization.
Expected weight change: Continued loss of 1 to 2.5 pounds per week. Total projected weight loss at 48 weeks is approximately 20 to 26% of starting body weight for most individuals.
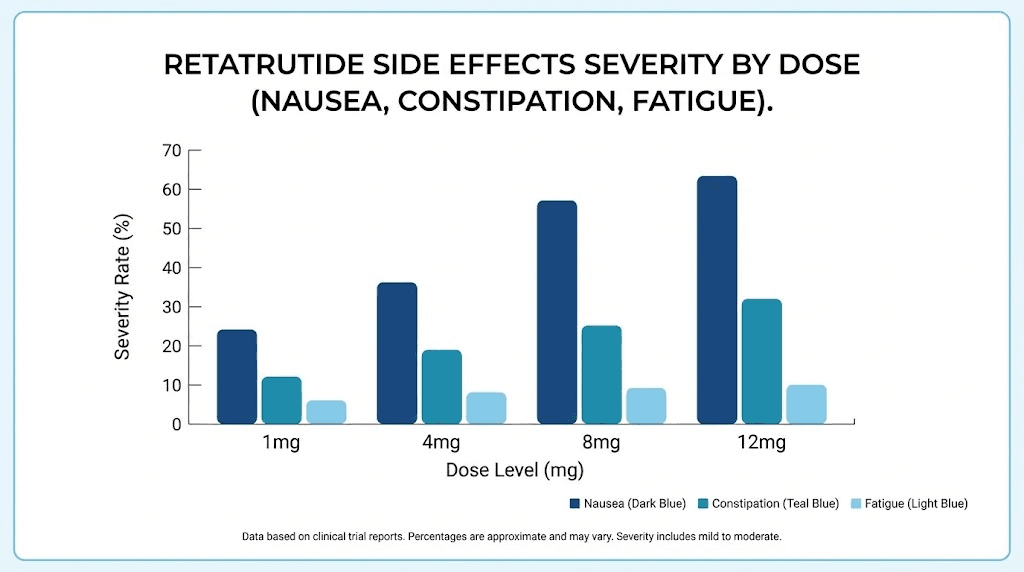
Side effects at each dose level and how to manage them
Understanding what to expect at each dose helps you differentiate normal adaptation from concerning symptoms. The side effect profile of retatrutide is primarily gastrointestinal, driven by all three receptor systems but most acutely by GLP-1 mediated gastric emptying delay.
Nausea: the most common side effect
Nausea affected approximately 27% of retatrutide users in clinical trials across all doses. The pattern is predictable. It is worst in the first 24 to 48 hours after each dose increase and typically improves with continued use at any given dose level. Most researchers report that nausea diminishes significantly by the second or third week at each new dose.
Management strategies that work include eating smaller, more frequent meals instead of large ones. Avoiding high-fat and greasy foods, which are harder to process with slowed gastric emptying. Staying well hydrated with small, frequent sips rather than large volumes. Timing your injection in the evening so the peak nausea window occurs during sleep. Ginger supplements or ginger tea before meals. And if necessary, medical options like ondansetron, which your prescriber can provide for severe cases.
The food guidance for GLP-1 agonists applies equally well to retatrutide. The principles of eating on these medications are consistent across the class.
Diarrhea and constipation
Retatrutide can cause either diarrhea or constipation, and some users experience both at different points in their protocol. Diarrhea is more common during dose escalation phases and usually resolves within one to two weeks. Constipation tends to develop more gradually and may persist, particularly at higher doses where gastric emptying is significantly slowed.
For diarrhea, the BRAT approach (bananas, rice, applesauce, toast) helps during acute episodes. Adequate hydration is critical. For persistent constipation, increasing fiber intake gradually, using magnesium supplements (400 to 600 mg daily), and maintaining adequate water intake (minimum 64 ounces daily) are first-line approaches. Stool softeners are a reasonable addition if dietary changes are insufficient.
Fatigue and energy fluctuations
Fatigue is common during the first two to three weeks at each new dose level. This results from the metabolic shift occurring as your body adapts to reduced caloric intake and enhanced fat oxidation. Think of it as your metabolism switching fuel sources. It takes time for the new energy pathways to fully optimize.
During this transition, maintaining adequate protein and calorie intake is critical. Many researchers make the mistake of eating too little when appetite suppression is strong, which creates a caloric deficit so aggressive that energy crashes are inevitable. Nutritional planning during GLP-1 agonist use is not optional. It is essential for tolerability and muscle preservation.
Hair thinning
Some users of GLP-1 and multi-agonist compounds report hair thinning, particularly during periods of rapid weight loss. This is almost always telogen effluvium, a stress-related hair shedding triggered by rapid metabolic change, caloric deficit, or protein inadequacy rather than the medication itself. The complete guide to GLP-1 and hair loss explains the mechanism and prevention strategies in detail.
Prevention focuses on three factors. Adequate protein intake (minimum 60 grams daily, ideally higher). Avoiding excessively rapid weight loss (no more than 2% of body weight per week sustained). And nutritional supplementation including biotin, zinc, and iron if blood levels indicate deficiency.
Injection site considerations
Retatrutide is administered subcutaneously once weekly. The recommended injection sites for retatrutide include the abdomen (at least 2 inches from the navel), front of the thigh, and back of the upper arm. Rotate sites with each injection to prevent lipodystrophy or irritation. The injection itself is typically well-tolerated, and most researchers report less injection site reaction with retatrutide than with some semaglutide formulations.
Reconstitution and preparation: getting the dose right
If you are working with lyophilized (freeze-dried) retatrutide, accurate reconstitution is essential for getting the correct dose. Errors in reconstitution are one of the most common reasons for unexpected side effects or underwhelming results. Too concentrated, and each injection delivers more than intended. Too dilute, and you are underdosing.
Standard reconstitution for a 10 mg vial
The most common research vial contains 10 mg of lyophilized retatrutide. The amount of bacteriostatic water you add determines your concentration. Here are the standard options.
Adding 2 mL of bacteriostatic water creates a concentration of 5 mg per mL. At this concentration, a 1 mg dose equals 0.2 mL (or 20 units on an insulin syringe). This is a good general-purpose concentration that makes measuring standard doses straightforward.
Adding 2.5 mL creates 4 mg per mL. A 1 mg dose equals 0.25 mL (25 units). This offers slightly easier measurement for doses that fall between standard unit markings.
Adding 3 mL creates approximately 3.33 mg per mL. A 1 mg dose equals 0.3 mL (30 units). This is the most common reconstitution volume because it makes a wider range of doses easy to measure accurately with standard insulin syringes.
The complete bac water guide for 10 mg retatrutide covers every scenario in detail, and the reconstitution chart provides a visual reference for all common vial sizes and water volumes. Our peptide reconstitution calculator can generate exact measurements for any vial size and target concentration.
Dose measurement with insulin syringes
Most researchers use standard U-100 insulin syringes (1 mL capacity, marked in units from 0 to 100). Each "unit" on the syringe equals 0.01 mL. If your concentration is 5 mg per mL, then 1 mg equals 20 units. If your concentration is 3.33 mg per mL, then 1 mg equals 30 units.
Here is a quick reference for common doses at the 5 mg/mL concentration.
Weekly dose | Volume to inject | Units on syringe |
|---|---|---|
1 mg | 0.2 mL | 20 units |
2 mg | 0.4 mL | 40 units |
4 mg | 0.8 mL | 80 units |
8 mg | 1.6 mL (split into 2 injections) | 80 units x 2 |
12 mg | 2.4 mL (split into 2-3 injections) | 80 units x 3 |
At higher doses, you will need to split injections because standard insulin syringes hold only 1 mL. This is normal. Many researchers prefer the 3.33 mg/mL concentration at higher doses because it reduces injection volume. The retatrutide peptide dosage chart shows all concentration and volume combinations.
Storage after reconstitution
Reconstituted retatrutide should be stored in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). The conservative shelf life for reconstituted peptide is 28 days, though some protocols document stability up to 60 days when stored properly. The peptide storage guide covers best practices for maintaining potency after reconstitution. Never freeze reconstituted peptide. Never leave it at room temperature for extended periods. And always check for discoloration or particles before each injection.
Retatrutide compared to semaglutide and tirzepatide: dose-for-dose
Understanding how retatrutide compares to other compounds helps contextualize the dosing question. If you have experience with semaglutide or tirzepatide, you already have a reference point for GLP-1 mediated effects. But retatrutide operates differently at every dose level because of the additional receptor systems.
Weight loss comparison at maximum doses
Compound | Max studied dose | Receptors targeted | Weight loss at max dose | Trial duration |
|---|---|---|---|---|
Semaglutide | 2.4 mg | GLP-1 | ~15 to 17% | 68 weeks |
Tirzepatide | 15 mg | GLP-1 + GIP | ~20 to 22% | 72 weeks |
Retatrutide | 12 mg | GLP-1 + GIP + Glucagon | ~24 to 29% | 48 to 68 weeks |
Retatrutide produces the most weight loss at maximum dose, and the gap is meaningful. The additional 4 to 7 percentage points compared to tirzepatide, and 9 to 12 percentage points compared to semaglutide, represent the contribution of glucagon receptor activation to energy expenditure and fat oxidation.
But maximum dose comparison does not tell the whole story. At moderate doses, the differences narrow. Retatrutide at 4 mg (17.1% weight loss) is comparable to tirzepatide at 10 to 15 mg. This suggests that the triple-agonist mechanism achieves equivalent results at lower relative doses, which has implications for side effect burden and cost.
Side effect comparison
The side effect comparison between semaglutide and tirzepatide shows that GI side effects are a class effect of GLP-1 agonism. Retatrutide adds the glucagon component, which can contribute to increased heart rate and potential blood glucose fluctuations in some individuals. However, the overall side effect profile in clinical trials was generally comparable to tirzepatide, with gastrointestinal events being the most common and dose-limiting factor.
The practical implication is that if you tolerated semaglutide or tirzepatide well, you will likely tolerate retatrutide at equivalent GLP-1 activity levels. The glucagon component introduces new considerations, but serious adverse events attributable specifically to glucagon receptor activation were rare in clinical trials.
Common dosing mistakes and how to avoid them
After analyzing hundreds of documented retatrutide protocols and community reports, several common mistakes consistently undermine results. Avoiding these gives you a significant advantage.
Mistake 1: escalating too fast because you feel fine
Feeling good at your current dose is not a reason to skip ahead. Some people take zero side effects at 1 mg and 2 mg as a signal that they can jump to 4 mg after just two weeks. This almost always backfires. The metabolic adaptation happening internally is not fully reflected in how you feel. Rushing escalation leads to more severe side effects at higher doses that could have been avoided with patience.
Mistake 2: chasing a number instead of listening to your body
Not everyone needs 12 mg. If you are losing weight consistently at 4 or 8 mg with manageable side effects and good quality of life, pushing to 12 mg for slightly faster results may not be worth the trade-off. The difference between 8 mg and 12 mg at 48 weeks was 1.4 percentage points in average weight loss. That is meaningful in a clinical trial population but may be negligible in your specific case if you are already responding well.
Mistake 3: not eating enough
Strong appetite suppression does not mean you should stop eating. Severe caloric restriction (under 800 to 1000 calories daily for extended periods) while on retatrutide leads to excessive muscle loss, hair shedding, fatigue, nutrient deficiencies, and metabolic adaptation that slows long-term progress. Even when you are not hungry, aim for minimum 1200 calories daily with at least 60 to 80 grams of protein. Think of eating on retatrutide as fueling your body, not feeding your appetite. The foods to avoid and diet planning guides for GLP-1 agonists apply here.
Mistake 4: inadequate hydration
GLP-1 receptor agonists can blunt thirst signals while simultaneously causing fluid loss through GI effects. Dehydration at any dose amplifies nausea, headaches, fatigue, and constipation. Minimum 64 ounces of water daily, and more if you experience diarrhea during escalation. Set a timer if needed. Do not rely on thirst to tell you when to drink.
Mistake 5: reconstitution errors
Using the wrong amount of bacteriostatic water throws off every dose measurement for the life of that vial. A 10% reconstitution error means every injection is 10% too high or too low. At higher doses like 8 or 12 mg, that error translates to meaningful clinical impact. The reconstitution chart and reconstitution calculator exist specifically to prevent this. Use them every time.
Mistake 6: comparing your timeline to others
Individual response to retatrutide varies enormously. Some people lose 5% of body weight in the first month. Others take 8 to 10 weeks to see the same result. Metabolic rate, starting body composition, hormonal status, stress levels, sleep quality, and countless other factors influence your personal response curve. The Phase 2 data shows averages, and averages hide enormous individual variation. Focus on your trend over weeks, not your daily scale readings.
How long should you stay on retatrutide
Duration of use is closely tied to dosing decisions. The Phase 2 trial ran for 48 weeks, and the Phase 3 TRIUMPH trials extend to 68 weeks and beyond. The data we have suggests that weight loss continues throughout the treatment period, with the rate gradually slowing as you approach a new equilibrium.
The 48-week data point
At 48 weeks, participants in the 12 mg group had lost an average of 24.2% of body weight and the trajectory had not fully plateaued. This suggests that longer treatment durations may produce additional weight loss beyond what the Phase 2 data captured. The TRIUMPH-4 Phase 3 data at 68 weeks showed 28.7% average weight loss, confirming that extended treatment does yield additional benefit.
What happens when you stop
Like all GLP-1 agonist compounds, discontinuing retatrutide results in some weight regain. The exact magnitude of regain with retatrutide has not been published in detail yet, but the general pattern from semaglutide and tirzepatide studies shows that approximately one-third to two-thirds of lost weight returns within 12 months of discontinuation, particularly without lifestyle modifications. The duration guide for semaglutide provides useful context for understanding this pattern, as the underlying biology is similar across the class.
This means that the dosing question is not just "how much" but "for how long." A researcher who reaches their goal weight at month 8 needs to consider whether to continue at a maintenance dose, reduce to a lower maintenance level, or taper off with the understanding that some regain is likely.
Maintenance dosing
While definitive maintenance dosing data for retatrutide is still forthcoming, the precedent from other GLP-1 agonists suggests that a reduced maintenance dose may sustain most of the weight loss while minimizing side effects and cost. A researcher who achieved their goal at 12 mg might maintain on 4 to 8 mg long-term. This approach has not been formally studied for retatrutide but aligns with the pharmacological logic and clinical precedent from the broader class. SeekPeptides provides detailed protocol guidance for transitioning between treatment and maintenance phases.
Special considerations that affect your dose
Beyond body weight and goals, several specific factors can influence how much retatrutide you should take and how you should approach the escalation.
Type 2 diabetes
Retatrutide has been studied in people with type 2 diabetes in a separate Phase 2 trial published in The Lancet. The dosing framework is similar but with additional glycemic monitoring requirements. The combination of GLP-1, GIP, and glucagon receptor activation produces complex effects on blood glucose. GLP-1 and GIP tend to lower glucose, while glucagon can raise it. In practice, the net effect in the clinical data was improved glycemic control, but individual responses vary.
If you have type 2 diabetes, work with a prescriber who can monitor HbA1c, fasting glucose, and post-prandial glucose throughout the titration. Hypoglycemia is a potential concern, particularly if you are on concurrent insulin or sulfonylurea medications. Dose adjustments to those medications may be needed as retatrutide improves insulin sensitivity.
Liver health
The MASLD trial data suggests retatrutide has particular benefits for liver health, with dramatic reductions in liver fat content. If you have fatty liver disease or elevated liver enzymes, retatrutide at moderate to high doses (8 to 12 mg) may provide both weight loss and direct hepatic benefits through glucagon receptor mediated fat oxidation in the liver. However, liver function should be monitored during treatment, particularly during dose escalation when the metabolic demands on the liver increase.
Gallbladder considerations
Rapid weight loss from any cause increases the risk of gallstone formation and gallbladder events. This is not unique to retatrutide but is relevant given the aggressive weight loss it produces. If you have a history of gallbladder disease or gallstones, discuss this with your prescriber before starting. The risk is highest during periods of rapid weight loss, which typically occur in the first 12 to 24 weeks of treatment at higher doses.
Thyroid considerations
GLP-1 receptor agonists carry a warning about thyroid C-cell tumors based on rodent studies. While this has not been confirmed in humans, retatrutide, like semaglutide and tirzepatide, is generally not recommended for people with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2. This is a prescriber-level decision, not a dose-level decision, but it influences whether retatrutide is appropriate for you at all.
Concurrent medications
Because retatrutide slows gastric emptying, it can affect the absorption of oral medications. If you take medications that require precise timing or have narrow therapeutic windows (such as thyroid medications, anticoagulants, or certain antibiotics), discuss the implications with your prescriber. Dose adjustments or timing changes may be needed for concurrent medications as your retatrutide dose increases.
Putting it all together: your personalized dosing decision
After absorbing all of this information, here is how to synthesize it into a practical dosing plan for your specific situation.
Step 1: Determine your starting point
Everyone starts at 1 mg. No exceptions. Regardless of body weight, prior experience, or impatience. The starting dose is not where results happen. It is where safety is established.
Step 2: Choose your escalation pace
Standard pace is four weeks per dose level. This works for most people. If you have no prior GLP-1 experience and weigh under 180 pounds, consider five to six weeks per level. If you have extensive prior experience with tirzepatide at high doses and weigh over 220 pounds, four weeks per level is appropriate, possibly three weeks at the 1 mg level only.
Step 3: Identify your likely maintenance dose
Based on your goals and body weight:
Moderate weight loss, lighter body weight: 4 to 8 mg
Moderate weight loss, heavier body weight: 8 mg
Aggressive weight loss, any body weight: 8 to 12 mg
Metabolic health focus: 4 to 8 mg
Maximum fat loss with muscle preservation: 4 to 8 mg with high protein intake
Step 4: Establish your monitoring protocol
At minimum, track weekly body weight (same time, same conditions), side effect type and severity, daily caloric and protein intake, hydration status, and energy levels. If possible, also track waist circumference weekly, body composition monthly (through DEXA or bioimpedance), and blood markers quarterly (fasting glucose, HbA1c, lipid panel, liver enzymes). The GLP-1 plotter can help you visualize your progress over time.
Step 5: Adjust based on response
The initial plan is not the final plan. If you reach 4 mg and are losing 2 pounds per week consistently with minimal side effects, staying at 4 mg is a perfectly valid choice. If you reach 8 mg and the side effects are making daily life difficult, dropping back to 4 mg and reassessing is not failure. It is intelligent protocol management.
The researchers who get the best long-term results with retatrutide are not the ones who race to 12 mg fastest. They are the ones who find the dose that produces consistent, tolerable, sustainable progress and stay there.
For researchers who want personalized protocol guidance beyond what a general guide can provide, SeekPeptides offers comprehensive dosing tools, dosage calculators, and access to detailed community protocols from thousands of documented experiences. The membership provides the granular, individual-level guidance that no blog post can fully replicate.
Frequently asked questions
What is the starting dose of retatrutide for everyone?
The starting dose is 1 mg subcutaneously once weekly, regardless of body weight, prior experience, or goals. Clinical trials consistently used 1 mg (or occasionally 2 mg) as the entry point, and starting higher significantly increases the risk of gastrointestinal side effects. Even researchers transitioning from high-dose tirzepatide should start at 1 mg because the glucagon receptor component is entirely new to their system.
Can I take retatrutide every day instead of once a week?
No. Retatrutide is designed for once-weekly subcutaneous injection. Its molecular structure includes a fatty diacid modification that extends the half-life to approximately six days, allowing weekly dosing. Taking it more frequently would lead to dangerous accumulation. The dose schedule should always be maintained at seven-day intervals, give or take 24 hours.
What happens if I miss a retatrutide dose?
If you miss a dose and it has been less than 4 days since you were supposed to inject, take the missed dose as soon as possible. If more than 4 days have passed, skip the missed dose and resume your regular schedule at the next planned injection day. Do not double up doses to compensate. The long half-life provides some buffer, but consistent weekly dosing produces the best results.
Is 12 mg always better than 8 mg?
Not necessarily. The 12 mg group lost an average of 24.2% body weight at 48 weeks versus 22.8% in the 8 mg group, a difference of 1.4 percentage points. For some individuals, the additional side effect burden at 12 mg is not worth that incremental improvement. If 8 mg produces consistent weight loss with good tolerability, it is an excellent maintenance dose. The decision depends on your personal risk-benefit assessment.
How long does it take for retatrutide to start working?
Most people notice appetite changes within the first week at 1 mg. Measurable weight loss typically begins by week 2 to 3. Significant weight loss becomes apparent by weeks 8 to 12 as the dose reaches the 4 mg range. The full thermogenic and metabolic effects of glucagon receptor activation become most pronounced at 8 to 12 mg, which is typically reached by weeks 13 to 17. The timeline for retatrutide to kick in provides more detailed week-by-week expectations.
Can I switch from semaglutide or tirzepatide to retatrutide directly?
Yes, though the transition requires starting retatrutide at 1 mg regardless of your previous dose. Most protocols suggest taking your last dose of semaglutide or tirzepatide and starting retatrutide one week later. There may be a brief gap in appetite suppression as you begin at the lower retatrutide dose, which is temporary. The switching guide between GLP-1 compounds covers transition strategies in detail.
Does body weight affect how much retatrutide I should take?
Body weight influences the pharmacokinetic exposure at each dose level, meaning lighter individuals receive a higher per-kilogram dose than heavier individuals at the same absolute milligram amount. Lighter people (under 150 lbs) may find that 4 to 8 mg is sufficient for significant results, while heavier individuals (over 250 lbs) typically benefit from escalating to 12 mg. The weight loss dosage chart provides guidance by weight range.
Is retatrutide FDA approved?
No. Retatrutide is currently in Phase 3 clinical trials through the TRIUMPH program conducted by Eli Lilly. Phase 3 results are expected throughout the remainder of the TRIUMPH program, with potential FDA approval estimated for 2027 at the earliest. Any retatrutide available outside of clinical trials is sourced from research suppliers and has not undergone regulatory review.
External resources
PubMed Central: Systematic Review and Meta-Analysis of Retatrutide Trials
Nature Medicine: Retatrutide for Metabolic Dysfunction-Associated Liver Disease
For researchers serious about optimizing their retatrutide protocol, SeekPeptides offers the most comprehensive resource available, with evidence-based dosing guides, proven escalation protocols, detailed calculators, and a community of thousands who have navigated these exact dosing decisions.
In case I do not see you, good afternoon, good evening, and good night. May your doses stay precise, your escalation stay patient, and your results stay consistent.