Feb 20, 2026

You just had a baby. The weight is not coming off. And somewhere between the midnight feedings and the pediatrician visits, you heard about tirzepatide. Maybe a friend mentioned it. Maybe your doctor brought it up. Maybe you saw it on social media, sandwiched between postpartum recovery tips and nursery decor ideas.
Here is the problem.
Most articles about tirzepatide and breastfeeding give you one of two answers. Either "absolutely not, it is too dangerous" or "probably fine, go for it." Neither answer is complete. Neither reflects what the research actually shows. And when you are making a decision that affects both your body and your baby, incomplete information is not good enough.
The truth sits somewhere more nuanced. A manufacturer-sponsored study of 11 lactating women found tirzepatide was undetectable in 164 of 171 breast milk samples. The remaining seven samples contained less than 0.02% of the maternal dose. That sounds reassuring, and in many ways it is. But the study only tested a single 5 mg dose, not the escalating weekly regimen most people actually follow. And it did not measure long-term effects on milk supply, infant growth, or nutritional composition of the milk itself.
This guide breaks down everything the research has found so far, what it has not studied yet, what the real risks are (hint: they are probably not what you think), and how to make an informed decision with your healthcare provider. Whether you are considering tirzepatide for weight loss, managing dosing protocols, or simply trying to understand the safety profile during lactation, every claim in this article comes from published data, clinical guidance, or expert medical opinion.
SeekPeptides tracks every development in peptide and GLP-1 research for breastfeeding mothers, and this guide reflects the most current evidence available.
How tirzepatide works and why breastfeeding changes the equation
Tirzepatide is not a simple weight loss pill. It is a dual-action peptide that activates two separate receptors in your body: GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1). This dual mechanism is what makes it different from semaglutide, which only targets GLP-1. Understanding how peptides work at this fundamental level matters because the mechanism directly affects whether and how the drug might reach your baby.
The dual GIP/GLP-1 mechanism explained
When you inject tirzepatide subcutaneously, following proper peptide injection techniques, it binds to GIP and GLP-1 receptors throughout your body. These receptors sit primarily in the pancreas, gut, and brain. The pancreatic effects enhance insulin secretion and reduce glucagon release. The gut effects slow gastric emptying, which is why you feel full longer and why understanding tirzepatide metabolic effects matters. The brain effects reduce appetite signals in the hypothalamus, creating the powerful appetite suppression that makes GLP-1 medications so effective for weight loss.
This combination produces more weight loss than GLP-1 alone. Clinical trials showed tirzepatide at the highest dose (15 mg weekly) produced an average of 22.5% body weight reduction, significantly more than semaglutide results at comparable timeframes. The most effective peptides for weight loss all work through GLP-1 pathways, but tirzepatide dual action sets it apart. For postpartum mothers carrying significant weight, that efficacy is appealing. But efficacy during breastfeeding requires a different kind of evaluation than efficacy alone.
Why the postpartum body is different
Breastfeeding changes everything about how your body processes medication. Your metabolic rate is elevated. You burn an additional 450 to 500 calories per day producing milk. Your hormonal profile is fundamentally altered, with prolactin and oxytocin driving milk production in ways that interact with appetite, metabolism, and energy balance. Women navigating peptide-based weight management understand that hormonal context matters enormously, and postpartum lactation represents one of the most complex hormonal states your body will experience.
Tirzepatide suppresses appetite. That is its job.
But during lactation, suppressed appetite can cascade into reduced caloric intake, which can cascade into reduced milk supply, which can cascade into inadequate infant nutrition. The drug itself may not cross into milk in meaningful amounts, but its effects on your eating behavior absolutely affect your baby. This indirect pathway is what most simplified articles completely miss, and it is arguably the bigger concern than direct drug transfer.
Your body is already in a catabolic state during breastfeeding. Adding a medication that further reduces caloric intake amplifies that catabolism. The foods you avoid on tirzepatide and the foods you should eat on tirzepatide become even more critical when you are also feeding another human being from your own nutritional reserves.
The molecular size advantage
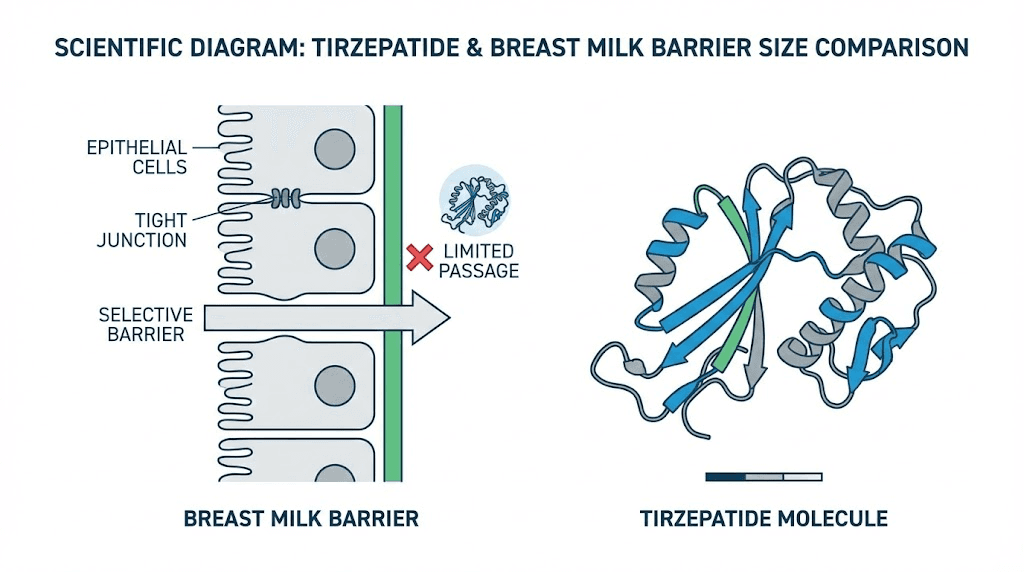
Here is where the science becomes somewhat reassuring. Tirzepatide has a molecular weight of approximately 4,814 Daltons. For context, most drugs that easily transfer into breast milk weigh less than 800 Daltons. The general pharmacological principle is straightforward: bigger molecules have a harder time crossing biological barriers, including the mammary epithelium that separates your bloodstream from your milk ducts.
At 4,814 Daltons, tirzepatide is roughly six times larger than the typical threshold for easy milk transfer. This does not mean zero transfer. It means very limited transfer, which is exactly what the studies have confirmed. Understanding what peptides are and how their size affects bioavailability helps explain why researchers expected low milk concentrations even before the studies began.
What the breast milk studies actually found
Two key datasets exist for tirzepatide and breast milk. Both come from manufacturer-sponsored research, which is worth noting, but also represents the most rigorous data available. Independent confirmation is still needed, but these findings form the foundation of current clinical guidance.
Study one: single-dose analysis in 11 women
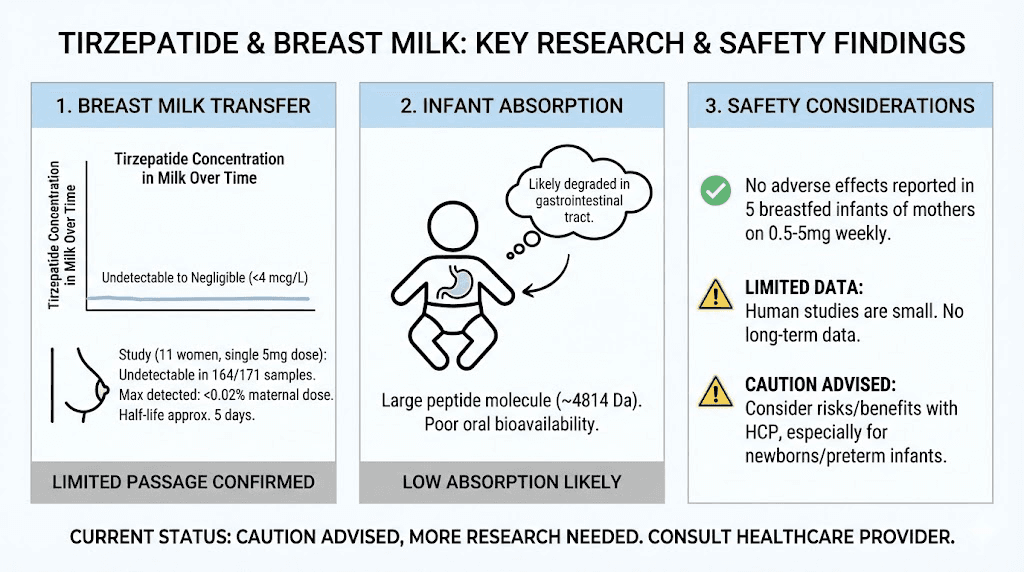
Eli Lilly conducted a study giving 11 healthy lactating women a single 5 mg subcutaneous dose of tirzepatide. Researchers then collected milk samples repeatedly over a 28-day period, yielding 171 total samples.
The results were striking. Of those 171 samples, 164 showed tirzepatide concentrations below the detectable limit of 4 mcg/L. That is 95.9% of all samples coming back as essentially zero. The remaining seven samples did show measurable levels, but the cumulative amount across all seven represented less than 0.02% of the maternal dose.
Less than 0.02%.
To put that number in perspective, lactation pharmacologists generally consider a relative infant dose below 10% to be compatible with breastfeeding. Tirzepatide came in at roughly 500 times below that threshold on a single-dose basis. The standard dosing protocols for tirzepatide start at 2.5 mg and escalate gradually, so this 5 mg test dose actually exceeds what most people take initially.
The measurable concentrations appeared approximately five days after the dose, which aligns with the timeline of tirzepatide activity in the body and its roughly five-day half-life.
Study two: weekly dosing in five women
A second dataset followed five women receiving tirzepatide at weekly doses ranging from 0.5 to 5 mg, which better reflects real-world use. Researchers collected multiple milk samples from each participant.
Every single sample came back with unquantifiable levels, below the limit of detection of 0.7 mcg/L. Not one sample from any of the five women showed measurable tirzepatide in breast milk during ongoing weekly treatment.
All five women continued breastfeeding throughout the study. None reported adverse effects in their infants attributable to tirzepatide. The babies fed normally. They grew normally. No gastrointestinal symptoms, no feeding difficulties, no observable changes.
These findings are encouraging. They are also limited. Five women over a relatively short period does not capture every possible scenario. Higher doses (10 mg and 15 mg) were not tested in these breastfeeding-specific studies. And the researchers did not measure milk composition changes, milk volume, or long-term infant developmental outcomes.
The LactMed database assessment
The National Institutes of Health maintain LactMed, a peer-reviewed database of drugs and lactation. Their assessment of tirzepatide reflects the available data: "If a mother requires tirzepatide, it is not a reason to discontinue breastfeeding." They add the caveat that caution should be used, "especially while nursing a newborn or preterm infant, until more data become available."
This is a carefully worded position. It does not say tirzepatide is definitively safe. It says the evidence does not support stopping breastfeeding solely because of tirzepatide use. The distinction matters. It acknowledges both the reassuring milk transfer data and the limitations of the current evidence base.
The clinical trial that could change everything
ClinicalTrials.gov lists study NCT05978713, a dedicated trial examining tirzepatide specifically in healthy lactating women. This Eli Lilly-sponsored study will provide more robust data on breast milk concentrations, infant exposure, and safety endpoints. When results are published, they will significantly expand our understanding of tirzepatide use during breastfeeding. Until then, the two smaller datasets above remain the primary evidence.
Understanding why so little tirzepatide reaches breast milk
The near-zero transfer of tirzepatide into breast milk is not random luck. It follows predictable pharmacological principles that apply to most large peptide molecules. Understanding these principles, much like understanding the basics of peptide dosage calculations, helps explain why lactation experts view tirzepatide differently than small-molecule drugs.
Molecular weight and the mammary barrier
Your mammary epithelium acts as a selective filter between blood and milk. Small molecules (under 200 Daltons) cross freely through gaps between cells. Medium molecules (200-800 Daltons) cross with varying efficiency depending on their chemical properties. Large molecules (over 800 Daltons) face significant barriers.
Tirzepatide at 4,814 Daltons is not just large. It is massive by drug standards. Compare this to common medications known to transfer well into breast milk: ibuprofen is 206 Daltons, acetaminophen is 151 Daltons, and even metformin is only 129 Daltons. Tirzepatide is 25 to 37 times larger than these freely-transferring drugs. For perspective, even collagen peptides used during pregnancy are broken down into smaller fragments that absorb differently than intact therapeutic peptides like tirzepatide.
The safety profile of peptides in general benefits from this size advantage. Most injectable peptides are large enough to face significant barriers when crossing biological membranes. This is why oral tirzepatide requires special formulation strategies, because the molecule does not absorb well through the gut on its own.
Protein degradation in the infant gut
Even the tiny amount of tirzepatide that might reach breast milk faces another barrier: the infant digestive system. Tirzepatide is a peptide, which means it is essentially a small protein chain. When an infant swallows breast milk containing trace peptide molecules, those molecules encounter stomach acid and digestive enzymes (pepsin, trypsin, chymotrypsin) that break proteins into individual amino acids.
Oral bioavailability of tirzepatide is practically nil without specialized absorption enhancers. This is the entire reason tirzepatide is given by injection rather than as a pill, and why developing tirzepatide tablets versus injections required significant pharmaceutical innovation. An infant swallowing trace amounts in breast milk would be exposed to even less than what an adult would fail to absorb orally.
There is one important exception.
Premature infants and newborns in the immediate neonatal period (first few days of life) have greater intestinal permeability. Their gut barrier is not yet fully developed. This "leaky gut" phase allows larger molecules to cross into the bloodstream more easily than in older infants. This is why every medical source adds extra caution for preterm and newborn nursing.
Protein binding and distribution
Tirzepatide is highly protein-bound in the bloodstream, with approximately 99% binding to albumin. Only the unbound fraction of a drug can cross into breast milk. With 99% bound and unavailable for transfer, only about 1% of circulating tirzepatide is even theoretically capable of reaching the mammary epithelium, and then it still faces the molecular size barrier.
This double protection, high protein binding plus large molecular size, explains why the actual measured milk concentrations are so extraordinarily low. It also explains why the appetite-suppressing effects of tirzepatide are so strong in the mother (the drug stays active in the bloodstream) while transfer to the infant is minimal.
The infant safety question: what we know and what we do not
Milk transfer data tells us how much drug reaches the baby. But "how much" is only half the question. The other half is "what does that amount do?"
What the current data shows about infant outcomes
Across both studies (16 total mother-infant pairs), no adverse effects were reported in any breastfed infant. No gastrointestinal symptoms. No feeding refusal. No growth abnormalities. No behavioral changes. Every infant continued feeding normally throughout the study period. This stands in contrast to the side effects mothers themselves experience, which can include muscle pain, headaches, and fatigue, none of which appeared in the nursing infants.
This is reassuring but incomplete. Sixteen infants over relatively short study periods cannot detect rare adverse events or subtle long-term effects. The studies were not designed to measure developmental milestones, cognitive outcomes, or metabolic programming effects that might only become apparent months or years later.
Theoretical risks to consider
GLP-1 and GIP receptors exist in the infant pancreas, gut, and developing brain. If tirzepatide were to reach the infant bloodstream in meaningful amounts (which current data suggests it does not), it could theoretically affect:
Insulin regulation: GLP-1 receptor activation enhances insulin secretion, which could affect infant blood sugar levels
Gastric motility: Slowed stomach emptying could reduce feeding volume or increase reflux
Appetite regulation: The developing hypothalamus is sensitive to metabolic signals during early life
Gut development: GIP and GLP-1 play roles in intestinal growth and maturation
These are theoretical concerns, not observed effects. The extremely low transfer levels make any of these outcomes unlikely at the doses studied. But they explain why experts recommend continued monitoring and why some clinicians prefer waiting until the infant is eating solid foods before the mother starts tirzepatide.
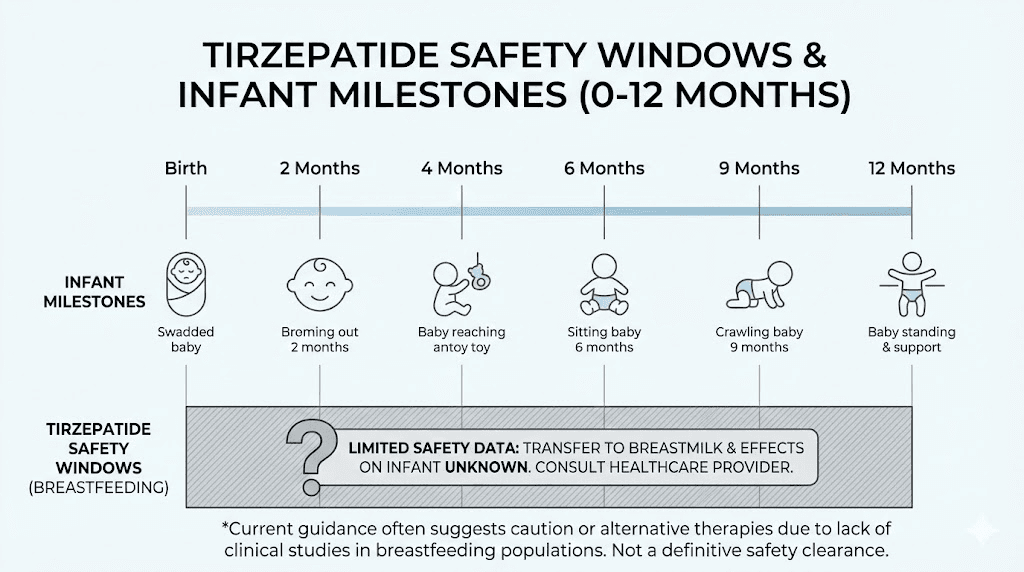
Risk stratification by infant age
Not all breastfeeding situations carry equal risk. Medical experts typically stratify the risk based on infant age and development:
Highest caution (0-4 weeks): Newborns have immature gut barriers, immature liver metabolism, and are exclusively dependent on breast milk. Most experts recommend avoiding tirzepatide entirely during this period. Any medication decisions in the immediate postpartum period should also consider the relationship between tirzepatide and reproductive health.
Moderate caution (1-6 months): The infant gut has matured somewhat but breastfeeding remains the primary nutrition source. If tirzepatide is medically necessary, close monitoring is recommended. Starting with the lowest possible dose via microdosing may reduce any theoretical risk.
Lower caution (6+ months): Once solid foods are introduced, the infant no longer depends exclusively on breast milk for nutrition. Breast milk volumes typically decrease, and the infant gut barrier is more mature. Many experts consider this the safest window to initiate tirzepatide if the mother chooses to continue breastfeeding.
Milk supply: the concern most people miss entirely
Here is what frustrates lactation consultants about the tirzepatide-breastfeeding conversation. Everyone focuses on whether the drug reaches the milk. Almost nobody talks about whether the drug affects how much milk you make.
This matters more than you might think.
How milk production actually works
Breast milk production follows a supply-and-demand model, but it requires adequate raw materials. Your body needs calories, fluids, and specific nutrients to manufacture milk. The average breastfeeding mother produces 750 to 800 mL of milk per day, requiring approximately 500 additional calories beyond her baseline metabolic needs.
Prolactin drives milk synthesis. Oxytocin triggers the let-down reflex. Tirzepatide does not directly target either hormone. There is no evidence that it interferes with the hormonal signaling that controls milk production.
But it does not need to target those hormones directly to cause problems.
The indirect pathway: appetite suppression and caloric deficit
Tirzepatide reduces appetite. Dramatically. Most users report significant decreases in hunger, food cravings, and portion sizes within the first few weeks of treatment. For someone trying to lose weight, this is the entire point. For someone who also needs to consume 500 extra calories daily to support milk production, this creates a dangerous tension.
If a breastfeeding mother on tirzepatide eats 1,200 calories per day instead of the 2,000 to 2,500 she needs, her body will prioritize survival over milk production. The result: gradually declining milk supply, reduced milk fat content, and potentially inadequate nutrition for the baby. Understanding proper nutrition planning on tirzepatide becomes absolutely essential during breastfeeding.
This is not hypothetical. Caloric restriction has well-documented effects on lactation. Studies show that severe caloric restriction (below 1,500 calories daily) can reduce milk volume by 15 to 25% within days. Even moderate restriction can alter the fatty acid composition of breast milk over weeks.
The tirzepatide meal planning resources take on new importance for nursing mothers. Every calorie counts not just for maternal nutrition but for infant nutrition as well.
What experts recommend for protecting supply
Lactation pharmacologists and registered dietitians who work with postpartum mothers on GLP-1 medications consistently emphasize three priorities:
Minimum caloric intake: Never drop below 1,800 calories per day while breastfeeding, regardless of how suppressed your appetite feels. Many practitioners recommend 2,000 to 2,300 calories as a safer floor. Use a structured eating plan designed for tirzepatide use but adjust the caloric targets upward for lactation.
Nutrient density over volume: When your appetite is suppressed, every bite needs to count. Prioritize protein (aiming for 1.2 to 1.5 grams per kilogram of body weight), healthy fats (essential for milk fat content and infant brain development), and complex carbohydrates. The supplements recommended alongside tirzepatide should include a comprehensive postnatal vitamin during breastfeeding.
Scheduled eating: Do not wait until you feel hungry. Set meal and snack times and eat on schedule even when tirzepatide has reduced your appetite signals. This is the opposite of how most people approach weight loss, but lactation demands it.
Hydration: the non-negotiable factor
Breast milk is approximately 87% water. An obvious fact with enormous implications for mothers taking tirzepatide.
Why tirzepatide and dehydration create a dangerous combination
GLP-1 medications can dull the sensation of thirst. Some users report not feeling thirsty at all during the first weeks of treatment, even when their body needs fluid. Combine this with the common gastrointestinal side effects of tirzepatide, including constipation, nausea, and occasional vomiting, and dehydration becomes a real risk.
For a breastfeeding mother, dehydration directly impacts milk volume. Your body cannot produce milk without adequate water. A reduction of even 10 to 15% in fluid intake can measurably reduce milk output within 24 to 48 hours.
Practical hydration targets
Breastfeeding mothers typically need 3.0 to 3.5 liters (about 100 to 120 ounces) of fluid daily. On tirzepatide, this number should be treated as a minimum, not a target. If you experience any GI side effects, increase fluid intake further to compensate for losses.
Practical strategies include keeping a large water bottle within reach during every feeding session, setting hourly hydration reminders on your phone, and tracking fluid intake the same way you might track medication doses. Some mothers find that protein shakes formulated for GLP-1 users serve double duty by providing both hydration and essential nutrients in a format that is easier to consume when appetite is suppressed.
The relationship between GLP-1 medication fatigue and dehydration is well documented. Postpartum mothers already dealing with sleep deprivation cannot afford additional fatigue from inadequate fluid intake.
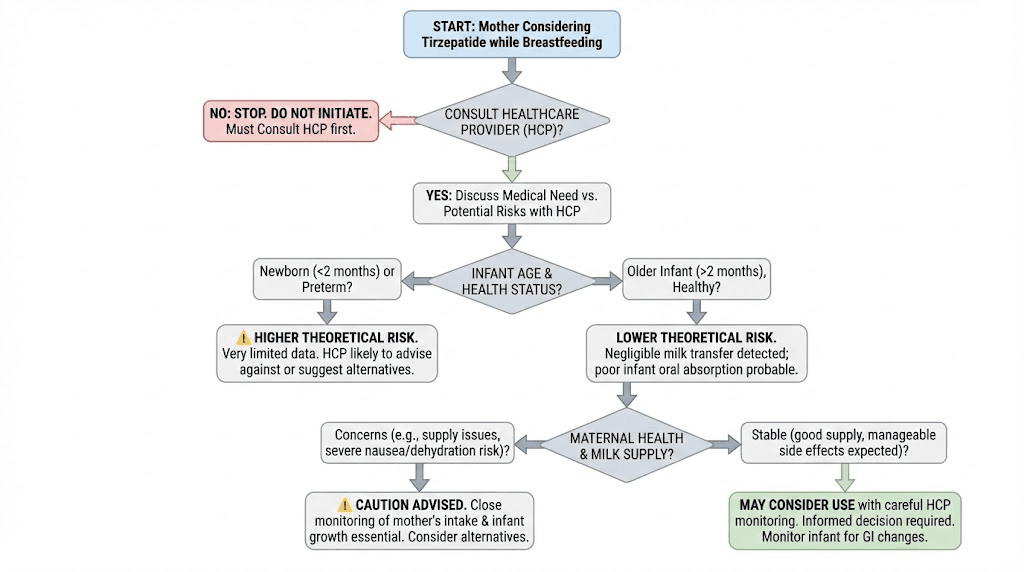
Timing: when experts say it is safer to start
If you have decided with your healthcare provider that tirzepatide is appropriate for your postpartum weight management, timing the start date matters significantly. The window you choose affects both your risk profile and your likelihood of maintaining an adequate milk supply.
The immediate postpartum period (0-6 weeks): wait
No medical organization recommends starting tirzepatide in the first six weeks postpartum. Your body is recovering from pregnancy and delivery. Your milk supply is being established. The infant gut barrier is at its most permeable. And your hormonal landscape is shifting dramatically.
Even outside of breastfeeding considerations, most physicians recommend waiting at least six weeks after delivery before starting any GLP-1 medication. This allows for postpartum recovery, baseline weight stabilization (some weight loss occurs naturally in the first weeks), and establishment of breastfeeding patterns.
The exclusive breastfeeding period (6 weeks to 6 months): proceed with caution
This is the gray zone where clinical judgment becomes critical. The breast milk transfer data is reassuring, but the infant is still exclusively dependent on breast milk for nutrition. If you start tirzepatide during this period, experts recommend:
Starting at the lowest dose (2.5 mg weekly) and escalating more slowly than standard protocols
Working with a registered dietitian to ensure caloric intake remains adequate
Monitoring milk supply closely through infant weight checks and feeding frequency
Watching the baby for any changes in feeding behavior, stool patterns, or temperament
Understanding how to measure the 2.5 mg starting dose accurately is critical during breastfeeding. Dosing errors carry higher stakes when you are also feeding an infant. A tirzepatide dosage calculator can help ensure precision.
After solid food introduction (6+ months): the recommended window
Most lactation pharmacologists and maternal health experts recommend waiting until the baby is eating solid foods before starting tirzepatide. By six months, several protective factors align:
The infant gut barrier has matured significantly. The baby is no longer exclusively dependent on breast milk for nutrition. Breast milk volumes are naturally decreasing. And the mother has had time to establish a stable milk supply that is more resilient to minor disruptions.
This is a recommendation, not a requirement. Individual circumstances, including the severity of maternal obesity, presence of obesity-related comorbidities, and the specific breastfeeding situation, may shift the risk-benefit calculation. A mother with type 2 diabetes and a BMI over 40 may have stronger medical reasons to start earlier than a mother seeking purely cosmetic weight loss.
Dosing considerations specific to breastfeeding mothers
If you and your provider decide to proceed with tirzepatide during lactation, the dosing approach should differ significantly from standard weight loss protocols. The goal shifts from maximum weight loss to adequate weight management with minimal risk.
Starting dose and escalation timeline
Standard tirzepatide protocols begin at 2.5 mg weekly for four weeks, then escalate to 5 mg, and eventually up to 10 or 15 mg based on response and tolerability. For breastfeeding mothers, most clinicians recommend a more conservative approach.
Start at 2.5 mg. Stay there for at least six to eight weeks instead of the standard four. Evaluate how your body responds, how your milk supply holds, and how the baby is growing before considering any increase. Some mothers find that 2.5 mg provides enough appetite regulation to support gradual weight loss without the intense suppression of higher doses.
If escalation is needed, move to 5 mg and again extend the evaluation period. The standard dose charts do not account for breastfeeding, so treat published escalation timelines as minimums, not targets. A dosage calculator can help determine the exact unit measurement for your specific concentration.
Many breastfeeding mothers choose to cap their dose at 5 mg, forgoing the higher doses entirely until nursing ends. The compounded dosage options available today make this flexibility possible, as some compounding pharmacies offer concentrations that simplify low-dose administration.
Injection timing and breastfeeding
Some mothers ask whether timing their injection relative to nursing sessions matters. Because of tirzepatide long half-life (approximately five days), the timing of your weekly injection has no meaningful impact on milk concentrations. Drug levels remain essentially constant throughout the week. Unlike short-acting medications where timing matters, you can inject on whatever day and time works best for your schedule.
Proper injection site selection and injection technique remain important for absorption consistency. Using the right injection equipment and following established GLP-1 injection protocols ensures consistent delivery. Inconsistent absorption could lead to variable drug levels, which is suboptimal for any user but particularly for breastfeeding mothers who want predictable, steady-state concentrations.
Compounded versus brand-name considerations
The breast milk studies used pharmaceutical-grade tirzepatide from Eli Lilly. If you are using compounded tirzepatide from pharmacies like Empower or other compounding sources, be aware that additional ingredients may be present. Glycine is a naturally occurring amino acid considered safe during breastfeeding. Vitamin B12 is also safe and potentially beneficial for postpartum recovery. Niacinamide (vitamin B3) is generally safe but has less specific breastfeeding data.
The overall safety profile of injectable peptides depends on both the active ingredient and the formulation. Discuss your specific compound formulation with your prescriber and lactation consultant. Resources like the SeekPeptides dosage calculator can help you verify exact measurements for any formulation. Some mothers prefer brand-name products during breastfeeding specifically because the safety data applies directly, while others find that the cost advantages of compounded tirzepatide make the minor uncertainty about excipients an acceptable trade-off. SeekPeptides members can access formulation comparison guides that detail the differences between major compounding pharmacies.
Why pump and dump does not work with tirzepatide
If you are familiar with alcohol and breastfeeding, you might know the "pump and dump" strategy: pump milk, throw it away, and wait for the substance to clear before nursing again. Some mothers wonder if the same approach works for tirzepatide.
It does not. The pharmacokinetics make it impossible.
The five-day half-life problem
Tirzepatide has a half-life of approximately five days. That means five days after your injection, half the drug is still circulating. After ten days, one quarter remains. After fifteen days, one eighth. After twenty days, roughly one sixteenth.
A complete clearance (considered to be five half-lives) takes approximately 25 days, nearly the entire interval between weekly doses. Since tirzepatide is dosed weekly, there is never a drug-free window. Steady-state concentrations maintain continuous levels in your bloodstream.
Pumping and dumping for a few hours, or even a day, accomplishes nothing because tirzepatide levels in your blood (and therefore in your milk) remain essentially constant throughout the week. This is fundamentally different from alcohol, which clears within hours, or from short-acting medications with half-lives measured in minutes.
Understanding tirzepatide dosing timing helps illustrate this point. Whether you take your dose on Monday or Tuesday, drug levels throughout the week remain remarkably stable after the first few doses.
The binary decision
With tirzepatide, the decision is binary. Either you take it while breastfeeding (with the reassuringly low transfer rates) or you do not take it until you finish breastfeeding. There is no middle ground where timed pumping creates a safe window, because that window does not exist.
This is not unique to tirzepatide. Most weekly-dosed medications share this characteristic. Semaglutide withdrawal follows a similar prolonged timeline, though with a slightly shorter half-life of about seven days for the weekly formulation.
Semaglutide versus tirzepatide: which is safer for nursing mothers?
If you are considering GLP-1 medications while breastfeeding, you might wonder whether semaglutide or tirzepatide has a better safety profile during lactation. The answer requires comparing the available data for each.
Semaglutide breast milk data
A study published in the journal Breastfeeding Medicine examined semaglutide transfer into breast milk. Researchers found no detectable levels of semaglutide in any breast milk samples from mothers receiving subcutaneous injections. The maximum projected relative infant dose was 1.26%, well below the 10% safety threshold.
Semaglutide has a molecular weight of approximately 4,113 Daltons, somewhat smaller than tirzepatide (4,814 Daltons) but still far above the 800-Dalton threshold for easy milk transfer. Both drugs share the same fundamental pharmacological advantage: they are too big to cross the mammary barrier efficiently. Understanding how semaglutide works over time provides useful comparison data for mothers weighing their options.
Key differences that matter for breastfeeding
A few differences between the two medications are relevant for nursing mothers:
Oral formulations: Semaglutide is available in oral form (Rybelsus), which contains salcaprozate sodium, an absorption enhancer. This chemical may enter breast milk and accumulate in infants. Oral semaglutide formulations should be avoided during breastfeeding. Only injectable forms of either medication should be considered. Similarly, oral versus injectable tirzepatide presents different considerations for lactating women.
Side effect profiles: The comparison between semaglutide and tirzepatide shows that both cause gastrointestinal side effects, but the intensity and pattern differ. Tirzepatide tends to produce more nausea during dose escalation phases, while semaglutide may cause more persistent GI symptoms at steady state. For breastfeeding mothers, any GI side effect that reduces food or fluid intake poses an indirect risk to milk supply.
Weight loss potency: Tirzepatide produces more weight loss at maximum doses. For a postpartum mother, faster weight loss means faster achievement of health goals, but also means a greater caloric deficit that must be carefully managed alongside breastfeeding demands. Reviewing the dosage comparison charts can help understand the relative potency at each dose level. If you are currently on semaglutide and considering switching between the two medications, the conversion charts provide guidance on equivalent dosing, though breastfeeding-specific conversion data does not yet exist.
The bottom line on comparison
Neither medication has a clearly superior safety profile for breastfeeding. Both show minimal milk transfer. Both pose indirect risks through appetite suppression and potential dehydration. The choice between them should be based on the same factors that guide non-lactating patients: individual response, side effect tolerance, and medical history, with the added consideration of how aggressively the caloric deficit needs to be managed.
Some clinicians prefer semaglutide during breastfeeding simply because it has slightly more published lactation data and a longer track record of clinical use. Others prefer tirzepatide for patients who need more robust weight loss, arguing that the milk transfer data is equally reassuring. Both positions are reasonable given the current evidence.
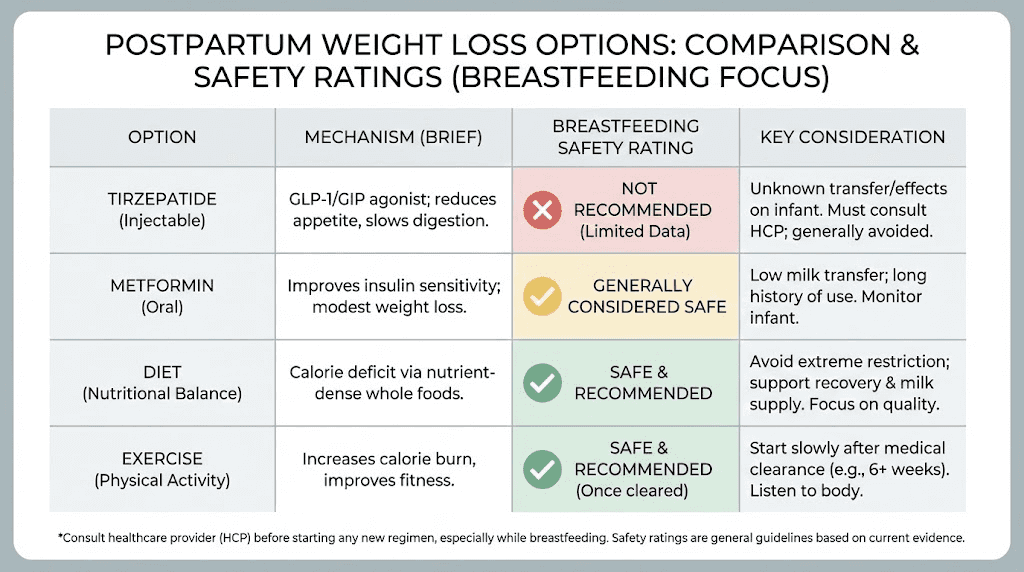
Alternative postpartum weight loss approaches
Tirzepatide is not the only option for postpartum weight management. Before committing to any medication during breastfeeding, consider the alternatives and their respective risk profiles.
Breastfeeding itself as weight management
Here is a fact that often gets lost in the medication discussion. Breastfeeding burns approximately 500 calories per day. Over six months of exclusive breastfeeding, that translates to roughly 90,000 additional calories burned, equivalent to approximately 25 pounds of fat tissue. Many women lose significant weight from breastfeeding alone, particularly when combined with reasonable dietary choices and attention to meal timing.
The catch is that this weight loss is gradual, typically 1 to 2 pounds per month beyond the initial postpartum loss. For women who gained 50 to 80+ pounds during pregnancy or who had pre-pregnancy obesity, this rate may feel frustratingly slow. But it is the safest approach with zero risk to the infant.
Metformin during breastfeeding
Metformin is widely considered safe during breastfeeding. Studies show very little transfers into breast milk, and it has decades of use in lactating women with type 2 diabetes. While metformin produces less weight loss than tirzepatide (typically 3 to 5% of body weight versus 15 to 22%), it carries virtually no lactation-specific concerns. The metabolic benefits of GLP-1 medications go beyond weight loss, but metformin addresses many of the same metabolic concerns with a much longer safety record during lactation.
For women with insulin resistance or polycystic ovary syndrome (PCOS) contributing to postpartum weight retention, metformin may address the underlying metabolic issue while posing minimal breastfeeding risk. Some providers also explore the comparison between phentermine and GLP-1 medications for postpartum patients, though phentermine carries more significant side effect concerns during breastfeeding than either semaglutide or tirzepatide.
Structured nutrition and exercise programs
Working with a registered dietitian who specializes in postpartum nutrition can produce meaningful weight loss without medication. Evidence-based approaches include:
The "tic-tac-toe" meal structure: pairing a lean protein (tic), a complex carbohydrate (tac), and a healthy fat (toe) at every meal. This approach supports milk production while creating a moderate caloric deficit that promotes weight loss without compromising supply.
Progressive return to exercise, starting with walking and gradually incorporating strength training, is safe for most women after the initial postpartum recovery period. Exercise does not reduce milk supply when accompanied by adequate caloric and fluid intake. For women who plan to eventually use peptide-based approaches, the exercise foundation built during breastfeeding makes the transition to peptide-supported weight loss or peptide stacking protocols more effective.
Waiting until weaning
The simplest approach is to wait. Start tirzepatide after breastfeeding ends, when there is zero concern about drug transfer or indirect effects on milk supply. This approach requires patience, but it eliminates all breastfeeding-related risks entirely.
Many women who choose this path use the breastfeeding period to establish healthy eating habits, build exercise routines, and work with healthcare providers to develop a comprehensive weight management plan that includes tirzepatide dosing protocols ready to implement as soon as breastfeeding concludes. This preparation time is also ideal for learning about peptide reconstitution, understanding bacteriostatic water requirements, and dosing fundamentals so you hit the ground running on day one after weaning.
Working with your healthcare team
The decision to use tirzepatide while breastfeeding should never be made alone. It requires coordination between multiple healthcare providers, each contributing different expertise to the decision.
Who should be involved
Your prescribing physician or endocrinologist: Evaluates your medical need for weight loss medication, considers obesity-related comorbidities, and determines whether the benefits of treatment outweigh the theoretical risks during lactation.
Your obstetrician or midwife: Understands your pregnancy history, delivery recovery, and postpartum health trajectory. They can contextualize weight management within your broader reproductive health picture.
A lactation consultant (IBCLC): Monitors milk supply, infant weight gain, and breastfeeding mechanics. If you start tirzepatide while nursing, regular lactation assessments can catch supply issues early before they affect the baby.
A registered dietitian: Creates a nutrition plan that meets both weight loss goals and lactation demands. This is not optional. The appetite suppression caused by tirzepatide makes professional dietary guidance essential during breastfeeding.
Questions to ask your provider
Before starting tirzepatide while breastfeeding, bring these questions to your appointment:
Given my specific medical history, do the benefits of starting now outweigh waiting until I finish breastfeeding?
What is the lowest effective dose we could start with, and can we escalate more slowly than standard protocols?
How often should we monitor the infant weight and feeding patterns?
What specific signs in the baby should prompt me to stop the medication?
Can you coordinate with a lactation consultant to track my milk supply?
What caloric intake do you recommend during treatment while breastfeeding?
Understanding unit measurements for tirzepatide dosing and proper injection technique matters for any user, but especially for breastfeeding mothers who need precise dosing to minimize unnecessary exposure.
Monitoring your baby for changes
If you choose to take tirzepatide while breastfeeding, vigilant monitoring of your infant becomes a critical safety measure. Even though the research is reassuring, ongoing observation provides an additional layer of protection.
Feeding behavior
Watch for changes in how your baby feeds. Is the baby nursing less frequently? Taking shorter feeds? Seeming less satisfied after feeding? Rejecting the breast? Any of these changes could indicate altered milk taste, reduced milk volume, or (very rarely) a reaction to trace medication in the milk.
Keep a feeding log for at least the first four weeks after starting tirzepatide. Note frequency, duration, and whether the baby seems satisfied afterward. This creates a baseline that makes it easier to identify changes.
Growth and weight gain
Infant weight gain is the most objective measure of adequate nutrition. Most pediatricians recommend monthly weight checks during the first six months. If you start tirzepatide while breastfeeding, consider increasing this to biweekly checks for the first month of treatment.
Expected weight gain varies by age. Newborns typically gain 5 to 7 ounces per week. By 4 to 6 months, this slows to 3 to 5 ounces per week. Any significant deviation from your infant established growth curve warrants immediate evaluation and possible discontinuation of tirzepatide.
Gastrointestinal symptoms
While unlikely given the minimal drug transfer, monitor for unusual GI symptoms in your baby: excessive spitting up, changes in stool frequency or consistency, unusual fussiness during or after feeding, or signs of abdominal discomfort. These could theoretically result from trace GLP-1 receptor activation in the infant gut, though no cases have been reported in the published literature.
General behavior and temperament
Note any changes in your infant overall alertness, sleep patterns, or temperament after you start tirzepatide. While it is extremely unlikely that trace amounts of the medication would cause behavioral changes in the infant, documenting baseline behavior makes it easier to identify any shifts. New parents often struggle to distinguish normal developmental changes from potential medication effects, which is another reason professional monitoring through your pediatrician is valuable. Resources on finding peptide-knowledgeable healthcare providers can help you build a team that understands both the medication and lactation considerations.
Practical nutrition strategies for nursing mothers on tirzepatide
If you and your healthcare team decide to proceed with tirzepatide while breastfeeding, nutrition planning becomes your most important daily task. The appetite suppression that makes tirzepatide effective for weight loss is the same property that threatens adequate nutrition for milk production.
Building a breastfeeding-compatible meal plan
Standard tirzepatide diet plans typically target 1,200 to 1,500 calories. During breastfeeding, this is dangerously low. You need to modify any standard plan to include at least 1,800 to 2,300 calories daily. Here is what that looks like in practice:
Breakfast (400-500 calories): Three eggs scrambled with spinach and avocado on whole grain toast. This provides protein, healthy fats, and complex carbohydrates in a format that is easy to eat even when appetite is low.
Mid-morning snack (200-300 calories): Greek yogurt with berries and a tablespoon of nut butter. High protein, calcium for milk production, and healthy fats.
Lunch (400-500 calories): Grilled chicken or salmon over quinoa with roasted vegetables and olive oil. Emphasize omega-3 fatty acids, which are critical for infant brain development and transfer directly into breast milk.
Afternoon snack (200-300 calories): Hummus with vegetables and whole grain crackers, or a smoothie made with milk, banana, protein powder, and flaxseed.
Dinner (500-600 calories): Lean protein (chicken, fish, lean beef, tofu) with sweet potato and a large salad with olive oil dressing. Include iron-rich foods to support postpartum recovery.
Evening snack (100-200 calories): Cottage cheese with fruit, or a small handful of nuts with dark chocolate.
The detailed tirzepatide meal plans provide additional structure, but always adjust upward for breastfeeding caloric needs. Reviewing the recommended foods for GLP-1 medication users offers additional ideas that work well during lactation.
Key nutrients for breastfeeding on tirzepatide
Certain nutrients deserve special attention when combining tirzepatide with breastfeeding:
Protein: Aim for 1.2 to 1.5 grams per kilogram of body weight daily. Protein supports milk production, postpartum recovery, and helps preserve lean muscle mass during weight loss. Most supplement recommendations for tirzepatide emphasize protein, and this is doubly important during lactation.
Calcium: Breastfeeding draws calcium from maternal stores. If dietary intake is insufficient, your bones supply the difference. Aim for 1,000 mg daily from food and supplements combined.
Iron: Postpartum iron needs are elevated, especially after significant blood loss during delivery. Iron deficiency worsens fatigue, which is already a concern with tirzepatide-related tiredness and normal postpartum sleep deprivation.
DHA (omega-3): Critical for infant brain and eye development. DHA transfers into breast milk in proportion to maternal intake. Aim for at least 200 to 300 mg of DHA daily through fatty fish or supplements.
Vitamin D: Breast milk is typically low in vitamin D. Most pediatricians recommend vitamin D supplementation for breastfed infants, but adequate maternal intake (600 to 1,000 IU daily) helps as well. The broader peptide and supplement dosage resources at SeekPeptides can help you understand optimal supplementation alongside any peptide protocol.
B vitamins: Many compounded tirzepatide formulations include vitamin B12 or B12 with glycine. B vitamins support energy metabolism and are important for both maternal health and milk quality. If your formulation does not include B12, consider a separate postnatal vitamin that covers the full B vitamin spectrum.
Managing nausea without compromising nutrition
Nausea is among the most common side effects during tirzepatide dose escalation, a reality that the broader GLP-1 side effect literature documents thoroughly. For a breastfeeding mother, nausea that prevents eating is a direct threat to milk supply. The early weeks on GLP-1 medications can be challenging even without breastfeeding added to the equation. Strategies to manage this include:
Eating smaller, more frequent meals instead of three large ones. Cold foods may be better tolerated than hot foods during nausea episodes. Ginger tea and peppermint can provide natural relief. Avoiding foods known to worsen GI symptoms on tirzepatide is especially important when you cannot afford to skip meals. Some mothers also find that avoiding alcohol entirely while on tirzepatide reduces nausea, which is already standard practice during breastfeeding. Understanding the broader relationship between GI side effect management and adequate nutrition helps mothers maintain both their comfort and their milk supply.
If nausea is severe enough to significantly reduce food intake, discuss dose adjustment with your prescriber. A microdosing approach that uses smaller, more gradual dose increases may reduce GI side effects enough to maintain adequate caloric intake during breastfeeding.
Some mothers find that glycine-enhanced tirzepatide formulations or niacinamide-containing compounds produce fewer GI side effects, though evidence for these benefits remains largely anecdotal.
Contraception and tirzepatide: a critical safety note
This topic warrants its own section because the implications are significant for postpartum women. Tirzepatide may reduce the effectiveness of oral contraceptive pills.
The mechanism involves delayed gastric emptying. Because tirzepatide slows how quickly your stomach processes food and medications, oral contraceptives may not absorb as efficiently. The UK Medicines and Healthcare products Regulatory Agency (MHRA) issued guidance recommending that all women taking tirzepatide use effective contraception, with non-oral methods preferred for reliability.
For breastfeeding mothers, this creates an important consideration. Many postpartum women rely on progestin-only oral contraceptives (the "mini-pill") because combined oral contraceptives can reduce milk supply. If tirzepatide reduces the effectiveness of even the mini-pill, alternative contraception (IUD, implant, injection, barrier methods) becomes essential.
Getting pregnant while on tirzepatide carries its own set of concerns, as the medication has not been adequately studied in pregnancy. Reliable contraception is not optional during tirzepatide treatment, and breastfeeding alone is not reliable contraception despite common misconceptions.
Long-term considerations and unknowns
Honest science acknowledges what it does not know. The tirzepatide-breastfeeding research, while encouraging, has significant gaps that deserve transparent discussion.
What we still do not know
Effects on milk composition: No study has measured whether tirzepatide alters the macro or micronutrient composition of breast milk independent of maternal diet changes. Could the drug affect milk protein profiles, immunoglobulin content, or growth factor concentrations? We do not know yet.
Higher dose data: The breast milk studies tested doses up to 5 mg. Many patients eventually use 10 or 15 mg weekly for optimal weight loss. Whether higher doses produce meaningfully different milk concentrations remains unknown, though the pharmacological principles suggest they should remain very low.
Long-term infant outcomes: No study has followed infants exposed to tirzepatide through breast milk for months or years after exposure. Questions about metabolic programming, growth trajectories, and developmental milestones remain unanswered.
Combined medication effects: Many postpartum women take multiple medications, including antidepressants, thyroid hormones, and supplements. How tirzepatide interacts with these medications in the context of lactation has not been studied. Understanding potential drug interactions with GLP-1 medications matters for all users, but the complexity increases during breastfeeding. The question of combining multiple peptides or medications becomes even more important when lactation adds another variable to the equation. Careful cycle planning should account for the breastfeeding timeline.
Supply over time: The current studies captured relatively short windows. Whether tirzepatide use over months of breastfeeding gradually affects supply through sustained caloric changes, even subtle ones, remains an open question.
The evolving evidence landscape
Research on GLP-1 medications and breastfeeding is accelerating. As millions of women of childbearing age use tirzepatide and semaglutide, the need for robust lactation data has become urgent. Several ongoing studies and registries are collecting data that should substantially improve our understanding within the next few years.
MotherToBaby, a service of the Organization of Teratology Information Specialists, is actively collecting data from women who use tirzepatide during pregnancy or breastfeeding. Participating in these registries helps build the evidence base that future mothers will rely on. Comprehensive peptide research tracking helps researchers and users alike stay current with the rapidly evolving evidence.
For now, the available data supports a cautious but not alarmist approach. The molecular properties of tirzepatide strongly favor minimal milk transfer. The limited human data confirms this prediction. And the practical strategies for protecting milk supply while using the medication are well-established, even if they require more effort and monitoring than typical tirzepatide use. Learning how peptide timelines and expectations work in general can help mothers plan their postpartum approach whether they start during or after breastfeeding.
Real-world decision making: putting it all together
Theory and studies matter. But real decisions happen in real lives, with real constraints and real competing priorities. Here is how the available evidence translates into practical decision-making frameworks.
When the case for tirzepatide during breastfeeding is strongest
The risk-benefit calculation most clearly favors treatment when:
The mother has severe obesity (BMI over 40) with related health conditions like type 2 diabetes, hypertension, or obstructive sleep apnea. The infant is older than six months and already eating solid foods. The mother has professional support from a dietitian and lactation consultant. And non-pharmaceutical approaches have been tried and proven insufficient. Understanding why some people do not lose weight on tirzepatide and what factors affect response helps set realistic expectations before starting.
In these cases, the maternal health benefits of weight loss may outweigh the small theoretical risks of tirzepatide exposure through breast milk. Severe obesity carries its own risks for postpartum health, including increased cardiovascular risk, worsened sleep apnea during a period of already disrupted sleep, and higher rates of postpartum depression.
When waiting is the better choice
The risk-benefit calculation favors waiting when:
The mother weight management goals are primarily cosmetic rather than medically urgent. The infant is under six months and exclusively breastfeeding. The mother does not have access to professional nutritional and lactation support. Or the mother has experienced milk supply issues in the past. In these cases, exploring hormonal health approaches or energy-boosting peptides that have better-established safety profiles during lactation may be more appropriate intermediate options.
Waiting does not mean doing nothing. The postpartum period is an excellent time to address common mistakes that new peptide users make, research how to get started safely, and prepare a comprehensive protocol with your healthcare team for when breastfeeding ends.
The middle ground
Some women and their providers choose a modified approach. Starting with the minimum effective dose. Escalating extremely slowly. Maintaining rigorous nutritional monitoring. And establishing clear stopping criteria: if milk supply drops below a defined threshold, if the baby weight gain falters, or if significant GI side effects prevent adequate nutrition, the medication is paused.
This approach acknowledges that tirzepatide can produce meaningful results even at lower doses, and that the weight loss trajectory does not require maximum dosing to begin showing benefits. Reviewing how quickly tirzepatide works at various dose levels can help set realistic expectations for a slower escalation protocol. Even men using tirzepatide at conservative doses report meaningful changes, suggesting that breastfeeding mothers can achieve similar benefits without pushing to maximum doses. If tirzepatide seems to stop working or plateau at a lower dose, discuss options with your provider before escalating, as the dynamics during breastfeeding differ from standard protocols.
Storage and handling considerations for nursing mothers
Proper medication storage takes on additional importance when there are infants in the home. Tirzepatide must be refrigerated properly and kept secure from curious toddlers if you have older children as well.
Key storage facts: tirzepatide lasts a specific duration in the refrigerator, and understanding how long it can remain at room temperature matters for travel and daily logistics. Learning to travel with GLP-1 medications is relevant for any new mother managing pediatric appointments and family obligations. Compounded formulations may have different stability profiles than brand-name products, so always check with your pharmacy. Knowing whether tirzepatide expires and how to verify potency is important for any user but especially for mothers who want to ensure consistent dosing. General peptide refrigeration guidelines and room temperature stability data provide useful context.
If you use compounded tirzepatide, understanding reconstitution procedures and post-reconstitution storage helps maintain medication integrity. Proper peptide storage practices ensure you are getting the full dose each injection, which matters when you and your provider have carefully calibrated the dose for breastfeeding safety.
Understanding the broader GLP-1 landscape during lactation
Tirzepatide does not exist in isolation. It belongs to a growing class of GLP-1 receptor agonists and related medications that postpartum women may consider. Understanding the broader landscape helps contextualize the tirzepatide-specific data.
GLP-1 medications and breastfeeding is a rapidly evolving area of research. Beyond tirzepatide and semaglutide, newer agents like mazdutide and orforglipron are entering the market with different pharmacological profiles. Some of these newer agents are oral formulations designed with better absorption, which may raise different questions about breast milk transfer.
The relationship between different GLP-1 medications can be confusing. Ozempic and Wegovy are both semaglutide but at different doses for different indications. Mounjaro and Zepbound are both tirzepatide but marketed for diabetes and weight loss respectively. Some weight loss medications like phentermine are not GLP-1 agonists at all and have completely different safety profiles during lactation.
For women interested in peptide-based weight loss approaches, the options extend beyond GLP-1 agonists. Some researchers explore AOD-9604 or other fat-loss peptides, but these have even less breastfeeding safety data than tirzepatide. Sticking with medications that have at least some lactation data, however limited, is the more responsible approach until research catches up. The best peptide options for women during the postpartum period should prioritize safety data over theoretical efficacy.
Common myths about tirzepatide and breastfeeding
Misinformation about GLP-1 medications and breastfeeding circulates widely on social media and in online forums. Separating fact from fiction is essential for making informed decisions.
Myth: tirzepatide will poison your breast milk
This is the most alarmist claim and the least supported by evidence. The word "poison" implies harmful concentrations reaching the infant. The actual data shows concentrations below the limit of detection in 95.9% of milk samples. The remaining samples contained less than 0.02% of the maternal dose. Even among peptide protocols commonly used by women, tirzepatide shows some of the lowest transfer rates into breast milk of any medication studied.
Myth: you need to wean before starting any GLP-1 medication
While some physicians take this conservative position, it is not supported by the available evidence. LactMed explicitly states that tirzepatide use is not a reason to discontinue breastfeeding. The decision should be individualized based on medical need, infant age, and available support, not made as a blanket rule. Understanding the broader landscape of safe peptide use for women provides context for this nuanced decision.
Myth: tirzepatide directly reduces milk production
No evidence shows that tirzepatide targets prolactin, oxytocin, or any other lactation hormone. The milk supply concern is entirely indirect, resulting from potential caloric restriction due to appetite suppression. With proper nutrition planning and the right dietary approach, this indirect effect can be managed. The energy effects of tirzepatide may actually help some postpartum mothers feel more capable of maintaining healthy eating routines.
Myth: oral tirzepatide is safer than injectable during breastfeeding
This is backwards. Oral tirzepatide formulations may contain absorption enhancers that have not been studied in lactation. Injectable tirzepatide is the formulation with breast milk safety data. If you are breastfeeding, stick with the injectable form unless your provider specifically recommends otherwise based on new data.
Myth: lower doses have no effect so there is no point starting small
Even the starting dose of 2.5 mg produces measurable appetite suppression and metabolic effects in most users. Many women achieve meaningful postpartum weight loss at doses of 2.5 to 5 mg, especially when combined with dietary changes. The timeline of tirzepatide effects shows that even at lower doses, most people notice changes within the first two to four weeks. You do not need the maximum dose to see results, and staying lower reduces any theoretical lactation concerns.
The emotional dimension: postpartum body image and medication decisions
No discussion of postpartum weight loss medication would be complete without acknowledging the emotional context. The pressure to "bounce back" after pregnancy is immense. Social media amplifies it. And the physical discomfort of carrying extra weight compounds the emotional burden.
These feelings are valid. They are also not a substitute for careful medical decision-making.
The desire to lose weight quickly after pregnancy is completely understandable. But breastfeeding is temporary. The window during which medication decisions affect your baby through breast milk is finite, usually 12 months or less. Tirzepatide will still be available when you finish nursing. The cost of compounded tirzepatide may even improve over time as more formulations become available. Exploring different tirzepatide sources and vendor options during breastfeeding means you will be ready to start immediately after weaning.
If postpartum depression or anxiety is a factor in your weight management concerns, address that first. Tirzepatide and anxiety have a complex relationship, and adding a new medication during an already vulnerable emotional period deserves careful consideration. Some women find that treating postpartum mood disorders actually improves their relationship with food and exercise, reducing the urgency of pharmaceutical weight loss intervention. The broader topic of peptides for mood support may also be worth discussing with your provider.
SeekPeptides encourages every reader to make medication decisions based on evidence and personal health needs, not external pressure or arbitrary timelines. Your body grew a human being. It deserves respect and informed care during recovery.
Frequently asked questions
Can I take tirzepatide while exclusively breastfeeding?
The available research shows that tirzepatide transfer into breast milk is extremely low, with the drug being undetectable in 95.9% of milk samples from a study of 11 women given a 5 mg dose. LactMed states that tirzepatide use "is not a reason to discontinue breastfeeding." However, most experts recommend waiting until the baby is eating solid foods if possible, and all recommend working closely with a healthcare provider to monitor dosing, nutrition, and infant growth. Our broader guide on GLP-1 medications during breastfeeding covers additional considerations.
Will tirzepatide reduce my milk supply?
Tirzepatide does not directly target prolactin or oxytocin, the hormones that control milk production. However, its appetite-suppressing effects can indirectly reduce supply if you do not eat enough calories. Breastfeeding requires approximately 500 extra calories daily. Follow a structured meal plan that accounts for both weight management and lactation needs, aiming for at least 1,800 to 2,300 calories daily.
Is semaglutide or tirzepatide safer during breastfeeding?
Neither has a clearly superior safety profile for breastfeeding. Both show minimal milk transfer due to their large molecular size. Both pose indirect risks through appetite suppression. The side effect profiles differ slightly, and the choice should be based on individual medical needs and provider recommendation rather than breastfeeding-specific safety differences.
Can I pump and dump to avoid exposing my baby to tirzepatide?
No. Tirzepatide has a five-day half-life and is dosed weekly, meaning drug levels in your blood remain essentially constant. There is no drug-free window during which pumping would produce medication-free milk. The decision is binary: either take tirzepatide while breastfeeding (with its very low transfer rates) or wait until breastfeeding ends.
What dose of tirzepatide is safest during breastfeeding?
The breast milk studies tested doses up to 5 mg. Most experts recommend starting at the lowest available dose (2.5 mg) and escalating more slowly than standard protocols. A microdosing approach may further minimize any theoretical risk while still providing meaningful appetite-suppressing effects.
How soon after giving birth can I start tirzepatide?
No medical organization recommends starting tirzepatide in the first six weeks postpartum. Most experts prefer waiting until the baby is eating solid foods (around six months) if the mother is breastfeeding. For women not breastfeeding, the timeline is determined by postpartum recovery status and medical readiness, which your provider can assess.
Should I avoid compounded tirzepatide while breastfeeding?
The breast milk studies used pharmaceutical-grade tirzepatide from Eli Lilly. Compounded tirzepatide formulations may contain additional ingredients (B12, glycine, niacinamide) whose breast milk transfer has not been specifically studied. The compounded GLP-1 medication landscape is evolving rapidly. If using compounded tirzepatide while breastfeeding, discuss the specific formulation with both your prescriber and your lactation consultant. The glycine and B12 compounds are generally considered safe during breastfeeding individually, but the combination has not been formally evaluated.
What signs in my baby should concern me while taking tirzepatide?
Monitor for changes in feeding behavior (refusing breast, shorter feeds, less satisfaction after feeding), altered stool patterns, unusual fussiness during or after nursing, decreased wet diapers, or any deviation from the established growth curve. Report any concerns to your pediatrician promptly. These signs are more likely related to milk supply changes from reduced caloric intake than from direct medication effects, but they warrant evaluation regardless of the cause. Understanding the full range of GLP-1 side effects helps you distinguish between medication-related changes in yourself versus changes in your baby.
External resources
Frontiers in Endocrinology - GLP-1 Agonists During Pregnancy and Lactation
CMAJ - GLP-1 Receptor Agonists During Pregnancy and Lactation
For researchers serious about navigating peptide protocols safely during every life stage, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members get access to detailed safety databases, expert-reviewed protocols, and personalized guidance for every stage of the peptide research journey.
In case I do not see you, good afternoon, good evening, and good night. May your milk supply stay abundant, your recovery stay steady, and your decisions stay informed.