Feb 15, 2026

You could be wasting hundreds of dollars on retatrutide that does not even contain the correct peptide. Testing data from over 1,700 samples across 132 vendors reveals purity ranging from 62% to 100%, with quantity discrepancies reaching 260% compared to what the label promised. That is not a minor inconsistency. That is a fundamental problem with the research peptide market, and it affects every person searching for retatrutide to purchase online.
The stakes here are unusually high. Retatrutide is not just another GLP-1 receptor agonist. It is the first triple-hormone receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, a mechanism that produced 24.2% mean body weight reduction in Phase 2 trials and a staggering 28.7% in Phase 3 data. Those numbers dwarf what semaglutide and tirzepatide achieve individually. But those results came from pharmaceutical-grade material manufactured under strict GMP conditions by Eli Lilly. What arrives in a research peptide vial from an online vendor is a completely different question.
This guide exists because the gap between clinical promise and marketplace reality has never been wider. We will walk through exactly how to evaluate retatrutide sources online, what purity testing actually tells you (and what it does not), how to read a Certificate of Analysis without getting fooled, what reconstitution and storage protocols look like for research-grade material, and where the entire retatrutide market stands regarding FDA approval timelines. Whether you are a researcher comparing vendors or someone trying to understand this peptide before it reaches pharmacies, everything you need is here.
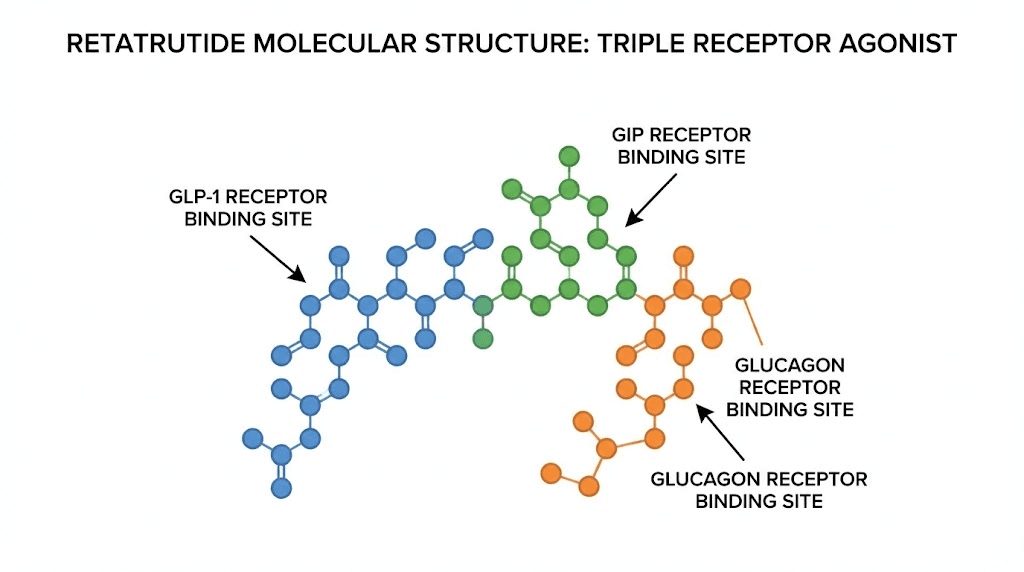
What makes retatrutide different from every other weight loss peptide
Before spending a dollar online, you need to understand what you are actually looking for. Retatrutide, also designated LY3437943, is a 39-amino-acid synthetic peptide engineered from a GIP peptide backbone. Eli Lilly developed it as a peptide that simultaneously activates three metabolic receptor systems. No other approved or late-stage investigational compound does this.
The triple agonist mechanism explained
Most people understand that peptides work by binding to specific receptors. Semaglutide hits one receptor. GLP-1. That single mechanism produces roughly 15-17% body weight loss in clinical settings. Tirzepatide targets two receptors, GLP-1 and GIP, and pushes weight loss to about 20-22%. Retatrutide adds a third. Glucagon.
Why does the glucagon receptor matter? Glucagon directly increases energy expenditure. It tells your body to burn stored fat for fuel, accelerates hepatic lipid oxidation, and raises resting metabolic rate. When you combine appetite suppression from GLP-1 activation with the insulin-sensitizing effects of GIP stimulation and the thermogenic boost from glucagon receptor engagement, you get a compounding metabolic effect that no single-receptor or dual-receptor agonist can match.
The numbers prove it. Phase 2 trials published in the New England Journal of Medicine showed the 12 mg dose group losing 24.2% of body weight at 48 weeks. Phase 3 data from the TRIUMPH-4 study, released in December of last year, showed 28.7% average body weight loss at the 12 mg dose over 68 weeks. That translates to roughly 71.2 pounds on average. For context, retatrutide outperforms semaglutide by a significant margin and exceeds tirzepatide as well.
Understanding this mechanism matters for online sourcing because it explains the molecular complexity. A 39-amino-acid peptide with a fatty diacid moiety attached for half-life extension is harder to synthesize correctly than shorter, simpler peptides. That complexity directly correlates with the quality variation you see across vendors.
Molecular weight and identification
The molecular weight of retatrutide is approximately 4,731.4 g/mol. This number is your first line of defense when evaluating a Certificate of Analysis. Mass spectrometry results on any legitimate COA should show an experimental molecular weight matching this theoretical value within a narrow tolerance. If the measured mass deviates significantly, the vial does not contain authentic retatrutide, regardless of what the label claims.
The peptide sequence and its fatty acid conjugation create a half-life of approximately six days, which enables once-weekly dosing in clinical protocols. This pharmacokinetic profile also means that storage and handling errors accumulate over time. A degraded sample does not just lose potency uniformly. It fragments into unpredictable breakdown products that mass spectrometry will detect as impurities.
Why this matters for the person searching online
When you search for retatrutide online, you are searching for a molecule that:
Is not yet FDA approved for any indication
Is currently in seven Phase 3 clinical trials run exclusively by Eli Lilly
Has a molecular weight of 4,731.4 g/mol requiring precise synthesis
Contains a fatty diacid moiety that complicates manufacturing
Must be stored at specific temperatures to maintain structural integrity
Every one of these factors increases the likelihood that what you receive from an online vendor differs from what was used in clinical trials. Understanding the science behind the molecule is not academic curiosity. It is practical protection against wasting money on a product that does not do what the research suggests the real molecule can do.
The current FDA and regulatory landscape
Before evaluating online sources, you need a clear picture of where retatrutide stands legally and regulatorily. Confusion about this drives many purchasing mistakes. People assume that because retatrutide is available from vendors, it has some form of regulatory approval. It does not.
Clinical trial status
Eli Lilly is running seven Phase 3 clinical trials evaluating retatrutide for obesity, type 2 diabetes, and related metabolic conditions. The TRIUMPH program includes studies examining weight loss outcomes, cardiovascular endpoints, and comorbidity improvements like osteoarthritis pain relief. Most of these trials are expected to complete enrollment and primary data collection during this year.
The first Phase 3 results came from the TRIUMPH-4 trial, announced in December. The 12 mg dose produced 28.7% weight loss over 68 weeks, and the 9 mg dose achieved 26.4%. Both doses met all primary and key secondary endpoints. These results exceeded analyst expectations and accelerated the projected timeline.
If all remaining Phase 3 trials report positive results, Eli Lilly could submit a New Drug Application to the FDA in late this year or early next year. Under standard review timelines, that places potential FDA approval somewhere around mid to late next year. Priority review could shorten that window, but no priority designation has been granted yet.
FDA enforcement actions
The FDA has taken a notably aggressive stance toward online retatrutide sales. In September of last year, the agency sent warning letters to multiple websites selling products described as compounded retatrutide. These letters cited violations of the Federal Food, Drug, and Cosmetic Act. The core issue is straightforward. Because retatrutide has no approved form, compounding pharmacies cannot legally produce it under the exemptions that apply to approved medications like semaglutide or tirzepatide.
This regulatory reality means that any product sold online as retatrutide falls into one of two categories. It is either marketed as a research chemical, labeled "not for human consumption," or it is being sold in violation of federal law. Neither category provides the safety assurances that pharmaceutical manufacturing does. Understanding this distinction is essential for anyone evaluating retatrutide costs and sourcing options.
Research chemical classification
The majority of retatrutide available online is sold under a "research use only" designation. This classification means the product is intended for in vitro laboratory research and is not manufactured, tested, or quality-controlled to pharmaceutical standards. The practical implications are significant. Research chemicals do not undergo sterility testing in most cases. They do not require endotoxin screening. Manufacturing environments may not meet GMP standards. And there is no regulatory body auditing production facilities.
Peptide legality varies by jurisdiction and intended use. In the United States, purchasing research peptides for legitimate laboratory use is generally legal. However, the line between research use and personal use is a legal gray area that enforcement agencies have increasingly scrutinized. The peptide regulation landscape continues to shift, and staying informed about current rules protects both your investment and your legal standing.
How to evaluate retatrutide vendors online
This section is the most important in this entire guide. Vendor evaluation determines whether you receive a legitimate research peptide or an expensive vial of uncertainty. The best peptide vendors differentiate themselves through testing transparency, manufacturing standards, and verifiable quality documentation.
Certificate of Analysis: what to look for
A Certificate of Analysis is the single most important document associated with any peptide purchase. It should accompany every batch, and any vendor who cannot provide a batch-specific COA on request should be immediately disqualified. But having a COA is not enough. You need to know how to read one.
The essential elements of a legitimate retatrutide COA include:
HPLC purity results. High-Performance Liquid Chromatography measures what percentage of the sample consists of the target peptide versus impurities. For retatrutide research, look for purity above 98%. Premium vendors achieve 99% or higher. The HPLC chromatogram should show a single dominant peak with minimal side peaks. Multiple significant peaks indicate synthesis errors, degradation products, or contamination.
Mass spectrometry data. This confirms molecular identity. The measured molecular weight should match retatrutide theoretical weight of 4,731.4 g/mol within a tight margin. Mass spec does not tell you about purity, but it definitively confirms whether the correct molecule was synthesized. If the mass is wrong, nothing else matters.
Appearance and solubility. Legitimate retatrutide in lyophilized form should appear as a white to off-white powder. Significant discoloration suggests degradation or contamination. The COA should note expected solubility characteristics in standard reconstitution solvents.
Batch number and date. Every COA must reference a specific production batch. Generic COAs that do not correspond to your specific vial are essentially meaningless. The production date matters because peptide stability is time-dependent even in lyophilized form.
Third-party testing: the gold standard
Vendor-provided COAs carry an inherent conflict of interest. The same company selling you the product is also telling you it is pure. Third-party testing eliminates this conflict. Independent analytical laboratories test the actual product and report results with no financial incentive to inflate quality claims.
Several approaches to third-party verification exist. Some vendors proactively submit samples to independent labs and publish results. Others offer to send samples from your specific batch upon request. The most transparent vendors encourage customers to submit samples to labs of their own choosing. This last approach provides the highest confidence level because neither the vendor nor their preferred lab controls the process.
Independent testing services like Finnrick Analytics have tested over 1,700 retatrutide samples across 132 vendors. Their data reveals the market reality. Purity varies from 62% to 100%, and actual peptide quantity in vials can deviate by up to 260% from the advertised amount. That means a vial labeled as 10 mg might contain anywhere from 3.8 mg to 26 mg of actual peptide. For anyone calculating retatrutide dosage, this variation makes precise research impossible without independent verification.
Red flags that should disqualify a vendor
Recognizing problematic vendors saves both money and risk. These warning signs should trigger immediate caution.
No COA available. Any vendor that cannot provide batch-specific analytical data is not worth considering. Period. Legitimate synthesis generates analytical data as part of quality control. If they cannot share it, either the data does not exist or the results are unfavorable.
Claims of pharmaceutical grade. Research peptide vendors do not produce pharmaceutical-grade products. That designation requires FDA-registered manufacturing facilities, validated processes, and regulatory oversight that research chemical companies do not have. Vendors making this claim are either misinformed or deliberately misleading.
Pricing far below market. The average price for a 10 mg vial of research-grade retatrutide hovers around $200-250. Significantly lower pricing, particularly below $100, suggests corners are being cut in synthesis, purification, or quality testing. Complex peptides like retatrutide have real manufacturing costs that cannot be circumvented without sacrificing quality.
No physical address or verifiable business identity. Legitimate research chemical suppliers maintain identifiable business addresses, customer service contacts, and often business registration records. Vendors operating through anonymous websites with only email contact present obvious accountability concerns.
Marketing for human consumption. Any vendor explicitly marketing retatrutide for human use, weight loss, or medical treatment is operating outside legal boundaries. This does not mean their product is necessarily lower quality, but it demonstrates a willingness to ignore regulatory requirements, which does not inspire confidence in their other quality claims.
Vendor evaluation checklist
Before purchasing from any online source, work through this checklist:
Batch-specific COA available with HPLC and mass spec data
Purity documented above 98% via HPLC
Mass spectrometry confirming correct molecular weight (4,731.4 g/mol)
Third-party testing results available or offered
Clear labeling as research use only
Verifiable business identity and contact information
Consistent pricing with market averages
Proper shipping conditions (temperature-controlled when necessary)
Return or replacement policy for quality issues
Responsive customer service that can answer technical questions
Checking every item does not guarantee a perfect product, but it eliminates the most common sources of quality problems. Peptide testing laboratories can verify any vendor claims independently if you want an additional layer of assurance.
Understanding purity testing methods
Reading a COA requires understanding what each test actually measures and, equally important, what it does not measure. Many researchers overestimate the information that standard purity tests provide while underestimating the gaps that remain even with favorable results.
HPLC purity analysis
HPLC separates the components of a sample based on their chemical properties and measures the proportion of each component. When a COA reports 99% purity by HPLC, it means that 99% of the UV-absorbing material in the sample was identified as the target peptide. The remaining 1% consists of other peptide-related impurities, typically truncated sequences, deletion sequences, or oxidized forms of the target peptide.
What HPLC does not tell you is significant. It does not detect non-peptide contaminants that do not absorb UV light at the detection wavelength. It does not identify endotoxins, residual solvents, or trace metals. It does not confirm that the peptide is biologically active. A sample could show 99% purity by HPLC while still containing problematic contaminants that a different analytical method would detect.
For peptide reconstitution and research applications, HPLC purity above 98% is generally considered acceptable. For sensitive cell-based assays, 99%+ is preferable. Below 95%, the impurity load becomes significant enough to potentially confound research results.
Mass spectrometry confirmation
Mass spectrometry measures the molecular weight of a compound with extreme precision. For retatrutide, the theoretical molecular weight of 4,731.4 g/mol serves as the reference standard. Mass spec results confirming this weight prove that the correct amino acid sequence was synthesized and that the fatty diacid moiety was properly attached.
A mass spectrum showing the correct molecular weight plus HPLC showing high purity gives you reasonable confidence that the vial contains what it claims. However, mass spec has its own limitations. It cannot distinguish between properly folded and misfolded peptides. It cannot detect all types of modifications or degradation products if they happen to have similar masses. And it provides a snapshot of the sample at the time of testing, which may not reflect the product condition after shipping and storage.
Endotoxin testing: the overlooked safety factor
This is where most vendors fail and most buyers do not even know to ask. Endotoxins are lipopolysaccharides from bacterial cell walls that can contaminate peptide products during manufacturing. They cause inflammatory reactions and can be dangerous even in small amounts. The safe limit for injectable products is 0.1 EU/mg.
Recent analysis suggests that 78% of research-grade retatrutide fails endotoxin screening, showing levels exceeding 100 EU/mg, which is 1,000 times the safe limit. This is not an abstract concern. Endotoxin contamination is a direct consequence of manufacturing environments that do not meet pharmaceutical cleanroom standards. Most research peptide facilities are not required to test for endotoxins, and most do not.
If a vendor does provide endotoxin testing data (typically via the LAL test), that vendor is demonstrating a higher level of quality consciousness than average. It does not make the product pharmaceutical grade, but it addresses one of the most significant safety gaps in the research peptide market.
Amino acid analysis
Amino acid analysis (AAA) measures the actual peptide content of a sample by hydrolysis and quantification of individual amino acids. This test tells you how much peptide is in the vial in absolute terms, not just purity percentages. A vial labeled as 10 mg with 99% HPLC purity might contain less actual peptide than expected due to residual moisture, counterions, or adsorption losses during manufacturing.
AAA is rarely included on standard COAs from research peptide vendors because it adds cost and frequently reveals that actual content is lower than label claims. When available, it provides the most accurate picture of what you are actually getting for your money.
Retatrutide versus semaglutide versus tirzepatide: understanding why people search online
The surge in online retatrutide searches is driven by clinical data that positions it as the most potent weight loss peptide in development. Understanding how it compares to currently available options explains both the demand and the willingness of researchers to navigate the unregulated market.
Head-to-head efficacy comparison
Retatrutide versus semaglutide is not even close in terms of raw weight loss numbers. Semaglutide at the 2.4 mg dose (the maximum approved for weight management) produces roughly 15-17% body weight reduction in clinical trials. Retatrutide at 12 mg produces 24-29% depending on the trial and duration. That gap of 10+ percentage points represents a fundamentally different outcome for many individuals.
Tirzepatide compared to semaglutide already showed that dual-receptor agonism outperforms single-receptor activation. At its highest dose, tirzepatide achieves approximately 20-22% weight loss. Retatrutide exceeds this by another 6-8 percentage points. The progression from one receptor (semaglutide) to two (tirzepatide) to three (retatrutide) follows a clear pattern of increasing efficacy.
Here is how the three compare across key parameters:
Parameter | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
Receptor targets | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
Max weight loss | 15-17% | 20-22% | 24-29% |
FDA approved | Yes | Yes | No (Phase 3) |
Dosing frequency | Weekly | Weekly | Weekly |
Max clinical dose | 2.4 mg | 15 mg | 12 mg |
Online availability | Prescription + research | Prescription + research | Research only |
The comparison reveals why online demand for retatrutide is so strong despite the regulatory and quality challenges. For researchers and individuals following the clinical data, retatrutide represents the most effective peptide for weight loss currently in the pipeline, and waiting for FDA approval means waiting at least another 18 months.
Beyond weight loss: metabolic benefits
Retatrutide clinical data extends beyond the scale. The glucagon receptor component drives metabolic improvements that dual agonists do not achieve as effectively. A randomized Phase 2a trial published in Nature Medicine demonstrated significant reductions in liver fat content in patients with metabolic dysfunction-associated steatotic liver disease (MASLD). At the 12 mg dose, liver fat reduction reached levels comparable to what bariatric surgery achieves.
The TRIUMPH-4 trial also showed substantial relief from knee osteoarthritis pain in participants with obesity. This finding makes sense because the magnitude of weight loss directly correlates with reduced mechanical stress on weight-bearing joints. But the glucagon-driven metabolic improvements, including better lipid profiles, reduced inflammation markers, and improved insulin sensitivity, suggest benefits beyond what weight loss alone would explain.
Researchers studying peptides for fat loss find retatrutide particularly interesting because the glucagon component appears to preferentially target visceral fat, the metabolically dangerous fat surrounding organs. This specificity could make retatrutide uniquely valuable for metabolic health beyond what the body weight numbers indicate.
Reconstitution, storage, and handling protocols
Assuming you have sourced retatrutide from a vendor that passes the evaluation criteria above, proper handling determines whether the peptide maintains its structural integrity and biological activity. The reconstitution process for retatrutide follows general peptide reconstitution principles but with specific considerations for its size and complexity.
What you need before reconstitution
Gather all materials before starting. You will need bacteriostatic water (BAC water containing 0.9% benzyl alcohol), sterile syringes, alcohol swabs, and proper refrigeration immediately available. The workspace should be clean and free from drafts or contaminants.
For a 10 mg vial, adding 2.0 mL of bacteriostatic water yields a concentration of 5.0 mg/mL. This is the most common reconstitution volume for research protocols. Our guide to bacteriostatic water amounts for retatrutide covers alternative volumes and concentrations for different vial sizes.
Step-by-step reconstitution
The process requires patience. Rushing any step risks damaging the peptide.
Step 1: Remove the vial from freezer storage and allow it to reach room temperature gradually. This takes 15-20 minutes. Do not attempt to speed the process with heat.
Step 2: Clean the vial stopper with an alcohol swab and allow it to dry completely. Residual alcohol entering the vial can interact with the peptide.
Step 3: Draw the calculated volume of bacteriostatic water into a sterile syringe. For a 10 mg vial reconstituted to 5 mg/mL, that is 2.0 mL.
Step 4: Insert the needle through the vial stopper and direct the stream of water against the glass wall, not directly onto the lyophilized powder. Direct impact can damage fragile peptide structures.
Step 5: Allow the water to flow down the glass wall and contact the powder gently. Do not inject the entire volume rapidly.
Step 6: Once all water is added, remove the syringe. Gently swirl the vial with a circular wrist motion. Never shake. The agitation from shaking creates foam and can denature the peptide through mechanical stress.
Step 7: If the powder does not dissolve immediately, place the vial in the refrigerator and check every 10-15 minutes. Most properly manufactured retatrutide dissolves within 5-10 minutes with gentle swirling. Persistent cloudiness or visible particles after 30 minutes may indicate a degraded product.
The reconstituted solution should be clear and colorless. Any yellow tint, cloudiness, or visible particulates is cause for concern. Our peptide reconstitution calculator can help you determine exact volumes for your specific vial size and desired concentration.
Storage requirements
Retatrutide stability depends heavily on proper storage at every stage. Before reconstitution, lyophilized retatrutide should be stored at -20 degrees Celsius (-4 degrees Fahrenheit) in a freezer. At this temperature, the powder remains stable for extended periods. Room temperature storage of lyophilized peptides gradually degrades potency, with losses accelerating over time.
After reconstitution, the solution must be refrigerated at 2-8 degrees Celsius (35.6-46.4 degrees Fahrenheit). Most protocols recommend using reconstituted retatrutide within 2-4 weeks. Beyond this window, degradation products accumulate and purity decreases progressively. Never freeze reconstituted peptide solutions, as the freeze-thaw cycle causes aggregation and loss of activity.
The guidelines for storing peptides after reconstitution apply across all research peptides, but the stakes with retatrutide are higher because of its molecular complexity. Larger peptides with post-translational modifications (like the fatty diacid moiety on retatrutide) are more susceptible to degradation than simpler sequences.
Protect the vial from light at all times. UV radiation accelerates oxidation of methionine and tryptophan residues in the peptide chain. Store in a dark section of your refrigerator or wrap the vial in foil. Peptide storage best practices apply universally, but they matter more with expensive, complex peptides where replacement cost is significant.
Dosage information from clinical trials
Any discussion of retatrutide dosing must start with a critical disclaimer. Retatrutide is not approved for human use. The dosage information below comes exclusively from published clinical trial data and is provided for educational context. Retatrutide dosage charts derived from clinical protocols offer reference points for understanding the research, not recommendations for self-administration.
Clinical trial dosing protocol
Phase 2 and Phase 3 trials used a titration approach, starting at lower doses and increasing gradually to minimize gastrointestinal side effects. The general escalation pattern from published protocols looks like this:
Initiation phase (weeks 1-4): Starting doses ranged from 0.5 mg to 2 mg once weekly, depending on the study arm. This low-dose introduction period allows the GI tract to adapt to the triple receptor activation.
Escalation phase (weeks 5-12): Doses increased by 2 mg every four weeks. This gradual titration was specifically designed to mitigate the nausea, vomiting, and diarrhea that occur more frequently with rapid dose increases.
Maintenance phase (week 13+): Target maintenance doses in the trials were 4 mg, 8 mg, and 12 mg once weekly. The 12 mg dose produced the most dramatic weight loss results but also the highest incidence of adverse events.
The detailed retatrutide dose schedule from clinical protocols emphasizes that slow titration is not optional. Rapid dose escalation significantly increases the severity and duration of gastrointestinal side effects. The complete retatrutide dose guide covers these protocols in greater detail.
Why titration matters more with triple agonists
Retatrutide activates three receptor systems simultaneously. Each system contributes its own set of physiological effects, and the combination creates a more intense metabolic shift than single or dual agonists. The glucagon component, in particular, adds effects that semaglutide and tirzepatide users do not experience, including increased thermogenesis and changes in hepatic glucose output.
Researchers who have experience with semaglutide dosing or tirzepatide dosing sometimes assume that retatrutide titration can be more aggressive because of their prior experience with GLP-1 receptor activation. This assumption is dangerous. The glucagon receptor activation introduces a novel set of effects that prior GLP-1 experience does not prepare you for.
Injection technique and site selection
Clinical trials administered retatrutide via subcutaneous injection, the same route used for semaglutide and tirzepatide. The injection site selection for retatrutide follows standard subcutaneous protocols. The abdomen (avoiding a 2-inch radius around the navel), the front of the thigh, and the back of the upper arm are all acceptable sites.
Site rotation matters for weekly injections. Using the same spot repeatedly can cause lipodystrophy, changes in the fatty tissue that affect absorption consistency. Rotating between at least three sites ensures even absorption and reduces local tissue reactions. For those new to peptide injections, starting with the abdomen typically provides the most consistent absorption and the easiest self-administration angle.
Clean the injection site with an alcohol swab and let it dry completely. Pinch a fold of skin, insert the needle at a 45-90 degree angle (depending on body composition), inject slowly, hold for 5-10 seconds, withdraw the needle straight, and apply gentle pressure if needed. Do not massage the site. Our guide on how to inject GLP-1 peptides covers the technique in full detail with illustrations.
Side effects and safety concerns
The clinical trial data provides a reasonably detailed picture of retatrutide side effects, though long-term safety data is still accumulating. Anyone researching retatrutide online should understand these risks thoroughly before making purchasing decisions.
Gastrointestinal effects
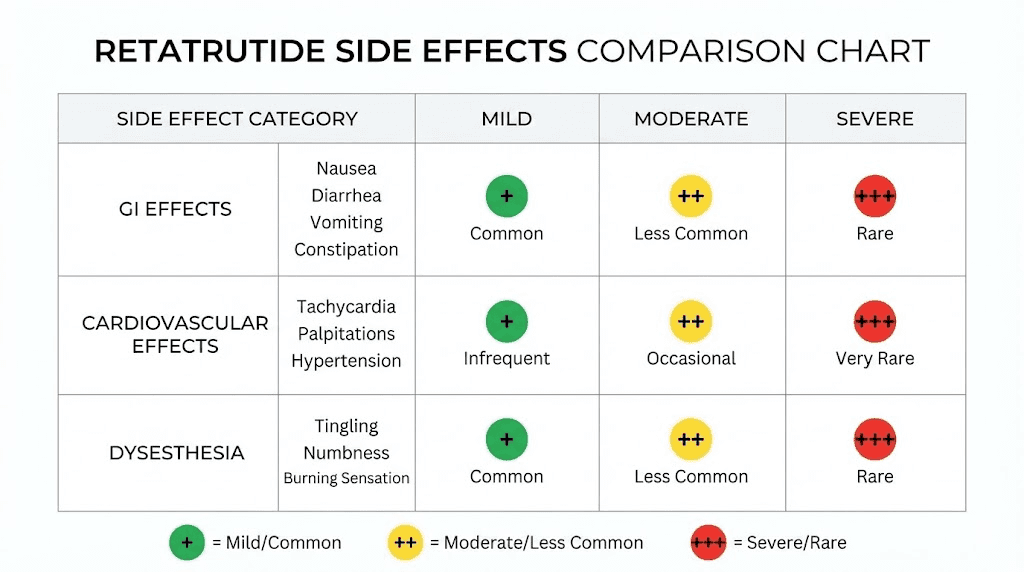
GI side effects dominate the adverse event profile, consistent with all GLP-1 receptor agonists. But the triple mechanism intensifies some of these effects compared to what semaglutide and tirzepatide side effects look like individually.
Nausea is the most frequently reported side effect, occurring at significantly higher rates across all dose levels compared to placebo. The incidence is dose-dependent, meaning higher doses produce more nausea. Importantly, slow titration partially mitigates this. Trials that started with lower initial doses and escalated more gradually showed reduced nausea severity compared to earlier protocols that escalated faster.
Diarrhea, vomiting, and constipation also occurred at higher rates in treatment groups. These effects were generally mild to moderate in severity and tended to diminish over time as the body adapted. Most participants who experienced GI side effects reported them during the dose escalation phase, with symptoms improving once a stable maintenance dose was reached.
For practical management of these effects, many of the strategies used with tirzepatide dietary adjustments and semaglutide dietary considerations apply. Smaller, more frequent meals, avoiding high-fat foods during the adjustment period, and maintaining hydration all help manage GI discomfort.
Cardiovascular effects
Heart rate increased in a dose-dependent manner during the first 24 weeks of treatment, with the largest average increase being approximately 6.7 beats per minute at the 12 mg dose. After 24 weeks, heart rate tended to decline toward baseline levels. This pattern is consistent with other GLP-1 receptor agonists, though the magnitude of the initial increase with retatrutide is slightly larger.
One serious cardiac adverse event, a case of prolonged QT syndrome, occurred in a trial participant. However, this event was associated with the concurrent use of ondansetron (an anti-nausea medication), not directly attributable to retatrutide itself. Cardiac arrhythmias reported across the trials were generally mild to moderate.
Dysesthesia: a new safety signal
The December TRIUMPH-4 data revealed a previously underrecognized side effect. Dysesthesia, described as skin sensitivity, tingling, or tenderness to the touch, affected approximately 20.9% of participants on the highest dose. This side effect has not been reported at similar rates with semaglutide or tirzepatide and appears to be related to the glucagon receptor activation component unique to retatrutide.
The mechanism behind this effect is not yet fully understood. Glucagon receptors are expressed in peripheral nerve tissue, and activation may alter sensory nerve signaling. For most affected participants, the symptoms were manageable and did not lead to treatment discontinuation, but it represents a novel consideration that researchers should be aware of.
Liver and pancreatic effects
Transient increases in liver enzymes (ALT) exceeding three times the upper normal limit occurred in roughly 1% of participants receiving retatrutide. These elevations resolved without intervention in most cases. Increases in amylase and lipase levels were also observed but were asymptomatic, with one exception: a single case of acute pancreatitis that was classified as a serious adverse event.
The liver enzyme pattern is noteworthy because retatrutide simultaneously shows potent fat-clearing effects in the liver (beneficial for MASLD/NAFLD) while occasionally triggering transient enzyme elevations. Monitoring liver function is standard practice in the clinical trials and would be expected in any future approved protocol.
What the research peptide market adds to the risk profile
Here is the part that most online discussions ignore. Every side effect and safety data point described above comes from pharmaceutical-grade retatrutide manufactured under GMP conditions with known purity, potency, and sterility. When you purchase retatrutide from an online research vendor, you are introducing additional variables.
Impurities from lower-purity synthesis can cause reactions unrelated to retatrutide itself. Endotoxin contamination triggers inflammatory responses. Incorrect peptide content means actual dosing may be much higher or lower than intended, which directly affects both efficacy and side effect risk. Degradation products from improper storage may have their own biological activity. These additional risks layer on top of the inherent risks of the molecule itself.
Understanding peptide safety and risks in general provides a foundation, but retatrutide online sourcing introduces quality variables that approved pharmaceutical products do not have.
Cost analysis and what to expect
The retatrutide cost landscape varies significantly depending on source, quantity, and quality. Understanding the pricing structure helps identify both reasonable deals and suspicious ones.
Research peptide market pricing
Based on current market data, research-grade retatrutide pricing generally falls into these ranges:
Vial size | Average price range | Price per mg |
|---|---|---|
5 mg | $120-180 | $24-36 |
10 mg | $200-300 | $20-30 |
20 mg | $350-500 | $17.50-25 |
30 mg | $450-650 | $15-22 |
Larger vials generally offer better per-milligram pricing. However, larger vials also mean committing more money to a single vendor before you can verify quality. A conservative approach is to order smaller quantities initially, test them, and scale up only after confirming quality through independent verification.
When comparing peptide costs across vendors, always normalize to price per milligram. A $150 vial that contains 8 mg of actual peptide (despite being labeled 10 mg) is more expensive per milligram than a $200 vial that truly contains 10 mg. This is why independent testing matters for cost evaluation, not just quality assurance.
Hidden costs to factor in
The vial price is not the complete cost. Factor in bacteriostatic water ($8-15 per vial), syringes ($15-30 for a box), alcohol swabs ($5-10), and potentially cold shipping surcharges ($15-30 during warm months). If you send samples for third-party testing, add $50-150 per test. These ancillary costs add 15-25% on top of the peptide price itself.
Our peptide cost calculator can help you model total research costs including all these factors.
Projected pharmaceutical pricing
When retatrutide eventually receives FDA approval, pharmaceutical pricing will likely be substantially higher than research peptide costs. Semaglutide (Wegovy) carries a list price exceeding $1,300 per month. Tirzepatide (Zepbound) is priced similarly. Retatrutide, as a novel triple agonist with potentially superior efficacy, will almost certainly be priced in the same range or higher.
This pricing gap between research peptides and pharmaceutical products is one of the primary drivers of the online market. However, the cost savings come with the quality, purity, and safety trade-offs discussed throughout this guide. It is not free money. It is a cost-risk trade-off that every researcher must evaluate individually.
How retatrutide online compares to other sourcing options
Online research peptide vendors are not the only pathway to retatrutide. Understanding all available options provides context for evaluating whether online sourcing is the right choice for your situation.
Clinical trials
Enrolling in one of Eli Lilly TRIUMPH trials provides access to pharmaceutical-grade retatrutide at no cost, with full medical supervision. The trade-off is that you may be randomized to placebo, you must meet specific eligibility criteria, and participation requires regular clinic visits and monitoring. ClinicalTrials.gov lists active retatrutide studies that are recruiting participants.
Compounding pharmacies
Following FDA warning letters, the compounding pharmacy route for retatrutide has essentially closed. Unlike semaglutide and tirzepatide, which can be compounded because they have FDA-approved reference products, retatrutide has no approved form to compound from. Any compounding pharmacy offering retatrutide is operating in a legally questionable space.
International sources
Some researchers source retatrutide from international suppliers, particularly those based in China where much of the global peptide synthesis capacity is located. Chinese peptide sourcing carries its own set of considerations, including longer shipping times, customs clearance uncertainties, communication barriers, and varying manufacturing standards. Quality can range from excellent to poor, making independent testing even more important with international sources.
Waiting for FDA approval
The most conservative approach is simply waiting. If Phase 3 trials complete successfully and Lilly submits an NDA in late this year, FDA approval could come by mid to late next year. At that point, retatrutide would be available through standard medical channels with guaranteed quality, proper dosing guidance, and medical supervision. The trade-off is time, potentially 18+ months from today.
For some people, the risk-benefit calculation clearly favors waiting. For others, particularly those with significant obesity-related health conditions, the potential benefits of earlier access justify the additional risks of the research market. This is an individual decision that should be made with full information about both the potential benefits and the real risks.
Protecting yourself when purchasing retatrutide online
If you have evaluated the risks and decided to source retatrutide from an online vendor, these practical steps minimize your exposure to quality and safety issues.
Start with a test order
Never make a large first purchase from any vendor. Order the smallest available quantity and submit a portion for independent testing before using any of the product. This approach costs more per milligram upfront but prevents the much larger cost of purchasing bulk quantities from a vendor whose quality turns out to be substandard.
Independent testing protocol
Select a third-party analytical laboratory that offers HPLC and mass spectrometry services for synthetic peptides. Send a sealed vial from your order directly to the lab. Request HPLC purity analysis, mass spectrometry identity confirmation, and endotoxin testing if available. Compare the results against the vendor-provided COA. Significant discrepancies between vendor claims and independent results tell you everything you need to know about that vendor future reliability.
Our guide to peptide testing laboratories covers how to find and work with reputable independent labs for this purpose.
Document everything
Maintain records of every purchase including vendor information, batch numbers, COAs, independent testing results, storage conditions, reconstitution details, and any observations during use. This documentation serves multiple purposes. It allows you to identify patterns across vendors and batches. It provides evidence in case of quality disputes. And it contributes to the broader community knowledge about vendor reliability.
Monitor your body
If you are using retatrutide for research involving biological systems, establish baseline measurements before beginning and track relevant parameters systematically. Any unexpected responses, particularly those inconsistent with published clinical data, may indicate product quality issues rather than true pharmacological effects. Knowing what to expect based on the clinical trial literature helps distinguish between expected effects and anomalies that warrant investigation.
Basic monitoring should include the parameters tracked in clinical trials: body weight, waist circumference, blood pressure, resting heart rate, and subjective GI symptom assessment. More comprehensive monitoring would add fasting glucose, HbA1c, liver enzymes, and lipid panel testing at baseline and regular intervals.
Have a plan for adverse reactions
Research peptides are not pharmaceutical products, and using them carries inherent risks. Before starting any research protocol, know how you would handle a significant adverse reaction. This means having access to medical care, being honest with healthcare providers about what you are using if medical attention becomes necessary, and being prepared to discontinue immediately if concerning symptoms arise.
The most common adverse reactions based on clinical trial data (nausea, diarrhea, reduced appetite) are generally manageable. The less common but more serious possibilities (pancreatitis symptoms, severe allergic reaction, significant heart rate changes) require immediate medical attention. Understanding the side effect profiles of related peptides provides additional context for what to watch for with GLP-1 class compounds.
The future of retatrutide availability
The retatrutide market will transform dramatically over the next 18-24 months. Understanding the trajectory helps frame current purchasing decisions in proper context.
Phase 3 trial completion timeline
Seven Phase 3 trials are expected to report results during this year. TRIUMPH-4 has already delivered positive topline results. As additional trials report, the picture of retatrutide efficacy and safety across different populations and indications will become increasingly complete. Each positive trial result moves the molecule closer to NDA submission.
Potential trial setbacks could delay the timeline. Safety signals in specific populations, insufficient efficacy in certain endpoints, or manufacturing challenges could all slow progress. However, the strength of TRIUMPH-4 data and the commercial incentive for Eli Lilly make significant delays unlikely barring an unexpected safety finding.
Projected approval and launch
Industry analysts and regulatory experts project the following scenario: NDA submission in Q4 of this year, FDA review during the first half of next year, and potential approval in Q3 or Q4 of next year. If Lilly receives Priority Review designation (possible given the unmet medical need in obesity), the review timeline could compress by several months.
Post-approval, Eli Lilly will face the same manufacturing and supply challenges that plagued the semaglutide and tirzepatide launches. Initial availability may be limited, with waiting lists and periodic shortages common in the first year. This dynamic could sustain demand for online research sources even after approval, as it did with both previous GLP-1 agonist launches.
Impact on the research peptide market
FDA approval will fundamentally change the online retatrutide market. Currently, research vendors operate in a gray area where demand exists but no legal pharmaceutical alternative is available. Once an approved version launches, the regulatory calculus shifts. The FDA will likely increase enforcement against vendors selling unapproved retatrutide, and the market may contract significantly.
However, the price differential between research peptides and pharmaceutical products will persist. As long as pharmaceutical retatrutide costs over $1,000 per month and research-grade material costs a fraction of that, demand for the research market will continue. This dynamic has played out with semaglutide already, where compounded semaglutide remains popular despite approved versions being available.
SeekPeptides continues to track the retatrutide approval timeline and will update our resources as new clinical data and regulatory developments emerge. Members get access to the latest analysis, protocol updates, and community discussions about retatrutide and other emerging peptides.
Stacking considerations and related peptides
Some researchers explore combining retatrutide with other peptides. This section addresses those considerations with the important caveat that retatrutide combination protocols have not been studied in clinical trials, and combining investigational compounds introduces compounding unknowns.
Retatrutide and other GLP-1 agonists
Combining retatrutide with semaglutide or tirzepatide is redundant and potentially dangerous. Retatrutide already activates GLP-1 receptors. Adding another GLP-1 agonist does not increase efficacy. It increases receptor overstimulation and the severity of side effects, particularly nausea and cardiovascular effects. There is no clinical rationale for this combination, and experienced researchers avoid it.
For those currently using semaglutide or tirzepatide who want to transition to retatrutide, a washout period between discontinuing the current peptide and initiating retatrutide is advisable. The switching between GLP-1 peptides guide covers transition protocols, though specific retatrutide switching data is limited to clinical trial enrollment procedures.
Retatrutide and non-overlapping peptides
Peptides targeting entirely different pathways present a different consideration. BPC-157 for gut healing, TB-500 for tissue repair, or peptides for hair growth target mechanisms unrelated to metabolic hormone signaling. Theoretical interaction risk is lower, but "lower" does not mean "zero." No clinical data exists for any of these combinations with retatrutide.
The peptide stacking guide covers general principles for combining peptides, and the peptide stack calculator helps model multi-peptide protocols. However, both resources should be applied with extra caution when one component is a potent triple agonist like retatrutide.
Supplements and supportive compounds
Clinical trial data shows that B vitamins, particularly B12, may help address some of the nutritional absorption changes that GLP-1 agonists can cause. Semaglutide with B12 is a common combination, and the rationale extends to retatrutide. Glycine supplementation has been explored with tirzepatide (semaglutide with glycine) for its potential anti-nausea and metabolic benefits.
Electrolyte supplementation becomes more important with any peptide that causes GI side effects. Vomiting and diarrhea deplete electrolytes rapidly, and maintaining hydration with electrolyte-containing fluids helps manage these common early-treatment effects.
Frequently asked questions
Is retatrutide FDA approved?
No. Retatrutide is currently in Phase 3 clinical trials conducted by Eli Lilly. No FDA approval application has been submitted yet. The earliest projected approval date is mid to late next year, assuming all ongoing trials complete successfully. Any product sold online as retatrutide is classified as a research chemical, not an approved pharmaceutical. Check our retatrutide availability guide for the latest regulatory updates.
How much weight can retatrutide help lose?
Phase 3 clinical trial data showed average weight loss of 28.7% of body weight (approximately 71.2 pounds) at the 12 mg dose over 68 weeks. Phase 2 data showed 24.2% weight loss at 48 weeks with the same dose. These results exceed both semaglutide (15-17%) and tirzepatide (20-22%) in comparable timeframes.
What is the difference between retatrutide and semaglutide?
Semaglutide targets only the GLP-1 receptor. Retatrutide targets three receptors simultaneously: GLP-1, GIP, and glucagon. This triple mechanism produces greater weight loss, potentially better metabolic improvements, and additional effects like increased energy expenditure from glucagon activation. The complete retatrutide versus semaglutide comparison covers all the differences in detail.
How do you reconstitute retatrutide?
Add bacteriostatic water to the lyophilized powder by directing the stream against the glass vial wall. For a 10 mg vial, adding 2 mL of BAC water yields 5 mg/mL concentration. Gently swirl (never shake) until dissolved. The solution should be clear and colorless. See our BAC water guide for retatrutide for detailed volumes and calculations.
Can you take retatrutide with tirzepatide?
This combination is not recommended and has no clinical data supporting it. Retatrutide already activates GLP-1 and GIP receptors, the same targets tirzepatide hits. Combining them would increase side effect risk without adding meaningful efficacy. If transitioning between the two, allow a washout period between stopping one and starting the other.
What are the main side effects of retatrutide?
The most common side effects are gastrointestinal: nausea, diarrhea, vomiting, and constipation. These are dose-dependent and usually mild to moderate in severity. A newer finding from Phase 3 data is dysesthesia (skin tingling or sensitivity) affecting about 21% of participants at the highest dose. The retatrutide side effects guide covers the full safety profile.
How should I store retatrutide?
Store lyophilized (powder) retatrutide at -20 degrees Celsius in a freezer. After reconstitution, refrigerate at 2-8 degrees Celsius and use within 2-4 weeks. Protect from light at all times. Never freeze reconstituted solution. Follow our peptide storage guide for detailed handling protocols.
How much does retatrutide cost online?
Research-grade retatrutide typically costs $120-180 for a 5 mg vial, $200-300 for 10 mg, and $350-500 for 20 mg. Prices vary significantly by vendor. The retatrutide cost guide provides detailed pricing analysis across the market.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members get access to detailed retatrutide protocol updates, vendor evaluation tools, and expert-reviewed dosing guidance that accounts for the real-world quality challenges the research market presents.
In case I do not see you, good afternoon, good evening, and good night. May your peptides stay pure, your vendors stay transparent, and your research stay informed.