Feb 18, 2026

You have been on tirzepatide for months. The weight came off. The appetite quieted down. Your protocol worked. But now something has shifted. Maybe the results have plateaued. Maybe you have heard about a triple agonist that targets an entirely different receptor pathway. Maybe your provider recommended retatrutide as the next step in your research.
Whatever brought you here, the question is the same. How long will it take for retatrutide to actually start working after you stop tirzepatide?
The short answer is four to eight weeks for noticeable effects, with the most significant results appearing between months three and six. But the full answer is more complex than that. It depends on your retatrutide dosage, your previous tirzepatide dose, whether you use a washout period, how your body responds to the titration schedule, and a handful of metabolic factors that make every transition unique. This guide covers all of it. The pharmacokinetics behind the switch. The week-by-week timeline you can actually expect. The dosing protocols that minimize side effects. The mistakes that delay results. And the specific strategies that experienced researchers use to make this transition as smooth as possible.
SeekPeptides has tracked hundreds of transition protocols, and the patterns are clear. Getting this switch right comes down to understanding a few critical variables that most guides completely ignore.
Why researchers switch from tirzepatide to retatrutide
Tirzepatide is a dual agonist. It targets two receptors: GLP-1 and GIP. These two pathways work together to suppress appetite, improve insulin sensitivity, and promote fat loss. For many researchers, tirzepatide delivers exceptional results. The before and after transformations speak for themselves.
But dual agonism has a ceiling.
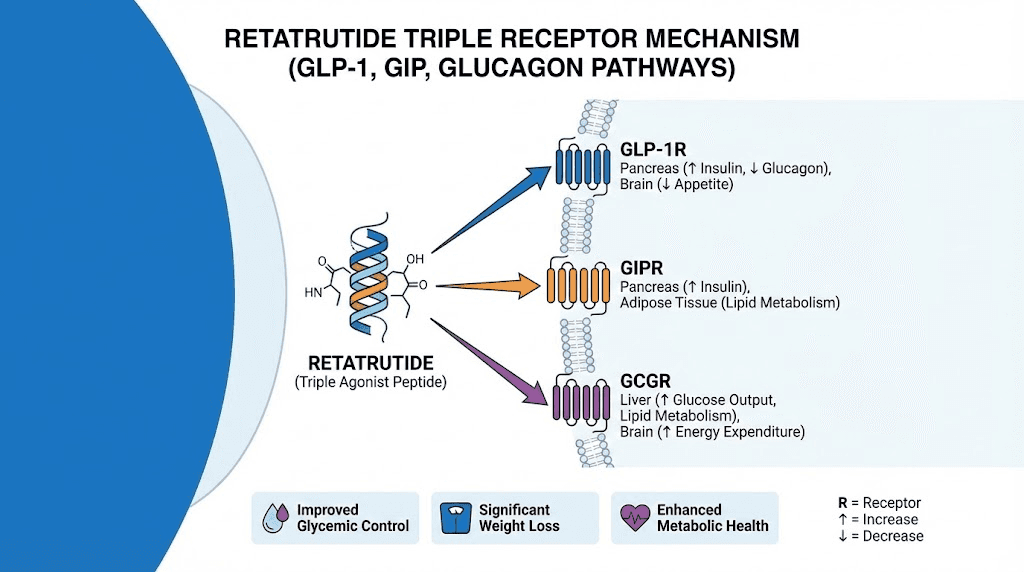
Retatrutide adds a third receptor to the equation. It activates GLP-1, GIP, and the glucagon receptor. That third pathway changes everything. Glucagon receptor activation promotes lipolysis, the direct breakdown of stored fat for energy. It increases hepatic fatty acid oxidation, meaning your liver burns fat more efficiently. It may even support modest increases in energy expenditure and thermogenesis, though human data on that point remains preliminary.
In Phase 2 clinical trials published in the New England Journal of Medicine, participants on retatrutide 12mg achieved an average weight reduction of 24.2% at 48 weeks. That compares to tirzepatide achieving up to 22.5% at 72 weeks in the SURMOUNT-1 trial. Retatrutide achieved comparable or better results in a shorter timeframe, and the Phase 3 TRIUMPH-4 trial pushed those numbers even further, with participants losing an average of 28.7% body weight (71.2 pounds) at 68 weeks.
The reasons researchers make this switch typically fall into a few categories.
Plateau breaking. After months on tirzepatide, weight loss can stall. The body adapts to dual agonism. Adding glucagon receptor activation introduces a new metabolic lever that the body has not yet adapted to. Researchers who hit a wall on tirzepatide often find that retatrutide restarts progress by engaging that third pathway.
Greater fat-specific loss. One notable finding from early retatrutide data is that it led to greater total fat mass reduction than dulaglutide, with a similar proportion of lean mass preserved. This matters. Losing weight is one thing. Losing fat while keeping muscle is something else entirely, and the glucagon pathway appears to preferentially target adipose tissue.
Liver health benefits. In a Phase 2a trial for metabolic dysfunction-associated steatotic liver disease (MASLD), retatrutide reduced liver fat by 81.4% to 86.0% from baseline at 48 weeks. Over 85% of patients achieved less than 5% liver fat. For researchers concerned about metabolic health beyond the scale, these liver outcomes are significant.
Enhanced metabolic markers. Retatrutide doses of 4mg and above improved insulin sensitivity dramatically, reducing fasting insulin by 37% to 71%, reducing HOMA2-IR by 36% to 69%, and increasing adiponectin by 30% to 99%. Triglycerides dropped 35% to 40%. These improvements go beyond what most dual agonists achieve alone.
Understanding the pharmacokinetics of the switch
Before you can understand when retatrutide will kick in, you need to understand what is happening in your body when you stop tirzepatide and start something new.
Tirzepatide has an elimination half-life of approximately 5 days. That means after your last injection, it takes about 5 days for the concentration in your blood to drop by half. After 10 days, roughly 25% remains. After 15 days, about 12.5%. After 20 to 25 days (four to five half-lives), tirzepatide is essentially cleared from your system.
This matters because tirzepatide does not just vanish the day you stop injecting it. Its effects on appetite suppression, gastric emptying, and insulin sensitivity continue to linger as the drug slowly clears. Many researchers notice that appetite remains suppressed for one to two weeks after their last tirzepatide dose, sometimes longer.
Retatrutide has a slightly longer half-life of approximately 6 days. With weekly dosing, it reaches steady-state concentrations in about 4 to 5 weeks. Maximum plasma concentration occurs 12 to 72 hours after each injection. And retatrutide demonstrates dose-proportional pharmacokinetics, meaning that doubling the dose roughly doubles the blood levels.
What does all this mean for the transition?
It means there is a pharmacological overlap period. If you start retatrutide immediately after your last tirzepatide dose, both compounds will be active in your system simultaneously for roughly two to three weeks. Some researchers prefer this overlap. Others prefer a clean washout. Both approaches have trade-offs, and we will cover both in detail.
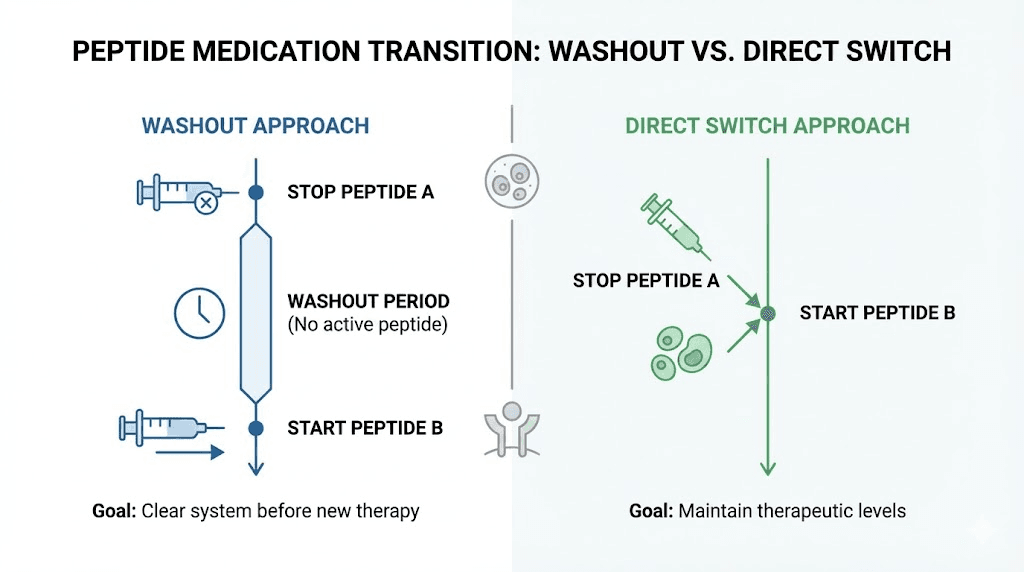
The washout period debate
This is where the research community splits into two camps. And both have legitimate points.
Camp 1: Use a washout period. A washout period of 7 to 14 days between your last tirzepatide injection and your first retatrutide injection allows tirzepatide to partially clear your system. This reduces the risk of compounding side effects from two active incretin-based compounds. It gives you a cleaner baseline to assess how retatrutide affects you independently. And it follows the more conservative pharmacological principle of allowing at least 4 to 5 half-lives (20 to 25 days) for complete elimination.
The downside is obvious. During the washout, you lose the appetite suppression and metabolic benefits of tirzepatide before retatrutide has built up to therapeutic levels. Researchers report increased hunger, potential weight regain, and frustration during this gap. Some describe it as going backwards before going forwards.
Camp 2: Direct switch, no washout. Because tirzepatide and retatrutide share similar receptor targets (both activate GLP-1 and GIP), some researchers argue that a lengthy washout is unnecessary. The body is already adapted to incretin-based signaling. Starting retatrutide while tirzepatide is still partially active provides continuity of appetite suppression and metabolic support during the transition.
The risk here is more pronounced gastrointestinal side effects. Tirzepatide side effects combined with new retatrutide side effects could mean more nausea, more digestive disruption, and a rougher first few weeks.
Most experienced researchers and providers land somewhere in the middle. A short washout of 5 to 7 days, roughly one tirzepatide half-life, seems to balance both concerns. This allows tirzepatide levels to drop by about 50% while still maintaining some residual activity, reducing but not eliminating the overlap period.
What the data suggests
There is no published clinical trial data specifically comparing washout versus direct switch protocols for tirzepatide-to-retatrutide transitions. The recommendations come from clinical experience, pharmacokinetic modeling, and extrapolation from similar drug class transitions.
What we do know is that in the original retatrutide clinical trials, participants were not transitioning from other GLP-1 medications. They started retatrutide from a drug-naive state. So the published timelines (appetite changes at 2 to 4 weeks, significant weight loss at 24 weeks) reflect a clean start, not a transition.
Researchers switching from tirzepatide often report faster initial appetite suppression with retatrutide, likely because their GLP-1 and GIP receptors are already sensitized to agonism. But the full metabolic effects, particularly the glucagon-mediated fat oxidation and liver benefits, still take 4 to 8 weeks to become apparent regardless of prior medication history.
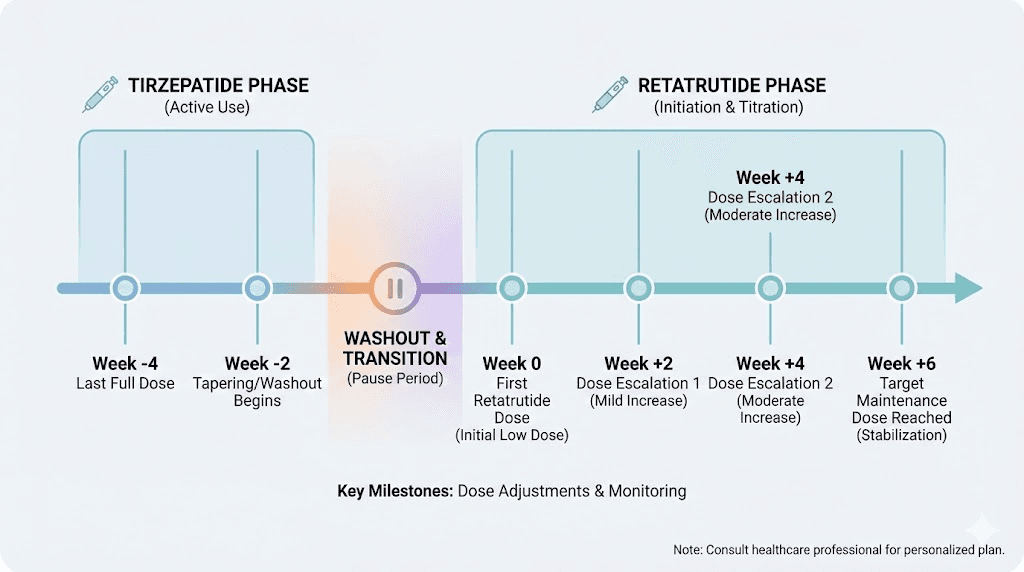
Week-by-week timeline: what to expect after starting retatrutide
Every body responds differently. But based on clinical trial data, community reports, and the pharmacokinetic profile of retatrutide, here is the general timeline most researchers experience when switching from tirzepatide.
Week 1: the adjustment begins
Your first retatrutide injection typically starts at 0.5mg to 1mg weekly. This is a deliberately low dose designed to let your body acclimate to the glucagon receptor activation, which is the genuinely new element in this equation.
During the first week, most researchers notice mild appetite changes. If you used a washout period, your appetite may have started returning before this first injection. Retatrutide begins reversing that within 24 to 72 hours of the first dose, though the effect is subtle at this low dose.
Common experiences during week one include mild nausea (especially in the first 24 hours after injection), slight reduction in appetite, possible fatigue as your body adjusts, and injection site reactions similar to what you experienced with tirzepatide injections.
Do not expect dramatic results in week one. You are laying the foundation.
Weeks 2 to 4: building the base
As retatrutide accumulates with weekly dosing, blood levels climb toward therapeutic range. By week 3, you are approaching roughly 50% of steady-state concentration. By week 4, you are at about 75%.
This is when most researchers notice meaningful appetite suppression returning. Not just the mild reduction from week one, but the kind of appetite control where food stops occupying your thoughts. Meals get smaller. Cravings for high-calorie foods diminish. The satiety signals kick in earlier during meals.
If you were on a higher dose of tirzepatide (10mg or above) before switching, you may find the appetite suppression during these early weeks less intense than what you were used to. That is normal. You are on a starting dose of retatrutide while your body previously had high-dose dual agonist support. The appetite suppression will intensify as you titrate up.
Weight changes during weeks 2 to 4 are typically modest. Clinical trial data shows approximately 2% to 5% body weight reduction by week 4 in drug-naive participants. Researchers transitioning from tirzepatide may see slightly different numbers depending on their starting point, washout approach, and metabolic adaptation status.
The key metric here is not the scale. It is how you feel. Energy levels stabilizing. Appetite normalizing. Gastrointestinal side effects settling down. These signs indicate that retatrutide is engaging its target receptors effectively.
Weeks 4 to 8: the inflection point
This is where things get interesting.
By week 4, most titration protocols increase the retatrutide dose from the starting level. A common schedule moves from 1mg to 2mg at week 5, following the standard 4-week titration intervals used in the clinical trials. This dose increase activates more of the glucagon receptor pathway, the mechanism that differentiates retatrutide from tirzepatide.
Researchers consistently report that weeks 4 to 8 are the inflection point. This is when the transition feels like it is actually working. The appetite suppression deepens. Energy expenditure may increase slightly (though this is difficult to measure subjectively). Clothes start fitting differently. The scale starts moving more consistently.
The glucagon component is the wild card. Unlike GLP-1 and GIP agonism, which primarily affect appetite and insulin signaling, glucagon receptor activation works on the supply side of the energy equation. It tells your body to mobilize and burn stored fat. This creates a fundamentally different metabolic environment than what tirzepatide provides.
Common experiences during weeks 4 to 8 include noticeable fat loss, particularly around the midsection, improved energy levels that many researchers describe as feeling more stable throughout the day, reduced cravings for carbohydrates and processed foods, better energy management compared to the fatigue some experienced on tirzepatide, and clearer hunger and satiety cues.
Side effects during this phase typically improve compared to the first few weeks. The gastrointestinal system adapts. Nausea decreases. Bowel patterns normalize. Most researchers describe weeks 4 to 8 as the period where retatrutide stops feeling like a new medication and starts feeling like part of their routine.
Weeks 8 to 12: reaching therapeutic range
By week 8, retatrutide has reached steady-state concentration at your current dose. If you have titrated according to the standard protocol, you may be at 2mg to 4mg weekly depending on your provider approach and tolerance.
This is when the full triple-agonist effect becomes apparent. All three receptor pathways are consistently activated. The metabolic effects compound. Fat oxidation accelerates. Compared to single or dual agonists, the body is now operating with three simultaneous metabolic interventions.
Researchers who transitioned from high-dose tirzepatide (12.5mg or 15mg) often report that weeks 8 to 12 are when retatrutide matches or exceeds their previous results. The initial weeks felt like a step back. Now it feels like a step forward. The glucagon pathway is delivering benefits that tirzepatide never could.
Visible changes by this point typically include 5% to 10% body weight reduction from the start of retatrutide (depending on starting dose and titration speed), measurable improvements in waist circumference, better metabolic markers if bloodwork is being tracked (fasting glucose, insulin, triglycerides), and continued or improved lean mass preservation.
Weeks 12 to 24: the acceleration phase
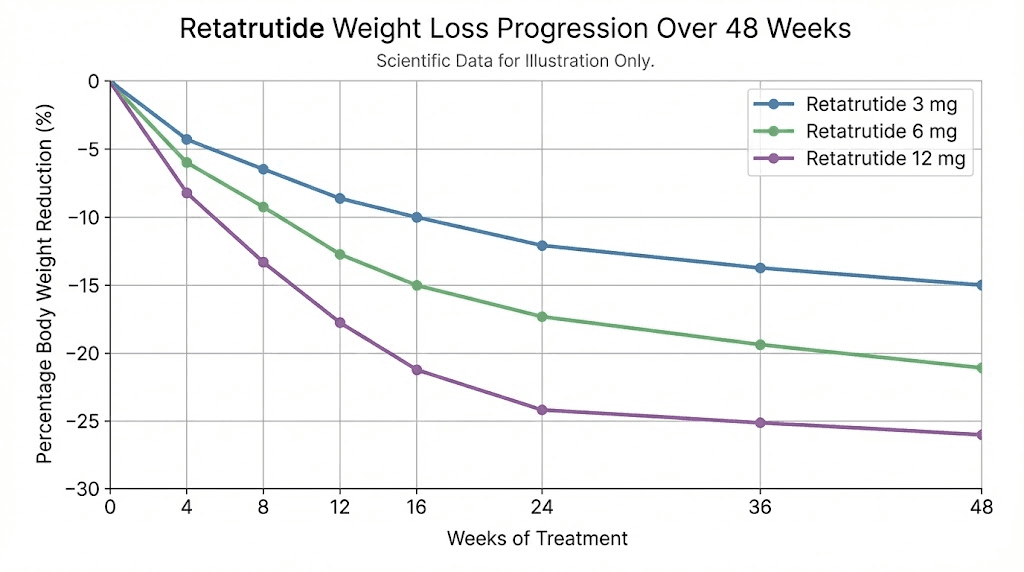
Between months 3 and 6, retatrutide delivers its most dramatic results. This aligns with the Phase 2 clinical trial data where participants on the 12mg dose showed 7.2% to 17.5% weight reduction at 24 weeks.
Titration continues during this phase. Many researchers reach their target dose of 8mg or 12mg by week 12 to 16, following the standard 4-week escalation intervals. At these higher doses, the glucagon pathway fully engages. Liver fat begins dropping dramatically, a benefit unique to retatrutide among this drug class.
For researchers who switched from tirzepatide specifically to break through a plateau, this is the vindication phase. The weight that stopped coming off on tirzepatide starts moving again. The metabolic markers that stalled begin improving. The third receptor pathway provides the novel stimulus that the body could not adapt to the way it adapted to dual agonism.
Phase 2 trial data from this period showed remarkable liver improvements. Participants on 8mg and 12mg retatrutide saw liver fat reductions of 81% to 86% from baseline. Over 85% of participants achieved less than 5% liver fat, effectively resolving fatty liver disease in the vast majority of cases.
The investment in retatrutide starts paying measurable dividends during this phase.
Weeks 24 to 48: sustained results
Long-term retatrutide data shows continued weight loss through 48 weeks, with the highest dose group achieving 24.2% average body weight reduction. The TRIUMPH-4 Phase 3 trial extended this further, showing 28.7% reduction at 68 weeks.
These are not just numbers. A 24% reduction for someone starting at 250 pounds means losing 60 pounds. At 300 pounds, that is 72 pounds. These are life-changing transformations, achieved through a structured dosing protocol with a medication that attacks fat from multiple angles simultaneously.
Researchers who track their progress carefully during this phase often note that the character of weight loss changes compared to tirzepatide. With tirzepatide, weight loss was primarily driven by reduced caloric intake through appetite suppression. With retatrutide, there is an additional component of enhanced fat mobilization and oxidation. The result is often a more favorable body composition shift, more fat lost relative to lean tissue.
The retatrutide titration protocol for tirzepatide switchers
Starting dose matters more than most researchers realize. The temptation after coming off a high dose of tirzepatide is to start retatrutide at a higher dose to maintain continuity. This is almost always a mistake.
Why you start low even after tirzepatide
Your body is adapted to GLP-1 and GIP agonism from tirzepatide. It is not adapted to glucagon receptor activation. Retatrutide introduces a completely new pharmacological stimulus through its glucagon component. Starting at a high dose means hitting an unadapted receptor pathway hard, which typically results in worse gastrointestinal side effects, nausea that is more intense than anything experienced with tirzepatide, and a higher likelihood of dropping out or reducing the dose prematurely.
Clinical trial data supports this approach. In the Phase 2 study, gastrointestinal symptom rates nearly doubled when participants were assigned directly to 8mg instead of titrating gradually from 1mg. Gradual escalation is not just a suggestion. It is the difference between tolerating the transition and abandoning it.
Standard titration schedule
The protocol used in retatrutide clinical trials provides a solid framework.
Weeks 1 to 4: 1mg weekly. This is the acclimation phase. Your body meets the glucagon receptor agonist for the first time. Side effects are usually mild. Appetite suppression begins but is not dramatic.
Weeks 5 to 8: 2mg weekly. The first dose increase. GLP-1 and GIP effects strengthen. Glucagon receptor activation intensifies. This is when most researchers start feeling the difference from tirzepatide, not necessarily stronger, but different. The metabolic profile shifts.
Weeks 9 to 12: 4mg weekly. Now all three pathways are meaningfully engaged. Appetite suppression is strong. Fat mobilization is measurably increased. Side effects from the earlier weeks have typically resolved. This is the dose where clinical trials showed significant improvements in insulin sensitivity, triglycerides, and other metabolic markers.
Weeks 13 to 16: 8mg weekly. For researchers targeting maximum efficacy, this dose begins approaching the range where the most dramatic weight loss results were observed in trials. The liver fat reduction data at this dose is remarkable.
Weeks 17 and beyond: 12mg weekly (if tolerated and desired). The highest dose tested in Phase 2 trials. This is where 24.2% average weight reduction was achieved at 48 weeks. Not every researcher needs or tolerates this dose. Some find that 4mg or 8mg provides the results they want with fewer side effects.
Modified protocol for researchers coming off high-dose tirzepatide
If you were on 10mg, 12.5mg, or 15mg of tirzepatide before switching, the standard protocol still applies, but with slightly different expectations.
At the 1mg starting dose, you will likely feel that appetite suppression is weaker than what you had on tirzepatide. This is expected. Do not try to compensate by increasing the dose faster than the 4-week intervals. The titration schedule exists for a reason, and that reason is the glucagon receptor. Your GLP-1 and GIP receptors may be ready for higher stimulation, but your glucagon receptors are not.
Some providers recommend a slightly modified approach for high-dose tirzepatide switchers. Starting at 2mg instead of 1mg (since your body is already adapted to strong GLP-1/GIP agonism), using a 3-week titration interval instead of 4 weeks (faster escalation for experienced users), and reaching the 4mg to 8mg range by week 8 to 10 instead of week 12. These modifications should only be made under clinical supervision.
Managing side effects during the transition
The transition from tirzepatide to retatrutide comes with a predictable set of challenges. Understanding them in advance makes them manageable instead of alarming.
Gastrointestinal effects
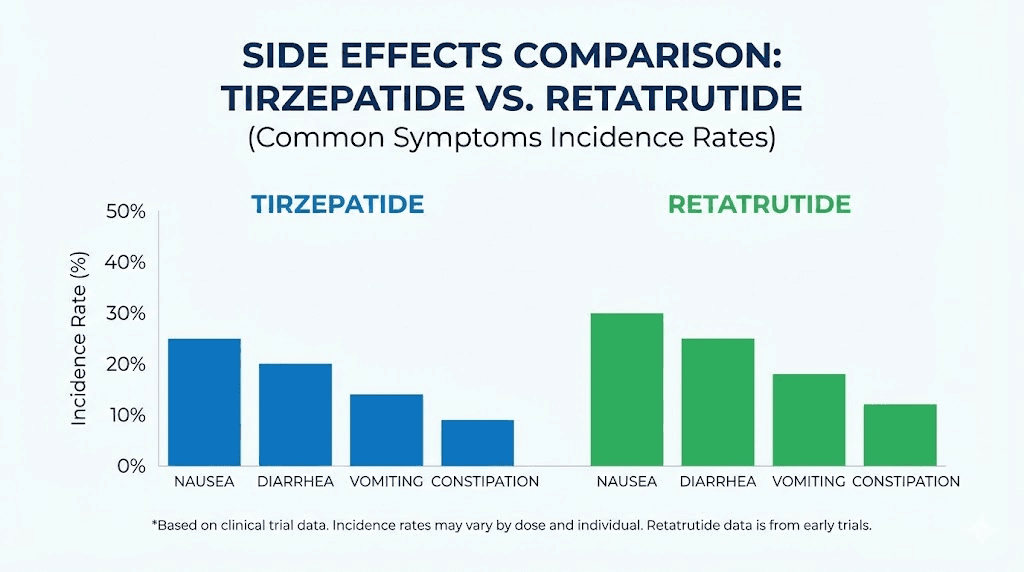
Nausea, diarrhea, vomiting, and constipation are the most common side effects with retatrutide, just as they were with tirzepatide side effects. Clinical trial data shows nausea affecting 14% of participants at 1mg and up to 60% at 12mg. Most GI symptoms resolve within 8 to 12 weeks of consistent dosing.
If you experienced significant GI issues on tirzepatide, you should expect similar or slightly worse effects during the first few weeks of retatrutide. The glucagon receptor activation can add to digestive disruption. However, researchers who tolerated tirzepatide well generally tolerate retatrutide well, provided they follow the titration schedule.
Management strategies that work:
Eat smaller, more frequent meals instead of large ones. Avoid high-fat and spicy foods during the first month, as they exacerbate nausea. Stay hydrated with steady sipping throughout the day, not large amounts at once. Consider taking your injection before bed so you sleep through the initial nausea window. The BRAT approach (bananas, rice, applesauce, toast) helps during acute episodes. If nausea prevents you from eating or drinking, or causes vomiting more than three times per day, contact your provider.
Appetite fluctuations
The transition period often brings unpredictable appetite patterns. One day you have no interest in food. The next day, hunger returns with surprising intensity. This inconsistency is normal during the switch and reflects the changing pharmacological environment as tirzepatide clears and retatrutide builds up.
Do not chase these fluctuations with caloric restriction or binging. Maintain a consistent eating pattern focused on protein-rich foods and whole nutrients. The appetite will stabilize as retatrutide reaches steady state, typically by week 4 to 6.
Energy changes
Some researchers report a temporary energy dip during the first two weeks, especially if they used a washout period. This makes sense. You have removed one metabolic intervention and not yet built up another. The body is adjusting.
Others report the opposite, a subtle energy increase once retatrutide reaches therapeutic levels. The glucagon pathway promotes fat mobilization, which means more available energy substrate. Researchers who experienced fatigue on tirzepatide sometimes find that retatrutide provides more stable energy throughout the day.
Heart rate considerations
Clinical trial data shows that retatrutide was associated with a greater increase in resting heart rate compared to tirzepatide. This is thought to be related to the glucagon receptor activation. Monitor your heart rate during the transition, particularly during dose increases. An increase of 5 to 10 beats per minute is common and generally not concerning. Increases beyond that warrant a conversation with your provider.
Injection site reactions
These are generally similar to what you experienced with tirzepatide. Rotating injection sites, using proper reconstitution technique, and allowing the solution to reach room temperature before injecting all help minimize reactions. The abdomen remains the most common injection site, but the thigh and upper arm are also appropriate.
Factors that affect how quickly retatrutide kicks in
Not everyone experiences the same timeline. Several factors influence how quickly you feel the effects of retatrutide after switching from tirzepatide.
Your previous tirzepatide dose
Researchers coming off low-dose tirzepatide (2.5mg to 5mg) tend to notice retatrutide effects faster. Their receptors are sensitized to incretin agonism but not maximally adapted. Starting retatrutide introduces a fresh stimulus.
Researchers coming off high-dose tirzepatide (10mg to 15mg) often describe a gap period where retatrutide at starting doses feels less effective than what they were used to. This is purely a dosing issue, not a sign that retatrutide is not working. Once titration reaches the 4mg to 8mg range, most high-dose switchers report that retatrutide matches or surpasses their tirzepatide experience.
Body composition and metabolic rate
Retatrutide distributes throughout body tissues. Researchers with higher body fat percentages may experience slightly different pharmacokinetics, with the drug distributing into a larger volume. This does not mean it works less well. It means the timeline to steady state may vary slightly.
Metabolic rate also plays a role. Researchers with higher baseline metabolic rates may clear the drug faster, which could affect the consistency of weekly dosing effects. If you notice that the effects seem to wear off before your next injection, discuss this with your provider.
Diet and lifestyle factors
Retatrutide works best as part of a comprehensive approach. Researchers who maintain adequate protein intake, stay physically active, manage stress, and sleep well consistently report faster onset of noticeable effects. This is not unique to retatrutide, but the glucagon pathway appears particularly responsive to metabolic context.
High-protein diets may actually enhance retatrutide effects. Glucagon is naturally secreted in response to protein ingestion. Supporting this pathway nutritionally while also pharmacologically activating the receptor could amplify results. Some researchers report that protein-focused meal plans during the transition accelerate the onset of fat loss.
Hydration and sleep
Dehydration worsens virtually every side effect of incretin-based medications and can delay the onset of positive effects. Aim for at least half your body weight in ounces of water daily. More if you exercise.
Sleep is equally critical. Growth hormone, cortisol, insulin sensitivity, and appetite-regulating hormones are all influenced by sleep quality. Poor sleep can delay the metabolic benefits of retatrutide and worsen side effects. Prioritize 7 to 9 hours nightly, especially during the first 8 weeks of transition.
Prior GLP-1 medication history
Researchers who have used multiple GLP-1 medications before switching to retatrutide sometimes report that each successive medication takes slightly longer to produce dramatic results. This may reflect progressive receptor adaptation or simply the diminishing returns of repeated weight loss cycles. If you have used semaglutide, tirzepatide, and now retatrutide in sequence, set realistic expectations for the timeline and focus on the unique benefits the glucagon pathway provides.
The glucagon advantage: what tirzepatide never did
This section matters because it explains why the wait is worth it.
Tirzepatide works primarily by reducing how much you eat. It suppresses appetite through GLP-1 and GIP receptor activation. It slows gastric emptying. It improves insulin signaling. These are powerful mechanisms. But they all work on the demand side of the energy equation, reducing caloric intake.
Retatrutide keeps all of those mechanisms and adds the supply side. Glucagon receptor activation tells your body to actively burn stored energy. It promotes lipolysis (fat breakdown), increases hepatic fatty acid oxidation (liver fat burning), reduces lipogenesis (new fat formation), and increases production of non-esterified fatty acids and ketone bodies (alternative fuel sources).
This is why retatrutide matters for tirzepatide switchers who have plateaued. Your body adapted to eating less. It downregulated metabolic rate to match reduced intake. Glucagon receptor activation bypasses that adaptation by telling the body to burn more, regardless of how much you eat.
In practical terms, researchers switching from tirzepatide to retatrutide often describe the weight loss as feeling different. Less about willpower and eating less. More about the body actively mobilizing and burning fat. The scale moves, but so does body composition. Researchers tracking body composition through the transition frequently report improving waist-to-hip ratios even when total weight loss is modest, suggesting preferential visceral fat reduction.
The liver data reinforces this point. Tirzepatide reduces liver fat through weight loss and improved insulin sensitivity. Retatrutide reduces liver fat through those same mechanisms plus direct glucagon-mediated hepatic fat oxidation. The result is liver fat reductions of 81% to 86%, numbers that dwarf what typical weight loss interventions achieve.
Common mistakes that delay retatrutide results
Knowing what not to do is as important as knowing what to do. These are the mistakes that consistently delay the transition timeline.
Mistake 1: starting at too high a dose
The logic seems sound. You were on 15mg of tirzepatide, so starting retatrutide at 4mg or 8mg should be fine, right? Wrong. Your glucagon receptors have not been pharmacologically activated before. Hitting them hard from day one leads to severe nausea, vomiting, diarrhea, and the very real possibility of abandoning the protocol entirely.
Start at the recommended dose. Be patient. The 4-week titration intervals exist because they work.
Mistake 2: expecting tirzepatide-level results in week one
If you were on 15mg of a mature, fully titrated tirzepatide protocol, you cannot expect 1mg of retatrutide to match those effects immediately. This comparison is unfair to both you and the medication. It takes time for any incretin-based drug to reach therapeutic levels, and you went through the same slow build with tirzepatide. Give retatrutide the same runway.
Mistake 3: abandoning the protocol during weeks 2 to 4
Weeks 2 to 4 are the hardest part of the transition for high-dose tirzepatide switchers. Appetite suppression has weakened compared to what you were used to. Side effects from retatrutide are present but the dramatic benefits have not appeared yet. This is the valley of disillusionment, and many researchers quit here.
Those who push through to weeks 6 to 8 are almost universally glad they did. The inflection point is real, and it is coming. Just not in week two.
Mistake 4: skipping the washout entirely at high doses
If you were on high-dose tirzepatide and start high-dose retatrutide simultaneously, you are asking for a rough experience. The overlapping GLP-1/GIP effects compound the gastrointestinal burden. At minimum, take a few days between your last tirzepatide injection and your first retatrutide dose. Your digestive system will thank you.
Mistake 5: not tracking your progress
Without data, you are guessing. Track your weight weekly. Measure your waist circumference biweekly. Log side effects daily for the first month. Note appetite levels on a 1-to-10 scale. Monitor energy throughout the day. Take photos monthly.
This data does two things. It shows you that retatrutide IS working even when it does not feel like it (the scale might not move but your waist measurement drops). And it gives your provider concrete information to optimize your dosage protocol.
Mistake 6: poor nutrition during the transition
Switching medications is not the time to also overhaul your diet. Keep your nutrition consistent during the transition. Focus on adequate protein (at least 0.7 to 1 gram per pound of lean body mass), sufficient fiber, and enough calories to support metabolic function. Extreme caloric restriction combined with a new medication is a recipe for muscle loss, fatigue, and poor outcomes.
Retatrutide vs tirzepatide: a direct comparison for switchers
Understanding how these two compounds differ helps set realistic expectations for the transition.
Factor | Tirzepatide | Retatrutide |
|---|---|---|
Receptor targets | GLP-1, GIP (dual) | GLP-1, GIP, Glucagon (triple) |
Half-life | ~5 days | ~6 days |
Time to steady state | ~4 weeks | ~4-5 weeks |
Max weight loss (trials) | 22.5% at 72 weeks | 24.2% at 48 weeks (Phase 2), 28.7% at 68 weeks (Phase 3) |
Liver fat reduction | Moderate (indirect) | 81-86% reduction (direct) |
Fat-specific loss | Standard | Enhanced (glucagon-mediated) |
FDA approval status | Approved | Investigational (Phase 3) |
Common doses | 2.5mg to 15mg weekly | 1mg to 12mg weekly |
GI side effects | Common | Common (slightly higher at equivalent doses) |
Heart rate effect | Minimal | Greater increase observed |
The comparison reveals why researchers switch. Retatrutide achieves more weight loss in less time, provides direct liver fat reduction that tirzepatide cannot match, and offers a novel metabolic pathway that can break through plateaus. The trade-offs are slightly more GI side effects during titration, a greater heart rate effect, and the fact that retatrutide remains investigational rather than FDA-approved.
For researchers serious about understanding every aspect of this comparison, the retatrutide versus semaglutide analysis and mazdutide versus tirzepatide comparison provide additional context on where these medications fit in the evolving landscape of incretin-based therapies.
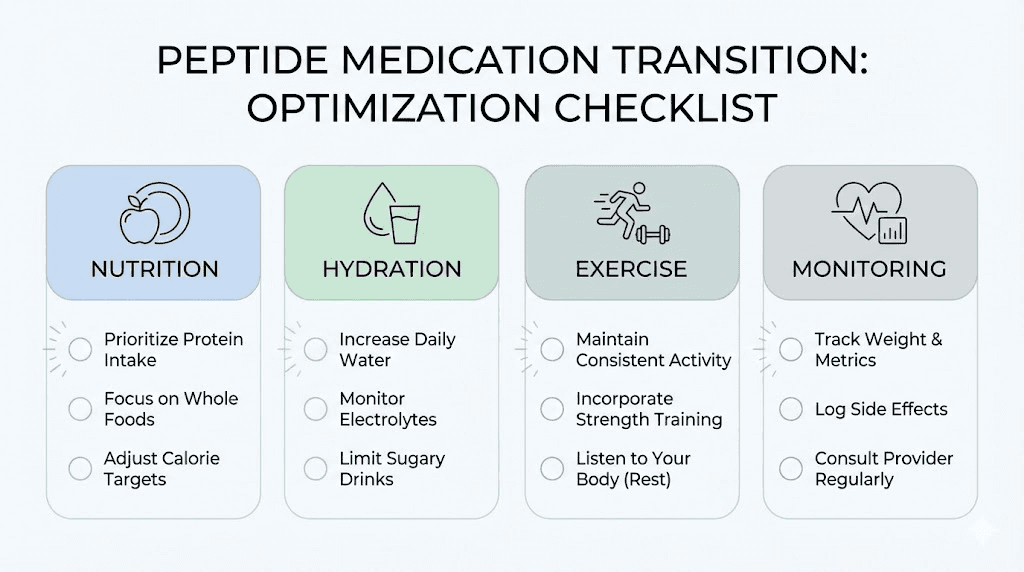
How to optimize your transition protocol
Beyond the basics, these strategies can help you get the most out of the switch.
Pre-transition bloodwork
Get comprehensive metabolic bloodwork before starting retatrutide. Fasting glucose, HbA1c, fasting insulin, comprehensive metabolic panel, lipid panel, liver enzymes (ALT, AST, GGT), thyroid function, and a complete blood count. This gives you a baseline to measure retatrutide effects against and helps identify any issues that might affect the transition.
Timing your injection
Many researchers find that injecting retatrutide in the evening, before bed, helps manage the initial nausea. You sleep through the worst of it. By morning, the acute post-injection nausea has usually passed. The injection timing strategies that work for other GLP-1 medications generally apply here as well.
Protein prioritization
Protein becomes even more important during the transition. The glucagon pathway promotes fat catabolism, and without adequate protein, you risk losing lean mass along with fat. Aim for a minimum of 100 grams of protein daily, ideally 130 to 160 grams for active researchers. Spread protein intake across 3 to 4 meals rather than loading it into one.
Protein shakes, lean meats, eggs, Greek yogurt, and cottage cheese are all excellent options. Some researchers find that high-protein meal planning makes the reduced appetite from retatrutide easier to manage because each smaller meal packs more nutritional value.
Exercise during the transition
Do not dramatically change your exercise routine during the first 4 weeks. Your body is adjusting to a new medication. Intense new workout programs on top of a drug transition often lead to excessive fatigue, poor recovery, and frustration.
Maintain your current activity level. If you are a resistance trainer, keep training but perhaps reduce volume by 10% to 15% during the first month. If you are primarily a cardio person, continue your routine but be willing to reduce intensity if energy levels dip. After week 4, when retatrutide is established, gradually return to or exceed your previous activity levels.
Hydration protocol
Retatrutide, like all incretin-based medications, increases the importance of hydration. The medication can cause mild dehydration through reduced food intake (less water from food), potential diarrhea or vomiting during titration, and subtle effects on renal function.
Aim for a minimum of 80 ounces of water daily. Add electrolytes if you exercise or experience diarrhea. Sip throughout the day rather than drinking large amounts at once. Many researchers find that proper hydration alone significantly reduces nausea and other side effects.
What the research community reports
While clinical trial data provides the scientific foundation, community reports from researchers who have actually made this switch add practical context that trials do not capture.
The most consistent finding is that patience pays off. Researchers who give retatrutide a full 8 to 12 weeks before judging its effectiveness are overwhelmingly positive about the transition. Those who expected immediate results matching their optimized tirzepatide protocol were consistently disappointed in the early weeks.
Another common observation is that the quality of weight loss feels different. Multiple researchers describe losing inches around the midsection faster than the scale number would predict. This aligns with the glucagon-mediated preferential fat mobilization observed in clinical data. The body composition changes can be dramatic even when total weight loss appears modest.
Side effects during transition are consistently described as manageable but real. Most researchers rate them as similar to or slightly worse than their initial tirzepatide experience. The key difference is that with retatrutide, there is a new type of GI disruption from the glucagon component that some describe as different from what they experienced with tirzepatide alone.
Energy levels are a positive surprise for many switchers. While the first 2 weeks often involve an energy dip, weeks 3 through 8 frequently bring energy levels that exceed what researchers experienced on tirzepatide. The glucagon pathway mobilizes stored energy, and many researchers feel this as a subtle but consistent improvement in daily energy and alertness.
Researchers who combine the transition with proper nutrition tracking, adequate protein, and consistent exercise report the fastest and most satisfying results. The medication is a tool. How you use the tool matters.
Special considerations for specific populations
Researchers with type 2 diabetes
If you are switching from tirzepatide to retatrutide for diabetes management, the transition requires additional monitoring. Retatrutide affects blood sugar through three receptor pathways instead of two, and the glucagon component adds complexity to glucose regulation.
Monitor blood glucose more frequently during the first 4 weeks. The combination of residual tirzepatide effects and emerging retatrutide effects can cause unpredictable glucose fluctuations. If you take other diabetes medications (metformin, sulfonylureas, insulin), discuss dose adjustments with your provider before starting retatrutide.
Researchers with liver concerns
For those switching specifically because of liver health concerns, the data is encouraging. Retatrutide MASLD trial results showed liver fat reductions of over 80% at 48 weeks. However, liver enzyme elevations can occur during the early weeks of treatment and do not necessarily indicate liver damage. Have your ALT and AST checked at baseline, 4 weeks, and 12 weeks into retatrutide to monitor trends.
Researchers over 50
Older researchers may experience slower onset of effects and greater sensitivity to side effects. The side effect profiles of incretin-based medications tend to be more pronounced in older populations. Consider starting at the lower end of the dosing range (0.5mg instead of 1mg) and extending titration intervals to 6 weeks instead of 4. The results will come. They may just take slightly longer.
Researchers with significant weight to lose
If you are starting retatrutide with a BMI above 40, the transition timeline may actually be faster for visible results. Higher starting weight means more absolute pounds lost per percentage point. The glucagon pathway has more adipose tissue to work with. Several researchers in this category report visible changes as early as week 3 to 4, even at starting doses.
Long-term planning after the switch
The transition from tirzepatide to retatrutide is not just about the first few months. Long-term success requires thinking ahead.
Finding your maintenance dose
Not everyone needs or wants to reach 12mg. Many researchers find their sweet spot at 4mg or 8mg, where side effects are minimal but efficacy is strong. Your maintenance dose is the lowest dose that continues producing meaningful results with acceptable side effects.
After reaching your target weight or metabolic goals, some researchers reduce from their peak dose to a lower maintenance level. This is an area where retatrutide data is still emerging, but the Phase 3 TRIUMPH program includes a 4mg maintenance dose arm that will provide guidance when results publish later in 2026.
Monitoring ongoing progress
Continue tracking key metrics monthly. Weight, waist circumference, body composition if possible, energy levels, appetite patterns, and any side effects. Quarterly bloodwork (metabolic panel, lipids, liver enzymes, HbA1c) provides objective data on how retatrutide is affecting your metabolic health beyond the scale.
SeekPeptides members access comprehensive tracking tools and protocol databases that make this ongoing monitoring systematic rather than ad hoc. For researchers navigating a complex transition like this, having structured support makes a measurable difference in outcomes.
When to consider further adjustments
If you have been on a stable retatrutide dose for 12 or more weeks with no continued progress and no side effects, it may be time to discuss a dose increase with your provider. If you are experiencing persistent side effects at your current dose that are not resolving, a dose reduction with slower re-titration may be appropriate.
The goal is sustainable, long-term metabolic improvement, not a sprint to the highest dose. Retatrutide works best as a marathon approach with consistent, patient adherence to the protocol.
Frequently asked questions
Can I take retatrutide the same day I stop tirzepatide?
Technically yes, but most providers recommend waiting at least 5 to 7 days after your last tirzepatide injection. This allows tirzepatide levels to drop by approximately 50%, reducing the risk of compounded GI side effects from two active incretin-based compounds. If you do switch immediately, start retatrutide at the lowest available dose and monitor closely for the first two weeks.
Will I gain weight during the washout period?
Some researchers report modest weight gain (1 to 3 pounds) during a washout period, primarily from increased food intake as appetite suppression fades and possible water retention changes. This is usually temporary and reverses quickly once retatrutide reaches therapeutic levels. The withdrawal effects from stopping incretin-based medications are well documented but typically mild and short-lived.
Is retatrutide stronger than tirzepatide?
Clinical data suggests retatrutide achieves greater average weight loss in shorter timeframes than tirzepatide. Phase 2 data showed 24.2% weight loss at 48 weeks with retatrutide 12mg versus tirzepatide 22.5% at 72 weeks. The TRIUMPH-4 Phase 3 trial showed 28.7% at 68 weeks. However, direct head-to-head comparison trials have not been conducted, so these numbers come from different study populations and designs.
What if retatrutide does not work after 8 weeks?
If you are not experiencing any appetite suppression or weight change after 8 weeks of properly titrated retatrutide, several factors should be evaluated. Confirm the reconstitution and dosing are correct. Verify storage conditions (retatrutide requires refrigeration). Review your diet for hidden caloric sources. Check whether you are actually at a therapeutic dose (8 weeks at 1mg is very different from 8 weeks that includes titration to 4mg). Discuss bloodwork with your provider to rule out thyroid or metabolic issues that could blunt response.
Can I switch back to tirzepatide if retatrutide does not work?
Yes. There is no pharmacological reason you cannot return to tirzepatide if retatrutide does not suit you. The same transition principles apply in reverse, with a recommended washout period of 7 to 14 days and restarting tirzepatide at a lower dose than where you left off, then titrating back up.
Does retatrutide cause more hair loss than tirzepatide?
Hair loss is reported with all rapid weight loss interventions, not specifically linked to any one medication. Retatrutide potentially greater rate of weight loss could theoretically increase the risk of telogen effluvium (temporary hair shedding related to rapid weight change). Adequate protein intake, biotin supplementation, and gradual rather than extreme weight loss rates can help minimize this risk. The retatrutide and hair loss data is still limited but aligns with what we see across the GLP-1 medication class.
How do I store retatrutide during the transition?
Store retatrutide in the refrigerator at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Protect from light. Do not freeze. Once reconstituted, most compounded retatrutide solutions remain stable for 28 to 30 days refrigerated. The storage requirements are similar to what you followed with tirzepatide, so your existing routine likely applies.
Will my insurance cover retatrutide?
As of early 2026, retatrutide is not FDA-approved and therefore not covered by insurance. It is available through research channels and compounding pharmacies. The TRIUMPH Phase 3 program results are expected throughout 2026, with potential FDA approval as early as late 2026 or 2027. Check the latest retatrutide availability updates for current access options.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your transitions stay smooth, your receptors stay responsive, and your protocols stay optimized.