Feb 18, 2026

You just saw the positive test. Two pink lines. A plus sign. Whatever the format, the message is clear. And your first thought was not celebration. It was panic. Because you are on tirzepatide, and nobody told you this could happen.
You are not alone. Thousands of women have discovered unexpected pregnancies while taking tirzepatide for weight loss or blood sugar management. The phenomenon has become so widespread that the media coined a term for it: "Ozempic babies." But this is not just a catchy headline. This is your life, your body, and a pregnancy you did not plan for. The questions flooding your mind right now are legitimate. Is your baby safe? Should you stop the medication immediately? Did the tirzepatide cause any harm during those early weeks before you knew?
This guide covers everything you need to know. Not vague reassurances. Not panic-inducing speculation. Real information drawn from FDA labeling, clinical research, reproductive endocrinology data, and the experiences of women who have walked this exact path before you. SeekPeptides created this resource because the intersection of tirzepatide protocols and reproductive health deserves more than a two-paragraph FAQ buried on a pharmaceutical website. You deserve a complete picture, along with clear next steps, so you can make informed decisions with your healthcare provider.
Why tirzepatide makes pregnancy more likely
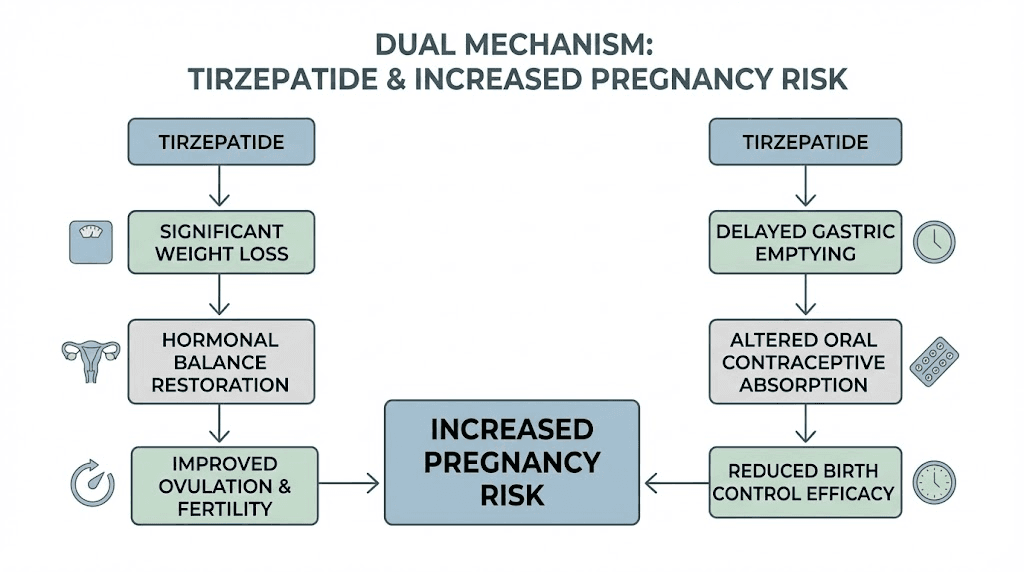
Before we talk about what to do, you need to understand why this happened. Tirzepatide increases your chances of getting pregnant through two separate mechanisms. Both are well documented. Neither requires a medical degree to understand.
The weight loss and fertility connection
The first mechanism is straightforward. Weight loss restores fertility.
Excess body fat produces estrogen. When estrogen levels rise too high, your brain receives a signal that resembles pregnancy. The hypothalamic-pituitary-ovarian axis, the communication loop between your brain and your ovaries, gets confused. Ovulation becomes irregular. Sometimes it stops entirely.
This is especially true for women with polycystic ovary syndrome (PCOS). PCOS affects roughly 10% of women of reproductive age, and obesity makes every symptom worse. Insulin resistance drives androgen production. High androgens disrupt follicle development. The ovaries produce cysts instead of mature eggs. Periods become unpredictable or disappear altogether.
Then you start tirzepatide.
The weight comes off. Sometimes quickly. Clinical trials showed average weight loss of 15-22% of body weight over 72 weeks. That is significant. And as the weight drops, several things happen simultaneously. Insulin sensitivity improves. Androgen levels fall. The hypothalamic-pituitary-ovarian axis recalibrates. Estrogen production normalizes.
And ovulation returns. Sometimes before your periods become regular again. This means you can become pregnant before you even realize your fertility has been restored. You might have gone months or years without ovulating. You might have been told you would need fertility treatments. Then tirzepatide changes your metabolic landscape, and your body quietly starts releasing eggs again.
Research shows that even modest weight loss of 5-10% of body weight can restore ovulation in women with obesity-related anovulation. Tirzepatide typically produces far more than 5-10% weight loss. The standard dosing protocol puts most women well beyond that fertility-restoration threshold within the first few months of treatment.
The birth control interference problem
The second mechanism is more alarming. Tirzepatide can reduce the effectiveness of your birth control pills.
Here is how it works. Tirzepatide is a dual GIP/GLP-1 receptor agonist. One of its primary effects is delayed gastric emptying. It slows down how quickly food and medications move through your stomach and into your small intestine. This is part of why it suppresses appetite so effectively. But it also means that oral medications, including oral contraceptives, get absorbed differently.
Clinical studies measured this directly. After a single 5 mg dose of tirzepatide, oral contraceptive absorption dropped by approximately 20%. The peak concentration of hormones in the bloodstream decreased. The time to reach peak concentration shifted. The overall exposure to the contraceptive hormones fell.
Twenty percent might not sound catastrophic. But oral contraceptives work on tight margins. They maintain a steady hormonal level that prevents ovulation. When that level drops by a fifth, the protection weakens. Combine reduced contraceptive efficacy with newly restored fertility from weight loss, and you have a recipe for unplanned pregnancy.
This is why the tirzepatide prescribing information includes a specific warning. If you use oral hormonal contraceptives, the FDA recommends switching to a non-oral method or adding a barrier method like condoms for four weeks after starting tirzepatide and for four weeks after each dose increase. The delayed gastric emptying effect is most pronounced right after initiation and after each dose escalation.
Many women never receive this warning. Or they receive it buried in a packet of paperwork they never read. Or their prescriber mentions it in passing and they forget. The result is the same: reduced protection at the exact moment their fertility is surging.
How tirzepatide differs from other GLP-1 medications
Worth noting: tirzepatide has a greater impact on oral contraceptive absorption than other GLP-1 receptor agonists like semaglutide. The dual GIP/GLP-1 mechanism produces a more significant delay in gastric emptying, particularly during the rapid dose escalation phase. The impact on medication absorption diminishes over time as your body adapts, but those early weeks and dose adjustment periods carry the highest risk.
If you were taking semaglutide before switching to tirzepatide, the contraceptive interference risk increased when you made that switch. Different mechanism. Different absorption profile. Your birth control was more compromised than you realized.
What to do right now if you just found out
Stop reading forums. Stop spiraling. Here is your action plan.
Step 1: stop taking tirzepatide immediately
Do not take your next scheduled dose. Do not taper. Just stop.
The FDA is clear on this point. Tirzepatide should be discontinued when pregnancy is recognized. There is no recommended tapering schedule for pregnancy-related discontinuation. The medication has a half-life of approximately five days, which means it takes about 25-30 days for tirzepatide to fully clear your system after your last injection.
You will not experience dangerous withdrawal symptoms from stopping tirzepatide abruptly. You may notice your appetite returning. You may experience some nausea as your gastric emptying speeds back up. These changes are manageable and temporary. Your pregnancy is the priority.
If you have questions about your current dose and how stopping might affect you, contact your prescribing physician. But do not wait for a callback to skip your next dose. Stop now.
Step 2: call your healthcare provider today
This is not a "schedule something for next week" situation. Call today. Tell the front desk you are newly pregnant and were taking tirzepatide. Most offices will prioritize this call.
Your provider needs to know:
Your last tirzepatide dose (date and amount)
How long you have been taking it
Your current dose level
When you estimate conception occurred
Whether you were taking oral contraceptives
Any other medications you are taking
Your provider will likely schedule an early ultrasound, possibly as soon as 6-8 weeks gestation. This is standard for pregnancies with potential medication exposure during early development. The ultrasound checks for normal fetal growth, heartbeat, and development.
Step 3: do not panic about early exposure
This is the part where you need to take a breath.
Here is the honest picture. Animal studies showed increased rates of pregnancy loss, reduced birth weight, and some developmental abnormalities when tirzepatide was administered during organogenesis (the critical period of organ formation in early pregnancy). These findings led to the pregnancy warnings on the label.
However. And this is important.
A 2023 study examining GLP-1 receptor agonist exposure during early pregnancy in humans found no increased risk of major birth defects compared to the general population. The Teratology Information Services, which track medication exposure during pregnancy across multiple countries, published data showing reassuring outcomes in women with first-trimester GLP-1 exposure.
Animal studies use doses and conditions that do not always translate directly to human outcomes. The weight loss itself, rather than the medication, may have contributed to some of the adverse outcomes seen in animal studies, since significant caloric restriction during pregnancy is independently harmful. The human data, while limited, has been more reassuring than the animal data predicted.
This does not mean there is zero risk. It means the situation is not as dire as the warning label might make you think. Your healthcare provider will monitor your pregnancy more closely because of the exposure, and that monitoring will catch any concerns early.
Step 4: expect closer monitoring
Your pregnancy will likely include:
An early dating ultrasound (6-8 weeks)
A detailed anatomy scan (18-20 weeks)
Potentially more frequent growth scans
Standard genetic screening (which you should do regardless)
Nutritional counseling (especially regarding adequate caloric intake after stopping a weight loss medication)
Tell every healthcare provider involved in your pregnancy about the tirzepatide exposure. Your OB/GYN, your midwife if you have one, your fertility specialist if applicable. They need the full picture.
The "Ozempic babies" phenomenon explained
You are part of a wave. An unexpected, unplanned, medically fascinating wave of pregnancies connected to GLP-1 medications.
The term "Ozempic babies" emerged from media reports and went viral. Strictly speaking, it refers to all GLP-1 related surprise pregnancies, not just those involving semaglutide (Ozempic). Tirzepatide (Mounjaro and Zepbound) pregnancies fall under the same umbrella.
How common is this?
We do not have exact numbers. Clinical trials excluded pregnant women and required contraception during participation. Post-market surveillance is still accumulating data. But the anecdotal reports are overwhelming. Fertility clinics report patients conceiving naturally after years of failed treatments, with the only change being GLP-1 medication and the resulting weight loss. Women who were told they might never conceive without IVF are getting pregnant within months of starting tirzepatide.
Michigan Medicine researchers highlighted that the connection between weight loss and restored fertility is well established in reproductive endocrinology. What is new is the speed and magnitude of weight loss that GLP-1 medications produce. Previous weight loss methods rarely achieved the 15-22% body weight reduction that tirzepatide delivers. The fertility implications of such rapid metabolic change are only now becoming apparent.
The PCOS connection
Women with PCOS are disproportionately affected. This makes medical sense.
PCOS is the most common cause of anovulatory infertility. It affects 6-12% of women of reproductive age. Obesity worsens PCOS through insulin resistance, which drives elevated androgen production and disrupts normal ovarian function.
Tirzepatide attacks PCOS from multiple angles simultaneously. It reduces body weight. It improves insulin sensitivity through direct GIP and GLP-1 receptor activation. It lowers the insulin levels that drive androgen production. Research published in Fertility and Sterility found that women with PCOS treated with metabolic agents showed significant reductions in BMI and total testosterone levels compared to placebo groups.
The result: women who have not ovulated regularly in years suddenly start ovulating. Their doctors prescribed tirzepatide for weight management or metabolic improvement. Nobody mentioned that the metabolic improvement might include restored fertility. Nobody suggested changing birth control methods. And pregnancies followed.
The timeline of restored fertility
Fertility can return faster than you think. Some women report conception within the first three months of tirzepatide treatment. The typical timeline looks something like this:
Weeks 1-4: Tirzepatide initiated at 2.5 mg starting dose. Weight loss begins. Insulin sensitivity starts improving. Gastric emptying delays. Birth control absorption potentially compromised.
Weeks 4-8: Dose escalation to 5 mg. Weight loss accelerates. Hormonal changes begin. Some women notice menstrual cycle changes (periods becoming more regular after being irregular). Birth control interference continues with dose increase.
Weeks 8-16: Further dose escalation. Significant weight loss achieved (often 5-10% of starting weight). Insulin resistance improving markedly. Androgen levels dropping. Ovulation may resume even before periods normalize. This is the high-risk window for unplanned pregnancy.
Weeks 16+: Continued weight loss and metabolic improvement. Full fertility restoration in many women who previously had anovulation. If using oral contraceptives without backup methods, pregnancy risk remains elevated.
The FDA position on tirzepatide and pregnancy
The FDA classifies the evidence as insufficient. Not dangerous. Not safe. Insufficient.
This matters because it shapes how you interpret the risk.
What the label actually says
The Mounjaro and Zepbound prescribing information states: "Available data with tirzepatide use in pregnant women are insufficient to evaluate for a drug-related risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes."
The label then references animal studies: "Based on animal reproduction studies, there may be risks to the fetus from exposure to tirzepatide during pregnancy."
And the recommendation: "Advise pregnant women of the potential risk to a fetus. Advise women of reproductive potential to inform their healthcare provider of a known or suspected pregnancy."
Notice what the label does not say. It does not say tirzepatide causes birth defects in humans. It does not say exposure during early pregnancy will harm your baby. It says the data is insufficient and that animal studies suggest potential risk. These are different statements, and the distinction matters for your mental health as you navigate this situation.
Animal study context
The animal studies that drive the pregnancy warnings used tirzepatide in pregnant rats and rabbits during organogenesis. The findings included:
Reduced fetal weight
Increased incidence of skeletal abnormalities
Higher rates of pregnancy loss
Some visceral malformations
Critical context: these studies used "clinically relevant exposures based on AUC," meaning the drug levels in animals were comparable to levels in humans at therapeutic doses. This is concerning. But animal-to-human translation is imperfect. Rats and rabbits metabolize drugs differently. The specific vulnerabilities of rodent embryos do not always predict human fetal risk.
More importantly, the weight loss caused by tirzepatide in these animals likely contributed to adverse outcomes independently of the drug itself. Caloric restriction during pregnancy is harmful regardless of the cause. Separating the effects of the drug from the effects of the drug-induced weight loss is methodologically challenging.
Human data so far
A multicenter observational study published in The Lancet Regional Health examined GLP-1 receptor agonist exposure during the first trimester of pregnancy. The study drew from databases of six Teratology Information Services across Europe. The findings: no elevated risk of major birth defects compared to unexposed pregnancies.
This study had limitations. The sample size was modest. Most exposures were to liraglutide and semaglutide rather than tirzepatide specifically. But the data provides a meaningful signal that first-trimester GLP-1 exposure may not be as harmful as animal studies predicted.
Additional reassurance comes from the thousands of "Ozempic baby" pregnancies now progressing through delivery. While systematic studies are still underway, the absence of alarming signals in post-market surveillance is cautiously encouraging.
How long tirzepatide stays in your system after stopping
Understanding the pharmacokinetics helps frame your risk window.
Half-life and clearance
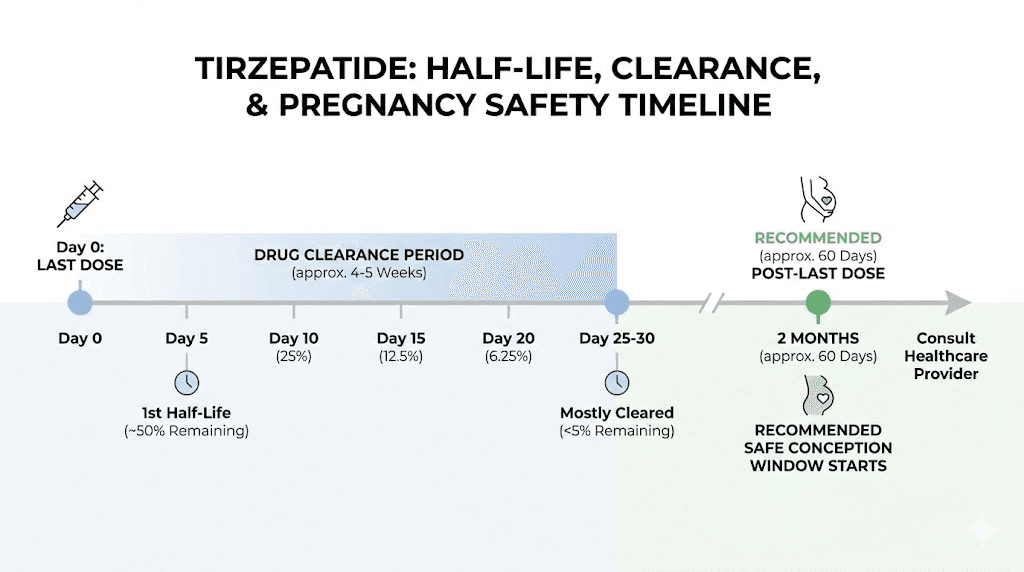
Tirzepatide has an elimination half-life of approximately 5 days. This means:
After 5 days: 50% of the drug remains
After 10 days: 25% remains
After 15 days: 12.5% remains
After 20 days: 6.25% remains
After 25 days: 3.125% remains
After 30 days: Less than 2% remains (considered functionally cleared)
Full clearance takes approximately 25-30 days from your last injection. This means that even after you stop tirzepatide upon discovering pregnancy, the drug will remain in your body at decreasing levels for about a month.
If you are in early pregnancy (4-6 weeks when most women discover it), the tirzepatide in your system will overlap with the beginning of organogenesis, which runs from approximately weeks 3-8 of gestation. This is the reality. But remember the human data: first-trimester GLP-1 exposure has not shown elevated birth defect rates in the observational studies conducted so far.
The recommended washout period for planned pregnancies
For women planning pregnancy, guidelines recommend stopping tirzepatide at least one to two months before attempting conception. The Canadian product guidelines specify four weeks. Many reproductive endocrinologists recommend two months (approximately 8-10 half-lives) for complete clearance during the critical early weeks of development.
If your pregnancy was unplanned, you obviously did not have the luxury of a washout period. This is the reality for most "Ozempic baby" pregnancies. The exposure happened. What matters now is monitoring, not guilt.
Birth control considerations while on tirzepatide
If you are reading this section because you want to prevent pregnancy while continuing tirzepatide treatment, here is what you need to know.
Why oral contraceptives are risky with tirzepatide
Tirzepatide delays gastric emptying. Oral contraceptives depend on consistent absorption through the gastrointestinal tract to maintain effective hormone levels. When gastric emptying slows, the rate and extent of contraceptive absorption changes. The result is lower peak hormone concentrations and reduced overall hormonal exposure.
The interaction is most significant during two periods:
Treatment initiation: When you first start tirzepatide, the gastric emptying effect is new and your body has not adapted
Dose escalation: Each time you increase your tirzepatide dose, the gastric emptying delay intensifies temporarily before your body adjusts
Over time, as your body adapts to each dose level, the impact on gastric emptying partially normalizes. But the first four weeks at any new dose are the highest-risk period for reduced oral contraceptive efficacy.
Recommended alternatives
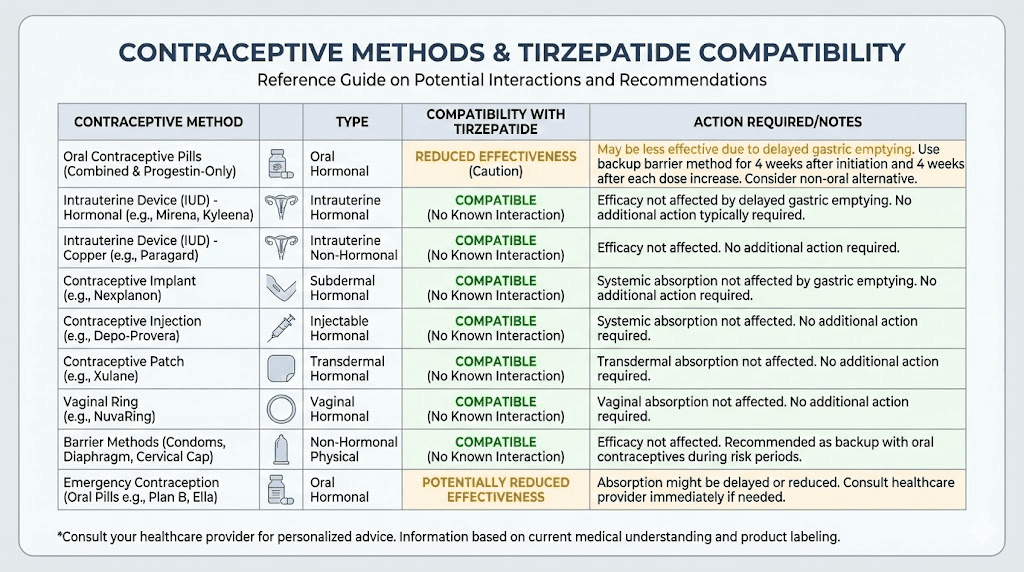
The FDA-approved prescribing information recommends one of two approaches:
Option 1: Switch to non-oral contraception entirely. Methods unaffected by gastric emptying include:
IUD (hormonal or copper)
Hormonal implant (Nexplanon)
Injectable contraceptive (Depo-Provera)
Contraceptive patch
Vaginal ring (NuvaRing)
These methods deliver hormones through routes other than the GI tract, so tirzepatide cannot interfere with their absorption.
Option 2: Keep oral contraceptives but add a barrier method. Use condoms in addition to your birth control pills for four weeks after starting tirzepatide and four weeks after each dose increase.
The most reliable approach is Option 1. Non-oral contraception eliminates the interaction entirely. If you are serious about preventing pregnancy while on tirzepatide, talk to your gynecologist about switching methods before or when you start treatment.
The timing trap
Many women start tirzepatide for weight loss without thinking about fertility at all. They may have struggled with infertility for years. They may have irregular periods and assume they cannot get pregnant easily. They may be on oral contraceptives and feel protected.
Then three things happen simultaneously. Their weight drops. Their fertility surges. Their birth control effectiveness decreases. Without any change in sexual behavior, their pregnancy risk has increased dramatically compared to pre-tirzepatide.
This is the trap. It catches women who were not trying to get pregnant. Women who thought they were protected. Women who had no reason to suspect their birth control was failing. If this describes you, understand that what happened was not carelessness. It was an inadequately communicated medication interaction.
Nutrition and weight management after stopping tirzepatide for pregnancy
Stopping tirzepatide during pregnancy creates a unique nutritional situation. Your body shifts from caloric restriction to needing increased nutrition for fetal development. This transition requires attention.
Appetite changes after discontinuation
When you stop tirzepatide, several things happen:
Your appetite returns. For many women, this happens within one to two weeks of the last dose. The appetite suppression that made weight loss feel effortless disappears. Food becomes interesting again. Cravings may intensify, particularly during pregnancy.
Gastric emptying normalizes. Meals move through your digestive system faster. You feel hungry sooner after eating. The fullness signals that tirzepatide amplified return to baseline levels.
This is actually helpful during pregnancy. Your body needs more calories to support fetal growth. The average pregnant woman needs an additional 340 calories per day in the second trimester and 450 additional calories per day in the third trimester. The return of normal appetite helps you meet these increased demands.
Weight regain concerns
Many women worry about weight regain after stopping tirzepatide. During pregnancy, some weight gain is not just normal. It is necessary.
The Institute of Medicine guidelines for pregnancy weight gain depend on your pre-pregnancy BMI:
Underweight (BMI under 18.5): 28-40 pounds recommended
Normal weight (BMI 18.5-24.9): 25-35 pounds recommended
Overweight (BMI 25-29.9): 15-25 pounds recommended
Obese (BMI 30+): 11-20 pounds recommended
If tirzepatide brought you from an obese BMI to a healthier range before pregnancy, you may be starting from a better metabolic position than if you had not taken the medication at all. The weight loss, even if some is regained during pregnancy, provided metabolic benefits that persist. Improved insulin sensitivity, reduced inflammation, and better cardiovascular markers do not disappear overnight.
Work with your OB/GYN or a registered dietitian to set appropriate pregnancy weight gain targets based on your current BMI. Do not try to maintain your tirzepatide weight loss during pregnancy. Your baby needs you to gain weight appropriately.
Key nutrients to prioritize
After stopping a medication that reduced food intake, certain nutritional deficiencies may need attention:
Folate/Folic acid: Critical for neural tube development. If you were not already taking a prenatal vitamin with at least 400-800 mcg of folic acid, start immediately. Some providers recommend 1 mg for women with higher BMI.
Protein: Essential for fetal growth. Aim for 75-100 grams daily. Collagen peptides can contribute to protein intake, though they should not be your primary source. Whole food proteins provide complete amino acid profiles.
Iron: Pregnancy increases iron demands significantly. If your diet was restricted on tirzepatide, your iron stores may be lower than ideal.
Calcium and Vitamin D: Needed for fetal bone development. Particularly important if your food intake was limited before discovering the pregnancy.
B vitamins: The B12 supplementation that some tirzepatide protocols include becomes even more important during pregnancy. B12 supports neural development and red blood cell formation.
Get bloodwork done early in your pregnancy to check for any nutritional deficiencies that may have developed during your tirzepatide treatment. Correcting deficiencies early gives your baby the best foundation.
Tirzepatide and breastfeeding after delivery
Looking ahead: many women want to know whether they can resume tirzepatide after delivery, particularly if they plan to breastfeed.
What the research shows
Recent research has provided reassuring data on this topic. A clinical study examining tirzepatide transfer into breast milk found that tirzepatide was undetectable in 164 of 171 milk samples collected from lactating women taking doses up to 5 mg. In the few samples where the drug was detected, the cumulative amount over 28 days represented less than 0.02% of the maternal dose.
Furthermore, even if tiny amounts reach the infant through breast milk, tirzepatide is a peptide. Peptides are proteins. The infant's gastrointestinal tract would likely break down tirzepatide before significant absorption could occur. This is the same reason why oral tirzepatide requires special formulation technology to survive adult GI digestion. An infant's digestive system would neutralize trace amounts.
Current recommendations
Despite the reassuring data, current guidelines still advise caution. Most healthcare providers recommend waiting until after breastfeeding to resume tirzepatide. The primary concern is not direct drug transfer, but indirect effects: tirzepatide-induced caloric restriction could reduce milk supply, and the appetite suppression might prevent mothers from consuming the extra 300-500 calories per day that breastfeeding demands.
If postpartum weight management is a priority, discuss the timing with your provider. Some women choose to resume tirzepatide after weaning. Others work with their doctors to restart at low doses during later stages of breastfeeding when the infant is receiving significant nutrition from solid foods. These decisions should be individualized based on your health needs and breastfeeding goals.
For more on the intersection of GLP-1 medications and lactation, see our guide on GLP-1 use while breastfeeding.
Emotional and psychological considerations
The medical facts only tell part of the story. Discovering an unplanned pregnancy while on a medication with pregnancy warnings creates a specific emotional experience that deserves acknowledgment.
The guilt spiral
Many women blame themselves. They think they should have known better. They should have used backup contraception. They should have asked more questions. They should have read the fine print.
Stop.
The contraception interaction warning is buried in prescribing information that most patients never see in full. Many prescribers do not emphasize the birth control interaction during consultations. The restored fertility from weight loss is discussed in medical literature but rarely communicated to patients starting weight loss medications. You did not fail. The communication system around these medications failed you.
The anxiety about fetal exposure
Worrying about your baby is natural. But persistent, debilitating anxiety about medication exposure can harm your pregnancy through elevated cortisol, disrupted sleep, and inadequate nutrition (if anxiety reduces your appetite).
Ground yourself in the actual data. The human studies available show no increased rate of major birth defects from first-trimester GLP-1 exposure. Your healthcare provider will monitor your pregnancy closely. Early ultrasounds will check development. Anatomy scans will evaluate organ formation. You are not flying blind.
If the anxiety becomes overwhelming, ask your provider about referral to a perinatal mental health specialist. They work specifically with pregnant women dealing with medication exposure anxiety and other pregnancy-related psychological concerns. There is no weakness in seeking this support.
The complicated feelings about the pregnancy itself
An unplanned pregnancy on tirzepatide can trigger complex emotions. You might feel happy and terrified simultaneously. You might feel resentful that your weight loss journey was interrupted. You might feel grateful if you wanted children but thought infertility would prevent it.
All of these feelings are valid. None of them are wrong. Give yourself permission to feel contradictory emotions without judgment. Talk to your partner, your friends, a therapist. Process the situation at your own pace.
What happens after delivery: resuming tirzepatide
Many women want to resume tirzepatide after pregnancy. The weight lost during treatment often returns during pregnancy and the postpartum period, especially when the medication is stopped abruptly. Planning for this is practical, not vain.
When to restart
The timeline depends on your situation:
If not breastfeeding: Most providers will consider restarting tirzepatide at the six-week postpartum checkup, once your body has recovered from delivery and any acute postpartum concerns have been addressed.
If breastfeeding: Wait until after weaning or until your baby is receiving most nutrition from solid foods (typically around 6-12 months). Discuss the specific timing with your provider based on your individual circumstances.
If planning another pregnancy soon: Consider whether the cycle of starting, stopping, and restarting makes sense for your situation. If you plan to conceive again within a year or two, your provider might recommend alternative weight management strategies during the interim.
Dose considerations when restarting
You will likely restart at the beginning of the dose escalation schedule (2.5 mg) regardless of your previous dose. Your body has been off the medication for months. Restarting at your previous maintenance dose could cause severe gastrointestinal side effects.
The dose escalation will be familiar. Your body may respond more quickly the second time around, since you have prior exposure experience. But follow your provider's guidance on titration speed.
Birth control planning for the restart
Before resuming tirzepatide, get your contraception sorted. If you were on oral contraceptives before and they failed you during tirzepatide treatment, consider switching to a non-oral method. Learn from the experience.
Non-oral options that will not interact with tirzepatide include IUDs, implants, injectable contraceptives, patches, and vaginal rings. Discuss these with your gynecologist and have the new method in place before restarting tirzepatide.
Planning a pregnancy while on tirzepatide
Some women reading this are not currently pregnant but want to be. They are taking tirzepatide and wondering how to transition safely to conception.
The recommended timeline
If you are currently taking tirzepatide and want to get pregnant:
Step 1: Talk to your prescriber about discontinuation. They can help you plan the transition and address any metabolic concerns.
Step 2: Stop tirzepatide at least one to two months before attempting conception. The Canadian prescribing information recommends four weeks minimum. Many reproductive specialists recommend eight weeks (two months) for a full washout that covers the critical first weeks of embryonic development.
Step 3: Start prenatal vitamins immediately if you have not already. Folic acid is most critical during the first weeks of pregnancy, often before a woman even knows she is pregnant.
Step 4: Use the washout period to stabilize your eating patterns. You will be transitioning from medication-assisted appetite suppression to natural hunger cues. Give yourself time to adjust before adding pregnancy's nutritional demands.
Step 5: Begin actively trying to conceive after the washout period is complete. Your fertility may be better than it was before tirzepatide treatment, thanks to the weight loss and metabolic improvements the medication provided.
Leveraging tirzepatide weight loss for fertility
Here is an interesting perspective that reproductive endocrinologists are starting to discuss. For women with obesity-related infertility, tirzepatide treatment before conception may actually improve pregnancy outcomes.
Maternal obesity increases risk of gestational diabetes, preeclampsia, macrosomia (large birth weight), cesarean delivery, and neural tube defects. By achieving significant weight loss before pregnancy, tirzepatide may reduce these risks substantially.
The ideal scenario: use tirzepatide to reach a healthier weight, stop the medication with adequate washout time, conceive at a lower BMI, and benefit from improved metabolic health throughout the pregnancy. Some reproductive endocrinologists are now incorporating GLP-1 medications into preconception weight loss protocols for exactly this reason.
This approach requires planning and coordination between your weight loss provider and your reproductive specialist. But for women with BMI-related fertility challenges, it represents a genuinely hopeful pathway.
Frequently asked questions
Can tirzepatide cause birth defects in humans?
No human studies have shown that tirzepatide causes birth defects. Animal studies showed developmental abnormalities at clinically relevant doses, but a 2023 multicenter human study of GLP-1 receptor agonist exposure during the first trimester found no elevated risk of major birth defects. The FDA classifies the data as insufficient rather than confirming harm.
How soon after stopping tirzepatide can I try to get pregnant?
Most guidelines recommend waiting at least one to two months after your last tirzepatide dose before attempting conception. This allows approximately 5-10 half-lives for the drug to clear your system. The Canadian prescribing information specifies four weeks minimum. Many reproductive specialists recommend eight weeks for a more conservative washout.
Does tirzepatide make you more fertile?
Tirzepatide does not directly increase fertility. However, the weight loss it produces can restore ovulation in women with obesity-related anovulation, particularly those with PCOS. Improved insulin sensitivity, reduced androgen levels, and normalized estrogen production collectively create conditions that support natural fertility. The effect can be dramatic for women who had not been ovulating.
Will my baby be okay if I was taking tirzepatide when I conceived?
The available human data is reassuring. Observational studies of first-trimester GLP-1 exposure have not shown increased rates of major birth defects compared to unexposed pregnancies. Your healthcare provider will monitor your pregnancy with additional ultrasounds and screening to ensure normal development. Most women with early tirzepatide exposure go on to have healthy pregnancies.
Do I need to stop tirzepatide immediately when I find out I am pregnant?
Yes. Discontinue tirzepatide as soon as pregnancy is confirmed. Do not take your next scheduled dose. Contact your healthcare provider the same day to discuss monitoring plans. There is no need to taper the medication. Stopping abruptly is the recommended approach.
Can tirzepatide affect my ability to breastfeed?
Tirzepatide is largely undetectable in breast milk at doses up to 5 mg, with less than 0.02% of the maternal dose appearing in milk over 28 days. However, most providers recommend not taking tirzepatide while breastfeeding due to potential indirect effects on milk supply from reduced caloric intake. See our GLP-1 and breastfeeding guide for detailed information.
Should I switch birth control methods before starting tirzepatide?
Yes, if you are using oral contraceptives and want to prevent pregnancy. Tirzepatide can reduce oral contraceptive absorption by approximately 20% due to delayed gastric emptying. The FDA recommends switching to non-oral contraception or adding a barrier method for four weeks after starting and four weeks after each dose increase. Non-oral methods like IUDs, implants, or injectable contraceptives are unaffected by tirzepatide.
Is the situation different with semaglutide versus tirzepatide?
The general concerns are similar, but tirzepatide has a greater impact on oral contraceptive absorption than semaglutide. The dual GIP/GLP-1 mechanism in tirzepatide produces more significant gastric emptying delays. Semaglutide manufacturers recommend an eight-week washout before conception, while tirzepatide guidelines suggest four weeks to two months. Both medications can restore fertility through weight loss.
What if I want to get pregnant and tirzepatide is helping my PCOS?
This is actually a promising scenario. Use tirzepatide to improve your metabolic health and achieve a healthier weight, then stop with adequate washout time before conceiving. The metabolic improvements, including better insulin sensitivity and lower androgens, persist for some time after stopping the medication. You may conceive more easily at your new weight than you would have before treatment. Coordinate with both your weight management provider and reproductive specialist.
Can I take tirzepatide for gestational diabetes during pregnancy?
No. Tirzepatide is not approved or recommended for use during pregnancy, including for gestational diabetes management. If you develop gestational diabetes, your provider will use pregnancy-safe treatments such as insulin, dietary modifications, and exercise. Do not self-prescribe tirzepatide for blood sugar management during pregnancy.
External resources
Fertility and Sterility: Treating Obesity and Fertility with GLP-1 RAs
Diabetes, Obesity and Metabolism: GLP-1 RA Safety in Pregnancy
For researchers serious about understanding the intersection of peptide protocols and reproductive health, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed tirzepatide protocols, safety information, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your pregnancies stay healthy, your decisions stay informed, and your families stay exactly the size you planned.